 Open Access
Open Access
ARTICLE
Enhancing Chickpea (Cicer arietinum L.) Resilience to Salinity through Proline and NaCl Seed Priming
1 Department of Biology, Faculty of Science, University of Sarajevo, Zmaja od Bosne 35, Sarajevo, Bosnia and Herzegovina
2 Department of Food Technology, University North, Trg. Dr. Žarka Dolinara 1, Koprivnica, Croatia
* Corresponding Author: Dunja Šamec. Email:
(This article belongs to the Special Issue: Abiotic Stresses and Plant Defences in Climate Change)
Phyton-International Journal of Experimental Botany 2026, 95(2), 13 https://doi.org/10.32604/phyton.2026.072517
Received 28 August 2025; Accepted 31 December 2025; Issue published 28 February 2026
Abstract
Salinity is one of the major abiotic stresses limiting chickpea (Cicer arietinum L.) productivity, particularly in arid and semi-arid regions where soil salinization is intensifying. Developing cost-effective and practical strategies to enhance seedling establishment and early vigor under saline conditions is therefore essential. In this study, we compared two seed-priming agents—1 mM proline and 25 mM NaCl—under identical hydroponic conditions to elucidate tissue-specific responses to 25 mM NaCl stress. Proline priming significantly improved shoot length (by ~23%), total chlorophyll content (by ~19%), and ascorbate peroxidase (ASPOX) activity. In contrast, NaCl priming enhanced root biomass retention (by ~38%) and peroxidase (POD) activity under salinity stress. Both priming treatments induced higher proline accumulation and antioxidant capacity, though with tissue-specific effects: proline favored aboveground resilience, while NaCl strengthened root ionic and oxidative balance. These findings highlight the complementary nature of proline and NaCl priming and support the concept of stress “memory,” whereby plants acquire enhanced readiness to cope with salinity. Integrating such priming strategies into chickpea cultivation could contribute to improved yield stability and sustainability in saline agroecosystems.Keywords
Chickpea (Cicer arietinum) is the second most important legume used for food worldwide, providing a significant source of carbohydrates, proteins, unsaturated fatty acids, minerals, vitamins, dietary fibers, and a range of isoflavones [1,2,3]. However, its cultivation is limited by various abiotic stresses, with salinity being one of the major factors affecting its productivity. Chickpeas are commonly grown in semi-arid and arid regions where saline soils prevail, resulting in substantial yield losses [4]. These saline conditions disrupt water and nutrient uptake, leading to osmotic stress, ion toxicity, and oxidative damage in the plant. Global studies estimate that the annual yield loss of chickpea due to salinity ranges from 8% to 10%, significantly impacting both the economic viability and food security in affected regions [5]. Despite its sensitivity to salinity, chickpea remains an important crop for regions with limited freshwater availability, highlighting the need for strategies to enhance its tolerance to salt stress.
One promising strategy to mitigate salt stress is seed priming, a pre-sowing treatment that enhances seed vigor and prepares seedlings for stressful conditions. This technique involves hydrating seeds before they begin to germinate, exposing them to mild stress-inducing factors, which can be natural, synthetic, or a combination of both, during the early stages of development. As a result, the induced mild stress improves the plant’s ability to withstand future stress conditions [6]. The pre-conditioning process helps establish both short-term and long-term stress memory, allowing the plant better adaptation when encountering additional stress [7]. The stress applied during seed priming can trigger epigenetic changes in the seed, increasing resilience to future environmental challenges [8]. Moreover, seed priming is an environmentally friendly practice that activates signaling molecules within the plant, enhancing its natural ability to tolerate salt stress and assisting in recovery from salt-induced damage [9,10].
Among various priming agents, osmoprotectants such as proline have gained attention for their ability to enhance stress tolerance by stabilizing cellular structures, maintaining osmotic balance, and activating antioxidant defence systems. Proline, an amino acid that naturally accumulates under various abiotic stresses, including salinity, functions as an osmolyte by stabilizing proteins and cellular membranes, regulating osmotic balance, and scavenging free radicals. In addition, proline plays a key role in modulating antioxidant defence mechanisms and mitigating oxidative damage caused by salt stress [11]. Several studies have demonstrated that exogenous application of proline enhances seed germination, plant growth, and performance under saline conditions through improved stress tolerance mechanisms [10,12,13]. These effects are largely attributed to enhanced nutrient uptake, better water absorption, and more efficient biological nitrogen fixation.
Similarly, sodium chloride (NaCl) seed priming has emerged as an effective, low-cost, and simple method for improving plant salinity tolerance. This technique strengthens antioxidant defences by enhancing the activities of key enzymes such as catalase (CAT), ascorbate peroxidase (ASPOX), and superoxide dismutase (SOD), which are critical for mitigating oxidative damage under salt stress [14,15]. In legumes, NaCl priming has also been associated with elevated glutathione S-transferase (GST) activity, contributing to redox homeostasis and improved stress adaptation across different cultivars [16].
The present study investigates the physiological and biochemical responses of chickpea to salt stress as influenced by seed priming with proline and NaCl. Specifically, we assessed proline accumulation, antioxidant enzyme activities (peroxidase—POD, ascorbate peroxidase—ASPOX), total soluble protein content, biomass production, and morphological traits such as root and shoot length. By comparing the effects of these distinct priming agents, the study aims to elucidate mechanisms underlying improved stress adaptation and to evaluate the potential of proline- and NaCl-based priming as practical strategies for enhancing chickpea performance in saline environments.
Although numerous studies have assessed hydro- and chemopriming in legumes, few have systematically compared two mechanistically distinct priming agents, an osmoprotectant (proline) versus an ionic inducer (NaCl), under identical conditions while resolving tissue-specific responses (shoots vs. roots). Understanding how these agents differentially modulate early growth, pigments, osmolytes, proteins and antioxidant enzymes at the tissue level can reveal complementary mechanisms of salt tolerance in chickpea. Here we test the hypotheses that (i) proline priming preferentially enhances shoot growth and photosynthetic/antioxidant readiness, whereas (ii) NaCl priming pre-conditions roots for ionic and osmotic regulation. We compare 1 mM proline and 25 mM NaCl priming under the same cultivation and stress conditions to investigate tissue-specific adaptive mechanisms and their practical potential for sustainable chickpea production [17,18,19,20,21].
2.1 Plant Material and Priming Treatments
The experiment was carried out using chickpea seeds (Cicer arietinum L.) obtained from the producer EuroCompany99 d.o.o. Ljubuški. Seed priming was performed by soaking healthy, undamaged seeds in a solution containing 1 mM proline or 25 mM NaCl for 24 h at 4°C. The selected doses (1 mM proline; 25 mM NaCl) were identified in preliminary screening as sub-toxic yet physiologically effective and fall within optimal ranges reported for chickpea/legumes to elicit priming without impairing germination or seedling vigor [17,19]. After priming seeds were dried to the initial moisture content and stored one-week prior cultivation.
2.2 Experimental Design and Seed Germination
Before cultivation surface Seeds were surface-sterilized in 70% ethanol (60 s), rinsed, then diluted sodium hypochlorite (1:4 v/v, 10–15 min), followed by five sterile water rinses. This ensured sterility without visibly impairing germination; nonetheless, milder/shorter treatments will be evaluated to minimize potential seed-coat injury. Filter paper was placed in Petri dishes, and 45 seeds from each priming treatment were evenly distributed on top. The paper was moistened with sterile distilled water. Each Petri dish was sealed with Parafilm along the edges to maintain sterility and minimize moisture loss through evaporation. Non-primed seeds were used as control and germinated in the same manner. The Petri dishes were placed in a growth chamber under long-day photoperiod conditions (16 h light) at 23°C; 70% humidity. Illumination inside the growth chamber was provided by Osram Fluora 8 lamps (2000 lux). Germination was observed every 48 h for a period of 15 days, and seeds were considered germinated when the radicle length exceeded 2 mm.
After the seedlings reached 15 days, they were moved to hydroponics into Hoagland solution for 4 weeks. Salt stressed plants were growing in Hoagland supplemented with 25 mM NaCl, while the control group growing in only Hoagland solution (Fig. 1). Hydroponics-Seedlings were grown in Hoagland (pH 6.2 ± 0.1), aerated continuously and renewed every 7 days. Macronutrients (mM): 6 KNO3, 4 Ca(NO3)2, 1 NH4H2PO4, 2 MgSO4; micronutrients and Fe-EDTA per standard formulation. Conditions: 23 ± 1°C, 16 h photoperiod (~2000 lux) Salt stress was applied by supplementing Hoagland’s solution with 25 mM NaCl, a moderate salinity level frequently used in legume salt tolerance studies to induce physiological responses without causing irreversible damage.
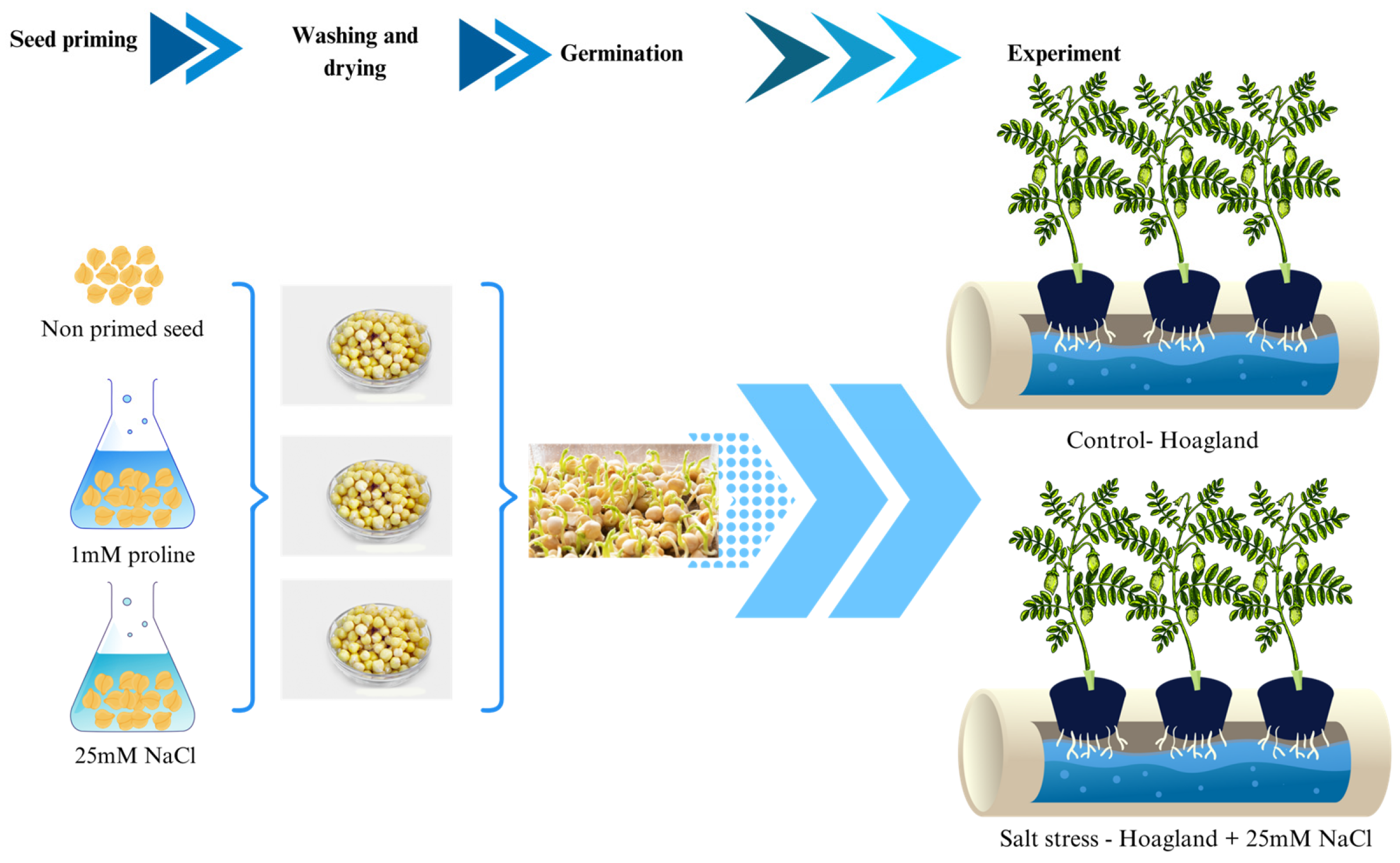
Figure 1: Scheme of the experimental setup.
After four weeks of growth and exposure to salt stress, root and shoot length of chickpea seedlings were measured to evaluate growth under control and salt stress conditions. Seedlings were carefully removed from hydroponics, placed on millimeter paper, photographed, and analyzed using ImageJ software (Image Processing and Analysis in Java; NIH, USA). Lengths were expressed in centimeters (cm) as the mean ± standard deviation of three biological replicates.
2.4 Analysis of Water Content (%)
Fresh weight (FW) and dry weight (DW) of shoots and roots were recorded. Dry weight was determined after drying the samples at 70°C for 72 h. Water content (%) was calculated using the formula:
Biomass production percentage was calculated relative to the corresponding control group:
E—fresh weight of experimental plants/stressed plants; C—fresh weight of control plants/non stressed plants.
In case of controlled conditions biomass % was calculated in relation to control where E is the fresh weight of experimental plants and C is the fresh weight of control plants. In case of salt stress exposure, biomass was calculated in relation to biomass produced under no stress conditions for each treatment, where E is the fresh weight of plants under salt stress and K is the fresh weight of plants grown under no stress of the same priming treatment.
2.5 Pigment Extraction and Quantification
Photosynthetic pigments were extracted from 0.1 g of fresh leaf tissue using 14 mL of 100% acetone. The homogenate was centrifuged for 20 min at 2100 rpm. Absorbance of the supernatant was measured at 661.6 nm (Chl a), 644.8 nm (Chl b), and 470 nm (carotenoids) using a UV–Vis spectrophotometer. Pigment concentrations were calculated according to Lichtenthaler [22].
In a cold mortar on ice, 0.25 g of plant material was homogenized with the addition of 1 mL of 0.1 M phosphate buffer (pH to 7). The mixture was transferred into an Eppendorf tube and centrifuged at ≈4700× g (5000 rpm) for 30 min at +4°C. The supernatant was used for subsequent analyses.
2.6.1 Analysis of Guaiacol Peroxidase (POD) Activity
Guaiacol peroxidase (POD) activity was determined according to a modified method of Angelini et al. [23] for plant extracts. The reaction mixture contained 0.1 M phosphate buffer (pH 7.0), 5.5 mM guaiacol, and 10 mM H2O2. Enzyme activity was measured spectrophotometrically at 436 nm every 30 s for 5 min. Results were calculated using the extinction coefficient of the guaiacol oxidation product tetraguaiacol (ε = 26.6 mM−1 cm−1) and expressed as enzyme units per milligram of protein. One unit of enzyme activity is defined as the amount of enzyme that catalyzes the oxidation of 1 μmol guaiacol per minute. The reaction was started by adding 10 μL of sample to 1 mL of reaction mixture.
2.6.2 Analysis of Ascorbate Peroxidase (ASPOX) Activity
The analysis of ascorbate peroxidase (ASPOX) activity was performed according to a modified method of Nakano and Asada [24], based on the decrease in absorbance at 290 nm over 1 min. The reaction mixture consisted of 0.2 M Tris-HCl buffer (1.55 g Tris-HCl dissolved in 40 mL H2O, adjusted to pH 7.8, and brought to a final volume of 50 mL), 0.25 mM ascorbic acid, and 0.5 mM H2O2. Enzyme activity measurement was initiated by adding 5 µL of the sample to 1 mL of the reaction mixture. ASPOX activity was calculated using the appropriate extinction coefficient (ε(290) = 2.8 mM−1 cm−1). One unit of ASPOX activity was defined as the amount of enzyme required to oxidize 1 μmol of ascorbic acid per minute under the assay conditions.
2.6.3 Determination of Total Protein Content
The total protein content was determined following the Bradford assay [25] by measuring absorbance at 595 nm using spectrophotometry, with bovine serum albumin (BSA) employed as the calibration standard. Protein content was expressed as mg g−1 fresh weight (mg g−1 FW) using a BSA calibration curve. The Bradford assay is based on the binding of Coomassie Brilliant Blue G-250 dye to proteins. Upon interaction, the dye undergoes a spectral shift from red to blue, which is measured spectrophotometrically at 595 nm. Protein amounts were calculated using the calibration factor derived from the BSA standard curve.
2.7 Determination of Proline Content
Determination of proline content in the sample was performed according to a modified method of Carillo et al. [26]. For proline quantification, 500 μL of 80% ethanolic extract was mixed with 1000 μL of the reaction mixture (1% ninhydrin in 60% acetic acid and 20% ethanol). The sample was then incubated in a water bath at 95°C for 20 min. Absorbance was measured at 520 nm using a blank as reference.
Proline content was calculated using the following formula:
A calibration curve was prepared using L-proline standards (0–100 µg mL−1; R2 > 0.99) to convert absorbance to quantitative proline content., expressed as mg proline per gram of dry weight (mg g−1 DW).
Each parameter was measured in three biological replicates, each comprising three technical replicates. Data were analyzed by one-way ANOVA followed by the Newman–Keuls post hoc test (p < 0.05). Different letters within a row/column indicate significant differences. Summary statistics are reported as mean ± SD; where informative, fold-changes are cited in the Results.
The present study demonstrated that seed priming using proline and NaCl differ-entially improved the tolerance of chickpea seedlings to salt stress, as evidenced by enhanced biomass accumulation, water and chlorophyll content, antioxidant enzyme activity, proline accumulation, and redox balance. These responses collectively point towards improved osmotic adjustment, tissue tolerance, and oxidative stress manage-ment as central mechanisms facilitated by priming.
The coordinated improvements in biomass, pigment retention, osmolyte levels and antioxidant capacity indicate that priming triggers anticipatory physiological programs encompassing osmotic adjustment (compatible solutes, membrane stabilization), ion homeostasis (enhanced Na+ handling and vacuolar sequestration in roots), and elements of stress-memory that accelerate ROS-scavenging upon challenge [27]. In our system, proline priming predominantly supported shoot functions (chlorophyll, ASPOX), while NaCl priming strengthened root defenses (POD, protein retention), pointing to complementary, tissue-resolved pathways.
3.1 Effect of Salt Stress and Priming on Root and Shoot Length, Fresh Mass, and Biomass Production
To evaluate how seed priming modulates morphophysiological responses under salinity, we measured shoot and root length, fresh mass, and calculated biomass production under control (0 mM NaCl) and salt stress (25 mM NaCl) conditions across different priming treatments (Table 1; Fig. 2).
Under control conditions, seedlings primed with 1 mM proline exhibited the greatest shoot (12.61 cm) and root (14.83 cm) length, indicating a stimulatory effect of proline on early vegetative growth. This observation supports previous findings that osmopriming enhances seedling vigour through improved water uptake and growth uniformity in legumes [28]. Under salt stress, shoot and root elongation decreased across all treatments, consistent with earlier reports on salinity-induced growth inhibition in chickpea cultivars [29,30]. Notably, proline-primed seedlings maintained the highest shoot length (8.78 cm) under stress, suggesting partial mitigation of salt effects, while NaCl-primed seedlings preserved the greatest root length (3.90 cm), reflecting improved root adaptability under saline conditions.
Patterns of fresh mass followed similar trends. Proline-primed seedlings accumulated the highest shoot (0.500 mg) and root (0.468 mg) fresh mass under control conditions, likely due to enhanced metabolic activity and osmotic balance [31]. Under salinity, NaCl-primed seedlings exhibited superior root fresh mass (0.138 mg), in line with previous evidence that NaCl priming promotes root system resilience by modulating ion homeostasis and maintaining water status [19].
Biomass production percentage (%BM), calculated relative to non-primed controls under 0 mM NaCl, further highlighted these differential responses. Proline priming under control conditions resulted in a slight reduction in shoot biomass (−5.79%) but enhanced root biomass (+9.52%), indicating improved belowground development. In contrast, NaCl priming under non-stress conditions led to reduced biomass in both shoots (−37.68%) and roots (−42.85%), likely reflecting energy investment in osmotic adjustments and stress-preparatory mechanisms.

Figure 2: Representative phenotypes of chickpea seedlings from primed seeds under control and salt stress (1 cm scale bar). Panels: (A) Control, 0 mM; (B) Control, 25 mM NaCl; (C) Proline, 0 mM; (D) Proline, 25 mM; (E) NaCl, 0 mM; (F) NaCl, 25 mM.
Under salt stress (25 mM NaCl), all treatments exhibited reduced biomass. However, proline-primed plants showed the smallest reduction in shoot biomass (−46.15%), indicating improved protection of aboveground tissues. Conversely, NaCl priming provided superior protection of root biomass (−45.83%), compared to proline-primed (−67.39%) and non-primed controls (−73.81%). This suggests that NaCl priming preferentially enhances root-based stress adaptation, possibly by stabilizing ion transport and promoting Na+ sequestration into vacuoles, thereby maintaining cellular osmotic balance [19].
These patterns highlight the complementary physiological effects of the two priming strategies. Proline priming promotes early shoot growth and mitigates shoot biomass loss under stress, while NaCl priming activates root-localized defence mechanisms that help sustain root biomass and water acquisition in saline environments [32]. This differential action aligns with the concept of “priming memory,” whereby treated seeds acquire a heightened state of alertness, enabling faster and stronger activation of stress-responsive pathways upon exposure to stress [33,34]. The observed tissue-specific responses underscore the potential for combining priming agents to target both root and shoot resilience in chickpea under abiotic stress. Overall, these results demonstrate a complementary role of NaCl and proline priming, with the former enhancing root-based stress tolerance and the latter supporting aboveground growth under both control and saline conditions.
Table 1: Effect of Seed Priming on Shoot and Root Length, Fresh Mass, and Biomass Production in Chickpea under Salt Stress.
| Parameter | Salt Stress Level (mM) | Control | 1 mM Proline | 25 mM NaCl |
|---|---|---|---|---|
| Shoot Length | 0 | 10.96 ± 1.81a | 12.61 ± 0.47a | 10.28 ± 1.99a |
| Root Length | 0 | 13.51 ± 1.73a | 14.83 ± 1.13a | 10.94 ± 0.60b |
| Shoot FW | 0 | 0.69 ± 0.03a | 0.65 ± 0.01a | 0.43 ± 0.03c |
| Root FW | 0 | 0.42 ± 0.03a | 0.46 ± 0.02a | 0.24 ± 0.01c |
| % Change in Shoot Biomass (vs. Control) | 0 | - | −5.79 ± 1.55a | −37.68 ± 2.22b |
| % Change in Root Biomass (vs. Control) | 0 | - | 9.52 ± 3.32a | −42.85 ± 4.21b |
| Shoot Length | 25 | 7.14 ± 0.17b | 8.78 ± 0.78a | 6.78 ± 0.69c |
| Root Length | 25 | 3.66 ± 0.44b | 2.41 ± 0.56c | 3.90 ± 0.98a |
| Shoot FW | 25 | 0.21 ± 0.01b | 0.35 ± 0.02a | 0.14 ± 0.01c |
| Root FW | 25 | 0.11 ± 0.01c | 0.15 ± 0.01a | 0.13 ± 0.01b |
| % Change in Shoot Biomass (vs. non stressed conditions) | 25 | −69.57 ± 2.51b | −46.15 ± 2.25a | −67.44 ± 3.44b |
| % Change in Root Biomass (vs. non stressed conditions) | 25 | −73.81 ± 3.54c | −67.39 ± 1.76b | −45.83 ± 4.12a |
3.2 Effect of Salt Stress and Priming on Photosynthetic Pigments
Chlorophyll content, including chlorophyll a (Chl a), chlorophyll b (Chl b), and total chlorophyll (Chl a + b), was significantly affected by both salt stress and priming treatments (Table 2). Under non-stress conditions (0 mM NaCl), proline-primed plants exhibited the highest levels of Chl a and total chlorophyll, exceeding values observed in both the non-primed control and NaCl-primed plants. In contrast, under 25 mM NaCl stress, a notable reduction in chlorophyll content was observed across all treatments, with proline-primed plants still maintaining relatively higher levels of Chl a and total chlorophyll compared to the NaCl-primed and control groups.
Relative to non-primed controls, proline priming increased shoot length by ~23% under control conditions, whereas NaCl priming increased root length by ~6% under salinity, illustrating tissue-specific benefits. Salt stress significantly impaired chlorophyll content in all treatments, corroborating previous findings that salinity disrupts chloroplast structure, affects enzyme activity involved in pigment biosynthesis, and enhances oxidative degradation of chlorophylls [35,36]. The decline in both Chl a and Chl b levels in control plants exposed to 25 mM NaCl confirms the sensitivity of the photosynthetic apparatus to ionic and osmotic stress.
However, the use of seed priming agents, especially proline, mitigated some of these adverse effects. Proline-primed plants retained higher total chlorophyll content under salt stress compared to other treatments. This finding aligns with studies by Farooq et al. [28] and Saini et al. [37], who demonstrated that priming enhances antioxidant defense, protects chloroplast membranes, and maintains pigment stability under stress conditions. The protective effect of proline may be attributed to its role as an osmoprotectant, stabilizing cellular structures and scavenging reactive oxygen species (ROS) [38,39].
Table 2: Effect of seed priming and salt stress on chlorophyll pigment composition in chickpea.
| Priming Treatment | Salt Stress Level (mM) | Chlorophyll a (mg g−1 FW−1) | Chlorophyll b (mg g−1 FW−1) | Total Chlorophyll (mg g−1 FW−1) |
|---|---|---|---|---|
| Control | 0 | 1.20 ± 0.02b | 0.64 ± 0.02a | 1.840 ± 0.03b |
| Control | 25 | 0.77 ± 0.02d | 0.60 ± 0.03b | 1.37 ± 0.05c |
| 25 mM NaCl | 0 | 0.86 ± 0.01c | 0.51 ± 0.01c | 1.37 ± 0.02c |
| 25 mM NaCl | 25 | 0.46 ± 0.02e | 0.34 ± 0.02d | 0.80 ± 0.02d |
| 1 mM Proline | 0 | 1.25 ± 0.01a | 0.60 ± 0.01b | 1.85 ± 0.03a |
| 1 mM Proline | 25 | 0.42 ± 0.01f | 0.31 ± 0.00d | 0.73 ± 0.01d |
Interestingly, NaCl priming under non-stress conditions led to a slight reduction in pigment levels compared to the control, suggesting that mild salt exposure during priming may affect pigment biosynthesis, possibly through hormetic effects. Under salt stress, however, NaCl-primed plants maintained better pigment retention than non-primed controls, confirming the stress-hardening effect of this priming method [40]. These results suggest that the improved pigment retention and photosynthetic potential in primed plants under salinity may be an early adaptive mechanism induced by seed priming. This priming-induced “memory” likely facilitates faster activation of defence responses and preservation of physiological integrity when plants are later exposed to environmental stressors [28,37].
3.3 Effect of Seed Priming and Salt Stress on Proline Accumulation in Chickpea
Proline content in both shoot and root tissues of chickpea varied significantly across priming treatments and salt stress conditions (Table 3). In non-stressed plants (0 mM NaCl), shoot proline levels ranged from 10.88 ± 0.99 mg g−1 FW in proline-primed plants to 16.18 ± 0.13 mg g−1 FW in NaCl-primed plants, with control plants showing intermediate values. Under salt stress (25 mM NaCl), all treatments exhibited a marked increase in shoot proline content, with the highest accumulation observed in proline-primed seedlings, followed by NaCl-primed and control plants.
In root tissues, control seedlings under non-stress conditions showed the highest baseline proline levels. Salt stress led to a decrease in root proline content in non-primed plants. However, priming with NaCl significantly enhanced root proline levels under salt stress, exceeding even the non-stressed control, while proline priming resulted in a moderate increase.
Proline accumulation is a well-documented physiological response to osmotic stress and plays a pivotal role in maintaining cellular homeostasis, osmotic adjustment, and protection against reactive oxygen species (ROS) [17,38]. In this study, both proline and NaCl priming substantially enhanced proline levels in chickpea tissues under salt stress, with distinct tissue-specific patterns. Proline-primed plants exhibited the highest shoot proline accumulation under stress, which is consistent with previous findings that exogenous proline enhances internal proline biosynthesis and activates associated signaling pathways [17,38].
This may contribute to improved shoot resilience by maintaining turgor and protecting the photosynthetic apparatus, as also observed through increased chlorophyll retention in primed plants. NaCl priming was more effective in enhancing root proline content, especially under salt stress, suggesting a preferential allocation or localized induction of proline biosynthesis in roots. This root-specific increase may be a key adaptive response for maintaining root hydraulic conductivity and facilitating ion compartmentalization [19]. The superior root proline content in NaCl-primed plants under salt stress supports the notion that mild ionic preconditioning via priming can enhance root stress tolerance mechanisms [40].
Table 3: Proline content (mg g−1 FW) in chickpea shoots and roots as affected by priming and salt stress.
| Priming Treatment | Salt Stress Level (mM) | Shoot | Root |
|---|---|---|---|
| Control | 0 | 12.36 ± 0.22d | 4.42 ± 0.13a |
| Control | 25 | 23.55 ± 0.03c | 2.87 ± 0.29c |
| 1 mM Proline | 0 | 10.88 ± 0.99d | 2.99 ± 0.07c |
| 1 mM Proline | 25 | 48.75 ± 7.27a | 3.65 ± 0.07b |
| 25 mM NaCl | 0 | 16.18 ± 0.13d | 3.20 ± 0.10b |
| 25 mM NaCl | 25 | 33.85 ± 0.89b | 4.50 ± 0.18a |
Interestingly, control plants under salt stress showed increased shoot proline but reduced root proline, indicating a stress-induced systemic response with limited root-level buffering capacity in the absence of priming. These findings reinforce the idea that priming not only enhances absolute proline levels but also modulates their spatial distribution within the plant, depending on the priming agent.
3.4 Effect of Priming on Protein, Peroxidase, Ascorbate Peroxidase Activity
Under 25 mM NaCl, NaCl priming elevated root POD activity by ~3.6-fold versus non-primed salt-stressed roots, while proline priming increased shoot ASPOX ~2.9-fold compared to controls. The activities of peroxidase, ascorbate peroxidase, and total protein content were significantly influenced by both seed priming and salinity levels in chickpea shoots and roots (Table 4).
Table 4: Effect of Priming and Salt Stress on Antioxidant Enzyme Activities (POD and ASPOX) and Protein Content in Chickpea Shoots and Roots.
| Priming Treatment | Salt Stress Level (mM) | POD Shoot (U mg−1 Protein) | POD Root (U mg−1 Protein) | ASPOX Shoot (U mg−1 Protein) | ASPOX Root (U mg−1 Protein) | Protein Shoot (mg g−1 FW) | Protein Root (mg g−1 FW) |
|---|---|---|---|---|---|---|---|
| Control | 0 | 336.04 ± 28.12a | 3971.14 ± 212.60bc | 298.21 ± 66.66d | 3513.61 ± 353.48cd | 9.07 ± 0.01a | 2.22 ± 0.01a |
| Control | 25 | 147.50 ± 15.29c | 2445.9 ± 320.87d | 1765.94 ± 113.11a | 5308.46 ± 146.21b | 4.50 ± 0.09e | 1.06 ± 0.06d |
| 1 mM Proline | 0 | 83.42 ± 12.69cd | 3481.99 ± 221.90c | 361.17 ± 70.96d | 3240.76 ± 150.74d | 6.46 ± 0.07b | 0.77 ± 0.06e |
| 1 mM Proline | 25 | 264.22 ± 24.33b | 6100.22 ± 162.79b | 858.36 ± 71.62c | 6205.33 ± 152.53a | 4.75 ± 0.06f | 0.66 ± 0.08f |
| 25 mM NaCl | 0 | 81.70 ± 14.00cd | 1729.96 ± 480.14e | 1287.99 ± 76.63b | 3888.41 ± 179.62c | 4.61 ± 0.04d | 1.29 ± 0.02c |
| 25 mM NaCl | 25 | 121.89 ± 10.10c | 8828.35 ± 270.63a | 888.66 ± 84.61c | 5745.31 ± 345.33b | 5.30 ± 0.07c | 2.10 ± 0.01b |
Under control conditions (0 mM NaCl), shoot POD activity was highest in non-primed plants, while all primed treatments showed lower levels. Under salt stress (25 mM NaCl), proline priming markedly enhanced shoot POD activity, nearly doubling the value observed in non-primed, salt-stressed plants. In roots, NaCl-primed plants under salt stress exhibited the highest POD activity, exceeding all other treatments.
ASPOX activity followed a similar trend. In shoots, the highest ASPOX activity occurred in salt-stressed, non-primed plants, with NaCl- and proline-primed, salt-stressed plants maintaining elevated values, significantly above the control. In roots, proline priming under salt stress led to the highest ASPOX activity, followed by NaCl priming. The lowest ASPOX levels were observed in non-primed roots under control conditions.
Total protein content declined under salt stress across all treatments. The highest shoot protein content was recorded in control plants, followed by proline-primed and NaCl-primed seedlings under non-stress conditions. In roots, salt-stressed NaCl-primed plants maintained relatively high protein levels, comparable to non-stressed controls, whereas proline-primed roots showed a sharper decline under salt stress.
Antioxidant enzymes such as POD and ASPOX play key roles in mitigating oxidative damage under abiotic stress, particularly salinity, by scavenging reactive oxygen species (ROS) such as H2O2 [33,37]. The present results show that both proline and NaCl seed priming significantly modulate antioxidant enzyme activities in chickpea seedlings, with distinct tissue-specific and treatment-specific effects.
Under salt stress (25 mM NaCl), NaCl priming resulted in the highest POD activity in roots (8828.35 ± 270.63 U mg−1 protein), indicating enhanced ROS detoxification in belowground tissues. This aligns with findings by Elradi et al. [32], where superior root antioxidant responses were linked to greater salt tolerance. The protective effect of NaCl priming likely reflects a “stress hardening” effect, enabling faster and stronger activation of defence responses under stress [35,40].
Similarly, proline priming enhanced both POD and ASPOX activities in roots and shoots, with particularly strong stimulation of ASPOX in roots. These results are consistent with prior reports [17,38], demonstrating that proline not only acts as a compatible solute under osmotic stress but also modulates antioxidant pathways. By supporting the ascorbate–glutathione cycle, proline increases ROS scavenging capacity and helps maintain cellular integrity, including membrane stability and photosynthetic efficiency [19,37].
Interestingly, non-primed salt-stressed plants showed a sharp increase in shoot ASPOX activit, likely reflecting an acute, compensatory response to unbuffered ROS accumulation. However, this response was insufficient to prevent reductions in root protein content, highlighting the physiological cost of uncontrolled stress.
Protein content trends further illustrate the protective role of priming. Under salt stress, NaCl-primed roots maintained high protein levels, comparable to control roots, indicating preserved metabolic function. In contrast, proline-primed roots showed lower protein content, suggesting that while proline enhances antioxidant defences, it may not fully sustain protein metabolism under prolonged stress. These observations align with the results of Rasool et al. [36] and Farooq et al. [34], who showed that salt stress triggers proteolysis unless antioxidant balance is tightly regulated.
Overall, these results point to a tissue-specific pattern: NaCl priming primarily enhances root resilience, promoting ion homeostasis and water uptake, while proline priming favours aboveground performance through improved antioxidant readiness [17,41]. This differentiation supports the concept of “priming memory,” where seeds develop lasting molecular and epigenetic adaptations that optimize stress responses [37].
Our findings support seed priming, particularly with NaCl or proline as an effective, low-cost strategy to improve salt stress tolerance in chickpea by strengthening antioxidant defences and preserving metabolic function. The complementary nature of the two treatments suggests that combined or sequential priming approaches may offer synergistic benefits, an avenue worth exploring in future applied research.
Recent studies have further highlighted that priming-induced enhancement of salt tolerance involves complex modulation of antioxidant pathways, ion transport, and osmotic adjustment mechanisms. For example Irshad et al. [42] demonstrated that NaCl priming boosts the glutathione–ascorbate cycle and photosystem II stability under salinity stress, which is in line with our findings of enhanced antioxidant enzyme activities in both shoots and roots. Similarly, Nasrallah [43] and Kaur [18] reported that proline priming improves salt tolerance in chickpea and other legumes through modulation of redox homeostasis and stabilization of key cellular structures, corroborating the tissue-specific enhancements we observed in our study. Furthermore, Liu et al. [27] showed that integrating priming with other stress management strategies can further amplify tolerance, suggesting that combining different priming agents or sequential treatments may represent a promising direction for future work. Our results align with these emerging insights, emphasizing that proline and NaCl priming promote complementary stress response mechanisms in chickpea by modulating both osmotic balance and antioxidant defences in a tissue-specific manner.
Recent studies corroborate priming-enhanced stress readiness across pathways: Wang et al. [21] showed small heat-shock proteins enhance tolerance via chaperone networks, and Ren et al. [20] demonstrated microbiome-mediated gains in nutrient/WUE under drought, reinforcing the systems-level nature of priming responses.
These comparisons further highlight the novelty of our work, which systematically contrasts two different priming agents under identical conditions and evaluates their distinct impacts on shoot- and root-based stress responses, an approach less commonly addressed in prior studies. From an agronomic perspective, root-focused hardening by NaCl priming (higher root POD and protein maintenance) would be expected to improve water acquisition and ion compartmentation during early establishment, whereas proline-driven shoot protection (chlorophyll retention, shoot biomass) should sustain photosynthetic start-up. Such root–shoot complementarity is consistent with improved stand establishment and yield stability under variable salinity; future field trials across genotypes should quantify these gains in farmers’ conditions.
Limitations and outlook—Our findings derive from a single genotype and single salinity level in hydroponics, which simplifies environmental noise but limits direct field extrapolation. Future work should test dose × duration, multiple genotypes, and soil-based/field conditions, including combined/sequential priming.
Proline and NaCl seed priming enhanced chickpea tolerance to moderate salinity through distinct yet complementary adjustments. Proline priming mainly protected shoot functions (chlorophyll, biomass, ASPOX), whereas NaCl priming reinforced root oxidative/ionic balance (POD, protein preservation). Quantitatively, proline improved shoot biomass and pigment stability, and NaCl increased root POD activity by ~3.6-fold under stress. These findings support combined or sequential priming to target both tissues. Next steps include multi-genotype, soil-based and field trials, optimization of dose × duration, and integration with breeding for salt-resilient chickpea ideotypes.
Acknowledgement:
Funding Statement: The authors received no specific funding for this study.
Author Contributions: The authors confirm contribution to the paper as follows: Conceptualization, Erna Karalija and Dunja Šamec; data curation, Sabina Dahija and Sajra Prijić; writing—original draft preparation, Erna Karalija; writing—review and editing, manuscript revision Dunja Šamec, Sabina Dahija and Sajra Prijić; visualization, Erna Karalija, Dunja Šamec. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: The authors confirm that the data supporting the findings of this study are available within the article.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Zia-Ul-Haq M , Iqbal S , Ahmad S , Imran M , Niaz A , Bhanger MI . Nutritional and compositional study of desi chickpea (Cicer arietinum L.) cultivars grown in Punjab, Pakistan. Food Chem. 2007; 105: 1357– 63. doi:10.1016/j.foodchem.2007.05.004. [Google Scholar] [CrossRef]
2. Jukanti AK , Gaur PM , Gowda CLL , Chibbar RN . Nutritional quality and health benefits of chickpea (Cicer arietinum L.): a review. Br J Nutr. 2012; 108( Suppl. 1): S11– S26. doi:10.1017/S0007114512000797. [Google Scholar] [CrossRef]
3. Xing Q , Dekker S , Kyriakopoulou K , Boom RM , Smid EJ , Schutyser MAI . Enhanced nutritional value of chickpea protein concentrate by dry separation and solid state fermentation. Innov Food Sci Emerg Technol. 2020; 59: 102269. doi:10.1016/j.ifset.2019.102269. [Google Scholar] [CrossRef]
4. Kheiri M , Kambouzia J , Deihimfard R , Yaghoubian I , Movahhed Moghaddam S . Response of rainfed chickpea yield to spatio-temporal variability in climate in the Northwest of Iran. Int J Plant Prod. 2021; 15: 499– 510. doi:10.1007/s42106-021-00153-5. [Google Scholar] [CrossRef]
5. Atieno J , Li Y , Langridge P , Dowling K , Brien C , Berger B , et al. Exploring genetic variation for salinity tolerance in chickpea using image-based phenotyping. Sci Rep. 2017; 7: 1300. doi:10.1038/s41598-017-01211-7. [Google Scholar] [CrossRef]
6. Paparella S , Araújo SS , Rossi G , Wijayasinghe M , Carbonera D , Balestrazzi A . Seed priming: state of the art and new perspectives. Plant Cell Rep. 2015; 34: 1281– 93. doi:10.1007/s00299-015-1784-y. [Google Scholar] [CrossRef]
7. Thakur A , Sharma KD , Siddique KHM , Nayyar H . Cold priming the chickpea seeds imparts reproductive cold tolerance by reprogramming the turnover of carbohydrates, osmo-protectants and redox components in leaves. Sci Hortic. 2020; 261: 108929. doi:10.1016/j.scienta.2019.108929. [Google Scholar] [CrossRef]
8. Marcos FC , Silveira NM , Marchiori PE , Machado EC , Souza GM , Landell MG , et al. Drought tolerance of sugarcane propagules is improved when origin material faces water deficit. PLoS One. 2018; 13: e0206716. doi:10.1371/journal.pone.0206716. [Google Scholar] [CrossRef]
9. Biswas S , Biswas AK , De B . Influence of sodium chloride on growth and metabolic reprogramming in nonprimed and haloprimed seedlings of blackgram (Vigna mungo L.). Protoplasma. 2020; 257: 1559– 83. doi:10.1007/s00709-020-01532-x. [Google Scholar] [CrossRef]
10. Guo X , Zhi W , Feng Y , Zhou G , Zhu G . Seed priming improved salt-stressed sorghum growth by enhancing antioxidative defense. PLoS One. 2022; 17: e0263036. doi:10.1371/journal.pone.0263036. [Google Scholar] [CrossRef]
11. Hosseinifard M , Stefaniak S , Ghorbani JM , Soltani E , Wojtyla L , Garnczarska M . Contribution of exogenous proline to abiotic stresses tolerance in plants: a review. Int J Mol Sci. 2022; 23: 5186. doi:10.3390/ijms23095186. [Google Scholar] [CrossRef]
12. Sharma S , Villamor JG , Verslues PE . Essential role of tissue-specific proline synthesis and catabolism in growth and redox balance at low water potential. Plant Physiol. 2011; 157: 292– 304. doi:10.1104/pp.111.183210. [Google Scholar] [CrossRef]
13. El Moukhtari A , Cabassa-Hourton C , Farissi M , Savouré A . How does proline treatment promote salt stress tolerance during crop plant development? Front Plant Sci. 2020; 11: 1127. doi:10.3389/fpls.2020.01127. [Google Scholar] [CrossRef]
14. Sen A , Puthur JT . Influence of different seed priming techniques on oxidative and antioxidative responses during the germination of Oryza sativa varieties. Physiol Mol Biol Plants. 2020; 26: 551– 65. doi:10.1007/s12298-019-00750-9. [Google Scholar] [CrossRef]
15. Zulfiqar F , Nafees M , Chen J , Darras A , Ferrante A , Hancock JT , et al. Chemical priming enhances plant tolerance to salt stress. Front Plant Sci. 2022; 13: 946922. doi:10.3389/fpls.2022.946922. [Google Scholar] [CrossRef]
16. Chatterjee P , Biswas S , Biswas AK . Sodium chloride primed seeds modulate glutathione metabolism in legume cultivars under NaCl stress. Am J Plant Physiol. 2018; 13: 8– 22. doi:10.3923/ajpp.2018.8.22. [Google Scholar] [CrossRef]
17. Dawood MG , Khater MA; El-Awadi ME . Physiological role of osmoregulators proline and glycinebetaine in increasing salinity tolerance of chickpea. Egypt J Chem. 2021; 64: 79. doi:10.21608/ejchem.2021.85725.4233. [Google Scholar] [CrossRef]
18. Kaur G , Sanwal SK , Sehrawat N , Kumar A , Sharma AK , Mann A . Morpho-physiological, biochemical, and transcript analysis revealed differential behavior of chickpea genotypes towards salinity. Appl Biochem Biotechnol. 2025; 197: 3668– 88. doi:10.1007/s12010-025-05192-6. [Google Scholar] [CrossRef]
19. Paul A , Mondal S , Mitra D , Chakraborty K , Biswas AK . Seed priming with NaCl boosted the glutathione-ascorbate pool to facilitate photosystem-II function and maintain starch in NaCl-primed chickpea under salt stress. Plant Physiol Biochem. 2025; 222: 109746. doi:10.1016/j.plaphy.2025.109746. [Google Scholar] [CrossRef]
20. Ren CG , Kong CC , Li SM , Wang XJ , Yu X , Wang YC , et al. Symbiotic microalgae and microbes: a new frontier in saline agriculture. Front Microbiol. 2025; 16: 1540274. doi:10.3389/fmicb.2025.1540274. [Google Scholar] [CrossRef]
21. Wang X , Ge J , He M , Li Q , Cai J , Zhou Q , et al. Enhancing crop resilience: understanding the role of drought priming in wheat stress response. Field Crops Res. 2023; 302: 109083. doi:10.1016/j.fcr.2023.109083. [Google Scholar] [CrossRef]
22. Lichtenthaler HK . Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. In: Methods enzymol. Vol. 148. New York, NY, USA: Academic Press; 1987. p. 350– 82. doi:10.1016/0076-6879(87)48036-1. [Google Scholar] [CrossRef]
23. Angelini R , Manes F , Federico R . Spatial and functional correlation between diamine-oxidase and peroxidase activities and their dependence upon deetiolation and wounding in chickpea stems. Planta. 1990; 182: 89– 96. doi:10.1007/BF00239989. [Google Scholar] [CrossRef]
24. Nakano Y , Asada K . Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 1981; 22: 867– 80. doi:10.1093/oxfordjournals.pcp.a076232. [Google Scholar] [CrossRef]
25. Bradford MM . A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976; 72: 248– 54. doi:10.1016/0003-2697(76)90527-3. [Google Scholar] [CrossRef]
26. Carillo P , Mastrolonardo G , Nacca F , Parisi D , Verlotta A , Fuggi A . Nitrogen metabolism in durum wheat under salinity: accumulation of proline and glycine betaine. Funct Plant Biol. 2008; 35: 412– 20. doi:10.1071/FP08108. [Google Scholar] [CrossRef]
27. Liu H , Able AJ , Able JA . Priming crops for the future: rewiring stress memory. Trends Plant Sci. 2022; 27: 699– 716. doi:10.1016/j.tplants.2021.11.015. [Google Scholar] [CrossRef]
28. Farooq M , Romdhane L , Al Sulti MK , Rehman A , Al-Busaidi WM , Lee DJ . Morphological, physiological and biochemical aspects of osmopriming-induced drought tolerance in lentil. J Agron Crop Sci. 2020; 206( 2): 176– 86. doi:10.1111/jac.12384. [Google Scholar] [CrossRef]
29. Ashraf M , Waheed A . Responses of some genetically diverse lines of chickpea (Cicer arietinum L.) to salt. Plant Soil. 1993; 154: 257– 66. doi:10.1007/BF00012531. [Google Scholar] [CrossRef]
30. Flowers TJ , Gaur PM , Gowda CL , Krishnamurthy L , Samineni S , Siddique KH , et al. Salt sensitivity in chickpea. Plant Cell Environ. 2010; 33: 490– 509. doi:10.1111/j.1365-3040.2009.02051.x. [Google Scholar] [CrossRef]
31. Nayyar H , Walia DP . Water stress induced proline accumulation in contrasting wheat genotypes as affected by calcium and abscisic acid. Biol Plant. 2003; 46: 275– 9. doi:10.1023/A:1022867030790. [Google Scholar] [CrossRef]
32. Elradi S , Suliman M , Zhou G , Nimir E , Nimir N , Zhu G , et al. Seed priming with β-aminobutyric acid alleviated salinity stress of chickpea at germination and early seedling growth. Chil J Agric Res. 2022; 82( 3): 426– 36. doi:10.4067/S0718-58392022000300426. [Google Scholar] [CrossRef]
33. Meloni DA , Oliva MA , Martinez CA , Cambraia J . Photosynthesis and activity of superoxide dismutase, peroxidase and glutathione reductase in cotton under salt stress. Environ Exp Bot. 2003; 49: 69– 76. doi:10.1016/S0098-8472(02)00058-8. [Google Scholar] [CrossRef]
34. Farooq M , Rehman A , Al-Alawi AKM , Al-Busaidi WM , Lee DJ . Integrated use of seed priming and biochar improves salt tolerance in cowpea. Sci Hortic. 2020; 272: 109507. doi:10.1016/j.scienta.2020.109507. [Google Scholar] [CrossRef]
35. Ceritoğlu M , Erman M . Mitigation of salinity stress on chickpea germination by salicylic acid priming. Uluslar Tarım Yaban Hayatı Bilim Derg. 2020; 6( 3): 582– 91. doi:10.24180/ijaws.774969. [Google Scholar] [CrossRef]
36. Rasool S , Ahmad A , Siddiqi TO , Ahmad P . Changes in growth, lipid peroxidation and some key antioxidant enzymes in chickpea genotypes under salt stress. Acta Physiol Plant. 2013; 35: 1039– 50. doi:10.1007/s11738-012-1142-4. [Google Scholar] [CrossRef]
37. Saini R , Das R , Adhikary A , Kumar R , Singh I , Nayyar H , et al. Drought priming induces chilling tolerance and improves reproductive functioning in chickpea (Cicer arietinum L.). Plant Cell Rep. 2022; 41( 10): 2005– 22. doi:10.1007/s00299-022-02905-7. [Google Scholar] [CrossRef]
38. Kaushal N , Gupta K , Bhandhari K , Kumar S , Thakur P , Nayyar H . Proline induces heat tolerance in chickpea (Cicer arietinum L.) plants by protecting vital enzymes of carbon and antioxidative metabolism. Physiol Mol Biol Plants. 2011; 17: 203– 13. doi:10.1007/s12298-011-0078-2. [Google Scholar] [CrossRef]
39. Verbruggen N , Hermans C . Proline accumulation in plants: a review. Amino Acids. 2008; 35: 753– 9. doi:10.1007/s00726-008-0061-6. [Google Scholar] [CrossRef]
40. Dadasoglu E , Turan M , Ekinci M , Argin S , Yildirim E . Alleviation mechanism of melatonin in chickpea (Cicer arietinum L.) under salt stress conditions. Horticulturae. 2022; 8( 11): 1066. doi:10.3390/horticulturae8111066. [Google Scholar] [CrossRef]
41. Aloui H , Mohamed Aymen E , Chérif H . Seed priming to improve seedling growth of pepper cultivars exposed to salt concentrations. Int J Veg Sci. 2017; 23( 6): 489– 507. doi:10.1080/19315260.2017.1326996. [Google Scholar] [CrossRef]
42. Irshad K , Siddiqui ZS , Chen J , Rao Y , Ansari HH , Wajid D , et al. Bio-priming with salt-tolerant endophytes improved crop tolerance to salt stress via modulating photosystem II and antioxidant activities in a sub-optimal environment. Front Plant Sci. 2023; 14: 1082480. doi:10.3389/fpls.2023.1082480. [Google Scholar] [CrossRef]
43. Nasrallah AK , Atia MA , Abd El-Maksoud RM , Kord MA , Fouad AS . Salt priming as a smart approach to mitigate salt stress in faba bean (Vicia faba L.). Plants. 2022; 11: 1610. doi:10.3390/plants11121610. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools