 Open Access
Open Access
ARTICLE
Cholecystokinin A Receptor Knockdown Diminishes Colon Cancer Cell Invasive Potential via Modulation of Integrin/FAK, EMT, and uPA/uPAR/MMP2 Axis
1 Institute of Medicine, College of Medicine, Chung Shan Medical University, Taichung, Taiwan
2 Precision Medicine Center, Chung Shan Medical University Hospital, Taichung, Taiwan
3 Division of Colorectal Surgery, Buddhist Tzu Chi Medical Foundation, Dalin Tzu Chi Hospital, Chiayi, Taiwan
4 School of Medicine, Tzu Chi University, Hualien, Taiwan
5 School of Medicine, Chung Shan Medical University, Taichung, Taiwan
6 Department of Surgery, Chung Shan Medical University Hospital, Taichung, Taiwan
7 Department of Medical Research, Chung Shan Medical University Hospital, Taichung, Taiwan
* Corresponding Authors: Hsiang-Lin Lee. Email: ; Shao-Hsuan Kao. Email:
# These authors contributed equally to this work as the first author
(This article belongs to the Special Issue: Signaling Pathway Crosstalk in Malignant Tumors: Molecular Targets and Combinatorial Therapeutics)
Oncology Research 2026, 34(4), 22 https://doi.org/10.32604/or.2026.074231
Received 06 October 2025; Accepted 05 February 2026; Issue published 23 March 2026
Abstract
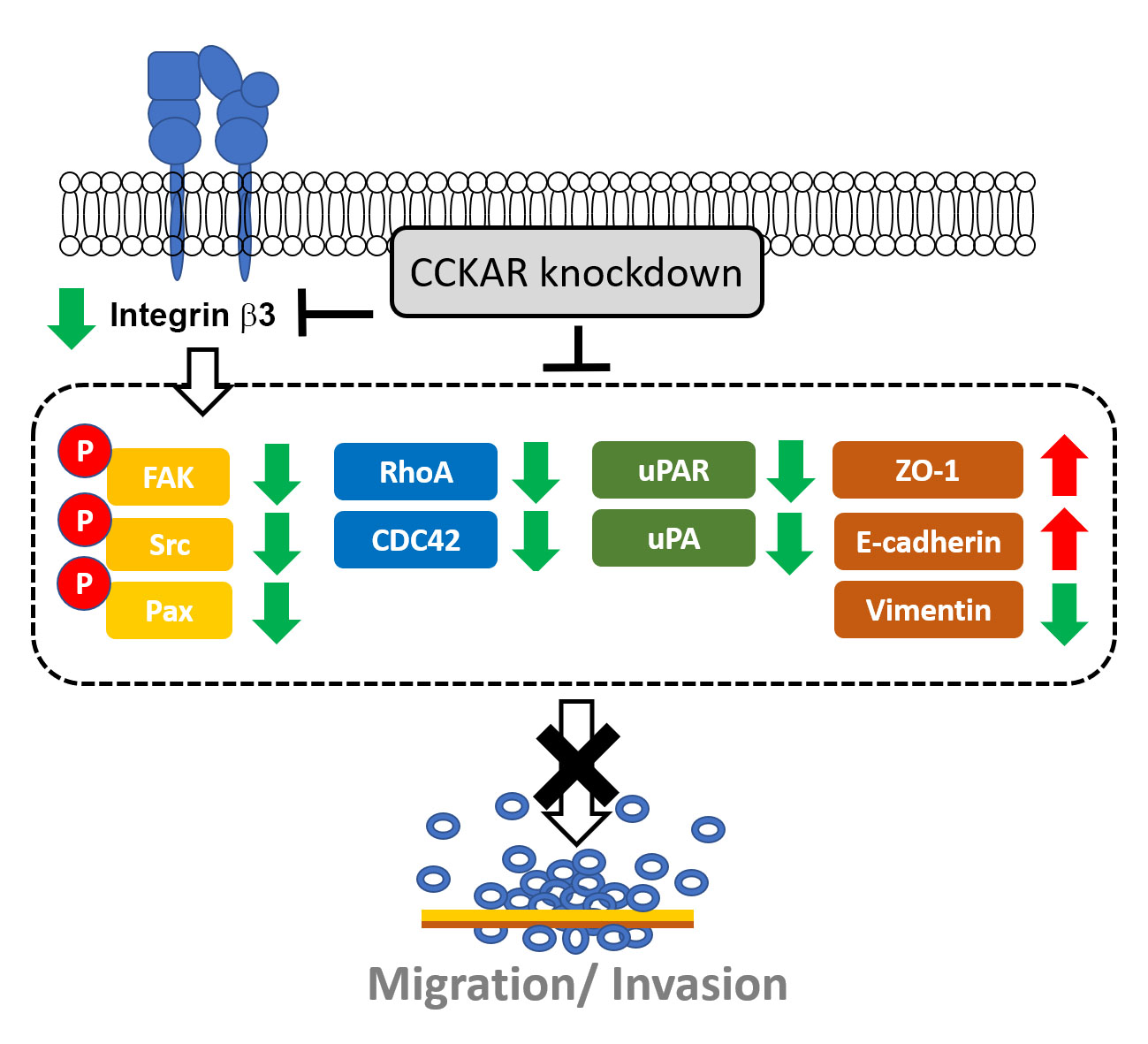
Objectives: Cholecystokinin A receptor (CCKAR) has been linked to poor prognosis in colon cancer patients, but the role of CCKAR in colon cancer cell invasiveness and the underlying mechanisms remain elusive. This study aimed to explore the effect of CCKAR on the invasive potential of colon cancer cells. Methods: Different human colon cancer cell lines were used. Gene expression was evaluated by reverse transcription polymerase chain reaction (RT-PCR) and quantitative real-time RT-PCR (qPCR), while protein expression and phosphorylation were assessed by Western blotting. Cell motility and invasiveness were examined through wound healing and invasion assays, respectively. Results: Our results showed that CCKAR expression levels varied across colon cancer cell lines, with DLD-1 and LoVo cells showing high expression. Knockdown of CCKAR significantly impaired the cell motility and invasiveness of DLD-1 and LoVo cells, downregulated integrin β3 expression, and diminished the phosphorylation levels of focal adhesion kinase (FAK), Src, and paxillin. In addition, CCKAR knockdown modulated epithelial-mesenchymal transition (EMT) markers ZO-1, E-cadherin, and vimentin and reduced urokinase-type plasminogen activator (uPA), uPA receptor (uPAR), Rho GTPase cell division control protein 42 (CDC42) and RhoA, and matrix metalloproteinase-2 (MMP-2). Conclusions: These findings indicate that CCKAR knockdown impairs the invasiveness of colon cancer cells, which may be attributed to modulating integrin/FAK/Rho GTPases, EMT markers, and the uPA/uPAR axis. It suggests that targeting CCKAR may represent a potential therapeutic strategy for colon cancer treatment.Graphic Abstract
Keywords
Supplementary Material
Supplementary Material FileCite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools