 Open Access
Open Access
ARTICLE
LASENet: BiLSTM-Attention-SE Network for High-Precision sEMG-Based Shoulder Joint Angle Prediction
College of Computer Science, Beijing University of Technology, Beijing, China
* Corresponding Author: Dan Wang. Email:
Computers, Materials & Continua 2026, 87(3), 46 https://doi.org/10.32604/cmc.2026.074554
Received 13 October 2025; Accepted 09 January 2026; Issue published 09 April 2026
Abstract
Accurate prediction of shoulder joint angles based on surface electromyography (sEMG) signals is critical in human–machine interaction and rehabilitation engineering. However, due to the shoulder joint’s complex degrees of freedom, dynamically varying muscle coordination patterns, and the susceptibility of sEMG signals to cross-talk and noise interference, achieving high-precision prediction remains challenging. In this study, LASENet (BiLSTM–Attention–SE Network) is proposed as an end-to-end deep learning framework that integrates a bidirectional long short-term memory network (BiLSTM), a multi-head self-attention (MHSA) mechanism, and a squeeze-and-excitation (SE) block to predict shoulder joint angles across three degrees of freedom directly from raw sEMG signals. By jointly modeling temporal dependencies, long-range feature interactions, and channel-wise importance, LASENet effectively captures motion-related patterns while suppressing redundant noise. Experimental results demonstrate that LASENet demonstrates outperforms baseline models in terms of root mean square error (RMSE) and correlation coefficient (CC), achieving superior prediction accuracy and stability. These findings demonstrate that LASENet is an effective solution for accurate shoulder joint angle prediction from sEMG signals.Keywords
Surface electromyography (sEMG) represents the summation of electrical activity generated by motor neurons that innervate skeletal muscle fibers, recorded non-invasively on the skin surface. It directly reflects muscle contraction and neuromuscular activation, making it a key signal for decoding upper-limb motion intentions [1,2].
Compared with traditional motion capture techniques—such as inertial measurement units (IMUs) or optical tracking systems—sEMG offers unique advantages. sEMG signals precede observable motion onset by tens of milliseconds, providing a temporal lead over kinematics-based sensors and enabling predictive control in real-time systems [3,4]. Moreover, sEMG demonstrates superior wearability and environmental robustness compared to optical systems [5]. It does not depend on external lighting conditions or line-of-sight visibility, maintaining stable performance even under visual occlusion or complex indoor environments [6]. Consequently, sEMG provides a safe, non-invasive, and intention-driven control signal for applications such as exoskeletons, prosthesis control, and robotic teleoperation [7–9].
Despite these advantages, significant challenges remain in decoding shoulder joint motion from sEMG signals due to the joint’s complex anatomy and signal characteristics. In contrast to single degrees of freedom (DOF) joints such as the elbow or wrist, the shoulder is among the most flexible and biomechanically intricate joints in the human body. It is composed of multiple sub-joints—including the acromioclavicular, sternoclavicular, scapulothoracic, and glenohumeral joints—that act in concert to achieve three rotational degrees of freedom: flexion/extension, abduction/adduction, and internal/external rotation [10,11]. This high degree of freedom necessitates dynamic coordination among multiple muscle groups rather than isolated activation of individual muscles [12]. At the signal level, the dense spatial distribution and overlap of shoulder muscles cause significant cross-talk and signal mixing among recording channels [13,14]. As a result, conventional motion recognition methods—typically designed for single-joint control—struggle to generalize to the shoulder, where accurate extraction and decoding of multi-DOF motion intentions remain an open and challenging problem in sEMG research.
To address these issues, researchers have increasingly applied deep learning techniques to continuous motion estimation and state recognition tasks for the upper limb and shoulder. Ma et al. proposed a bidirectional long short-term memory (BiLSTM)-based decoding framework for continuous upper-limb motion estimation from sEMG signals, which effectively captured the temporal dependencies of muscle activity and achieved high prediction accuracy for both elbow and shoulder angles [15]. Wen et al. introduced the SCA-LSTM model, combining sparse component analysis (SCA) with LSTM to estimate continuous joint angles [16]. While SCA-LSTM effectively suppressed redundant information through signal decomposition, its performance relied heavily on handcrafted preprocessing modules, limiting its end-to-end trainability and flexibility.
Recent studies have explored the incorporation of attention and transformer mechanisms to enhance temporal and inter-channel modeling in shoulder motion decoding. Bai et al. proposed SWCTNet, a model that integrates convolutional neural networks (CNNs) with channel–temporal attention-based transformers, using multimodal IMU and sEMG data to predict muscle activation and shoulder motion dynamics, achieving superior performance in rehabilitation scenarios [17]. Another study adopted a transformer-based framework that fused multiple biosignals to predict rotational accelerations of the shoulder and elbow, demonstrating improved accuracy and stability over traditional LSTM and recurrent neural network (RNN) models [18].
In addition to architectural advances, channel optimization has also been explored as an effective strategy to enhance recognition performance. Bai et al. employed 64-channel high-density sEMG recordings of shoulder movements and applied multiclass common spatial pattern (CSP) and wavelet packet decomposition for spatial–spectral feature extraction and channel selection. Their study demonstrated that retaining only 4–5 optimized channels after CSP filtering could achieve over 92% classification accuracy for shoulder motion patterns [19]. However, such feature-dependent channel selection relies heavily on prior expert knowledge, and the designed strategy must often be reconfigured when task conditions or motion classes change, limiting generalizability.
To overcome these limitations, LASENet (BiLSTM–Attention–SE Network) is proposed, an end-to-end deep learning model for shoulder joint angle prediction during single-degree-of-freedom isokinetic tasks. LASENet employs BiLSTM to extract deep temporal representations directly from raw sEMG signals and integrates a multi-head self-attention mechanism to enhance cross-channel and temporal feature perception, thereby enabling high-dimensional decoding of coordinated multi-muscle activation patterns and establishing a precise nonlinear mapping from sEMG to shoulder kinematics. Crucially, LASENet incorporates a squeeze-and-excitation (SE) block to dynamically weight informative channels and suppress redundant noise features, maintaining strong representational capacity even under high cross-talk or low electrode density conditions. By directly mapping raw sEMG inputs to three shoulder joint angles—flexion/extension, abduction/adduction, and internal/external rotation—through end-to-end training, LASENet eliminates the need for extensive feature engineering or manual preprocessing. Experimental results demonstrate that LASENet consistently outperforms baseline models in root mean square error (RMSE) and correlation coefficient (CC), achieving superior accuracy and stability in multi-DOF periodic shoulder motion prediction.
In this study, LASENet (BiLSTM–Attention–SE Network) is proposed, a deep learning architecture that integrates multidimensional temporal feature extraction with channel-adaptive enhancement mechanisms (Fig. 1). The model is designed to improve both the accuracy and stability of shoulder joint angle prediction based on sEMG signals. LASENet primarily consists of three key modules: a BiLSTM network, a multi-head self-attention (MHSA) mechanism, and a SE block, followed by a multi-layer perceptron (MLP) for continuous joint angle regression.
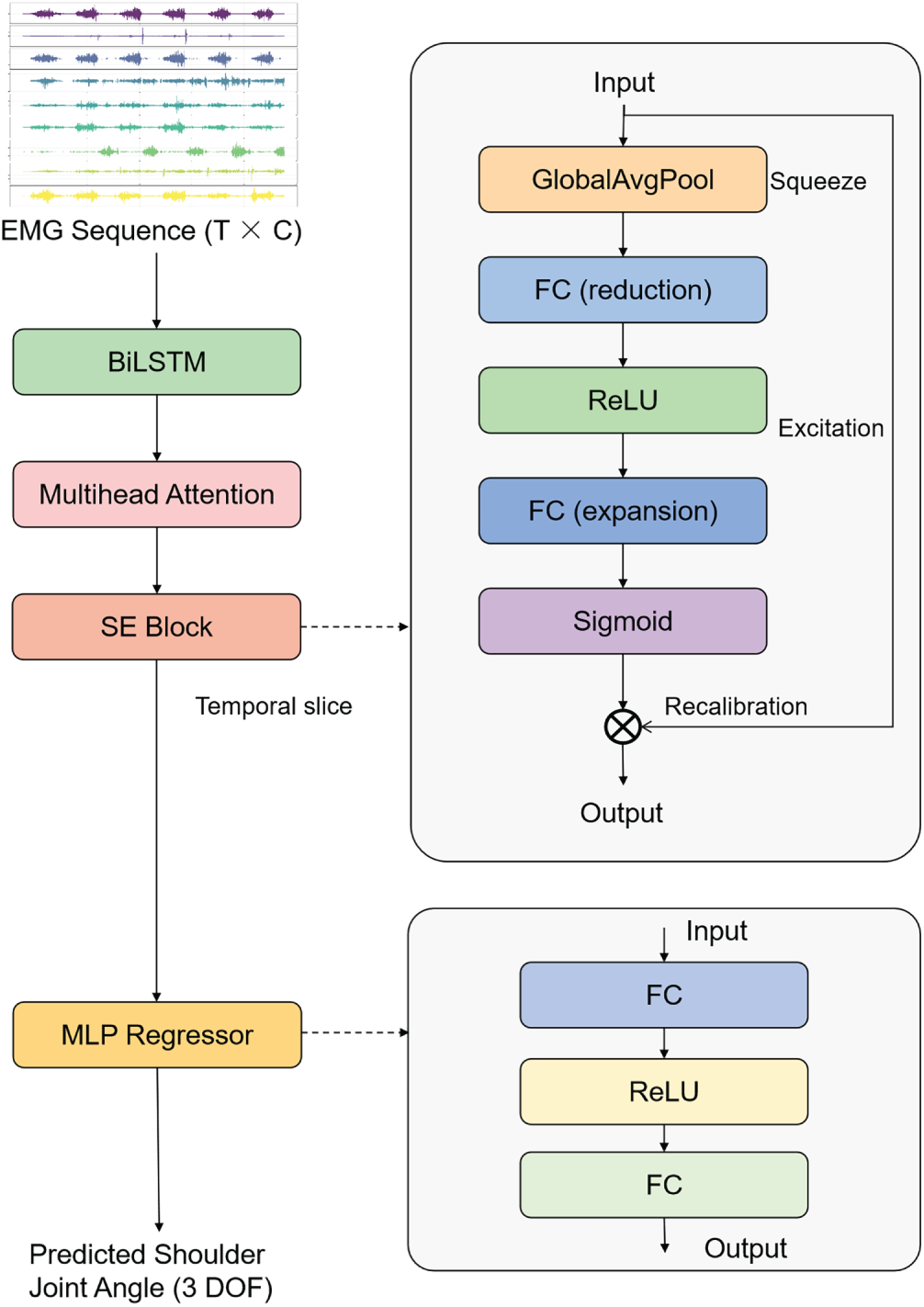
Figure 1: Overall architecture of the proposed LASENet.
In the feature extraction stage, a BiLSTM network with 256 hidden units in each direction (resulting in a 512-dimensional bidirectional output), introducing 273,408 trainable parameters and accounting for 18.8% of the total model size, is employed to capture the temporal dependencies and inter-muscle coordination patterns inherent in sEMG signals. Processing sequences in both forward and backward directions enables the model to construct a more comprehensive representation of the temporal dynamics underlying muscle activation. Subsequently, a MHSA module is introduced, configured with an embedding dimension of 512 and 32 attention heads, contributing 1,048,576 trainable parameters (72.2% of the total model size), primarily arising from the linear projection matrices of the attention mechanism, which allows the network to model long-range temporal dependencies across time steps. This attention mechanism enables adaptive weighting of salient temporal segments that contribute most to motion transition cues, thereby improving feature discriminability and enhancing the model’s generalization capability across various dynamic shoulder joint angle movement patterns.
Furthermore, the SE block explicitly models inter-channel correlations through a squeeze–excitation operation with a reduction ratio r = 8, contributing 65,536 trainable parameters (4.5% of the total model size) from the two fully connected layers. Given a 512-dimensional feature channel, global average pooling first compresses the temporal dimension to produce a compact channel descriptor of size 512. This descriptor is then passed through two fully connected layers, undergoing a dimensionality reduction from 512
Finally, the feature representation from the last time step is fed into a regression MLP composed of two fully connected layers (512
Through coordinated optimization across three levels—temporal modeling, dependency representation, and channel weighting—LASENet effectively exploits the motion intention information embedded within sEMG signals. This architecture provides an efficient and physiologically interpretable deep learning framework for continuous shoulder joint angle prediction.
The SE Block is an efficient channel attention mechanism designed to explicitly model inter-channel dependencies and enhance feature representation through adaptive recalibration. First proposed by Hu et al. [20], this module has been widely adopted in various domains due to its simplicity and superior performance, including computer vision [21] and temporal signal modeling [22,23].
As illustrated in Fig. 2, the SE Block operates through three main stages: Squeeze, Excitation, and Recalibration.
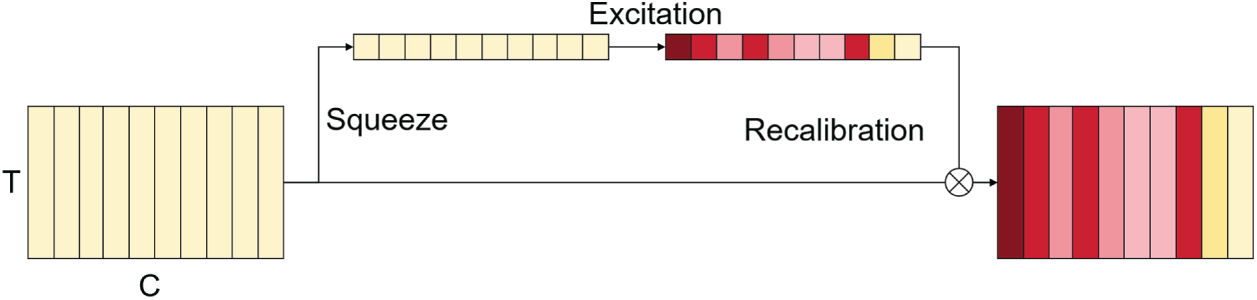
Figure 2: Structure of the SE block.
In the Squeeze stage, the input feature map
where T denotes the number of time steps, C represents the number of channels, and
In the Excitation stage, channel-wise dependencies are modeled through a two-layer fully connected gating mechanism, as defined in Eq. (2). The first layer performs dimensionality reduction followed by a nonlinear transformation using the ReLU activation function, while the second layer restores the original channel dimension and applies a Sigmoid function to generate normalized channel weights:
where
Finally, in the Recalibration stage, the obtained channel weights
where
Through this squeeze–excitation recalibration mechanism, the SE block adaptively reweights channel-wise features, enhancing the representation of informative muscle activation patterns while suppressing redundant or noisy channels, with minimal additional computational overhead. In this study, the SE Block is integrated after the MHSA module to adaptively enhance high-dimensional feature representations at the channel level, further improving feature selection and information utilization efficiency in sEMG-based modeling.
A total of twelve healthy male subjects aged between 18 and 25 years were recruited for this study. All subjects underwent a comprehensive health screening prior to the experiment to ensure the absence of upper-limb motor dysfunction, neuromuscular disorders, or any recent shoulder injuries or treatments. Informed consent was obtained from all subjects before data collection.
The shoulder joint exhibits three rotational degrees of freedom, which are generated through coordinated contractions and torque coupling of the relevant muscle groups. For shoulder flexion/extension, the Anterior Deltoid and Biceps Brachii primarily drive flexion, whereas extension is mainly powered by the Posterior Deltoid and Latissimus Dorsi. Meanwhile, the Pectoralis Major provides auxiliary force and stabilizes the joint during anteriorly directed movements. The Lateral Triceps contributes to the shoulder–elbow extension synergy, and the Brachioradialis reflects elbow–forearm stabilization in shoulder-driven tasks, thereby capturing upper-limb dynamic coupling patterns closely related to shoulder kinematics. In shoulder abduction/adduction, the Middle Deltoid serves as the primary mover for abduction, while adduction is mainly generated by the Pectoralis Major and Latissimus Dorsi, which produce forces toward the trunk. With respect to shoulder internal/external rotation, the Infraspinatus is the key muscle for external rotation, whereas internal rotation is primarily mediated by the Pectoralis Major and Latissimus Dorsi, with the Posterior Deltoid playing a crucial role in rotational stability. The functional differentiation and coordinated activation of these muscles form the physiological basis for the sEMG acquisition in this study [24].
Based on this functional understanding of the selected muscles, sEMG signals were recorded using a multi-channel acquisition system (Noraxon USA Inc., Scottsdale, AZ, USA). Electrodes were placed on several key muscle groups of the right upper limb, as detailed in Table 1. Prior to electrode placement, the skin was carefully prepared by shaving, exfoliating, and cleaning with alcohol to reduce skin impedance and improve signal quality [25]. The sEMG sampling frequency was set to 2000 Hz, and raw signals were processed using a 10–500 Hz band-pass filter to retain the main physiological frequency components, along with a 50 Hz notch filter to remove power-line interference.

The motion tasks were performed using an isokinetic dynamometer (BIODEX System, Biodex Medical Systems, Shirley, NY, USA), which can be connected to the sEMG system host to generate rising-edge trigger signals, enabling synchronized acquisition between the dynamometer and the sEMG system. Subjects were seated and securely fixed with straps across the trunk, waist, and the non-tested arm to minimize compensatory movements, ensuring that only single-degree-of-freedom shoulder motions were executed during testing. The experimental tasks consisted of shoulder flexion/extension, abduction/adduction, and internal/external rotation (as shown in Fig. 3). Each movement was conducted at three angular velocities—
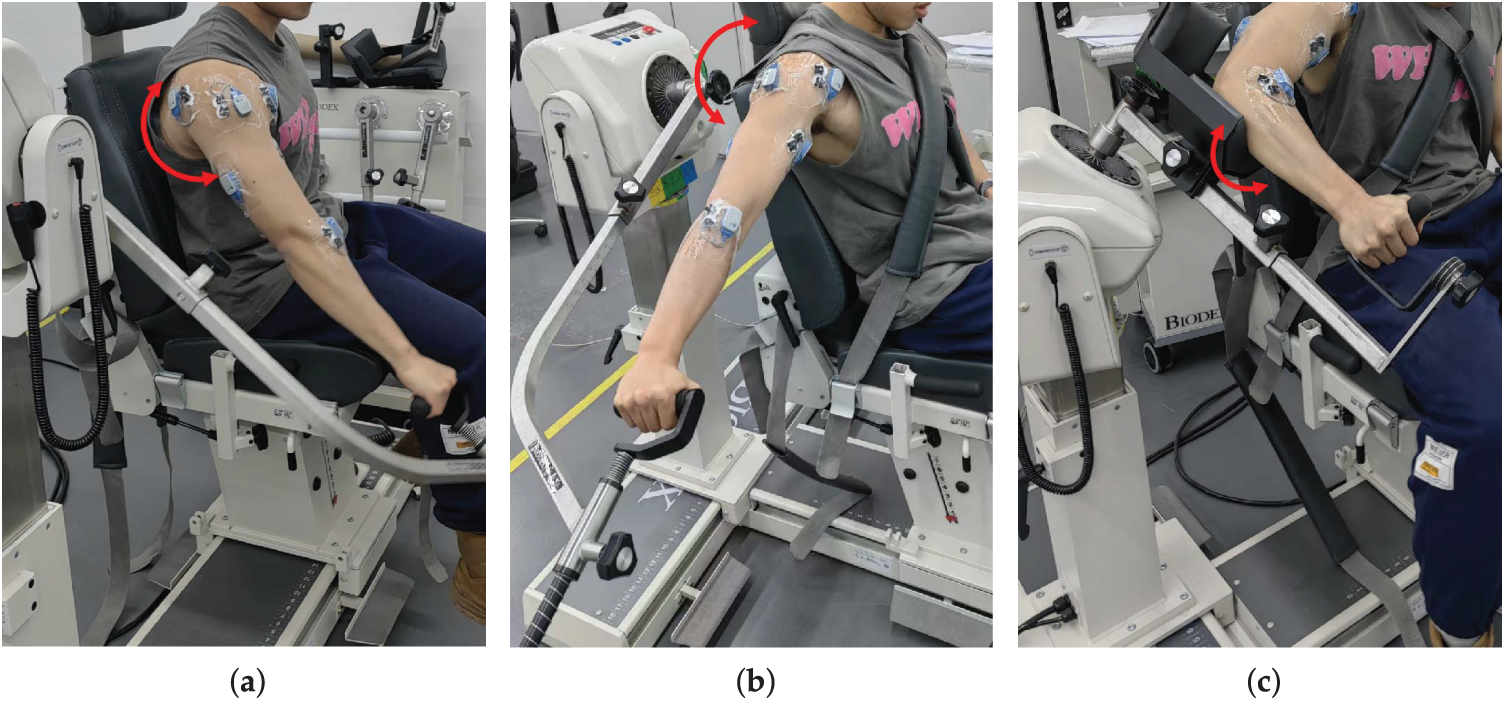
Figure 3: Experimental setup for joint angle collection: (a) shoulder flexion/extension; (b) shoulder abduction/adduction; (c) shoulder internal/external rotation.
Multichannel sEMG signals (9 channels) were segmented using a sliding window approach to construct the dataset, with a window length of 200 ms and 50% overlap (i.e., 100 ms step size). This window length balances temporal resolution and signal stationarity, providing sufficient information on muscle activation while avoiding excessive fluctuations or noise, as supported by previous studies on sEMG-based control [26,27]. The 50% overlap increases the number of training samples, better captures the continuous variations in movement, and reduces information loss at window boundaries. For each window, the 9-channel, 200 ms sEMG segment was used as the input feature x, and the joint angle corresponding to the final time point of the window was taken as the supervisory signal y. After segmentation, each sEMG channel was independently standardized using z-score normalization, with the mean and standard deviation computed exclusively from the training set after cross-validation splitting (see Section 2.5). These statistics were subsequently applied to the validation and test sets, ensuring consistent normalization while preventing data leakage and distribution shift [28].
This study employed a subject-wise leave-one-turn-out (LOTO) cross-validation strategy, which is a structured variant of the conventional leave-one-out (LOO) approach, where the held-out unit corresponds to a trial (turn) rather than a sample. Each “task–speed” combination was defined as an independent movement pattern. Accordingly, each subject had a total of 3 (tasks)
The optimization objective was defined using the Smooth L1 Loss (Huber Loss), which behaves as the Mean Squared Error (MSE) for small residuals to enhance convergence stability, and transitions to the Mean Absolute Error (MAE) for larger residuals, effectively reducing the impact of outliers on model optimization [29]. The Adam optimizer was adopted with an initial learning rate of
To evaluate the effectiveness of the proposed LASENet model in shoulder joint angle prediction and to quantify the contribution of each component module, a series of systematic ablation experiments were conducted. Several representative deep learning architectures were compared, including the baseline BiLSTM, Transformer, BiLSTM with Attention, and the proposed LASENet model integrating both Attention and SE modules. Tables 2 and 3 present the correlation coefficient (CC) and root mean square error (RMSE) results, respectively, for the X-axis (flexion/extension), Y-axis (abduction/adduction), and Z-axis (internal/external rotation) of the shoulder joint.
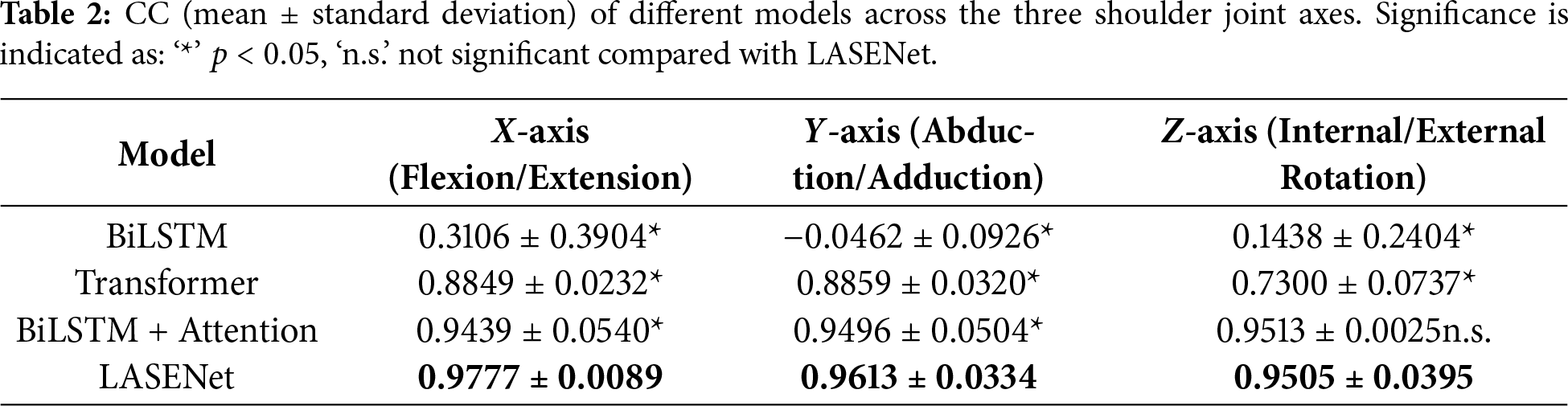
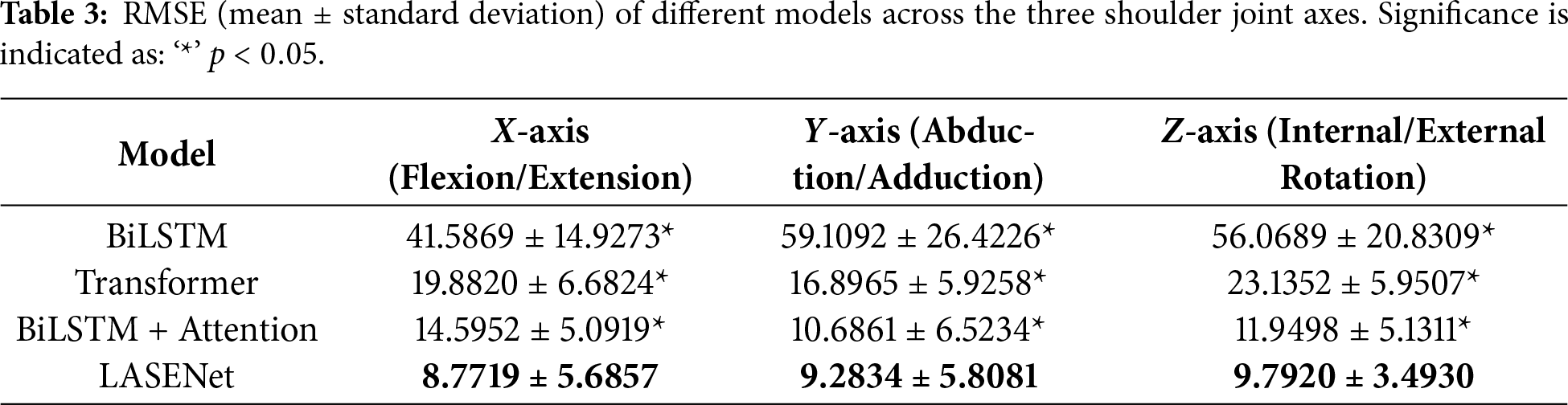
As shown in Table 2, LASENet (BiLSTM + Attention + SE) achieved the highest correlation across all three axes, with CC values of 0.9777, 0.9613, and 0.9505 for the X, Y, and Z directions, respectively. All differences between LASENet and the other models were statistically significant (
The RMSE results in Table 3 further confirm the superiority of LASENet, which achieved the lowest prediction errors of
Overall, LASENet achieved consistently high correlation and low prediction error across all three degrees of freedom, demonstrating its superiority in capturing multi-muscle coordination and cross-channel dynamic features. By integrating the bidirectional temporal dependency modeling of BiLSTM, the temporal saliency extraction capability of the Attention mechanism, and the channel-wise adaptive enhancement of the SE block, LASENet effectively establishes a more accurate mapping between sEMG signals and kinematic parameters, validating its strong predictive performance and robustness.
To examine training stability and potential overfitting, we evaluated LASENet’s total parameter count (1,453,571) in the context of the effective training set per LOTO round (68,890 windows), noting that each window is a high-dimensional segment of 9 channels
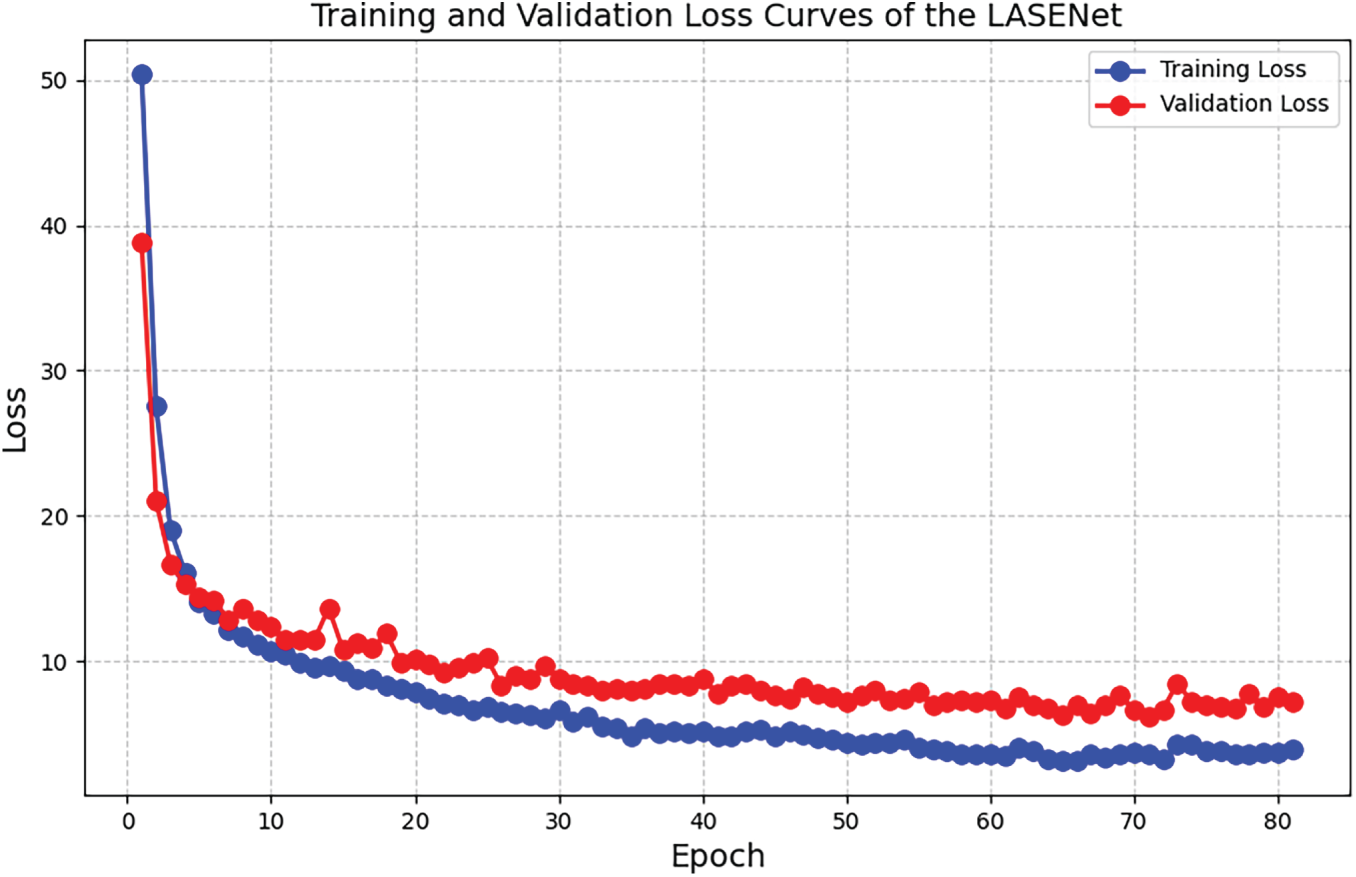
Figure 4: Training and validation loss curves of the LASENet across epochs.
4.1 Model Mechanism and Performance Analysis
In this study, LASENet demonstrated substantially superior performance over all comparative models in predicting shoulder joint angles across three degrees of freedom—flexion/extension, abduction/adduction, and internal/external rotation. The model achieved high correlation and low prediction error in all directions, benefiting from its multi-level fusion architecture and adaptive enhancement mechanisms. Compared with the conventional BiLSTM model, LASENet not only captures bidirectional temporal dependencies in sEMG signals but also leverages the attention mechanism to focus on critical temporal segments during motion transitions. This enables the model to more effectively represent the dynamic dependencies among muscle groups during coordinated contractions. This design philosophy aligns with findings by Abdelhady et al. [32], who proposed a GRU + Attention model, and Zangene et al. [33], who developed an Attention–BiLSTM approach—both confirming that attention mechanisms can significantly enhance accuracy and stability in continuous joint angle estimation. Similarly, Lv et al. [34] verified the efficacy of temporal attention for handling nonstationary biosignals in knee joint angle prediction.
To provide a more intuitive comparison of model performance, the results from one representative subject were selected, comparing three models with relatively high correlation—Transformer, BiLSTM+Attention, and LASENet. As shown in Fig. 5, the prediction curves of LASENet (subplots g–i) align most closely with the actual joint angles across all three degrees of freedom. Particularly during motion direction transitions, LASENet produces smoother trajectories with reduced latency compared with the Transformer (subplots a–c) and BiLSTM+Att (subplots d–f) models.
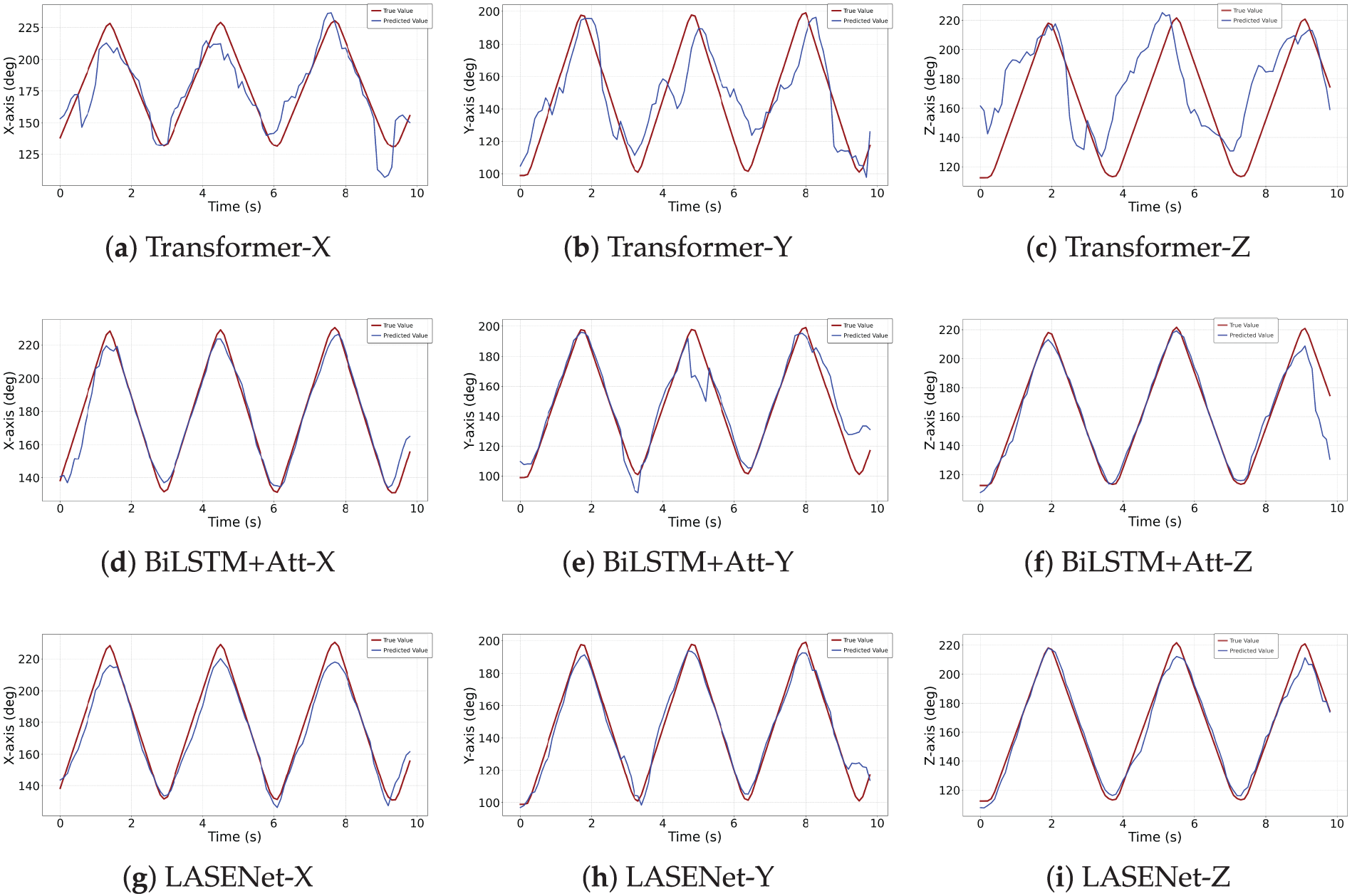
Figure 5: Comparison between predicted and actual shoulder joint angles for each model across three motion axes: (X) flexion/extension, (Y) abduction/adduction, and (Z) internal/external rotation.
It is worth noting that the baseline BiLSTM achieved significantly low CC and high RMSE values in this study, likely due to the absence of explicit feature engineering. While BiLSTM has demonstrated good performance in other upper-limb joint angle prediction studies, its success largely depended on handcrafted features, such as RMS or median frequency (MDF) representations derived from variational mode decomposition (VMD) and wavelet packet transform (WPT). In contrast, LASENet attains high correlation and low prediction errors directly from raw sEMG signals, highlighting its capability to learn robust temporal and cross-channel dependencies without relying on feature engineering. This comparison further underscores the effectiveness of integrating bidirectional temporal modeling, attention-based temporal saliency, and channel-wise adaptive enhancement in capturing complex neuromuscular dynamics.
However, relying solely on temporal feature extraction remains insufficient to address the spatial coupling and redundancy present in shoulder sEMG signals. To overcome this, LASENet incorporates a Squeeze-and-Excitation (SE) module, which performs adaptive feature reweighting through a “squeeze–excitation–recalibration” process. This mechanism dynamically amplifies signals from key muscle channels while suppressing redundant or noisy information, thereby enhancing cross-muscle feature discrimination. When originally proposed for visual recognition, Hu et al. [20] validated the SE module’s feature enhancement capability. Similarly, Xu et al. [35] integrated SE with attention mechanisms for sEMG-based gesture recognition and confirmed the effectiveness of channel recalibration for modeling complex biosignals. The present study further demonstrates that the SE module significantly improves prediction stability and feature separability when processing multi-muscle signals. Compared with conventional approaches that rely on hand-crafted time–frequency or statistical features [36–38], LASENet learns joint temporal–channel–spatial representations directly from raw sEMG data via end-to-end training, enhancing modeling accuracy while removing reliance on manually designed features, which may introduce subjectivity and variability. Overall, the BiLSTM–Attention–SE architecture effectively models temporal, inter-channel, and spatial dependencies in sEMG signals, offering physiologically informed interpretability and robust performance for high-degree-of-freedom upper-limb motion decoding.
4.2 Physiological Interpretation of Prediction Difficulty across Axes
Although LASENet substantially improves overall prediction accuracy, the distribution of errors across different motion directions reveals that prediction along the Z-axis (internal/external rotation) remains more challenging than along the X-axis (flexion/extension) and Y-axis (abduction/adduction). This trend, consistent across all compared models, reflects both the physiological complexity of shoulder rotation and the high noise characteristics of sEMG signals. Specifically, internal/external rotations are primarily driven by deep rotator cuff muscles such as the infraspinatus, teres minor, and subscapularis. These muscles are small in volume, produce low-amplitude signals, and exhibit high spatial overlap, resulting in substantial inter-muscle crosstalk and reduced signal-to-noise ratio (SNR), which diminish model discriminability [31,39,40]. Additionally, the shoulder’s rotational axis shifts dynamically during movement due to the coupled motion of the humerus and scapula, causing significant variation in muscle activation patterns at different joint postures. This further increases the nonlinear complexity of the mapping between sEMG and joint kinematics [41,42]. Despite these challenges, LASENet achieved an RMSE of
This study proposes a deep learning architecture—LASENet (BiLSTM-Attention-SE Network)—for multi-degree-of-freedom shoulder joint angle prediction based on sEMG. The proposed model integrates temporal modeling and channel-adaptive enhancement mechanisms by combining a BiLSTM network, a MHSA mechanism, and a SE Block. Through this synergistic design, LASENet achieves a coordinated fusion of temporal dependency modeling, global feature aggregation, and channel-level feature optimization. By adopting an end-to-end learning strategy, the model automatically extracts multi-level dynamic features related to shoulder motion directly from raw sEMG signals, effectively eliminating the dependence on handcrafted feature engineering and extensive signal preprocessing required by traditional approaches.
Experimental results demonstrate that LASENet demonstrates superior performance relative to other models—including BiLSTM, Transformer, and BiLSTM+Attention—in predicting shoulder joint angles across three degrees of freedom: flexion/extension, abduction/adduction, and internal/external rotation. Specifically, LASENet achieves the highest correlation coefficients (CC) and the lowest root mean square errors (RMSE) across all axes, validating its superior performance under conditions of high channel crosstalk and low signal-to-noise ratio. The findings further confirm that the incorporation of the SE module enhances the model’s ability to adaptively identify and reweight channel importance, thereby strengthening its sensitivity to key muscle activations while suppressing redundant noise—significantly improving recognition of coordinated activation patterns among multiple shoulder muscles.
In summary, the proposed LASENet model achieves breakthroughs in both prediction accuracy and robustness while providing a physiologically interpretable and scalable framework for modeling sEMG signals in high-degree-of-freedom joint motion. Future work will further explore the potential of LASENet in multimodal fusion (e.g., combining sEMG with IMU or EEG signals) and personalized model transfer, to facilitate its practical deployment in intelligent rehabilitation, exoskeleton control, and neuroengineering applications.
Despite strong performance across various dynamic shoulder joint angle movement patterns, this study is limited to within-subject evaluations, leaving cross-subject generalization untested. Future work will explore domain adaptation or transfer learning for inter-subject scenarios and incorporate multimodal signals to improve accuracy and robustness, aiming for personalized, generalizable sEMG-based joint angle estimation.
Acknowledgement: Thanks to all the members of our research group, their suggestions and support have provided important help and profound impact on our research work. Additionally, we confirm that AI-assisted tools were used solely for language polishing and grammatical improvement to enhance clarity and readability. No AI tools were used for structural modification, data analysis, experimental design, model development, result interpretation, or scientific decision-making. All intellectual content, methodologies, and conclusions are entirely the work of the authors.
Funding Statement: The authors received no specific funding.
Author Contributions: The authors confirm contribution to the paper as follows: Study conception and design: Dan Wang and Ruida Liu; Analysis of results: Ruida Liu and Meng Xu; Draft manuscript preparation: Ruida Liu and Jiaming Chen. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: Data available on request from the authors.
Ethics Approval: All subjects provided written informed consent prior to the experiment. This study does not involve any human or animal clinical trials.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Merletti R, Farina D. Surface electromyography: physiology, engineering, and applications. Hoboken, NJ, USA: John Wiley & Sons; 2016. doi:10.1002/9781119082934. [Google Scholar] [CrossRef]
2. Avila ER, Williams SE, Disselhorst-Klug C. Advances in EMG measurement techniques, analysis procedures, and the impact of muscle mechanics on future requirements for the methodology. J Biomech. 2023;156(1):111687. doi:10.1016/j.jbiomech.2023.111687. [Google Scholar] [PubMed] [CrossRef]
3. Guo W, Sun Y, Xu Y, Qiao Z, Yang Y, Xiong H. SpGesture: source-free domain-adaptive sEMG-based gesture recognition with jaccard attentive spiking neural network. Adv Neural Inf Process Syst. 2024;37:36717–47. doi:10.52202/079017-1157. [Google Scholar] [CrossRef]
4. Xu D, Wang Q. Noninvasive human-prosthesis interfaces for locomotion intent recognition: a review. Cyborg Bionic Syst. 2021;2021(3):9863761. doi:10.34133/2021/9863761. [Google Scholar] [PubMed] [CrossRef]
5. Suo X, Tang W, Li Z. Motion capture technology in sports scenarios: a survey. Sensors. 2024;24(9):2947. doi:10.3390/s24092947. [Google Scholar] [PubMed] [CrossRef]
6. Salter S, Warren R, Schlager C, Spurr A, Han S, Bhasin R, et al. emg2pose: a large and diverse benchmark for surface electromyographic hand pose estimation. Adv Neural Inf Process Syst. 2024;37:55703–28. doi:10.52202/079017-1770. [Google Scholar] [CrossRef]
7. Chen B, Zhou Y, Chen C, Sayeed Z, Hu J, Qi J, et al. Volitional control of upper-limb exoskeleton empowered by EMG sensors and machine learning computing. Array. 2023;17:100277. doi:10.1016/j.array.2023.100277. [Google Scholar] [CrossRef]
8. Shah G, Sharma A, Joshi D, Rathor AS. Revolutionizing prosthetic hand control using non-invasive sensors and intelligent algorithms: a comprehensive review. Comput Electr Eng. 2025;123(3):110094. doi:10.1016/j.compeleceng.2025.110094. [Google Scholar] [CrossRef]
9. Song T, Yan Z, Guo S, Li Y, Li X, Xi F. Review of sEMG for robot control: techniques and applications. Appl Sci. 2023;13(17):9546. [Google Scholar]
10. Menze J, Croci E, Andersen MS, Hess H, Lund ME, De Pieri E, et al. Advancing musculoskeletal shoulder modeling: reflecting glenohumeral translation with bony, ligamentous, and muscular stability constraints. Front Bioeng Biotechnol. 2025;13:1441530. doi:10.3389/fbioe.2025.1441530. [Google Scholar] [PubMed] [CrossRef]
11. Dalla Pria P. Biomechanics of the shoulder joint. In: Human orthopaedic biomechanics. Amsterdam, The Netherlands: Elsevier; 2022. p. 285–303. doi:10.1016/b978-0-12-824481-4.00017-2. [Google Scholar] [CrossRef]
12. Kuniki M, Yokoyama H, Konishi R, Iwamoto Y, Yamagiwa D, Kuwahara D, et al. Shoulder kinematics and muscle synergy during multi-plane humeral elevation and lowering. J Biomech. 2025;186(2):112735. doi:10.1016/j.jbiomech.2025.112735. [Google Scholar] [PubMed] [CrossRef]
13. Bedoy EH, Guirola Diaz EA, Dalrymple AN, Levy I, Hyatt T, Griffin DM, et al. Improving localization and measurements of M-waves using high-density surface electromyography. J Neurophysiol. 2025;133(1):299–309. doi:10.1152/jn.00354.2024. [Google Scholar] [PubMed] [CrossRef]
14. Arghadeh R, Alizadeh MH, Minoonejad H, Sheikhhoseini R, Asgari M, Jaitner T. Electromyography of shoulder muscles in individuals without scapular dyskinesis during closed kinetic chain exercises on stable and unstable surfaces: a systematic review and meta-analysis. Front Sports Act Living. 2024;6:1385693. doi:10.3389/fspor.2024.1385693. [Google Scholar] [PubMed] [CrossRef]
15. Ma C, Lin C, Samuel OW, Guo W, Zhang H, Greenwald S, et al. A bi-directional LSTM network for estimating continuous upper limb movement from surface electromyography. IEEE Robot Autom Lett. 2021;6(4):7217–24. doi:10.1109/lra.2021.3097272. [Google Scholar] [CrossRef]
16. Wen L, Xu J, Li D, Pei X, Wang J. Continuous estimation of upper limb joint angle from sEMG based on multiple decomposition feature and BiLSTM network. Biomed Signal Process Control. 2023;80(4):104303. doi:10.1016/j.bspc.2022.104303. [Google Scholar] [CrossRef]
17. Bai A, Song H, Wu Y, Dong S, Feng G, Jin H. Sliding-window CNN + channel-time attention transformer network trained with inertial measurement units and surface electromyography data for the prediction of muscle activation and motion dynamics leveraging IMU-only wearables for home-based shoulder rehabilitation. Sensors. 2025;25(4):1275. doi:10.3390/s25041275. [Google Scholar] [PubMed] [CrossRef]
18. Bai Y, Guan X, He L, Wang Z, Li Z, Zhu M. Estimating rotational acceleration in shoulder and elbow joints using a transformer algorithm and a fusion of biosignals. Sensors. 2024;24(6):1726. doi:10.3390/s24061726. [Google Scholar] [PubMed] [CrossRef]
19. Bai D, Chen S, Yang J. Upper arm motion high-density sEMG recognition optimization based on spatial and time-frequency domain features. J Healthc Eng. 2019;2019(4):3958029. doi:10.1155/2019/3958029. [Google Scholar] [PubMed] [CrossRef]
20. Hu J, Shen L, Sun G. Squeeze-and-excitation networks. In: Proceedings of the 2018 IEEE/CVF Conference on Computer Vision and Pattern Recognition; 2018 Jun 18–23; Salt Lake City, UT, USA. p. 7132–41. doi:10.1109/cvpr.2018.00745. [Google Scholar] [CrossRef]
21. Mei K, Jiang A, Li J, Ye J, Wang M. An effective single-image super-resolution model using squeeze-and-excitation networks. In: Neural information processing. Cham, Switzerland: Springer International Publishing; 2018. p. 542–53. doi:10.1007/978-3-030-04224-0. [Google Scholar] [CrossRef]
22. Li Y, Liu Y, Cui WG, Guo YZ, Huang H, Hu ZY. Epileptic seizure detection in EEG signals using a unified temporal-spectral squeeze-and-excitation network. IEEE Trans Neural Syst Rehabil Eng. 2020;28(4):782–94. doi:10.1109/tnsre.2020.2973434. [Google Scholar] [PubMed] [CrossRef]
23. Chen W, Shi K. Multi-scale attention convolutional neural network for time series classification. Neural Netw. 2021;136(2):126–40. doi:10.1016/j.neunet.2021.01.001. [Google Scholar] [PubMed] [CrossRef]
24. Neumann DA, Kelly ER. Kinesiology of the musculoskeletal system: foundations for rehabilitation. London, UK: Mosby; 2010. [Google Scholar]
25. Stjerna S, Alatalo P, Mäki J, Vanhatalo S. Evaluation of an easy, standardized and clinically practical method (SurePrep) for the preparation of electrode-skin contact in neurophysiological recordings. Physiol Meas. 2010;31(7):889–901. doi:10.1088/0967-3334/31/7/002. [Google Scholar] [PubMed] [CrossRef]
26. Smith LH, Hargrove LJ, Lock BA, Kuiken TA. Determining the optimal window length for pattern recognition-based myoelectric control: balancing the competing effects of classification error and controller delay. IEEE Trans Neural Syst Rehabil Eng. 2011;19(2):186–92. doi:10.1109/tnsre.2010.2100828. [Google Scholar] [PubMed] [CrossRef]
27. Nazmi N, Abdul Rahman M, Yamamoto SI, Ahmad S, Malarvili MB, Mazlan S, et al. Assessment on stationarity of EMG signals with different windows size during isotonic contractions. Appl Sci. 2017;7(10):1050. doi:10.3390/app7101050. [Google Scholar] [CrossRef]
28. Al-Faiz MZ, Ibrahim AA, Hadi SM. The effect of Z-Score standardization (normalization) on binary input due the speed of learning in back-propagation neural network. Iraqi J Inf Commun Technol. 2019;1(3):42–8. doi:10.31987/ijict.1.3.41. [Google Scholar] [CrossRef]
29. Wang Q, Ma Y, Zhao K, Tian Y. A comprehensive survey of loss functions in machine learning. Ann Data Sci. 2022;9(2):187–212. doi:10.1007/s40745-020-00253-5. [Google Scholar] [CrossRef]
30. Al-Kababji A, Bensaali F, Dakua SP. Scheduling techniques for liver segmentation: ReduceLRonPlateau vs. OneCycleLR. In: Intelligent systems and pattern recognition. Cham, Switzerland: Springer International Publishing; 2022. p. 204–12. doi:10.1007/978-3-031-08277-1. [Google Scholar] [CrossRef]
31. Barron SM, Ordonez Diaz T, Pozzi F, Vasilopoulos T, Nichols JA. Linear relationship between electromyography and shear wave elastography measurements persists in deep muscles of the upper extremity. J Electromyogr Kinesiol. 2022;63(1):102645. doi:10.1016/j.jelekin.2022.102645. [Google Scholar] [PubMed] [CrossRef]
32. Abdelhady M, Damiano DL, Bulea TC. Attention-based deep recurrent neural network to estimate knee angle during walking from lower-limb EMG. In: Proceedings of the 2023 International Conference on Rehabilitation Robotics (ICORR); 2023 Sep 24–28; Singapore. p. 1–6. doi:10.1109/icorr58425.2023.10304604. [Google Scholar] [PubMed] [CrossRef]
33. Zangene AR, Williams Samuel O, Abbasi A, Nazarpour K, McEwan AA, Li G. An attention-based bidirectional LSTM model for continuous cross-subject estimation of knee joint angle during running from sEMG signals. In: Proceedings of the 2023 45th Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC); 2023 Jul 24–27; Sydney, Australia. p. 1–4. doi:10.1109/embc40787.2023.10340791. [Google Scholar] [PubMed] [CrossRef]
34. Lv J, Huang B, Qiang L. Continuous prediction of knee joint angle in lower limbs based on sEMG: a method combining an improved ZOA optimizer and attention-enhanced GRU. J King Saud Univ Comput Inf Sci. 2025;37(6):149. doi:10.1007/s44443-025-00164-6. [Google Scholar] [CrossRef]
35. Xu Z, Yu J, Xiang W, Zhu S, Hussain M, Liu B, et al. A novel SE-CNN attention architecture for sEMG-based hand gesture recognition. Comput Model Eng Sci. 2023;134(1):157–77. doi:10.32604/cmes.2022.020035. [Google Scholar] [CrossRef]
36. Huang Y, Chen K, Zhang X, Wang K, Ota J. Motion estimation of elbow joint from sEMG using continuous wavelet transform and back propagation neural networks. Biomed Signal Process Control. 2021;68(5):102657. doi:10.1016/j.bspc.2021.102657. [Google Scholar] [CrossRef]
37. Liang J, Shi Z, Zhu F, Chen W, Chen X, Li Y. Gaussian process autoregression for joint angle prediction based on sEMG signals. Front Public Health. 2021;9:685596. doi:10.3389/fpubh.2021.685596. [Google Scholar] [PubMed] [CrossRef]
38. Lu Z, Chen S, Yang J, Liu C, Zhao H. Prediction of lower limb joint angles from surface electromyography using XGBoost. Expert Syst Appl. 2025;264(6):125930. doi:10.1016/j.eswa.2024.125930. [Google Scholar] [CrossRef]
39. Backus SI, Tomlinson DP, Vanadurongwan B, Lenhoff MW, Cordasco FA, Chehab EL, et al. A spectral analysis of rotator cuff musculature electromyographic activity: surface and indwelling. HSS J. 2011;7(1):21–8. doi:10.1007/s11420-010-9178-8. [Google Scholar] [PubMed] [CrossRef]
40. Kim D, Park S. Selective activation of the subscapularis muscle: a cross-sectional observational study. Healthcare. 2025;13(11):1349. doi:10.3390/healthcare13111349. [Google Scholar] [PubMed] [CrossRef]
41. Temporiti F, Furone R, Cescon C, Barbero M, Gatti R. Dispersion of helical axes during shoulder movements in young and elderly subjects. J Biomech. 2019;88(15):72–7. doi:10.1016/j.jbiomech.2019.03.018. [Google Scholar] [PubMed] [CrossRef]
42. Williamson PM, Hanna P, Momenzadeh K, Lechtig A, Okajima S, Ramappa AJ, et al. Effect of rotator cuff muscle activation on glenohumeral kinematics: a cadaveric study. J Biomech. 2020;105:109798. doi:10.1016/j.jbiomech.2020.109798. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools