 Open Access
Open Access
ARTICLE
Bonding Properties of the Graphene/Aluminum Interface with Transition Metal Coating: A First-Principles Study
1 School of Materials Science and Engineering, Shenyang Ligong University, Shenyang, China
2 China Institute of Atomic Energy, Beijing, China
* Corresponding Authors: Tianfu Li. Email: ; Haicheng Liang. Email:
Computers, Materials & Continua 2026, 87(3), 39 https://doi.org/10.32604/cmc.2026.078760
Received 07 January 2026; Accepted 05 March 2026; Issue published 09 April 2026
Abstract
Graphene has excellent mechanical, electrical and optical properties, which make it an ideal reinforcement phase for aluminum matrix composites. However, graphene is easy to agglomerate and has poor wettability with the aluminum matrix, resulting in unsatisfactory effects when added to the aluminum matrix. In this paper, the effects of transition metals (Cu, Ni, Co) on the bonding properties at the graphene/aluminum interface were systematically investigated using first-principles calculations. The computational results reveal significant differences in the effects of various metals and their crystal plane orientations on interface stability and bonding strength. Among Cu, Ni, Co coatings, Co exhibits the most pronounced modification effect. Its most stable Co(110) crystal plane forms an interface with Al(111)/Gr(0001), exhibiting the highest adhesion work of 3.755 J/m2. This trend indicates that the interfacial bonding strength follows the order Co > Ni > Cu. The calculated density of states and differential charge density show that graphene primarily undergoes weak physical adsorption on the Cu(100) plane, with negligible charge transfer, resulting in limited enhancement of interfacial bonding. In contrast, nickel and cobalt modifications introduce strong chemical bonding. Simulated tensile tests reveal that the cobalt-modified interface, characterized by strong chemical bonding, exhibits optimal mechanical properties, achieving a tensile strength of 7.715 GPa and an elongation at break of approximately 21%. Compared to the clean Al(111)/Gr interface (4.329 GPa, 15%), this represents a strength enhancement exceeding 78%. The copper-coated interface, which primarily relies on physical adsorption, shows limited strength improvement (5.493 GPa) and exhibits brittle fracture behavior. Both nickel- and cobalt-coated interfaces demonstrate distinct ductile fracture characteristics.Keywords
Aluminum matrix composites (AMCs) have broad application prospects in the aerospace, electronics, marine, and automotive industries due to their low density, low coefficient of thermal expansion, high specific strength, good ductility, and excellent machinability [1]. Graphene (Gr) is a two-dimensional material composed of a single layer of carbon atoms arranged in a hexagonal lattice. This unique structure endows it with numerous exceptional properties [2]. However, with the rapid advancement of the aerospace, defense, and automotive sectors [3,4], there are increasingly stringent demands on the comprehensive performance of graphene-reinforced aluminum matrix composites. The reinforcing effect of graphene within the aluminum alloy matrix largely depends on its uniform dispersion and interfacial bonding properties with the matrix [5]. Studies have shown that graphene tends to agglomerate in aluminum matrix composites [6–8] and readily forms brittle Al4C3 phases at the interfaces, significantly reducing load-transfer efficiency and degrading material performance [9–11]. In contrast, ideal interfacial bonding can not only effectively suppress graphene agglomeration but also enhance material properties by optimizing stress distribution [12]. Graphene/aluminum interfaces exhibit significant potential in advanced composites and electronics due to their high strength-to-weight ratio and thermal stability. However, the intrinsic weak van der Waals interactions at these interfaces often limit interfacial bonding strength and charge transfer efficiency. Until now, graphene/aluminum composites still face widespread challenges such as weak interfacial bonding and non-uniform distribution of the reinforcing phase, which severely limit improvements in mechanical properties and practical applications [13–15]. Therefore, effective interfacial modification of graphene-reinforced aluminum matrix composites has become a key focus of current research.
Theoretical and computational studies suggest that transition metal atoms can chemisorb onto graphene, forming strong bonds with aluminum substrates. For instance, ab initio calculations indicate that Pt or Ni coating can induce charge redistribution at the interface, leading to enhanced adhesion energy (typically increasing by 30%–50% compared to pristine interfaces) and improved electronic coupling due to orbital hybridization between the transition metal d-states and graphene π-orbitals [16]. However, challenges remain in atomic-scale interactions and their evolution mechanisms within the interfacial micro-region, which is difficult to observe in situ through experimental methods [17]. Therefore, it is essential to employ first-principles calculations to investigate the interfacial bonding characteristics between matrix atoms and reinforcing particles within the composite’s micro-regions, thereby providing a theoretical foundation for regulating the macroscopic properties of the composites.
Numerous researchers have employed first-principles calculations to investigate interfacial bonding characteristics and mechanical responses under tensile loading in materials. Huang et al. [18] systematically examined the influence of Al(100), Al(110), and Al(111) surfaces on the bonding stability of graphene, revealing that the Al(111) surface exhibits the highest interfacial adhesion energy with pristine graphene. Zhang and Wang [19] simulated various metal/graphene interfacial structures to explore the electronic orbital interactions and bonding mechanisms at the interface. Additionally, Huang et al. [20] investigated the effects of different alloying elements at the graphene/aluminum interface, demonstrating that such modifications can enhance interfacial bonding properties. Xie et al. [21] calculated the tensile strength of Mo-doped diamond (111)/Al(Cu)(111) interfaces using first-principles methods and elucidated how interfacial interactions influence tensile performance. These studies provide a crucial theoretical foundation for understanding and optimizing the interfacial behavior of graphene/aluminum composites.
The theoretical understanding of wetting and interfacial bonding at transition metal-coated graphene/aluminum interfaces remains limited. In this work, first-principles calculations based on density functional theory (DFT) are employed to investigate the evolution of interfacial bonding properties and electronic structures at graphene/copper, nickel, and cobalt/aluminum interfaces, as well as the tensile mechanical behavior of the corresponding composite materials. The results aim to provide theoretical insights and guidance for the interface design of graphene-reinforced aluminum matrix composites.
Structural optimization, energy calculations, and electronic structure analyses based on a plane-wave basis within the density functional theory (DFT) framework were performed using the Quantum-ESPRESSO software package [22]. Ultrasoft pseudopotentials [23] were employed, with the exchange-correlation functional implemented in the GGA-PBE form [24]. The DFT-D3 type of vdWforce correction was included in our calculations [25]. K-point sampling in the Brillouin zone utilized a Monkhorst-Pack grid [26]. The energy cutoff for the plane-wave basis was set to 500 eV for structural optimizations of all bulk and surface models. Specific K-point grid settings were as follows: for bulk and surface calculations of aluminum, copper, nickel, and cobalt, 6 × 6 × 6 and 4 × 4 × 2 grids were used, respectively; for graphene bulk and surface calculations, 6 × 6 × 6 and 6 × 6 × 3 grids were applied, respectively. Subsequent interface model studies—including optimal interfacial spacing tests, structural optimization, and interfacial tensile simulations—uniformly employed a 500 eV energy cutoff and a 5 × 5 × 1 K-point grid. The exchange-correlation functional used for interface models was GGA-PBESOL. Self-consistent field convergence was achieved at 1 × 10−5 eV/atom, with atomic forces relaxed to below 0.03 eV/Å, stress errors not exceeding 0.05 GPa, and displacement errors below 0.001 Å.
First, structural optimization was performed on the unit cell models of aluminum, copper, nickel, cobalt, and graphene. The calculated unit cell parameters are compared with literature-reported values in Table 1. As shown, the parameters obtained in this study exhibit strong agreement with the literature, indicating that the computational methods and parameters employed are appropriate. This provides guidance for the subsequent structural optimization calculations of the interface structure in aluminum matrix composites.
To ensure the reliability of subsequent Al/Gr interface models and computational results, two critical preliminary calculations must be performed on the Al substrate before constructing the interface. First, layer number convergence testing determines the minimum number of Al layers required to accurately simulate bulk properties. Second, systematic calculations of surface energies for all low-index crystal planes of Al assess the thermodynamic stability of different surface orientations, thereby providing a theoretical foundation for the rational design of the interface model. The surface energy is calculated using the following expression,
where
First, the surface energies of the Al(100), Al(110), and Al(111) surfaces were calculated using Eq. (1). The surface energies are 0.720 J/m2 for Al(111), 0.938 J/m2 for Al(110), and 0.832 J/m2 for Al(100). Based on these results, the surface energies of the Al crystal planes are clearly ranked as Al(110) > Al(100) > Al(111). This conclusion aligns with previous studies [18], demonstrating the reliability and accuracy of the calculations.
To minimize computational costs, the Al(111) surface was chosen for layer number convergence testing because surfaces with lower surface energies are more stable. A systematic comparison of the surface energies of 3-layer, 5-layer, 7-layer, and 9-layer Al(111) surface models is presented, with the results shown in Fig. 1.
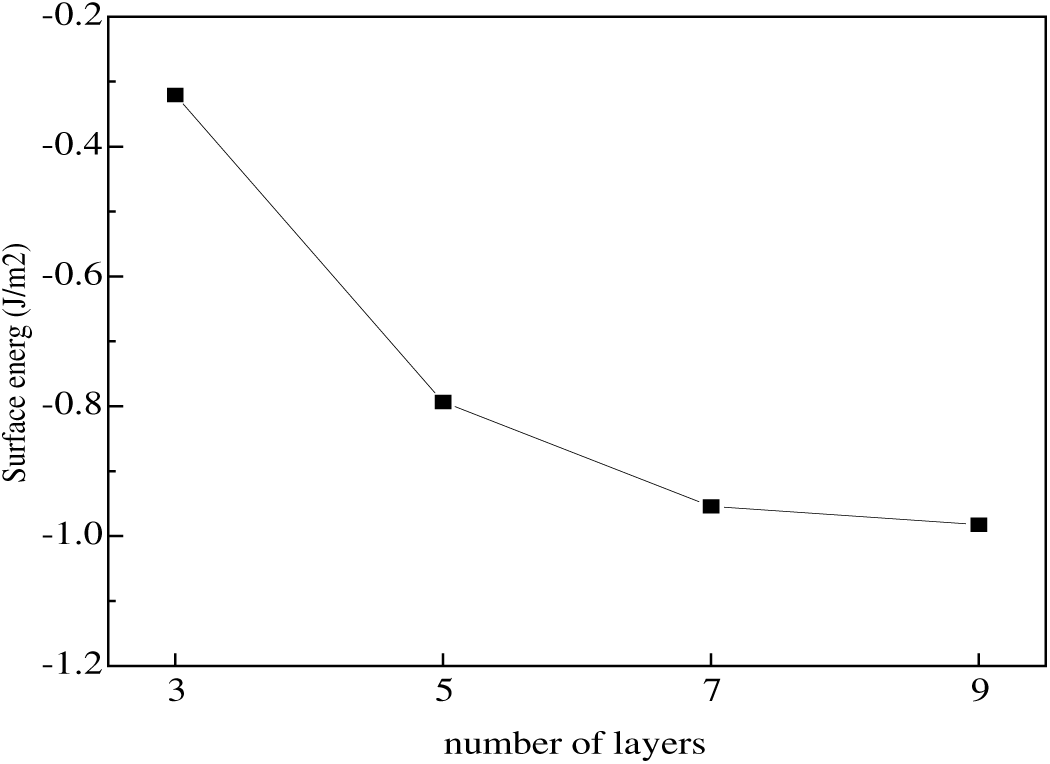
Figure 1: Surface energy of Al(111) with different atomic layers.
The surface energies of 3-, 5-, 7-, and 9-layer Al(111) surface models were systematically compared, with the results shown in Fig. 1. It was found that the surface energy of the Al surface model with a 7-layer slab converged to 1.02 J/m2, a value consistent with other theoretical predictions [36,37] ranging from 0.9 to 1.1 J/m2. This study employs a seven-atom-layer-thick Al(111) film to construct an Al(111)/graphene(0001) interface model. To simulate a metallic bulk environment, the bottom five Al atomic layers are fixed at their ideal lattice positions, while only the two Al atomic layers near the interface (i.e., the region closest to graphene or the overlay layer) are allowed to undergo full geometric relaxation. This constraint effectively reflects the bulk material’s influence on the interface structure while accurately describing atomic rearrangement and energy optimization processes near the interface.
To investigate the bonding mechanism at the aluminum/graphene (Al/Gr) interface, Al/Gr interface models with different crystallographic plane indices were constructed, as shown in Fig. 2. The interface binding energy
where,
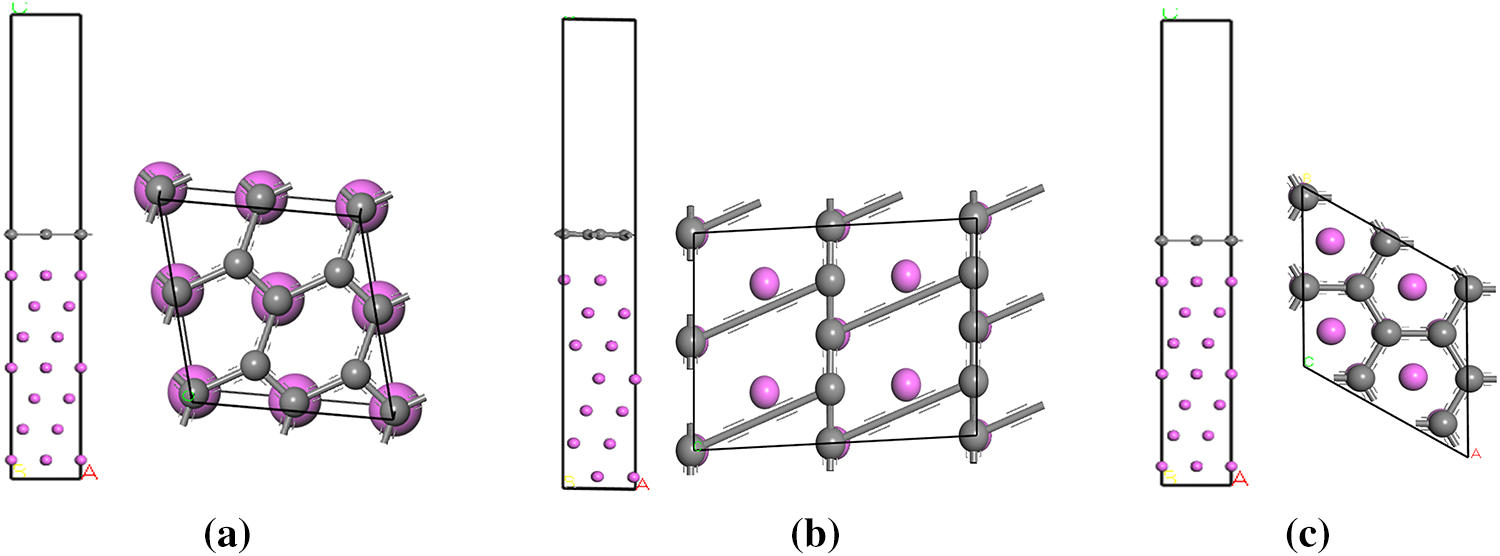
Figure 2: Al/Gr(0001) interface models, (a) Al(100)/Gr(0001), (b) Al(110)/Gr(0001), (c) Al(111)/Gr(0001).
As shown in Fig. 3a, for Al/Gr interface structures with different crystal plane indices, all the interface binding energies (
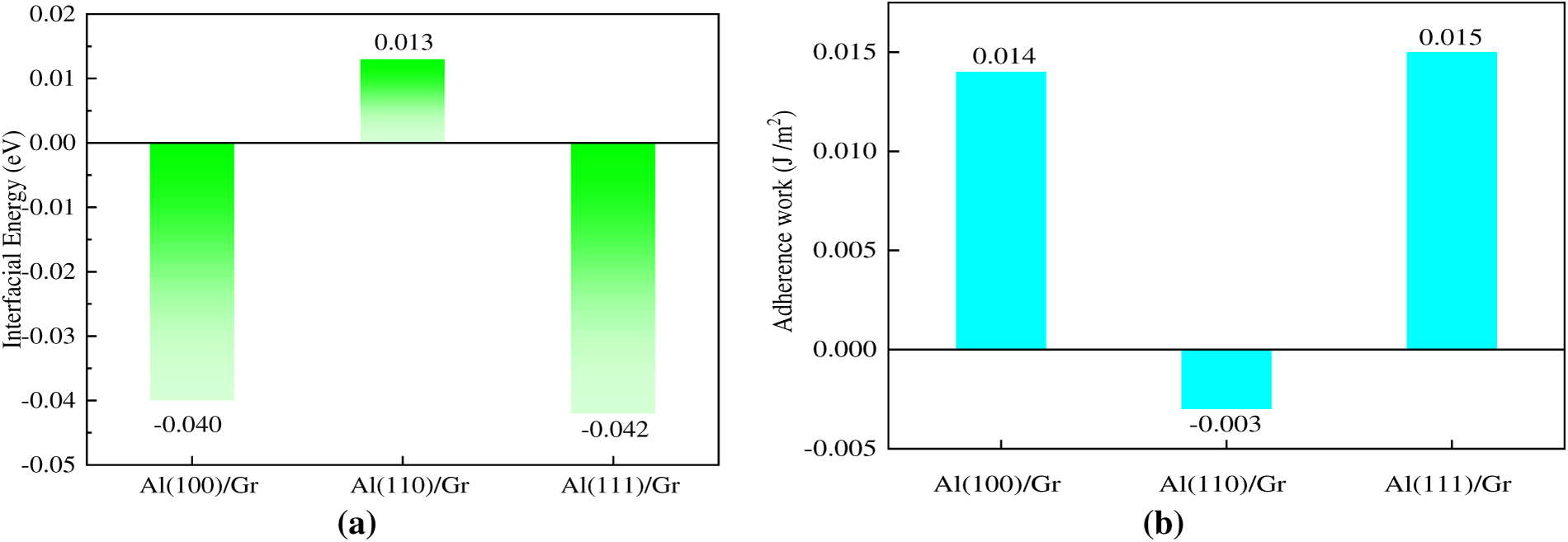
Figure 3: Calculated interfacial energy and adhesion work for Al/Gr composite interfaces with various low-index crystal planes, (a) Interfacial energy, (b) Adhesion work.
Surface energy calculations indicate that the surface energy stabilizes at seven atomic layers of aluminum; therefore, a 7-layer Al surface model was adopted for interface construction. To prevent mirror interactions between layers under periodic boundary conditions, a 15 Å vacuum layer was introduced. During structural optimization, the bottom five layers of Al atoms were fixed to simulate the constraints of the bulk material, while the top two layers of Al atoms were allowed to relax along with the modified metal atoms. This approach more accurately reflects the interfacial bonding behavior. Based on this optimization strategy, the study constructed an Al(111)/X/Gr(0001) interface model by coating a single layer of metal atoms (X = Cu, Ni, Co) onto the seven-layer Al(111) surface and matching it with a single layer of the Gr(0001) surface, as shown in Fig. 4. We choose the in-plane lattice constant of graphene equal to its optimized PBE value, a = 2.46 Å, adapting the lattice constants of the metals accordingly. The graphene honeycomb lattice then matches the triangular lattice of the metal (111) surfaces in the lateral unit cells shown in Fig. 4. The 2 × 2 graphene unit cell was adjusted to the 2 × 2 unit cell of Al, Cu, Ni and Co(111) surfaces. The lattice constant mismatches for Al, Cu, Ni and Co(111) surfaces were −0.81%, −4.06%, −1.22% and −1.83%, respectively. Negative lattice mismatch represents compression of metal lattice.
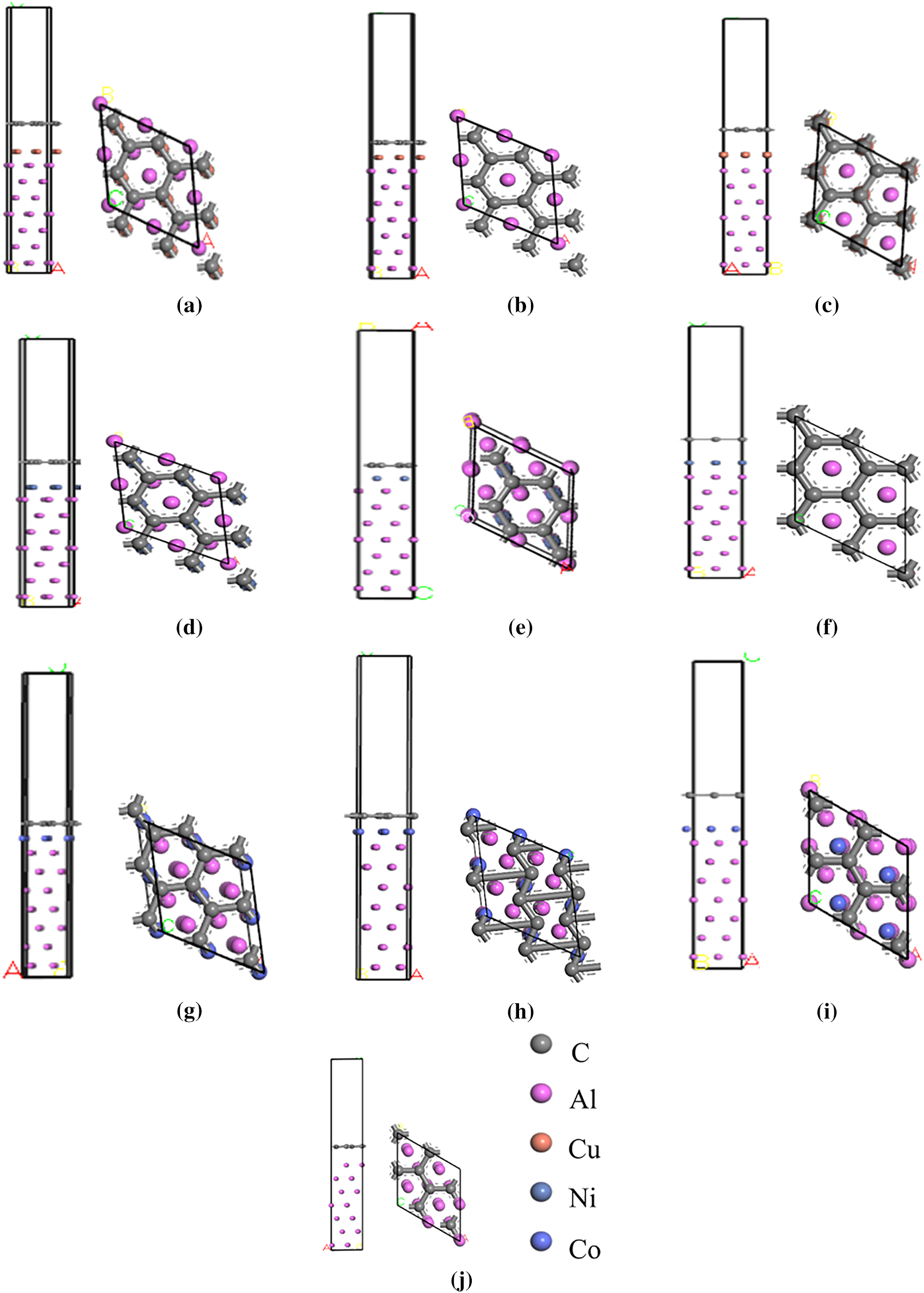
Figure 4: Al(111)/X/Gr(0001) interface model (a) Al(111)/Cu(100)/Gr(0001), (b) Al(111)/Cu(110)/Gr(0001), (c) Al(111)/Cu(111)/Gr(0001), (d) Al(111)/Ni(100)/Gr(0001), (e) Al(111)/Ni(110)/Gr(0001), (f) Al(111)/Ni(111)/Gr(0001), (g) Al(111)/Co(100)/Gr(0001), (h) Al(111)/Co(110)/Gr(0001), (i) Al(111)/Co(111)/Gr(0001), (j) Al(111)/Gr(0001).
For composite structures with two interfaces, the interface adhesion work, Wad, is as follows [28]
where
The calculated
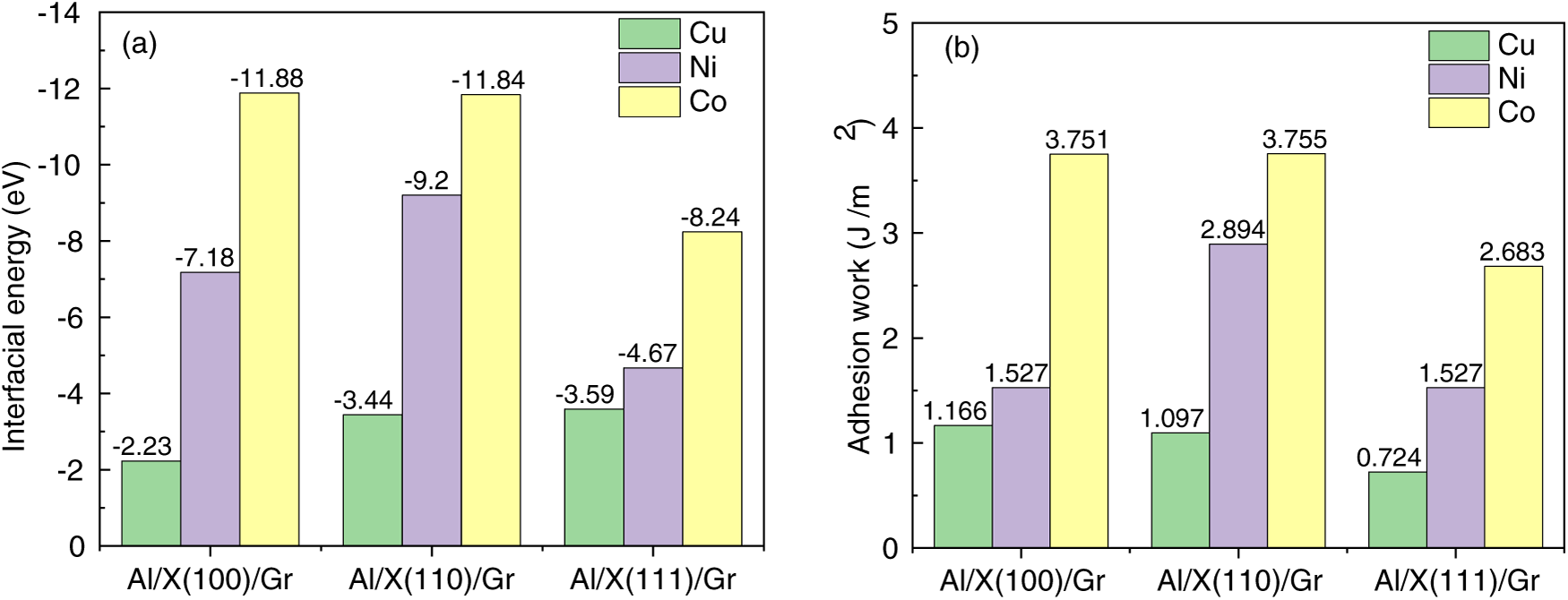
Figure 5: Interface binding energy and adhesion work at the Al(111)/X/Gr(0001) interface, (a) Interfacial energy, (b) Adhesion work.
Comparing the most stable crystal planes across the modified systems reveals significant variations in interfacial bonding strength. The Co-coated Al/Gr interface exhibits the highest stability, followed by Ni, while the Cu-coated Al/Gr interface demonstrates the lowest stability. Regarding adhesion work, the interfacial bonding strengths are ranked as follows: Al/Co(110)/Gr > Al/Ni(110)/Gr > Al/Cu(100)/Gr, indicating that the Co(110) plane provides the most pronounced interface strengthening effect.
To clarify the nature of interatomic bonding, the partial density of states (PDOS) for atoms in the interface layers of the Al(111)/Gr, Al(111)/Cu(100)/Gr, Al(111)/Ni(110)/Gr, and Al(111)/Co(110)/Gr interface models is shown in Fig. 6. For the Al(111)/Gr interface model, no overlapping peaks were observed between the orbital electrons of Al and C atoms (Fig. 6a), indicating no bonding tendency between Al and C atoms.
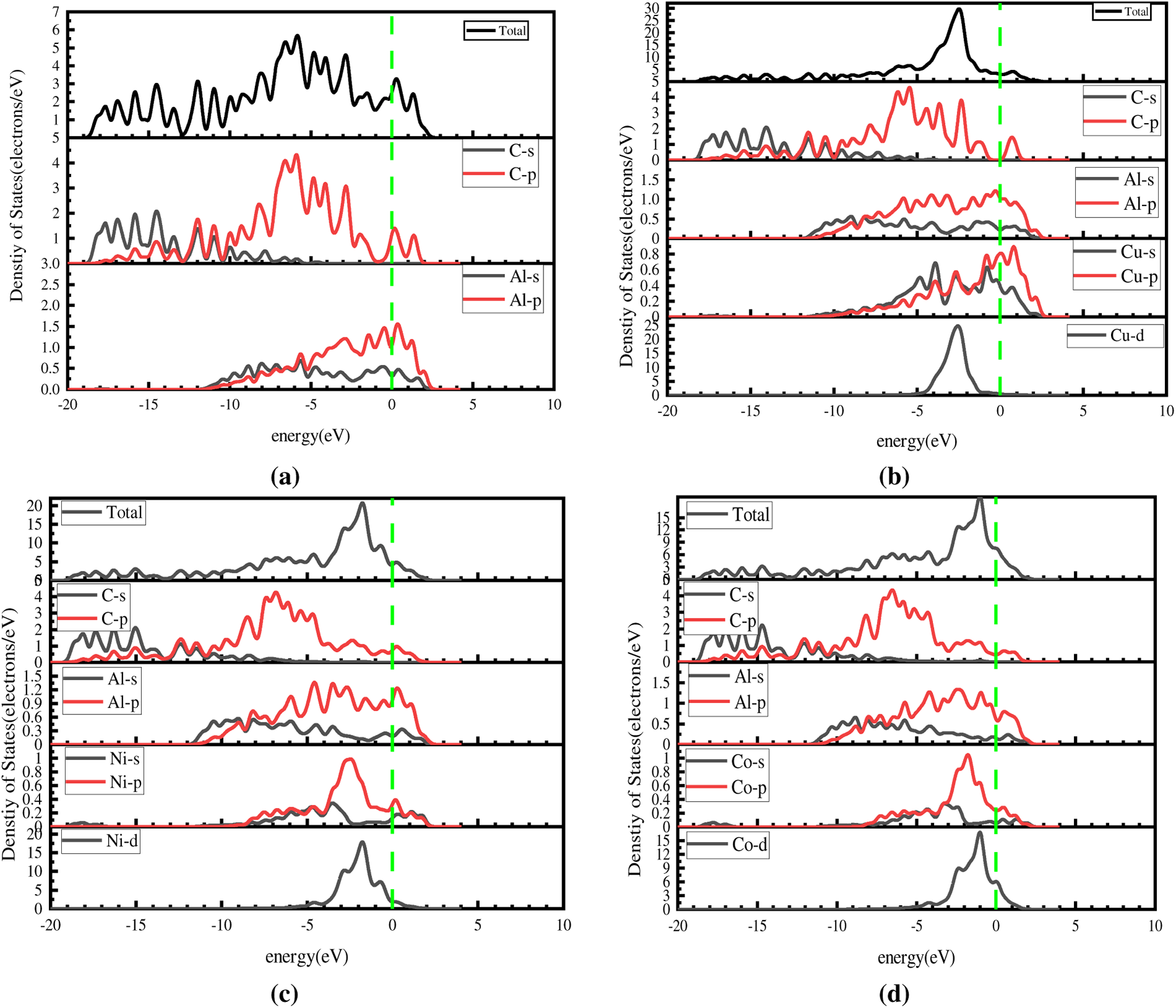
Figure 6: Partial density of states (PDOS) at the interface of the four models, (a) Al(111)/Gr, (b) Al(111)/Cu(100)/Gr, (c) Al(111)/Ni(110)/Gr, (d) Al(111)/Co(110)/Gr.
In the Al(111)/Cu(100)/Gr interface model, no significant interaction was observed between Cu and C atoms, and no substantial shift was detected in the electronic density of states peaks of the C atoms, as shown in Fig. 6b. In contrast, Cu and Al atoms exhibit a large overlap region and multiple resonance peaks between approximately −3 and 0 eV. This corresponds to hybridization among the s, p, and d orbitals of Cu and the s and p orbitals of Al, indicating strong interactions between the two elements and a pronounced tendency toward covalent bond formation.
In the Al/Ni(Co)(110)/Gr interface model (Fig. 6c,d), multiple resonance peaks appear between Ni(Co) atoms and C atoms within the energy range of −5 to −2 eV. The s, p, and d orbitals of Ni(Co) atoms exhibit multiple couplings with the p orbitals of C atoms, indicating a tendency toward covalent bond formation between them. Furthermore, under the influence of the Ni(Co) metal, the density of states near the Fermi level in the graphene layer is no longer zero, indicating that its electronic properties have transformed from nonmetallic to metallic. Simultaneously, numerous resonance peaks exist between Ni(Co) atoms and Al atoms in the energy range from −8 eV to the Fermi level. Extensive hybridization occurs between the s, p, and d orbitals of Ni(Co) and the s and p orbitals of Al, consistent with the formation mechanism of metallic bonds.
3.3 Mulliken Population Analysis and Differential Charge Density
Mulliken population analysis combined with differential charge density analysis provides insights into bonding, charge distribution, and charge transfer between atoms. The results of Mulliken population analysis and differential charge density calculations for the Al(111)/Gr, Al/Cu(100)/Gr, Al/Ni(110)/Gr, and Al/Co(110)/Gr interface models are presented in Table 2 and Fig. 7. Table 2 shows that the Mulliken occupancies of C and Al atoms in the Al(111)/Gr model remain largely unchanged, with net charges of only −0.03 and 0.04 e, respectively. Fig. 7a also reveals no significant charge transfer or accumulation between these atoms, indicating that the physical adsorption interface between Al and C atoms is primarily governed by van der Waals forces.
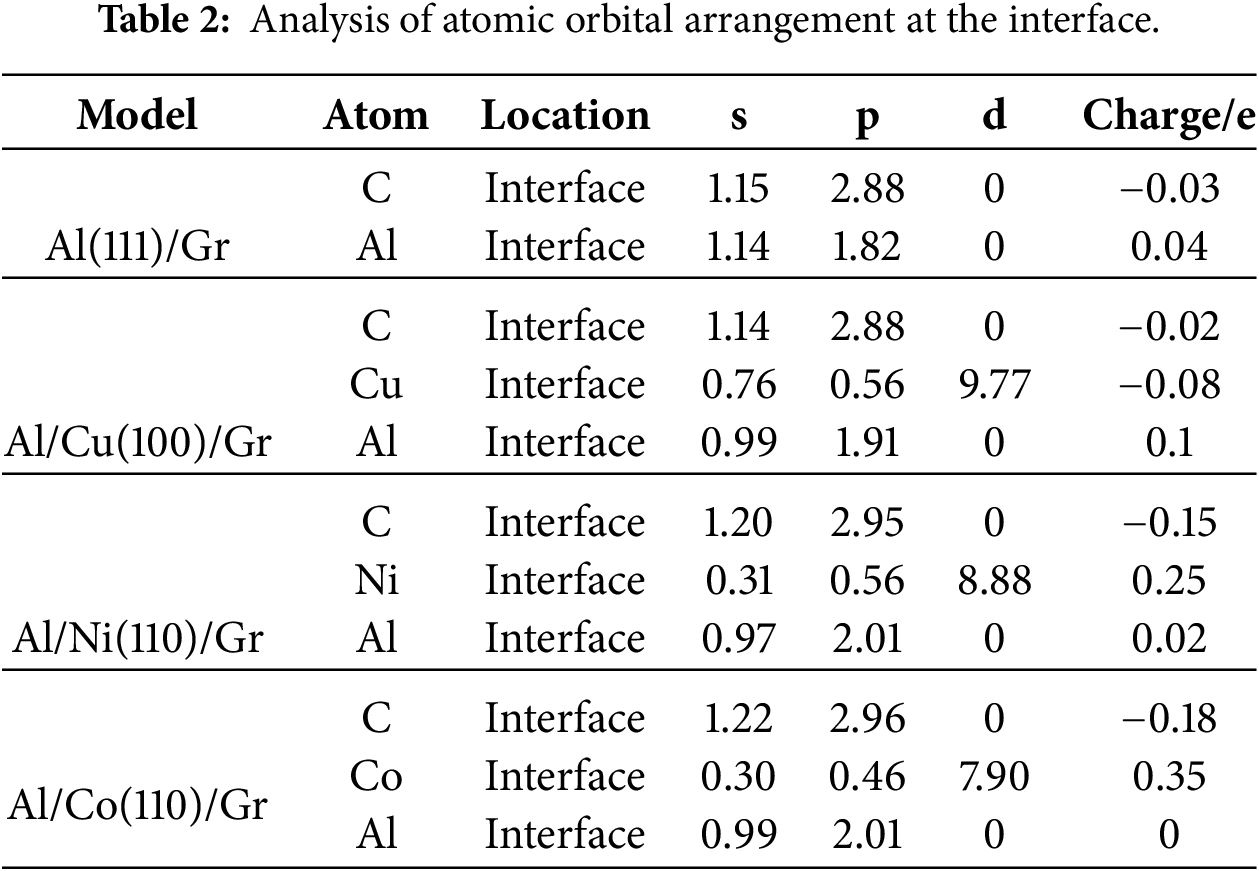
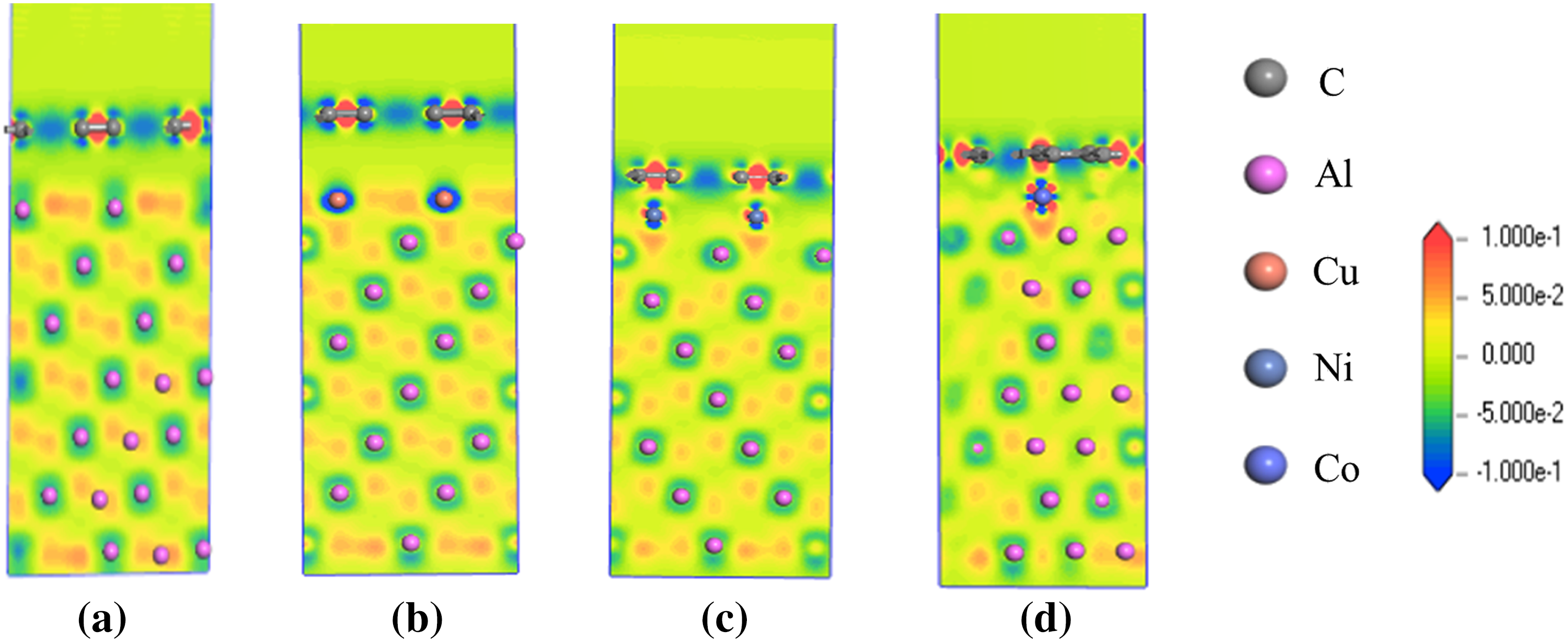
Figure 7: Differential charge density of the three interface structures, (a) Al(111)/Gr, (b) Al(111)/Cu(100)/Gr, (c) Al(111)/Ni(110)/Gr, (d) Al(111)/Co(110)/Gr.
In the Al/Cu(100)/Gr model, charge transfer between Cu and C atoms at the interface is negligible, with net charges of −0.02 and −0.08 e respectively, indicating virtually no effective interaction between them. This result is consistent with the conclusions from the electronic density of states analysis of the corresponding interface region (Fig. 7b). For the Al/Ni(110)/Gr model (Fig. 7c), significant hybridization occurs among the s, p, and d orbitals of the Ni valence electron layer, exhibiting an overall electron-deficient character with a net charge of +0.25 e. Conversely, the C atom gains electrons in both its s and p orbitals, resulting in a net charge of −0.15 e. This indicates electron sharing between Ni atoms at the interface and multiple neighboring C atoms, consistent with a covalent bond formation mechanism. The calculated C–Ni bond occupancy is 0.07, with a bond length of 2.15 Å, confirming that this covalent interaction contributes to enhancing the interfacial bonding strength.
In the Al/Co(110)/Gr model (Fig. 7d), the valence electron layer of the Co atom also undergoes s, p, and d orbital hybridization, with a net charge of +0.35 e. This higher electron loss compared to the Ni atom indicates the formation of stronger covalent bonds between Co and C atoms. Calculations show that the C–Co bond has a 0.03 higher occupation number than the C–Ni bond, while its bond length is 0.1 Å shorter, corresponding to a higher interfacial bonding strength.
As shown in Fig. 7b–d, at the Al/Cu(100)/Gr, Al/Ni(110)/Gr, and Al/Co(110)/Gr interface models, Al atoms in the interfacial region share electrons with Cu, Ni, and Co atoms, respectively. In the Al/Cu(100)/Gr model, the Al atom carries a net charge of 0.1 e and tends to form covalent bonds with Cu atoms. In contrast, for the Al/Ni(110)/Gr and Al/Co(110)/Gr interface models, the net charge of Al atoms is nearly zero, indicating that Al bonds with these metal atoms through metallic bonding. The interfacial strength provided by this type of bonding is essentially consistent across the different models.
Based on the above analysis, to elucidate the mechanisms by which metal modifications such as Cu, Ni, and Co influence the interfacial strength of graphene-reinforced aluminum matrix composites, this study systematically compares different interfacial models. In the Al(111)/Gr model, the bonding potential between the Al(111) surface and graphene is relatively low, primarily manifesting as physical adsorption at the interface, resulting in the weakest bonding strength. This finding indirectly supports Zou’s [39] conclusion regarding SiC/Al composites: during actual high-temperature fabrication, the system readily reacts to form an Al4C3 interphase, which enhances interfacial bonding. However, Al4C3 is a brittle compound prone to cracking under external forces, ultimately leading to complete interfacial separation. For the Al/Cu(100)/Gr interface, the bonding tendency between the Cu(100) plane and graphene is weaker, with the interface still exhibiting physical bonding and limited bonding strength. However, due to chemical bonding between the Cu(100) and Al(111) planes, the formation of Al2Cu during preparation—where the Cu coating encapsulates the Al coating—prevents the formation of the harmful Al4C3 phase, thereby improving the Al/Gr interface bonding [40]. Consequently, the overall interfacial strength is enhanced to some extent. In the Al/Ni(110)/Gr and Al/Co(110)/Gr models, the Al(111) plane readily forms metallurgical bonds with both Ni(110) and Co(110) planes. Simultaneously, chemical bonding occurs between graphene and both metal surfaces, significantly enhancing the overall interfacial strength. Notably, charge transfer between the Co(110) surface and graphene is more pronounced, resulting in stronger Co–C bonds than Ni–C bonds. Consequently, the Al/Co(110)/Gr interface exhibits optimal stability. Since Ni and Co coatings are more stable than the Al layer relative to graphene, carbon readily forms compounds with Ni and Co. This prevents the formation of Al4C3, thereby improving the Al/Gr interface. This can be confirmed by the comparison between Figs. 3a and 5a. The interfacial energy of Gr/Al interface is greatly reduced after modification with metal coatings (Ni, Cu, Co). It shows that the interface stability is significantly improved, which is not conducive to the formation of Al4C3. Moreover, the mechanism of coating modification is also related to the bonding mode at the interface. When the interfaces between Ni and Co coatings and the graphene layer exhibit chemical bonding, they more readily encapsulate the graphene layer, thereby preventing the formation of harmful Al4C3 species. whereas when the Cu coating forms a physical bond with the graphene layer but a chemical bond with the Al layer, it more readily encapsulates the Al layer, thereby eliminating the generation of harmful Al4C3.
The results indicate that the differences in interfacial bonding strength primarily arise from the bonding characteristics and the degree of charge transfer between graphene and various metal surfaces. The interfacial bonding strengths of Cu, Ni, and Co coatings, as well as the Al/graphene interface, also explain the differing modification mechanisms.
3.4 Simulation Stress-Strain Analysis
Based on first-principles calculations, this study initially constructed a fully relaxed interface unit cell model to determine the lowest-energy stable structure. Subsequently, uniaxial tensile stress was applied along the Z-axis, perpendicular to the interface, uniformly stretching the interface in fixed increments of 1% strain per step. The Poisson’s ratio contraction effect parallel to the interface was neglected, and the lateral lattice parameters of the supercell were held constant. The tensile strength obtained by this method corresponds to the theoretical ideal strength of the interface at 0 K. The first abrupt drop in the stress-strain curve indicates interface failure. Subsequent stress fluctuations primarily reflect local debonding and atomic rearrangements within the aluminum metal matrix, no longer representing the true failure mechanism of the original interface and thus having limited physical significance. For computational efficiency and to maintain focus on the problem, the calculation was terminated upon the initial stress drop and the onset of the first plateau. The stress-strain relationship during this process can be calculated using:
where
The tensile stress-strain results for the Al(111)/Gr, Al/Cu(100)/Gr, Al/Ni(110)/Gr, and Al/Co(110)/Gr interfaces are presented in Fig. 8. As shown in Fig. 8, the tensile stress–strain curves of Gr/Al composites with varying metal coating reveal a clear trend: the overall tensile strength of the composites decreases progressively in the following order: Al(111)/Gr < Al/Cu(100)/Gr < Al/Ni(110)/Gr < Al/Co(110)/Gr. This demonstrates that the coating on the Gr/Al interface exerts a pronounced influence on the tensile mechanical performance of Gr/Al composites.
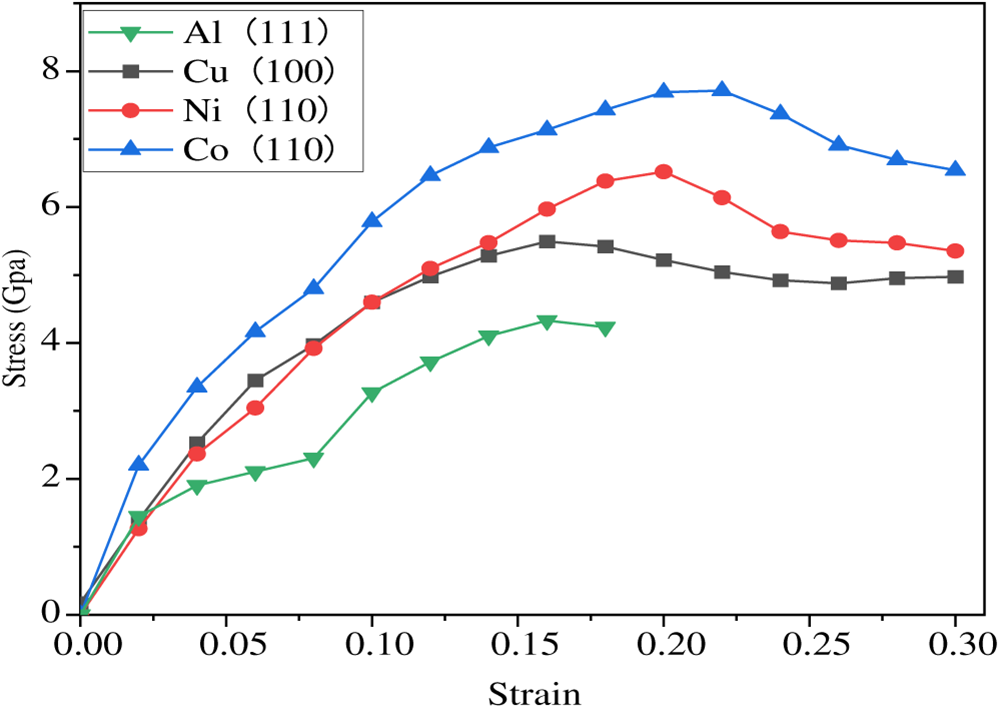
Figure 8: Stress-strain curves of Al(111)/Gr, Al/Cu(100)/Gr, Al/Ni(110)/Gr, and Al/Co(110)/Gr interfaces.
As shown, the Al(111)/Gr interface exhibits the lowest tensile strength of 4.329 GPa, with an elongation at fracture of approximately 15%. The Al/Cu(100)/Gr interface demonstrates an elongation at fracture of about 16%, achieving a tensile strength of 5.493 GPa—a 27% increase over the Al(111)/Gr interface. Following Ni(110) modification, the tensile strength of the Al/Ni(110)/Gr interface rises to 6.251 GPa, with elongation at break increasing to approximately 20%, representing a 44.4% improvement in tensile strength compared to the Al(111)/Gr interface. The Al/Co(110)/Gr interface exhibits the most favorable overall mechanical properties, with tensile strength and elongation at break of 7.715 GPa and 21%, respectively. This corresponds to a 78.2% increase in tensile strength relative to the Al(111)/Gr interface.
Notably, during tensile deformation, different interface models exhibit distinctly varied mechanical responses: the Al(111)/Gr and Al/Cu(100)/Gr interfaces detach at relatively low strains, whereas the Al/Ni(110)/Gr and Al/Co(110)/Gr interfaces demonstrate superior ductility accompanied by significant stress reduction. Analysis of charge density distributions at the moment of fracture for each interface (Fig. 8) offers further insight into their micro-failure mechanisms.
At the Al(111)/Gr interface (Fig. 9a), the stress discontinuity arises from localized unbonding and atomic rearrangement within the aluminum substrate. At this point, the graphene layer exhibits significant wrinkling, indicating a weak bond between aluminum and graphene. This finding is consistent with conclusions drawn from the interface electronic structure analysis. In the Al/Cu(100)/Gr model (Fig. 9b), the copper coating deforms substantially along with the aluminum layer while maintaining a distinct separation from the graphene layer. This observation further supports the earlier conclusion that the Cu coating forms stronger bonds with the Al layer. Although the tensile strength increased markedly, the elongation at break remained unchanged. While the bonding strength between Cu and graphene improved, it remained a physical bond. In summary, these results indicate that when the metal and graphene layers are connected by a physical bond, the fracture behavior at the interface manifests as a distinct brittle fracture in the composite material.
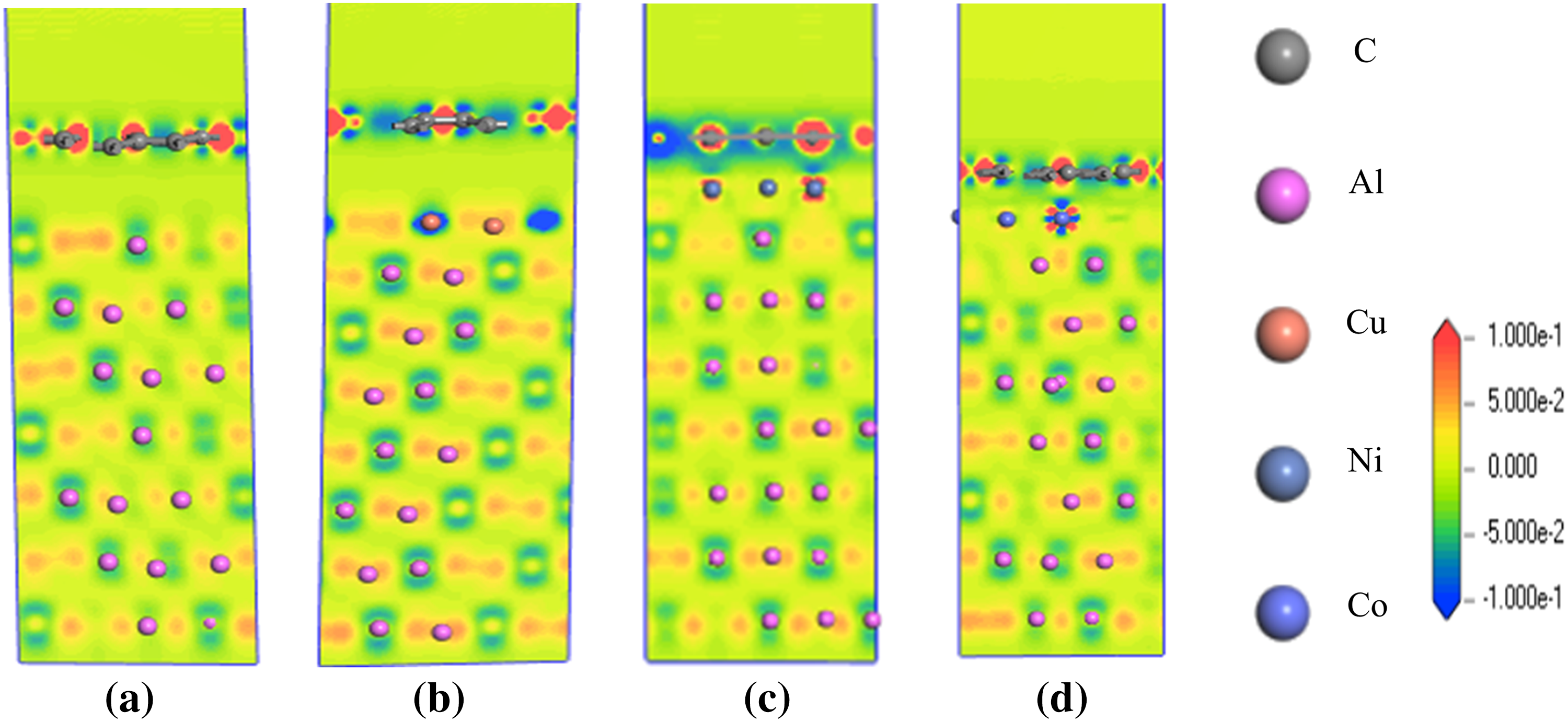
Figure 9: Differential charge density of the interface models during the tensile process, (a) Al(111)/Gr, (b) Al(111)/Cu(100)/Gr, (c) Al(111)/Ni(110)/Gr, (d) Al(111)/Co(110)/Gr.
At the Al/Ni(110)/Gr and Al/Co(110)/Gr interfaces (Fig. 9c,d), no significant electron transfer occurs between the Ni/Co atoms and the C atoms. This indicates that the interfaces are separated and that the Ni/Co coatings did not deform along with the Al layer. This finding further confirms the earlier observation that the Ni/Co coatings bond with the graphene layer. The Al/Ni(110)/Gr and Al/Co(110)/Gr interfaces exhibit not only higher tensile strength but also increased elongation at break. Their stress decay patterns demonstrate pronounced ductile fracture characteristics. This suggests that when the metal layer chemically bonds with the graphene layer, the fracture behavior at the interface manifests as distinct brittle fracture in the composite material.
Based on the preceding analysis, these results clearly demonstrate that the enhanced interfacial bonding between the modified metal and graphene significantly improves the macroscopic tensile strength of the composite material. Furthermore, the nature of the interfacial bonding between the modified metal and graphene influences the fracture mechanism of the composite.
Liu et al. [42] demonstrated that during the hot-pressing process of copper-coated graphite flake-reinforced aluminum matrix composites, the copper layer diffused into the aluminum matrix, forming a transition zone dominated by aluminum-copper intermetallic compounds. This transition zone effectively suppressed the formation of brittle phases such as Al-C-O at the interface, utilizing Al-Cu compounds as bonding bridges to enhance the mechanical and thermal properties of glass fiber/aluminum composites. Concurrently, Liu et al. [43] experimentally fabricated nickel-coated graphene-reinforced aluminum (NGR-Al) composites. They observed a 132% increase in strength compared to the unreinforced aluminum matrix, attributed to optimized interfacial bonding and uniform graphene distribution. There is currently no direct experimental data available for Co coatings, which underscores the forward-looking nature of this prediction and highlights a key direction for future experiments.
In summary, although the idealized model employed in this study predicts theoretical performance limits, the physical mechanisms it reveals and the performance ranking trends across different systems show good qualitative and semi-quantitative agreement with existing experimental reports. This further validates the reliability of the computational model and demonstrates its significant reference value in guiding the interface design and performance optimization of practical aluminum-based composites.
This study systematically investigates the effects of various metal-coated crystal planes on the interfacial interactions and mechanical properties of graphene/aluminum composites using first-principles calculations. The principal conclusions are as follows.
(1) The Cu(100), Ni(110), and Co(110) planes were identified as the optimal surfaces for interface modification. The interfacial bonding strength, ranked according to the calculated work of adhesion, follows this order: Al/Co(110)/Gr (3.755 J/m2) > Al/Ni(110)/Gr (2.894 J/m2) > Al/Cu(100)/Gr (1.097 J/m2). This indicates that the Co(110) coating layer provides the most significant interfacial strengthening effect.
(2) From the bonding mechanism perspective, the Al(111) surface exhibits chemical bonding with the Cu metal surface, while forming metallurgical bonding with both Ni and Co metal surfaces. The difference in overall interface stability primarily stems from the nature of interactions between graphene and metal surfaces: Gr(0001)/Cu(100) exhibits weak physical adsorption, whereas strong chemical bonding forms with Ni(110) and Co(110). The net charge of Co atoms in the graphene/Co(110) interface is more than that of Ni atoms in graphene/Ni(110) interface, which resulting in the formation of stronger covalent bonds between Co and C atoms than between Ni and C atoms.
(3) Regarding mechanical performance, the clean Al(111)/Gr interface exhibited the lowest tensile strength of 4.329 GPa, with a fracture elongation of approximately 15%. The tensile strength increased to 5.493 GPa (16% elongation) for the Al/Cu(100)/Gr interface, 6.251 GPa (20% elongation) for the Al/Ni(110)/Gr interface, and reached an optimum of 7.715 GPa (21% elongation) for the Al/Co(110)/Gr interface. These results consistently demonstrate that enhancing the chemical bonding between the coating layer and graphene directly correlates with improved macroscopic mechanical properties of the composite.
In summary, selecting appropriate crystallographic planes and introducing metal coating layers that can form chemical bonds with graphene significantly enhance both the interfacial bonding strength and the mechanical properties of graphene/aluminum composites. Among the systems studied, the Co(110) coating layer was the most effective in improving interfacial strength.
Acknowledgement: Not applicable.
Funding Statement: This research was funded by Future Industry Frontier Technology Project in Liaoning Province in 2025 (2025JH2/101330141), Key Research and Development Plan Projects of Liaoning Province in 2025 (2025JH2/101800416), and Fundamental Research Project of China National Nuclear Corporation (No. FK010261123429).
Author Contributions: The authors confirm contribution to the paper as follows: Conceptualization: Xiaoming Du and Tianfu Li; Methodology: Tianfu Li, Gaohan Liao and Jiahui Guo; Formal analysis: Gaohan Liao, Jiahui Guo and Xiaoming Du; Writing (reviewing and editing): Xiaoming Du, Gaohan Liao, Jiahui Guo and Haicheng Liang; Project administration: Xiaoming Du, Haicheng Liang and Tianfu Li. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: The authors confirm that the data supporting the findings of this study are available within the article.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Sun Y, Zhang W, Zhou Q, Liu M, Hu W. A coupled micromechanics and elastoplastic phase field method for fracture in graphene/metal composites. Thin Walled Struct. 2025;208(2):112826. doi:10.1016/j.tws.2024.112826. [Google Scholar] [CrossRef]
2. Zhou W, Chen C, Mikulova P, Dong M, Fan Y, Kikuchi K, et al. Thermal expansion behaviors of few-layered graphene-reinforced Al matrix composites. J Alloys Compd. 2019;792:988–93. doi:10.1016/j.jallcom.2019.04.140. [Google Scholar] [CrossRef]
3. Boppana SB, Dayanand S, Anil Kumar M, Kumar V, Aravinda T. Synthesis and characterization of nano graphene and ZrO2 reinforced Al6061 metal matrix composites. J Mater Res Technol. 2020;9(4):7354–62. doi:10.1016/j.jmrt.2020.05.013. [Google Scholar] [CrossRef]
4. Zhou J, Shen J, Yue W, Liu Y, Chen Z. Molecular dynamics simulation of reinforcement mechanism of graphene/aluminum composites and microstructure evolution. J Mater Res Technol. 2023;23:2147–59. doi:10.1016/j.jmrt.2023.01.161. [Google Scholar] [CrossRef]
5. Sadhu KK, Mandal N, Sahoo RR. SiC/graphene reinforced aluminum metal matrix composites prepared by powder metallurgy: a review. J Manuf Process. 2023;91(57):10–43. doi:10.1016/j.jmapro.2023.02.026. [Google Scholar] [CrossRef]
6. Smyrak B, Gniełczyk M. Study of the effect of graphene content on the electrical and mechanical properties of aluminium-graphene composites. Materials. 2025;18(3):590. doi:10.3390/ma18030590. [Google Scholar] [PubMed] [CrossRef]
7. Liao GH, Du XM. First-principles computational study of the Cu-modified graphene/aluminum interface. J Shenyang Ligong Univ. 2025;44(6):49–56. (In Chinese). [Google Scholar]
8. Sun Y, Zhang W, Hu W, Liu M. Tailoring the strength and ductility of graphene/metal composites with percolation network. Int J Solids Struct. 2024;286-287:112533. doi:10.1016/j.ijsolstr.2023.112533. [Google Scholar] [CrossRef]
9. Su J, Teng J. Recent progress in graphene-reinforced aluminum matrix composites. Front Mater Sci. 2021;15(1):79–97. doi:10.1007/s11706-021-0541-0. [Google Scholar] [CrossRef]
10. Wakhi Anuar NFB, Salleh MS, Omar MZ, Zamri WFHW, Md Ali A, Samat S. Wear properties of graphene-reinforced aluminium metal matrix composite: a review. Rev Adv Mater Sci. 2023;62(1):20220326. doi:10.1515/rams-2022-0326. [Google Scholar] [CrossRef]
11. Zhu P, Zhang Q, Gou HS, Wang PP, Shao PZ, Kobayashi E, et al. First-principles calculation of diamond/Al interface properties and study of interface reaction. Acta Phys Sin. 2021;70(17):257–67. (In Chinese). doi:10.7498/aps.70.20210341. [Google Scholar] [CrossRef]
12. Hu Z, Tong G, Lin D, Nian Q, Shao J, Hu Y, et al. Laser sintered graphene nickel nanocomposites. J Mater Process Technol. 2016;231:143–50. doi:10.1016/j.jmatprotec.2015.12.022. [Google Scholar] [CrossRef]
13. Li Y, Du XM, Liu FG. Microstructure and mechanical properties of graphene-reinforced 6061 aluminum matrix composites. J Shenyang Ligong Univ. 2023;42(2):49–55. (In Chinese). doi:10.15251/djnb.2020.152.407. [Google Scholar] [CrossRef]
14. Luo Y, Huang Y, Wang W, Yu S, Chen Q. The influence of interface products on the mechanical and electrical properties of graphene aluminum composites. Surf Interfaces. 2024;46:104164. doi:10.1016/j.surfin.2024.104164. [Google Scholar] [CrossRef]
15. Arsha AG, Ghaderi O, Rajan TPD, Rohatgi PK. Solidification processing of reduced graphene oxide dispersed aluminum composites by squeeze casting. Int J Met. 2025;19(2):664–73. doi:10.1007/s40962-024-01348-y. [Google Scholar] [CrossRef]
16. Benedikt PK, Elizabeth HS, Lukas R, Griffin MR, Samuel JH, Spencer JC, et al. Enhanced bonding of pentagon-heptagon defects in graphene to metal surfaces: insights from the adsorption of azulene and naphthalene to Pt(111). Chem Mater. 2020;32(3):1041–53. doi:10.1021/acs.chemmater.9b03744. [Google Scholar] [CrossRef]
17. Wang Y, Li M, Peng P, Gao H, Wang J, Sun B. Preferred orientation at the Al/graphene interface: first-principles calculations and experimental observation. J Alloys Compd. 2022;900:163304. doi:10.1016/j.jallcom.2021.163304. [Google Scholar] [CrossRef]
18. Huang J, Liu Y, Lai Z, Hu J, Zhou F, Zhu J. A systematic study of interface properties and fracture behavior of graphene/aluminum: insights from a first-principles study. Vacuum. 2022;204:111346. doi:10.1016/j.vacuum.2022.111346. [Google Scholar] [CrossRef]
19. Zhang X, Wang S. Interfacial strengthening of graphene/aluminum composites through point defects: a first-principles study. Nanomaterials. 2021;11(3):738. doi:10.3390/nano11030738. [Google Scholar] [PubMed] [CrossRef]
20. Huang J, Wang K, Li M, Cheng Y, Lai Z, Hu J, et al. Influence of alloy atoms on the electronic structure and interfacial properties of graphene/aluminum composites: theoretical calculation and experimental verification. Vacuum. 2023;215(11):112253. doi:10.1016/j.vacuum.2023.112253. [Google Scholar] [CrossRef]
21. Xie H, Chen Y, Zhang T, Zhao N, Shi C, He C, et al. Adhesion, bonding and mechanical properties of Mo doped diamond/Al(Cu) interfaces: a first principles study. Appl Surf Sci. 2020;527:146817. doi:10.1016/j.apsusc.2020.146817. [Google Scholar] [CrossRef]
22. Giannozzi P, Andreussi O, Brumme T, Bunau O, Buongiorno Nardelli M, Calandra M, et al. Advanced capabilities for materials modelling with Quantum ESPRESSO. J Phys Condens Matter. 2017;29(46):465901. doi:10.1088/1361-648X/aa8f79. [Google Scholar] [PubMed] [CrossRef]
23. Vanderbilt D, Resta R. Chapter 5 quantum electrostatics of insulators: polarization, wannier functions, and electric fields. In: Conceptual foundations of materials—a standard model for ground- and excited-state properties. Amsterdam, The Netherlands: Elsevier; 2006. p. 139–63. doi:10.1016/s1572-0934(06)02005-1. [Google Scholar] [CrossRef]
24. Engel E, Chevary JA, MacDonald LD, Vosko SH. Asymptotic properties of the exchange energy density and the exchange potential of finite systems: relevance for generalized gradient approximations. Z Für Phys D at Mol Clust. 1992;23(1):7–14. doi:10.1007/BF01436696. [Google Scholar] [CrossRef]
25. Johnson ER, Becke AD. A post-Hartree-Fock model of intermolecular interactions. J Chem Phys. 2005;123(2):24101. doi:10.1063/1.1949201. [Google Scholar] [PubMed] [CrossRef]
26. Hilliker PR, Hakim TM. Dynamic contribution of the Brillouin zone center to the floating transition of adsorbed monolayers. J Chem Phys. 1990;93(3):2080–1. doi:10.1063/1.459085. [Google Scholar] [CrossRef]
27. Wang W, Cui C, Xia F, Xu W, Gang T, Chen L. A first principles investigation into the mechanical properties and the strengthening mechanism of the graphene/aluminum interface structure. Sci Rep. 2025;15(1):38571. doi:10.1038/s41598-025-22443-y. [Google Scholar] [PubMed] [CrossRef]
28. Bourbia A, Draissia M, Bedboudi H, Boulkhessaim S, Debili MY. X-ray and optical crystallographic parameters investigations of high frequency induction melted Al-(alpha-Al2O3) alloys. J Xray Sci Technol. 2010;18(2):201–19. doi:10.3233/XST-2010-0245. [Google Scholar] [PubMed] [CrossRef]
29. Hu Z, Wu Z, Luo S, Wang X, Nian Q, Chen Y, et al. Large scale production of graphene aluminum composites by stir casting: process, microstructure and properties. J Mater Res Technol. 2023;27:681–91. doi:10.1016/j.jmrt.2023.09.298. [Google Scholar] [CrossRef]
30. Novoselov KS, Geim AK, Morozov SV, Jiang D, Zhang Y, Dubonos SV, et al. Electric field effect in atomically thin carbon films. Science. 2004;306(5696):666–9. doi:10.1126/science.1102896. [Google Scholar] [PubMed] [CrossRef]
31. Chen W, Zheng H, Fan L, Li J, Ding Z, Guo X, et al. Graphene/copper composite films: interface regulation for enhanced electrical performance. Mater Charact. 2024;210:113790. doi:10.1016/j.matchar.2024.113790. [Google Scholar] [CrossRef]
32. Lejaeghere K, Van Speybroeck V, Van Oost G, Cottenier S. Error estimates for solid-state density-functional theory predictions: an overview by means of the ground-state elemental crystals. Crit Rev Solid State Mater Sci. 2014;39(1):1–24. doi:10.1080/10408436.2013.772503. [Google Scholar] [CrossRef]
33. Zhang B, Han P, Mei J. Theoretical insight on mechanically robust graphene-nickel interfaces using chromium-substituted nickel and boron-doped graphene. Appl Surf Sci. 2022;593(1):153356. doi:10.1016/j.apsusc.2022.153356. [Google Scholar] [CrossRef]
34. Abdallah FS, Bouamama K, Farhat S, Chérif SM. First-principles calculations study of the configurations, structural, electronic and magnetic properties of graphene and h-BN monolayers and bilayers adsorbed on Co0001 surface. Phys B Condens Matter. 2021;611(5):412755. doi:10.1016/j.physb.2020.412755. [Google Scholar] [CrossRef]
35. Häglund J, Fernández GA, Grimvall G, Körling M. Theory of bonding in transition-metal carbides and nitrides. Phys Rev B. 1993;48(16):11685–91. doi:10.1103/physrevb.48.11685. [Google Scholar] [PubMed] [CrossRef]
36. Qi Y, Hector LG Jr, Ooi N, Adams JB. A first principles study of adhesion and adhesive transfer at Al(111)/graphite(0001). Surf Sci. 2005;581(2–3):155–68. doi:10.1016/j.susc.2005.02.048. [Google Scholar] [CrossRef]
37. Fiolhais C, Almeida LM, Henriques C. Extraction of aluminium surface energies from slab calculations: perturbative and non-perturbative approaches. Prog Surf Sci. 2003;74(1–8):209–17. doi:10.1016/j.progsurf.2003.08.017. [Google Scholar] [CrossRef]
38. Gong C, Lee G, Shan B, Vogel EM, Wallace RM, Cho K. First-principles study of metal-graphene interfaces. J Appl Phys. 2010;108(12):123711. doi:10.1063/1.3524232. [Google Scholar] [CrossRef]
39. Zou AH, Zhou XL, Kang ZB, Wu KY, Su YQ. Effect of discontinuous interfacial phase Al4C3 on interface bonding of SiC/Al composites: a first-principle and experiment. Acta Mater Compos Sin. 2021;38:824–31. (In Chinese). doi:10.15541/jim20180540. [Google Scholar] [CrossRef]
40. Luo Y, Huang Y, Liu J, Chen Q. Copper coated graphene reinforced aluminum composites with enhanced mechanical strength and conductivity. Vacuum. 2023;218:112610. doi:10.1016/j.vacuum.2023.112610. [Google Scholar] [CrossRef]
41. Nielsen O, Martin R. Quantum-mechanical theory of stress and force. Phys Rev B. 1985;32(6):3780–91. doi:10.1103/physrevb.32.3780. [Google Scholar] [PubMed] [CrossRef]
42. Liu J, Sun Z, Du X, Yang Q, Shi H. Fabrication, mechanical and thermal properties of copper-coated graphite flakes reinforced aluminum matrix composites. Compos Interfaces. 2026;33(1):1–21. doi:10.1080/09276440.2025.2505358. [Google Scholar] [CrossRef]
43. Liu G, Zhao N, Shi C, Liu E, He F, Ma L, et al. In-situ synthesis of graphene decorated with nickel nanoparticles for fabricating reinforced 6061Al matrix composites. Mater Sci Eng A. 2017;699:185–93. doi:10.1016/j.msea.2017.05.084. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF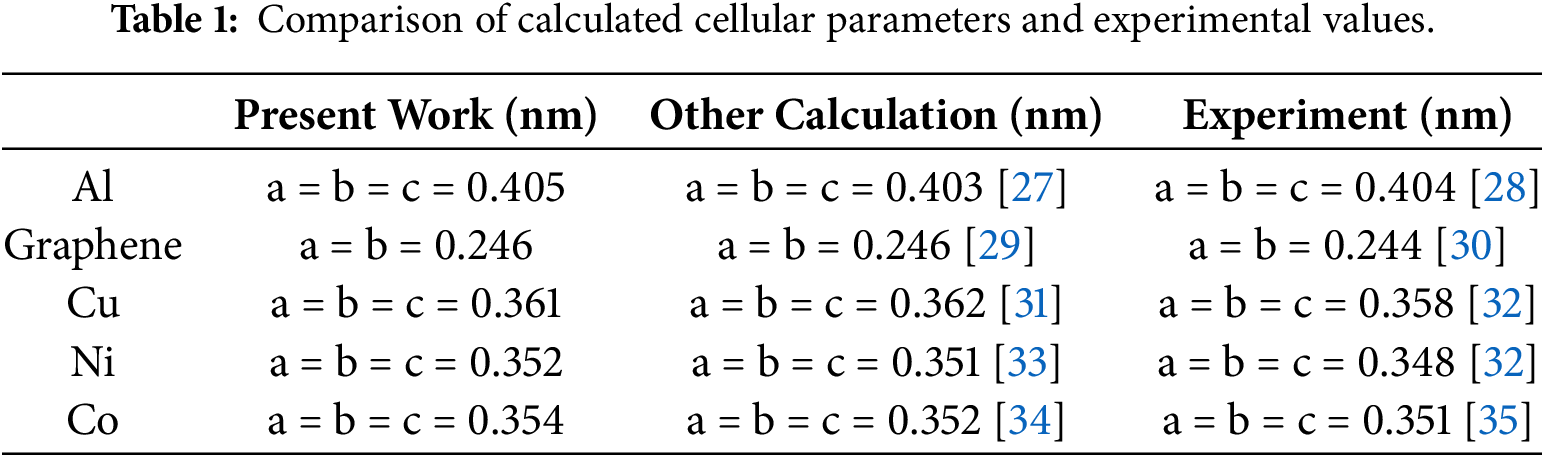
 Downloads
Downloads
 Citation Tools
Citation Tools