 Open Access
Open Access
REVIEW
CO2 Capture in Construction Materials: Review of Uptake Approaches and Energy Considerations
1 c5Lab–Sustainable Construction Materials Association, Rua Central Park 6, 2795-242 Linda-a-Velha, Portugal
2 CERENA–Centro de Recursos Naturais e Ambiente, Instituto Superior Técnico, Av. Rovisco Pais 1, 1049-001 Lisbon, Portugal
3 LNEC–National Laboratory for Civil Engineering, Av. do Brasil 101, 1700-066 Lisbon, Portugal
4 CERIS–Instituto Superior Técnico, University of Lisbon, Av. Rovisco Pais, 1049-001 Lisbon, Portugal
* Corresponding Author: Cinthia Maia Pederneiras. Email:
(This article belongs to the Special Issue: Advancing Carbon Mitigation Strategies for a Sustainable Future)
Energy Engineering 2026, 123(4), 6 https://doi.org/10.32604/ee.2026.074246
Received 06 October 2025; Accepted 17 December 2025; Issue published 27 March 2026
Abstract
The construction industry is a significant contributor to global CO2 emissions, and urgent innovation is needed to mitigate its environmental impact. This paper provides a comprehensive review of scalable approaches for CO2 uptake in construction materials, including the injection of CO2 into fresh concrete, the CO2 curing of precast concrete, and the use of ceramics as CO2 sinks. Among these three approaches, CO2 curing methods for concrete represent the most advanced and widely adopted strategies within industrial practice, with substantial research supporting their effectiveness and scalability. The comparison of carbonation mineralisation across three distinct material groups reveals that the direct injection of CO2 into fresh concrete mixes results in CO2 uptake of less than 3 kg/m3. For the precast concrete elements, the CO2 uptake ranges from 30 to 350 kg/m3, while ceramics can achieve uptake efficiencies up to 23 wt.% under pilot-scale conditions. Achieving efficient CO2 uptake in fresh and precast concrete without compromising mechanical properties relies on precise control over the CO2 dose, a tailored mix design, and optimised curing conditions, while avoiding excessive carbonation that could reduce alkalinity or durability. Valorisation of carbonated materials as supplementary cementitious components or aggregates is identified as an important circular solution, though further research is needed to address regeneration, performance, and standardisation. The review highlights ongoing gaps in life-cycle assessment and industrial-scale validation, and recommends future work on durability and techno-economic optimisation for robust decarbonisation in the cement and concrete industries.Graphic Abstract

Keywords
The cement industry accounts for approximately 8% of global anthropogenic CO2 emissions, primarily due to limestone calcination (50%–60% of emissions), fossil fuel combustion for high-temperature processing (30%–40% of emissions), and electricity consumption (≈20% of emissions). Meeting carbon-neutrality targets by 2050, in accordance with international climate commitments, requires emission reductions throughout the cement production chain from raw material extraction to the end-of-life of cement-based structures. In response, the cement industry has adopted multiple decarbonization strategies, including the partial substitution of clinker with alternative materials such as Supplementary Cementitious Materials (SCMs) [1,2], development of alternative binders such as Limestone Calcined Clay Cement (LC3) [3,4], and alkali-activated binders [5], and recycled cement [6]. Additional measures include the use of alternative fuels, the electrification of production processes [7,8], and the implementation of Carbon Capture and Utilisation (CCU) technologies [9]. Still, the urgent need highlights the demand for effective CO2 sinks to mitigate carbon emissions from the cement industry. It is known that cementitious materials naturally absorb atmospheric CO2 through slow carbonation reactions with hydrated cement phases [10]. The natural carbonation process starts at the exposed surface and gradually moves inward in a slow, diffusion-controlled manner [11]. Since the carbonated layer reduces CO2 diffusion and limits the inherent carbonation potential of cement-based materials, the carbonation is initially restricted to a relatively thin layer near the exposed surface [12]. Therefore, the carbonation rate is insufficient to deliver a substantial reduction in emissions within the immediate timelines for achieving carbon neutrality [13]. Accelerated carbonation, by contrast, intentionally intensifies this reaction under controlled conditions, enabling rapid and considerable CO2 uptake and positioning this approach as a practical short-term decarbonization pathway for the cement industry [14,15].
In this context, the present review aims to identify the most effective methods for CO2 uptake using construction material, addressing the urgent need to reduce the cement industry’s carbon footprint. CO2 uptake using concrete with a specific focus on (i) CO2 injection into fresh concrete and (ii) CO2 curing of concrete will be evaluated. Additionally, the potential of ceramic waste materials as a considerable fraction of construction and demolition waste (CDW) in CO2 sequestration will be assessed. Moreover, this review assesses the industrial adaptations required for each CO2 capture technology, focusing on the process modifications needed for large-scale implementation while accounting for both technical and economic factors. The final goal and key innovation of this work is to identify sustainable, actionable strategies that can significantly reduce the environmental impact of cement production by integrating CO2 absorption into the cementitious product throughout the value chain. It is worth noting that, among the three techniques evaluated in this review, the use of concrete for CO2 capture is the most extensively investigated, whereas the other approaches are more recent and remain less explored.
2 Methodology of Literature Review
As mentioned previously, this work aims to identify the most effective methods for CO2 uptake using construction materials, with a specific focus on CO2 injection into fresh concrete, CO2 curing of concrete, and CO2 absorption by ceramic waste materials.
Therefore, first, the literature searches focused on the mechanism of CO2 capture by cementitious materials. Then, advances in CO2 capture using cementitious materials were explored to cover all innovative solutions in the field. Lastly, the industrial implementation of the three techniques was covered. The literature exploration was conducted using the Web of Science, Scopus, and Google Scholar databases, focusing on original research published between 2000 and 2025 that reported quantitative or qualitative data on CO2 sequestration in cementitious materials, concrete, or ceramics. Search keywords included combinations of CO2 capture, CO2 curing, CO2 uptake, CO2 sequestration, CO2 absorption mechanism, chemical absorption, CO2 capture efficiency, cement industry, concrete industry, fresh mix, fresh concrete, concrete curing, and ceramic waste. As a result, a wide spectrum of references using CO2 mineralisation methods has been retrieved for cementitious materials, including accelerated carbonation of aggregates, direct aqueous carbonation of industrial byproducts and waste, bio-mineralisation via microorganisms, end-of-life concrete carbonation, CO2 capture and mineralisation by ceramics, especially via incorporating ceramic demolition waste into cementitious systems, and enforced carbonation during mixing. To follow the objective of this review, the inclusion criteria were studies that directly investigated cementitious or ceramic-based materials for CO2 capture, sequestration, or curing. Exclusion criteria comprised works unrelated to concrete/ceramics, unrelated to CO2 capture, or lacking outcomes relevant to sequestration performance. After removing duplications, the initial search yielded a set of 350 documents linking to the subject. A two-step screening process was applied. First, titles, abstracts, and conclusions were screened for relevance. Second, an accurate classification process began, in which whole-text articles were assessed against the inclusion and exclusion criteria. A literature search using more specific keywords was repeated during the writing and revision processes whenever contradictory or unclear statements were encountered. This careful evaluation yielded a refined set of 129 research studies related to the topic; all published between 2000 and 2025. Of these, 62.5% were published between 2020 and 2025, 23% between 2015 and 2019, and only 7.5% between 2000 and 2014.
As mentioned previously, cement and concrete can naturally absorb atmospheric CO2 through slow carbonation reactions. CO2 uptake by concrete over its service life contributes directly to the reduction of greenhouse gas emissions and, in theory, enhances material strength by decreasing porosity [10]. Nevertheless, according to research carried out in Nordic countries, it would take 100 years to store 33%–57% of the CO2 emissions from cement production in the year 2003, by natural carbonation [16]. Therefore, forced carbonation technologies in which cementitious materials are exposed to elevated concentrations of CO2 are considered a more viable approach to contribute to short-term climate change mitigation than natural carbonation and have a positive impact on reducing CO2 emissions [17,18].
Two established pathways for utilising CO2 as a feedstock in concrete are: (i) injection of CO2 into fresh concrete during the mixing stage, and (ii) curing of precast concrete in a CO2-rich atmosphere. In both processes, the gaseous CO2 is diffused and dissolved into the alkaline pore solution. The dissolved CO2 further hydrolyses to bicarbonate (HCO3−) and carbonate (CO32−) ions, which then react with calcium ions (Ca2+) released from hydrates into the pore solution and predominantly convert into solid calcium carbonate (CaCO3). The carbonation of calcium hydroxide (Ca(OH)2), which is generated during the hydration of tricalcium silicate (C3S) and dicalcium silicate (C2S) phases in Portland cement shown in Eq. (1) [10].
CO2 can also react with calcium silicate hydrates (C–S–H), a primary product of cement hydration responsible for the strength and durability of concrete, forming silica gel according to Eq. (2) [16].
Likewise, CO2 react with unhydrated C3S and C2S, resulting at formation of CaCO3 and silica gel according to the Eqs. (3) and (4) [10].
It is worth mentioning that the reactions described by Eqs. (2)–(4) may adversely affect the mechanical properties of concrete, as the excessive precipitation of CaCO3 within the pore structure may increase porosity or provoke the formation of microcracks. Moreover, early-age exposure to CO2, when hydration is still in progress, can disrupt matrix development, restrict the formation of essential hydration products such as C–S–H, and lead to incomplete microstructural development [19,20]. Thus, precise control of parameters such as CO2 partial pressure, temperature, relative humidity, exposure duration, and timing relative to hydration is crucial to balance CO2 uptake with material quality. The influence of these parameters will be addressed in detail in the following chapters, providing a framework for optimising CO2 curing to maximise both environmental and mechanical benefits. Fig. 1 shows the schematic representation of the carbonation reaction in cementitious materials.
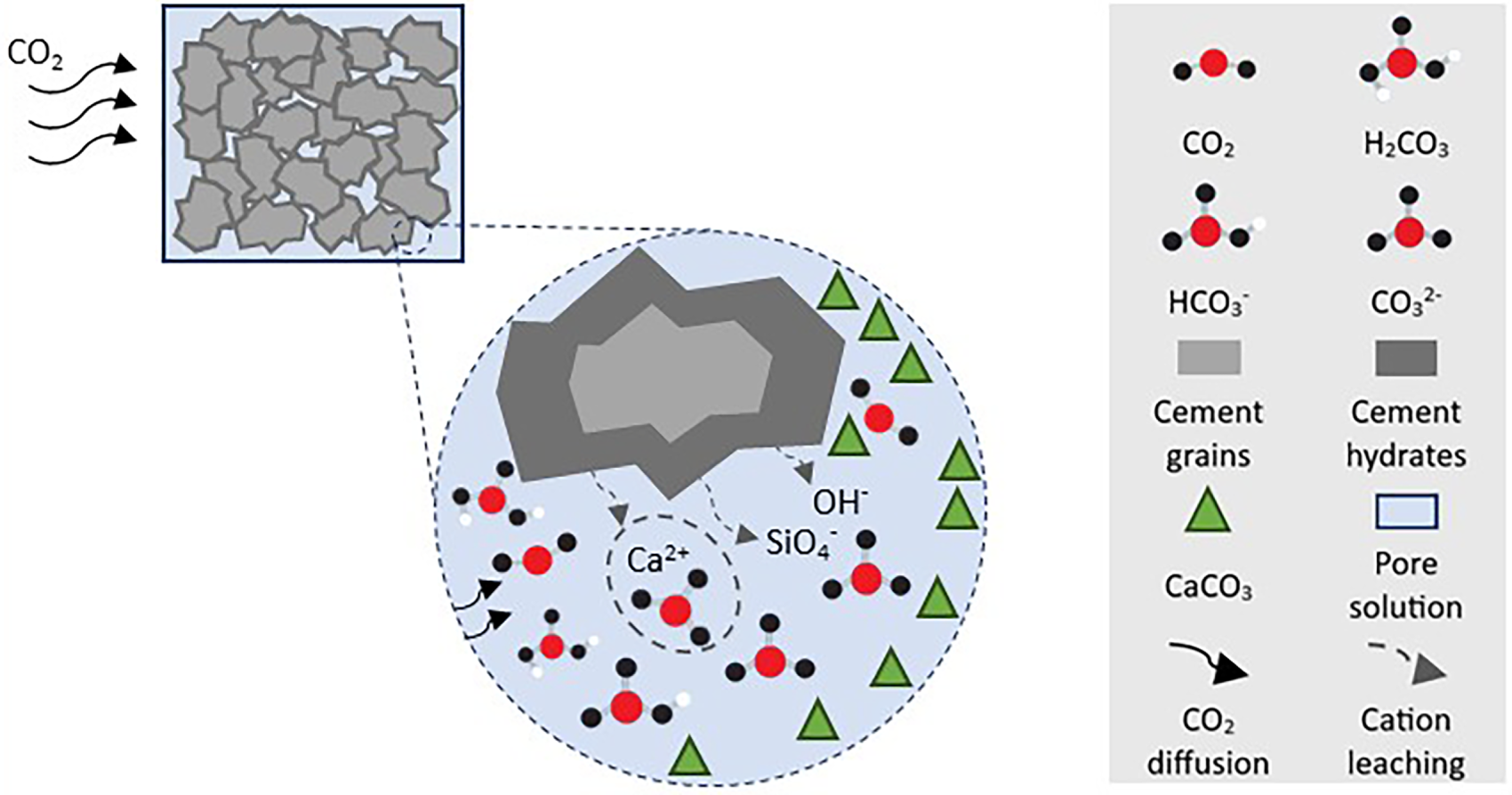
Figure 1: Schematic representation of carbonation mechanism in cementitious materials
3.1 CO2 Injection into Fresh Concrete
CO2 injection into fresh concrete or mortar during mixing creates a carbonating environment for cementitious materials. Continuous stirring of the fresh mix in the mixer enhances reagent interaction, facilitating more efficient CO2 diffusion by eliminating porous solid barriers that form during CaCO3 precipitation, thereby increasing the rate of CO2 absorption [14]. To inject CO2 into freshly mixed cement-based materials, the literature reports the use of carbonated water (Fig. 2a), in which CO2 is initially dissolved in water, and then used to prepare the cementitious mixture [21,22]. Nevertheless, since CO2 is poorly soluble in water, the expected CO2 uptake by concrete is low, limiting the contribution of this approach to the cement industry’s carbon neutrality. To compensate for the low CO2 uptake by water, Kwasny et al. [22] and Fu et al. [23] added an extra CO2 injection step, such as injecting CO2 into a cement suspension or a concrete mix. However, the most common method involves injecting CO2 directly into the cementitious mixture during the mixing stage (Fig. 2b) [24,25], resulting in the formation of amorphous nanocrystalline CaCO3 [26].
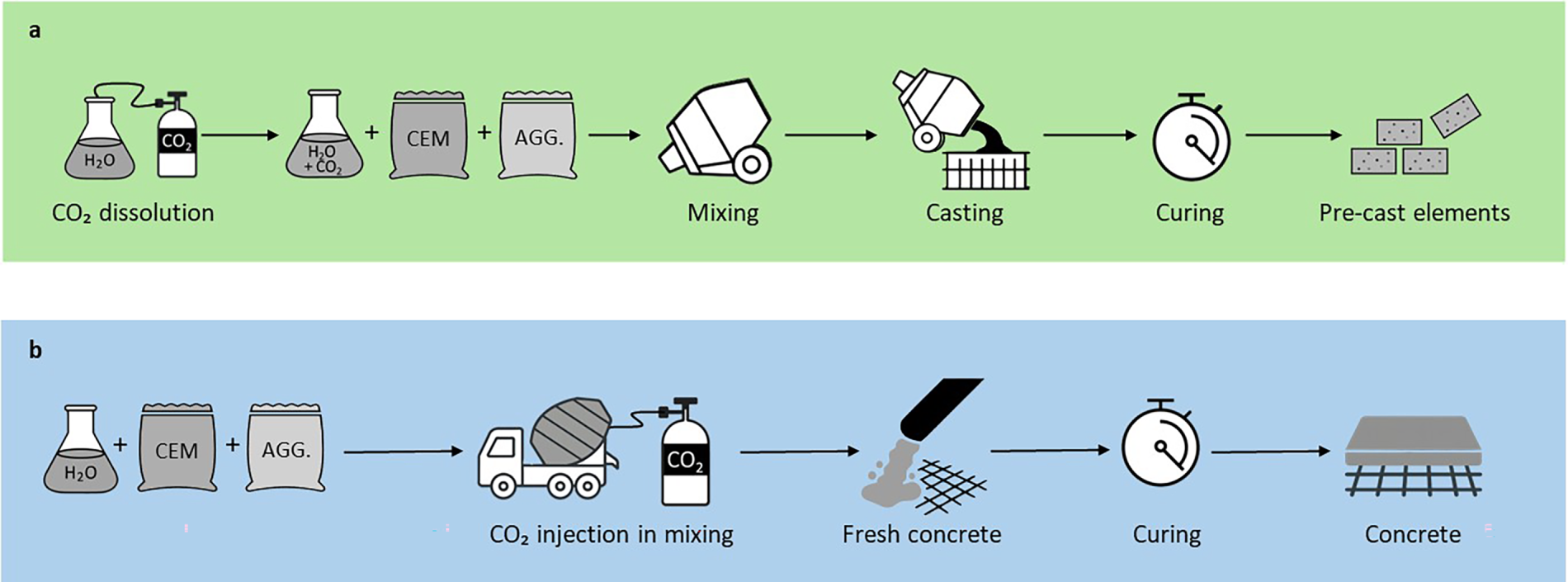
Figure 2: Schematic representation of CO2 injection into fresh concrete: (a) dissolution of CO2 in water, and (b) direct injection of CO2 into the fresh mix
The formation of these amorphous CaCO3 accelerates the hydration reaction and increases the compressive strength [23,25]. The acceleration of the hydration reaction during CO2 injection into fresh concrete has been explained through three key mechanisms: (i) the filler effect, (ii) the nucleation effect, and (iii) the chemical effect [26]. The filler effect involves rapid CO2 mineralisation, producing fine CaCO3 particles that occupy nano-scale pores in the concrete mix, improving microstructural density and potentially increasing durability. The nucleation effect occurs as these CaCO3 particles provide additional nucleation sites for hydration products, such as C–S–H, thereby accelerating the development of early-age strength and hydration kinetics. The chemical effect involves CO32− ions participating in cement hydration reactions, which results in a reduced calcium-to-silicon (Ca/Si) ratio within the C–S–H gel. Additionally, these CO32− ions promote the formation of carboaluminate phases through their reaction with aluminate-bearing components in the cement matrix [27]. When CO2 dosage and timing are carefully optimised, these mechanisms collectively improve hydration efficiency, microstructural quality, and early strength, while reducing the overall carbon footprint of concrete production [26]. According to Zajac et al. [28] while only a small amount of CO2 is stored in concrete (0.1–1 wt.%), the increased strength allows for a reduction in cement content [23,24,26], which indirectly reduces the carbon footprint of concrete production. Nevertheless, some contradictory results have been reported regarding the reduction in strength after CO2 injection [29]. According to Zajac et al. [28] and Silva et al. [30], the carbonation of fresh concrete should sequester less than 1 wt.% of CO2, as higher CO2 uptake would negatively impact the hydraulic reactivity and performance of the hardened concrete [28,30]. The reasons for the low CO2 uptake and negative effect on the mechanical properties in such technology have been discussed recently by Silva et al. [30]. Based on this study, low CO2 absorption during mixing is attributed to the fact that, at this initial stage, the primary cement hydration products, particularly Ca(OH)2, which is the most reactive with CO2, are not yet formed in significant quantities. As a result, freshly mixed concrete primarily contains unhydrated clinker phases with limited direct reactivity toward CO2. The same authors explain that the reduction in mechanical properties observed when CO2 is injected into fresh concrete is related to changes in cement hydration and particle morphology. Exposure of anhydrous cement to CO2 in the presence of water accelerates the dissolution of small unhydrated particles (mostly C2S), resulting in the complete consumption of a larger portion of the finer particles. This particle redistribution produces a coarser pore structure, which compromises the paste’s early-age mechanical performance, although partial strength recovery can occur at later curing stages [30]. Therefore, the low CO2 absorption of CO2 injection into the fresh mix is the major drawback of this strategy.
Winnefeld et al. [31] reported the possibility of using the same approach to storing CO2 in the concrete slurry waste. Concrete slurry waste generated during the cleaning of concrete mixers and trucks contains high levels of reactive calcium-bearing phases such as Ca(OH)2 and partially hydrated cement, making it a suitable feedstock for mineral carbonation. This composition enables 12% CO2 storage per solid content in concrete slurry waste (≈0.8% CO2 absorption in the liquid slurry) by converting these phases into stable CaCO3. Winnefeld et al. [31] also reported that the carbonated concrete slurry waste was reused as a SCM, partially replacing Portland cement in new mixes and delivering dual benefits of permanent CO2 immobilisation and reduced clinker demand, while also offering some improvements in hydration and early strength. Sim et al. [32] reported that the supercritical CO2 process demonstrated that near-complete carbonation of concrete slurry waste can be achieved rapidly (≤30 min) at a solids content of approximately 5%, producing a stable, carbon-rich sludge that can be dried and blended into fresh concrete. Despite these advantages, both studies highlight a critical limitation: concrete slurry waste accounts for only 1%–3% of total ready-mix output, so while the process is technically viable and aligns with carbon-neutrality goals, its large-scale impact on cement-sector decarbonisation is constrained by limited feedstock availability [31,32]. Despite the low CO2 uptake of CO2 injection into the fresh mix as a major drawback of this strategy, commercialised technologies for this approach have been reported by CarbonCure [24,26].
CarbonCure ready-mix technology injects a precise amount of recycled CO2 (captured from industrial operations) into fresh concrete during mixing, after approximately half of the cement and water contents have been discharged. The liquified CO2, when injected into the mix, reacts with the cement compounds, forming nano-sized CaCO3 particles that become permanently bound within cementitious material, allowing it to mineralise while preserving its properties [26]. Both CO2 dosage control and the timing of CO2 injection are key factors in CarbonCure’s success in CO2 injection into the fresh mix, without compromising the mechanical properties or negatively affecting the high alkalinity required for steel reinforcement protection [26,33]. Using CarbonCure technology, the CO2 uptake based on cement mass was approximately 1.4 wt.%. Overall, the technology is reported to save 7–11 kg of CO2 per cubic meter (kg/m3) of ready-mix concrete, 12–18 kg/m3 of precast concrete, and 0.5 kg of CO2 per 30 standard masonry blocks [34]. Additionally, Zhang et al. [16] and Monkman et al. [35] have been claimed that upon carbonation, the fast formation of nano-CaCO3 results in the shorter initial setting of concrete by 40% and improves the 1- and 3-day strength by 14% and 10%, respectively. Nonetheless, at 28 days, CO2 curing can reduce the compressive strength of concrete compared with conventional curing, as reported by Ravikumar et al. [36]. Therefore, this technology requires further in-depth investigation.
Monkman et al. [15] investigated the use of CO2 injection into fresh-mix concrete (CarbonCure technology) with reduced binder content. Compared to a standard mix, the binder-reduced mixture exhibited lower compressive strength; however, this loss was offset when CO2 was introduced during curing. The approach demonstrated that binder content can be lowered while preserving mechanical performance. In a large-scale implementation spanning over ten months, CO2 was injected into approximately 130,000 m3 of concrete, resulting in a 5% reduction in cement consumption. This reduction corresponds to savings of about 600 tonnes of cement and 530 tonnes of CO2 emissions, without compromising performance.
There are also other commercialised solutions that use CO2 in fresh concrete, such as Concrete 4 Change (C4C) [37]. C4C has developed an innovative technology for CO2 uptake in cementitious materials by mineralising captured CO2 from industrial flue gases using abundant waste calcium sources to create a reactive CaCO3 additive. A low-cost, porous, granular absorbent material derived from waste streams such as recycled wood, glass, ceramics, and plastics is used as a CO2 carrier. The CO2 carrier is placed in an autoclave and loaded with CO2, where the gases are absorbed. This approach turns waste into value, promotes a circular economy, and minimises the carbon footprint. These carriers are then added as an additive into fresh concrete mixes, without altering conventional production processes, resulting in a well-controlled carbonation of the concrete. The carrier slowly releases CO2 inside the concrete for permanent mineralisation. This approach, which results in permanent CO2 sequestration, increases concrete strength and reduces cement content by 20%–50%. Recent pilot projects and ongoing commercial development indicate strong scalability and cost-effectiveness. The technology distinguishes itself by enabling superior CO2 uptake efficiency, broad compatibility with existing concrete types, and a circular production model, positioning it as a key industrial pathway to achieving net-zero targets in the construction sector. This technology is currently in the pilot trial phase and is expected to undergo a full-scale rollout by 2026–2027. The total emission savings are 50 kg of CO2 per cubic meter of concrete, while the concrete CO2 emission (of this technology) of C4C concrete is 230.63 kg CO2/m3 [38]. It is worth mentioning that recently NEG8 Carbon, a company specialising in direct air capture, reported successful CO2 uptake in wet mix concrete by injecting captured atmospheric CO2 directly into fresh concrete during batching [39]. Table 1 summarises the main features of the CarbonCure and C4C technologies, including their TRL, key advantages and limitations, and commercial status.

3.2 CO2 Curing of Pre-Cast Concrete
Carbonation curing of precast concrete under a CO2-rich atmosphere is another approach to decreasing carbon footprint that has attracted the attention of the cement and concrete industries. The approach of CO2 curing precast concrete is reported as a promising method for permanently storing CO2 in concrete [24,28]. To date, carbonation curing of non-reinforced precast concrete under a CO2-rich atmosphere is recommended [40]. In this approach, after mixing and casting, CO2 is introduced into the concrete to accelerate curing (Fig. 3). It allows for rapid strength gain and has been shown to enhance product density and durability [16,41]. The rapid strength gain also enables the precast industries to achieve a high production turnover [42]. It is worth noting that carbonation curing is self-limiting, as the formation of a dense carbonated layer restricts further reaction. Even so, studies consistently report significant early-age benefits of CO2-curing, compared to non-carbonated samples, although values typically converge by 28 days [40]. Similarly, another study reported strength improvements upon the CO2 curing, ranging from 20%–100% at early ages and 5%–20% after subsequent 28-day water curing [28].
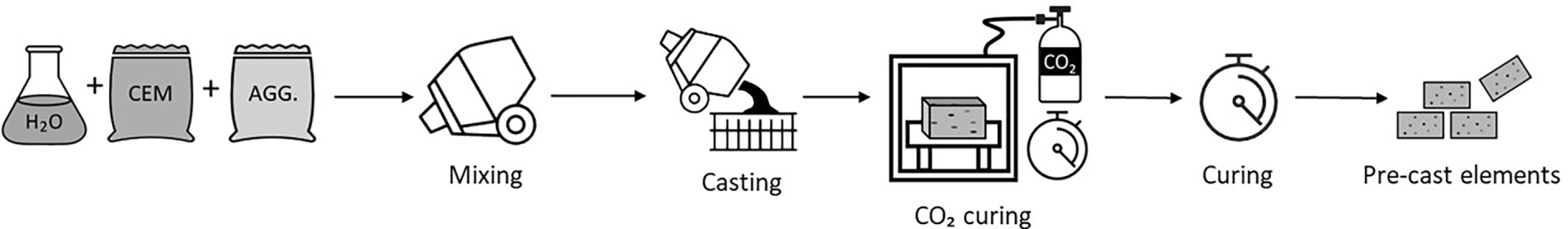
Figure 3: Schematic representation of CO2 curing of pre-cast concrete elements
The CO2 curing process can be performed in a closed chamber or flow-through system, where temperature, humidity, CO2 concentration, and curing time need to be precisely controlled to maximise the carbonation reaction and ensure the quality of the final product [43]. The concrete mix design, e.g., water-to-binder ratio (w/b) [44] and aggregate particle size, is a crucial factor in the success of CO2 curing [45,46]. In this respect, two types of precast materials, dry-mix and wet-mix, can be subjected to CO2 curing. The carbonation curing process for precast concrete typically involves three stages: pre-conditioning, carbonation, and post-conditioning [47]. The pre-conditioning stage aims to reduce pore water content, thereby optimising CO2 diffusion through the concrete matrix during carbonation, as saturated pores may hinder CO2 penetration [17]. This step is usually performed at 20°C–25°C and 40%–60% relative humidity (RH) [17]. While pre-conditioning is a crucial step for wet-mix concrete to achieve optimum humidity and to balance hydration and carbonation reactions, it should be avoided for dry-mix concrete, as over-drying may cause water starvation, which inhibits the hydration process [16]. The low humidity level during carbonation is also a critical parameter, as the literature reports its negative effect on CO2 uptake and carbonation rate in cementitious materials [16]. Additionally, pre-conditioning time is an essential parameter for the CO2 curing of cement-based materials. As such, a wide range of curing conditions (wet, in mould, off mould, open air, and steam curing) and various curing durations (3–72 h) have been investigated in the literature [47,48]. Nevertheless, evidence suggests that CO2 absorption is primarily governed by the degree of moisture loss, rather than by the duration of pre-conditioning [16].
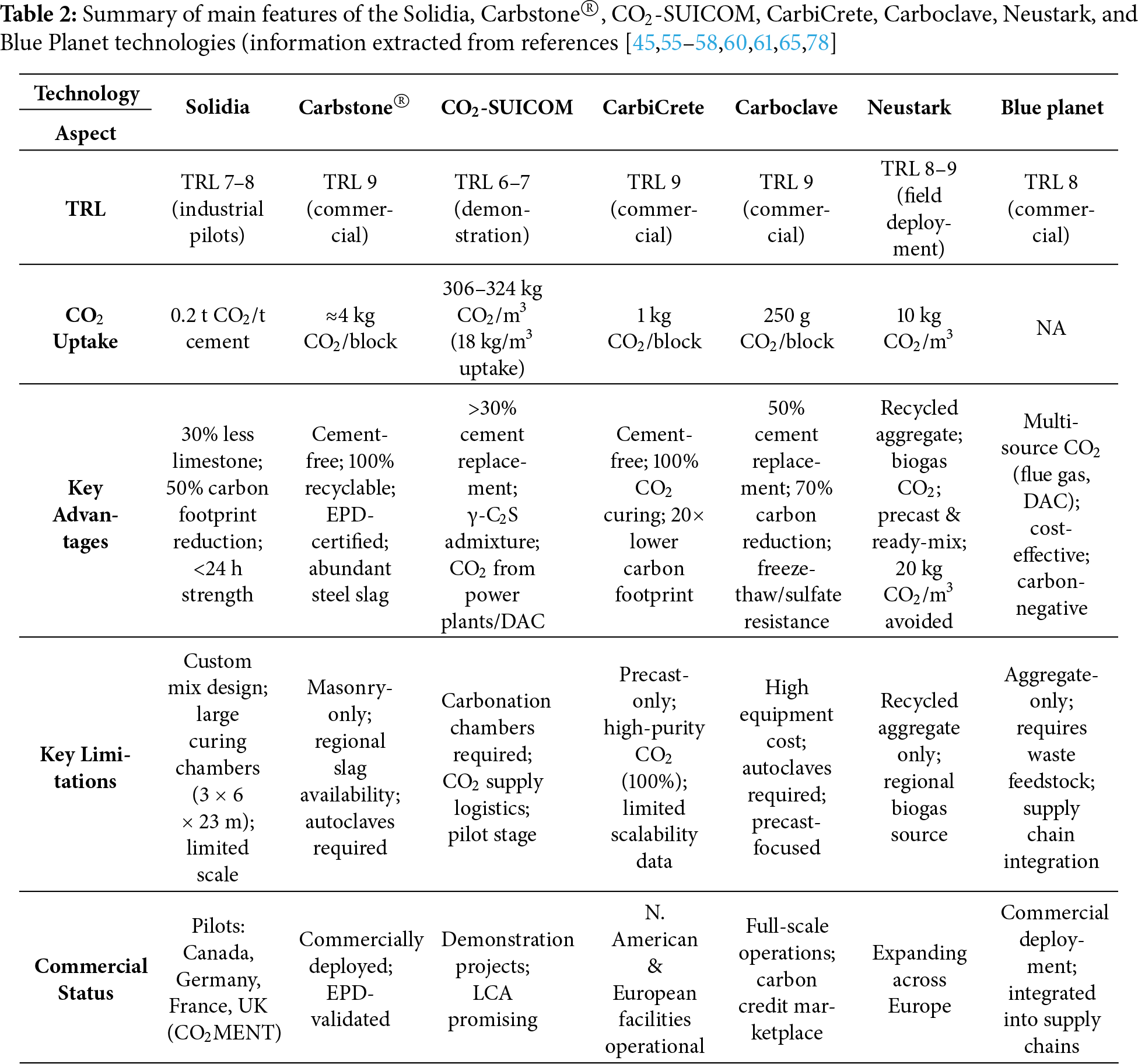
During the carbonation stage, to accelerate hydration and promote early-age strength development, concrete is exposed to CO2 under controlled conditions [47]. CO2 concentration, curing time and temperature, RH, and pressure are crucial parameters that affect the CO2 uptake of cement-based materials [49,50]. Literature reported on 25%–100% CO2 concentration [44,51], 4–96 h of curing time, 23°C–95°C of curing temperature, 30%–90% RH [40,44], and 0.1–13.9 bar of CO2 pressure [52] to maximise the CO2 uptake by concrete. It is worth mentioning that, based on the literature, 50%–60% RH and room temperature are optimum conditions, which maximise carbonation efficiency [47,53]. In the post-conditioning stage, concrete is stored under controlled environmental conditions at 20°C–25°C and RH of 40%–95%, which allows further hydration of unreacted cement particles by consuming the remaining free water in the concrete, thereby stabilising concrete properties [51]. Nevertheless, most experimental studies kept the carbonated samples in a humidity-controlled chamber at 25°C and 95% RH for 27 days [51,54]. The study by Ravikumar et al. [36] reveals that the CO2 uptake potential in CO2 curing for precast concrete applications is significantly higher than that in CO2 injection into fresh-mix concrete. For this reason, commercialised technologies for this approach have been reported by several companies, namely Solidia Technologies [55], Carbostone® [56], CO2-SUICOM [57], CarbiCrete [58], Neustark [59], Carboclave [60], and Blue Planet [61]. These successful cases applied CO2 curing technology in non-reinforced concrete prefabricated elements.
Solidia Technologies, headquartered in the United States, offers a patented process in which concrete is cured using CO2 rather than conventional water/air curing. This technology has been applied to concrete masonry and paving units, enabling the permanent storage of CO2 within precast products [16]. The proprietary mix design and dedicated curing strategy have led to notable environmental and performance improvements. Specifically, the process enables lower production temperatures and approximately 30% less limestone usage, resulting in a 30% reduction in CO2 emissions and a decrease in the overall carbon footprint of up to 50% [16].
Demonstrations and industrial-scale applications have shown that at least 0.2 tonnes of CO2 can be absorbed per tonne of Solidia Cement, which is formulated with wollastonite and rankinite as principal alternatives to Portland cement, when used in precast concrete. For industrial deployment, Solidia has developed curing chambers with dimensions of 3.0 m × 6.0 m × 23.0 m [40]. The curing protocol involves exposing ground clinker and gypsum compositions to a controlled CO2 atmosphere within these chambers. During the process, water is removed, and CO2 is incorporated, facilitating strength development in less than 24 h and resulting in a significant improvement over the conventional 28-day hydration period [62]. The CO2MENT project, a collaboration between LafargeHolcim, Solidia Technologies, and Svante, has established a demonstration unit at the Richmond Cement Plant in British Columbia. Using Svante’s capture technology, CO2 is removed from flue gas and injected into Solidia’s curing chambers for mineralisation within precast concrete. Industrial pilots based on this process are underway in Canada [62], USA [60,61], Germany [59,62,63], France [62], and the UK [62], representing a significant advancement in CCU for cement production.
Carbstone®, developed through collaborative research initiated by VITO in Belgium in 2007, represents a leading example of industrial-scale CO2 mineralisation for sustainable construction. The technology converts calcium-rich steel slag into cement-free building blocks [56,64]. Production involves compacting the slag into shaped products and curing them in autoclaves under elevated CO2 pressure and temperature. During curing, reactive oxides convert into stable carbonates, permanently uptaking CO2 and producing high-performance construction materials [56]. The resulting masonry products offer strength and durability comparable to conventional concrete. Carbstone® blocks are certified CO2-negative and fully recyclable, and their commercial deployment with Environmental Product Declaration (EPD) validation demonstrates their suitability as an alternative to cement-based materials. Each block captures about 2 kg of CO2, with an additional 2 kg avoided by eliminating cement-related emissions; a full truckload saves 6–7 tonnes of CO2 compared to standard blocks, making the products carbon-negative over their lifetime. Steel slag feedstock is also widely available, with more than 40 million tonnes produced annually in the EU and several hundred thousand tonnes in Portugal [65,66].
In CO2-SUICOM technology, an advanced concrete formulation enabling net zero or carbon-negative performance through the synergistic use of clinker substitution and accelerated carbonation curing [57,67,68]. In this process, Portland cement is replaced by more than 30 wt.% with a blend of industrial byproducts, such as ground granulated blast furnace slag (GGBFS) and a gamma dicalcium silicate (γ-C2S) admixture, the latter synthesised from waste calcium hydroxide, which is calcined at significantly lower temperatures to substantially reduce process CO2 emissions. The reactivity of γ-C2S enables hardening through a direct reaction with CO2 under controlled conditions. Under controlled CO2-rich curing conditions, γ-C2S rapidly reacts with CO2 to form stable CaCO3 phases, achieving approximately 50% conversion within 24 h. It is worth noting that C3S exhibits a much lower affinity for direct carbonation, while it is primarily responsible for early strength development under standard hydration conditions in Portland cement [69]. Carbonation curing is conducted in chambers where temperature, RH, and CO2 can be precisely regulated. Often, CO2 is sourced from the exhaust of thermal power plants or generated by direct air capture technologies, with concentrations typically ranging from 10% to 20%. CO2-SUICOM reported a mean CO2 emissions reduction of 306–324 kg CO2/m3 of concrete, resulting from both reduced cement usage and CO2 uptake during curing, equivalent to a reduction of 18 kg CO2/m3. CO2-SUICOM exhibits compaction and porosity characteristics comparable to those of conventional concrete and can meet the required flexural strength and durability specifications for infrastructure applications. Further enhancements in cost efficiency and scalability depend on optimising carbonation chamber operation and the logistics of high-purity CO2 supply, with demonstration projects and life-cycle assessments indicating promising results for large-scale deployment across both precast and cast-in-place segments [57,68].
Another commercialised technology for CO2 uptake using precast products is CarbiCrete [58]. It is a cement-free concrete technology in which steel slag serves as the binder once again. After mixing with aggregates and water, the fresh precast products are placed in curing chambers, where CO2 at high concentration (often up to 100% CO2 by volume) is introduced, reacting with the slag to form stable carbonates and driving strength development without the need for hydraulic cement. This process avoids the significant CO2 emissions associated with Portland cement production and permanently mineralises up to 1 kg of CO2 per standard block, resulting in overall reductions in global warming potential of 150 kg CO2 per metric tonne of concrete. EPDs indicate that utilising CarbiCrete technology for masonry production results in carbon footprint values up to 20 times lower than those of conventional concrete masonry units. CarbiCrete’s technology is already in commercial use at several North American and European precast facilities, demonstrating robust mechanical properties and durability while enabling scalable, permanent carbon sequestration in construction materials [58].
Carboclave, founded in Canada in 2016, has developed and commercialised an advanced CO2 curing system for precast concrete products, including masonry and paving units [60]. The technology supports both retrofitting of existing low-pressure curing chambers and the construction of purpose-built autoclaves for high-pressure CO2 curing. The system is fully automated, ensuring precise control of gas distribution, pressure, and safety for both adapting conventional installations and installing new high-efficiency units. Carboclave’s technology enables up to a 50% replacement of cement with supplementary cementitious materials, resulting in a carbon footprint reduction of up to 70% compared to conventional units. It can durably sequester up to 250 g of CO2 per standard 19 cm × 19 cm × 39 cm masonry block. Products fabricated with this system exhibit higher early-age compressive strength, enhanced resistance to freeze-thaw, sulfate attack, and efflorescence, as well as improved durability and reduced permeability [70]. Recent industrial deployments have confirmed Carboclave’s commercial readiness, with ongoing full-scale operations and planned participation in the voluntary carbon credit marketplace through a verified third-party life-cycle assessment. User reports highlight consistent product quality, rapid curing, cost savings, and the successful 100% replacement of virgin aggregates with recycled materials in masonry units, all of which support the industry’s shift toward low-carbon, resilient construction materials [16,60,71].
Recently, the properties and roles associated with the reuse of recycled aggregates, including carbonated recycled aggregates, have been extensively investigated and reviewed in the literature [72–74]. A comprehensive synthesis of these findings falls outside the scope of the present review and can be found in dedicated sources [75–77]. Nonetheless, it should be noted that, to date, two commercialised technologies have been reported for CO2-curing recycled aggregates to mitigate CO2 emissions in the construction industry.
As the most recent technology, Neustark has developed a commercial solution for CO2 mineralisation in recycled concrete aggregates sourced from construction and demolition waste sites [63,78]. The company captures and liquefies CO2 from regional biogas facilities and distributes it to nearby aggregate plants. At these locations, CO2 is injected directly into recycled aggregates through a controlled, accelerated mineralisation process. This technique permanently binds CO2 within the aggregate matrix, enabling its subsequent use in concrete production. Industrial trials and recent field deployments have demonstrated that this approach enables the permanent sequestration of up to 10 kg of CO2 per cubic meter of produced concrete. In addition to this direct storage, further reductions in cement content are achieved, resulting in total avoided emissions of approximately 20 kg CO2/m3 compared to conventional concrete mixtures. Performance monitoring and life-cycle assessments have demonstrated that this process produces durable, strong masonry products while reducing carbon emissions. Neustark is now expanding its technology across Europe, and it works well both for factory-made (precast) and site-made (ready-mix) concrete products [59].
In line with the Neustart technology, Blue Planet Systems (a US-based company) has developed a patented technology that captures CO2 from a wide range of sources, including flue gases and direct air capture, and mineralises it into synthetic limestone aggregate for use in concrete. The Geomimetic® process permanently sequestrates CO2 by combining it with calcium-rich industrial waste, such as cement kiln dust, steel slag, demolition concrete, and fly ash, producing durable construction materials. Unlike conventional CCU approaches that rely on purification, Blue Planet’s method directly incorporates CO2 into aggregate, making the process cost-effective and scalable. Concrete produced with this aggregate is certified as carbon-negative, and the CO2 remains secured in the mineral structure even after demolition. Commercial deployment is underway, with facilities validating large-scale production and successfully integrating it into conventional concrete supply chains, thereby helping drive progress toward net-zero targets within the built environment [61,79].
It is worth mentioning that the influence of carbonation on the long-term durability of CO2-cured concrete remains a subject of debate. Cai et al. [80] demonstrated that CO2 curing can reduce drying shrinkage by 49%–53% in alkali-activated slag concrete through pore refinement and CaCO3 precipitation. Similarly, Liu et al. [81] observed that carbonation curing not only reduces early-age shrinkage but also enhances long-term mechanical strength and durability in manufactured concrete. In contrast, Zhu et al. [82] and Xian et al. [83] reported that carbonation curing can increase dry shrinkage due to water consumption during CO2 injection and the development of heterogeneous moisture gradients, which was attributed to the non-uniform distribution of carbonation products within the material. Additionally, Humad et al. [84] linked increased creep in carbonated alkali-activated systems to microcracking and decalcification of C–S–H gel, which coarsens the pore structure and reduces long-term dimensional stability. These divergent outcomes underscore that the net effects on shrinkage and creep are highly sensitive to curing parameters (pre-curing duration, curing temperature, relative humidity), water-to-binder ratio, binder composition (including activator type and MgO content), and the extent of carbonation. Accelerated carbonation generally improves resistance to chloride ingress and sulfate attack by densifying pores and forming a carbonate-rich surface layer that reduces permeability and pore connectivity. Rostami et al. [85] demonstrated that carbonation-cured concrete exhibited superior resistance to chloride permeability, sulfate attack, and freeze-thaw damage compared to conventional curing methods, while Su et al. [86] confirmed enhanced sulfate resistance through microstructure refinement. However, Mi et al. [87] identified a competing mechanism: carbonation releases previously bound chloride ions by decomposing chloride-bearing hydration products (Friedel’s salt), thereby increasing the concentration of free chlorides available to initiate steel corrosion. This effect is particularly pronounced when carbonation penetrates beyond the protective surface layer into the bulk matrix. Additionally, prolonged or excessive carbonation that causes C–S–H decalcification can degrade sulfate resistance, as reported by Zhang et al. [88] in studies of fly ash concrete exposed to combined carbonation and sulfate attack. Nevertheless, the main durability challenge for carbonation-cured reinforced concrete is the rapid drop in pore solution pH from ≈13 to 8.5–9.5, which compromises the passive oxide film protecting the steel. Depassivation typically occurs at pH 9.4–10.0, depending on carbonate ion concentration and moisture conditions. Importantly, early-age carbonation applied only to the concrete surface (as in precast applications) can maintain core alkalinity above the threshold required for passivation, provided adequate cover depth is specified. Wu et al. [89] demonstrated that steel slag incorporation delays passive film formation but may sustain long-term passivity through controlled carbonation depth. Xian et al. [54] reported that ambient-pressure CO2-curing reduces rebar mass loss by approximately 60% and improves chloride resistance vs. conventional curing, attributed to the dense carbonate-rich surface layer that restricts ingress. Despite promising short-term results (typically <5 years), the long-term durability (>10 years) of carbonation-cured reinforced concrete under real-world exposure conditions remains insufficiently documented. Key uncertainties include: (i) the interaction between early-age accelerated carbonation and subsequent atmospheric carbonation over service life; (ii) performance under combined degradation mechanisms (carbonation + chloride ingress + freeze-thaw cycling); (iii) optimal carbonation parameters (depth, CO2 pressure, curing duration) that maximize CO2 uptake and mechanical performance while preserving adequate alkalinity for reinforcement protection; and (iv) the influence of SCMs on long-term alkalinity buffering capacity in carbonation-cured systems [81,90]. Field trials and accelerated aging tests that simulate long-term exposure are essential to validate laboratory durability predictions and develop performance-based guidelines for carbonation-cured structural concrete.
Table 2 summarises the main features of Solidia, Carbstone®, CO2-SUICOM, crCrete, Carboclave, Neustark, and Blue Planet technologies, including their TRL, key advantages and limitations, and commercial status.
4 Ceramic Waste Materials as a CO2 Sink
In recent years, ceramic materials have been investigated as potential CO2 uptake materials due to their favourable chemical composition and structural characteristics. As discussed previously, CO2, when dissolved in water, forms an acidic solution that can react with basic alkali and alkaline-earth ceramics to form stable carbonates [91].
Therefore, researchers hypothesise that the alkali and alkaline-earth ceramics could be a potential material for CO2 uptake [92,93]. The most studied ceramics include calcium and magnesium oxides (CaO and MgO, respectively), lithium orthosilicate (Li4SiO4), lithium metazirconate (Li2ZrO3), and sodium metazirconate (Na2ZrO3), which have demonstrated a capacity to capture CO2 through adsorption or chemisorption mechanisms [91]. In the chemisorption mechanism, materials tend to be densified due to the formation of carbonation product layers (e.g., Na2CO3, Li2CO3) at the surface or interface, which can affect further CO2 diffusion and uptake kinetics [94]. Sodium zirconosilicate (Na2ZrSiO5) [95], lithium silicates (e.g., Li4SiO4), and Na2ZrO3 are some examples of ceramics with the capacity of CO2 sequestration by chemisorption [96].
Eqs. (5)–(7) demonstrate the reactions of CO2 uptake through chemisorption with Na2ZrO3, Na2ZrSiO5, and Li4SiO4, respectively. In these reactions, the acid properties of CO2 enable the extraction of an oxygen anion (O2−) from the ceramic, forming the CO32− anion, which further reacts with the alkali or alkaline-earth cation (Na and Li) to form the corresponding carbonate salt. It is worth noting that the temperature ranges for the CO2 uptake of Na2ZrO3, Na2ZrSiO5, and Li4SiO4 are reported to be 400°C–800°C, 1000°C–1100°C, and 500°C–600°C, respectively [97,98].
The process of CO2 uptake begins at the ceramic material’s surface, where CO2 molecules chemically react with the surface, forming strong chemical bonds (surface chemisorption).
Ida et al. [99] and Venegas et al. [100] explained the process of CO2 chemisorption in ceramics using a dual-shell model, which accounts for the biphasic kinetic behaviour observed in ceramic CO2 uptake. This model describes two sequential stages: (i) a rapid, chemically controlled surface chemisorption phase, and (ii) a slower, diffusion-controlled uptake phase. Based on this model, CO2 absorption begins at the ceramic surface, where CO2 molecules react chemically to form a layer of carbonate species (e.g., Li2CO3, Na2CO3) through direct surface chemisorption. This initial stage is characterised by fast reaction kinetics, driven by the strong chemical affinity between CO2 and surface oxygen anions. Activation energies for this phase are typically in the range of 50–100 kJ/mol, depending on the cation type and particle size. In the second phase, as the carbonate product layer thickens, the rate-limiting step shifts from surface reaction to solid-state diffusion of CO2 through the increasingly dense product shell. During this diffusion-controlled stage, lithium ions (Li+) and carbonate ions must migrate through the Li2CO3 product layer and any remaining intermediate silicate phases (e.g., Li2SiO3), which are generally less permeable than the original oxide precursor. Experimental and theoretical studies using TGA, XRD, and in situ FTIR confirm the formation of two distinct carbonate regions: an outer layer consisting primarily of surface carbonates (often mixed Li2CO3/Na2CO3) and an inner layer composed of residual unreacted oxide or mixed oxide–carbonate material. This structural evolution directly influences CO2 diffusivity by creating pathways that facilitate or, in some cases, hinder ion transport.
It is worth noting that several factors, including the active surface area, particle size, composition, pressure, humidity, and gas mixture composition, affect the CO2 uptake of ceramic materials. Calcium oxide (CaO) is considered to have a high potential due to its balance between its reactivity and industrial scalability. Additionally, other lithium- and sodium-based ceramic materials also demonstrate potential for CO2 uptake [101].
While chemisorption through ceramic materials has potential for CO2 uptake, it is important to consider whether regeneration of the ceramic material is intended, as it requires significant energy due to the strength of the chemical bonds formed [91,102]. If regeneration is not intended, carbonated ceramics can be directly repurposed as functional additives, SCM, or aggregates in construction materials (Fig. 4). This valorisation pathway offers a practical and circular solution for carbon management in the construction sector [103,104].
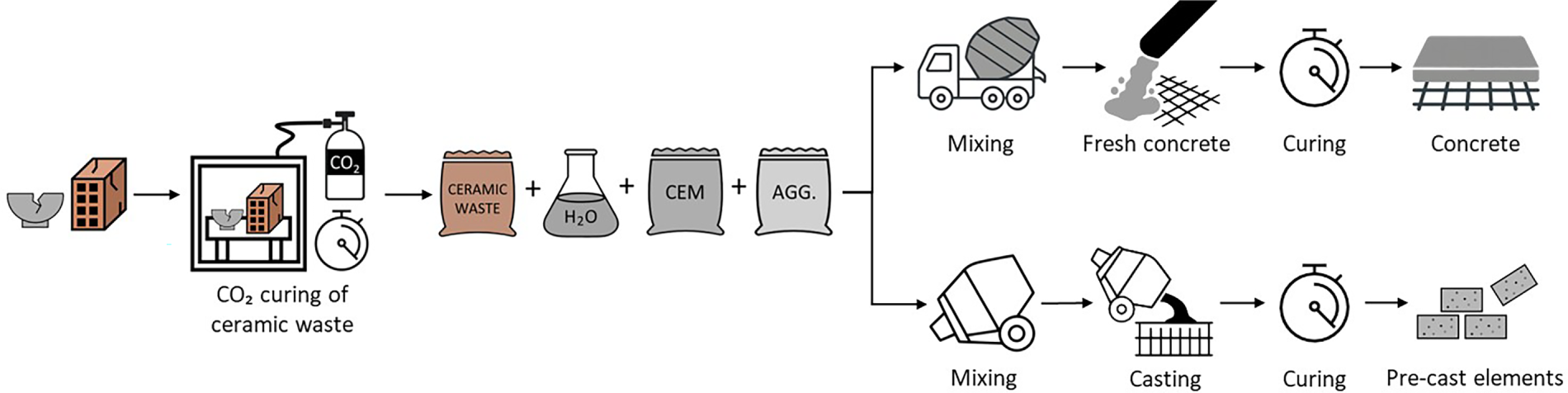
Figure 4: Schematic representation of CO2 curing of ceramic waste and the possibility of re-utilising the carbonated waste in the construction industry
Ceramic materials can also capture CO2 via adsorption. In this process, CO2 molecules adhere to the surface of the ceramic material, forming weaker bonds such as Van der Waals or hydrogen bonds, making the process typically reversible. An example of this mechanism is represented in Eq. (8) [101].
Ramírez-Moreno et al. [101] reported that the adsorption temperature of CO2 on ceramic materials is typically below 400°C for porous materials with a large surface area (e.g., Layered Double Hydroxides). In such a condition, a significant anion-exchange capacity of 2–3 milliequivalents per gram (meq/g) and optimal thermal stability can be achieved.
Recently, studies have focused on the potential of ceramic waste materials for CO2 uptake. These ceramics contain a high concentration of calcium- and magnesium-silicates, which react with CO2. The silicate framework facilitates ion exchange and carbonate formation. The ion exchange mechanism during carbonation of silicate frameworks relies on the ability of the silicate mineral structure to release and replace cations. As mentioned previously, when CO2 dissolves in water, it generates CO32− and hydrogen ions (H+). These ions can be replaced by divalent cations such as Ca2+ and Mg2+ within the silicate lattice. These released cations then react with CO32− ions in solution to form stable mineral carbonates, such as CaCO3 and MgCO3 [105,106] (Eqs. (9)–(11)).
Martín et al. [107] investigated the carbonation of ceramic bricks surrounded by common clay (marl) in a hermetically sealed reactor. The experimental procedure involved maintaining a CO2 pressure of 0.5 bar, a temperature of 23°C, and a solid-to-liquid ratio of 4:1 (wet carbonation) for 5 months. The results showed the formation of calcite as the main carbonated phase, with wollastonite and anhydrite as the principal phases transformed during the reaction. A maximum CO2 sequestration of 10 wt.% was obtained under these conditions.
Afterwards, Martín et al. [108] conducted a similar study in a pilot-scale system to simulate the carbonation of ceramic waste bricks in a reclaimed quarry environment. The system was evaluated at 5, 7, 9, and 12 months, and the results showed a progressive increase in CO2 uptake. After CO2 exposure, CO2 uptakes of 17 and 23 wt.% were reported at 5 and 12 months, respectively. Mineralogical analyses confirmed the transformation of wollastonite and the formation of calcite during the carbonation process. Microstructural characterisation indicated that macroporosity increased due to mineral dissolution by carbonic acid, while microporosity decreased due to the precipitation of carbonate phases. These findings demonstrate that ceramic brick waste is suitable for CO2 uptake under the conditions examined [108]. No studies have been identified on the reuse of carbonated ceramic waste in cementitious materials. However, two notable projects in Italy have explored the material’s potential for CCU within the ceramic industry.
The LIFE12 ENV/IT/000424 project, supported by the European Union and conducted in partnership with Ceramica Alta and the Department of Industrial Engineering at the University of Padova, aimed to mitigate CO2 emissions from the ceramic firing process by implementing oxy-fuel combustion technology in place of traditional air combustion [109]. This modification altered the composition of exhaust gases, producing a stream of carbon dioxide and water vapour that facilitated the recovery and separation of CO2. The recovered CO2 was subsequently evaluated for reuse, including applications in greenhouse cultivation and the mineralisation of ceramic waste for recycling. The adoption of oxy-fuel combustion technology yielded notable benefits, including reductions in CO2 and NOx emissions and lower energy consumption at the pilot scale. Additionally, tiles produced from recycled carbonated materials exhibited properties comparable to those of tiles produced by the conventional process [109]. A more recent project, Carbon Capture Storage and CO2 Mineralisation for the Ceramic Industry (CCS4CER), launched in February 2024 and is coordinated by the Ceramic Centre of Italy [104]. The project objectives include developing approaches to separate CO2 emissions and convert hazardous ceramic process wastes, such as spent lime, into carbonate-based products via mineralisation. The project also aims to optimise gas-liquid operating conditions to enhance the production of materials such as CaCO3, CaF2, and CaSO4. CCS4CER further intends to validate the use of these synthesised materials in carbon-negative mortars and eco-cements, thereby supporting circular economy practices, while ensuring that all solutions are both energy- and cost-efficient to facilitate the transition of the ceramic tile industry toward climate neutrality. The CCS4CER project is investigating three primary approaches for CO2 uptake in ceramic manufacturing: oxy-fuel combustion, which generates a concentrated CO2 and water vapor stream to facilitate separation; post-combustion chemical absorption, such as amine scrubbing, for removal of CO2 from exhaust gases; and direct process integration, where captured CO2 is utilized for mineralizing ceramic wastes through reactions that produce valuable carbonates and other compounds. To date, no specific outcomes have been reported from the project [104]. It is worth noting that, in addition to ceramic waste as an alkaline residue, other alkaline industrial by-products, such as air-pollution control residues, cement kiln dust, and municipal solid-waste incineration ashes, have also been utilised in CO2 mineralisation processes, with some pathways already reaching commercial deployment. Among these, the most advanced is the accelerated carbonation technology developed by Carbon8 Systems (UK), which has achieved a Technology Readiness Level (TRL) of 8–9, signifying full commercial operation. In September 2020, Vicat implemented the first European cement-industry CO2ntainer system at its Montalieu-Vercieu plant in France, where CO2 from kiln exhaust gases is captured and reacted with bypass dust to produce a lightweight aggregate with thermal-insulating properties. This process permanently sequesters approximately 10–15 wt.% CO2 as stable carbonates while simultaneously converting hazardous industrial residues into marketable construction materials. The technology is undergoing global scale-up following a 2021 partnership between Carbon8 Systems and FLSmidth, a major provider of cement-processing technologies. Notably, this pathway eliminates the energy penalty typically associated with sorbent regeneration, as the carbonated product serves as the final commercial material rather than an intermediate requiring thermal cycling [110]. This example illustrates that ceramic-based materials’ carbonation technologies can successfully transition from laboratory research to commercial deployment through two principal innovations: (i) valorisation of carbonated products as SCMs or aggregates instead of regenerating sorbents, and (ii) seamless integration with existing kiln operations without the need for dedicated CO2 capture infrastructure.
5 Cost of CO2 Injection into Cementitious Materials
The costs associated with CO2 injection in concrete production are highly variable and depend on several factors, including the scale of implementation, the origin and purity of CO2, and the specific technology employed. A key cost component is CO2 capture from industrial sources, which can be substantial and varies with the method used (e.g., amine scrubbing, membrane separation). Nevertheless, in line with legislative initiatives, near-term access to large volumes of captured CO2 at reduced costs is expected to enhance the competitiveness of carbonation curing processes, thereby strengthening the competitive advantage of implementing them. Associated costs include capital investments for CO2 injection equipment and retrofitting existing installations, as well as operational expenses related to CO2 injection and system maintenance [16].
Additional expenses arise from CO2 purification, compression, transportation, and storage [111]. Typically, pure CO2 gas is used to accelerate diffusion. However, some studies aim to capture CO2 directly from flue gas. The maximum CO2 uptake will be strongly influenced by the CO2 concentration (typically 20%–25% in cement plants) [112]. He et al. [113] investigated the carbonation of cementitious materials using flue gas. The lower reaction efficiency was observed due to reduced CO2 concentration in flue gas. Nevertheless, the necessary strength was achieved in 5 h (using seven injection cycles), enabling CO2 uptake of 5%–6%, with strength gains comparable to those achieved with pure CO2 carbonation. Therefore, the study demonstrates the feasibility of connecting cement plants and concrete plants for effective CO2 emission reduction through direct flue gas utilisation. It was not possible to obtain the specific investment and operating costs for CO2 injection into fresh concrete. However, Monkman et al. [114] explored the use of CO2 as an accelerating additive in the concrete mixing process, with CarbonCure, comparing it to samples utilising a conventional accelerator (non-chloride accelerator). CO2 injection expedites concrete setting without compromising durability, offering a cost-effective alternative to chemical accelerators. In 2025, the escalated price of industrial CO2 is approximately €508 per tonne, with per-truckload (8 m3) expenses ranging from €0.64 to €3.76. By contrast, calcium nitrate costs €188 per tonne, and conventional non-chloride admixtures total €16.28-€32.56 per truckload. These findings confirm the economic advantage of CO2-based acceleration under current market conditions.
Regarding CO2 curing of precast products, operational costs are primarily related to controlling curing conditions, particularly during the pre-curing phase, when energy is required to maintain the desired humidity for optimal carbonation efficiency. Compared with conventional steam curing, CO2 curing typically requires only about one-fifth of the energy demand [48].
Fortunato et al. [40] conducted a feasibility study on the implementation of CO2 curing in the Brazilian concrete block industry. The proposed curing chamber had internal dimensions of 4.0 m × 4.0 m × 4.0 m, corresponding to a total volume of 64.0 m3. To achieve chamber saturation, 52.94 m3 of CO2 were injected per cycle, equivalent to approximately 109 kg. Based on a daily production rate of 5760 blocks, corresponding to 126,720 blocks per month, distributed across six curing chambers, the total CO2 demand was estimated at 654 kg per day. Under these conditions, the cumulative CO2 consumption amounted to approximately 14,388 tonnes per month, assuming three curing cycles per chamber operating over an 8-h daily schedule.
Fortunato et al. [40] reported that a 2 h CO2 curing cycle enables the sequestration of approximately 6827 kg of CO2 per month. Accounting for Chemical Engineering Plant Cost Index (CEPCI) escalation to 2025, the capital investment for a new curing chamber is estimated at €9383 and retrofitting an existing unit costs €10,342. The higher retrofit cost primarily results from increased labour, additional insulation, and technical upgrades required for airtight CO2 curing, often making retrofits less cost-efficient than new installations. Monthly CO2 supply expenses, dependent on supplier distance, range from €2537 to €8458, contributing to a 4%–14% increase in the unit price of blocks. Direct utilisation of flue gas from cement plants may present a more economical alternative for CO2 supply.
At a commercial scale, Wang et al. [115] implemented a retrofitted CO2 mineralisation curing system in China, achieving an annual CO2 sequestration capacity of 11 kton. After over 72 h of continuous curing, the concrete specimens reached a CO2 uptake of 5.3 wt.% and a conversion efficiency of 98.97%. The process yielded an adjusted economic benefit of €31.65 per tonne of CO2 and reduced the product’s carbon footprint by 178 kg CO2-eq/m3, with approximately 65% attributable to direct CO2 sequestration.
Commercialisation costs and production economics for novel CO2 mineralisation concrete technologies exhibit significant variability. Solidia reports production costs comparable to or lower than those of conventional Portland cement, owing to process efficiencies such as rapid curing, lower energy requirements, and the elimination of steam autoclaving. After adjustment for CEPCI escalation and currency conversion, capital investment for initial pilot and demonstration facilities is estimated at €1.13 million and €0.44 million, respectively [116].
Carbstone®, utilising stainless steel slag in a high-CO2 environment, requires an initial investment of approximately €540,000 (accounting for CEPCI escalation from 2004 to 2025) for curing chamber retrofits. However, it subsequently achieves product costs comparable to those of standard blocks, with further savings realised through full recyclability and avoided landfill charges [56,117]. VITO and Orbix have built Carbstone® pilot plants and commercial demonstration lines in Flanders and Wallonia, Belgium. These sources discuss operational success and market penetration but do not provide installation costs. Industrial articles note that residual materials, CO2, and labour are the main cost factors, and emphasise lower raw material and energy costs (compared to conventional cement blocks), but capital investment is not disclosed [118].
CO2-SUICOM has reached industrial scale in Japan by employing waste-derived binders and carbon credit incentives, resulting in competitive cost parity for precast applications; however, the main operational expenses remain in carbonation equipment and CO2 procurement. Although specific capital expenditure figures for CO2-SUICOM carbonation equipment remain proprietary and undisclosed, the technology has demonstrated competitive cost parity for precast applications through carbon credit mechanisms and substantial cement replacement (>50%) with waste-derived binders, thereby reducing operational expenses and enhancing economic viability [68,119].
Carbicrete has secured investments exceeding €19.2 million for commercial-scale-up, with production costs reported to match those of conventional concrete blocks following precast chamber retrofits and savings from eliminating cement [58].
Carboclave retrofits pressure vessels for CO2 curing and claims lower costs than autoclave-cured blocks, primarily driven by energy savings, while the final retail price is generally at or slightly below the standard block price [120]. Neustark’s process, implemented in Europe, involves sizable infrastructure investment (secured approximately €61.4 million for expansion), yet its cost per ton of CO2 removed remains favourable in carbon markets, with the process typically incurring only a slight premium over recycled aggregate but benefiting from carbon removal revenues and project-scale investment. In summary, all technologies can achieve cost parity or an advantage where supportive carbon markets or material incentives exist, and major costs are concentrated in the initial process adaptation, curing equipment, or CO2 supply [121].
Recent techno-economic analyses of CO2 mineralisation using ceramic wastes have clarified several critical factors that determine feasibility and scalability. The concentration of CO2 in ceramic kiln exhausts is lower than that in traditional power plants, resulting in higher capture costs and lower process efficiencies. However, integrating waste heat recovery and energy management, such as utilising low-temperature heat from kiln operations, can partially offset these challenges, improving the energy profile of CO2 capture systems installed in ceramics manufacturing. Indirect carbonation methods, especially those employing pH-swing mineralisation, have been found to offer carbon efficiencies approaching 95% when acid and base recycling are optimised, with reported process costs ranging from €445 to €712 per tonne CO2 under the best scenarios. In less favourable setups that lack integrated recycling or employ more energy-intensive process steps, the cost can rise as high as €1602 per tonne CO2; the carbonate precipitation stage consistently represents the largest expense in these workflows [122,123].
The proximity and availability of suitable ceramic wastes for mineralisation are essential to minimise transportation and handling costs and support viable circular economy models. Techno-economic models consistently emphasise the importance of valorising products generated, such as precipitated CaCO3 and MgCO3, which can be utilised in carbon-negative mortars or incorporated into eco-cement formulations, thereby generating economic returns and offsetting process expenses. Nevertheless, upfront capital costs for large-scale deployment of CO2 mineralisation plants in ceramics remain substantial, and technical obstacles, including slow leaching kinetics, efficient process integration, and reliable supply of reactive alkalinity, must still be adequately addressed. Overall, while the utilisation of ceramic wastes for CO2 mineralisation is increasingly supported by life cycle and cost-benefit assessments, widespread implementation will depend on continued advances in process optimisation, product market integration, and policy incentives to overcome both technical and economic barriers [123,124]. It is worth noting that the reported value in this chapter was updated to 2025 using the CEPCI using Eq. (12):
6 Summary of Potential Approaches for CO2 Uptake Using Fresh Mix and Precast Concrete and Ceramic Waste Materials
In this work, potential approaches for CO2 uptake using construction materials were reviewed. The specific attention was paid to those technologies that directly inject flue gas or CO2 captured and stored for further re-use from the cement and concrete industry. Based on the literature, CarbonCure and C4C are the most advanced commercialised technologies for purifying CO2, either directly or indirectly (via CO2 carriers), in fresh mix concrete. As shown in Table 3, the CO2 uptake is permanent but of low quantitative significance. In turn, the novel technologies that inject CO2 into precast products reported higher CO2 uptake in their products. It is crucial to note that the CO2 uptake or CO2 avoided in most of these technologies was reported as the sum of CO2 uptake and CO2 emission avoided through the substitution of some part of cement or natural aggregates with industry by-products, wastes, or recycled aggregates. CO2 curing in precast concrete achieves substantially higher CO2 uptake compared to CO2 injection into fresh concrete mixtures. This increase results from the optimised reaction conditions and extended exposure during precast curing, which enhance the mineralisation efficiency and lead to greater permanent sequestration of CO2 within the concrete matrix.
Recent research on ceramics as promising materials for CO2 uptake in construction applications was also assessed. According to the literature, alkali and alkaline-earth ceramics, particularly those containing calcium, magnesium, or lithium, exhibit favourable chemical reactivity, enabling them to form stable carbonates by chemisorption at elevated temperatures. Both laboratory and pilot-scale studies demonstrate the capacity of ceramic waste to capture significant amounts of CO2 via mineral carbonation, with capture efficiencies reaching up to 23 wt.% under optimised conditions. It is worth mentioning that the CO2 uptake values reported in this review use multiple units (kg/m3, wt.%, and % of theoretical maximum) to accurately reflect the diversity of measurement conventions established in the source literature. This variation reflects differences in material form, experimental methods, and the distinct reporting practices of the chemical engineering and civil engineering communities.
Table 4 presents the commercialisation status and estimated implementation costs of the technologies reported in Table 3. Implementation costs for carbon utilisation technologies are drawn from recent industry reports and published literature, with values reflecting direct investments, operating expenses, and funding sources. CarbonCure operates under a licensing model that entails no upfront capital expenditure (CAPEX), as the equipment is provided through retrofit installations with no purchase costs for the client. All recurring expenses, including the annual supply of CO2 and related operational activities, are classified as operating expenditure (OPEX). Based on updated industry data and the CEPCI for 2025, typical CO2 supply costs now range from approximately €4700 to €18,800 per year. The market rate for delivered CO2 is currently between €470 and €560 per tonne, reflecting recent pricing trends and escalation factors from the current CEPCI, which has increased slightly over the past year.
C4C secured approximately €2.8 million in seed funding in January 2024, bringing the total investment to €5.1 million, including €1.7 million in UK government grants. The seed round, led by Zacua Ventures and Counteract with strategic investment from Siam Cement Group and Goldbeck, positions C4C for pilot-to-commercial scale-up and a planned Series A round of €5.67–11.34 million 2025–2026.
Solidia secured €1.97 million from the U.S. Department of Energy’s Office of Energy Efficiency and Renewable Energy (EERE) in May 2022.
CarbiCrete’s commercial plant retrofit was funded at €5.89 million through a partnership between NGen (€2.5 million) and industry partners Patio Drummond and Innovobot Labs (€3.39 million).
Neustark’s expansion to a projected one million tonne annual CO2 removal capacity by 2030 is supported by €64.75 million raised in June 2024 from Decarbonization Partners (BlackRock-Temasek), Blume Equity, and strategic partners. Carbstone, CO2-SUICOM, Carboclave, and Blue Planet have reported large-scale installations or partnerships; however, exact per-plant costs remain unpublished. Available figures and budgets are consistent with sector norms for industrial retrofit and project-scale deployment.
It is worth mentioning that despite the recent advances in CO2 capture technologies in construction materials, the deployment of such a technology faces critical barriers that must be overcome to transition from laboratory demonstrations to widespread industrial implementation, particularly for CO2 injection in fresh concrete mixes, CO2 uptake by precast elements, and CO2 utilization in ceramic waste systems.
CO2 injection in fresh concrete faces fundamental challenges related to reaction kinetics and durability trade-offs. Insufficient CO2 volumes result in unstable carbonate polymorphs, vaterite and aragonite, which transform to calcite. This conversion causes volume changes and cracking, weakening the concrete’s long-term compressive strength. Maintaining mechanical performance while integrating CO2 injection into high-volume industrial mixing requires precise control of injection parameters (flow rate, pressure, duration), which adds energy and capital costs and complicates economic feasibility. Furthermore, CO2-induced carbonation interacts unpredictably with SCMs such as slag or fly ash; while these materials can enhance CO2 uptake and refine pore structures at later ages, they may also coarsen early-age porosity, reduce early strength development, and complicate quality control [23,127].
Precast concrete carbonation benefits from controlled factory environments that enable higher CO2 uptake through optimized curing protocols, yet scalability challenges persist. Achieving uniform carbonation depth across larger or thicker precast elements remains difficult, as CO2 penetration is limited by the interplay between moisture content, relative humidity, and pore connectivity. Additionally, extending CO2 curing to structural elements demands rigorous validation of durability performance, including resistance to chloride ingress, sulfate attack, and alkali-silica reaction (ASR), under service conditions [36,47].
Ceramic waste as a CO2-binding material presents opportunities for dual circularity but faces reactivity and standardization gaps. Variability in ceramic waste chemistry (calcium oxide and reactive silica content), particle size distributions, and pozzolanic activity directly influences both the rate and extent of CO2 uptake, with optimal performance typically requiring milling to reduce the particle sizes [128].
Three key barriers limit the deployment of all three technologies. First, standardized protocols for measuring CO2 uptake are missing, making it harder to compare results and gain regulatory approval. Second, LCA and TEA often reveal that the energy needed to capture, transport, compress, and heat CO2, especially from distant sources or requiring cryogenic cooling, can cancel out the gains from carbon sequestration [34,129].
This review critically examines recent advancements in carbon dioxide uptake technologies for construction materials, with a particular emphasis on methods offering feasible implementation and scalability to substantially mitigate CO2 emissions from the cement industry. Innovation in this field has accelerated to address the pressing need to reduce the carbon footprint associated with cement and concrete production. Special attention is given to technologies targeting CO2 uptake in fresh concrete mixtures and precast concrete elements, as these approaches offer the greatest practical potential for industry-wide adoption and impact. Recent innovations are summarised in comparative tables, highlighting critical metrics such as country of origin, CO2 uptake per cubic meter, uptake mechanism, commercialisation status, and detailed implementation costs for each technology. The CO2 uptake values for each technology presented in this review are as follows: CarbonCure achieves 1.0–1.5 kg CO2 per cubic meter of concrete, C4C up to 3.0 kg/m3, Solidia approximately 30–50 kg/m3, Carbstone 350 kg/m3, CO2-SUICOM 324 kg/m3, Carbicrete between 80–150 kg/m3, Carboclave 100–180 kg/m3, Neustark 20–100 kg/m3, and Blue Planet up to 440 kg/m3. These data reflect permanent mineralisation capacities reported under optimised operational conditions. In ceramics, the formation of stable carbonates through mineral carbonation can reach up to 23 wt.% in optimised laboratory and pilot studies. Cost data and deployment scales are provided where available, illustrating investment requirements and the current commercial reach of leading technologies. Despite significant progress, notable gaps remain, particularly in standardised life cycle assessments, long-term durability studies, transparent cost benchmarks at industrial scale, and protocols for quantifying permanent CO2 sequestration. Future research and innovation must prioritize the synergistic integration of CO2 uptake through advanced SCM blends and tailored mix designs that maximize CO2 uptake without compromising mechanical or durability performance. Developing robust, industry-ready carbonation protocols with harmonized measurement standards is essential to enable regulatory acceptance and cross-comparison of technologies. Scale-up efforts should focus on pilot-to-industrial transitions, including optimized supply chains, process automation, and cost-reduction strategies that leverage co-location of CO2 sources with production facilities. For ceramic and other solid waste materials, advancing mechanistic understanding of activation pathways and particle engineering will be critical to achieving higher reactivity and consistent performance. Additionally, comprehensive durability testing under realistic service conditions, spanning chloride and sulfate exposure, freeze-thaw cycling, and ASR susceptibility is imperative to ensure that CO2-enhanced materials meet or exceed conventional concrete benchmarks over decades-long service lives. By addressing these challenges through interdisciplinary collaboration, standardization, and rigorous benchmarking of LCA and techno-economic metrics, the construction sector can translate the substantial theoretical potential of CO2 capture technologies into reliable, scalable, and economically viable pathways for decarbonization.
Acknowledgement: The authors acknowledge c5Lab-Sustainable Construction Materials Association.
Funding Statement: This research was funded by the Recovery and Resilience Plan (PRR) to support Collaborative Laboratories (CoLABs), Interface Mission [Notice No. 01/C05-i02/2022].
Author Contributions: The authors confirm contribution to the paper as follows: Conceptualization, Mahboobeh Attaei, Maria Vieira, Cinthia Maia Pederneiras and Rosário Veiga; methodology, Mahboobeh Attaei, Cinthia Maia Pederneiras and Rosário Veiga; formal analysis, Mahboobeh Attaei, Cinthia Maia Pederneiras and Rosário Veiga; writing—original draft preparation, Mahboobeh Attaei, Maria Vieira, Cinthia Maia Pederneiras, Filipa Clara Coimbra and David Bastos; writing—review and editing, Mahboobeh Attaei, Cinthia Maia Pederneiras, Filipa Clara Coimbra and Rosário Veiga; supervision, Rosário Veiga. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials: Not applicable.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest to report regarding the present study.
References
1. Snellings R. Assessing, understanding and unlocking supplementary cementitious materials. RILEM Tech Lett. 2016;1:50–5. doi:10.21809/rilemtechlett.2016.12. [Google Scholar] [CrossRef]
2. Zajac M, Skocek J, Gołek Ł, Deja J. Supplementary cementitious materials based on recycled concrete paste. J Clean Prod. 2023;387:135743. doi:10.1016/j.jclepro.2022.135743. [Google Scholar] [CrossRef]
3. Khan MSH, Nguyen QD, Castel A. Carbonation of limestone calcined clay cement concrete. In: Calcined clays for sustainable concrete. Dordrecht, The Netherlands: Springer; 2017. p. 238–43. doi:10.1007/978-94-024-1207-9_38. [Google Scholar] [CrossRef]
4. Avet F, Scrivener K. Investigation of the calcined kaolinite content on the hydration of Limestone Calcined Clay Cement (LC3). Cem Concr Res. 2018;107:124–35. doi:10.1016/j.cemconres.2018.02.016. [Google Scholar] [CrossRef]
5. Merta I, Poletanovic B, Dragas J, Carevic V, Ignjatovic I, Komljenovic M. The influence of accelerated carbonation on physical and mechanical properties of hemp-fibre-reinforced alkali-activated fly ash and fly ash/slag mortars. Polymers. 2022;14(9):1799. doi:10.3390/polym14091799. [Google Scholar] [PubMed] [CrossRef]
6. Sousa V, Bogas JA, Real S, Meireles I. Industrial production of recycled cement: energy consumption and carbon dioxide emission estimation. Environ Sci Pollut Res. 2023;30(4):8778–89. doi:10.1007/s11356-022-20887-7. [Google Scholar] [PubMed] [CrossRef]
7. Beguedou E, Narra S, Afrakoma Armoo E, Agboka K, Damgou MK. Alternative fuels substitution in cement industries for improved energy efficiency and sustainability. Energies. 2023;16(8):3533. doi:10.3390/en16083533. [Google Scholar] [CrossRef]
8. Quevedo Parra S, Romano MC. Decarbonization of cement production by electrification. J Clean Prod. 2023;425:138913. doi:10.1016/j.jclepro.2023.138913. [Google Scholar] [CrossRef]
9. Cormos AM, Cormos CC. Reducing the carbon footprint of cement industry by post-combustion CO2 capture: techno-economic and environmental assessment of a CCS project in Romania. Chem Eng Res Des. 2017;123:230–9. doi:10.1016/j.cherd.2017.05.013. [Google Scholar] [CrossRef]
10. Winnefeld F, Leemann A, German A, Lothenbach B. CO2 storage in cement and concrete by mineral carbonation. Curr Opin Green Sustain Chem. 2022;38:100672. doi:10.1016/j.cogsc.2022.100672. [Google Scholar] [CrossRef]
11. Basheer L, Kropp J, Cleland DJ. Assessment of the durability of concrete from its permeation properties: a review. Constr Build Mater. 2001;15(2–3):93–103. doi:10.1016/S0950-0618(00)00058-1. [Google Scholar] [CrossRef]
12. Maia Pederneiras C, Brazão Farinha C, Veiga R. Carbonation potential of cementitious structures in service and post-demolition: a review. CivilEng. 2022;3(2):211–23. doi:10.3390/civileng3020013. [Google Scholar] [CrossRef]
13. Attaei M, Condeço J, Mateus MM, Nogueira R, Bogas JA. Amine-based additives to enhance the carbonation rate of cement-based materials: a pathway to CO2 neutrality. J Clean Prod. 2025;520:145940. doi:10.1016/j.jclepro.2025.145940. [Google Scholar] [CrossRef]
14. Silva A, Nogueira R, Bogas JA. Strategies for OPC paste carbonation: relationship between microstructure, performance and net CO2 balance. Sustainability. 2024;16(1):361. doi:10.3390/su16010361. [Google Scholar] [CrossRef]
15. Monkman S, MacDonald M. On carbon dioxide utilization as a means to improve the sustainability of ready-mixed concrete. J Clean Prod. 2017;167:365–75. doi:10.1016/j.jclepro.2017.08.194. [Google Scholar] [CrossRef]
16. Zhang D, Ghouleh Z, Shao Y. Review on carbonation curing of cement-based materials. J CO2 Util. 2017;21(3):119–31. doi:10.1016/j.jcou.2017.07.003. [Google Scholar] [CrossRef]
17. El-Hassan H. Accelerated carbonation curing as a means of reducing carbon dioxide emissions. In: Cement industry—optimization, characterization and sustainable application. London, UK: IntechOpen; 2020. p. 117–42. doi:10.5772/intechopen.93929. [Google Scholar] [CrossRef]
18. Fante M, Ribeiro FRC, Nunes DB, Modolo RCE, de Souza Kazmierczak C, Mancio M, et al. Behavior of cementitious mixtures with filler carbonate subjected to accelerated carbonation. Case Stud Constr Mater. 2022;17:e01300. doi:10.1016/j.cscm.2022.e01300. [Google Scholar] [CrossRef]
19. Rostami V, Shao Y, Boyd AJ, He Z. Microstructure of cement paste subject to early carbonation curing. Cem Concr Res. 2012;42(1):186–93. doi:10.1016/j.cemconres.2011.09.010. [Google Scholar] [CrossRef]
20. Zhang D, Cai X, Jaworska B. Effect of pre-carbonation hydration on long-term hydration of carbonation-cured cement-based materials. Constr Build Mater. 2020;231:117122. doi:10.1016/j.conbuildmat.2019.117122. [Google Scholar] [CrossRef]
21. Silva A, Nogueira R, Bogas JA, Rodrigues M. Influence of carbon dioxide as a mixture component on the cement hydration. In: International RILEM Conference on Microstructure Related Durability of Cementitious Composites. Delft, The Netherlands: Delf; 2021. p. 181–91. [Google Scholar]
22. Kwasny J, Basheer PAM, Russell M. CO2 sequestration in cement-based materials during mixing process using carbonated water and gaseous CO2. In: Proceedings of the 4th International Conference on the Durability of Concrete Structures; 2014 Jul 24–26; West Lafayette, IN, USA. p. 72–9. doi:10.5703/1288284315385. [Google Scholar] [CrossRef]
23. Fu X, Guerini A, Zampini D, Rotta Loria AF. Storing CO2 while strengthening concrete by carbonating its cement in suspension. Commun Mater. 2024;5(1):109. doi:10.1038/s43246-024-00546-9. [Google Scholar] [CrossRef]
24. De la Varga I, Sargam Y, Martins Alexio D. CO2 mineralization in the production of sustainable concrete. Rev Ing Constr. 2024;39:1–9. doi:10.7764/ric.00134.21. [Google Scholar] [CrossRef]
25. Fernández Bertos M, Simons SJR, Hills CD, Carey PJ. A review of accelerated carbonation technology in the treatment of cement-based materials and sequestration of CO2. J Hazard Mater. 2004;112(3):193–205. doi:10.1016/j.jhazmat.2004.04.019. [Google Scholar] [PubMed] [CrossRef]
26. Zatzman J. Role of CO2 injection in concrete: cement hydration reaction enhancer [Internet]. Dartmouth, NS, Canada: CarbonCure Technologies. [cited 2025 Oct 1]. Available from: https://www.carboncure.com/resources/role-of-co2-injection-in-concrete-cement-hydration-reaction-enhancer/. [Google Scholar]
27. Bonavetti VL, Rahhal VF, Irassar EF. Studies on the carboaluminate formation in limestone filler-blended cements. Cem Concr Res. 2001;31(6):853–9. doi:10.1016/S0008-8846(01)00491-4. [Google Scholar] [CrossRef]
28. Zajac M, Skocek J, Ben Haha M, Deja J. CO2 mineralization methods in cement and concrete industry. Energies. 2022;15(10):3597. doi:10.3390/en15103597. [Google Scholar] [CrossRef]
29. He Z, Li Z, Shao Y. Effect of carbonation mixing on CO2 uptake and strength gain in concrete. J Mater Civ Eng. 2017;29(10):04017176. doi:10.1061/(asce)mt.1943-5533.0002031. [Google Scholar] [CrossRef]
30. Silva A, Nogueira R, Gomes MG, Bogas JA. Carbon utilization during concrete production: reasons for the low CO2 uptake. J Build Eng. 2025;107:112766. doi:10.1016/j.jobe.2025.112766. [Google Scholar] [CrossRef]
31. Winnefeld F, Tiefenthaler J, Leemann A. Carbonated concrete slurry waste as supplementary cementitious material. J Adv Concr Technol. 2024;22(8):485–98. doi:10.3151/jact.22.485. [Google Scholar] [CrossRef]
32. Sim SR, Ryu DW. Fundamental studies on CO2 sequestration of concrete slurry water using supercritical CO2. Materials. 2022;15(1):94. doi:10.3390/ma15010094. [Google Scholar] [PubMed] [CrossRef]
33. Thomas M. Impact of CO2 utilization in fresh concrete on corrosion of steel reinforcement. Dartmouth, NS, Canada: CarbonCure Technologies Inc.; 2019. [Google Scholar]
34. CarbonCure. CarbonCure technology [Internet]. [cited 2025 Oct 1]. Available from: https://www.carboncure.com/Concrete-Producers/. [Google Scholar]
35. Monkman S, MacDonald M. Carbon dioxide upcycling into industrially produced concrete blocks. Constr Build Mater. 2016;124:127–32. doi:10.1016/j.conbuildmat.2016.07.046. [Google Scholar] [CrossRef]
36. Ravikumar D, Zhang D, Keoleian G, Miller S, Sick V, Li V. Carbon dioxide utilization in concrete curing or mixing might not produce a net climate benefit. Nat Commun. 2021;12(1):855. doi:10.1038/s41467-021-21148-w. [Google Scholar] [PubMed] [CrossRef]
37. Concrete4change. Net-zero concrete technology [Internet]. [cited 2025 Jul 31]. Available from: https://www.concrete4change.com/technology. [Google Scholar]
38. Li N, Unluer C. Towards a net zero built environment: decarbonisation of the UK cement industry. npj Mater Sustain. 2025;3(1):10. doi:10.1038/s44296-025-00052-0. [Google Scholar] [CrossRef]
39. Elroy DM. Storage CO2 in concrete for sustainable construction [Internet]. [cited 2025 Oct 1]. Available from: https://www.climeco.com/?s=Storage+CO2+in+concrete+for+sustainable+construction. [Google Scholar]
40. Fortunato LR, Parsekian GA, Neves A Jr. Feasibility analysis for implementing CO2 curing in a concrete block industry in the São Paulo Region. Rev IBRACON Estrut Mater. 2022;15(6):e15607. doi:10.1590/s1983-41952022000600007. [Google Scholar] [CrossRef]
41. Rostami V, Shao Y, Boyd AJ. Durability of concrete pipes subjected to combined steam and carbonation curing. Constr Build Mater. 2011;25(8):3345–55. doi:10.1016/j.conbuildmat.2011.03.025. [Google Scholar] [CrossRef]
42. Zajac M, Irbe L, Bullerjahn F, Hilbig H, Ben Haha M. Mechanisms of carbonation hydration hardening in Portland cements. Cem Concr Res. 2022;152:106687. doi:10.1016/j.cemconres.2021.106687. [Google Scholar] [CrossRef]
43. Zhang S, Yuan Q, Ni J, Zheng K, Xu Y, Zhang J. CO2 utilization and sequestration in ready-mix concrete—a review. Sci Total Environ. 2024;907:168025. doi:10.1016/j.scitotenv.2023.168025. [Google Scholar] [PubMed] [CrossRef]
44. Bastos D, Attaei M, Pederneiras CM, Veiga R, Santos Silva A. On the influence of mix design and pre-conditioning on the CO2—uptake of cementitious materials. MATEC Web Conf. 2025;409:15003. doi:10.1051/matecconf/202540915003. [Google Scholar] [CrossRef]
45. Zajac M, Skocek J, Skibsted J, Ben Haha M. CO2 mineralization of demolished concrete wastes into a supplementary cementitious material—a new CCU approach for the cement industry. RILEM Tech Lett. 2021;6:53–60. doi:10.21809/rilemtechlett.2021.141. [Google Scholar] [CrossRef]
46. Han SH, Jun Y, Shin TY, Kim JH. CO2 curing efficiency for cement paste and mortars produced by a low water-to-cement ratio. Materials. 2020;13(17):3883. doi:10.3390/ma13173883. [Google Scholar] [PubMed] [CrossRef]
47. Liu Z, Meng W. Fundamental understanding of carbonation curing and durability of carbonation-cured cement-based composites: a review. J CO2 Util. 2021;44:101428. doi:10.1016/j.jcou.2020.101428. [Google Scholar] [CrossRef]
48. Liu B, Qin J, Shi J, Jiang J, Wu X, He Z. New perspectives on utilization of CO2 sequestration technologies in cement-based materials. Constr Build Mater. 2021;272:121660. doi:10.1016/j.conbuildmat.2020.121660. [Google Scholar] [CrossRef]
49. Liu W, Teng L, Rohani S, Qin Z, Zhao B, Xu CC, et al. CO2 mineral carbonation using industrial solid wastes: a review of recent developments. Chem Eng J. 2021;416:129093. doi:10.1016/j.cej.2021.129093. [Google Scholar] [CrossRef]
50. von Greve-Dierfeld S, Lothenbach B, Vollpracht A, Wu B, Huet B, Andrade C, et al. Understanding the carbonation of concrete with supplementary cementitious materials: a critical review by RILEM TC 281-CCC. Mater Struct. 2020;53(6):136. doi:10.1617/s11527-020-01558-w. [Google Scholar] [CrossRef]
51. Gill DS, Mariam Abraham S. Feasibility of CO2 sequestration in concrete containing recycled aggregates. Mater Today Proc. 2023;534:631. doi:10.1016/j.matpr.2023.03.186. [Google Scholar] [CrossRef]
52. El-Hassan H, Shao Y. Early carbonation curing of concrete masonry units with Portland limestone cement. Cem Concr Compos. 2015;62:168–77. doi:10.1016/j.cemconcomp.2015.07.004. [Google Scholar] [CrossRef]
53. Xuan D, Zhan B, Poon CS. A maturity approach to estimate compressive strength development of CO2-cured concrete blocks. Cem Concr Compos. 2018;85:153–60. doi:10.1016/j.cemconcomp.2017.10.005. [Google Scholar] [CrossRef]
54. Xian X, Zhang D, Lin H, Shao Y. Ambient pressure carbonation curing of reinforced concrete for CO2 utilization and corrosion resistance. J CO2 Util. 2022;56:101861. doi:10.1016/j.jcou.2021.101861. [Google Scholar] [CrossRef]
55. Solidia concrete—a sustainable method for cement production and CO2 utilization, solidia technologies [Internet]. [cited 2025 Dec 16]. Available from: https://www.eralberta.ca/projects/details/solidia-concrete-a-sustainable-method-for-cement-production-and-co2-utilization/. [Google Scholar]
56. Carbstone Team. Carbonation: an ancient but forgotten application [Internet]. [cited 2025 Sep 11]. Available from: https://www.carbstone.be/en/technology/. [Google Scholar]
57. CO2-SUICOM Team. Carbon-capturing concrete: capturing CO2 like trees [Internet]. [cited 2025 Sep 11]. Available from: https://www.kajima.co.jp/english/techc_sus_con/technogy01/index.html. [Google Scholar]
58. CarbiCrete Site. CarbiCrete secure $12 million USD in first close of series a round [Internet]; 2022 [cited 2025 Sep 11]. Available from: https://carbicrete.com/secures-12-million/. [Google Scholar]
59. Neustark. We remove CO2 from the atmosphere—and store it permanently [Internet]. [cited 2025 Jul 31]. Available from: https://www.neustark.com/. [Google Scholar]
60. Carboclave. Carbon-sequestering technology with a concrete impact [Internet]. [cited 2025 Jul 31]. Available from: https://www.carboclave.com. [Google Scholar]
61. Blue Planet Team. Carbon negative, climate positive [Internet]. [cited 2025 Sep 11]. Available from: https://www.blueplanetsystem.com/. [Google Scholar]
62. HOLCIM. LafargeHolcim ramps up partnership with solidia technologies to capture CO2 in building materials [Internet]. 2025 [cited 2025 Jul 31]. Available from: https://www.holcim.com/media/media-releases/lafargeholcim-partnership-solidia-technologies-capture-CO2-building-materials. [Google Scholar]
63. Neustark Team. Construction & demolition waste recycles [Internet]. [cited 2025 Sep 11]. Available from: https://www.neustark.com/en. [Google Scholar]
64. Li N, Mo L, Unluer C. Emerging CO2 utilization technologies for construction materials: a review. J CO2 Util. 2022;65(9):102237. doi:10.1016/j.jcou.2022.102237. [Google Scholar] [CrossRef]
65. EUROFER. European steel in Figs. 2020. Brussels, Belgium: EUROFER; 2020. [Google Scholar]
66. Correia AG, Ferreira S, Roque A, Cavalheiro A. Portuguese steel slags. a new geomaterial. In: Proceedings of the 17th International Conference on Soil Mechanics and Geotechnical Engineering. Amsterdam, The Netherlands: IOS Press; 2009. p. 15–8. doi:10.3233/978-1-60750-031-5-15. [Google Scholar] [CrossRef]
67. Yoshioka K, Obata D, Nanjo H, Yokozeki K, Torichigai T, Morioka M, et al. New ecological concrete that reduces CO2 emissions below zero level: new method for CO2 capture and storag. Energy Proc. 2013;37:6018–25. doi:10.1016/j.egypro.2013.06.530. [Google Scholar] [CrossRef]
68. Kajima Corporation Team. Trial construction project of the “CUCO-SUICOM Dome” completed, achieving a 70% reduction in CO2 emissions: constructing an environmentally-friendly concrete dome for Expo 2025 Osaka, Kansai [Internet]. 2024 [cited 2025 Oct 1]. Available from: https://www.cuco-2030.jp/english/pressrelease/pdf/20240313.pdf. [Google Scholar]
69. Sun H, Luo X, Ren Z, Zhong J, Jin Y, Cui H, et al. Effect of carbonation curing on C3S, C2S, C3A and C4AF phases: microstructure and reactivity mechanisms. J Build Eng. 2025;112:113819. doi:10.1016/j.jobe.2025.113819. [Google Scholar] [CrossRef]
70. Hargestzaid PW, Al-Ghouleh Z, inventors. CO2-laden concrete precast products and the method of making the same. United States patent US 2019/0047175A1; 2019 Feb 14. [Google Scholar]
71. Driver JG, Bernard E, Patrizio P, Fennell PS, Scrivener K, Myers RJ. Global decarbonization potential of CO2 mineralization in concrete materials. Proc Natl Acad Sci U S A. 2024;121(29):e2313475121. doi:10.1073/pnas.2313475121. [Google Scholar] [PubMed] [CrossRef]
72. Hu R, Zhou Y, Xing F. Strategies to improve the life cycle net CO2 benefit of recycled aggregate concrete. Engineering. 2025;2025:1–19. doi:10.1016/j.eng.2024.11.040. [Google Scholar] [CrossRef]
73. Bastos D, Brazão Farinha C, Maia Pederneiras C, Veiga R, Bogas JA, Infante Gomes R, et al. Pathway to carbon neutrality in the cement industry: CO2 uptake by recycled aggregates from construction and demolition waste. Appl Sci. 2024;14(12):5224. doi:10.3390/app14125224. [Google Scholar] [CrossRef]
74. Bastos D, Brazão Farinha C, Pederneiras CM, Veiga R, Bogas JA, Infante Gomes R, et al. Towards industrial implementation: experimental campaign based on variations in temperature, humidity, and CO2 concentration in forced carbonation reactions of recycled aggregates. Appl Sci. 2025;15(9):5005. doi:10.3390/app15095005. [Google Scholar] [CrossRef]
75. Gu Z, Jiang Y, Ma Z, Shen P, Poon CS. A review on the utilization of recycled concrete powders: preparation, activity excitation and application. Mater Rep Solidwaste Ecomater. 2025;1:9520002. doi:10.26599/mrse.2025.9520002. [Google Scholar] [CrossRef]
76. Aceituno D, Zhang X, Hao H. A comprehensive review on carbon utilization pathways in concrete from conventional to improved strategies. Carbon Capture Sci Technol. 2025;16:100467. doi:10.1016/j.ccst.2025.100467. [Google Scholar] [CrossRef]
77. Shuvo AK, Sarker PK, Shaikh FUA. Efficacy of various accelerated carbonation techniques to improve recycled concrete aggregates: a comprehensive review. J Build Eng. 2024;95(1):110257. doi:10.1016/j.jobe.2024.110257. [Google Scholar] [CrossRef]
78. Gasós A, Tschurtschenthaler L, Tiefenthaler J, Mazzotti M. Industrial demonstration of indirect mineralization at 100 kg of CO2 per hour scale: process operation and performance using various industrial feedstocks. In: Proceedings of the 17th International Conference on Greenhouse Gas Control Technologies; 2024 Oct 20–24; Calgary, AB, Canada. p. 1–8. [Google Scholar]
79. Constantz BR, Lightbown V, Eng P, Zhou J, Geol P. Carbon capture and mineralogic sequestration—addressing the world wide epidemic on a world wide scale [Internet]. 2016 [cited 2025 Oct 1]. Available from: https://www.eralberta.ca/wp-content/uploads/2023/05/K130111-Blue-Planet-Final-Report-PUBLIC.pdf. [Google Scholar]
80. Cai Y, Yu L, Yang Y, Gao Y, Yang C. Effect of early age-curing methods on drying shrinkage of alkali-activated slag concrete. Materials. 2019;12(10):1633. doi:10.3390/ma12101633. [Google Scholar] [PubMed] [CrossRef]
81. Liu C, Liu P, Tang K, Guan S, Luo X, Zhang L, et al. Carbonation curing and long-term shrinkage performance of manufactured sand concrete with different strength grades from tunnel muck. Constr Build Mater. 2025;467:140406. doi:10.1016/j.conbuildmat.2025.140406. [Google Scholar] [CrossRef]
82. Zhu J, Qu Z, Liang S, Li B, Du T, Wang H. Macroscopic and microscopic properties of cement paste with carbon dioxide curing. Materials. 2022;15(4):1578. doi:10.3390/ma15041578. [Google Scholar] [PubMed] [CrossRef]
83. Xian X, Logan C, Shao Y. Dimensional stability of cement paste and concrete subject to early-age carbonation curing. Mater Struct. 2022;55(3):94. doi:10.1617/s11527-022-01926-8. [Google Scholar] [PubMed] [CrossRef]
84. Humad AM, Provis JL, Habermehl-Cwirzen K, Rajczakowska M, Cwirzen A. Creep and long-term properties of alkali-activated Swedish-slag concrete. J Mater Civ Eng. 2021;33(2):04020475. doi:10.1061/(asce)mt.1943-5533.0003381. [Google Scholar] [CrossRef]
85. Rostami V, Shao Y, Boyd AJ. Carbonation curing versus steam curing for precast concrete production. J Mater Civ Eng. 2012;24(9):1221–9. doi:10.1061/(asce)mt.1943-5533.0000462. [Google Scholar] [CrossRef]
86. Su A, Chen T, Gao X, Li Q, Qin L. Effect of carbonation curing on durability of cement mortar incorporating carbonated fly ash subjected to Freeze-Thaw and sulfate attack. Constr Build Mater. 2022;341:127920. doi:10.1016/j.conbuildmat.2022.127920. [Google Scholar] [CrossRef]
87. Mi T, Li Y, Liu W, Dong Z, Gong Q, Min C, et al. The effect of carbonation on chloride redistribution and corrosion of steel reinforcement. Constr Build Mater. 2023;363:129641. doi:10.1016/j.conbuildmat.2022.129641. [Google Scholar] [CrossRef]
88. Zhang S, Ghouleh Z, Azar A, Shao Y. Improving concrete resistance to low temperature sulfate attack through carbonation curing. Mater Struct. 2021;54(1):37. doi:10.1617/s11527-021-01626-9. [Google Scholar] [CrossRef]
89. Wu M, Chen D, Lv Z, Xiang T, Liu C, Cui L. Passivation behavior of carbon steel in carbonation-cured cement-based materials incorporating steel slags. Constr Build Mater. 2025;477:141354. doi:10.1016/j.conbuildmat.2025.141354. [Google Scholar] [CrossRef]
90. Jeon SM, Liyew G, Kim HK. Service life assessment of accelerated carbonation-cured concrete considering weathering carbonation and chloride ingression. J Build Eng. 2025;103:112094. doi:10.1016/j.jobe.2025.112094. [Google Scholar] [CrossRef]
91. González-Varela D, Hernández-Fontes C, Wang N, Pfeiffer H. State of the art and perspectives of the CO2 chemisorption in ceramics with its simultaneous or subsequent chemical transformation. Carbon Capture Sci Technol. 2023;7:100101. doi:10.1016/j.ccst.2023.100101. [Google Scholar] [CrossRef]
92. Tsiotsias AI, Georgiadis AG, Charisiou ND, Hussien AGS, Dabbawala AA, Polychronopoulou K, et al. Mid-temperature CO2 adsorption over different alkaline sorbents dispersed over mesoporous Al2O3. ACS Omega. 2024;9(10):11305–20. doi:10.1021/acsomega.3c07204. [Google Scholar] [PubMed] [CrossRef]
93. Chu J, Guo J, Guo Y, Guo B, Huang P, Zhang Z, et al. Solid-state synthesis of alkaline lithium ceramics for integrated CO2 capture and conversion to syngas. J Environ Chem Eng. 2025;13(1):115105. doi:10.1016/j.jece.2024.115105. [Google Scholar] [CrossRef]
94. Gammie C, Hesse F, Kennedy B, Bos JG, Sanna A. Encoding CO2 adsorption in sodium zirconate by neutron diffraction. Molecules. 2024;29(16):3798. doi:10.3390/molecules29163798. [Google Scholar] [PubMed] [CrossRef]
95. Mendoza-Nieto JA, Martínez-Hernández H, Pfeiffer H, Gómez-García JF. A new kinetic model for CO2 capture on sodium zirconate (Na2ZrO3an analysis under different flow rates. J CO2 Util. 2022;56:101862. doi:10.1016/j.jcou.2021.101862. [Google Scholar] [CrossRef]
96. Alcántar-Vázquez B, Duan Y, Pfeiffer H. CO oxidation and subsequent CO2 chemisorption on alkaline zirconates: Li2ZrO3 and Na2ZrO3. Ind Eng Chem Res. 2016;55(37):9880–6. doi:10.1021/acs.iecr.6b02257. [Google Scholar] [CrossRef]
97. Lubbe SJ, Crouse PL. Zircon as feedstock for zirconium chemicals, ceramics, the metal and alloys: a review. Suid Afr Tydskr Vir Natuurwetenskap En Tegnol. 2023;42(1):43–63. doi:10.36303/satnt.2023.42.1.963e. [Google Scholar] [CrossRef]
98. Stefanelli E, Vitolo S, Malquori S, Puccini M. Li4SiO4-based adsorbents for CO2 capture at high temperature: performance enhancement by ball milling process. Chem Eng Trans. 2023;100:193–8. doi:10.3303/CET23100033. [Google Scholar] [CrossRef]
99. Ida JI, Lin YS. Mechanism of high-temperature CO2 sorption on lithium zirconate. Environ Sci Technol. 2003;37(9):1999–2004. doi:10.1021/es0259032. [Google Scholar] [PubMed] [CrossRef]
100. Venegas MJ, Fregoso-Israel E, Escamilla R, Pfeiffer H. Kinetic and reaction mechanism of CO2 sorption on Li4SiO4: study of the particle size effect. Ind Eng Chem Res. 2007;46(8):2407–12. doi:10.1021/ie061259e. [Google Scholar] [CrossRef]
101. Ramírez-Moreno MJ, Romero-Ibarra IC, Ortiz-Landeros J, Pfeiffer H. Alkaline and alkaline-earth ceramic oxides for CO2 capture, separation and subsequent catalytic chemical conversion. In: CO2 sequestration and valorization. London, UK: IntechOpen; 2015. p. 253–300. doi:10.5772/57444. [Google Scholar] [CrossRef]
102. Elazar-Mittelman H, Tsapatsis M, Erlebacher J. Carbon dioxide derived carbon-ceramic composites by chemical vapor infiltration. Chem Eng J. 2025;514:163165. doi:10.1016/j.cej.2025.163165. [Google Scholar] [CrossRef]
103. Karatas E, Al-Mutwalli SA, Taher MN, Shirazi MMA, Koseoglu-Imer DY. Reusability of discarded tubular ceramic membranes for CO2 removal: a case study for membrane circularity. ACS Omega. 2023;8(32):29225–33. doi:10.1021/acsomega.3c02568. [Google Scholar] [PubMed] [CrossRef]
104. Ceramics of Italy. Converting waste into a resource using CO2 [Internet]. [cited 2025 Jul 24]. Available from: https://www.ceramica.info/en/articoli/converting-waste-into-a-resource-using-co2/. [Google Scholar]
105. Rais A, Vandenabeele D, Doppelhammer N, Asselman K, Wangermez W, Kirschhock CEA, et al. Ion exchange selectivity governs phase selection in zeolite synthesis. Mater Horiz. 2025;12(22):9601–7. doi:10.1039/d5mh01044f. [Google Scholar] [PubMed] [CrossRef]
106. Alqahtani A, Addassi M, Hoteit H, Oelkers E. Rapid CO2 mineralization by zeolite via cation exchange. Sci Rep. 2025;15(1):958. doi:10.1038/s41598-024-82520-6. [Google Scholar] [PubMed] [CrossRef]
107. Martín D, Aparicio P, Galán E. Mineral carbonation of ceramic brick at low pressure and room temperature. A simulation study for a superficial CO2 store using a common clay as sealing material. Appl Clay Sci. 2018;161:119–26. doi:10.1016/j.clay.2018.04.021. [Google Scholar] [CrossRef]
108. Martín D, Aparicio P, Galán E. Time evolution of the mineral carbonation of ceramic bricks in a simulated pilot plant using a common clay as sealing material at superficial conditions. Appl Clay Sci. 2019;180:105191. doi:10.1016/j.clay.2019.105191. [Google Scholar] [CrossRef]
109. European Comission. Zero emission firing strategies for ceramic tiles by oxy-fuel burners and CO2 sequestration with recycling of byproducts [Internet]. [cited 2025 Jul 24]. Available from: https://webgate.ec.europa.eu/life/publicWebsite/project/LIFE12-ENV-IT-000424/zero-emission-firing-strategies-for-ceramic-tiles-by-oxy-fuel-burners-and-co2-sequestration-with-recycling-of-byproducts. [Google Scholar]
110. Carey P. Carbonation of alkaline residues in the manufacture of lightweight aggregate [Internet]; 2022 [cited 2025 Sep 17]. Available from: https://www.carbon8.co.uk/. [Google Scholar]
111. Antzaras AN, Papalas T, Heracleous E, Kouris C. Techno–economic and environmental assessment of CO2 capture technologies in the cement industry. J Clean Prod. 2023;428:139330. doi:10.1016/j.jclepro.2023.139330. [Google Scholar] [CrossRef]
112. Shao Y, Zhou X, Monkman S. A new CO2 sequestration process via concrete products production. In: Proceedings of the 2006 IEEE EIC Climate Change Conference; 2006 May 10–12; Ottawa, ON, Canada. p. 1–6. doi:10.1109/EICCCC.2006.277189. [Google Scholar] [CrossRef]
113. He Z, Wang S, Mahoutian M, Shao Y. Flue gas carbonation of cement-based building products. J CO2 Util. 2020;37:309–19. doi:10.1016/j.jcou.2020.01.001. [Google Scholar] [CrossRef]
114. Monkman S, MacDonald M, Hooton RD, Sandberg P. Properties and durability of concrete produced using CO2 as an accelerating admixture. Cem Concr Compos. 2016;74:218–24. doi:10.1016/j.cemconcomp.2016.10.007. [Google Scholar] [CrossRef]
115. Wang T, Yi Z, Song J, Zhao C, Guo R, Gao X. An industrial demonstration study on CO2 mineralization curing for concrete. iScience. 2022;25(5):104261. doi:10.1016/j.isci.2022.104261. [Google Scholar] [PubMed] [CrossRef]
116. EU Life Project Team. Solidia low CO2 cement: from cement production to precast industry [Internet]. 2022 [cited 2025 Aug 20]. Available from: https://webgate.ec.europa.eu/life/publicolidia-low-co2-cement-from-cement-production-to-precast-industry. [Google Scholar]
117. VITO. Carbstone: converting CO2 to building materials [Internet]. [cited 2025 Oct 1]. Available from: https://vito.be/en/applications/carbstone. [Google Scholar]
118. Hanifa M, Agarwal R, Sharma U, Thapliyal PC, Singh LP. A review on CO2 capture and sequestration in the construction industry: emerging approaches and commercialised technologies. J CO2 Util. 2023;67:102292. doi:10.1016/j.jcou.2022.102292. [Google Scholar] [CrossRef]
119. Carbon-negative concrete: a game changer for sustainable future [Internet]. [cited 2025 Oct 1]. Available from: https://www.japan.go.jp/kizuna/2022/08/carbon-negative_concrete.html. [Google Scholar]
120. ASTM International. Environmental product declaration-normal-weight and light-weight concrete masonry units as manufactured by members of Canadian concrete masonry producers association (CCMPA) [Internet]; 2017 [cited 2025 Oct 1]. Available from: https://ccmpa.ca/wp-content/uploads/2016/12/CCMPA_EPD_2016-08-24.pdf. [Google Scholar]
121. Renveska S. Neustark secures $69M to scale promising carbon removal technology [Internet]; 2024 [cited 2025 Oct 1]. Available from: https://carbonherald.com/Neustark-Secures-69m-to-Scale-Promising-Carbon-Removal-Technology/. [Google Scholar]
122. NGK beings CO2 capture demonstration for CCUS: capturing CO2 from exhaust gas of ceramics firing kiln to be utilized for methanation. 2023 [cited 2025 Oct 1]. Available from: https://www.ngk-insulators.com/en/news/20231108_1.html. [Google Scholar]
123. Gomes KV, Woodall CM, Pilorgé H, Psarras P, Wilcox J. Techno-economic analysis of indirect carbonation processes for carbon sequestration using mining waste. Energy Adv. 2025;4(3):435–46. doi:10.1039/d4ya00567h. [Google Scholar] [CrossRef]
124. Wang F, Dreisinger D. Status of CO2 mineralization and its utilization prospects. Miner Miner Mater. 2022;1:4. doi:10.20517/mmm.2022.02. [Google Scholar] [CrossRef]
125. Wescombe A, Lyons N, Parkinson C, Pintimalli F, Dirckze G, Dando R, et al. Carbon capture utilisation and storage technologies and concrete: permanent sequestration of CO2 through cabron injected concrete and carbon injected reclaimed water [Internet]. 2024 [cited 2025 Oct 1]. Available from: https://roads.org.au/wp-content/uploads/Group-5-CCUS-Technologies-and-concrete.pdf. [Google Scholar]
126. Shearer A, Molinaro M, Montazerian M, Sly JJ, Miola M, Baino F, et al. The unexplored role of alkali and alkaline earth elements (ALAEs) on the structure, processing, and biological effects of bioactive glasses. Biomater Sci. 2024;12(10):2521–60. doi:10.1039/d3bm01338c. [Google Scholar] [PubMed] [CrossRef]
127. Hargis CW, Chen I, Wang Y, Maraghechi H, Gilliam RJ, Monteiro PJM. Microstructure development of calcium carbonate cement through polymorphic transformations. Cem Concr Compos. 2024;153:105715. doi:10.1016/j.cemconcomp.2024.105715. [Google Scholar] [CrossRef]
128. Murali G, Hassas N, Abdelgader HS. Ceramic waste as a sustainable cementitious resource: pathways to cleaner and high-performance concrete. Clean Mater. 2025;18:100352. doi:10.1016/j.clema.2025.100352. [Google Scholar] [CrossRef]
129. Gusca J, Blumberga D. Simplified dynamic life cycle assessment model of CO2 compression, transportation and injection phase within carbon capture and storage. Energy Proc. 2011;4:2526–32. doi:10.1016/j.egypro.2011.02.149. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF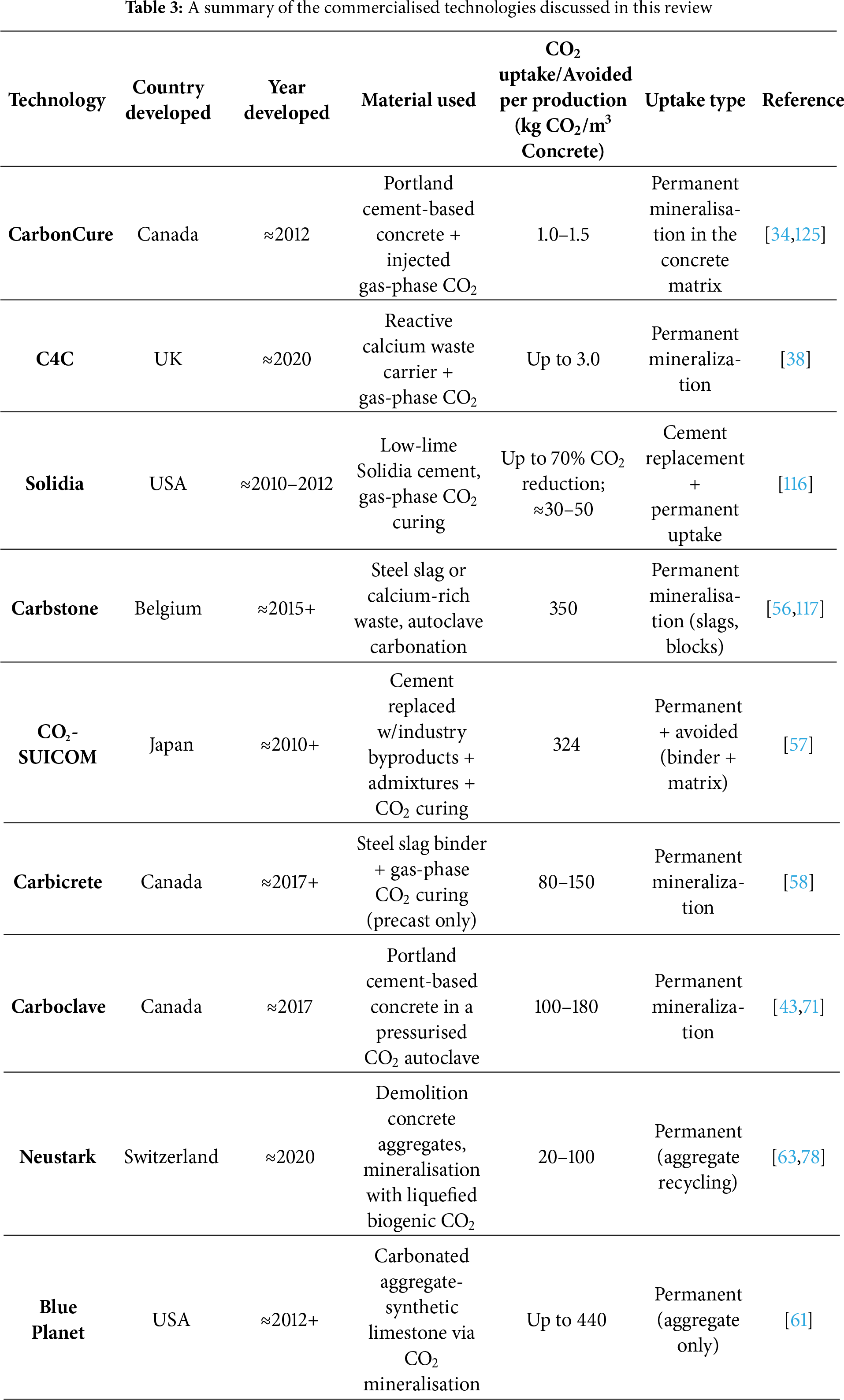
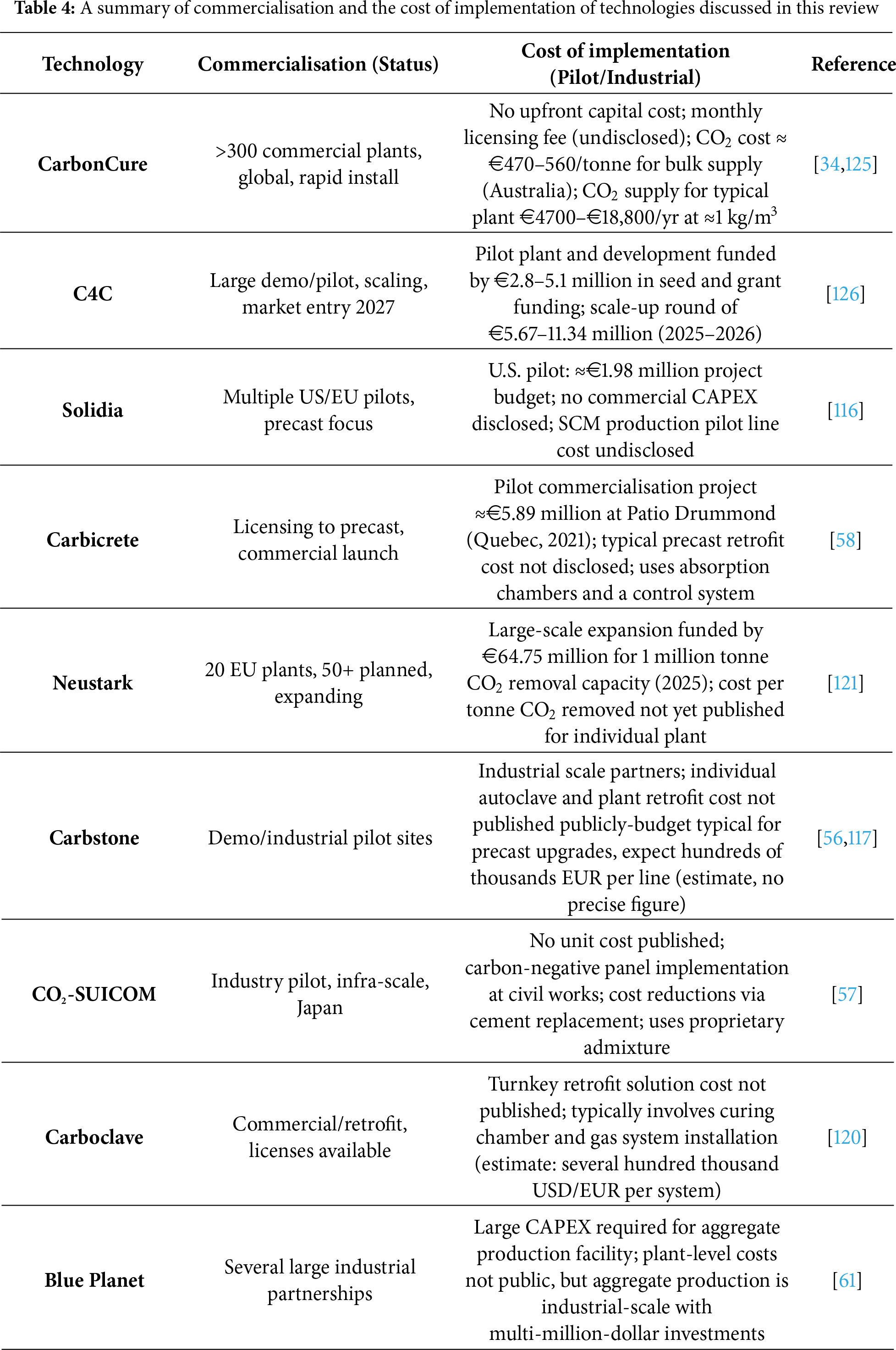
 Downloads
Downloads
 Citation Tools
Citation Tools