 Open Access
Open Access
CASE REPORT
Late Embolization of a Ductal Stent in Pulmonary Atresia with Intact Ventricular Septum Successfully Managed by Urgent Central Shunt Reconstruction: A Case Report
1 Department of Pediatric and Congenital Heart Surgery, Hospital Regional de Alta Especialidad del Bajío, Servicios de Salud del Instituto Mexicano del Seguro Social para el Bienestar (IMSS-BIENESTAR), León, Mexico
2 Department of Pediatric Cardiology, Hospital Regional de Alta Especialidad del Bajío, Servicios de Salud del Instituto Mexicano del Seguro Social para el Bienestar (IMSS-BIENESTAR), León, Mexico
3 Department of Cardiothoracic Surgery, Hospital Regional de Alta Especialidad del Bajío, Servicios de Salud del Instituto Mexicano del Seguro Social para el Bienestar (IMSS-BIENESTAR), León, Mexico
4 Department of Pediatric Cardiac and Congenital Heart Surgery, Instituto Nacional de Cardiología Ignacio Chávez, Ciudad de México, Mexico
5 Department of Research, Hospital Regional de Alta Especialidad del Bajío, Servicios de Salud del Instituto Mexicano del Seguro Social para el Bienestar (IMSS-BIENESTAR), León, Mexico
* Corresponding Authors: Edel Rafael Rodea-Montero. Email: ,
Structural and Congenital Heart Disease 2026, 21(1), 6 https://doi.org/10.32604/schd.2026.071470
Received 06 August 2025; Accepted 18 March 2026; Issue published 31 March 2026
Abstract
We report a full-term newborn with pulmonary atresia with an intact ventricular septum who developed acute hypoxemia after ductal stent embolization. Emergency surgery was performed to remove the stent and create a central systemic-to-pulmonary shunt using a 3.5 mm GORE-TEX® graft via a modified Gates, Laks, and Johnson technique. Postoperative recovery was favorable. At 24 months, the shunt remained patent with adequate pulmonary artery growth. This case underscores prompt surgical rescue and supports central shunting as an effective mid-term solution.Keywords
Pulmonary atresia with an intact ventricular septum (PAIVS) is a rare congenital heart defect characterized by complete pulmonary valve obstruction and a hypoplastic right ventricle, without a ventricular septal defect [1,2]. Its estimated prevalence ranges from 4 to 8 per 100,000 live births [3], with a slight male predominance [4]. Most cases present in the neonatal period with central cyanosis and hypoxemia. Without intervention, progressive ductal closure leads to circulatory collapse, requiring immediate prostaglandin infusion to maintain ductal patency.
Initial palliation in duct-dependent pulmonary circulation includes ductal stent placement or a modified Blalock-Taussig-Thomas shunt [5]. Ductal stenting has become increasingly adopted in centers with interventional expertise, particularly in neonates with favorable ductal anatomy, representing a substantial proportion of initial palliations. Stenting is less invasive and associated with lower morbidity [6]. However, late stent embolization, although rare (0.84%), is a life-threatening complication requiring prompt intervention [7].
A full-term female neonate, born to a 25-year-old mother with two prior unexplained pregnancy losses, was delivered by scheduled cesarean section at 38 weeks of gestation. No detailed information regarding the cardiac status of the previous fetuses was available, and no fetal autopsy or genetic testing had been performed. The current neonate had a birth weight of 3100 g, length 51 cm, with an Apgar score of 9 and Silverman Anderson score of 0. The initial postnatal course was unremarkable, and the patient was discharged at 24 h.
This case report was prepared in accordance with the ethical principles of the Declaration of Helsinki. The study was approved by the Hospital Regional de Alta Especialidad del Bajío (HRAEB) Research Ethics Committee (code: CEI-049-2025). Informed consent for publication was obtained from the patient’s parents.
On day five of life, she developed central cyanosis, tachypnea, feeding difficulty, and drowsiness. At a secondary care facility, oxygen saturation was 74%. Chest radiography revealed Grade III cardiomegaly with poor pulmonary circulation. Echocardiography confirmed pulmonary atresia with intact ventricular septum (PAIVS), a hypoplastic right ventricle, a wide atrial septal defect, and a patent ductus arteriosus. Prostaglandin E1 infusion (0.02 μg/kg/min) was initiated and the patient was transferred to our tertiary center.
Upon admission, vital signs were as follows: heart rate 158 bpm, respiratory rate 69 breaths per minute, weak pulses, blood pressure 71/34 mmHg, temperature 37.4°C, and oxygen saturation 86%. Laboratory tests showed hemoglobin 14.9 g/dL, leukocytosis (20,190/μL), and normal renal function. Arterial blood gas analysis demonstrated pH 7.36, PCO2 30 mmHg, PaO2 44 mmHg, lactate 0.8 mmol/L, and oxygen saturation 75%. Echocardiography confirmed PAIVS, pulmonary branches of adequate size, absence of collaterals, and a large ductus arteriosus with left-to-right shunting. The ductus arteriosus was elongated and conical in shape, with distal narrowing, arising immediately distal to the origin of the left subclavian artery and coursing with mild tortuosity before inserting into the origin of the left pulmonary artery. The duct measured 9.05 mm at aortic end and 3.40 mm at pulmonary end, with a total length of 16.30 mm. The straight-line distance between the aortic origin and the pulmonary insertion was 14.20 mm, resulting in a ductal curvature index (DCI) of 0.13, indicating mild tortuosity. Distal narrowing was quantified as a 62% reduction from the aortic to the pulmonary end.
The right ventricle showed no evidence of right ventricular–dependent coronary circulation, although intramyocardial sinusoids were present. It was bipartite, comprising the inlet and trabecular portions, with complete valvular pulmonary atresia and moderate tricuspid regurgitation. The tricuspid valve annulus measured 5 mm (Z-score −4.65), and the pulmonary valve annulus measured 2 mm (Z-score −10.2). Right ventricular systolic pressure was estimated at 80 mmHg.
Once ductal patency was confirmed, prostaglandin infusion was cautiously weaned. During the observation period, the patient developed desaturation and bradycardia, consistent with ductal dependence. The prostaglandin infusion was therefore reinstated to stabilize the patient prior to cardiac catheterization.
Cardiac catheterization was performed via a retrograde femoral approach using a 4 Fr introducer sheath. Heparin dose of 10 U/kg was infused. The aorta was reached, and the ductus arteriosus was cannulated through the aortic end. A guidewire was advanced into the left pulmonary artery, and the coronary catheter with the mounted stent (Xience SierraTM, Abbott, 23 mm length, 4 mm diameter), was advanced over the wire; the 4 mm diameter was selected based on the patient’s weight at the time of catheterization (2700 g) in accordance with institutional protocol and published recommendations indicating that the duct is stented to 3.5–4 mm in normal term infants weighing 2.5–4 kg [8]. The stent was then deployed with balloon inflation at 12 atmospheres for three seconds. Angiography confirmed stent in position within the ductus arteriosus immediately after implantation, Fig. 1A, and adequate bilateral pulmonary artery flow. Prostaglandin infusion was not resumed after stent implantation, and acetylsalicylic acid (5 mg/kg/day) was started once hemodynamic stability and the absence of bleeding were confirmed.
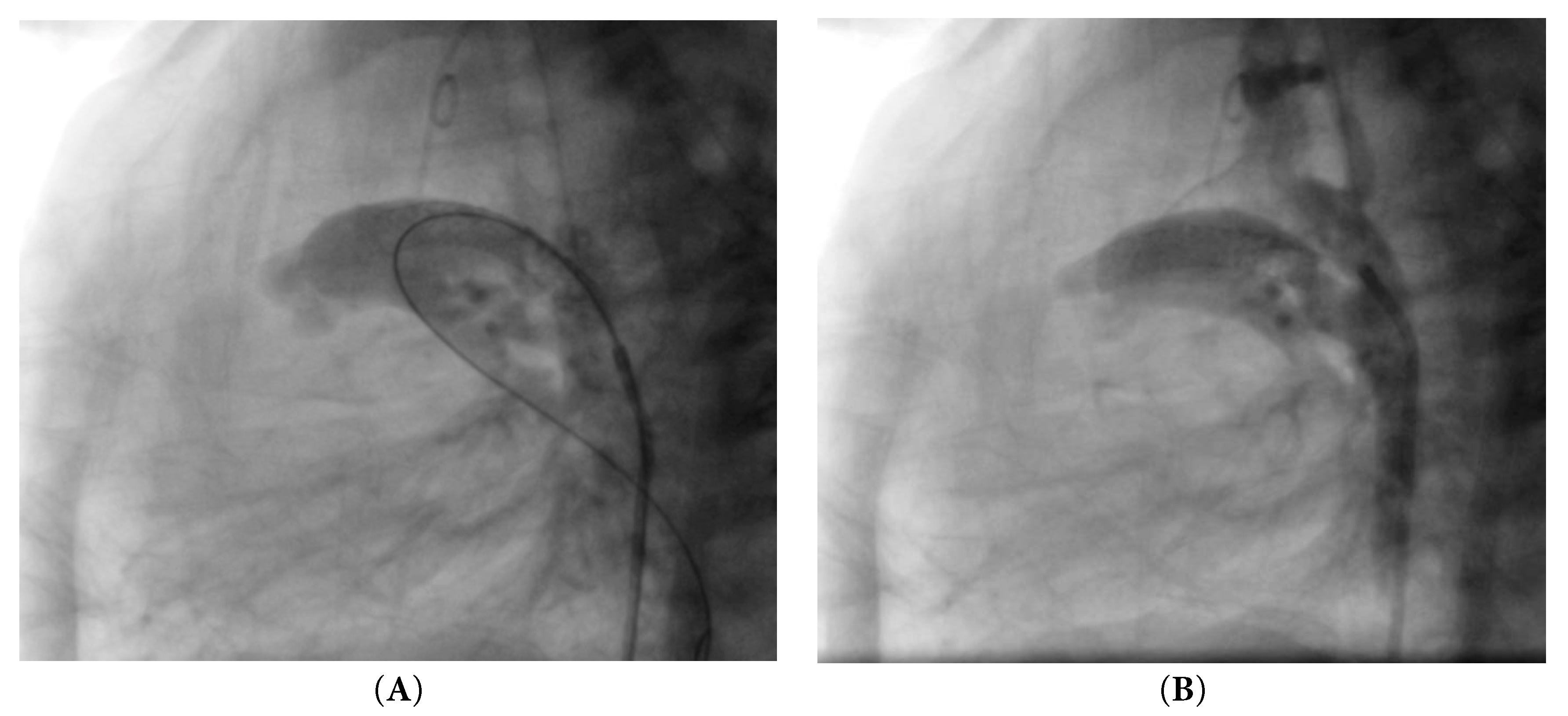
Figure 1: Angiography. (A) Stent in position immediately after implantation. (B) Stent embolization.
Six hours later, the patient deteriorated, presenting with tachycardia (166 bpm), hypotension (61/29 mmHg), desaturation (67%), along with a respiratory rate of 46 bpm and a temperature of 36.3°C. Arterial blood gas analysis revealed pH 7.34, PCO2 46 mmHg, PaO2 42 mmHg, lactate 5.70 mmol/L, and oxygen saturation 64%. Urgent echocardiography and subsequent angiography showed complete embolization of the stent into the main pulmonary artery, Fig. 1B, resulting in cessation of ductal flow. Given the patient’s profound instability, characterized by severe desaturation and bradycardia following stent embolization into the main pulmonary artery, immediate surgical intervention was prioritized. The critical condition did not allow for an attempt at a second stent, and the patient was therefore taken urgently to the operating room.
Urgent surgery was performed by a median sternotomy. Heparin dose of 300 U/kg was infused, and after bicaval and aortic cannulation, the ductus arteriosus was cinched, and cardiopulmonary bypass was started under normothermic conditions. The embolized stent was carefully removed by a longitudinal incision in the lateral aspect of the pulmonary trunk; the length of the incision was similar to the diameter of the 3.5 mm GORE-TEX® graft (WL Gore & Associates, Flagstaff, AZ, USA).
A central systemic-to-pulmonary shunt was constructed with the GORE-TEX® graft using 7–0 polypropylene running suture, as shown in Fig. 2. C-clamps were placed with anastomosis between each of the terminal ends of the graft and the lateral aspect of the pulmonary trunk and the anterolateral aspect of the ascending aorta, maintaining a perpendicular relationship with the graft for minimizing the risk of kinking. The graft was de-aired, and its patency was verified before the clamps were removed and the sutures were adjusted.
Successful pump weaning and reversion were achieved with protamine (4 mg/kg). A palpable continuous flow and an audible murmur were documented with an oxygen saturation of 78%. Double ligation of the ductus arteriosus was performed. Upon completion, the patency of the shunt was confirmed by echocardiography.
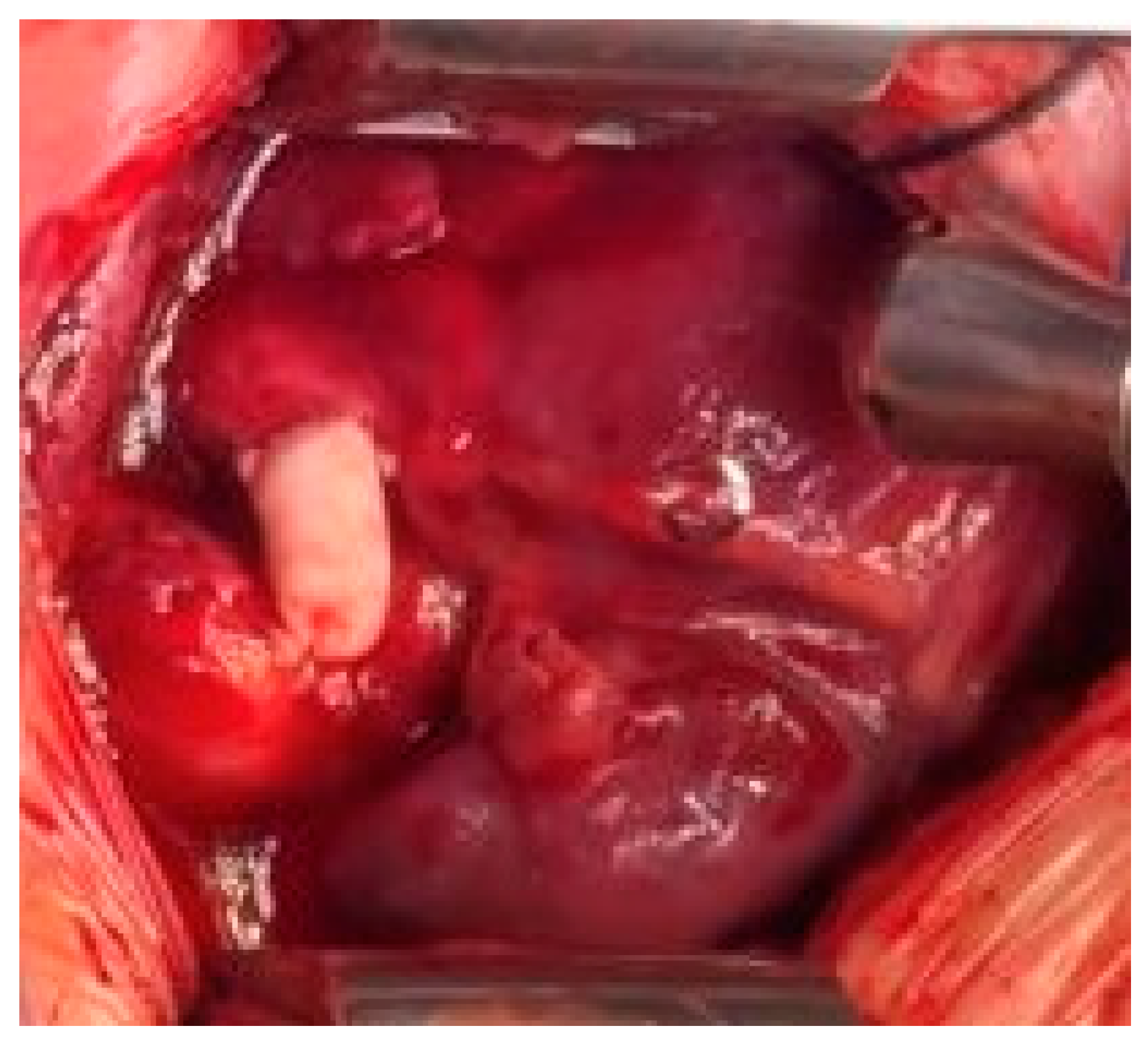
Figure 2: Intraoperative photograph of the central systemic-to-pulmonary shunt with a 3.5 mm GORE-TEX® graft.
Postoperatively, the patient was admitted to the ICU and was administered dopamine (5 μg/kg/min), which was progressively withdrawn. Intravenous furosemide (3 mg/kg/h) was administered for the first six hours, and the patient was extubated in the first 12 h. Acetylsalicylic acid was reinitiated after bleeding was ruled out.
The echocardiographic and clinical follow-up revealed a patent shunt, without signs of overload, with stable saturation (75–85%) until hospital discharge, with monthly multidisciplinary follow-up in cardiology and nutrition.
The choice of a central shunt over a Modified Blalock-Taussig-Thomas shunt was dictated by several factors. First, the patient’s profound hemodynamic instability required institution of cardiopulmonary bypass, which not only allowed safe retrieval of the embolized stent but also enabled construction of the central shunt through the same sternotomy and pulmonary trunk incision, thereby minimizing additional vascular trauma. Second, the patient’s low weight (2700 g) and the relatively larger caliber of the ascending aorta and main pulmonary artery provided more favorable vessel dimensions compared to the smaller right subclavian and right pulmonary arteries. Third, especially in neonates, the modified Blalock-Taussig-Thomas shunt is associated with an increased incidence of iatrogenic pulmonary artery deformation. Finally, the right pulmonary artery was preserved to maintain optimal surgical options for future staged palliation, such as Glenn or Fontan procedures, should a univentricular pathway be required.
In this patient with PAIVS and a severely hypoplastic right ventricle, the decision to avoid initial right ventricular decompression was based on objective severity markers. The right ventricular was bipartite, and the tricuspid valve annulus (5 mm; Z-score −4.65) together with the pulmonary valve annulus (2 mm; Z-score −10.2) confirmed severe inlet and outlet hypoplasia. In our institutional protocol, right ventricular decompression is not pursued when the tricuspid valve Z-score is <−3 or when the pulmonary valve annulus <−4. Therefore, a central systemic-to-pulmonary shunt was chosen over upfront right ventricular decompression due to the limited size and capacity of the right ventricle to support antegrade pulmonary flow. This strategy allowed stabilization, maintenance of pulmonary blood flow, and facilitation of pulmonary artery growth. At 14 months postoperatively, right ventricular decompression was performed as a second-stage procedure, resulting in adequate antegrade flow and further growth of the right ventricle.
At 24 months postsurgery, when the patient had a weight of 10 kg and a height of 80 cm, diagnostic catheterization was performed as part of the protocol for the transition to definitive palliation or repair. A patent central shunt with well-developed pulmonary branches (right 7.51 mm and left 9.38 mm) was observed (McGoon 2.59; Nakata 238.5 mm2/m2). The abdominal aorta measured 6.52 mm. Pulmonary artery systolic and diastolic pressures were 26 and 13 mmHg, respectively, with a mean pressure of 17 mmHg and a calculated pulmonary vascular resistance to systemic vascular resistance (PVR/SVR) of 0.57. The right ventricle remained hypoplastic, with a measured systolic excursion of the tricuspid annulus of 10 mm, indicating persistent reduced right ventricular systolic function. These findings suggested adequate pulmonary artery growth but limited right ventricular contribution to forward flow. A staged single-ventricle pathway was therefore considered the most likely course, with a bidirectional Glenn anticipated as the next step pending further assessment of right ventricular growth and function.
This case illustrates a rare but relevant outcome in patient with a PAIVS initially managed with a ductal stent. Recent analyses, such as that of Mini et al. [5], have shown that although ductal stenting offers advantages over surgical shunts (including less invasiveness and more physiological pulmonary artery growth), complications such as stent instability or embolization can significantly worsen clinical status and impair the outcomes of emergent surgical shunts, increasing adverse events. This contextualizes the severity of the complication observed in our patient and underscores the importance of prompt recognition and timely surgical rescue. Stent embolization represents a critical complication; some options for its treatment are percutaneous repositioning or placement of a second stent. However, in cases of hemodynamic instability, urgent surgical intervention may be needed [9].
The ductus arteriosus in this patient was nearly straight, with a ductal curvature index (DCI) of 0.13, indicating mild tortuosity, which is generally favorable for stent placement [10]. However, the duct exhibited marked distal narrowing, with a 62% reduction from the aortic to the pulmonary end. While the low DCI suggested technical feasibility, the significant taper posed a risk for stent migration and suboptimal distal flow. In addition to this anatomical configuration, other procedural factors may have contributed to the late embolization. The 4 mm stent, although commonly used in neonates, may have provided limited anchorage at the broader aortic end, raising the possibility of relative undersizing in this segment. Likewise, variations in intraprocedural anticoagulation strategies or in the timing of antiplatelet initiation may influence early thrombus formation and stent stability. Finally, withdrawal of prostaglandin after the intervention may have altered ductal wall tone and geometry, decreasing the forces maintaining stent position. Together, these anatomical and periprocedural elements likely contributed to the embolization observed, underscoring the need for careful stent selection and planning in ducts with severe distal narrowing despite minimal curvature.
The surgical urgency was based on avoiding circulatory collapse secondary to the loss of pulmonary flow. Surgical management by means of median sternotomy with cardiopulmonary bypass was selected because of the hemodynamic instability of the patient, as described by Brooks et al. [9]. This allowed safe dissection, extraction of the stent and creation of the central systemic-to-pulmonary shunt. The creation of a central systemic-to-pulmonary shunt, although technically demanding, constitutes an effective solution since, in addition to restoring systemic oxygenation, it facilitates the growth of pulmonary branches, optimizing the conditions for future palliation or repair [10].
The central systemic-to-pulmonary shunt has proven to be feasible and effective in neonates with pulmonary atresia and diminutive pulmonary arteries, as it promotes more balanced pulmonary arterial development compared to the modified Blalock–Taussig-Thomas shunt, which in some cases may lead to branch distortion [11,12]. Moreover, the site of graft anastomosis directly influences pulmonary artery growth: patients undergoing a modified Blalock–Taussig-Thomas shunt often demonstrate disproportionate enlargement of the right pulmonary artery with concomitant hypoplasia of the left, whereas the central shunt is associated with symmetric and homogeneous growth [13]. Furthermore, this technique has been shown to be safe, with low mortality rates, a reduced incidence of thrombosis, and minimal risk of pulmonary overcirculation or subsequent pulmonary hypertension [14,15].
The diameter of the GORE-TEX® graft (3.5 mm) was chosen on the basis of the weight of the patient at the time of surgery (2700 g) to avoid hyperflow and minimize the risk of heart failure, which is consistent with that suggested by Barozzi et al. [10] and in accordance with that described by Myers et al. [14]. These patients are considered high-risk patients if their weight is less than 3000 g, and better results are obtained with grafts of 3.5 to 4 mm.
To construct the shunt, a modification of the central aorto-pulmonary shunt technique originally described by Gates, Laks, and Johnson was used because of the need for a precise anastomosis with the fewest possible incisions [11]. This approach minimizes vascular trauma and allows optimal graft orientation. First, owing to the location of the stent, an incision was made at the base of the pulmonary trunk toward its lateral aspect for extraction, and taking advantage of this incision, pulmonary anastomosis was performed with the terminal end of the graft, avoiding a new incision in the pulmonary trunk. Second, the central systemic-to-pulmonary shunt was completed in the lateral aspect of the aorta with an anastomosis of the terminal part of the graft and not with its lateral aspect. Through this modification, an adequate perpendicular position in this region of the aorta was sought, avoiding kinking of the graft.
The 24-month hemodynamic profile (mean pulmonary artery pressure 17 mmHg; PVR/SVR 0.57) together with favorable pulmonary artery indices (McGoon 2.59; Nakata 238.5 mm2/m2) indicate adequate pulmonary arterial development despite persistent right ventricle hypoplasia and reduced systolic function. These findings support continuation along a staged single-ventricle pathway, with a bidirectional Glenn as the next planned stage to reduce systemic venous burden and promote further pulmonary arterial remodeling. A one-and-a-half ventricular repair would be considered only if subsequent right ventricle growth and functional recovery permit meaningful contribution to pulmonary blood flow; Fontan completion would be contemplated later only if standard Fontan criteria are met (acceptable ventricular function, unobstructed pulmonary arteries, and sufficiently low pulmonary vascular resistance).
This case highlights several important lessons. First, ductal stent embolization in the setting of PAIVS, although uncommon, must always be considered and rapidly recognized, since delayed action may result in circulatory collapse. Second, in patients with hemodynamic instability, immediate surgical rescue is a life-saving alternative to percutaneous strategies, which are often further limited in low-weight neonates because of the small caliber of the vessels and the increased technical difficulty of catheter-based approaches. Finally, the technical nuances of the modified Gates, Laks, and Johnson shunt were determined by the location of the migrated stent and the need to minimize vascular trauma, underscoring the importance of tailoring the surgical approach to the anatomical and physiological conditions of each neonate, particularly in those with low body weight.
Ductal stent embolization in the context of a PAIVS is a rare complication that can compromise the clinical outcome if it is not identified and treated promptly. Given respiratory and hemodynamic instability, the immediate surgical approach can be used to avoid ischemic injuries or neonatal death. Salvage surgery with removal of the stent and central systemic‒to‒pulmonary shunt is an effective and safe strategy to reestablish pulmonary circulation. This case underscores the importance of early recognition of this complication and highlights the role of prompt surgical rescue as a feasible alternative in critically unstable neonates.
Acknowledgement:
Funding Statement: This research did not receive any specific grant from any funding agency in the public, commercial or not-for-profit sectors. Open Access funding for this article was supported by Servicios de Salud del Instituto Mexicano del Seguro Social para el Bienestar (IMSS-BIENESTAR).
Author Contributions: The authors confirm contribution to the paper as follows: study conception and design: Ricardo Garcia-Mora, Gunther Hernández-Morales, Miguel Ángel Chagolla-Santillán, Iliana Acevedo-Bañuelos, Jorge Luis Cervantes-Salazar, and Edel Rafael Rodea-Montero; data collection: Ricardo Garcia-Mora, Gunther Hernández-Morales, Miguel Ángel Chagolla-Santillán, Iliana Acevedo-Bañuelos, Jorge Luis Cervantes-Salazar, and Edel Rafael Rodea-Montero; analysis and interpretation of results: Ricardo Garcia-Mora, Gunther Hernández-Morales, Miguel Ángel Chagolla-Santillán, Iliana Acevedo-Bañuelos, Jorge Luis Cervantes-Salazar, and Edel Rafael Rodea-Montero; draft manuscript preparation: Ricardo Garcia-Mora, Gunther Hernández-Morales, Miguel Ángel Chagolla-Santillán, Iliana Acevedo-Bañuelos, Jorge Luis Cervantes-Salazar, and Edel Rafael Rodea-Montero; preparation of visual materials: Ricardo Garcia-Mora, Gunther Hernández-Morales, Miguel Ángel Chagolla-Santillán, Iliana Acevedo-Bañuelos, Jorge Luis Cervantes-Salazar, and Edel Rafael Rodea-Montero. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: The data that support the findings of this study are available from the Corresponding Author, Edel Rafael Rodea-Montero, upon reasonable request.
Ethics Approval: This study was reviewed and approved by Hospital Regional de Alta Especialidad del Bajío (HRAEB) Research-Ethics board (code: CEI-049-2025). All procedures were performed in accordance with the ethical standards of the institutional research committee and with the Declaration of Helsinki. Informed consent for the publication was obtained from the patient’s parents.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Hogan WJ , Grinenco S , Armstrong A , Devlieger R , Dangel J , Ferrer Q , et al. Fetal cardiac intervention for pulmonary atresia with intact ventricular septum: International fetal cardiac intervention registry. Fetal Diagn Ther. 2020; 47( 10): 731– 9. doi:10.1159/000508045. [Google Scholar] [CrossRef]
2. Cheung EW , Mastropietro CW , Flores S , Amula V , Radman M , Kwiatkowski D , et al. Procedural outcomes of pulmonary atresia with intact ventricular septum in neonates: A multicenter study. Ann Thorac Surg. 2023; 115( 6): 1470– 7. doi:10.1016/j.athoracsur.2022.07.055. [Google Scholar] [CrossRef]
3. Norbisrath KS , Labarinas S , Meliones J , McMann D , Patel M , Vasquez Choy A , et al. Pulmonary atresia intact ventricular septum with anomalous left coronary artery from pulmonary artery. Ann Thorac Surg Short Rep. 2024; 2( 4): 884– 7. doi:10.1016/j.atssr.2024.06.015. [Google Scholar] [CrossRef]
4. Sukhavasi A , McHugh-Grant S , Glatz AC , Mondal A , Griffis H , Burnham N , et al. Pulmonary atresia with intact ventricular septum: Intended strategies. J Thorac Cardiovasc Surg. 2022; 164( 5): 1277– 88. doi:10.1016/j.jtcvs.2021.11.104. [Google Scholar] [CrossRef]
5. Mini N , Schneider MBE , Asfour B , Mikus M , Zartner PA . Duct stenting vs. modified blalock-taussig shunt: New insights learned from high-risk patients with duct-dependent pulmonary circulation. Front Cardiovasc Med. 2022; 9: 933959. doi:10.3389/fcvm.2022.933959. [Google Scholar] [CrossRef]
6. Manhem S , Odermarsky M , Wåhlander H , Ekman-Joelsson BM . Pulmonary atresia with intact ventricular septum, a national comparison between interventional and surgical approach, in combination with a systemic literature review. Pediatr Cardiol. 2025; 46( 6): 1523– 36. doi:10.1007/s00246-024-03566-x. [Google Scholar] [CrossRef]
7. Santoro G , Gaio G , Giugno L , Capogrosso C , Palladino MT , Iacono C , et al. Ten-years, single-center experience with arterial duct stenting in duct-dependent pulmonary circulation: Early results, learning-curve changes, and mid-term outcome. Catheter Cardiovasc Interv. 2015; 86( 2): 249– 57. doi:10.1002/ccd.25949. [Google Scholar] [CrossRef]
8. Qureshi SA . Catheterization in neonates with pulmonary atresia with intact ventricular septum. Catheter Cardiovasc Interv. 2006; 67( 6): 924– 31. doi:10.1002/ccd.20690. [Google Scholar] [CrossRef]
9. Brooks A . Systemic-pulmonary artery shunts in infants: Modified Blalock-Taussig and central shunt procedures. Multimed Man Cardiothorac Surg. 2014; 2014: mmu007. doi:10.1093/mmcts/mmu007. [Google Scholar] [CrossRef]
10. Barozzi L , Brizard CP , Galati JC , Konstantinov IE , Bohuta L , d’Udekem Y . Side-to-side aorto-GoreTex central shunt warrants central shunt patency and pulmonary arteries growth. Ann Thorac Surg. 2011; 92( 4): 1476– 82. doi:10.1016/j.athoracsur.2011.05.105. [Google Scholar] [CrossRef]
11. Gates RN , Laks H , Johnson K . Side-to-side aorto-gore-tex central shunt. Ann Thorac Surg. 1998; 65( 2): 515– 6. doi:10.1016/s0003-4975(97)01126-0. [Google Scholar] [CrossRef]
12. Potapov EV , Alexi-Meskishvili VV , Dähnert I , Ivanitskaia EA , Lange PE , Hetzer R . Development of pulmonary arteries after central aortopulmonary shunt in newborns. Ann Thorac Surg. 2001; 71( 3): 899– 905; discussion 905–6. doi:10.1016/s0003-4975(00)02442-5. [Google Scholar] [CrossRef]
13. Batra AS , Starnes VA , Wells WJ . Does the site of insertion of a systemic-pulmonary shunt influence growth of the pulmonary arteries? Ann Thorac Surg. 2005; 79( 2): 636– 40. doi:10.1016/j.athoracsur.2004.07.062. [Google Scholar] [CrossRef]
14. Myers JW , Ghanayem NS , Cao Y , Simpson P , Trapp K , Mitchell ME , et al. Outcomes of systemic to pulmonary artery shunts in patients weighing less than 3 kg: Analysis of shunt type, size, and surgical approach. J Thorac Cardiovasc Surg. 2014; 147( 2): 672– 7. doi:10.1016/j.jtcvs.2013.09.055. [Google Scholar] [CrossRef]
15. Simsek B , Ozyuksel A , Saygi M , Demiroluk S , Basaran M . Revisiting the central aortopulmonary shunt procedure. Turk Gogus Kalp Damar Cerrahisi Derg. 2023; 31( 2): 207– 14. doi:10.5606/tgkdc.dergisi.2023.24247. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools