 Open Access
Open Access
ARTICLE
Studying the Electrocatalytic Hydrogen Evolution Reaction Performance of L10-NiM Intermetallic Compounds by DFT Calculation
1 Science and Technology Development Corporation, Shenyang Ligong University, Shenyang, China
2 Ordos Institute, Liaoning Technical University, Ordos, China
3 College of Materials Science and Engineering, Liaoning Technical University, Fuxin, China
4 Key Laboratory of Anisotropy and Texture of Materials (Ministry of Education), Northeastern University, Shenyang, China
* Corresponding Authors: Chun Wu. Email: ; Runqing Liu. Email:
(This article belongs to the Special Issue: Advances in Computational Materials Science: Focusing on Atomic-Scale Simulations and AI-Driven Innovations)
Computers, Materials & Continua 2026, 87(3), 28 https://doi.org/10.32604/cmc.2026.077864
Received 18 December 2025; Accepted 03 March 2026; Issue published 09 April 2026
Abstract
The intermetallic compounds with modulated electronic structure can provide more catalytically active sites and enhance electrocatalytic performance. In this study, the first-principles calculation method has been employed to investigate the potential of L10-NiM (M = Mn, Fe, Co, Cu, Zn, Mo) intermetallic compounds for electrocatalytic hydrogen evolution reaction (HER). Firstly, the L10-NiM present a homogenized charge transfer environment, where the Bader charge difference on the catalyst surface is below 0.13 e, significantly mitigating the locally strong adsorption of adsorbates in Ni. Additionally, the L10-NiM also fine-tunes the antibonding orbital interactions with adsorbates, facilitating both water dissociation and proton reduction. Furthermore, the L10-NiCu exhibits better HER electrocatalytic activity, with a water dissociation energy barrier of 0.49 eV and a Gibbs free energy of hydrogen adsorption of −0.524 eV. A scaling relationship analysis reveals a good linear correlation between HER activity and adsorption descriptors across the investigated L10-NiM intermetallic compounds, providing a theoretical foundation for the development of low-cost catalysts.Keywords
Recently, the establishment of a sustainable, zero-emission energy system has attracted more and more attention [1]. Compared with steam methane reforming and coal gasification, the production of green hydrogen by water electrolysis plays a crucial role in hydrogen energy application and in reducing environmental pollution [2]. The hydrogen evolution reaction (HER) is one of the key reactions in water electrolysis, and it is the common pursuit of researchers to develop HER electrocatalysts with high activity, enhanced chemical stability, and excellent selectivity [3].
Nickel-based alloy catalysts are widely utilized in low-cost alkaline electrolyzers. Compared with pure Ni catalysts, the NiM (M = Mn, Fe, Co, Cu, etc.) alloys can improve the electrocatalytic HER performance by controlling the exposure of active atoms, adjusting the reaction potentials, and enhancing thermal stability at high temperatures [4–6]. Alloying also improves the d-band centre and changes the hydrogen adsorption Gibbs free energy of Ni; the NiCu electrocatalyst is reported to exhibit an approximately optimal ΔGH when compared with Ti, V, Mn, Fe, and Co. The overpotential is only 23 mV at a current density of 10 mA/cm2 [6]. In addition, alloying Ni with similar thermal workability elements (such as Fe, Co, Cu, and so on) can induce the formation of the intermetallic L10-phase to improve catalytic activity [7–10]. The formation of an orderly L10-phase can change the electronic structure, provide more active sites, promote H2O dissociation, and facilitate H adsorption, which significantly enhances the efficiency of the HER and the oxygen reduction reaction [7–10]. However, the research on ordered intermetallic compounds in electrocatalysis mainly focuses on Pt-based noble metal alloys. Typically, the Fe became more stable in the core-shell structure of L10-FePt/Pt with a 5 Å Pt shell, which enhances the electrocatalysis performance under fuel cell operating conditions by Pt shell compression [11]. The ordered NiPt alloy can be prepared at a lower temperature by tuning the strength of the metal bond, which can be used in fuel cells [12]. Optimizing the composition, size, and ordering degree of Pt-based intermetallic compounds to promote electrocatalysis performance has been widely studied [13–16]. The excellent electrocatalytic performance of ordered Pt-based alloys encourages us to develop ordered non-precious metal catalysts, especially Ni-based intermetallic compounds. Therefore, it is of great significance to analyze the effect of Ni-based intermetallic compounds on HER, revealing the catalytic mechanism will provide a reference for the development of a new high-performance Ni-based electrocatalyst.
In this research, the electrocatalytic HER properties of typical L10-NiM (M = Mn, Fe, Co, Cu, Zn, Mo) intermetallic compounds were studied by First-principles calculations based on Density Functional Theory (DFT). Through key measurement methods such as geometric configuration reorganization, electronic structure changes, and the free energy of the reaction rate-determining step, a comprehensive analysis and discussion of ordered L10-NiM intermetallic compounds is conducted. By understanding the contribution degree of the outer orbitals and the electron migration ability of the active surface, the regularity of HER activity is expounded, expecting to provide effective guidance for the design and development of ordered intermetallic compounds in combination with scaling relationships.
The DFT calculations were performed using the Vienna Ab initio Simulation Package (VASP) version 5.4.4 [17]. The projective additive wave (PAW) method based on the generalized gradient approximation (GGA) and the Perdew-Burke-Ernzerhof (PBE) exchange-correlation energy function was applied [18]. The super-cell models used in this paper were a 2 × 2 × 2 expansion of Ni with fcc structure and ordered L10-NiM (M = Mn, Fe, Co, Cu, Zn, Mo) alloys with a fct structure. The surface models were the (100) and (111) faces of fcc-Ni and L10-NiM. The cutoff energy and K-point were selected as 700 eV and 4 × 4 × 1 by convergence testing. The spin polarization was turned on during calculations to ensure structural stability [19]. To precisely calculate the density of states (DOS), the K-point grid was expanded to 8 × 8 × 1 [20]. Meanwhile, to avoid the mutual interference of the periodic arrangement and eliminate the weak force of interlayer atoms, the thickness of the vacuum layer was selected as 15 Å, and the atom layer is set as 4. To ensure the accuracy of the calculations, the geometry optimization was stopped when the force reached 0.02 eV/Å, the energy reached 10−6 eV, and the maximum displacement tolerance was 0.02 Å. Free energy corrections for all intermediate species were performed using VASPKIT [21]. The implicit solvation model VASPsol was employed to determine the computational PZCs, where a bulk dielectric constant εk = 78.4, dielectric cavity width σ = 0.6, cutoff charge density ρcut = 0.0025 Å–3, and a surface tension parameter of 0.525 meV/Å2 were used [22]. The LDA + U electronic correlation was used to adopt strong localization in transition metals, the LDAU was set as TRUE, and the initial Hubbard U = 4 eV and J = 1 eV were applied for Ni 3d, Mn 3d, Fe 3d, Co 3d, Cu 3d, Zn 3d, and Mo 4d orbitals. Figures were visualized and analyzed using VESTA [23]. The climbing image nudged elastic band (CI-NEB) method was used to obtain the transition states, where the spring constant K was set as –5, and the transition states were verified through vibrational frequency analysis [24].
The electronic and catalytic parameters obtained in this paper were calculated as follows. The energy required for surface dissociation ΔEd = −ΔEA + ΔEB + ux, where ΔEA is the total energy of the normal surface, ΔEB is the total energy of the vacant surface, and ux is the chemical potential of the x atom (uNi = −4.51 eV) [25]. The Gibbs free energy for H adsorption (ΔGH*) in HER followed the definition described by Nørskov, ΔGH* = ΔEH + ΔEZPE − TΔSH, where ΔEZPE = 0.04 eV was the value of hydrogen adsorption on different metallic surfaces, ΔSH was the entropy difference of H between the adsorption state and the gas phase, T was the room-temperature 298.15 K, and TΔSH was determined to be −0.20 eV [26,27]. The effectiveness of the hydrogen catalytic improved with increasing exchange current density (i0), and the i0 at pH = 0 is calculated as i0 = −ek0/[1 + exp(−ΔGH*/kBT)], where the rate constant k0 is set to 1, and the kB is the Boltzmann constant [28]. The d-band center was determined as
Fig. 1a illustrates the supercell models of L10-NiM (M = Mn, Fe, Co, Cu, Zn, Mo) intermetallic compounds, where Ni and M atoms are periodically arranged along the <001> direction. The (111) crystal plane with a lower index of Ni-based alloys is easy to expose on the surface of nanoparticles, which is used to analyze the electrocatalytic HER performance [6,30]. After geometric optimization, the lattice parameters a and c of L10-NiM are shown in Fig. 1b. The a of L10-NiM is unequal to c, which is different from the fcc structure [31]. The a and c of L10-NiM are related to the atomic radius and Ni-M bond length. All the a and c values are around 0.35 nm, falling within the range of 0.33~0.37 nm, which is consistent with the previous calculation results for NiM alloys [6,32]. As the Mn, Fe, and Co atoms show similar atomic radius to Ni, the a-axis is compressed, and the c-axis is stretched when forming the L10-phase. In contrast, both the a-axis and c-axis are stretched in L10-NiCu, L10-NiZn, and L10-NiMo.
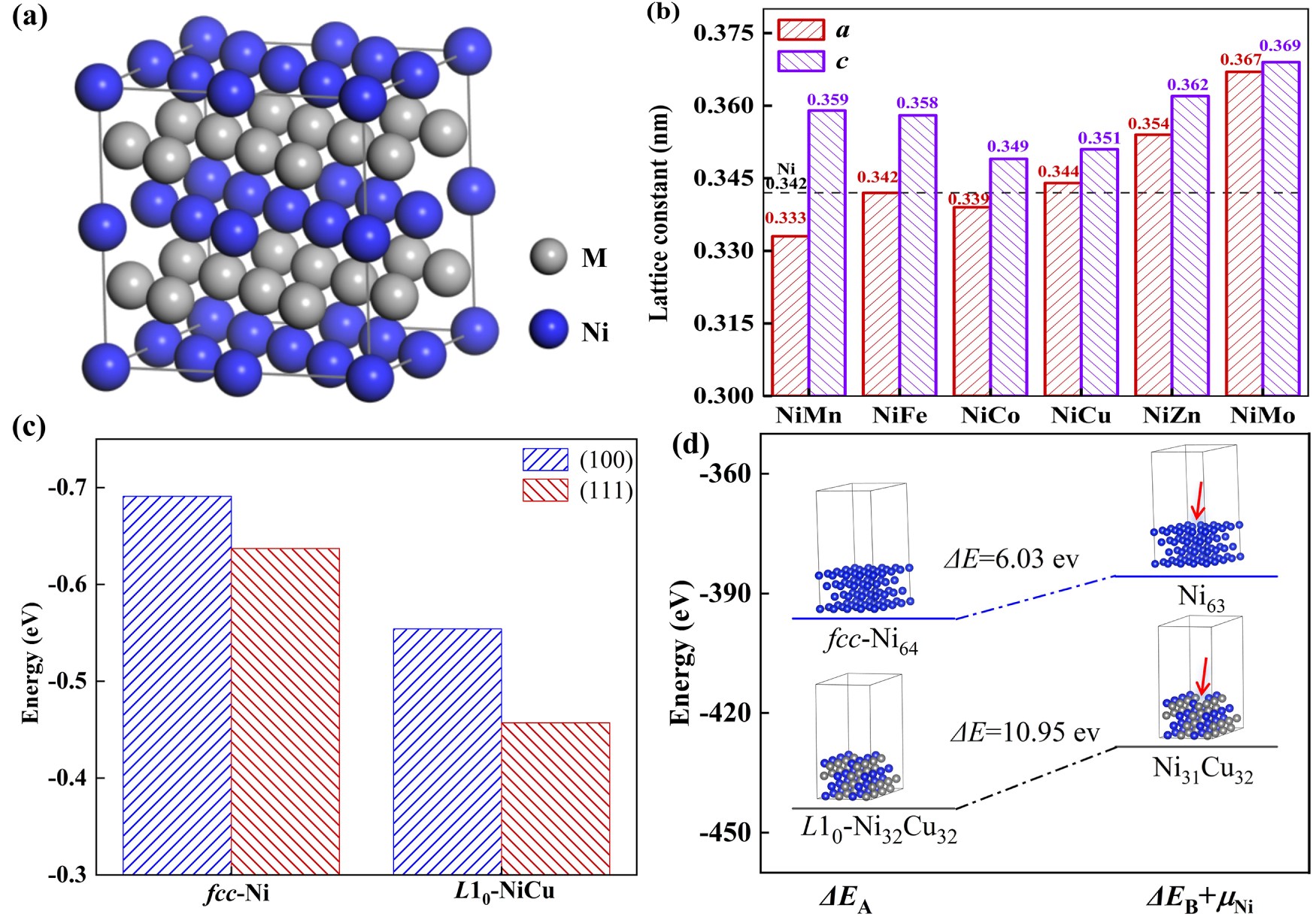
Figure 1: (a) Supercell models and (b) lattice parameters of L10-NiM intermetallic compounds. (c) The H2O dissociation barrier of fcc-Ni and L10-NiCu on both (111) and (100) faces. (d) The energy evolutions of fcc-Ni and L10-NiCu (111) face to dissociate a Ni atom.
In the process of electrocatalytic alkaline HER, the adsorption of H2O on the catalyst surface initiates the reaction, with the dissociation of H2O + e− → OH− + *H (Volmer step) being a rate-determining step [4,5,10]. Then, the generation of H2 follows two typical routes, the Heyrovsky step H2O + e− +*H → OH− + H2, and the Tafel step *H +*H → H2. Fig. 1c shows the H2O dissociation energy of fcc-Ni and L10-NiCu on both (111) and (100) faces. Clearly, the H2O dissociation energy of L10-NiCu is lower than that of fcc-Ni in both faces, and the H2O dissociation energy from (111) faces is lower than that from (100) faces in both alloys. It means the (111) face is a more important electrochemically active center when compared with the (100) face. Fig. 1d shows the energy evolutions of fcc-Ni and L10-NiCo (111) face to dissociate a Ni atom. Clearly, the total energy ΔEA of L10-NiCo is lower than that of fcc-Ni, and the energy required for surface dissociation ΔEd of L10-NiCo is higher than that of fcc-Ni. Both results illustrate that the (111) face of L10-NiCo is more stable and harder to dissociate, revealing a stronger stability in the electrocatalytic HER process.
Fig. 2a illustrates the transition state barrier of L10-NiM intermetallic compounds in the H2O dissociation process. The transition state energy barrier of H2O dissociation of L10-NiFe is about 0.718 eV, slightly higher than that of fcc-Ni (0.706 eV). After the addition of Mn, Co, Cu, Zn, and Mo elements, the H2O dissociation transition state barriers of L10-NiM compounds are decreased. These elements enhance the H2O dissociation in the HER process [9,30]. The H2O dissociation transition state barrier of L10-NiCu is the lowest, which indicates that adding the Cu element can promote the H2O dissociation process more effectively. The releasing energy of L10-NiCu for dissociation of H2O to *H+ and *OH− is the lowest, which also indicates that the L10-NiCu is the most suitable intermetallic compound for electrocatalytic H2O dissociation. Another important step in the electrocatalytic alkaline HER process is the reduction of *H + (Tafel step), which is usually the rate-determining step of electrocatalytic HER in an acidic environment [30–32]. Fig. 2b shows the H-adsorption Gibbs free energy (∆GH*) of the L10-NiM intermetallic compounds. The ∆GH* of L10-NiFe is about −0.736 eV, lower than that of fcc-Ni (−0.708 eV). Upon adding Mn, Co, Cu, Zn, and Mo elements, the ∆GH* of L10-NiM is increased. Similar results can also be found in fcc-NiM alloys, alloying Mo increases the ∆GH* from −0.22 to −0.13 eV [33], and alloying Cu increases the ∆GH* from −0.39 to −0.29 eV [34] or from −0.27 to −0.16 eV [35]. The ∆GH* of L10-NiCu is the highest at −0.524 eV, closer to 0 eV, which means the L10-NiCu is the most suitable intermetallic compound for electrocatalytic H reduction [36,37].
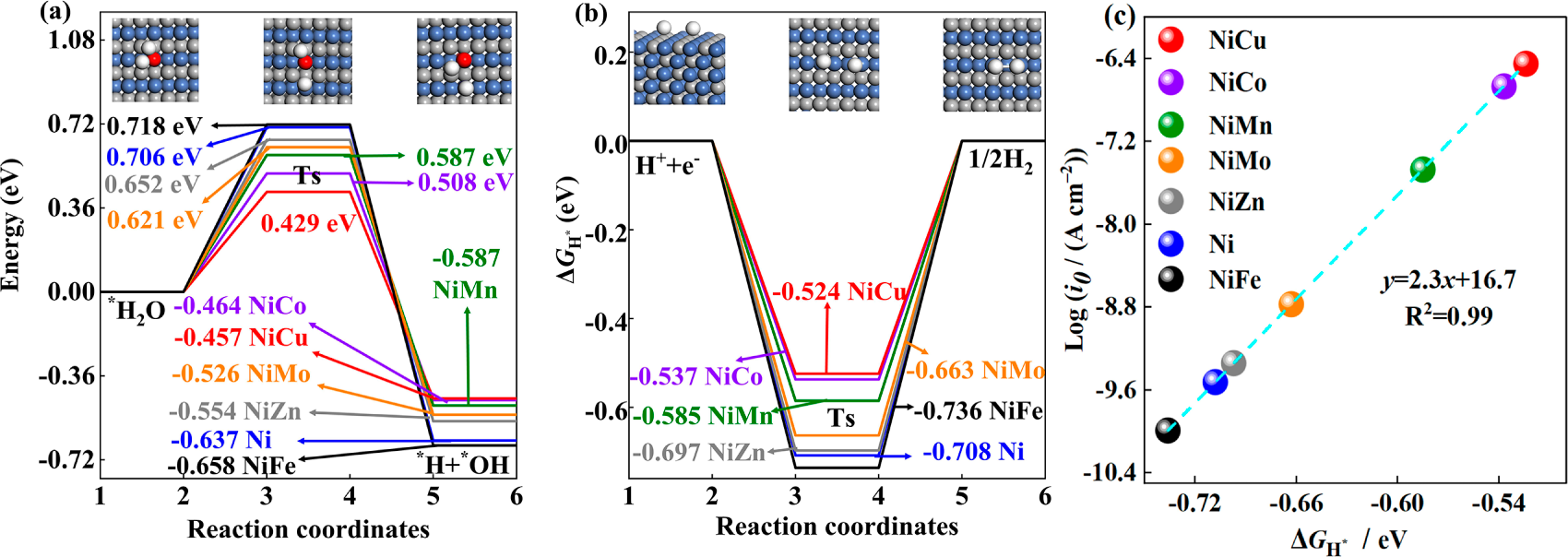
Figure 2: (a) H2O dissociation energy barrier, (b) H-adsorption Gibbs free energy, and (c) scale relationship between ∆GH* and exchange current density of L10-NiM intermetallic compounds.
The scale relationship between ∆GH* and exchange current density is established in Fig. 2c, which can visually compare the electrocatalytic HER performance of L10-NiM intermetallic compounds. The goodness of linear fitting R2 is as high as 0.99, consistent with the “volcanic plot” on the side of ∆GH* < 0. The L10-NiCu presents the highest exchange current density and better electrocatalytic H reduction ability [38]. Indeed, electrocatalytic HER is a complex process that is influenced by various factors such as the electrolysis potential, acid or alkaline environment, and concentration. The H2O dissociation barrier and single-site ∆GH* are effective parameters for evaluating the electrocatalytic HER activity [4,5,10,31]. Combining the electrocatalytic H2O dissociation and H adsorption abilities of L10-NiM intermetallic compounds, it can be concluded that L10-NiCu presents better electrocatalytic alkaline HER performance, followed by L10-NiCo, L10-NiMn, L10-NiMo, and L10-NiZn. The linear scale relationship between ∆GH* and exchange current density further confirms that the L10-NiM intermetallic compounds present promising applications toward alkaline electrolyzers, which provides a reference for developing similar catalytic systems.
Fig. 3 shows the state densities and the d-band centers of the L10-NiM intermetallic compounds after hydrogen evolution. According to the Sabatier principle, the adsorption of intermediates should not be too strong or too weak, which will lead to the enhancement or weakening of the bonding of the outer electrons. Neither of them is conducive to the formation of electron interaction and directional transport between the adsorbate and the catalysts [39]. The state density of fcc-Ni is mainly located below the Fermi level, demonstrating a strong H adsorption ability and confining the electrocatalytic HER performance. The d-band center of fcc-Ni is calculated to be −1.72 eV, which is comparable with other reports [6–8,10]. Alloying with Mn, Cu, and Mo raises the d-band center to the near Fermi level, which will reduce the strong adsorption effect of Ni on proton H+ and provide favorable conditions for the detachment of H-H* from the surface of the catalyst. In addition, except for L10-NiZn and L10-NiFe, electrons gradually shift in the direction of the empty orbitals of Ni atoms, to achieve a charge-energy equilibrium, when the outer d-orbital electrons of the other alloying elements couple with the Ni atoms’ empty orbitals. At this stage, the antibond orbitals will increase, decreasing the surface capacity to adsorb *H [40]. It also indicates that the introduction of Zn increases the adsorption strength of H, so that the center of the d-band of the alloy will move down from −2.48 to −4.53 eV, which is not conducive to the formation of H2.
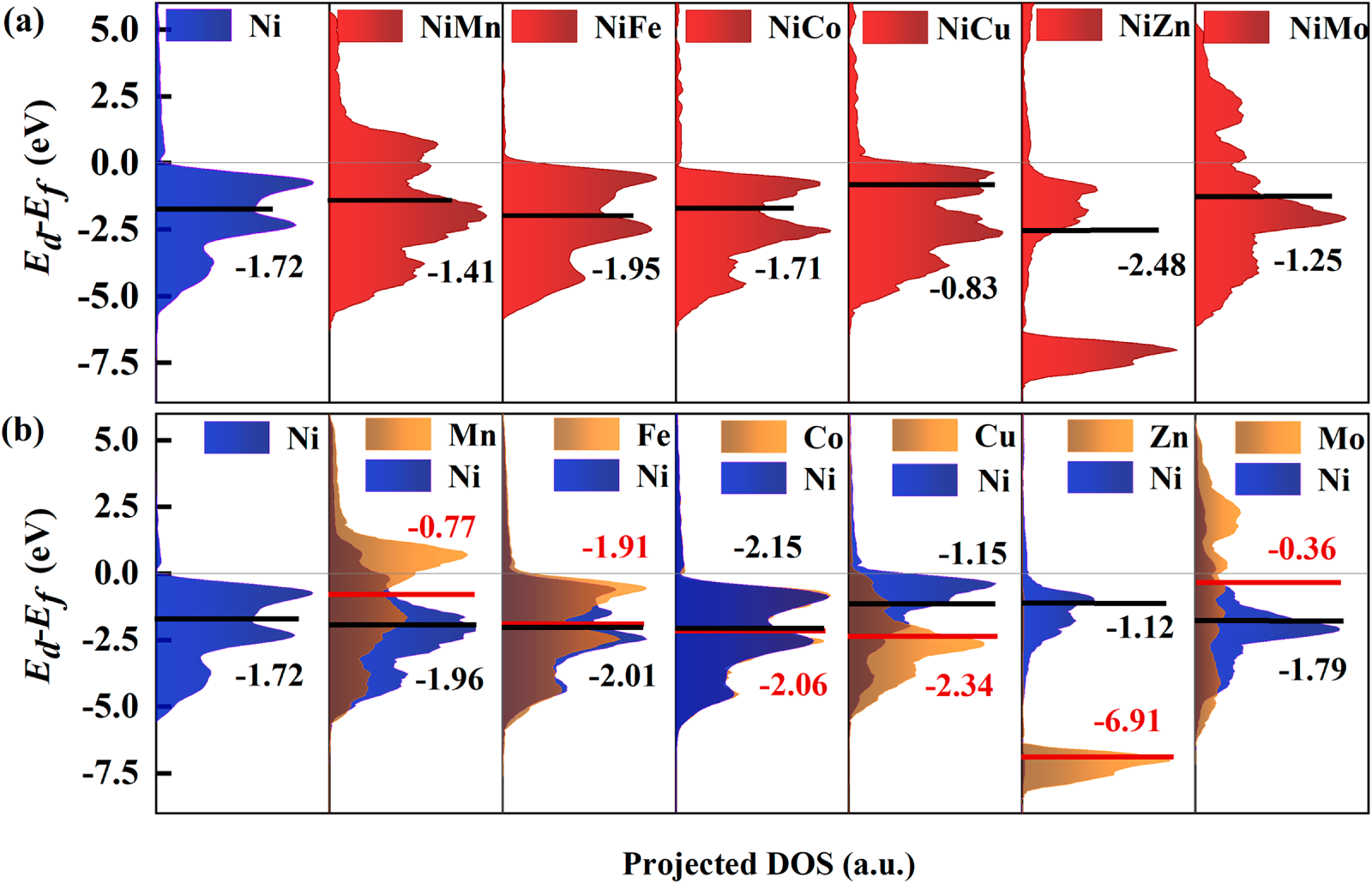
Figure 3: (a) States densities and (b) the d-band centers of L10-NiM intermetallic compounds.
The d orbital states of the various atoms were examined following the adsorption of H (Fig. 3b). The peaks of the 3d orbitals of Ni and Zn atoms do not intersect when both of them are alloyed, suggesting that there is no interaction between the two atoms, and the alloying effect in L10-NiZn is insufficient. Additionally, the 3d orbital of the Zn atom is too poor, indicating that the L10-NiZn catalyst has an insufficient surface adsorption capacity, which may have a substantial effect on surface charge transport [6]. After alloying with Mo and Mn atoms, the peak of the Ni-3d orbital moves in the positive direction across the Fermi level, and some electrons are filled in the antibonding orbital, resulting in stronger adsorption of the catalyst. The analogous electronic configurations of Co, Fe, and Ni atoms lead to equivalent peaks in the Ni-3d, Co-3d, and Fe-3d orbitals, rendering the electronic structures of L10-NiCo and L10-NiFe quite similar compared to that of Ni, thus resulting in a negligible impact on adsorption regulation. The adsorption of catalyst performance reaches the maximum in L10-NiCu, because the Ni-3d orbital becomes flatter and positively shifted, while the Cu-3d orbital is appropriately negatively evacuated (−2.34 eV), the electrons then flow to the bonding orbital. The Mo and Mn atoms positively shift in the center of the d-band of the alloy catalyst, and the adsorption increases, which is not conducive to improving catalytic activity. Only the d-band center of the Cu atom (−2.12 eV) is appropriately negatively displaced, indicating that the t2g-filled electrons are enhanced and the antibond orbital is dragged down (Fig. 4a) [28]. Further analysis of the bonding characteristics can be conducted by observing the band structure of L10-NiCu [41]. The electrocatalytic performance of L10-NiCu alloy was enhanced, and the adsorption strength of the alloy surface to *H was tuned while the alloy structure stabilized itself. Although it is hard to establish a quantitative relationship between the d-band center and adsorption behavior, it can be deduced that the L10-NiCu alloy presents a better d-band for electrocatalytic HER.
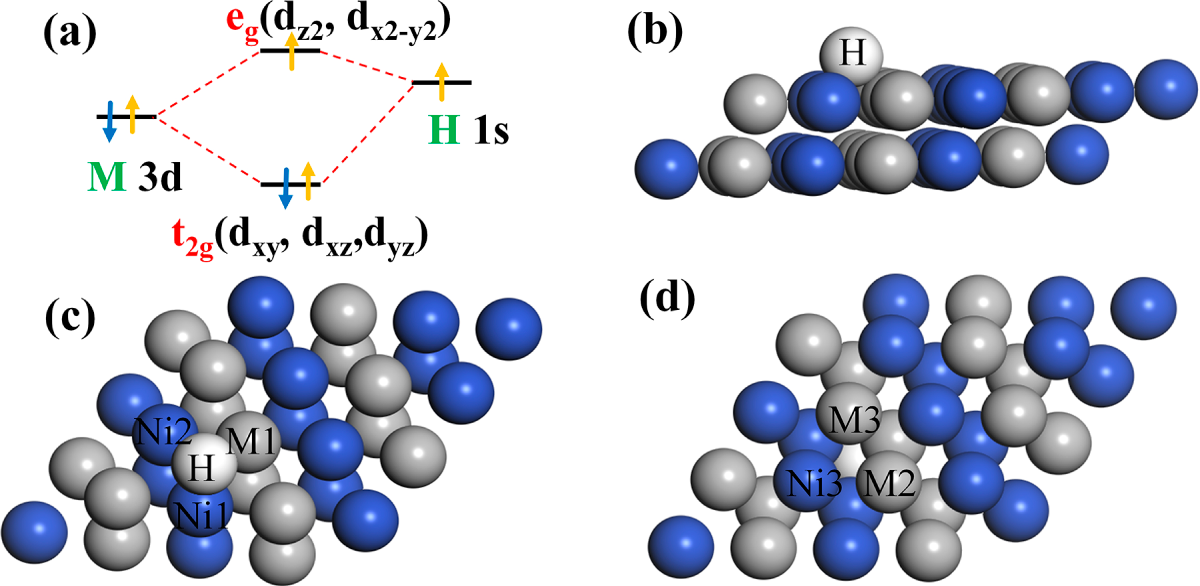
Figure 4: (a) Schematic diagram of orbital coupling. Models of Bader charge atoms (b) H atom, (c) H, Ni1, Ni2 and M1 atoms, (d) Ni3, M2 and M3 atoms.
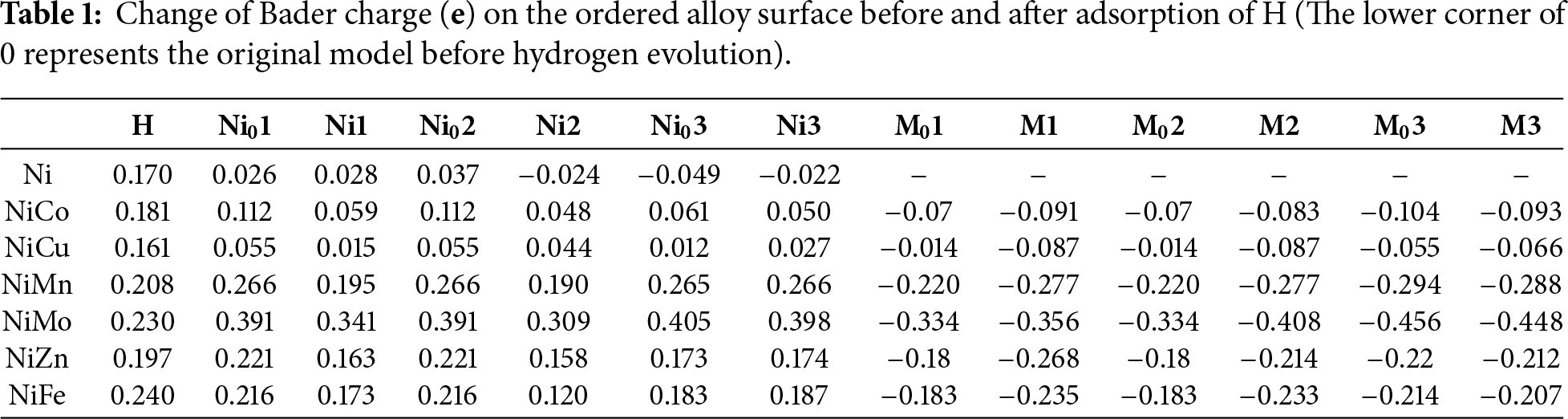
During H dissociation, proton H can interact electronically with multiple adjacent atoms simultaneously, illustrating that the adsorption binding energy of H is influenced by interactions with several atoms [42]. Fig. 5a–g demonstrates the differential charge densities when *H is formed on the surface of the ordered L10-NiM catalyst, respectively, and the H atoms are also adsorbed in the hollow position (located at the center of every three atoms on the surface, and there are no atoms at this position one layer below the site). The charge distribution shows the characteristics of homogeneity and symmetry. Combined with the analysis in Table 1 (the atomic number is shown in Fig. 4), this law can also be proved by the Bader charge before the adsorption of H. When Mo, Mn, and Fe elements are added, the yellow area of the differential charge region is increased, and the accumulated Bader charge number of *H is increased, from 0.17 e to 0.2~0.24 e of Ni. It means the adsorption of H is more stable, with enhanced charge transfer and interaction between H atoms and the catalyst surface, while the strong adsorption of Ni for H remained intact. When Co atoms are added, the yellow area of the differential charge region is all gathered around the Ni atoms, the charge migration degree of Co atoms is low, and the charge amount on H does not decrease. After the addition of Cu element, the yellow region of the differential charge region is attracted by the Cu element, which alleviates the strong adsorption of Ni atoms, and the Bader charge on *H is the smallest of 0.161 e, which promotes the adsorption and charge transfer of H atoms. Although it is hard to establish a quantitative relationship between the Bader charge and energy barriers, it also indicates that the alloy surface could improve the HER activity at this stage.
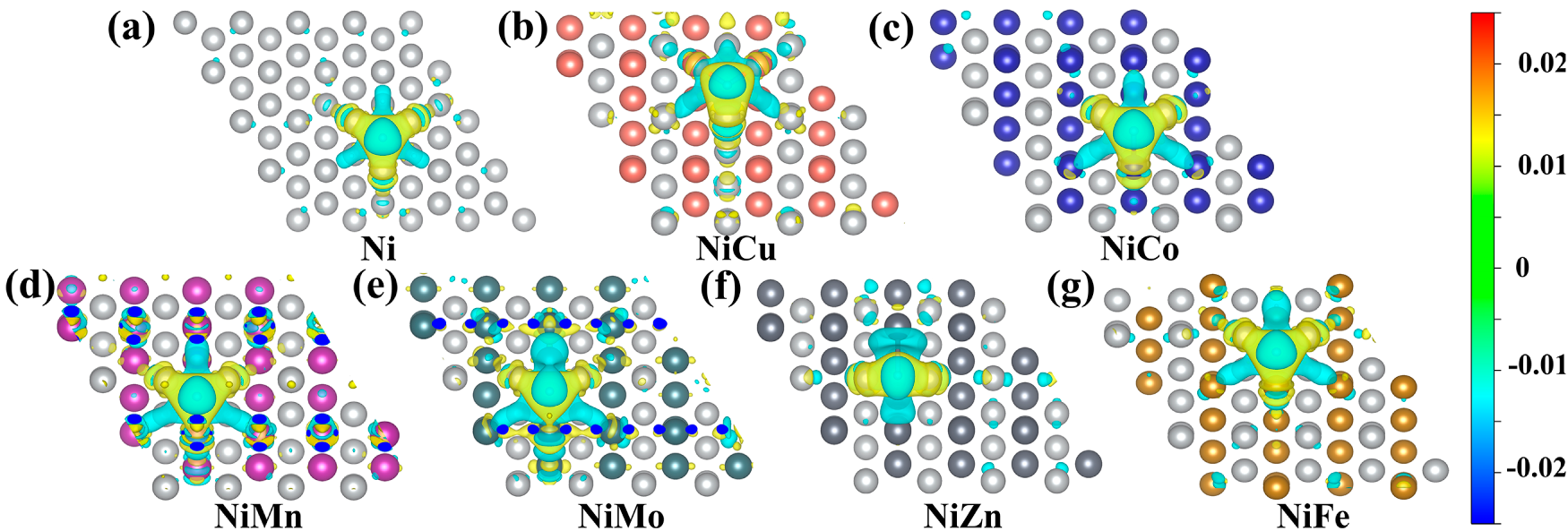
Figure 5: Differential charge density diagram of L10-NiM intermetallic compounds. (a) fcc-Ni, (b) L10-NiCu, (c) L10-NiCo, (d) L10-NiMn, (e) L10-NiMo, (f) L10-NiZn, and (g) L10-NiFe.
The electron location function (ELF) is used to quantitatively illustrate the peripheral electrons of fcc-Ni and L10-NiCu. The atom models and two-dimensional ELFs from (111) faces of fcc-Ni and L10-NiCu are shown in Fig. 6a,b, respectively. Both the ELF values are lower than 0.5, which indicates that the peripheral electrons of the nucleus have a free distribution and typical metal bonding in fcc-Ni and L10-NiCu. The maximum ELF value of L10-NiCu (0.332) is lower than that of fcc-Ni (0.353), which means the freedom degree and delocalization of Ni peripheral electrons are enhanced by the formation of L10-NiCu. This is why the L10-NiCu can exhibit better electrocatalytic performance.
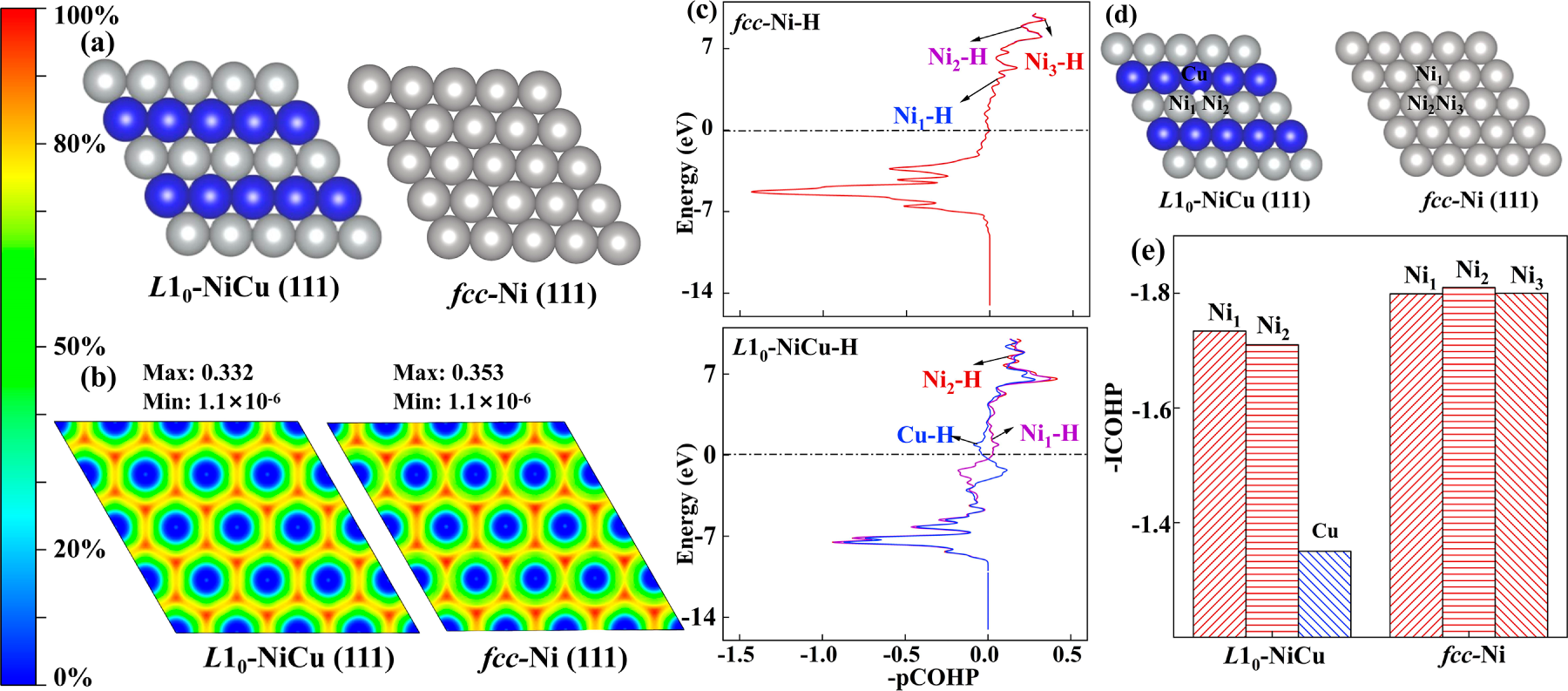
Figure 6: ELF calculation of fcc-Ni and L10-NiCu intermetallic compounds. (a) Models, (b) ELFs. COHP calculation of fcc-Ni and L10-NiCu intermetallic compounds. (c) COHP diagrams, (d) models, and (e) bonding strength.
The crystal orbital Hamilton population (COHP) is used to quantitatively evaluate the bond strengths of *H on the surface of the catalyst. The COHP diagrams and atom models from (111) faces of fcc-Ni and L10-NiCu are shown in Fig. 6c,d, respectively. The COHP diagrams of three Ni atoms in fcc-Ni show no significant changes, and the bonding strength is about −1.80 eV (Fig. 6e). On the contrary, the COHP diagrams of Ni and Cu atoms in L10-NiCu are quite different, the bonding strength of Cu-H (−1.35) is lower than that of Ni-H (−1.72). It means the Cu-H bond is easier to break, which facilitates the electrocatalytic HER process. In addition, the average bonding strength of L10-NiCu is lower than that of fcc-Ni, the strong adsorption effect of Ni is thus weakened by the formation of L10-NiCu. Both quantitative results indicate that the L10-NiCu presents better electrocatalytic HER performance, consistent with the analysis of reaction barriers, hydrogen adsorption free energies, d-band, and Bader charge.
This research evaluates the fundamental hydrogen evolution capacity of L10-NiM intermetallic compounds at a theoretical level. In L10-NiM (M = Mn, Fe, Co, Cu, Zn, Mo) intermetallic compounds, the introduction of alloy elements can effectively modify the position of the electron orbitals, and the ordered L10-structure can better homogenize the electrons on the surface. The homogeneous electron distribution can effectively improve the hydrogen evolution activity, and the good charge distribution will not make the adsorbate strongly adsorbed on the surface of the catalyst. Therefore, there is no polarization of the non-homogeneous charge, which in turn causes strong adsorption of H. The addition of Cu, Co, Mo, and Mn elements in L10-NiM can weaken the binding of adsorbates by lowering the d-band center and inducing the reduction of the antibonding orbital. In this way, the H2O dissociation barrier can be reduced, and the H adsorption Gibbs free energy can be increased to close to 0 eV, thereby improving the activity and stability of electrocatalytic HER. It was found that the L10-NiCu could achieve the highest HER activity, with free energy and hydrolysis energy barriers of −0.524 and 0.49 eV, respectively. It was proven that a suitable d-band center and homogenized Bader charge could effectively optimize the HER process and accelerate the HER kinetics. This study provides an effective scientific foundation for the application of intermetallic compounds in HER and related catalytic reactions.
Acknowledgement: Not applicable.
Funding Statement: This work was financially supported by the National Natural Science Foundation of China (Grant No. 52371179), the China Postdoctoral Science Foundation (Grant No. 2023M732392), the Basic Research Project for Universities of Education Department of Liaoning Province (Grant No. LJ212510147002), the Liaoning Province Special Program for Guiding and Supporting Municipal Science and Technology Development (Grant No. 20240337), and Liaoning Provincial Natural Science Foundation Joint Funding Program (Grant No. 2025-Z0010).
Author Contributions: The authors confirm contribution to the paper as follows: Conceptualization, Chun Wu, Runqing Liu, and Wenli Pei; Methodology, Chun Wu and Zhiqiang Ma; Software, Zhiqiang Ma and Lina Dong; Validation, Xuhui Wang, Changsheng Lou, and Runqing Liu; Writing—original draft preparation, Chun Wu and Zhiqiang Ma; Writing—review and editing, Chun Wu and Runqing Liu; Visualization, Changsheng Lou; Funding acquisition, Chun Wu and Wenli Pei. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: The data that support the findings of this study are available from the corresponding authors.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Turner JM. The matter of a clean energy future. Science. 2022;376(6600):1361. doi:10.1126/science.add5094. [Google Scholar] [CrossRef]
2. Seh ZW, Kibsgaard J, Dickens CF, Chorkendorff I, Nørskov JK, Jaramillo TF. Combining theory and experiment in electrocatalysis: insights into materials design. Science. 2017;355(6321):eaad4998. doi:10.1126/science.aad4998. [Google Scholar] [PubMed] [CrossRef]
3. Feng L, Meng YY, Chen YZ, Zhu ZK, Zou YM, Zhang WB, et al. Configurational-entropy driven formation of single-phase high entropy carbide nanoparticles for efficient hydrogen evolution. Rare Met. 2025;44(10):7360–72. doi:10.1007/s12598-025-03468-8. [Google Scholar] [CrossRef]
4. He S, Zhao M, Zhao P, Ma J, Liu C, Xiao H, et al. Optimizing electrochemical microenvironment of Ni sites by constructing Ni-WO2 heterostructure for promoting electrocatalytic conversion of methanol to formate via direct electrooxidation path. Rare Met. 2025;44(11):8593–608. doi:10.1007/s12598-025-03544-z. [Google Scholar] [CrossRef]
5. Hu C, Lv C, Zeng N, Liu A, Liu Y, Hu L, et al. Recent advances in Ni-based electrocatalysts for hydrogen evolution reaction. Energy Tech. 2023;11(1):2201048. doi:10.1002/ente.202201048. [Google Scholar] [CrossRef]
6. Wang J, Xin S, Xiao Y, Zhang Z, Li Z, Zhang W, et al. Manipulating the water dissociation electrocatalytic sites of bimetallic nickel-based alloys for highly efficient alkaline hydrogen evolution. Angew Chem Int Ed. 2022;61(30):e202202518. doi:10.1002/anie.202202518. [Google Scholar] [PubMed] [CrossRef]
7. Zhou J, Chen X, Guo M, Hu W, Huang B, Yuan D. Enhanced catalytic activity of bimetallic ordered catalysts for nitrogen reduction reaction by perturbation of scaling relations. ACS Catal. 2023;13(4):2190–201. doi:10.1021/acscatal.2c05877. [Google Scholar] [CrossRef]
8. Liu P, Li L, Yu S, Ai S, Long M, Chen D. Effect of transition metal element additions on the mechanical and electronic properties of L10 CoNi alloys. Extreme Mech Lett. 2021;42:101128. doi:10.1016/j.eml.2020.101128. [Google Scholar] [CrossRef]
9. Liu Y, Fu B, Fu F, He Y, Li D. Extension of inducing effect of support coordination on Ni-based ordered alloys catalyst for selective hydrogenation. Chem Eng Sci. 2022;260:117852. doi:10.1016/j.ces.2022.117852. [Google Scholar] [CrossRef]
10. Wu C, Wang X, Huang M, Meng C, Chang L, Xu D, et al. Design and fabrication of intermetallic NiCo electrocatalysts for the alkaline HER. Nanoscale. 2024;16(32):15148–57. doi:10.1039/d4nr02519a. [Google Scholar] [PubMed] [CrossRef]
11. Li J, Xi Z, Pan YT, Spendelow JS, Duchesne PN, Su D, et al. Fe stabilization by intermetallic L10-FePt and Pt catalysis enhancement in L10-FePt/Pt nanoparticles for efficient oxygen reduction reaction in fuel cells. J Am Chem Soc. 2018;140(8):2926–32. doi:10.1021/jacs.7b12829. [Google Scholar] [PubMed] [CrossRef]
12. Liang J, Wan Y, Lv H, Liu X, Lv F, Li S, et al. Metal bond strength regulation enables large-scale synthesis of intermetallic nanocrystals for practical fuel cells. Nat Mater. 2024;23(9):1259–67. doi:10.1038/s41563-024-01901-4. [Google Scholar] [PubMed] [CrossRef]
13. Dong S, Ding D, Wu C, Zhou K, Wang Q, Liu R, et al. Solid-state reaction synthesizing of intermetallic L12-(FeCoZn)Pt3 nanoparticles for efficient methanol electrooxidation. J Alloys Compd. 2025;1012:178416. doi:10.1016/j.jallcom.2024.178416. [Google Scholar] [CrossRef]
14. Wu C, Zhou K, Huang M, Ding D, Ma Z, Liu R, et al. Synthesis of intermetallic L10-Fe40Ni5M5Pt50 (M = Mn, Co, Cu) nanoparticles for electrocatalytic methanol oxidation. J Mater Chem A. 2025;13(6):4257–64. doi:10.1039/d4ta07338j. [Google Scholar] [CrossRef]
15. Wu C, Li Z, Dong S, Zhou K, Ding D, Liu R, et al. Small-size intermetallic FeMnPt nanoparticles electrocatalyst for HER under acidic and alkaline conditions. ACS Appl Nano Mater. 2024;7(7):8093–101. doi:10.1021/acsanm.4c00570.s001. [Google Scholar] [CrossRef]
16. Dong S, Li Z, Wang X, Wu C, Chang L, Liu R, et al. Optimizing Co content in intermetallic L10-FeCoPt nanoparticles to enhance the electrocatalytic performance. J Alloys Compd. 2024;973:172924. doi:10.1016/j.jallcom.2023.172924. [Google Scholar] [CrossRef]
17. Kresse G, Furthmüller J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys Rev B. 1996;54(16):11169–86. doi:10.1103/physrevb.54.11169. [Google Scholar] [PubMed] [CrossRef]
18. Blöchl P. Projector augmented-wave method. Phys Rev B. 1994;50(24):17953–79. doi:10.1103/physrevb.50.17953. [Google Scholar] [PubMed] [CrossRef]
19. Bučko T, Lebègue S, Hafner J, Ángyán JG. Tkatchenko-Scheffler van der Waals correction method with and without self-consistent screening applied to solids. Phys Rev B. 2013;87(6):064110. doi:10.1103/physrevb.87.064110. [Google Scholar] [CrossRef]
20. Monkhorst HJ, Pack JD. Special points for Brillouin-zone integrations. Phys Rev B. 1976;13(12):5188–92. doi:10.1103/physrevb.13.5188. [Google Scholar] [CrossRef]
21. Wang V, Xu N, Liu JC, Tang G, Geng WT. VASPKIT: a user-friendly interface facilitating high-throughput computing and analysis using VASP code. Comput Phys Commun. 2021;267(1):108033. doi:10.1016/j.cpc.2021.108033. [Google Scholar] [CrossRef]
22. Mathew K, Sundararaman R, Letchworth-Weaver K, Arias TA, Hennig RG. Implicit solvation model for density-functional study of nanocrystal surfaces and reaction pathways. J Chem Phys. 2014;140(8):084106. doi:10.1063/1.4865107. [Google Scholar] [PubMed] [CrossRef]
23. Momma K, Izumi F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J Appl Crystallogr. 2011;44(6):1272–6. doi:10.1107/S0021889811038970. [Google Scholar] [CrossRef]
24. Wang HF, Liu ZP. Comprehensive mechanism and structure-sensitivity of ethanol oxidation on platinum: new transition-state searching method for resolving the complex reaction network. J Am Chem Soc. 2008;130(33):10996–1004. doi:10.1021/ja801648h. [Google Scholar] [PubMed] [CrossRef]
25. Liang Z, Luo M, Chen M, Qi X, Liu J, Liu C, et al. Exploring the oxygen electrode bi-functional activity of Ni-N–C-doped graphene systems with N, C co-ordination and OH ligand effects. J Mater Chem A. 2020;8(39):20453–62. doi:10.1039/d0ta06335e. [Google Scholar] [CrossRef]
26. Nørskov JK, Bligaard T, Rossmeisl J, Christensen CH. Towards the computational design of solid catalysts. Nat Chem. 2009;1(1):37–46. doi:10.1038/nchem.121. [Google Scholar] [PubMed] [CrossRef]
27. Valdés Á., Qu ZW, Kroes GJ, Rossmeisl J, Nørskov JK. Oxidation and photo-oxidation of water on TiO2 surface. J Phys Chem C. 2008;112(26):9872–9. doi:10.1021/jp711929d. [Google Scholar] [CrossRef]
28. Nørskov JK, Bligaard T, Logadottir A, Kitchin JR, Chen JG, Pandelov S, et al. Trends in the exchange current for hydrogen evolution. J Electrochem Soc. 2005;152(3):J23. doi:10.1149/1.1856988. [Google Scholar] [CrossRef]
29. Yin WJ. Density functional theory-free descriptor for the practical discovery of perovskite catalysts. Comput Mater Sci. 2021;193:110342. doi:10.1016/j.commatsci.2021.110342. [Google Scholar] [CrossRef]
30. Meng C, Wang X, Li Z, Wu C, Chang L, Liu R, et al. Synthesis of FeCoNiCuPt high-entropy alloy nanoparticle electrocatalysts with various Pt contents by a solid-state reaction method. Mater Adv. 2024;5(2):719–29. doi:10.1039/d3ma00834g. [Google Scholar] [CrossRef]
31. Li D, Zhang B, Li Y, Chen R, Hu S, Ni H. Boosting hydrogen evolution activity in alkaline media with dispersed ruthenium clusters in NiCo-layered double hydroxide. Electrochem Commun. 2019;101:23–7. doi:10.1016/j.elecom.2019.01.014. [Google Scholar] [CrossRef]
32. Mete S, Sengar MS, Dhayal M, Kumar V, Singh SK. Lattice strain-induced electronic effects on a heteroatom-doped nickel alloy catalyst for electrochemical water splitting. J Mater Chem A. 2024;12(46):32371–84. doi:10.1039/d4ta05604c. [Google Scholar] [CrossRef]
33. Song J, Jin YQ, Zhang L, Dong P, Li J, Xie F, et al. Phase-separated Mo-Ni alloy for hydrogen oxidation and evolution reactions with high activity and enhanced stability. Adv Energy Mater. 2021;11(16):2003511. doi:10.1002/aenm.202003511. [Google Scholar] [CrossRef]
34. Li Z, Yu C, Wen Y, Gao Y, Xing X, Wei Z, et al. Mesoporous hollow Cu-Ni alloy nanocage from core-shell Cu@Ni nanocube for efficient hydrogen evolution reaction. ACS Catal. 2019;9(6):5084–95. doi:10.1021/acscatal.8b04814. [Google Scholar] [CrossRef]
35. Wei C, Sun Y, Scherer GG, Fisher AC, Sherburne M, Ager JW, et al. Surface composition dependent ligand effect in tuning the activity of nickel-copper bimetallic electrocatalysts toward hydrogen evolution in alkaline. J Am Chem Soc. 2020;142(17):7765–75. doi:10.1021/jacs.9b12005. [Google Scholar] [PubMed] [CrossRef]
36. Saini S, Halldin Stenlid J, Abild-Pedersen F. Electronic structure factors and the importance of adsorbate effects in chemisorption on surface alloys. npj Comput Mater. 2022;8(1):163. doi:10.1038/s41524-022-00846-z. [Google Scholar] [CrossRef]
37. Li Y, Dong S, Hu X, Hou W, Zhang J, Zhang X, et al. Effect of molybdenum phosphorus-based single/double-atom catalysts on hydrogen evolution reaction: first principles. Int J Hydrogen Energy. 2024;51:957–69. doi:10.1016/j.ijhydene.2023.07.119. [Google Scholar] [CrossRef]
38. Nørskov JK. Theory nof chemisorption and heterogeneous catalysis. Phys B+C. 1984;127(1–3):193–202. doi:10.1016/S0378-4363(84)80030-3. [Google Scholar] [CrossRef]
39. Hu S, Li WX. Sabatier principle of metal-support interaction for design of ultrastable metal nanocatalysts. Science. 2021;374(6573):1360–5. doi:10.1126/science.abi9828. [Google Scholar] [PubMed] [CrossRef]
40. Wang X, Liu X, Fang J, Wang H, Liu X, Wang H, et al. Tuning the apparent hydrogen binding energy to achieve high-performance Ni-based hydrogen oxidation reaction catalyst. Nat Commun. 2024;15(1):1137. doi:10.1038/s41467-024-45370-4. [Google Scholar] [PubMed] [CrossRef]
41. Kaviani S, Piyanzina I, Nedopekin OV, Tayurskii DA. A DFT-D3 investigation on Li, Na, and K decorated C6O6Li6 cluster as a new promising hydrogen storage system. Int J Hydrogen Energy. 2023;48(77):30069–84. doi:10.1016/j.ijhydene.2023.04.179. [Google Scholar] [CrossRef]
42. Qi B, Chang W, Xu Q, Jiang L, An S, Chu JF, et al. Regulating hollow carbon cage supported NiCo alloy nanoparticles for efficient electrocatalytic hydrogen evolution reaction. ACS Appl Mater Interfaces. 2023;15(9):12078–87. doi:10.1021/acsami.3c00385. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools