 Open Access
Open Access
REVIEW
Light Regulation across Strawberry Growth Stages: A Comprehensive Review
1 Department of Agronomy and Horticulture, Jiangsu Vocational College of Agriculture and Forestry, Jurong, China
2 College of Horticulture, Qingdao Agricultural University, Qingdao, China
3 Center for Environment, Health and Field Sciences, Chiba University, Kashiwa, Japan
4 College of Agricultural Engineering, Jiangsu University, Zhenjiang, China
5 Horticulture Research Institute, Sichuan Academy of Agricultural Science, Chengdu, China
6 Jiangsu Engineering and Technology Center for Modern Horticulture, Zhenjiang, China
* Corresponding Authors: Yuanhua Wang. Email: ; Zhengnan Yan. Email:
Phyton-International Journal of Experimental Botany 2026, 95(2), 1 https://doi.org/10.32604/phyton.2026.077381
Received 08 December 2025; Accepted 19 January 2026; Issue published 28 February 2026
Abstract
As a pivotal environmental factor, light, comprising intensity, photoperiod, and spectrum, governs the entire life cycle of strawberries by mediating alterations in the plant’s morphological, physiological, and biochemical traits. Although extensive research has been conducted on light-mediated growth regulation in horticultural crops, most reviews focus primarily on leafy and fruiting vegetables, with limited attention given to berry crops such as strawberries. Additionally, most existing reviews concentrate on one or several growth stages, failing to systematically characterize light’s effects throughout the entire growth cycle and postharvest stage. This review briefly summarizes the regulatory roles of light across key stages of strawberry growth, including seedling propagation, vegetative growth, reproductive growth, and postharvest stages. It seeks to address the knowledge gap by systematically organizing research findings across these developmental phases. The integrated analysis provides a theoretical foundation for designing stage-specific lighting strategies to improve strawberry yield and quality.Keywords
Light, through its spectral composition, light intensity, and photoperiod, governs fundamental processes in plant growth and development, from seed germination to morphogenesis photosynthetic characteristics, physiological metabolism, reproductive activity, and ultimately yield and quality [1]. With the rapid development of modern agriculture, there is increasing emphasis on regulating the photic environment to optimize plant growth. Considering the challenges in precisely controlling natural daylight, the application of artificial light sources to tailor the light environment has become a common practice [2].
Conventional light sources, including have several drawbacks, such as non-tunable spectral characteristics, high thermal radiation, and inconsistent light intensity [3,4] In contrast, light-emitting diodes (LEDs) offer advantages such as narrow bandwidth, low energy consumption, high photoelectric efficiency, minimal thermal output, compact size, enabling precise manipulation of spectral characteristics, luminous intensity, and photoperiod [5]. This has facilitated the widespread application of LEDs across various agricultural fields [6,7,8]. Statistical data indicate that the LED lighting industry is geographically concentrated in Europe and North America [9]. The United States, China, and Canada collectively supply nearly half of all LED grow lights globally, while production in Europe is more evenly distributed among countries [9].
In higher plants, chlorophylls, carotenoids, and anthocyanins serve as the three primary light-absorbing pigments [10]. Red and blue light are most effective in driving photosynthesis, as chlorophylls exhibit maximal absorption within these spectral ranges [11]. Nevertheless, other wavelengths, including green, far-red, and moderate-intensity ultraviolet (UV) light, also significantly influence plant growth [3]. For instance, green light can penetrate leaves more deeply [12], and increase anthocyanin [13] and chlorophyll [14] concentrations while reducing nitrate content in plants [15]. Far-red light can enhance the photosynthetic rate [16], but may decrease anthocyanins [17] and carotenoids [18] levels in some plant species. UV light can benefit postharvest quality [19] and enhance antioxidant levels [20]. A novel study has demonstrated for the first time that manipulating light quality with LED lights can increase the inhibitory activity of strawberry fruit extracts against digestive enzymes, thereby enhancing the functional properties of the fruit [21]. Beyond light quality, light intensity and photoperiod also critically affect plant growth. The suitable light environment for plant growth varies among species and cultivars, necessitating tailored strategies for maximum yield and optimal quality.
Strawberry (Fragaria × ananassa Duch.) is a common and valuable crop cultivated worldwide. Strawberry fruits are abundant in fiber, vitamins, micronutrients, and flavonoids [22], offering significant health benefits. Over the past decade, global strawberry cultivation area and yield have grown substantially, reaching approximately 0.435 million hectares and 10.49 million tonnes in 2023, respectively (FAOSTAT). The compact plant architecture, short fruit development cycle, and high genetic transformation efficiency also make strawberries a model species for fruit crop research [23,24]. However, strawberry production and nutritional quality are highly sensitive to environmental conditions [25] and vulnerable to multiple fungal pathogens, including Botrytis cinerea, Podosphaera aphanis, and Colletotrichum spp. [26,27,28].
Several reviews have summarized progress in areas like flower initiation and secondary metabolite accumulation [25,29,30], while few have focused on the effects of light across the entire strawberry lifecycle, from seedling propagation to postharvest storage. This review therefore aims to fill this gap by providing a holistic synthesis of the effects of light spectrum (Table 1), photoperiod (Table 2), and light intensity (Table 3) on strawberries across four key developmental stages: (1) seedling propagation, (2) vegetative growth, (3) reproductive growth, and (4) postharvest period (Fig. 1). By providing systematic information on the stage-specific light regulations on the growth and development of strawberries, our work offers valuable theoretical guidance for optimizing strawberry production.
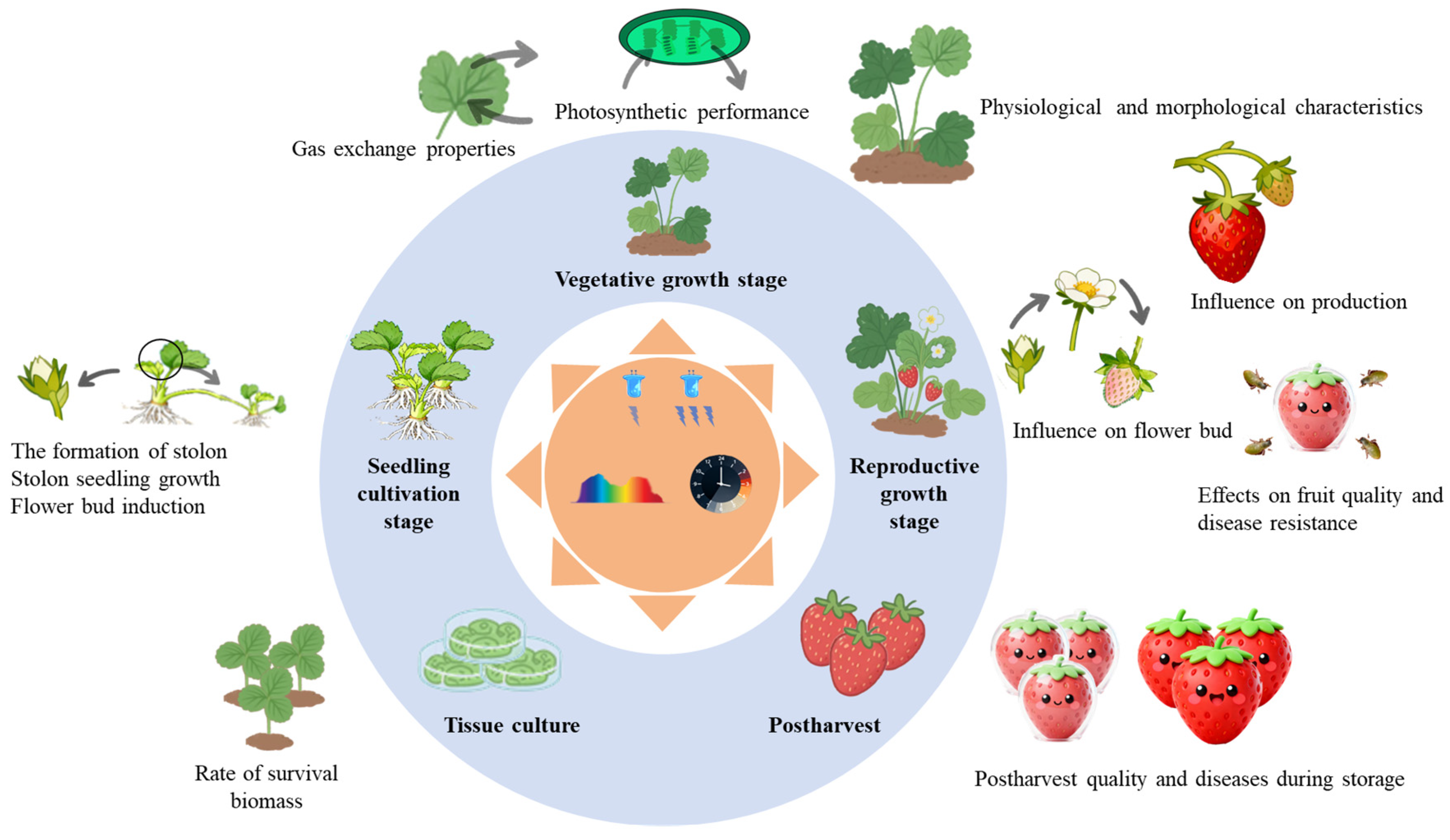
Figure 1: Overview of the effects of light on morphological traits, photosynthetic performance, and quality preservation of strawberry at different growth stages.
2 The Role of Light during the Seedling Propagation
2.1 Impact of Light on Strawberry Plantlets in Tissue Culture
Light is a critical environmental factor influencing the growth of strawberry plantlets in vitro. Studies show that light quality regulates the growth of strawberry tissue-cultured seedlings. For instance, red and green plastic films (the films were used specifically to create defined spectra) promoted the highest shoot regeneration, accompanied by elevated chlorophyll b levels and antioxidant enzyme activities [31]. During the in vitro multiplication of ‘Sabrosa’ strawberries, red and green LEDs enhanced shoot multiplication [32]. Experiments on multiple cultivars using colored cellophane filters observed that red and green light promote adventitious bud regeneration and shoot proliferation, while blue and yellow light enhanced total chlorophyll content [33]. For various cultivars like ‘Akihime’ and ‘Camarosa’, mixtures of red and blue LEDs have been found optimal for plantlet growth [34,35]. Current research on light in strawberry tissue culture focuses on quality, showing red light consistently promoting optimal growth and development across monochromatic and mixed light regimes.
2.2 Role of Light in Cultivating Vigorous Strawberry Transplants
2.2.1 Photosynthesis and Biomass
Light directly regulates photosynthesis, the energy source for plants [36]. During the nursery phase of strawberry plants, red light (660 nm) induced higher net photosynthetic rates compared to blue light (450 nm) [37]. Combinations of blue and red LEDs (ratio of 1:6) plus 100 mg L−1 of nitrogen enhanced photosynthesis and transpiration in ‘Praratchatan 80’, leading to high fruit yield [38]. In ‘Albion’, increases in photosynthetic photon flux density (PPFD) resulted in significant enhancements in plant growth parameters like crown diameter and biomass [39]. Supplementing red and blue light with far-red light also increased leaf area and shoot dry mass of ‘Monterey’ [40].
2.2.2 Runner Formation and Runner Plant Growth
Indoor cultivation with artificial lighting enables high-quality, year-round strawberry production with enhanced yield and consistency [41,42]. Artificial lighting during transplant production enhances runner formation and subsequent plantlet growth [43]. For example, in ‘Maehyang’, elevated light intensity significantly enhanced runner formation and runner plant growth under fluorescent lighting [44]. In ‘Albion’, the number, dry mass, and fresh mass of daughter plants were found to increase with light intensity rising from 250 μmol m−2 s−1 to 450 μmol m−2 s−1 [45]. For ‘Benihoppe’, elevating light intensity from 90 to 270 μmol m−2 s−1 increased crown diameter, shoot dry weight, root dry weight, and root to shoot ratio [43]. Applying high light to stock plants, rather than runners, more effectively enhanced runner growth, providing a new strategy for strawberry propagation [41]. Photoperiod is another critical factor regulating runner formation and elongation in strawberries. In several strawberry cultivars, including ‘Natsuakari’, ‘Camarosa’, and ‘Camaroga’, extended photoperiods have been shown to enhance runner production, elongation, and plantlet formation [46,47]. These effects may be attributed to photoperiodic effects on epidermal cell length and number [48]. However, no significant differences were detected in the number of primary runners, secondary runners, and total runners of strawberry plants subjected to a photoperiod extended from 12 to 16 h d−1; in contrast, runner length was significantly shorter under the 16 h d−1 photoperiod [43]. Spectral composition also modulates strawberry seedling growth. Full-spectrum LEDs (e.g., white LEDs) are increasingly used in controlled environments. Advances in LED chip technology have made diverse white LEDs (e.g., warm, neutral, cool, and mint-white) with distinct spectra commercially available [49]. Mint-white LEDs, with high efficacy in photosynthesis, enhanced runner formation and vegetative growth of ‘Maehyang’ strawberry in controlled environments [50]. Supplementing mint-white LEDs with optimal blue wavelengths improved growth in both stock plants and runner offspring [51]. White LEDs promoted dry mass accumulation in ‘Akihime’ strawberry runners, whereas adding green light to red-blue LED combinations boosted runner plant formation and proliferation [52].
Interactions among light parameters further illustrate the critical impact of light regimes on strawberry seedling development. Daily light integral (DLI), determined by light intensity and photoperiod was shown in ‘Benihoppe’ strawberries to suppress runner elongation and reduce photosynthetic capacity under high DLI conditions [43]. For ‘Sulhyang’, 20 μmol m−2 s−1 blue light maximized runner formation irrespective of photoperiod (long-day/short-day) or supplemental lighting strategy [53]. Blue light under non-runnering short-day conditions upregulated key gibberellin pathway genes, potentially triggering runner formation. Furthermore, photoperiod beyond 16 h or night interruption lighting with various wavelengths enhanced runner induction in ‘Sulhyang’ and ‘Maehyang’ [54]. A novel side lighting strategy also improved morphophysiological traits and runner formation by stimulating photosynthesis in cuttings, offering a cost-effective approach for strawberry propagation in closed cultivation systems [42].
Flower bud induction is a critical step during the seedling stage. Low-intensity red light irradiation during cold storage could maintain chlorophyll and carbohydrate levels in ‘Nyoho’ strawberry seedlings [55]. Similarly, the accumulation of carbohydrates and the ratio of photosynthetic pigments were enhanced under mixed red and blue LEDs in cold-stored ‘Elkat’ [56]. A high far-red/red light ratio promoted floral bud induction [57,58], likely by inactivating phytochrome B and activating the photoperiodic flowering pathway [59]. Both far-red and blue light accelerate floral induction in multiple strawberry cultivars [60,61]. Extended photoperiods supplemented with far-red light also promoted flower bud development [62]. However, a study conducted by Sidhu et al. [63] demonstrated that photoperiod conditioning exerted no significant effects on the flower stalk development and bud induction of strawberry plants. Crucially, there is a strong interaction between temperature and photoperiod during floral induction. High temperatures inhibit strawberry flowering and bud development [64], and seedling age affects induction [65].
3 The Effects of Light during the Vegetative Growth Period
3.1 Photosynthetic Characteristics
3.1.1 Photosynthetic Pigment Content
Studies on cultivars such as ‘Elsanta’, ‘Akihime’, and ‘Daewang’ generally report higher chlorophyll levels under red light than blue light, potentially due to blue light-induced suppression of chlorophyll b synthesis [66,67,68]. Compared to monochromatic light, broad-spectrum or mixed red-blue light often enhances chlorophyll content, while the influence is cultivar-specific [28,69]. Monochromatic blue light or a higher proportion of blue light generally promotes an increase in the chlorophyll a/b ratio [67,68]. Carotenoid content has been reported to decline under blue light relative to red light in some strawberry cultivars [67,68], red and blue light combinations can elevate it [70]. Studies have indicated that blue and red light can enhance the expression of genes encoding enzymes related to carotenoid pigments [71].
3.1.2 Gas Exchange Characteristics
Blue light or cool white lamps (with higher blue light proportion) have been reported to enhance photosynthetic rates and stomatal conductance in strawberry plants [72,73,74], likely due to modifications in leaf biochemistry and enhanced stomatal conductance [74,75]. An optimal blue to red light ratio (e.g., 1:6) can exhibit the highest photosynthetic and transpiration rates due to synergistic interactions [38,76]. Green light also benefits plant growth and physiology [77]. A combination of 20% green, 20% blue, and 60% red light enhanced net photosynthesis, transpiration rate, and stomatal conductance in strawberry plants, but exceeding 20% green light reduced these parameters [78]. Plant gas exchange characteristics of strawberry plants are also influenced by leaf age and the orientation of light exposure [79].
3.1.3 Chlorophyll Fluorescence Parameters
Blue LED morning supplementation enhanced the maximum quantum efficiency of PSII in ‘Maehyang’ and ‘Seolhyang’ [73]. In ‘Daeselect’, the effective quantum yield of PSII photochemistry Y(II), photochemical quenching coefficient, and electron transport rate were optimized under 250 μmol m−2 s−1 supplemental LED light (50% red, 20% green, and 30% blue) with a 12 h photoperiod [70]. In ‘Benihoppe’, shading reduced leaf minimal fluorescence, while red/blue LED light elevated it [80]. Red film enhanced photosynthesis in ‘Akihime’ by elevating photosynthetic pigment content, quantum yield, electron transfer efficiency, reaction center density [68].
Essential nutrients regulate plant growth, influenced by species, stage, and environment. Blue and red light promote mineral accumulation, but responses are differential and cultivar-specific [69,81]. For example, in ‘Toyonoka’, leaf iron and magnesium levels were higher under a high red:blue ratio [82], while potassium decreased under a low red:blue ratio. In ‘Praratchatan 80’, leaf nitrogen peaked under 3:1 red:blue LED, while potassium content increased with higher red-light ratios [38]. Green light can also impact leaf nutrient status in strawberries. For instance, a high proportion (27%) resulted in elevated calcium, potassium, and phosphorus [78], possibly attributed to increased root biomass. Moreover, blue morning light best boosted strawberry carbohydrates [73]. Supplemental lighting with distinct spectral compositions also modulates antioxidant enzyme activities and oxidative stress markers. In ‘Elsanta’, red light has been shown to enhance antioxidant enzyme activity but also increase levels of hydrogen peroxide and malondialdehyde [83].
3.3 Biomass and Morphological Indices
Crop yield generally responds positively to increased PPFD, photoperiod, and DLI. Total plant dry weight in ‘Toyonok’ and ‘Fukuoka S6’ increased with PPFD elevation [82,84]. ‘Albion’ strawberry exhibited linear biomass gains as PPFD rose, and root biomass increased with photoperiod extension [39]. For ‘HS138’, dry matter accumulation was greater under high DLI [85]. Far-red light addition to red and blue light combinations increased shoot fresh and dry weights in ‘Monterey’, whereas no significant effects on ‘Albion’ [40]. Appropriate combinations of red and blue light enhanced biomass in various cultivars such as ‘Toyonoka’, ‘Darselect’, and ‘Albion’ [70,78,82,86].
Morphological characteristics of strawberry plants are also influenced by light. Supplemental lighting enhanced leaf area in ‘Clery’, ‘San Andreas’, ‘Albion’ and ‘Sabrina’ [87,88]. In ‘Monterey’, the addition of far-red light to red-blue light combinations resulted in an increase in leaf area [40]. Studies conducted on ‘Akihime’ revealed increased petiole length and leaf area when plants were grown under red plastic film [68]. Supplemental LED lighting also increased the specific leaf weight [89], which reflects leaf thickening and is likely driven by the accumulation of photosynthetic assimilation products.
3.4 Light-Mediated Enhancement of Strawberry Resilience to Stress
3.4.1 Biotic Stress Resistance
Plant diseases markedly curtail agricultural productivity, especially for high-value edible fruit crops such as strawberries. Light is a key regulator of plant-pathogen interactions [67,90]. A low red: far-red ratio can suppress immunity, while red light enhances biotic stress tolerance [91,92,93]. In strawberries, specific red light regimens significantly boost leaf resistance to B. cinerea [94,95], correlating with the induction of antioxidant systems and defense pathways [67,83]. In contrast, blue light appears to exert a more direct inhibitory effect on B. cinerea infection [94]. Strawberry responsiveness to B. cinerea is under circadian control [95], and correlates with leaf developmental stage [67]. This age-dependent resistance pattern may associate with differential levels of hydrogen peroxide, chlorophyll, and carotenoids across leaf ages. UV light, particularly UV-C, has been shown to modulate susceptibility to B. cinerea and Colletotrichum in a cultivar-dependent manner [96,97,98,99]. UV-C-induced pathogen resistance may involve enhanced biosynthesis of secondary metabolites, increased defense-related enzyme activity, and accumulation of phenolic compounds [98,99,100]. Beyond UV-C, UV-B irradiation has also been documented to enhance plant disease resistance against pathogens (e.g., Colletotrichum spp.) [28,101]. These findings position light as an eco-friendly alternative to pesticides for controlling strawberry diseases. However, further optimization for specific pathogens and cultivars is needed.
3.4.2 Abiotic Stress Resistance
Strawberry is highly sensitive to salinity and alkalinity [102]. Recent research indicates that light radiation can effectively mitigate abiotic stresses like salinity and alkalinity [103,104]. In ‘Sabrina’, under salinity and alkalinity stress, combined blue and red LED treatments enhanced leaf chlorophyll content and improved the efficiency of the photosynthetic apparatus [102], which is likely due to the upregulation of key genes encoding enzymes involved in chlorophyll biosynthesis [105]. Studies on ‘Camarosa’ strawberry demonstrated that combined blue and red light significantly increased leaf area under salinity and alkalinity stress [106]. In ‘Sabrina’, blue, red, and combined light spectra enhanced mineral content, phenolics, antioxidants, and crown starch, thereby likely improving stress tolerance to salinity and alkalinity [107]. Regulating light regimes enhances stress resistance, yet the effects of light intensity and photoperiod warrant further investigation.
4 The Influence of Light during the Reproductive Growth Period
4.1 Effect of Light on Strawberry Flowering
Strawberry flowering is regulated by genotype and environmental factors [108,109]. Strawberries are primarily categorized into seasonal (short-day) and perpetual (long-day or day-neutral) flowering types based on their photoperiodic responses and cropping season [110]. In seasonal flowering cultivars, flower bud initiation, which marks the transition from vegetative to reproductive growth, is typically induced under low temperatures (15–25°C) and short-day conditions (10–12 h of daylight) [111]. In contrast, perpetual flowering types initiate flower buds under long-day conditions (>12 h of daylight) or independently of day length (day-neutral), enabling continuous fruit production under suitable temperatures [111]. A previous study reported by Muñoz-Avila et al. [112] demonstrated conserved function of the MADS-box transcription factor FaSOC1 as a potent flowering repressor in woodland and cultivated strawberries, and its key role in photoperiodic repression of FLOWERING LOCUS T (FT) genes FaFT2/FaFT3—with FaTFL1 upregulation less prominent than in the woodland strawberry. Limitations of outdoor cultivation are prompting a shift to controlled environments, which improve stability and efficiency, including through supplemental or sole-source LED lighting [40]. For example, supplementing with 13 h of combined red and blue LEDs commencing 1 h before sunset in greenhouses increased flowering up to a 300% [113]. Under monochromatic supplementary light, blue LEDs produced the highest number of flower clusters, while red LEDs produced the fewest [113], potentially due to differential effects on hormone levels [114]. Within mixed red-blue spectra, higher red light proportions can promote inflorescence initiation in some cultivars such as ‘Albion’ and ‘Praratchatan 80’ [38,86]. This effect may be attributed to red light enhancing sugar synthesis and nutrient allocation while blue light accelerates flowering through photoperiodic signaling and increased carbohydrate availability [115,116], achieving an optimal balance between vegetative and reproductive growth. Furthermore, supplementing a red-blue background lighting with far-red light reduced the time to first harvest in some long-day strawberry cultivars like ‘Albion’ [40]. Light intensity and photoperiod are also crucial. Reduced light intensity during flowering can induce stamen abortion [117], while elevated light intensity accelerates flowering and promotes uniformity [109,118]. In ‘Albion’, extending the photoperiod from 12 to 16 h under relatively high light intensity (PPFD ≥ 300 μmol m−2 s−1) accelerated flowering [39], likely attributable to increased carbohydrate accumulation in the crown and roots, a process that supports floral initiation [119,120]. To maximize profits, achieving early, uniform, and stable flowering through light environment regulation is essential in strawberry production, as it ensures timely harvests and efficient management.
4.2 Effect of Light on Strawberry Yield
Studies on ‘Elsanta’ strawberries demonstrated that blue LED light enhanced fruit set and final yield compared to red LEDs or fluorescent lamps [121]. For ‘Albion’, a light spectrum comprising 20% green, 20% blue, and 60% red light achieved high fruit yield, but increasing green light beyond 20% reduced productivity [78]. This phenomenon may be attributable to the higher crown biomass observed under the 20% green light regime, consistent with previous findings correlating larger crown size with increased yield [122]. Similarly, another study reported that optimal yields under a spectrum of 20% green, 30% blue, and 50% red light were achieved at a PPFD of 250 μmol m−2 s−1, whereas lower intensities reduced yield despite the inclusion of green light [70]. For ‘Akihime’, red-transmitting film achieved superior fruit weight, suggesting excellent cultivation suitability [68]. Furthermore, light directly regulates photosynthetic processes in plants. LED supplemental lighting boosts strawberry fruit weight, number, and marketable yield across cultivars through enhanced photosynthesis and carbohydrate accumulation that drives fruit development [21,69,123,124]. Notably, supplemental lighting generally exhibits more pronounced effects during the winter season under cloudy weather and low solar radiation conditions [125]. However, once solar radiation exceeds a critical threshold, supplemental lighting shows reduced effectiveness and diminished economic viability.
4.3 Effect of Light on Strawberry Fruit Quality
Anthocyanins are ubiquitous, vital secondary metabolites that contribute to fruit coloration in a wide range of higher plants [126,127]. Blue, red, and far-red light have been shown to enhance anthocyanin accumulation in fruits [21,128]. Similarly, wavelength-selective plastic films (e.g., red or yellow filters) and light-conversion films (which convert ultraviolet to blue light) also demonstrate that light quality regulates anthocyanin content in strawberry fruits [68,74,126]. Research has indicated that light quality affects anthocyanin accumulation in strawberries by regulating genes (e.g., PHOT2, LAR, ANR, MYBs, bHLHs) associated with anthocyanin biosynthesis and/or modifying enzyme activities [68,129]. UV light exposure during the nighttime cultivation period also increases anthocyanin content [128,130].
Hardness is a critical parameter for evaluating fruit quality, as it serves as an indicator of the maturation stage [131]. Supplemental lighting like red, blue, and white light combination can reduce strawberry firmness, thereby accelerating ripening [131]. However, some studies indicate that preharvest UV irradiation enhances strawberry firmness and extends shelf life [132,133], possibly by downregulating the transcription of genes associated with cell-wall degradation [134].
The rich nutritional value is one of the reasons why strawberries are preferred by consumers. Exposure to UV light during the cultivation of strawberries has been demonstrated to enhance the content of ascorbic acid and ellagic acid in the fruits [130]. Similarly, another study reported that illumination using LED lights comprising red, blue, and green wavelengths led to an increase in ascorbic acid levels, with direct irradiation of the fruits being more effective than irradiation of the leaves [135]. Light quality also influences the content of phenolic compounds in strawberry fruits. For example, significant increases in phenolic content have been observed under UV-B, red light, far-red light, and blue light [21,128,136]. Exposure to red and far-red supplemental light promoted the synthesis of certain individual polyphenols in strawberry fruits [137]. Supplemental lighting (e.g., UV-B) can elevate the total soluble solids content in strawberry fruits [131], while the red light-quality selective plastic film enhances soluble protein levels [68].
Taste-driven consumer choices highlight the importance of flavor in strawberries, which is primarily determined by sugars, organic acids, and aroma [135,138,139]. Supplementary lighting enhances strawberry sugar content by increasing photosynthate accumulation [89,140]. Besides, UV light and red-light selective films alter the sugar-acid ratio [68,130]. Furthermore, red mulch enhances strawberry aroma [68,138], as its reflected red/far-red light acts through the natural phytochrome system to modify gene expression, increasing aroma compounds. This finding provides novel insights for enhancing strawberry aroma, which can improve the quality of both fresh fruits and derived products, thereby benefiting consumer health.
4.4 Effect of Light on Preharvest Disease Resistance of Strawberry
Strawberries are highly susceptible to numerous pathogens, which can cause significant yield losses both in the field and during postharvest storage [141]. UV light is the main light source applied for preharvest disease control. UV-B radiation suppresses powdery mildew by upregulating defense-related genes and stimulating biosynthesis of antifungal compounds [141]. Nighttime UV-C irradiation enhances efficacy against powdery mildew [26,142]. Visible light, such as red light, can also enhance pathogen tolerance in strawberry fruit by upregulating cell wall development-related genes (e.g., FaPE41) and inhibiting fungal enzymes [83]. The use of light to suppress preharvest diseases aligns with the principles of sustainable agriculture and addresses growing consumer preferences for safer, high-quality produce grown with minimal chemical inputs.
5 The Role of Light on Strawberries during the Postharvest Stage
5.1 Role of Light in Fruit Preservation and Quality Maintenance
Strawberry postharvest quality is regulated by both physiological and environmental factors. Light exposure significantly influences this process, with different wavelengths inducing distinct effects. Red light (e.g., 525 nm) promotes coloration by increasing anthocyanins, maintains ascorbic acid, and enhances soluble solids [130,143,144]. In contrast, blue light (e.g., 470 nm) elevates total phenols and ascorbic acid by boosting antioxidant enzyme activity and free radical scavenging capacity, but it also accelerates ripening by increasing respiration and ethylene production [130]. Similarly, violet-blue light at 405 nm increases anthocyanin and phenol content, though without affecting firmness [145]. However, not all lighting is beneficial; white LED light, for instance, can accelerate quality deterioration by doubling transpiration and promoting ascorbic acid degradation [146]. Thus, light quality must be carefully selected to improve postharvest strawberry quality.
Light has been employed both alone and synergistically to extend strawberry shelf life. The combination of low temperature (8°C) and blue light was more effective than either alone, enhancing antioxidant defense and delaying cell wall degradation [147]. Similarly, integrating blue light with salicylic acid soaking better preserved weight, firmness, and soluble protein content [148]. Intense pulsed light (2.0 Hz, 60 s) under storage conditions of 25°C, 80% relative humidity, and 7 days, significantly reduced strawberry decay rate, stabilized color, and regulated flavor compounds via the lipoxygenase pathway [149].
Strawberry aroma, a key quality trait, is regulated by the interaction of light and temperature, influencing the biosynthesis of esters and phenylpropanoids [150]. Research has characterized the comprehensive features and genetic mechanisms of this light- and temperature-mediated aroma regulation, though its effects are cultivar-dependent [150]. For instance, postharvest blue light improved the sensory quality and key volatiles in ‘Festival’ strawberries but had no significant effect on ‘Florida Radiance’, highlighting the role of genetic variation [8].
5.2 Inhibitory Effects of Light on Postharvest Diseases of Fruits
Strawberry shelf life is challenged by postharvest diseases, which UV irradiation can address [151]. Far-UV-C shows superior bactericidal activity to UV-C (254 nm) but can impair plant physiology [26]. Combining UV with other methods, such as peracetic acid or heat treatment, more effectively reduces pathogens (e.g., Salmonella), delays ripening, and preserves flavor without negative sensory impact [132,152].
High-power pulsed light mitigates microbial contamination on produce. It is more effective on smooth surfaces (e.g., blueberries) than rough ones (e.g., strawberries), but excessive doses cause damage [153]. A study using specific parameters (0–3.9 J/cm2 UV dose, 0–200 s) reduced strawberry surface microbial contamination, extended shelf life by 2 days without affecting nutrients [154]. Light and temperature interact to inhibit pathogens. Blue light at 4°C reduced powdery mildew incidence by 70%, whereas it was ineffective at 21°C [155].
Table 1: Effects of light spectrum on growth and secondary metabolites accumulation of strawberry at different growth stages.
| Growth Stages | Light Spectrum | Photoperiod | Cultivars | Effects | Reference |
|---|---|---|---|---|---|
| Seedling propagation stage | Red, green, and yellow plastic films | 16 h | Toyonoka | Achieved the highest regeneration rate under red and green plastic films and increased chlorophyll b content; the activities of SOD/CAT: red film > green film > yellow film. | [31] |
| Red light | 16 h | Sabrosa | Increased concentrations of photosynthetic pigments. | [32] | |
| 16 h | Festival, Sweet Charlie, Tudla | Demonstrated the highest adventitious shoot regeneration frequency; elevated levels of N+ in plant tissues. | [33] | ||
| 24 h | HS138 | Increased the dry matter during the nursery period. | [37] | ||
| Green light | 16 h | Sabrosa | Decreased concentrations of photosynthetic pigments. | [32] | |
| 16 h | Festival, Sweet Charlie, Tudla | Increased adventitious shoot regeneration frequency. Increased levels of K+ in plant tissues. | [33] | ||
| Blue light | 16 h | Festival, Sweet Charlie, Tudla | Increased chlorophyll content and elevated levels of phosphorus in plant tissues. | [33] | |
| 10 h/14 h | Sulhyang | In non-runnering short-day conditions, blue light stimulated the expression of FaGRAS32 and FaGA20ox. | [53] | ||
| Yellow cellophane films | 16 h | Festival, Sweet Charlie, Tudla | Exhibited the higher total chlorophyll content. | [33] | |
| Red light + blue light | / | Akihime | Red:blue (7:3), 60 μmol m−2 s−1: demonstrated optimal growth. | [34] | |
| 16 h | Camarosa | Red:blue (9:1): the most effective in vitro shoot tip development. | [35] | ||
| 16 h | Elkat | In cryopreserved stock plants, carbohydrate accumulation and the ratio of photosynthetic pigments were enhanced. | [56] | ||
| Red light + blue light + far-red light | 18 h | Monterey | Increased the leaf area and shoot dry mass. | [40] | |
| White light | 16 h | Maehyang | Enhanced runner formation. | [49] | |
| 16 h | Akihime | Promoted dry matter accumulation. | [52] | ||
| Blue light+ white light | 16 h | Maehyang | Enhanced the growth performance of both mother and daughter plants. | [51] | |
| Vegetative growth stage | Blue light | 16 h | Elsanta, Akihime | Increased the leaf carotenoid content. | [67] |
| 12 h/16 h | Darselect | Inhibited elongation growth. | [70] | ||
| Red light | 16 h | Elsanta, Akihime | Enhanced plant resistance to pathogens. | [67] | |
| 5 h | Elsanta | Enhanced antioxidant enzyme activities. | [83] | ||
| Red colored light-quality selective plastic films | / | Akihime | Increased petiole length and leaf area in the plants. | [68] | |
| Red light + blue light | 18 h | Albion, Monterey | Enhanced biomass. | [40] | |
| 16 h | Darselect | Red:blue (3:7): increased chlorophyll a and chlorophyll b content. | [66] | ||
| 11 h | Camarosa | Enhancing CO2 uptake under salinity stress conditions while under alkaline stress. | [103] | ||
| UV light | 49 s, 73 s, 104 s, 159 s, 208 s | Candiss, Charlotte, Cirafine | UV-C: modulated their susceptibility to B. cinerea. | [99] | |
| 3 h | multiple varieties | UV-B: low plant injury. | [101] | ||
| Reproductive growth stage | Red light + blue light | 9 h | Praratchatan 80 | Red:blue (6:1), nitrogen application of 100 mg L−1: advanced the flowering time. | [38] |
| Red light + blue light + far-red light | 18 h | Albion | Reduced the time to first harvest in certain long-day. | [40] | |
| 18 h | Monterey | Increased leaf area. | [40] | ||
| Red light | / | Akihime | Increased the weight of individual fruits; enhanced strawberry production. | [68] | |
| 16 h | Toyonaka | Increased phenolic content. | [129] | ||
| / | Seolhyang | Red light-selective plastic mulch increased soluble protein levels. | [130] | ||
| Red light + blue light + green light | / | Albion | Green:blue:red (2:2:6): achieved high fruit yield. | [78] | |
| Blue light | 13 h | Fortuna | Increased the number of flower clusters. | [113] | |
| 14 h | Elsanta | Improved production. | [121] | ||
| UV light | 14 h | Albion | Enhanced strawberry firmness. | [128] | |
| / | Seolhyang | Increased anthocyanin content in strawberry fruits. | [130] | ||
| Postharvest stage | Blue light | 24 h | Florida Radiance | Enhanced the sensory quality. | [8] |
| 24 h | Fengguang | Increased antioxidant enzyme activity; increased antioxidant content; improved fruit color; promoted fruit ripening. | [143] | ||
| UV light | / | Camarosa | UV-C: inhibited microbial growth on strawberries. | [151] |
Table 2: Effects of photoperiod on growth and secondary metabolites accumulation of strawberry at different growth stages.
| Growth Stages | Photoperiod | Cultivars | Effects | Reference |
|---|---|---|---|---|
| Seedling propagation stage | 12 h/d and 16 h/d | Benihoppe | Runner length decreased linearly with increasing DLI. | [43] |
| 12, 14, 16, 18, 20, and 22 h/d | Sulhyang, Maehyang | Photoperiod exceeding 16 h induced strawberry runner formation. | [54] | |
| Vegetative growth stage | 12 and 16 h/d | Albion | 16 h/d: Increased in root biomass. | [39] |
| 12, 14, and 16 h/d | Darselect | 12 h/d: Optimal performance in photosystem II parameters. | [70] | |
| Reproductive growth stage | 12 and 16 h/d | Albion | 16 h/d: Earlier flowering period, increased strawberry fruit production. | [39] |
| 16 and 24 h/d | Elan | Extended photoperiod promoted flower bud development. | [64] | |
| Supplemental 13-h LED lighting | Fortuna | Increased Fruit quality. | [113] |
Table 3: Effects of light intensity on growth and secondary metabolites accumulation of strawberry at different growth stages.
| Growth Stages | Light Intensity | Cultivars | Effects | Reference |
|---|---|---|---|---|
| Seedling propagation stage | 45, 60, and 75 μmol m−2 s−1 | Akihime | The optimal light intensity was 60 μmol m−2 s−1 and higher light intensity improved the growth of seedlings. | [34] |
| 200, 250, 300, and 350 μmol m−2 s−1 | Benihoppe | Length of runners decreased under photoperiod of 16 h/d. | [45] | |
| 137.4, 217.0, and 274.7 μmol m−2 s−1 | Maehyang | About 280 μmol m−2 s−1 improved the number of strawberry seedlings and the total yield of stolons. | [44] | |
| 250, 350, and 450 μmol m−2 s−1 | Albion | The number, total dry mass, and total fresh mass of daughter plants increased with the increase in light intensity. | [43] | |
| Vegetative growth stage | 200, 300, and 450 μmol m−2 s−1 | Albion | Increased strawberry plant growth with increased light intensity. | [39] |
| 150, 200, and 250 μmol m−2 s−1 | Darselect | The light intensity of 250 μmol m−2 s−1 showed a better performance in terms of key photosynthetic indicators. | [70] | |
| 149, 269, and 567 μmol m−2 s−1 | Toyonoka | Improvement of PPFD increased the total dry weight of plants. | [82] | |
| Reproductive growth stage | 200, 300, and 450 μmol m−2 s−1 | Albion | Improve PPFD advanced the flowering period and the harvest period. | [39] |
| Supplementary light | Red Face | High light intensity improved the sugar content and strawberry production. | [140] |
This review outlines how regulating light intensity, photoperiod, and spectrum influences the entire strawberry growth cycle (Fig. 2). Studies show these parameters affect propagation, growth, flowering, fruit quality, and postharvest preservation primarily by modulating photosynthesis, morphology, physiology, and defense responses. Notably, the effects of light regulation are developmental stage-specific and cultivar-dependent. While current research predominantly focuses on single growth stages or spectral factors, we propose that establishing precise, stage-specific light regulation strategies represents a promising approach to simultaneously enhance yield, quality, and stress resistance in strawberry production. Future research and commercial applications should prioritize: (1) developing integrated, cost-effective “light recipes” that dynamically adapt to different cultivars and growth stages to maximize resource-use efficiency; (2) investigating the synergistic optimization of the light environment across the entire growth cycle and explore the interaction mechanisms between light signaling and other environmental factors (e.g., temperature, CO2, humidity); and (3) establishing sensor-based, automated lighting control systems that respond in real-time to plant phenotypic and physiological feedback.
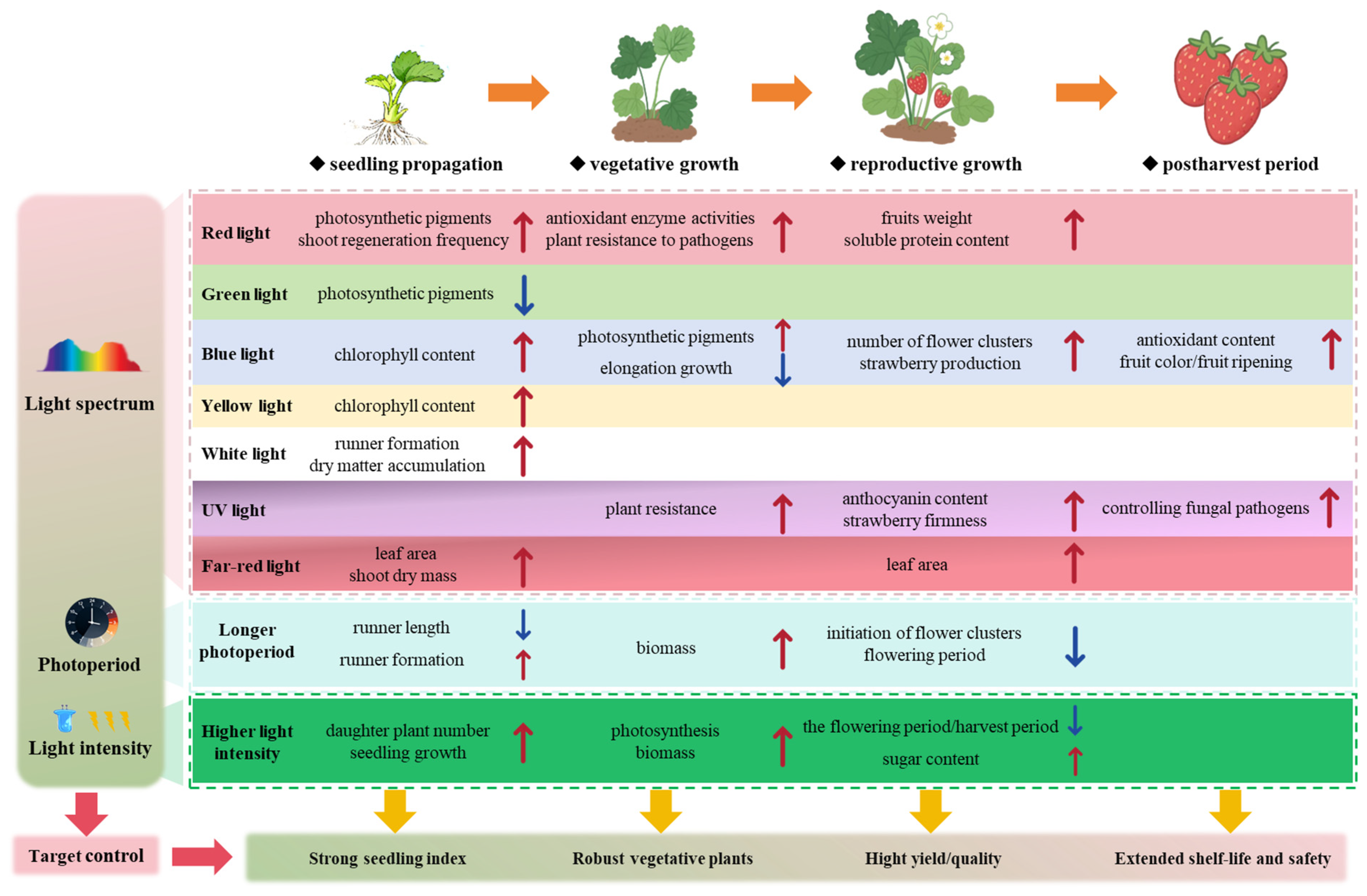
Figure 2: Schematic diagram of the effects of light spectrum, intensity, and photoperiod on key growth and developmental parameters of strawberry at different stages. Please increase the smallest letter size inside the figure to make it readable.
Acknowledgement:
Funding Statement: This work was supported by National Key Research and Development Program of China (2023YFF1001700), the Unveiling and Leading Projects (2022kj05) and Yafu Technology Innovation Team of Jiangsu Vocational College of Agriculture and Forestry (2024kj02), the Innovation & Entrepreneurship Training Program for College Students of Qingdao Agricultural University (QNDC20250149).
Author Contributions: Conceptualization, Geng Zhang, Zhengnan Yan; Supervision, Geng Zhang, Duyen T. P. Nguyen, Jinxiu Song, Zejin Zhang, Zhiming Yan, Yuanhua Wang, Zhengnan Yan; Funding acquisition, Geng Zhang, Zhiming Yan; Project management, Geng Zhang, Zhiming Yan, Yuanhua Wang; Writing—original draft, Geng Zhang, Xiangyu Ding, Zhengnan Yan; Methodology, Xiangyu Ding, Zidan Wen, Chao Liu, Duyen T. P. Nguyen, Jinxiu Song, Zejin Zhang, Zhengnan Yan, Yuanhua Wang; Writing—review & editing, Geng Zhang, Xiangyu Ding, Zidan Wen, Chao Liu, Yuanhua Wang, Zhengnan Yan. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: Not applicable.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Zoratti L , Karppinen K , Luengo Escobar A , Häggman H , Jaakola L . Light-controlled flavonoid biosynthesis in fruits. Front Plant Sci. 2014; 5: 534. doi:10.3389/fpls.2014.00534. [Google Scholar] [CrossRef]
2. Singh D , Basu C , Meinhardt-Wollweber M , Roth B . LEDs for energy efficient greenhouse lighting. Renew Sustain Energy Rev. 2015; 49: 139– 47. doi:10.1016/j.rser.2015.04.117. [Google Scholar] [CrossRef]
3. D’Souza C , Yuk HG , Khoo GH , Zhou W . Application of light-emitting diodes in food production, postharvest preservation, and microbiological food safety. Compr Rev Food Sci Food Saf. 2015; 14( 6): 719– 40. doi:10.1111/1541-4337.12155. [Google Scholar] [CrossRef]
4. Ngcobo BL , Bertling I . An overview of the recent developments and current status on the preharvest application of LED technology in controlled environment agriculture. J Hortic Sci Biotechnol. 2024; 99( 5): 531– 8. doi:10.1080/14620316.2024.2341906. [Google Scholar] [CrossRef]
5. Nassarawa SS , Abdelshafy AM , Xu Y , Li L , Luo Z . Effect of light-emitting diodes (LEDs) on the quality of fruits and vegetables during postharvest period: a review. Food Bioprocess Technol. 2021; 14( 3): 388– 414. doi:10.1007/s11947-020-02534-6. [Google Scholar] [CrossRef]
6. Nanya K , Ishigami Y , Hikosaka S , Goto E . Effects of blue and red light on stem elongation and flowering of tomato seedlings. Acta Hortic. 2012; 956: 261– 6. doi:10.17660/ActaHortic.2012.956.29. [Google Scholar] [CrossRef]
7. Kaiser E , Ouzounis T , Giday H , Schipper R , Heuvelink E , Marcelis LFM . Adding blue to red supplemental light increases biomass and yield of greenhouse-grown tomatoes, but only to an optimum. Front Plant Sci. 2019; 9: 2002. doi:10.3389/fpls.2018.02002. [Google Scholar] [CrossRef]
8. Campbell SM , Sims CA , Bartoshuk LM , Colquhoun TA , Schwieterman ML , Folta KM . Manipulation of sensory characteristics and volatile compounds in strawberry fruit through the use of isolated wavelengths of light. J Food Sci. 2020; 85( 3): 771– 80. doi:10.1111/1750-3841.15044. [Google Scholar] [CrossRef]
9. Paucek I , Appolloni E , Pennisi G , Quaini S , Gianquinto G , Orsini F . LED lighting systems for horticulture: business growth and global distribution. Sustainability. 2020; 12( 18): 7516. doi:10.3390/su12187516. [Google Scholar] [CrossRef]
10. Zheng L , He H , Song W . Application of light-emitting diodes and the effect of light quality on horticultural crops: a review. HortScience. 2019; 54( 10): 1656– 61. doi:10.21273/HORTSCI14109-19. [Google Scholar] [CrossRef]
11. Chen M , Chory J , Fankhauser C . Light signal transduction in higher plants. Annu Rev Genet. 2004; 38: 87– 117. doi:10.1146/annurev.genet.38.072902.092259. [Google Scholar] [CrossRef]
12. Terashima I , Fujita T , Inoue T , Chow WS , Oguchi R . Green light drives leaf photosynthesis more efficiently than red light in strong white light: revisiting the enigmatic question of why leaves are green. Plant Cell Physiol. 2009; 50( 4): 684– 97. doi:10.1093/pcp/pcp034. [Google Scholar] [CrossRef]
13. Samuolienė G , Sirtautas R , Brazaitytė A , Duchovskis P . LED lighting and seasonality effects antioxidant properties of baby leaf lettuce. Food Chem. 2012; 134( 3): 1494– 9. doi:10.1016/j.foodchem.2012.03.061. [Google Scholar] [CrossRef]
14. Muneer S , Kim EJ , Park JS , Lee JH . Influence of green, red and blue light emitting diodes on multiprotein complex proteins and photosynthetic activity under different light intensities in lettuce leaves (Lactuca sativa L.). Int J Mol Sci. 2014; 15( 3): 4657– 70. doi:10.3390/ijms15034657. [Google Scholar] [CrossRef]
15. Samuolienė G , Brazaitytė A , Sirtautas R , Novičkovas A , Duchovskis P . Supplementary red-LED lighting affects phytochemicals and nitrate of baby leaf lettuce. J Food Agric Environ. 2011; 9( 3–4): 271– 4. [Google Scholar]
16. Emerson R , Chalmers R , Cederstrand C . Some factors influencing the long-wave limit of photosynthesis. Proc Natl Acad Sci U S A. 1957; 43( 1): 133– 43. doi:10.1073/pnas.43.1.133. [Google Scholar] [CrossRef]
17. Stutte GW , Edney S , Skerritt T . Photoregulation of bioprotectant content of red leaf lettuce with light-emitting diodes. HortScience. 2009; 44( 1): 79– 82. doi:10.21273/HORTSCI.44.1.79. [Google Scholar] [CrossRef]
18. Li J , Hikosaka S , Goto E . Effects of light quality and photosynthetic photon flux on growth and carotenoid pigments in spinach (Spinacia oleracea L.). In: Proceedings of the VI International Symposium on Light in Horticulture; 2009 November 15–19; Tsukuba, Japan. Leuven, Belgium: ISHS; 2011. p. 105– 10. doi:10.17660/ActaHortic.2011.907.12. [Google Scholar] [CrossRef]
19. Perkins-Veazie P , Collins JK , Howard L . Blueberry fruit response to postharvest application of ultraviolet radiation. Postharvest Biol Technol. 2008; 47( 3): 280– 5. doi:10.1016/j.postharvbio.2007.08.002. [Google Scholar] [CrossRef]
20. Li Q , Kubota C . Effects of supplemental light quality on growth and phytochemicals of baby leaf lettuce. Environ Exp Bot. 2009; 67( 1): 59– 64. doi:10.1016/j.envexpbot.2009.06.011. [Google Scholar] [CrossRef]
21. Pérez-Romero LF , Stirling PJ , Hancock RD . Light-Emitting Diodes improve yield, quality and inhibitory effects on digestive enzymes of strawberry. Sci Hortic. 2024; 332: 113192. doi:10.1016/j.scienta.2024.113192. [Google Scholar] [CrossRef]
22. Giampieri F , Alvarez-Suarez JM , Mazzoni L , Romandini S , Bompadre S , Diamanti J , et al. The potential impact of strawberry on human health. Nat Prod Res. 2013; 27( 4–5): 448– 55. doi:10.1080/14786419.2012.706294. [Google Scholar] [CrossRef]
23. Symons GM , Chua YJ , Ross JJ , Quittenden LJ , Davies NW , Reid JB . Hormonal changes during non-climacteric ripening in strawberry. J Exp Bot. 2012; 63( 13): 4741– 50. doi:10.1093/jxb/ers147. [Google Scholar] [CrossRef]
24. Kumar R , Khurana A , Sharma AK . Role of plant hormones and their interplay in development and ripening of fleshy fruits. J Exp Bot. 2013; 65( 16): 4561– 75. doi:10.1093/jxb/eru277. [Google Scholar] [CrossRef]
25. Warner R , Wu BS , MacPherson S , Lefsrud M . A review of strawberry photobiology and fruit flavonoids in controlled environments. Front Plant Sci. 2021; 12: 611893. doi:10.3389/fpls.2021.611893. [Google Scholar] [CrossRef]
26. Janisiewicz WJ , Takeda F , Nichols B , Glenn DM , Jurick WM II , Camp MJ . Use of low-dose UV-C irradiation to control powdery mildew caused by Podosphaera aphanis on strawberry plants. Can J Plant Pathol. 2016; 38( 4): 430– 9. doi:10.1080/07060661.2016.1263807. [Google Scholar] [CrossRef]
27. Salehi F . Application of pulsed light technology for fruits and vegetables disinfection: a review. J Appl Microbiol. 2022; 132( 4): 2521– 30. doi:10.1111/jam.15389. [Google Scholar] [CrossRef]
28. Smith BJ , Stafne ET , Sakhanokho HF , Sampson BJ . Intensity of supplemental greenhouse lighting affects strawberry plant growth, anthracnose infection response, and Colletotrichum pathogen development in culture. HortScience. 2023; 58( 1): 127– 33. doi:10.21273/HORTSCI16888-22. [Google Scholar] [CrossRef]
29. Afrin S , Gasparrini M , Forbes-Hernandez TY , Reboredo-Rodriguez P , Mezzetti B , Varela-López A , et al. Promising health benefits of the strawberry: a focus on clinical studies. J Agric Food Chem. 2016; 64( 22): 4435– 49. doi:10.1021/acs.jafc.6b00857. [Google Scholar] [CrossRef]
30. Yan JW , Ban ZJ , Lu HY , Li D , Poverenov E , Luo ZS , et al. The aroma volatile repertoire in strawberry fruit: a review. J Sci Food Agric. 2018; 98( 12): 4395– 402. doi:10.1002/jsfa.9039. [Google Scholar] [CrossRef]
31. Qin Y , Zhang S , Asghar S , Zhang L , Qin Q , Chen K , et al. Regeneration mechanism of Toyonoka strawberry under different color plastic films. Plant Sci. 2005; 168( 6): 1425– 31. doi:10.1016/j.plantsci.2004.11.016. [Google Scholar] [CrossRef]
32. Rocha PS , Oliveira RP , Scivittaro WB , Santos UL . Light-emitting diodes and BAP concentrations in the in vitro strawberry multiplication. Ciência Rural. 2010; 40: 1922– 8. doi:10.1590/S0103-84782010000900011. [Google Scholar] [CrossRef]
33. Mohamed FH , Omar GF , Ismail MA . In vitro regeneration, proliferation and growth of strawberry under different light treatments. Acta Hortic. 2017; 1155: 361– 8. doi:10.17660/ActaHortic.2017.1155.53. [Google Scholar] [CrossRef]
34. Nhut DT , Takamura T , Watanabe H , Okamoto K , Tanaka M . Responses of strawberry plantlets cultured in vitro under superbright red and blue light-emitting diodes (LEDs). Plant Cell Tissue Organ Cult. 2003; 73( 1): 43– 52. doi:10.1023/A:1022638508007. [Google Scholar] [CrossRef]
35. Hung CD , Hong CH , Jung HB , Kim SK , Van Ket N , Nam MW , et al. Growth and morphogenesis of encapsulated strawberry shoot tips under mixed LEDs. Sci Hortic. 2015; 194: 194– 200. doi:10.1016/j.scienta.2015.08.016. [Google Scholar] [CrossRef]
36. Wu W , Chen L , Liang R , Huang S , Li X , Huang B , et al. The role of light in regulating plant growth, development and sugar metabolism: a review. Front Plant Sci. 2025; 15: 1507628. doi:10.3389/fpls.2024.1507628. [Google Scholar] [CrossRef]
37. Yoshida H , Mizuta D , Fukuda N , Hikosaka S , Goto E . Effects of varying light quality from single-peak blue and red light-emitting diodes during nursery period on flowering, photosynthesis, growth, and fruit yield of everbearing strawberry. Plant Biotechnol. 2016; 33( 4): 267– 76. doi:10.5511/plantbiotechnology.16.0216a. [Google Scholar] [CrossRef]
38. Salisu Jibia S , Panjama K , Inkham C , Sato T , Ohtake N , Ruamrungsri S . Interactive effects of LED spectrum and nitrogen levels on physiological changes and yield of strawberry (Fragaria × ananassa Duch.). Plants. 2024; 14( 1): 89. doi:10.3390/plants14010089. [Google Scholar] [CrossRef]
39. Park Y , Sethi R , Temnyk S . Growth, flowering, and fruit production of strawberry ‘Albion’ in response to photoperiod and photosynthetic photon flux density of sole-source lighting. Plants. 2023; 12( 4): 731. doi:10.3390/plants12040731. [Google Scholar] [CrossRef]
40. Ries J , Park Y . Far-red light in sole-source lighting can enhance the growth and fruit production of indoor strawberries. HortScience. 2024; 59( 6): 799– 805. doi:10.21273/HORTSCI17729-24. [Google Scholar] [CrossRef]
41. Park SW , Kwack Y , Chun C . Growth of runner plants grown in a plant factoryas affected by light intensity and CONTAINERVOLUME. Hortic Sci Technol. 2017; 35( 4): 439– 45. doi:10.12972/kjhst.20170047. [Google Scholar] [CrossRef]
42. Yang J , Song J , Jeong BR . Side lighting enhances morphophysiology and runner formation by upregulating photosynthesis in strawberry grown in controlled environment. Agronomy. 2022; 12( 1): 24. doi:10.3390/agronomy12010024. [Google Scholar] [CrossRef]
43. Zheng J , He D , Ji F . Effects of light intensity and photoperiod on runner plant propagation of hydroponic strawberry transplants under LED lighting. Int J Agric Biol Eng. 2019; 12( 6): 26– 31. doi:10.25165/j.ijabe.20191206.5265. [Google Scholar] [CrossRef]
44. Kim SK , Jeong MS , Park SW , Kim MJ , Na HY , Chun CH . Improvement of runner plant production by increasing photosynthetic photon flux during strawberry transplant propagation in a closed transplant production system. Hortic Sci Technol. 2010; 28( 4): 535– 9. [Google Scholar]
45. Xu X , Hernández R . The effect of light intensity on vegetative propagation efficacy, growth, and morphology of “Albion” strawberry plants in a precision indoor propagation system. Appl Sci. 2020; 10( 3): 1044. doi:10.3390/app10031044. [Google Scholar] [CrossRef]
46. Hamano M , Yamazaki H , Morishita M , Imada S . Effect of chilling and day length on runner of everbearing type strawberry. Acta Hortic. 2009; 842: 671– 4. doi:10.17660/ActaHortic.2009.842.144. [Google Scholar] [CrossRef]
47. Hasan SM , Isam AM , Aziz A , Yusoff WAB . Effect of photoperiod on propagation of strawberry (Fragaria × ananassa Duch.). J Hortic For. 2011; 3( 8): 259– 63. [Google Scholar]
48. Nishizawa T . Effects of photoperiods on the length and number of epidermal cells in runners of strawberry plants. J Jpn Soc Hortic Sci. 1994; 63( 2): 347– 52. doi:10.2503/jjshs.63.347. [Google Scholar] [CrossRef]
49. Ohno Y . Spectral design considerations for white LED color rendering. Opt Eng. 2005; 44( 11): 111302. doi:10.1117/1.2130694. [Google Scholar] [CrossRef]
50. Lee H , Park SW , Pham MD , Hwang H , Chun C . Effect of the light spectrum of white LEDs on the productivity of strawberry transplants in a plant factory with artificial lighting. Hortic Environ Biotechnol. 2020; 61( 6): 971– 9. doi:10.1007/s13580-020-00284-0. [Google Scholar] [CrossRef]
51. Lee H , Park SW , Cui M , Lee B , Minh Pham D , Hwang H , et al. Improvement of strawberry transplant production efficiency by supplementary blue light in a plant factory using white LEDs. Hortic Environ Biotechnol. 2023; 64( 2): 233– 44. doi:10.1007/s13580-022-00493-9. [Google Scholar] [CrossRef]
52. Chen J , Ji F , Gao R , He D . Reducing red light proportion in full-spectrum LEDs enhances runner plant propagation by promoting the growth and development of mother plants in strawberry. Front Plant Sci. 2024; 15: 1465004. doi:10.3389/fpls.2024.1465004. [Google Scholar] [CrossRef]
53. Yang J , Song J , Jeong BR . Flowering and runnering of seasonal strawberry under different photoperiods are affected by intensity of supplemental or night-interrupting blue light. Plants. 2024; 13( 3): 375. doi:10.3390/plants13030375. [Google Scholar] [CrossRef]
54. Li Y , Xiao J , Hu J , Jeong BR . Critical photoperiod and optimal quality of night interruption light for runner induction in June-bearing strawberries. Agronomy. 2021; 11( 10): 1996. doi:10.3390/agronomy11101996. [Google Scholar] [CrossRef]
55. Nishizawa T , Shishido Y , Kudo M , Kumakura H , Hamamoto H . Petiole length, chlorophyll and carbohydrate levels, and photosynthetic rates of June-bearing strawberry plants as influenced by red-light irradiation during storage under cool conditions. Sci Hortic. 1997; 72( 1): 25– 33. doi:10.1016/S0304-4238(97)00113-1. [Google Scholar] [CrossRef]
56. Samuolienė G , Brazaitytė A , Urbonavičiūtė A , Šabajevienė G , Duchovskis P . The effect of red and blue light component on the growth and development of frigo strawberries. Zemdirb-Agric. 2010; 97: 99– 104. [Google Scholar]
57. Black BL , Swartz HJ , Deitzer GF , Butler B , Chandler CK . The effects of conditioning strawberry plug plants under altered red/far-red light environments. HortScience. 2005; 40( 5): 1263– 7. doi:10.21273/HORTSCI.40.5.1263. [Google Scholar] [CrossRef]
58. Takeda F , Glenn DM , Stutte GW . Red light affects flowering under long days in a short-day strawberry cultivar. HortScience. 2008; 43( 7): 2245– 7. doi:10.21273/HORTSCI.43.7.2245. [Google Scholar] [CrossRef]
59. Verhage L , Angenent GC , Immink RGH . Research on floral timing by ambient temperature comes into blossom. Trends Plant Sci. 2014; 19( 9): 583– 91. doi:10.1016/j.tplants.2014.03.009. [Google Scholar] [CrossRef]
60. Yoshida H , Hikosaka S , Goto E , Takasuna H , Kudou T . Effects of light quality and light period on flowering of everbearing strawberry in a closed plant production system. In: Proceedings of the VII International Symposium on Light in Horticultural Systems; 2012 Oct 15–18; Wageningen, The Netherlands. Leuven, Belgium: ISHS; 2012. p. 107– 12. doi:10.17660/ActaHortic.2012.956.9. [Google Scholar] [CrossRef]
61. Rantanen M , Kurokura T , Mouhu K , Pinho P , Tetri E , Halonen L , et al. Light quality regulates flowering in FvFT1/FvTFL1 dependent manner in the woodland strawberry Fragaria vesca. Front Plant Sci. 2014; 5: 271. doi:10.3389/fpls.2014.00271. [Google Scholar] [CrossRef]
62. Yanagi T , Okuda N , Okamoto K . Effects of light quality and quantity on flower initiation of Fragaria chiloensis L. CHI-24-1 grown under 24h day-length. Sci Hortic. 2016; 202: 150– 5. doi:10.1016/j.scienta.2016.02.035. [Google Scholar] [CrossRef]
63. Sidhu V , Bernier-English V , Lamontagne-Drolet M , Gravel V . Effect of light quality and extended photoperiod on flower bud induction during transplant production of day-neutral strawberry cultivars. Can J Plant Sci. 2022; 102( 2): 356– 67. doi:10.1139/cjps-2021-0081. [Google Scholar] [CrossRef]
64. Tsuruyama J , Shibuya T . Effects of far-red light and photoperiod during early growth stages on flower bud development of seed-propagated strawberry seedlings. Sci Hortic. 2023; 317: 112051. doi:10.1016/j.scienta.2023.112051. [Google Scholar] [CrossRef]
65. Zahedi SM , Sarikhani H . Effect of far-red light, temperature, and plant age on morphological changes and induction of flowering of a ‘June-bearing’ strawberry. Hortic Environ Biotechnol. 2016; 57( 4): 340– 7. doi:10.1007/s13580-016-0018-8. [Google Scholar] [CrossRef]
66. Choi HG , Moon BY , Kang NJ . Effects of LED light on the production of strawberry during cultivation in a plastic greenhouse and in a growth chamber. Sci Hortic. 2015; 189: 22– 31. doi:10.1016/j.scienta.2015.03.022. [Google Scholar] [CrossRef]
67. Meng L , Höfte M , Van Labeke MC . Leaf age and light quality influence the basal resistance against Botrytis cinerea in strawberry leaves. Environ Exp Bot. 2019; 157: 35– 45. doi:10.1016/j.envexpbot.2018.09.025. [Google Scholar] [CrossRef]
68. Peng X , Wang B , Wang X , Ni B , Zuo Z . Effects of different colored light-quality selective plastic films on growth, photosynthetic abilities, and fruit qualities of strawberry. Hortic Sci Technol. 2020; 38( 4): 462– 73. doi:10.7235/HORT.20200044. [Google Scholar] [CrossRef]
69. Roosta HR , Bikdeloo M , Ghorbanpour M . The growth, nutrient uptake and fruit quality in four strawberry cultivars under different Spectra of LED supplemental light. BMC Plant Biol. 2024; 24( 1): 179. doi:10.1186/s12870-024-04880-5. [Google Scholar] [CrossRef]
70. Dos Santos Suzana Guiamba H , Zhang X , Sierka E , Lin K , Ali MM , Ali WM , et al. Enhancement of photosynthesis efficiency and yield of strawberry (Fragaria ananassa Duch.) plants via LED systems. Front Plant Sci. 2022; 13: 918038. doi:10.3389/fpls.2022.918038. [Google Scholar] [CrossRef]
71. Ma G , Zhang L , Kato M , Yamawaki K , Kiriiwa Y , Yahata M , et al. Effect of blue and red LED light irradiation on β-cryptoxanthin accumulation in the flavedo of Citrus fruits. J Agric Food Chem. 2012; 60( 1): 197– 201. doi:10.1021/jf203364m. [Google Scholar] [CrossRef]
72. Wu CC , Yen YH , Chang MY , Fang W . Effects of light quality and CO2 concentration on diurnal photosynthetic characteristics of strawberry. In: Proceedings of the VII International Symposium on Light in Horticultural Systems; 2012 Oct 15–18; Wageningen, The Netherlands. Leuven, Belgium: ISHS; 2012. p. 247– 53. doi:10.17660/ActaHortic.2012.956.27. [Google Scholar] [CrossRef]
73. Wei H , Liu C , Hu J , Jeong BR . Quality of supplementary morning lighting (SML) during propagation period affects physiology, stomatal characteristics, and growth of strawberry plants. Plants. 2020; 9( 5): 638. doi:10.3390/plants9050638. [Google Scholar] [CrossRef]
74. El Horri H , Vitiello M , Ceccanti C , Lo Piccolo E , Lauria G , De Leo M , et al. Ultraviolet-to-blue light conversion film affects both leaf photosynthetic traits and fruit bioactive compound accumulation in Fragaria × ananassa. Agronomy. 2024; 14( 7): 1491. doi:10.3390/agronomy14071491. [Google Scholar] [CrossRef]
75. Landi M , Zivcak M , Sytar O , Brestic M , Allakhverdiev SI . Plasticity of photosynthetic processes and the accumulation of secondary metabolites in plants in response to monochromatic light environments: a review. Biochim Biophys Acta Bioenerg. 2020; 1861( 2): 148131. doi:10.1016/j.bbabio.2019.148131. [Google Scholar] [CrossRef]
76. Ogawa T , Ishikawa H , Shimada K , Shibata K . Synergistic action of red and blue light and action spectra for malate formation in guard cells of Vicia faba L. Planta. 1978; 142( 1): 61– 5. doi:10.1007/BF00385121. [Google Scholar] [CrossRef]
77. Bian Z , Yang Q , Li T , Cheng R , Barnett Y , Lu C . Study of the beneficial effects of green light on lettuce grown under short-term continuous red and blue light-emitting diodes. Physiol Plant. 2018; 164( 2): 226– 40. doi:10.1111/ppl.12713. [Google Scholar] [CrossRef]
78. Avendaño-Abarca VH , Alvarado-Camarillo D , Valdez-Aguilar LA , Sánchez-Ortíz EA , González-Fuentes JA , Cartmill AD . Response of strawberry to the substitution of blue light by green light in an indoor vertical farming system. Agronomy. 2023; 13( 1): 99. doi:10.3390/agronomy13010099. [Google Scholar] [CrossRef]
79. Mochizuki Y , Sekiguchi S , Horiuchi N , Aung T , Ogiwara I . Photosynthetic characteristics of individual strawberry (Fragaria × ananassa Duch.) leaves under short-distance lightning with blue, green, and red LED lights. HortScience. 2019; 54( 3): 452– 8. doi:10.21273/HORTSCI13560-18. [Google Scholar] [CrossRef]
80. Li P , Wang Z , Wang X , Liu F , Wang H . Changes in phytohormones and transcriptomic reprogramming in strawberry leaves under different light qualities. Int J Mol Sci. 2024; 25( 5): 2765. doi:10.3390/ijms25052765. [Google Scholar] [CrossRef]
81. Brazaitytė A , Miliauskienė J , Vaštakaitė-Kairienė V , Sutulienė R , Laužikė K , Duchovskis P , et al. Effect of different ratios of blue and red LED light on Brassicaceae microgreens under a controlled environment. Plants. 2021; 10( 4): 801. doi:10.3390/plants10040801. [Google Scholar] [CrossRef]
82. Talukder MR , Asaduzzaman M , Tanaka H , Asao T . Light-emitting diodes and exogenous amino acids application improve growth and yield of strawberry plants cultivated in recycled hydroponics. Sci Hortic. 2018; 239: 93– 103. doi:10.1016/j.scienta.2018.05.033. [Google Scholar] [CrossRef]
83. Lauria G , Lo Piccolo E , Ceccanti C , Paoli L , Giordani T , Guidi L , et al. Supplemental red light more than other wavebands activates antioxidant defenses in greenhouse-cultivated Fragaria × ananassa var. Elsanta plants. Sci Hortic. 2023; 321: 112319. doi:10.1016/j.scienta.2023.112319. [Google Scholar] [CrossRef]
84. Yoneda A , Yasutake D , Hidaka K , Muztahidin N , Miyoshi Y , Kitano M , et al. Effects of supplemental lighting during the period of rapid fruit development on the growth, yield, and energy use efficiency in strawberry plant production. Int Agrophys. 2020; 34( 2): 233– 9. doi:10.31545/intagr/117623. [Google Scholar] [CrossRef]
85. Miyazawa Y , Hikosaka S , Goto E , Aoki T . Effects of light conditions and air temperature on the growth of everbearing strawberry during the vegetative stage. In: Proceedings of the VI International Strawberry Symposium; 2008 Mar 3–7; Huelva, Spain. Leuven, Belgium: ISHS; 2008. p. 817– 20. doi:10.17660/ActaHortic.2009.842.180. [Google Scholar] [CrossRef]
86. Naznin MT , Lefsrud M , Gravel V , Hao X . Using different ratios of red and blue LEDs to improve the growth of strawberry plants. In: Proceedings of the VIII International Symposium on Light in Horticulture; 2016 May 22–26; East Lansing, MI, USA. Leuven, Belgium: ISHS; 2016. p. 125– 30. doi:10.17660/ActaHortic.2016.1134.17. [Google Scholar] [CrossRef]
87. Van Delm T , Melis P , Stoffels K , Vanderbruggen R , Baets W . Advancing the strawberry season in Belgian glasshouses with supplemental assimilation lighting. In: Proceedings of the VIII International Symposium on Light in Horticulture; 2016 May 22–26; East Lansing, MI, USA. Leuven, Belgium: ISHS; 2016. p. 147– 54. doi:10.17660/ActaHortic.2016.1134.20. [Google Scholar] [CrossRef]
88. Stuemky A , Uchanski ME . Supplemental light-emitting diode effects on the growth, fruit quality, and yield of two greenhouse-grown strawberry (Fragaria × ananassa) cultivars. HortScience. 2020; 55( 1): 23– 9. doi:10.21273/HORTSCI14113-19. [Google Scholar] [CrossRef]
89. Hidaka K , Dan K , Imamura H , Miyoshi Y , Takayama T , Sameshima K , et al. Effect of supplemental lighting from different light sources on growth and yield of strawberry. Environ Control Biol. 2013; 51( 1): 41– 7. doi:10.2525/ecb.51.41. [Google Scholar] [CrossRef]
90. Demkura PV , Ballaré CL . UVR8 mediates UV-B-induced Arabidopsis defense responses against Botrytis cinerea by controlling sinapate accumulation. Mol Plant. 2012; 5( 3): 642– 52. doi:10.1093/mp/sss025. [Google Scholar] [CrossRef]
91. de Wit M , Spoel SH , Sanchez-Perez GF , Gommers CMM , Pieterse CMJ , Voesenek LACJ , et al. Perception of low red: far-red ratio compromises both salicylic acid- and jasmonic acid-dependent pathogen defences in Arabidopsis. Plant J. 2013; 75( 1): 90– 103. doi:10.1111/tpj.12203. [Google Scholar] [CrossRef]
92. Xu H , Fu YN , Li TL , Wang R . Effects of different LED light wavelengths on the resistance of tomato against Botrytis cinerea and the corresponding physiological mechanisms. J Integr Agric. 2017; 16( 1): 106– 14. doi:10.1016/S2095-3119(16)61435-1. [Google Scholar] [CrossRef]
93. Gallé Á , Czékus Z , Tóth L , Galgóczy L , Poór P . Pest and disease management by red light. Plant Cell Environ. 2021; 44( 10): 3197– 210. doi:10.1111/pce.14142. [Google Scholar] [CrossRef]
94. Meng L , Van Labeke MC , Höfte M . Timing of light quality affects susceptibility to Botrytis cinerea in strawberry leaves. J Photochem Photobiol B Biol. 2020; 211: 111988. doi:10.1016/j.jphotobiol.2020.111988. [Google Scholar] [CrossRef]
95. Meng L , Höfte M , van Labeke MC . Varying the time of red light exposure influences leaf resistance to different Botrytis cinerea isolates in strawberry. J Plant Dis Prot. 2023; 130( 1): 163– 8. doi:10.1007/s41348-022-00687-4. [Google Scholar] [CrossRef]
96. Magerøy MH , Kowalik EH , Folta KM , Shinkle J . Evidence of physiological phototropin1 (phot1) action in response to UV-C illumination. Plant Signal Behav. 2010; 5( 10): 1204– 10. doi:10.4161/psb.5.10.12413. [Google Scholar] [CrossRef]
97. Ouhibi C , Attia H , Nicot P , Lecompte F , Vidal V , Lachaâl M , et al. Effects of nitrogen supply and of UV-C irradiation on the susceptibility of Lactuca sativa L to Botrytis cinerea and Sclerotinia minor. Plant Soil. 2015; 393( 1): 35– 46. doi:10.1007/s11104-015-2458-9. [Google Scholar] [CrossRef]
98. Vàsquez H , Ouhibi C , Lizzi Y , Azzouz N , Forges M , Bardin M , et al. Pre-harvest hormetic doses of UV-C radiation can decrease susceptibility of lettuce leaves (Lactuca sativa L.) to Botrytis cinerea L. Sci Hortic. 2017; 222: 32– 9. doi:10.1016/j.scienta.2017.04.017. [Google Scholar] [CrossRef]
99. Forges M , Vàsquez H , Charles F , Sari DC , Urban L , Lizzi Y , et al. Impact of UV-C radiation on the sensitivity of three strawberry plant cultivars (Fragaria × ananassa) against Botrytis cinerea. Sci Hortic. 2018; 240: 603– 13. doi:10.1016/j.scienta.2018.06.063. [Google Scholar] [CrossRef]
100. Darras AI , Demopoulos V , Bali I , Tiniakou C . Photomorphogenic reactions in Geranium (Pelargonium × hortotum) plants stimulated by brief exposures of ultraviolet-C irradiation. Plant Growth Regul. 2012; 68( 3): 343– 50. doi:10.1007/s10725-012-9722-2. [Google Scholar] [CrossRef]
101. Smith BJ , Rezazadeh A , Stafne ET , Sakhanokho HF . Effect of light-emitting diodes, ultraviolet-B, and fluorescent supplemental greenhouse lights on strawberry plant growth and response to infection by the anthracnose pathogen Colletotrichum gloeosporioides. HortScience. 2022; 57( 8): 856– 63. doi:10.21273/HORTSCI16591-22. [Google Scholar] [CrossRef]
102. Malekzadeh MR , Roosta HR , Esmaeilizadeh M , Dabrowski P , Kalaji HM . Improving strawberry plant resilience to salinity and alkalinity through the use of diverse spectra of supplemental lighting. BMC Plant Biol. 2024; 24( 1): 252. doi:10.1186/s12870-024-04984-y. [Google Scholar] [CrossRef]
103. Esmaeilizadeh M , Malekzadeh Shamsabad MR , Roosta HR , Dąbrowski P , Rapacz M , Zieliński A , et al. Manipulation of light spectrum can improve the performance of photosynthetic apparatus of strawberry plants growing under salt and alkalinity stress. PLoS One. 2021; 16( 12): e0261585. doi:10.1371/journal.pone.0261585. [Google Scholar] [CrossRef]
104. Razzak MA , Asaduzzaman M , Tanaka H , Asao T . Effects of supplementing green light to red and blue light on the growth and yield of lettuce in plant factories. Sci Hortic. 2022; 305: 111429. doi:10.1016/j.scienta.2022.111429. [Google Scholar] [CrossRef]
105. Okamoto H , Ducreux LJM , Allwood JW , Hedley PE , Wright A , Gururajan V , et al. Light regulation of chlorophyll and glycoalkaloid biosynthesis during Tuber greening of potato S. tuberosum. Front Plant Sci. 2020; 11: 753. doi:10.3389/fpls.2020.00753. [Google Scholar] [CrossRef]
106. Malekzadeh Shamsabad MR , Esmaeilizadeh M , Roosta HR , Dąbrowski P , Telesiński A , Kalaji HM . Supplemental light application can improve the growth and development of strawberry plants under salinity and alkalinity stress conditions. Sci Rep. 2022; 12( 1): 9272. doi:10.1038/s41598-022-12925-8. [Google Scholar] [CrossRef]
107. Malekzadeh MR , Roosta HR , Kalaji HM . Enhancing strawberry resilience to saline, alkaline, and combined stresses with light spectra: impacts on growth, enzymatic activity, nutrient uptake, and osmotic regulation. BMC Plant Biol. 2024; 24( 1): 1038. doi:10.1186/s12870-024-05755-5. [Google Scholar] [CrossRef]
108. Sønsteby A , Opstad N , Heide OM . Environmental manipulation for establishing high yield potential of strawberry forcing plants. Sci Hortic. 2013; 157: 65– 73. doi:10.1016/j.scienta.2013.04.014. [Google Scholar] [CrossRef]
109. Wang R , Eguchi M , Gui Y , Iwasaki Y . Evaluating the effect of light intensity on flower development uniformity in strawberry (Fragaria × ananassa) under early induction conditions in forcing culture. HortScience. 2020; 55( 5): 670– 5. doi:10.21273/HORTSCI14917-20. [Google Scholar] [CrossRef]
110. Harsonowati W , Masrukhin , Narisawa K . Prospecting the unpredicted potential traits of Cladophialophora chaetospira SK51 to alter photoperiodic flowering in strawberry, a perennial SD plant. Sci Hortic. 2022; 295: 110835. doi:10.1016/j.scienta.2021.110835. [Google Scholar] [CrossRef]
111. Ren C , Jiang L , Chen W , Wang Z . Effect of different post-flowering photoperiods on main agronomic traits of strawberry (Fragaria × ananassa Duch. cv. Akihime). Agronomy. 2024; 14( 9): 2039. doi:10.3390/agronomy14092039. [Google Scholar] [CrossRef]
112. Muñoz-Avila JC , Prieto C , Sánchez-Sevilla JF , Amaya I , Castillejo C . Role of FaSOC1 and FaCO in the seasonal control of reproductive and vegetative development in the perennial crop Fragaria × ananassa. Front Plant Sci. 2022; 13: 971846. doi:10.3389/fpls.2022.971846. [Google Scholar] [CrossRef]
113. Díaz-Galián MV , Torres M , Sanchez-Pagán JD , Navarro PJ , Weiss J , Egea-Cortines M . Enhancement of strawberry production and fruit quality by blue and red LED lights in research and commercial greenhouses. S Afr N J Bot. 2021; 140: 269– 75. doi:10.1016/j.sajb.2020.05.004. [Google Scholar] [CrossRef]
114. D’Aloia M , Bonhomme D , Bouché F , Tamseddak K , Ormenese S , Torti S , et al. Cytokinin promotes flowering of Arabidopsis via transcriptional activation of the FT paralogue TSF. Plant J. 2011; 65( 6): 972– 9. doi:10.1111/j.1365-313X.2011.04482.x. [Google Scholar] [CrossRef]
115. Kadowaki M , Yano A , Ishizu F , Tanaka T , Noda S . Effects of greenhouse photovoltaic array shading on Welsh onion growth. Biosyst Eng. 2012; 111( 3): 290– 7. doi:10.1016/j.biosystemseng.2011.12.006. [Google Scholar] [CrossRef]
116. Wang S , Liu X , Liu X , Xue J , Ren X , Zhai Y , et al. The red/blue light ratios from light-emitting diodes affect growth and flower quality of Hippeastrum hybridum ‘Red Lion’. Front Plant Sci. 2022; 13: 1048770. doi:10.3389/fpls.2022.1048770. [Google Scholar] [CrossRef]
117. Smeets L . Effect of the light intensity during flowering on stamen development in the strawberry cultivars ‘Karina’ and ‘Sivetta’. Sci Hortic. 1980; 12( 4): 343– 6. doi:10.1016/0304-4238(80)90049-7. [Google Scholar] [CrossRef]
118. Yoshida Y , Ozaki E , Murakami K , Goto T . Flower induction in June-bearing strawberry by intermittent low temperature storage. J Japan Soc Hort Sci. 2012; 81( 4): 343– 9. doi:10.2503/jjshs1.81.343. [Google Scholar] [CrossRef]
119. Eshghi S , Tafazoli E , Dokhani S , Rahemi M , Emam Y . Changes in carbohydrate contents in shoot tips, leaves and roots of strawberry (Fragaria × ananassa Duch.) during flower-bud differentiation. Sci Hortic. 2007; 113( 3): 255– 60. doi:10.1016/j.scienta.2007.03.014. [Google Scholar] [CrossRef]
120. Cocco C , Gonçalves MA , Reisser Junior C , Marafon AC , Antunes LEC . Carbohydrate content and development of strawberry transplants from Rio grande do Sul and imported. Rev Bras Frutic. 2016; 38( 4): e-581. doi:10.1590/0100-29452016581. [Google Scholar] [CrossRef]
121. Nadalini S , Zucchi P , Andreotti C . Effects of blue and red LED lights on soilless cultivated strawberry growth performances and fruit quality. Eur J Hortic Sci. 2017; 82( 1): 12– 20. doi:10.17660/eJHS.2017/82.1.2. [Google Scholar] [CrossRef]
122. Torres-Quezada EA , Zotarelli L , Whitaker VM , Santos BM , Hernandez-Ochoa I . Initial crown diameter of strawberry bare-root transplants affects early and total fruit yield. HortTechnology. 2015; 25( 2): 203– 8. doi:10.21273/HORTTECH.25.2.203. [Google Scholar] [CrossRef]
123. Hidaka K , Dan K , Imamura H , Takayama T , Sameshima K , Okimura M . Variety comparison of effect of supplemental lighting with LED on growth and yield in forcing culture of strawberry. Environ Control Biol. 2015; 53( 3): 135– 43. doi:10.2525/ecb.53.135. [Google Scholar] [CrossRef]
124. Verma P , Singh G , Singh SK , Ahmad Mirza A , Bakshi M , Anmol , et al. Modulating productivity of strawberries (Fragaria × ananassa Duch.) through artificial full-spectrum light in indoor vertical farming. J Soil Sci Plant Nutr. 2025; 25( 1): 1219– 33. doi:10.1007/s42729-024-02197-8. [Google Scholar] [CrossRef]
125. Nakayama M , Nakazawa Y . Effects of environmental control and LED supplemental lighting on strawberry growth and yield in a subtropical climate. Sci Hortic. 2023; 321: 112349. doi:10.1016/j.scienta.2023.112349. [Google Scholar] [CrossRef]
126. Miao L , Zhang Y , Yang X , Xiao J , Zhang H , Zhang Z , et al. Colored light-quality selective plastic films affect anthocyanin content, enzyme activities, and the expression of flavonoid genes in strawberry (Fragaria × ananassa) fruit. Food Chem. 2016; 207: 93– 100. doi:10.1016/j.foodchem.2016.02.077. [Google Scholar] [CrossRef]
127. Guan L , Dai Z , Wu BH , Wu J , Merlin I , Hilbert G , et al. Anthocyanin biosynthesis is differentially regulated by light in the skin and flesh of white-fleshed and teinturier grape berries. Planta. 2016; 243( 1): 23– 41. doi:10.1007/s00425-015-2391-4. [Google Scholar] [CrossRef]
128. Zhu X , Trouth F , Yang T . Preharvest UV-B treatment improves strawberry quality and extends shelf life. Horticulturae. 2023; 9( 2): 211. doi:10.3390/horticulturae9020211. [Google Scholar] [CrossRef]
129. Zhang Y , Jiang L , Li Y , Chen Q , Ye Y , Zhang Y , et al. Effect of red and blue light on anthocyanin accumulation and differential gene expression in strawberry (Fragaria × ananassa). Molecules. 2018; 23( 4): 820. doi:10.3390/molecules23040820. [Google Scholar] [CrossRef]
130. Kim SK , Bae RN , Chun CH . Changes in bioactive compounds contents of ‘Maehyang’ and ‘Seolhyang’ strawberry fruits by UV light illumination. Hortic Sci Technol. 2011; 29( 3): 172– 80. [Google Scholar]
131. Tang N , Wang J , Zhang B , Chen H , Qiu M . Chromatic effects of supplemental light on the fruit quality of strawberries. Horticulturae. 2023; 9( 12): 1333. doi:10.3390/horticulturae9121333. [Google Scholar] [CrossRef]
132. Pan J , Vicente AR , Martínez GA , Chaves AR , Civello PM . Combined use of UV-C irradiation and heat treatment to improve postharvest life of strawberry fruit. J Sci Food Agric. 2004; 84( 14): 1831– 8. doi:10.1002/jsfa.1894. [Google Scholar] [CrossRef]
133. Xie Z , Fan J , Charles MT , Charlebois D , Khanizadeh S , Rolland D , et al. Preharvest ultraviolet-C irradiation: influence on physicochemical parameters associated with strawberry fruit quality. Plant Physiol Biochem. 2016; 108: 337– 43. doi:10.1016/j.plaphy.2016.07.026. [Google Scholar] [CrossRef]
134. Pombo MA , Dotto MC , Martínez GA , Civello PM . UV-C irradiation delays strawberry fruit softening and modifies the expression of genes involved in cell wall degradation. Postharvest Biol Technol. 2009; 51( 2): 141– 8. doi:10.1016/j.postharvbio.2008.07.007. [Google Scholar] [CrossRef]
135. Hanenberg MA , Janse J , Verkerke W . LED light to improve strawberry flavour, quality and production. In: Proceedings of the International Symposium on Innovation in Integrated and Organic Horticulture (INNOHORT); 2015 Jun 8–12; Avignon, France. p. 207– 12. doi:10.17660/ActaHortic.2016.1137.29. [Google Scholar] [CrossRef]
136. Xu X , Zhang Y , Li Y , Liao S , Sun Y , Liu F . Supplemental light and silicon improved strawberry fruit size and sugars concentration under both full and deficit irrigation. Sci Hortic. 2023; 313: 111912. doi:10.1016/j.scienta.2023.111912. [Google Scholar] [CrossRef]
137. Pérez-Romero LF , Hancock RD . Polyphenols content of strawberry fruit grown under Light-Emitting Diodes (LED). Sci Hortic. 2025; 354: 114298. doi:10.1016/j.scienta.2025.114298. [Google Scholar] [CrossRef]
138. Loughrin JH , Kasperbauer MJ . Aroma of fresh strawberries is enhanced by ripening over red versus black mulch. J Agric Food Chem. 2002; 50( 1): 161– 5. doi:10.1021/jf010950j. [Google Scholar] [CrossRef]
139. Wang SY , Zheng W , Galletta GJ . Cultural system affects fruit quality and antioxidant capacity in strawberries. J Agric Food Chem. 2002; 50( 22): 6534– 42. doi:10.1021/jf020614i. [Google Scholar] [CrossRef]
140. Qiu J , Cai C , Shen M , Gu X , Zheng L , Sun L , et al. Responses of growth, yield and fruit quality of strawberry to elevated CO2, LED supplemental light, and their combination in autumn through spring greenhouse production. Plant Growth Regul. 2024; 102( 2): 351– 65. doi:10.1007/s10725-023-01065-2. [Google Scholar] [CrossRef]
141. Kanto T , Matsuura K , Ogawa T , Yamada M , Ishiwata M , Usami T , et al. A new UV-B lighting system controls powdery mildew of strawberry. In: Proceedings of the VII International Strawberry Symposium; 2012 Feb 18–22; Beijing, China. Leuven, Belgium: ISHS; 2014. p. 655– 60. doi:10.17660/ActaHortic.2014.1049.101. [Google Scholar] [CrossRef]
142. Onofre RB , Gadoury DM , Stensvand A , Bierman A , Rea M , Peres NA . Use of ultraviolet light to suppress powdery mildew in strawberry fruit production fields. Plant Dis. 2021; 105( 9): 2402– 9. doi:10.1094/PDIS-04-20-0781-RE. [Google Scholar] [CrossRef]
143. Xu F , Shi L , Chen W , Cao S , Su X , Yang Z . Effect of blue light treatment on fruit quality, antioxidant enzymes and radical-scavenging activity in strawberry fruit. Sci Hortic. 2014; 175: 181– 6. doi:10.1016/j.scienta.2014.06.012. [Google Scholar] [CrossRef]
144. Wang L , Luo Z , Yang M , Liang Z , Qi M , Dong Y , et al. The action of RED light: specific elevation of pelargonidin-based anthocyanin through ABA-related pathway in strawberry. Postharvest Biol Technol. 2022; 186: 111835. doi:10.1016/j.postharvbio.2022.111835. [Google Scholar] [CrossRef]
145. Chong L , Ghate V , Zhou W , Yuk HG . Developing an LED preservation technology to minimize strawberry quality deterioration during distribution. Food Chem. 2022; 366: 130566. doi:10.1016/j.foodchem.2021.130566. [Google Scholar] [CrossRef]
146. Hoffmann TG , Finardi S , de Oliveira JT , Mueller E , Bertoli SL , de Souza CK . Comparative study of white LED light and dark condition in domestic refrigerator on reducing postharvest strawberries waste: south Brazil. Future Food J Food Agric Soc. 2022; 10( 2): 1– 10. [Google Scholar]
147. Lu W , Li W , Zhao K , Bai X , Zhang Y , Li Q , et al. Blue light irradiation combined with low-temperature storage further enhances postharvest quality of strawberries through improving antioxidant defense and cell wall metabolic activities. Food Chem X. 2024; 25: 102115. doi:10.1016/j.fochx.2024.102115. [Google Scholar] [CrossRef]
148. Zhang Y , Li S , Deng M , Gui R , Liu Y , Chen X , et al. Blue light combined with salicylic acid treatment maintained the postharvest quality of strawberry fruit during refrigerated storage. Food Chem X. 2022; 15: 100384. doi:10.1016/j.fochx.2022.100384. [Google Scholar] [CrossRef]
149. Liu Q , Ding H , Zhang T , Zhou D , Zhu T , Pan L , et al. Effect on lipoxygenase pathway and flavor stabilization of postharvest strawberries by intense pulsed light treatment. Postharvest Biol Technol. 2023; 204: 112472. doi:10.1016/j.postharvbio.2023.112472. [Google Scholar] [CrossRef]
150. Fu X , Cheng S , Zhang Y , Du B , Feng C , Zhou Y , et al. Differential responses of four biosynthetic pathways of aroma compounds in postharvest strawberry (Fragaria × ananassa Duch.) under interaction of light and temperature. Food Chem. 2017; 221: 356– 64. doi:10.1016/j.foodchem.2016.10.082. [Google Scholar] [CrossRef]
151. Ortiz Araque LC , Rodoni LM , Darré M , Ortiz CM , Civello PM , Vicente AR . Cyclic low dose UV-C treatments retain strawberry fruit quality more effectively than conventional pre-storage single high fluence applications. LWT. 2018; 92: 304– 11. doi:10.1016/j.lwt.2018.02.050. [Google Scholar] [CrossRef]
152. Ortiz-Solà J , Colás-Medà P , Nicolau-Lapeña I , Alegre I , Abadias M , Viñas I . Pathogenic potential of the surviving Salmonella Enteritidis on strawberries after disinfection treatments based on ultraviolet-C light and peracetic acid. Int J Food Microbiol. 2022; 364: 109536. doi:10.1016/j.ijfoodmicro.2022.109536. [Google Scholar] [CrossRef]
153. Cao X , Huang R , Chen H . Evaluation of food safety and quality parameters for shelf life extension of pulsed light treated strawberries. J Food Sci. 2019; 84( 6): 1494– 500. doi:10.1111/1750-3841.14613. [Google Scholar] [CrossRef]
154. Luksiene Z , Buchovec I , Viskelis P . Impact of high-power pulsed light on microbial contamination, health promoting components and shelf life of strawberries. Food Technol Biotechnol. 2013; 51( 2): 284. [Google Scholar]
155. Suthaparan A , Zhou G , Veerabagu M , Zhu P . Blue lighting combined with cold storage temperature can suppress gray mold in strawberry fruit. Postharvest Biol Technol. 2024; 218: 113148. doi:10.1016/j.postharvbio.2024.113148. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools