 Open Access
Open Access
ARTICLE
Evaporation of a CO2 Droplet in a High Temperature, Supercritical Pressure Environment
1 Department of Physics, Faculty of Sciences, Université de Lomé, Lomé, 01 BP 1515, Togo
2 Centre d’Excellence Régional Pour la Maîtrise de l’Electricité (CERME), Université de Lomé, Lomé, 01 BP 1515, Togo
3 Laboratoire d’Analyse de Modélisation Mathématique et Applications (LAMMA), Université de Lomé, Lomé, 01 BP 1515, Togo
* Corresponding Author: Yendoubouame Lare. Email:
(This article belongs to the Special Issue: Heat and Mass Transfer on A Small Temporal and Spatial Scale)
Frontiers in Heat and Mass Transfer 2026, 24(1), 12 https://doi.org/10.32604/fhmt.2025.074506
Received 13 October 2025; Accepted 15 December 2025; Issue published 28 February 2026
Abstract
This study presents a numerical investigation of the transient relaxation dynamics of a near-critical CO2 droplet immersed in a warmer supercritical environment composed of the same fluid. Three thermodynamic regimes were analysed: quasi-critical (Keywords
Nomenclature
| Symbol | Definition Unit |
| Radial distance from droplet center | |
| Instantaneous droplet radius | |
| Radial velocity component | |
| Time | |
| Temperature | |
| Critical temperature of CO2 (31.1°C) | |
| Pressure | |
| Critical pressure of CO2 (73.8 bar) | |
| Density | |
| Critical density of CO2 (467 kg· | |
| Specific heat at constant pressure | |
| Specific heat at constant volume | |
| Thermal conductivity | |
| Specific gas constant | |
| Van der Waals constants | |
| Acoustic time scale | |
| Diffusion time scale | |
| Mach number - | |
| Ratio of specific heats - |
Droplet evaporation plays a central role in the performance of liquid-fuel combustion systems, including diesel engines, gas turbines, and rocket thrusters. In these systems, fuel is injected into the combustion chamber as a cloud of droplets, where it vaporizes and oxidizes to release thermal energy [1,2]. When temperature and pressure exceed the fluid’s critical point, a supercritical environment emerges, fundamentally altering thermodynamic properties and eliminating the distinct liquid gas interface [3].
Under such conditions, evaporation mechanisms become unconventional and difficult to model. The nonlinear coupling of mass, momentum, and energy transport, combined with the absence of a sharp interface, challenges the validity of classical approaches. Since the 1950s, several models have been proposed including the well-known d2-law of Spalding [4] and Godsave [5], which assumes a linear decrease of droplet diameter squared with time. However, this law, even when extended to supercritical regimes, relies on assumptions that break down at elevated pressures [6–8].
Experimental studies have further revealed phenomena that classical models cannot fully explain, such as thermal instabilities, pseudo-boiling phenomena, and discontinuities in flow fields, often linked to incomplete combustion and increased pollutant emissions [9–11]. Spalding [6] proposed that under critical conditions, the droplet behaves as a point source of fluid. Sánchez-Tarifa et al. [12] introduced a three-zone model (cold droplet, transition layer, quasi-static outer region), enabling estimation of the recession rate of the intermediate layer via temperature profile analysis. These studies confirmed the partial applicability of the d2-law in supercritical regimes, while highlighting its limitations near the critical point [13–15].
In response to these challenges, recent research has advanced the modelling of transcritical and supercritical evaporation. Ly et al. [16] identified four distinct regimes governing the transition from subcritical evaporation to supercritical vaporization and dense fluid–gas mixing, using a diffuse interface method coupled with molecular simulations. Cheng and Xia [17] emphasized the potential of supercritical CO2 as a thermal transport fluid in advanced energy systems, citing its high thermal conductivity, diffusivity, and low viscosity. Despite these advances, several studies have shown that instability and experimental difficulties persist. Min et al. [18] reported non-monotonic profiles and transient instabilities in near-critical regimes, while Kang et al. [19] emphasized the efficient heat transfer characteristics of deep supercritical conditions. Majumdar et al. [20] observed ultrafast molecular reorganization, confirming the singularity of supercritical regimes. Simeoni et al. [21], and Lopes et al. [22] further underlined the experimental challenges in characterizing thermal instabilities and pseudo-boiling phenomena. In addition to these studies, recent reviews have broadened the context of advanced fluids and supercritical CO2 applications. Furthermore, Awais et al. [23] reviewed nanofluid heat transfer and pressure drop performance, underlining the trade-off between enhanced thermal conductivity and increased flow resistance. While nanofluids rely on particle-driven mechanisms, the present study focuses on pure CO2 droplets, thereby contributing complementary insights into high-performance thermal systems. These results point to the need for a unified framework capable of bridging unstable near-critical dynamics with stabilized deep supercritical behavior, while distinguishing physical instabilities from numerical artifacts.
Taken together, these findings underline that important gaps remain in our understanding of droplet evaporation under supercritical conditions. Specifically: (i) the lack of quantitative validation of numerical models against experimental data near the critical point, (ii) the difficulty of distinguishing physical instabilities from numerical artifacts, and (iii) the absence of a unified framework capable of capturing transient relaxation dynamics in supercritical regimes. These bottlenecks limit predictive capability and hinder the design of efficient propulsion and energy systems.
To address these gaps, the present study introduces a multi-order perturbation framework based on the compressible Navier Stokes equations and a Van der Waals thermodynamic formulation. This approach captures non-ideal fluid behaviour, including molecular interactions and density fluctuations near the critical point. CO2 is selected as the working fluid due to its chemical stability, low viscosity, and favourable critical coordinates coordinates (Tc = 31.1°C, Pc = 73.8 bar) [24]. The objective is to analyse the transient relaxation dynamics of a cold CO2 droplet immersed in a warmer supercritical environment, focusing on the structure and evolution of the interfacial transition layer and the spatiotemporal behavior of velocity, density, and temperature fields. A numerical approach is implemented, solving the conservation equations with appropriate discretization and parametric control of evaporation conditions. By explicitly addressing the limitations of classical models and experimental bottlenecks, this work aims to provide new insights into the mechanisms governing droplet evaporation in supercritical regimes.
We consider a cold droplet of pure carbon dioxide (CO2) suddenly introduced into an infinite hotter supercritical environment composed of the same fluid. The droplet is initially at rest, with uniform temperature equal to the critical temperature
Let
The following assumptions are adopted to simplify the physical model:
• The fluid is pure carbon dioxide (CO2), treated as a single component fluid.
• The droplet is perfectly spherical and isolated, allowing the problem to be reduced to a radial geometry in spherical coordinates.
• Viscosity, gravity, and surface tension are neglected; pressure is assumed spatially uniform.
• The regime is supercritical: T > Tc = 31.1°C; P > Pc = 73.8 bar.
• Evaporation is driven purely by thermal gradients; vapor diffusion is neglected.
• The thermodynamic behavior is modeled using a realistic Van der Waals equation of state, accounting for molecular interactions and excluded volume effects.
• The heat capacity at constant volume, Cv is assumed invariant and equal to that of an ideal gas.
Under these assumptions, the governing equations reduce to the spherically symmetric inviscid conservation laws for mass, momentum, and energy, expressed as follows [13,15]:
The parameters
These relations ensure that the non-ideal equation of state reproduces the correct critical point of CO2. Based on these definitions, the governing conservation equations are subsequently reduced to a one-dimensional (1D) dimensionless form under spherical symmetry [13]:
In the critical and supercritical regimes, the thermal conductivity of CO2 is expressed as follows [25]:
Eq. (3) expresses effective thermal conductivity as a function of reduced temperature and density. The first term
A constant temperature
where
First approximation system Eq. (4):
Second approximation system Eq. (5):
where:
•
•
•
•
with the initial conditions:
the boundary conditions:
A constant wall-temperature boundary condition was imposed to isolate the intrinsic effects of thermodynamic regimes, without introducing additional convective coupling. This choice highlights the fundamental differences between thermal confinement, relaxation, and homogenization across the regimes studied. Consequently, the simulations were carried out using CO2 as the working fluid, with temperature-dependent thermophysical properties near the critical point.
The system is nondimensionalized using critical fluid properties, and an asymptotic expansion with respect to the Mach number yields zeroth- and first-order perturbation systems. Due to the nonlinearity of the governing equations, no analytical solution was possible, therefore, the equations were solved numerically via a coupled finite volume approach [27–29] with second-order centered spatial discretization and an Crank-Nicolson scheme ensuring stability at large time steps. The domain is discretized into concentric radial cells, refined near the droplet-fluid interface to capture steep thermal gradients, while convective terms were linearized using a Newton-Raphson method. A mesh independence study was conducted across three refinement levels, with the convergence criterion defined as:
where (N) and (N′) are two successive meshes, and
The numerical simulations reveal distinct evaporation behaviors across the three thermodynamic regimes. In the quasi-critical regime, the droplet maintains a sharp thermal gradient, and the transition layer remains thin and well-defined. As the system enters the transitional regime, the interfacial layer broadens, and nonlinear coupling between temperature and velocity fields becomes pronounced, leading to nonmonotonic relaxation profiles and localized thermal waves. It is worth noting that, although the model relies on spherical symmetry, the observed instabilities suggest that, if this symmetry were broken, they would manifest anisotropically, leading to non-uniform evaporation rates and asymmetric redistribution of mass and heat.
4.1 Velocity Evolution in the Droplet
Figs. 1 and 2 illustrate the spatiotemporal evolution of the flow velocity within a carbon dioxide droplet immersed in a supercritical environment, evaluated at zeroth and first order, respectively. Both figures correspond to reduced temperature
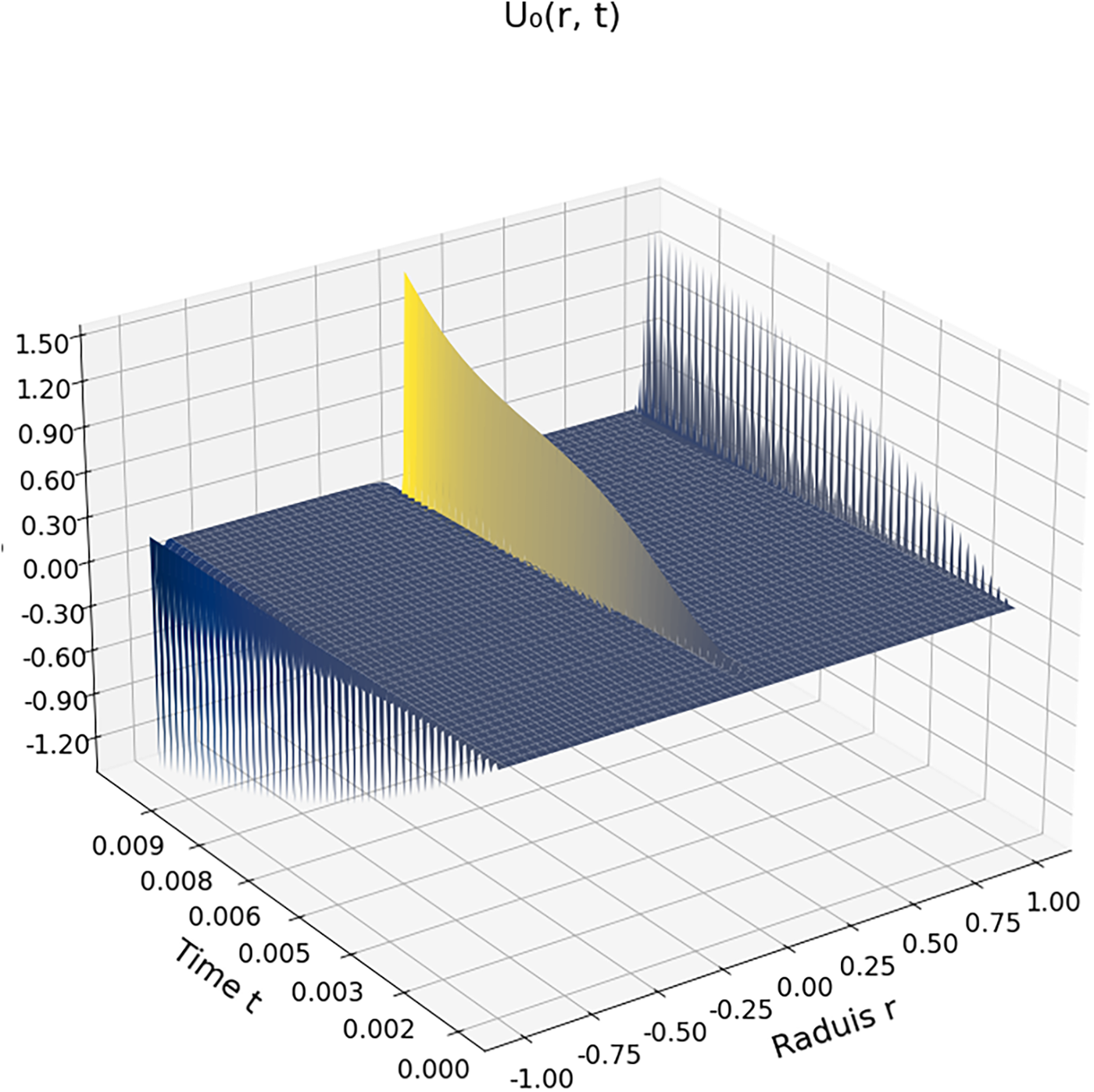
Figure 1: Zeroth-order velocity field
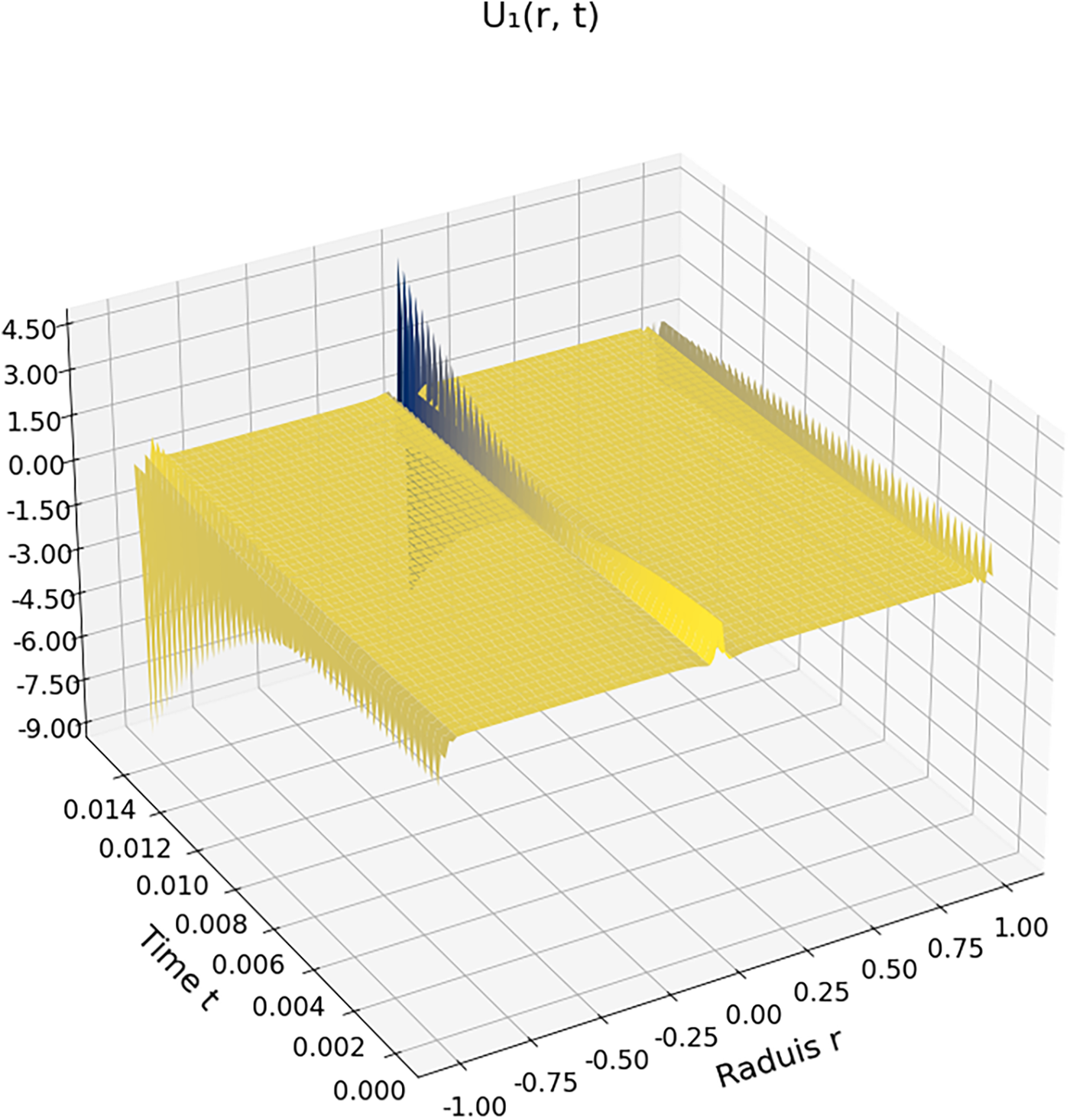
Figure 2: First-order velocity correction
Fig. 1 shows the zeroth-order velocity field
Fig. 2 presents the first-order velocity correction
Fig. 3 compares radial velocity profiles
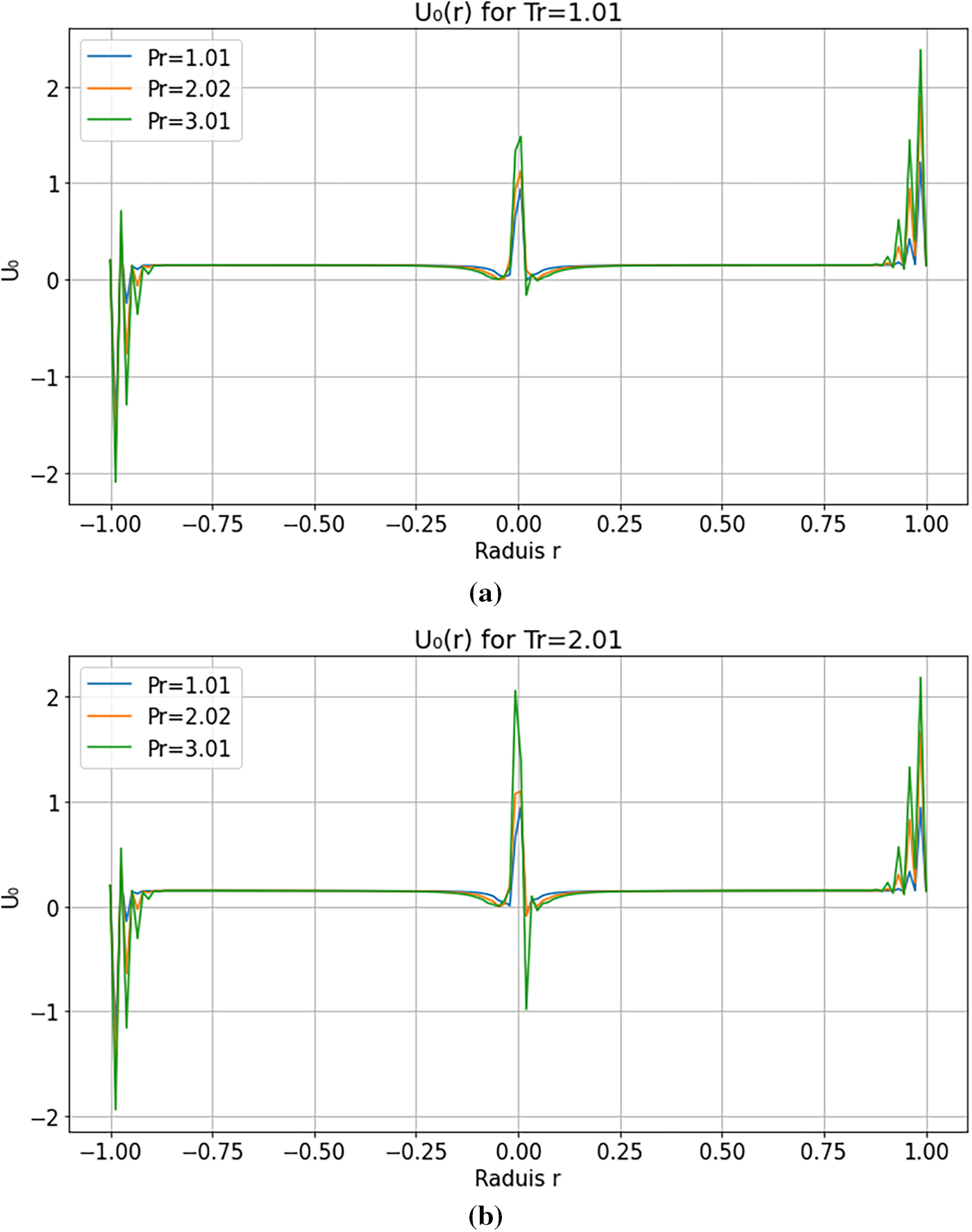
Figure 3: (a): Radial velocity profiles
Sharp velocity gradients often centred near
Thermodynamic instabilities manifest through non-monotonic velocity profiles and coherent relaxation fronts, in agreement with previous observations in supercritical fluids (e.g., Simeoni et al. [21]). In contrast, numerical instabilities are sensitive to discretization parameters such as time step and mesh resolution. Convergence and mesh-independence tests were performed, confirming that the oscillations and gradients reported here are robust and of physical origin.
Overall, the radial velocity field emerges as a key indicator of transient regimes and coupled mechanisms involving thermal diffusion, mechanical compression, and interfacial instability. These results reinforce the relevance of multi-order modelling approaches for capturing the complex interplay between thermodynamic gradients, compressibility, and transient flow structures in supercritical droplet dynamics.
The quasi-critical regime exhibits localized perturbations and radial fronts propagating toward the interface. These structures can be interpreted as signatures of internal recirculation, contributing to non-monotonic velocity profiles and influencing the redistribution of mass and heat.
The spatiotemporal evolution of the density field within a carbon dioxide droplet immersed in a supercritical environment reveals distinct dynamic regimes. Fig. 4 shows the zeroth-order density field
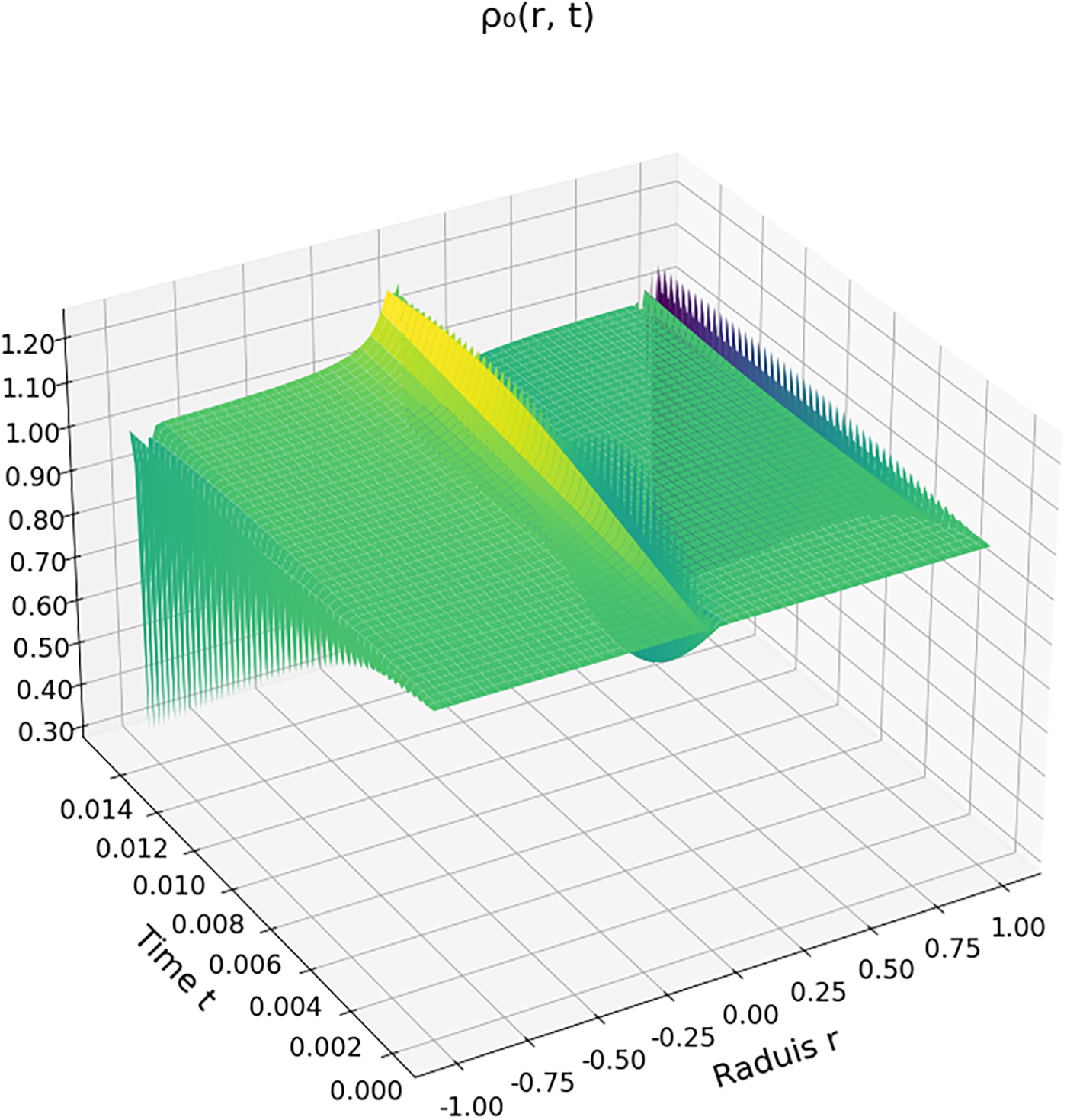
Figure 4: Zeroth-order density field
Fig. 5 presents the first-order correction
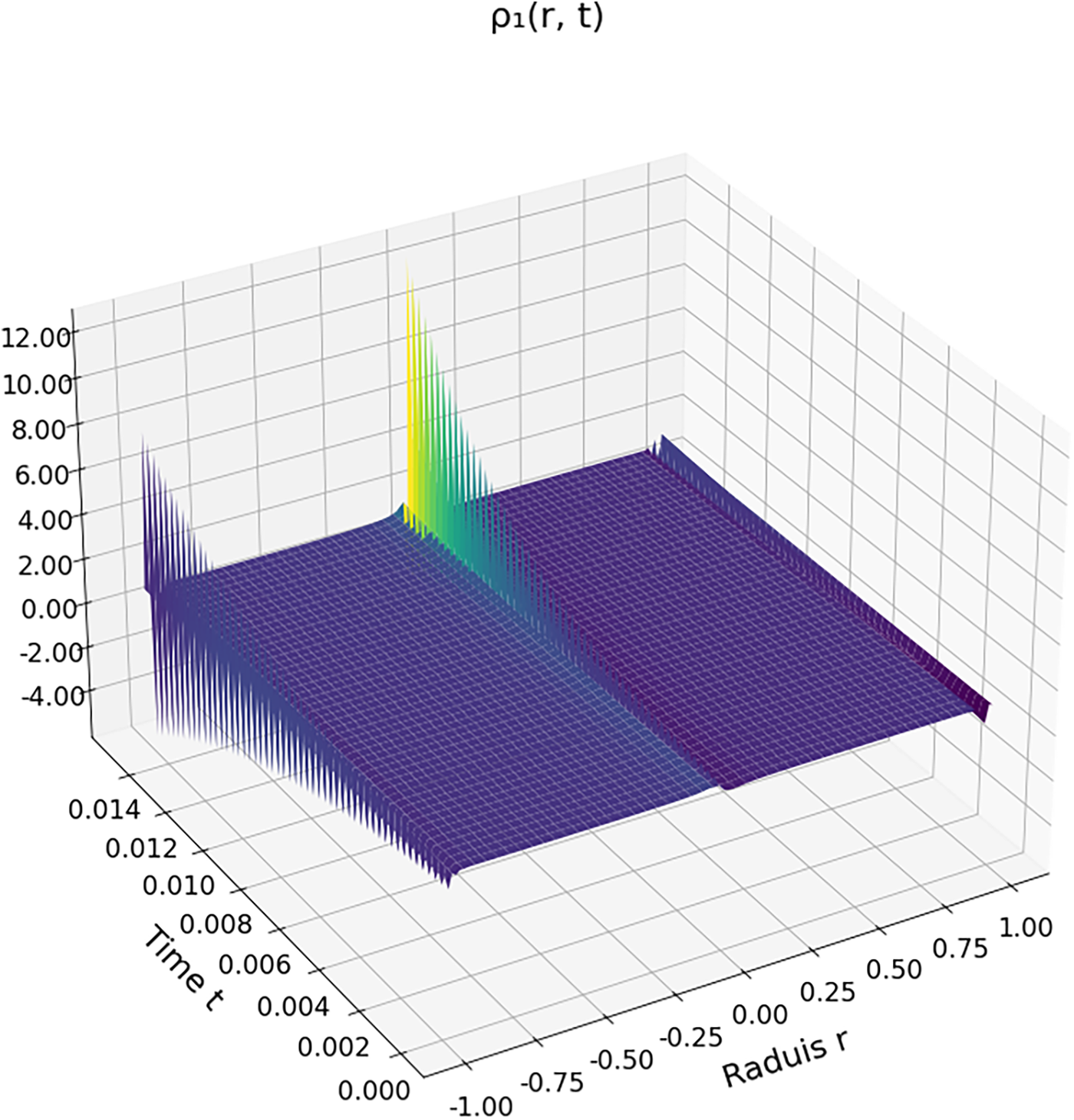
Figure 5: First-order density field
Figs. 6 and 7 illustrate the radial density profiles
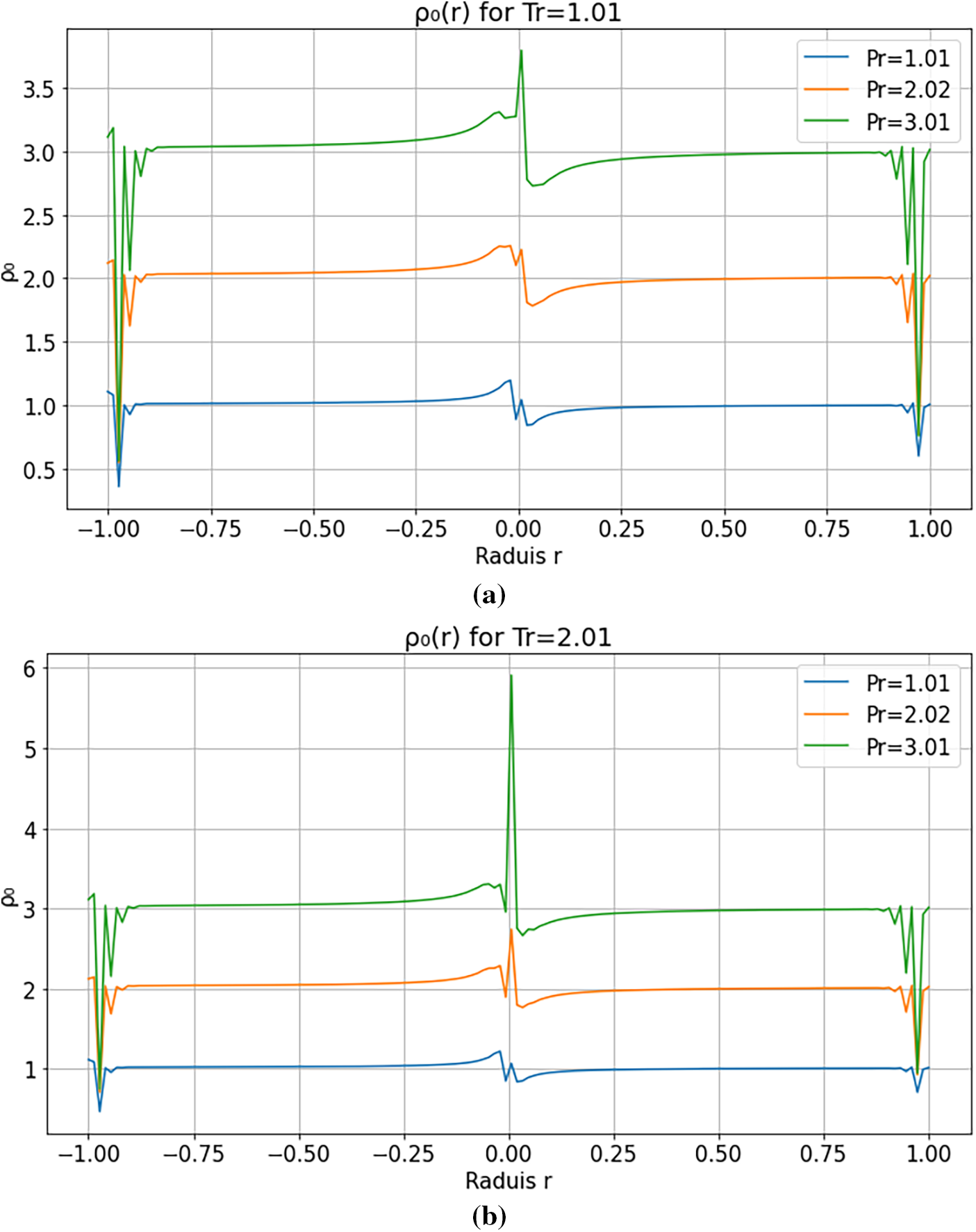
Figure 6: (a): Transition from unstable to confined regimes radial density profiles
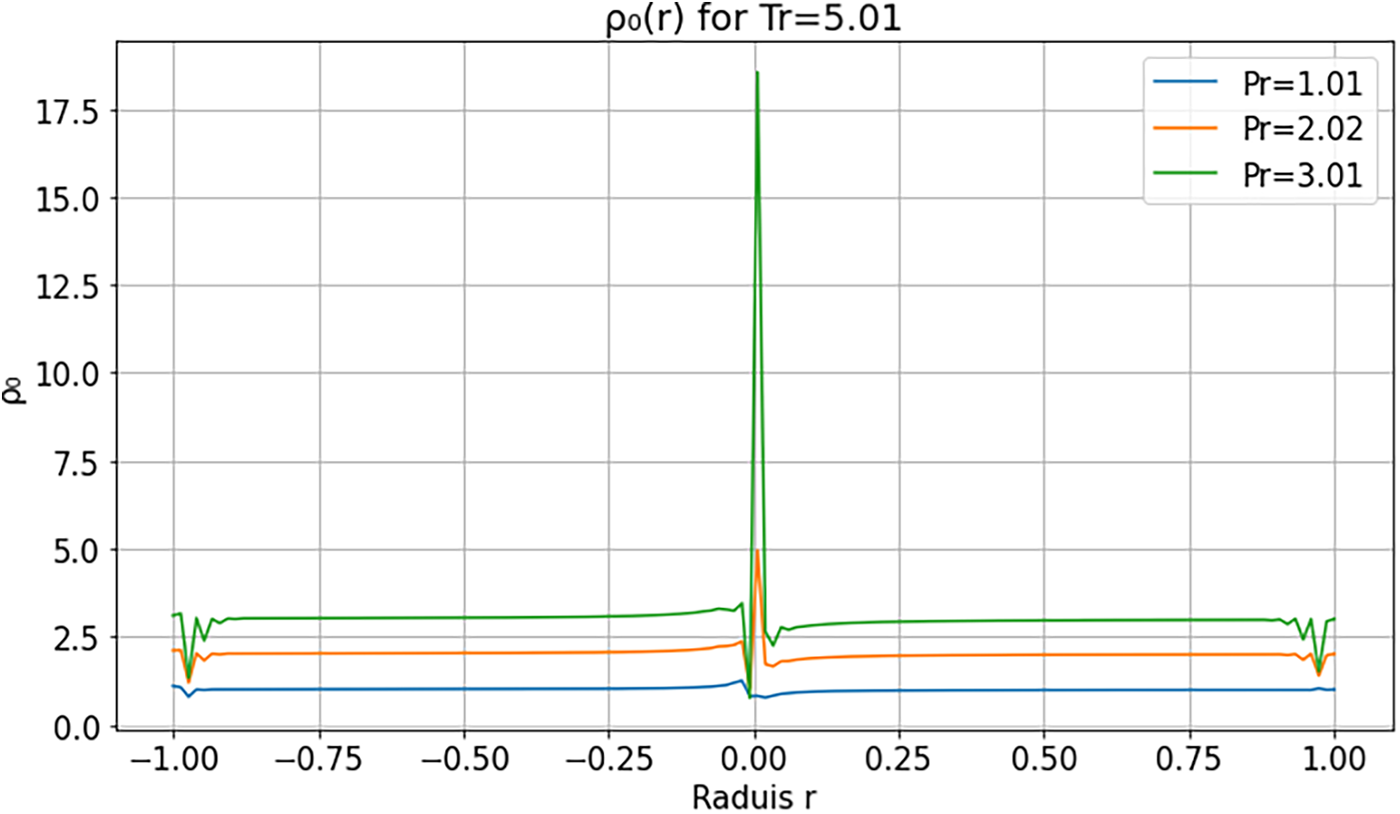
Figure 7: Radial density profile
At moderate and high reduced temperatures (
These results are consistent with previous studies [9,30], which reported nonmonotonic density profiles and interfacial instabilities in supercritical droplets. They underscore the relevance of multi-order modeling for capturing transient regimes and phase-transition dynamics in supercritical environments.
The observed velocity and density gradients reflect a competition between thermal diffusion, compressibility, and mass confinement. At low reduced pressure/temperature, enhanced compressibility amplifies the system’s sensitivity to perturbations, producing peripheral oscillations and radially propagating relaxation fronts. At higher reduced pressure/temperature, stronger molecular cohesion and more uniform thermal expansion damp local disturbances, yielding a confined and stable regime. Non-monotonic profiles therefore indicate an out-of-equilibrium response where diffusion does not instantaneously offset compressibility-induced gradients. These interpretations are consistent with experimental reports [2,31].
4.3 Evolution of the Temperature in the Droplet
Figs. 8–14 illustrate the temperature field evolution within a CO2 droplet subjected to various supercritical regimes, governed by changes in reduced temperature and pressure.
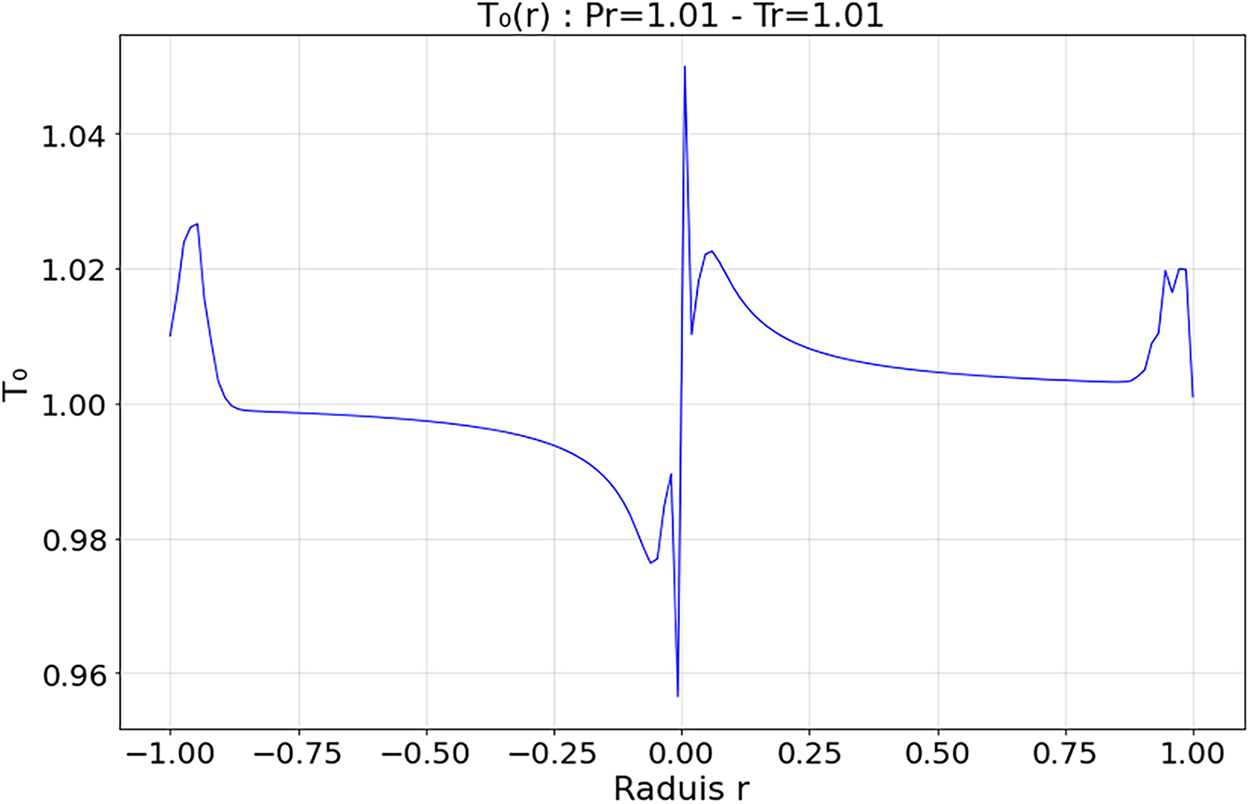
Figure 8: Spatial evolution zeroth-order heat flux within the droplet at Tr = 1.01 and Pr = 1.01
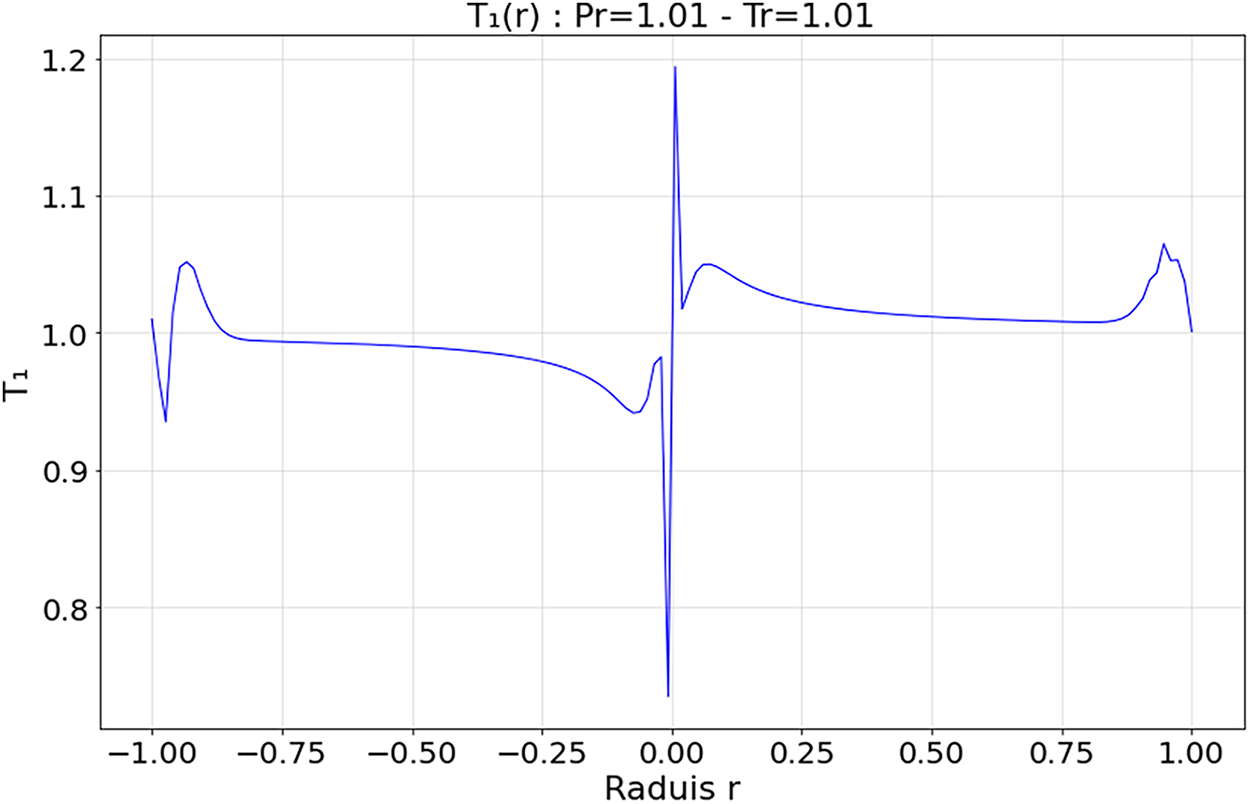
Figure 9: Spatial evolution first-order heat flux within the droplet at Tr = 1.01 and Pr = 1.01
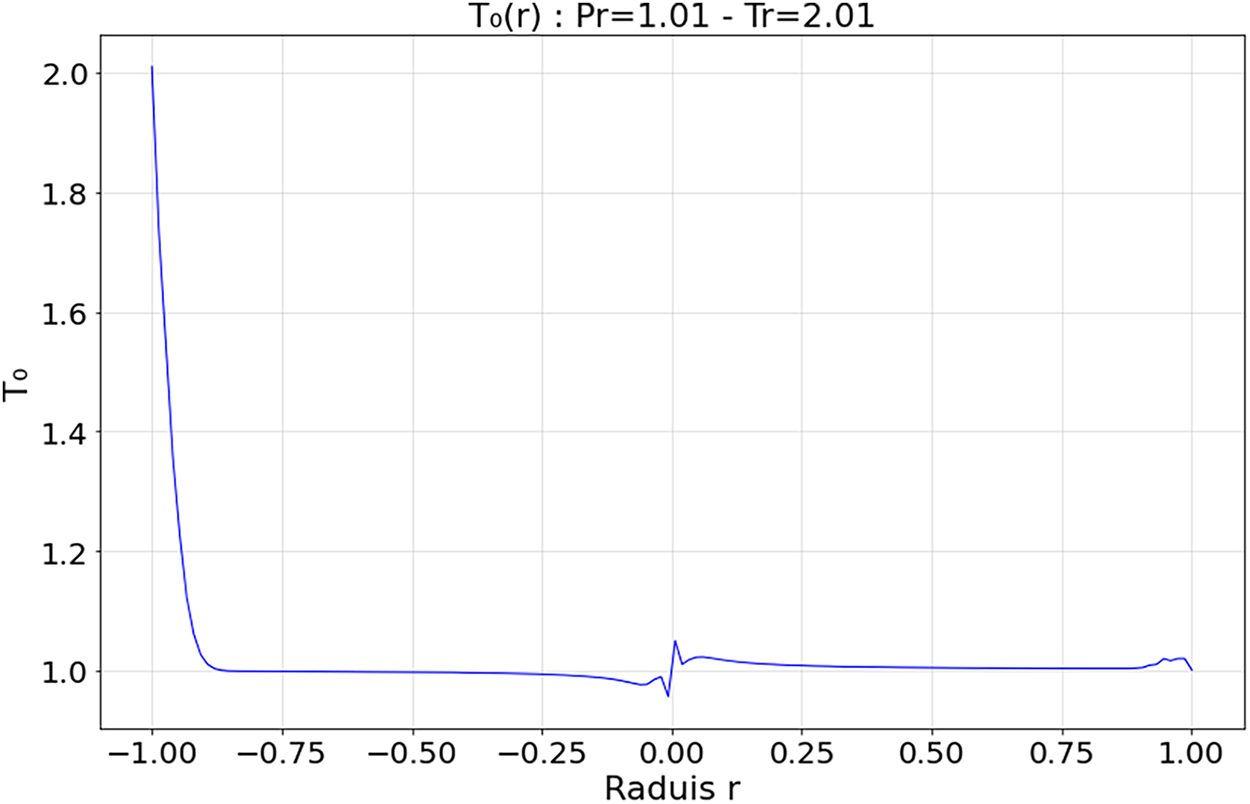
Figure 10: Spatial evolution zeroth-order heat flux within the droplet at Tr = 2.01 and Pr = 1.01
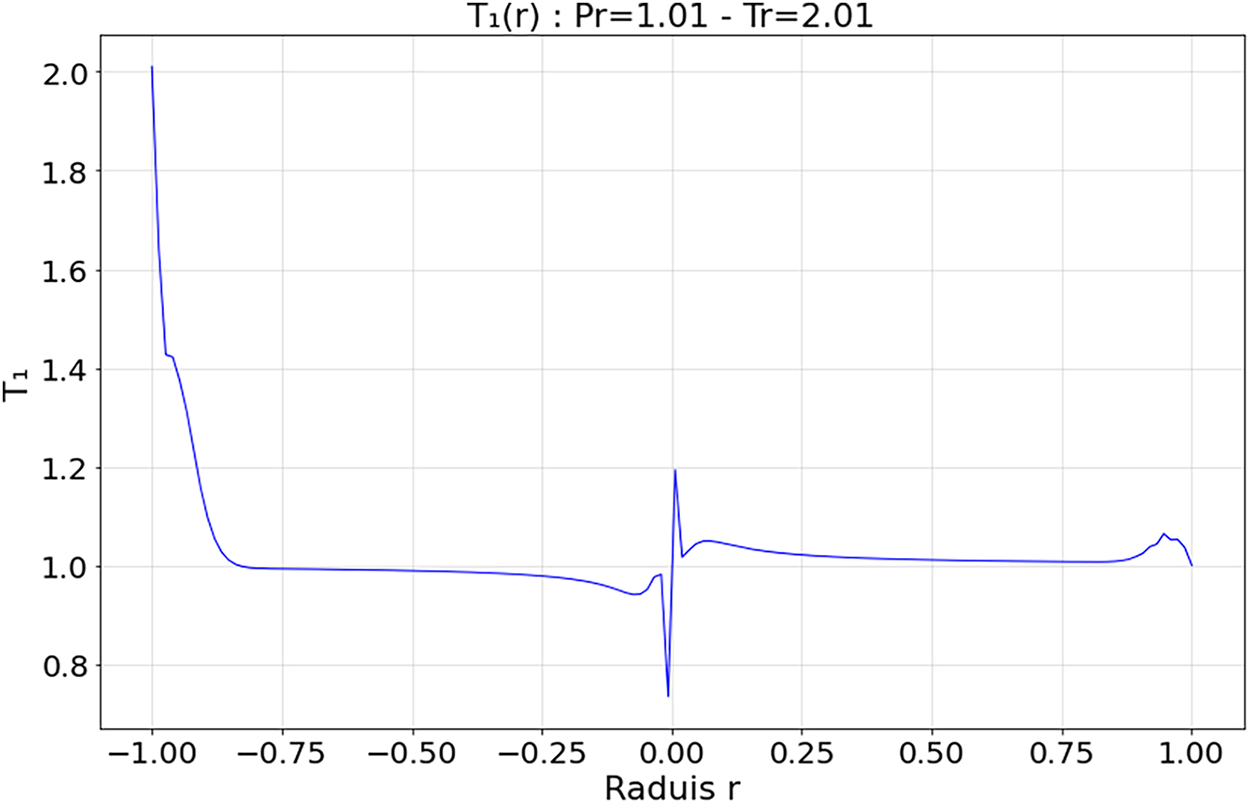
Figure 11: Spatial evolution first-order heat flux within the droplet at Tr = 2.01 and Pr = 1.01
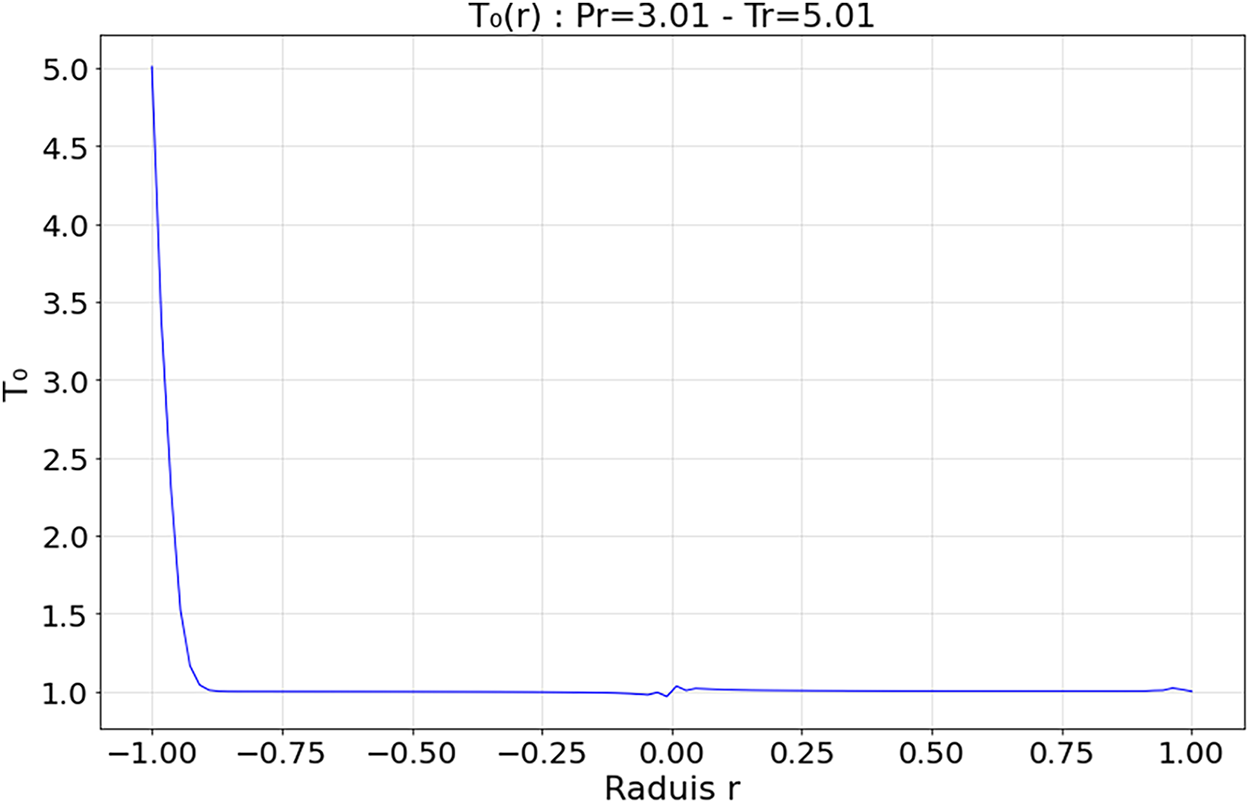
Figure 12: Spatial evolution zeroth-order heat flux within the droplet at Tr = 5.01 and Pr = 3.01
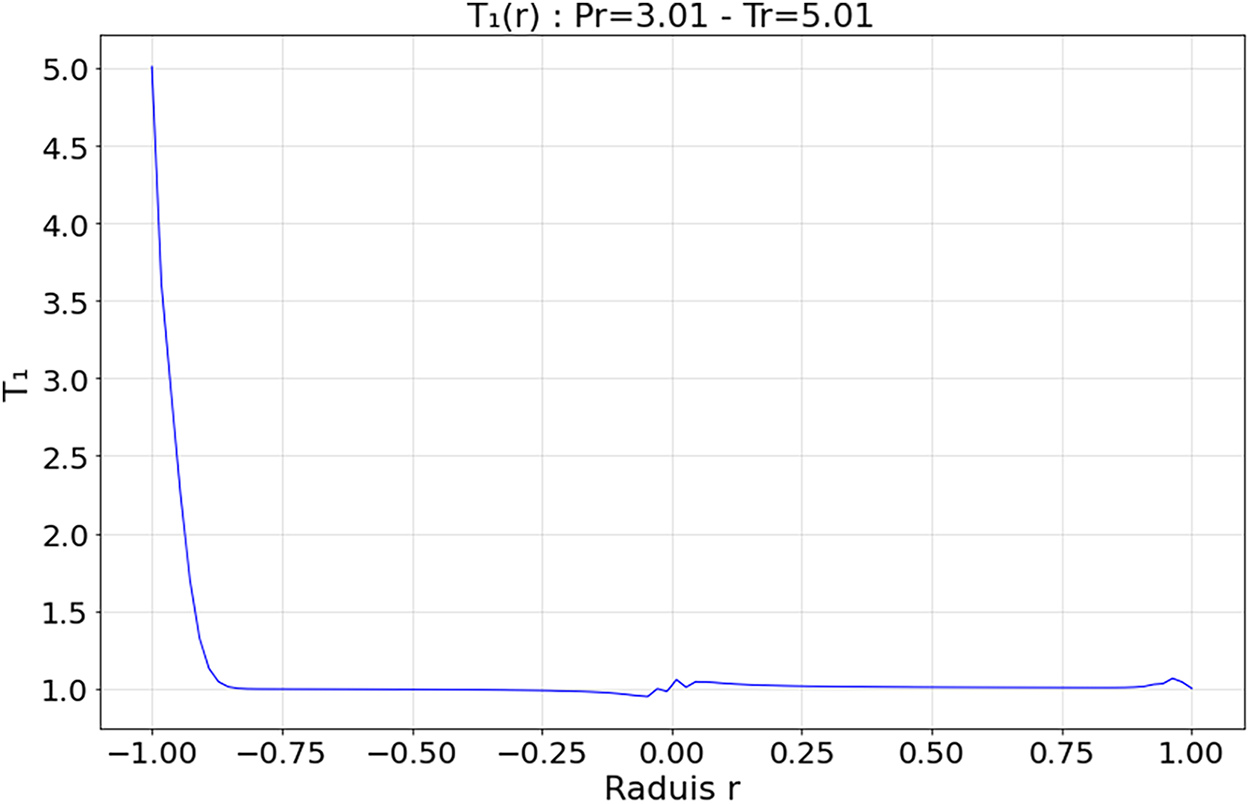
Figure 13: Spatial evolution first-order heat flux within the droplet at Tr = 5.01 and Pr = 3.01
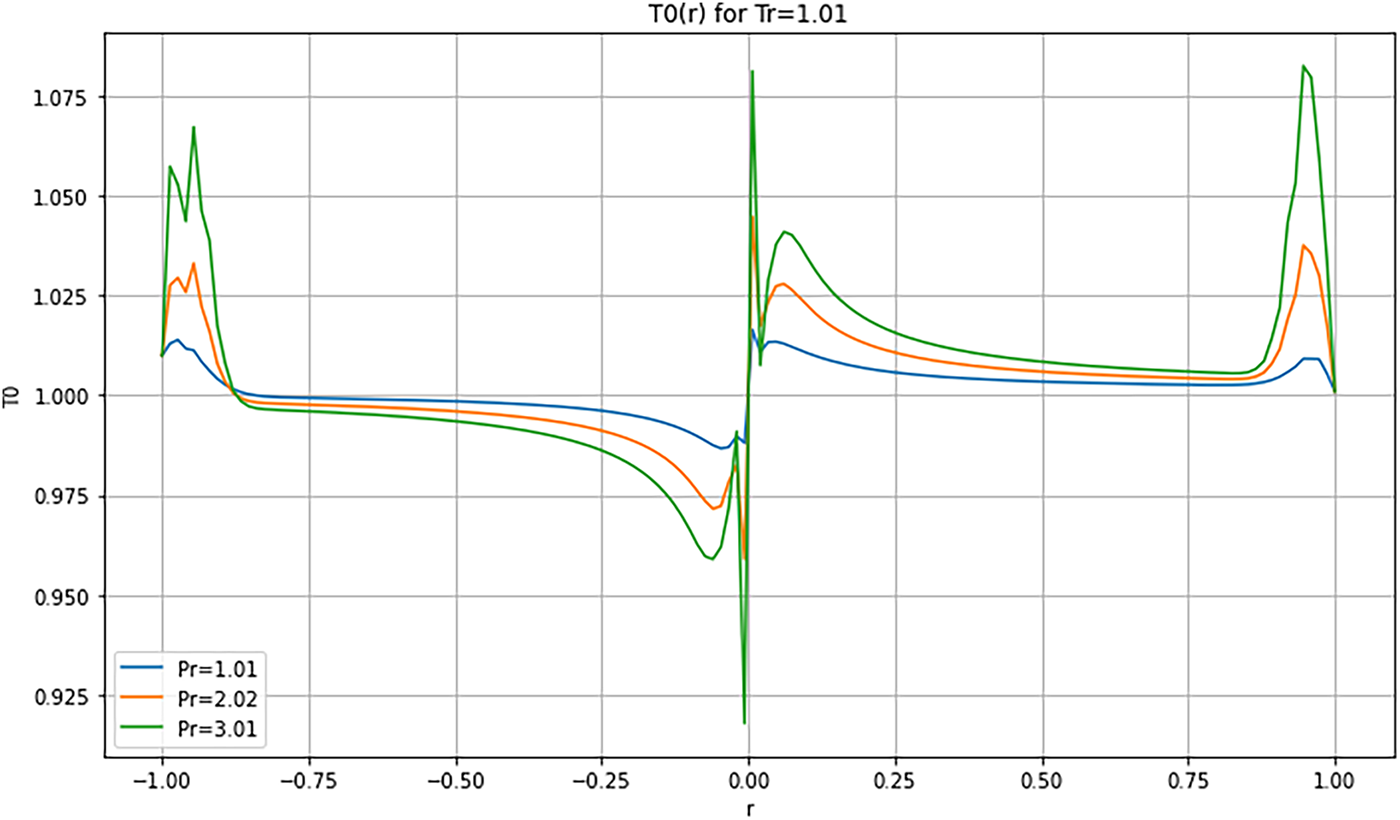
Figure 14: Radial Temperature distribution
4.3.1 Near-Critical Regime—Figs. 8 and 9 (
The zeroth-order temperature field
The first-order field
4.3.2 Transitional Regime—Figs. 10 and 11 (
The zeroth-order field
Simeoni et al. [21] and Majumdar et al. [20] confirmed that this intermediate regime is marked by moderate thermal gradients and relaxation dynamics without phase rupture, characteristic of pseudo-boiling phenomena in supercritical CO2.
4.3.3 Deep Supercritical Regime—Figs. 12 and 13 (
The zeroth-order field
The absence of a distinct interface between the droplet and the surrounding medium, limits the formation of shock fronts or transition zones or discontinuities, ensuring structural stability. Kang et al. [19] highlighted the efficiency and stability of deep supercritical regimes for heat exchange and solute dissolution, particularly in energy and space systems. Overall, this regime is characterized by low thermal gradients and homogeneous diffusion, confirming its suitability for high-performance applications under extreme conditions.
4.3.4 Radial Profiles—Figs. 14 and 15
At low reduced temperature (
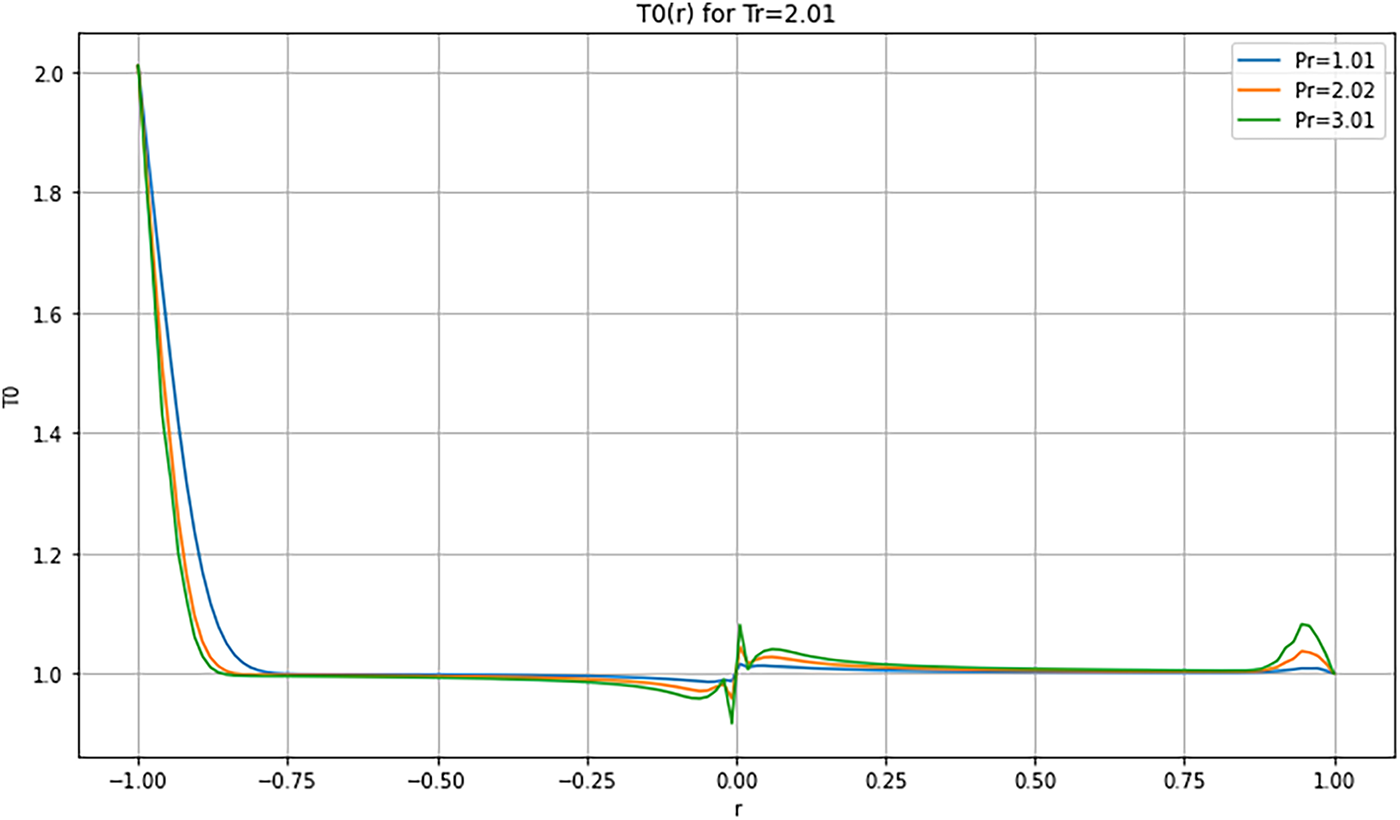
Figure 15: Radial Temperature distribution
These results confirm that the choice of reduced parameters strongly influences the internal thermal dynamics of the droplet. Multi-order analysis enables accurate resolution of diffusion, relaxation, and stabilization mechanisms. Beyond regime-specific observations, these temperature fields highlight general mechanisms that govern supercritical droplet dynamics. Thermodynamic instabilities are identified through non-monotonic profiles and relaxation fronts, consistent with previous studies (e.g., Simeoni et al. [21]). In contrast, numerical instabilities depend on discretization parameters such as time step and mesh resolution. Convergence and mesh-independence tests were performed, confirming that the oscillations and gradients reported here are robust and of physical origin. Temperature profiles reveal the same competition between thermal diffusion, compressibility, and mass confinement observed in the velocity field. In the quasi-critical regime, the droplet sustains a sharp thermal gradient with early relaxation fronts, reflecting diffusion that lags the initial compressive transient. Entering the transitional regime, the interfacial layer broadens, and temperature becomes locally non-monotonic, with overshoot or plateau features indicative of nonlinear T-u-ρ coupling and thermally driven radial fronts. At higher reduced pressure/temperature, stronger cohesion and more uniform thermal expansion damp local disturbances, yielding smoother temperature profiles and a less reactive interface. These trends support a coupled picture in which diffusion progressively dominates over compressibility, while internal recirculation can transiently recycle heat and sustain localized structures.
It should be noted, however, that the present model relies on idealized assumptions, such as neglecting viscous losses, variations in heat transfer efficiency, and material aging. While these simplifications may limit direct applicability to real systems, they provide a tractable framework to highlight the dominant physical mechanisms. Future work will incorporate non-ideal effects to improve practical relevance.
Through detailed numerical simulations, this work characterizes the relaxation behaviour of a CO2 droplet across three distinct thermodynamic regimes. In the quasi-critical case (
In the transitional regime (
The numerical framework based on second-order spatial discretization, Crank-Nicolson time integration, and mesh refinement near the interface proves robust and accurate. Validation against prior studies by Ly et al. [16] confirms the model’s ability to reproduce key features of supercritical and transcritical droplet evaporation.
Overall, this study advances the understanding of transition layer dynamics in supercritical fluids and contributes to the development of predictive models for jet formation, transient evaporation, and high-performance thermal systems using CO2 as a working fluid. The present results have direct industrial relevance. Regime-dependent thermal behaviors provide guidance for optimizing combustion and propulsion systems, where improved control of thermal instabilities can reduce fuel consumption and enhance energy efficiency. They also support the design of safe CO2 supercritical storage and transport facilities, and the improvement of supercritical extraction processes in pharmaceutical, food, and materials industries. These perspectives underline that the proposed modeling approach offers practical insights that extend beyond theoretical analysis. Nevertheless, future work should address feasibility aspects such as energy demand and containment materials, as well as technical challenges including viscous dissipation, variable transport properties, and material aging, to strengthen the predictive capability and industrial applicability of the system.
Acknowledgement: Not applicable.
Funding Statement: The authors received no specific funding for this study.
Author Contributions: Conceptualization: Yendoubouame Lare, Koffi Sagna; methodology: Yendoubouame Lare; software: Yendoubouame Lare; validation: Koffi Sagna, Amah Séna d’Almeida; formal analysis: Yendoubouame Lare, Koffi Sagna, Amah Séna d’Almeida; investigation: Yendoubouame Lare; resources: Koffi Sagna, Amah Séna d’Almeida; data curation, Yendoubouame Lare, Koffi Sagna, Amah Séna d’Almeida; writing original draft preparation, Yendoubouame Lare; writing review and editing: Koffi Sagna, Amah Séna d’Almeida; visualization, supervision: Koffi Sagna, Amah Séna d’Almeida; project administration: Koffi Sagna, Amah Séna d’Almeida; funding acquisition: Yendoubouame Lare, Koffi Sagna, Amah Séna d’Almeida. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials: Not applicable.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest to report regarding the present study.
Statement of Novelty: This work introduces a novel multi-order modeling framework to analyze the spatiotemporal evolution of velocity, density, and temperature fields within a carbon dioxide droplet exposed to supercritical conditions. Unlike previous studies that rely on simplified thermodynamic assumptions or macroscopic droplet behavior, this approach incorporates first- and second-order perturbation dynamics to resolve the internal structure of the transition layer. The explicit separation of zeroth- and first-order contributions to heat flux and density evolution reveals non-trivial couplings between initial perturbations and the base state. These results offer new insights into droplet stability and phase transition mechanisms, with direct implications for modeling supercritical fluid behavior and optimizing propulsion systems. Controlling evaporation-induced discontinuities may significantly enhance fuel efficiency and combustion stability in high-performance applications.
References
1. Chauveau C, Birouk M, Gökalp I. An analysis of the d2-law departure during droplet evaporation in microgravity. Int J Multiph Flow. 2011;37(3):252–9. doi:10.1016/j.ijmultiphaseflow.2010.10.009. [Google Scholar] [CrossRef]
2. He P, Li Y, Zhao L. Evaporation of liquid fuel droplet at supercritical conditions. Sci China Technol Sci. 2011;54(2):369–74. doi:10.1007/s11431-010-4231-x. [Google Scholar] [CrossRef]
3. Ju D, Huang L, Zhang K, Ye M, Huang Z, Yi G. Comparison of evaporation rate constants of a single fuel droplet entering subcritical and supercritical environments. J Mol Liq. 2022;347(5):118346. doi:10.1016/j.molliq.2021.118346. [Google Scholar] [CrossRef]
4. Spalding DB. Combustion of fuel particles. Fuel. 1951;30(1):121–30. [Google Scholar]
5. Godsave GAE. Studies of the combustion of drops in a fuel spray—the burning of single drops of fuel. Symp Int Combust. 1953;4(1):818–30. doi:10.1016/S0082-0784(53)80107-4. [Google Scholar] [CrossRef]
6. Spalding DB. Theory of particle combustion at high pressures. ARS J. 1959;29(11):828–35. doi:10.2514/8.4918. [Google Scholar] [CrossRef]
7. Williams FA. On the assumptions underlying droplet vaporization and combustion theories. J Chem Phys. 1960;33(1):133–44. doi:10.1063/1.1731068. [Google Scholar] [CrossRef]
8. Wieber PR. Calculated temperature histories of vaporizing droplets to the critical point. AIAA J. 1963;1(12):2764–70. [Google Scholar]
9. Sagna K. A study of droplet evaporation. Am J Mod Phys. 2013;2(2):71. doi:10.11648/j.ajmp.20130202.17. [Google Scholar] [CrossRef]
10. Lare Y, Sagna K, Apeke KS, Afodanyi DK, Lare Y, d’Almeida AS. Study of the thermodynamic properties of a drop in an environment under subcritical conditions during evaporation. Int J Phys. 2023;11(3):97–105. [Google Scholar]
11. Sagna K, Amou KA, Boroze TT, Kassegne D, d’Almeida A, Napo K. Environmental pollution due to the operation of gasoline engines: exhaust gas law. Int J Oil Gas Coal Eng. 2017;5(4):39–43. doi:10.11648/j.ogce.20170504.11. [Google Scholar] [CrossRef]
12. Sánchez Tarifa C, Crespo Martínez A, Fraga E. A theoretical model for the combustion of droplets in supercritical conditions and gas pockets. In: International Colloquium on Gasdynamics of Explosions; 1971 Sep 12–17; Marseille, France. [Google Scholar]
13. Sagna K, d’Almeida A. Supercritical evaporation of a drop. Int J Res Eng Sci. 2014;2(7):41–7. [Google Scholar]
14. Prud’Homme R, d’Almeida AS. Evaporation de gouttes: lois de récession du rayon. In: 19ème Congrès Français de Mécanique; 2009 Aug 24–28; Marseille, France. [Google Scholar]
15. Préau S, Prud’homme R, Ouazzani J, Zappoli B. Supercritical density relaxation as a new approach of droplet vaporization. Phys Fluids. 2004;16(11):4075–87. doi:10.1063/1.1790231. [Google Scholar] [CrossRef]
16. Ly N, Majumdar A, Ihme M. Regimes of evaporation and mixing behaviors of nanodroplets at transcritical conditions. Fuel. 2023;331:125870. doi:10.1016/j.fuel.2022.125870. [Google Scholar] [CrossRef]
17. Cheng L, Xia G. Progress and prospects for research and technology development of supercritical CO2 thermal conversion systems for power, energy storage, and waste heat recovery. Heat Transf Eng. 2024;45(20–21):1836–53. doi:10.1080/01457632.2023.2282765. [Google Scholar] [CrossRef]
18. Min I, Kang SG, Huh C. Instability analysis of supercritical CO2 during transportation and injection in carbon capture and storage systems. Energies. 2018;11(8):2040. doi:10.3390/en11082040. [Google Scholar] [CrossRef]
19. Kang X, Mao L, Shi J, Liu Y, Zhai B, Xu J, et al. Supercritical carbon dioxide systems for sustainable and efficient dissolution of solutes: a review. Environ Chem Lett. 2024;22(2):815–39. doi:10.1007/s10311-023-01681-4. [Google Scholar] [CrossRef]
20. Majumdar A, Li H, Muhunthan P, Späh A, Song S, Sun Y, et al. Direct observation of ultrafast cluster dynamics in supercritical carbon dioxide using X-ray photon correlation spectroscopy. Nat Commun. 2024;15:10540. doi:10.1038/s41467-024-54782-1. [Google Scholar] [PubMed] [CrossRef]
21. Simeoni GG, Bryk T, Gorelli FA, Krisch M, Ruocco G, Santoro M, et al. The Widom line as the crossover between liquid-like and gas-like behaviour in supercritical fluids. Nature Phys. 2010;6(7):503–7. doi:10.1038/nphys1683. [Google Scholar] [CrossRef]
22. Lopes NC, Chao Y, Dasarla V, Sullivan NP, Ricklick MA, Boetcher SKS. Comprehensive review of heat transfer correlations of supercritical CO2 in straight tubes near the critical point: a historical perspective. J Heat Transf. 2022;144(12):120801. doi:10.1115/1.4055345. [Google Scholar] [CrossRef]
23. Awais M, Ullah N, Ahmad J, Sikandar F, Ehsan MM, Salehin S, et al. Heat transfer and pressure drop performance of nanofluid: a state-of-the-art review. Int J Thermofluids. 2021;9(1):100065. doi:10.1016/j.ijft.2021.100065. [Google Scholar] [CrossRef]
24. Sarrade S, Benaissi K. Le CO2 supercritique et ses applications industrielles. l’Actualité Chim. 2013;371-372:72–7. [Google Scholar]
25. Arias-Zugasti M, García-Ybarra PL, Castillo JL. Unsteady effects in droplet vaporization lifetimes at subcritical and supercritical conditions. Combust Sci Technol. 2000;153(1):179–91. doi:10.1080/00102200008947259. [Google Scholar] [CrossRef]
26. Accary G, Raspo I, Bontoux P, Zappoli B. An adaptation of the low Mach number approximation for supercritical fluid buoyant flows. Comptes Rendus Mécanique. 2005;333(5):397–404. doi:10.1016/j.crme.2005.03.004. [Google Scholar] [CrossRef]
27. Versteeg HK, Malalasekera W. An introduction to computational fluid dynamics: the finite volume method. 2nd ed. London, UK: Pearson Education; 2007. [Google Scholar]
28. Patankar S. Numerical heat transfer and fluid flow. Abingdon, UK: Hemisphere Publishing Corporation; 1980. [Google Scholar]
29. Zhang H. Evaporation of a suspended droplet in forced convective high-pressure environments. Combust Sci Technol. 2003;175(12):2237–68. doi:10.1080/714923282. [Google Scholar] [CrossRef]
30. Sagna K. Étude de l’évaporation d’une goutte liquide isolée à pressions subcritique et supercritique. Lomé, Togo: Université de Lomé; 2014. [Google Scholar]
31. Préau S, Prud’Homme R, Ouazzani J, Zappoli B. Pertinence de la comparaison à la loi en d2 en «évaporation» supercritique. Comptes Rendus Mécanique. 2004;332(5–6):397–402. doi:10.1016/j.crme.2004.02.009. [Google Scholar] [CrossRef]
32. Chai L, Tassou SA. Recent progress on high temperature and high pressure heat exchangers for supercritical CO2 power generation and conversion systems. Heat Transf Eng. 2023;44(21–22):1950–68. doi:10.1080/01457632.2022.2164683. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools