 Open Access
Open Access
ARTICLE
Electroosmotic Transport and Entropy Generation in ZnO-Williamson Nanoblood Flow through a Converging/Diverging Tapered Stenosed Artery
1 Institute of Mathematical Sciences, Faculty of Science, Universiti Malaya, Kuala Lumpur, Malaysia
2 Center for Data Analytics Consultancy and Services (UM-CDACS), Faculty of Science, Universiti Malaya, Kuala Lumpur, Malaysia
3 Department of Mechanical Engineering, College of Engineering, Prince Mohammad Bin Fahd University, Al Khobar, Saudi Arabia
4 Department of Mathematics, Faculty of Natural Sciences, HITEC University, Taxila Cantt, Pakistan
5 Department of Biosciences, COMSATS University, Islamabad, Pakistan
* Corresponding Author: Noor Fadiya Mohd Noor. Email:
(This article belongs to the Special Issue: Mathematical and Computational Modeling of Nanofluid in Biofluid Systems)
Computer Modeling in Engineering & Sciences 2026, 146(3), 24 https://doi.org/10.32604/cmes.2026.075694
Received 06 November 2025; Accepted 06 February 2026; Issue published 30 March 2026
Abstract
Electroosmotic transport and entropy generation play a decisive role in regulating efficiency, stability, and energy cost of non-Newtonian nanoblood flows in stenosed arteries, particularly with tapered geometries. This study develops a unified model to analyze ZnO–Williamson nanoblood flow through a stenosed artery with converging, diverging, and non-tapered configurations, incorporating electroosmosis, viscous dissipation, and entropy production. The arterial walls are assumed to be electrically charged with a no-slip condition to induce electroosmotic propulsion along the endothelial surface. The partial differential equations are nondimensionalized to a coupled system of nonlinear ordinary differential equations, which are solved numerically using a MATLAB-based shooting technique. Parametric investigation is conducted for Brinkman, Grashof, and Weissenberg numbers, ZnO fractional volume, volumetric flow rate, and Helmholtz–Smoluchowski velocity to quantify their influences on axial velocity, wall shear stress, impedance resistance, temperature distribution, entropy generation, Bejan number, and streamline topology. The axial velocity decreases radially with increasing Brinkman number for all arterial geometries. Increasing ZnO nanoparticles improves thermal transport owing to enhanced effective thermal conductivity but simultaneously elevates entropy generation due to increased viscous dissipation. Higher Weissenberg numbers suppress entropy production by promoting elastic stress redistribution and lowering shear-induced irreversibility. Impedance resistance decreases with increasing stenosis height but increases with stenosis shape parameter and ZnO fractional volume. Streamline analysis shows that buoyancy and viscoelasticity significantly distort flow near the stenosis, while increasing electroosmotic velocity stabilizes streamlines, suppresses recirculation, and reduces local shear stress and pressure fluctuations. In conclusion, electroosmotic actuation is most effective in reducing flow resistance in the converging tapered artery, particularly at lower ZnO volume fractions. Overall, the findings highlight the potential of optimized electroosmotic actuation and controlled nanoparticle loading to minimize thermodynamic losses, regulate shear stress, and improve flow uniformity in stenosed vessels, with promising implications for electro-assisted drug delivery, nanotherapeutics, and bio-inspired vascular microfluidic systems.Keywords
The transport of blood in a living human is braced by a convolution of vessels, comprised of veins, arteries, and capillaries, heaved by the beats of the heart. The right atrium, at the upper chamber of the heart, collects deoxygenated blood from the body before it is pumped by the right ventricle in the lower chamber and transported to the lungs via pulmonary arteries. This allows exchange of carbon dioxide with oxygen in the alveoli at the end of the bronchioles within the lungs. The resulting oxygenated blood is then transported back to the left atrium via pulmonary veins. Next, the oxygenated blood is pumped out by the left ventricle through the aorta to allow delivery of oxygen and nutrients to other parts of the body in concurrent reception of heat and wastes from the system. These two primary interconnected pulmonary and systemic circulatory loops repeat rhythmically with each heartbeat, to keep a human being alive every 1 to 2 s, successively. Ergo, any disease, blockage, or disruption in the transport of blood is a life-threatening reality that must be duly resolved, treated, and contained [1–4].
Tapered artery refers to an artery with walls that narrow or dilate gradually and naturally, normally or abnormally. Normal tapering occurs as the artery departs into tinier capillaries in a normal systemic circulation, assisting in blood rate regulation. In contrast, abnormal tapering happens as the artery is elastically enlarged radially due to aneurysm, vascular diseases, or aging factors. Yet, for most cases, abnormal tapering frequently happens in stenosed arteries where the channel is narrowed due to aggregation of fatty plaques, calcium, and particles at the internal lining of the arteries or due to other structural, inherited, or non-inherited causes. Existing treatments for stenosis require the patients’ consumption of several traditional drugs to control blood pressure, to lower bad cholesterol, and to prevent blood clotting in the stenosed arteries. Direct surgical treatments to tackle the stringent conditions are also available. These include endarterectomy and atherectomy procedures to clear the plaques from the stenosed artery, manually or via a catheter [5] with blades/laser, placing a nano-stent over an inflated balloon in the stenosed artery, as well as creating a new blood path from healthy tissues surgically to bypass the severely narrowed artery. Fortunately, there are emerging alternative treatments under progressive practice and research, one of which is by exploiting nanomedicine [6,7].
Nanomedicine defines a revolutionary spectrum of leveraging nanoparticles in medications to detect, to cure, and to contain the progression of diseases as part of nanotechnology advances in medical fields. Nanoparticles such as silver (Ag), gold (Au), zinc oxide (ZnO), iron oxide (Fe2O3), magnetite (Fe3O4), and graphene-based nanofilms are promising drug-delivery carriers to outstrip many side effects and low absorption issues in traditional medicines. Hybrid silver and alumina (Ag-Al2O3) nanoparticles can alter the hemodynamic properties of unsteady blood patterns through an artery with stenosis and aneurysm [8] while copper (Cu) and diamond (ND) nanoparticles with a shape factor regulate the peristaltic pumping in a vertical microchannel, aided by an electromagnetic mechanism [9]. On the other hand, the combination of magnetohydrodynamic effects and zinc oxides as anti-microbial and anti-cancer agents tends to control the bioheat transfer along the tapered blood artery due to stenosis/thrombosis [10]. These innovative nanomaterials can also be devised to trace and obliterate site-specific plaque build-ups along the stenosed artery in atherosclerosis treatments. Besides, thrombosis and restenosis risks can be obstructed by instilling specific nano-sized particles with anti-clotting and anti-inflammatory features in the nanocoating of the stents placed at the stenosed section of the coronary artery in an angioplasty procedure.
Electroosmosis [11] and osmosis are two analogies in sound and word, yet largely distinguished by the content-specific. Albeit osmosis conveys the idea of molecular water dynamics passing through permeable substances, passively driven by concentration gradients, electroosmosis addresses the duo-dynamics of ions and fluids across electrically charged surfaces with a greater impact on nanomedicine prevalence for enhanced drug transports in blood vessels [12] and for mitigation of plaque formation [13] in stenosed arteries. Essentially, the electroosmosis force is instilled in the blood vessels by placing an external electrode adjacent to the stenosed artery to establish electric fields along the axial direction for arterial stenosis dismissal. The nanoparticles’ surface [14] can also be functionalized with electric voltage to induce the electroosmosis force [15], assisting the drugs' penetration into the stenosed regions of the arterial walls. Likewise, modifications in hybrid magnetite nanoparticles and embedding nanoparticles in the surface coating of low-voltage-supplied nano-stents can be put forth in achieving similar objectives.
Blood is a sophisticated bio-suspension of cells, platelets, and plasma, rather than a simple fluid. It is more accurately described by a non-Newtonian shear-thinning model due to its variable viscosity dependency on shear rate, microstructure, and flow conditions. The non-Newtonian Herschel-Bulkley model is preferred to impersonate blood as a yield stress fluid having blood clots, while the blood elasticity is imitated by the Mazumdar model and the Rubinow-and-Keller model along an arterial stenosis [16]. The non-Newtonian Jeffrey models have been examined to mimic the viscoelasticity of red blood cells for an exact solution [17] and for a deionized nanoblood finite-element solution [18] of the pulsatile peristaltic flows. A simultaneous rise in stream function but a decline in wall shear stress is observed as Casson fluid parameter, microchannel inclination angle, Helmholtz–Smoluchowski velocity, zeta potential, and Reynolds number augment in the non-Newtonian membrane pumping-driven electroosmotic transport under Debye-Huckle linearization assumption [19]. The mathematical models of electroosmotic nanoblood flows have been formulated based on various non-Newtonian shear-thinning models, such as third-grade fluid [20], Papanastasiou modified Bingham fluid [21], Prandtl fluid [22], while Williamson [23], micropolar [24], and Sutterby [25] models have been reported as the best models for nanoblood flows.
ZnO-Williamson nanoblood refers to the dynamics of blood, adhering to the non-Newtonian law of the Williamson shear-thinning model, dispersed with zinc oxides as the nanoscale enhancements in the flow thermal, mass, and bio-transport properties. Despite intense research in bio-nanomedicine have been eagerly tended to date, specific studies on zinc oxide as an emerging smart drug-delivery agent for stenosis remedies in non-Newtonian Williamson blood flow are still very much limited. ZnO nanoparticles can be functionalized as a smart drug nanocarrier owing to their attributable properties of low-cost, electrical-conductive, anti-oxidant, anti-microbial, bio-compatible, biodegradable, pro-inflammatory cytokines to activate immune cells, and requirements of minimal invasive and mild heat to combat stenosis in the region-targeted blood vessels. A recent experimental study by Paul et al. [26] confirmed the attributes of ZnO nanoparticles for remarkable biodegradable (photocatalysis) property, antibacterial quality, high thermal stability, and crystallinity, ensuring decent use of the nanomaterials for both medical and environmental applications, especially to combat pollution and bacterial contamination. With respect to overcoming the drawbacks of zinc oxides as nano-agents for the transport of medicines and the dislodgment of arterial stenosis, electroosmosis aids in vanishing the nanoparticles’ clumps that could worsen the state of the stenosis at the cost of minimal entropy protraction and drug/viscosity loss. Recent studies on stenosed artery ZnO nanoblood flows with electroosmosis using artificial intelligence methods are reported in [27,28]. Hence, combining the appealing characteristics of ZnO [26] in the electroosmotic Williamson nanoblood flow model for biomedicine applications, such as in [23], would be a wise choice to begin with for a new investigation.
In biological fluid systems, particularly blood flow in arteries, entropy generation represents the degree of irreversibility and energy loss occurring during physiological transport processes. From a biological perspective, entropy generation provides a quantitative measure of how efficiently the cardiovascular system transports blood, heat, and nutrients. Entropy generation arises primarily due to viscous dissipation, which reflects internal friction between blood layers and between blood and the arterial walls. Elevated entropy generation indicates increased irreversibility, defining a greater portion of mechanical energy that is irretrievably converted into thermal energy rather than contributing to effective blood transport. In stenosed arteries, this irreversibility is amplified due to flow acceleration, recirculation, and elevated shear rates. Blood acts as a carrier of metabolic heat generated by tissues. Entropy generation associated with heat transfer reflects the inefficiency of thermal regulation within the vascular system. High entropy production may correspond to excessive thermal gradients, suggesting increased metabolic demand or impaired heat dissipation, which can influence tissue viability and vascular health. From a physiological standpoint, lower entropy generation is associated with more efficient cardiovascular function, whereas higher entropy generation implies increased cardiac workload to maintain adequate perfusion. In pathological conditions such as arterial stenosis, elevated entropy generation may signal compromised flow efficiency and increased energy expenditure by the heart. Regions of high entropy generation often coincide with elevated wall shear stress and temperature gradients, which can affect endothelial cell function, promote inflammation, and influence platelet activation and red blood cell deformation. These effects are closely linked to vascular remodeling and disease progression. Some studies in entropy generation focused on hemodynamic peristaltic nanofluid [9], non-Newtonian nanofluid with MHD and thermal radiation [18], utilization of ZnO in non-Newtonian electroosmotic stenosed artery blood flows [10,27,28], and application of the shooting method to solve the non-Newtonian Williamson models [29–31].
1.1 Problem Statement and Research Gaps
The mathematical modelling of Williamson non-Newtonian fluid is developed from lubrication theory and the second law of thermodynamics [23]. The Williamson nanofluid model is widely regarded as more appropriate than the Casson nanofluid model [29,31,32] for investigating electroosmotic blood flow through stenosed arteries with converging and diverging geometries, and entropy generation, owing to its superior ability to represent the zero-threshold shear-thinning behavior of blood across a broad range of shear rates. Unlike the Casson model, which primarily accounts for yield stress effects [19] and may oversimplify viscosity variations in high-shear regions near stenoses and tapered arterial walls, the Williamson model provides a more flexible and realistic rheological framework. Its formulation enables accurate characterization of velocity gradients, viscous dissipation, and electroosmotic [23] forcing, all of which are critical for quantifying thermal transport and entropy generation [29,30] driven by fluid friction and heat transfer irreversibility. Moreover, the Williamson model allows a more effective incorporation of nanoparticle-induced enhancements in thermal conductivity [31] and flow modulation, thereby improving predictions of heat transfer performance and energy dissipation in nanoblood systems. Previous studies have consistently shown that Williamson nanofluids yield stronger velocity, thermal, and mass transfer responses than Casson nanofluids under electrokinetic, magnetic, and radiative influences [31,32]. Consequently, the Williamson nanofluid model offers a more comprehensive and reliable framework for analyzing electroosmotic stenosed artery nanoblood flow with tapering effects and entropy generation, making it particularly suitable for advanced biomedical and therapeutic applications [23].
The discussed literature collectively indicates that while considerable progress has been made in modeling electroosmotic blood flow through stenosed arteries using nanoparticles and non-Newtonian models, existing studies are limited in scope, typically addressing no more than four coupled phenomena at a time. While advanced techniques such as artificial neural networks [27,28] have been employed to capture velocity profiles, wall shear stress, electroosmosis, and entropy generation, a unified and computationally efficient framework integrating all these aspects remains absent. In particular, no prior work has systematically analyzed the combined effects of stenosed arterial geometry with converging/diverging tapering, electroosmotic actuation, entropy generation, and ZnO–Williamson nanofluid transport using a semi-numerical approach such as the shooting method. This gap underscores the need for a comprehensive, reliable, and tractable model that can simultaneously capture the multi-physical interactions of governing nanoblood flow models in stenosed arteries, providing mechanistic insights and practical guidance for biomedical applications. Table 1 clearly presents the research gaps between the present work and the closest existing literature. All of these pave the motivation and justification of the present study.

The present study embarks on the following research questions:
• How does electroosmotic actuation modify the velocity field, wall shear stress, and flow resistance of a ZnO–Williamson nanoblood flow through a stenosed artery with converging and diverging walls?
• What is the role of non-Newtonian shear-thinning (Weissenberg number) in regulating entropy generation and thermodynamic irreversibility in an electroosmotic nanoblood flow?
• How do ZnO nanoparticle loading (volume fraction) and viscous dissipation (Brinkman number) interact to control thermal transport, entropy production, and Bejan number distributions in stenosed arterial geometries?
• In what way do electroosmotic velocity, buoyancy, and non-Newtonian Williamson shear-thinning influence the flow streamlines, hence the hemodynamics?
• Can a reduced-order semi-analytical framework based on the shooting technique reliably capture the coupled electro-thermo-hydrodynamic behavior of non-Newtonian nanoblood flows in complex stenosed geometries?
1.3 Present Research Objectives
Following the literature reviews, the current study aims to investigate the behaviors of electroosmotic ZnO-Williamson nanoblood flows through a stenosed artery with non-tapered and converging/diverging tapered walls on the hemodynamics and heat transport, Bejan number and entropy profiles, impedance resistance and stress of shear, as well as the streamline profiles. In particular, the present study embarks on the following research objectives:
• To formulate the governing nonlinear partial differential equations for electroosmotic ZnO–Williamson nanoblood flow in a stenosed artery with converging, diverging, and non-tapered geometries, incorporating entropy generation and thermal transport.
• To solve the resulting boundary value problem using a MATLAB-based shooting technique across selected physiologically relevant parameter ranges.
• To determine the effects of key dimensionless parameters: Brinkman number, Grashof number, Weissenberg number, ZnO nanoparticle volume fraction, volumetric flow rate, and Helmholtz–Smoluchowski parameter on axial velocity, wall shear stress, impedance resistance, temperature distribution, entropy generation, and Bejan number.
• To assess the role of electroosmosis (Helmholtz-Smoluchowski no-slip parameter), buoyancy (Grashof number), and non-Newtonian Williamson shear-thinning (Weissenberg number) in improving the nanoblood hemodynamics near the stenosis throat based on the streamline patterns.
• To minimize entropy production while maintaining favorable hemodynamic and thermodynamic performance in the stenosed arterial ZnO-Williamson nanoblood flows.
The core physical insights from the present study are as follows:
• Blood flow in stenosed arteries exhibits strong coupling between non-Newtonian rheology, wall geometry, thermal transport, and entropy generation, resulting in pronounced shear localization and irreversible energy losses.
• The Williamson shear-thinning model offers a realistic yet analytically efficient representation of blood viscoelasticity, enabling systematic examination of elastic stress redistribution and its influence on shear-dependent irreversibility.
• ZnO nanoparticles elevate thermal conductivity and intensify heat diffusion, lowering temperature gradients near the core region. However, increased nanoparticle volume fraction raises viscous dissipation, resulting in higher total entropy unless mitigated by electroosmotic actuation.
• Electroosmotic actuation introduces a near-wall body force that alters the momentum balance and provides a mathematically tractable mechanism for flow regulation under electrically charged boundary conditions.
• The present study provides theoretical insights into geometry-induced nonlinear interactions, entropy minimization mechanisms, and parameter-sensitive governing electroosmotic nanofluid transport in complex arterial configurations. These insights are physically relevant for predicting shear-controlled transport, thermal regulation, and energy-efficient flow manipulation in electro-assisted vascular and microfluidic systems.
The present model is developed under the following assumptions:
• Blood is treated as a single-phase ZnO–Williamson nanofluid with uniform nanoparticle dispersion and thermal equilibrium.
• The flow is assumed to be steady, axisymmetric, and laminar with a sufficiently small Reynolds number
• A weak ionic solution enabling electroosmosis with extremely low generated zeta potential (<25 mV) is considered so that the Poisson-Boltzmann equation can be simplified using Debye-Huckle linearization principle [18,25].
• The artery has prescribed converging, diverging, or non-tapered geometries and is mechanically rigid.
• The arterial wall is electrically charged, with a no-slip boundary condition and electroosmotic effects.
• Entropy generation is used to quantify the thermodynamic irreversibility.
• Viscous dissipation and nanoparticle-enhanced thermal transport are considered.
• Pulsatile flow, multiphase interactions, and full electric double-layer dynamics are neglected.
• Joule heating is neglected to simplify the energy equation without losing accuracy, as viscous dissipation and heat transfer dominate entropy generation in the artery. This assumption is widely accepted in bio-nanofluid modeling and electroosmotic blood flow studies.
The overall assumptions isolate the fundamental coupling between electroosmosis, non-Newtonian rheology, nanoparticle-enhanced thermal transport, and entropy generation while maintaining mathematical tractability.
2.2 Stenosed Artery Wall Configuration
Let us postulate an incompressible mixed convective flow of blood-based zinc oxide (ZnO) nanoparticles through a tapered stenosed artery having a length.
in which
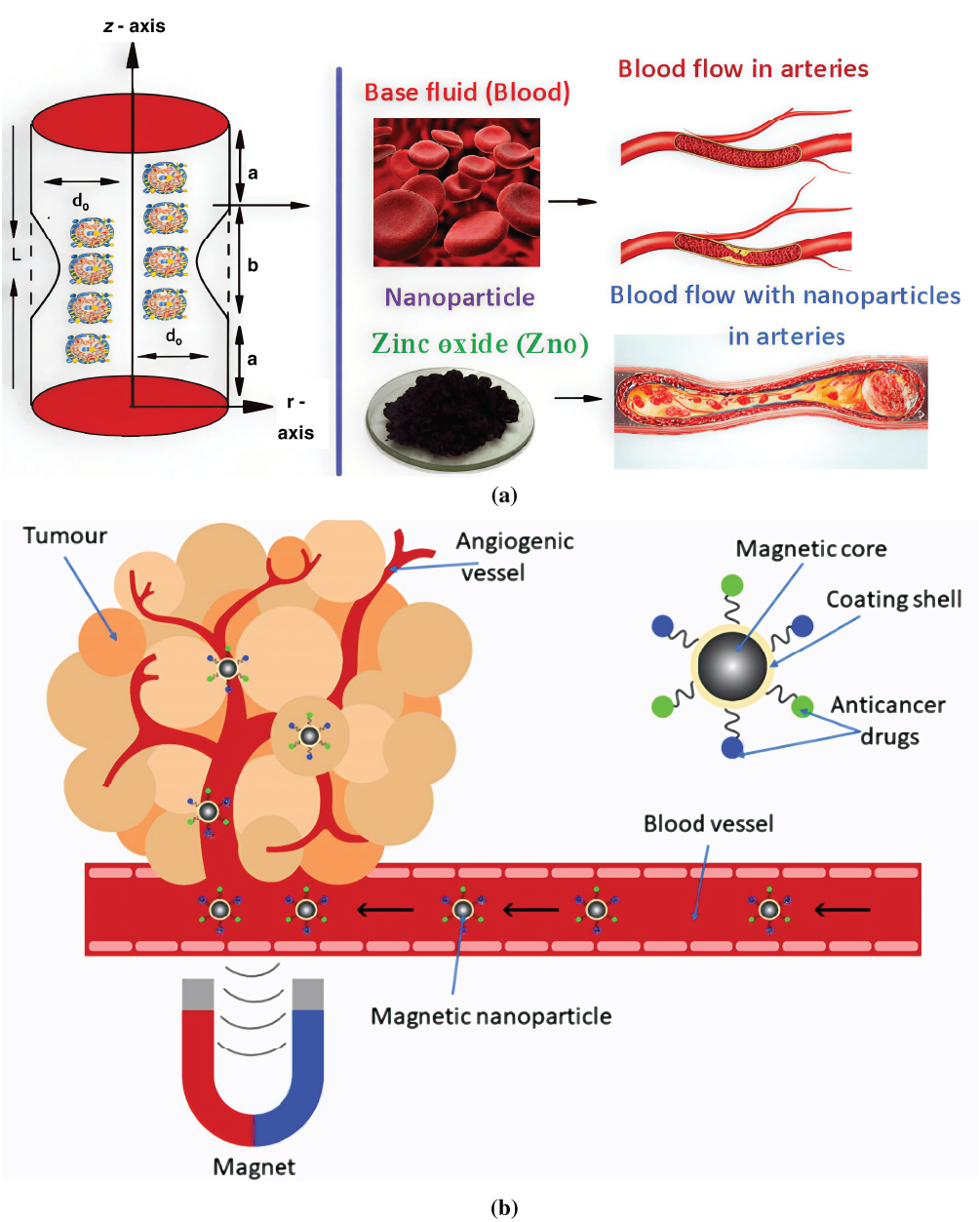
Figure 1: (a) Blood flow with interaction of ZnO nanoparticles in arteries and (b) The application of electroosmosis where nanoparticles are used to target tumor in the blood artery.
The equations dominating the flow and heat properties of blood blow through the stenosed tapered artery are formulated as [6,10,27]
In the previous relations,
where the terms
The electroosmotic phenomenon happens when the ionic solution comes across a surface, and it gets charged. The charged surface pulls the opposite ionic species towards itself, making an electric double layer (EDL) near the solid surface. Upon introducing an external electric field, the ionic species in EDL are pushed along the direction of the electric field, and so they carry the fluid along with them. The electric potential generated as a result of this process is mathematically represented by the Poisson equation, in which the charge number density of both species is calculated from the Boltzmann relation. As a result, the combined distribution is named the Poisson-Boltzmann distribution. Under a weak ionic solution and for most of such solutions, the generated zeta potential lies within the range of 25 mV. Therefore, Poisson-Boltzmann distribution can be simplified under the assumption of low zeta potential using Debye-Huckle linearization principle [18,25].
Now introducing the Poisson equation that accounts for measuring the electric potential
This gives
in which
Let us impose the no-slip and symmetry boundary conditions as
and introduce the following dimensionless quantities
Nondimensionalizing the Eqs. (2)–(5) using Eq. (15) and imposing assumption of laminar flow, i.e.,
The corresponding simplified boundary conditions are now
where

Integrating Eq. (19) subject to Eqs. (20) and (21), the electric potential is found in terms of modified Bessel function
Furthermore, the resistance impedance can be computed as
where
Analyzing the process of entropy generation provides an insight into the irreversibility or energy loss during the given process. In the problem under consideration, the entropy generated by nanoparticles and viscous forces is given by
After converting Eq. (25) into dimensionless form and imposing the mild stenosis assumption, the dimensionless expression for entropy is obtained as
Bejan number as the measure of contribution of thermal irreversibility in overall irreversibility is finally introduced as
Our flow equations in this article are coupled non-linear PDEs with associated boundary conditions, which were initially converted into first-order ODEs by introducing new scaling variables. After the conversion of the boundary value problem (BVP) to an initial value problem (IVP) took place, a shooting technique in MATLAB [29,31,32] is leveraged to guess the missing initial constraints iteratively until they are satisfied to evaluate the final solutions. The overall steps for implementing the procedure are depicted in Fig. 2. Since the momentum and energy equations are coupled and the stress terms are nonlinear, the shooting method is especially suitable for this fluid mechanics problem. The shooting method uses high-order adaptive integration techniques (such as Runge-Kutta-Fehlberg, RKF45) to resolve abrupt gradients within the boundary layer with great accuracy [29,30], in contrast to finite difference approaches that necessitate the inversion of massive sparse matrices [22,32]. Stable convergence of the shooting method can be preserved by using a continuation strategy (parameter stepping). To get a reliable first estimate, the solution is firstly calculated for a simplified situation (e.g., Newtonian fluid
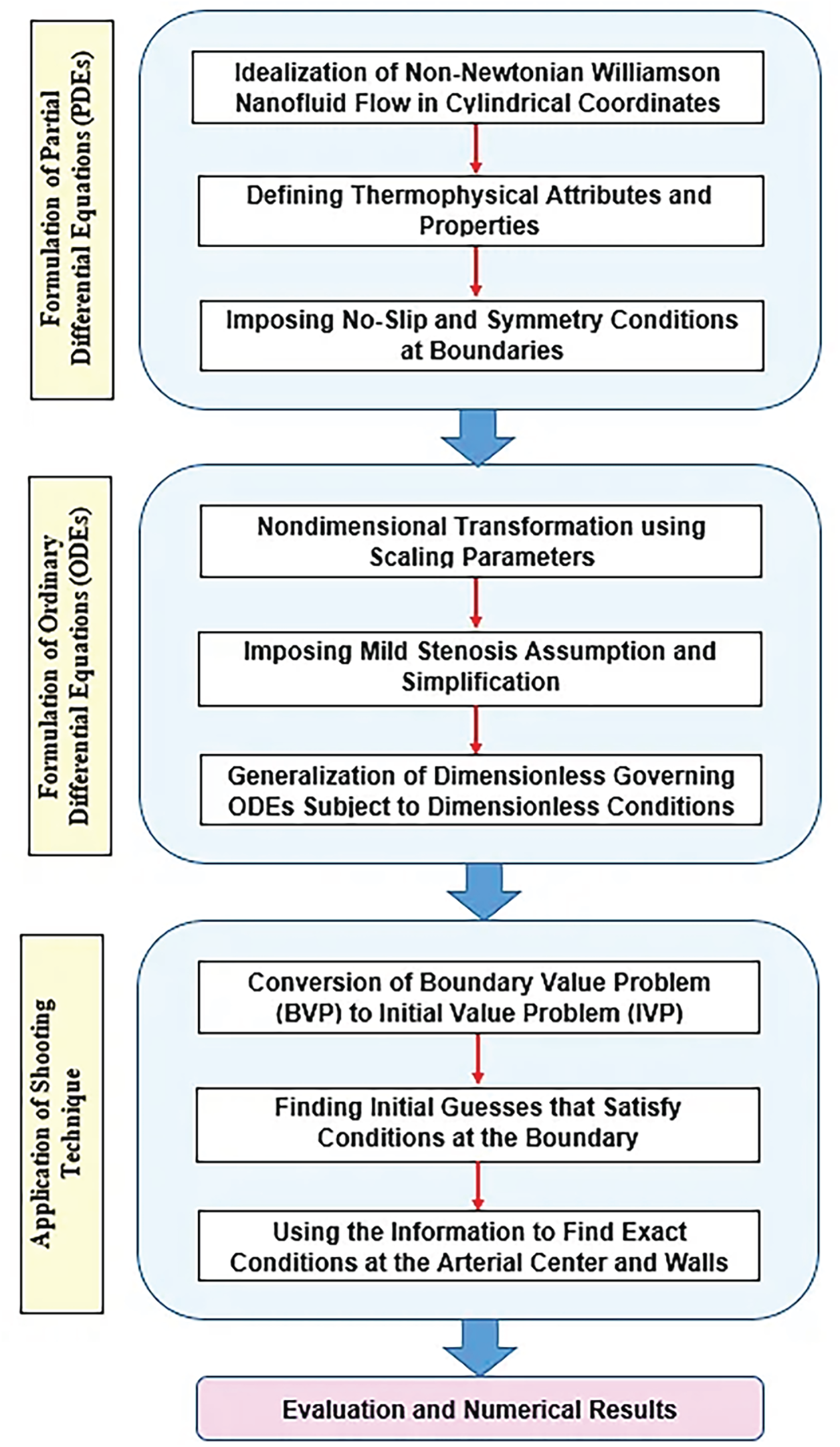
Figure 2: Methodology description.
In the present context of solving boundary value problems (BVPs) with the shooting method, mesh distribution refers to how the independent variables (in the form of axial coordinates) are discretized over which the ordinary differential equations (ODEs) are integrated [31]. Unlike finite difference or finite element methods [22,32], the shooting method does not require a fixed mesh for the solution itself, because it essentially converts the boundary value problem (BVP) into an initial value problem (IVP) [31]. However, the mesh is still needed for numerical integration of the IVP using methods like Runge–Kutta. With relative and absolute tolerances set to
By imposing the following BVP-IVP transforms
we get
The transformed conditions are
whilst the resistance impedance can be evaluated as
Boundary Shearing Stress
Boundary shearing stress (often called wall shear stress) is the tangential force per unit area exerted by a flowing fluid on a solid boundary due to viscous effects. It arises because of the no-slip condition, which states that the fluid velocity at a solid boundary is equal to the velocity of the boundary itself. When a fluid flows over a surface (such as blood flowing along an arterial wall or a fluid moving through a pipe), layers of fluid slide past one another. The layer in direct contact with the boundary has zero velocity relative to the wall, while adjacent layers move faster. This velocity gradient generates a shearing force at the boundary, known as boundary shearing stress.
The expression for boundary shearing stress considered in the present work is depicted as:
The wall shear at the greatest height of the stenosis, where the stress caused by shear at the narrowing of the throat located at
The discussions onwards are crumbled into five subsections. In the first three subsections, hemodynamics and heat transport, entropy and Bejan number, as well as impedance resistance and stress of shear are profiled for the stenosed artery nanoblood flows according to three arterial wall tapering scenes: converging, none, and diverging. The outcomes can be observed from Figs. 3–8, respectively. Eventually, individual dominances of Grashof number Gr, Weissenberg number We and Helmholtz-Smoluchowski velocity
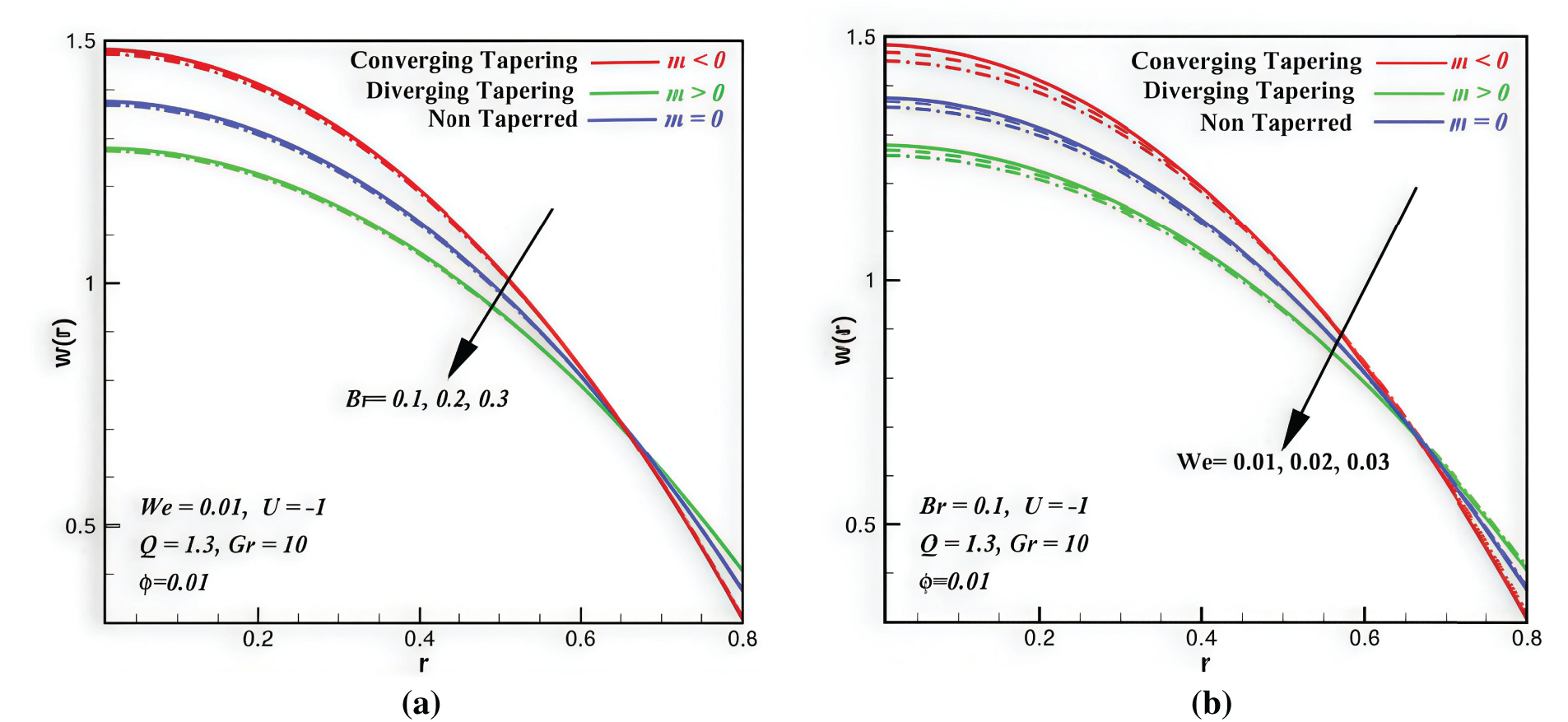
Figure 3: Velocity profiles vs. space coordinate,
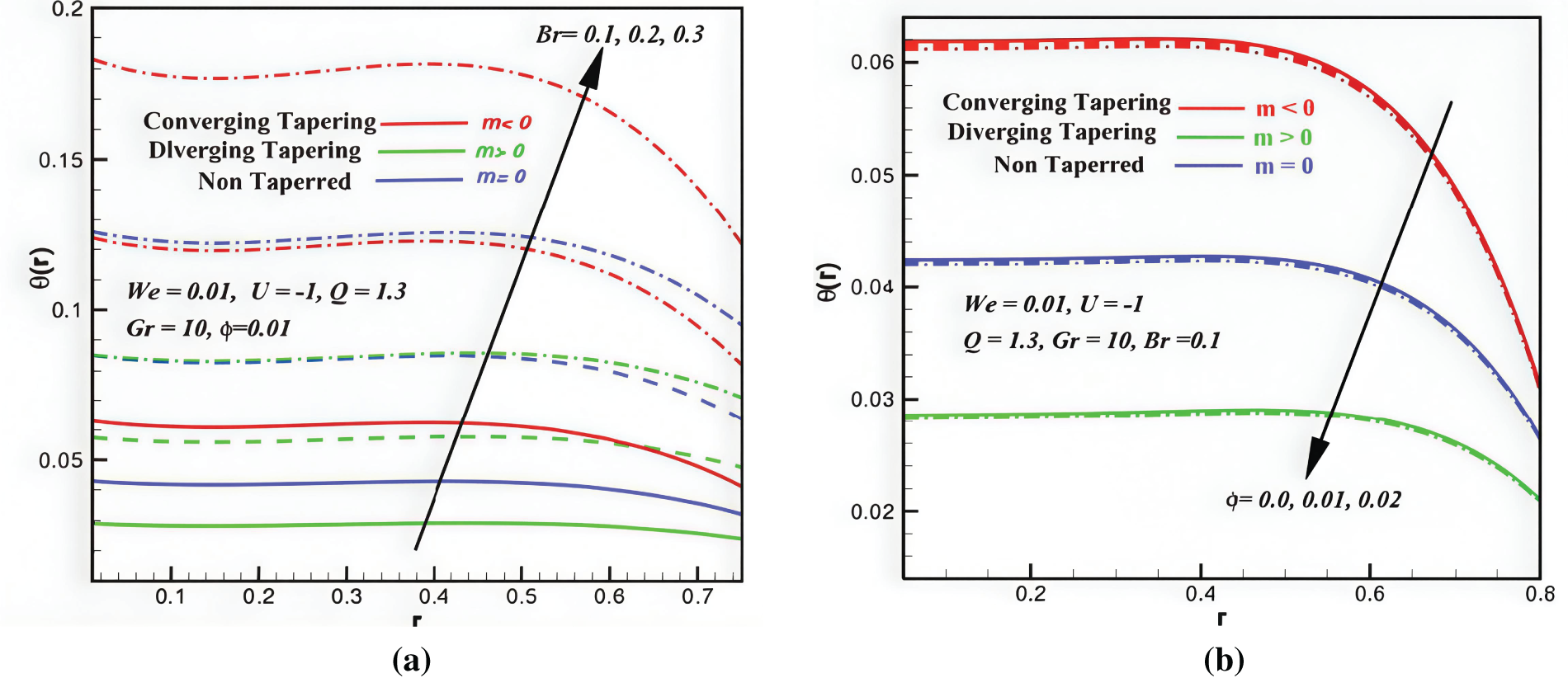
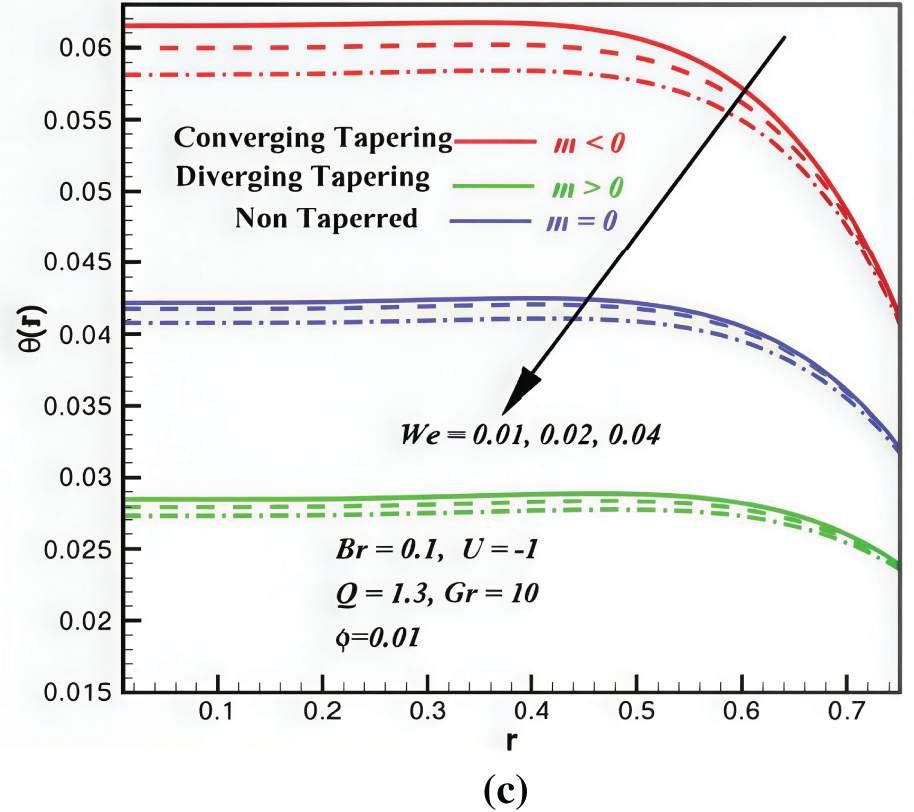
Figure 4: Temperature profiles vs. space coordinate,
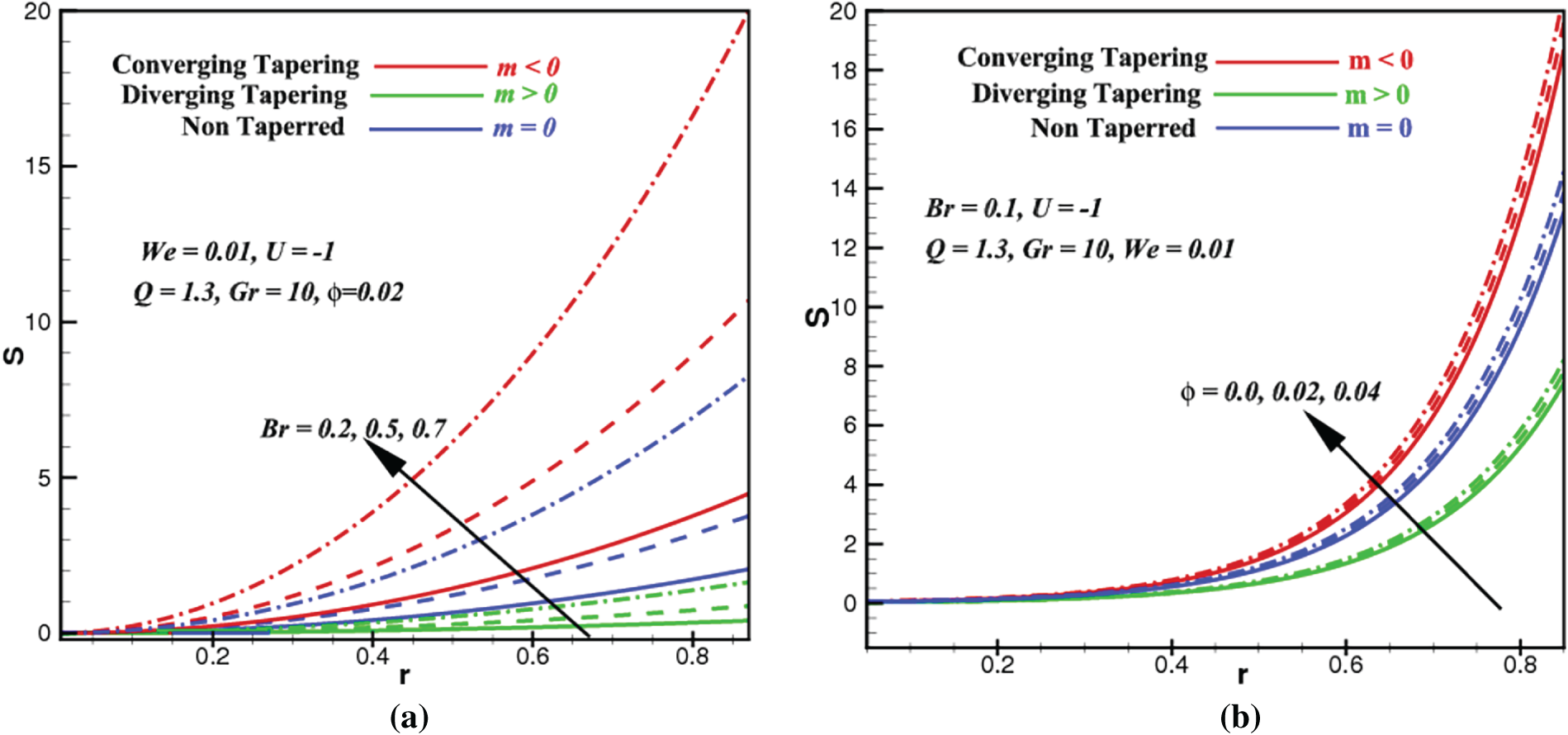
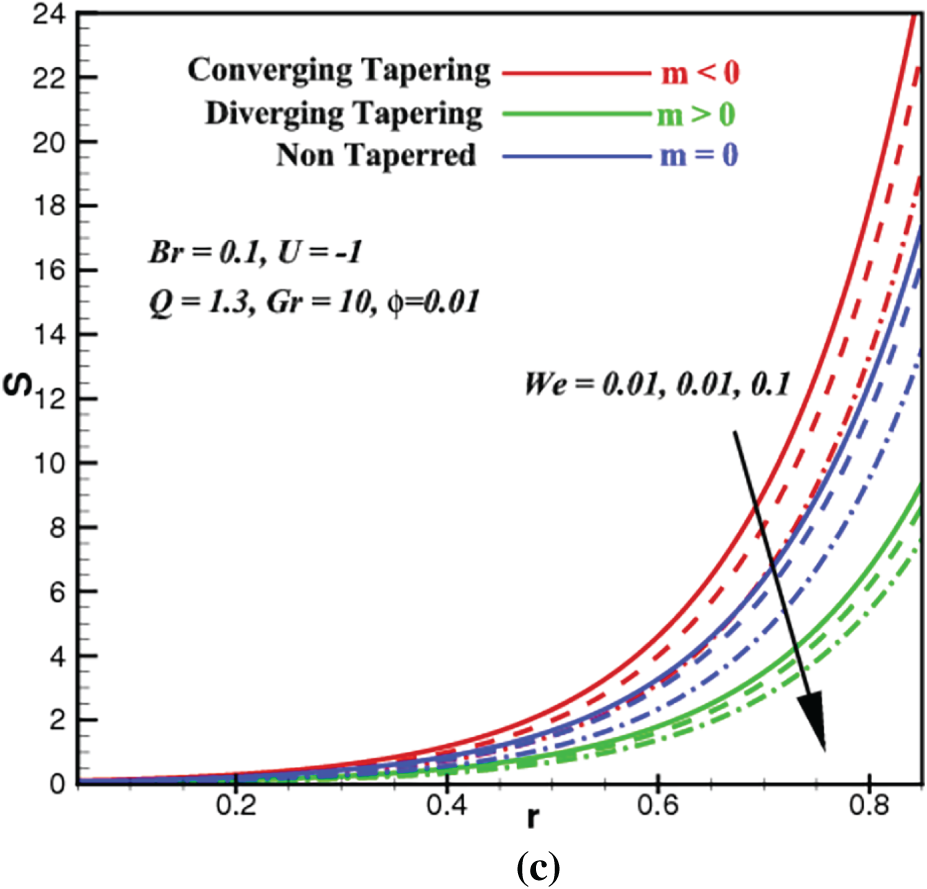
Figure 5: Entropy profiles vs. space coordinate,
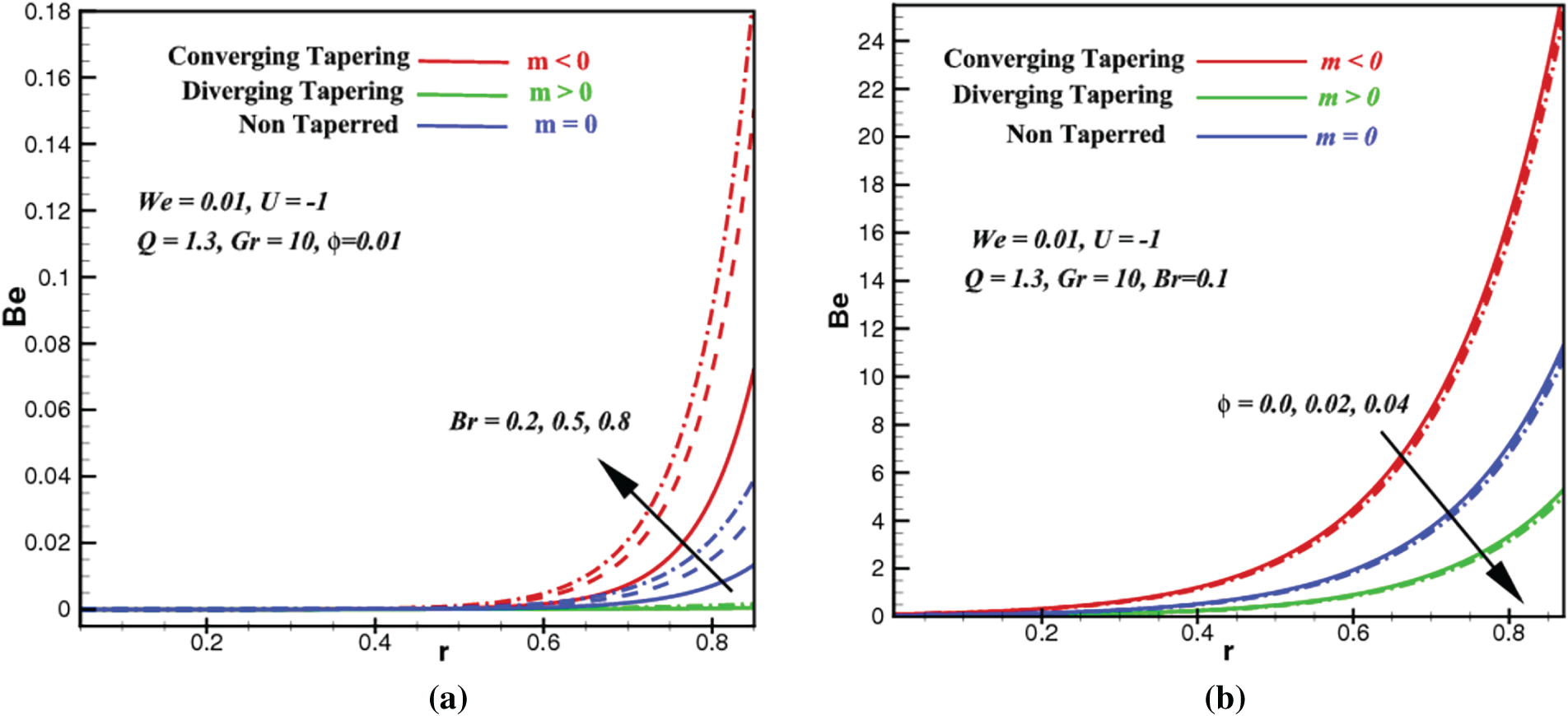
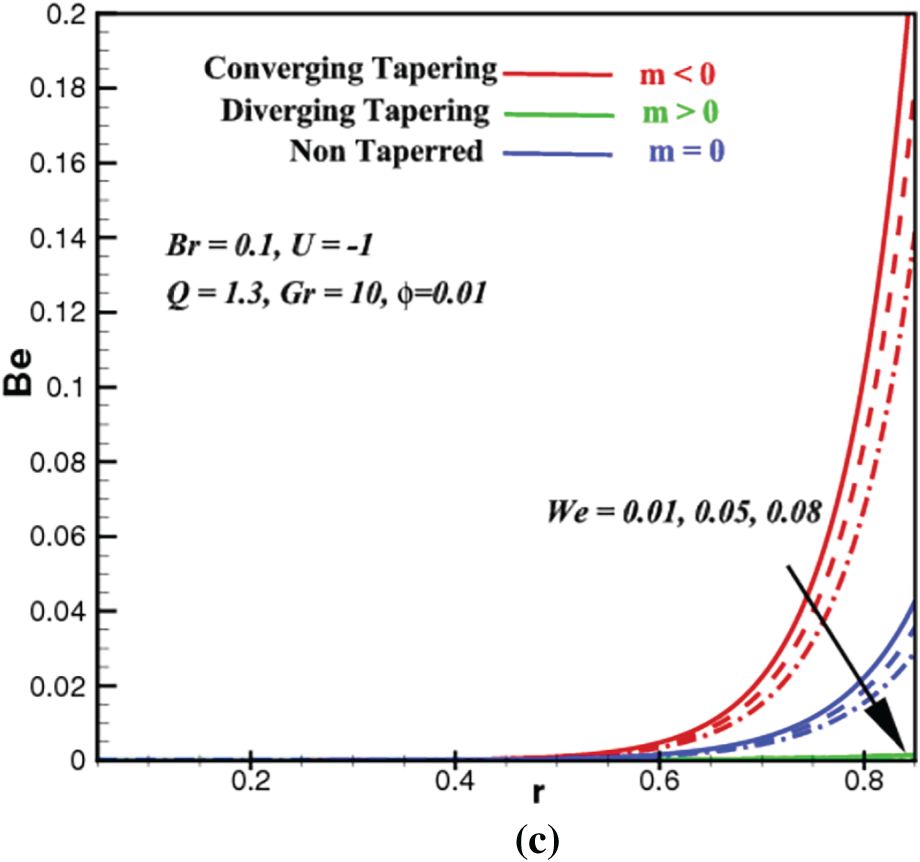
Figure 6: Bejan number profiles vs. space coordinate,
Figure 7: Impedance resistance profiles vs. stenosis height,
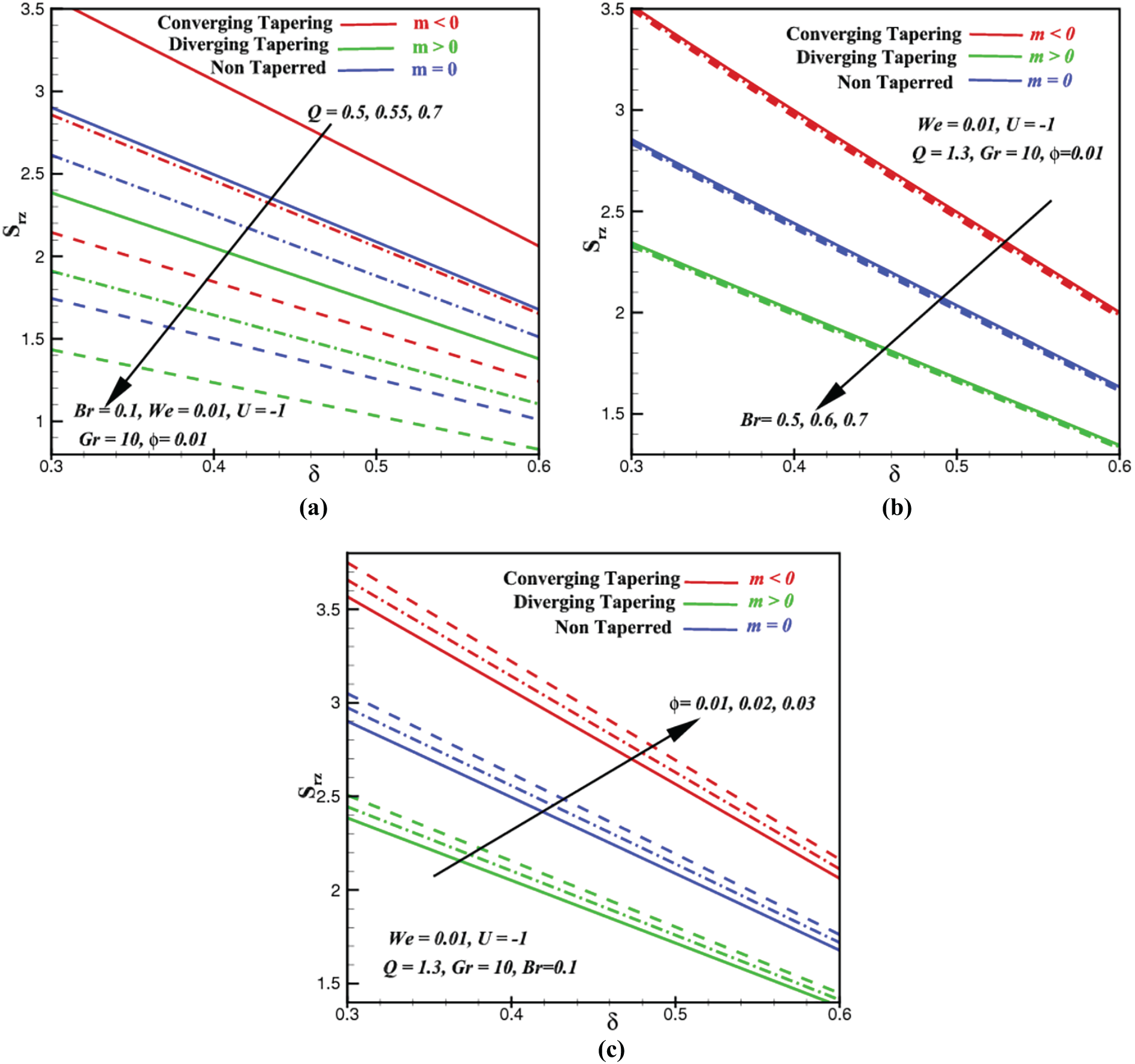
Figure 8: Shear stress profiles vs. stenosis height,
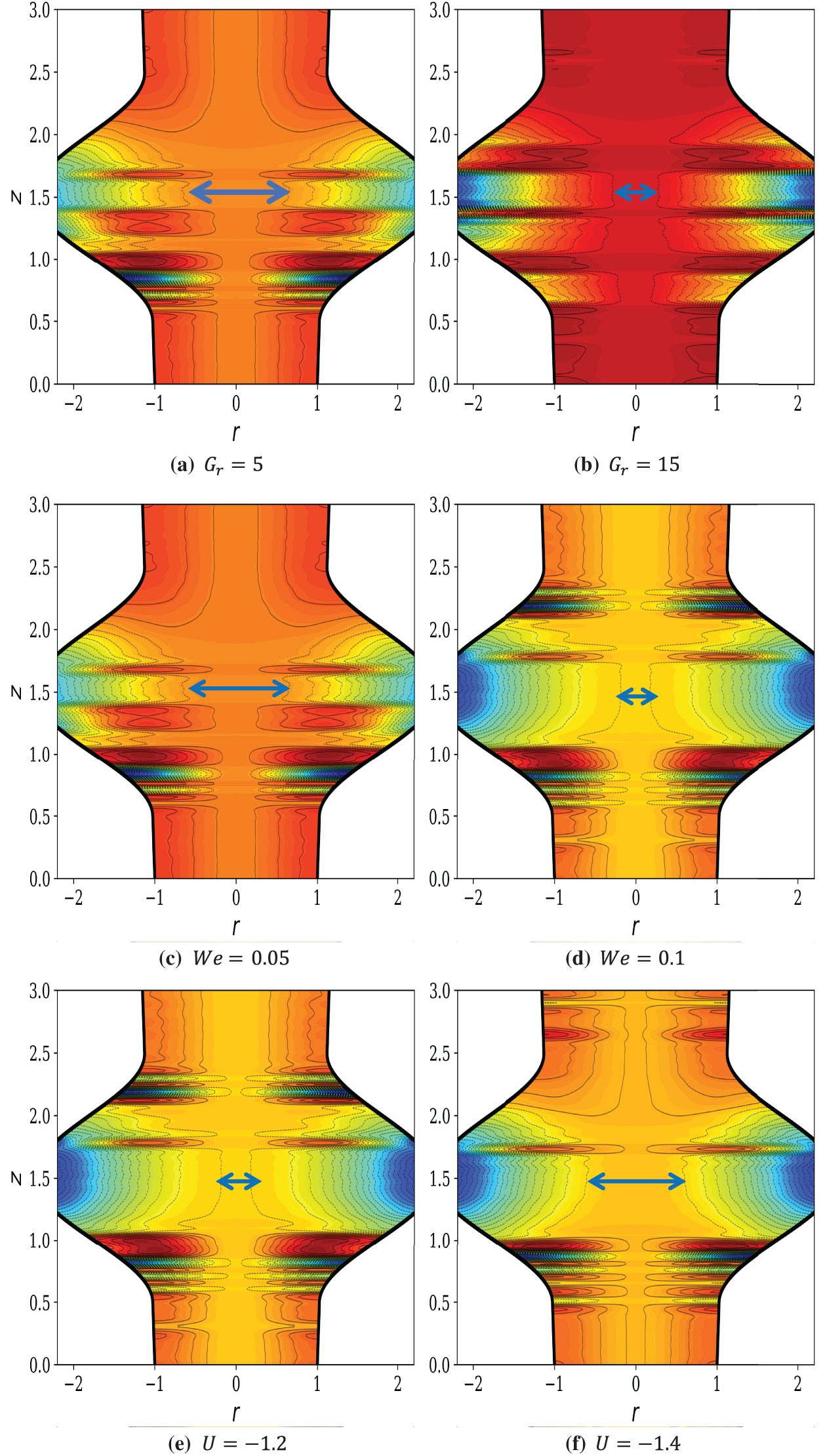
Figure 9: Streamlines versus space coordinate,
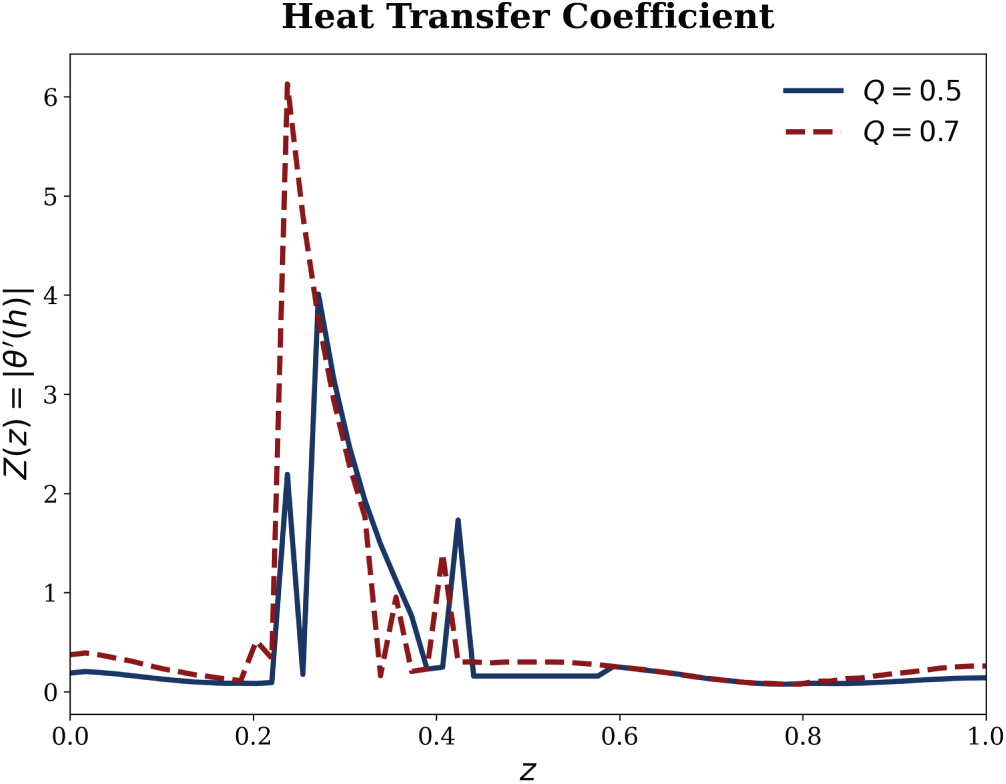
Figure 10: Relationship between the volumetric flow rate Q and the heat transfer coefficient.
5.1 Hemodynamics and Heat Transport
Fig. 3 outlines the nanoblood hemodynamic profiles with variation in Br and We, along the radial direction with r = 0 indicates the centerline of the stenosed artery, where the maximum flow velocity would possibly occur while r
Next, we intend to uncover how the nanoblood temperatures can be affected by Br, We and the ZnO volume fraction
Entropy production, S quantifies the potential of how much energy can be destructed or wasted during a process. This is why an efficient system should have the least entropy production. Bejan number, Be constitutes how much the proportion of entropy produced by heat transport as compared to the overall entropy production (dissipation of viscosity + heat transfer, etc.) in the system. In this subsection, we investigate how the Brinkman number, Br, ZnO volume fraction,
First of all, we notice that all the profiles of S and Be are insignificant at the core of the artery but are exclusively conspicuous at the radial region approaching the arterial walls. As the Brinkman number Br rises in Fig. 5a, the entropy from heat transfer (part of total entropy S) rises prominently near the arterial wall due to added supplies of energy from viscous or frictional heating. Since the heat transfer entropy appears both in the numerator and denominator of the Be ratio in Eq. (27), similar outcomes are postulated in Fig. 6a. Apparently, the influences of
5.3 Impedance Resistance and Stress of Shear
Fig. 7a illustrates the variation of impedance resistance,
In Fig. 8, we consider the behavior of the shearing boundary (Eq. (34)) with respect to volumetric flow rate, Q (Fig. 8a), Brinkman number Br (Fig. 8b) and the ZnO fractional volume
Next, we would like to understand why raising the Brinkman number, Br shares a similar impact as rising Q (Fig. 8a) to the shearing stress in Fig. 8b. Essentially, it is notable that when Br is magnified in Fig. 8b, the proportion of viscous heating becomes greater than the proportion of conductive heating in Br. Naturally, this excess of heat will either be absorbed as enthalpy, be irreversibly converted into another potential energy, or, in many cases, the heat will just be released back to its environment. As the loss of heat increases with increasing Br, this causes the stress of shear to decline, as evidenced in Fig. 8b. On the other hand, adding the ZnO fractional volume into the blood increases the concentration, aggregation, and collisions of the blood substances. As a result, the shearing stress increases with the inclining
In the present study, the ZnO-Williamson nanoblood flows through a stenosed artery are considered under the assumptions of having a small Reynolds number and a mild stenosis case for Br = 0.3, Q = 2 and
Grashof number, Gr quantifies the relative strength of buoyancy forces induced by temperature gradients as compared to viscous forces. Based on Fig. 9a, the streamlines remain relatively smooth and aligned with the axial flow direction, indicating that viscous effects dominate and buoyancy-induced convection is weak. Flow remains largely symmetric about the artery centerline, with limited secondary motion near the stenosed region. In contrast, at a higher Grashof number (Fig. 9b), buoyancy forces become significant, leading to pronounced streamline distortion and curvature, particularly near and downstream of the stenosis throat. On the other hand, the Weissenberg number, We, represents the ratio of elasticity to viscosity forces in the non-Newtonian Williamson nanoblood shear-thinning flow. At a lower Weissenberg number (Fig. 9c), elastic effects are weak, and the streamlines exhibit nearly Newtonian-like behavior with mild deformation around the stenosis. The flow adjusts smoothly to geometric constriction, and recirculation zones remain limited. However, as We increases (Fig. 9d), viscoelastic effects become dominant, resulting in substantial streamline stretching, asymmetry, and compression near the stenosis throat. The intensified streamline deformation indicates elastic energy storage and delayed stress relaxation characteristics of Williamson-type fluids. This enhances shear localization, modifies near-wall transport, and significantly alters the hemodynamic response, which may increase flow resistance while amplifying thermal gradients and entropy production due to elastic dissipation.
In electroosmotic blood flow, the electroosmotic velocity arises due to the interaction between the applied electric field and the charged ions within the electrical double layer adjacent to the arterial wall. This velocity component, represented by Helmholtz–Smoluchowski velocity, is typically confined to a thin region near the wall and is proportional to the electric field strength and the zeta potential. In contrast, the axial flow velocity is primarily driven by the pressure gradient and dominates the bulk transport of blood through the artery. From a physical perspective, the magnitude of the electroosmotic velocity is generally one to two orders of magnitude smaller than the axial flow velocity under physiological electric field strengths. Consequently, electroosmosis does not replace pressure-driven flow but rather acts as a secondary modulation mechanism, influencing near-wall velocity gradients, wall shear stress, and nanoparticle transport. In stenosed arteries where velocity gradients are intensified, electroosmosis can locally alter the flow structure by slightly flattening the velocity profile near the wall, thereby reducing frictional resistance and modifying entropy generation. However, the axial velocity remains the dominant contributor to overall hemodynamics and volumetric flow rate of the nanoblood flows.
Fig. 9e,f demonstrates the impact of the Helmholtz–Smoluchowski parameter (no-slip electroosmotic velocity) on the present ZnO-Williamson nanoblood flows. At a lower magnitude of the electroosmotic velocity (Fig. 9e), pressure-driven flow dominates, resulting in noticeable streamline curvature near the stenosis and partial recirculation zones adjacent to the arterial walls. These features indicate higher shear stress concentration and non-uniform velocity distribution. When the magnitude of the Helmholtz–Smoluchowski velocity is increased in Fig. 9f, electroosmotic forcing significantly reorganizes the flow, producing more uniformly distributed, nearly parallel streamlines across the artery cross-section. The suppression of flow reversal and reduction of recirculation zones reflect the formation of a plug-like electroosmotic profile, which stabilizes hemodynamics, enhances nanoparticle dispersion, and promotes efficient thermal transport. Consequently, electroosmosis plays a crucial role in mitigating stenosis-induced disturbances while improving heat transfer and entropy regulation.
Based on Fig. 9a–d, it is obvious that imposing higher values of Gr and We increases the pressure of the ZnO nanoblood flow at the throat hence causing the stenosis throat to be tighter. This phenomenon could be rooted from increments in the product of thermal expansion and temperature gradient that is directly proportional to Gr. Besides, as We inclines, the radial distance in the denominator of We is decreased and so the relative stenosis height. Being the no-slip velocity that defines an electroosmosis property of the ZnO nanoblood flow over an electrically charged surface,
In summary, the streamline distributions reveal that increasing the Grashof number, Gr significantly enhances buoyancy-driven convection, leading to pronounced streamline curvature and intensified mixing near and downstream of the stenosis. This not only promotes heat transfer but also increases local velocity gradients, thereby elevating shear stress and contributing to higher entropy generation. At higher Weissenberg number, We, viscoelastic effects dominate, causing substantial streamline distortion, elastic stretching, and shear localization around the stenosed region. These effects reflect delayed stress relaxation and heightened non-Newtonian behavior characteristic of Williamson shear-thinning nanoblood, which in turn amplifies the pressure gradients required to maintain the flow continuity. In contrast, increasing the Helmholtz–Smoluchowski no-slip velocity,
5.5 Brief Comparative Analysis and Other Discussion
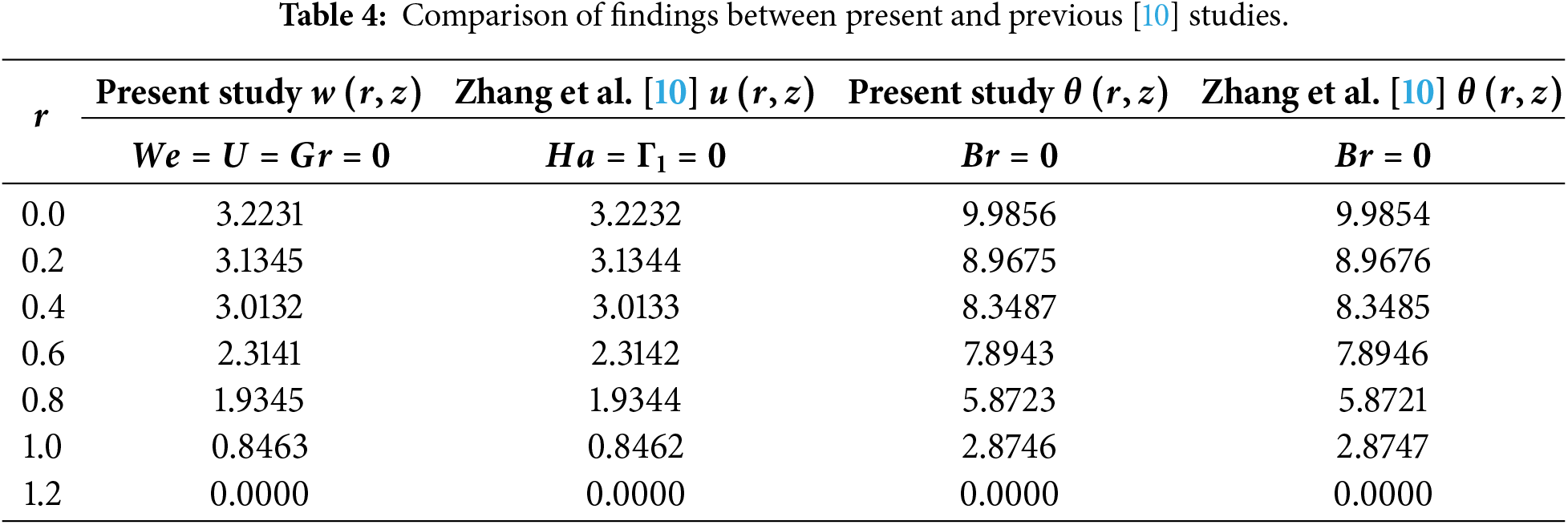
In this subsection, other analyses of interest are discussed. Table 4 presents brief comparisons between present and previous findings [10]. The numerical results are evaluated for a special case when the non-Newtonian Jeffery model parameters in [10] and the non-Newtonian Williamson model parameters in the present study are set to zero, using a powerful data visualization and analysis software, Tecplot. It is obvious that both numerical findings achieved a good agreement, i.e., up to 3 decimal places.
On the other hand, it is found that the volumetric flow rate Q is directly related to heat transfer coefficient based on Fig. 10. This is a fact because convective movement of thermal energy inside the fluid is improved by an increase in the flow rate Q. Physically speaking, a higher flow rate means that fluid particles travel through the channel or artery more quickly, continuously substituting cooler fluid from the core region for the fluid next to the hot wall. By decreasing the thermal boundary layer’s thickness, this procedure raises the temperature gradient at the wall. Consequently, the heat transfer coefficient rises with the increasing volumetric flow rate since it is directly proportional to this wall temperature gradient.
The present study explored the electroosmotic transport and entropy generation characteristics of ZnO–Williamson nanoblood flow through a stenosed artery under converging, diverging, and non-tapered geometries. The governing nonlinear equations were solved using a shooting technique where the effects of key dimensionless parameters such as Brinkman number, Grashof number, Weissenberg number, ZnO nanoparticle volume fraction, and electroosmotic velocity were systematically examined. The main conclusions are summarized as follows:
• The axial velocity of nanoblood decreases radially with higher Brinkman numbers across all tapering configurations. ZnO nanoparticles enhance heat dissipation due to their high thermal conductivity, thereby producing a cooling effect in the flow with increasing nanoparticle concentration.
• Entropy generation and Bejan number remain minimal at the artery core but increase significantly near the wall, primarily due to viscous dissipation and thermal gradients. Larger Brinkman and ZnO nanoparticle volume fractions intensify total entropy, while higher Weissenberg numbers suppress entropy and Bejan values owing to improved fluid elasticity and reduced shear friction.
• Among all wall geometries, the converging tapering produces the most pronounced hemodynamic and thermodynamic responses, followed by the non-tapered and diverging scenes.
• Impedance resistance and stress of shear consistently decrease with an increase in the stenosis height, yet are significantly intensified by rising stenosis shape parameter and ZnO volume fraction. The converging tapering provides more favorable hemodynamic and thermal transport conditions for the Williamson nanoblood flows under intensified stenosis shape parameter and/or at the lowest ZnO volume fraction.
• Streamline analysis reveals that increasing the electroosmotic (Helmholtz–Smoluchowski no-slip) velocity stabilizes the flow and mitigates recirculation, while higher Grashof and Weissenberg numbers enhance buoyancy-driven curvature, elastic stretching, and shear localization near the stenosis.
The findings further suggest that electroosmotic actuation could be harnessed in practical biomedical devices, such as microcatheters, targeted nanoparticle-assisted drug delivery systems and lab-on-a-chip vascular platforms, to actively control flow rates, to improve nanoparticle dispersion and to reduce localized shear stress.
6.1 Limitations of the Present Study
The present analysis is conducted under the assumptions of no-slip boundary conditions, mild stenosis, and sufficiently small Reynolds number, restricting the applicability of the results to steady, laminar flow regimes. The arterial geometry is prescribed with converging, diverging, and non-tapered configurations and is treated as mechanically rigid, thereby excluding wall deformation and fluid–structure interaction effects. Blood is modeled as a single-phase steady laminar non-Newtonian Williamson shear-thinning nanofluid with uniform ZnO nanoparticle dispersion, while pulsatile flows, multiphase interactions, and detailed electric double-layer dynamics are neglected to preserve the mathematical tractability. These idealizations were intentionally adopted so that the fundamental coupling between electroosmosis, non-Newtonian rheology, nanoparticle-induced thermal transport, and entropy generation can be focused on.
Future works may extend the present framework by incorporating pulsatile flows to capture transient electroosmotic and entropy-generation dynamics under physiological cardiac cycles. Accounting for arterial wall compliance through fluid–structure interaction would enable analysis of moderate-to-severe stenosis and pressure–deformation coupling. Multiphase modeling of blood–nanoparticle interactions, including particle migration and agglomeration, may further enhance the hemodynamic and thermal transport realism. Resolving electric double-layer dynamics via Poisson–Boltzmann formulations and validating predictions through microfluidic or experimental studies would further strengthen the model fidelity and support translation toward electro-assisted drug delivery and bio-inspired vascular flow control applications.
Acknowledgement: This work is supported by the Ministry of Higher Education, Malaysia, under the Fundamental Research Grant Scheme FRGS/1/2023/STG06/UM/02/4 (Project FP069-2023).
Funding Statement: This work is funded by the Ministry of Higher Education, Malaysia, under the Fundamental Research Grant Scheme FRGS/1/2023/STG06/UM/02/4 (Project FP069-2023).
Author Contributions: Noor Fadiya Mohd Noor: Conceptualization, Literature Review, Formal Analysis, Writing—Original Draft, Writing—Review and Editing, Funding Acquisition. Noreen Sher Akbar: Formal Analysis, Investigation, Methodology, Visualization, Writing—Original Draft. Rashid Mehmood: Data Curation, Investigation, Resources. Muhammad Bilal Habib: Software, Validation, Resources. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: Data available upon reasonable request from the authors.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
Nomenclature
| Bejan number | |
| Brinkman number | |
| Specific heat capacity | |
| Gradient of pressure per axial unit length (Pa | |
| Dimensionless electrostatic potential | |
| Electric potential in EDL (V) | |
| Eckert number | |
| Electronic charge (C) | |
| Gravitational acceleration | |
| Grashof number | |
| Boltzmann constant ( | |
| Debye-Hückle parameter | |
| Measure of artery wall non-uniformity | |
| Shape factor of the stenosis | |
| Charge number density of cationic species | |
| Charge number density of anionic species | |
| Pressure (Pa) | |
| Volumetric flow rate | |
| Dimensionless radial coordinate | |
| Radius of artery | |
| Reynolds number | |
| Total entropy production | |
| Arterial wall shearing stress | |
| Helmholtz-Smoluchowski velocity | |
| Weissenberg number | |
| Dimensionless axial coordinate | |
| Valency of ionic species | |
| Greek symbol | |
| Wall roughness ratio | |
| Thermal expansion parameter | |
| Williamson constant parameter | |
| Relative stenosis height | |
| Maximum height of stenosis (m) | |
| Nanoblood dielectric constant | |
| Viscosity at zero shear rate | |
| Viscosity at infinite shear rate | |
| Nanofluid dynamic viscosity | |
| Dimensionless temperature | |
| Density ( | |
| Charge number density | |
| ZnO fractional volume | |
| Abbreviations | |
| BVP | Boundary Value Problem |
| EDL | Electric Double Layer |
| IVP | Initial Value Problem |
| ODEs | Ordinary Differential Equations |
| PDEs | Partial Differential Equations |
| ZnO | Zinc Oxide |
References
1. Liu B, Tang D. A numerical simulation of viscous flows in collapsible tubes with stenoses. Appl Numer Math. 2000;32(1):87–101. doi:10.1016/S0168-9274(99)00015-X. [Google Scholar] [CrossRef]
2. Hsu RY, Lo HY, Chen CH, Wu YJ, Chan DC, Wu CC, et al. Blood pressure targets, medication considerations and special concerns in elderly hypertension: focus on atherosclerotic cardiovascular diseases, atrial fibrillation, heart failure, and aortic stenosis. J Formos Med Assoc. 2025;124(Suppl 1):S49–54. doi:10.1016/j.jfma.2025.05.007. [Google Scholar] [PubMed] [CrossRef]
3. Nejad AA, Talebi Z, Cheraghali D, Shahbani-Zahiri A, Norouzi M. Pulsatile flow of non-Newtonian blood fluid inside stenosed arteries: investigating the effects of viscoelastic and elastic walls, arteriosclerosis, and polycythemia diseases. Comput Methods Programs Biomed. 2018;154(5):109–22. doi:10.1016/j.cmpb.2017.11.016. [Google Scholar] [PubMed] [CrossRef]
4. Kumar A, Tripathi D, Tiwari AK, Seshaiyer P. Magnetic field modulation of electroosmotic-peristaltic flow in tumor microenvironment. Phys Fluids. 2025;37(4):043109. doi:10.1063/5.0264693. [Google Scholar] [CrossRef]
5. Wajihah SA, Sankar DS, Nagar AK. Influence of thrombosis, stenosis and catheter on rheological characteristics of blood: a systematic review. Arch Appl Mech. 2023;93(12):4279–304. doi:10.1007/s00419-023-02496-z. [Google Scholar] [CrossRef]
6. Mandal B, Shit GC. Blood flow through a deformable artery and magnetically targeted drug delivery. J Magn Magn Mater. 2025;629(1):173325. doi:10.1016/j.jmmm.2025.173325. [Google Scholar] [CrossRef]
7. Abdelsalam SI, Mekheimer KS, Zaher AZ. Alterations in blood stream by electroosmotic forces of hybrid nanofluid through diseased artery: aneurysmal/stenosed segment. Chin J Phys. 2020;67(11):314–29. doi:10.1016/j.cjph.2020.07.011. [Google Scholar] [CrossRef]
8. Zaman A, Ali N, Khan AA. Computational biomedical simulations of hybrid nanoparticles on unsteady blood hemodynamics in a stenotic artery. Math Comput Simul. 2020;169(1):117–32. doi:10.1016/j.matcom.2019.09.010. [Google Scholar] [CrossRef]
9. Sridhar V, Ramesh K, Gnaneswara Reddy M, Azese MN, Abdelsalam SI. On the entropy optimization of hemodynamic peristaltic pumping of a nanofluid with geometry effects. Waves Random Complex Medium. 2025;35(3):4403–23. doi:10.1080/17455030.2022.2061747. [Google Scholar] [CrossRef]
10. Zhang L, Bhatti MM, Marin M, Mekheimer SK. Entropy analysis on the blood flow through anisotropically tapered arteries filled with magnetic zinc-oxide (ZnO) nanoparticles. Entropy. 2020;22(10):1070. doi:10.3390/e22101070. [Google Scholar] [PubMed] [CrossRef]
11. Chen J, Sun Y, Hu D, Yao H, Shen X, Zhang C, et al. Performance modeling and mechanism study of proton exchange membrane water electrolyzer coupled with water electroosmosis. Energy Convers Manag. 2024;315:118753. doi:10.1016/j.enconman.2024.118753. [Google Scholar] [CrossRef]
12. Al Nuwairan M, Souayeh B. Simulation of gold nanoparticle transport during MHD electroosmotic flow in a peristaltic micro-channel for biomedical treatment. Micromachines. 2022;13(3):374. doi:10.3390/mi13030374. [Google Scholar] [PubMed] [CrossRef]
13. Irfan M, Siddique I, Nazeer M, Ali W. Theoretical study of silver nanoparticle suspension in electroosmosis flow through a nonuniform divergent channel with compliant walls: a therapeutic application. Alex Eng J. 2024;86(1):443–57. doi:10.1016/j.aej.2023.11.083. [Google Scholar] [CrossRef]
14. Gubbiotti A, Baldelli M, Di Muccio G, Malgaretti P, Marbach S, Chinappi M. Electroosmosis in nanopores: computational methods and technological applications. Adv Phys X. 2022;7(1):2036638. doi:10.1080/23746149.2022.2036638. [Google Scholar] [CrossRef]
15. Baldelli M, Di Muccio G, Sauciuc A, Morozzo Della Rocca B, Viola F, Balme S, et al. Controlling electroosmosis in nanopores without altering the nanopore sensing region. Adv Mater. 2024;36(33):e2401761. doi:10.1002/adma.202401761. [Google Scholar] [PubMed] [CrossRef]
16. Awad AM, Mekheimer KS, Elkilany SA, Zaher AZ. Leveraging elasticity of blood stenosis to detect the role of a non-Newtonian flow midst an arterial tube: mazumdar and Keller models. Chin J Phys. 2022;77(2):2520–40. doi:10.1016/j.cjph.2022.04.006. [Google Scholar] [PubMed] [CrossRef]
17. Abd-Alla AM, Abo-Dahab SM, Salah DM, Bayones FS, Abdelhafez MA. Magneto-hydrodynamic peristaltic flow of a Jeffery fluid in the presence of heat transfer through a porous medium in an asymmetric channel. Sci Rep. 2023;13(1):21088. doi:10.1038/s41598-023-48137-x. [Google Scholar] [PubMed] [CrossRef]
18. Akbar NS, Akram J, Hussain MF, Maraj EN, Muhammad T. Thermal storage study and enhancement of heat transfer through hybrid Jeffrey nanofluid flow in ducts under peristaltic motion with entropy generation. Therm Sci Eng Prog. 2024;49(4):102463. doi:10.1016/j.tsep.2024.102463. [Google Scholar] [CrossRef]
19. Pandey A, Jangid P, Tripathi D, Sharma K. Membrane pumping-driven electroosmotic flow of Casson fluid in inclined microchannel. Discov Appl Sci. 2025;7(10):1147. doi:10.1007/s42452-025-07735-2. [Google Scholar] [CrossRef]
20. Dolui S, Bhaumik B, De S, Changdar S. Nanoparticle aggregation and electro-osmotic propulsion in peristaltic transport of third-grade nanofluids through porous tube. Comput Biol Med. 2024;176(4):108617. doi:10.1016/j.compbiomed.2024.108617. [Google Scholar] [PubMed] [CrossRef]
21. Abdelmoneim M, Eldabe NT, Abouzeid MY, Ouaf ME. Modified Darcy’s law and couple stress effects on electro-osmotic flow of non-Newtonian nanofluid with peristalsis. Int J Appl Electromagn Mech. 2023;72(3):253–77. doi:10.3233/jae-220287. [Google Scholar] [CrossRef]
22. Abbasi A, Mabood F, Farooq W, Khan SU. Radiation and joule heating effects on electroosmosis-modulated peristaltic flow of Prandtl nanofluid via tapered channel. Int Commun Heat Mass Transf. 2021;123:105183. doi:10.1016/j.icheatmasstransfer.2021.105183. [Google Scholar] [CrossRef]
23. Ajmal M, Mehmood R, Akbar NS, Muhammad T. Electroosmotic base magnetic field study of peristaltic transport in a non-uniform wavy ciliated channel for Williamson fluid with enhancing nanoparticles. ZAMM J Appl Math Mech/Z Für Angew Math Und Mech. 2024;104(12):e202400296. doi:10.1002/zamm.202400296. [Google Scholar] [CrossRef]
24. Eldabe NT, Abouzeid MY, Abdelmoneim MM, Ouaf ME. Impacts of activation energy and electroosmosis on peristaltic motion of micropolar Newtonian nanofluid inside a microchannel. Mod Phys Lett B. 2025;39(3):2450407. doi:10.1142/s0217984924504074. [Google Scholar] [CrossRef]
25. Yadav PK, Singh A. Biomedical simulations of electroosmotic non-Newtonian hybrid nanofluid (blood) with hematocrit viscosity through a porous overlapping irregular stenosed artery. Comput Biol Med. 2025;196(Pt B):110575. doi:10.1016/j.compbiomed.2025.110575. [Google Scholar] [PubMed] [CrossRef]
26. Paul P, Robel FN, Bahadur NM, Tabassum S, Ahmed MF, Dey SS, et al. Amplification of photocatalytic and antimicrobial activities with eco-friendly prepared ZnO nanoparticles. Hybrid Adv. 2025;11:100557. doi:10.1016/j.hybadv.2025.100557. [Google Scholar] [CrossRef]
27. Akbar NS, Maraj EN, Ijaz S, Muhammad T, Hussain MF, Habib MB, et al. Artificial neural network analysis for entropy-driven electroosmotic blood cell transport with drug-loaded ZnO nanoparticles in a stenosed tapered artery. Int Commun Heat Mass Transf. 2025;169(1):109786. doi:10.1016/j.icheatmasstransfer.2025.109786. [Google Scholar] [CrossRef]
28. Akbar NS, Maraj EN, Ijaz S, Habib MB, Muhammad T, Jahan Z, et al. Artificial Neural Network (ANN) optimized entropy ornamented effects on electroosmotic blood cell manipulation with drugged interacting zinc oxide nanoparticles in an anisotropically stenosed tapered artery. Int Commun Heat Mass Transf. 2026;171(4):110085. doi:10.1016/j.icheatmasstransfer.2025.110085. [Google Scholar] [CrossRef]
29. Gomathi N, Poulomi D. Entropy optimization on EMHD Casson Williamson penta-hybrid nanofluid over porous exponentially vertical cone. Alex Eng J. 2024;108(5):590–610. doi:10.1016/j.aej.2024.07.092. [Google Scholar] [CrossRef]
30. Rammoorthi R, Mohanavel D. Entropy generation and sensitivity analysis for a squeezed flow of a Williamson hybrid nanofluid under the supervision of microcantilever sensor. Results Eng. 2024;24(5–6):103464. doi:10.1016/j.rineng.2024.103464. [Google Scholar] [CrossRef]
31. Jyothi N, Avula Golla VK. Study of radiative heat and mass intensification with magnetic field for Casson and Williamson nanofluid flow model over a porous stretching sheet with higher-order chemical reaction: application of solar energy. J Therm Anal Calorim. 2024;149(13):6783–96. doi:10.1007/s10973-023-12536-z. [Google Scholar] [CrossRef]
32. Supriya P, Pullepu B, Chamkha AJ, Senthil R. Numerical approach of unsteady free convective Casson–Williamson nanofluid flow over the radiative vertical cone surface with cross-diffusion. Int J Mod Phys B. 2025;39(17):2550150. doi:10.1142/s0217979225501504. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools