

 | Journal of Renewable Materials |  |
DOI: 10.32604/jrm.2022.020965
ARTICLE
Insecticidal Effect of Green Bimetallic Nanoparticles from Crotalaria longirostrata on Cotton Mealybug, Phenacoccus solenopsis
1Facultad de Medicina de la Universidad Autónoma de Baja California, Dr. Humberto Torres Sanginés S/N, Centro Cívico, Mexicali, Baja California, México
2Instituto de Ingeniería de la Universidad Autónoma de Baja California, Calle de la Normal S/N y Boulevard Benito Juárez, Mexicali, Baja California, México
3Instituto Tecnológico de Conkal, División de Estudios de Posgrado e Investigación, Antigua Carretera Mérida-Motul, Conkal, Yucatán, México
4Facultad de Medicina Veterinaria y Zootecnia de la Universidad Autónoma de Chiapas, Chiapas, México
5Universidad Autónoma de Baja California, Instituto de Ciencias Agrícolas, Baja California, México
*Corresponding Author: Daniel González-Mendoza. Email: danielg@uabc.edu.mx
Received: 21 December 2021; Accepted: 14 April 2022
Abstract: The present study was conducted to evaluate the impact of bimetallic nanoparticles (NPs) of copper (Cu) and zinc (Zn) from Crotalaria longirostrata on the insect mortality and cell viability of Phenacoccus solenopsis. The biosynthesis of bimetallic nanoparticles (Cu/Zn-NPs) from C. longirostrata was confirmed by the presence of a single peak around 378 nm. The hydrodynamic diameter of the Cu/Zn nanoparticles varied from 59 to 119 nm and their zeta potential was found to be −30.90 mV. The EDX analysis showed a strong signal for Cu and Zn (8 and 8.6 keV, respectively). The Cu/Zn-NPs showed an effective insecticide activity against P. solenopsis with 63% of insect mortality after 96 h treatment. However, further studies about nanotoxicity of Cu/Zn-NPs from C. longirostra are needed to reveal the exact mechanism of nanoparticles toxicity in insects.
Keywords: Phytonanoparticles; chipilin; biocontrol; agronanotechnology
The Nanotechnology involve the design, characterization, and study of organic, inorganic or hybrid materials at the nanoscale in the rage of 1–100 nm. These materials receipt the term of nanoparticles and are characterized by theirs great versatile and having a wide spectrum of applications [1]. Nanostructures, nanomaterials and nanocomposites can be obtain using two different process: “bottom-up” and “top-down” in the first, the nanostructures are fabricated by building upon single atoms or molecules including the miniaturization of materials components (up to atomic level) with further self-assembly process leading to the formation of nanomaterials (2–10 nm size range). The methods used in bottom-up approach include plasma arcing, chemical vapor deposition process, metal organic decomposition, laser or spray pyrolysis, sol-gel method and self-assembly processes and green synthesis [2]. On the other hand, “top down” represent the most conventional nanomaterial manufacturing in which a larger sized material is ground or milled to smaller particle sizes. For example, some graphenes in which graphite is mechanically reduced to nanoscale dimension [3,4]. However, the top-down approach has several disadvantages such as development of imperfections in processed materials, high cost and requirement of high surface finished materials [5]. The chemical and physical synthesis of nanomaterials are characterized by high energy consumption, low yield, high cost, and environmental damage by employing harsh reducing agents [6]. In this sense, the green synthesis a bottom-up approach is a promising and novelty tool in agro-bionanotechnology. This method provides economic and environmental benefits as an alternative to chemical and physical methods [7].
The green synthesis is clean, cost-effective and environmentally friendly because nontoxic biosafe reagents are used. This synthesis involves different natural resources available in nature such as microorganisms (e.g., fungi, bacteria, algae, and viruses) and plant extracts as reducing agents. Although, the microorganisms can be used to produce nanomaterials, their pathogenicity issue, slow range of synthesis, limited number of sizes/shapes nanoparticles and requirements of large cultures limit their use compared to routes involving plant-based materials [8]. The aqueous extracts from plants produce more stable nanoparticles compared to other means and it is very straightforward to scale up with lower risk of contamination. Aqueous extracts from plants are generally a mixture of different active biomolecules like proteins, carbohydrates, vitamins, polymers and different metabolites that provide high stability and enhanced dispersity to the synthesized nanoparticles [9]. The use of the bio-nanotechnology (combination of biological principles with different chemical and physical methods) could be presented as a potential alternative for reducing the use of chemicals in agriculture. For example, the larvicidal activity of biosynthesized silver and gold nanoparticles using the leaf extracts as potential source of alternative tools against insects vector (Anopheles stephensi, Aedes aegypti and Culex quinquefasciatus) of human diseases [10,11], have been previously reported. In the case of pest of plants, Murugan et al. [12] evaluated the efficacy of green nanoparticles (ZnO) fabricated using Sargassum wightii against Helicoverpa armigera Hubner an important polyphagous agricultural insect pest in the word. However, information about the action of nanoparticles against insects such as P. solenopsis (cotton mealybug) are limited. P. solenopsis is a devastating pest of cotton causing tremendous loss in the yield of crops each year in the word. On the other hand, Chipilín (C. longirostrata Hook. & Arn.) is a vegetable used in the traditional cuisine of Central America and Southern Mexico [13]. This vegetable is rich in secondary metabolites, whose leaf extract has demonstrated antibacterial and antifungal properties [14]. Chipilín represented a biotechnological alternative by their significantly content of bioactive compounds such as: tannins, phenols and flavonoids [15]. Today investigations about the use of Chipilin in the bio production of bimetallic nanoparticles are limited. Therefore, the aim of this study was focused in the pesticide potential against P. solenopsis of nanoparticles of copper and zinc from Chipilin extracts.
2.1 Preparation of Leaf Extract
Healthy leaves of Chipilín (C. longirostrata) were collected from Suchiapa, Chiapas, Mexico (16° 13′ N, 93° ′ W). Random leaf samples were pulverized and 5 g were transferred into 100 mL Erlenmeyer flask containing 50 mL distilled water and stirred for 30 min at 60°C in a heating water bath. The particulate matter in the C. longirostrata extracts were removed by centrifugation at 6000 rpm for 10 min. Then the samples were refrigerated at −4°C in 100 mL Erlenmeyer flasks for further experiments.
2.2 Biosynthesis of Phyto-Nanoparticles
For the synthesis of bimetallic nanoparticles (Cu/Zn-NPs), an aliquot of 10 mL aqueous extract of C. longirostrata was mixed with 50 mL of 10 mM solution of copper sulfate/zinc nitrate (1:1) in 100 mL Erlenmeyer flask and heated at 60°C for 30 min. The process of bio reduction of Cu/Zn-NPs, was determinate by the color change in the reaction mixture which indicates the formation of Cu/Zn-NPs.
The bioreduction and formation of C. longirostrata Cu/Zn nanoparticles (Cu/Zn-NPs) was monitored using the UV/VIS Spectrophotometer, (DR6000™ USA) at a resolution of 5 nm from 340 to 650 nm. The synthesized Cu/Zn-NPS were purified through centrifugation at 10000 g for 15 min and the pellet was washed with sterile distilled water and transferred to freeze dryer. The powder obtain was used in assays and further characterization.
2.3 Green Cu/Zn Nanoparticles Characterization
The size and morphology and elemental composition of synthesized Cu/Zn-NPs was realized by scanning electron microscopy–energy-dispersive spectra (SEM–EDS) (JEOL 64000; Tokyo, Japan). The Fourier transform infrared spectrum (FTIR) was used to identify the molecules in C. longirostrata extracts responsible for formation of Cu/Zn-NPs. The infrared spectra of plant extract and Cu/Zn-NPs were recorded using KBr pellet technique in Bruker IFS 66 V model FTIR spectrometer in the region 4000–400 cm−1. The frequencies for all sharp bands were accurate to 0.001 cm−1. The Dynamic Light Scattering (DLS) of Cu/Zn-NPs, was realized using a Nanotrac Wave II instrument (Microtrac) and analyzing using Microtrac FLEX operating software.
2.4 Evaluation of Insecticidal Activity of Cu/Zn-NPs
The insecticidal activity of Cu/Zn-NPs from C. longirostrata was evaluate used P. solenopsis (Tinsley) (Hemiptera: Pseudococcidae) previously collected from infested chili “habanero” plants (Capsicum chinense Jacq.) in Mexicali valley, Mexico during summer 2020. Twenty insects were carefully transferred to Petri dishes each (100 × 15 mm). The Petri dishes containing the insects were sprayed (using a small volume hand atomizer) with 2 ml aqueous solution of Cu/Zn-NPs (100 ppm) (Treatment 1) and same procedure was followed for the control group, which consisted of distilled water (Treatment 2) and Cu/Zn solution (100 ppm) (Treatment 3). All Petri dishes were incubated in a growth chamber at 30 ± 2°C with 12/12 h day-night period with 70% relative humidity. The number of insects survivors (% mortality) was recorded counted after 24, 48, 72 and 96 h of initial application by using Abotts formula. Death of insect individuals was determined based on the coordinated muscle response of mealybugs to gentle prodding with an insect needle [15].
2.5 Determination of Cell Viability Using Evans Blue Staining
The cell viability of mealybugs treated with Cu/Zn-NPs were evaluated by spectrophotometric assay using Evans blue staining as described previously by Mendez-Trujillo et al. [15] with minor modifications. The insects of each treatment were stained with 0.25% aqueous Evans blue solution for 30 min at room temperature. Then the insects were washed with distilled water for 10 min to remove excess and unbound dye. The Evans blue trapped on insects was removed using 50% ethanol with 1% SDS solution at 60°C for 30 min and quantified spectrophotometric at 600 nm.
All the tests were realized by triplicate and the data collected were subjected to analysis of variance (ANOVA) using Statistica software version 9.0.3.
3.1 Biosynthesis of Phyto-Nanoparticles
In the present study, the function of C. longirostrata extract as reducing and stabilizing agents was monitored by UV-visible spectrum analysis in the range of 280–800 nm. The spectrum showed a peak at 378 nm, which can be attributed to absorption by Cu/Zn NaPs. Similar results were also reported to Cu/Zn nanoparticles obtain from Borassus flabellifer and Azardirachta indica which showed an absorption peak between 338 and 400 nm wavelength, respectively [16,17]. Even though the color change was observed in the biogenic Cu/ZN-NPs synthesis (Fig. 1), the mechanism of bio-reduction of Cu/Zn by C. longirostrata extract is not known yet. In this sense, some studies suggest that biological activity from this plant can be attributed to the presence of functional groups such as alkaloids, saponins and flavonoids. Such molecules can act as reducers and stabilizers in the stabilization process green synthesis of bimetallic nanoparticles [14,15]. In this sense the probable mechanism of the biosynthesis Cu/Zn-NPs using C. longirostrata, take place in two main steps as follows: 1) the reduction and nucleation by effect phytomolecules in the plant extract solution; 2) a growth phase during which the small adjacent nanoparticles spontaneously coalesce into larger size particles accompanied by an increase in the thermodynamic stability of green nanoparticles.
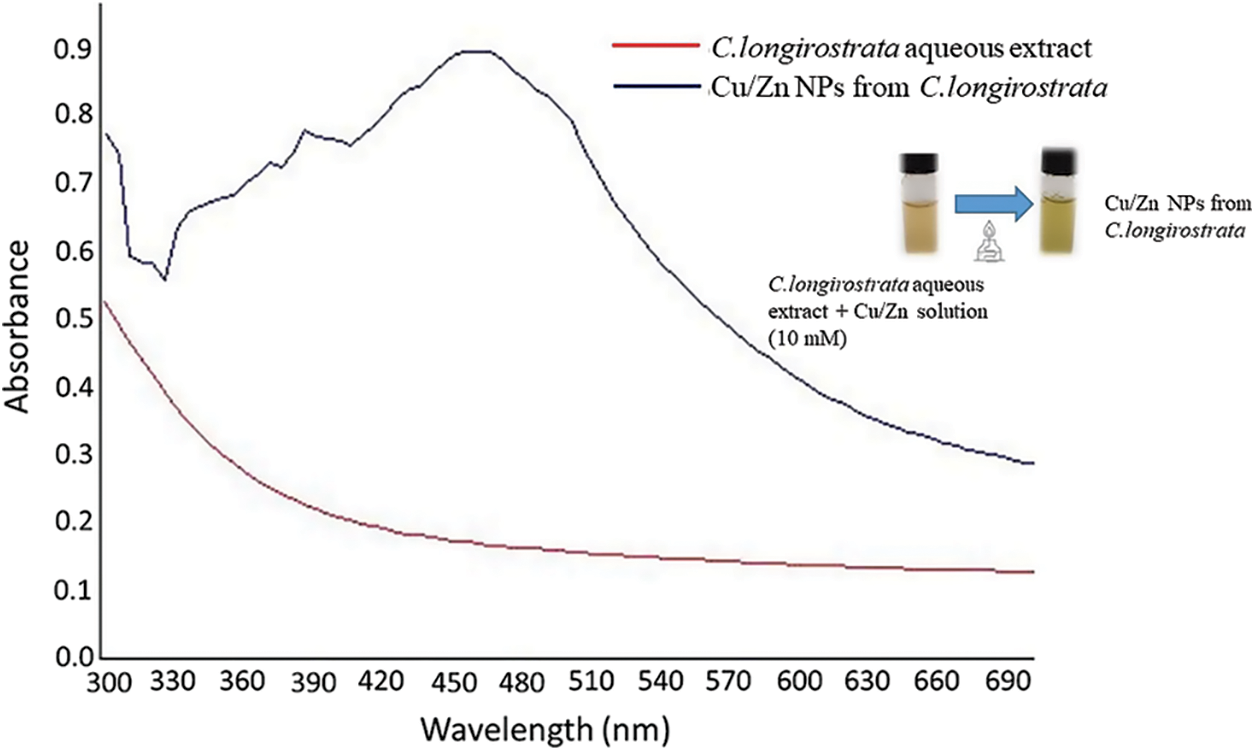
Figure 1: UV-vis absorption spectrum of Cu/Zn nanoparticles from C. longirostrata and their green synthesis
3.2 Green Cu/Zn Nanoparticles Characterization
In the present study the hydrodynamic size, polydispersity index (PDI) and zeta potential of the Cu/Zn-NPs synthesis were determined using dynamic light scattering (DLS) analysis. The hydrodynamic diameter of the Cu/Zn nanoparticles varied between 59 to 119 nm at pH 7 (Fig. 2). The zeta potential can be related with the electrostatic repulsive force between the NPs depends on the charge that resides on the surface of nanoparticles [18]. Therefore, when the charge of the NPs is negative, it does not cause the aggregation of these nanoparticles, causing long-term stability. In this sense, the zeta potential of the Cu/Zn-NPs in the present investigation was found to be −30.90 mV with PDI of 2.81 and it can be inferred that the synthesis of Cu/Zn-NPs are stable but not disperse according to the Ostwald ripening phenomena, which can broaden particle size distribution [19,20]. The SEM analyses of synthesized Cu/Zn-NPs clearly showed spherical aggregates with size ranging from 41 to 67 μm (Fig. 3a). Moreover, Vinayagam et al. [21] mentioned that the aggregate formation of NPs may be due to the procedures involved during sample preparation and the electrostatic interactions between the phytomolecules of plant extract and NPs synthetized.
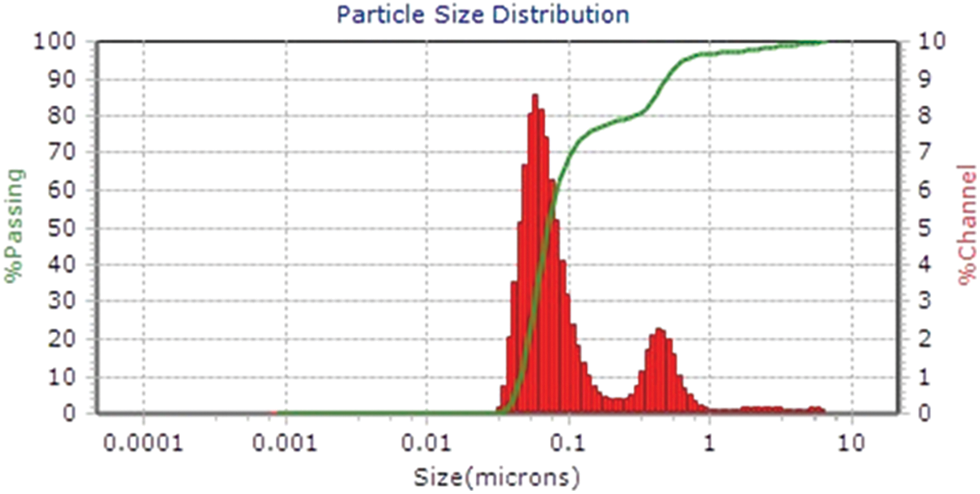
Figure 2: Particle size distribution of Cu/Zn nanoparticles from C. longirostrata using dynamic light scattering measurements (DLS)
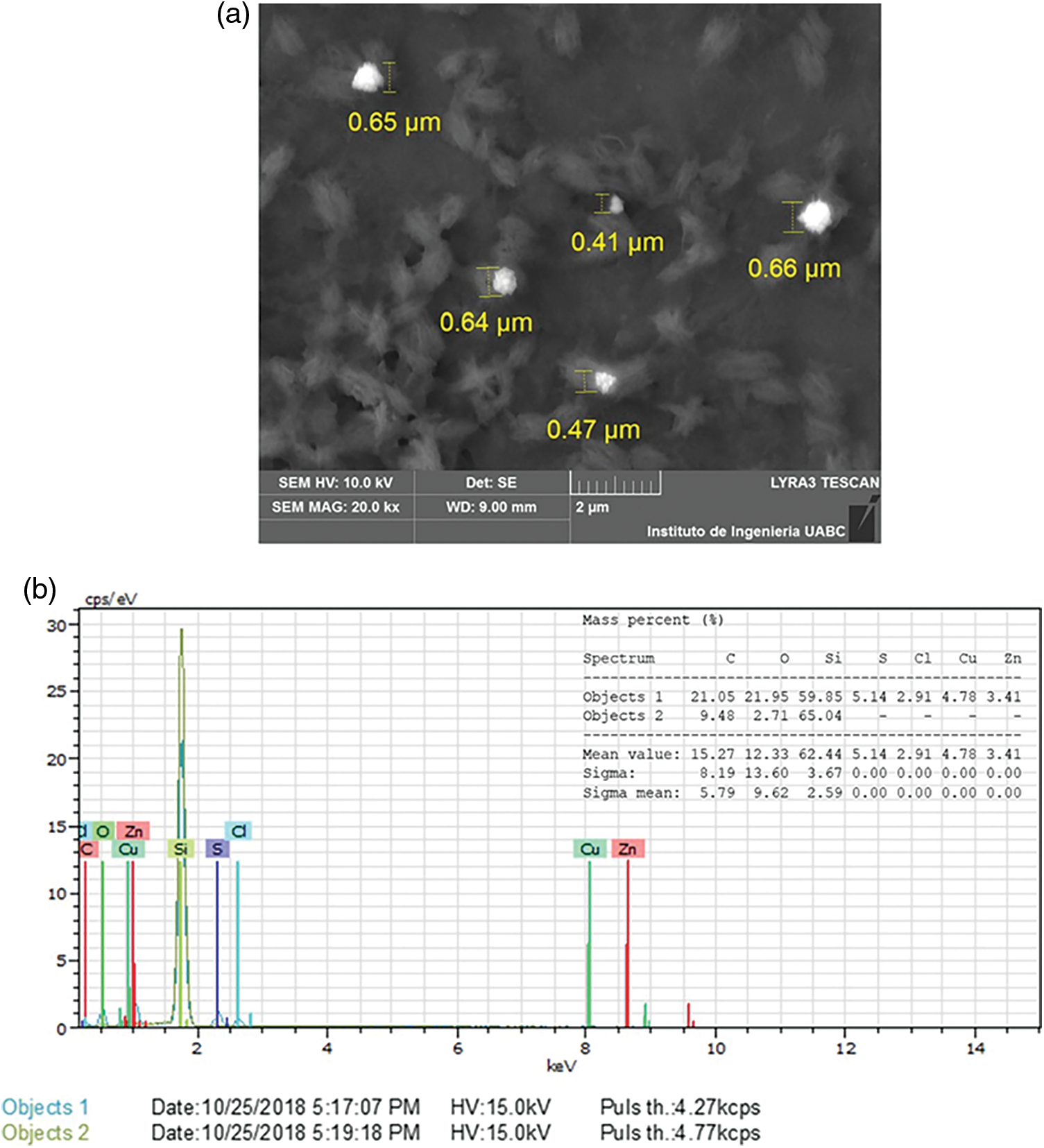
Figure 3: Analysis of SEM (a) and EDX (b) image of Cu/Zn nanoparticles from C. longirostrata
The elemental composition of the Cu/Zn-NPs was determined by EDX analysis. Fig. 3b reveals the strong signal for Cu and Zn (8 and 8.6 keV, respectively). This finding ascertained the existence of metallic Cu and Zn in the sample. The presence of other peaks, such as C (15%), O (12.33%), Si (62.44%), S (5.14%) and Cl (2.91%) may be due to the glass slide that bears the sample and the mineral composition of plant extract [9]. Spectra of the FTIR analysis both Cu/Zn-NPs and the plant extract are presented in Fig. 4. The infrared spectrum of C. longirostrata showed characteristic bands, 3260.71, 1588.33, 1389.07 and 1038 cm−1 that can associate with OH (alcohol), C≡C (aromatic ring), C–O and C–H stretching vibrations (Fig. 4a).
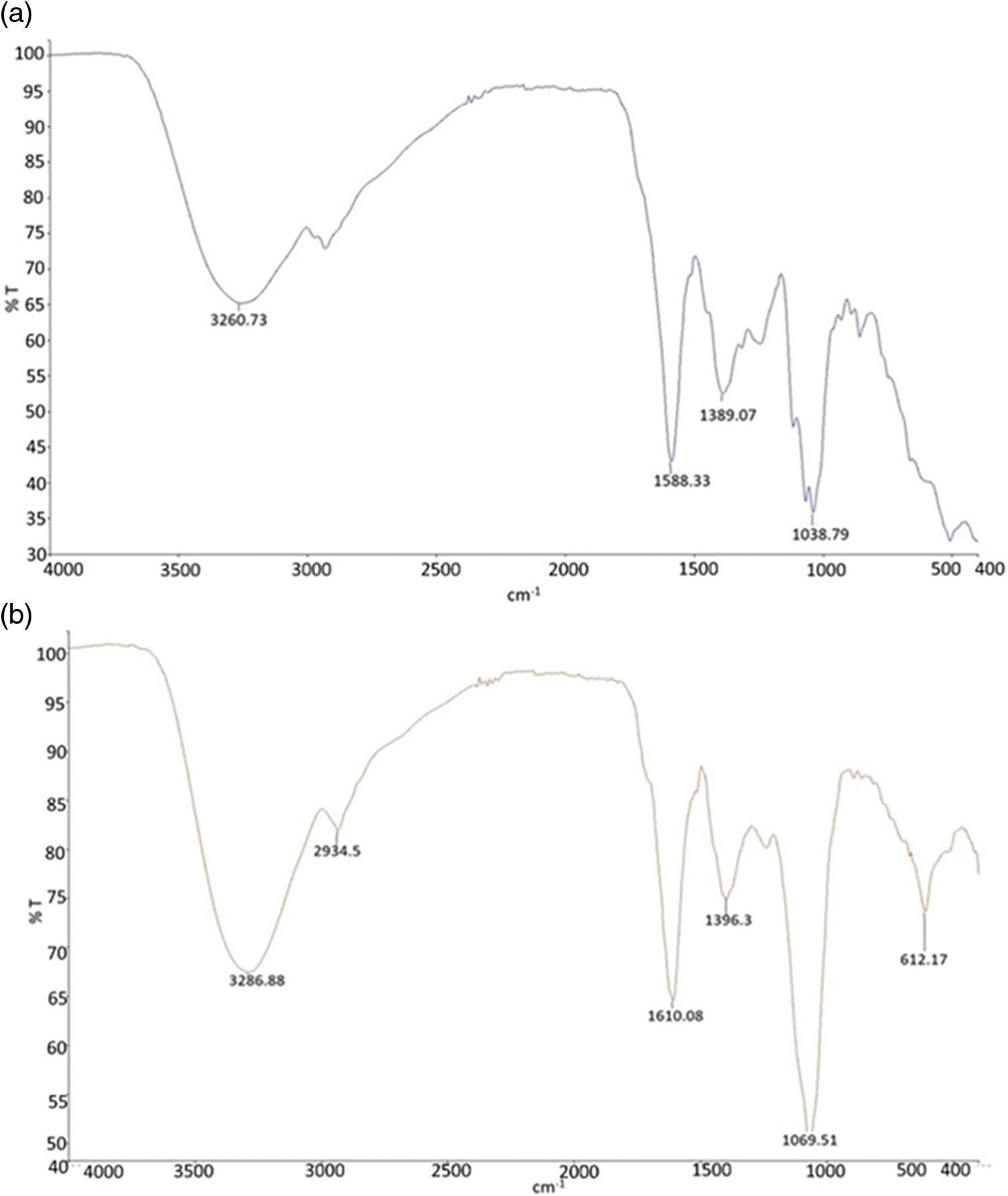
Figure 4: FTIR spectra of aqueous extract (a) and Cu/Zn-NPs (b), extracted from C. longirostrata
However, these bands were not evident with that of Cu/Zn-NPs. In the IR spectrum of Cu/Zn-NPs, exhibited a variation in the size of bands in region 3286, 2934, 1610, 1396, 1069 and 612 cm−1 (Fig. 4b). In this sense, 3286 cm−1 corresponded O–H (hydroxyl group) and 2964 cm−1 for C–H stretching of alkanes. The band at 1610 cm−1 corresponded to formation of C=C double bond of alkenes and the band 612 cm−1 can be related a reduction of o C–H bending of alkynes during the formation of nanoparticles [22].
3.3 Insect Mortality of P. solenopsis
The results of the mortality test for Cu/Zn-NPs are showed in Fig. 5. The applications of Cu/Zn-NPs show significantly changes in the insect mortality with respect to application of Cu/Zn solution and C. longirostrata extract. For example, insect treated with Cu/Zn-NPs the mortality was of 63% after 96 h of treatment. In contrast, insect treated with bimetallic solution (Cu/Zn) at 10 mM and plant extract did not show significant toxicity at any time interval. Although the toxicity in this insect by bimetallic nanoparticles has been reported previously.
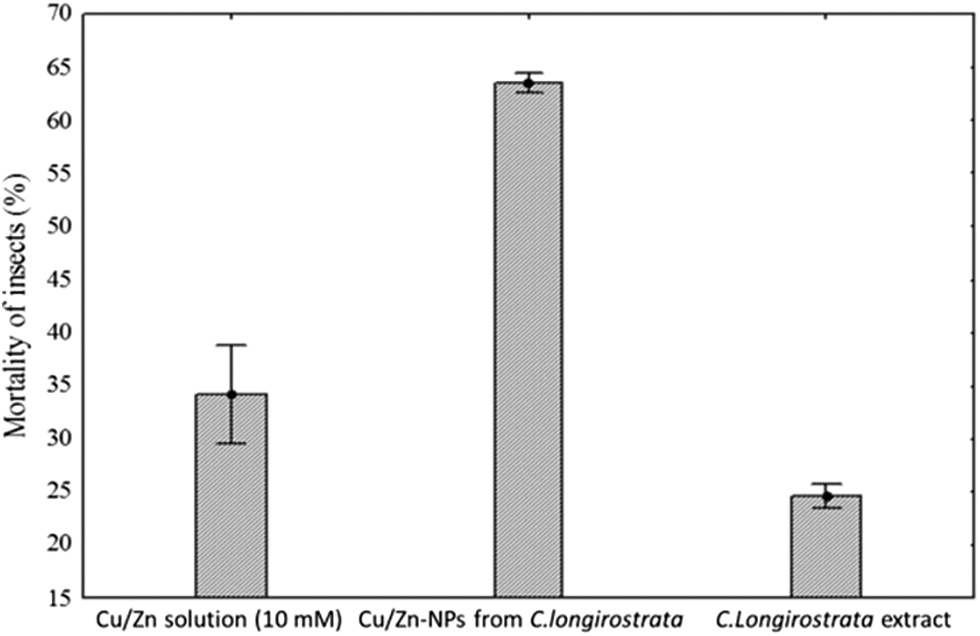
Figure 5: Insect mortality of Phenococcus solenpsis after 96 h exposure with Cu/Zn-NPs from C. longirostrata
Our study showed that Cu/Zn nanoparticles from C. longirostrata were more effective than reported by Mendez-Trujillo et al. [15]. The presence of diverse metabolites in the different plants enhances the toxicity, functional and chemical reactivity of Cu/Zn nanoparticles [23]. Additionally, Minal et al. [16] mentioned that the formulation of different bimetallic nanoparticles combinations possess distinct properties as compared with their monometallic forms. Kumaravel et al. [24] observed that bimetallic zinc oxide and titanium dioxide nanoparticles from Metarhizium anisopliae show pupicidal and antifeedant activity against Spodoptera frugiperda an important pest of several crops. According to Benelli [25], several studies have been realized to evaluate the toxic potential of nanoparticles against a wide number of arthropods pest. However, information on the mechanisms of action of nanoparticles against insects such as P. solenopsis is limited. In this sense, our results suggest that Cu/Zn-NPs have a significant impact on the stability of membrane (cell viability) of P. solenopsis that might be attributable to a distortion of the lipid layers of the membrane proteins by exposition to bimetallic nanoparticles (Figs. 6 and 7). Similar results were reported by Leon-Jimenez et al. [22] and Mendez-Trujillo et al. [15] who observed a decreasing cell viability by the generation of reactive oxygen species (ROS) in P. solenopsis treated with bimetallic (100 ppm) and monometallic nanoparticles (300 ppm).
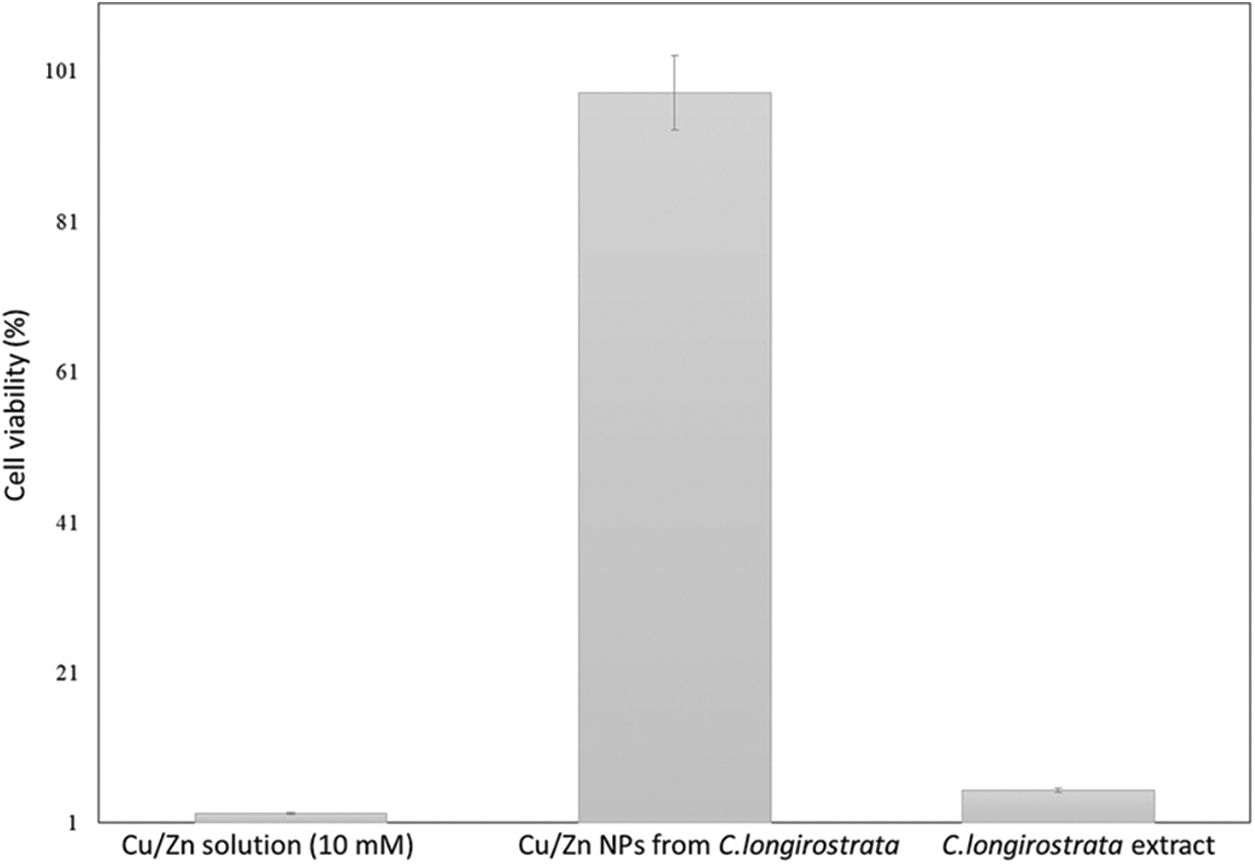
Figure 6: Cell viability of P. solenopsis after 96 h exposure with Cu/Zn-NPs from C. longirostrata
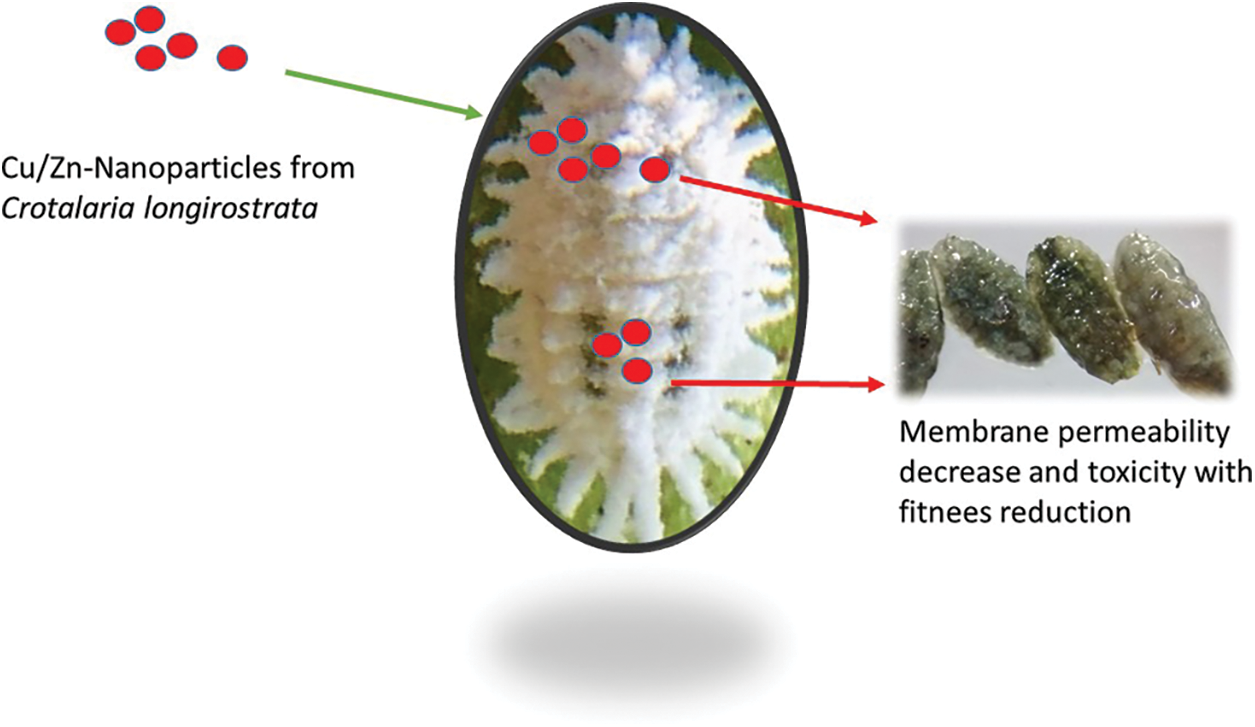
Figure 7: Possible mode action of Cu/Zn nanoparticles from C. longirostrata in P. solenopsis
In our investigation, we successfully biosynthesized NPs-Cu/Zn using the C. longirostrata extract and characterized the NPs using UV–vis spectroscopy, SEM-EDX, FT-IR, and DLS analyses. We found that the biosynthesized NPs exhibited high efficacy against P. solenopsis Tinsley (Hemiptera: Pseudococcidae), an invasive insect pest of multiple crops in the world. These promising results could represent a crucial step toward developing efficient nanoinsecticides for sustainable plague control of this and other insects. Further studies about nanotoxicity of Cu/Zn-NPs from C. longirostra are needed to elucidate the mechanism of toxicity of this nanoparticles in P. solenopsis.
Acknowledgement: The authors gratefully acknowledge Instituto de Ingenieria de la Universidad Autonoma de Baja California for the XRD, EDS, FESEM and FTIR analysis.
Funding Statement: The authors would like to thank Universidad Autonoma de Baja California.
Conflicts of Interest: The authors declare that they have no conflicts of interest to report regarding the present study.
1. Singh, J., Kumar, S., Alok, A., Upadhyay, S. K., Rawat, M. et al. (2019). The potential of green synthesized zinc oxide nanoparticles as nutrient source for plant growth. Journal of Cleaner Production, 214, 1061–1070. DOI 10.1016/j.jclepro.2019.01.018. [Google Scholar] [CrossRef]
2. Singh, J., Dutta, T., Kim, K. H., Rawat, M., Samddar, P. et al. (2018). Green synthesis of metals and their oxide nanoparticles: Applications for environmental remediation. Journal Nanobiotechnology, 16, 84. DOI 10.1186/s12951-018-0408-4. [Google Scholar] [CrossRef]
3. Boverhof, D. R., Bramante, C. M., Butala, J. H., Clancy, S. F., Lafranconi, M. et al. (2015). Comparative assessment of nanomaterial definitions and safety evaluation considerations. Regulatory Toxicology Pharmacology Journal, 73, 137–150. DOI 10.1016/j.yrtph.2015.06.001. [Google Scholar] [CrossRef]
4. Hoseini-Ghahfarokhi, M., Mirkiani, S., Mozaffari, N., Abdolahi Sadatlu, M. A., Ghasemi, A. et al. (2020). Applications of graphene and graphene oxide in smart drug/gene delivery: Is the world still flat? International Journal Nanomedicine, 15, 9469–9496. DOI 10.2147/IJN.S265876. [Google Scholar] [CrossRef]
5. Aryal, S., Park, H., Leary, J. F., Key, J. (2019). Top-down fabrication-based nano/microparticles for molecular imaging and drug delivery. International Journal Nanomedicine, 14, 6631–6644. DOI 10.2147/IJN.S212037. [Google Scholar] [CrossRef]
6. Jeyaraj, M., Gurunathan, S., Qasim, M., Kang, M. H., Kim, J. H. (2019). A comprehensive review on the synthesis, characterization, and biomedical application of platinum nanoparticles. Nanomaterials, 9, 1719. DOI 10.3390/nano9121719. [Google Scholar] [CrossRef]
7. Hussain, I., Singh, N. B., Singh, A., Singh, H., Singh, S. C. (2016). Green synthesis of nanoparticles and its potential application. Biotechnology Letter, 38, 545–60. DOI 10.1007/s10529-015-2026-7. [Google Scholar] [CrossRef]
8. Drummer, S., Madzimbamuto, T., Chowdhury, M. (2021). Green synthesis of transition-metal nanoparticles and their oxides: A review. Materials, 14, 2700. DOI 10.3390/ma14112700. [Google Scholar] [CrossRef]
9. Bernardo-Mazariegos, E., Valdez-Salas, B., González-Mendoza, D., Abdelmoteleb, A., Tzintzun-Camacho, O. et al. (2019). Silver nanoparticles from Justicia spicigera and their antimicrobial potentialities in the biocontrol of foodborne bacteria and phytopathogenic fungi. Revista Argentina de Microbiologia, 51, 103–109. DOI 10.1016/j.ram.2018.05.002. [Google Scholar] [CrossRef]
10. Namita, S., Soam, P. (2014). Green nanoparticles for mosquito control. The Scientific World Journal, 2014, 496362. DOI 10.1155/2014/49636. [Google Scholar] [CrossRef]
11. Pilaquinga, F., Morejón, B., Ganchala, D., Morey, J., Piña, N. et al. (2019). Green synthesis of silver nanoparticles using Solanum mammosum L. (Solanaceae) fruit extract and their larvicidal activity against Aedes aegypti L. (Diptera: Culicidae). PLoS One, 14(10), e0224109. DOI 10.1371/journal.pone.0224109. [Google Scholar] [CrossRef]
12. Murugan, K., Roni, M., Panneerselvam, C., Aziz, A. T., Suresh, U. et al. (2018). Sargassum wightii-synthesized ZnO nanoparticles reduce the fitness and reproduction of the malaria vector Anopheles stephensi and cotton bollworm Helicoverpa armigera. Physiological and Molecular Plant Pathology, 101, 202–213. DOI 10.1016/j.pmpp.2017.02.004. [Google Scholar] [CrossRef]
13. Camarillo-Castillo, F., Mangan, F. X. (2020). Biological nitrogen fixation in chipilin (Crotalaria longirostrata Hook. & Arn.a sustainable nitrogen source for commercial production. Revista Chapingo Serie Horticultura, 26, 125–141. [Google Scholar]
14. Miranda-Granados, J., Chacón, C., Ruiz-Lau, N., Vargas-Díaz, M. E., Zepeda, L. G. et al. (2018). Alternative use of extracts of chipilín leaves (Crotalaria longirostrata Hook. & Arn) as antimicrobial. Sustainability, 10, 883. DOI 10.3390/su10030883. [Google Scholar] [CrossRef]
15. Mendez-Trujillo, V., Valdez-Salas, B., Carrillo-Beltran, M., Curiel-Alvarez, M. A., Tzintzun-Camacho, O. et al. (2019). Green synthesis of bimetallic nanoparticles from Prosopis juliflora (Sw) DC., and its effect against cotton mealybug, Phenacoccus solenopsis (Hemiptera: Pseudococcidae). Phyton-International Journal of Experimental Botany, 88, 269–275. DOI 10.32604/phyton.2019.07316. [Google Scholar] [CrossRef]
16. Minal, S. P., Prakash, S. (2020). Laboratory analysis of Au-Pd bimetallic nanoparticles synthesized with citrus limon leaf extract and its efficacy on mosquito larvae and non-target organisms. Scientific Reports, 10(1), 21610. DOI 10.1038/s41598-020-78662-y. [Google Scholar] [CrossRef]
17. Merugu, R., Gothalwal, R., Deshpande, P. K., Mandal, S. D., Padala, G. et al. (2021). Synthesis of Ag/Cu and Cu/Zn bimetallic nanoparticles using toddy palm: Investigations of their antitumor, antioxidant and antibacterial activities. Materials Today: Proceedings, 44, 99–105. DOI 10.1016/j.matpr.2020.08.027. [Google Scholar] [CrossRef]
18. Ruiz-Romero, P., Valdez-Salas, B., González-Mendoza, D., Mendez-Trujillo, V. (2018). Antifungal effects of silver phytonanoparticles from Yucca shilerifera against strawberry soil-borne pathogens: Fusarium solani and Macrophomina phaseolina. Mycobiology, 46, 47–51. DOI 10.1080/12298093.2018.1454011. [Google Scholar] [CrossRef]
19. Raj, S., Chand Mali, S., Trivedi, R. (2018). Green synthesis and characterization of silver nanoparticles using Enicostemma axillare (Lam.) leaf extract. Biochemical and Biophysical Research Communications, 503, 2814–2819. DOI 10.1016/j.bbrc.2018.08.045. [Google Scholar] [CrossRef]
20. El-Saadony, M. T., Abd El-Hack, M. E., Taha, A. E., Fouda, M., Ajarem, J. S. et al. (2020). Ecofriendly synthesis and insecticidal application of copper nanoparticles against the storage pest Tribolium castaneum. Nanomaterials, 10, 587. DOI 10.3390/nano10030587. [Google Scholar] [CrossRef]
21. Vinayagam, R., Varadavenkatesan, T., Selvaraj, R. (2018). Green synthesis, structural characterization and catalytic activity of silver nanoparticles stabilized with Bridelia retusa leaf extract. Green Processing and Synthesis, 7, 30–37. DOI 10.1515/gps-2016-0236. [Google Scholar] [CrossRef]
22. Leon-Jimenez, E., Valdez-Salas, B., Gonzalez-Mendoza, D., Tzintzun-Camacho, O. (2019). Synthesis and insecticide activity of Cu-nanoparticles from Prosopis juliflora (Sw) DC and Pluchea sericea (Nutt.) on Phenacoccus solenopsis Tinsley (Hemiptera: Pseudococcidae). Revista Sociedad Entomologica Argentina, 78, 12–21. DOI 10.25085/rsea. [Google Scholar] [CrossRef]
23. Gonzalez-Mendoza, D., Valdez-Salas, B., Bernardo-Mazariegos, E., Tzintzun-Camacho, O., Ruiz-Vladiviezo, V. et al. (2019). Influence of monometallic and bimetallic phytonanoparticles on physiological status of mezquite. Open Life Sciences, 20(14), 62–68. DOI 10.1515/biol-2019-0008. [Google Scholar] [CrossRef]
24. Kumaravel, J., Lalitha, K., Arunthirumeni, M., Shivakumar, M. S. (2021). Mycosynthesis of bimetallic zinc oxide and titanium dioxide nanoparticles for control of Spodoptera frugiperda. Pesticide Biochemistry and Physiology, 178, 104910. DOI 10.1016/j.pestbp.2021.104910.E. [Google Scholar] [CrossRef]
25. Benelli, G. (2018). Mode of action of nanoparticles against insects. Environmental Science and Pollution Research, 25, 12329–12341. DOI 10.1007/s11356-018-1850-4. [Google Scholar] [CrossRef]
 | This work is licensed under a Creative Commons Attribution 4.0 International License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |