 Open Access
Open Access
ARTICLE
Impact of a Multifaceted Prevention Program on Ventilator-Associated Pneumonia in a Surgical Pediatric Cardiac ICU
1 Department of Pediatric Intensive Care Unit, National Center for Cardiovascular Disease and Fuwai Hospital, Chinese Academy of Medical Sciences, Peking Union Medical College, Beijing, China
2 Beijing Key Laboratory of Surveillance, Early Warning and Pathogen Research on Emerging Infectious Diseases, Beijing Center for Disease Prevention and Control, Beijing, China
3 School of Public Health, Capital Medical University, Beijing, China
4 Department of Infection Control, Peking University First Hospital, Beijing, China
5 Department of Infection Control, National Center for Cardiovascular Disease and Fuwai Hospital, Chinese Academy of Medical Sciences, Peking Union Medical College, Beijing, China
* Corresponding Author: Xu Wang. Email:
# These authors contributed equally to this paper as first author
Structural and Congenital Heart Disease 2026, 21(1), 4 https://doi.org/10.32604/schd.2026.077612
Received 13 December 2025; Accepted 06 March 2026; Issue published 31 March 2026
Abstract
Background: This study evaluated the impact of a comprehensive prevention program, which integrated eight evidence-based measures consistent with current clinical guidelines and practice standards, on ventilator-associated pneumonia (VAP) rates in a pediatric cardiac surgical intensive care unit (ICU). Methods: A quasi-experimental study was conducted from 2023 to 2024. We compared VAP rates across a 5-month pre-intervention period, a 12-month intervention period, and a 7-month post-intervention period in patients receiving mechanical ventilation for over 48 h. Additional outcomes, including postoperative length of stay were also assessed before and after the intervention. Results: Among 829 at-risk patients and 5677 ventilator-days, the VAP rates per 1000 ventilator-days were 25.7, 11.3, and 10.8 in the pre-intervention, intervention, and post-intervention periods, respectively. Poisson regression identified the intervention and post-intervention periods as protective factors for VAP. After adjusting for age, weight, emergency surgery, and cardiopulmonary bypass duration, VAP rates decreased by 56% during the intervention period (adjusted incidence rate ratio (IRR) 0.382, 95% CI 0.212–0.691; p = 0.001) and by 58% in the post-intervention period (adjusted IRR 0.452, 95% CI 0.232–0.882; p = 0.020). Postoperative length of stay also declined significantly from 22 (16, 35) days pre-intervention to 21 (15, 28) days post-intervention (p = 0.040). Conclusions: Implementation of the multidisciplinary prevention program was associated with a sustained reduction in VAP rates in a surgical pediatric cardiac ICU and may contribute to shorter postoperative hospital stays.Keywords
Ventilator-associated pneumonia (VAP) ranks among the most prevalent nosocomial infections in intensive care units (ICUs) [1]. Roughly 10% of patients receiving mechanical ventilation for over 48 h go on to develop VAP [2]. VAP is associated with elevated mortality rates [3], prolonged hospital stays, and increased healthcare costs [4]. A growing body of research has demonstrated reduced VAP incidence rates linked to the implementation of preventive care bundles [5,6,7].
Congenital heart disease ranks as the leading cause of morbidity and mortality among all birth defects worldwide [8]. Surgical intervention constitutes a cornerstone of treatment for this condition. While all patients receive mechanical ventilation intraoperatively, most can be extubated shortly after surgery; however, a subset of patients with complex lesions or critical illness requires prolonged mechanical ventilation for over 48 h [9]. Following cardiac surgery, patients often experience surgical stress-induced dysregulated inflammatory responses, which lead to systemic impairments including compromised immune function and consequently heighten their susceptibility to various infections [10]. VAP represents the most prevalent healthcare-associated infection in the postoperative period of cardiac surgery [11] and is strongly associated with adverse postoperative outcomes [12].
Therefore, infection prevention and control exert a significant impact on postoperative outcomes in pediatric cardiac surgery patients. Developed countries have established sophisticated infection prevention and control systems, yielding favorable outcomes in the management of congenital heart disease [13]. In recent years, notable progress has been made in healthcare-associated infection control in China [14]. However, a research gap persists globally, whether in developed or developing nations, regarding the efficacy of VAP prevention measures in pediatric cardiac ICUs.
Pediatric VAP research has predominantly focused on neonatal populations, particularly on infection prevention and control strategies in neonatal ICUs, such as training programs, hand hygiene adherence, sterile equipment handling, infant positioning to prevent gastroesophageal reflux, and continuous assessment of extubation readiness [15,16]. For non-neonatal pediatric patients, additional attention should be directed toward monitoring endotracheal tube cuff pressure [17]. Notably, no studies have been conducted on pediatric patients following cardiac surgery. Nevertheless, these evidence-based VAP prevention bundles are presumably applicable to this population and hold promise for reducing VAP incidence.
The primary objective of this prospective, interventional single-center study which incorporates eight preventive measures was to assess the effect of implementing a multifaceted VAP prevention program on VAP incidence rates in a pediatric cardiac surgical ICU. The secondary objectives were to evaluate the program’s impact on postoperative length of stay and mortality.
This study adopted a quasi-experimental, uncontrolled before-and-after design to analyze the effects of implementing a VAP prevention bundle. Only patients who received mechanical ventilation for more than 48 h (defined as the duration until first extubation) were consecutively enrolled in the analysis. Patients who declined to provide informed consent (or their guardians) and those with preoperative infections were excluded from the study. For eligible patients who underwent postoperative reintubation, the total duration of mechanical ventilation was calculated as the sum of all ventilation episodes. The flowchart of this study is shown in Fig. 1.
The study spanned two years and consisted of three periods. The duration of each study phase was predefined based on pilot data and ethical considerations to ensure methodological rigor and clinical relevance:
Pre-intervention period (January 2023–May 2023; 5 months): Our preliminary pilot study indicated that the primary outcome would stabilize at a baseline level within 5 months, providing a reliable reference for subsequent intervention comparisons. Extending this period was unnecessary, as baseline characteristics remained consistent beyond this timeframe, and prolonged baseline surveillance would not enhance data quality but would increase resource consumption.
Intervention period (June 2023–May 2024; 12 months): This duration was chosen to ensure full implementation, staff training, and consolidation of the multifaceted VAP prevention bundle. It allowed clinical teams to integrate the eight preventive measures into routine practice, address implementation barriers, and achieve stable compliance with the bundle components.
Post-intervention period (June 2024–December 2024; 7 months): A longer post-intervention period was designed for two key purposes: (1) To evaluate the sustainability of the intervention effect, as the maintenance of preventive behaviors relied on institutionalized protocols and staff training after the formal intervention ceased, a 7-month follow-up was necessary to confirm that reductions in VAP rates were not transient. (2) To capture potential time-dependent effects, including delayed benefits or rebound effects.
This study has been reviewed and approved by the Ethics Committee of the National Center for Cardiovascular Disease and Fuwai Hospital (ID: 2022-1859). The informed consent forms were obtained from all patients/statutory guardians. The ethical principles outlined in the 2013 Helsinki Declaration were followed throughout the study. The work has been reported in line with the STROCSS criteria [18].
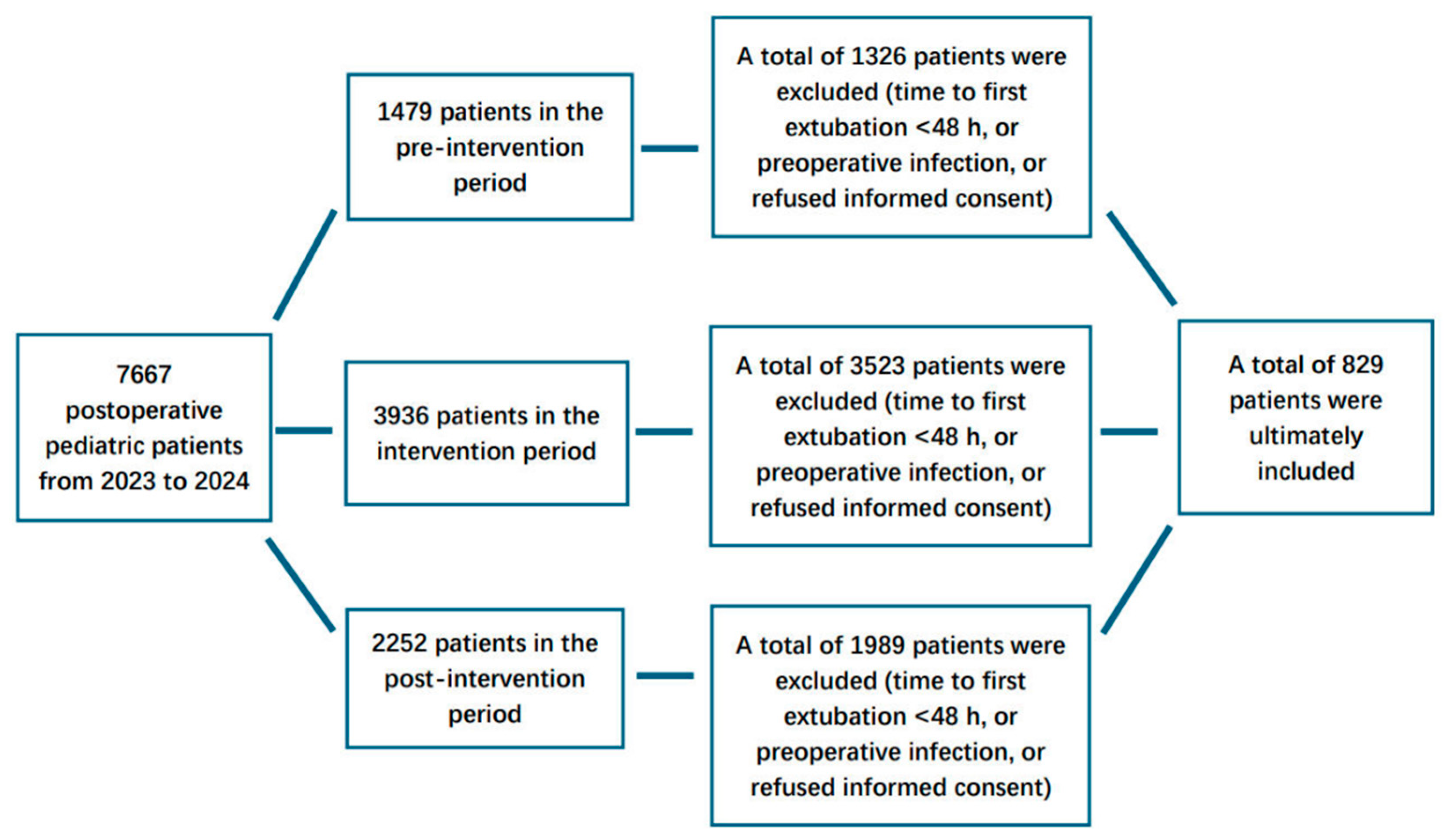
Figure 1: The flowchart of the study.
Our hospital is a tertiary teaching hospital specializing in cardiovascular diseases. The Pediatric Cardiac Surgery Center performs approximately 3500–4000 cardiac surgeries annually. All patients receive postoperative care in the 46-bed pediatric cardiac ICU. Staffing levels are dynamically adjusted based on patient census, with minimal fluctuation across shifts or over time. The nursing team maintains a minimum headcount of 120 members, with each nurse assigned to care for no more than two patients at any given time. Similarly, the physician team consists of at least 20 clinicians, with each physician responsible for a maximum of nine patients. Notably, two physicians and one nurse serve as part-time infection prevention and control personnel, collaborating closely with full-time infection prevention and control staff. Additionally, the hospital’s infection prevention and control department has designated a dedicated full-time specialist to be stationed at our ICU.
Our hospital initiated active surveillance of VAP in pediatric patients in January 2023, which included VAP incidence rates and relevant variables such as age, gender, weight, genetic abnormalities, underweight status, emergency surgery, duration of mechanical ventilation, cardiopulmonary bypass duration, Risk Adjustment in Congenital Heart Surgery (RACHS-1) classification for the surgical procedure, economic burden, dates of ICU admission and discharge, ventilation initiation and cessation dates, VAP onset dates, microorganism profiles, and mortality. During the initial phase of the study (pre-intervention period, January to May 2023), due to insufficient experience in VAP management, only compliance with hand hygiene practices was monitored.
Subsequently, we engaged two experts to provide professional expertise and technical support, while also having them participate fully in the program implementation. By leveraging a questionnaire survey to integrate current clinical guidelines with our institutional practice, these experts assisted us in formulating a tailored VAP prevention bundle. Subglottic suctioning was excluded from the bundle, as our pediatric tracheal intubation devices were not equipped to support this procedure. Furthermore, the two experts guided us in conducting monthly VAP process surveillance, took part in our monthly VAP prevention education sessions and feedback meetings, and remained available during working hours to address any questions raised by our management team. This period, spanning from June 2023 to May 2024, was designated as the intervention phase of the program.
During the intervention period, in addition to consulting two external experts, our team (the ICU-based internal infection control team and the hospital’s full-time infection control personnel) completed nationally accredited healthcare-associated infection control training and obtained relevant professional certification. This equipped the hospital’s full-time and part-time infection control staff with the capability to independently maintain the same rigorous VAP bundle monitoring and management standards during the post-intervention period, without the need to rely on external experts.
During the program’s follow-up period (post-intervention phase, June 2024 to December 2024), the ICU team took primary responsibility for ongoing VAP surveillance, while the hospital’s Infection Control Department conducted monthly VAP process surveillance, as well as delivering regular training sessions and comprehensive feedback. For staff training, a multimodal protocol integrating lectures, assessments and scenario-based practical operations was implemented to improve adherence to the VAP prevention care bundles.
We adopted the VAP definitions established by the National Health Safety Network, as this standard is particularly well-suited for our cohort of postoperative pediatric patients. Per these criteria, VAP is diagnosed in mechanically ventilated patients who present with new or progressive infiltrates, consolidations, cavitations, or pleural effusions on chest radiography. In addition, the patient must meet at least one of the following clinical and microbiological criteria: (1) new onset of purulent sputum or a change in sputum character; (2) positive blood cultures for a pathogenic organism; (3) isolation of an etiologic agent from specimens obtained via tracheal aspiration, bronchial brushing, bronchoalveolar lavage, or tissue biopsy [19]. All VAP diagnoses were made by two specially trained physicians from our ICU team. Throughout the two-year study duration, the diagnostic criteria were strictly standardized and consistently applied, with the same team of physicians responsible for all diagnoses to ensure diagnostic uniformity. Moreover, all patients received treatment under the direct supervision of the ICU director, thereby guaranteeing consistency in therapeutic strategies.
Other VAP criteria, such as the Clinical Pulmonary Infection Score, the Hospital in Europe Link for Infection Control through Surveillance, or the Johanson criteria, were not adopted. These systems assign significant weight to abnormal body temperature and white blood cell count. However, following cardiac surgery, patients typically exhibit transient but marked leukocytosis, and their body temperature is actively managed through various interventions. Thus, these criteria are not suitable in this specific postoperative context.
2.5 VAP Bundle Components [5,6,7,20]
- (1)Adherence to hand-hygiene guidelines
- (2)Maintenance of patients in a semi-recumbent position (30–45° elevation of the head of the bed)
- (3)Performance of daily assessments of readiness to wean and the use of weaning protocols
- (4)Performance of comprehensive regular oral care with 0.2% chlorhexidine-gluconate aqueous solution, every 8 h
- (5)Maintenance of an endotracheal cuff pressure 20–30 cm H2O
- (6)Removal of the condensate from ventilator circuits and keeping the ventilator circuit closed during condensate removal; change of the ventilator circuit only when visibly soiled or malfunctioning
- (7)Daily active mobilization (Conducted daily by bedside nurses, physicians and rehabilitation therapist to confirm the patient’s readiness for mobilization, excluding the following contraindications: unstable blood pressure, active postoperative bleeding, uncorrected arrhythmias, fraction of inspired oxygen (FiO2) >0.6, deep vein thrombosis. For patients with muscle weakness [Grade 0–2] or those unable to follow commands [due to young age or sedation], perform passive, movement or body touch [infants/toddlers] twice daily, 15 min per session. Deliver neuromuscular electrical stimulation as an adjunct for insufficient muscle strength, with patient repositioning every 2 h. For command-following patients, implement active movement combined with griping, throwing, cycling, sitting up in the bed. Conduct these training sessions twice daily, 20 min per session, and reposition the patient every 2 h. Safety: Vital signs [including heart rate, blood pressure, oxygen saturation, and respiratory rate] were monitored throughout the procedure. The intervention was immediately discontinued if intolerance occurred, such as a >5% drop in oxygen saturation, excessive heart rate, or significant fatigue)
- (8)Avoidance of gastric overdistention (Feeding Management: For enteral nutrition, intermittent feeding is adopted, with feeding intervals of 3 to 4 h. Symptom Monitoring: Nurses measure residual volume via the feeding tube using a syringe before each feeding, and monitor abdominal circumference, vomiting, or diarrhea. Intervention Threshold: Residual volume >50% of the previous feeding volume. If the threshold is exceeded, the impending feed is withheld once, and the physician is notified to assess for gastrointestinal motility disorders); avoidance of histamine receptor 2-blocking agents and proton pump inhibitors. Since we were unable to determine in advance whether patients would require mechanical ventilation for more than 48 h following their transfer from the operating room to the ICU, we implemented standardized VAP prevention measures for all patients.
Statistical analysis was performed using the SPSS software (version 26.0; IBM Corporation, USA). The Kolmogorov-Smirnov test was used to assess the conformity of the continuous variables to the normal distribution. Normal distribution was expressed as the mean plus or minus the standard deviation, whereas a non-normal distribution was expressed as the median accompanied by the interquartile range. Categorical variables are displayed in a numerical form, accompanied by their respective percentages. The Wilcoxon rank-sum test was used for nonnormally distributed variables. Categorical variables were analyzed using the chi-squared test, correction for continuity, or Fisher’s exact test. We computed the number of VAP episodes per 1000 ventilator-days at risk (until VAP diagnosis or extubation). We analyzed the effect of the intervention on the rates of VAP episodes by segmented regression analysis using a Poisson model. The model included an intercept and three main period effects (before–during intervention–after intervention). All statistical tests were two-tailed and a p-value < 0.05 was considered statistically significant.
2.7 Sample Size and Statistical Power
A sample size calculation was performed using PASS software (version 15.0; NCSS Corporation, USA), with the primary outcome as the core reference and secondary outcomes incorporated to ensure adequate power for all key endpoints (α = 0.05, two-tailed; power = 0.80):
Primary outcome (VAP rate per 1000 ventilator-days): Based on preliminary data showing a VAP rate of 25/1000 ventilator-days and a clinically meaningful 40% relative reduction (to 15/1000 ventilator-days), accounting for a 10% loss to follow-up, the required sample size was ≥780 at-risk patients.
Secondary outcome 1 (postoperative length of stay): Based on preliminary data indicating a mean length of stay of 22 days (SD = 15), and targeting a 15% reduction (to a mean of 18.7 days, SD = 12), the required sample size was estimated to be at least 620 at-risk patients.
Secondary outcome 2 (postoperative mortality): Based on preliminary data indicating a mortality rate of 3.5%, and aiming for a clinically relevant reduction to 1%, the required sample size was calculated to be at least 1096 at-risk patients.
Of the 7667 total postoperative pediatric patients admitted to our ICU, 829 at-risk patients were observed, accounting for 5677 ventilation days at risk. A total of 84 patients were diagnosed with VAP, of whom 9 cases (10.7%) were confirmed via bronchoalveolar lavage. The detailed pathogen distribution of VAP cases is presented in Table 1. Stratified by study phase, the pre-intervention period included 153 at-risk patients and 1399 ventilation days at risk; the intervention period included 413 at-risk patients and 3349 ventilation days at risk; and the post-intervention period included 263 at-risk patients and 929 ventilation days at risk. Statistically significant differences were observed in age, body weight, emergency surgery status, and cardiopulmonary bypass duration among patients across the three study periods. Detailed demographic and surgery-related parameters are summarized in Table 2.
Table 1: The details of pathogen distribution of ventilator-associated pneumonia cases.
| Pathogens | Cases (n = 84) |
|---|---|
| Stenotrophomonas maltophilia | 17 (20.23%) |
| Burkholderia cepacia | 11 (13.09%) |
| Acinetobacter baumannii | 10 (11.90%) |
| Klebsiella pneumoniae | 9 (10.71%) |
| Pseudomonas aeruginosa | 8 (9.52%) |
| Escherichia coli | 7 (8.33%) |
| Staphylococcus aureus | 6 (7.14%) |
| Candida parapsilosis | 4 (4.76%) |
| Enterobacter cloacae | 4 (4.76%) |
| Candida albicans | 3 (3.57%) |
| Serratia marcescens | 2 (2.38%) |
| Elizabethkingia meningoseptica | 1 (1.19%) |
| Enterococcus Faecium | 1 (1.19%) |
| Enterococcus faecalis | 1 (1.19%) |
Table 2: The details of demographic and surgery-related parameters in the three periods.
| Variable | Pre-Intervention (n = 153) | Intervention (n = 413) | Post-Intervention (n = 263) | p-Value |
|---|---|---|---|---|
| Age (month) | 8 (3, 29) | 5 (2, 15) | 5 (2, 10) | 0.004 |
| Gender Male (case, %) | 82 (54) | 223 (54) | 162 (62) | 0.114 |
| Weight (kg) | 7.4 (5.4, 12) | 6.2 (4.6, 9.1) | 5.9 (4.3, 8.9) | 0.005 |
| Genetic abnormalities (case, %) | 1 (0.7) | 0 (0) | 3 (1.1) | 0.107 |
| Underweight (case, %) | 13 (8.5) | 33 (7.9) | 15 (5.7) | 0.452 |
| Emergency surgery (case, %) | 0 (0) | 3 (0.7) | 7 (2.7) | 0.025 |
| Duration of mechanical ventilation (hours) | 65 (48, 120) | 50 (48, 119) | 50 (48, 113) | 0.821 |
| Duration of cardiopulmonary bypass (minutes) | 113 (97, 175) | 119 (83, 162) | 133 (95, 168) | 0.027 |
| Cardiopulmonary bypass surgery (case, %) | 147 (96.1) | 387 (93.7) | 239 (90.9) | 0.109 |
| RACHS-1 category | 3 (2, 3) | 3 (2, 3) | 3 (2, 3) | 0.445 |
| RACHS-1 category ≥ 4 (case, %) | 23 (15) | 57 (14) | 44 (17) | 0.582 |
| Economic burden (1000 Chinese yuan) | 154.5 (117.7, 213.7) | 143.2 (110.3, 210.1) | 148.0 (112.2, 205.3) | 0.460 |
A total of 183, 449, and 293 hand hygiene compliance evaluations were conducted during the pre-intervention, intervention, and post-intervention periods, respectively. For other preventive measures, a total of 2001 and 1371 evaluations were performed in the intervention and post-intervention periods, respectively. Detailed information on all preventive measures is presented in Table 3.
Table 3: The details of preventive measures in the three periods.
| Pre-Intervention | Intervention | Post-Intervention | |
|---|---|---|---|
| Hand-hygiene | |||
| Compliant/Total (number) | 105/183 | 324/449 | 192/293 |
| Compliance rate (%) | 57.4 | 72.1 | 65.5 |
| Semi-recumbent position | - | ||
| Compliant/Total (number) | 178/218 | 137/164 | |
| Compliance rate (%) | 81.7 | 83.5 | |
| Oral care | - | ||
| Compliant/Total (number) | 400/449 | 261/293 | |
| Compliance rate (%) | 89.1 | 89.1 | |
| Active mobilization | - | ||
| Compliant/Total (number) | 186/231 | 109/129 | |
| Compliance rate (%) | 80.5 | 84.5 | |
| Cuff pressure | - | ||
| Compliant/Total (number) | 180/218 | 144/164 | |
| Compliance rate (%) | 82.6 | 87.8 | |
| Daily trial of ventilation weaning | - | ||
| Compliant/Total (number) | 182/218 | 149/164 | |
| Compliance rate (%) | 83.5 | 90.9 | |
| Ventilator circuit | - | ||
| Compliant/Total (number) | 178/218 | 137/164 | |
| Compliance rate (%) | 81.7 | 83.5 | |
| Avoidance of gastric overdistention | - | ||
| Compliant/Total (number) | 360/449 | 216/293 | |
| Compliance rate (%) | 80.3 | 73.8 |
The VAP rates (expressed as events per 1000 ventilator days) across the three study periods were 25.7, 11.3, and 10.8, respectively. Time-series plots depicting the dynamic changes in VAP rates are presented in Fig. 2. For the Poisson regression analysis, we incorporated variables that exhibited statistically significant differences across the three periods, namely age, body weight, emergency surgery status, and cardiopulmonary bypass duration. The regression results indicated that only the intervention and post-intervention phases served as independent protective factors against VAP. After adjusting for age, body weight, emergency surgery status, and cardiopulmonary bypass duration, the VAP rate was reduced by 56% in the intervention period (adjusted incidence rate ratio (IRR) = 0.382, 95% confidence interval (CI): 0.212–0.691; p = 0.001). In the post-intervention period, the VAP rate decreased by 58% (adjusted IRR = 0.452, 95% CI: 0.23–0.882; p = 0.020). Detailed results of the Poisson regression analysis are summarized in Table 4.
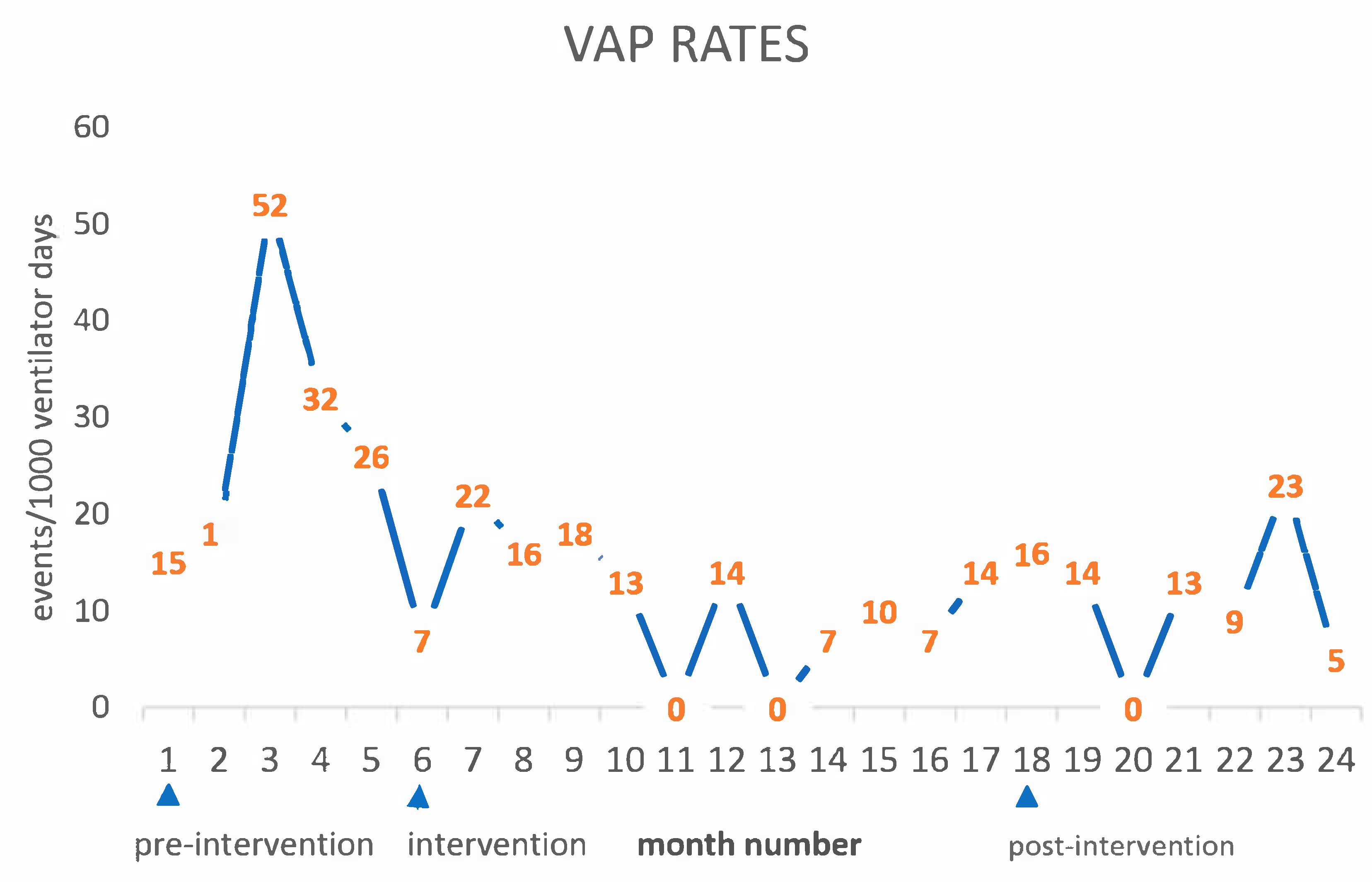
Figure 2: The time-series plots of ventilator-associated pneumonia rates.
Table 4: The results of Poisson regression.
| Variable | HR | 95% CI | p-Value |
|---|---|---|---|
| Intervention period | 0.382 | 0.212–0.691 | 0.001 |
| Post-intervention period | 0.452 | 0.232–0.882 | 0.020 |
| Age | 0.939 | 0.862–1.022 | 0.145 |
| Weight | 1.361 | 0.886–2.089 | 0.159 |
| Emergency surgery | 0.781 | 0.452–1.348 | 0.375 |
| cardiopulmonary bypass | 1.005 | 0.988–1.021 | 0.581 |
The analysis of outcome measures across the pre-intervention, intervention, and post-intervention periods revealed that the postoperative length of stay was 22 (16, 35) days, 22 (15, 34) days, and 21 (15, 28) days respectively. A significant decrease occurred between the pre- and post-intervention phases, with a p-value of 0.040. Regarding postoperative mortality, the number of cases and corresponding percentages were 5/153 (3.3%), 4/413 (0.97%), and 4/263 (1.5%) in each period respectively. Although there was a decline of over 50% in mortality from the pre- to post-intervention period, no statistically significant differences were observed (p = 0.410). Additionally, the hospitalization costs for patients during the pre-intervention, intervention, and post-intervention phases were 154.5 (117.7, 213.7), 143.2 (110.3, 210.1), and 148.0 (112.2, 205.3) ×1000 Chinese yuan, respectively. Although the values showed a decrease, the results were not statistically significant (p = 0.460).
Our data indicate that the implementation of a multifaceted VAP prevention program was associated with a sustained reduction in VAP incidence rates. A key distinctive feature of this study is the emphasis on the long-term sustainability of these VAP prevention measures. While numerous studies have demonstrated that infection control interventions can reduce VAP rates, far fewer have documented long-term, sustained improvements in outcomes [5,21]. One plausible explanation for this sustained efficacy may be attributed to the internal champion team composed of ICU staff. This part-time team was established out of professional commitment and interest in infection control, with no additional financial compensation provided. The presence of this clinical team ensured the consistent implementation of infection control measures; moreover, through close collaboration with the hospital’s full-time infection control department, it helped maintain the sustainability of infection control gains. This finding is highly consistent with the conclusions of existing implementation and survey studies [22,23]. Of course, supervision and feedback from the infection control department were equally critical. Relying solely on the ICU’s internal infection control team, despite improvements in professional skills through training, would not yield optimal infection control outcomes due to the absence of external oversight “pressure” [24].
The critical role of the VAP prevention bundle also merits emphasis. During the pre-intervention period, although hand hygiene compliance was already approaching a relatively high level (approximately 60%) [25], the VAP rate remained suboptimal, which we attribute to the absence of standardized VAP surveillance and management protocols. Nevertheless, following the implementation of the multifaceted VAP prevention bundle, the VAP rate decreased by over 50%, representing a clinically significant improvement. Post-intervention, the VAP rate was reduced to approximately 11 events per 1000 ventilator days; however, we refrain from interpreting the clinical significance of this absolute value. This is primarily because there is currently a paucity of published data on VAP rates in this specific population of pediatric patients following congenital heart disease surgery, precluding direct comparisons with findings from other clinical centers.
Published literature on VAP in pediatric populations reports VAP rates ranging from 8.1 to 12 events per 1000 ventilator days [1,26], while studies focusing on adult patients following cardiac surgery document a VAP rate of 15.9 events per 1000 ventilator days [6]. Given that pediatric patients undergoing congenital heart disease surgery exhibit impaired immune function and are consequently more susceptible to infections [10], we can infer that the outcomes achieved in our study represent satisfactory infection control efficacy. It is also critical to emphasize, however, that VAP diagnostic criteria vary substantially across different studies. For this reason, direct comparisons of VAP rates derived from distinct investigations should be avoided. The key highlight of this study remains that our VAP prevention program yielded sustained reductions in VAP rates during both the intervention and post-intervention periods.
During the evaluation process, we also observed that owing to the unique clinical characteristics of postoperative pediatric patients, certain components of the VAP prevention bundle including the maintenance of a semi-recumbent position, daily active mobilization, and daily trials of ventilation weaning did not achieve optimal compliance. This suboptimal adherence was not attributable to procedural errors, but rather to unavoidable clinical factors such as unstable hemodynamic status, elevated risk of postoperative hemorrhage, and generalized debilitation following major cardiac surgery. Nevertheless, resolving these cardiac surgery-specific infection control challenges will undoubtedly elevate our VAP prevention capabilities to a new level. This interdisciplinary initiative can be effectively spearheaded by our ICU-based infection control team. Our part-time infection prevention and control personnel, through their bedside immersion in critical care settings, are uniquely positioned to conduct granular observations of infection control practices, strike an optimal balance between infection prevention protocols and patient safety priorities, and thereby improve overall compliance with the VAP prevention bundle.
Numerous factors are associated with VAP development in pediatric patients following cardiac surgery, such as young age and the high complexity of surgical procedures [27]. In our study, although statistically significant differences existed across the three study periods in terms of age, body weight, emergency surgery status, and cardiopulmonary bypass duration, subsequent Poisson regression analysis demonstrated that only the intervention and post-intervention phases were identified as protective factors against VAP. Nationwide congenital heart disease screening in China has reduced older patient volumes and lowered diagnostic ages. Infants and young children undergoing cardiac surgery mostly have severe/complex defects, and delayed intervention would threaten their growth and survival. With local hospitals now managing simple defects, our center only receives complex or critically ill referrals, increasing surgical complexity and emergency cases. This high-risk patient profile prompted us to enhance infection control measures. Despite greater patient severity, our comprehensive infection control bundle effectively reduced VAP rates, which is the core clinical value of our study.
In addition, this study demonstrated that the VAP prevention bundle reduced the mortality rate from 3.3% to a range of 0.97%–1.5%. Despite this greater than 50% reduction, the difference did not reach statistical significance. Analysis of the reasons indicates that the actual enrolled sample size in this study was 829 cases, which is lower than the expected 1096 cases for mortality, and this may have contributed to the failure to achieve a statistically significant difference. Therefore, extending the follow-up period or increasing the sample size could potentially detect meaningful differences. It should be noted that, for severe complications such as mortality, even in the absence of statistical significance, every single reduction in cases translates to meaningful improvements in surgical outcomes from a clinical perspective. A similar trend was noted for hospitalization costs: although the reduction did not achieve statistical significance, it still holds potential economic benefits for individual families and public health insurance systems.
Our study was not blinded, and it is well known that VAP definitions carry some subjectivity. Although we used a standard definition and the methodology did not change over time, this inherent subjectivity still poses a risk of misclassification. Misclassification of VAP cases could lead to inaccurate calculation of VAP rates and potentially distort the results of the study.
Due to the potential influence of the Hawthorne effect, we cannot confirm that compliance remained consistent during periods when observations were not conducted. If adherence was in fact lower during these unobserved intervals, the reported decline in VAP rates may be overestimated. Future studies can mitigate this limitation by implementing electronic monitoring systems and consumables-based compliance tracking, both of which minimize observer bias and allow continuous, objective assessment.
As a single-center study conducted in a tertiary teaching hospital with specialized resources (e.g., dedicated infection control personnel, a high nurse-to-patient ratio), the findings may not be generalizable to smaller healthcare facilities or resource-limited settings. To address this issue, we have provided as detailed an account as possible of the VAP prevention and control process, enabling hospitals at various levels to implement prevention measures according to their own circumstances. In addition, this problem can be solved by conducting multi-center research in future research.
While our study was conducted in a resource-rich tertiary center, the core components of the VAP bundle can be adapted for resource-limited settings to enhance generalizability. Regarding low-cost alternatives, manual manometers or the minimal occlusive volume technique can replace expensive electronic devices for cuff pressure monitoring. Furthermore, task-sharing strategies hold promise in settings with lower nurse-to-patient ratios; for instance, training junior staff to assist with patient repositioning, head-of-bed elevation, and basic mobilization under professional supervision. Such adaptations ensure that the essential elements of VAP prevention remain feasible and sustainable across diverse clinical environments.
Our study adopted a quasi-experimental, uncontrolled before-and-after design. Owing to the lack of randomization and a concurrent control group, we cannot rule out the potential impact of confounding factors, such as secular trends in healthcare quality or advancements in surgical techniques, which may have contributed to the observed reduction in VAP rates. Consequently, this limits our ability to draw definitive causal inferences from the study findings. These limitations highlight the necessity of future research employing more rigorous, higher-evidence study designs, such as randomized controlled trials.
Another limitation is the lack of subgroup analyses on the efficacy of individual VAP bundle components, preventing us from disentangling their independent contributions to VAP reduction. Given the unique physiological and clinical characteristics of postoperative pediatric cardiac patients, the effectiveness of different bundle measures may vary, and this information is crucial for clinical optimization. Future studies should incorporate subgroup analyses based on compliance with each component to inform the development of personalized VAP prevention strategies.
Our VAP prevention program was associated with a sustained reduction in VAP rates. Additionally, it may exert a beneficial effect on shortening postoperative length of stay.
Acknowledgement:
Funding Statement: This study was supported by the National High Level Hospital Clinical Research Funding (2025-GSP-QN-7, 2025-GSP-GG-19 and 2025-GSP-GG-11).
Author Contributions: Xu Wang and Xiaofeng Wang contributed to the conception and design of the study; Xiaofeng Wang, Qian Zhang and Ya Gao contributed to the acquisition of the data; Da Huo and Shuo Li contributed to Statistical analysis of the data; Da Huo and Shuo Li contributed to the interpretation of the data; Xiaofeng Wang and Wenlong Wang wrote the first draft of the article; Xu Wang, Da Huo and Shuo Li revised the article. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: The data that support the findings of this study are available from the corresponding author, Xu Wang, upon reasonable request.
Ethics Approval: This study has been reviewed and approved by the Ethics Committee of the National Center for Cardiovascular Disease and Fuwai Hospital (ID: 2022-1859). The informed consent forms were obtained from all patients/statutory guardians. The ethical principles outlined in the 2013 Helsinki Declaration were followed throughout the study.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Osman S , Al Talhi YM , AlDabbagh M , Baksh M , Osman M , Azzam M . The incidence of ventilator-associated pneumonia (VAP) in a tertiary-care center: Comparison between pre- and post-VAP prevention bundle. J Infect Public Health. 2020; 13( 4): 552– 7. doi:10.1016/j.jiph.2019.09.015. [Google Scholar] [CrossRef]
2. Metersky ML , Wang Y , Klompas M , Eckenrode S , Mathew J , Krumholz HM . Temporal trends in postoperative and ventilator-associated pneumonia in the United States. Infect Control Hosp Epidemiol. 2023; 44( 8): 1247– 54. doi:10.1017/ice.2022.264. [Google Scholar] [CrossRef]
3. Vacheron CH , Lepape A , Savey A , Machut A , Timsit JF , Comparot S , et al. Attributable mortality of ventilator-associated pneumonia among patients with COVID-19. Am J Respir Crit Care Med. 2022; 206( 2): 161– 9. doi:10.1164/rccm.202202-0357OC. [Google Scholar] [CrossRef]
4. Nanao T , Ishikawa KB , Ikeda S , Yamazaki T . Clinical and economic impact of ventilator-associated pneumonia in intensive care units in Japan. Infect Prev Pract. 2025; 7( 1): 100440. doi:10.1016/j.infpip.2025.100440. [Google Scholar] [CrossRef]
5. Landelle C , Nocquet Boyer V , Abbas M , Genevois E , Abidi N , Naimo S , et al. Impact of a multifaceted prevention program on ventilator-associated pneumonia including selective oropharyngeal decontamination. Intensive Care Med. 2018; 44( 11): 1777– 86. doi:10.1007/s00134-018-5227-4. [Google Scholar] [CrossRef]
6. Su KC , Kou YR , Lin FC , Wu CH , Feng JY , Huang SF , et al. A simplified prevention bundle with dual hand hygiene audit reduces early-onset ventilator-associated pneumonia in cardiovascular surgery units: An interrupted time-series analysis. PLoS One. 2017; 12( 8): e0182252. doi:10.1371/journal.pone.0182252. [Google Scholar] [CrossRef]
7. DeLuca LA , Walsh P , Davidson DD , Stoneking LR , Yang LM , Grall KJH , et al. Impact and feasibility of an emergency department–based ventilator-associated pneumonia bundle for patients intubated in an academic emergency department. Am J Infect Control. 2017; 45( 2): 151– 7. doi:10.1016/j.ajic.2016.05.037. [Google Scholar] [CrossRef]
8. Su Z , Zou Z , Hay SI , Liu Y , Li S , Chen H , et al. Global, regional, and national time trends in mortality for congenital heart disease, 1990-2019: An age-period-cohort analysis for the Global Burden of Disease 2019 study. EClinicalMedicine. 2022; 43: 101249. doi:10.1016/j.eclinm.2021.101249. [Google Scholar] [CrossRef]
9. Epstein R , Ohliger SJ , Cheifetz IM , Malay S , Shein SL . Trends in time to extubation for pediatric postoperative cardiac patients and its correlation with changes in clinical outcomes: A virtual PICU database study. Pediatr Crit Care Med. 2022; 23( 7): 544– 54. doi:10.1097/PCC.0000000000002950. [Google Scholar] [CrossRef]
10. Li M , Nie Y , Yang Z . The association between systemic immune-inflammation index (SII) and early nosocomial infections after cardiopulmonary bypass surgery in children with congenital heart disease. BMC Cardiovasc Disord. 2024; 24( 1): 698. doi:10.1186/s12872-024-04378-w. [Google Scholar] [CrossRef]
11. Yu X , Chen M , Liu X , Chen Y , Hao Z , Zhang H , et al. Risk factors of nosocomial infection after cardiac surgery in children with congenital heart disease. BMC Infect Dis. 2020; 20( 1): 64. doi:10.1186/s12879-020-4769-6. [Google Scholar] [CrossRef]
12. Klompas M , Branson R , Cawcutt K , Crist M , Eichenwald EC , Greene LR , et al. Strategies to prevent ventilator-associated pneumonia, ventilator-associated events, and nonventilator hospital-acquired pneumonia in acute-care hospitals: 2022 Update. Infect Control Hosp Epidemiol. 2022; 43( 6): 687– 713. doi:10.1017/ice.2022.88. [Google Scholar] [CrossRef]
13. Pasquali SK , He X , Jacobs ML , Hall M , Gaynor JW , Shah SS , et al. Hospital variation in postoperative infection and outcome after congenital heart surgery. Ann Thorac Surg. 2013; 96( 2): 657– 63. doi:10.1016/j.athoracsur.2013.04.024. [Google Scholar] [CrossRef]
14. Su C , Zhang Z , Zhao X , Peng H , Hong Y , Huang L , et al. Changes in prevalence of nosocomial infection pre- and post-COVID-19 pandemic from a tertiary Hospital in China. BMC Infect Dis. 2021; 21( 1): 693. doi:10.1186/s12879-021-06396-x. [Google Scholar] [CrossRef]
15. Elsaeed BIK , Atta MHR , Fouda ME , Ahmed HAE , El Demerdash D , Elzlbany GAM . Effect of implementing training programme for nurses about care bundle on prevention of ventilator-associated pneumonia among newborns. Nurs Crit Care. 2025; 30( 2): e70000. doi:10.1111/nicc.70000. [Google Scholar] [CrossRef]
16. Alriyami A , Kiger JR , Hooven TA . Ventilator-associated pneumonia in the neonatal intensive care unit. Neoreviews. 2022; 23( 7): e448– 61. doi:10.1542/neo.23-7-e448. [Google Scholar] [CrossRef]
17. Antalová N , Klučka J , Říhová M , Poláčková S , Pokorná A , Štourač P . Ventilator-associated pneumonia prevention in pediatric patients: Narrative review. Children. 2022; 9( 10): 1540. doi:10.3390/children9101540. [Google Scholar] [CrossRef]
18. Rashid R , Sohrabi C , Kerwan A , Franchi T , Mathew G , Nicola M , et al. The STROCSS 2024 guideline: Strengthening the reporting of cohort, cross-sectional, and case-control studies in surgery. Int J Surg. 2024; 110( 6): 3151– 65. doi:10.1097/JS9.0000000000001268. [Google Scholar] [CrossRef]
19. Rahimibashar F , Miller AC , Yaghoobi MH , Vahedian-Azimi A . A comparison of diagnostic algorithms and clinical parameters to diagnose ventilator-associated pneumonia: A prospective observational study. BMC Pulm Med. 2021; 21( 1): 161. doi:10.1186/s12890-021-01527-1. [Google Scholar] [CrossRef]
20. Guanche-Garcell H , Morales-Pérez C , Rosenthal VD . Effectiveness of a multidimensional approach for the prevention of ventilator-associated pneumonia in an adult intensive care unit in Cuba: Findings of the International Nosocomial Infection Control Consortium (INICC). J Infect Public Health. 2013; 6( 2): 98– 107. doi:10.1016/j.jiph.2012.11.009. [Google Scholar] [CrossRef]
21. Álvarez-Lerma F , Palomar-Martínez M , Sánchez-García M , Martínez-Alonso M , Álvarez-Rodríguez J , Lorente L , et al. Prevention of ventilator-associated pneumonia: The multimodal approach of the Spanish ICU “pneumonia zero” program. Crit Care Med. 2018; 46( 2): 181– 8. doi:10.1097/CCM.0000000000002736. [Google Scholar] [CrossRef]
22. Pence BW , Gaynes BN , Udedi M , Kulisewa K , Zimba CC , Akiba CF , et al. Two implementation strategies to support the integration of depression screening and treatment into hypertension and diabetes care in Malawi (SHARP): Parallel, cluster-randomised, controlled, implementation trial. Lancet Glob Health. 2024; 12( 4): e652– 61. doi:10.1016/S2214-109X(23)00592-2. [Google Scholar] [CrossRef]
23. Wang X , Li S , Huo D , Wang S , Wang W , He H , et al. Healthcare-associated infection management in 62 ICUs for patients with congenital heart disease in China: A survey study. Int J Surg. 2024; 110( 4): 2025– 33. doi:10.1097/JS9.0000000000001138. [Google Scholar] [CrossRef]
24. Bénet T , Allaouchiche B , Argaud L , Vanhems P . Impact of surveillance of hospital-acquired infections on the incidence of ventilator-associated pneumonia in intensive care units: A quasi-experimental study. Crit Care. 2012; 16( 4): R161. doi:10.1186/cc11484. [Google Scholar] [CrossRef]
25. Mouajou V , Adams K , DeLisle G , Quach C . Hand hygiene compliance in the prevention of hospital-acquired infections: A systematic review. J Hosp Infect. 2022; 119: 33– 48. doi:10.1016/j.jhin.2021.09.016. [Google Scholar] [CrossRef]
26. Rosenthal VD , Álvarez-Moreno C , Villamil-Gómez W , Singh S , Ramachandran B , Navoa-Ng JA , et al. Effectiveness of a multidimensional approach to reduce ventilator-associated pneumonia in pediatric intensive care units of 5 developing countries: International Nosocomial Infection Control Consortium findings. Am J Infect Control. 2012; 40( 6): 497– 501. doi:10.1016/j.ajic.2011.08.005. [Google Scholar] [CrossRef]
27. Tang CW , Liu PY , Huang YF , Pan JY , Lee SS , Hsieh KS , et al. Ventilator-associated pneumonia after pediatric cardiac surgery in southern Taiwan. J Microbiol Immunol Infect. 2009; 42( 5): 413– 9. [Google Scholar]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools