 Open Access
Open Access
CASE REPORT
Progressive Right Ventricular Outflow Tract Obstruction Following Perimembranous VSD Closure with Lifetech™ Konar-Multifunctional Occluder (MFO) Device: A Rare Case of Valve–Device Interaction
1 Department of Pediatric Cardiology, Koc University Hospital, Istanbul, Turkey
2 Koc University School of Medicine, Koc University Hospital, Istanbul, Turkey
3 Graduate School of Health Sciences, Koc University Hospital, Istanbul, Turkey
4 Department of Pediatric Cardiovascular Surgery, Koc University Hospital, Istanbul, Turkey
5 Nemours Children’s Health, Orlando, FL, USA
* Corresponding Author: Boran Cakan. Email:
Structural and Congenital Heart Disease 2026, 21(1), 5 https://doi.org/10.32604/schd.2026.074189
Received 05 October 2025; Accepted 23 March 2026; Issue published 31 March 2026
Abstract
Background: Transcatheter closure of perimembranous ventricular septal defects (pmVSDs) with Lifetech™ Konar-Multifunctional Occluder (MFO) has demonstrated high procedural success and safety. However, long-term complications due to valve–device interaction are rarely reported. We describe a pediatric patient who developed progressive right ventricular outflow tract (RVOT) obstruction and severe tricuspid regurgitation 20 months after MFO closure, highlighting mechanisms, management, and outcomes. Case Description: A 13-year-old girl underwent successful MFO closure of a 6-mm pmVSD. Early follow-up showed trivial tricuspid regurgitation and mild subpulmonic stenosis. Pre-procedural imaging revealed right ventricular hypertrophy caused by long-standing jet-related turbulence from the pmVSD, with no indications of a double-chambered right ventricle. Initial follow-up was uneventful, but serial echocardiography revealed rising RVOT gradients and mild tricuspid regurgitation. Sixteen months later, she developed exercise intolerance with a 60-mmHg gradient, although device position remained stable without migration. At 20 months, severe obstruction necessitated surgery. Intraoperatively, the device was found to entrap the anterior and septal tricuspid leaflets, displace papillary muscles, and induce fibrotic remodeling at the device-valve interference. Surgical management included excision of fibrotic tissue, pericardial patch augmentation of the tricuspid valve, and Kay annuloplasty. The patient recovered uneventfully, and follow-up confirmed resolution of obstruction with preserved valve function. Conclusions: This case demonstrates a rare long-term complication of MFO closure, in which chronic leaflet traction and increasing perimembranous fibrosis led to significant RVOT obstruction. Careful evaluation of anatomical risk factors, including VSD rim shape and leaflet proximity, can help identify patients at higher risk. Although MFO closure of pmVSDs is effective and typically safe, rare late complications involving the tricuspid valve and RVOT may occur. Pediatric cardiologists should carefully monitor valve–device interactions during implantation, and any leaflet restriction should prompt repositioning of the device. Follow-up must be thorough, with particular attention to Doppler gradients. If progressive obstruction develops, prompt surgical intervention can restore valve function and normal blood flow, leading to favorable outcomes.Keywords
Transcatheter closure of perimembranous ventricular septal defects (pmVSDs) is an accepted alternative to surgical repair in selected pediatric patients, reducing surgical burden and shortening recovery times [1]. The proximity of the perimembranous septum to the conduction system, the aortic valve, and the tricuspid valve creates challenges during device deployment [2].
The Lifetech™ Konar-Multifunctional Occluder (MFO) was developed to address these issues through its flexible nitinol design and low-profile discs [2]. For instance, a recent meta-analysis of 19 studies (n = 839) demonstrated a high procedural success rate of 94.2% for MFO implantation, with low rates of acute complications, including complete atrioventricular block (2.3%), aortic regurgitation (4.4%), and tricuspid regurgitation (3.7%) [3]. However, although early and mid-term results demonstrate high procedural success and a favorable safety profile, data regarding long-term complications remain limited [4,5].
We present a patient who developed progressive right ventricular outflow tract (RVOT) obstruction and severe tricuspid regurgitation 20 months after transcatheter pmVSD closure using an MFO device. The case is illustrated with detailed intraoperative findings and imaging, providing insight into the underlying mechanism of delayed valve–device interaction.
This study was conducted in accordance with the Declaration of Helsinki and relevant ethical guidelines. Ethical approval was obtained from the Koç University Institutional Review Board (IRB No: 2025.589.IRB1.083). Written informed consent was obtained from the patient’s legal guardian for participation and for the publication of clinical data and images. All data were anonymized to safeguard patient confidentiality. The signed consent form is available upon request.
A 13-year-old girl was referred with a 6-mm pmVSD and aneurysmal tissue formation. Transthoracic echocardiography (TTE) revealed a significant left-to-right shunt, with a peak systolic interventricular gradient of 100 mmHg. No significant Doppler gradient was detected across the RVOT. Given her results, transcatheter closure was recommended. One month after her initial presentation, percutaneous closure was performed when the patient weighed 42 kg and was 160 cm in height. The defect was considerable, and since previous studies reported a relatively high early residual shunt with MFO, a device 12 × 10 mm was selected [5]. An MFO device was deployed via a retrograde approach. Transesophageal echocardiography (TEE) confirmed stable device position, with no residual shunt or aortic regurgitation. The procedure was uncomplicated. The intra-procedural TEE video, available in the Supplementary Material S1, shows trivial tricuspid regurgitation and an unobstructed RVOT after deployment of the MFO device.
At the one-week follow-up, TTE demonstrated a normal sinus rhythm without any pattern suggestive of atrioventricular block or bundle-branch conduction delay. The device remained well-positioned and stable, with an unobstructed left ventricular outflow tract and no evidence of aortic regurgitation. Tricuspid valve assessment was unchanged from the intra-procedural evaluation, revealing only trivial regurgitation without any additional abnormalities. Three months later, TTE showed the device well seated, with no residual flow but mild tricuspid regurgitation and mild subpulmonic stenosis (30 mmHg gradient). Nine months later, RVOT obstruction had progressed to a 65-mmHg gradient, with a right ventricular systolic pressure (RVSP) of 60 mmHg. Close follow-up was recommended since her current right ventricular (RV) gradient was below half of the left ventricular systolic pressure.
Sixteen months later, the patient developed exercise intolerance. Doppler echocardiography confirmed worsening obstruction with a 75-mmHg gradient, RVSP of 65 mmHg, persistent RV hypertension, and moderate tricuspid regurgitation. Progressive worsening of the patient’s exercise intolerance, in conjunction with the increase in tricuspid regurgitation, prompted the decision to proceed with surgical intervention.
The patient underwent tricuspid valve repair with Kay annuloplasty under cardiopulmonary bypass 20 months after pmVSD closure, with minimal anticipated risk of future annular restriction; studies showed preserved annular growth and satisfactory tricuspid valve function [6]. Intraoperative findings provided direct evidence of the mechanisms underlying this progression. Entrapment of the anterior and septal tricuspid leaflets, displacement of papillary muscles to the periphery, and fibrosis across the perimembranous septum were seen. These changes impaired the continuity of right ventricular inlet–outlet flow and promoted obstruction and regurgitation. Surgical excision of fibrotic tissue and pericardial patch augmentation restored valve competency and unobstructed flow. The intraoperative lesions, including device–valve adhesions and fibrotic bands, are demonstrated in Fig. 1. Fibrotic tissue, seen in Fig. 2, arising from the anterior leaflet papilla, which impeded the continuity between the right ventricular inlet and outlet, was excised, and the defect on the tricuspid valve was subsequently reconstructed (augmented) using an autologous pericardial patch. Notably, the surgical assessment confirmed that the device was stable, properly positioned, and not protruding into or directly narrowing the RVOT. Therefore, removing the device and patching the VSD was unnecessary due to the recognized risk of atrioventricular conduction disturbance associated with device explantation. Instead, targeted resection of fibrotic tissue and tricuspid valve reconstruction effectively restored RVOT patency and valve function. The patient was weaned from bypass uneventfully, and intraoperative TEE confirmed mild tricuspid regurgitation with complete relief of the obstruction. The postoperative course was uncomplicated.
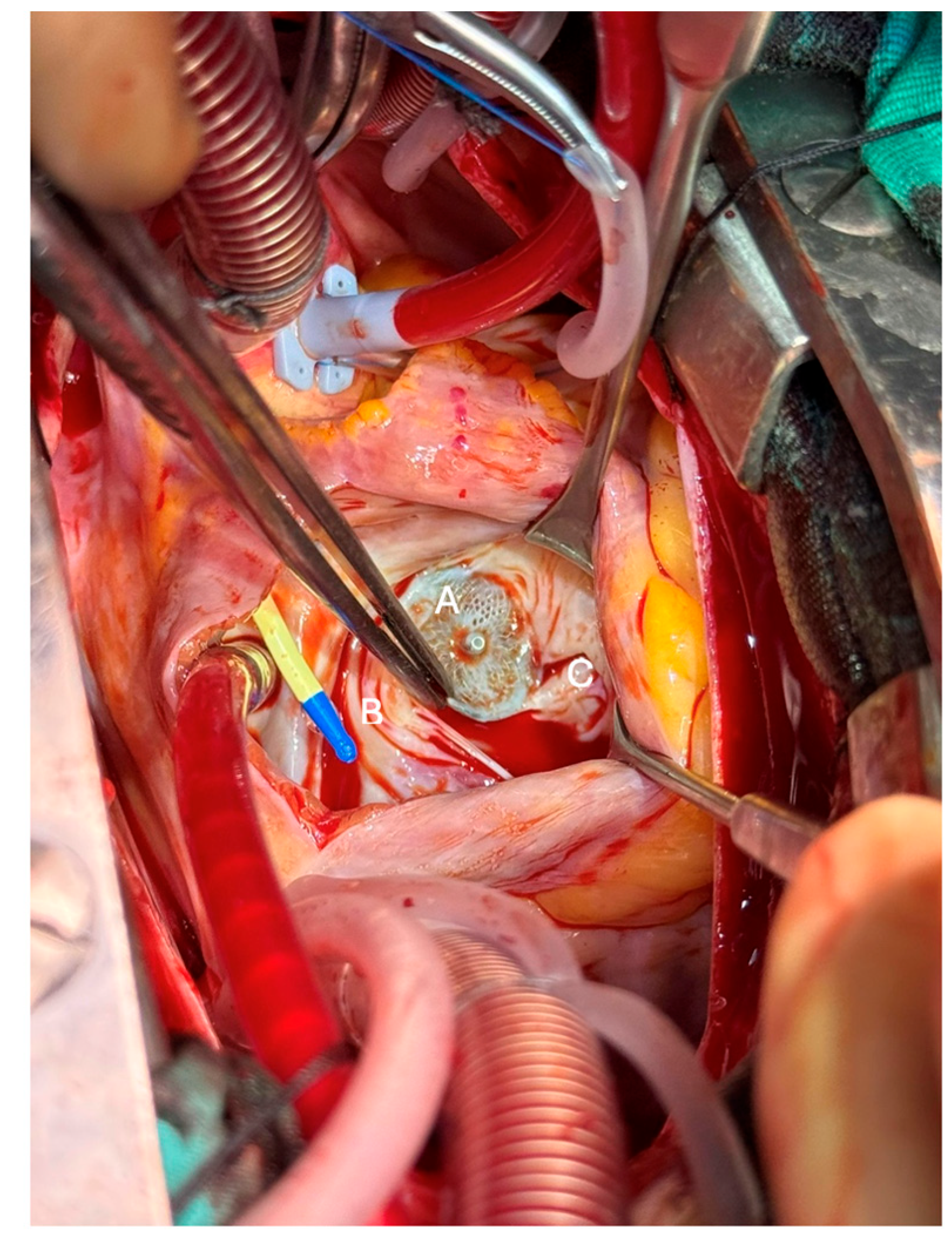
Figure 1: Intraoperative image from right atriotomy view showing interaction of the MFO device (A) with the anterior and septal leaflets of the tricuspid valve. Entrapment of leaflets that are pushed to the periphery by the device (B) and associated displacement of papillary muscles (C) are demonstrated.
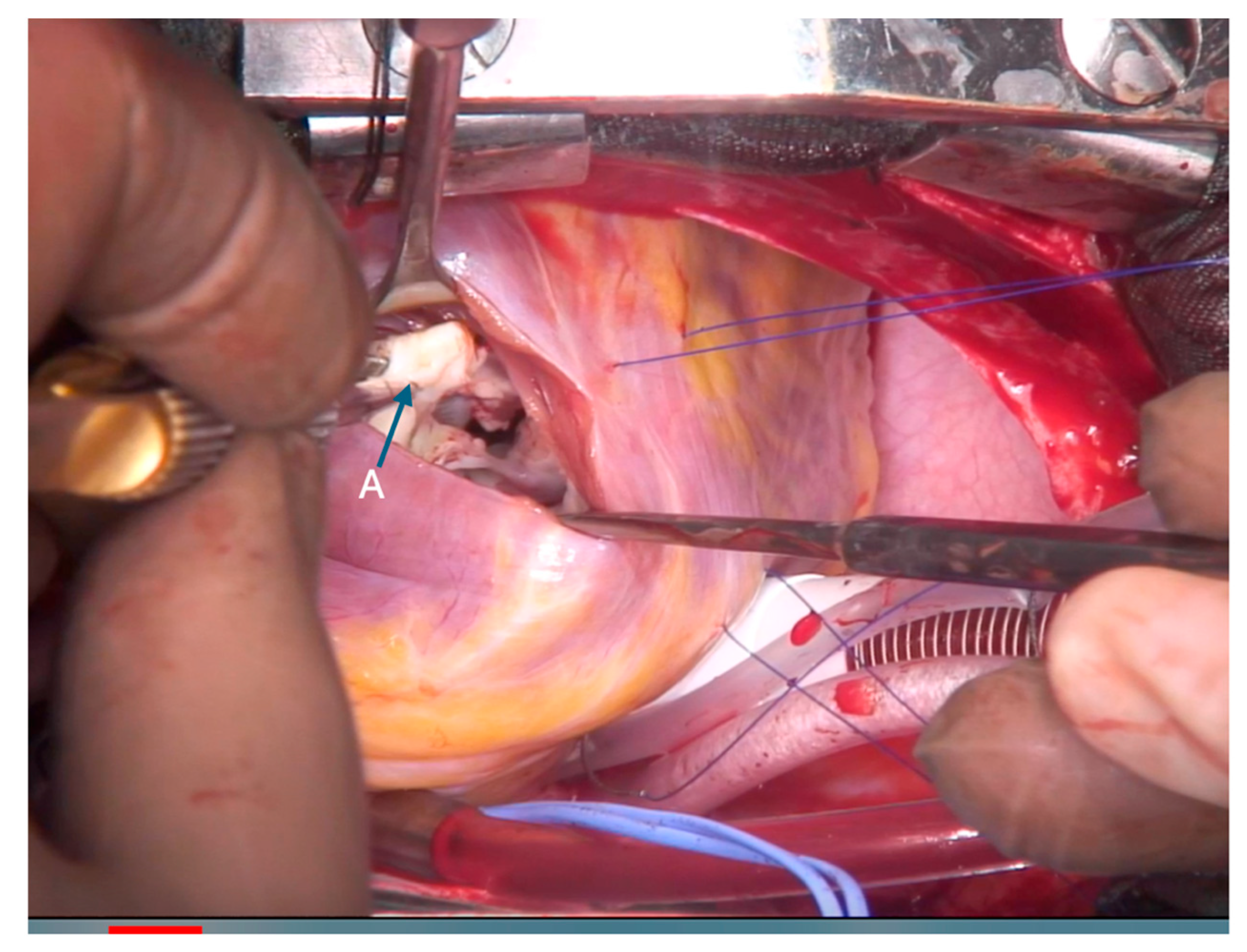
Figure 2: Right ventricular outflow tract view demonstrating fibrotic tissue (A) excised from the anterior papillary muscle–leaflet interface, which impaired continuity of RV inlet–outlet flow.
At the three-month postoperative follow-up, TTE demonstrated mild tricuspid regurgitation, no evidence of RVOT obstruction, and a stable device position. This case illustrates a rare but clinically significant late complication of MFO closure, characterized by progressive RVOT obstruction due to combined device–valve entrapment, perimembranous septal fibrosis, and papillary muscle hypertrophy. To the best of our knowledge, this combination of late complications has scarcely been documented in the long-term follow-up of MFO closures, highlighting the novelty and clinical relevance of this case.
The Lifetech™ Konar-Multifunctional Occluder (MFO) has a low-profile design that reduces radial pressure on the atrioventricular conduction system, thereby decreasing the risk of atrioventricular block, and is associated with high procedural success and safety [3,4,5]. However, its discs may still contact nearby valvular structures, especially the aortic and tricuspid valves, leading to acute and delayed complications [7]. For instance, new-onset tricuspid regurgitation may result from distortion of the tricuspid subvalvular apparatus caused by the MFO [7]. Notably, the right-sided disc of the MFO, which is approximately 1–2 mm larger than that of the Lifetech™ Cera VSD occluder, may enhance interaction with the tricuspid valve apparatus and increase the risk of tricuspid regurgitation [7]. Consequently, a higher ratio of right disc diameter to body weight has been identified as an independent risk factor for TR (OR: 2.816), underscoring the significance of precise device sizing, particularly in lighter patients [8]. Nonetheless, tricuspid regurgitation is usually well tolerated and does not progress over time [8]. Although intra-procedural TEE in our patient (available in Supplementary Material S1) revealed only minimal tricuspid regurgitation with preserved leaflet motion and no evidence of entrapment or chordal distortion, subtle abnormalities may not be immediately apparent at the time of implantation [9,10,11]. Therefore, the mechanism underlying the delayed progressive tricuspid regurgitation observed in our patient is best explained by chronic device–leaflet interaction and ongoing fibrotic remodeling.
To understand the mechanism of late-onset RVOT obstruction in our patient, it is essential to consider the baseline RV structure and the long-term hemodynamic effects of the pmVSD prior to device closure. Pre-procedural imaging showed RV hypertrophy due to long-standing jet-related turbulence from the pmVSD, with no signs of a double-chambered right ventricle. Given that the patient was 13 years old, the RV had likely been exposed to chronic turbulent flow for many years, which could have led to hypertrophic remodeling [12]. Although preoperative echocardiography showed the RVOT to be open, the right ventricular free wall was hypertrophic. It is possible that, despite the MFO device’s low profile, ongoing turbulence in this area caused progressive fibrosis around the edges of the device, increased RVOT narrowing, and worsening tricuspid regurgitation over time [9,13,14]. Therefore, the later development of significant tricuspid regurgitation, fibrosis, and RVOT obstruction is most consistent with a delayed interaction between the device’s right-sided disc and the tricuspid subvalvular apparatus, rather than an underlying anatomical condition or unrecognized acute complication at the time of implantation.
Minimal or mild tricuspid regurgitation alone is not considered a contraindication to device implantation, as several series have demonstrated that trivial tricuspid regurgitation is a common and clinically acceptable finding after transcatheter pmVSD closure [15]. In contrast, acute device-related tricuspid leaflet entrapment during deployment, resulting in significant regurgitation, indicates that the device cannot be safely positioned and should not be released. Importantly, early signs of device–leaflet interaction may be absent at implantation, as even subtle abnormalities can progress over time and lead to clinically significant chronic valvular dysfunction [9,10,11]. For this reason, in some patients with VSD (especially those with subpulmonic defects), the hemodynamic and anatomical presentation can resemble features of a double-chambered right ventricle. Therefore, meticulous echocardiographic evaluation is vital in this subgroup, as these presentations fundamentally change the therapeutic approach and may shift management from transcatheter closure to surgical intervention.
Device-related late complications, such as those observed in our case, are rare after transcatheter pmVSD closure [15]. In large long-term series, severe valve-related complications occur in approximately 2–3% of patients and are reported more frequently with coil-based systems, primarily due to chordal entrapment or leaflet impingement, whereas single- and double-disc devices, including the MFO, are generally associated with lower rates of residual shunt and valve dysfunction [15]. Although the MFO’s low-profile, flexible design is intended to minimize valve interference, isolated cases of delayed valve–device interaction and progressive outflow obstruction, as seen in our patient, highlight that late complications can still occur and warrant long-term surveillance.
While closing pmVSDs with the MFO is effective and generally safe, rare long-term complications involving the tricuspid valve and RVOT can happen. During the procedure, any signs of tricuspid valve–device interaction should prompt careful reassessment and, if needed, repositioning of the device. Even when implantation appears satisfactory, pediatric cardiologists should remain vigilant and follow up, especially by monitoring Doppler gradients to detect and manage any developing obstruction.
Acknowledgement:
Funding Statement: The authors received no specific funding for this study.
Author Contributions: The authors confirm their contributions to the paper as follows: Mete Han Kizilkaya, Mehmet Bicer, and Ender Odemis; Boran Cakan conceived the project. Boran Cakan wrote the main manuscript text and prepared figures and videos with the help of Mete Han Kizilkaya. Mete Han Kizilkaya, Boran Cakan, Ender Odemis, and Mehmet Bicer critically revised the work. Ender Odemis and Mehmet Bicer approved the final manuscript and agree to be accountable for all aspects of the work. All authors contributed to the revision process. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: The authors will supply the relevant data in response to adequate requests.
Ethics Approval: This study was conducted in accordance with the Declaration of Helsinki and relevant ethical guidelines. Ethical approval was obtained from the Koç University Institutional Review Board (IRB No.: 2025.589.IRB1.083). Written informed consent was obtained from the patient’s legal guardian for participation and for the publication of clinical data and images. All data were anonymized to safeguard patient confidentiality. The signed consent form is available upon request.
Conflicts of Interest: The authors declare no conflicts of interest.
Supplementary Materials: The supplementary material is available online at https://www.techscience.com/doi/10.32604/schd.2026.074189/s1.
References
1. Santhanam H , Yang L , Chen Z , Tai BC , Rajgor DD , Quek SC . A meta-analysis of transcatheter device closure of perimembranous ventricular septal defect. Int J Cardiol. 2018; 254: 75– 83. doi:10.1016/j.ijcard.2017.12.011. [Google Scholar] [CrossRef]
2. Tanidir IC , Baspinar O , Saygi M , Kervancioglu M , Guzeltas A , Odemis E . Use of LifetechTM Konar-MF, a device for both perimembranous and muscular ventricular septal defects: A multicentre study. Int J Cardiol. 2020; 310: 43– 50. doi:10.1016/j.ijcard.2020.02.056. [Google Scholar] [CrossRef]
3. Kabadayı B , Eren ZB , Odemis E . Efficacy and safety of the LifeTechTM Multifunctional Occluder (Konar-MFO) in transcatheter closure of ventricular septal defects: A systematic review and meta-analysis. Catheter Cardiovasc Interv. 2025; 106( 1): 325– 35. doi:10.1002/ccd.31560. [Google Scholar] [CrossRef]
4. Laha S , Gangopadhyay D , Roy M , Singh A , Nandi D , Dutta J . Procedural outcomes of percutaneous closure of perimembranous and other ventricular septal defects using Konar-MF occluder and short-term follow-up. Ann Pediatr Cardiol. 2024; 17( 2): 101– 8. doi:10.4103/apc.apc_201_23. [Google Scholar] [CrossRef]
5. Odemis E , Kizilkaya MH . Early and mid-term outcomes of transcatheter closure of perimembranous ventricular septal defects using the LifetechTM Konar-MF Occluder device (MFO). Cardiol Young. 2023; 33( 10): 2021– 6. doi:10.1017/S1047951122003547. [Google Scholar] [CrossRef]
6. Tchantchaleishvili V , Rajab TK , Cohn LH . Posterior suture annuloplasty for functional tricuspid regurgitation. Ann Cardiothorac Surg. 2017; 6( 3): 262– 5. doi:10.21037/acs.2017.05.04. [Google Scholar] [CrossRef]
7. Gozar JD , Cheng DED , Del Rosario JJD . Procedural and short-term outcomes of transcatheter closure of ventricular septal defect using Lifetech multifunctional occluder: Initial experience. Cardiol Young. 2021; 31( 3): 435– 45. doi:10.1017/S1047951120004229. [Google Scholar] [CrossRef]
8. Jiang D , Yi Y , Zhao L , Wang J , Wang Y , Lv J , et al. Risk factors and long-term outcomes of tricuspid regurgitation after transcatheter closure of pediatric perimembranous ventricular septal defects. J Am Heart Assoc. 2025; 14( 6): e039443. doi:10.1161/JAHA.124.039443. [Google Scholar] [CrossRef]
9. Mediratta A , Addetia K , Yamat M , Moss JD , Nayak HM , Burke MC , et al. 3D echocardiographic location of implantable device leads and mechanism of associated tricuspid regurgitation. JACC Cardiovasc Imaging. 2014; 7( 4): 337– 47. doi:10.1016/j.jcmg.2013.11.007. [Google Scholar] [CrossRef]
10. Matyal R , Wang A , Mahmood F . Percutaneous ventricular septal defect closure with Amplatzer devices resulting in severe tricuspid regurgitation: VSD closure resulting in severe TR. Catheter Cardiovasc Interv. 2013; 82( 6): E817– 20. doi:10.1002/ccd.24803. [Google Scholar] [CrossRef]
11. Butera G , Massimo C , Mario C . Late complete atriovenous block after percutaneous closure of a perimembranous ventricular septal defect. Catheter Cardiovasc Interv. 2006; 67( 6): 938– 41. doi:10.1002/ccd.20696. [Google Scholar] [CrossRef]
12. Turner ME , Bouhout I , Petit CJ , Kalfa D . Transcatheter closure of atrial and ventricular septal defects: JACC focus seminar. J Am Coll Cardiol. 2022; 79( 22): 2247– 58. doi:10.1016/j.jacc.2021.08.082. [Google Scholar] [CrossRef]
13. Chin CY , Chen CA , Fu CM , Hsu JY , Lin HC , Chiu SN , et al. Risk factors of long-term sequelae after transcatheter closure of perimembranous ventricular septal defect in young children. Circ J. 2024; 88( 5): 663– 71. doi:10.1253/circj.CJ-23-0891. [Google Scholar] [CrossRef]
14. Wang J , Zuo J , Yu S , Yi D , Yang X , Zhu X , et al. Effectiveness and safety of transcatheter closure of perimembranous ventricular septal defects in adults. Am J Cardiol. 2016; 117( 6): 980– 7. doi:10.1016/j.amjcard.2015.12.036. [Google Scholar] [CrossRef]
15. Promphan W , Wongwaitaweewong K , Eleena A , Roymanee S , Jarutach J , Buntharikpornpun R , et al. Long-term outcomes following transcatheter closure of small perimembranous ventricular septal defects. Pediatr Cardiol. 2026; 47( 2): 886– 97. doi:10.1007/s00246-025-03857-x. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools