

 | Phyton-International Journal of Experimental Botany |  |
DOI: 10.32604/phyton.2022.020540
REVIEW
The Primacy of Moringa (Moringa oleifera Lam.) in Boosting Nutrition Status and Immunity Defence Amidst the COVID-19 Catastrophe: A Perspective
1ICAR-Central Agroforestry Research Institute, Jhansi, 284003, India
2ICAR-Indian Grassland and Fodder Research Institute, Jhansi, 284003, India
3Department of Biosciences, COMSATS University Islamabad (CUI), Islamabad, 45550, Pakistan
4Agricultural College, Professor Jayashankar Telangana State Agricultural University, Warangal, 506006, India
5ICAR-Central Institute for Arid Horticulture, Bikaner, 334006, India
6Department of Biological Sciences, Faculty of Science, King Abdulaziz University, Jeddah, 22233, Saudi Arabia
7Princess Dr. NajlaBint Saud Al-Saud Center for Excellence Research in Biotechnology, King Abdulaziz University, Jeddah, 21589, Saudi Arabia
8Department of Public Health, Daffodil International University, Dhaka, 1341, Bangladesh
9Department of General Surgery, Faculty of Medicine, Siirt University, Siirt, 56100, Turkey
10Faculty of Agriculture, Department of Field Crops, Siirt University, Siirt, 56100, Turkey
11Department of Agronomy, Faculty of Agriculture, Kafrelsheikh University, Kafrelsheikh, 33156, Egypt
*Corresponding Authors: Khalid Rehman Hakeem. Email: kur.hakeem@gmail.com; Ayman EL Sabagh. Email: ayman.elsabagh@agr.kfs.edu.eg
Received: 29 November 2021; Accepted: 15 March 2022
Abstract: A severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) led novel coronavirus disease (COVID-19) outbreak spread through China has become the biggest global public health challenge today. The virus upon several mutations has led to the resurgence of more infectious and lethal variants infecting over 298 million people with more than 5.46 million deaths worldwide by the end of December, 2021. Though vaccines are available, various preventive measures particularly a high body immunity is still extremely important which determines the likelihood of disease severity and subsequent recovery in the current and future pandemics. This review acknowledges the potentiality of miraculous Moringa oleifera Lam. against recently evolved novel coronavirus and accompanying health complications. Moringa a well-proven super-food, densely packed with an abundant quantity of 92 minerals, several vitamins, 46 antioxidants, and numerous bioactive compounds, thus own a massive therapeutic potential for healing all levels of nutritional deficiencies and poor immunities and cure above 300 diseases. Moringa acts as anti-asthmatic, anti-cancerous, anti-diabetic, anti-inflammatory, hypotensive, hepatic, renal and cardio-protective, and anti-viral in nature. Thus it may reduce the severity of COVID-19 infections and associated serious medical emergencies. In addition, self-isolation at home or the workplace has put people at increased risk of physical and mental sicknesses, which could be simply addressed by integrating this wonderful plant into everyday diet. Furthermore, the immune-modulatory properties and viral inhibiting nature of moringa contribute to reduced risk of COVID-19 infection and quicker recovery from its symptoms. As per the existing pieces of literature, it is a great time to harness the esteemed moringa for safeguarding people from the terrible ongoing COVID-19 situation and other future pandemics.
Keywords: COVID-19; SARS-CoV-2; Moringa oleifera; immune-modulatory properties; pandemics
Coronaviruses of the coronaviridae family cause a range of lethal diseases in mammals and birds [1]. Recently, one of these viruses viz., severe acute respiratory syndrome coronavirus (SARS-CoV-2, formerly 2019-nCoV) triggered a severe respiratory disease outbreak in Wuhan, China [2,3] when the first human coronavirus was identified in 1965 [4]. Within no time, the virus has spread to several countries with high morbidity and mortality. The World Health Organization (WHO) immediately declared it as a ‘Global Health Emergency’ on January 30, 2020, and a ‘Global Pandemic’ on March 11, 2020. Soon, this virus has invaded in almost every country or territory across the world. As on January 03, 2022, it has infected over 298 million people and resulted in more than 5.46 million deaths worldwide [5]. The virus was reported to follow a droplet-borne transmission particularly through droplets of saliva or any discharge from the nose of infected individuals upon sneezing or coughing, with an incubation period of 2 to 14 days. Infected patients initially experienced mild respiratory illnesses, dry cough, tiredness, and fever and shortly started developing serious symptoms like breathing difficulty or shortness of breath, chest pain, loss of speech or taste dysfunction, multi-organ infections, which when left uncured, has led to severe medical complications or death. However, most of the COVID-19 patients (>80%) recover by themselves without requiring any special treatments or medications [6]. The virus is extremely contagious, infecting people of all age groups with a variety of symptoms. Furthermore, numerous studies have undoubtedly reported the higher sensitivity of people with already underlying medical conditions like asthma, chronic obstructive pulmonary diseases, influenza or pneumonia-like infections, hypertension, cancer, diabetes, cardiac or liver diseases, obesity, pregnancy, poor immunity, a habit of smoking, etc. towards COVID-19 infections [7]. Generally, viruses widespread in the population get an opportunity to replicate at an exceptionally high rate and undergo random genetic mutations to produce new variants with altered survivability, transmissibility, and infectivity [8,9]. Such variants or strains can be termed as sub-types of any major virus species with different characteristics. Being the RNA virus, it has soon evolved itself into several new strains with inherently better infection potential and affinity to escaping antibodies. These ‘variants of a great concern’ were reported in different countries imposing a great challenge to humanity particularly in a populated country like India. Though the SARS-CoV-2 virus upon continuous mutations pops up every day into several new strains, not all of them are important [10,11]. The deadly second COVID-19 wave has made India the world’s second worst-hit country, after the United States of America, leaving all its hospitals and crematoriums overwhelmed. India has alone reported above 35 million confirmed COVID-19 cases with more than 0.48 million fatalities so far according to WHO. Like earlier ‘Spanish flu’ caused by the H1N1 virus in 1918–19, the advanced surges of the COVID-19 were observed to be more devastating, posing a greater risk on public health [12]. The second and third waves of COVID-19 have challenged the whole medical system by infecting more than 0.8 million people every day worldwide (Fig. 1). By the end of 2021, the resurgence of this virus in the form of a new variant viz., “OMICRON” has become a major concern recently which again received the global attention.
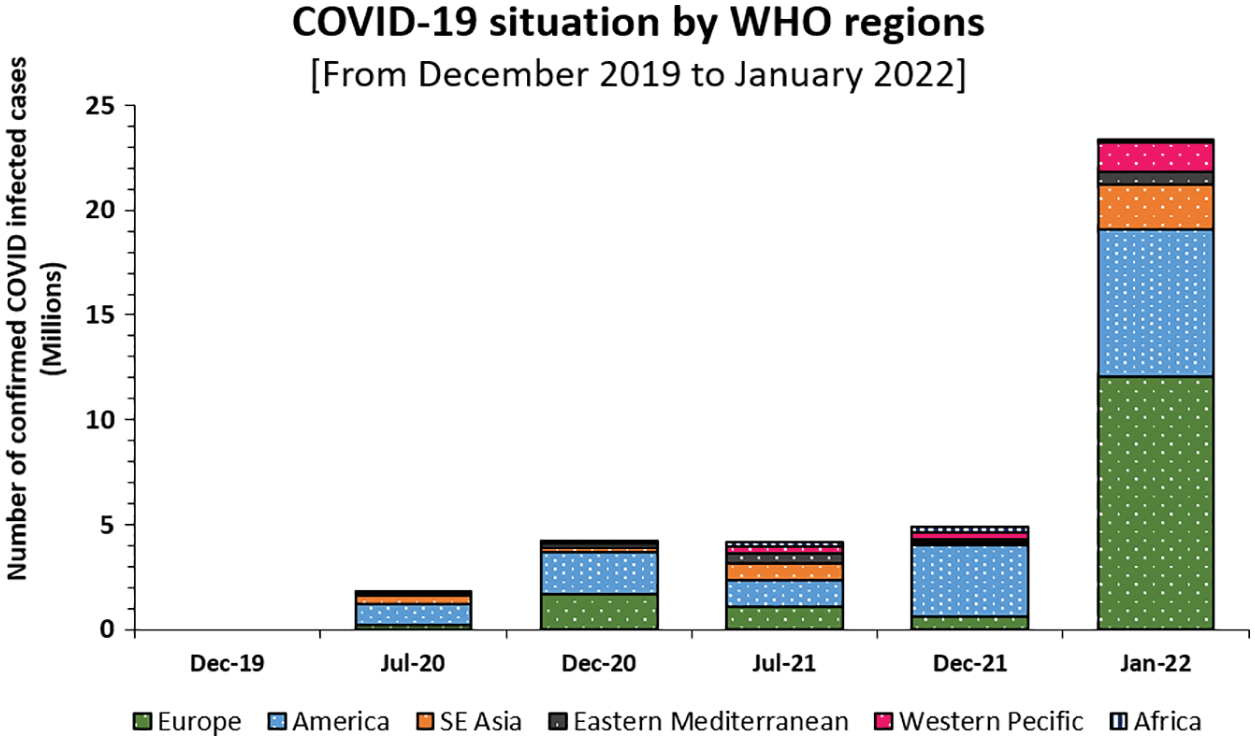
Figure 1: Global COVID-19 cases and death by WHO region as on January, 2022 (Source: WHO, 2022)
Another major task is to stop the re-infection of COVID-19 after getting vaccinated with the recommended doses [13–15]. Although, the vaccine is an effective means of preventing COVID-19 infections, no specific drugs or vaccines are available to cure the COVID-19 infections. Thus, it is extremely crucial to maintain a strong and healthy body immunity throughout the pandemic to avoid and recover any sort of medical complications [16,17]. Besides, people confined at home during the pandemic have been reported to be more lethargic, with a lot of confusion and anxiety over having COVID-19 infections [18–20], which has led to a reduction in all forms of physical activities and a rise of about 28% in everyday sitting and unhealthy food intake patterns [21–23]. This added tension makes people more susceptible to different physical and psychological conditions [24]. The consumption of selective foods enriched with essential minerals, vitamins, antioxidants, and bioactive compounds not only keep overall health maintained but also enhances the body immunity and develop confidence by protecting against the myriad of potentially hazardous diseases [12,25–27]. The psychological and dietary interventions are in turn vital determinants for increasing the effectiveness of approaches to public health vis-a-vis COVID-19 and future pandemics [28,29].
An unhealthy dietary pattern is often a major cause of a variety of chronic and Non-Communicable Diseases (NCDs) including cardiovascular diseases, strokes, cancer, diabetes, obesity, hypertension, etc. which are becoming more prominent global public health concerns [21,30–32]. Moreover, the changed food habit on the account of corona-phobia (fear of COVID-19) has further increased the risks of health issues [33]. On the contrary, regular healthy dietary intake not only optimizes proper body functioning but also strengthens the immune system, which subsequently reduces the development of serious chronic symptoms linked with COVID-19 [34]. Also, strong immunity increases the possibility of rapid recovery after COVID-19 infection [16]. Vitamins A, C, D, E, riboflavin, folic acid, cyanocobalamin, and minerals like copper (Cu), iron (Fe), selenium (Se), and zinc (Zn) are the most crucial micronutrients required for boosting the efficacy of T-cell mediated immunity and adaptive antibody responses [35]. Equally, consuming raw and unprocessed foods fortified with essential minerals, vitamins, amino acids, dietary fibers, and antioxidants greatly reduces the danger of NCDs which lead directly to increased mortality amid COVID-19 [16]. Usually, the energy intake should be by energy expenditure. Unhealthy weight gain could be avoided by keeping daily fat consumption below 30% of total energy intake avoids [36] particularly, saturated fats (<10%) and trans-fats (<1%) [37]. Free sugars below 10% of total daily energy intake do not permit diabetes and obesity [38], while daily salt intake of less than 5 g helps to prevent hypertension and the chances of heart diseases in the adult population [39].
There are several evidences that the ethnobotanical herbs enriched with ample immuno-modulatory bioactive compounds exhibit anti-viral properties by inhibiting either viral attachment, cell penetration, or viral RNA and protein synthesis. Additionally, plant-based foods increase the useful bacterial population in the intestinal which contributes up to 85% of body immunity [16]. During COVID-19, people were recommended to consume citrus fruits and red bell peppers for vitamin C [40], garlic, ginger, and turmeric [41] for antioxidants and anti-inflammation, oysters, cashews, and clams for zinc, and legumes, nuts, green leafy vegetables, and whole grains for magnesium, which lessen anxiety levels and quickly overcome infections through boosted immunity [16,42–44]. Studies have shown that some people have used Moringa oleifera for COVID-19 prevention, and its active ingredients can be used for treatment/prevention of COVID-19 [45–47]. Though several good sources of vitamins, minerals, and antioxidants are available worldwide, one of the most wonderful trees is M. oleifera, a promising super food that is packed with several times higher amounts of micronutrients, antioxidants, and bioactive compounds of vital nutritional and pharmacological applications [48]. These bioactive ingredients have multi-mechanistic modes of action against different types of viruses. All of the assumptions mentioned herewith are based on the idea that the mechanism of immune responses to COVID-19 are more or less similar to those of other coronaviruses and can be verified by future SARS-CoV-2 findings. This review highlights a unique opportunity of exploiting the potential of M. oleifera and its products for boosting the nutrition status and body immunity for effective prevention and cure of COVID-19 infections.
The information on the topic was retrieved from peer reviewed articles downloaded through various search engines, including PubMed, Google Scholar, Research gate and Scopus and many more. Different keyword combinations were used for retrieving the desired piece of information. Based on the scientific studies done on nutritional and therapeutic potential of M. oleifera, a perspective have been made in this review for boosting nutrition status and immunity defence amidst the ongoing COVID-19 disaster.
3 Moringa oleifera: An Overview
Moringa oleifera Lam. (Synonymously M. pterygosperma Gaertn.) is a fast-growing, drought-tolerant, perennial tree of the moringaceae family [49–53]. It is often nicknamed as ‘moringa’, ‘drumstick’, ‘horseradish’ ‘ben oil tree’ and has been considered as ‘nature’s gift to mankind’ since it is potentially the most nutrient-rich plant ever discovered on the planet. Though this shrubby plant is indigenous to the Southern Himalayan tract of North-Western India [54], it eventually got spread worldwide owing to its usefulness and high adaptive nature across diversified climates [55]. The genus Moringa with just 13 species from all across the world (Fig. 2) forms one of the most phenotypically and morphologically diverse groups among angiosperms that contain smaller plants to giant trees. However, M. oleifera is the most popular, studied, and widely distributed species for thousands of years [56,57]. In the sub-Himalayan region of India, Pakistan, Afghanistan, and Bangladesh, it was first reported as a medicinal herb around 2000 years Before Christ (BC) [58] and even before the first century Anno Domini (AD) as ‘Shigon’ in ancient literature called ‘Shushruta Sanhita’ [59]. Traditional uses of moringa have been well documented in Romans, Greeks, Egyptians, Unani, and Siddha medical systems [60–62]. In the recent few decades, moringa got ‘rediscovered’ because of its incredible multi-dimensional health care applications and henceforth retitled as miracle tree’, ‘tree of life’, ‘wonder tree’, ‘mother’s milk’ or ‘mother’s best friend’ [63–65]. Every part of this plant, i.e., leaves, pods, seeds, flowers, bark, and roots is edible and can be consumed by cooking or in the form of traditional medicines as it has got a great potential to treat malnutrition particularly in developing countries [65–69].
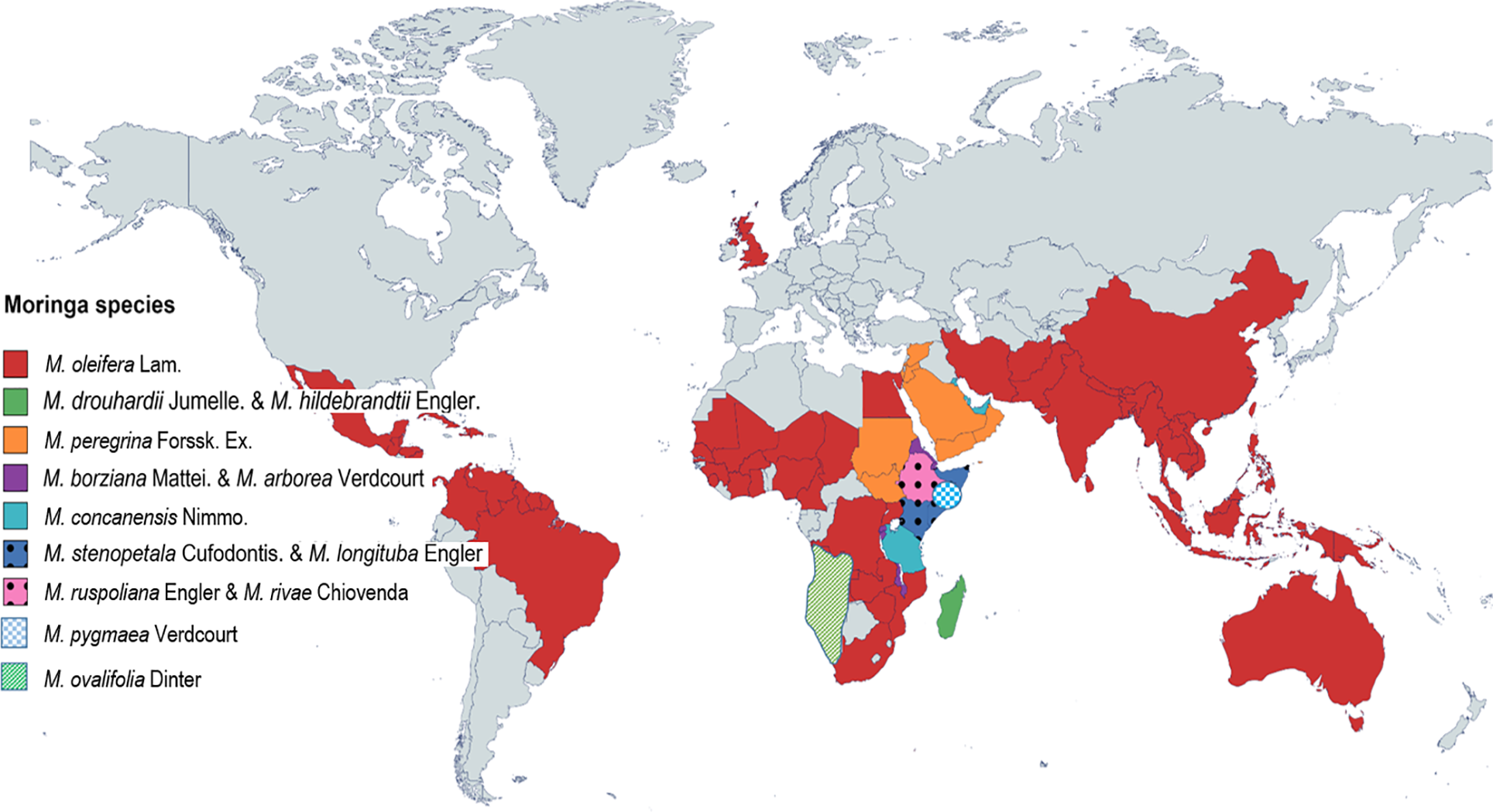
Figure 2: Worldwide geographical distribution of different species of Moringa [70]
Moringa leaves and pods are commonly consumed as vegetables in many countries. Leaves can be dried and crushed into a powder which is quite easy to store and consume by the users without losing any nutrition [71–73]. Seeds are also useful as they contain ‘Ben oil’ which is enriched with oleic acid (∼76%) conferring its resistance to oxidative degradation and a potential source for biodiesel and cosmetic industries [74,75]. Flowers also directly form part of the regular diet as they have hypo-cholesterolemic and anti-cancerous properties [76,77]. Roots and bark can also be consumed as a pickle or any other form which have hepato-protective, anti-inflammatory, anti-ulcer, and antimicrobial properties [50,54,76].
Morphologically, the moringa tree grows up to a height of 5–15 m, with a thick, straight and corky trunk; small, pale green, tri-pinnate compound leaves; 15–90 cm long pods, and a tuberous tap root system [78,79] as shown in Fig. 3. The yellowish-white, hermaphrodite and fragrant flowers (1–3 cm in size) are present in clusters of 15–25 cm area, and composed of sepals (05), petals (05), stamens (05) with anthers, and one pistil. Flowers primarily exhibit the anemophilous and entomophilous modes of pollination [80]. Fruits or pods are angular trilobite capsules containing up to 10–30, winged or wingless seeds [81]. It can be well adapted to a range of humid or dry hot climates and almost all kinds of soils with an annual rainfall of 250–3000 mm [82–84], and it is hence sometimes also referred to as ‘Never Die’ plant [85,86]. It has a true diploid set of chromosomes (2n = 28) and a genome size of ∼315 Mb [55,87,88]. As moringa species has a significant importance in terms of enhanced biodiversity, ethnobotanical, nutritional, pharmacological, and socio-economical perspectives, there is huge opportunity for commercial and industrial exploitation. Interestingly, India is a leading producer of moringa in the world with an annual production of over 2.2 million tonnes of tender fruits and 80% of the global moringa leaf supplier from a 43,600-hectare area [89]. Also, the Indian moringa products have predominantly captured the global market of over USD 8 billion worth) and are expected to grow more in the near future [89].

Figure 3: (a) Plant, (b) leaves, (c) mature pods, (d) tender pods, (e) seeds, (f & g) flowers, and (h) roots of Moringa oleifera
Despite the huge multi-dimensional progress made during the 21st century, the world has almost 690 million people (approximately 8.9%) undernourished [5]. The United Nations has declared the ongoing decade (2016–2025) as a ‘Decade of Action on Nutrition to make people understand and eliminate malnutrition in all its forms’ [90]. A tree like M. oleifera has proven itself as a superfood because it acts as a storehouse of nutrients, vitamins, minerals, antioxidants, and essential amino acids (Table 1). It is enriched with 92 minerals, 46 types of antioxidants, 36 anti-inflammatory agents, and vitamins [91–93]. From leaves to roots, moringa offers good quantities of important phytonutrients required for the overall growth and development of the body in addition to boosting the immunity [66,82,94,95]. In addition, it retains a larger number of electrolyte minerals, which keep the body hydrated even under dry conditions [96]. Altogether, it acts as an exceptionally rich source of low-cost nutrition. However, the nutritional content of the moringa tree varies based on the type of species, geographical location, season, and environmental conditions [97,98].
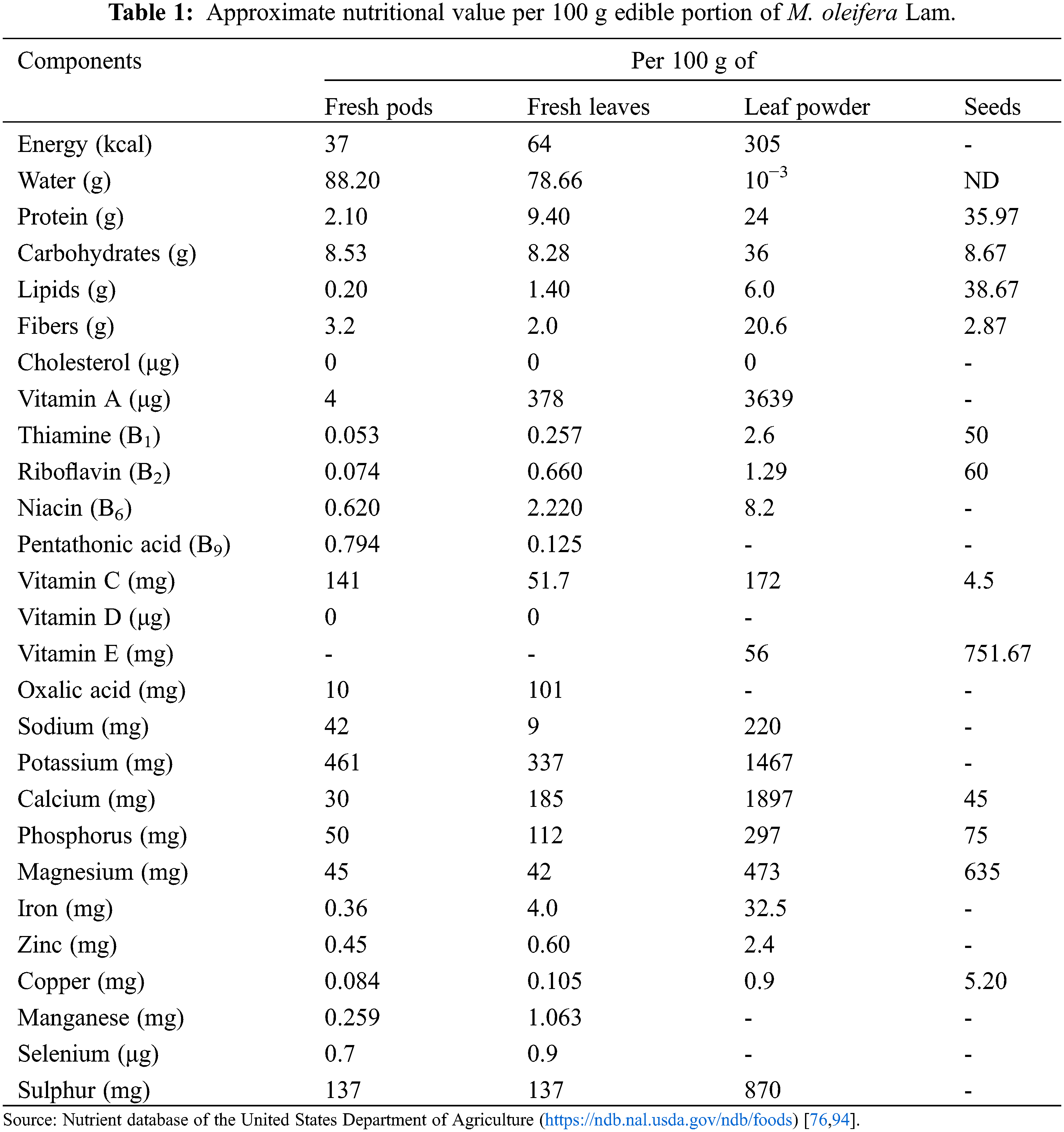
Essential nutrients are not synthesized in the body therefore, they are supplemented through diet. Moringa is an incredibly rich source of micronutrients besides having high quantities of proteins, carbohydrates, fibers, vitamins, and organic acids. Importantly, it is easily accessible to various third-world countries which are seriously struggling to combat malnutrition [98]. Further, numerous popular and scientific literatures in last few decades have highlighted the importance of moringa and thus, it has gained a huge focus worldwide [99]. The leaves are loaded with minerals like calcium, potassium, iron, and zinc [93,100] which can directly substitute the mineral tablets/capsules/syrup for supplementing the body metabolism and functions. The quantity of minerals in moringa is so high that 100 g of its dried leaf powder contains twice the fiber content than in wheat, 17 times more calcium than in milk, 9 times more proteins than in yogurt, 15 times more potassium than in bananas, 14 times more iron than in beef, 25 times more iron than in spinach, and 36 times more magnesium than in eggs [85,101–103] as shown in Fig. 4. Right from the child to adolescent to an old aged person, anybody can consume it in the way they like. Malnourished children (around 3 years’ age) deprived of breast milk can easily be cured by just feeding 100 g of its leaf powder that supplies around 75% and 50% of its daily iron and protein requirements, respectively, besides essential minerals, vitamins, and amino acids. Its high iron content provides a strong choice against anemia. A pregnant mother would just require around 6 spoonfuls of moringa leaf powder to fulfil her daily iron and calcium requirements [72]. Higher fiber (∼46.78%) and protein (∼20.66%) content of immature pods cures digestive problems and colon cancer [93,97]. Further, the amino acid profile of moringa is equivalent to milk and eggs which makes it as outstanding source of plant based-proteins for vegans and vegetarian as shown in the Table 2. Leaves, pods, and flowers contains up to 44%, 30%, and 31% of amino acid content. The flowers are also a good source of dietary fibers and antioxidants [101]. Beside this, moringa serves as a vital source of essential fatty acids, a prerequisite for optimal cellular health. The matured seeds contain odourless, sweet, non-drying, and healthy vegetable oil (35%–45% with 76% PUFA) which is equivalent to the olive oil rich in omega-3 fatty acids and proteins, and has a high thermal and oxidative stability [77,102,103]. This alternative source of oil becomes important as most of Africa’s and Asia’s poor communities are obliged to use cheaper quality oil due to inaccessibility to a healthier cooking oils and may pose severe health issues [65]. Another interesting fact is that the vegetables lose their nutrient content upon cooking or processing but it has been seen that the raw, cooked and dried leaves of moringa can be preserved with no refrigeration for a long time without sacrificing much of their nutrients [71].
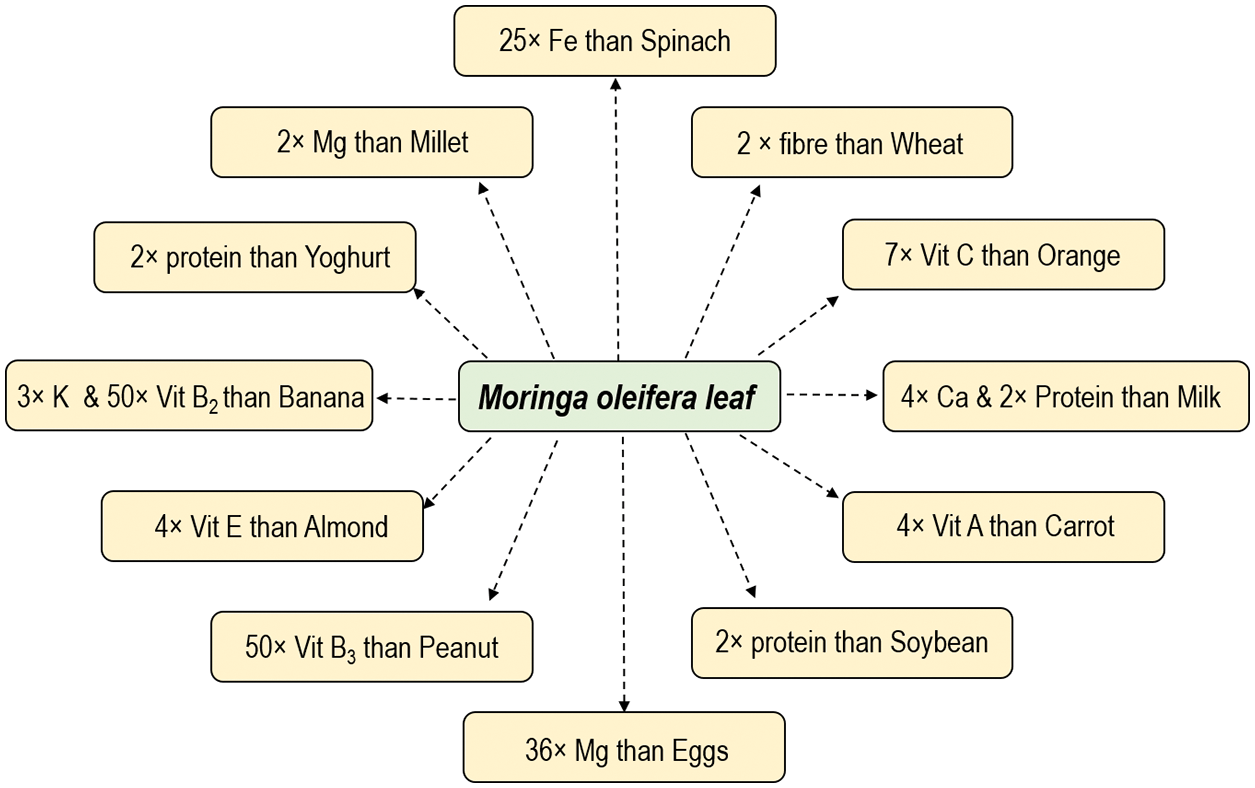
Figure 4: Comparative nutritional value of moringa and other important nutritional sources per 100 g of edible portion [97–99]

Vitamins are the organic molecules required essentially by the body metabolism but have to be supplemented through external diets [98,104]. Moringa has been observed to have exceptionally high quantities of vitamins like β-carotene (Vit A), B complexes viz., thiamine (Vit B1), riboflavin (Vit B2), niacin (Vit B3), pyridoxine (Vit B6), and folates (Vit B9), ascorbic acids (Vit C), and alpha-tocopherol (Vit E). A 100 g of dry leaf powder of moringa holds 7 times higher vitamin C than oranges, 10 times higher β-carotene than carrots, and 4 times more vitamin E than almonds [97–99]. A child can fulfil all his/her vitamins A and C requirements just by consuming 20 g of moringa leaves. The availability of β-carotene and vitamin C also induces antioxidant and anti-inflammatory activities in the body [105]. Sterols present in the pods potentially inhibit the formation of free radicles. However, the seasonal variation can significantly impact the vitamin content as a higher abundance of β-carotene was found in the hot and humid season, while ascorbic acid and iron were more abundant in the cool and dry season [106].
Antioxidants are the substances that delay the aging of cells and the body by preventing the formation and neutralization of unstable molecules which otherwise lead to heart diseases, cancer, and other serious illnesses [112]. They enhance the life of an individual by acting as ‘free radicle scavengers’. Moringa owns a wide range of vital antioxidants which in combination are proven to be more effective than a single antioxidant due to synergistic mechanisms [88,113–115]. Compounds like ascorbic acid, flavonoids, phenols, carotenoids, terpenoids, saponins, and tannins play a major role in antioxidant activities in the body [116]. Among phenolics, gallic acid and chlorogenic acid whereas, among flavonoids, rutin, luteolin, quercetin, apigenin, and kaempferol are the potent natural antioxidants available in moringa [104]. Flavonoids intake protects against various chronic diseases accompanied by oxidative stress. Similarly, polyphenols act as major plant molecules for reducing oxidative damage in tissues through scavenging free radicals. Quercetin is also a robust antioxidant that exhibits numerous therapeutic properties. The high quantities of antioxidant compounds also enhance the overall shelf-life of lipid containing foods by preventing lipid oxidation [117,118]. Thus, moringa particularly its leaves are unambiguously the rich source of cost-effective, natural antioxidants particularly for nutritionally deprived societies.
The traditional medicine practitioners were well acquainted with the remedial properties of plants against various sicknesses even without knowing the bioactive molecules present in them. The modern medical researchers are more focused on the plant-based bioactive molecules for treating the chronic diseases and disorders as they pose almost no side effects [118]. In general, these bioactive molecules or secondary metabolites are responsible for adaptation and survival mechanisms of the plants but could be well used in developing medicines for curing various human and animal diseases. A well-known phrase, “Give the body what it needs and it will heal itself” cites well particularly when it comes to the highly esteemed plant like moringa, which is a hub of medicinal properties for curing more than 300 diseases in human [78,94,119] as listed in Fig. 5. Numerous scientific studies on humans have expounded its health curing properties. Either leaves, fruits, seeds, flowers, roots or its bark, all contains a plentiful of pharmacologically useful compounds [120]. People in India and African countries consider moringa as a folk medicine for ages.
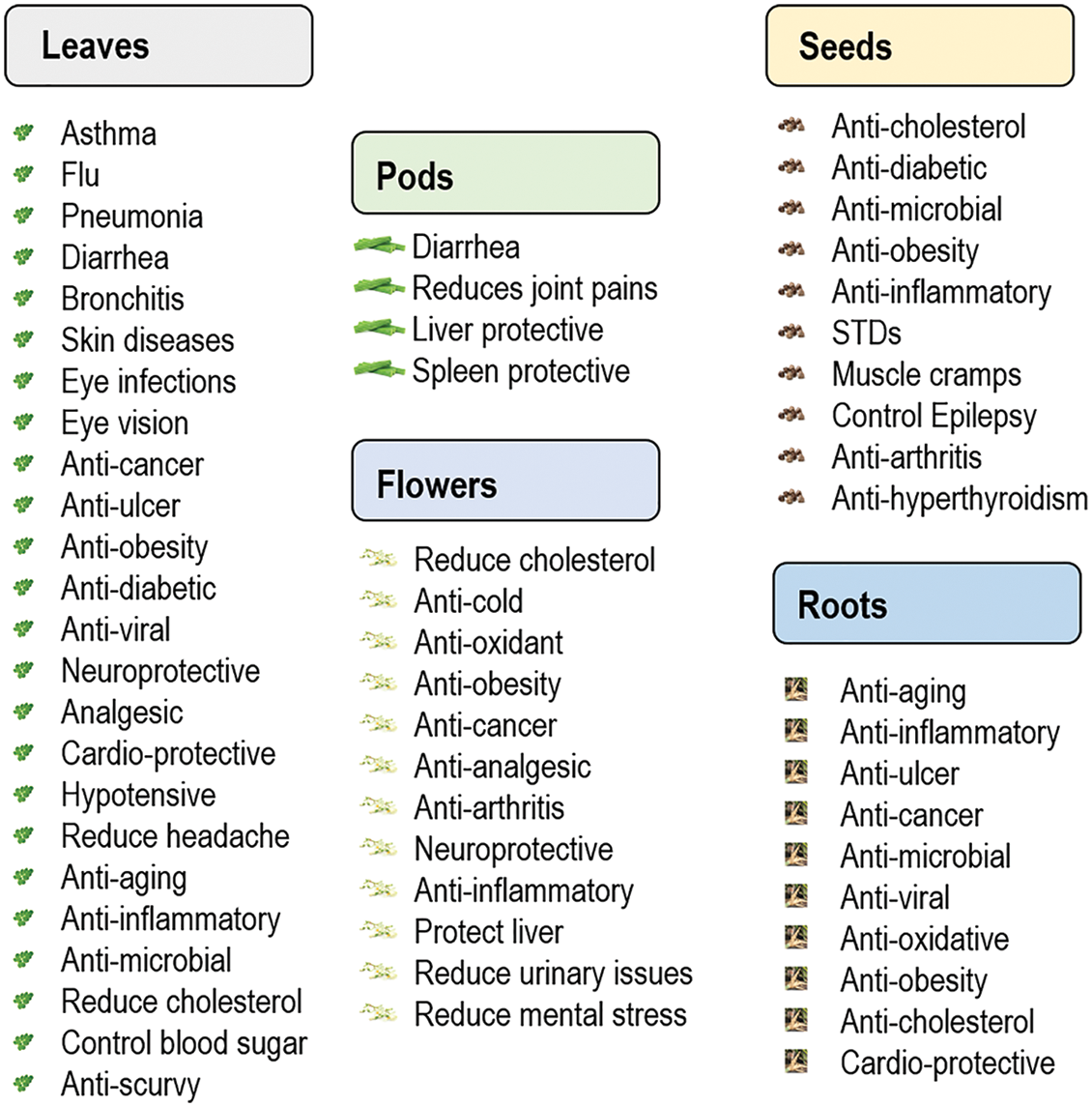
Figure 5: Spectrum of major human disease curing potential of moringa plant parts [76,94,119–128]
3.2.1 Bioactive Compounds and Their Biological Activities
The therapeutic potential of moringa is due to abundance of a wide repertoire of minerals, vitamins, antioxidants, phytochemicals viz., alkaloids, carotenoids, flavonoids, glucosinolates, glycosides, iso-thiocyanates, phenols, polyphenols, sterols, saponins, tannins, terpenes, and their derivatives [51,103,120,122–124] as shown in Table 3. Several studies have highlighted the usefulness of moringa leaves, pods, flowers, seed, seed oil, roots, and bark against prominent diseases like cancer, diabetes, hypertension, hyper-glycemia, hyper-cholesterol, high blood pressure, depression, atherosclerosis, tuberculosis, rheumatoid arthritis, chronic anaemia, haemorrhage, infertility, inflammation, ulcers, tumours, epilepsy, skin allergies, digestive or gastrointestinal disorders, diarrhea, asthma and other respiratory diseases, knee join disorders, memory disorders, fever, wounds, body and tooth aches, cough, gouts, cramp, syphilis, and sexually transmitted diseases and microbes like bacteria, fungus and viruses, as well as regulates diseases related to kidney, lever, heart, eyes, and thyroids, etc. [48,63,68,76,78,93,94,104,120,121,124–129,139]. In Ayurveda, the tri-doshas or illnesses like Vata (space & air), i.e., energy of movement, pitta (fire & water), i.e., energy of digestion and metabolism, and Kapha (earth & water), i.e., energy of structure and lubrication, are accounted for all kind of physical, physiological, mental, and emotional health of an individual [140,141]. Moringa is said to heal up the vitiation of these illnesses naturally [94,142]. Among diseases, cancer is a very prominent one that has grasped millions of the people worldwide. Compounds like benzyl isothiocyanate, glucosinolates, hexadecanoic acid ethyl ester, niazimicin, and rhamnosides in moringa potentially exhibit a protective role against DNA damage in cells associated with cancer and tumor progression [104,142,143].

Consuming moringa leaves cures pancreatic and breast cancers [89,140]. It acts well against diabeties and treats both Type-1 diabetes, i.e., non-production of insulin and Type-2 diabetes i.e., insulin resistance [76,144–146]. The flavonoids particularly quercetin and phenols scavenge ROS in mitochondria and protect the beta cells which controls the body glucose level [147,148]. A high quantity of phenols and phenolic derivatives present in moringa are responsible for the antioxidant and neuroprotective activities [114,149]. Moringa reduces the acidity in gastric ulcers (∼86.15% at 500 mg concentration), thus acts as an antiulcer agent [150,151]. Phenols and flavonoids inhibit the pancreatic cholesterol esterase activities and thereby play significant roles in regulating hyperlipidemia [148,152]. Hypertension is a growing sickness among people worldwide possibly due to imbalanced physical, mental, and dietary activities. Compounds like thiocarbamate, isothiocyanate, niazinin, niazimicin, hydroxybenzoate, and β-sitosterols from leaves and pods of moringa exert the lowering effects on high blood pressure and hypertension [122,153,154]. The leaf extract exhibits a significant anti-inflammatory and immunomodulatory effects through ethyl acetate, flavonoids, and phenols which majorly inhibit the NF-κB pathways [155,156]. Quercetin present in the leaves shows hepatic and renal-protective properties by lowering hepatic lipids and lipid peroxidation activities [157].
Almost all parts of moringa plant exhibit anti-microbial activities particularly due to alkaloids, flavonoids, gallic acid, isothiocyanates, phenols, pyterigospermin, moringine, saponins, and tannins present in it. Development of bacteria like Enterococcus faecalis, Streptococcus aureus, Streptococcus mutans, Candida albicans, Staphylococcus aureus, Staphylococcus epidermidis, Salmonella enterica, Pseudomonas aeruginosa, Klebsiella pneumonia, Escherichia coli, fungi like Aspergillus spp., Candida spp., Fusarium solani, Hortaea werneckii, Microsporum canis, Mucor spp., Penicillium sp. Rhizopus sp., Trichoderma spp., and Trichophyton spp. can be checked through moringa [119]. Besides this, several reports have highlighted its antiviral activities as well such as against Immunodeficiency Viruses (HIV/AIDS), Herpes Simplex Virus type 1 (HSV), Hepatitis B Virus (HBV) in humans, and Epstein Barr Virus (EBV), Foot and Mouth Disease Virus (FMDV), and Newcastle Disease Virus (NDV) in animals, particularly due to the action of niaziminin, a thiocarbamate which prevents RNA viruses [158]. Recently, new compounds viz., glucomoringin and vitexin have been extracted from moringa which exhibited strong inhibitory properties against the H1N1 virus [135]. The extract of moringa plant parts was also reported to have the anti-hyperthyroidism, [159–163], anti-trypanosomal, a parasitic protozoan [164], anti-leishmanial [165], antihelmintic against Pheritima posthuma [166], anti-atherosclerotic [167], anti-spasmodic [123], and analgesic [168] functions. Its leaves and fruits contain high quantities of β-carotene, thus can prevent cataracts and night blindness [120]. Consuming moringa flowers can help in curing urinary problems and also increase the mother’s milk production. Moringa seed oil is useful against diarrhoea and also has laxative properties. Further, the extracts of moringa have also been reported with the hepato-protective [43], renal-protective, and cardiovascular-protective [76,120,169,170] functions. The root extract inhibits the development of uterus and blastocyst implantation, therefore, traditional Indian women often used this as a natural oral contraceptive [171,172]. The therapeutic functions of different bioactive compounds of moringa have been proven through numerous scientific experiments and all of them have supported the traditional medicinal utility followed since pre-historic time in countries like India. In general, moringa offers a natural, reliable, safe, and cost-effective means of preventing and curing several major chronic diseases in humans and animals and is therefore considered as a miracle or wonder plant [68].
4 Protective Role of M. oleifera against COVID-19 and Comorbidities
Confining at home and restricting regular activities during pandemics like COVID-19 significantly impacted the health of a common man. Ensuring a healthy lifestyle through adequate nutrition is highly anticipated during the COVID-19 pandemic particularly for people with poor immunity or COVID infection [41]. The sufficient intake of minerals particularly zinc and iron, as well as vitamins like A, B6, B12, C, and E are the pre-requisite for keeping the body immunity strengthened during the pandemic to fight against various health issues. Zinc is an essential constituent of the human proteome and is involved in a variety of signalling events of antiviral responses, whereas iron forms an integral part of haemoglobin that carries oxygen to all the body parts [173]. Ascorbic acid is an important water-soluble vitamin required for strengthening the general body immunity. In this backdrop, several scientific studies have the M. oleifera as a storehouse of proteins, carbohydrates, lipids, dietary fibers, minerals, vitamins, antioxidants, and hundreds of bioactive compounds, thus constituting a part of optimum nutrition for ensuring the body against any assault by the virus [45]. As every part of moringa plant is densely packed with essential nutrients, even a small quantity of it in the regular diet can fulfil the daily nutritional requirements. It supplies vitamin C in higher quantity than the daily dietary requirement for men (90 mg/d) and women (75 mg/d) [99]. The daily requirements of almost all important minerals and vitamins can also be easily achieved by consuming moringa on daily basis. This protect the individual’s metabolic health perfectly against any underlying chronic illnesses [81]. Furthermore, consuming moringa reduces the physical and mental fatigue, and improves the mental clarity of the people self-isolated at home. Effective hand-washing product from the moringa leaves has also been reported that exhibited strong microbial inhibition. Using such natural hand-washing products could be a good alternative to synthetic and chemical-based products which may have some adverse effects in the long-term.
Particularly, COVID-19 which is mainly associated with immune-inflammatory injury and enhanced oxidative stress that alters the regular functioning of multiple body organs. Antibodies and cell-mediated immune responses through CD8 and CD4 T cells are primarily required to overcome such infections. Acquiring tolerance against COVID-19 definitely requires an elevated immune response, and moringa acts as a potent immune-stimulant for significantly enhancing the number of immune responsive cells even at low concentrations viz., 0.1 μg/mL of extract [113]. Thus, its potentiality in boosting the body’s immunity and treating numerous underlying diseases makes it a promising candidate for both easier preventing as well as curing the suspected COVID-19 patients besides sustaining a good physical and mental health [53,94,113,174–177]. The antioxidants and secondary metabolites in moringa detoxify the body and repair the damaged body cells in the course of COVID-19 infection. The leaves are clinically used for treating bronchitis, sore throat, headaches, and constipation, etc., which corresponds to the symptoms of COVID-19 infections [178]. Studies have shown antiviral properties of biomolecules like kaempferol, pterygospermin, morphine, quercetin, rutin, β-sitosterol, niazirin, niaziminin, niazimicin, isorhamnetin, apigenin, etc. [76,94,176,179,180]. These bioactive molecules either prevent the virus to enter the cell or do not allow their viral RNA or protein formation, thus checking their attack in the body. Moreover, the abundance of ascorbic acid and 46 types of antioxidants in moringa diminishes the chances of inflammation, hypertension, cancer, cardiovascular diseases, oxidative damage, and many other diseases in the body which have been reported to increase the severity of COVID-19 symptoms with higher mortality [48]. These compounds from moringa plant can exhibit significant therapeutic activities against COVID-19 by blocking a wide range of signalling pathways as depicted in Fig. 6. However, comprehensive in-depth studies may be required to have a better understanding on these aspects. Importantly, the molecular docking studies have identified ellagic acid and apigenin in moringa as potentially antiviral compounds particularly against the SARS-CoV-2 virus. Recently, the S protein and SARS-CoV-2 main proteinase 3CL Mpro are two essential proteins required by SARS-CoV-2 for its multiplication. The inhibition of 3CL Mpro by natural inhibitors from M. oleifera have been recently demonstrated [175,181–183] which indicates its potential as a natural drug candidate against COVID-19.

Figure 6: Schematic illustration of multi-site inhibitory action of bioactive compounds of Moringa oleifera against SARS-CoV-2
5 Ways to Consume Moringa oleifera
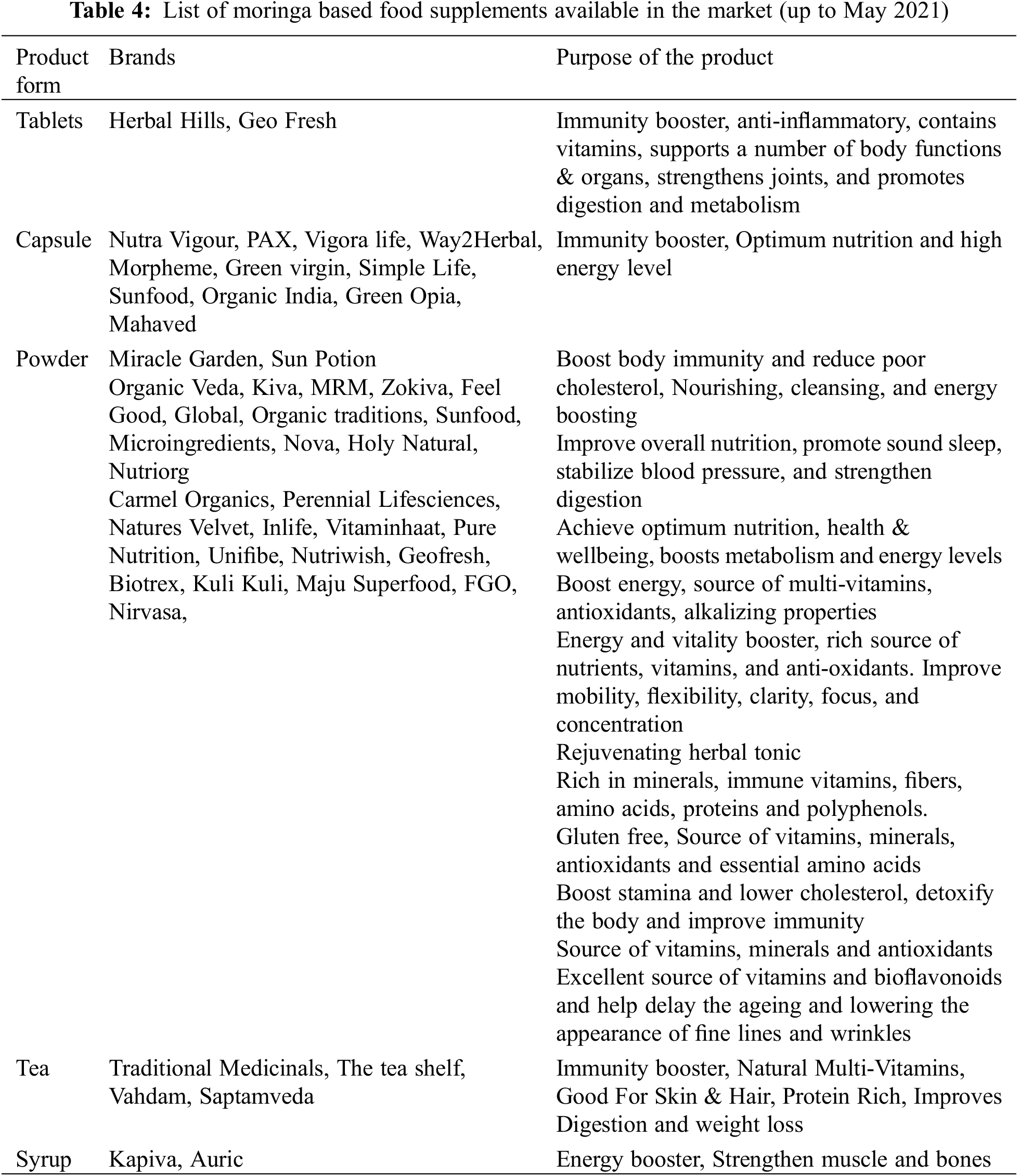
A popular phrase, “What’s old is new again” suits well to the age-old moringa, as recently it has been brought into mainstream nutrition and therapeutic care in several countries. Every part of moringa plant can be consumed in several ways [94]. Leaves are consumed either as leafy green vegetables or as powder, whereas fresh, young pods are consumed by boiling or frying in different dishes. The young seeds in the pod are quite soft and have a great potential to detoxify the bloodstream [77]. Flower buds are highly nutritious and consumed as fried snacks or added to a range of dishes [73]. Tuberous roots are used to prepare pickles and powder which can be consumed in any way. Though the moringa has a strong food safety profile, it does not exhibit any considerable side effects. Studies have suggested ∼70 g or 10–12 teaspoons of moringa as a single dose or 7–8 g/day for 28 days to be a safe dose for consumption [184]. A number of moringa based products are available in the market (Table 4). The various ways to consume moringa are as follows;
Steep as tea: Just like tea leaves, add some fresh or dry moringa leaves or powder to the boiling water and consume it as usual tea. The taste can be improved by adding honey and lemon as desired.
Stir into water: Moringa leaf powder can be directly dissolved in cold or warm water and drank afterwards.
Blend in juice/shake/soup: Moringa powder is slightly bitter and sweet in taste, which can directly be added in shakes or soup to boost nutrition intake.
Mixed in smoothie: Moringa fresh leaves or powder well combines with cooked food work as a smoothie for high quality and flavour it may be blended with suitable fruit.
Augment in curries: Leaves can be simply consumed as green leafy vegetables, while tender pods can be cut into pieces and mixed in a wide range of delicious curries/recipes. It adds extra nutrition, flavour and taste in the dish.
Bake with anything: Moringa leaves or powder can be directly added to a vast array of baked items. At home, it can be simply added to the wheat flour just like coriander leaves.
As salad: Slightly roasted fresh moringa leaves or powder can be directly added or sprinkled onto the salad or mixed with cooked rice for improved flavour and digestion.
As pickles: Moringa pods or roots can be consumed as nutritious and tasty pickles.
Capsules/tablets: Moringa powders are available in the form of capsules and tablets. Usually, 1-4 capsules or tablets twice a day can be directly consumed with warm water. It is simple, convenient and the most popular way to consume moringa.
Moringa syrup: The readymade moringa syrups are available in the market or can be prepared at home. Drinking 1–2 teaspoons of syrup twice a day will help in reducing blood pressure and other chronic diseases.
Moringa oil: Moringa seed oil can be used as cooking oil or essential skin or hair oil for deep hydration.
6 Safety and Efficacy of Consuming Moringa oleifera
People have been using moringa as a folk medicine since ages [185]. In last few decades, numerous scientific experiments on the positive and negative effects of moringa on human and animals, particularly rodents, have been carried out [72,154,186,187] and efficacy and risks of different extracts have been assessed. A very high degree of safety was established at certain dose in most of the experiments [51,180]. However, the safety of consuming moringa may depend upon the dosage, plant part used, and the type of preparation to be administered [188]. In general, no toxicity or adverse effects were observed in human-based studies at lower doses.
The general recommendation is starting with a small dose and increasing slowly as per the need up to a maximum limit. However, preference to any superfood supplement requires to check the brand’s recommended dosage for optimal effectiveness. The bioactive compounds in moringa do not have any mutagenic effects and liver injury. Therefore, they could be effectively utilized for drug development by the industries. However, the dosages significantly above the quantity recommended may have some negative effects [186,189,190]. Therefore, more scientific investigations are required to understand the nature of adverse effects of doses administered orally in humans [191]. As a cost-effective and safe pharmacologic and nutritional alternative for public health and marginal farmers, moringa has achieved a significant international hype and trade growth during the last few decades [192].
COVID-19 has emerged as a global epidemic outbreak leaving probably none of the countries unaffected. In a small period of time, the virus has mutated itself several times and resulted into a more infectious and deadly forms which led to the death of millions of people worldwide. Although people are getting vaccinated, the risk of infection still remains high particularly in the populated nations. The level of immunity determines the likelihood of disease severity and accompanying symptoms; thus, the selection of a balanced diet is crucial for an enhanced nutrition and robust body immunity, which certainly reduces the menace of disease complications including death. Moringa oleifera serves as a socio-economically important plant with a plentiful of nutritional, pharmacological, and industrial applications. It has a multi-faceted usage in bioscience besides curing a range of general health problems, chronic diseases, vitamin and nutritional deficiencies. Several scientific studies have suggested that the high quantities of nutrients, vitamins, antioxidants, and bioactive molecules in moringa can prevent the risk of viral attack and heal up the body during viral attack. Further, the molecular docking studies highlighted its potentiality as natural drug candidate against COVID-19. This would act as inexpensive and eco-friendly alternative to synthetic medicines against COVID-19 particularly for a poor and vulnerable sector of the society who get a limited access to costlier modern technological resources. Its high level of safety further confirms the effectiveness of moringa in healing the body and achieving a high level of confidence. Therefore, it is a great time to exploit the immune-modulatory properties of this esteemed and high-quality gift of nature either through direct consumption or pharmacological development to safeguard people from the scariest ongoing and future pandemics. As a matter of fact, the saying ‘prevention is better than cure’ is always important to be considered. Therefore, developing a strong immune system by adding these wonderfull tree products to the human lifestyle can be a commendable move towards building a reliable first wall of defence to fight the current and future pandemics rather than becoming a prey of them immediately.
Authorship: The authors confirm contribution to the paper as follows: study conception and design: HA, RKS, AS, AB, AR, AKH, AA; data collection: HA, RKS, LC, ST, KR; analysis and interpretation of results: HA, RKS, YT, HY, KRH, DÖ, AELS; draft manuscript preparation: HA, RKS, AS, AB. All authors reviewed the results and approved the final version of the manuscript.
Acknowledgement: Authors are thankful to the Director, ICAR-Central Agroforestry Research Institute for his continuous support and motivation during conceptualizing the idea and formulating the manuscript.
Funding Statement: The authors received no specific funding for this study.
Conflicts of Interest: The authors declare that they have no conflicts of interest to report regarding the present study.
1. El-Sayed, A., Kamel, M. (2021). Coronaviruses in humans and animals: The role of bats in viral evolution. Environmental Science and Pollution Research, 28(1), 1–12. DOI 10.1007/s11356-021-12553-1. [Google Scholar] [CrossRef]
2. CDC (2019). Novel coronavirus. Wuhan, China, CDC. https://www.cdc.gov/coronavirus/2019-ncov/about/index.html. [Google Scholar]
3. Gorbalenya, A. E., Baker, S. C., Baric, R., Groot, R. J. D., Drosten, C. et al. (2020). Severe acute respiratory syndrome-related coronavirus: The species and its viruses–A statement of the coronavirus study group. bioRxiv. DOI 10.1101/2020.02.07.937862. [Google Scholar] [CrossRef]
4. Fong, S. J., Dey, N., Chaki, J. (2021). Artificial Intelligence for Coronavirus Outbreak, Springer Nature Singapore. DOI 10.1007/978-981-15-5936-5_1. [Google Scholar] [CrossRef]
5. WHO (2021). Coronavirus disease (COVID-19) pandemic. https://www.who.int/emergencies/diseases/novel-coronavirus-2019. [Google Scholar]
6. Balachandar, V., Mahalaxmi, I., Subramaniam, M., Kaavya, J., Senthil Kumar, N. et al. (2020). Follow-up studies in COVID-19 recovered patients–Is it mandatory? The Science of the Total Environment, 729, 139021. DOI 10.1016/j.scitotenv.2020.139021. [Google Scholar] [CrossRef]
7. CDC (2021). Underlying medical conditions associated with high risk for severe COVID-19: Information for healthcare providers. https://www.cdc.gov/coronavirus/2019-ncov/hcp/clinical-care/underlyingconditions.html. [Google Scholar]
8. Li, Q., Wu, J., Nie, J., Zhang, L., Hao, H. et al. (2020). The impact of mutations in SARS-CoV-2 spike on viral infectivity and antigenicity. Cell, 182(5), 1284–1294. DOI 10.1016/j.cell.2020.07.012. [Google Scholar] [CrossRef]
9. Paital, B. (2020). Nurture to nature via COVID-19, a self-regenerating environmental strategy of environment in global context. Science of the Total Environment, 729, 139088. DOI 10.1016/j.scitotenv.2020.139088. [Google Scholar] [CrossRef]
10. Das, K., Pingali, M. S., Paital, B., Panda, F., Pati, S. G. et al. (2021). A detailed review of the outbreak of COVID-19. Frontier in Bioscience, 26, 149–170. DOI 10.52586/4931. [Google Scholar] [CrossRef]
11. Martin Webb, L., Matzinger, S., Grano, C. (2021). Identification of and surveillance for the SARS-CoV-2 variants B.1.427 and B.1.429–Colorado, January–March 2021. Morbidity and Mortal Weekly Report, 70(19), 717–718. DOI 10.15585/mmwr.mm7019e2. [Google Scholar] [CrossRef]
12. Knorr, D., Khoo, C. S. H. (2020). COVID-19 and food: Challenges and research needs. Frontiers in Nutrition, 7. DOI 10.3389/fnut.2020.598913. [Google Scholar] [CrossRef]
13. Kim, A. Y., Gandhi, R. T. (2020). Reinfection with severe acute respiratory syndrome coronavirus 2: What goes around may come back around. Clinical Infectious Diseases, 73(9), e3009–e3012. DOI 10.1093/cid/ciaa1541. [Google Scholar] [CrossRef]
14. Ledford, H. (2020). COVID-19 reinfection: Three questions scientists are asking. Nature, 585(7824), 168–169. DOI 10.1038/d41586-020-02506-y. [Google Scholar] [CrossRef]
15. Cavanaugh, A. M., Spicer, K. B., Thoroughman, D., Glick, C., Winter, K. (2021). Reduced risk of reinfection with SARS-CoV-2 after COVID-19 vaccination—Kentucky. Morbidity and Mortality Weekly Report, 70(32), 1081. DOI 10.15585/mmwr.mm7032e1. [Google Scholar] [CrossRef]
16. Arshad, M. S., Khan, U., Sadiq, A., Khalid, W., Hussain, M. et al. (2020). Coronavirus disease (COVID-19) and immunity booster green foods: A mini review. Food Science and Nutrition, 8(8), 3971–3976. DOI 10.1002/fsn3.1719. [Google Scholar] [CrossRef]
17. Calder, P. C. (2021). Nutrition and immunity: Lessons for COVID-19. European Journal of Clinical Nutrition, 75, 1–10. DOI 10.1038/s41430-021-00949-8. [Google Scholar] [CrossRef]
18. Ingram, J., Maciejewski, G., Hand, C. J. (2020). Changes in diet, sleep, and physical activity are associated with differences in negative mood during COVID-19 lockdown. Frontiers in Psychology, 11, 2328. DOI 10.3389/fpsyg.2020.588604. [Google Scholar] [CrossRef]
19. Paital, B., Das, K., Parida, S. K. (2020). Internation social lockdown versus medical care against COVID-19, a mild environmental insight with special reference to India. Science of the Total Environment, 728, 138914. DOI 10.1016/j.scitotenv.2020.138914. [Google Scholar] [CrossRef]
20. Paital, B., Agrawal, P. K. (2020). Air pollution by NO2 and PM2.5 explains COVID-19 infection severity by overexpression of angiotensin-converting enzyme 2 in respiratory cells: A review. Environmental Chemistry Letters, 19(1), 25–42. DOI 10.1007/s10311-020-01091-w. [Google Scholar] [CrossRef]
21. Kaur, H., Singh, T., Arya, Y. K., Mittal, S. (2020). Physical fitness and exercise during the COVID-19 pandemic: A qualitative enquiry. Frontiers in Psychology, 11, 590172. DOI 10.3389/fpsyg.2020.590172. [Google Scholar] [CrossRef]
22. Labrague, L., Ballad, C. A. (2020). Lockdown fatigue among college students during the COVID-19 pandemic: Predictive role of personal resilience, coping behaviours, and health. Perspectives in Psychiatric Care, (5), 1–8. DOI 10.1111/ppc.12765. [Google Scholar] [CrossRef]
23. Ammar, A., Brach, M., Trabelsi, K., Chtourou, H., Boukhris, O. et al. (2020a). Effects of COVID-19 home confinement on eating behaviour and physical activity: Results of the ECLB-COVID19 international online survey. Nutrients, 12, 1583. DOI 10.3390/nu12061583. [Google Scholar] [CrossRef]
24. di Santo, S. G., Franchini, F., Filiputti, B., Martone, A., Sannino, S. (2020). The effects of COVID-19 and quarantine measures on the lifestyles and mental health of people over 60 at increased risk of dementia. Frontiers in Psychiatry, 11, 1–14. DOI 10.3389/fpsyt.2020.578628. [Google Scholar] [CrossRef]
25. de Souza Monnerat, J. A., de Souza, P. R., da Fonseca Cardoso, L. M., Mattos, J. D., de Souza Rocha, G. et al. (2021). Micronutrients and bioactive compounds in the immunological pathways related to SARS-CoV-2 (adults and elderly). European Journal of Nutrition, 60(2), 559–579. DOI 10.1007/s00394-020-02410-1. [Google Scholar] [CrossRef]
26. Bhushan, I., Sharma, M., Mehta, M., Badyal, S., Sharma, V. et al. (2021). Bioactive compounds and probiotics–A ray of hope in COVID-19 management. Food Science and Human Wellness, 10(2), 131–140. DOI 10.1016/j.fshw.2021.02.001. [Google Scholar] [CrossRef]
27. Cámara, M., Sánchez-Mata, M. C., Fernández-Ruiz, V., Cámara, R. M., Cebadera, E. et al. (2021). A review of the role of micronutrients and bioactive compounds on immune system supporting to fight against the COVID-19 disease. Foods, 10(5), 1088. DOI 10.3390/foods10051088. [Google Scholar] [CrossRef]
28. Naja, F., Hamadeh, R. (2020). Nutrition amid the COVID-19 pandemic: A multi-level framework for action. Europian Journal of Clinical Nutrition, 74, 1117–1121. DOI 10.1038/s41430-020-0634-3. [Google Scholar] [CrossRef]
29. Maffoni, S. I., Kalmpourtzidou, A., Cena, H. (2020). The potential role of nutrition in mitigating the psychological impact of COVID-19 in healthcare workers. Nutrition and Food Science, 22, 6–8. DOI 10.1016/j.nfs.2020.12.002. [Google Scholar] [CrossRef]
30. Jayedi, A., Soltani, S., Abdolshahi, A., Shab-Bidar, S. (2020). Healthy and unhealthy dietary patterns and the risk of chronic disease: An umbrella review of meta-analyses of prospective cohort studies. The British Journal of Nutrition, 124(11), 1133–1144. DOI 10.1017/S0007114520002330. [Google Scholar] [CrossRef]
31. Yamada, M., Hapsari, E. D., Matsuo, H. (2020). Behaviors toward non-communicable diseases prevention and their relationship with physical health status among community-dwelling, middle-aged and older women in Indonesia. International Journal of Environmental Research and Public Health, 17(7), 2332. DOI 10.3390/ijerph17072332. [Google Scholar] [CrossRef]
32. Idris, I. B., Azit, N. A., Ghani, S. R. A., Nor, S. F. S., Nawi, A. M. (2021). A systematic review on noncommunicable diseases among working women. Industrial Health, 59(3), 146–160. DOI 10.2486/indhealth.2020-0204. [Google Scholar] [CrossRef]
33. Hassen, B., El Bilali, T., S Allahyari, H., M. (2020). Impact of COVID-19 on food behavior and consumption in Qatar. Sustainability, 12(17), 6973. DOI 10.3390/su12176973. [Google Scholar] [CrossRef]
34. Cena, H., Calder, P. C. (2020). Defining a healthy diet: Evidence for the role of contemporary dietary patterns in health and disease. Nutrients, 12(2), 334. DOI 10.3390/nu12020334. [Google Scholar] [CrossRef]
35. Kumar, P., Kumar, M., Bedi, O., Gupta, M., Kumar, S. et al. (2021). Role of vitamins and minerals as immunity boosters in COVID-19. Inflammopharmacology, 24(4), 1001–16. DOI 10.1007/s10787-021-00826-7. [Google Scholar] [CrossRef]
36. Hooper, L., Abdelhamid, A., Bunn, D., Brown, T., Summerbell, C. D. et al. (2015). Effects of total fat intake on body weight. Cochrane Database Systematic Reviews, 8, CD011834. DOI 10.1002/14651858.CD011834. [Google Scholar] [CrossRef]
37. WHO (2018). Guidelines: Saturated fatty acid and trans-fatty acid intake for adults and children. Geneva. [Google Scholar]
38. FAO (2010). Fats and fatty acids in human nutrition: Report of an expert consultation. FAO Food and Nutrition Paper, 91. Rome. [Google Scholar]
39. WHO (2012). Guideline: Sodium intake for adults and children. Geneva. [Google Scholar]
40. Jain, S. (2021). Diet and nutrition recommendations during the COVID-19 pandemic. IP Journal of Nutrition, Metabolism and Health Science, 3(4), 114–118. DOI 10.18231/ijnmhs. [Google Scholar] [CrossRef]
41. Zahedipour, F., Hosseini, S. A., Sathyapalan, T., Majeed, M., Jamialahmadi, T. et al. (2020). Potential effects of curcumin in the treatment of COVID-19 infection. Phytotherapy Research, 34(11), 2911–2920. DOI 10.1002/ptr.6738. [Google Scholar] [CrossRef]
42. Das, R., Kumar, M. (2020). Need for promoting vitamin C rich foods for combating detrimental effects of COVID-19. Biotica Research Today, 2(8), 742–744. [Google Scholar]
43. Mishra, S., Patel, M. (2020). Role of nutrition on immune system during COVID-19 pandemic. Journal of Food Nutrition and Health, 3(2), 20–33. [Google Scholar]
44. Sarkar, S., Dey, J. K. (2021). Role of food to fight against COVID-19. Biotica Research Today, 3(6), 544–546. [Google Scholar]
45. Kretchy, I. A., Boadu, J. A., Kretchy, J. P., Agyabeng, K., Passah, A. A. et al. (2021). Utilization of complementary and alternative medicine for the prevention of COVID-19 infection in Ghana: A national cross-sectional online survey. Preventive Medicine Reports, 24, 101633. DOI 10.1016/j.pmedr.2021.101633. [Google Scholar] [CrossRef]
46. Sen, D., Bhaumik, S., Debnath, P., Debnath, S. (2021). Potentiality of Moringa oleifera against SARS-CoV-2: Identified by a rational computer aided drug design method. Journal of Biomolecular Structure and Dynamics, 7, 1–18. DOI 10.1080/07391102.2021.1898475. [Google Scholar] [CrossRef]
47. Ullah, A., Ullah, K. (2021). Inhibition of SARS-CoV-2 3CL mpro by natural and synthetic inhibitors: Potential implication for vaccine production against COVID-19. Frontiers in Molecular Biosciences, 8, 211. DOI 10.3389/fmolb.2021.640819. [Google Scholar] [CrossRef]
48. Fajri, M. (2021). The potential of Moringa oleifera as immune booster against COVID 19. IOP Conference Series: Earth and Environmental Science, 807(2), 022008. DOI 10.1088/1755-1315/807/2/022008. [Google Scholar] [CrossRef]
49. Jacques, A. S., Arnaud, S. S., Jacques, D. T. (2020). Review on biological and immunomodulatory properties of Moringa oleifera in animal and human nutrition. Journal of Pharmacognosy and Phytotherapy, 12(1), 1–9. DOI 10.5897/JPP. [Google Scholar] [CrossRef]
50. Ojeda-López, J., Marczuk-Rojas, J. P., Polushkina, O. A., Purucker, D., Salinas, M. et al. (2020). Evolutionary analysis of the Moringa oleifera genome reveals a recent burst of plastid to nucleus gene duplications. Scientific Reports, 10(1), 1–15. DOI 10.1038/s41598-020-73937-w. [Google Scholar] [CrossRef]
51. Xiao, X., Wang, J., Meng, C., Liang, W., Wang, T. et al. (2020). Moringa oleifera Lam and its therapeutic effects in immune disorders. Frontiers in Pharmacology, 11, 2188. DOI 10.3389/fphar.2020.566783. [Google Scholar] [CrossRef]
52. Balakumbahan, R., Sathiyamurthy, V. A., Janavi, G. J. (2020). Moringa leaf–A super food. Biotica Research Today, 2(6), 438–440. [Google Scholar]
53. Singh, A. K., Rana, H. K., Tshabalala, T., Kumar, R., Gupta, A. et al. (2020). Phytochemical, nutraceutical and pharmacological attributes of a functional crop Moringa oleifera Lam: An overview. South African Journal of Botany, 129, 209–220. DOI 10.1016/j.sajb.2019.06.017. [Google Scholar] [CrossRef]
54. Sultana, S. (2020). Nutritional and functional properties of Moringa oleifera. Metabolism Open, 8, 100061. DOI 10.1016/j.metop.2020.100061. [Google Scholar] [CrossRef]
55. Gandji, K., Chadare, F. J., Idohou, R., Salako, V. K., Assogbadjo, A. E. et al. (2018). Status and utilisation of Moringa oleifera Lam: A review. African Crop Science Journal, 26(1), 137–156. DOI 10.4314/acsj.v26i1.10. [Google Scholar] [CrossRef]
56. Padayachee, B., Baijnath, H. (2020). An updated comprehensive review of the medicinal, phytochemical and pharmacological properties of Moringa oleifera. South African Journal of Botany, 129, 304–316. DOI 10.1016/j.sajb.2019.08.021. [Google Scholar] [CrossRef]
57. Khan, S., Amin, R., Ali, S., Zeb, T. F., Haider, M. S. et al. (2020). Comparative analysis of the nutritional components of different parts of Moringa oleifera from Karachi, Pakistan. Pakistan Journal of Science, 72(4), 271. [Google Scholar]
58. Matic, I., Guidi, A., Kenzo, M., Mattei, M., Galgani, A. (2018). Investigation of medicinal plants traditionally used as dietary supplements: A review on Moringa oleifera. Journal of Public Health in Africa, 9(3), 841. DOI 10.4081/jphia.2018.841. [Google Scholar] [CrossRef]
59. Bose, C. K. (2007). Possible role of Moringa oleifera Lam. root in epithelial ovarian cancer. Medscape General Medicine, 9(1), 26. [Google Scholar]
60. Fahey, J. (2005). Moringa oleifera: A review of the medical evidence for its nutritional, therapeutic, and prophylactic properties. Part I. Trees Life Journal, 1(5), 1–15. DOI 10.1201/9781420039078.ch12. [Google Scholar] [CrossRef]
61. Mahajan, S., Banerjee, A., Chauhan, B., Padh, H., Nivsarkar, M. et al. (2009). Inhibitory effect of n-butanol fraction of Moringa oleifera Lam. seeds on ovalbumin-induced airway inflammation in a Guinea pig model of asthma. International Journal of Toxicology, 28(6), 519–527. DOI 10.1177/1091581809345165. [Google Scholar] [CrossRef]
62. Senthilkumar, A., Karuvantevida, N., Rastrelli, L., Kurup, S. S., Cheruth, A. J. (2018). Traditional uses, pharmacological efficacy, and phytochemistry of Moringa peregrina (Forssk.) fiori: A review. Frontiers in Pharmacology, 9, 465. DOI 10.3389/fphar.2018.00465. [Google Scholar] [CrossRef]
63. Estrella, C. P., Mantaring, J. B. V., Davis, G. Z., Taup, M. A. (2000). A Double-blind randomized controlled trial on the use of malunggay (Moringa oleifera) for augmentation on the volume of breast milk among non-nursing mothers of preterm infants. The Philippine Journal of Pediatrics, 49(1), 3–6. [Google Scholar]
64. Dawn, C. P., Ambrose, D., L, A. (2015). Moringa oleifera mothers best friend: An ideal health food. Beverage and Food World, 40(2), 33–34. [Google Scholar]
65. Palada, M. C. (2019). The miracle tree: Moringa oleifera. Philippines: Xlibris Corporation. [Google Scholar]
66. Debajyoti, D., Dipsundar, S., Dinesh, B., Chandreyee, R., Sanatan, R. et al. (2017). Moringa olifera (shigruA miracle tree for its nutritional, ethnomedicinal and therapeutic importance. International Journal of Development Research, 7(11), 16823–16827. [Google Scholar]
67. Tshabalala, T., Ndhlala, A. R., Ncube, B., Abdelgadir, H. A., van Staden, J. (2020). Potential substitution of the root with the leaf in the use of Moringa oleifera for antimicrobial, antidiabetic and antioxidant properties. South African Journal of Botany, 129, 106–112. DOI 10.1016/j.sajb.2019.01.029. [Google Scholar] [CrossRef]
68. Keshri, A., Dixit, S., Sharma, R., Leitanthem, V. K., Patil, A. K. et al. (2021). Moringa and its medicinal properties: A review. The Pharma Innovation Journal, 10(8), 647–652. [Google Scholar]
69. Trigo, C., Castello, M. L., Ortola, M. D., Garcia-Mares, F. J., Desamparados Soriano, M. (2021). Moringa oleifera: An unknown crop in developed countries with great potential for industry and adapted to climate change. Foods, 10(1), 31. DOI 10.3390/foods10010031. [Google Scholar] [CrossRef]
70. Tejas, G. H., Umang, J. H., Payal, B. N., Tusharbinu, D. R., Pravin, T. R. (2012). A panoramic view on pharmacognostic, pharmacological, nutritional, therapeutic and prophylactic values of Moringa olifera Lam. International Research Journal of Pharmacy, 3(6), 1–7. [Google Scholar]
71. Olson, M. E., Sankaran, R. P., Fahey, J. W., Grusak, M. A., Odee, D. et al. (2016). Leaf protein and mineral concentrations across the “Miracle tree” genus Moringa. PLoS One, 11(7), 1–17. DOI 10.1371/journal.pone.0159782. [Google Scholar] [CrossRef]
72. Ogbe, A. O., Affiku, J. P. (2021). Proximate study, mineral and anti-nutrient composition of Moringa oleifera leaves harvested from Lafia, Nigeria: Potential benefits in poultry nutrition and health. Journal of Microbiology, Biotechnology and Food Sciences, 1(3), 296–308. [Google Scholar]
73. Sreeja, M., Jayasri, P., Keerthi, N., Yeshashwini, J., Praveen, J. (2021). Moringa oleifera: A review on nutritive importance and its potential use as nutraceutical plant. Journal of Medicinal Plants, 9(2), 15–17. DOI 10.22271/plants. [Google Scholar] [CrossRef]
74. Anwar, F., Ashraf, M., Bhanger, M. I. (2005). Inter-provenance variation in the composition of Moringa oleifera oil seeds from Pakistan. Journal of American Oil Chemistry Society, 82, 45–51. DOI 10.1007/s11746-005-1041-1. [Google Scholar] [CrossRef]
75. Granella, S. J., Bechlin, T. R., Christ, D., Coelho, S. R. M., de Oliveira Paz, C. H. (2021). An approach to recent applications of Moringa oleifera in the agricultural and biofuel industries. South African Journal of Botany, 137, 110–116. DOI 10.1016/j.sajb.2020.10.006. [Google Scholar] [CrossRef]
76. Gopalakrishnan, L., Doriya, K., Kumar, D. S. (2016). Moringa oleifera: A review on nutritive importance and its medicinal application. Food Science and Human Wellness, 5(2), 49–56. DOI 10.1016/j.fshw.2016.04.001. [Google Scholar] [CrossRef]
77. Rajbhar, Y. P., Rajbhar, G., Rawat, P. L., Shardulya, S., Kumar, M. (2018). Grow moringa (Moringa oleiferathe miracle tree on the earth. Horticulture International Journal, 2(4), 166–172. DOI 10.15406/hij.2018.02.00047. [Google Scholar] [CrossRef]
78. Ashraq, M., Basra, S. M. A., Ashfaq, U. (2012). Moringa: A miracle plant for agroforestry. Journal of Agriculture, Forestry and Social Sciences, 8(2), 115–122. [Google Scholar]
79. Sharma, A., Dhiman, G., Lal, P. S., Godiyal, R. D., Thapliyal, B. P. (2021). Characterization of Moringa oleifera (drumstick) wood for pulp and paper making. Cellulose Chemistry and Technology, 55(3–4), 255–262. DOI 10.35812/CelluloseChemTechnol.2021.55.25. [Google Scholar] [CrossRef]
80. Zhang, J., Lin, M., Chen, H., Zhu, Q., Chen, X. (2018). Floral biology and pistil receptivity of the drumstick tree (Moringa oleifera Lam.). Archives of Biological Sciences, 70(2), 299–305. DOI 10.2298/ABS170205046Z. [Google Scholar] [CrossRef]
81. Leone, A., Spada, A., Battezzati, A., Schiraldi, A., Aristil, J. et al. (2016). Moringa oleifera seeds and oil: Characteristics and uses for human health. International Journal of Molecular Sciences, 17(12), 2141. DOI 10.3390/ijms17122141. [Google Scholar] [CrossRef]
82. Anwar, F., Latif, S., Ashraf, M., Gilani, A. H. (2007). Moringa oleifera: A food plant with multiple medicinal uses. Phytotherapy Research, 21, 17–25. DOI 10.1002/ptr.2023. [Google Scholar] [CrossRef]
83. Thurber, M. D., Fahey, J. W. (2010). Adoption of Moringa oleifera to combat under-nutrition viewed through the lens of the diffusion of innovations theory. Ecology of Food and Nutrition, 48(1), 1–13. DOI 10.1080/03670240902794598. [Google Scholar] [CrossRef]
84. Vaknin, Y., Eisikowitch, D., Mishal, A. (2021). Floral and pollen traits of Moringa oleifera Lam. and Moringa peregrina (Forssk.) fiori provide reproductive adaptations for arid conditions. Agronomy, 11(6), 1090. DOI 10.3390/agronomy11061090. [Google Scholar] [CrossRef]
85. Fuglie, L. J. (2001). Introduction to the multiple uses of Moringa. In: The miracle tree: The multiple attributes of moringa. Church World Service: New York, NY, USA. [Google Scholar]
86. Verma, R., Chauhan, A., Shandilya, M., Li, X., Kumar, R. et al. (2020). Antimicrobial potential of Ag-doped ZnO nanostructure synthesized by the green method using Moringa oleifera extract. Journal of Environmental Chemical Engineering, 8(3), 103730. DOI 10.1016/j.jece.2020.103730. [Google Scholar] [CrossRef]
87. Tian, Y., Zeng, Y., Zhang, J., Yang, C., Yan, L. et al. (2015). High-quality reference genome of drumstick tree (Moringa oleifera Lam.a potential perennial crop. Science China Life Sciences, 58(7), 627–638. DOI 10.1007/s11427-015-4872-x. [Google Scholar] [CrossRef]
88. Zhang, J., Pian, R., Yang, E., Zhou, W., He, Q. et al. (2020). In vitro induction and characterization of tetraploid drumstick tree (Moringa oleifera Lam.). Open Life Sciences, 15(1), 840–847. DOI 10.1515/biol-2020-0087. [Google Scholar] [CrossRef]
89. Venkatesan, N., Sekhar, C., Murugananthi, M. (2018). Marketing and price spread analysis of Moringa in Tamil Nadu, India. Horticulture International Journal, 2(4), 212–221. DOI 10.15406/hij.2018.02.00055. [Google Scholar] [CrossRef]
90. Mahy, L., Wijnhoven, T. (2020). Is the decade of action on nutrition (2016–2025) leaving a footprint? taking stock and looking ahead. American Journal of Public Health, 44, e73. DOI 10.26633/RPSP.2020.73. [Google Scholar] [CrossRef]
91. Mbikay, M. (2012). Therapeutic potential of Moringa oleifera leaves in chronic hyperglycemia and dyslipidemia: A review. Frontiers in Pharmacology, 3, 1–12. DOI 10.3389/fphar.2012.00024. [Google Scholar] [CrossRef]
92. Abbasi-Parizad, P., de Nisi, P., Adani, F., Pepé Sciarria, T., Squillace, P. et al. (2020). Antioxidant and anti-inflammatory activities of the crude extracts of raw and fermented tomato pomace and their correlations with aglycate-polyphenols. Antioxidants, 9(2), 179. DOI 10.3390/antiox9020179. [Google Scholar] [CrossRef]
93. Milla, P. G., Peñalver, R., Nieto, G. (2021). Health benefits of uses and applications of Moringa oleifera in bakery products. Plants, 10(2), 318. DOI 10.3390/plants10020318. [Google Scholar] [CrossRef]
94. Meireles, D., Gomes, J., Lopes, L., Hinzmann, M., Machado, J. (2020). A review of properties, nutritional and pharmaceutical applications of Moringa oleifera: Integrative approach on conventional and traditional asian medicine. Advances in Traditional Medicine, 20(4), 1–21. DOI 10.1007/s13596-020-00468-0. [Google Scholar] [CrossRef]
95. Divya, K. G., Rubeena, M., Andalil, R., Erni, B., Sundaramoorthy, B. et al. (2019). Identity profile of Moringa oleifera Lam. flower. International Journal of Botany Studies, 4(4), 90–99. [Google Scholar]
96. Dasat, G. S., Danjuma, G., Chundusu, E. S. (2020). Evaluation of the nutritionally valuable mineral composition of Moringa oleifera leaf. European Journal of Nutrition & Food Safety, 12(10), 46–53. DOI 10.9734/ejnfs/2020/v12i1030300. [Google Scholar] [CrossRef]
97. Moyo, B., Masika, P., Hugo, A., Muchenje, V. (2011). Nutritional characterization of Moringa (Moringa oleifera Lam.) leaves. African Journal of Biotechnology, 10, 12925–12933. DOI 10.5897/AJB. [Google Scholar] [CrossRef]
98. Lanjwani, A. H., Ghanghro, A. B., Khuhawar, T. M. J., Lanjwani, M. F. (2021). Nutritional assessment and biological activity of Moringa oleifera. Biological Sciences, 64(1), 1–6. [Google Scholar]
99. Raja, S., Bagle, B. G., More, T. A. (2013). Drumstick (Moringa oleifera Lam.) improvement for semiarid and arid ecosystem: Analysis of environmental stability. Journal of Plant Breeding and Crop Science, 5, 164–170. DOI 10.5897/JPBCS. [Google Scholar] [CrossRef]
100. Kasolo, J. N., Bimenya, G. S., Ojok, L., Ochieng, J., Ogwal-okeng, J. W. (2010). Phytochemicals and uses of Moringa oleifera leaves in Ugandan rural communities. Journal of Medicinal Plants Research, 4, 753–757. [Google Scholar]
101. Oduro, I., Ellis, W. O., Owusu, D. (2008). Nutritional potential of two leafy vegetables: Moringa oleifera and Ipomoea batatas leaves. Scientific Research Essay, 3, 57–60. [Google Scholar]
102. Rockwood, J. L., Anderson, B. G., Casamatta, D. A. (2013). Potential uses of Moringa oleifera and an examination of antibiotic efficacy conferred by M. oleifera seed and leaf extracts using crude extraction techniques available to underserved indigenous populations. International Journal of Phototherapy Research, 3(2), 61–71. [Google Scholar]
103. Saini, R. K., Sivanesan, I., Keum, Y. S. (2016). Phytochemicals of Moringa oleifera: A review of their nutritional, therapeutic and industrial significance. 3 Biotech, 6(203), 1–14. DOI 10.1007/s13205-016-0526-3. [Google Scholar] [CrossRef]
104. Alegbeleye, O. O. (2018). How functional is Moringa oleifera? A review of its nutritive, medicinal, and socioeconomic potential. Food and Nutrition Bulletin, 39(1), 149–170. DOI 10.1177/0379572117749814. [Google Scholar] [CrossRef]
105. Madane, P., Das, A. K., Pateiro, M., Nanda, P. K., Bandyopadhyay, S. et al. (2019). Drumstick (Moringa oleifera) flower as an antioxidant dietary fibre in chicken meat nuggets. Foods, 8(8), 307. DOI 10.3390/foods8080307. [Google Scholar] [CrossRef]
106. Eman, N. A., Muhamad, K. N. S. (2016). Comparison of Moringa oleifera seeds oil characterization produced chemically and mechanically. IOP Conference Series: Earth and Environmental Science, 36(1012063. IOP Publishing, Malaysia. [Google Scholar]
107. Duarte, A. M., Aquino, J. S., Queiroz, N., Dantas, D. L., Maciel, G. S. et al. (2018). A comparative study of the thermal and oxidative stability of Moringa oil with olive and canola oils. Journal of Thermal Analysis and Calorimetry, 134(3), 1943–1952. DOI 10.1007/s10973-018-7651-7. [Google Scholar] [CrossRef]
108. Combs, G. F., McClung, J. P. (2016). The vitamins: Fundamental aspects in nutrition and health. London, UK; San Diego, CA, USA: Academic Press. [Google Scholar]
109. Ahanger, M. A., Tomar, N. S., Tittal, M., Argal, S., Agarwal, R. M. (2017). Plant growth under water/salt stress: ROS production; antioxidants and significance of added potassium under such conditions. Physiology and Molecular Biology of Plants, 23(4), 731–744. DOI 10.1007/s12298-017-0462-7. [Google Scholar] [CrossRef]
110. Carr, A. C., Rowe, S. (2020). Factors affecting vitamin C status and prevalence of deficiency: A global health perspective. Nutrients, 12(7), 1963. DOI 10.3390/nu12071963. [Google Scholar] [CrossRef]
111. Freiberger, C. E., Vanderjagt, D. J., Pastuszyn, A., Glew, R. S., Mounkaila, G. et al. (1998). Nutrient content of the edible leaves of seven wild plants from Niger. Plant Foods for Human Nutrition, 53, 57–69. DOI 10.1023/A:1008080508028. [Google Scholar] [CrossRef]
112. Abrams, B., Duncan, D., Hertz Piccioto, I. (1993). A prospective study of dietary intake and acquired immune deficiency syndrome in HIV sero-positive homosexsual men. Journal of Acquired Immune Deficiency Syndrome, 8, 949–58. [Google Scholar]
113. Bhattacharjee, M. (2020). Moringa oleifera: A potent immune booster in the catastrophe of COVID-19. Ecology, Environment and Conservation, 26, 202–209. [Google Scholar]
114. Gulcin, İ (2020). Antioxidants and antioxidant methods: An updated overview. Archives of Toxicology, 94(3), 651–715. DOI 10.1007/s00204-020-02689-3. [Google Scholar] [CrossRef]
115. Ferreira, P. M. P., Farias, D. F., Oliveira, J. T. D. A., Carvalho, A. D. F. U. (2008). Moringa oleifera: Bioactive compounds and nutritional potential. The Revista de Nutricao, 21, 431–437. DOI 10.1590/S1415-52732008000400007. [Google Scholar] [CrossRef]
116. Olaoye, A. B., Ologunde, C. A., Molehin, O. R., Nwankwo, I. (2021). Comparative antioxidant analysis of Moringa oleifera leaf extracts from south western states in Nigeria. Future Journal of Pharmaceutical Sciences, 7, 68. DOI 10.1186/s43094-021-00204-8. [Google Scholar] [CrossRef]
117. Siddhuraju, P., Becker, K. (2003). Antioxidant properties of various solvent extracts of total phenolic constituents from three different agro-climatic origins of drumstick tree (Moringa oleifera Lam.) leaves. Journal of Agricultural Food Chemistry, 51, 2144–55. DOI 10.1021/jf020444+. [Google Scholar] [CrossRef]
118. Dash, S., Panda, M. K., Singh, M. C., Jit, B. P., Singh, Y. D. et al. (2020). Bioactive molecules from alpinia genus: A comprehensive review. Current Pharmaceutical Biotechnology, 21, 1. DOI 2174/1389201021666200510002409. [Google Scholar]
119. Rani, A., Husain, N. Z., Kumolosasi, K., E. (2018). Moringa genus: A review of phytochemistry and pharmacology. Frontiers in Pharmacology, 9, 108. DOI 10.3389/fphar.2018.00108. [Google Scholar] [CrossRef]
120. Vergara-Jimenez, M., Almatrafi, M. M., Fernandez, M. L. (2017). Bioactive components in Moringa oleifera leaves to protect against chronic disease. Antioxidants, 6(4), 91. DOI 10.3390/antiox6040091. [Google Scholar] [CrossRef]
121. Berkovich, L., Earon, G., Ron, I., Rimmon, A., Vexler, A. et al. (2013). Moringa oleifera aqueous leaf extract down-regulates nuclear factor-kappa B and increases cytotoxic effect of chemotherapy in pancreatic cancer cells. BMC Complementary and Alternative Medicines, 13, 212–219. DOI 10.1186/1472-6882-13-212. [Google Scholar] [CrossRef]
122. Leone, A., Spada, A., Battezzati, A., Schiraldi, A., Aristil, J. et al. (2015). Cultivation, genetic, ethnopharmacology, phytochemistry and pharmacology of Moringa oleifera leaves: An overview. International Journal of Molecular Science, 16, 12791–12835. DOI 10.3390/ijms160612791. [Google Scholar] [CrossRef]
123. Caceres, A., Saravia, A., Rizzo, S., Zabala, L., de Leon, E. et al. (1991). Pharmacologic properties of Moringa oleifera. 2: Screening for antispasmodic, anti-inflammatory and diuretic activity. Journal of Ethnopharmacology, 36(3), 233–237. DOI 10.1016/0378-8741(92)90049-W. [Google Scholar] [CrossRef]
124. Giuberti, G., Rocchetti, G., Montesano, D., Lucini, L. (2021). The potential of Moringa oleifera in food formulation: A promising source of functional compounds with health-promoting properties. Current Opinion in Food Science, 42, 257–269. DOI 10.1016/j.cofs.2021.09.001. [Google Scholar] [CrossRef]
125. Islam, Z., Islam, S. M., Hossen, F., Mahtab-ul-Islam, K., Hasan, M. et al. (2021). Moringa oleifera is a prominent source of nutrients with potential health benefits. International Journal of Food Science, 2021, 1–11. DOI 10.1155/2021/6627265. [Google Scholar] [CrossRef]
126. Falowo, A. B., Mukumbo, F. E., Idamokoro, E. M., Lorenzo, J. M., Afolayan, A. J. et al. (2018). Multi-functional application of Moringa oleifera Lam. in nutrition and animal food products: A review. Food Research International, 106, 317–334. DOI 10.1016/j.foodres.2017.12.079. [Google Scholar] [CrossRef]
127. Dhakad, A. K., Ikram, M., Sharma, S., Khan, S., Pandey, V. V. et al. (2019). Biological, nutritional, and therapeutic significance of Moringa oleifera Lam. Phytotherapy Research, 33(11), 2870–2903. DOI 10.1002/ptr.6475. [Google Scholar] [CrossRef]
128. Brilhante, R. S. N., Sales, J. A., Pereira, V. S., Castelo, D. D. S. C. M., de Aguiar Cordeiro, R. et al. (2017). Research advances on the multiple uses of Moringa oleifera: A sustainable alternative for socially neglected population. Asian Pacific Journal of Tropical Medicine, 10(7), 621–630. DOI 10.1016/j.apjtm.2017.07.002. [Google Scholar] [CrossRef]
129. Faizi, S., Siddiqui, B., Saleem, R., Aftab, K., Shaheen, F. et al. (1998). Hypotensive constituents from the pods of Moringa oleifera. Planta Medica, 64, 225–228. DOI 10.1055/s-2006-957414. [Google Scholar] [CrossRef]
130. Jung, I. L. (2014). Soluble extract from Moringa oleifera leaves with a new anti-cancer activity. PLoS One, 9, 1–10. DOI 10.1371/journal.pone.0095492. [Google Scholar] [CrossRef]
131. Fischer, A., Sellner, M., Neranjan, S., Smieško, M., Lill, M. A. (2020). Potential inhibitors for novel coronavirus protease identified by virtual screening of 606 million compounds. International Journal of Molecular Sciences, 21(10), 3626. DOI 10.3390/ijms21103626. [Google Scholar] [CrossRef]
132. Fuglie, L. J. (2005). The Moringa tree: A local solution to malnutrition church world service in senegal. Dakar, Senegal. http://www.moringanews.org/documents/Nutrition. [Google Scholar]
133. Nair, S., Varalakshmi, K. N. (2011). Anticancer, cytotoxic potential of Moringa oleifera extracts on HeLa cell line. Journal of Natural Pharmacology, 2, 138–142. DOI 10.4103/2229-5119.86260. [Google Scholar] [CrossRef]
134. Ijarotimi, O. S., Adeoti, O., Ariyo, O. (2013). Comparative study on nutrient composition, phytochemical, and functional characteristics of raw, germinated, and fermented Moringa oleifera seed flour. Food Science and Nutrition, 1, 452–463. DOI 10.1002/fsn3.70. [Google Scholar] [CrossRef]
135. Xiong, Y., Rajoka, M. S. R., Zhang, M. X., He, Z. (2020). Isolation and identification of two new compounds from the seeds of Moringa oleifera and their antiviral and anti-inflammatory activities. Natural Product Research, 1–10. DOI 10.1080/14786419.2020.1851218. [Google Scholar] [CrossRef]
136. Sutalangka, C., Wattanathorn, J., Muchimapura, S., Thukham-mee, W. (2013). Moringa oleifera mitigates memory impairment and neurodegeneration in animal model of age-related dementia. Oxidative Medicine and Cellular Longevity, 2013, 1–9. DOI 10.1155/2013/695936. [Google Scholar] [CrossRef]
137. Adeyemi, O. S., Elebiyo, T. C. (2014). Moringa oleifera supplemented diets prevented nickel-induced nephrotoxicity in wistar rats. Journal of Nutrition and Metabolism, 2014, 1–8. DOI 10.1155/2014/958621. [Google Scholar] [CrossRef]
138. Monera, T. G., Maponga, C. C. (2012). Prevalence and patterns of Moringa oleifera use among HIV positive patients in Zimbabwe: A cross-sectional survey. Journal of Public Health in Africa, 3(1), 22–24. DOI 10.4081/jphia.2012.e6. [Google Scholar] [CrossRef]
139. Patel, S., Klagholz, S., Peterson, C. T., Weiss, L., Chopra, D. et al. (2019). Psychosocial effects of a holistic ayurvedic approach to well-being in health and wellness courses. Global Advances in Health and Medicine, 8. DOI 10.1177/2164956119843814. [Google Scholar] [CrossRef]
140. Rajput, D. S. (2020). Evolution, ayurveda, immunity, and preventive aspects for emerging infectious diseases such as COVID-19. International Journal of Research in Pharmaceutical Sciences, 11(2), 86–93. DOI 10.26452/ijrps.v11iSPL1.2227. [Google Scholar] [CrossRef]
141. Soumya, M. C., Sinimol, T. P., Sumedhan, V. (2019). Pharmacological aspects of Sigru (Moringa oleifera Lam.) in view of Ayurveda. Journal of Ayurveda and Integrated Medical Sciences, 4(1), 39–42. DOI 10.21760/jaims.4.1.7. [Google Scholar] [CrossRef]
142. Pal, S. K., Mukherjee, P. K., Saha, B. P. (1995). Studies on the antiulcer activity of Moringa oleifera leaf extract on gastric ulcer models in rats. Phytotherapy Research, 9, 463–465. DOI 10.1002/ptr.2650090618. [Google Scholar] [CrossRef]
143. Aja, P. M., Agu, P. C., Ezeh, E. M., Awoke, J. N., Ogwoni, H. A. et al. (2021). Prospect into therapeutic potentials of Moringa oleifera phytocompounds against cancer upsurge: De novo synthesis of test compounds, molecular docking, and ADMET studies. Bulletin of the National Research Centre, 45(1), 1–18. DOI 10.1186/s42269-021-00554-6. [Google Scholar] [CrossRef]
144. Sidker, K., Sinha, M., Das, N., Das, D. K., Datta, S. et al. (2013). Moringa oleifera leaf extract prevents in vitro oxidative DNA damage. Asian Journal of Pharmaceutical and Clinical Research, 6, 159–163. [Google Scholar]
145. Kumari, D. J. (2010). Hypoglycaemic effect of Moringa oleifera and Azadirachta indica in type 2 diabetes mellitus. Bioscan, 5(20), 211–214. [Google Scholar]
146. Oyedepo, T. A., Babarinde, S. O., Ajayeoba, T. A. (2013). Evaluation of the antihyperlipidemic effect of aqueous leaves extract of Moringa oleifera in alloxan induced diabetic rats. International Journal of Biochemistry Research & Review, 3, 162–170. DOI 10.9734/IJBCRR/2013/3639. [Google Scholar] [CrossRef]
147. Kamalakkannan, N., Prince, P. S. M. (2006). Antihyperglycaemic and antioxidant effect of rutin, a polyphenolic flavonoid, in streptozotocin-induced diabetic wistar rats. Basic and Clinical Pharmacology and Toxicology, 98, 97–103. DOI 10.1111/j.1742-7843.2006.pto_241.x. [Google Scholar] [CrossRef]
148. Tavsan, Z., Kayali, H. A. (2019). Flavonoids showed anticancer effects on the ovarian cancer cells: Involvement of reactive oxygen species, apoptosis, cell cycle and invasion. Biomedicine and Pharmacotherapy, 116, 109004. DOI 10.1016/j.biopha.2019.109004. [Google Scholar] [CrossRef]
149. Kirisattayakul, W., Wattanathorn, J., Tong-Un, T., Muchimapura, S., Wannanon, P. et al. (2013). Cerebroprotective effect of Moringa oleifera against focal ischemic stroke induced by middle cerebral artery occlusion. Oxidative Medicine and Cellular Longevity, 2013, 10–13. DOI 10.1155/2013/951415. [Google Scholar] [CrossRef]
150. Choudhary, M. K., Bodakhe, S. H., Gupta, S. K. (2013). Assessment of the antiulcer potential of Moringa oleifera root-bark extract in rats. JAMS Journal of Acupuncture Meridian Studies, 6, 214–220. DOI 10.1016/j.jams.2013.07.003. [Google Scholar] [CrossRef]
151. Abdu, H., Ashiru Garba, A. (2021). Proximate analysis and anti-ulcer activity of methanolic extract of Moringa oleifera. African Scholar Journal of Agriculture and Agricultural Technology, 20(1),188–201. [Google Scholar]
152. Adisakwattana, S., Chanathong, B. (2011). Alpha-glucosidase inhibitory activity and lipid-lowering mechanisms of Moringa oleifera leaf extract. European Review for Medical and Pharmacological Sciences, 15(7), 803–808. [Google Scholar]
153. Dubey, D. K., Dora, J., Kumar, A., Gulsan, R. K. (2013). A multipurpose tree-Moringa oleifera. International Journal of Pharmaceutical and Chemical Sciences, 2(1), 415–423. [Google Scholar]
154. Mabrouki, L., Rjeibi, I., Taleb, J., Zourgui, L. (2020). Cardiac ameliorative effect of Moringa oleifera leaf extract in high-fat diet-induced obesity in rat model. BioMed Research International, 2020, 1–10. DOI 10.1155/2020/6583603. [Google Scholar] [CrossRef]
155. Rao, K. S., Mishra, S. H. (1998). Anti-inflammatory and anti-hepatoxic activities of the roots of Moringa pterygosperma gaertn. Indian Journal of Pharmaceutical Sciences, 60(1), 12–16. [Google Scholar]
156. Arulselvan, P., Tan, W. S., Gothai, S., Muniandy, K., Fakurazi, S. et al. (2016). Anti-inflammatory potential of ethyl acetate fraction of Moringa oleifera in downregulating the NF-κB signaling pathway in lipopolysaccharide-stimulated macrophage. Molecules, 21, 1452–1465. DOI 10.3390/molecules21111452. [Google Scholar] [CrossRef]
157. Alkhudhayri, D. A., Osman, M. A., Alshammari, G. M., Al Maiman, S. A., Yahya, M. A. (2021). Moringa peregrina leaf extracts produce anti-obesity, hypoglycemic, anti-hyperlipidemic, and hepatoprotective effects on high-fat diet fed rats. Saudi Journal of Biological Sciences, 28(6), 3333–3342. DOI 10.1016/j.sjbs.2021.02.078. [Google Scholar] [CrossRef]
158. Biswas, D., Nandy, S., Mukherjee, A., Pandey, D. K., Dey, A. (2020). Moringa oleifera Lam. and derived phytochemicals as promising antiviral agents: A review. South African Journal of Botany, 129, 272–282. DOI 10.1016/j.sajb.2019.07.049. [Google Scholar] [CrossRef]
159. Lin, M., Zhang, J., Chen, X. (2018). Bioactive flavonoids in Moringa oleifera and their health-promoting properties. Journal of Functional Foods, 47, 469–479. DOI 10.1016/j.jff.2018.06.011R. [Google Scholar] [CrossRef]
160. Murthy, H. N., Bapat, V. A. (2019). Bioactive compounds in underutilized fruits and nuts, pp. 1–15. New York, NY: Springer International Publishing. [Google Scholar]
161. Jahn, S. A., Musnad, H. A., Burgstaller, H. (1986). The tree that purifies water: Cultivating multipurpose Moringaceae in the Sudan. Unasylva FAO, 38(152), 23–28. [Google Scholar]
162. Tahiliani, P., Kar, A. (2000). Role of Moringa oleifera leaf extract in the regulation of thyroid hormone status in adult male and female rats. Pharmacology Research, 41, 319–323. DOI 10.1006/phrs.1999.0587. [Google Scholar] [CrossRef]
163. Alam, M. A., Quamri, M. A., Haider, N. (2021). Efficacy and safety of barg-e-sahajna (Moringa olifera Lam.) in primary hypothyroidism. Drug Metabolism and Personalized Therapy (Preprint). DOI 10.1515/dmdi-2021-0136. [Google Scholar] [CrossRef]
164. Ibrahim, M. A., Mohammed, A., Isah, M. B., Aliyu, A. B. (2014). Anti-trypanosomal activity of African medicinal plants: A review update. Journal of Ethnopharmacology, 154, 26–54. DOI 10.1016/j.jep.2014.04.012. [Google Scholar] [CrossRef]
165. Singh, M. K., Paul, J., De, T., Chakraborti, T. (2015). Bioactivity guided fractionation of Moringa oleifera Lam. flower targeting Leishmania donovani. Indian Journal of Experimental Biology, 53, 747–752. [Google Scholar]
166. Nilani, P., Pinaka, M. K., Duraisamy, B., Dhamodaran, P., Jeyaprakash, M. R. (2012). Anthelmintic activity of Moringa oleifera seed oil: Validation of traditional use. Journal of Advanced Science and Research, 3(2), 65–66. [Google Scholar]
167. Chumark, P., Khunawat, P., Sanvarinda, Y., Phornchirasilp, S., Morales, N. P. et al. (2008). The in vitro and ex vivo antioxidant properties, hypolipidaemic and antiatherosclerotic activities of water extract of Moringa oleifera Lam. leaves. Journal of Ethnopharmacology, 116, 439–446. DOI 10.1016/j.jep.2007.12.010. [Google Scholar] [CrossRef]
168. Bhattachrya, A., Agrawal, D., Sahu, P. K., Kumar, S., Mishra, S. S. et al. (2014). Analgesic effect of ethanolic leaf extract of Moringa oleifera on albino mice. Indian Journal of Pain, 28(2), 89–94. DOI 10.4103/0970-5333.132846. [Google Scholar] [CrossRef]
169. Laksmiani, N. L., Larasanty, L. P., Santika, A. J., Prayoga, P. A., Dewi, A. K. et al. (2020). Active compounds activity from the medicinal plants against SARSCoV-2 using in silico assay. Biomedical and Pharmacology Journal, 13(2), 873–881. DOI 10.13005/bpj/1953. [Google Scholar] [CrossRef]
170. Bennett, R. N., Mellon, F. A., Foidl, N., Pratt, J. H., Dupont, M. S. et al. (2003). Profiling glucosinolates and phenolics in vegetative and reproductive tissues of the multi-purpose trees Moringa oleifera L. (Horseradish tree) and Moringa stenopetala L. Journal of Agricultural Food Chemistry, 51, 3546–3553. DOI 10.1021/jf0211480. [Google Scholar] [CrossRef]
171. Nath, D., Sethi, N., Singh, R. K., Jain, A. K. (1992). Commonly used Indian abortifacient plants with special reference to their teratologic effects in rats. Journal of Ethnopharmacology, 36(2), 147–154. DOI 10.1016/0378-8741(92)90015-J. [Google Scholar] [CrossRef]
172. Agrawal, S. S., Vishal, D., Sumeet, G., Shekhar, C., Ashish, N. et al. (2018). Antifertility activity of ethanol leaf extract of Moringa oleifera Lam in female wistar rats. Indian Journal of Pharmaceutical Sciences, 80(3), 565–570. DOI 10.4172/pharmaceutical-sciences. [Google Scholar] [CrossRef]
173. Gromova, O. A., Torshin, I. Y. (2020). The importance of zinc in maintaining the activity of antiviral innate immunity proteins: Analysis of publications on COVID-19. The Russian Journal of Preventive Medicine, 23(3), 125–133. DOI 10.17116/profmed202023031131. [Google Scholar] [CrossRef]
174. Moustafa, A. A., Mansour, S. R. (2020). Moringa peregrina a natural medicine for increasing immunity defense against the COVID-19. Medicinal and Aromatics Plants, 9, 358. DOI 10.35248/2167-0412.20.9.358. [Google Scholar] [CrossRef]
175. Nair, A., James, T. J. (2020). Computational screening of phytocompounds from Moringa oleifera leaf as potential inhibitors of SARS-CoV-2 Mpro. Research Square (Preprint). DOI 10.21203/rs.3.rs-71018/v1. [Google Scholar] [CrossRef]
176. Shaji, D. (2020). Computational identification of drug lead compounds for COVID-19 from Moringa oleifera. ChemRxiv (Preprint). DOI 10.26434/chemrxiv.12535913.v1. [Google Scholar] [CrossRef]
177. Arifan, F., Broto, R. W., Sapatra, E. F., Pujiastuti, A. (2021). Utilization of Moringa oleifera leaves for making hand sanitizers to prevent the spread of COVID-19 virus. IOP Conference Series: Earth and Environmental Science, 623(1), 012015. IOP Publishing, Indonesia. [Google Scholar]
178. Ninivaggi, F. (2010). Ayurveda: A comprehensive guide to traditional indian medicine for the west. Lanham: Rowman and Littlefeld Publishers. [Google Scholar]
179. Fitriani, I. N., Utami, W., Zikri, A. T., Santoso, P. (2020). In silico approach of potential phytochemical inhibitor from Moringa oleifera, Cocos nucifera, Allium cepa, Psidium guajava, and Eucalyptus globulus for the treatment of COVID-19 by molecular docking. Research Square (Preprint). DOI 10.21203/rs.3.rs-42747/v1. [Google Scholar] [CrossRef]
180. Kaur, M., Choudhary, A., Saraf, I., Singh, I. P., Kaur, S. (2021). Efficacy of Moringa oleifera (Lam.) extract against Spodoptera litura (Fabricius(Lepidoptera: Noctuidae). International Journal of Tropical Insect Science, 42(1), 103–108. DOI 10.1007/s42690-021-00522-7. [Google Scholar] [CrossRef]
181. Stohs, S. J., Hartman, M. J. (2015). Review of the safety and efficacy of Moringa oleifera. Phytotherapy Research, 29, 796–804. DOI 10.1002/ptr.5325. [Google Scholar] [CrossRef]
182. Kneller, D. W., Phillips, G., O’Neill, H. M., Jedrzejczak, R., Stols, L. et al. (2020). Structural plasticity of SARS-CoV-2 3CL mpro active site cavity revealed by room temperature X-ray crystallography. Nature Communications, 11, 3202. DOI 10.1038/s41467-020-16954-7. [Google Scholar] [CrossRef]
183. Muhammad, S., Hassan, S. H., Al-Sehemi, A. G., Shakir, H. A., Khan, M. et al. (2021). Exploring the new potential antiviral constituents of Moringa oliefera for SARS-COV-2 pathogenesis: An in silico molecular docking and dynamic studies. Chemical Physics Letters, 767, 138379. DOI 10.1016/j.cplett.2021.138379. [Google Scholar] [CrossRef]
184. Asiedu-Gyekye, I. J., Frimpong-Manso, S., Awortwe, C., Antwi, D. A., Nyarko, A. K. (2014). Micro and macro elemental composition and safety evaluation of the nutraceutical Moringa oleifera leaves. Journal of Toxicology, 2014, 1–13. DOI 10.1155/2014/786979. [Google Scholar] [CrossRef]
185. Chiu, B. C., Olson, M. E., Fahey, J. W. (2021). Exploring the use of Moringa oleifera as a vegetable in Agua Caliente Nueva, Jalisco, Mexico: A qualitative study. Food Frontiers, 2(3), 294–304. DOI 10.1002/fft2.103. [Google Scholar] [CrossRef]
186. Abdel-Daim, M. M., Khalil, S. R., Awad, A., Abu Zeid, E. H., El-Aziz, R. A. et al. (2020). Ethanolic extract of Moringa oleifera leaves influences NF-κB signaling pathway to restore kidney tissue from cobalt-mediated oxidative injury and inflammation in rats. Nutrients, 12(4), 1031. DOI 10.3390/nu12041031. [Google Scholar] [CrossRef]
187. Sodvadiya, M., Patel, H., Mishra, A., Nair, S. (2020). Emerging insights into anticancer chemopreventive activities of nutraceutical Moringa oleifera: Molecular mechanisms, signal transduction and in vivo efficacy. Current Pharmacology Reports, 6(2), 38–51. DOI 10.1007/s40495-020-00210-z. [Google Scholar] [CrossRef]
188. Bailey-Shaw, Y., Rodney, S., Golding, C., Foster, S., Randle, M. et al. (2021). The potential applications of Moringa oleifera leaves in functional herbal beverages: A review of its nutritional, medicinal, and toxicity profiles. Current Nutrition and Food Science, 17(3), 272–287. DOI 10.2174/1573401316999200802182952. [Google Scholar] [CrossRef]
189. Adedapo, A., Mogbojuri, O., Emikpe, B. (2009). Safety evaluations of the aqueous extract of the leaves of Moringa oleifera in rats. Journal of Medicinal Plants Research, 3, 586–591. [Google Scholar]
190. Asare, G. A., Gyan, B., Bugyei, K., Adjei, S., Mahama, R. et al. (2012). Toxicity potentials of the nutraceutical Moringa oleifera at supra-supplementation levels. Journal of Ethnopharmacology, 139(1), 265–272. DOI 10.1016/j.jep.2011.11.009. [Google Scholar] [CrossRef]
191. Pham, D. C., Shibu, M. A., Mahalakshmi, B., Velmurugan, B. K. (2020). Effects of phytochemicals on cellular signaling: Reviewing their recent usage approaches. Critical Reviews in Food Science and Nutrition, 60(20), 3522–3546. DOI 10.1080/10408398.2019.1699014. [Google Scholar] [CrossRef]
192. Ma, Z. F., Ahmad, J., Zhang, H., Khan, I., Muhammad, S. (2020). Evaluation of phytochemical and medicinal properties of moringa (Moringa oleifera) as a potential functional food. South African Journal of Botany, 129, 40–46. DOI 10.1016/j.sajb.2018.12.002. [Google Scholar] [CrossRef]
 | This work is licensed under a Creative Commons Attribution 4.0 International License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |