 Open Access
Open Access
ARTICLE
Analysis of Relevant Factors Associated with Postoperative Recovery after Anomalous Origin of the Left Coronary Artery from Pulmonary Artery Surgery in Children
1 Department of CICU, Guangzhou Women and Children’s Medical Center, Guangzhou Medical University, Guangzhou, China
2 Department of Neonatology, Guangzhou Women and Children’s Medical Center, Liuzhou Hospital, Liuzhou, China
3 Department of Emergency, Women and Children’s Medical Center, Shenzhen Maternity and Child Healthcare Hospital, Southern Medical University, Shenzhen, China
4 Department of Cardiac Surgery, Guangzhou Women and Children’s Medical Center, Guangzhou Medical University, Guangzhou, China
5 Department of Pediatric Cardiac Surgery, Center of Perinatology and Pediatric Cardica Surgery, Almaty, Kazakhstan
* Corresponding Author: Na Zhou. Email:
# These authors contributed equally to this work as the first author
Structural and Congenital Heart Disease 2026, 21(1), 3 https://doi.org/10.32604/schd.2026.076648
Received 24 November 2025; Accepted 06 March 2026; Issue published 31 March 2026
Abstract
Backgorund: Anomalous origin of the left coronary artery from the pulmonary artery (ALCAPA) is a rare congenital anomaly of coronary artery anatomy, usually diagnosed in infancy, but adults may also be affected by this deformity. Objectives: The aim of this study is to examine long-term outcomes in patients with ALCAPA and analyze the relevant factors influencing postoperative outcomes. Methods: The records of patients with ALCAPA admitted from January 2015 to December 2024 were retrospectively reviewed. Clinical data of the patients were retrieved from the records. The follow-up data included mortality rates and complications. Kaplan-Meier survival curves were used to analyze the occurrence of death and reoperation during the follow-up of patients with ALCAPA, and the log-rank test was used for comparisons between groups. A Cox regression model was used to analyze the risk factors for adverse events in patients with clinical indicators of an unfavorable prognosis. Results: 48 patients met the study criteria. Postoperative death or reoperation was defined as an unfavorable prognosis. Patients were divided into two groups: favorable prognosis (n = 41) and unfavorable prognosis (n = 7). The differences among the Kaplan–Meier survival curves for the different subgroups were statistically significant. Feeding difficulties (p < 0.001), preoperative left ventricular end-diastolic diameter (LVEDD) Z score (p = 0.049), preoperative mechanical ventilation (p < 0.001), preoperative vasoactive drugs (p < 0.001), ECMO (p < 0.001), and delayed sternal closure (p < 0.001) were associated with an unfavorable prognosis. Multivariate Cox proportional hazard analysis revealed that preoperative LVEF (HR: 0.711; 95% CI: 0.542–0.932; p = 0.013), CPB time (HR: 1.021; 95% CI: 1.003–1.04; p = 0.022) and length of stay (HR: 1.064; 95% CI: 1.015–1.116; p = 0.01) were associated with an unfavorable prognosis in patients, and age at surgery (HR: 1.092; 95% CI: 0.991–1.203; p = 0.077) was closely related to an unfavorable prognosis. Conclusions: A younger surgical age, a reduced preoperative LVEF, a longer CPB time, and a longer length of stay were associated with an unfavorable prognosis in this cohort.Keywords
Anomalous origin of the left coronary artery from the pulmonary artery (ALCAPA) is a rare congenital anomaly of coronary artery anatomy, with an incidence rate of 1 in 300,000 [1]. As pulmonary vascular resistance decreases during the first few weeks after birth, myocardial ischemia and/or infarction may occur. ALCAPA causes severe myocardial ischemia, overall left ventricular dysfunction, and dilation of the mitral valve annulus, resulting in varying degrees of mitral regurgitation (MR) [2]. ALCAPA is usually diagnosed in infancy, but adults may also be affected by this deformity, although it is extremely rare. Most infants who do not undergo surgical treatment die within one year [3]. Some patients may do not present symptoms, whereas others may present with manifestations such as left ventricular dysfunction, malignant arrhythmias, or sudden cardiac death [4]. Therefore, early diagnosis and surgery can significantly reduce the mortality rate.
Previous studies have reported that a low preoperative left ventricular ejection fraction (LVEF), age at surgery, and severe preoperative MR findings are risk factors for postoperative mortality after ALCAPA surgery [5,6]. In this study, long-term follow-up was conducted on patients with ALCAPA to analyze the factors influencing prognosis in children and to provide a strong basis for clinical treatment decisions and prognosis assessment.
Patients with ALCAPA admitted to Guangzhou Women and Children’s Medical Center, Guangzhou Medical University, from January 2015 to December 2024 were retrospectively reviewed. The inclusion criteria were age < 18 years, a diagnosis of ALCAPA through echocardiography and enhanced cardiac CT, and the exclusion of cardiomyopathy, complex congenital heart disease, and other coronary artery origin abnormalities.
Ethical approval for the present study was granted by the Ethics Committee of Guangzhou Women and Children’s Medical Center, Guangzhou Medical University (No. 212A01), and the parents of the patients signed the informed consent form. This retrospective study was conducted in accordance with the Declaration of Helsinki.
2.2 Surgical Technique and Strategies of CPB
Surgical technique was coronary artery reimplantations. All patients underwent the procedure under normothermic cardiopulmonary bypass (CPB) (rectal temperature of 35.5 to 37°C) using standard aortic and bicaval venous cannulations. During CPB, the flow rate was maintained at 250 mL·m−2·min−1 or greater and the hematocrit level was kept between 32% and 36%. An alpha-stat pH blood gas management was used in all patients. Serum lactate levels were monitored continuously throughout the operation.
Patients were followed under sedation and paralysis at least 48 h with a continuous infusion of sufentanyl and midazolam. The use and duration of postoperative inotropes, length of total hospitalization, and mortality were determined.
Clinical data retrieved from patient records included weight Z score, symptoms, and clinical signs (for example, shortness of breath and growth retardation). Definitions of feeding difficulties refer to various problems that occur during the eating process that affect the intake of nutrients, age at surgery, perioperative condition, whether it was a second surgery, prognosis, and follow-up data after discharge. The follow-up data included mortality rates and complications, and the follow-up methods included outpatient visits and telephone follow-up. The echocardiographic parameters included the LVEF, MR degree and left ventricular end-diastolic diameter (LVEDD). Z-scores were extrapolated based on age and body surface area [7].
All statistical analyses were performed using SPSS software, version 29.0 for Windows (IBM Corp., Armonk, NY, USA), GraphPad Prism 10 (GraphPad Software Inc., Boston, MA, USA), was used for plotting. Continuous variables are reported as the mean ± SD or median and interquartile range, according to a normal or non-normal distribution according to the Shapiro–Wilk normality test. Groups were compared by use of the unpaired Student’s t test when normally distributed, whereas the Mann–Whitney U test was applied to those with a non-normal distribution. The Wilcoxon matched-paired signed-rank test was used to analyze paired data at different time points. Categorical variables were compared via Fisher’s exact test. Kaplan–Meier survival curves were used to analyze the occurrence of death and reoperation during the follow-up of patients with ALCAPA, and the log-rank test was used for comparisons between groups. A Cox regression model was used to analyze the risk factors for adverse events in patients with clinical indicators of an unfavorable prognosis. A two-tailed p < 0.05 was considered statistically significant.
Of the 48 patients who met the study criteria, the median age at surgery was 5 months (range 0–13 years), and the median weight Z-score was −1.7 ± 1.2. Among all the patients, 4 had other cardiac malformations, such as ventricular septal defects, tetralogy of Fallot, transitional atrioventricular septal defects, and aortic coarctation. All the patients underwent left coronary artery reimplantation, and among them, 24 underwent mitral valvuloplasty simultaneously. Postoperative death or reoperation was defined as an unfavorable prognosis. Patients were divided into two groups: favorable prognosis (n = 41) and unfavorable prognosis (n = 7). The baseline characteristics are described in Table 1. There were no significant differences in other demographic characteristics and clinical manifestations between the two groups. Only the difference in feeding difficulties was found to be significant (p = 0.008). The preoperative echocardiogram showed, preoperative LVEF (p < 0.001) was significantly lower than that in the favorable prognosis group, and their preoperative LVEDD Z-score (p = 0.011) was higher than that in the favorable prognosis group. Preoperative mechanical ventilation and vasoactive drugs were significantly more commonly used in the unfavorable prognosis group than in the favorable prognosis group (p = 0.018).
Table 1: Demographic characteristics and perioperative data with ALCAPA.
| Favorable Prognosis Group (n = 41) | Unfavorable Prognosis Group (n = 7) | p | |
|---|---|---|---|
| Demographic data, n (%) | |||
| Sex | 0.412 | ||
| Male | 15 (36.6) | 4 (57.1) | |
| Female | 26 (63.4) | 3 (42.9) | |
| Weight Z-score | −1.7 ± 1.0 | −1.54 ± 1.55 | 0.707 |
| Age at surgery, months | 5 (3.5, 33) | 5 (2, 13) | 0.509 |
| Presenting symptoms, n (%) | |||
| Shortness of breath | 24 (58.5) | 3 (42.9) | 0.683 |
| Feeding difficulties | 1 (2.4) | 3 (42.9) | 0.008* |
| Growth retardation | 11 (26.8) | 1 (14.3) | 0.662 |
| Preoperative NT-proBNP, pg/mL | 4979 (353.5, 21,303.5) | 33,739 (26,355, 35,000) | 0.013* |
| Electrocardiograph, at admission, n (%) | |||
| Abnormal Q wave | 7 (17.1) | 1 (14.3) | 0.999 |
| ST-T change | 19 (46.3) | 3 (42.9) | 0.999 |
| T-wave change | 13 (31.7) | 3 (42.9) | 0.672 |
| Preoperative echocardiogram, n (%) | |||
| LVEF, % | 39 (30.5, 59.5) | 26 (20, 28) | <0.001* |
| Degree of MR | 0.233 | ||
| None or mild MR | 19 (46.3) | 1 (14.3) | |
| Moderate severe MR | 10 (24.4) | 2 (28.6) | |
| Severe MR | 12 (29.3) | 4 (57.1) | |
| LVEDD Z-score | 4.22 ± 2.28 | 6.58 ± 1.18 | 0.011* |
| Preoperative mechanical ventilation, n (%) | 2 (4.9) | 3 (42.9) | 0.018* |
| Preoperative vasoactive drugs, n (%) | 2 (4.9) | 3 (42.9) | 0.018* |
| CPB time, min | 140 (119.5, 169) | 157 (121, 205) | 0.267 |
| Aortic cross-clamp time, min | 68 (59.5, 86) | 70 (60, 90) | 0.599 |
| MR repair, n (%) | 19 (46.3) | 4 (57.1) | 0.696 |
| Delayed sternal closure, n (%) | 3 (7.3) | 3 (42.9) | 0.033* |
| ECMO, n (%) | 1 (2.4) | 2 (28.6) | 0.052 |
| Length of stay, days | 17 (13, 21) | 18 (9, 49) | 0.988 |
3.2 Mortality and Complications
The mortality rate of the patients was 10.42% (5/48). Two patients died of heart failure (on the 9th and 49th days after surgery), among whom one patient underwent repeat mitral valvuloplasty due to severe MR. Two patients died of malignant arrhythmia (on the 7th and 18th days after surgery), and one died of respiratory failure due to severe pulmonary infection (49 days after surgery). All deaths occurred in the postoperative period.
A follow-up was available for 43 patients; the median follow-up was 687 days (range 1–2961 days), and no patients died after discharge. Reoperation was required in two patients. Reasons for reoperation included MR repair and pulmonary artery balloon dilation for the pulmonary artery trunk and right pulmonary artery stenosis. The estimated rates of freedom from unfavorable prognosis at 2 months, 6 months, 1 year, 2 years, and 3 years were 91.62%, 89.44%, 89.44%, 86.46%, and 86.46%, respectively (Fig. 1). Echocardiography during the follow-up period revealed that the LVEF of the surviving patients with low preoperative LVEF was completely restored. The median time to normalization of left ventricular function was 118 days (range 7–604 days) (Fig. 2). The differences among the Kaplan–Meier survival curves for the different subgroups were statistically significant (Fig. 3A–F). Feeding difficulties (p < 0.001), higher preoperative LVEDD Z-scores (p = 0.049), preoperative mechanical ventilation (p < 0.001), preoperative vasoactive drug use (p < 0.001), extracorporeal membrane oxygenation (ECMO) (p < 0.001), and delayed sternal closure (p < 0.001) were associated with an unfavorable prognosis.
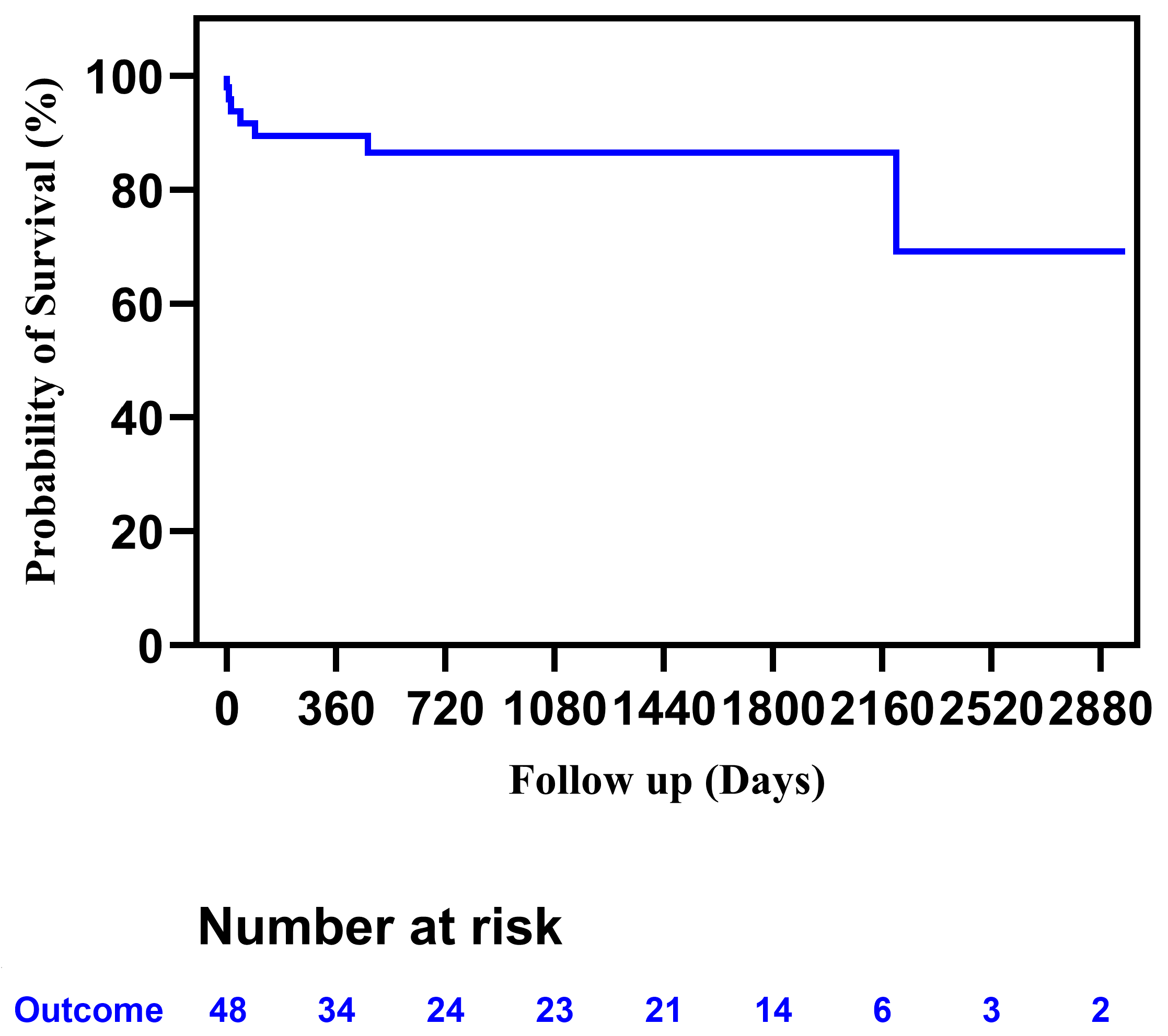
Figure 1: Kaplan-Meier curves for favorable outcomes in patients with ALCAPA. ALCAPA, Anomalous origin of the left coronary artery from the pulmonary artery.
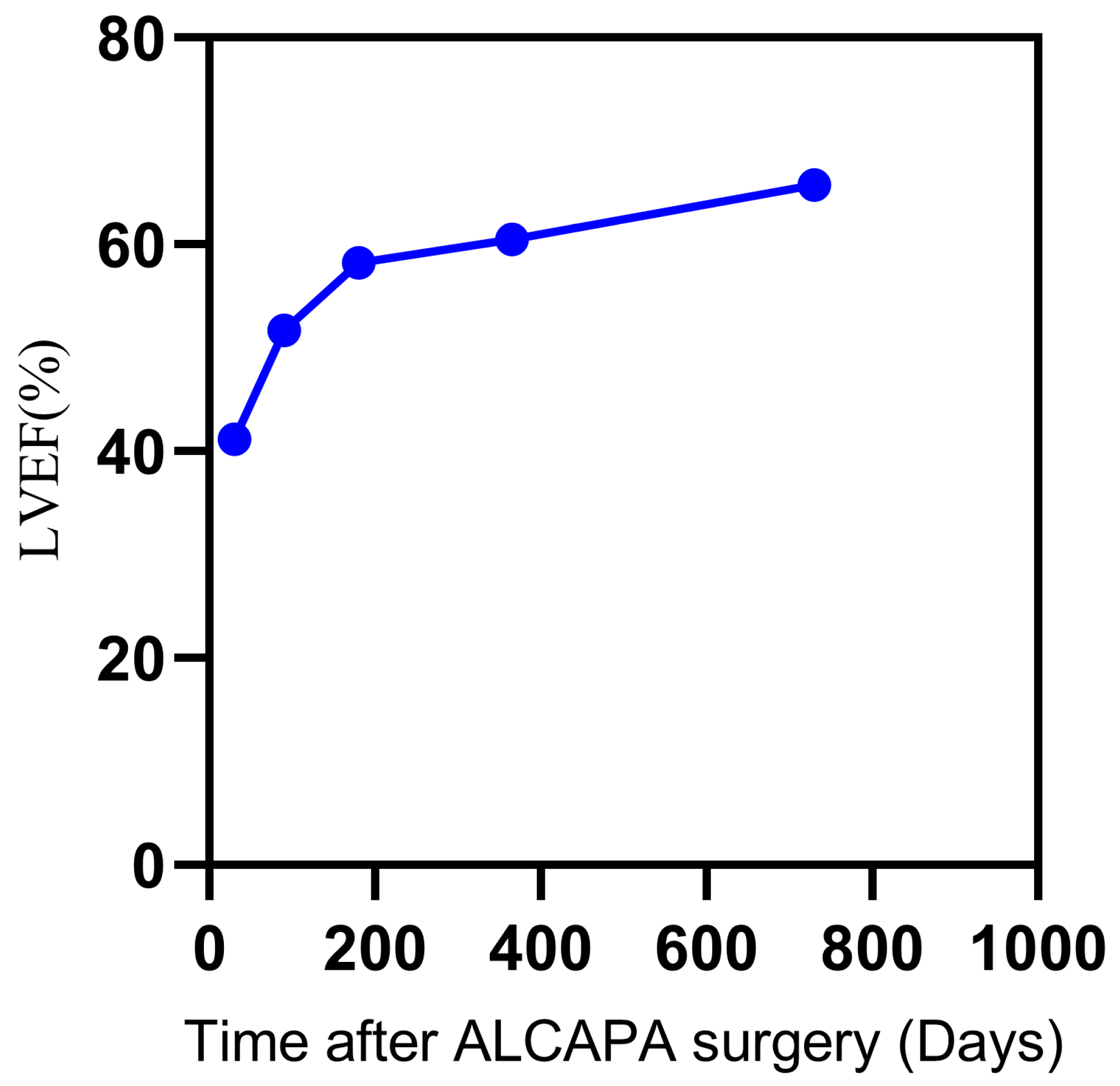
Figure 2: Dynamic change of LVEF of the surviving patients with preoperative low LVEF. LVEF, Left ventricular ejection fraction.
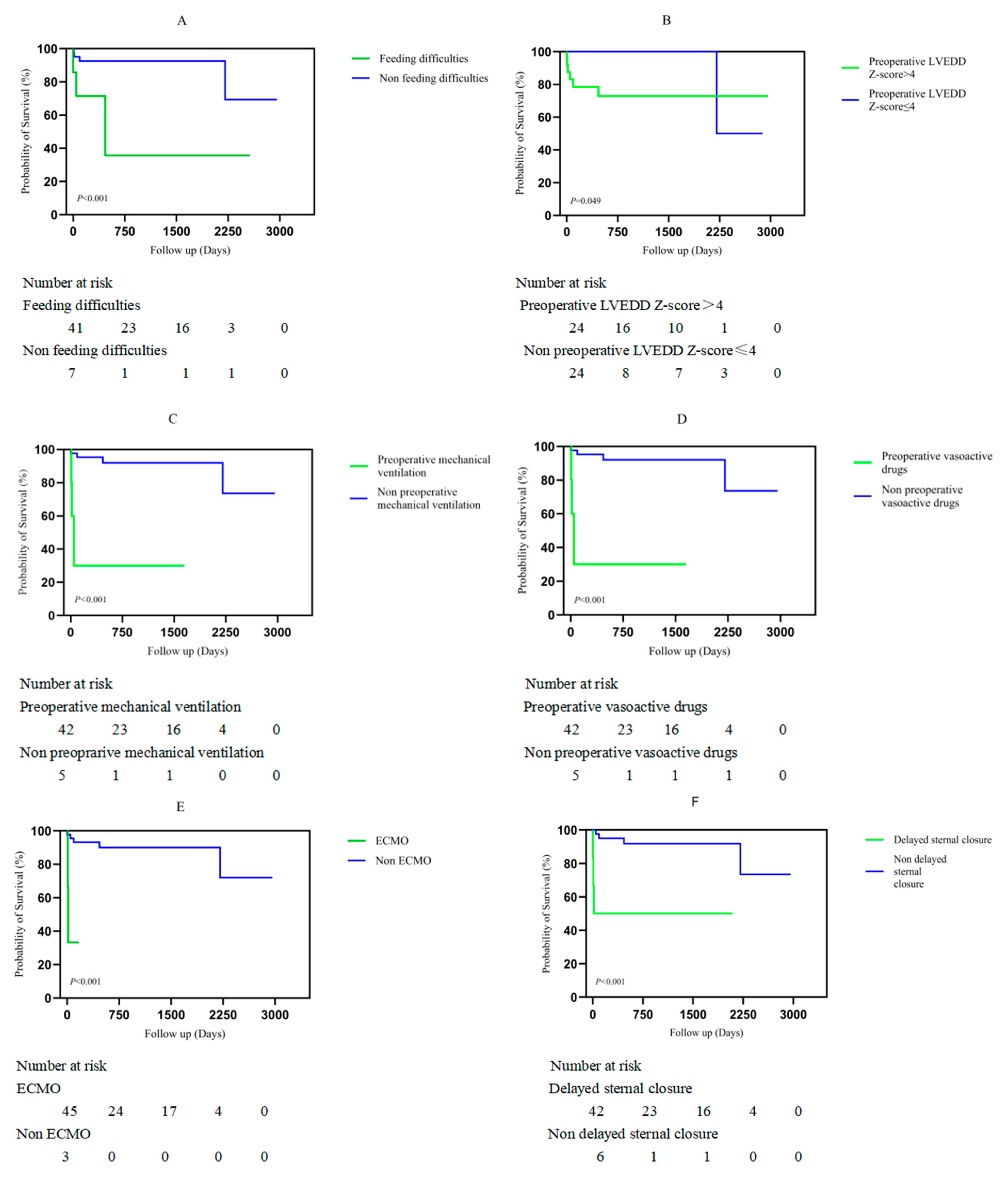
Figure 3: Kaplan–Meier analysis of factors associated with unfavourable prognosis in patients with ALCAPA. (A) Feeding difficulties. (B) Preoperative left ventricular end-diastolic diameter Z-score (LVEDD-Z). (C) Preoperative mechanical ventilation. (D) Preoperative vasoactive drug support. (E) Use of extracorporeal membrane oxygenation (ECMO). (F) Delayed sternal closure. ALCAPA, Anomalous origin of the left coronary artery from the pulmonary artery.
3.4 Cox Proportional Hazard Analyses
Univariate and multivariate Cox proportional hazard analyses for independent risk factors for prognosis are shown in Table 2. Multivariate Cox proportional hazard analysis revealed that preoperative LVEF (Hazard ratio (HR): 0.711; 95% confidence interval (CI): 0.542–0.932; p = 0.013), CPB time (minutes) (HR: 1.021; 95% CI: 1.003–1.04; p = 0.022), and length of stay (days) (HR: 1.064; 95% CI: 1.015–1.116; p = 0.01) were associated with unfavorable prognosis in patients, and age at surgery (HR: 1.092; 95% CI: 0.991–1.203; p = 0.077) was closely related to unfavorable prognosis. The influence of the variables on unfavorable prognosis is shown in Fig. 3A–F.
Table 2: Cox proportional hazard analysis for factors for the prognosis among ALCAPA patients.
| Risk Factor | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|
| Hazard Ratio (95% CI) | p | Hazard Ratio (95% CI) | p | |
| Sex | 0.467 [0.104–2.091] | 0.32 | ||
| Weight, Z-score | 1.334 [0.632–2.814] | 0.45 | ||
| Age at surgery | 0.948 [0.906–0.992] | 0.022 | 1.092 [0.991–1.203] | 0.077 |
| Shortness of breath, n (%) | 0.563 [0.126–2.518] | 0.452 | ||
| Growth retardation, n (%) | 0.565 [0.066–4.841] | 0.603 | ||
| Preoperative NT-proBNP > 5000 pg/mL, n (%) | 6.064 [0.726–50.637] | 0.096 | ||
| Preoperative mechanical ventilation, n (%) | 16.252 [3.043–86.789] | 0.001 | ||
| Preoperative vasoactive drugs, n (%) | 16.252 [3.043–86.789] | 0.001 | ||
| Preoperative LVEDD Z-score | 1.813 [1.105–2.975] | 0.019 | ||
| Preoperative LVEF, n (%) | 0.82 [0.721–0.933] | 0.003 | 0.711 [0.542–0.932] | 0.013 |
| Preoperative moderat-severe MR, n (%) | 3.656 [0.436–30.670] | 0.232 | ||
| Abnormal Q wave, n (%) | 0.767 [0.092–6.374] | 0.806 | ||
| ST-T change, n (%) | 0.72 [0.153–3.392] | 0.678 | ||
| T-wave change, n (%) | 1.326 [0.295–5.954] | 0.712 | ||
| CPB time, min | 1.009 [1–1.017] | 0.038 | 1.021 [1.003–1.04] | 0.022 |
| Aortic cross-clamp time, min | 1.008 [0.982–1.035] | 0.548 | ||
| Delayed sternal closure, n (%) | 12.056 [2.34–62.113] | 0.003 | ||
| ECMO, n (%) | 15.849 [2.529–99.31] | 0.003 | ||
| Length of stay, days | 1.031 [1.003–1.059] | 0.027 | 1.064 [1.015–1.116] | 0.01 |
Fetuses with ALCAPA usually have no symptoms because the diastolic pressures of the pulmonary artery and aorta are similar during prenatal circulation. However, after birth, when pulmonary vascular resistance begins to decline, most infants start to present clinical symptoms, such as poor development, excessive sweating, breathing difficulties, and pallor, due to retrograde flow in the left coronary artery [8]. Moreover, coronary artery retrograde flow can lead to coronary steal, which further aggravates myocardial ischemia and thereby worsens symptoms. Prompt surgical correction of ALCAPA is required as soon as the clinical diagnosis is confirmed, regardless of the patient’s age and cardiac function. The anomalous coronary artery is reimplanted into the aorta to reconstruct the dual coronary artery [9]. The prognosis following this operation is usually good. The postoperative mortality rate ranges from 0% to 16% [10], and 3% of patients require long-term reinterventions [1]. MR and pulmonary artery stenosis are the main reasons for reoperation [1]. In this study, the postoperative mortality rate was 10.4% (5/48), and the rate of reoperation during the follow-up period was 4.7% (2/43), which is consistent with previous reports.
Since the clinical manifestations of this disease are not specific, our study revealed that most patients presented with symptoms such as shortness of breath, and developmental delays. However, in the group with unfavorable prognosis, feeding difficulties were more common. Younger patients are more likely to experience respiratory distress (p = 0.04), whereas older patients are more likely to experience chest pain as the initial symptom (p = 0.004) [9]. In some centers, younger surgical age may also be associated with death, possibly due to insufficient development of coronary artery collateral vessels, more severe ventricular ischemia or dysfunction, or both [11]. Multivariate Cox regression analysis revealed that age at surgery (p = 0.077) was closely related to unfavorable prognosis.
For the cardiac malformations of ALCAPA, there are various surgical strategies, include coronary artery reimplantations, anomalous coronary artery ligation, Takeuchi tunneling, coronary artery bypass grafting, anastomosis to the subclavian artery. At present, the preferred method is coronary artery reimplantations [12]. Dehaki et al. [13] analyzed 21 patients of ALCAPA. The surgical methods included aortic reimplantation (n = 10), ostial closure (n = 8), and ligation (n = 3). The results showed that there was no significant difference in the postoperative mortality rate and medium-to-long-term prognosis among the patients. In this study, all the surgeries were coronary artery reimplantations. All the surgeries were performed by the same team of surgeons who were qualified to carry out ALCAPA surgeries, so there was no impact on prognosis due to the technical skills or operational proficiency of the surgeons.
Whether to simultaneously repair the mitral valve during ALCAPA surgery has always been controversial. Some scholars [14] believe that in patients with ALCAPA, MR is associated with ischemic left ventricular dilation, circular enlargement, and papillary muscle dysfunction. After restoring dual coronary artery blood supply, left ventricular function improves and the degree of MR decreases. There is no evidence suggesting that MR is related to early death. To shorten the aortic cross-clamping time and simultaneously treat mitral valve diseases. However, Bicer et al. [15] reported that although the rate of mitral valve reintervention is relatively low, it is still necessary to pay attention if MR exceeds moderate severity before the operation. Early implementation of mitral valve repair can improve early postoperative cardiac output and facilitate early recovery of cardiac function [1]. Thomas et al. [16] found in the long-term follow-up of 228 children with ALCAPA who underwent surgical correction, among patients with moderate or severe MR, that the risk of death was 28% lower when undergoing mitral valvuloplasty. Although some studies have reported that MR is a risk factor for postoperative death, the degree of MR did not predict any of our postoperative outcomes [17]. In this study, it was found that the degree of MR was not significantly associated with prognosis. 28 patients had moderate or severe MR before the operation. Among them, 23 patients underwent mitral valve repair simultaneously. Only one patient had severe MR due to gradually worsening mitral regurgitation 6 years after the operation and thus underwent MR repair again. Five patients did not undergo mitral valve repair; two of them died, and three survived. During the follow-up period, MR results recovered to a mild state in these three patients.
In this study, a longer CPB time (p = 0.022) and length of stay (p = 0.01) were risk factors for an unfavorable prognosis. This is the first report indicating that a longer CPB time and longer length of stay may be related to postoperative prognosis. In the Cox model, an interaction term between CPB time and the preoperative LVEF value was added. The result showed no significance.
Early mortality is significantly associated with left ventricular dysfunction. While advancements in surgical techniques and pharmacological therapies have mitigated the adverse effects on left ventricular function, numerous studies have demonstrated that preoperative left ventricular dysfunction remains an independent predictor of early mortality [10,15]. This study corroborates these findings, showing that lower preoperative LVEF values adversely affect prognosis. If there is no left ventricular dysfunction, the prognosis after surgery is favorable [18]. In a retrospective study of infants and children with ALCAPA, Kudumula et al. [19] reported that 88% of patients achieved left ventricular function recovery during a long-term follow-up of 20 years. The literature reports a median duration of left ventricular recovery of 4 months, with some cases requiring nearly one year for complete functional restoration [20]. Occasionally, prolonged recovery may indicate the gradual reversal of these adaptive cellular changes. Postoperative myocardial depression may sharply exacerbate cardiac dysfunction, that is, persistent transient mechanical dysfunction that persists after reperfusion and the restoration of normal coronary artery blood flow [21]. In this study, five patients died, all of whom presented with significantly reduced preoperative LVEFs. During the postoperative follow-up period, echocardiography revealed that the left ventricular function of all the surviving patients, especially those with a low LVEF before the operation, had fully recovered. The median time to LVEF recovery was 118 days, and the longest was 604 days.
The main limitations of this study include its retrospective design, single-center nature, relatively small sample size, and potential selection bias due to the exclusion of patients with complex congenital heart disease. Additionally, the exclusive reliance on echocardiography for cardiac function assessment may not provide a comprehensive evaluation of left ventricular contractility.
ALCAPA is a rare congenital anomaly of coronary artery anatomy. Early surgical intervention achieves satisfactory outcomes and improves left ventricular function in survivors. Younger surgical age, reduced preoperative LVEF, longer CPB time, and longer length of stay are the key risk factors that affect the prognosis in patients.
Acknowledgement:
Funding Statement: Guangzhou Major Clinical Technology Project (2023C-ZD10). Guangzhou Science and Technology Bureau Project (2024A03J1165). The Basic and Applied Basic Research Project of Guangzhou Municipal Science and Technology Bureau (2025A03J4265).
Author Contributions: Jia Yuan was responsible for the writing of the article. Yanxing Lv was responsible for the analyzed data. Xinyuan Ding performed the collected preoperative data. Li Ma, Kamil Bildebayev and Boiko Yuliya Nikolaevna were responsible for the surgery. Yunyi Zeng, Hang Yang and Lin Jiang were responsible for registering the postoperative follow-up data of patients. Na Zhou designed the research study. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Ethics Approval: This study was reviewed and approved by Guangzhou Women and Children’s Medical Center, Guangzhou Medical University (Approval No.212A01), and informed consent was obtained from the parents of the patients. This retrospective study was conducted in accordance with the Declaration of Helsinki.
Conflicts of Interest: The authors declare no conflicts of interest.
Abbreviation
| ALCAPA | Anomalous origin of the left coronary artery from the pulmonary artery |
| MR | Mitral regurgitation |
| LVEF | Left ventricular ejection fraction |
| LVEDD | Left ventricular end diastolic diameter |
| NT-proBNP | N-terminal pro-B-type natriuretic peptide |
| CPB | Cardiopulmonary bypass |
| ECMO | Extracorporeal membrane oxygenation |
References
1. Yu J, Ren Q, Liu X, Chen T, Rong L, Wen S, et al. Anomalous left coronary artery from the pulmonary artery: outcomes and management of mitral valve. Front Cardiovasc Med. 2022;9:953420. doi:10.3389/fcvm.2022.953420. [Google Scholar] [CrossRef]
2. Brancaccio G, Chinali M, Dionisi S, Trezzi M, Esposito C, Iacobelli R, et al. A meta-analysis of mitral surgery in patients undergoing surgery for anomalous left coronary artery: when to perform repair? Pediatr Cardiol. 2025;46(8):2531–41. doi:10.1007/s00246-024-03712-5. [Google Scholar] [CrossRef]
3. Ivanauskiene T, Zuoziene G, Zakarkaite D, Tarutis V, Glaveckaite S. The anterior mitral valve leaflet prolapse as a key to diagnosis of ALCAPA syndrome. J Am Coll Cardiol. 2024;84(9):868–73. doi:10.1016/j.jacc.2024.06.021. [Google Scholar] [CrossRef]
4. Lotman EM, Karu K, Mikkel M, Elmet M. Late adult presentation of ALCAPA syndrome: need for a new clinical classification? A case report and literature overview. Eur Heart J Case Rep. 2020;4(6):1–5. doi:10.1093/ehjcr/ytaa318. [Google Scholar] [CrossRef]
5. Kazmierczak PA, Ostrowska K, Dryzek P, Moll JA, Moll JJ. Repair of anomalous origin of the left coronary artery from the pulmonary artery in infants. Interact CardioVascular Thorac Surg. 2013;16(6):797–801. doi:10.1093/icvts/ivt061. [Google Scholar] [CrossRef]
6. Mishra A. Surgical management of anomalous origin of coronary artery from pulmonary artery. Indian J Thorac Cardiovasc Surg. 2021;37(1):131–43. doi:10.1007/s12055-021-01147-8. [Google Scholar] [CrossRef]
7. Pettersen MD, Du W, Skeens ME, Humes RA. Regression equations for calculation of z scores of cardiac structures in a large cohort of healthy infants, children, and adolescents: an echocardiographic study. J Am Soc Echocardiogr. 2008;21(8):922–34. doi:10.1016/j.echo.2008.02.006. [Google Scholar] [CrossRef]
8. Ismail M, Jijeh A, Alhuwaymil RM, Alahmari R, Alshahrani R, Almutairi R, et al. Long-term outcome of the anomalous origin of the left coronary artery from the pulmonary artery (ALCAPA) in children after cardiac surgery: a single-center experience. Cureus. 2020;12(12):e11829. doi:10.7759/cureus.11829. [Google Scholar] [CrossRef]
9. Straka N, Gauvreau K, Huang Y, DeWitt E, Allan C, Thiagarajan R, et al. Analysis of perioperative and long-term outcomes among presentations of anomalous left coronary artery from the pulmonary artery diagnosed beyond infancy versus during infancy. Pediatr Cardiol. 2025;46(1):139–47. doi:10.1007/s00246-023-03344-1. [Google Scholar] [CrossRef]
10. Zhang C, Zhang H, Yan J, Hua Z, Song Y, Sun H, et al. Mid-term outcome for anomalous origin of the left coronary artery from the pulmonary artery. Heart Lung Circ. 2020;29(5):766–71. doi:10.1016/j.hlc.2019.04.018. [Google Scholar] [CrossRef]
11. Azakie A, Russell JL, McCrindle BW, Van Arsdell GS, Benson LN, Coles JG, et al. Anatomic repair of anomalous left coronary artery from the pulmonary artery by aortic reimplantation: early survival, patterns of ventricular recovery and late outcome. Ann Thorac Surg. 2003;75(5):1535–41. doi:10.1016/s0003-4975(02)04822-1. [Google Scholar] [CrossRef]
12. George A, Pavithran S, Sivakumar K, Agarwal R. Anomalous left coronary artery origin from the nonfacing sinus of pulmonary artery—successful repair of a rare entity. Ann Pediatr Cardiol. 2024;17(4):288–91. doi:10.4103/apc.apc_129_24. [Google Scholar] [CrossRef]
13. Dehaki MG, Al-Dairy A, Rezaei Y, Ghavidel AA, Omrani G, Givtaj N, et al. Mid-term outcomes of surgical repair for anomalous origin of the left coronary artery from the pulmonary artery: in infants, children and adults. Ann Pediatr Cardiol. 2017;10(2):137–43. doi:10.4103/0974-2069.205140. [Google Scholar] [CrossRef]
14. Wang Z, Ma K, Li S. Progress of surgical treatment of anomalous left coronary artery from the pulmonary artery. Cardiol Young. 2024;34(7):1411–23. doi:10.1017/S1047951124025381. [Google Scholar] [CrossRef]
15. Biçer M, Korun O, Yurdakök O, Çiçek M, Dedemoğlu M, Özdemir F, et al. Anomalous left coronary artery from the pulmonary artery repair outcomes: preoperative mitral regurgitation persists in the follow-up. J Card Surg. 2021;36(2):530–5. doi:10.1111/jocs.15247. [Google Scholar] [CrossRef]
16. Thomas AS, Chan A, Alsoufi B, Vinocur JM, Kochilas L. Long-term outcomes of children operated on for anomalous left coronary artery from the pulmonary artery. Ann Thorac Surg. 2022;113(4):1223–30. doi:10.1016/j.athoracsur.2021.07.053. [Google Scholar] [CrossRef]
17. Brown JW, Ruzmetov M, Parent JJ, Rodefeld MD, Turrentine MW. Does the degree of preoperative mitral regurgitation predict survival or the need for mitral valve repair or replacement in patients with anomalous origin of the left coronary artery from the pulmonary artery? J Thorac Cardiovasc Surg. 2008;136(3):743–8. doi:10.1016/j.jtcvs.2007.12.065. [Google Scholar] [CrossRef]
18. Bichali S, Giroux N, Benbrik N, Liet JM. Unexplained near-drowning can reveal ALCAPA in children. Arch Pediatr. 2021;28(3):252–4. doi:10.1016/j.arcped.2020.12.001. [Google Scholar] [CrossRef]
19. Kudumula V, Mehta C, Stumper O, Desai T, Chikermane A, Miller P, et al. Twenty-year outcome of anomalous origin of left coronary artery from pulmonary artery: management of mitral regurgitation. Ann Thorac Surg. 2014;97(3):938–44. doi:10.1016/j.athoracsur.2013.11.042. [Google Scholar] [CrossRef]
20. Hu R, Zhang W, Yu X, Zhu H, Zhang H, Liu J. Midterm surgical outcomes for ALCAPA repair in infants and children. Thorac Cardiovasc Surg. 2022;70(1):2–9. doi:10.1055/s-0041-1725978. [Google Scholar] [CrossRef]
21. Schwartz ML, Jonas RA, Colan SD. Anomalous origin of left coronary artery from pulmonary artery: recovery of left ventricular function after dual coronary repair. J Am Coll Cardiol. 1997;30(2):547–53. doi:10.1016/s0735-1097(97)00175-7. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools