Oncology Research is committed to publishing high-quality, innovative research that is focused on the entire range of basic, translational, and clinical cancer research, with a particular interest in cancer therapeutics, providing a new platform for the understanding, prevention, diagnosis, and treatment of cancer.
Science Citation Index Expanded (Clarivate Analytics): 2024 Impact Factor: 4.1; Scopus CiteScore (Impact per Publication 2025): 4.1; SNIP (Source Normalized Impact per Paper 2025): 0.767; Embase; PubMed Central; MEDLINE; EBSCO; Google Scholar; Proquest; Portico, etc.
 Open Access
Open Access
REVIEW
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.076088 - 21 May 2026
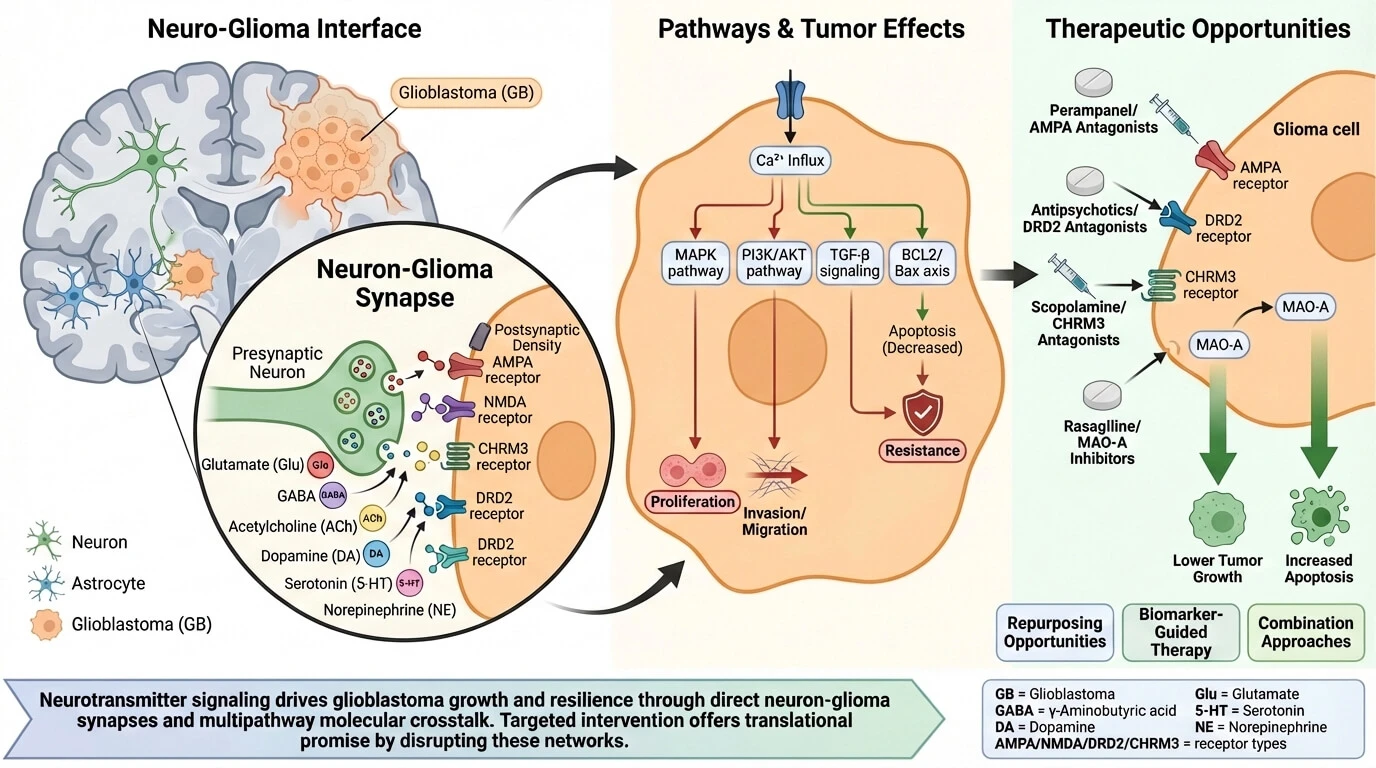
Abstract Glioblastoma (GB) is the most common primary malignant brain tumor of adulthood, and despite optimal safe resection and chemoradiation, it is still lethal. Neuroscience of cancer has shown that neuronal activities, as well as neurotransmitters, play an active role in the glioma microenvironment. This article aims to integrate the existing literature on the role of neurotransmitters and their receptors in glioblastoma, as well as other gliomas, highlighting areas of therapeutic intervention in the neuron-tumor interface. We will describe the neuro–glioma interface, including functional neuron–glioma synapses and activity-dependent tumor growth. We will also discuss major neurotransmitter… More >
Graphic Abstract
 Open Access
Open Access
REVIEW
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.076509 - 21 May 2026
(This article belongs to the Special Issue: Rewiring Metabolism for Cancer Treatment: Emerging Approaches)
Abstract Checkpoint kinase 1 (CHK1), a key regulator of cell cycle checkpoints, plays a central role in the DNA damage response network, serving as a critical mediator that links DNA damage detection to DNA repair mechanisms. In recent years, several other cellular functions of CHK1 have gradually been discovered. As well as monitoring genomic integrity, CHK1 coordinates the timing of DNA replication with the availability of metabolic resources. This prevents unscheduled DNA synthesis from exceeding the cell’s metabolic capacity and causing DNA damage. CHK1 activity also contributes to tumour immune surveillance and the modulation of immune… More >
 Open Access
Open Access
REVIEW
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.078650 - 21 May 2026
Abstract Over the past decade, the therapeutic paradigm of cutaneous melanoma has been transformed strongly, driven by advances in immuno-oncology and precision medicine. Building on the success of immune checkpoint blockade and targeted therapy, new treatment strategies now aim to improve efficacy, overcome resistance, and prolong the durability of responses. Clinical trials on neoadjuvant therapy supporting its clinical use are presented. Furthermore, the latest progress in combinatorial immune checkpoint inhibition such as dual anti-LAG-3 or anti-TIGIT with anti-PD-1 blockade, next-generation bispecific antibody development, mRNA-based vaccines in clinical practice, and intralesional therapies are summarized. Additionally, it outlines More >
 Open Access
Open Access
REVIEW
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.074215 - 21 May 2026
(This article belongs to the Special Issue: Advances in Targeted and Precision Medicine in Breast Oncology)
Abstract Objectives: Triple-negative breast cancer (TNBC) accounts for approximately 15% of all invasive breast cancers and is characterized by aggressive behavior, limited therapeutic options, and poor clinical outcomes. Due to the absence of hormone receptors and HER2 expression, systemic treatment relies predominantly on chemotherapy, which is associated with high rates of early recurrence and mortality. Emerging evidence suggests that alterations in the microbiome can contribute to TNBC progression and influence therapeutic response, particularly affecting the efficacy of chemotherapy and immunotherapy through immune-mediated mechanisms; however, its role in TNBC remains incompletely understood. This systematic review aims to explore… More >
 Open Access
Open Access
REVIEW
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.075346 - 21 May 2026
(This article belongs to the Special Issue: Advances in Targeted and Precision Medicine in Breast Oncology)
Abstract Long non-coding RNAs (lncRNAs) have emerged as key regulators of drug resistance in human epidermal growth factor receptor 2 (HER2)-positive breast cancer, a subtype in which both intrinsic and acquired resistance to HER2-targeted therapies remain major clinical challenges. Although mechanistic studies have begun to reveal how lncRNAs modulate signaling pathways, interact with microRNAs, and influence the tumor microenvironment, dedicated investigations in HER2-positive disease are still limited. This review synthesizes current evidence across epigenetic, transcriptional, and post-transcriptional mechanisms of resistance, including competing endogenous RNA (ceRNA) networks, RNA-binding protein interactions, and exosome-mediated intercellular communication. Particular emphasis is… More >
 Open Access
Open Access
REVIEW
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.076937 - 21 May 2026
(This article belongs to the Special Issue: Advancements in Hepatocellular Carcinoma Treatment)
Abstract Hepatocellular carcinoma (HCC) represents a critical global health challenge, standing as a leading cause of cancer mortality with a significant and projected increasing incidence worldwide. A primary hurdle in HCC management is late diagnosis, often attributable to the absence of early symptoms. Despite considerable advancements in therapeutic strategies over the past decade, including immune checkpoint inhibitors and targeted therapies, mortality rates remain high, underscoring the urgent need for more effective novel approaches. The inherent molecular complexity and heterogeneity of HCC, where only a minority of tumors possess readily targetable drivers, contribute to treatment resistance and More >
 Open Access
Open Access
REVIEW
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.073660 - 21 May 2026
(This article belongs to the Special Issue: Advances in Cancer Therapeutics)
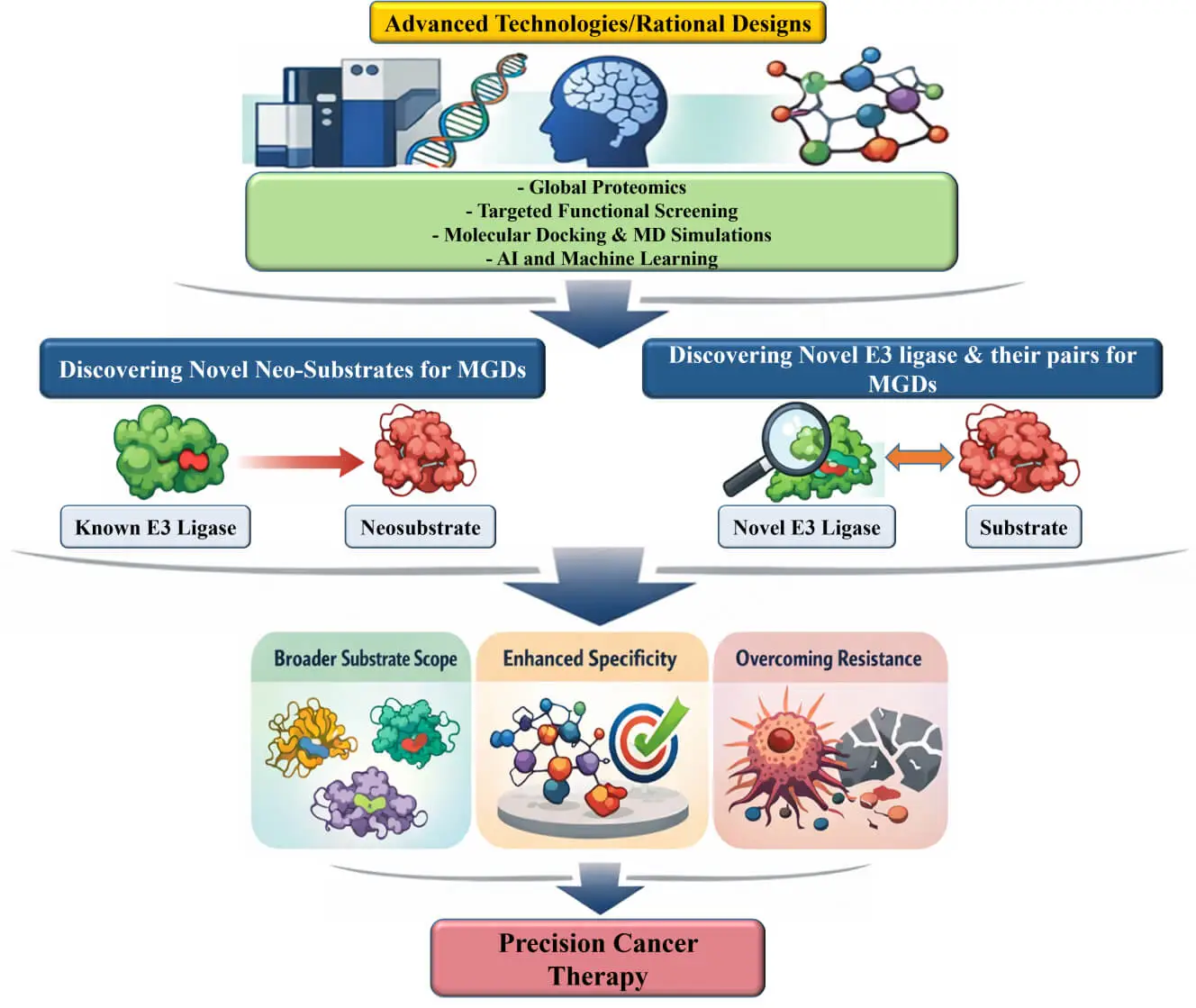
Abstract Molecular glue degraders (MGDs) are an emerging class of small molecules that promote selective protein degradation by inducing neomorphic interactions between E3 ubiquitin ligases and non-native substrates, referred to as neosubstrates. Clinically validated examples include thalidomide analogs that recruit cereblon (CRBN) to degrade IKAROS family zinc finger 1/3 in multiple myeloma, and arylsulfonamide-based MGDs that promote the degradation of RNA-binding protein 39 in acute myeloid leukemia and solid tumors. These molecules highlight the therapeutic potential of this modality in oncology. These findings underscore the promise of MGDs for eliminating oncogenic proteins previously considered undruggable and… More >
Graphic Abstract
 Open Access
Open Access
REVIEW
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.077445 - 21 May 2026
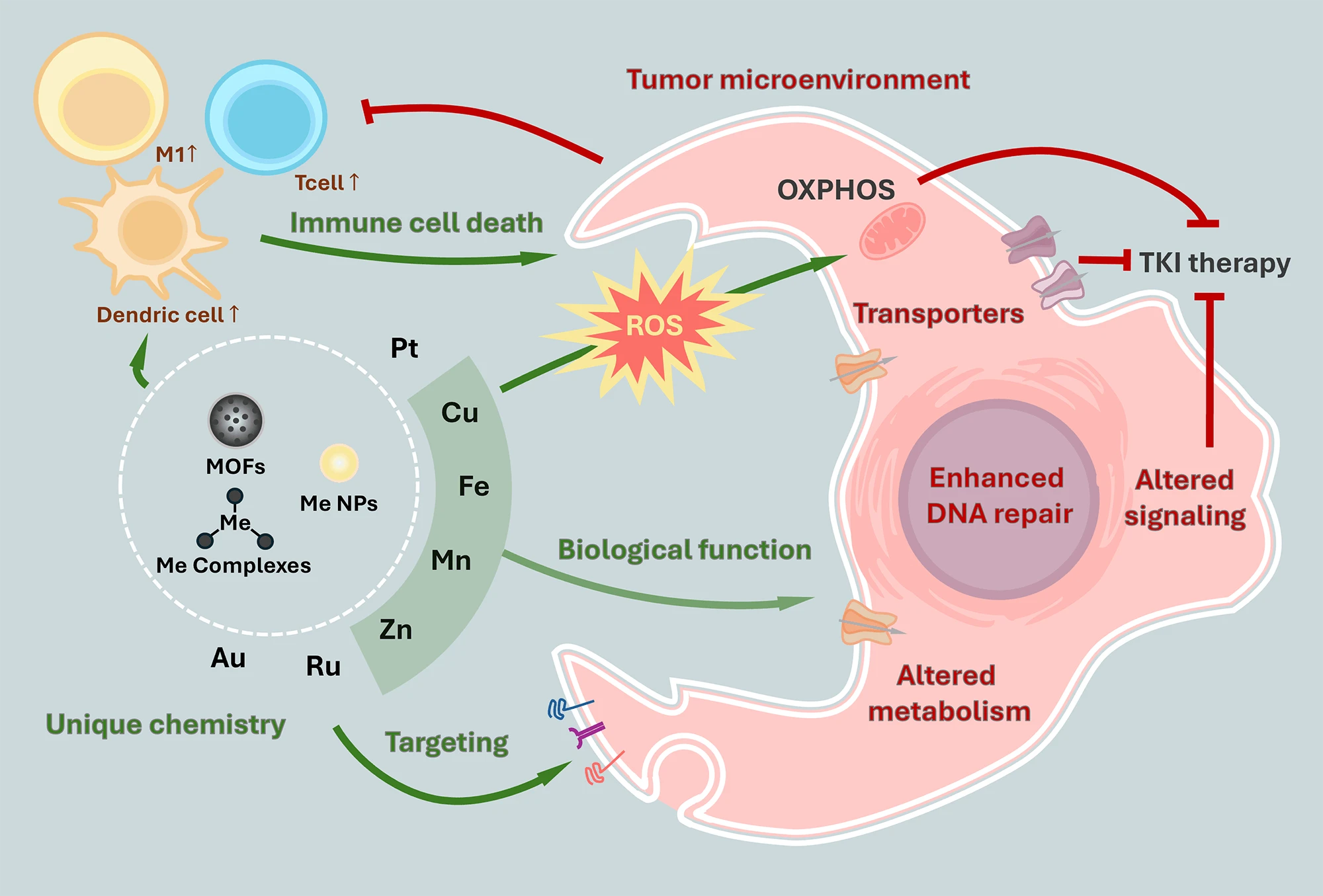
Abstract The formation of drug resistance poses the ultimate threat in modern oncology. Targeted therapy lacks versatility, while conventional therapy is famous for its side effects. However, for the new therapeutics to address the challenge of drug resistance, such compounds should combine properties of both modalities. In this review, we argue that metal-based therapeutics are paramount substances for achieving this goal. The unique physico-chemical properties and metabolism of these compounds, as well as metals themselves, allow to realize unique activities in normal and cancer cells, including precise targeting, non-apoptotic cell death, and disruption of critical signaling More >
Graphic Abstract
 Open Access
Open Access
REVIEW
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.077913 - 21 May 2026
(This article belongs to the Special Issue: Advances in Skin Cancer Management: From Molecular Targets to Innovative Treatments)
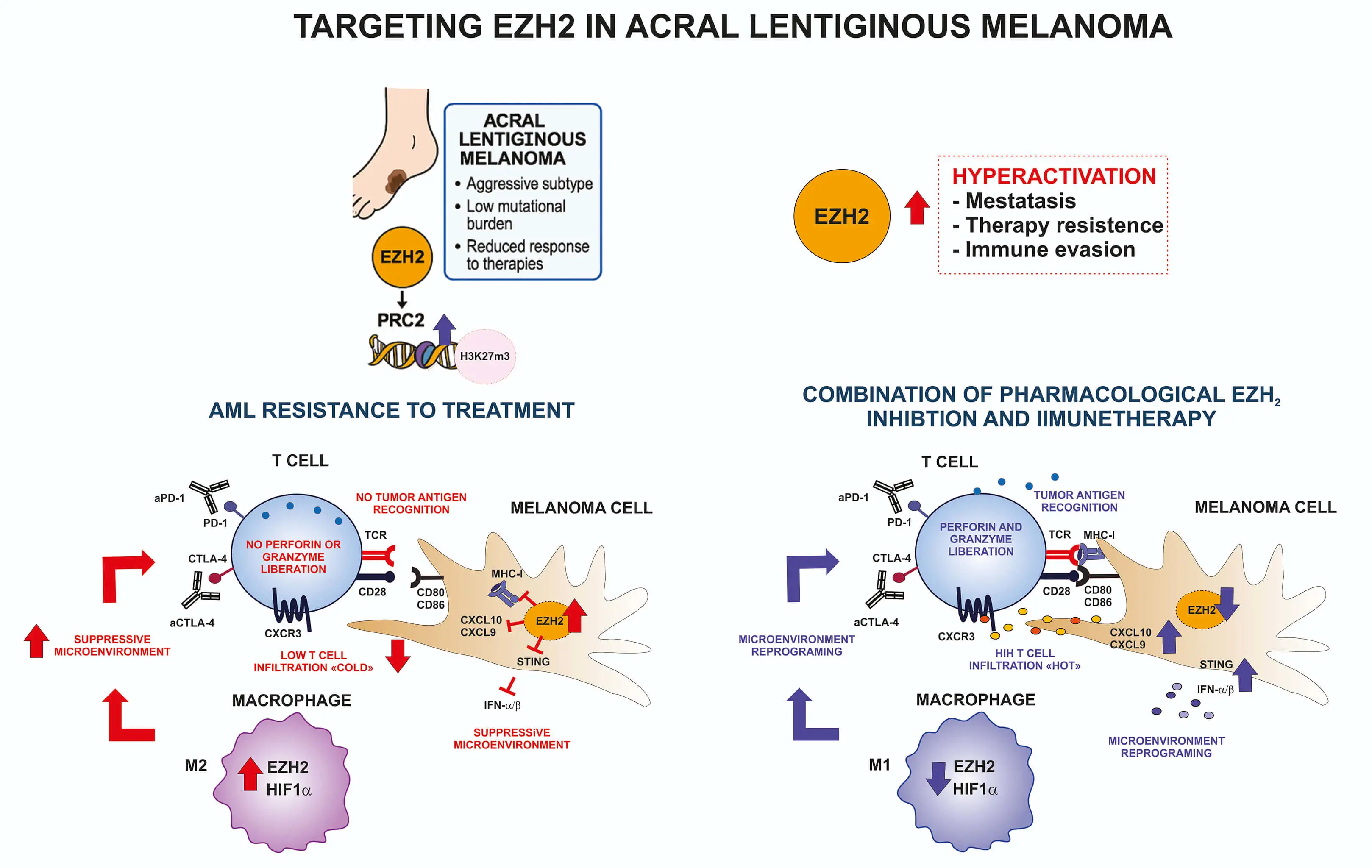
Abstract Acral lentiginous melanoma (ALM) is characterized by a low mutational burden, frequent chromosomal rearrangements, and profound epigenetic dysregulation, distinguishing it from ultraviolet (UV)-induced melanoma. Among the epigenetic regulators, Enhancer of Zeste Homolog 2 (EZH2), the catalytic component of the Polycomb Repressive Complex 2 (PRC2), plays a central role in chromatin compaction and transcriptional repression through trimethylation of histone H3 on lysine 27 (H3K27me3). EZH2 overexpression or hyperactivation contributes to tumor progression, immune evasion, and therapeutic resistance. Recent multi-omic studies have highlighted the importance of EZH2 in regulating melanoma plasticity, immune modulation, and metabolic reprogramming. In… More >
Graphic Abstract
 Open Access
Open Access
REVIEW
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.076499 - 21 May 2026
(This article belongs to the Special Issue: New Insights in Drug Resistance of Cancer Therapy: A New Wine in an Old Bottle)
Abstract Liquid-liquid phase separation (LLPS) is an emerging biophysical principle that governs subcellular organization through the formation of dynamic, membraneless biomolecular condensates. This review aims to elucidate the multifaceted mechanisms by which dysregulated LLPS drives cancer drug resistance and to explore therapeutic strategies targeting oncogenic biomolecular condensates for improved anticancer outcomes. We synthesize evidence demonstrating that dysregulated LLPS drives cancer drug resistance through diverse mechanisms, including sustaining oncogenic transcription despite targeted therapies, creating physical barriers against chemotherapeutics, modulating immune checkpoint activity, enhancing DNA damage repair, promoting cancer stemness and radioresistance. By integrating insights from cell cycle More >
 Open Access
Open Access
REVIEW
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.076300 - 21 May 2026
(This article belongs to the Special Issue: Novel Biomarkers and Treatment Strategies in Solid Tumor Diagnosis, Progression, and Prognosis (Ⅱ))
Abstract Hormone Receptor-positive/Human Epidermal Growth Factor Receptor 2-negative (HR+/HER2−) breast cancer treatment has made a breakthrough due to the introduction of cyclin-dependent kinases 4 and 6 (CDK4/6) inhibitors. This article mainly reviews the mechanisms of action, clinical efficacy, and current application status of CDK4/6 inhibitors, including Palbociclib, Ribociclib, Abemaciclib, and the emerging Dalpiciclib. The advantages and limitations of different treatment stages are also discussed. CDK4/6 inhibitors have excellent efficacy in prolonging progression-free survival (PFS) and overall survival (OS), and have become a key option for HR+/HER2− breast cancer first-line and adjuvant treatment. The issues of drug More >
 Open Access
Open Access
REVIEW
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.078219 - 21 May 2026
(This article belongs to the Special Issue: Novel Drug Targets and Combination Strategies in Gynecologic Cancers)
Abstract Cervical cancer remains a major global health challenge despite advances in human papillomavirus (HPV) vaccination, screening, and treatment. Persistent infection with high-risk HPV types, particularly HPV16 and HPV18, is a necessary cause of cervical cancer; however, only a small fraction of infections progress to malignancy, indicating the importance of additional cofactors. Increasing evidence identifies estrogen signaling as a critical modifier of HPV-driven carcinogenesis. Estrogen acts synergistically with HPV oncogenes E6 and E7 to promote genomic instability, immune evasion, and tumor progression, largely through effects on the tumor microenvironment (TME). This review aims to clarify and… More >
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.075600 - 21 May 2026
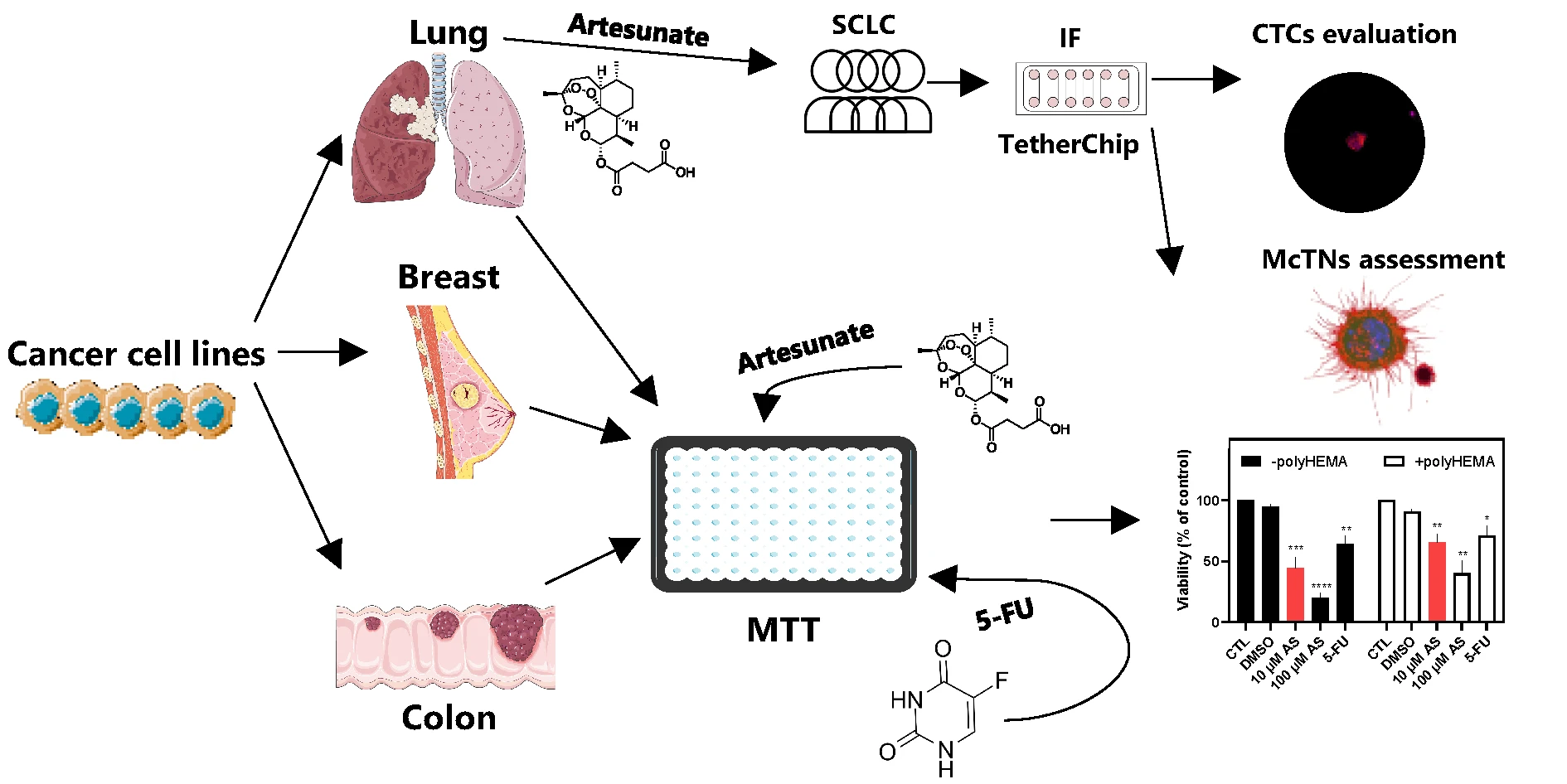
Abstract Objectives: Circulating tumor cells (CTCs) drive metastasis and exhibit resistance to conventional therapies, making them crucial therapeutic targets. Artesunate (AS), a derivative of artemisinin, displays anticancer activity, including inhibition of JunB proto-oncogene (JUNB) and programmed death ligand-1 (PD-L1) and upregulation of Vimentin (VIM), markers related to poor prognosis in CTCs. This study aimed to evaluate the effects of AS on adherent and non-adherent cancer cell lines (breast, lung, colon), the patient-derived colon cancer CTC-MCC-41 line, and CTCs from small-cell lung cancer (SCLC) patients. Methods: AS’s effect was evaluated using TetherChip technology. Cell viability was measured using… More >
Graphic Abstract
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.075609 - 21 May 2026
(This article belongs to the Special Issue: Gastroenteropancreatic Tumors: From Basic Research to Therapeutic Approach)
Abstract Background: Claudin 18.2 (CLDN18.2) has become a clinically relevant therapeutic target in gastric adenocarcinoma (GC), with zolbetuximab now approved for use in CLDN18.2-positive, HER2-negative advanced disease. The aim of this study was to evaluate the prevalence, intratumoral reproducibility, and clinicopathologic associations of CLDN18.2 expression in a Western cohort of resected GC. Methods: CLDN18.2 expression was evaluated by immunohistochemistry in 204 resected GCs arranged in tissue microarrays containing duplicate tumor cores corresponding to the tumor center and invasive front. Correlations between paired cores, clinicopathologic parameters, additional biomarkers (E-cadherin, HER2, p53, mismatch repair (MMR)), and clinical outcomes were… More >
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.078309 - 21 May 2026
(This article belongs to the Special Issue: Molecular Targeting Therapy for Anticancer Treatment)
Abstract Objectives: Genetic risk models have substantially advanced our understanding of germline pathogenic variants (GPVs) in some malignancies, whereas their clinical significance in lung cancer remains unclear. The present study aimed to better understand potential contribution of GPVs to lung cancer etiology. Methods: A targeted sequencing panel of 143 cancer-related genes was applied to analyze 26 distinct lung adenocarcinoma (LUAD) tumors from 11 patients histopathologically diagnosed with multiple primary lung cancers (MPLC). Tumor classification was performed through integrated evaluation of mutation profiles, and variants shared among tumor lesions were further validated as likely germline or somatic mutations… More >
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.072421 - 21 May 2026
(This article belongs to the Special Issue: Emerging Insights into the Tumor Microenvironment: Mechanisms, Therapeutic Targets, and Clinical Applications)
Abstract Objectives: Deregulated plasticity is involved in initiation, progression, metastasis, and resistance to therapy of various cancers. Our study aimed to present new checkpoints involved in complex biological processes that sustain epithelial-mesenchymal transition (EMT) variability and heterogeneity in prostate tumor cell plasticity. Methods: Dysregulated cell signaling pathways involved in prostate EMT heterogeneity were analyzed by intrinsic and extrinsic factors such as cell cycle phases by propidium iodide (PI) stain, apoptosis by caspase-3/7 biochemical cascade DEVDase enzyme activity by Magic Red stain (DEVD-MR)/propidium iodide stain, autophagy and nuclear shrinkage by Hoechst/acridine orange stain, evasion of immune surveillance by… More >
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.076944 - 21 May 2026
Abstract Objectives: Pancreatic ductal adenocarcinoma (PDAC) is a leading cause of cancer-related mortality and mainly affects older adults, who frequently experience polypharmacy. While systemic therapy may improve outcomes in selected older patients, the use of multiple drugs increases the risk of potential drug–drug interactions (pDDIs). This study aimed to evaluate the prevalence and characteristics of pDDIs in older patients with PDAC receiving first-line systemic therapy and their potential impact on clinical outcomes. Methods: We conducted a retrospective single-center study including patients aged ≥ 75 years with PDAC who initiated first-line systemic therapy between December 2011 and January… More >
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.076384 - 21 May 2026
Abstract Background: Germline breast cancer susceptibility gene 1/2 (BRCA1/2) variants guide breast cancer treatment, but their clinical relevance in metastatic triple-negative breast cancer (mTNBC) treated with sacituzumab govitecan (SG) remains unclear. The study aimed to evaluate the association between BRCA status and outcomes in SG-treated mTNBC. Methods: We retrospectively analyzed 264 patients with mTNBC and known germline BRCA1/2 (gBRCA1/2) status who received SG between August 2021 and May 2025 across multiple oncology centers in Poland, the Czech Republic and Slovakia. Survival outcomes were compared between patients with gBRCA1/2 mutations (gBRCA1/2m) and those with gBRCA1/2 wild-type (gBRCA1/2wt) using Kaplan–Meier estimates, the log-rank… More >
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.070445 - 21 May 2026
(This article belongs to the Special Issue: Tumor Biomarkers for Diagnosis, Prognosis and Targeted Therapy)
Abstract Background: Glycosylation and inflammation are pivotal in tumor progression, yet the specific glycosyltransferases bridging these processes remain poorly defined. This study investigated Exostosin-1 (EXT1), a key enzyme in heparan sulfate (HS) biosynthesis, as a mechanistic bridge connecting inflammation, stromal remodeling, and immune evasion-driven cancers. Methods: We used a multi-omics approach including Least Absolute Shrinkage and Selection Operator (LASSO) Cox regression on The Cancer Genome Atlas (TCGA) pan-cancer cohorts, transcriptomics, survival, single-cell RNA sequencing (scRNA-seq), DNA methylation profiling, pathway enrichment analysis (MetaCore), molecular docking, and immunohistochemistry (IHC) on pancreatic adenocarcinoma (PAAD) and lung adenocarcinoma (LUAD) tissue microarrays. Results:… More >
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.079120 - 21 May 2026
(This article belongs to the Special Issue: Advances in Pediatric and Adolescent Oncology: From Bench to Bedside)
Abstract Objectives: Germline variants in cancer predisposition genes have been increasingly recognized in pediatric cancers. However, their spectrum in East Asian children with central nervous system (CNS) tumors remains insufficiently defined. This study investigated the prevalence and clinical significance of pathogenic or likely pathogenic (P/LP) germline mutations in Korean children, adolescents, and young adults (AYAs) with CNS tumors. Methods: We performed targeted next-generation sequencing of 358 cancer-associated genes using peripheral blood DNA from 108 patients. Germline variants were classified according to ACMG/AMP guidelines and curated using ClinVar and relevant literature. Results: Among 108 patients, 17 (15.7%) carried P/LP… More >
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.077076 - 21 May 2026
(This article belongs to the Special Issue: Tumor Biomarkers for Diagnosis, Prognosis and Targeted Therapy)
Abstract Objective: Glioblastoma (GBM) is the most common primary malignant brain tumor and is characterized by significant intratumoral heterogeneity. This study aimed to investigate the clinical and genomic landscapes of GBM across diverse ethnic populations to identify potential prognostic markers. Methods: Leveraging The Cancer Imaging Archive (TCIA) and bioinformatics modeling, White, African, and Asian American cohorts were analyzed. Patients were stratified according to the 2021 WHO classification of central nervous system (CNS) tumors. Population-specific genomic drivers and phenotypic markers were evaluated for their impact on outcomes. Survival rates across age, sex, and ethnicity were estimated using the… More >
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.078376 - 21 May 2026
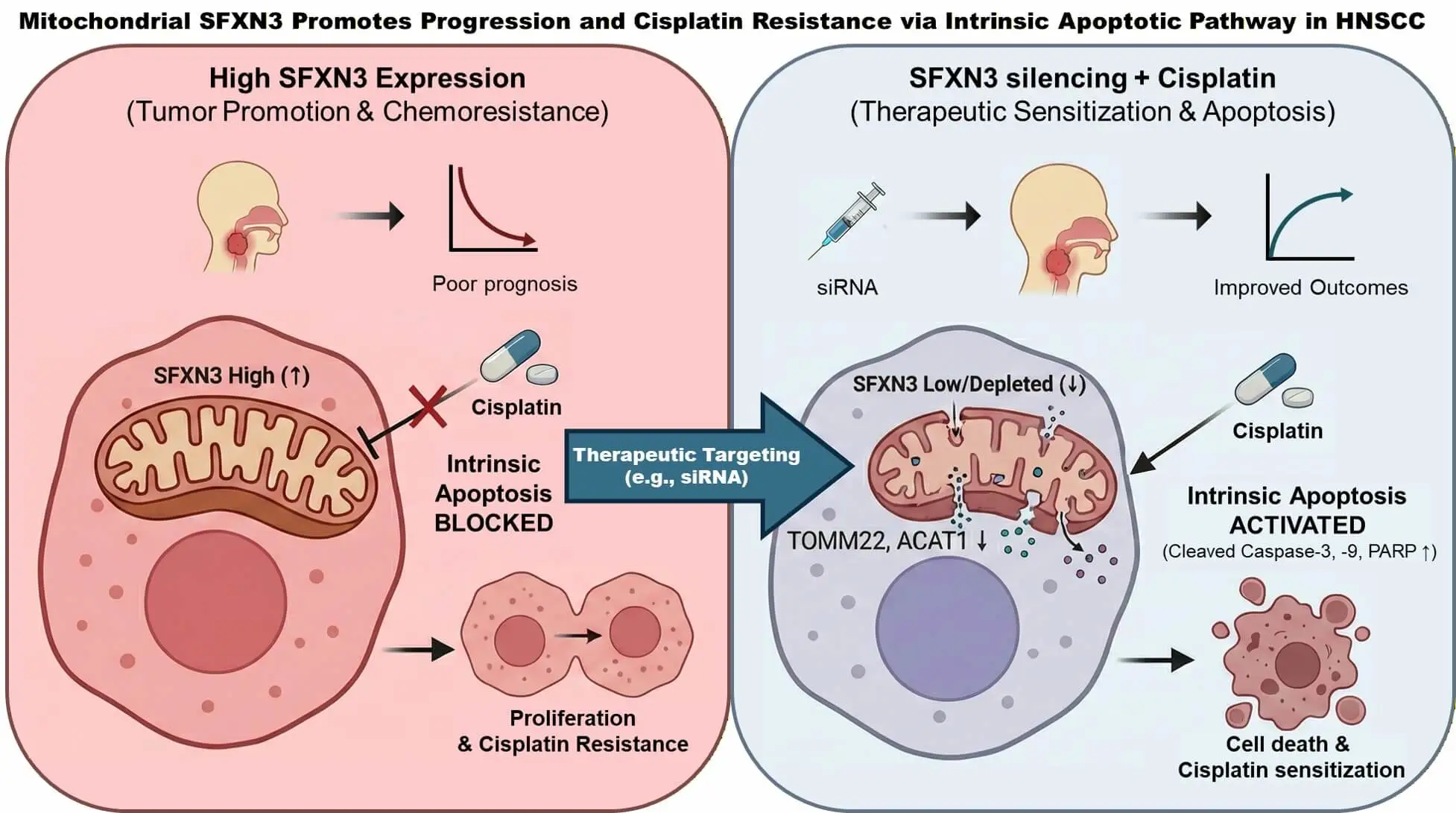
Abstract Objective: The systematic evaluation of expansive genomic databases facilitates the discovery of clinically vital biomarkers. While Sideroflexin 3 (SFXN3) consistently displays elevated expression in head and neck squamous cell carcinoma (HNSCC), its specific pathobiological functions and prognostic value remain insufficiently characterized. This study aims to delineate the clinical and functional significance of SFXN3 in HNSCC. Methods: We interrogated SFXN3 expression patterns, patient survival outcomes, and immune cell infiltration characteristics utilizing multiple independent repositories, including the cancer genome atlas (TCGA) and gene expression omnibus (GEO). The prognostic independence of SFXN3 was verified via multivariate Cox regression. These… More >
Graphic Abstract
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.077514 - 21 May 2026
(This article belongs to the Special Issue: Advances in Cancer Immunotherapy)
Abstract Objective: Studies on the comprehensive utility of complete blood count-derived inflammatory biomarkers (CBC-IBs) as biomarkers in pembrolizumab-treated advanced non-small-cell lung cancer (NSCLC) are scarce. This study aimed to investigate the clinical relevance of a panel of CBC-IBs as potential predictive biomarkers and assess whether integrating the systemic immune-inflammation index (SII) with programmed death-ligand 1 (PD-L1) expression could overcome the limitations of PD-L1 as a standalone predictive biomarker. Methods: Our real-world preliminary study was conducted on a cohort of patients with advanced NSCLC. Patients who had undergone PD-L1 immunohistochemistry testing at the time of diagnosis, and had… More >
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.079463 - 21 May 2026
(This article belongs to the Special Issue: The Neural Niche in the Tumor Microenvironment: From Mechanistic Insights to Therapeutic Targeting)
Abstract Objectives: Gamma-aminobutyric acid type B (GABAB) receptors are involved in tumor progression, and baclofen exerts broad-spectrum antitumor effects in various cancers. Nevertheless, its specific function and underlying molecular mechanisms in glioma are still largely unclear. This study aimed to evaluate the effects of baclofen on glioma cells and elucidate the associated signaling pathways. Methods: The antitumor effects of baclofen were evaluated in glioma cell lines, and its underlying molecular mechanisms were explored using transcriptome sequencing integrated with Western blotting. The in vivo antitumor efficacy of baclofen was further verified in animal models. Results: In vitro functional assays revealed that… More >
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.074145 - 21 May 2026
(This article belongs to the Special Issue: AI-Guided Phenotypic Response Surfaces for Precision Oncology: From Model Systems to Clinical Dosing)
Abstract Objectives: Hepatocellular carcinoma (HCC) arising in metabolic dysfunction–associated steatotic liver disease (MASLD) develops under lipid-rich stress and inflammatory remodeling, which can alter therapeutic windows. We aimed to determine whether phenotypic response surface–guided optimization (PRS-OPT) can nominate hepatocyte-sparing propolis–metformin–regorafenib (PMR) dose windows that retain antitumor activity under MASLD-like fatty-acid (FA) stress and translate to an in vivo immune endpoint. Methods: PMR combinations were profiled in hepatoma cell lines (PLC/PRF/5 and HepG2) and non-malignant hepatocytes (THLE-2) under FA-free and FA-enriched conditions. Quadratic response surfaces were fitted and used for constrained dose nomination, followed by in vitro validation. Cell-death contributions were… More >
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.078651 - 21 May 2026
(This article belongs to the Special Issue: Identification of potential targets and biomarkers for cancers and the exploration of novel molecular mechanisms of tumorigenesis and metastasis)
Abstract Objectives: Uridine-cytidine kinase 2 (UCK2) plays a crucial role in the pyrimidine salvage pathway, but its function in lung adenocarcinoma (LUAD) is still largely unclear. The study aimed to investigate the expression, prognostic value, biological functions, and molecular mechanisms of UCK2 in LUAD. Methods: Bioinformatic analyses were performed using The Cancer Genome Atlas (TCGA), Gene Set Cancer Analysis (GSCA), Gene Expression Omnibus (GEO), and Genotype Tissue Expression (GTEx) datasets. In vitro assays evaluated the effect of UCK2 overexpression on LUAD cells. Co-immunoprecipitation and pathway analyses were utilized to explore the underlying mechanism. Immune landscape and drug sensitivity… More >
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.073934 - 21 May 2026
Abstract Objectives: Cisplatin resistance is a major obstacle in the treatment of renal cell carcinoma (RCC), severely compromising therapeutic efficacy and patient prognosis. This study aimed to clarify the role and molecular mechanism of cyclin-dependent kinase 4 (CDK4) in cisplatin resistance of RCC. Methods: Immunohistochemistry (IHC) was used to detect the expression of CDK4 in cisplatin-resistant RCC tissues. In RCC cells and their drug-resistant sublines, CDK4 overexpression/knockdown assays were performed to evaluate the effects on cisplatin resistance and malignant progression. An in vivo model was established, to verify the in vivo function of CDK4. Transcriptome sequencing (RNA-seq), Cleavage Under… More >
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.078665 - 21 May 2026
(This article belongs to the Special Issue: Long noncoding RNAs as Tumorigenic Drivers and Therapeutic Targets)
Abstract Objective: Osimertinib can selectively inhibit both epidermal growth factor receptor (EGFR) sensitizing and T790M gatekeeper mutations, and has shown remarkable therapeutic effects in patients with lung adenocarcinoma. However, almost all patients inevitably develop drug resistance. Herein, we sought to clarify the roles of exosomal lncRNA H19 in modulating osimertinib resistance, focusing on the PI3K-PTEN-Akt signaling axis. Methods: Functional assays, including cell viability assay, colony formation, cell apoptosis and xenograft mouse, employed in evaluate the effects of exosomal lncRNA H19 on cell growth and apoptosis. RNA quantitation and western blot were adopted to demonstrate the regulatory roles of… More >
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.078833 - 21 May 2026
Abstract Background: Pediatric sarcomas are aggressive malignancies characterized by marked biological heterogeneity and a high risk of relapse. Standard surveillance relies on imaging and invasive biopsies, which may fail to detect early molecular disease. Liquid biopsy using circulating tumor DNA (ctDNA) and microRNAs offers a minimally invasive strategy for longitudinal monitoring. This study aimed to evaluate dynamic changes in ctDNA and circulating microRNAs during treatment and examined their associations with treatment response, disease recurrence, and survival outcomes. Methods: This prospective cohort study included 100 pediatric patients with histologically confirmed sarcomas. Serial peripheral blood samples were collected at… More >
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.075185 - 21 May 2026
Abstract Objectives: Although claudin-1 (CLDN1) interacts with Cluster of Differentiation 81 (CD81) in various cell types, the specific mechanism underlying this interaction and its functional implications in colorectal cancer (CRC) cells remain poorly understood. This study outlines the regulatory role of CLDN1 in CRC cell tumorigenicity through its interaction with CD81, elucidating the underlying signaling cascade. Methods: Changes in the expression of CLDN1 and CD81, as well as their correlation with the survival of CRC patients, were analyzed using samples from The Cancer Genome Atlas database, the Kaplan‒Meier plotter database, and tissue microarrays. CLDN1 and CD81 were… More >
 Open Access
Open Access
ARTICLE
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.078278 - 21 May 2026
(This article belongs to the Special Issue: Novel Biomarkers and Treatment Strategies in Solid Tumor Diagnosis, Progression, and Prognosis (Ⅱ))
Abstract Background: Breast cancer treatment is often hampered by the immunosuppressive tumor microenvironment (TME). To improve therapeutic efficacy, this study developed a folic acid-chitosan (FA-CS)-modified liposomal formulation co-delivering doxorubicin (DOX) and resiquimod (R848) for combined chemotherapy and immune modulation. Methods: The FA-CS-R848/DOX@Lip liposomes were prepared by rotary evaporation and characterized for morphology, particle size, zeta potential, drug encapsulation efficiency (EE), drug loading (DL) capacity, and drug release profiles. Cellular uptake and cytotoxicity were determined to assess the biological effects of the formulation. Antitumor efficacy and biosafety were assessed in an EO771 tumor-bearing mouse model. The macrophage phenotype,… More >
 Open Access
Open Access
CASE REPORT
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.067876 - 21 May 2026
Abstract Background: Orbital metastases are rare in breast cancer, representing only 3–10% of ocular metastases. This report highlights a case where orbital involvement was the first indicator of systemic metastatic spread. Case Presentation: A 72-year-old woman with a history of Estrogen Receptor (ER)-positive (5%), Progesterone Receptor (PR)-negative, Human epidermal growth factor receptor-2 (HER2)-negative breast cancer (diagnosed 3 years prior) presented with right orbital pain, diplopia, and periorbital swelling. Imaging revealed multiple myositis of the extraocular muscles, compressive displacement of the optic nerve, and right periorbital edema. Bone scintigraphy identified multifocal skeletal metastases. A navigation-assisted biopsy confirmed metastatic invasive More >
 Open Access
Open Access
RETRACTION
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.085502 - 21 May 2026
Abstract This article has no abstract. More >
 Open Access
Open Access
RETRACTION
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.085235 - 21 May 2026
Abstract This article has no abstract. More >
 Open Access
Open Access
RETRACTION
Oncology Research, Vol.34, No.6, 2026, DOI:10.32604/or.2026.085239 - 21 May 2026
Abstract This article has no abstract. More >