
Oncology Research
ISSN: 0965-0407 (Print)
ISSN: 1555-3906 (Online)
Shortcuts
Cover Letter
Templates
General Format of Articles
Declarations
Research Data and Supplementary Materials
Artificial Intelligence Policy
Editors and Journal Staff as Authors
Authors’ Guideline
All manuscripts must be submitted via the online system, and manuscripts submitted for publication must be prepared according to the guidelines given below.
Template in MS Word: Microsoft Word template
References Style file for EndNote: Vancouver
This guideline is intended to assist authors as they prepare their manuscripts. To avoid any delay and time-consuming restructuring, Oncology Research asks and encourages authors to read the guidelines before writing the manuscript.
Article Types
Oncology Research publishes Research Articles, Reviews, Case Reports, Editorial, Analysis, Letters and other types of manuscripts. All papers must be written in English and must follow a clear concise style. The language editors may have to check the language and grammar of your submitted manuscript and make editorial changes if deemed necessary.
Research Articles
A Research article is a detailed technical report of an original study that is likely to impact its field. It is a primary report where authors collect and analyze data and draw conclusions from the results leading to an original study in the literature. Research articles incorporate a comprehensive list of elements i.e., Title, Keywords, Authors and Affiliations, Abstract, a substantive Introduction, Material and Methods, Results, Discussion and Conclusion. There is no specific word count limitation; however, manuscripts must be as concise as possible.
Review Articles
A Review article is a paper based on other published research. It is a secondary source. It does not report original research but rather critically evaluate previously published material. Typically, a review article analyzes or synthesizes existing literature on a subject with the aim of expanding on its current understanding or sums up the already existing work to relate it to its present status and suggest new research directions. Systematic reviews and meta-analyses should use the same structure as research articles and adhere to the PRISMA guidelines, and authors should also include a completed PRISMA checklist and PRISMA flow diagram as supporting files.
Case Reports
A Case report is a detailed article that describes and interprets symptoms, signs, diagnosis, treatment, and follow-up on a patient(s). It often covers an unusual or novel medical occurrence in the form of a narrative. Case Reports should include a succinct Introduction about the patient’s general medical condition or symptoms to be discussed, a case Presentation which includes all of the relevant de-identified demographic, descriptive Information about the patient(s), a description of the symptoms, diagnosis, treatment, and outcomes, a discussion providing context and interpretation of specific treatment decisions, and finally a Conclusion briefly outlining observations and recommendations. Case reports should be prepared according to the CARE guidelines, with the CARE checklist provided as an additional supplementary file.
Editorials
Editorials are short personal perspectives about topics relevant to the journal’s aims and gateways. Editorials are not formally peer-reviewed and must not include new research and data. They are evaluated by the editorial team in-house, if necessary, in consultation with advisory board members.
Analysis
An Analysis article reports comprehensive comparative analyses of technologies, methods or reagents of key importance for a field of research, leading to important practical conclusions about their performances. Analysis articles may also report on new analysis of existing large datasets that lead to a novel, exciting or arresting conclusion. The main text (excluding Abstract, Online Methods, References and Figure legends) is approximately 3,000 words. As a general guideline, an analyses report is limited to a total of 50 references.
Cover Letter
A submitted manuscript must be accompanied by a cover letter. The cover letter must clearly state that the manuscript is an original work with its merit, has not been previously published in whole or in part, and is not being considered for publication elsewhere. It should also include statements indicating that all authors have read the final manuscript, have approved the submission to the journal, and have accepted full responsibilities pertaining to the manuscript’s delivery and contents. If any ethical, copyright, disclosure issues come with the manuscript, please reveal them in the cover letter. In the cover letter, the authors need to declare that there is no conflict of interest or disclose all the conflicts of interest regarding the manuscript submitted.
Templates
Use the Microsoft Word template or LaTeX template to prepare your manuscript.
Authors are strongly encouraged to use either the Microsoft Word or LaTeX template to prepare their manuscript. Using the provided template will significantly speed up the copy-editing and publication process for accepted manuscripts. The total file size for all submissions must not exceed 200 MB. If the file size exceeds this limit, please contact the Editorial Office at oncologyresearch@techscience.com. Accepted file formats are:
● Microsoft Word: Manuscripts prepared in Microsoft Word must be submitted as a single file. When using Microsoft Word, we recommend using the Microsoft Word template (each section has a predefined style, accessible via the “Styles” gallery in Word). Figures should be inserted into the main text immediately after the paragraph where they are first cited.
● LaTeX: Manuscripts prepared in LaTeX should be submitted in a single ZIP folder containing all source files and images, allowing the Editorial Office to recompile the PDF. We recommend using the LaTeX template for LaTeX manuscripts.
● Supplementary Files: These can be in any format, though it is recommended to use common, non-proprietary formats whenever possible.
General Format of Articles
Manuscripts should comprise:
Front matter: Title, Author list, Affiliations, Abstract, Keywords.
Main text: For Articles, a structured format, e.g., Introduction, Results, Discussion, Methods, Conclusions, is recommended, while Reviews may use a more flexible structure.
Back matter: Acknowledgment, Funding Statement, Author Contributions, Availability of Data and Materials, Ethics Approval, Conflict of Interests, Supplementary Materials (if any), Glossary (if any), Appendices (if any), and References.
Front Matter
● Title
The title of your manuscript should be precise, clear, and directly related to the study. It should indicate whether the research involves human or animal trial data, or if it is a systematic review, meta-analysis, or replication study. Avoid including short forms like running titles or headers, as these will be removed by the Editorial Office. They can be used in abstracts only if the related expanded form is provided (just after the acronym, in parentheses) the first time they are used.
● Author Information
Please provide the full names of all authors, including first and last names, with middle name initials included if necessary. Affiliations should follow the PubMed/MEDLINE standard format, including detailed address information such as city, postal code, state/province, and country. At least one author must be designated as the corresponding author, whose email address will be publicly displayed in the published article. Please note that after acceptance, changes to author names or affiliations may not be permitted. For authors with equal contributions, use a superscript symbol (#) for identification and include the following statement below the affiliation: “These authors contributed equally to this work.” Authors with equal contributions should, in principle, be listed consecutively in the author list. Additionally, equal contributions should be clearly detailed in the author contributions section. Please review the relevant criteria to ensure authorship qualifications are met.
● Author Affiliations
All authors must include their current affiliation as well as the institution where the majority of the research for the manuscript was conducted. The primary affiliation should typically reflect the institution that provided the most support or where the bulk of the research was performed, but authors are encouraged to confirm any specific requirements with their institution, particularly regarding contractual agreements.
Accurate author names and affiliations are crucial to ensure proper attribution, citation, and to avoid issues related to recognition, promotions, or funding. Once an article is published, requests for updates or corrections to author details, including affiliations, may not be accommodated.
For Independent Researchers
Authors who are not currently associated with any university, institution, or organization, and were not during the development of the manuscript, should identify themselves as “Independent Researchers.”
● Abstract
Abstracts of a research paper should be typically 200 to 400 words in length, and 150 to 300 words for a review paper. Abstracts shall be running continuously and shall not include referencecitations. Abbreviations that appear only once in the abstract should be defined in full. If abbreviations appear more than once, the full definitions should be provided first before they can be used elsewhere.
● Keywords
Please list 3 to 10 pertinent keywords specific to the article yet reasonably common within the subject discipline.
Main Text
The text should be written in clear, concise English, and it should be easily understandable to a broad readership. Sentences should not begin with abbreviations or numerals. Main headings should be Introduction, Material and Methods, Results (or Systematic Description) and Discussion. Combining Results and Discussion is not allowed.
● Materials and Methods
Any commercialized kits, reagents, instruments, software, antibodies, etc. used in the research, shall be provided with their full name, along with the information of the Manufacturers/suppliers/software details (Name, City, Province/State, Country).
Accession numbers of RNA, DNA and protein sequences used in the manuscript should be provided.
● Figures and Tables
Order
1. Figures and tables should be numbered consecutively using Arabic numerals and placed within the text immediately following their first citation to maintain a seamless flow and clarity in the presentation, and the first citation of figures and tables in the main text must follow a sequential order (as well as text Boxes and Algorithms). In the published article, the figures are inserted based on the placement of the first citation and caption.
2. The lettered subpanels of whole figures may be cited in any order in the text following the first mention of each whole figure in numerical order. For example, any subpart of Fig. 3 may be cited in any order (e.g., Fig. 3C before Fig. 3A) provided that Figs. 1 and 2 have already been cited.
Content
1. The figure content should be complete and the characters should not be masked. Unnecessary marks such as red wavy lines and hard (soft) returns are not allowed in figures;
2. Any special characters or icons in an image/table (e.g., *, **, ***, #, ...) need to have a corresponding explanation (can be added in the image or caption);
3. Please remove all non-English terms or add a definition for them;
4. References in the form of “[xx]” are not allowed in images. “Author + Year” format can be used in the image, and all mentioned references must be cited in the caption if necessary;
5. No specific feature of an image should be augmented, altered, enhanced, obscured, moved, or removed. The focus should be on the data rather than its presentation (e.g., background, imperfections, and non-specific bands should not be “cleaned up”);
Resolution and Format
1. Figures should be scaled to a maximum width of 16.51 cm (6.50 in) and height of 20 cm (7.87 in), preserving their original proportions without distortion. Any excess white space surrounding a figure should be removed before calculating its size. The preferred format is .tif, with RGB color space, a DPI of 500+ (accepted image resolutions: Line Art ≥ 900 dpi, Halftone ≥ 300 dpi, Combo ≥ 600 dpi), no alpha channels, and flattened layers;
| Image Type | Description | Recommended Format | Resolution |
| Line Art | An image composed of lines and text, which does not contain tonal or shaded areas | tif or eps | 900–1200 dpi |
| Halftone | A continuous tone photograph, which contains no text | tif | 300 dpi |
| Combo | Image contains halftone + text or line art elements | tif or eps | 500–900 dpi |
2. To avoid any errors during position changes, please provide the combined image instead of editable pieces in the figure;
3. Please provide an editable table in MS Word format, not images (including algorithms, listings, etc.);
4. RGB (8 bit/channel), CMYK, or greyscale mode are acceptable;
5. Do not use Photoshop or such software to change the color or appearance of figures.
Labels and Captions
1. Figure labels must be sized in proportion to the image, sharp, and legible. The label size should be no smaller than 8-point and no larger than the font size of the main text;
2. Labels must be saved using standard fonts (Arial, Helvetica or Symbol font) and should be consistent for all the figures;
3. All labels should be in black, and should not be overlapped, faded, broken or distorted, feature unnecessary gaps or irregular spacing, or appear condensed, expanded, or otherwise distorted either horizontally or vertically;
4. Space must be inserted before measurement units. The first letter of each phrase, not each word, must be capitalized.
5. Provide a short title (in the legend, not on the figure itself) and an explanation in brief but sufficient detail to make the figure intelligible without reference to the text. Statistical evaluations should indicate the test used.
6. Each panel of a multi-panel figure (referred to as, e.g., Figs. 1A, 1B, 1C, and 1D in the text) should be logically connected to the other panels, and all of the panels should be assembled into a single file on a single page. Images that contain large amounts of information should be broken down into multiple figures to ensure that all of the information is visible. To repeat, multiple panels must be assembled and submitted as a single file rather than as separate files.
7. The sublabels for panels (again, referred to as, e.g., Fig. 1A, 1B, 1C, and 1D in the text) should be placed in the top left-hand corner of the panels and contrast clearly with the background. Note that each panel should be labeled with only a letter (e.g., A, B, C, and D—not 1A, 1B, 1C, and 1D).
Scale Bar
1. A scale bar, rather than magnification, must be provided for any micrographs. The scale bar should be explicitly and prominently displayed on each figure; one cannot simply add a line on the figures without any scale bar description or simply add a description of the scale in the figure legend without a scale bar in the figure. This practice is mandatory for all micrographs and petri dish diagrams.
Copyright of Figures and Tables
1. Ensure that permission has been obtained and there is no copyright issue. If copyright is needed, please provide a citation in the following format: “Reprinted/adapted with permission from reference [xx]. Copyright year, copyright owner’s name”. All figures, tables, and images will be published under a Creative Commons CC-BY license, and permission must be obtained for the use of copyrighted material from other sources.
2. If a figure or table has been published previously (even by an author of the manuscript being submitted for review), copyright permission for reuse of the figure or table will often be required. The acknowledgment and written permission from the copyright holder will be required where necessary. It is the responsibility of the authors to acquire the licenses, follow any citation instructions requested by third-party rights holders, and cover any supplementary charges.
3. For any figures (or tables) that contain data from a public database (e.g., Gene Ontology/KEGG), the source should be cited in the caption, legend, or title explicitly. For publicly available DNA sequences, the accession number should be provided.
Images of Gels and Blots
1. Raw Data Accessibility: Ensure that raw, unprocessed images are available to readers. Authors are encouraged to provide the original images without cropping or any other processing in the supplementary materials.
2. Minimize Cropping: Figures should not be over-cropped around the bands of interest. A small background area should be retained above and below the bands. Non-specific bands, which are common, should be included in the figure and explained in the figure caption or legend.
3. Molecular Weight Markers: Each Western Blot figure should include at least one molecular weight marker above and below the protein of interest.
4. Same Gel for Comparative Analysis: When presenting comparative analysis of bands, all relevant samples should be run on the same gel or blot. If data from multiple blots or gels are used, they should be clearly distinguished as separate panels, and the caption should clarify that multiple gels, blots, or exposures are being presented.
5. Control Samples: Each figure should include relevant controls. Control samples, when appropriate, should be run on the same blot or gel alongside the experimental samples. If controls are run on different gels, this must be acknowledged in the caption or legend.
6. Avoid Composite Images: Figures should not include composite images of bands from multiple blots, exposures, or gels unless clearly indicated. Any rearrangement or splicing of lanes should be indicated with vertical black lines, and the figure caption or legend should explain how the figure was prepared.
7. Data Presentation with Dot Plots: It is preferable to present data using dot plots, as they convey information on sample size and data spread.
8. Quantitative Comparison Across Gels: Quantitative comparisons of samples across multiple gels or blots are strongly discouraged. If such a comparison is necessary, the figure legend must state whether the samples are from the same or parallel experiments, and whether the gels or blots were processed in parallel.
9. Lane Rearrangement: Any rearrangement of lanes within a single blot or gel must be clearly indicated. If lanes are non-adjacent, the boundary between the rearranged lanes should be marked, and this should be acknowledged in the figure caption.
10. Detailed Methods Reporting: Authors should provide a detailed description of the experimental methods, including the amount of total protein loaded onto the gel, the membrane blocking protocol, antibody identifiers, antibody labeling protocols, type and number of replicates performed, and any other relevant procedural details.
11. Image Quality: High-contrast images should be avoided, as overexposure may mask additional bands. Authors should ensure that images are clear, well-lit, and properly exposed to show all relevant data.
12. Duplication Check: Authors must carefully check their figures for duplication. They should also ensure that any cropped images retain all critical bands and match the unprocessed scans provided.
For further examples and templates on how to improve Western Blot figure reporting, please refer to the following resources:
Figure Samples
From: Monica RD, Cuomo M, Visconti R, Mauro A, Buonaiuto M, Costabile D, et al. Evaluation of MGMT Gene Methylation in Neuroendocrine Neoplasms. Oncol. Res. 2020;28(9):837–45.
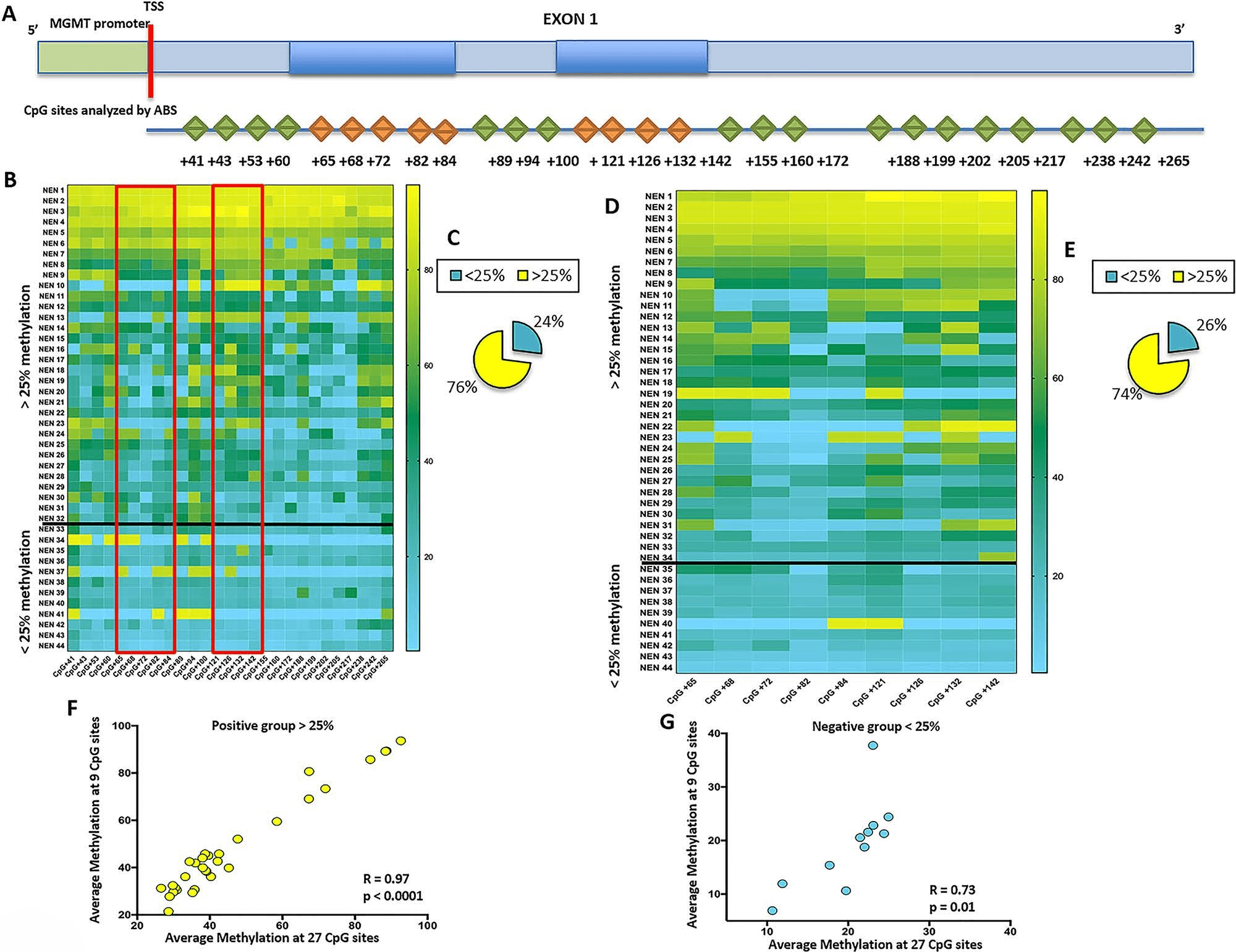
Figure 1: (A) Graphic representation of the MGMT gene regulatory region analyzed by methylation-specific polymerase chain reaction (PCR) (MSP) and amplicon bisulfite sequencing (ABS). Upper figure: MGMT gene promoter (green) and exon 1 (light blue) are represented. The regions spanned by MSP primers are highlighted within exon 1 (dark blue). Lower figure: The CpG sites analyzed by ABS are shown as rhombuses; in orange, the CpG sites interrogated also by the MSP test. CpG numbering is referred to nucleotide positions from the transcriptional start site (TSS). (B) DNA methylation heatmap of the 42 analyzed neuroendocrine neoplasms (NENs); in rows, the ID of each tumor sample, and in columns, each CpG site analyzed by ABS, with nucleotide position from TSS. Among CpGs, those in the red box are the CpG sites analyzed by both MSP and ABS. Colors related to grade of methylation (from light blue, nonmethylation, to yellow, high methylation) are shown on the right. The horizontal black line represents the cutoff value (25% of average methylation). (C) Graphic representation of the percentage of positive and negative samples based on all analyzed CpG. In yellow, the percentage of samples with average methylation at all 27 CpG sites >25%; in light blue, the percentage of samples with average methylation at all 27 CpG sites <25%. (D) DNA methylation heatmap of the CpG sites covered by both ABS and MSP; in rows, the ID of each sample, and in columns, each CpG site. The adjacent line indicates colors related to grade of methylation (from light blue, nonmethylation, to yellow, high methylation). The horizontal black line represents the cutoff value (25% of average methylation) and divides the map in two zones: methylated samples (over the cutoff line) and nonmethylated samples (under the cutoff line). (E) Graphic representation of the percentage of positive and negative samples based on nine CpG sites covered by both ABS and MSP: in yellow, the percentage of samples with average methylation at nine CpG sites >25%; in light blue, the percentage of samples with average methylation at nine CpG sites <25%. (F) Correlation between the average methylation at all 27 analyzed CpG sites and the average methylation at 9 analyzed CpG in positive samples (>25%). Correlation plot indicates on the x axis the average methylation at 27 CpG sites for each sample and on the y axis the average methylation at 9 CpG sites for each sample with a total average methylation >25%. (G) Correlation between the average methylation at all 27 analyzed CpG sites and the average methylation at 9 analyzed CpG in negative samples (<25%). Correlation plot indicates on the x axis the average methylation at 27 CpG sites for each sample and on the y axis the average methylation at 9 CpG sites for each sample with a total average methylation <25%
From: Song Q, Zhang J, Zhang Q, Liu J, Lv K, et al. Exosomes derived from circBCRC-3-knockdown mesenchymal stem cells promoted macrophage polarization. BIOCELL. 2020;44(4):623–29.
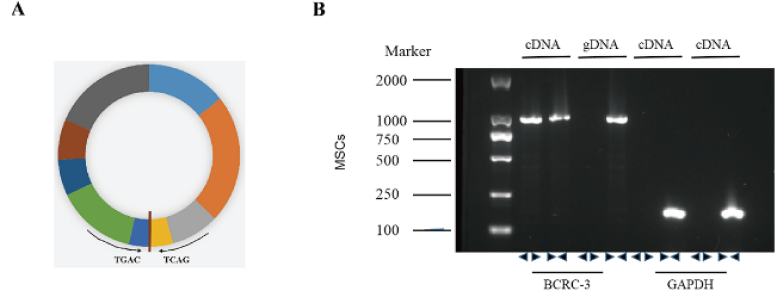
Figure 1: Identification of circBCRC-3 in MSCs. (A). The genomic structure indicates that circular RNA BCRC-3 consists of nine exons (1002 bp) from the PSMD1 gene. (B). Agarose gel electrophoresis analysis of PCR product with divergent and convergent primers of circBCRC-3 in cDNA and gDNA. GAPDH was used as the negative control
From: Zhang J, Huang Y, Liu W, Li L, Chen L. Chaperone-mediated autophagy targeting chimeras (CMATAC) for the degradation of ERα in breast cancer. BIOCELL. 2020;44(4):591–95.
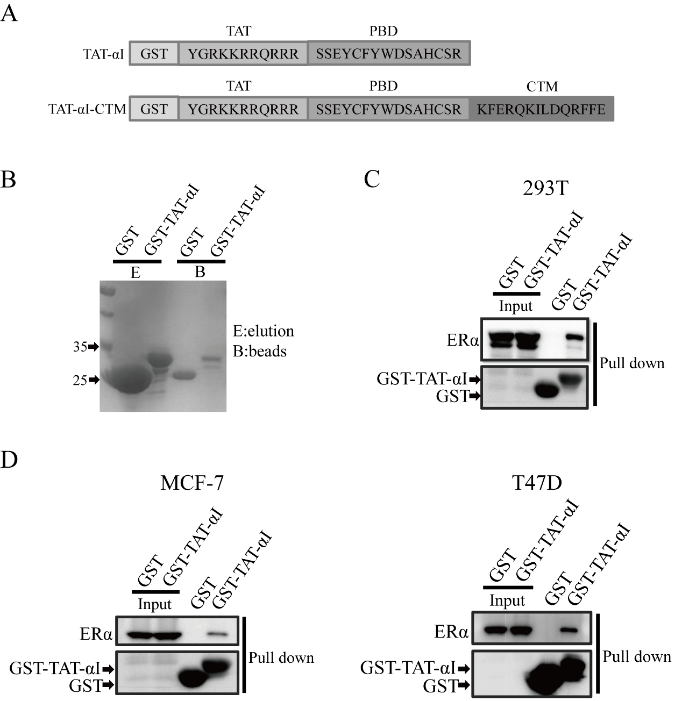
Figure 2: The binding of peptide αI to ERα. (A) Design TAT-αI-CTM and TAT-αI peptides. (B) Production of GST and GST-TAT-αI using an E. coli expression system. Coomassie blue staining after SDS-PAGE assessed their purity. (C) Pull-down of TAT-αI and ERα. HEK 293T cells were transiently transfected with plasmids pEGFP-N2-ERα. 48 h after transfection, cell lysates were subjected to GST pull down, and the pull-down fractions were immunoblotted analyzed. (D) Pull-down of TAT-αI and ERα in ERα-positive breast cancer cell lines, MCF-7 and T47D
From: Fu S, Yun T, Ma D, Zheng B, Jiang D, et al. Thylakoid Transit Peptide Is Related to the Expression and Localization of NdhB Subunits in Soybean. Phyton-Int J. Exp Bot. 2021;90(1):99–110.
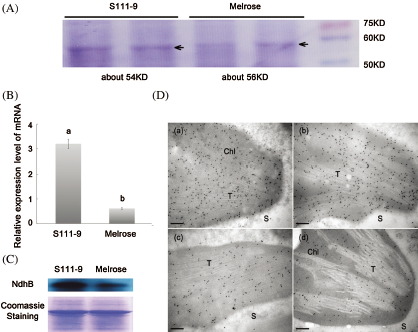
Figure 3: Comparisons of sizes, relative abundance of mRNA and protein, and immunolocalization for NdhB subunits in Melrose and S111-9. (A) Sizes of NdhB subunits were analyzed by SDS-PAGE. NdhB proteins were isolated and purified from the leaves of Melrose and S111-9 extracted with Crosslink IP Kit. (B) Relative mRNA expression level of ndhB in Melrose and S111-9. Bars represent the mean ± SD of three biological replicates. Student t-test was applied to assess difference of means between two varieties. Different letters indicate a significant difference between varieties (p < 0.05). (C) The content of NdhB were analyzed using western blotting in the leaves of Melrose and S111-9. The equal loading of lower pictures is shown by coomassie staining. (D) Immunolocalization of NdhB subunits in S111-9 (a,b) and Melrose (c,d). All immunogold particles were mainly concentrated in chloroplast, not in the cytosol. T, Thylakoid; S, Cytosol Stroma. Scale bars = 0.2 μm
From: Yang Y, Xu K, Zhao B, Liu N, Zhou J. The Bacteria Absorption-based Yolk-Shell Ni3P-Carbon @ Reduced Graphene Oxides for Lithium-Ion Batteries. J Renew Mater. 2021;9(5):855–65. Doi:10.32604/jrm.2021.014525.
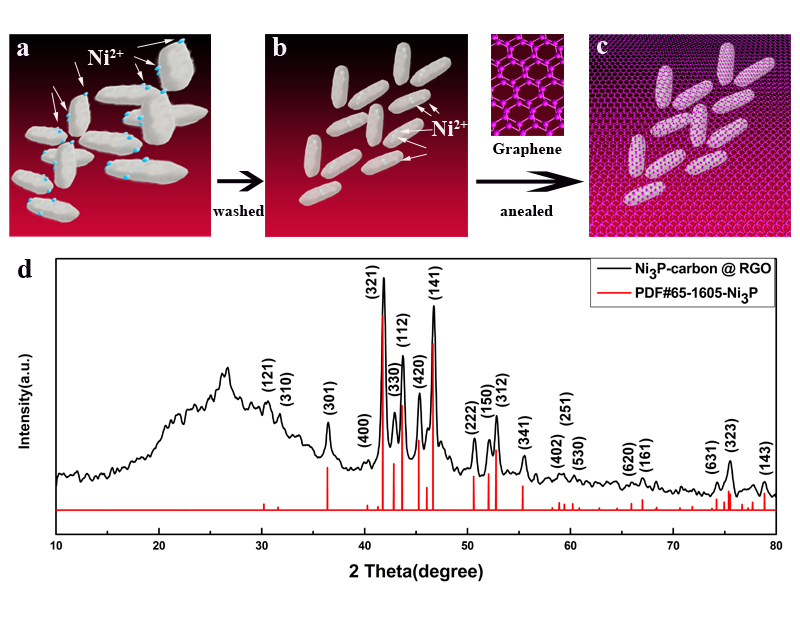
Figure 1: Schematic diagram of material synthesis process (a–c) and XRD pattern of the NPC @ RGO (d)
● Acronyms/Abbreviations/Initialisms
Acronyms, abbreviations, and initialisms must be defined upon their first use in three sections: the abstract, the main text, and the first figure or table. When first introduced, include the abbreviation or acronym in parentheses following the full term. Accepted abbreviations for statistical parameters are: P, n, SD, SEM, df, ns, ANOVA, t.
● International System of Units
The use of SI Units (International System of Units) is required. Whenever possible, convert imperial, US customary, or other units to SI equivalents.
1. There should be a space between the unit and Arabic number: 5 mm NOT 5mm.
2. There should be a space before and after the operator: 3 cm × 5 cm NOT 3 cm×5 cm.
3. Please use Arabic number and SI Units (International System of Units) in the manuscript: 5 kg NOT five kilograms or 5 kilograms or five kg.
4. Do not use hyphen/dash or any connector symbol between the value and its unit: 5 kg NOT 5-kg.
5. Please clarify all units during a calculation or a mathematical relationship: 3 cm × 5 cm NOT 3 × 5 cm, 123 g ± 2 g or (123 ± 2) g NOT 123 ± 2 g, 70%–85% NOT 70–85%.
● Symbols
Greek letters must be inserted using the correct Greek symbol (using Times, Helvetica or Symbol font), NOT written in full, i.e., alpha: α; beta: β, ß, (available in Times and Helvetica); and gamma: γ, etc.
● Equations
For equations, if preparing your manuscript in Word, use the Microsoft Equation Editor or the MathType add-on. Ensure that equations are editable and not inserted as images.
● Statistical Analysis
Appropriate statistical treatment of the data is essential. When statistical analysis is performed, the name of the statistical test used, the number for each analysis, the comparisons of interest, the alpha level and the actual p-value for each test should be provided.
· When the P value is less than 0.001, report as P < 0.001.
· When the P value is greater than 0.99, report as P > 0.99.
· Special care should be taken when reporting P values of P = 0 or P = 1, as these values are rare and may require additional clarification.
● Scientific Naming and Formatting Rules
Linnean scientific names should be in italics, while higher than generic taxa should not. The generic name of drugs, as well as all other common names, should be written in lower case. Gene designations should be in lower case and in italics, while protein designations should be in regular capital letters. All the p in p value, whether in the text or on the figure, should be lowercase and italic.
For more details and examples, please refer to the template.
Back Matter
● Declarations
Please note that the 6 pieces of information (Acknowledgement, Funding Statement, Author Contributions, Availability of Data and Materials, Ethics Approval, Conflicts of Interest) need to be truthfully provided at the end of the article.
Acknowledgement
This section is intended for acknowledging any support not covered under the Author Contributions or Funding Statement sections. This may include administrative and technical assistance, as well as in-kind contributions such as materials or equipment provided for the research. Please be aware that the specific funding grant number should only appear in the Funding Statement. If there are no acknowledgments to be made, please use “Not applicable”.
Funding Statement
Authors should describe sources of funding that have supported the work, including specific grant numbers, initials of authors who received the grant, and the URLs to sponsors’ websites: “This research was funded by Name of Funder, grant number xxx” or “The APC was funded by xxx”. If there is no funding support, please write “The author(s) received no specific funding for this study”.
Author Contributions
The Author Contributions statement is mandatory for research articles, except for papers with a single author. It should represent all the authors and is to be included upon submission. All listed authors must have substantially contributed to the manuscript and have approved the final submitted version, which should include a description of each author’s specific work and contribution. We suggest the following format for the contribution statement:
“The authors confirm contribution to the paper as follows: Conceptualization, First-name Lastname1 and First-name Lastname2; methodology, First-name Lastname1; software, First-name Lastname1; validation, First-name Lastname1, First-name Lastname2 and First-name Lastname3; formal analysis, First-name Lastname1; investigation, First-name Lastname1; resources, First-name Lastname1; data curation, First-name Lastname1; writing—original draft preparation, First-name Lastname1; writing—review and editing, First-name Lastname1; visualization, First-name Lastname1; supervision, First-name Lastname1; project administration, First-name Lastname1; funding acquisition, First-name Lastname1. All authors reviewed the results and approved the final version of the manuscript”.
Please turn to the CRediT role descriptors—CRediT for the term explanation.
Availability of Data and Materials
This statement should inform readers where the data supporting the findings can be accessed, or explain why access is restricted. Please select the most appropriate category from the list below and adapt the suggested wording.
1. Data Available in a Public Repository
Use when data are fully open and archived in a recognized repository.
Statement Template:
“The data supporting the findings of this study are openly available in [repository name] at [DOI/URL].”
2. Data Available within the Article or Supplementary Material
Use when all data are included directly within the manuscript or its supplementary files.
Statement Template:
“All data supporting the results of this study are included within the article and/or its Supplementary Materials.”
3. Data Available upon Reasonable Request
Use when data can be shared with researchers on request.
Statement Template:
“The data supporting the findings of this study are available from the corresponding author upon reasonable request.”
4. Data Not Available Due to Ethical, Legal, or Privacy Restrictions
Use when data cannot be shared due to participant confidentiality, legal agreements, proprietary issues, or other restrictions.
Statement Template:
“Restrictions apply to the availability of these data due to [ethical/legal/privacy/commercial] reasons. Data may be available from the corresponding author upon reasonable request.”
5. Data Not Available Due to Third-Party Restrictions
Use when the study is based on datasets obtained from an external source.
Statement Template:
“The data supporting this study were obtained from [third party name]. Access to these data requires permission from [third party name].”
6. Data Not Currently Accessible (Restricted Availability)
For datasets that exist but cannot be shared at the time of publication because of ongoing research, incomplete processing, technical limitations, or planned future release.
Statement Template:
“The datasets supporting this article are not currently available due to ongoing analyses. Requests for access may be directed to the corresponding author.”
7. Data Under Embargo
Use when data will become available after a specified period (e.g., commercialization, pending patent).
Statement Template:
“The data supporting the findings of this study will be made available in [repository name] at [URL/DOI] after an embargo period ending on [date].”
8. Data Sharing Not Applicable (No New Data Generated)
Used for studies that produce no original dataset. This applies to theoretical papers, conceptual analyses, or reviews based solely on existing literature.
Statement Template:
“No new data were created or analyzed in this study. Data sharing is not applicable.”
9. Data Derived from Public Domain Resources
Use when analysis is based on pre-existing publicly available datasets.
Statement Template:
“This study used publicly available data from [repository/resource] at [URL/DOI]. The dataset is accessible via reference number [number].”
Ethics Approval
Guidelines for ethical approval statements may differ based on the journal, a standard ethical approval statement will usually include:
1. Whether or not the study included human or animal subjects. In all cases, the ethical approval status of the work should be stated in the ethical approval statement.
2. The committee which approved the study.
3. The compliance documents. What policies, declarations, acts, etc.
4. Persistent identifier: reference or approval number. Include the registration ID/reference number if applicable.
5. “Not applicable.” for studies not involving humans or animals.
Conflicts of Interest
Declare conflicts of interest or state: “The author(s) declare(s) no conflicts of interest to report regarding the present study”.
● Supplementary Materials
In addition to the data, computer code, and research materials transparency guidelines, TSP encourages authors to provide supplementary materials that complement their main articles and enhance the readers' understanding of the research. Supplementary materials may include additional data, figures, tables, multimedia content, or relevant information.
1. Supplementary Materials Submission: Authors should submit supplementary materials along with their main article during the manuscript submission process. These materials should be in a separate section and clearly labeled as "Supplementary Materials.".
2. Content Relevance: All supplementary materials should be directly relevant to the main research article and provide valuable additional insights or data that support or expand upon the article's findings. Supplementary materials should not duplicate information already presented in the main text.
3. File Formats: Supplementary materials can be submitted in various formats, such as Word, PDF, Excel, CSV, images (JPEG or PNG), audio (MP3), video (MP4), or any other appropriate format for the content type.
4. Supplementary Data: Authors can provide raw data or additional data that support the article's findings but are not included in the main text due to space constraints. Data should be well-organized, properly labeled, and accompanied by clear explanations of the data's context and significance.
5. Supplementary Figures and Tables: Authors may include supplementary figures or tables to complement the main article, which should be clear, readable (with a minimum resolution of 300 dpi), and accompanied by accurate legends. These materials must be referenced in the main text using the prefix "S" (e.g., Fig. S1, Eq. (S2), Table S1) and should be submitted without tracked changes, highlights, comments, or line numbers.
● Appendices
The appendix is an optional section that can contain details and data supplemental to the main text. Authors that need to include an Appendix section should place it before the References section. Multiple appendices should all have headings in the style used for above. They should be ordered as such: A, B, and C, etc.
Appendix sections must be referenced in the main text. Within the appendices, figures, tables, and other elements should be labeled starting with “A”—e.g., Figure A1, Figure A2, etc.
● References
Tech Science Press (TSP) recommends editors and authors to utilize professional reference management tools such as EndNote for academic writing and literature formatting. EndNote is a reference management software from Clarivate Analytics. It is designed to manage bibliographies and references and available for Windows and MacOS. For authors, EndNote offers a convenient and efficient way to format their references according to a particular journal's guidelines. For journals and publishers, EndNote can save editorial time, and also shortens production time potentially.
All references should be cited in the main text sequentially (including citations in tables and legends) and listed individually at the end of the manuscript. We recommend preparing the references with a bibliography software package, such as EndNote, Mendeley or Zotero, to avoid typing mistakes and duplicated references. Include the digital object identifier (DOI) for all references where available.
For citations of references, please use square brackets and consecutive numbers, e.g., [1], [2,3], [4–6]. For embedded citations in the text with pagination, use both parentheses and brackets to indicate the reference number and page numbers; for example [5] (p. 10), or [6] (p. 101–105). When a cited reference is the subject of a sentence, use the author’s last name (e.g., Rhee [1]) or “Reference/Ref.” (e.g., Reference [1]). For multiple authors, use the first author followed by et al. (e.g., Al-Khshali et al. [2] or Refs. [4–6]). It is not recommended to cite more than 5 consecutive references. Please include the first 6 authors' names before using “et al.” in the references. Citations and references in the Supplementary Materials are permitted if they also appear in the reference list here.
The following are examples of the reference style (Vancouver style), which should be strictly adhered to (More details of style can refer to: Citing Medicine—NCBI Bookshelf):
● Journals
1. Author AA, Author BB. Title of article. Abbreviated Journal Name. Year;volume(issue):pagination.
2. Author AA Jr, Author BB 2nd, Author CC, Author DD, Author EE, Author FF, et al. Title of article. Abbreviated Journal Name. Year;volume(issue):pagination.
● Books
3. Author AA, Author BB. Title of the book. Publisher Location: Publisher; Year. Pagination (Optional).
4. Editor AA, Editor BB, editors. Title of the book. Publisher Location: Publisher; Year. Pagination (Optional).
5. Author AA, Author BB. Title of the book. xth ed. (if not first) Vol. x (if any). Publisher Location: Publisher; Year. Pagination (Optional).
6. Author AA, Author BB. Title of the book. xth ed. Translator AA, translator. Publisher Location: Publisher; Year. Pagination (Optional).
7. Author AA, Author BB. Title of the book. Publisher Location: Publisher; Year. Section/Table/Charts/… x; Pagination (Required).
8. Author AA, Author BB. Title of the chapter. In: Editor AA, Editor BB, editors. Title of the book. xth ed. (if not first) Publisher Location: Publisher; Year. Pagination (Required).
9. Author AA, Author BB. English Title of the book. Publisher Location: Publisher; Year. Pagination (In Original Language).
● Conferences
10. Editor AA, Editor BB, editors. Book title (Optional). Conference Title: (Proceedings of the) xth Name of Conference; Date of Conference; Location of the Conference (Optional). Publisher Location: Publisher; Year of publication. Pagination (Optional).
11. Author AA, Author BB. Title of the paper. In: Editor AA, Editor BB, editors. Book title (Optional). Conference Title: (Proceedings of the) xth Name of Conference; Date of Conference; Location of the Conference (Optional). Publisher Location: Publisher; Year of publication. Pagination (Required).
12. Author AA, Author BB. Title of the paper/Poster session. Paper/Poster session presented at: Name of the conference; Date of Conference; Location of the Conference (Optional).
● Dissertations and Theses
13. Author AA. Title of dissertation [dissertation/master’s thesis]. Location: Institution Name; Year of publication. Pagination (Optional).
● Web Sites
14. Author AA/Organization (Optional). Title of electronic publication/webpage [Internet/Video/…]. Location: Publisher (Optional); Date of publication (Optional) [cited 2024 Jan 1]. Available from: http://URL.
● Patents
15. Inventor AA, Inventor BB, inventors; Assignee AA, assignee. Title of the patent. Country of patent Patent number. Issue date/grant date.
Research Data and Supplementary Materials
In addition to the data, computer code, and research materials transparency guidelines, TSP encourages authors to provide supplementary materials that complement their main articles and enhance the readers' understanding of the research. These supplementary materials may include additional data, figures, tables, multimedia content, or relevant information.
1. Supplementary Materials Submission: Authors should submit supplementary materials along with their main article during the manuscript submission process. These materials should be in a separate section and clearly labeled as "Supplementary Materials."
2. Content Relevance: All supplementary materials should be directly relevant to the main research article and provide valuable additional insights or data that support or expand upon the article's findings. Supplementary materials should not duplicate information already presented in the main text.
3. File Formats: Supplementary materials can be submitted in various formats, such as Word, PDF, Excel, CSV, images (JPEG or PNG), audio (MP3), video (MP4), or any other appropriate format for the content type.
4. Supplementary Data: Authors can provide raw data or additional data that support the article's findings but are not included in the main text due to space constraints. Data should be well-organized, properly labeled, and accompanied by clear explanations of the data's context and significance.
5. Supplementary Figures and Tables: Authors may include extra figures or tables that complement those in the main article. These should be numbered separately (e.g., Supplementary Figure S1, Supplementary Table S1) and referred to in the main text.
Chemical Compounds
Chemical and Chemical Nomenclature and Abbreviations
Authors should provide the exact structure of the chemical compound, and if there are appeared as new chemical compounds, authors should submit the small-molecule crystallographic data to the Cambridge Structural Database (CSD) and deposit relevant information to PubChem. The final version of the manuscript should contain the accession codes. When possible, authors should use systematic nomenclature to identify chemical compounds, and biomolecules using IUPAC is preferred. Standard chemical and chemical abbreviations should be used.
Combinatorial Compound Libraries
The authors should include standard characterization data for a diverse panel of library components when describing the preparation of combinatorial libraries in the manuscript.
Chemical Structures for Organic and Organometallic Compounds
Chemical structures for organic and organometallic compounds should be established through spectroscopic analysis. The authors should provide standard peak listings for both 1H NMR and proton-decoupled 13C NMR for all new compounds. Other NMR data, when appropriate, such as 31P NMR, 19F NMR, etc. should be reported. For the identification of functional groups, both UV and IR spectral data should be reported when appropriate. For crystalline materials, melting-point ranges should be included. For the analysis of chiral compounds, specific rotations should be reported. For known compounds, authors should provide detailed references.
Spectral Data
Detailed spectral data for new compounds should be provided in the Materials and methods section. The authors should explain how specific, unambiguous NMR assignments were made in the Materials and methods section.
Crystallographic Data for Small Molecules
For crystallographic data for small molecules, authors should provide a standard crystallographic information file (CIF) and a structural figure with probability ellipsoids. The authors should check the CIF using the International Union of Crystallography (IUCr) checkCIF. For the structure, the structure factors must be included either in the main CIF or in a separate CIF. Crystallographic data for small molecules should be submitted to the Cambridge Crystallographic Data Centre (CCDC), and the accession number must be referenced in the manuscript.
Biomolecular Materials
Manuscripts reporting new biomolecular structures should contain a table summarizing structural and refinement statistics. If suitable, high-field NMR or X-ray crystallography may also be used. For new biopolymeric materials (e.g., oligosaccharides, peptides, nucleic acids, etc.), if it is not possible for structural analysis by NMR spectroscopic methods. Authors must provide evidence of the identity based on sequence (when appropriate) and mass spectral characterization.
Biological Constructs
Authors should provide sequencing or functional data that validates the identity of their biological constructs (plasmids, fusion proteins, site-directed mutants) upon request.
Polymers
For new materials, as well as 1H NMR and 13C NMR, the mass spectral analysis should be used to support the identification of molecular weight. Ideally, high-resolution mass spectral (HRMS) data are preferred.
Nanomaterials
The authors must provide a detailed characterization of both individual objects and bulk composition.
Data Sharing and Deposition
Data Sharing
Researchers share their data so that other researchers can replicate and build on their published claims. We encourage authors to share the data described and discussed in their articles. Research data can be uploaded to repositories and the access information provided in a published article or appended to the article in supplementary files. Any restrictions on the availability of research materials or information must be disclosed to the editors directly at the time of submission and in the submitted manuscript. Read more about TSP’s Data Sharing Policies.
Reporting Requirements for Research in the Life Sciences, Behavioral and Social Sciences, and Ecology, Evolution, and Environmental Sciences
Authors of research articles in the life sciences, behavioral and social sciences, and ecology, evolution, and environmental sciences may be required to make available details about aspects of experimental and analytical design to the editors and reviewers for the purpose of assessing the manuscript.
At Tech Science Press, we value open scientific exchange and believe in promoting transparency and reproducibility in research. To ensure consistency across TSP journals, authors are required to include a Data Availability Statement in all submissions. For examples of acceptable statements, please refer to the templates provided in the Availability of Data and Materials section above.
1. Data sharing may be inappropriate when ethical, legal, or privacy considerations arise. In such cases, authors must clearly outline any limitations in the Data Availability Statement during manuscript submission. It is the author's responsibility to ensure that any shared data adhere to the consent obtained from participants regarding the use of confidential information.
2. Data Repositories
Authors are strongly encouraged to deposit their research data in reputable and discipline-specific data repositories. Preferred data repositories should be recognized and indexed by organizations like DataCite, re3data, or other relevant repositories in their respective fields.
3. Data Citation
Research data should be cited in the main article to give proper credit and acknowledge the efforts of data creators. Authors must properly reference the deposited data in their reference list, including the dataset's persistent identifier (DOI, accession number, etc.).
Analytic Methods (Code) Transparency
For work where novel computer code was developed, authors should release it either by depositing it in a recognized, public repository such as GitHub or uploading it as supplementary information to the publication. The name, version, corporation and location information for all software used should be clearly indicated. Please include all the parameters used to run software/program analyses.
1. Code Availability
Authors are strongly encouraged to share the computer code and software used to generate results presented in their articles. The availability of code enhances research reproducibility and allows other researchers to build upon the work.
2. Code Repositories
Authors should deposit their code and software in well-established and reputable code repositories such as GitHub, GitLab, Bitbucket, or other relevant platforms. Providing a link to the code repository should be included in the Data Availability Statement.
3. Code Documentation
Authors must ensure that the deposited code is well-documented, readable, and easy to understand. Sufficient comments and explanations should be provided within the code to facilitate its usage by others.
4. Citation
Authors must provide a citation for the code in the article's reference list. Include the code's persistent identifier (e.g., DOI or URL) to facilitate proper acknowledgment and citation by other researchers.
Data Deposition and Suggested Repositories
Prior to manuscript submission, please choose the appropriate repository, below are recommended data repositories for your research:
Dataverse, Dryad, figshare, GigaScience, Mendeley Data, Zenodo
You may also visit DataCite, re3data to identify registered data repositories for your data sharing.
For journals with health research subjects, the deposition of sequence information to the community-endorsed, public repository is necessary. Accession numbers and other relevant, unique identifiers provided by the database should be included in the submitted manuscript.
DNA and RNA Sequences: GenBank, European Nucleotide Archive (ENA), DDBJ, Protein DataBank, UniProt
DNA Sequencing Data: Gene Expression Omnibus (GEO), ArrayExpress, NCBI Sequence Read Archive, ENA Sequence Versions Archive
New microarray: Gene Expression Omnibus (GEO), ArrayExpress
Genetic polymorphisms: dbSNP, dbVar
Linked genotype and phenotype data: dbGaP, European Genome-phenome Archive (EGA)
Protein sequences: UniProt
Flow cytometry: FlowRepository
Chemical Compound Screening and Assay Data: PubChem
Suggesting Reviewers
Authors are welcome and encouraged to suggest reviewers when they submit their manuscripts by using the submission system. Authors should make sure they are totally independent and without conflicts of interest in any way. When suggesting reviewers, the Corresponding Author must provide an institutional email address for each suggested reviewer.
English Editing Service
Clear and concise language enables both the journal editors and reviewers to concentrate on the scientific content of your manuscript. In order to facilitate a proper peer review process and ensure that submissions are judged exclusively on academic merit, Oncology Research strongly encourages authors to prepare the language of their manuscripts with the utmost care. The use of the recommended language polishing service on your manuscript does not indicate the acceptance of your manuscript for publication in Oncology Research.
If you are an author whose native language is not English—or have any concerns regarding the language quality of your manuscript—we recommend having your manuscript professionally edited by a qualified English-speaking researcher in your field prior to submission.
Tech Science Press offers paid language editing services, or you may choose to use an alternative service that provides a confirmation certificate.
Editorial Policies
Authorship and Contribution
Authorship
Tech Science Press follows the International Committee of Medical Journal Editors (ICMJE) guidelines regarding authorship and contributions. Authorship should be based on the following 4 criteria:
1. Substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work; AND
2. Drafting the work or reviewing it critically for important intellectual content; AND
3. Final approval of the version to be published; AND
4. Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
All those designated as authors should meet all four criteria for authorship, and those who do not meet all four criteria should be acknowledged in the acknowledgement section.
The corresponding author is the one individual who takes primary responsibility for communication with the journal during the manuscript submission, peer-review, and publication process. The corresponding author typically ensures that all the journal’s administrative requirements, such as providing details of authorship, ethics committee approval, clinical trial registration documentation, and disclosures of relationships and activities are properly completed and reported, although these duties may be delegated to one or more co-authors. The corresponding author should be available throughout the submission and peer-review process to respond to editorial queries in a timely way, and should be available after publication to respond to critiques of the work and cooperate with any requests from the journal for data or additional information should questions about the paper arise after publication.
When the work has to be conducted by a large multi-author group, it is advised that the list of authors be decided before the work starts and confirmed before the manuscript submission. All members of that group listed as authors should have met all the above four criteria for authorship with final approval of the manuscript, and should be able to take public responsibility for the work with full confidence in the accuracy and integrity of the work of all group authors. As such, they will be required as individuals to complete conflict-of-interest disclosure forms.
Submissions by any individual other than one of the listed authors will strictly not be considered. All authors will take responsibility for the content of the manuscript they submitted, and ensure they are familiar with the other authors individual contribution.
Non-author Contributor
Contributors who meet fewer than all 4 of the above criteria for authorship should not be listed as authors, but they should be acknowledged. Examples of activities that alone (without other contributions) do not qualify a contributor for authorship are acquisition of funding; general supervision of a research group or general administrative support; and writing assistance, technical editing, language editing, and proofreading. Those whose contributions do not justify authorship may be acknowledged individually or together as a group under a single heading (e.g. "Clinical Investigators" or "Participating Investigators"), and their contributions should be specified (e.g., "served as scientific advisors," "critically reviewed the study proposal," "collected data," "provided and cared for study patients," "participated in writing or technical editing of the manuscript").
Alteration to Authorship
Requests made for an authorship change after submission must be made to the editorial office with an explanation for the change, include the signature of all authors, and be submitted by the corresponding author.
TSP places significant importance on maintaining the integrity and transparency of authorship contributions, and TSP journals do not accept any requests to change the first author or corresponding author during any stage of manuscript processing. Any insistence on altering the first author or corresponding author will result in the rejection of the manuscript without further review or consideration.
Please note that if you have changed affiliation during the course of the research, your new affiliation could be acknowledged in a note. TSP does not normally take requests for changes to affiliations after the acceptance of manuscripts.
Authorship issues found after publication may result in a correction. If and when the authors are unable to resolve among themselves an authorship-related dispute, TSP may raise the issue with the authors’ institution(s) and abide by its/their guidelines.
Authorship Contribution Statement
The Author Contributions statement is mandatory for research articles, except for papers with a single author. It should represent all the authors and is to be included upon submission. All listed authors must have substantially contributed to the manuscript and have approved the final submitted version, which should include a description of each author’s specific work and contribution. We suggest the following format for the contribution statement:
“The authors confirm contribution to the paper as follows: Conceptualization, First-name Lastname1 and First-name Lastname2; methodology, First-name Lastname1; software, First-name Lastname1; validation, First-name Lastname1, First-name Lastname2 and First-name Lastname3; formal analysis, First-name Lastname1; investigation, First-name Lastname1; resources, First-name Lastname1; data curation, First-name Lastname1; writing—original draft preparation, First-name Lastname1; writing—review and editing, First-name Lastname1; visualization, First-name Lastname1; supervision, First-name Lastname1; project administration, First-name Lastname1; funding acquisition, First-name Lastname1. All authors reviewed the results and approved the final version of the manuscript”.
Please turn to the CRediT role descriptors—CRediT for the term explanation.
Artificial Intelligence Policy
TSP acknowledges the evolving impact of Generative Artificial Intelligence (GenAI), including tools such as ChatGPT and other large language models (LLMs), on scholarly publishing. In line with guidance issued by the STM Association and the Committee on Publication Ethics (COPE), we have established the following policy to promote transparency and uphold academic integrity.
1. Use of AI in Manuscript Preparation
Authors who employ GenAI tools during the preparation of their manuscripts—for tasks such as text generation, graphical output, data analysis, study design, or interpretation—are required to declare such usage upon submission. Specific details regarding the nature and scope of AI tool usage should be disclosed in the “Materials and Methods” section. Additionally, the tool’s name and version must be acknowledged in the “Acknowledgments” section.
Acknowledgment statement example:
“During the preparation of this manuscript, the authors utilized [tool name, version] for [brief description of use]. The authors have carefully reviewed and revised the output and accept full responsibility for all content.”
When AI tools are used solely for linguistic editing—such as grammar correction, spelling checks, punctuation, style improvements, or formatting adjustments—this falls outside the policy's requirements and does not need to be declared. This allowance applies only to editorial refinements and does not extend to generative content creation or authorship.
Authors remain fully accountable for the accuracy, originality, integrity, and ethical compliance of all manuscript content, including any output generated or supported by GenAI tools. All materials must conform to TSP’s Publication Ethics, including but not limited to, policies concerning plagiarism, data integrity, and image authenticity.
GenAI tools cannot be cited as references, as such tools do not constitute verifiable or accountable contributors to scholarly work.
TSP reserves the right to request clarification or supporting information and will make editorial decisions in accordance with TSP’s Editorial Policies and Terms and Conditions.
2. Authorship
GenAI tools and LLMs do not fulfill TSP’s Authorship Criteria and cannot be listed as authors.
3. Use of AI in Peer Review
Peer reviewers should not employ GenAI tools for the generation or structuring of review reports. Such use may compromise confidentiality, data protection, and proprietary information. Limited application of AI for improving the linguistic clarity of a review report (e.g., grammar or punctuation) may be permitted but must be disclosed upon submission.
Under no circumstances may reviewers upload manuscripts (in full or in part), images, tables, or related materials to any AI platform, since such actions breach TSP’s Privacy Policy. Violations of this policy will result in the rejection of the review report.
4. Use of AI in Editorial Decision-Making
The Editors (including Editors-in-Chief, Associate Editors, Guest Editors, or Editorial Board Members) are strictly prohibited from using GenAI tools in the editorial evaluation or decision-making process. Uploading any confidential manuscript materials to AI platforms constitutes a breach of TSP’s Privacy Policy.
TSP is committed to revisiting Artificial Intelligence Policy in alignment with emerging best practices from STM Association, COPE, and the wider academic community.
Editors and Journal Staff as Authors
In the circumstances where Editors or editorial staff of the journal submit their own studies to the journal, they shall not be involved in the reviewing process, and the review process must be made transparently and rigorously. Submissions authored by editors or editorial staff of the journal will be handled by another editor who has least COIs with the authors to minimize the bias.
Conflicts of Interest
Conflicts of interest (COIs, also referred to as “competing interests”) may indicate the potential to influence the validity or objectivity of research. Editors, authors, and reviewers may be involved into COIs, and Oncology Research considers it essential to identify and seek to mitigate them so as to ensure the integrity of its role in the dissemination and preservation of knowledge. Failure to declare competing interests may result in decline of a manuscript.
Authors must declare all potential conflicts of interest; if they have none to declare, they should state plainly, “The author(s) declare(s) no conflicts of interest to report regarding the present study”.
In order to limit COIs, all roles involved in the peer review process must identify and declare any personal circumstances or associations that may be perceived as having such influence and acknowledge all funding sources for the work. However, COI statements relating to public funding sources, such as government agencies and charitable or academic institutions, need not be supplied.
To be specific, Oncology Research defines a COI as any relationship that may have an impact on the authors, reviewers, or editors of a manuscript during the peer review process, on the making of editorial decisions, or generally on any stage in the path toward publication.
Thus, COIs may include (but not limited to):
Financial COIs
Stock or share ownership
Patent applications
Research grants
Consultancies
Royalties
Non-financial COIs
Affiliation with the same institution;
Personal relationships, e.g., between thesis advisers and their students, friends, family members, etc.;
Academic relationships, e.g., among co-authors, collaborators, or competitors;
Government employees;
Members of Oncology Research editorial board of a TSP journal.
COIs are not considered permanent; such relationships that have ended more than two years prior to the submission of a manuscript need not be identified as sources of potential conflict.
Authors
Oncology Research requires a declaration from all authors of a manuscript regarding any potential COIs that could be relevant to the integrity or reliability of the scientific and professional judgment presented therein, as well as that of otherwise unassociated studies in the same journal. Potential conflict, unless already declared, will be held in confidence while the paper is under review. If the article is accepted for publication, the potential conflict of interest will be included in the acknowledgments. If there is, in fact, no conflict of interest, the authors should state plainly.
Reviewers
Reviewers should declare any COIs when they are assigned a manuscript and disclose this information to the editor, who will then assess whether they should proceed with the review process.
Editors
Editors, including Editors-in-Chief, Associate Editors and Guest Editors should be aware of their own potential COIs. If the Editors have authored or coauthored the manuscripts submitted to Oncology Research, Editors might be perceived to be influenced by the relationship. Oncology Research expects the Editor(s) to declare any COIs or potential COIs.
Copyright and Licensing
Oncology Research publishes all articles under an open-access license, which means that the articles remain accessible to all without charge and without technical or legal barriers and that they can be reused with proper acknowledgment and citation. Financial support for the open access publication is provided by the authors’ institutions or by research funding agencies in the way of article processing charge (APC) once manuscripts have been accepted. More specifically, Oncology Research publishes articles under the Creative Commons Attribution (CC BY) License. Oncology Research is committed to open access publishing as a means to foster the exchange of research among scientists, especially across disciplines.
The copyright and other proprietary rights related to papers published by Oncology Research are retained by the authors. If the authors reproduce any text, figures, tables, or illustrations from the papers published by Oncology Research in their own future research, they must cite the originally published version. They are further asked to inform Oncology Research’s editorial office of any exceptional circumstances in this regard at the time of submission, for which exceptions may be granted at the discretion of the publisher.
Articles published in Oncology Research are likely to contain material republished with permission under a more restrictive license. When this situation arises, it should be indicated; it is the responsibility of the authors to seek permission for reuse from the copyright holder.
Repository Policy
Tech Science Press allows authors the use of the published version, accepted version and submitted version of an article for self-archiving and/or archiving in an institutional repository (on a non-profit server) without embargo time. The published source must be acknowledged. For articles, a link to the journal home page or articles' DOI must be set.
Corrections & Retractions
TSP journals will issue corrections, and/or retraction statements, when deemed proper.
Corrections
Oncology Research aims to publish every article online in its final form. Upon receiving the proofs of their accepted manuscripts, authors will have an opportunity to check for errors and oversights. Occasionally, a mistake is pointed out in a published article, necessitating the issuance of a correction statement. A correction is a statement rectifying an error or an omission, Authors or readers may submit such a statement either through the journal’s online manuscript submission system (https://www.techscience.com/or), or by sending an email, along with the submission ID, to the journal’s editorial office (oncologyresearch@techscience.com). A correction notice, published and linked to the corresponding article, is freely accessible to all readers.
When making corrections to the original articles, the original article both in PDF and XML versions are corrected and bi-directionally linked to and from the published amendment notice that details the original error. Any changes made to the original articles affect data in figures, tables or text, the amendment notice will reproduce the original data. If it is not possible to correct the original article in both PDF and XML versions, the article will remain unchanged but will contain links that direct to and from the published correction notice.
Author’s Correction: An Author’s Correction may be published to correct an important error(s) made by the author that affects the scientific integrity of the published article, the publication record, or the reputation of the authors or the journal. The Managing Editor of that manuscript will be responsible for handling the correction process.
Publisher’s Correction: A Publisher’s Correction may be published to correct an important error(s) made by the journal that affects the scientific integrity of the published article, the publication record, or the reputation of the authors or of the journal.
Retractions
A retraction is a notice that a previously published paper should no longer be regarded as part of the published literature. The primary purpose of a retraction is to ensure the integrity and completeness of scholarly records by withdrawing any manuscript which is found to contain infringements of professional ethical codes, major errors, or where its main conclusion is seriously undermined as a result of new evidence coming to light.
Violations of professional ethical codes include multiple submissions without proper citations or permission, redundant publications, fake claims of authorship, plagiarism, fraudulent use of data, etc. Major errors cover any or all miscalculations or experimental errors, intentionally or due to honest mistakes.
The retraction will be referred to the Editors-in-Chief, Associate Editors, and the Managing Editor who have handled the paper. Retracted articles will not be removed from the printed copies of the journal (e.g., from libraries) nor from the electronic archives. Their retracted status will be indicated as clearly as possible. Bibliographic information about the article will be retained to ensure the permanence and integrity of the published scientific record. When an article is retracted, in most of the cases, the original manuscript is corrected and is bi-directionally linked (to and from) the published retraction notice which details the original error. For the purpose of transparency, when corrections made to the original article affect any data, figures, tables or texts, the retraction notice will display the original data alongside the corrected version. When a correction is not possible, all existing versions of the article will remain unchanged but will contain the bi-directional links, to and from, the published retraction notice.
The notice of retraction is permanently linked to its corresponding retracted article and is freely available and accessible by all readers.
Articles may be retracted by their Author(s), by the Journal Editors, or by the Publisher, i.e., Tech Science Press. In all instances, the retraction should indicate the reason for the action as well as the entity behind the decision. A retraction made without the unanimous agreement of the authors is feasible and indicated as such.
Article Withdrawal
Article Withdrawal is only used for articles in press, which represent early versions of articles and sometimes contain errors, or may have been accidentally submitted twice. Occasionally, an article may contain infringements of professional ethical codes, such as multiple submissions, bogus claims of authorship, plagiarism, and fraudulent use of data or the like. Articles that include errors or are discovered to be accidental duplicates of other published article(s), or are determined to violate our publishing ethics guidelines in the view of the editors (such as multiple submissions, bogus claims of authorship, plagiarism, fraudulent use of data or the like), may be “Withdrawn” by the article author or the journal editor.
Removal of Published Content
Under special circumstances, TSP reserves the right to remove an article, book or other content from TSP’s website and submission system. Such action may be taken when:
There are evidence indicating that the published content is defamatory, infringes on intellectual property rights, privacy rights, other legal rights, or is plainly unlawful;
A court or government order requires removal of such content;
The content, if acted upon, would pose an immediate and serious risks to health. Removal may be temporary or permanent. A statement will be published explaining the decision behind the removal.
Addressing Post-publication Issues
TSP is fully committed to maintaining the integrity and completeness of the scientific record and recognizes its importance to researchers and the academic community at large. As such, TSP will thoroughly investigate concerns that are directly raised with us by authors and/or readers. Authors are strongly encouraged to address any raised issues. During our investigation, we may request original raw data, and consult with experts and other scholars in the field. Depending on the seriousness of the issues, the following outcomes may ensue:
A manuscript still under consideration may be rejected and returned to the author.
A published online article, depending on the nature and severity of the issues, may result in a correction notice or a retraction notice.
Issues deemed to be serious may prompt TSP to inform the authors’ institution and related affiliations.
Our actions are driven by our dedicated aim for transparent notification to our readers and unabated commitment to the integrity of the published record, and not by any motivation to sanction individuals or attribute responsibility to specific named individuals. We may refer readers to the institutional investigations’ reports if they are publicly available. While we are committed to addressing post-publications issues and correcting the record swiftly, investigations typically take some time to reach resolutions given the complexity of the discussions, the diligence in our process and the need to obtain original data and consult with experts. We will issue and regularly update relevant Editor’s Notes and/or Editor’s Expression of Concern as interim notifications to alert our readership of any of concerns with published material.
Appeals and Complaints
TSP’s appeal and complaint procedures pertain to grievances against editorial decisions, discontent with procedural inaccuracies (such as tardiness in manuscript handling), and complaint regarding publishing ethics.
Queries of appeal and complaint must be accompanied by comprehensive justifications, and authors are requested to submit appeals and complaints in writing to TSP Feedback Center at https://ijs.tspsubmission.com/user/feedback. The editorial office will provide a prompt response upon receipt of a formal appeal or complaint, and endeavor to resolve the matter within a reasonable time frame.
Concerns regarding ethical misconducts may also be reported to TSP’s Editorial Integrity Team (https://www.techscience.com/ndetail/contact). The Editorial Integrity team adheres to COPE Guidelines, and subsequently determines a suitable course of action, authorizing editorial offices to furnish the complainant with feedback.