 Open Access
Open Access
ARTICLE
Comparative SPH Simulation of Shock-Induced Exothermic Reactions in Al-Based Energetic Mixtures Including Gas-Phase Effects
Tomsk Scientific Center of the Siberian Branch of the Russian Academy of Sciences, Tomsk, Russia
* Corresponding Author: Oksana Ivanova. Email:
(This article belongs to the Special Issue: Perspective Materials for Science and Industrial: Modeling and Simulation)
Computers, Materials & Continua 2026, 87(2), 17 https://doi.org/10.32604/cmc.2026.075451
Received 01 November 2025; Accepted 12 February 2026; Issue published 12 March 2026
Abstract
This study presents an investigation into shock-induced exothermic reactions within three distinct aluminum-based energetic mixtures: aluminum/sulfur (Al/S), aluminum/copper oxide (Al/CuO), and aluminum/polytetrafluoroethylene (Al/PTFE). A challenge in current modeling efforts is accurately capturing the complex physical and chemical coupling under extreme loading, especially the influence of rapidly forming gaseous products in Al/PTFE mixtures on material integrity. To address this, a wide-range numerical model based on the Smoothed Particle Hydrodynamics (SPH) method was developed. This mesh-free approach manages large deformations and incorporates elastic-plastic flow, heat transfer, component diffusion, and chemical kinetics simulated using both zero- and first-order reaction schemes, favoring the latter for surface-reaction mechanisms. The proposed model takes into account gaseous reaction products, specifically aluminum fluoride (AlF3) to assess their impact on ampoule fracture dynamics. Numerical simulations, validated against experimental data, demonstrated that reaction rate, local pressure, and temperature are the primary controlling factors governing energy release and structural response. Comparative analysis revealed that although Al/CuO initiates reaction more readily (lower critical pressure/temperature), the Al/S mixture exhibits superior overall reaction efficiency under shock-wave loading, highlighting the significance of post-initiation kinetic factors. Furthermore, simulations using the conical ampoule geometry confirmed its effectiveness in generating a continuous pressure gradient, enabling systematic characterization of pressure-dependent reaction kinetics. This validated SPH model provides a powerful and predictive tool for understanding the complex behavior of energetic materials under shock-wave loading and aids in optimizing material composition for desired performance characteristics.Keywords
The control of chemical reactions induced by shock waves presents significant opportunities for the development of novel materials exhibiting unique properties. These materials are highly demanded in diverse fields, including the defense industry, aerospace engineering, and energy production. Since the late 1990s, research efforts have increasingly focused on high-density energetic materials (HDEMs), specifically reactive materials (RMs) and high-density reactive materials (HDRMs) [1–3]. RMs are particularly attractive due to their high stored energy and the potential for controlled energy release [4–7]. Thermite mixtures, intermetallic compounds, and metastable intermolecular composites (MICs) have received considerable attention due to their high energy content and capacity for enhanced explosive effects [8–12]. Recent advances focus on optimizing these high-energy mixtures [13–17].
Understanding and optimizing the performance of aluminum and polytetrafluoroethylene (Al/PTFE) mixtures is a particularly active area of research [18–22]. However, a comprehensive numerical model that accurately accounts for the influence of gaseous reaction products on the reaction dynamics and structural integrity remains a significant challenge.
Despite significant advances in the field, existing numerical models often oversimplify the complex physical and chemical processes that occur during shock-wave loading of these materials. This simplification is most evident in the modeling of Al/PTFE mixtures, where the formation and subsequent expansion of gaseous products can dramatically influence the overall process dynamics and potentially lead to fracture of ampoules. Ren et al. [23] conducted a detailed investigation of the mechanical behavior of Al/PTFE composites under varying strain rates, their analysis did not incorporate the complex kinetic processes that govern the chemical reaction itself—an omission when attempting to accurately model material behavior under shock-wave loading. Other studies have explored the use of Al/PTFE as an additive to enhance the performance of explosives [24], but a fundamental understanding of the exothermic reaction kinetics within Al/PTFE, Al/CuO, and Al/S mixtures remains incomplete. This work focuses on providing new insights into these fundamental processes.
Current detonation models, such as those based on the JWL equation-of-state, offer effective descriptions of macroscopic detonation behavior, they often fail to capture the underlying microscopic reaction dynamics. In particular, these models typically do not fully account for the effects of diffusion and the dependence of reaction rates on the gas phase environment. Guo et al. [25] developed a phenomenological shock reaction model for Al/PTFE composites and implemented it within the LS-DYNA software. While this model successfully captures the coupling between mechanical, thermal, and chemical phenomena, its reliance on a phenomenological approach may limit its ability to accurately represent the underlying microscopic reaction dynamics, especially the explicit role of gaseous product expansion.
To address these limitations, the current study employs a meshless method known as Smoothed Particle Hydrodynamics (SPH). SPH is well-suited for simulating chemical kinetics in complex, high-temperature, and high-pressure environments, especially when the gas-phase dynamics play a significant role, as it handles the large deformations and material mixing without mesh constraints. SPH inherently accounts for the multicomponent nature of the reacting medium, making it a powerful computational tool for simulating a wide range of problems involving shock-wave loading [26–28].
Accurate determination of the kinetic parameters and reaction mechanisms that govern chemical reactions under shock-wave and explosive loading is of fundamental importance to the field of condensed matter chemistry. Traditionally, such reactions have been modeled using simplified phenomenological models based on zero-order kinetics due to their ease of implementation. However, these models often lack the necessary precision to accurately represent real processes, particularly in the case of complex reactions such as those occurring in Al/PTFE mixtures.
The primary objective of this study is to develop a comprehensive numerical model, utilizing SPH, to simulate the exothermic reactions in powder mixtures of aluminum and various oxidizers, specifically Al/CuO, Al/S, and Al/PTFE, under shock-wave loading conditions. A key innovation of this research is the inclusion of the gaseous phase of reaction products, with particular emphasis on aluminum fluoride (AlF3), to assess its influence on the dynamics of ampoule fracture. Furthermore, the developed model incorporates a kinetic scheme that considers both zero- and first-order reactions, enabling a more accurate representation of reaction kinetics and energy release. This model serves two primary purposes: to provide a deeper understanding of the complex processes occurring within energetic materials subjected to extreme loading conditions and to facilitate the optimization of material composition and parameters for achieving desired performance characteristics.
2.1 Numerical Model: Shock-Wave Initiation in Powder Mixtures
To study the details of reaction processes under high-pressure and high-temperature conditions, a wide-range numerical model was developed to simulate elastic-plastic deformation of mixed materials coupled with chemical reactions. The model is based on a weak variational formulation of elastic-plastic flow [29]:
where
The components of the strain tensor in (1) were expressed through the components of the velocity vector:
Consequently, the components of the velocity vector gradient incorporated in (2) were determined using the Liu approximation, a method that exhibits first-order accuracy at all nodes:
where
The stresses in the medium are described by the following relations:
where
Y0 is the yield strength.
The motion of the elastic-plastic medium was calculated using a first-order accurate conservative scheme, which has the following form:
To simulate chemical reactions in the mixture, a smoothed particle hydrodynamics (SPH) scheme was developed, accounting for the significant heat release from exothermic reactions. SPH is a Lagrangian, mesh-free method that is particularly well-suited for simulating problems involving large deformations, material mixing, and complex geometries [26–28]. In this study, the SPH method was implemented using a cubic spline kernel function. This kernel provides a good balance between accuracy and computations. The smoothing length, h, which defines the size of the kernel’s support domain, was chosen to ensure that each particle had approximately 30 neighboring particles within its support domain. The smoothing length was dynamically adjusted during the simulation to maintain a constant number of neighbors. The derivatives are computed using the standard SPH approach, which is corrected using the correction matrix described above.
Two possible mechanisms for the chemical reactions are considered: zero-order and first-order reaction kinetics.
A zero-order reaction has the following form:
where
The following model was proposed for a first-order reaction:
Since the reaction occurs at the contact surfaces of the particles and the reaction rate is determined by the rate of diffusion of reactants to the reaction surface, it is assumed that temperature does not affect the reaction rate. In this case, the dependence of the chemical reaction rate on the pressure increase can be described by a simple linear equation:
where K0 (1 − η) is the rate of the first-order reaction, which is proportional to the concentration of reactants; K1 (P − Pcrit) is the reaction acceleration factor due to the pressure increase, and K1 (P − Pcrit) = 0 when P < Pcrit.
Diffusion of chemical components and heat conduction are described by Fick’s law:
where Cp is the specific heat capacity, ρ is the density, and Q is the volumetric heat generation; C is the concentration of the chemical component, D is the diffusion coefficient, and
The temperature dependence of the diffusion coefficient is described by the Arrhenius equation:
where
Heat generation is caused by two factors: plastic deformation of the materials and the thermal effect of the reaction:
where
The heat transfer calculation was performed using a first-order approximation of the temperature derivatives with respect to time:
A first-order accurate time integration scheme was used for the heat transfer calculation:
For the Laplace operator, the Morris approximation was used [29].
An implicit finite difference scheme for calculating unsteady thermal fields is derived from Eqs. (14) and (15):
The system of linear equations arising from (15) was solved using the simple iteration method. Because the solution of this system changes smoothly and slowly over time, the iterative process converges quickly, and more efficient and complex methods are unnecessary.
The pressure is determined using a Mie-Grüneisen equation of state, as follows:
where
where
The numerical scheme for calculating the elastoplastic flow of chemically reacting mixtures uses the smoothed particle hydrodynamics method, employing a “kernel approximation” for spatial derivatives. The simulated volume is discretized using a set of approximating points, each characterized by its position, mass, density, and smoothing length. The smoothing length is selected to ensure overlap between each particle’s smoothing domain and those of its neighbors, while limiting the number of overlapping neighbors (typically to less than 30).
To validate the numerical model, a convergence study was performed. The accuracy of the calculations was verified by monitoring the conservation of total mechanical energy, momentum, and angular momentum. In addition, the model was validated by comparing simulation results with experimental data on the shock-induced reaction in Al/CuO mixtures [10].
We investigated the shock-wave loading of energetic mixtures (Al/S, Al/CuO, Al/PTFE) within a conical steel ampoule with dimensions L = 12 cm, R1 = 0.75 cm, R2 = 1.5 cm, and H = 1 cm (Fig. 1). The steel has a density of 7.87 g/cm3, an elastic modulus of 200 GPa, and a shear modulus of 70 GPa. The conical geometry was chosen to create varying conditions for reaction initiation and propagation by modifying the intensity and magnitude of the shock wave.
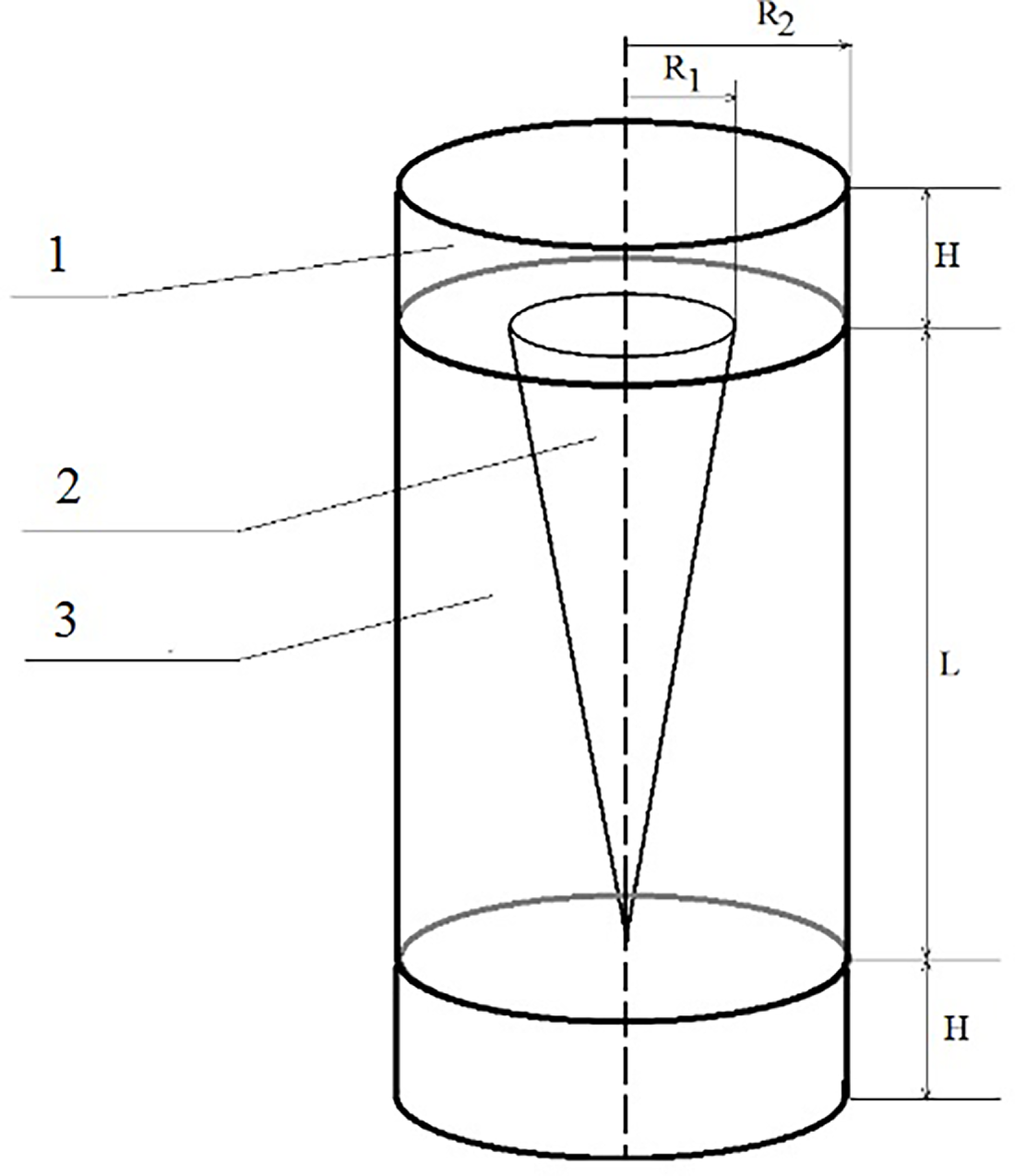
Figure 1: Schematic of the numerical experiment and configuration of the conical ampoule containing the thermite mixture at time t = 0: 1—impactor; 2—conical sample of the mixture; 3—ampoule walls.
The reaction within the conical ampoule is initiated by the impactor striking its wider section. This generates a high-intensity shock wave (SW), triggering the chemical reaction of the powder mixture components. Two primary wave types propagate within the ampoule: a shock wave in the ampoule walls and a shock wave propagating through the powder mixture.
Propagation of the shock wave within the ampoule walls is accompanied by a gradual decrease in its intensity. This is attributable to several factors. First, as the wave propagates along the ampoule, the wall thickness increases, causing the initial energy to be distributed over a larger cross-sectional area. Second, the shock wave propagates faster in the metal walls than in the powder mixture. Consequently, a convergent shock wave forms from the ampoule walls along the axial direction, which can also initiate a chemical transformation within the powder mixture.
The converging nature of the shock wave (SW) within the powder mixture results in its gradual attenuation along the ampoule’s axis. Consequently, the reaction initiation conditions change smoothly: the SW intensity decreases. Simultaneously, the pressure on the wave front may increase as it propagates inward from the walls towards the center of the powder mixture.
Thus, the conical ampoule provides unique conditions for the reaction to proceed under various regimes. High pressure from the shock wave primarily initiates the reaction in the wider section, while temperature becomes dominant as the wave propagates toward the narrower section. Exceeding a critical pressure increases the reaction rate, further complicating the process kinetics and potentially leading to interesting results [30–32]. This configuration allows for varying the initiation and propagation conditions, enabling the investigation of different kinetic regimes and their influence on the process.
The procedure for the numerical simulation was as follows:
1. Initial conditions: Definition of the computational domain geometry, initial velocities, temperatures, and material properties.
2. Neighbor search for SPH particles.
3. Calculation of material properties considering temperature.
4. Calculation of elastic-plastic flow of materials using the SPH method:
a strain rates, temperature gradients, and heat fluxes;
b stress increments per time step;
c influence of artificial viscosity;
d nodal accelerations and their displacements per time step.
5. Calculation of chemical reactions:
a calculation of changes in component concentrations per time step;
b calculation of new properties for the mixture of reacting components;
c calculation of heat release from the chemical reaction;
d calculation of the equations of state for the reacting components and equilibrium pressure values in the multicomponent mixture for the new component composition.
6. Temperature calculation: accounting for heat release from chemical reactions, plastic deformation, and changes in the properties of reacting components.
7. Calculation of new stresses, coordinates, temperatures, and other parameters of the nodes.
8. Calculation of the time step based on the of the Friedrichs–Courant–Lewy (FCL) condition.
9. Conservation law monitoring: energy and momentum conservation.
10. Next time step.
3.1 Exothermic Reactions in Al/S and Al/CuO Mixtures
Calculations were performed to simulate zero- and first-order chemical reactions occurring in Al/S and Al/CuO thermite mixtures under shock-wave loading. The impact velocity was varied from 250 to 350 m/s, in increments of 25 m/s. The parameters governing reaction initiation and progression were as follows: critical pressure [2 GPa (Al/S), 1.6 GPa (Al/CuO)], critical temperature [933 K (Al/S), 708 K (Al/CuO)], reaction rate constant (K0) [0.06 μs−1 (Al/S), 0.058 μs−1 (Al/CuO)], and heat of reaction [4820 J/g (Al/S), 4080 J/g (Al/CuO)]. Beyond the critical pressure, the reaction exhibits accelerated kinetics. Acceleration constants Kp = 2.5, 5, and 10 were used to model the zero-order reaction. For the first-order reaction, acceleration reaction constants K1 = 0.025/GPa, 0.05/GPa, 0.1/GPa, 2.5/GPa, 5/GPa, and 10/GPa were used.
Fig. 2 shows the average reaction depth in the Al/S mixture vs. impact velocity. The calculations (Fig. 2) show a similar pattern of reaction product accumulation for both zero- and first-order reactions. In the wide section of the conical ampoule, where the pressure behind the shock front significantly exceeds the critical pressure, the reaction in the mixture proceeds at an accelerated regime directly in the shock wave front. However, the conical geometry of the ampoule causes the shock wave amplitude to decay as it propagates toward the narrow end. Below a critical pressure, the shock wave front can no longer sustain the accelerated regime of reaction. In this case, the reaction continues to proceed at a slower rate due to the elevated (critical) temperature. Due to the further decrease of the shock wave amplitude as it moves along the cone, the reaction depth of the mixture decreases, and eventually, when the temperature falls below the critical temperature, the reaction stops completely.
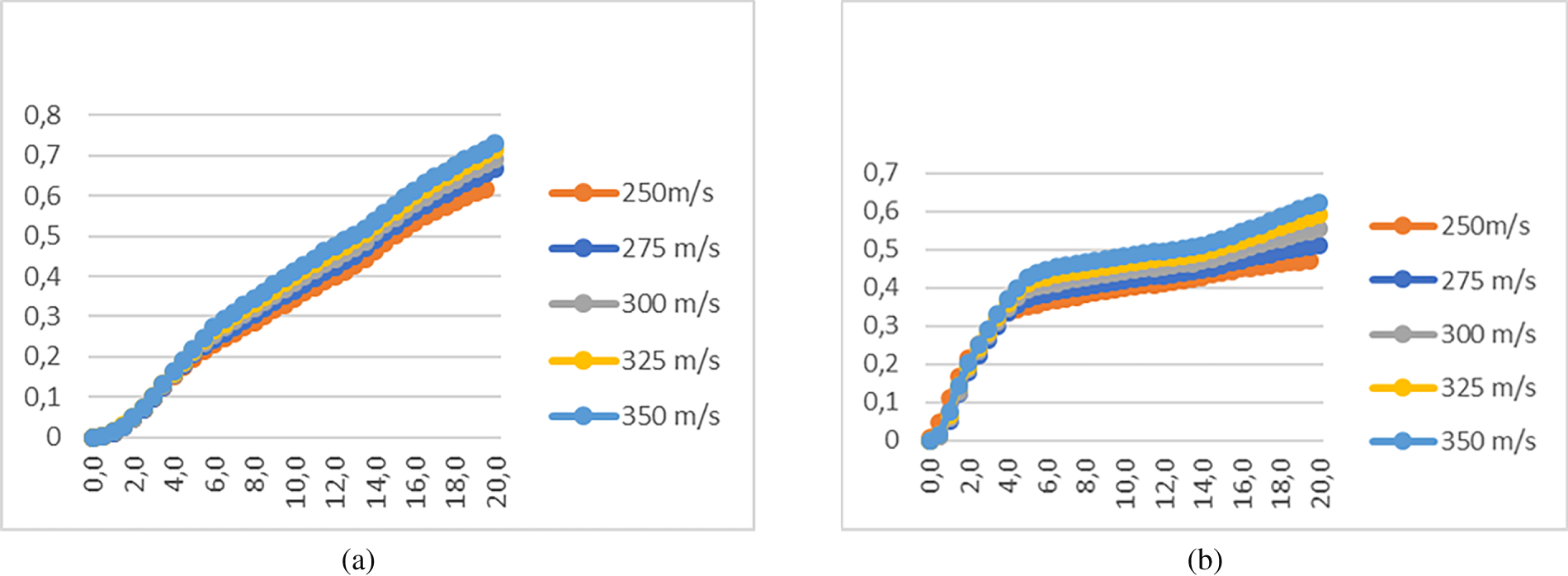
Figure 2: Average reaction depth in the Al/S mixture under shock-wave loading for the zero-order chemical reaction (Kp = 2.5, (a)) and the first-order chemical reaction (K1 = 2.5/GPa, (b)).
Fig. 3 shows the average reaction depth for the Al/CuO mixture vs. impact velocity.
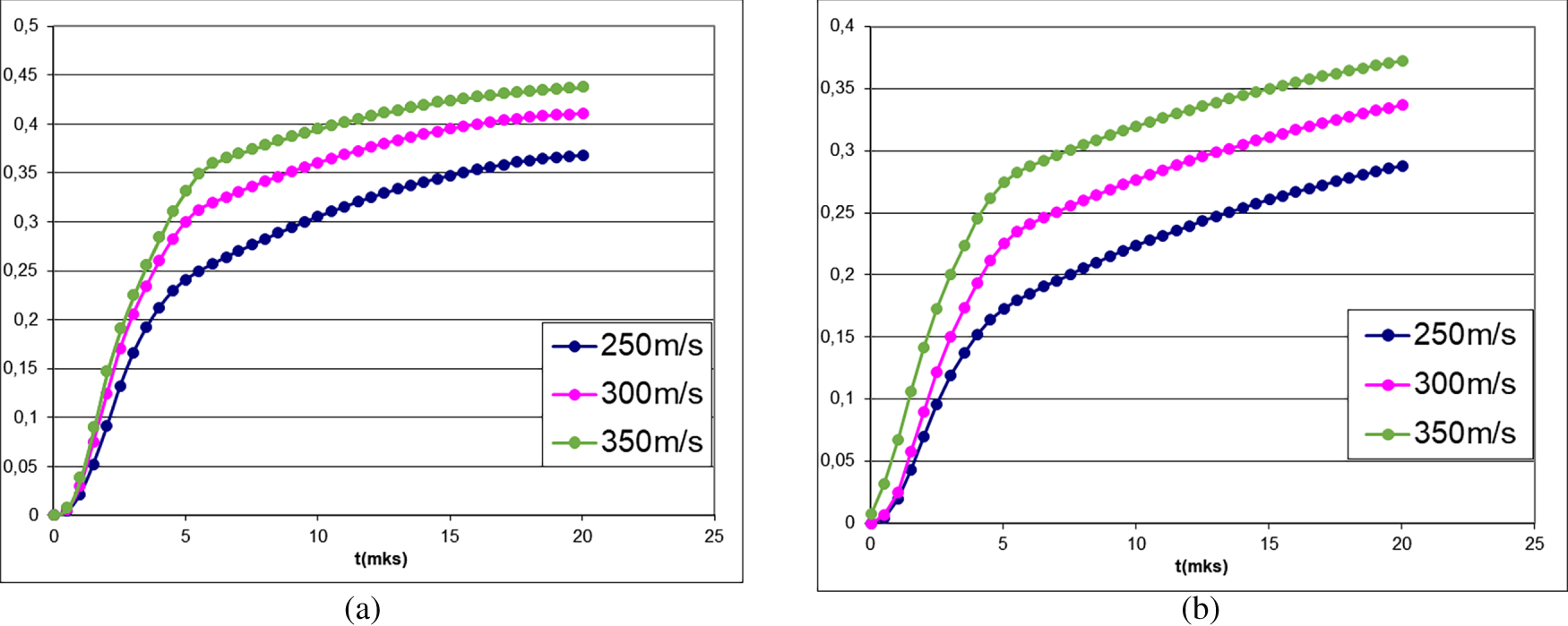
Figure 3: Average reaction depth in the Al/CuO mixture under shock-wave loading for the zero-order chemical reaction (Kp = 2.5, (a)) and the first-order chemical reaction (K1 = 2.5/GPa, (b)).
The lower reaction initiation temperature and pressure for Al/CuO (708 K and 1.6 GPa) compared to Al/S (933 K and 2 GPa) suggest that Al/CuO should be more sensitive to low shock wave pressures and exhibit a higher response even at lower impact velocities.
However, analysis of Fig. 3 reveals that despite satisfying the pressure criterion and having a similar initiation of reaction, the Al/CuO mixture exhibits a lower reaction depth (0.45 for zero-order and 0.38 for first-order reactions) than Al/S (0.75 and 0.65, respectively, at an impact velocity of 350 m/s).
This indicates that while the reaction in Al/CuO initiates more readily, subsequent steps are slower or less efficient compared to Al/S. This may be due to both the slower reaction rate and lower energy release of Al/CuO, as well as factors limiting contact between the reactants. For example, the formation of a dense layer of copper or aluminum oxide on the surface of reacting particles may impede further propagation of the reaction. In contrast, the products of the Al/S reaction likely have a morphology that does not so strongly inhibit further reaction.
Analyzing Figs. 2 and 3, the following conclusions can be drawn:
– Al/CuO reacts more readily, but reacts incompletely compared to Al/S.
– The lower values of reaction initiation temperature and pressure for Al/CuO are not the only determining factors for the reaction.
– Kinetic factors such as reaction rate and product morphology play a significant role.
– The Al/S system likely transfers the shock wave energy to the reactants more efficiently and maintains a higher reaction rate at all stages.
3.2 Exothermic Reactions in Al/PTFE Mixtures
In contrast to the reactions of aluminum with sulfur and copper oxide, which proceed without significant volume change or gas release, permitting the collection of reaction products in quantities sufficient to study the shock-wave loading of ampoules, the reaction with polytetrafluooethylene (PTFE) shows a significantly different behavior (Figs. 4 and 5).

Figure 4: Combustion of Al/PTFE under ambient atmospheric conditions (in air, without shock loading).

Figure 5: Images of two steel cylindrical ampoules containing Al/PTFE, broken during the experiment under shock-wave loading: (a) Al/PTFE porosity of 50%; (b) Al/PTFE porosity of 4.5%; detonation velocity of 3.85 km/s.
Polytetrafluoroethylene, (C2F4)n, serves as a source of carbon and fluorine, which react exothermically with aluminum to form aluminum carbides and fluorides. Thermodynamic calculations [33] indicate that temperatures during this reaction may reach 3000 K, resulting in the formation of both molten carbides and gaseous fluorides, notably aluminum fluoride (AlF3). The energy released significantly exceeds that of the trinitrotoluene decomposition reaction by a factor of two. Experiments on shock-induced initiation of the Al/PTFE reaction in steel ampoules demonstrate ampoule fracture, directly evidencing the formation of a gaseous phase during the reaction [34]. It is noteworthy that aluminum fluoride (AlF3), while being a crystalline solid with a density of 2.88 g/cm3 (exceeding that of the initial mixture) under ambient conditions, forms in the gaseous phase during the high-temperature shock reaction.
Due to the extreme conditions accompanying the Al/PTFE reaction, several unresolved questions remain regarding its mechanism. Such reactions are not sufficiently studied, as its characteristic time is comparable to the shock wave’s duration (~tens of microseconds). It should also be noted that the experimental setup using a cylindrical ampoule—in the case of ampoule fracture—does not allow for the acquisition of sufficient data to evaluate the reaction (Fig. 5). As a possible alternative for a more informative experiment with such mixtures We propose using conical ampoules as a potential alternative for more informative experiments with these mixtures.
Therefore, accurate modeling of the Al/PTFE reaction under shock-wave loading necessitates the determination of critical reaction parameters: the reaction rate, total heat effect, critical pressure, and reaction onset temperature, along with the effect of pressure on reaction rate. Furthermore, determining the equation of state parameters for the reaction products, particularly gaseous aluminum fluoride, is essential. Unfortunately, these data are currently absent from the available literature.
3.3 Choice of Parameters for Al/PTFE Reaction
For modeling the Al/PTFE reaction, the following parameters were selected: critical pressure
A key feature of the Al/PTFE mixture is that aluminum fluoride, a reaction product, exists in the gaseous state under critical reaction temperatures and pressures. Therefore, a multicomponent mixture model was employed in the numerical simulation [31], incorporating the reacting mixture’s variable composition, the gaseous phase of the reaction products (aluminum fluoride), and the distinct properties of each component. Using these parameters, the simulations accurately predicted ampoule wall destruction, consistent with experimental observations. The reaction terminates roughly 3.5 cm from the cone’s apex (Fig. 6), where the pressure wave intensity drops below the critical initiation threshold. At this location, heat from the reaction zone is efficiently dissipated into the surrounding conical ampoule material, thus reducing the reactant temperature below the ignition point.
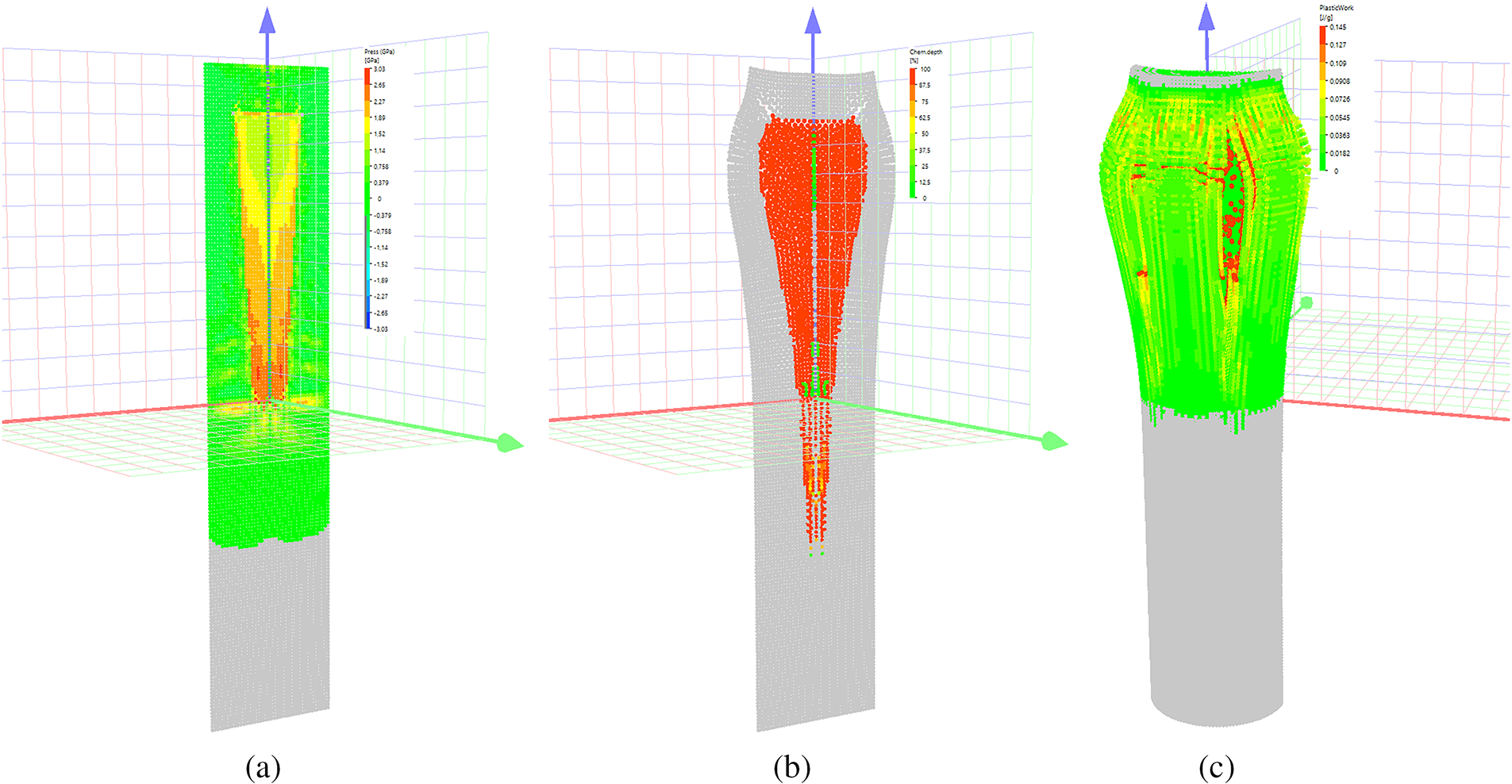
Figure 6: Ampoule configurations: (a) at t = 10 μs, showing pressure in color; (b) t = 40 μs, showing chemical conversion depth in color; (c) Crack propagation in the ampoule walls at t = 50 μs.
As shown in Fig. 7, as the shock wave propagates along the axis of the ampoule, the maximum pressure within the reacting mixture decreases. This leads to a reduction in the reaction rate (which is pressure-dependent) and a decrease in the maximum conversion achieved under shock-wave loading. Approximately in the center of the ampoule, conditions are met where a portion of the material remains unreacted. Furthermore, the volume of this unreacted material is dependent on the reaction parameters, allowing experiments with a conical ampoule to be used to obtain more detailed information on the effect of pressure on the initiation and progression of reactions in the reacting mixtures.
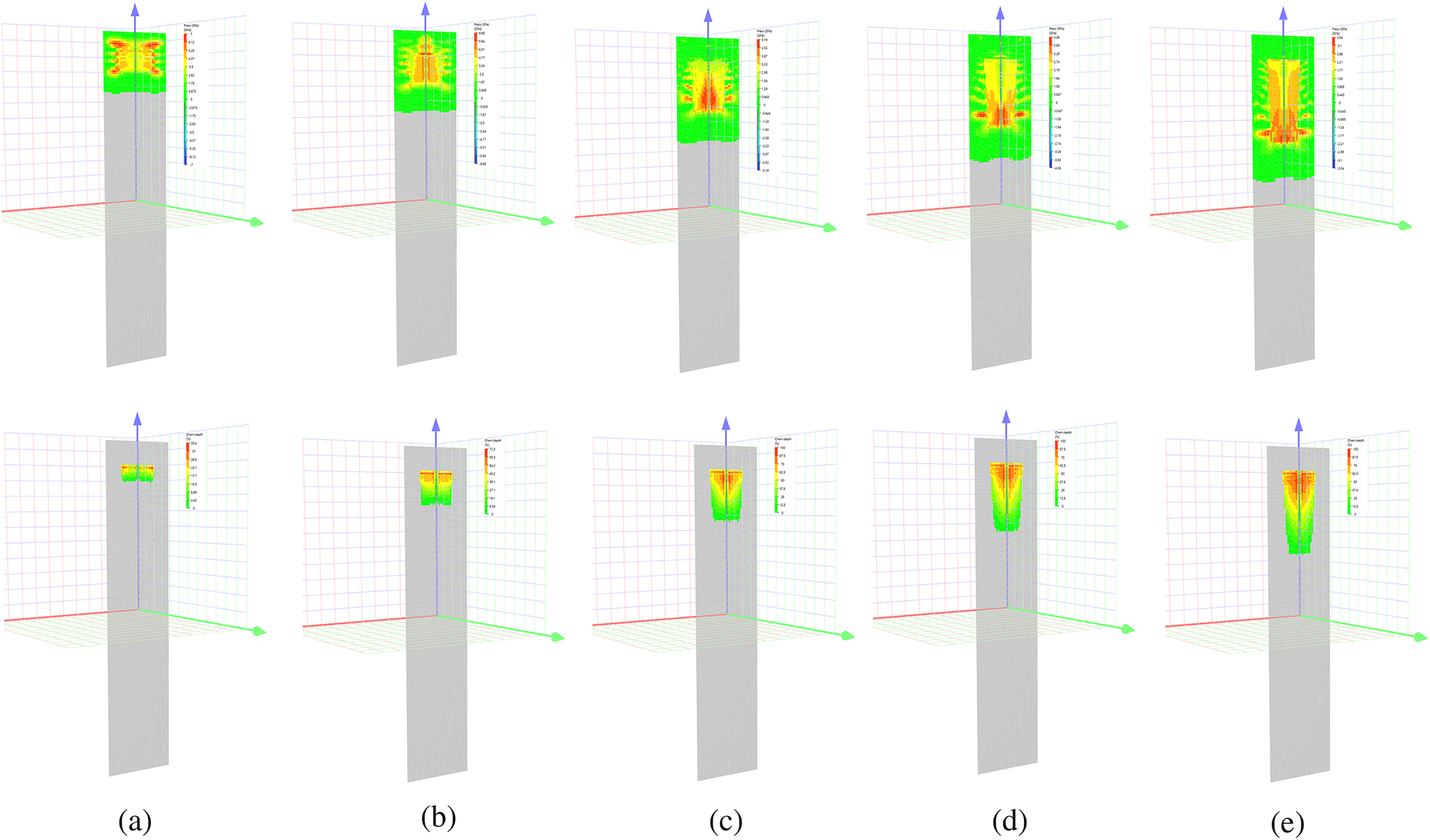
Figure 7: Ampoule configurations: (a) at t = 1 μs, (b) at t = 2 μs (c) at t = 3 μs (d) at t = 4 μs (e) at t = 5 μs showing pressure in color (first line) and reaction depth (second line).
The maximum calculated temperature of the reaction products was 2630 K, which is in close proximity to the adiabatic combustion temperature of the Al/PTFE mixture (2700 K) reported in [33]. This finding suggests that the model is realistic. It is possible to adjust the position of the reaction stopping point in the calculations and the ampoule fracture by varying the numerical parameters of the model. These numerical parameters include the reaction rate, critical pressure and reaction onset temperature, as well as the reaction acceleration constant. Given the relative ease of experimentally determining these parameters, the reaction model can be refined by comparing simulation results with experimental data. This approach will yield a more accurate and predictive model of Al/PTFE mixture behavior under extreme conditions.
In this study we developed and validated a numerical model using the Smoothed Particle Hydrodynamics (SPH) method to simulate shock-induced exothermic reactions in Al/S, Al/CuO, and Al/PTFE energetic mixtures. The model effectively couples elastic-plastic mechanics, thermal effects, component diffusion, and chemical kinetics (including both zero- and first-order mechanisms).
The conducted analysis confirmed that, while initial reaction tendency in Al/CuO is higher than in Al/S (due to lower critical conditions), the Al/S mixture demonstrates superior reaction efficiency under shock-wave loading, suggesting that post-initiation kinetic factors and product morphology play a crucial role in overall energy release.
The incorporation of the gaseous phase dynamics—specifically the formation of aluminum fluoride (AlF3)—into the Al/PTFE simulation is essential for accurately predicting ampoule fracture mechanisms observed experimentally.
The use of the conical ampoule was numerically justified, as the changing shock wave intensity along the ampoule axis generates a gradient of reaction conditions (Pmax, rate, conversion). This gradient confirms the proposed experimental geometry for systematically characterizing the pressure dependence of reaction initiation and propagation kinetics, which is often lacking in standard planar shock experiments.
The developed model, calibrated against experimental data, offers a predictive tool capable of capturing complex phenomena such as reaction quenching due to shock wave decay and coupling between mechanical fracture and chemical energy release.
In summary, this work contributes to the fundamental understanding of exothermic mixtures under shock-wave loading and offers a tool for developing and optimizing novel energetic materials.
Acknowledgement: Not applicable.
Funding Statement: This work was conducted under the public contract for the Tomsk Scientific Center of the Siberian Branch of the Russian Academy of Sciences (Project No. FWRF-2024-0009).
Author Contributions: The authors confirm contribution to the paper as follows: Conceptualization, Oksana Ivanova; methodology, Oksana Ivanova and Roman Cherepanov; software, Roman Cherepanov; validation, Roman Cherepanov; formal analysis, Oksana Ivanova and Roman Cherepanov; investigation, Oksana Ivanova and Roman Cherepanov; resources, Sergey Zelepugin; data curation, Roman Cherepanov; writing—original draft preparation, Oksana Ivanova and Roman Cherepanov; writing—review and editing, Oksana Ivanova and Sergey Zelepugin; visualization, Roman Cherepanov; supervision, Oksana Ivanova and Sergey Zelepugin; project administration Sergey Zelepugin; funding acquisition, Sergey Zelepugin. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: Data available on request from the authors. The data that support the findings of this study are available from the Corresponding Author, [Oksana Ivanova], upon reasonable request.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Liu WH, Liu QJ, Liu FS, Liu ZT. Machine learning approaches for predicting impact sensitivity and detonation performances of energetic materials. J Energy Chem. 2025;102:161–71. doi:10.1016/j.jechem.2024.10.035. [Google Scholar] [CrossRef]
2. Gu F, Xiao J. First-principles study on crystal structure, mechanical thermodynamics, and electronic properties of perovskite energetic materials under high pressure. Chem Phys. 2025;598(4):112802. doi:10.1016/j.chemphys.2025.112802. [Google Scholar] [CrossRef]
3. Zhang Z, Tian W, Wang T, Liu Z, Yang Y, Ge C, et al. Impact-induced energy release of typical HCP metal/PTFE/W reactive materials: experimental study and predictive modeling via machine learning. Def Technol. 2025;47(20):124–38. doi:10.1016/j.dt.2025.01.006. [Google Scholar] [CrossRef]
4. Yan T, Li H, Xi D, Sun L, Liu L, Jin D. Pressure-induced structural phase transition of energetic material 4-aminobenzamide. Chem Phys Lett. 2025;869:142072. doi:10.1016/j.cplett.2025.142072. [Google Scholar] [CrossRef]
5. Duran AV, Ramazani A, Sundararaghavan V. Multi-scale modeling of shock wave propagation in energetic solid-state composites. Int J Solids Struct. 2023;285(1):112535. doi:10.1016/j.ijsolstr.2023.112535. [Google Scholar] [CrossRef]
6. Liu R, Wang KY, Feng JR, Huang LL, Geng HH, Ge C, et al. Effects of shock-induced chemical reaction on equation of state for Ni/Al energetic structural material. Energ Mater Front. 2023;4(2):93–102. doi:10.1016/j.enmf.2023.06.001. [Google Scholar] [CrossRef]
7. Ye C, Zhang Q. High-speed flow and multiphase detonation of energetic mixture. Energy. 2025;314:134238. doi:10.1016/j.energy.2024.134238. [Google Scholar] [CrossRef]
8. Wan C, Chen S, Liu X, Xiong Y, Qin Z, Li J, et al. Enhancing energy release and controlled combustion of B-based MICs by the synergistic effect between polyvinyl pyrrolidone and nitrocellulose. Chem Eng J. 2024;500(5):157474. doi:10.1016/j.cej.2024.157474. [Google Scholar] [CrossRef]
9. Sullivan K, Young G, Zachariah MR. Enhanced reactivity of nano-B/Al/CuO MIC’s. Combust Flame. 2009;156(2):302–9. doi:10.1016/j.combustflame.2008.09.011. [Google Scholar] [CrossRef]
10. Dolgoborodov AY, Kirilenko VG, Brazhnikov MA, Grishin LI, Kuskov ML, Valyano GE. Ignition of nanotHermites by a laser diode pulse. Def Technol. 2022;18(2):194–204. doi:10.1016/j.dt.2021.01.006. [Google Scholar] [CrossRef]
11. Sanders VE, Asay BW, Foley TJ, Tappan BC, Pacheco AN, Son SF. Reaction propagation of four nanoscale energetic composites (Al/MoO3, Al/WO3, Al/CuO, and B12O3). J Propuls Power. 2007;23(4):707–14. doi:10.2514/1.26089. [Google Scholar] [CrossRef]
12. Tichtchenko E, Magliano H, Wu T, Pillemont L, Simonin O, Bédat B, et al. Numerical modeling and experimental study of self-propagating flame fronts in Al/CuO tHermite reactions. Int J Therm Sci. 2025;211(5):109687. doi:10.1016/j.ijthermalsci.2025.109687. [Google Scholar] [CrossRef]
13. Geng H, Wang J, Xiong W, Ma L, Hou X, Chi R, et al. Dynamic behavior and shock-induced energy release characteristics of Ni/Al energetic structural materials prepared by cold spray additive manufacturing. J Mater Res Technol. 2025;35(8):7393–404. doi:10.1016/j.jmrt.2025.03.073. [Google Scholar] [CrossRef]
14. Liu R, Zhang W, Wang K, Chen P, Ge C, Wang H. Multiscale thermodynamics of Ni/Al energetic structural materials under shock. J Appl Phys. 2024;135(13):135103. doi:10.1063/5.0195672. [Google Scholar] [CrossRef]
15. Xiao J, Zhang J, Ma J, An D, Wei R, Liu S. Mechanics–thermotics–chemistry coupling response model and numerical simulation of reactive materials under impact load. Adv Eng Softw. 2024;192(1):103647. doi:10.1016/j.advengsoft.2024.103647. [Google Scholar] [CrossRef]
16. Cai Y, Feng X, He C, Zhang S, Li S, Liu J. A 7.62 mm energetic bullet filled with PTFE-Mg-based reactive materials for anti-drone application. J Mater Res Technol. 2024;30:8749–59. doi:10.1016/j.jmrt.2024.05.240. [Google Scholar] [CrossRef]
17. Yetter RA. Progress towards nanoengineered energetic materials. Proc Combust Inst. 2021;38(1):57–81. doi:10.1016/j.proci.2020.09.008. [Google Scholar] [CrossRef]
18. Wang T, Cai Y, Guo L, Wang C, He Y, He Y. Energy release characteristics of Al/PTFE reactive materials under laser ignition experiment. Int J Therm Sci. 2025;211:109693. doi:10.1016/j.ijthermalsci.2025.109693. [Google Scholar] [CrossRef]
19. Qiang W, Xiao Z, Tang E. Mechanical behavior of polytetrafluoroethylene/aluminum energetic materials under high and low temperature conditions. J Mater Eng Perform. 2023;32(24):11192–204. doi:10.1007/s11665-023-07936-z. [Google Scholar] [CrossRef]
20. Ren X, Li Y, Huang J, Wu J, Wu S, Liu Q, et al. Effect of addition of HTa to Al/PTFE under quasi-static compression on the properties of the developed energetic composite material. RSC Adv. 2021;11(15):8540–5. doi:10.1039/d0ra09084k. [Google Scholar] [PubMed] [CrossRef]
21. Geng H, Liu R, Ren Y, Chen P, Ge C, Wang H. Dynamic compression-shear ignition mechanism of Al/PTFE reactive materials. Compos Struct. 2024;331(5):117908. doi:10.1016/j.compstruct.2024.117908. [Google Scholar] [CrossRef]
22. Saikov I, Seropyan S, Malakhov A, Saikova G, Denisov I, Petrov E. Energetic materials based on W/PTFE/Al:thermal and shock-wave initiation of exothermic reactions. Metals. 2021;11(9):1355. doi:10.3390/met11091355. [Google Scholar] [CrossRef]
23. Ren X, Liu J, Wu J, Li Y, Tu Y, Meng Z. A wide range of strain rate sensitivity and impact-induced behavior of Al/PTFE reactive materials: experimental and numerical investigation. Compos Part A Appl Sci Manuf. 2025;192(2):108797. doi:10.1016/j.compositesa.2025.108797. [Google Scholar] [CrossRef]
24. Jiang F, Sun P, Bu Y, Niu Y, Li Y, Zhang K, et al. Energy output performance of aluminized explosive containing Al/PTFE reactive materials. RSC Adv. 2024;14(46):34348–55. doi:10.1039/d4ra01476f. [Google Scholar] [PubMed] [CrossRef]
25. Guo BY, Ren KR, Ma XY, Li G, Huang CM, Li ZB, et al. Shock reaction model for impact energy release behavior of Al/PTFE reactive material. Energ Mater Front. 2024;5(4):329–42. doi:10.1016/j.enmf.2024.09.004. [Google Scholar] [CrossRef]
26. Cherepanov R, Ivanova O, Zelepugin S. SPH simulation of shock-induced chemical reactions in reactive powder mixtures. Mater Today Commun. 2024;38(2):108261. doi:10.1016/j.mtcomm.2024.108261. [Google Scholar] [CrossRef]
27. Siva Prasad AVS, Basu S. Numerical modelling of shock-induced chemical reactions (SICR) in reactive powder mixtures using smoothed particle hydrodynamics (SPH). Model Simul Mater Sci Eng. 2015;23(7):075005. doi:10.1088/0965-0393/23/7/075005. [Google Scholar] [CrossRef]
28. Chen Y, Kulasegaram S. Numerical modelling of fracture of particulate composites using SPH method. Comput Mater Sci. 2009;47(1):60–70. doi:10.1016/j.commatsci.2009.06.014. [Google Scholar] [CrossRef]
29. Schwaiger HF. An implicit corrected SPH formulation for thermal diffusion with linear free surface boundary conditions. Int J Numer Meth Eng. 2008;75(6):647–71. doi:10.1002/nme.2266. [Google Scholar] [CrossRef]
30. Zelepugin SA, Ivanova OV, Yunoshev AS, Sil’vestrov VV. The development of the aluminum sulfide synthesis reaction on explosive loading of a cylindrical ampoule. Dokl Phys Chem. 2010;434(2):172–6. doi:10.1134/s0012501610100052. [Google Scholar] [CrossRef]
31. Ivanova O, Zelepugin S, Yunoshev A, Silvestrov V. A multicomponent medium model for reacting porous mixtures under shock wave loading. J Energ Mater. 2010;28(sup1):303–17. doi:10.1080/07370652.2010.505940. [Google Scholar] [CrossRef]
32. Ivanova OV, Cherepanov RO, Zelepugin SA. Numerical simulation of solid-phase chemical transformations in tHermite mixtures under shock-wave loading. J Phys Conf Ser. 2022;2154(1):012005. doi:10.1088/1742-6596/2154/1/012005. [Google Scholar] [CrossRef]
33. Saikov IV, Seropyan SA, Saikova, GR, Alymov MI. Shock-wave initiation of exothermic reactions in W–Al–PTFE reactive materials. Dokl Phys Chem. 2020;492(1):60–3. doi:10.1134/S0012501620350018. [Google Scholar] [CrossRef]
34. Zelepugin SA, Ivanova OV, Yunoshev AS, Zelepugin AS. Destruction of cylindrical ampoules containing solid phase reaction mixtures under explosive loading. Lett Mater. 2015;5(4):468–72. doi:10.22226/2410-3535-2015-4-468-472. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools