

 | Journal of Renewable Materials |  |
DOI: 10.32604/jrm.2022.018142
REVIEW
A Review of Various Sources of Chitin and Chitosan in Nature
1Higher Institution Center of Excellence (HICoE), Institute of Tropical Aquaculture and Fisheries, Universiti Malaysia Terengganu, Kuala Nerus, 21030, Malaysia
2Department of Fisheries and Aquaculture, Joseph Sarwuan Tarka University Makurdi, Makurdi, 2373, Nigeria
*Corresponding Authors: Benedict Terkula Iber. Email: benedictiber@gmail.com; Nor Azman Kasan. Email: norazman@umt.edu.my
Received: 01 July 2021; Accepted: 06 August 2021
Abstract: Chitin was first discovered by its name from the Greek word “chiton”, which means “mail coat”. It is indeed a polysaccharide made up of naturally occurring acetyl-D-glucosamine monomers. Hatchett was the first researcher who extracted chitin from the shells of mollusks (crabs and lobsters), prawns, and crayfish in 1799. Later in 1811, Henri Braconnot discovered chitin in the cell walls of mushrooms and called it “fungine”. Chitin and chitosan are abundant in the biosphere as essential components of many organisms’ exoskeletons and as by-products of the global seafood industry. The biopolymer must be deacetylated before chitosan can be produced. It can also be extracted using microbes in a biological extraction procedure. The development of products that take advantage of the bioactivities of the existing primary commercial source of chitin (crustacean) has lagged expectations. Also, the disadvantages of the present commercial source such as seasonality and competition for other uses among others has been one of the driving forces towards seeking alternative sources of chitin and chitosan in nature. This review highlights some of the efforts made by environmental scholars to locate possible commercial sources of chitin and chitosan in nature over time.
Keywords: Chitin; chitosan; crustaceans; insects; fungi
Due to wastewater production and the manufacture of polymeric materials through different industrial processes, industrial development has posed numerous environmental challenges [1]. Water and land contamination may result from wanton dumping of this waste (with or without adequate treatment) and the polymer. Not withstanding the heinous negative effects on soil and water [2] this impacts negatively on the wellbeing of living creatures [3]. This has been a major source of concern at all levels of government because it poses a danger to humanity’s survival.
Around the world, up to 140,106 tonnes of synthetic polymers are produced each year. On the other hand, these synthetic polymersare fairly stable and their biodegradation is minimal [4]. This requires the use of biodegradable polymers that are considered as environmentally friendly. Chitin and chitosan are two biopolymers that have piqued the interest of researchers and industrial players. This is as a result of their various possible utilization in bioengineering, agriculture, papermaking, food industry, and textile products [5]. Chitin and chitosan have a wide spectrum of uses due to their abundance as well as their non poisonous effects and good biocompatibility [1,4]. Other synthetic polymers such as nylon, polyethylene, polyester, teflon, and epoxy do not have many applications and therefore may not draw quite so much interest. As a result, research on the potential uses of biopolymers is highly recommended.
Biopolymers such as chitin and chitosan enter the world via a variety of pathways, one of which is crustaceans, a group of invertebrates. Around 6.1 million tonnes of crustaceans are caught in both coastal and inland waters [6]. Other than crustaceans, food intake from the sea will result in waste amounting up to 40–50 percent of total weight [7]. Chitin and calcium carbonate are found in about 40% of the waste produced [8]. Gortari et al. [9] found out that this volume of chitin contained in waste is a good source for chitin and chitin products. Unfortunately, most of these large quantities of chitin-containing wastes end up in the environment as pollutants [10]. Since most studies focus on marine water sources, there are few reports on the amount of these same types of waste generated by freshwater bodies.
Chitin and chitosan have piqued the interest of several scholars, who have published numerous studies on their origins and production. An immense number of living creatures produce these polymers [11–13]. Chitin and chitosans are abundant in the biosphere as essential components of many organisms’ exoskeletons and as waste from global seafood industries [14–16]. Numerous studies have been conducted in the last 20 years, highlighting the growing interest in chitin and chitosan production from crustacean shell [17,18], insect cuticles, Melolonthamelolontha [19,20], Orthoptera species [21], wings of cockroach [22], grasshopper species [22], medicinal fungus [18], larvae and adult of Colorado potato beetle aquatic invertebrates [18], bat guano [19], resting eggs of Daphnia longispina [18], spider species [18], Daphnia magna resting eggs [23], lastly in green algae and fungi cell wall. The yield of chitin and chitosan as well as physical and chemical properties were, however, reported to be dependent on the source used. This review observed, however, that none of these approaches by these researchers have made it to the industrial scale.
Chitosan is a cationic polymer gained by deacetylation of chitin that is particularly abundant in crustacean, bug, arthropod exoskeletons, and mollusk exoskeletons [24]. Similarly, Shaala et al. [25] and Ehrlich et al. [7] stated that deproteinization, demineralization, and decoloration are all steps in the chemical extraction platform for isolating chitin. Chitin must be deacetylated to produce chitosan. These polymers can also be processed using a biological extraction process that employs microbes. Based on their specific application, chitosan and its variants can be used as gels, beads, membranes, films, or sponges [24,26]. Reusing waste from the shipping industry is not recommended, and a substantial proportion of waste biomass is dumped directly into the atmosphere without being treated [27]. The seafood industry produces about 106 tonnes of waste per year, where the majority of which is composted or turned into low-value-added products such as animal feed and fertilizers. Approximately 2000 tonnes of chitosan are processed per year, with most of the material coming from shrimp and crab shell remnants [28,29].
The primary large scale source of chitin and chitosan are reportedly exoskeleton of crustacean, which collect as a massive waste material from the sea food industry [20,30]. However, the research questions which need to be addressed are:
1. What are the examples of species of crustaceans suitable for chitin and chitosan production?
2. Are there other sources of chitin and chitosan aside crustaceans that could be explored on a commercial basis?
3. How do we identify some of the shortcomings like seasonality in supply, environmental contamination, and inadequate supply inherent in some of the reported sources of chitin and chitosan?
Thus, this review focuses on addressing the above questions in addition to a brief insight into the history and structure of chitin and chitosan as well as their chemical differences.
2 History of Chitin and Chitosan
Chitin (C8H13O5N) comes from the Greek word “chiton”, which means “coat of mail”. It’s a polysaccharide made up of-(1-4)-N acetyl-D-glucosamine monomers that exist in nature. Hatchett extracted chitin from the shells of mollusks (crabs and lobsters), prawns, and crayfish using mineral acids for the first time in 1799 [31]. Hatchett observed that they had a mild effervescence but were flexible and plastic of a yellowish nature, similar to cartilage, and that they retained their initial form after a short period of time.While this is the first description of calcified chitin in invertebrates, chitin was firstly identified in 1811 by a chemist Henri Braconnot, who called it “fungine” after discovering it in the cell walls of mushrooms. Odier [32] who extracted a hornlike substance after treating cockchafer elytra with potassium hydroxide about thirty years before the discovery of cellulose, renamed fungine as chitin in 1823 [33,34].
Chitosan, a major of chitin variant, was reported approximately 40 years after chitin was identified. Chitosan’s origins can be traced back to Charles Rouget’s study in 1859, when he produced the material by heating chitin in an alkaline medium. However, Felix Hoppe-Seyler coined the word “chitosan” in 1894, and the chemical structure was discovered only in 1950 [35]. Even after this earlier detection, industrial chitin and chitosan processing and commercialization began in the 1970’s [20,36]. Chitosan has piqued scientific and industrial curiosity in the United States, Korea, China, Canada, Norway, Australia, France, the United Kingdom, Poland, and Germany since the late 1970’s dueto its distinctive macromolecular structure, biocompatibility, biodegradability, and other fundamental chemical composition [37,38]. In 2015, Japan experienced significant growth which controlled about 35% of the global market. Undoubtedly, Japan has made significant progress in the technology, commercialization, and application of these biopolymers since the 1980’s [39,40]. This chitosan industry consumes about 700–800 tonnes per year [41].
Despite their versatility, accessibility, and marketability in many countries of the world, chitin and chitosan have only been industrially isolated from crustaceans and by extension, fungi [20]. Over one thousand billions tones of chitin have been reported annually in the biospher [42]. After its discovery, chitin and chitosan have witnessed tremendous research works as a result of their diverse biological, chemical, and physical characteristics and uses [12,37]. For the many years, the chief source of chitin and chitosan has been crab and shrimp shell. Nevertheless, more scientific works sprung up when it was revealed that the isolation of chitin and chitosan from the cell wall of fungi generated less waste and little allergenic substances. This also opened doors for numerous biotechnological applications [4,43].
3 Structure of Chitin and Chitosan
Due to the extreme identical chemical backbone, chitin is frequently confused with cellulose, however, rather than a hydroxyl group, chitin has acetamide groups (CH3CONH2) at the carbonyl group (Fig. 1) [44]. Its crystalline nature consists of poly (1, 4)-linked N-acetyl-2-amino-2-deoxy-D-glucose (GlcNAc) with some 2-amino-2-deoxy-D-glucose remnants, it has white, nitrogenous, non-elastic, and tough material properties. Due to their huge abundance as waste from the marine industry worldwide, chitin and chitosan are considered as significant marine renewable materials. Chitin abundance is projected to be over 10 billion tonnes per year [16,45,46].

Figure 1: Structure of chitin (A) and chitosan (B) [47]
α-chitin, β-chitin, and γ-chitin are three crystalline allomorphic types of chitinthat can be found in nature. Anti-parallel chains are found in α-chitin, where it is the major form and accountable for the stiffness of the polymer. Most of crab and shrimp shells contain α-chitin [48]. On the other hand, β-chitin is made up of parallel chains that form monocyclic crystals with intramolecular (hydrogen bonds) as well as intermolecular interactions [10]. For instance, diatom spines, squid pens, and pogonophoran tubes are all known to contain β-chitin. γ-chitin is a combination of parallel and anti-parallel chains with properties similar to α-chitin and β-chitin which can be found in fungi, yeasts, and insect cocoons [22,49,20].
The chitinous materials obtained from crustaceans, invertebrates, and arthropods, particularly some possessing tough outer parts are typically in the α-shape. This is due to the anti-parallel nature of the chains, which makes the arrangement relatively compact due to greater and thus more stable hydrogen bonding. Furthermore, β-form chains are parallel, and the majority of chitin sources for this β-form come from mollusks such as squid pens [50]. Chitin with the γ-shape is motlypresent in insect cocoons which have two parallel strings and one anti-parallel string. It is possible to convert from the α-form to the β-form, but not the other way around. It has been discovered that the crystalline, purity and polymer chain composition of chitin isolated from various sources are varies depending on the source and the proportion of chitin inherent there in [51].
Chitin is pellucid, flexible, elastic and very strong in its natural state. In very many arthropods, nevertheless, it is frequently modified, and it is often found as part of a mixed substance, like sclerotin, a tanned protein-like matrix that makes up most of an insect’s exoskeleton. Chitin makes an even tougher composite when fused with calcium carbonate (CaCO3), as seen in crustacean and mollusk exoskeleton. This composite material is stronger and less fragile than pure CaCO3, smoother and more rigid as compared to unmixed chitin. Another clear distinction between mixed and unmixed type of chitin could be seen when comparing caterpillar and beetle. While the former’s body wall is supple, the later’s is non-pliable and posseses a greater amount of sclerotin [52–55].
In another study, chitin and chitosan resemble heparin, chondroitin sulphate, and hyaluronic acid, all of which are biologically essential mucopolysaccharides in all mammals. Because of the substituent carboxyl and sulfuryl groups, these mucopolysaccharides are anionic polymers [51]. Chitosan, however, is perhaps the only cationic polysaccharide found naturally, it is safe and non-toxic and biodegradable in humans [56]. In terms of biomedical applications, this unique property is worth noting. Chitosan’s biomedical use is restricted, nevertheless, because it is insoluble in neutral or simple aqueous media. The chemical modification of chitosan produces substances that dissolve easily at pH 7 and above. This helps to bind other functional groups and regulates other essential properties that are cationic, hydrophobic and anionic in nature [51].
α-Chitin has also been found in the cell walls of yeast and fungal organisms, shrimp shells, insect cuticles, crab as well as lobster’s tendons and shells [57,58]. According to Kaur et al. [59], polysaccharide chains in chitin are organized in an antiparallel manner, allowing for maximal bonds. Consequently, α-chitin is by far the most composite structure of chitin in existence, leading to high crystallinity chitin fibrils (80 percent). β-Chitin can be found in squid pens as well as the tubes produced by vestimentiferan worms and pogonophorans. It can also be found in aphrodite chaetae and lorica, which are formed by protozoa or sea grasses. Diatom, Thalassiosirafuviatilis has also been reported to discharge a more pure type of β-chitin in its monocrystalline spine. Polymer chains are also linked together to form arrangement in β-chitin, and the crystallinity index of chitin fibrils is about 70%. This type is much more sensitive and dissolves in solvents due to the greater gap across neighboring polymer chains [60]. γ-Chitin is made up of a mixture of α-and β chitin-type configurations, where two parallel polymer chains alternate with one antiparallel chain.
Chitosan’s physical and chemical structure has also been revealed. It is a linear polysaccharide made up of deacetylated and acetylated d-glucosamine units connected by (1, 4) glycosidic bonds, as earlier shown in Fig. 1 [16,46]. Deacetylation of chitin produces acetate ions and a −NH2 group, which is derived by hydrolysis of the acetamide group. The degree of deacetylation in chitosan is usually defined by the ratio of glucosamine to N-acetyl glucosamine. Chitin has a greater proportion of N-acetyl glucosamine than glucosamine, whereas chitosan has a larger proportion of glucosamine than N-acetyl glucosamine [61,62]. Depending on the experimental circumstances, the degree of deacetylation affects chemicalproperties (e.g., tensile strength, solubility, surface area, viscosity, conductivity, porosity, and flexibility) as well as biological properties (e.g., adsorption enhancer, biodegradability, antioxidant, bioavailability, and biocompatibility) [63,64].
In comparison to commercial biopolymers, chitosan from shrimp shell displayed superior antibacterial activity against Gram negative bacteria, Escherichia coli [65]. Chitosan has also been identified as a linear polysaccharide made up of randomly distributed b-(1-4)-linked D-glucosamine and N-acetyl-Dglucosamine extracted by minimal deacetylation of chitin [11,65]. Chitin and chitosan also have a high nitrogen content, ranging from 2% to 8%, making them more appealing to a variety of industrial applications [66,67]. Due to its solid structure and strong intra and intermolecular hydrogen bonds, chitin is immiscible with water and most organic solvents. On the other hand, chitosandissolves readily in acidic solutions (e.g., hydrochloric acid, citric acid, and acetic acid) with a pH below 6 [68,42]. Chitin’s stiffness and insoluble condition have restricted its uses in comparison to chitosan, which is more elastic and has a higher solubility.
4 Chemical Differences Influence the Application of Chitin and Chitosan
Chitin and its main constituent, chitosan, are the most efficient and promising aquatic biopolymers on the planet, and their physicochemical properties vary depending on their source and extraction process, [16,69]. Since there is no specific procedure for chitin production, several studies have been performed to produce natural chitin without altering the biopolymer’s physicochemical properties [9,10].
Chitin’s physicochemical properties have had a major impact on its uses. Because of its acetyl groups, chitin has minimal applications; however, chitin can be transformed into chitosan via the deacetylation process [70]. The acetyl group in chitin is transformed into hydroxyl (-OH) and amino (-NH2) groups in chitosan during the deacetylation process. The ability to modify the reactive functional groups in chitosan opens the door to a wide range of applications [71,72]. Chitin has a low solubility and is insoluble in most different solvents. In comparison to chitosan, chitin’s solubility challenge limits its application and uses [6,10].
Furthermore, chitin is a glycosidic polymer with a significant degree of polymerization, multiple intramolecular and intermolecular hydrogen bonds, semi-crystalline structure, and nearly complete acetylation [70]. Chitin is made using well-known chemical or biotechnology production routes that begin with fish waste, which is an inexpensive and renewable material. Furthermore, chitin purification produces calcium carbonate-or protein-rich side streams which can be subsequently refined, boosting chitin manufacturing economics [45]. On the other hand, chitosan is a chitin product with a smaller specific weight and acetylation degree that is primarily formed by chemical conversion of chitin. The transformation reaction involves the use of a highly concentrated alkaline solution at an elevated temperature, making the process non-sustainable. Since the acetamido groups are configured in a trans-configuration with respect to the hydroxyl group at C3, harsh conditions are necessary [73].
In order to develop a more sustainable chitosan processing method, the enzymatic deacetylation of chitin under mild reaction conditions is recommended. The possibility of a cost-effective manufacturing process leads to the expectation that chitosan will have a brighter future than chitin, particularly in the treatment of effluent water [74]. In contrast to chitin, Younes and Rinaudostated that chitosan has a broader range of applications [75]. This is due to the fact thatamine grouphad high abundance and availability, as well as their lower intermolecular forces and acidic aqueous solubility. The low solubility of chitin, which results in less leaching and hence more frequent use in all media is indeed perculiar to it. However, processes and fictionalizations for increasing chitosan tolerance in acidic media have been defined: chitosan can be used as a powder, flakes, or gel in membranes or beads. Freeze-drying, ionotropic gelation, neutralization, crosslinking, and solvent evaporation are some of the gel preparation methods. At least one of the last four approaches includes measures that end in a stronger chitosan derivative, composite, or blend [76]. Notwithstanding, in acidic media, chitosan could be used as a condensed solution, allowing for homogeneous modification reactions or heavy metal adsorption at the free amine functionalities.
Similarly, the chitin’s dense acetyl groups act as an electrostatic repulsion, preventing reagents from properly approaching nitrogen. Chitin’s ineffective adsorption of heavy metal ions has been demonstrated [77,78]. Besides that, as in chitosan, the higher the degree of deacetylation, the higher the density of available primary amine groups, which are primarily responsible for the electrostatic interaction. Chitosan’s higher proportion of amorphous regions improves availability and sorption capability, as well as making it ideal for a versatile linker between different colloids in coagulation/flocculation process [79,77].
5 Preparation of Chitin and Its Subsequent Conversion to Chitosan
After cellulose, chitin is the most abundant natural polysaccharide which is predominantly found in crustaceous shells or cell walls of fungi, crustaceans, mollusks, arthropods, and some seaweed (Fig. 2) [50,67]. Nevertheless, it has not been commonly used for industrial purposes because it is insoluble across many solvents, and it is comparatively difficult to separate in pure state from natural sources and to process in a repeatable manner within favorable economic conditions [35].

Figure 2: Sources of chitin and chitosan
In most cases, the production of chitin and chitosan comes under 4 basic processing steps. These include: deproteinization, demineralization, decolorization and deacetylation. The deproteinization involves the removal of protein using a strong base, while demineralization entails a step when calcium carbonates and calcium phosphates are removed using a strong acid. Furthermore, the removal of pigment and conversion of chitin to chitosan are referred to as decolorization and deacetylation respectively. Decolorization may be achieved using an alcohol e.g ethanol, while deacetylation process is accomplished using a strong alkali such as NaOH (Fig. 3) [35].

Figure 3: Conversion of chitin to chitosan [47]
Wang et al. [80] and Yang et al. [81] had presented a technique for deproteinizing crustacean chitin wastes using microorganisms or proteolytic enzymes (Fig. 4). This allows for a more cost-effective processing of chitin and chitosan. The alkaline deacetylation of chitin with a powerful alkaline solution is the most common technique for extracting chitosan. The raw material is usually smashed, rinsed with water or detergent, and then chopped. Since the mineral content of the exoskeleton from different sources varies, alternative metrics can be used [82].

Figure 4: Standard protocols for chitin and chitosan extraction
Regardless of the source, deacetylation of chitin to yield chitosan is a challenging reaction due to chitin’s low solubility across most solvents, necessitating large amounts of sodium hydroxide and heat. Chitin deacetylation usually happens in tandem with polymer chain depolymerization, reducing exposure to high molecular weight chitosan [83]. Di Nardo [83] amorphizes commercially obtained chitin by milling it in a zirconia milling jar with a zirconia ball, which allows for easier access to the N-acetyl groups for deacetylation. The deacetylation was activated by homogeneously mixing the amorphized chitin with sodium hydroxide in a polytetrafluoroethylene jar with a zirconia ball. An ageing method using humidity-controlled chambers of 43, 75, and 98 percent relative humidity was used because it was presumed that excessive moisture content would enable the depolymerization of chitin/chitosan. This makes it possible for a regulated water supply during the deacetylation step, reducing depolymerization [83]. This process has been shown to be flexible and appropriate to a variety of chitin sources, such as shrimp shell, crab, and lobster, fly larva, and fly larva treated with 1-ethyl-3methylimidazolium acetate [84].
Chitin was efficiently isolated from pupae shells and adult black soldier fly (BSF) via demineralization, deproteinisation, and decoloration techniques [31]. By using a deacetylation process, the extracted chitins were efficiently transformed into chitosan. By adjusting the concentration, voltage, flow rate, and tip-to-collector range, commercial chitosan was electrospun into nanofibers. Fourier-transform infrared spectroscopy (FTIR) was used to analyse the spectral patterns and peaks relating to the stretching and vibrations of different functional groups, X-ray Powder Diffraction (XRD) to analyze the crystalline structure, Scanning electron microscope (SEM) to investigate the morphology, and Thermogravimetric Analyser (TGA) to study the thermal stability of the synthesised and fabricated materials. To quantify the level of acetylation and deacetylation, an elemental analysis was performed. To evaluate the consistency of the extracted substances, commercial shrimp chitin and chitosan were matched. The structure, crystallinity, and thermal stability of electrospun chitosan nanofibers were compared to bulk chitosan to determine how they had changed following the electrospinning phase.The highest optimized parameters for demineralisation and deproteinisation were 1 M hydrochloric acid (HCl), 100 min and 50°C for demineralisation and 1 M sodium hydroxide (NaOH), 10 h and 85°C for deproteinisation, accordingly, yielding the maximum final dry weight yields of 13% and 5% for pupae shells and adult BSF chitin. The chitins with a degree of acetylation of 115.1 percent for pupae shells and 91.5 percent for adult BSF are of suitable quality, according to the findings by elemental analysis. Furthermore, both chitosans with a deacetylation level of 67 percent for pupae shells and 69 percent for adult BSF are pure enough [85]. In contrast to most crustacean sources, the BSF is ideal for chitin and chitosan processing; it is not periodic in availability. The BSF pupa has a high yield, making it a high-potential commercial source of chitin and chitosan.
Chitosan is typically produced from shrimp shell waste at two deacetylation temperatures (90°C and 40°C) [86]. As opposed to chitosan processed at 40°C with a degree of deacetylation of 88 percent and a molecular weight of 353 KDa, the chitosan extracted at 90°C had greater physicochemical characteristics. Even though this process generated chitosan with a high degree of acetylation (DA), the high amount of heat needed made it unsustainable. Thus, more research is required to come up with a method for producing high-quality chitosan which needs less energy.
Other attempts have been made to establish more sustainable methods for chitin and chtosan preparation. Chitin can be converted to chitosan through an enzymatic or chemical process [87,88]. Chemical processes, on the other hand, are widely used for commercial chitosan production due to their suitability for large scale production. Alkali or acid are used to deacetylate chitin in the chemical process of deacetylation. As glycosidic bonds are sensitive to acid, alkali is suggested as a better option [89]. Chitin deacetylationprocess can be carried out in a heterogeneous or homogeneous condition. In the heterogeneous phase, chitin is subjected to a strong alkaline NaOH solution for a few hours, resulting in an insoluble chitosan filtrate in the form of 85%–99% deacetylated chitin. Alkali chitin is made using the homogeneous method, which involves discharging chitin in a concentrated NaOH solution at 25°C for 3 h. At 0°C, the alkali chitin is suspended in ice cubes. This approach typically resulted in the development of soluble chitosan with an average DA of 48–55 percent. This method produces chitosan with a deacetylation degree of 100% and homogeneously dispersed acetyl groups along the chains after 58 h [88]. According to Aiba [90], the deacetylation reaction resulted in an unequal distribution of d-glucosamine and N-acetyl-d-glucosamine residues along polymeric chains within heterogeneous conditions.
6 Previous Attempts towards Uncovering Potential Sources of Chitin and Chitosan
6.1 Chitosan from Crustacean Sources
Numerous sources of crustacean chitin have been identified for decades and are still being discovered. Chemical treatments of shrimp shells were effective in obtaining chitin and chitosan. The impacts of various particle sizes (50–1000 mm) of the raw material on particle size, demineralization, deproteinization, and deacetylation of chitin, as well as the chitosan extraction method, were investigated. To extract chitin, a particle size range of 800–1000 mm was chosen. When compared to commercial biopolymers, the extracted chitin and chitosan from shrimp shell exhibit great antibacterial activity against Gram negative bacteria, Escherichia coli [17,18,64].
The primary source of large scale chitin and chitosan today comes from marine orgsnism waste, mostly crustacean exoskeletons [8]. In 2016, the total worldwide supply of crustaceans for human utilization was projected to be 8 million tones [91], with 40% of waste exoskeletons [92] containing 15–40% chitin [93]. Nevertheless, since industrial exploitation of crustacean begins in the spring after the spawning season, this makes the supply of fishery waste seasonal [94]. Furthermore, the long-term viability of crustacean cultivation is presently being debated [92,95].
Turunen et al. [96] found that the efficacy of biopolymer-based natural coagulants/flocculants (e.g., tannin, starch, chitosan) in the treatment of two related agricultural sewage with varying levels of phosphorus contamination and turbidity. At doses of 5–10 mL/L, tannin and chitosan (C6H11NO4)n from crustacean shells (e.g.;crabs and prawns) coagulants ranked the highest. Chitosan is a biopolymer that has been shown in wastewater treatment to incorporate the characteristics of both coagulants and flocculants over time. This implies that the polymer’s cationic properties can destabilize suspended colloids in solution, forcing them to coagulate and flocculate, resulting in increased flocs that can be filtered out.
According to a related study, chitin is typically obtained from the exoskeleton of crustaceans, especially shrimps and crabs, where α-chitin is yielded [97]. A more deacetylable form of chitin is the β-chitin. An essential source of this form of chitin is squid. Owing to the much weaker intermolecular hydrogen bonding as a result of the parallel structure of the main chains, β-chitin exhibit greater solubility, reactivity, affinity for solvents, and swelling than α-chitin [97,98]. The development of chitosan from crustacean shells collected as food waste is cost-effective, particularly if carotenoids are retrieved [82].
Many researchers of chitin and chitosan have shown that crustaceans are the first choice as far as commercial sources of the biopolymer are concerned. The exoskeletons of mud crabs from Abbottabad were demineralized with 2 mol/dm3 sulphuric acid (H2SO4) solution for 4 h and then deproteinized with 2mol/dm3 NaOH solution for 4 h at room temperature in hopes of finding a method of preparation and sources of chitin and chitosan from crustaceans. The output of raw chitin was 78 percent, and chitosan was obtained by deacetylating it with a 55 percent NaOH solution at 110°C for 4 h. The output of pure chitosan from the crab shell was 39% after precipitation [8,19,99]. Likewise, Ali et al. [100] described shrimp, crabs, and lobsters as main sources of chitin and chitosan, but noted that the properties of chitosan varied depending on the degree of deacetylation and molecular weight. Arbia et al. [8] reported that the amount of chitin differs from one crustacean organism to another, estimating that chitin extracted from crustacean crabs ranges from 60 to 70 percent. This review found significant differences in the methods for preparing chitin and chitosan among the various authors listed thus far. This has raised serious doubts about whether the effect on chitin and chitosan yields is due to the methodologies used rather than the differences in sources as stated by these scholars.
Many countries with longer coastlines are now encouraging commercial farming of crustacean animals, the majority of which live in the sea. Indonesia is one of these nations. In the fields of agriculture, plantation, mining, and fisheries, Indonesia is abundant in mineral resources. Indonesia’s geographical position also contributes to the vastness of the coastal region, resulting in enormous opportunities in the fishing field [101]. Shrimp is one of the plentiful capture fisheries in the Province of West Nusa Tenggara (NTB), particularly in Lombok. Shrimp use has thus far been restricted to the contents for eating, with the shell or skin being discarded, resulting in waste and environmental contamination. The production of chitosan followed the usual conventional process of demineralization, deproteinization and deacetylation using HCL and NaOH, respectively [82,95]. The shaped chitosan weighed 0.1 gram (as many as five samples). Chitosan can be produced from compounds that contain chitin, such as crustaceans [101].
Shrimp trading has a significant social and economic effect in Brazil, and its production produces a lot of garbage [102]. During processing, shrimp body is peeled and the head removed. This result to over fourty percent lost in the total weight of the shrimp. The processing and disposal of these shells is a perfect illustration of the fishing industry’s pollution. Because of their foul odor and the insects they draw, the shells cause environmental concerns. Furthermore, due to the extreme untreated waste that is dumped, this condition can endanger human health. Chemical and microwave approaches were used to obtain chitin and chitosan from Pacific whiteleg shrimp, Litopenaeusvannamei garbage. The study found that a particle size of 32 mesh and six 5-min pulses were successful in deacetylation with a degree of 92 percent and a chitosan yield of 52.2 percent [102–104]. According to Elieh-Ali-Komi et al. [69], the exoskeleton of crustaceans posses varying degrees of chitin, protein, calcium carbonate, phosphate, lipid and ash; however, the proportion of these parameters differ with species (Table 1).
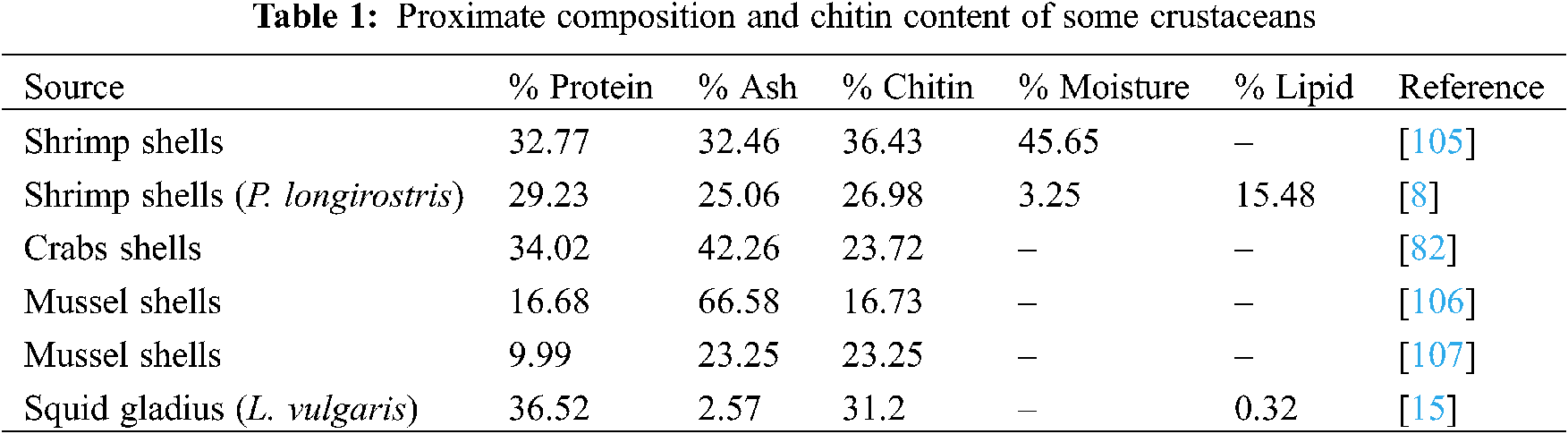
Some authors have started to recognize freshwater crustacean organisms to step away from the marine crustacean sources of chitin and chitosan that have gained so much attention so far. The freshwater crab is one of these species. Potamonalgerienseis a freshwater crab that belongs to the Potamidae family, and is the biggest of all freshwater crab families, with 95 genera and over 505 species [10,108,109]. P. algeriense is only present in three countries in North Africa: Morocco, Algeria, and Tunisia [108]. In Morocco, this species has indeed been recorded from the Oued Laou watershed near Chefchaouen, the watershed of Moulouya in the northeast, and the Oued OumRbia watershed near Khenifra in the Middle Atlas. Despite its widespread penetration, P. algeriense has yet to be commercially assessed [109]. For the first time, a standardized and updated chemical process is used to remove and characterise chitin from the shell of the freshwater crab species Potamonalgeriense [24]. Up to 62.12 percent of the ground shell was extracted after cooking, drying, and crushing. After demineralization of field crab shell, a harvest of 40.92 percent was achieved, though after deproteinization, a yield of 8.74 percent was gained. After decolorization, 8.27 percent of raw chitin was produced, with 5.89 percent of chitosan derived from crab shells [24,110,111].
Finally, another report on the crustacean source of chitin and chtosan worth mentioning in this review is on snow crab, Chionoecetes opilio. The shell waste of the snow crab and the northern prawn,Pandalus borealis was believed to harbor 17–32.2 percent of chitin [112,113,108,109,114–116]. The chitin content of blue crab has been estimated to be 14 percent. TheGrey shrimp, Crangoncrangon contains 17.8 percent chitin, whereas the speckled shrimp, Metapenaeusmonoceros did contain 4.5–7 percent chitin [113]. Water fleas, tadpole shrimp, fairy shrimp (branchiopods), copepods, seed shrimp (ostracods), scuds (amphipods), water sow bugs (isopods), shrimp, crayfish, clams, and crabs are some of the crustaceans found in inland water bodies. This review discovered that only a few of these species, such as shrimp, prawns, and crabs, have been explored for chitin and chitosan production thus far. More research on other numerous species of freshwater crustaceans is therefore encouraged to identify other potential commercial sources of chitin and chitosan.
6.2 Chitosan from Fungal Sources
Braconnot [117] detected chitin from Agaricusvolvaceus, Agaricus acris, Agaricuscantarellus, Agaricuspiperatus, Hydnumrepandum, Hydnumhybridum, and Boletus viscidus [118]. Knorr [33] succeeded in making and isolating chitin and chitosan from the cuticles of insects and mushrooms during the nineteenth century. Hu et al. [119] devised a procedure for extracting fungal chitosan [42]. This procedure outlines the steps required for chitin production [119]. Among the fungal classes studied for chitin and chitosan manufacturing are Absidiacoerulea, Absidia glauca, Absidiablakesleeana, Mucor rouxii, Aspergillus niger, Phycomycesblakesleeanus, Trichoderma reesei, Colletotrichum lindemuthianum, Gongronellabutleri and Pleurotussajo [120–122]. The quantity of chitin in the fungal cell wall varies depending on the species, climatic circumstances, and age of the fungus, [4].
In their study, Abdel-Gawad et al. [95] also stated that the breakthrough in the discovery of chitin was sequel to an experiment carried out on some fungi such as Agaricusvolvaceus, Acris cantarellus, Acris piperatus, Hydnumrepandum, H. hybridum, and Boletus viscidus. The fungi were made pure by bubbling through potassium hydroxide (KOH). This process was reported to aid the removal of pigments and proteinaceous substances [4]. This study, which was published in English by Children (1824), gained high public recognition. In another development; Lassaigne [123] purified Coleopteran elytra and Bombyx mori exuviae, and subjected the residue through potassium at hot conditions, yielding potassium cyanide, which although dangerous, unambiguously revealed the existence of nitrogen in chitin [1]. The authors further confirmed the absence of chitin in higher mamals also emphasized that although Knorr [33], came up with the term “chitin”, he did not revealits identity.
Further research has been conducted in order to demonstrate the true identity of chitin visa-vis fungi, and a few discoveries have been reported. Chitin is a distinctive constituent of the mushroom taxonomic groups Zygomycetes, Ascomycetes, Basidiomycetes and Deuteromycetes [44]. The Food and Agriculture Organization of the United Nations revised the global mushroom manufacturing in 2009. China is the largest producer (>1.5 million metric tonnes in 2007, with a 65 percent increase in ten years) [95]. The United States and Canada came in second and third place, with 390,000 and 81,500 tonnes, respectively. Due to public knowledge about the health advantages of mushrooms in terms of prebiotics and polysaccharides, Israel and India demonstratedbest outputs over the aforementioned decade [124]. The most widely planted mushroom for chitin production globally are Auricula bisporus (button mushroom), followed by Lentinus edodes (shiitake), Pleurotusspp (oyster mushrooms), Auricula auricula (wood ear mushroom), Flamulinavelutipes (winter mushroom), and Volvariellavolvacea (straw mushroom) [125]. Kalac [126] and Muzzarelli et al. [124] evaluated the chemical properties and nutritional value of European species of wild mushrooms. Even though fungi are a good source of chitin, recent research advances in their capability as a raw material source to produce essential medicinal products such as vitamins, pigments, lipids, glycolipids, polysaccharides, and polyhydric alcohols have also made them less appealing as a commercial source of chitin and chitosan. Fungi such as Saccharomyces crevisiae can also be used in the production of leavened bread and fermented juices.
According to Jardine et al. [127], chitin and chitosan are predicted to hit 4.2 billion US dollars by 2021 in the world market. These authors further added that chitin has so far shown a compound yearly growth rate of only fifteen percent. This has necessitated the development of new sources of chitin to overcome the rising demand. Fungi are the second most common source of chitin after crustaceans. Several studies have concentrated on fungi and their economic value, as they have gained popularity as a vegan alternative to chitin and chitosan [95]. Chitin accounts for 1 to 15% of the weight of fungal cell walls and its composition is similar to that of crustaceans [128]. Chitin is commonly represented in different fungal phyla like Basidiomycota, Ascomycota, and Zygomycota, despite the fact that it is not found in all fungi [128]. To extract chitosan from fungal chitin, comparable to crustacean chitin, extreme conditions are needed. Chitosan, on the other hand, can be extracted directly from the cell wall of certain fungal species without the need for acetyl group cleavage. Absidia spp. (zygomycetes), A. niger (ascomycetes), rouxii (zygomycetes), Rhizophusoryzae (zygomycetes), and Lentinus edodes (basidiomycetes) are among the most studied species for direct chitosan processing [121,122,129,130]. Despite this, industrial-scale processing of fungal chitin and chitosan is yet to be achieved [96].
In light of their (chitin and chitosan) rising demand, the need for industrial production of fungal species to satisfy other conflicting priorities as well as provide an adequate quantity for chitin and chitosan production has arisen. Fungal biopolymers have several characteristics that make them superior to biopolymers derived from marine waste. Chitin and chitosan are not found in all fungal species’ cell walls. Chitin and chitosan are found in the cell walls of Basidiomycetes, Ascomycetes, Zygomycetes, and Deuteromycetes, among other fungi [4,5]. Furthermore, fungal chitin and chitosan will provide non-seasonable and reliable raw material sources as well as consistent product characteristics [13].
Chitin is commonly found in several fungi groups, namely Ascomycetes, Basidiomycetes, and Phycomycetes [4]. Fungal chitin is also found in the functional membranes and cell walls of mycelia, stalks, and spores. Nevertheless, has been reported absence in some fungi, and may also be missing in similar species. Chitin has been recognised as a main element in primary septa in both mother and daughter cells in Saccharomyces cerevisiae [131]. A few mushroom species have been highly toxic and lethal including eathcap (Amanita phalloides), Death cap fungus (Conocybefilaris, Conocybefilaris),Webcaps (Cortinarius species), Webcap mushroom, Autumn skullcap (Galerinamarginata) mushroom, Destroying angels (Amanita species), Podostromacornu-damae (Lepiotabrunneoincarnata) [132]. Despite the fact that these species are not edible and may be less competitive in their use, their utilization as a possible commercial source of chitin should be approached with caution due to their highly poisonous nature.
To emphasize the causes of the increasing demand for fungal chitin and chitosan, Abdel-Abdel-Gawad et al. [95] stated that chitosan produced by fungi has outstanding physico-chemical characteristics to that produced by conventional crustacean sources. Chitosan was extracted from Aspergillus niger biomass under various alkali deproteinization environments, and the effect of alkali concentration, temperature, and time on chitosan output, degree of deacetylation (DD), and molecular weight was investigated using a Box-Benhken experimental design (MW). Various sets of processing factors resulted in chitosan with various physico-chemical characteristics. The highest performance combinations of responses were then predicted using the desirability function. The best resolution permitted for chitosan recovery with a yield of 7.0 percent (w/w), DD 83.64 percent, and MW 2.70–104 Da, all of which were close to the expected amounts.
Due to the extreme seasonal and restricted supply of crustacean shells, the expensive demand, and ambiguous physico-chemical characteristics such as protein contamination, inconsistent levels of deacetylation, and high molecular weight, the conventional method of chitin and chitosan production has restricted potential for industrial adoption [133]. Fungal chitosan has been the subject of recent studies [134]. Chitosan produced from fungi developed within controlled circumstances has a higher chance of being reliable. The material sources of fungal chitin are easily accessible at any time of the year. These biomass materials may be harvested at low cost by simple fermentation. It is customary with crustacean and other sources of chitin to undergo deminerization to obtain chitosan. However, Dhillon et al. [134] reported that production of chitosan from fungal sources does not require deminerization process. These authors further added that only mild treatment with alkali and acid is enough to obtain good quality chitosan thereby making it a green process.
Fungal chitosan also contains no allergenic shrimp protein and has a medium-low molecular weight with greater bioactivity. The methods for producing chitosan from fungi, on the other hand, have not been expanded to an industrial standard. Rhizopus oryzae, Mucor rouxii, Absidia glauca, Aspergillus niger, Gongronellabutleri, Pleurotussajor-caju, and Lentinus edodes have all had their mycelium studied as a potential source of chitosan [84]. A. niger biomass was used in research to optimise chitosan output. Numerous efforts to grow fungi solely for the purpose of maximising chitosan performance have been made, with yields ranging from 2.0 percent to 12.7 percent depending on a variety of variables such as fungal strain, fermentation process, temperature, and culture media composition [95].
6.3 Chitosan from Insect Sources
Insects have long been regarded as a reliable food source, with 2 billion people worldwide eating 1900 diverse species of insects for human nutrition [135]. Southeast Asia, the Pacific, Sub-Saharan Africa, and Latin America represent major insect consumers. Insects contain 30–45 percent protein, 25–40 percent fat, and 10–15 percent chitin in total [30].
According to a study by Spranghers et al. [30], between 1998 and 2020, nearly 67 scientific studies with the phrases “insect chitin and chitosan” were printed and classified in scientific journals and databases. Most of the studies were conducted in Turkey (28 percent), China (24 percent), and South Korea (7%), accounting for 59 percent of all reported scientific studies, whereas 4% of the studies were conducted in Egypt, Iran, Russia, and Brazil, and 3% of the research was conducted in Japan, Poland, Malaysia, and India. Just only one study was found in Mexico, Spain, Slovakia, Italy, Thailand, Bulgaria, and Belgium [132].
The chitin polymer has been discovered in the integuments of arthropods, nematodes, and mollusks, as well as the gut linings of insects, as a building element. Chitin is normally extracted from the exoskeletons of arthropods’ chitin-based tissue with 30 percent to 40 percent protein, 30 percent to 50 percent calcium carbonate, and 20 percent to 30 percent chitin [136]. Tenebrio molitor chitosan was contrasted to crustacean chitosan (Penaeus monodon) for antimicrobial capability against various pathogenic microorganisms of interest in food security. Pathogenic bacteria, Salmonellasp and was found to be by far the most resistant bacteria, and insect-derived chitosan was less effective than crustacean-derived chitosan, particularly against Salmonellasp [137,138]. Insects are a credible alternative source of chitin, but due to scarcity, they have not been used in the past. Insect cultivation, on the other hand, is currently being heavily conducted for the long-term production of animal feed. Huge quantities of insect biomass and chitin-rich industrial byproducts such as exuviae and exoskeletons are becoming more widely available [95].
Deacetylation of chitin, which is found in the exoskeletons of crustaceans and insects, as well as in the cell walls of many of these fungi and some algae, is used to make chitosan on a large scale [139]. Though its main source of chitin is crab, prawn, crayfish, and shrimp residues [139], insect chitosan is essential because insects serve as a reliable source of protein [138]. This means that commercial insect processing will not only solve the problem of having a reliable source of chitin and chitosan, but it will also solve the problem of having a reliable source of chitin and chitosan. It will also deal with the issue of malnutrition in human diets.
In particular, a variety of studies have been conducted using insects to demonstrate their viability as chitin and chitosan sources (Table 2). In several parts of the world, the Field cricket, Gryllusbimaculatus is eaten as a delicacy [64]. Nowak et al. [65] characterised the chitosan obtained from Field crickets and to determine its possible use in the increasing functional market, the molecular mass of Field cricket chitosan was less than that of industrial chitosan. Nevertheless, the DA, FTIR, and XRD spectra, on the other hand, were all quite close. The Field cricket chitosan had a better particle size (208.27 3.47 nm), zeta potential (35.72 1.29 mV), and polydispersity index (PDI: 0.27 0.03) than the industrial chitosan. Chitin makes up about 5.1 percent of the dry weight of crickets,. About 2.04 g of chitin was obtained from 40 g of dried cricket powder as a raw material, yielding a chitosan yield of 41.75 percent [64]. Based on the species, the chitin level in crustacean shells used for industrial chitin ranged from 7 to 40% [140]. As a result, if the output could be increased, Field cricket (G. bimaculatus) would be a viable choice for commercial purposes as a source of chitin and chitosan [64].
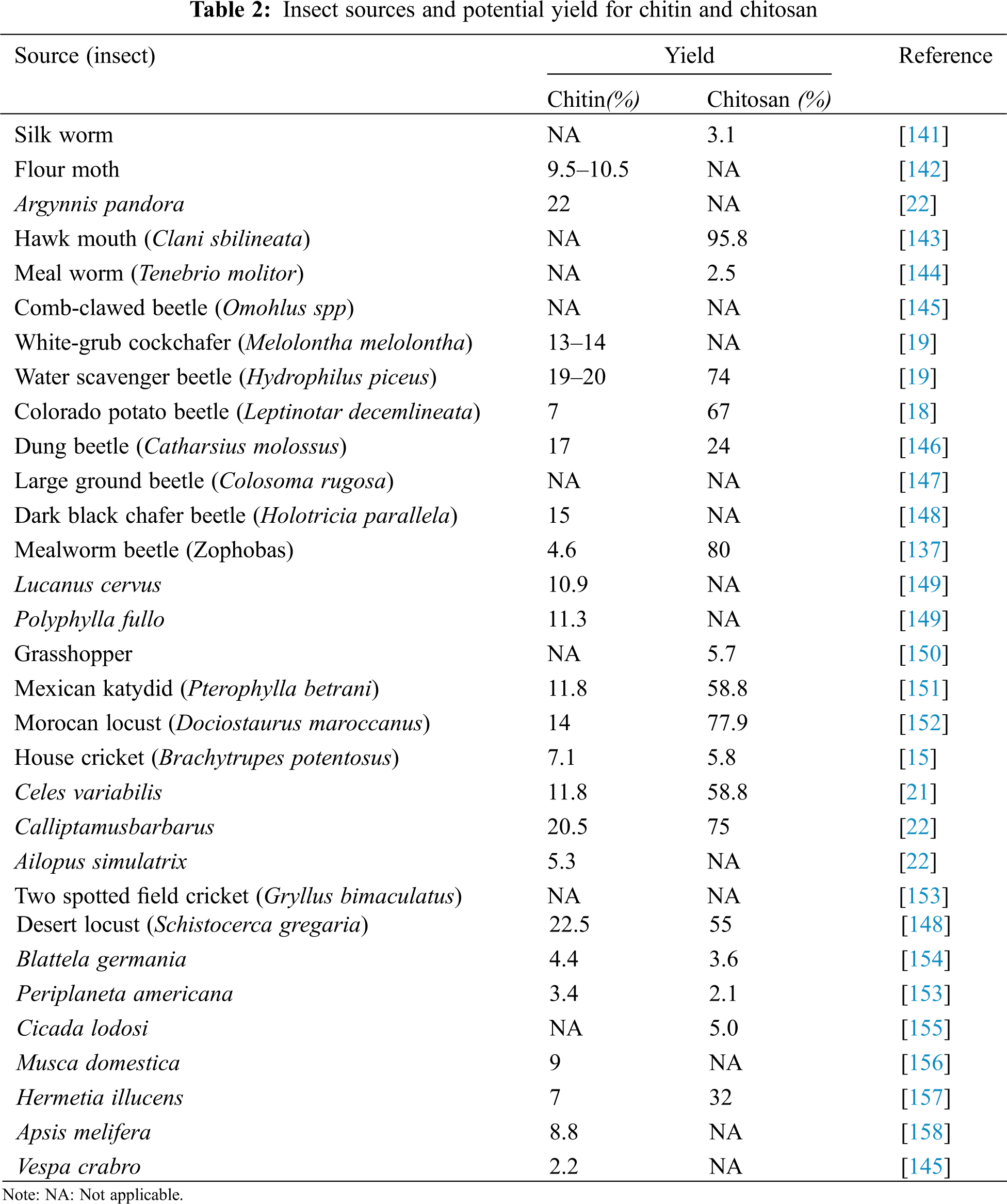
Insects are also a viable and reliable source of chitin and chitosan, even though little attention has been given to them so far [159]. Insects have certain benefits over crustaceans in that they are not seasonal and can be readily breed due to their high fertility and reproductive rate [150]. Additionally, insect breeding centres are springing up all over the world [135]. Insects can be used as a viable option for crustaceans as a source of chitin and chitosan for larger ecological and economic sustainability as bioconverterswhich arereared for organic waste management and animal feed processing [160]. Centipedes [18,159] and woodlice [19,145] have been studied as chitin sources. Wide centipede body parts are well suited to the creation of three-dimensional chitin rings [145]. Chitin has also been isolated from poriferans [161], bryozoans [21], tardigrades [162], and insectivorous bat guano [19,20,162].
A new source of insect chitin has been suggested in a new study by Jang et al. [49]. For demineralization and deproteinization, 1M HCl and 1M NaOH were used to extract insect chitin from cicada sloughs. The oxidising agent, 6 percent sodium hypochlorite, was used to extract the brown colour of this chitin from cicada sloughs. The insect chitin isolated from cicada sloughs was known to have a greater percentage yield than the chitin obtained from rice-field crab shells. Crab, shrimp, and krill shells, which are waste from the production of ocean food, are the conventional commercial sources of chitin. Chitin has lately been collected from unusual sources, such as insects and fungal mycelia [148,158,152,163,164]. Despite this, few studies on the preparation and characterization of insect chitin have been conducted.Crab and shrimp chitin has been the subject of most previous research. Regrettably, research on the composition of insect chitin has just recently been conducted on the cuticle of beetles and the pupa exuvia of silkworms (Bombyx mori) [164]. Other insect chitin sources include honeybees (Apis mellifera) [158], bumblebees (Bombus terrestris), scorpions, worms, brachiopods, ants, and cockroaches [163], scorpions, spiders, brachiopods, ants, and cockroaches [165,149,166], the chitin monomer of the silkworm had a molecular weight of C8H13O5N = 203 [47].
Further research into the comparative advantages of insect-derived chitin and chitosan over conventional sources reveals that chitin is a major biopolymer among the different components in insects [156], and that extracting chitin and chitosan from insects is more beneficial in terms of extraction techniques, chemical consumption, time, and output as compared to traditional sources [151]. Conversely, the amount of chitin in each species varies depending on its life cycle. Adult Tenebrio molitor and Hermetiaillucens species generate up to 5% chitin, while pre-pupa/pupa stages of black soldier flies, Tebo worms, Turkestan cockroaches, and house flies produce 21, 11.1, 6.7, and 11.9 g/kg of chitin [167].
According to Huet et al. [168], chitin of various purity grades (45 percent, 89.7%, and 93.3 percent) were efficiently extracted and thoroughly physicochemicallycharacterised from Bombyx eri larvae.The recorded information revealed that insect chitins had identical crystallographic structures, thermal stability, and degree of acetylation (>87 percent) to commercially accessible and isolated-chitin from shrimp shell. The crystallinity indexes (66 percent vs. 75 percent for shrimp chitin) and morphological structures were the most significant variations. Insects are a useful protein source that is easily accessible for human and animal consumption, making them a promising substitute or supplement to feed the world’s rising population [169]. In this context, the United Nations’ Food and Agriculture Organization (FAO) and the European Community unanimously implemented the use of insects as animal biomass for food and feed (farm fish), promoting insect farming as a new source of proteins [135,170]. This development can thus be used by commercial chitin and chitosan manufacturers to identify possible sources of chitosan from a variety of insect classes.
Insects are eaten and a component of the diets of at least 2 billion people, with over 1900 insect species recorded and used for human consumption [135,171], especially in Southeast Asia and the Pacific, Sub-Saharan Africa, and Latin America [135,171,172]. It is worth noting that insect chitin contains less calcium carbonate (6%) than crustacean chitin (30–50%), allowing for easier extraction and a more eco-sustainable process [173]. Over the last few years, numbers of scientific papers have been published on the processing of chitin and subsequent conversions into bioproducts or biomaterials from various insect species and sections of their bodies [19,21,22,48,147,153,174,175].
Some microalgae species can possess chitin and even chitosan in their cell walls [23,176,177,178]. As a result, Żółtowska-Aksamitowska et al. [179] developed a screening method for precisely detecting each of these polymers among the large variety of microalgae species in Greenaltech’s library in order to test these hypotheses.Native chitins and chitosans have been discovered in many genera according to this screening process. The isolation and physicochemical characterization of chitins and chitosans from Chlorella sp. were used to support the technique. Simultaneously, the enzymatic deacetylation mechanism underlying Chlorella’s natural development of chitosan was investigated, with a focus on identifying active chitin deacetylases. Eventually, the antimicrobial, wound healing, and nanocapsule-forming properties of microalgal chitosans were investigated as part of an exploratory biofunctional analysis. These activities are demonstrated the utility of microalgal chitin [179].
Other experiments investigating the green microalgae and yeast cell walls [180,177], marine diatoms, coralline algae, and mollusks by chemical processes using strong acids and bases [7,24,25,111,133], also yielded positive results of chitin and chitosan. These sources of chitin and chitosan outperformed crustacean chitin and chitosan. However, unlike the extraction of chitin and chitosan from algae sources mentioned so far in this study, no other methods other than chemical processes have been attempted.
Finally, fish scales have recently gained recognition as a possible source of chitin and chitosan [177]. Organic chitosan generated from fish scales and food-grade chitosan were studied as possible corrosion inhibitors on ASTM A36 iron in a 3.5 percent NaCl solution using the potentiodynamicpolarisation process. The analysis found that food-grade chitosan and chitosan synthesised from fish scales also prevented the corrosion of ASTM A36 iron in a 3.5 percent NaCl solution. The chitosan’s action and ability were unaffected by the source of the chitosan. The two forms of chitosan had an effective level of 3000 ppm [181]. This review shows that the use of fish scale in the development of chitin and chitosan is a field that has yet to receive adequate attention. Largeamount of wastes containing various types of fish scales, which are typically produced by fish processing industries, may be used as commercial sources of chitin. Additionally, it is necessary to determine which fish species and scale types are the best sources of chitin and chitosan.
Despite the fact that chitin and chitosan have been dubbed our “last biomass resource” and are anticipated to usher in a new usable polymer, their application and exploration are limited. Several fascinating biological activities have been identified over time; functional implementation tends to lag. One of the chief factors for this is that these biological processes are not exclusive to chitin and chitosan; they occur in other materials as well. The second explanation is cost, as chitosan is relatively costly (20–30 US dollars per kilogramme). If a specific biological activity was discovered to be particular to chitosan materials, practical use, notwithstanding the expense, would be promoted, particularly for biomedical applications.
Although fungal sources of chitin and chitosan were the first to be found in history, crustacean chitins are still the most commercially available. Despite the commercial existence of crustacean chitin and chitosan, research has shown that insect sources of chitin are a more promising source of the substance. Finally, the production of products that take advantage of the bioactivities of the current main commercial source of chitin and chitosan (crustacean) has tended to lag behind projections, according to this review study. The key explanation for this is that the biological effects of chitosans are difficult to reproduce due to a harsh chemical deacetylation process that alters the structure of the polysaccharides, causing variation. Another explanation is the immune responses that these polymers can elicit when traces of crustacean proteins are present. As a result, it is important to collect chitin and chitosan from non-animal sources using a more eco-sustainable and reliable method.
Acknowledgement: The authors would like to acknowledge Long Term Research Grant Scheme (LRGS/1/2018/USM/01/1/1)(LRGS/2018/USM-UKM/EWS/01) granted by Ministry of Higher Education Malaysia for funding this research project.
Funding Statement: This study is funded by the Long Term Research Grant Scheme (LRGS/1/2018/USM/01/1/1)(LRGS/2018/USM-UKM/EWS/01) granted by Ministry of Higher Education Malaysia for funding this research project.
Conflicts of Interest: The authors declare that they have no conflicts of interest to report regarding the present study.
1. Islam, S., Bhuiyan, M. A. R., Islam, M. N. (2017). Chitin and chitosan: Structure, properties and applications in biomedical engineering. Journal of Polymers and the Environment, 25(3), 854–866. DOI 10.1007/s10924-016-0865-5. [Google Scholar] [CrossRef]
2. Chhonkar, P. K., Datta, S. P., Joshi, H. C., Pathak, H. (2000). Impact of industrial effluents on soil health and agriculture-Indian experience: Part I–Distillery and paper mill effluents. Journal of Scientific and Industrial Research, 59(5), 350–361. [Google Scholar]
3. Gautam, S., Saini, G. (2020). Use of natural coagulants for industrial wastewater treatment. Global Journal of Environmental Science and Management, 6(4), 553–578. DOI 10.22034/gjesm.2020.04.10. [Google Scholar] [CrossRef]
4. Abo Elsoud, M. M., El Kady, E. M. (2019). Current trends in fungal biosynthesis of chitin and chitosan. Bulletin of the National Research Centre, 43(1), 59. DOI 10.1186/s42269-019-0105-y. [Google Scholar] [CrossRef]
5. Akila, R. M. (2014). Fermentative production of fungal chitosan, a versatile biopolymer (perspectives and its applications). Advances in Applied Science Research, 5(4), 157–170. [Google Scholar]
6. FAO (2010). Part 1: World review of fisheries and agriculture. The State of World Fisheries and Agriculture, Marine Policy, 36(3746–752. DOI 10.1016/j.marpol.2011.10.021. [Google Scholar] [CrossRef]
7. Ehrlich, H., Shaala, L. A., Youssef, D. T., Żółtowska-Aksamitowska, S., Tsurkan, M. et al. (2018). Discovery of chitin in skeletons of non-verongiid Red Sea demosponges. PLoS One, 13(5), e0195803. DOI 10.1371/journal.pone.0195803. [Google Scholar] [CrossRef]
8. Arbia, W., Arbia, L., Adour, L., Amrane, A. (2013). Chitin extraction from crustacean shells using biological methods–A review. Food Technology and Biotechnology, 51(1), 12–25. [Google Scholar]
9. Gortari, M. C., Hours, R. A. (2013). Biotechnological processes for chitin recovery out of crustacean waste: A mini-review. Electronic Journal of Biotechnology, 16(3), 12–25. DOI 10.2225/vol16-issue3-fulltext-10. [Google Scholar] [CrossRef]
10. Ni’mah, Y. L., Harmami, H., Ulfin, I., Suprapto, S., Welny Saleh, C. (2019). Water-soluble chitosan preparation from marine sources. Malaysian Journal of Fundamental and Applied Sciences, 15(2), 159–163. DOI 10.11113/mjfas.v15n2.971. [Google Scholar] [CrossRef]
11. Rinaudo, M. (2006). Chitin and chitosan: Properties and applications. Progress in Polymer Science, 31(7), 603–632. DOI 10.1016/j.progpolymsci.2006.06.001. [Google Scholar] [CrossRef]
12. Bhuiyan, M. R., Shaid, A., Bashar, M. M., Haque, P., Hannan, M. A. (2013). A novel approach of dyeing jute fiber with reactive dye after treating with chitosan. Open Journal of Organic Polymer Mater, 3(4), 87–91. DOI 10.4236/ojopm.2013.34014. [Google Scholar] [CrossRef]
13. Yeul, V. S., Rayalu, S. S. (2013). Unprecedented chitin and chitosan: A chemical overview. Journal of Polymers and the Environment, 21(2), 606–614. DOI 10.1007/s10924-012-0458-x. [Google Scholar] [CrossRef]
14. Sampantamit, T., Ho, L., Lachat, C., Sutummawong, N., Sorgeloos, P. et al. (2020). Aquaculture production and its environmental sustainability in Thailand: Challenges and potential solutions. Sustainability, 12(5), 2010. DOI 10.3390/su12052010. [Google Scholar] [CrossRef]
15. El Knidri, H., Belaabed, R., Addaou, A., Laajeb, A., Lahsini, A. (2018). Extraction, chemical modification and characterization of chitin and chitosan. International Journal of Biological Macromolecules, 120, 1181–1189. DOI 10.1016/j.ijbiomac.2018.08.139. [Google Scholar] [CrossRef]
16. Casadidio, C., Peregrina, D. V., Gigliobianco, M. R., Deng, S., Censi, R. et al. (2019). Chitin and chitosans: Characteristics, eco-friendly processes, and applications in cosmetic science. Marine Drugs, 17(6), 369. DOI 10.3390/md17060369. [Google Scholar] [CrossRef]
17. A., M., Abdel-Rahman, R. M., Hrdina, R., Imramovsky, A., Burgert, L. et al. (2012). Antibacterial cotton fabrics treated with core-shell nanoparticles. International Journal of Biolological Macromolecules, 50(5), 1245–1253. DOI 10.1016/j.ijbiomac.2012.03.018. [Google Scholar] [CrossRef]
18. Kaya, M., Baran, T., Mentes, A., Asaroglu, M., Sezen, G. et al. (2014). Extraction and characterization of α-chitin and chitosan from six different aquatic invertebrates. Food Biophysics, 9(2), 145–157. DOI 10.1007/s11483-013-9327-y. [Google Scholar] [CrossRef]
19. Kaya, M., Baublys, V., Can, E., Šatkauskienė, I., Bitim, B. et al. (2014). Comparison of physicochemical properties of chitins isolated from an insect (Melolontha melolontha) and a crustacean species (Oniscus asellus). Zoomorphology, 133(2014), 285–293. DOI 10.1007/s00435-014-0227-6. [Google Scholar] [CrossRef]
20. Hahn, T., Roth, A., Ji, R., Schmitt, E., Zibek, S. (2020). Chitosan production with larval exoskeletons derived from the insect protein production. Journal of Biotechnology, 310, 62–67. DOI 10.1016/j.jbiotec.2019.12.015. [Google Scholar] [CrossRef]
21. Kaya, M., Baublys, V., Satkauskiene, I., Akyuz, B., Bulut, E. et al. (2015). First chitin extraction from Plumatella repens (Bryozoa) with comparison to chitins of insect and fungal origin. International Journal of Biolological Macromolecules, 79, 126–132. DOI 10.1016/j.ijbiomac.2015.04.066. [Google Scholar] [CrossRef]
22. Kaya, M., Lelesius, E., Nagrockaite, R., Sargin, I., Arslan, G. et al. (2015). Differentiations of chitin content and surface morphologies of chitins extracted from male and female grasshopper species. PLoS One, 10(1), e0115531. DOI 10.1371/journal.pone.0115531. [Google Scholar] [CrossRef]
23. Kaya, M. I., Sargin, K. Ö., Tozak, T., Baran, S., Erdogan, G. S. (2013). Chitin extraction and characterization from Daphnia magna resting eggs. International Journal of Biolological Macromolecules, 61, 459–464. DOI 10.1016/j.ijbiomac.2013.08.016. [Google Scholar] [CrossRef]
24. Klinger, C., Żółtowska-Aksamitowska, S., Wysokowski, M., Tsurkan, M. V., Galli, R. et al. (2019). Express method for isolation of ready-to-use 3D chitin scaffolds from Aplysina archeri (Aplysineidae: Verongiida) demosponge. Marine Drugs, 17(3), 131. DOI 10.3390/md17020131. [Google Scholar] [CrossRef]
25. Shaala, L. A., Asfour, H. Z., Youssef, D. T., Żółtowska-Aksamitowska, S., Wysokowski, M. et al. (2019). New source of 3D chitin scaffolds: The Red Sea demosponge pseudoceratinaarabica (Pseudoceratinidae, Verongiida). Marine Drugs, 17(2), 92. DOI 10.3390/md17020092. [Google Scholar] [CrossRef]
26. Wysokowski, M., Bazhenov, V. V., Tsurkan, M. V., Galli, R., Stelling, A. L. et al. (2013). Isolation and identification of chitin in three-dimensional skeleton of aplysinafistularis marine sponge. International Journal of Biological Macromolecules, 62, 94–100. DOI 10.1016/j.ijbiomac.2013.08.039. [Google Scholar] [CrossRef]
27. Wang, S. L., Nguyen, V. B. (2019). Production of potent antidiabetic compounds from shrimp head powder via Paenibacillus conversion. Process Chemistry, 76, 18–24. DOI 10.1016/j.procbio.2018.11.004. [Google Scholar] [CrossRef]
28. Muñoz, I., Rodríguez, C., Gillet, D., Moerschbacher, B. M. (2018). Life cycle assessment of chitosan production in India and Europe. International Journal of Life Cycle Assessment, 23(2018), 1151–1160. DOI 10.1007/s11367-017-1290-2. [Google Scholar] [CrossRef]
29. Santos, V. P., Marques, N. S. S., Maia, P. C. S. V., Lima, M. A. B., de Franco, L. et al. (2020). Seafood waste as attractive source of chitin and chitosan production and their applications. International Journal of Molecular Sciences, 21(12), 4290. DOI 10.3390/ijms21124290. [Google Scholar] [CrossRef]
30. Spranghers, T., Ottoboni, M., Klootwijk, C., Ovyn, A., Deboosere, S. et al. (2017). Nutritional composition of black soldier fly (Hermetiaillucens) prepupae reared on different organic waste substrates. Journal of the Science of Food and Agriculture, 97(8), 2594–2600. DOI 10.1002/jsfa.8081. [Google Scholar] [CrossRef]
31. Hatchett, C. (1799). Experiments and observations on shell and bone. In: Bowyer, W., Nichols, J. (Eds.Transactions of the royal society of London, vol. 89, pp. 315–334, London: Royal Society. [Google Scholar]
32. Odier, A. (1823). Mémoir sur la composition chimique des parties cornées des insectes. Mémoirs de la Societéd’Histoire Naturelle, 1, 29–42. [Google Scholar]
33. Knorr, D. (1984). Use of chitinous polymer in food–A challenge for food research and development. Food Technology, 38(1), 85–97. [Google Scholar]
34. Kumari, S., Rath, P. K. (2014). Extraction and characterization of chitin and chitosan from (Labeorohit) fish scales. Procedia Materials Science, 6(2014), 482–489. DOI 10.1016/j.mspro.2014.07.062. [Google Scholar] [CrossRef]
35. Ruiz, G. A. M., Corrales, H. F. Z. (2017). Chitosan, chitosan derivatives and their biomedical applications. In: Shalaby, E. (Eds.Biological activities and application of marine polysaccharides, pp. 87–106. InTech. London, UK. [Google Scholar]
36. Crini, G. (2019). Historical review on chitin and chitosan biopolymers. Environmental Chemistry Letters, 17(4), 1623–1643. DOI 10.1007/s10311-019-00901-0. [Google Scholar] [CrossRef]
37. Ferraro, V., Cruz, I. B., Jorge, R. F., Malcata, F. X., Pintado, M. E. et al. (2010). Valorisation of natural extracts from marine source focused on marine by-products: A review. Food Research International, 43(9), 2221– 2233. DOI 10.1016/j.foodres.2010.07.034. [Google Scholar] [CrossRef]
38. New, N., Furuike, T., Tamura, H. (2011). Chitosan from aquatic and terrestrial organisms and microorganisms. Production, properties and applications. In: Johnson, B. M., Berkel, Z. E. (Eds.Biodegradable materials, pp. 29–50. New York: Nova Science Publishers Inc. [Google Scholar]
39. Badawy, M., Rabea, E. I. (2017). Chitosan and its modifcations as biologically active compounds in diferent applications. In: Masuell, M., Renard, D. (Eds.Advances in physicochemical properties of biopolymers, pp. 1–108. Sharjah: Bentham Science Publishers. [Google Scholar]
40. Bonecco, M. B., Martínez Sáenz, M. G., Bufa, L. M. (2017). Chitosan, from residue to industry. In: Masuell, M., Renard, D. (Eds.Advances in physicochemical properties of biopolymers, pp. 224–256. Sharjah: Bentham e-Books, Bentham Science Publishers. [Google Scholar]
41. Morin-Crini, N., Lichtfouse, E., Torri, G., Crini, G. (2019). Applications of chitosan in food, pharmaceuticals, medicine, cosmetics, agriculture, textiles, pulp and paper biotechnology and environmental chemistry. Environmental Chemistry Letters, 17(4), 1667–1692. DOI 10.1007/s10311-019-00904-x. [Google Scholar] [CrossRef]
42. Revathi, M., Saravanan, R., Shanmugam, A. (2012). Production and characterization of chitinase from Vibrio species, a head waste of shrimp metapenaeusdobsonii (Miers, 1878) and chitin of sepiellainermis orbigny, 1848. Advances in Bioscience and Biotechnology, 3(4), 392–397. DOI 10.4236/abb.2012.34056. [Google Scholar] [CrossRef]
43. Chien, R., Yen, M., Mau, J. (2016). Antimicrobial and antitumor activities of chitosan from shiitakestipes, compared to commercial chitosan from crab shells. Carbohydrates Polymers, 138(1), 259–264. DOI 10.1016/j.carbpol.2015.11.061. [Google Scholar] [CrossRef]
44. Dutta, J., Tripathi, S., Dutta, P. K. (2012). Progress in antimicrobial activities of chitin, chitosan and its oligosaccharides: A systematic study needs for food applications. Food Science and Technology International, 18(1), 3–34. DOI 10.1177/1082013211399195. [Google Scholar] [CrossRef]
45. Zargar, V., Asghari, M., Dashti, A. (2015). A review on chitin and chitosan polymers: Structure, chemistry, solubility, derivatives, and applications. ChemBioEng Reviews, 2(3), 204–226. DOI 10.1002/cben.201400025. [Google Scholar] [CrossRef]
46. Yan, N., Chen, X. (2015). Sustainability: Don’t waste seafood waste. Nature, 524(7564), 155–157. DOI 10.1038/524155a. [Google Scholar] [CrossRef]
47. Berezina, N. (2016). Production and application of chitin. Physical Sciences Reviews, 1(9), 1527. DOI 10.1515/psr-2016-0048. [Google Scholar] [CrossRef]
48. Sajomsang, W., Gonil, P. (2010). Preparation and characterization of ⊍-chitin from cicada sloughs. Materials Science and Engineering, 30(3), 357–363. DOI 10.1016/j.msec.2009.11.014. [Google Scholar] [CrossRef]
49. Jang, M. K., Kong, B. G., Jeong, Y. I., Lee, C. H., Nah, J. W. (2004). Physicochemical characterization of α-chitin, β-chitin, and γ-chitin separated from natural resources. Journal of Polymer Science Part A–Polymer Chemistry, 42(14), 3423–3432. DOI 10.1002/(ISSN)1099-0518. [Google Scholar] [CrossRef]
50. Muzzarelli, R. A. (2011). Chitin nanostructure in living organisms. In: Gupta, N. (EdsIn: Chitin: Formation and diagenesis, pp. 1–34. Netherland: Dordrecht. [Google Scholar]
51. Zainol Abidin, N. A., Kormin, F., ZainolAbidin, N. A., Mohamed Anuar, N. A. F., Abu Bakar, M. F. (2020). The potential of insects as alternative sources of chitin: An overview on the chemical method of extraction from various sources. International Journal of Molecular Sciences, 21(14), 4978. DOI 10.3390/ijms21144978. [Google Scholar] [CrossRef]
52. Azuma, K., Ifuku, S., Osaki, T., Okamoto, Y., Minami, S. (2014). Preparation and biomedical applications of chitin and chitosan nanofibers. Journal of Biomedical Nanotechnology, 10(10), 2891–2920. DOI 10.1166/jbn.2014.1882. [Google Scholar] [CrossRef]
53. Andrew, C. A., Wan, A. C., Tai, B. C. (2013). Chitin—A promising biomaterial for tissue engineering and stem cell technologies. Biotechnology Advances, 31(8), 1776–1785. DOI 10.1016/j.biotechadv.2013.09.007. [Google Scholar] [CrossRef]
54. Jayakumar, R., Prabaharan, M., Nair, S. V., Tamura, H. (2010). Novel chitin and chitosan nanofibers in biomedical applications. Biotechnology Advances, 28(1), 142–150. DOI 10.1016/j.biotechadv.2009.11.001. [Google Scholar] [CrossRef]
55. Shah, P., Jogani, V., Mishra, P., Mishra, A. K., Bagchi, T. et al. (2007). Modulation of gancioclovir intestinal absorption in presence of absorption enhancers. Journal of Pharmaceutical Sciences, 96(10), 2710–2722. DOI 10.1002/jps.20888. [Google Scholar] [CrossRef]
56. Pilai, K. S., Paul, W., Sharma, C. P. (2009). Chitin and chitosan polymers: Chemistry, solublity and fiber formation. Progress in Polymer Science, 34, 641–678. DOI 10.1016/j.progpolymsci.2009.04.001. [Google Scholar] [CrossRef]
57. Merzendorfer, H., Zimoch, L. (2003). Chitin metabolism in insects: Structure, function and regulation of chitin synthases and chitinases. Journal of Experimental Biology, 206(24), 4393–4412. DOI 10.1242/jeb.00709. [Google Scholar] [CrossRef]
58. Schmitz, C., Auza, L. G., Koberidze, D., Rasche, S., Fischer, R. et al. (2019). Conversion of chitin to defined chitosan oligomers: Current status and future prospects. Marine Drugs, 17(8), 452. DOI 10.3390/md17080452. [Google Scholar] [CrossRef]
59. Kaur, S., Dhillon, G. S. (2014). The versatile biopolymer chitosan: Potential sources evaluation of extraction methods and applications. Critical Reviews in Microbiology, 40(2), 155–175. DOI 10.3109/1040841X.2013.770385. [Google Scholar] [CrossRef]
60. João, C. F. C., Silva, J. C., Borges, J. P. (2015). Chitin-based nanocomposites: Biomedical applications. In: Eco-friendly polymer nanocomposites, pp. 439–457. New Delhi: Springer. [Google Scholar]
61. Ramírez, M. A., Rodriguez, A. T., Alfonso, L., Peniche, C. (2010). Chitin and its derivatives as biopolymers with potential agricultural applications. Biotecnología Aplicada, 27(4), 270–276. [Google Scholar]
62. Benhabileset, M. S., Salah, R., Lounici, H., Drouiche, N., Goosen, M. F. A. et al. (2012). Antibacterial activity of chitin, chitosan and its oligomers prepared from shrimp shell waste. Food Hydrocolloids, 29(1), 48–56. DOI 10.1016/j.foodhyd.2012.02.013. [Google Scholar] [CrossRef]
63. Park, B. K., Kim, M. M. (2010). Applications of chitin and its derivatives in biological medicine. International Journal of Molecular Sciences, 11(12), 5152–5164. DOI 10.3390/ijms11125152. [Google Scholar] [CrossRef]
64. Fan, W., Yan, W., Xu, Z., Ni, H. (2012). Formation mechanism of monodisperse, low molecular weight chitosan nanoparticles by ionic gelation technique. Colloids and Surfaces B: Biointerfaces, 90, 21–27. DOI 10.1016/j.colsurfb.2011.09.042. [Google Scholar] [CrossRef]
65. Nowak, V., Persijn, D., Rittenschober, D., Charrondiere, U. R. (2016). Review of food composition data for edible insects. Food Chemistry, 193(2), 39–46. DOI 10.1016/j.foodchem.2014.10.114. [Google Scholar] [CrossRef]
66. Chae, K. S., Shin, C. S., Shin, W. S. (2018). Characteristics of cricket (Gryllus bimaculatus) chitosan and chitosan-based nanoparticles. Food Science and Biotechnology, 27(3), 631–639. DOI 10.1007/s10068-018-0314-4. [Google Scholar] [CrossRef]
67. Lichtfouse, E., Morin-Crini, N., Fourmentin, M., Zemmouri, H., Oliveira do Carmo Nascimento, I. et al. (2019). Chitosan for direct bioflocculation processes. In: Crini, G., Lichtfouse, E. (Eds.Sustainable agriculture reviews, vol. 36, pp. 335–380. Berlin, Germany: Springer International Publishing. [Google Scholar]
68. Abdel-Rahman, R. M., Hrdina, R., Abdel-Mohsen, A. M., Fouda, M. M. G., Soliman, A. Y. et al. (2015). Chitin and chitosan from Brazilian Atlantic Coast: Isolation, characterization and antibacterial activity. International Journal of Biological Macromolecules, 80, 107–120. DOI 10.1016/j.ijbiomac.2015.06.027. [Google Scholar] [CrossRef]
69. Elieh-Ali-Komi, D., Hamblin, M. R. (2016). Chitin and chitosan: Production and application of versatile biomedical nanomaterials. International Journal of Advanced Research, 4(3), 411–427. [Google Scholar]
70. Kumirska, J. M., Czerwicka, Z., Kaczyński, A., Bychowska, K., Brzozowski, J. et al. (2010). Application of spectroscopic methods for structural analysis of chitin and chitosan. Marine Drugs, 8(5), 1567–1636. DOI 10.3390/md8051567. [Google Scholar] [CrossRef]
71. Islam, M. S., Khan, S., Tanaka, M. (2004). Waste loading in shrimp and fish processing effluents: Potential source of hazards to the coastal and nearshore environments. Marine Pollution Bulletine, 49(1–2), 103–110. DOI 10.1016/j.marpolbul.2004.01.018. [Google Scholar] [CrossRef]
72. Rhazi, M., Tolaimate, A., Habibi, Y. (2012). Interactions of chitosan with metals for water purification. In: Polysaccharide building blocks, pp. 127–141. Hoboken, New Jersey, United States: John Wiley & Sons, Inc. [Google Scholar]
73. Kumari, S., Kishor, R. (2020). Chitin and chitosan: Origin, properties, and applications. In: Handbook of chitin and chitosan, pp. 1–33. Amsterdam, Netherlands: Elsevier. [Google Scholar]
74. Rangel-Mendez, J. R., Barrios, V. A. E., Davila-Rodriguez, J. L. (2010). Chitin based biocomposites for removal of contaminants from water: A case study of fluoride adsorption. In: Elnashar, M. (Eds.Biopolymers. InTech. London, UK. [Google Scholar]
75. Younes, I., Rinaudo, M. (2015). Chitin and chitosan preparation from marine sources. Marine Drugs, 13(3), 1133–1174. DOI 10.3390/md13031133. [Google Scholar] [CrossRef]
76. Krajewska, B. (2005). Membrane-based processes performed with use of chitin/chitosan materials. Separation and Purification Technology, 41(3), 305–312. DOI 10.1016/j.seppur.2004.03.019. [Google Scholar] [CrossRef]
77. Inoue, K., Baba, Y. (2007). Chitosan: A versatile biopolymer for separation, purification, and concentration of metal ions. In: Senguta, A. K. (Eds.Ion exchange and solvent extraction, vol. 18. Boca Raton: CRC Press. [Google Scholar]
78. Hahn, T., Zibek, S. (2018). Sewage polluted water treatment via chitosan: A review. In: Dongre, R. S. (Eds.Chitin-Chitosan—Myriad functionalities in science and technology. InTech. London, UK. [Google Scholar]
79. Ioelovich, M. (2014). Crystallinity and hydrophility of chitin and chitosan. Research and Reviews: Journal of Chemistry, 3(3), 7–14. [Google Scholar]
80. Wang, S. L., Kaoc, T. Y., Wang, Y. H., Yen, Y. H., Chern, T. Y. et al. (2006). Solvent stable metalloprotease produced by Bacillus sp. TKU004 and its application in deproteinization of squid pen for bchitin preparation. Enzyme and Microbial Technology, 39(4), 724–727. DOI 10.1016/j.enzmictec.2005.12.007. [Google Scholar] [CrossRef]
81. Yang, J. K., Shih, I. L., Tzeng, Y. M., Wang, S. L. (2000). Production and purification of protease from a Bacillus subtilis that can deproteinize crustacean wastes. Enzyme and Microbial Technology, 26(5–6), 406–413. DOI 10.1016/S0141-0229(99)00164-7. [Google Scholar] [CrossRef]
82. Abdou, E. S., Nagy, K. S., Elsabee, M. Z. (2008). Extraction and characterization of chitin and chitosan from local sources. Bioresource Technology, 99(5), 1359–1367. DOI 10.1016/j.biortech.2007.01.051. [Google Scholar] [CrossRef]
83. Di Nardo, T. (2018). Deacetylation by mechanochemistry and aging as a pathway to high molecular weight chitosan from chitin (Masters Thesis). Montreal, Canada: McGill University Canada. [Google Scholar]
84. Yadav, M., Goswami, P., Paritosh, K., Kumar, M., Pareek, N. et al. (2019). Seafood waste: A source for preparation of commercially employable chitin/chitosan materials. Bioresources and Bioprocessing, 6(1), 1–20. DOI 10.1186/s40643-019-0243-y. [Google Scholar] [CrossRef]
85. Zimri, M. N. (2018). Preparation and electrospinning of chitosan from waste black soldier fly biomass (PhD. Thesis). Bellville, South Africa: University of the Western Cape. [Google Scholar]
86. Boudouaia, N., Bengharez, Z., Jellali, S. (2019). Preparation and characterization of chitosan extracted from shrimp shells waste and chitosan film: Application for eriochrome black T removal from aqueous solutions. Applied Water Science, 9(4), 1–12. DOI 10.1007/s13201-019-0967-z. [Google Scholar] [CrossRef]
87. Tokuyasu, K., Mitsutomi, M., Yamaguchi, I., Hayashi, K., Mori, Y. (2000). Recognition of chitooligosaccharides and their N-acetyl groups by putative subsites of chitin deacetylase from a deuteromycete, colletotrichum lindemuthianum. Biochemistry, 39(30), 8837–8843. DOI 10.1021/bi0005355. [Google Scholar] [CrossRef]
88. Philibert, T., Lee, B. H., Fabien, N. (2017). Current status and new perspectives on chitin and chitosan as functional biopolymers. Applied Biochemistry and Biotechnology, 181(4), 1314–1337. DOI 10.1007/s12010-016-2286-2. [Google Scholar] [CrossRef]
89. Hajji, S., Younes, I., Ghorbel-Bellaaj, O., Hajji, R., Rinaudo, M. et al. (2014). Structural differences between chitin and chitosan extracted from three different marine sources. International Journal of Biological Macromolecules, 65(4), 298–306. DOI 10.1016/j.ijbiomac.2014.01.045. [Google Scholar] [CrossRef]
90. Aiba, S. I. (1991). Studies on chitosan: Evidence for the presence of random and block copolymer structures in partially N-acetylated chitosans. International Journal of Biological Macromolecules, 13(1), 40–44. DOI 10.1016/0141-8130(91)90008-I. [Google Scholar] [CrossRef]
91. Tacon, A. G. J. (2018). Global trends in aquaculture and compound aquafeed production. Magazine of the World Aquaculture Society, 49(2), 33–46. [Google Scholar]
92. Gillett, R. (2008). Global study of shrimp fisheries. Rome: FAO, FAO Fisheries Technical Paper, 331. [Google Scholar]
93. Kurita, K. (2006). Chitin and chitosan: Functional biopolymers from marine crustaceans. Marine Biotechnology, 8(3), 203–226. DOI 10.1007/s10126-005-0097-5. [Google Scholar] [CrossRef]
94. Pittman, S. J., McAlpine, C. A. (2003). Movements of marine fish and decapod crustaceans: Process, theory and application. Advances in Marine Biology, 44(1), 205–294. DOI 10.1016/S0065-2881(03)44004-2. [Google Scholar] [CrossRef]
95. Abdel-Gawad, K. M., Hifney, A. F., Fawzy, M. A., Gomaa, M. (2017). Technology optimization of chitosan production from Aspergillus niger biomass and its functional activities. Food Hydrocolloids, 63(2), 593–601. DOI 10.1016/j.foodhyd.2016.10.001. [Google Scholar] [CrossRef]
96. Turunen, J., Karppinen, A., Ihme, R. (2019). Effectiveness of biopolymer coagulants in agricultural wastewater treatment at two contrasting levels of pollution. SN Applied Sciences, 1(3), 210. DOI 10.1007/s42452-019-0225-x. [Google Scholar] [CrossRef]
97. Pawadee, M., Malinee, P., Thanawit, P., Junya, P. (2003). HetrogeneousNdeacetylation of squid chitin in alkaline solution. Carbohydrate Polymers, 52(2), 119–123. DOI 10.1016/S0144-8617(02)00300-4. [Google Scholar] [CrossRef]
98. Chandumpaia, A., Singhpibulpornb, N., Faroongsarngc, D., Sornprasit, P. (2004). Preparation and physico-chemical characterization of chitin and chitosan from the pens of the squid species, loligolessoniana and loligoformosana. Carbohydrate Polymers, 58(4), 467–474. DOI 10.1016/j.carbpol.2004.08.015. [Google Scholar] [CrossRef]
99. Sakthivel, D., Vijayakumar, N., Anandan, V. (2015). Extraction of chitin and chitosan from Mangrove crab (Sesarmaplicatum) from Thengaithittu Estuary Pondicherry Southeast Coast of India. Human Journals, 4(1), 12–24. [Google Scholar]
100. Ali, M., Shakeel, M., Mehmood, K. (2019). Extraction and characterization of high purity chitosan by rapid and simple techniques from mud crabs taken from Abbottabad. Pakistan Journal of Pharmaceutical Sciences, 32(1), 171–175. [Google Scholar]
101. Prasetya, D. S. B., Ahzan, S. (2019). Time stirring effect of chitosan from shrimp shells as gold absorbent. Lensa JurnalKependidikan Fisika, 7(2), 46–50. DOI 10.33394/j-lkf.v7i2.2685. [Google Scholar] [CrossRef]
102. Almeida, L., Rodrigues, W. L., Aguiar, N. V., Silva, R. S., Moreira, C. K. P. (2015). Extração de quitina, síntese e caracterização de quitosanaobtidaatravés de resíduos de camarão (Macrobrachiumamazonicum). Blucher Chemical Engineering Proceedings, 1(3), 2272–2278. DOI 10.5151/chemeng-cobeqic2015-477-34107-262119. [Google Scholar] [CrossRef]
103. Santos, V. P., Maia, P., Alencar, N., de, S., Farias, L. et al. (2019). Recovery of chitin and chitosan from shrimp waste with microwave technique and versatile application. Arquivos Do Instituto Biológico, 86(5), 1–7. DOI 10.1590/1808-1657000982018. [Google Scholar] [CrossRef]
104. Souza, F. M., Ferreira, R. M. S., Barbosa, R. C. (2015). Utilização da casca de camarão para produção de quitina. 1–11, Revista Scire. [Google Scholar]
105. Mahdy Samar, M., El-Kalyoubi, M. H., Khalaf, M. M., Abd El-Razik, M. M. (2013). Physicochemical, functional, antioxidant and antibacterial properties of chitosan extracted from shrimp wastes by microwave technique. Annals of Agricultural Sciences, 58(1), 33–41. DOI 10.1016/j.aoas.2013.01.006. [Google Scholar] [CrossRef]
106. Abdelmalek, B. E., Sila, A., Haddar, A., Bougatef, A., Ali, M. (2017). Chitin and chitosan from squid gladius: Biological activities of chitosan and its application as clarifying agent for apple juice. International Journal of Biological Macromolecules, 104, 953–962. DOI 10.1016/j.ijbiomac.2017.06.107. [Google Scholar] [CrossRef]
107. Abdulwadud Abdulkarim, A. O. A., Muhammed, T. I., Surajudeen, A., Abubakar, J. M. (2013). Extraction and characterisation of chitin and chitosan from civ. Environmental Research, 3(2), 108–115. [Google Scholar]
108. Cumberlidge, N., Daniels, S. R. (2008). A conservation assessment of the freshwater crabs of western Africa (Brachyura: Potamonautidae). African Journal of Ecology, 46(1), 74–79. DOI 10.1111/j.1365-2028.2007.00815.x. [Google Scholar] [CrossRef]
109. Yeo, D. C. J., Ng, P. K. L., Cumberlidge, N., Magalhaes, C., Daniels, S. R. et al. (2007). Global diversity of crabs (Crustacea: Decapoda: Brachyura) in freshwater. Freshwater Animal Diversity Assessment. Hydrobiologia, 595(2007), 275–286. [Google Scholar]
110. Fadlaoui, S., Mohammed, L., Omari, A., Melhaoui, M., Sihame, A. et al. (2019). Isolation and characterization of chitin from shells of the freshwater crab potamon algeriense. Progress on Chemistry and Application of Chitin and Its Derivatives, 14, 23–25. DOI 10.15259/PCACD.24.002. [Google Scholar] [CrossRef]
111. Synowiecki, J., Al-Khateeb, N. A. (2003). Production, properties, and some new applications of chitin and its derivatives. Critical Reviews in Food Science and Nutrition, 43(2), 145–171. DOI 10.1080/10408690390826473. [Google Scholar] [CrossRef]
112. Tharanathan, R. N., Kittur, F. S. (2003). Chitin-the undisputed biomolecule of great potential. Critical Reviews in Food Science and Nutrition, 43(1), 61–87. DOI 10.1080/10408690390826455. [Google Scholar] [CrossRef]
113. Chakrabarti, R. (2002). Carotenoprotein from tropical brown shrimp shell waste by enzymatic process. Food biotechnology, 16(1), 81–90. DOI 10.1081/FBT-120004202. [Google Scholar] [CrossRef]
114. No, H. K., Lee, K. S., Meyers, S. P. (2000). Correlation between physicochemical characteristics and binding capacities of chitosan products. Journal of Food Science, 65(7), 1134–1137. DOI 10.1111/j.1365-2621.2000.tb10252.x. [Google Scholar] [CrossRef]
115. Elasri, O., Salem, M., Ramdani, M., Zaraali, O., Latrach, L. (2018). Effect of increasing inoculum ratio on energy recovery from chicken manure for better use in Egyptian agricultural farms. Chemical and Biological Technologies in Agriculturure, 17(5), 2–7. DOI 10.1186/s40538-018-0129-9. [Google Scholar] [CrossRef]
116. Cárdenas, G., Cabrera, G., Taboada, E., Miranda, S. P. (2004). Chitin characterization by SEM, FTIR, XRD, and 13C cross polarization/mass angle spinning NMR. Journal of Applied Polymer Science, 93(4), 1876–1885. DOI 10.1002/app.20647. [Google Scholar] [CrossRef]
117. Braconnot, M. H. (1811). LXXXIII. Chemical analysis of the green shell of the walnut. The Philosophical Magazine, 38(164), 447–451. DOI 10.1080/14786441108638687. [Google Scholar] [CrossRef]
118. Krčmář, M. (2018). Chitosan-glukanovýkomplexizolovanýzeSchizophyllum commune.: The chitosan-glucan complex isolated from Schizophyllum commune (Thesis). Brno-střed, Czechia: Brno University of Technology. http://hdl.handle.net/11012/4480. [Google Scholar]
119. Hu, K. J., Yeung, K. W., Ho, K. P., Hu, J. L. (1999). Rapid extraction of high-quality chitosan from mycelia of Absidia glauca. Journal of Food Biochemistry, 23(2), 187–196. DOI 10.1111/j.1745-4514.1999.tb00013.x. [Google Scholar] [CrossRef]
120. New, N., Chandrkrachang, S., Stevens, W. F., Maw, T., Tan, T. K. et al. (2002). Production of fungal chitosan by solid state and submerged fermentation. Carbohydrates Polymers, 49(2), 235–237. DOI 10.1016/S0144-8617(01)00355-1. [Google Scholar] [CrossRef]
121. Pochanavanich, P., Suntornsuk, W. (2002). Fungal chitosan production and its characterization. Letters in Applied Microbiology, 35(1), 17–21. DOI 10.1046/j.1472-765X.2002.01118.x. [Google Scholar] [CrossRef]
122. Suntornsuk, W., Pochanavanich, P., Suntornsuk, L. (2002). Fungal chitosan production on food processing by-products. Process Biochemistry, 37(7), 727–729. DOI 10.1016/S0032-9592(01)00265-5. [Google Scholar] [CrossRef]
123. Lassaigne, I. L. (1843). Sur les tissues tégumentaires des insectes de differentsordres. Comptes Rendus des Séances de l’Académie des Sciences, 16, 1087–1089. DOI 10.1007/978-3-030-16538-3_2. [Google Scholar] [CrossRef]
124. Muzzarelli, R. A. A., Boudrant, J., Meyer, D., Manno, N., DeMarchis, M. et al. (2012). Current views on fungal chitin/chitosan, human chitinases, food preservation, glucans, pectins and inulin: A tribute to Henri Braconnot, precursor of the carbohydrate polymers science, on the chitin bicentennial. Carbohydrate Polymers, 87(2), 995–1012. DOI 10.1016/j.carbpol.2011.09.063. [Google Scholar] [CrossRef]
125. Aida, F. M., Shuhaimi, N. A., Yazid, M., Maaruf, A. G. (2009). Mushroom as a potential source of prebiotics: A review. Trends in Food Science & Technology, 20(11–12), 567–575. DOI 10.1016/j.tifs.2009.07.007. [Google Scholar] [CrossRef]
126. Kalac, P. (2009). Chemical composition and nutritional value of European species of wild growing mushrooms: A review. Food Chemistry, 113(1), 9–16. DOI 10.1016/j.foodchem.2008.07.077. [Google Scholar] [CrossRef]
127. Jardine, A., Sayed, S. (2018). Valorisation of chitinous biomass for antimicrobial applications. Pure and Applied Chemistry, 90(2), 293–304. DOI 10.1515/pac-2017-0707. [Google Scholar] [CrossRef]
128. Peter, M. G. (2005). Chitin and chitosan in fungi. In Wiley Biopolymers Online. pp. 123–132. DOI 10.1002/3527600035.bpol6005. [Google Scholar] [CrossRef]
129. Kim, W. J., Lee, W. G., Theodore, K., Chang, H. N. (2001). Optimization of culture conditions and continuous production of chitosan by the fungi, Absidia coerulea. Biotechnology and Bioprocess Engineering, 6(1), 6–10. DOI 10.1007/BF02942243. [Google Scholar] [CrossRef]
130. Chatterjee, S., Adhya, M., Guha, A. K., Chatterjee, B. P. (2005). Chitosan from Mucor rouxii: Production and physico-chemical characterization. Process Biochemistry, 40, 395–400. DOI 10.1016/j.procbio.2004.01.025. [Google Scholar] [CrossRef]
131. Sbrana, C., Avio, L., Giovannetti, M. (1995). The occurrence of calcofluor and lectin binding polysaccharides in the outer wall of arbuscular mycorrhizal fungal spores. Mycological Research, 99(10), 1249–1252. DOI 10.1016/S0953-7562(09)80287-6. [Google Scholar] [CrossRef]
132. Mohan, K., Ganesan, A. R., Muralisankar, T., Jayakumar, R., Sathishkumar, P. et al. (2020). Recent insights into the extraction, characterization, and bioactivities of chitin and chitosan from insects. Trends in Food Science & Technology, 105(4), 17–42. DOI 10.1016/j.tifs.2020.08.016. [Google Scholar] [CrossRef]
133. Gapsari, F., Dewi, F. G., Wijaya, P. H. S., Hidayatullah, S. (2020). The effectiveness of fish scale waste-synthesized chitosan and food-grade chitosan as corrosion inhibitor. Journal of Southwest Jiaotong University, 55(2), 1–10. DOI 10.35741/issn.0258-2724.55.2.47. [Google Scholar] [CrossRef]
134. Dhillon, G. S., Kaur, S., Brar, S. K., Verma, M. (2013). Green synthesis approach: Extraction of chitosan from fungus mycelium. Critical Reviews in Biotechnology, 33(4), 379–403. DOI 10.3109/07388551.2012.717217. [Google Scholar] [CrossRef]
135. Van Huis, A. (2013). Potential of insects as food and feed in assuring food security. Annual Review of Entomology, 58(1), 563–583. DOI 10.1146/annurev-ento-120811-153704. [Google Scholar] [CrossRef]
136. Shamshina, J. L., Berton, P., Rogers, R. D. (2019). Advances in functional chitin materials: A review. ACS Sustainable Chemistry & Engineering, 7(7), 6444–6457. DOI 10.1021/acssuschemeng.8b06372. [Google Scholar] [CrossRef]
137. Shin, C. S., Kim, D. Y., Shin, W. S. (2019). Characterization of chitosan extracted from Mealworm beetle (Tenebrio molitor, Zophobasmorio) and Rhinoceros beetle (Allomyrinadichotoma) and their antibacterial activities. International Journal of Biological Macromolecules, 15(125), 72–77. [Google Scholar]
138. Ibañez-Peinado, D., Ubeda-Manzanaro, M., Martínez, A., Rodrigo, D. (2020). Antimicrobial effect of insect chitosan on Salmonella Typhimurium, Escherichia coli O157: H7 and Listeria monocytogenes survival. PLoS One, 15(12), e0244153. DOI 10.1371/journal.pone.0244153. [Google Scholar] [CrossRef]
139. Hu, Z. (2018). Challenges and opportunities related to the use of chitosan as food preservative. Journal of Applied Microbiology, 126(5), 1318–1331. DOI 10.1111/jam.14131. [Google Scholar] [CrossRef]
140. Tolaimate, A., Desbrieres, J., Rhazi, M., Alagui, A. (2003). Contribution to the preparation of chitins and chitosans with controlled physicochemical properties. Polymer, 44(26), 7939–7952. DOI 10.1016/j.polymer.2003.10.025. [Google Scholar] [CrossRef]
141. Battampara, P., Sathish, T. N., Reddy, R., Guna, V., Nagananda, G. S. et al. (2020). Properties of chitin and chitosan extracted from silkworm pupae and egg shells. International Journal of Biological Macromolecules, 15(161), 1296–1304. DOI 10.1016/j.ijbiomac.2020.07.161. [Google Scholar] [CrossRef]
142. Mehranian, M., Pourabad, R. F., Bashir, N. S., Taieban, S. (2017). Physicochemical characterization of chitin from the Mediterranean flour moth, Ephestiakuehniella Zeller (Lepidoptera: Pyralidae). Journal of Macromolecular Science, Part A, 54(10), 720–726. DOI 10.1080/10601325.2017.1332461. [Google Scholar] [CrossRef]
143. Wu, S. (2012). Preparation of chitooligosaccharides from clanis bilineata larvae skin and their antibacterial activity. International Journal of Biological Macromolecules, 51(5), 1147–1150. DOI 10.1016/j.ijbiomac.2012.08.035. [Google Scholar] [CrossRef]
144. Song, Y. S., Kim, M. W., Moon, C., Seo, D. J., Han, Y. S. et al. (2018). Extraction of chitin and chitosan from larval exuvium and whole body of edible mealworm, Tenebrio molitor. Entomological Research, 48(3), 227–233. DOI 10.1111/1748-5967.12304. [Google Scholar] [CrossRef]
145. Kaya, M., Mulerčikas, P., Sargin, I., Kazlauskaitė, S., Baublys, V. et al. (2016). Three-dimensional chitin rings from body segments of a pet diplopod species: Characterization and protein interaction studies. Materials Science and Engineering C, 68, 716–722. DOI 10.1016/j.msec.2016.06.046. [Google Scholar] [CrossRef]
146. Ma, B., Herzog, E. L., Lee, C. G., Peng, X., Lee, C. M. et al. (2015). Role of chitinase 3-like-1 and semaphorin 7a in pulmonary melanoma metastasis. Cancer Research, 75(3), 487–496. DOI 10.1158/0008-5472.CAN-13-3339. [Google Scholar] [CrossRef]
147. Marei, N., Elwahy, A. H., Salah, T. A., El Sherif, Y., Abd El-Samie, E. (2019). Enhanced antibacterial activity of Egyptian local insects’ chitosan-based nanoparticles loaded with ciprofloxacin-HCl. International Journal of Biological Macromolecules, 126(12), 262–272. DOI 10.1016/j.ijbiomac.2018.12.204. [Google Scholar] [CrossRef]
148. Liu, X., Cooper, A. M., Yu, Z., Silver, K., Zhang, J. et al. (2019). Progress and prospects of arthropod chitin pathways and structures as targets for pest management. Pesticide Biochemistry and Physiology, 161, 33–46. DOI 10.1016/j.pestbp.2019.08.002. [Google Scholar] [CrossRef]
149. Kabalak, M., Aracagök, D., Torun, M. (2020). Extraction, characterization and comparison of chitins from large bodied four Coleoptera and Orthoptera species. International Journal of Biological Macromolecules, 145, 402–409. DOI 10.1016/j.ijbiomac.2019.12.194. [Google Scholar] [CrossRef]
150. Luo, Q., Wang, Y., Han, Q., Ji, L., Zhang, H. et al. (2019). Comparison of the physicochemical, rheological, and morphologic properties of chitosan from four insects. Carbohydrate Polymers, 209(1), 266–275. DOI 10.1016/j.carbpol.2019.01.030. [Google Scholar] [CrossRef]
151. Torres-Castillo, J. A., Sinagawa-García, S. R., Lara-Villalón, M., Martínez-Ávila, G. C. G., Mora-Olivo, A. et al. (2015). Evaluation of biochemical components from Pterophylla beltrani (Bolívar & Bolívar) (Orthoptera: TettigoniidaeA forest pest from northeastern Mexico. Southwestern Entomologist, 40(4), 741–751. DOI 10.3958/059.040.0402. [Google Scholar] [CrossRef]
152. Erdogan, S., Kaya, M. (2016). High similarity in physicochemical properties of chitin and chitosan from nymphs and adults of a grasshopper. International Journal of Biological Macromolecules, 89, 118–126. DOI 10.1016/j.ijbiomac.2016.04.059. [Google Scholar] [CrossRef]
153. Ibitoye, E. B., Lokman, I. H., Hezmee, M. N. M., Goh, Y. M., Zuki, A. B. Z. et al. (2018). Extraction and physicochemical characterization of chitin and chitosan isolated from house cricket. Biomedical Materials, 13(2), 025009. DOI 10.1088/1748-605X/aa9dde. [Google Scholar] [CrossRef]
154. Kim, Y. H., Park, S. K., Hur, J. Y., Kim, Y. C. (2017). Purification and characterization of a major extracellular chitinase from a biocontrol bacterium, Paenibacilluselgii HOA73. The Plant Pathology Journal, 33(3), 318–328. DOI 10.5423/PPJ.FT.01.2017.0022. [Google Scholar] [CrossRef]
155. Mol, A., Kaya, M., Mujtaba, M., Akyuz, B. (2018). Extraction of high thermally stable and nanofibrous chitin from cicada (Cicadoidea). Entomological Research, 48(6), 480–489. DOI 10.1111/1748-5967.12299. [Google Scholar] [CrossRef]
156. Caligiani, A., Marseglia, A., Leni, G., Baldassarre, S., Maistrello, L. et al. (2018). Composition of black soldier fly prepupae and systematic approaches for extraction and fractionation of proteins, lipids and chitin. Food Research International, 105, 812–820. DOI 10.1016/j.foodres.2017.12.012. [Google Scholar] [CrossRef]
157. Khayrova, A., Lopatin, S., Varlamov, V. (2019). Black soldier fly Hermetia illucens as a novel source of chitin and chitosan. International Journal of Sciences, 8(4), 81–86. DOI 10.18483/ijSci.2015. [Google Scholar] [CrossRef]
158. Tsaneva, D., Petkova, Z., Petkova, N., Stoyanova, M., Stoyanova, A. et al. (2018). Isolation and characterization of chitin and biologically active substances from honeybee (Apis mellifera). Journal of Pharmaceutical Sciences and Research, 10(4), 884–888. [Google Scholar]
159. Bulut, E., Sargin, I., Arslan, O., Odabasi, M., Akyuz, B. et al. (2017). In situ chitin isolation from body parts of a centipede and lysozyme adsorption studies. Materials Science and Engineering: C, 70(2017), 552–563. DOI 10.1016/j.msec.2016.08.048. [Google Scholar] [CrossRef]
160. Jucker, C., Lupi, D., Moore, C. D., Leonardi, M. G., Savoldelli, S. (2020). Nutrient recapture from insect farm waste: Bioconversion with Hermetia illucens (L.) (Diptera: Stratiomyidae). Sustainability, 12(1), 1–14. DOI 10.3390/su12010362. [Google Scholar] [CrossRef]
161. Petrenko, I., Bazhenov, V. V., Galli, R., Wysokowski, M., Fromont, J. et al. (2017). Chitin of poriferan origin and the bioelectrometallurgy of copper/copper oxide. Internatoinal Journal of Biological Macromolecules, 104, 1626–1632. DOI 10.1016/j.ijbiomac.2017.01.084. [Google Scholar] [CrossRef]
162. Greven, H., Kaya, M., Baran, T. (2016). The presence of α-chitin in tardigrada with comments on chitin in the Ecdysozoa. Journal of Experimental Zoology, 264, 11–16. DOI 10.1016/j.jcz.2016.06.003. [Google Scholar] [CrossRef]
163. Nemtsev, S. V., Zueva, O. Y., Khismatullin, M. R., Albulov, A. I., Varlamov, V. P. (2004). Isolation of chitin and chitosan from Honeybees. Applied Biochemistry and Microbiology, 40, 39–43. DOI 10.1023/B:ABIM.0000010349.62620.49. [Google Scholar] [CrossRef]
164. Zhang, M., Haga, A., Sekiguchi, H., Hirano, S. (2000). Structure of insect chitin isolated from beetle larva cuticle and silkworm (Bombyx mori) pupa exuvia. Internatoinal Journal of Biological Macromolecules, 27(1), 99–105. DOI 10.1016/S0141-8130(99)00123-3. [Google Scholar] [CrossRef]
165. Basseri, H., Bakhtiyari, R., Hashemi, S. J., Baniardelani, M., Shahraki, H. et al. (2019). Antibacterial/antifungal activity of extracted chitosan from American cockroach (Dictyoptera: Blattidae) and German cockroach (Blattodea: Blattellidae). Journal of Medical Entomology, 56(5), 1208–1214. DOI 10.1093/jme/tjz082. [Google Scholar] [CrossRef]
166. Majtán, J., Bíliková, K., Markovič, O., Gróf, J., Kogan, G. et al (2007). Isolation and characterization of chitin from bumblebee (Bombus terrestris). International Journal of Biological Macromolecules, 40(3), 237–241. DOI 10.1016/j.ijbiomac.2006.07.010. [Google Scholar] [CrossRef]
167. Finke, M. D. (2013). Complete nutrient content of four species of feeder insects. Zoo Biology, 32(1), 27–36. DOI 10.1002/zoo.21012. [Google Scholar] [CrossRef]
168. Huet, G., Hadad, C., Husson, E., Laclef, S., Lambertyn, V. et al. (2020). Straightforward extraction and selective bioconversion of high purity chitin from bombyx eri larva: Toward an integrated insect biorefinery. Carbohydrate Polymers, 228, 115382. DOI 10.1016/j.carbpol.2019.115382. [Google Scholar] [CrossRef]
169. Marei, N. H., Abd El-Samie, E., Salah, T., Saad, G. R., Elwahy, A. H. (2016). Isolation and characterization of chitosan from different local insects in Egypt. International Journal of Biological Macromolecules, 82(7), 871–877. DOI 10.1016/j.ijbiomac.2015.10.024. [Google Scholar] [CrossRef]
170. Henry, M., Gasco, L., Piccolo, G., Fountoulaki, E. (2015). Review on the use of insects in the diet of farmed fish: Past and future. Animal Feed Science and Technology, 203, 1–22. DOI 10.1016/j.anifeedsci.2015.03.001. [Google Scholar] [CrossRef]
171. FAO (2012). Expert consultation; assesses the potential of insects as food and feed in assuring food security. Technical Consultation Meeting, pp. 23–25. Rome, Italy. [Google Scholar]
172. Hanboonsong, Y., Jamjanya, T., Durst, P. B. (2013). Six-legged livestock: Edible insect farming, collection and marketing in Thailand. RAP Publication, 3, 8–21. [Google Scholar]
173. Kaya, M., Mujtaba, M., Ehrlich, H., Salaberria, A. M., Baran, T. et al. (2017). On chemistry of γ-chitin. Carbohydrate Polymers, 176(5), 177–186. DOI 10.1016/j.carbpol.2017.08.076. [Google Scholar] [CrossRef]
174. Liu, S., Sun, J., Yu, L., Zhang, C., Bi, J. et al. (2012). Extraction and characterization of chitin from the beetle holotrichia parallela motschulsky. Molecules, 17(4), 4604–4611. DOI 10.3390/molecules17044604. [Google Scholar] [CrossRef]
175. Badariotti, F., Thuau, R., Lelong, C., Dubos, M. P., Favrel, P. (2007). Characterization of an atypical family 18 chitinase from the oyster crassostrea gigas: Evidence for a role in early development and immunity. Developmental & Comparative Immunology, 31(6), 559–570. DOI 10.1016/j.dci.2006.09.002. [Google Scholar] [CrossRef]
176. Rumpold, B. A., Schlüter, O. K. (2013). Nutritional composition and safety aspects of edible insects. Molecular Nutrition & Food Research, 57(5), 802–823. DOI 10.1002/mnfr.201200735. [Google Scholar] [CrossRef]
177. Latgé, J. P. (2007). The cell wall: A carbohydrate armour for the fungal cell. Molecular Microbiology, 66(2), 279–290. DOI 10.1111/j.1365-2958.2007.05872.x. [Google Scholar] [CrossRef]
178. Berger, R., Christina, T., de Miranda, A., Pessoa, P., Barbosa, M. A. et al. (2017). Chitosan produced from mucorales fungi using agroindustrial by-products and its efficacy to inhibit colletotrichum species. International Journal of Biological Macromolecules, 108(2017), 635–641. DOI 10.1016/j.ijbiomac.2017.11.178. [Google Scholar] [CrossRef]
179. Żółtowska-Aksamitowska, S., Shaala, L. A., Youssef, D. T., Elhady, S. S., Tsurkan, M. V. et al. (2018). First report on chitin in a non-verongiid marine demosponge: The mycale euplectellioides case. Marine Drugs, 16(2), 68. DOI 10.3390/md16020068. [Google Scholar] [CrossRef]
180. Croisier, F., Jérôme, C. (2013). Chitosan-based biomaterials for tissue engineering. European Polymer Journal, 49(4), 780–792. DOI 10.1016/j.eurpolymj.2012.12.009. [Google Scholar] [CrossRef]
181. Khor, E., Wu, H., Lim, L. Y., Guo, C. M. (2011). Chitin-methacrylate: Preparation, characterization and hydrogel formation. Materials, 4(10), 1728–1746. DOI 10.3390/ma4101728. [Google Scholar] [CrossRef]
 | This work is licensed under a Creative Commons Attribution 4.0 International License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |