 Open Access
Open Access
ARTICLE
Humic Acid Biodegradable Film Increased Yam Yield and Total Soluble Sugar Content by Reshaping Soil Microbial Community Structure
1 State Key Laboratory of Vegetable Biobreeding, Institute of Vegetables and Flowers, Chinese Academy of Agricultural Sciences, Beijing, China
2 Wencheng Academy of Agriculture and Forestry Science, Wenzhou Vocational College of Science and Technology, Wenzhou, China
3 Southern Zhejiang Key Laboratory of Crop Breeding, Wenzhou Vocational College of Science and Technology, Wenzhou, China
4 College of Agriculture and Biotechnology, Wenzhou Vocational College of Science and Technology, Wenzhou, China
* Corresponding Authors: Yansu Li. Email: ; Yan Yan. Email:
# These authors contributed equally to this study
Phyton-International Journal of Experimental Botany 2026, 95(2), 4 https://doi.org/10.32604/phyton.2026.074706
Received 16 October 2025; Accepted 08 January 2026; Issue published 28 February 2026
Abstract
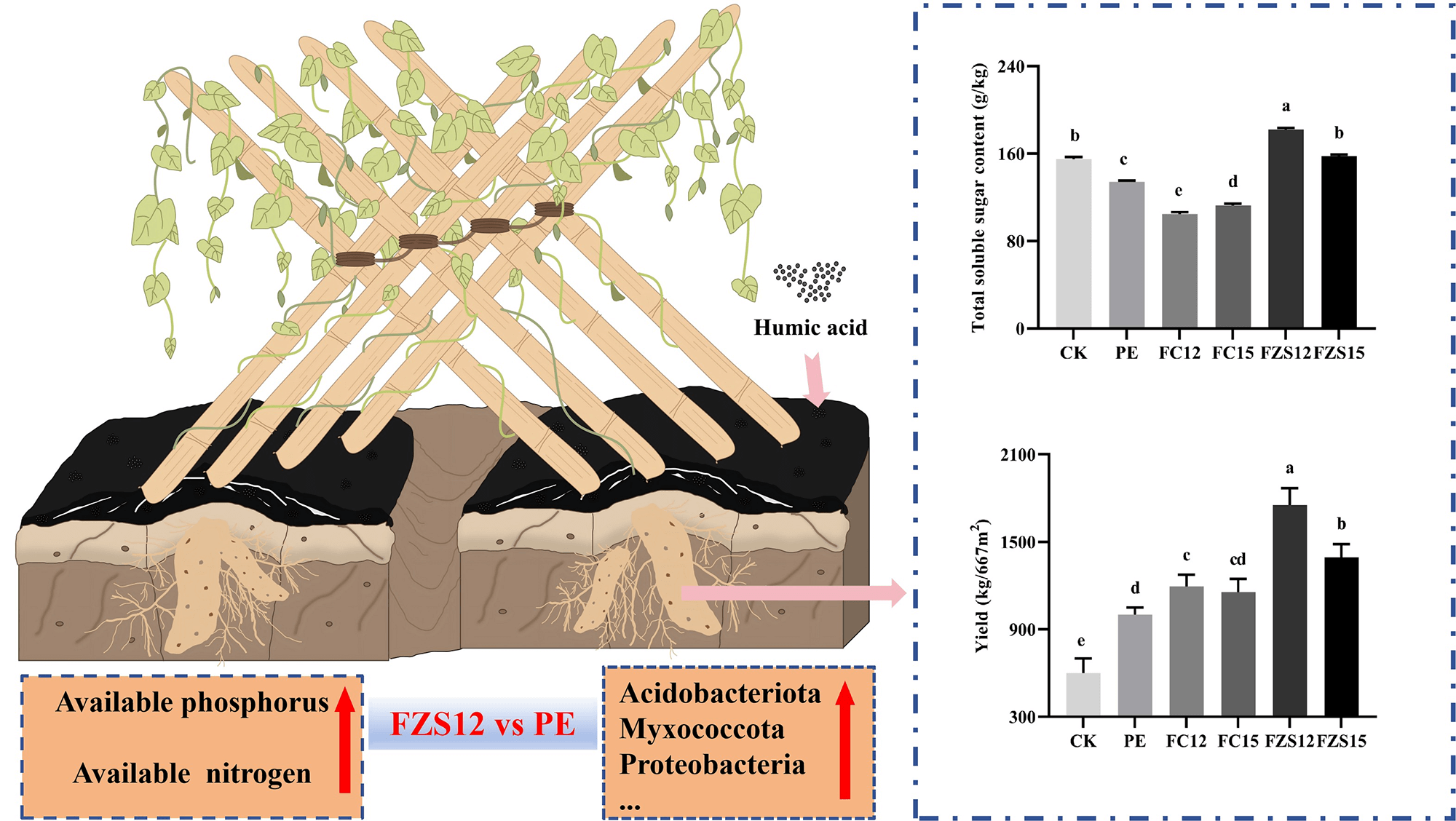
To address the issue of residual pollution caused by polyethylene mulch, this study explored the effects of different mulching methods on the soil environment of the yam field, as well as on yam yield and quality. The experiment comprised six treatments in total: one non-mulched treatment served as the control (CK), along with five different film-mulched treatments, namely PE, FZS12, FZS15, FC12, and FC15. The degradation of these films and their effects on soil physicochemical properties, microbial community, yam yield and quality were compared. The results showed that the FZS12 treatment achieved grade 5 degradation by the end of the planting period. Compared with PE treatment, the total soluble sugar content and yield of yam treated with FZS12 were significantly increased by 35.78% and 74.97%, respectively (p < 0.05). Compared with CK and PE treatments, FZS12 significantly increased soil available nitrogen by 31.62% and 6.20%, respectively (p < 0.05), and significantly increased soil available phosphorus by 8.58% and 4.45%, respectively (p < 0.05). Soil pH, available nitrogen, and available phosphorus were the main environmental factors affecting the soil bacterial community. The FZS12 treatment significantly increased the relative abundances of soil bacteria phylum including Acidobacteriota, Myxococcota, Patescibacteria, and Proteobacteria compared with the CK and PE treatments. Functional prediction using Picrust2 revealed that the FZS12 treatment had significantly higher levels of signal transduction and amino acid metabolism than the CK and PE treatments. In conclusion, covering with 12 μm PBAT/PLA humic acid biodegradable film enhances yam yield and total soluble sugar content by shaping beneficial soil microbial communities, activating soil nutrients.Graphic Abstract
Keywords
Yam (Dioscorea opposite Thunb.) is an annual or perennial twining vine plant belonging to the genus Dioscorea in the family Dioscoreaceae [1]. China is one of the countries with the richest yam resources in the world, accounting for 17% of the global yam tuber yield [2]. Glutinous yam is highly glutinous, with a soft, sticky, and sweet taste. It is rich in starch, polysaccharides, proteins, allantoin, saponins, vitamins, and trace elements [3,4], and has both edible and medicinal values. Wencheng glutinous rice yam, a Chinese national geographical indication agricultural product, has effectively driven the development of industries such as characteristic medicinal diets and the integration of agriculture and tourism.
Mulch film is a crucial agricultural production material, widely used in agricultural practices for its functions of heat and moisture retention, improving water and fertilizer use efficiency, enhancing crop quality, increasing yields, and ensuring the stable supply of agricultural products [5,6,7]. China’s mulch film usage and coverage area both account for approximately 70% of the world’s total. From 1993 to 2021, the usage of mulch film increased from 374,600 t to 1,320,300 t, and the coverage area increased from 5.7219 million hm2 to 17.2822 million hm2 [8]. Polyethylene, the primary component of traditional agricultural mulch films, is a high-molecular-weight compound characterized by high stability and strong resistance to degradation in natural environments [9]. This has led to a year-by-year increase in residual agricultural mulch films, with the average residual amount reaching as high as 200 kg·hm−2 in some provinces [10], which has a serious impact on soil quality. Plastic film residue not only destroys the structure of farmland topsoil, but also reduces the permeability of topsoil [11]. Plastic film residue and its microplastics also affect the content and mobility of heavy metals in soil [12]. Microplastics derived from residual mulch films not only alter soil physicochemical properties [13] and soil microbial diversity [14], but also affect soil nutrient cycling, cause crop yield reduction [15], and endanger human health [16]. In addition, the accumulated residual film will also increase the risk of microplastic pollution in farmland [8]. The governance of mulch film residual pollution in China is difficult and costly, so promoting biodegradable film has become one of the important paths for mulch film pollution prevention and control.
Biodegradable films are primarily made from biodegradable materials such as PBAT, PLA, PHA, PHB, PCL, etc. Biodegradable films not only possess the functions of traditional polyethylene films-such as heat and moisture preservation, crop quality improvement and yield increase, and weed suppression [17], but also can be mineralized by microbes in natural environments, ultimately decomposing into small inorganic molecules like CO2 and H2O [18]. After degradation, they can also boost soil microbial activity [19]. Studies have shown that there is no significant difference in agronomic traits and quality between PBAT biodegradable mulch and polyethylene mulch in cabbage planting [20]. In addition, Compared with polyethylene film, covering PBAT/PLA lignin biodegradable film in greenhouse melon planting can significantly increase soluble sugar content and yield [21]. Covering PBAT/PLA humic acid biodegradable film in facility tomato planting can significantly increase, lycopene, soluble sugar content and tomato yield [6,7]. They can effectively reduce the pollution of residual films to soil and the environment in agricultural production, making them one of the important approaches for developing green and sustainable agriculture.
However, the choice of PBAT/PLA is mainly attributed to the following reasons: PBAT combines the characteristics of both PBA and PBT, offering good ductility and elongation at break, as well as favorable heat resistance and impact resistance, all while maintaining excellent biodegradability. Blending PLA with PBAT not only enhances the toughness of the mulch film material but also allows the hydrolysis products of PLA to catalyze the degradation of PBAT, facilitating the regulation of the material’s biodegradability [22].
Humic acid is a natural organic matter formed by the decomposition of animal and plant residues, which is widely found in soil, coal and water. Humic acid can improve soil, activate insoluble mineral elements, promote plant absorption of nutrients and growth and development, increase production and improve product quality [23,24,25]. Sodium nitrophenolate has been shown to stimulate vegetative growth, enhance photosynthetic capacity, improve plant tolerance to various abiotic stresses, and improve crop quality [26,27].
The purpose of this study was to compare the effects of polyethylene mulch and biodegradable film on yam yield, quality, soil nutrients and microbial community, in order to screen out biodegradable film products suitable for yam production, alleviate the residual pollution of mulch, and provide theoretical basis and technical support for green sustainable agricultural production. It is clear whether the coverage of biodegradable film can increase the beneficial flora by changing the soil microbial community structure, thereby improving the soil nutrient availability, and ultimately improving the yield and quality of yam, so as to fill the research deficiencies in this field.
2.1 Experimental Materials and Design
Initial physical and chemical properties of soil: pH: 5.40; available nitrogen: 251.79 mg/kg; available phosphorus: 131.43 mg/kg; organic matter: 29.12 g/kg.
The trial was conducted in Wencheng County, Wenzhou City, Zhejiang Province from May to November 2024 using the yam variety ‘Wennuo No.1’ as test material. A total of 6 treatments were set up: CK (no mulch film), PE (polyethylene mulch, thickness 10 μm), FZS12 (humic acid biodegradable film, thickness 12 μm), FZS15 (humic acid biodegradable film, thickness 15 μm), FC12 (humic acid + sodium nitrophenolate biodegradable film, thickness 12 μm), and FC15 (humic acid + sodium nitrophenolate biodegradable film, thickness 15 μm). The color of the film is black. Each treatment was repeated four times, with 14 plants planted in each replicate.
2.2 Measurement Items and Methods
After mulching, the degradation status of the mulch films was observed every 30 days, and the damage situation of the mulch films in each treatment (whether cracks or fractures appeared, and the degree of fragmentation) was recorded. The degradation grading index of mulching film refers to the standards set by Shen et al. [5].
Total starch content (containing resistant starch), and total soluble sugar: a Thermo Fisher (USA) multifunctional microplate reader (model: Multiskan GO) was used for quantitative analysis of the target substances. Each treatment was set with 3 replicates.
Ten yams were mixed and weighed for each replicate. Finally, the yield was calculated according to 800 plants/667 m2.
2.2.3 Physicochemical Properties and Enzyme Activity of Soil
At the end of the planting period, soil samples from the 0–20 cm plow layer were collected using the “S”—shaped sampling method, with 3 replicates for each treatment. pH value: the soil pH value was determined by the potentiometric method with reference to NY/T 1121.2-2006. Available nitrogen: the content of hydrolyzable nitrogen was determined by the alkali extraction-incubation-volumetric method with reference to LY/T 1228-2015. Available phosphorus: the content of available phosphorus was determined by the ammonium fluoride-hydrochloric acid extraction-molybdenum antimony anti-colorimetric method with reference to NY/T 1121.7-2014. Organic matter: they were determined by the potassium dichromate oxidation method with reference to NY/T 1121.6-2006.
2.2.4 Targeted Quantitative Determination of Sugar Content
The detection of sugar content by MetWare (http://www.metware.cn/) is based on the Agilent 8890-5977B platform. The specific procedure is as follows: Weigh 20 mg of powder into a centrifuge tube with a corresponding number. Add 500 μL of extraction solution (methanol: isopropanol: water = 3:3:2, v/v/v) to the sample, vortex for 3 min, and perform ultrasonic treatment in a 4°C water bath for 30 min. Centrifuge at 12,000 r/min at 4°C for 3 min, suction 50 μL of the supernatant, add 20 μL of internal standard solution with a concentration of 1000 μg/mL, and then dry the mixture by nitrogen blowing and lyophilization. Add 100 μL of methoxylamine pyridine (15 mg/mL), incubate at 37°C for 2 h, then add 100 μL of BSTFA and incubate at 37°C for 30 min to obtain the derivatized solution. Transfer 50 μL of the derivatized solution, dilute to 1 mL with n-hexane, filter through a 0.22 μm filter membrane, and store the filtrate in a brown sample vial for GC-MS analysis. The mass spectrometry scanning mode for sugar detection is selected ion monitoring (SIM). Data processing is performed using qualitative and quantitative software.
Soil samples from the 0–20 cm layer were collected using the “S”—shaped sampling method. The numbered samples were brought back to the laboratory and stored at −80°C. Each treatment was set with 3 replicates. Sequencing: After extracting the total DNA of the sample, PCR amplification was performed and the products were purified, quantified and homogenized to form a sequencing library. The constructed library was first subjected to library quality inspection, and the qualified library was sequenced with Illumina NovaSeq 6000. Sequencing was performed by Beijing Biomarker Technologies Co., Ltd. Primer information: F:ACTCCTACGGGAGGCAGCA; R:GGACTACHVGGGTWTCTAAT.
The data were processed by Microsoft Excel 2010 software, plotted by GraphPad Prism 9.5.1, and analyzed by DPS2005.12.26 for one-way ANOVA.
When the yam is harvested, the film degradation photo is shown in Fig. 1A. The FC12, FC15, FZS12, and FZS15 mulches took 60 d, 90 d, 60 d, and 90 d respectively to reach grade 1 degradation. At the time of yam harvest, the FC12, FC15, FZS12, and FZS15 mulches achieved grade 5, 4, 5 and 4 degradation respectively. The PE mulch showed no degradation throughout the entire growth period of yam (Fig. 1B).
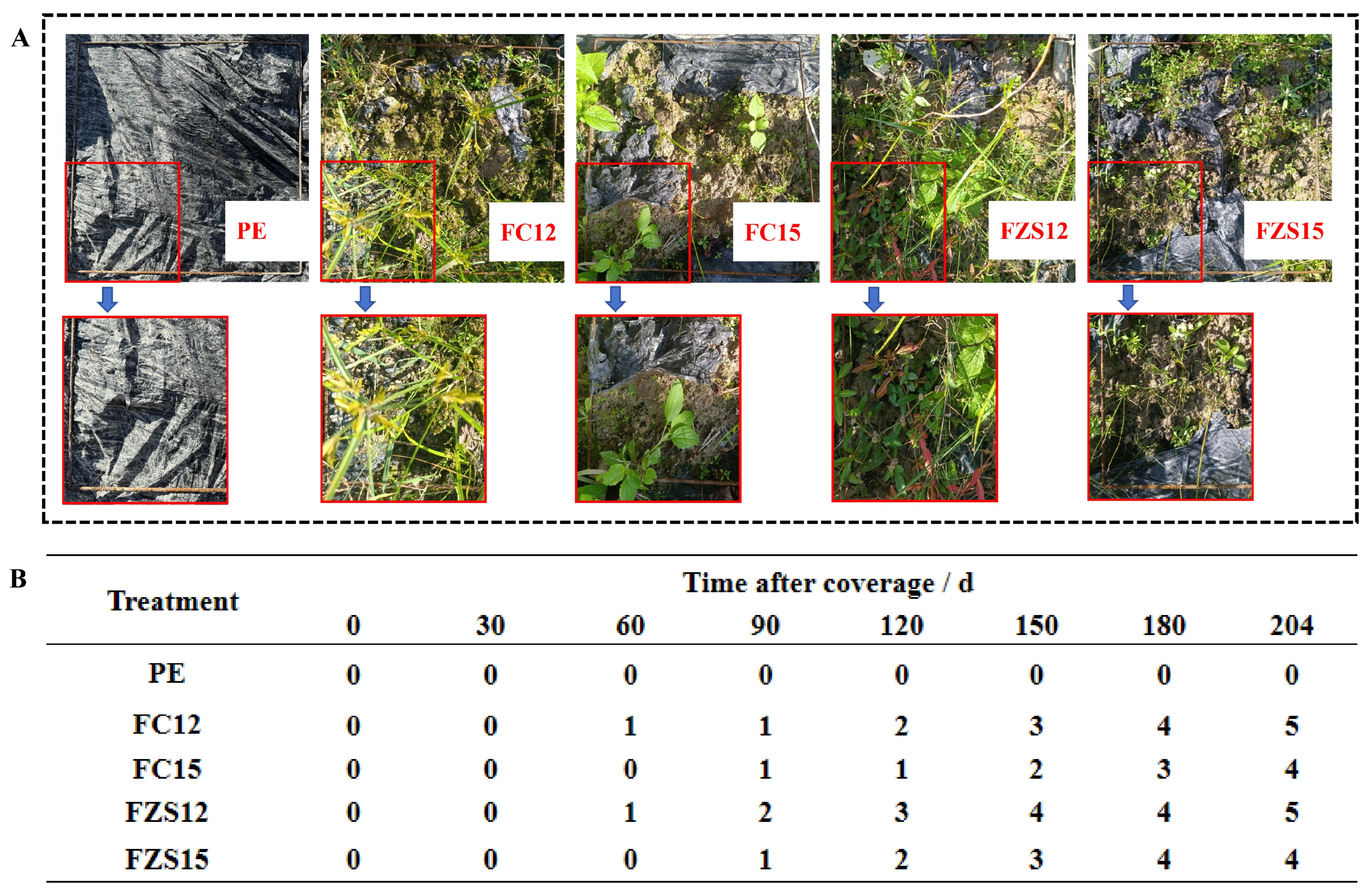
Figure 1: Mulch film degradation. Note: n = 3, (A) Degradation of plastic film in field; (B) Degradation classification of plastic film, 0–5 represents the classification index of film degradation. A greater value indicates a higher degree of film degradation.
The growth status of yam is shown in Fig. 2A. The total starch content of yam in the PE and FC12 treatments was significantly higher than that in other treatments (Fig. 2B) (p < 0.05). Compared with PE treatment, the total soluble sugar content and yield of yam treated with FZS12 were significantly increased by 35.78% and 74.97%, respectively (Fig. 2C,D) (p < 0.05).
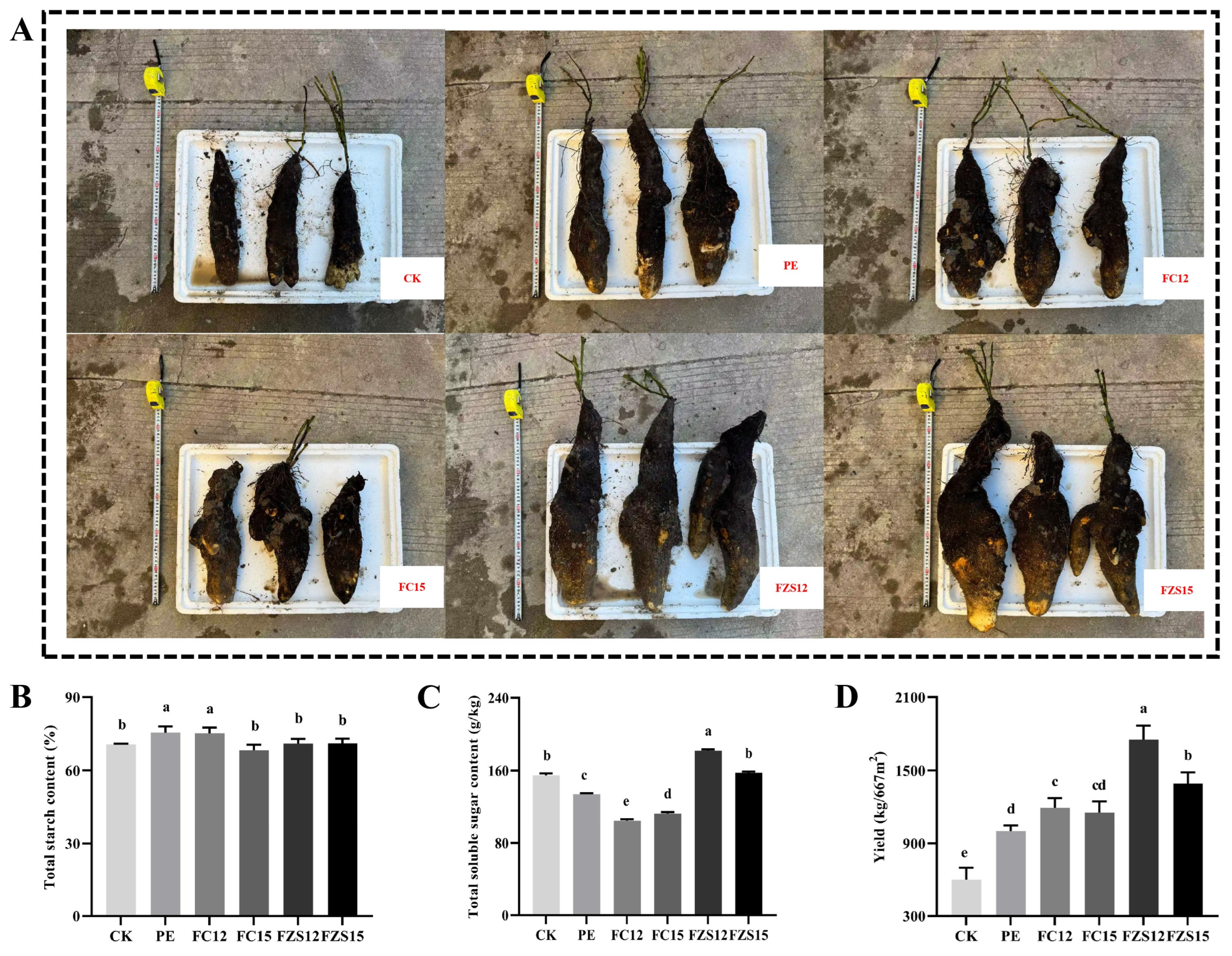
Figure 2: Effects of different treatments on yam yield and quality. Note: n = 3, (A) Yam pictures; (B) Total starch content; (C) Total soluble sugar content; (D) Yield. Different lowercase letters indicate significant differences between treatments (p < 0.05).
Combined with plastic film degradation, yam yield and quality. we selected the CK, PE, and FZS12 treatments for further analysis of yam sugar metabolism, soil physicochemical properties, and microbial community structure.
3.3 Targeted Quantitative Analysis of Sugar Content
A total of 22 sugar metabolites were detected in the CK, PE, and FZS12 treatments, including 19 monosaccharides and 3 disaccharides. The monosaccharides are: Barium D-ribose-5-phosphate, D-Mannose, L-Rhamnose, D-Ribono-1,4-lactone, D-Ribose, D-Glucuronic acid, D-Sorbitol, Xylitol, D-Xylose, Glucose, D-Galacturonic acid, D-Galactose, L-Fucose, D-Fructose, 2-Acetamido-2-deoxy-D-glucopyranose, D-Arabinose, D-Arabinitol, Levoglucosan, and Inositol. The disaccharides are: Sucrose, Phenylglucoside, and Trehalose (Fig. 3A). There were 1, 4, and 2 differential metabolites in the FZS12 vs. CK, FZS12 vs. PE, and PE vs. CK comparison groups, respectively, with 1 common differential metabolite among the three comparison groups (Fig. 3B). In the FZS12 vs. CK group, the significantly upregulated differential metabolite was D-Ribose; in the FZS12 vs. PE group, the significantly upregulated differential metabolites were D-Ribose, D-Xylose, Glucose, and D-Fructose; in the PE vs. CK group, the significantly downregulated differential metabolite was Glucose, and the upregulated one was D-Ribose (Fig. 3C).
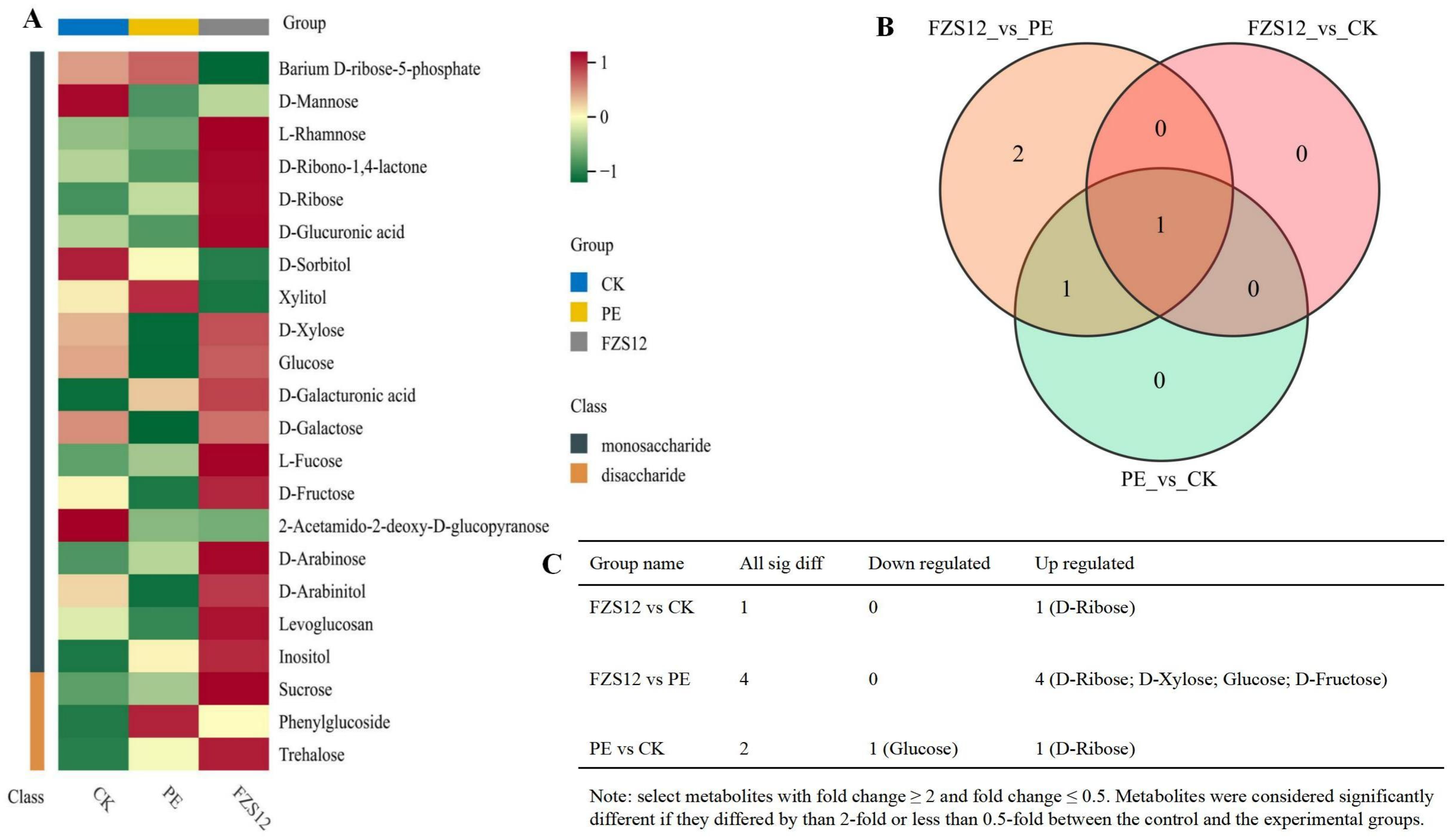
Figure 3: Targeted quantitative metabolomics study of sugars in yam. Note: n = 3, (A) Heatmap; (B) Differential metabolite venn map; (C) Statistical analysis of differential metabolites.
3.4 Soil pH, Nutrients and Microorganisms
Compared to CK treatment, the soil pH of PE and FZS12 treatments decreased significantly by 4.44% and 7.52%, respectively (Fig. 4A) (p < 0.05). Compared with CK and PE treatments, FZS12 significantly increased soil available nitrogen by 31.62% and 6.20%, respectively (Fig. 4B) (p < 0.05), and significantly increased soil available phosphorus by 8.58% and 4.45%, respectively (Fig. 4C) (p < 0.05). There was no significant difference in soil organic matter among the three treatments (Fig. 4D).
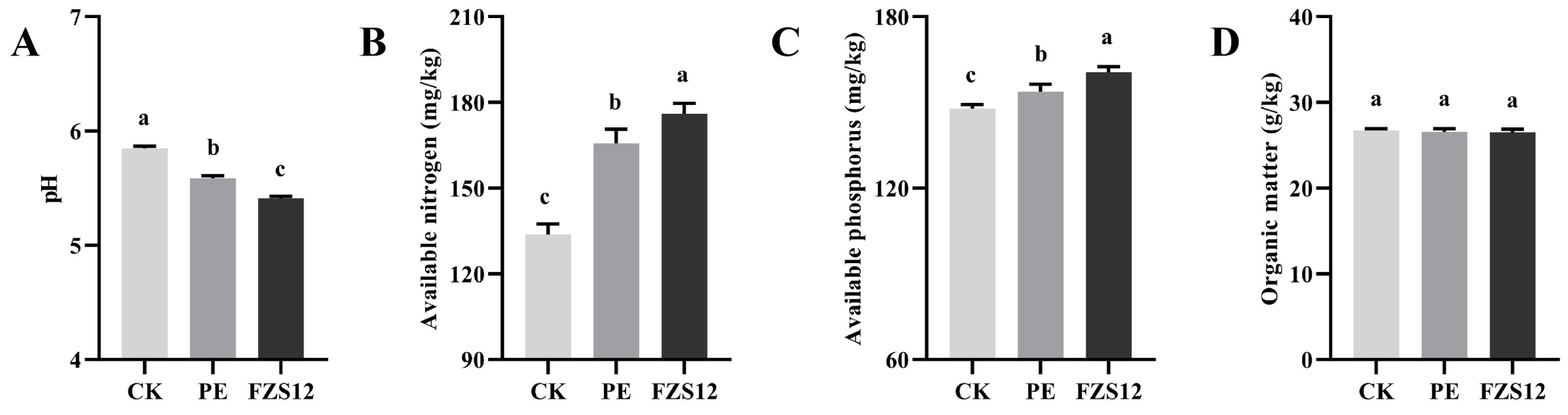
Figure 4: Effects of different treatments on soil pH and nutrients. Note: n = 3, (A) pH; (B) Available nitrogen; (C) Available phosphorus; (D) Organic matter. Different lowercase letters indicate significant differences between treatments (p < 0.05).
The soil bacterial Chao1 index in the FZS12 treatment was significantly higher than that in the CK and PE treatments, and the Shannon index was significantly higher than that in the PE treatment (Fig. 5A,B). The PCA analysis of soil fungal communities showed that the first and second axes explained 89.56% and 8.96% of the variance among samples, respectively. Soil samples from the CK, PE, and FZS12 treatments clustered separately, indicating small differences among replicates within each mulch treatment (Fig. 5C). The number of unique bacterial OTUs in the CK, PE, and FZS12 treatments was 3116, 2454, and 4980, respectively, with 115 shared bacterial OTUs among the three treatments (Fig. 5D). At the phylum level, the relative abundances of Acidobacteriota, Myxococcota, Patescibacteria, and Proteobacteria in the FZS12 treatment were significantly higher than those in the CK and PE treatments (Fig. 5E). At the genus level, the relative abundances of unclassified_Acidobacteriales and uncultured_forest_soil_bacterium in the FZS12 treatment were significantly higher than those in the CK and PE treatments (Fig. 5F). The taxonomic cladogram, under the screening criterion of an LDA threshold of 4, showed that at the genus level, the biomarker bacterial genera enriched in the CK treatment were unclassified_Bacteria, unclassified_Gemmatimonadaceae, and unclassified_Vicinamibacterales; those enriched in the PE treatment were Lactobacillus, Limosilactobacillus, unclassified_Muribaculaceae, Ligilactobacillus, Alistipes, Bacteroides, and Alloprevotella; and those enriched in the FZS12 treatment were uncultured_forest_soil_bacterium, Bordetella, Chujaibacter, unclassified_Acidobacteriales, and unclassified_Gaiellales (Fig. 5G).
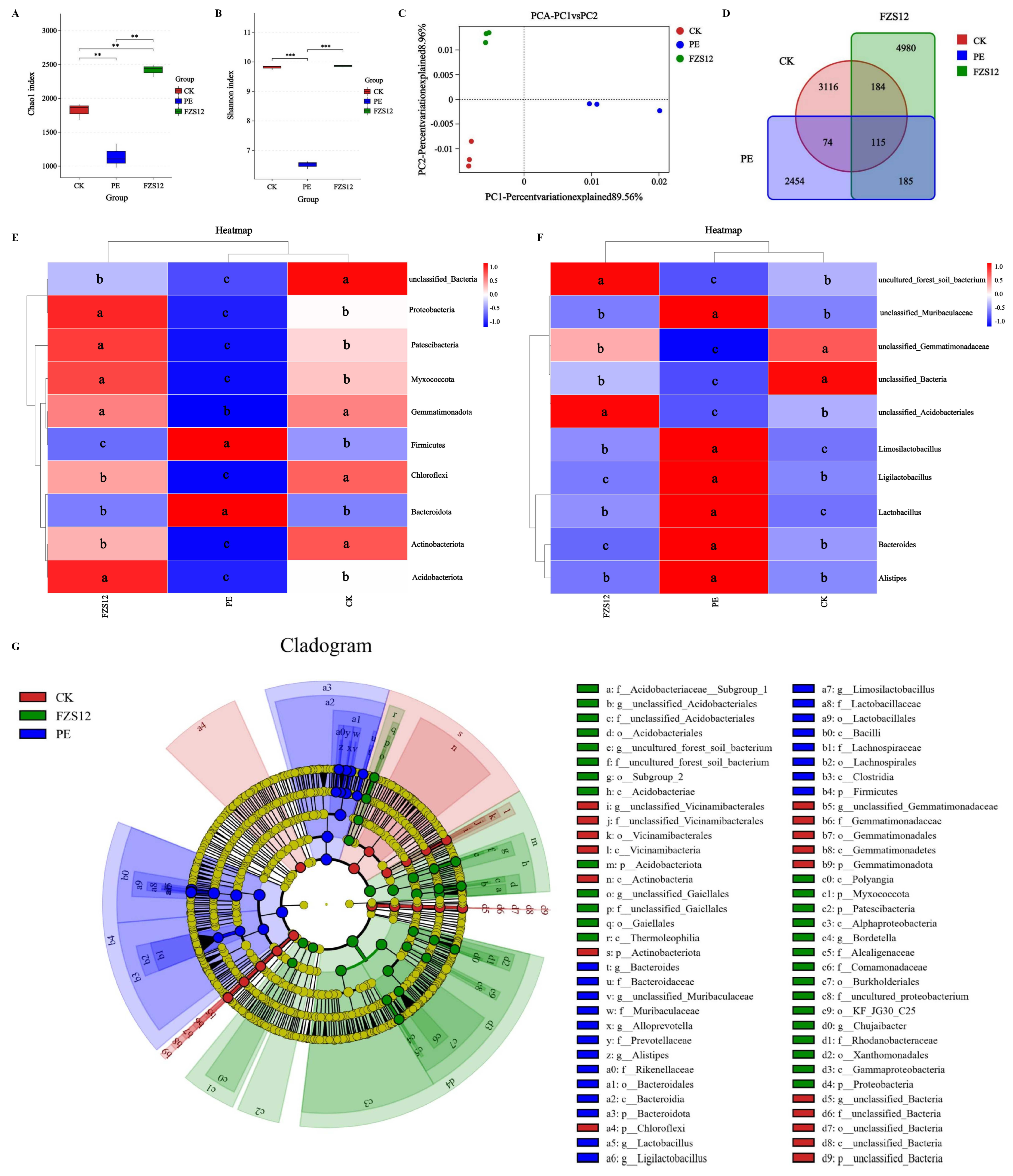
Figure 5: Soil bacterial community structure. Note: n = 3, (A) Chao1 index; (B) Shannon index; (C) PCA analysis; (D) Venn map of OTU level; (E) Species richness heatmap at phylum level; (F) Species abundance heatmap at genus level; (G) LEfSe analysis. **indicates p < 0.01, ***indicates p < 0.001. In Fig. 5E,F, different lowercase letters indicate significant differences between treatments (p < 0.05).
The Picrust2 functional prediction of soil bacteria showed that the functions of membrane transport, translation, carbohydrate metabolism, replication and repair, and nucleotide metabolism in the PE treatment were significantly higher than those in the CK treatment (Fig. 6A). The functions of signal transduction and amino acid metabolism in the FZS12 treatment were significantly higher than those in the CK treatment (Fig. 6B). Compared with the PE treatment, the FZS12 treatment had significantly higher functions in signal transduction, metabolism of cofactors and vitamins, energy metabolism, amino acid metabolism, and global and overview maps (Fig. 6C).
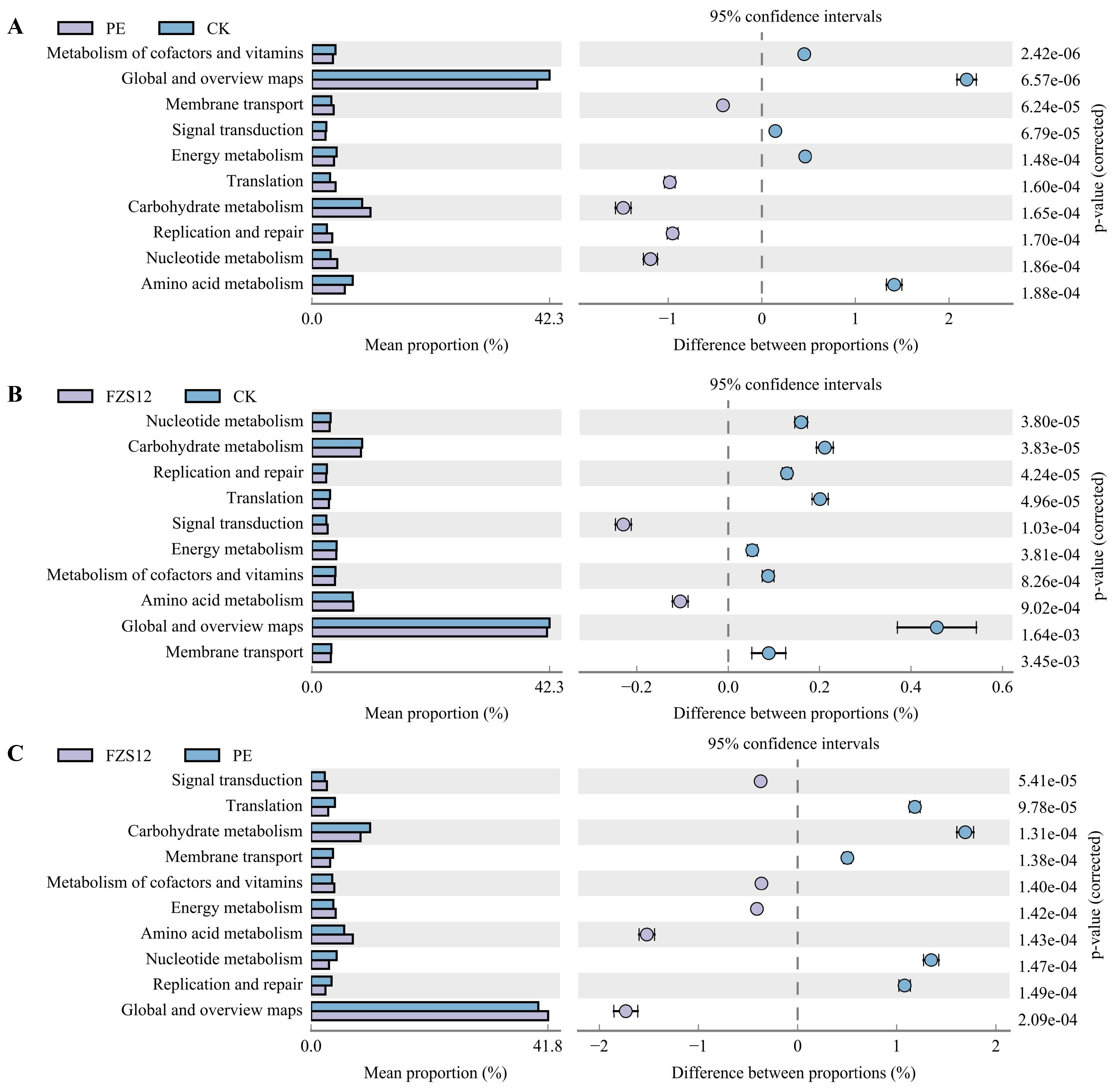
Figure 6: Functional prediction of Picrust2. Note: n = 3, (A) PE and CK; (B) FZS12 and CK; (C) FZS12 and PE.
Soil pH, available nitrogen, and available phosphorus are the main environmental factors affecting the bacterial community (Fig. 7A). There was no significant correlation between the top 10 soil bacterial genera and environmental factors (Fig. 7B).
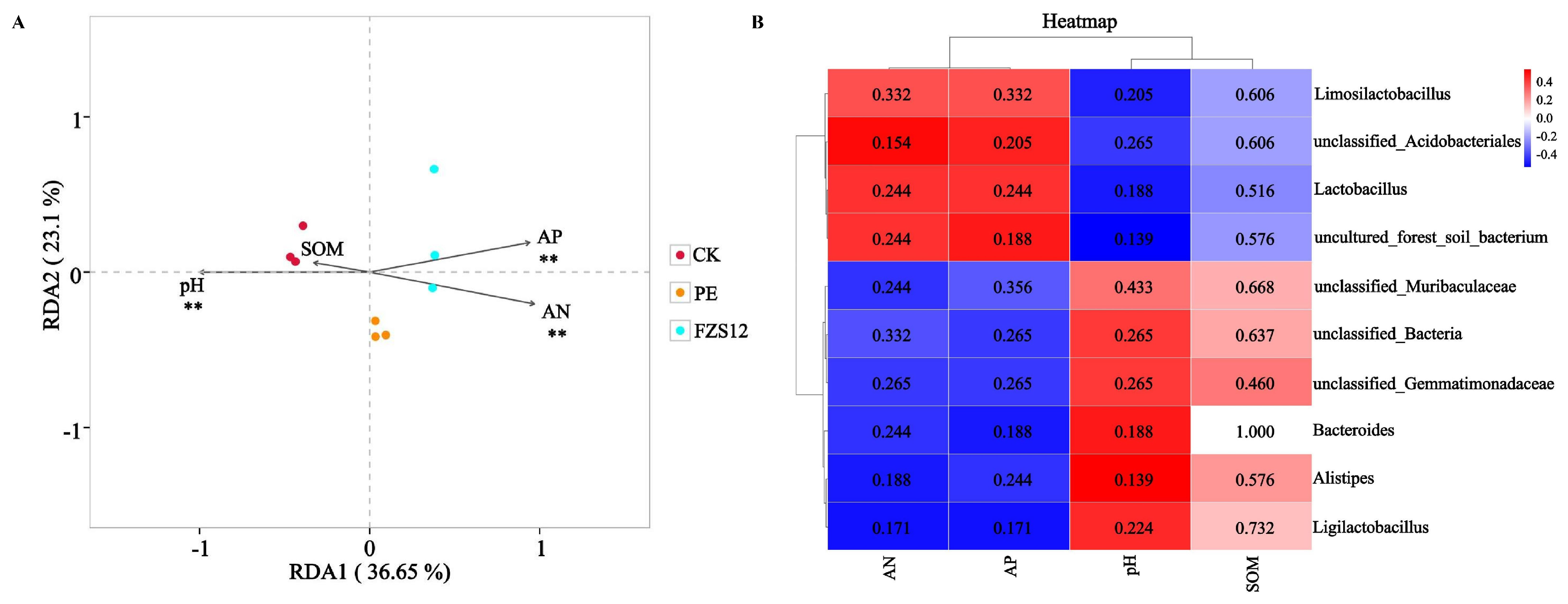
Figure 7: Correlation analysis. Note: n = 3, (A) RDA analysis; (B) Correlation heatmap. **indicates p < 0.01; the numbers shown in Fig. 7B are p values. AN: available nitrogen; AP: available phosphorus; pH: soil pH; SOM: soil organic matter.
The ‘white pollution’ of farmland has become an important problem restricting the green and sustainable development of yam industry. The fully biodegradable film is one of the important ways to solve the residual pollution of plastic film. However, in actual production, different kinds of biodegradable films showed different degrees of degradation, which is attributed to factors such as chemical composition, material thickness, environmental conditions, and others can influence the degradation rate and extent of biodegradable films [28]. The polyethylene mulch film showed no degradation because polyethylene compounds possess properties such as high molecular weight, stable performance, resistance to chemical erosion, and ability to buffer cold and heat. They are difficult to undergo photodegradation and thermal degradation under natural conditions, and are also not easily degraded by biological way such as bacteria and enzymes [29].
In this study, the FZS12 treatment significantly increased yam yield, which may be attributed to the following reasons: First, plastic mulching technology can effectively increase crop yield and improve crop quality. Second, the humic acid in FZS12 mulch can promote plant growth, increase crop yield and improve soil fertility [30]. Additionally, humic acid may contain organic biostimulants such as cytokinin enzymes, which can activate the physiological responses of plants. By affecting changes in the permeability of plant cell membranes, these biostimulants promote the synthesis of plant proteins and cell growth, thereby increasing yield [31]. The FZS12 treatment significantly increased the total soluble sugar content of yam, possibly because humic acid can stimulate plant growth, promote crops’ nutrient absorption, and improve quality [32]. Furthermore, previous studies have shown that covering with humic acid biodegradable film can effectively increase the soluble sugar content of tomatoes [6], which is consistent with the results of this study. Under the condition of high temperature and high humidity rainfall, covering polyethylene film can easily lead to the decrease of soil permeability, but the water vapor permeability of biodegradable film is higher than that of polyethylene film [21]. As a rhizome crop, the growth and development of yam is closely related to the soil temperature and humidity environment [33]. Under the high temperature and rainy weather conditions in Wencheng County in the summer of 2024, covering biodegradable mulch may help improve soil temperature and humidity conditions [6,21]. However, good soil permeability can promote the growth and development of the underground part of the crop [34], this may be one of the reasons for increasing production.
In the process of using humic acid biodegradable film, condensation under the film can precipitate humic acid, which helps to improve the yield and quality of yam. Humic acid fertilizer can increase soil organic matter by improving soil physical and chemical properties [35], and the change of soil environment will lead to the change of soil microbial community structure [36]. In this study, the FZS12 treatment increased the relative abundances of the soil bacteria phylum Acidobacteriota, Myxococcota, Patescibacteria, and Proteobacteria. The alteration in soil microbial community structure may be attributed to changes in soil temperature, humidity, enzyme activity, and nutrient content caused by covering with biodegradable film [6,37]. Previously, numerous studies have confirmed that biodegradable film mulching can alter soil physicochemical properties and soil microbial community structure [21]. In addition, humus can be used as a nutrient source for microorganisms, which can effectively improve the living environment of soil microorganisms, thereby promoting the growth of native microbial communities [38], and changing the diversity of soil microorganisms [39]. Secondly, humus can change the soil C/N ratio and create more favorable conditions for microbial growth [40]. For example, humic acid, as an organic macromolecular substance, can provide abundant carbon sources for soil microorganisms, thereby increasing the number of soil microorganisms [41].
The FZS12 treatment significantly reduced soil pH, because humic acid in the FZS12 mulch is an acidic substance that can lower soil pH through acid-base neutralization reactions. Meanwhile, humic acid can promote the activity of soil microorganisms. During their life activities, microorganisms continuously release CO2 into the surrounding environment and secrete some organic acids, thus leading to a decrease in soil pH [42]. The FZS12 treatment significantly increased the contents of soil available nitrogen and phosphorus. This may be due to the fact that: in the carbon and nitrogen cycle driven by soil microorganisms, different functional groups collaborate to maintain nutrient transformation and community stability in the ecosystem. Among them, microorganisms carrying the key gene cbbM of the Calvin cycle (such as Myxococcota) may play an important role in shaping microbial carbon assimilation [43]; meanwhile, phyla such as Acidobacteriota are mainly involved in the nitrogen cycle [44]. However, Proteobacteria are not only active in soil nitrogen and phosphorus cycles, but also play a key role in driving functional changes in eutrophic soils, and are crucial for maintaining the stability and function of soil microbial community structure [45,46]. Moreover, Proteobacteria plays an important role in decomposing organic matter, and soil nutrients cycling [47]. However, humic acid can reduce soil pH to a certain extent, which is beneficial to the effective transformation of phosphorus in the soil [42].
Other studies have shown that humic acid has the functions of adsorption, complexation and chelation, which can reduce the volatilization loss of soil nitrogen in the form of NH3-N and convert it into available nitrogen. At the same time, it can also complex with metal ions such as Al3+ and Fe3+ to reduce or avoid their fixation of phosphorus [41]. In addition, it can also convert insoluble P, K, medium and trace elements in soil into available nutrients and improve the availability of ineffective nutrients in soil [48]. In addition, humic acid fertilizer can stimulate the bacterial populations in Acidobacteria and Actinobacteria, which can decompose organic matter and regulate the effectiveness of mineral nutrients in soil, thereby enhancing the absorption and utilization of nutrients by plants [49]. Compared with conventional fertilization without organic fertilizer, 10% reduction of chemical fertilizer combined with humic acid fertilizer could increase the relative abundance of Proteobacteria [50]. This is consistent with the results of this study that covering humic acid biodegradable film can increase the relative abundance of Acidobacteria and Proteobacteria, and can increase the content of available nitrogen and available phosphorus in soil, which preliminarily verifies the role of humic acid in improving soil nutrient utilization.
Therefore, we speculate that in yam cultivation, this microbial reorganization enhances key ecological functions of the soil, especially by increasing the relative abundance of microorganisms such as Acidobacteria and Proteobacteria that participate in soil nitrogen and phosphorus cycling. This accelerates soil nutrient activation, thereby improving the availability of nutrients such as nitrogen and phosphorus to plants. Ultimately, through the synergistic effects of improving soil environment and enhancing nutrient supply efficiency, crop nutrient absorption was jointly driven, resulting in increased yam yield and total soluble sugar content.
Covering with FZS12 film can alleviate the “white pollution” caused by polyethylene mulch, increase the content of soil available nutrients, improve yam yield and quality, and alter the soil microbial community structure. These altered microorganisms can activate soil nutrients, thereby promoting yams’ absorption of nutrients —this may be the mechanism by which covering with FZS12 film improves yam yield and quality. Specifically, compared with the other two treatments (no mulching, CK; polyethylene mulch, PE), using an FZS12 film can significantly increase the relative abundance of bacterial phylum such as Acidobacteriota, Myxococcota, and Proteobacteria in the soil. This promotes the activation of soil nutrients, significantly increases the contents of soil available nitrogen and phosphorus, thereby significantly improving yam yield and total soluble sugar content.
Acknowledgement:
Funding Statement: This work was supported by the Wencheng County Science and Technology Plan Project (2023NKY03); Earmarked Fund for Modern Agro-industry Technology Research System (Grant Number CARS-24-B04; CARS-23-B05); Additional support was provided by Key Laboratory of Biology and Genetic Improvement of Horticultural Crops (Vegetables), Ministry of Agriculture and Rural Affairs, China.
Author Contributions: Study conception and design: Yan Yan, Yansu Li, Wenwu Zou; data collection: Lu Lu, Zewei Lu, Wenwu Zou, Jing Liu, Jiahao Pan; analysis and interpretation of results: Lu Lu, Zewei Lu, Kun Li, Mintao Sun, Jun Wang, Yansu Li, Yan Yan; draft manuscript preparation: Lu Lu, Yan Yan. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials: The datasets analyzed during the current study are available from the corresponding author on reasonable request.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflict of interest.
References
1. Luo GF , Podolyan A , Kidanemariam DB , Pilotti C , Houliston G , Sukal AC . A review of viruses infecting yam (Dioscorea spp.). Viruses. 2022; 14( 4): 662. doi:10.3390/v14040662. [Google Scholar] [CrossRef]
2. Zeng X , Liu D , Huang L . Metabolome profiling of eight Chinese yam (Dioscorea polystachya Turcz.) varieties reveals metabolite diversity and variety specific uses. Life. 2021; 11( 7): 687. doi:10.3390/life11070687. [Google Scholar] [CrossRef]
3. Chen Y , Zhou XZ , Ma LN , Lin YS , Huang XG . Chinese yam yield is affected by soil nutrient levels and interactions among N, P, and K fertilizers. Chin Herb Med. 2023; 15( 4): 588– 93. doi:10.1016/j.chmed.2022.11.006. [Google Scholar] [CrossRef]
4. Chen JN , Gao Q , Liu CJ , Li DJ , Liu CQ , Xue YL . Comparison of volatile components in 11 Chinese yam (Dioscorea spp.) varieties. Food Biosci. 2020; 34: 100531. doi:10.1016/j.fbio.2020.100531. [Google Scholar] [CrossRef]
5. Shen LX , Zhang YM , Lan YC , Li RF . Effects of degradable films with different degradation cycles on soil temperature, moisture and maize yield. Int J Agric Biol Eng. 2019; 12( 3): 36– 44. doi:10.25165/j.ijabe.20191203.4065. [Google Scholar] [CrossRef]
6. Han Y , Lu L , Wang L , Liu ZT , Huang PF , Chen SC , et al. In-situ straw return, combined with plastic film use, influences soil properties and tomato quality and yield in greenhouse conditions. Agric Commun. 2024; 2( 1): 100028. doi:10.1016/j.agrcom.2024.100028. [Google Scholar] [CrossRef]
7. Han Y , Duan Y , Niu YJ , Li YS , He CX , Sun MT , et al. Transcription metabolic mechanism of humic acid biodegradable plastic film to improving the fruit quality of tomato in the greenhouse. Acta Hortic Sin. 2024; 51( 8): 1758– 72. (In Chinese). doi:10.16420/j.issn.0513-353x.2024-0116. [Google Scholar] [CrossRef]
8. Yan CR , Hou JY , Xu YY , Hong ZJ , Ma ZG , Cui JX . Plastic mulch film application and management:experience from developed countries. J Agro-Environ Sci. 2024; 43( 06): 1288– 93. (In Chinese). doi:10.11654/jaes.2024-0068. [Google Scholar] [CrossRef]
9. Fu Q , Lai JL , Ji XH , Luo ZX , Wu G , Luo XG . Alterations of the rhizosphere soil microbial community composition and metabolite profiles of Zea mays by polyethylene-particles of different molecular weights. J Hazard Mater. 2022; 423( PA): 127062. doi:10.1016/j.jhazmat.2021.127062. [Google Scholar] [CrossRef]
10. Zhao Y , Chen XG , Wen HJ , Zheng X , Niu Q , Kang JM . Research status and prospect of control technology for residual plastic film pollution in farmland. Trans Chin Soc Agric Mach. 2017; 48( 06): 1– 14. (In Chinese). [Google Scholar]
11. Thidar M , Gong DZ , Mei XR , Gao LL , Li HR , Hao WP , et al. Mulching improved soil water, root distribution and yield of maize in the loess plateau of Northwest China. Agric Water Manag. 2020; 241( 1): 106263. doi:10.1016/j.agwat.2020.106340. [Google Scholar] [CrossRef]
12. Feng XY , Wang QL , Sun YH , Zhang SW , Wang FY . Microplastics change soil properties, heavy metal availability and bacterial community in a Pb-Zn-contaminated soil. J Hazard Mater. 2022; 424( PA): 127364. doi:10.1016/j.jhazmat.2021.127364. [Google Scholar] [CrossRef]
13. Kim SW , Liang Y , Zhao TT , Rillig MC . Indirect effects of microplastic-contaminated soils on adjacent soil layers: vertical changes in soil physical structure and water flow. Front Environ Sci. 2021; 9: 681934. doi:10.3389/fenvs.2021.681934. [Google Scholar] [CrossRef]
14. Chang N , Chen L , Wang N , Cui QL , Qiu TY , Zhao SL , et al. Unveiling the impacts of microplastic pollution on soil ecosystems: A comprehensive review. Sci Total Environ. 2024; 951: 175643. doi:10.1016/j.scitotenv.2024.175643. [Google Scholar] [CrossRef]
15. Hoang VH , Nguyen MK , Hoang TD , Ha MC , Huyen NTT , Bui VKH , et al. Sources, environmental fate, and impacts of microplastic contamination in agricultural soils: a comprehensive review. Sci Total Environ. 2024; 950: 175276. doi:10.1016/j.scitotenv.2024.175276. [Google Scholar] [CrossRef]
16. Zha H , Han SY , Tang RQ , Cao D , Chang K , Li LJ . Polylactic acid micro/nanoplastic-induced hepatotoxicity: Investigating food and air sources via multi-omics. Environ Sci Ecotech. 2024; 21: 100428. doi:10.1016/j.ese.2024.100428. [Google Scholar] [CrossRef]
17. Deng L , Yu Y , Zhang HY , Wang Q , Yu RD . The effects of biodegradable mulch film on the growth, yield, and water use efficiency of cotton and maize in an arid region. Sustainability. 2019; 11( 24): 7039. doi:10.3390/su11247039. [Google Scholar] [CrossRef]
18. Sintim HY , Bandopadhyay S , English ME , Bary AI , DeBruyn JM , Schaeffer SM , et al. Impacts of biodegradable plastic mulches on soil health. Agric Ecosyst Environ. 2019; 273: 36– 49. doi:10.1016/j.agee.2018.12.002. [Google Scholar] [CrossRef]
19. Men J , Liu H , Jin T , Cai GX , Cao HZ , Cernava T , et al. The color of biodegradable mulch films is associated with differences in peanut yield and bacterial communities. Environ Res. 2024; 248: 118342. doi:10.1016/j.envres.2024.118342. [Google Scholar] [CrossRef]
20. Zhang W , Ma JJ , Cui ZL , Xu LT , Liu Q , Li JB , et al. Effects of biodegradable plastic mulch film on cabbage agronomic and nutritional quality traits, soil physicochemical properties and microbial communities. Agronomy. 2023; 13( 5): 1220. doi:10.3390/agronomy13051220. [Google Scholar] [CrossRef]
21. Wang YJ , Jia XF , Olasupo IO , Feng Q , Wang L , Lu L , et al. Effects of biodegradable films on melon quality and substrate environment in solar greenhouse. Sci Total Environ. 2022; 829: 154527. doi:10.1016/j.scitotenv.2022.154527. [Google Scholar] [CrossRef]
22. Xue YH , Sun ZX , Ju XH , Xi B , Jin T , Jia T . Current status of research and applications of degradable materials for agricultural soil films. China Plast. 2020; 34( 05): 87– 96. (In Chinese). [Google Scholar]
23. Amoah-Antwi C , Kwiatkowska-Malina J , Szara E , Fenton O , Thornton SF , Malina G . Assessing factors controlling structural changes of humic acids in soils amended with organic materials to improve soil functionality. Agronomy. 2022; 12: 283. doi:10.3390/agronomy12020283. [Google Scholar] [CrossRef]
24. Zhao KQ , Yang Y , Peng H , Zhang LH , Zhou YY , Zhang JC , et al. Silicon fertilizers, humic acid and their impact on physicochemical properties, availability and distribution of heavy metals in soil and soil aggregates. Sci Total Environ. 2022; 822: 153483. doi:10.1016/j.scitotenv.2022.153483. [Google Scholar] [CrossRef]
25. Lazarev VI , Minchenko ZN . Evaluation of the efficacy of the use of humic fertilizers in the cultivation of spring barley in the soil and climatic conditions of the central chernozem region. Russ Agric Sci. 2024; 50( 2): 125– 30. doi:10.3103/S1068367424700083. [Google Scholar] [CrossRef]
26. Liu ZP , Han P , Zhao RJ , Wu YY , Wei WX , He FH , et al. Compound sodium nitrophenolate (CSN) improves photo-synthesis and forage quality in hemarthria compressa. Agronomy. 2025; 15( 11): 2526. doi:10.3390/agronomy15112526. [Google Scholar] [CrossRef]
27. Zhou MD , Di QH , Yan Y , He CX , Wang J , Li YS , et al. Multi-omics reveal the molecular mechanisms of Sodium Nitrophenolate in enhancing cold tolerance through hormonal and antioxidant pathways in cucumber. Plant Physiol Bioch. 2025; 223: 109836. doi:10.1016/j.plaphy.2025.109836. [Google Scholar] [CrossRef]
28. Wang ZH , Wu Q , Fan BH , Zhang JZ , Li WH , Zheng XR , et al. Testing biodegradable films as alternatives to plastic films in enhancing cotton (Gossypium hirsutum L.) yield under mulched drip irrigation. Soil Till Res. 2019; 192: 196– 205. doi:10.1016/j.still.2019.05.004. [Google Scholar] [CrossRef]
29. Passos TM , Marconato JC , Franchetti SMM . Biodegradation of films of low density polyethylene (LDPE), poly(hydroxibutyrate-co-valerate) (PHBV), and LDPE/PHBV (70/30) blend with Paecilomyces variotii. Polímeros. 2015; 25( 1): 29– 34. doi:10.1590/0104-1428.1432. [Google Scholar] [CrossRef]
30. Wandansari NR , Kurniawan S , Suntari R , Soemarno . Improving soil fertility and maize growth in suboptimal land through application of humic acid. Int J Des Nat Ecodyn. 2022; 17( 5): 679– 90. doi:10.18280/ijdne.170505. [Google Scholar] [CrossRef]
31. Ayman M , Kamar M , Khalid M . Amino and humic acids promote growth, yield and disease resistance of faba bean cultivated in clayey soil. Aust J Basic Appl Sci. 2009; 3( 2): 731– 9. [Google Scholar]
32. Galambos N , Compant S , Moretto M , Sicher C , Puopolo G , Wäckers F , et al. Humic acid enhances the growth of tomato promoted by endophytic bacterial strains through the activation of hormone-, growth-, and transcription-related processes. Front Plant Sci. 2020; 11: 582267. doi:10.3389/fpls.2020.582267. [Google Scholar] [CrossRef]
33. Kumar SS , Wani OA , Prasad B , Banuve A , Mua P , Sharma AC , et al. Effects of mulching on soil properties and yam production in tropical region. Sustainability. 2024; 16( 17): 7787. doi:10.3390/su16177787. [Google Scholar] [CrossRef]
34. Zhao JY , Yang KJ , Shock CC , Yang B , Dai JL , Wang FX . Small soil wetted proportion and emitter flow rate of drip irrigation enhance potato yield by improving soil water and aeration in the sandy loam in arid Northwest China. Field Crop Res. 2024; 316: 109498. doi:10.1016/j.fcr.2024.109498. [Google Scholar] [CrossRef]
35. Liu ML , Wang C , Liu XL , Lu YC , Wang YF . Saline-alkali soil applied with vermicompost and humic acid fertilizer improved macroaggregate microstructure to enhance salt leaching and inhibit nitrogen losses. Appl Soil Ecol. 2020; 156: 103705. doi:10.1016/j.apsoil.2020.103705. [Google Scholar] [CrossRef]
36. Alguacil MM , Torrecillas E , Lozano Z , Roldán A . Arbuscular mycorrhizal fungi communities in a coral cay system (Morrocoy, Venezuela) and their relationships with environmental variables. Sci Total Environ. 2015; 505: 805– 13. doi:10.1016/j.scitotenv.2014.10.030. [Google Scholar] [CrossRef]
37. Yang C , Huang YZ , Long BB , Gao XH . Effects of biodegradable and polyethylene film mulches and their residues on soil bacterial communities. Environ Sci Pollut Res Int. 2022; 29( 59): 89698– 711. doi:10.1007/s11356-022-22014-y. [Google Scholar] [CrossRef]
38. Jiang N , Wu M , Li GL , Petropoulos E , Sun FF , Wang X , et al. Humic substances suppress Fusarium oxysporum by regulating soil microbial community in the rhizosphere of cucumber (Cucumis sativus L.). Appl Soil Ecol. 2022; 174: 104389. doi:10.1016/j.apsoil.2022.104389. [Google Scholar] [CrossRef]
39. Pathma J , Sakthivel N . Microbial diversity of vermicompost bacteria that exhibit useful agricultural traits and waste management potential. SpringerPlus. 2012; 1( 1): 26. doi:10.1186/2193-1801-1-26. [Google Scholar] [CrossRef]
40. Kallenbach C , Grandy SA . Controls over soil microbial biomass responses to carbon amendments in agricultural systems: a meta-analysis. Agr Ecosyst Environ. 2011; 144( 1): 241– 52. doi:10.1016/j.agee.2011.08.020. [Google Scholar] [CrossRef]
41. Lu ZL , Zhou HY , Zhang NM . A brief overview of research progress on humic acid in fertilizer applications. Humic Acid. 2025;( 03): 14– 20. (In Chinese). doi:10.19451/j.cnki.issn1671-9212.2025.03.002. [Google Scholar] [CrossRef]
42. Wu RP , Xue JH , Wang Y . Effect of weathered coal humic acid on soil physical and chemical properties and heavy metal absorption in lead contaminated soil. Environ Sci Surv. 2018; 37( 02): 56– 61. (In Chinese). doi:10.13623/j.cnki.hkdk.2018.02.015. [Google Scholar] [CrossRef]
43. Jiang HM , Han SY , Zhang HJ , Liu TC , Huang SH , Zhu XY , et al. Calvin cycle driven autotrophic CO2-fixation traits and autotrophic microbial communities in paddy (Anthrosol) and upland (Vertisol) soils: rhizosphere effects and impacts of biochar. Biochar. 2025; 7( 1): 118. doi:10.1007/s42773-025-00538-z. [Google Scholar] [CrossRef]
44. Li YS , Liu MQ , Yu ZH , Liu CK , Hu XJ , Liu JJ , et al. Long-term application of cattle manure alters functional N cycling genes and improves maize yield and nitrogen use efficiency. Geoderma. 2025; 460: 117398. doi:10.1016/j.geoderma.2025.117398. [Google Scholar] [CrossRef]
45. Bai XL , Wu JM , Zhang BY , Zhao H , Tian F , Wang B . Metagenomics reveals functional profiles of soil nitrogen and phosphorus cycling under different amendments in saline-alkali soil. Environ Res. 2024; 267: 120686. doi:10.1016/j.envres.2024.120686. [Google Scholar] [CrossRef]
46. Shi YL , Li TT , Zheng L , Jing XK , Hussain HA , Zhang QW . Enhancing soil multifunctionality through restoring erosion environment and microbial functions combined with organic manure and straw mulching. Agric Ecosyst Environ. 2025; 383: 109515. doi:10.1016/j.agee.2025.109515. [Google Scholar] [CrossRef]
47. Dai ZM , Su WQ , Chen HH , Barberán A , Zhao HC , Yu MJ , et al. Long-term nitrogen fertilization decreases bacterial diversity and favors the growth of Actinobacteria and Proteobacteria in agro-ecosystems across the globe. Glob Change Biol. 2018; 24( 8): 3452– 61. doi:10.1111/gcb.14163. [Google Scholar] [CrossRef]
48. Li YN . Research progress of humic acid fertilizer on the Soil. J Phys Conf Ser. 2020; 1549( 2): 022004. doi:10.1088/1742-6596/1549/2/022004. [Google Scholar] [CrossRef]
49. Jin Q , Zhang YY , Wang QX , Li MJ , Sun H , Liu N , et al. Effects of potassium fulvic acid and potassium humate on microbial biodiversity in bulk soil and rhizosphere soil of Panax ginseng. Microbiol Res. 2022; 254: 126914. doi:10.1016/j.micres.2021.126914. [Google Scholar] [CrossRef]
50. Sun ZJ , Zhong GX , Zhang SB , Yang X , Peng JL , Guo W , et al. Effects of fertilizer reduction combined with organic fertilizer on physicochemical properties and microbial community structure of tobacco-planting soil. Acta Agric Boreali-Sin. 2024; 39( 03): 146– 58. (In Chinese). doi:10.7668/hbnxb.20194778. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools