 Open Access
Open Access
ARTICLE
Prolonged survival and risk factors for reintervention after inflatable penile prosthesis implantation
1 Department of Urology, Andrology, and Kidney Transplantation, Pasteur 2 Hospital, University Hospital of Nice, Nice, 06000, France
2 Clinique Saint-Jean, Cagnes-Sur-Mer, 06800, France
3 INSERM U1081-CNRS UMR 7284, University of Nice Côte d’Azur, Nice, 06000, France
* Corresponding Author: Skander Zouari. Email:
Canadian Journal of Urology 2026, 33(1), 173-184. https://doi.org/10.32604/cju.2025.064281
Received 11 February 2025; Accepted 30 September 2025; Issue published 28 February 2026
Abstract
Background: Inflatable Penile Prosthesis (IPP) is the treatment for erectile dysfunction (ED) refractory to pharmacological therapies. Long-term data on factors associated with prosthesis survival remain unclear. This study aimed to analyze the long-term survival of penile prostheses and identify risk factors associated with survival without reintervention. Methods: This is a retrospective, single-center study of patients who underwent IPP implantation between January 2014 and December 2022. Preoperative data related to the patient and the etiology of ED, as well as perioperative data, were collected. The primary outcome was survival without reintervention, defined as prosthesis revision or explantation due to mechanical dysfunction or infection. We conducted survival analyses without reintervention and searched for risk factors using a multivariate Cox model. Results: In total, 33 out of 137 patients underwent reintervention (24.1%), including 24 (17.5%) prosthesis revisions and 9 (6.6%) had explantations. Median follow-up was 39 months with an interquartile range (IQR) of 9.00 to 62.00. Median survival without reintervention was 7 years. In univariate analysis, downsizing (p = 0.046) was associated with reintervention. Smoking (p = 0.003) and age (p = 0.034) were associated with prosthesis explantation. The number of implantations (p = 0.009) was associated with prosthesis revision. Multivariate analysis by the Cox model did not identify any independent predictive factors for reintervention. Conclusion: Smoking may play a role in infection post-IPP implantation. Primary implantations seem to be associated with better survival. Adjusting cylinder size, known as downsizing, is likely to be a proxy for the complexity of the procedure and thus linked to earlier reintervention.Keywords
Inflatable Penile Prosthesis (IPP) implantation is the main treatment for erectile dysfunction (ED) that is refractory to pharmacological therapies.1 Thanks to advancements in surgical techniques and prosthesis design, patient satisfaction has improved, and complication rates have decreased.2,3 Despite these improvements, challenges remain, particularly in managing complications such as infection and mechanical malfunction. These complications not only affect patients’ well-being but also have implications for healthcare costs and long-term outcomes.4 Among these, postoperative infection has drawn the most attention due to its severity and impact.
Preventing infection is key to improving outcomes in IPP procedures. Many risk factors have been identified and are broadly categorized as either patient-related or procedure-related. These include immunosuppression, diabetes, spinal cord injury, revision surgery, and concurrent procedures. Surgical techniques and perioperative protocols like antibiotic use, skin preparation, and cylinder placement also play a role.5 However, the literature remains inconclusive. Indeed, despite numerous studies, no consensus exists on which risk factors predict infection or mechanical failure the most strongly. Many exclude patients undergoing revision surgeries or do not clearly link revision to infection versus mechanical failure.6–9 There is also a lack of research into whether surgical variables influence outcomes differently in primary versus revision cases.10
We hypothesized that both patient-related and surgical procedure-related factors, including intraoperative adjustments, are associated with increased risk of postoperative complications, including infection and mechanical dysfunction, following IPP implantation. In this study, we aimed to explore the overall complication rate following IPP implantation in a real-world setting and to identify factors associated with infection and mechanical malfunction. We included a heterogeneous patient population, including revision cases, to better reflect everyday clinical practice.
Our study comprised a retrospective, single-center cohort of patients who underwent IPP implantation between January 2014 and December 2022 at the Pasteur 2 Hospital, University Hospital of Nice in France. All data were collected retrospectively from electronic and paper medical records. Inclusion criteria were all patients with erectile dysfunction who either failed or had an insufficient response to pharmacological treatments, defined as a course of medical treatment with PDE5 inhibitors; the use of prostaglandin E1 via intracavernosal injection, topical application, or intraurethral administration. We excluded patients with incomplete surgical records, those lost to postoperative follow-up, and individuals who had undergone IPP implantation at other institutions.
This study does not require ethical review, since it used anonymized retrospective data, complied with national guidelines, and the data is kept strictly confidential.
All patients underwent the placement of a three-piece IPP, which was either an AMS 700™ CX (Boston Scientific Corp., Boston, MA, USA) or a Titan® device (Coloplast Corp., Minneapolis, MN, USA). A preoperative urine culture was systematically performed, and positive cultures were treated for at least 48 h before surgery with an antibiotic based on the local antibiogram. The surgical approach was penoscrotal, with or without an inguinal counter incision for fluid reservoir placement. Antibiotic prophylaxis was systematic and in accordance with the current national guidelines. Cefazolin 2 g IV was administered 30 min before incision. Intraoperative irrigation was performed with gentamicin (80 mg) and rifampicin (600 mg) diluted in saline. For patients receiving standard prophylaxis, no additional postoperative antibiotics were given. In cases of prosthesis infection, vancomycin (15 mg/kg IV every 12 h) and gentamicin (5 mg/kg IV once daily) were initiated 48 h prior to explantation and subsequently tailored according to culture results.
The Foley catheter was routinely removed on postoperative day 1.
Outcomes and influencing factors
The primary outcome is the overall rate of complications requiring prosthesis revision or explantation, due to either infection or mechanical failure.
Secondary outcomes included the time to complication and identification of pre- or intraoperative factors associated with reintervention, as well as predictive factors for prosthesis removal or reoperation due to infection or mechanical dysfunction.
Factors potentially influencing prosthesis lifespan were categorized into preoperative, intraoperative, and postoperative variables. Preoperative factors included age, history of hypertension, etiology of erectile dysfunction, smoking status, body mass index (BMI), and whether the procedure was a primary implantation or a revision. Intraoperative variables included operative time, type of prosthesis, length of the corpus cavernosum, cylinder size, use and length of rear-tip extenders, presence of concomitant procedures, and use of an inguinal counter-incision for reservoir placement. Postoperative factors included the length of hospital stay and the occurrence of postoperative complications.
We summarized categorical variables using counts and percentages. Quantitative variables were reported using means ± standard deviation or medians with interquartile ranges (IQRs), depending on data distribution. Group comparisons were performed using Student’s t-test or Mann-Whitney U test for continuous variables, and chi-square or Fisher’s exact test for categorical variables. Kaplan-Meier survival analysis was used to estimate time to complication, with comparisons made via the log-rank test. Cox proportional hazards regression was performed to identify factors independently associated with reintervention. Variables with a p-value < 0.20 in univariate analysis were considered in the multivariate model. Proportional hazard assumptions were verified using Schoenfeld residuals, and no violations were found. Hazard ratios (HR) with 95% confidence intervals (CI) were calculated to assess the strength of the association between variables and the risk of reintervention or complications. In all statistical tests, the significance threshold was set at 0.05. The final model adjusted for potential confounders and interactions as necessary. All statistical analyses were performed using Microsoft Excel for Windows (Microsoft Corp., Redmond, WA, USA).
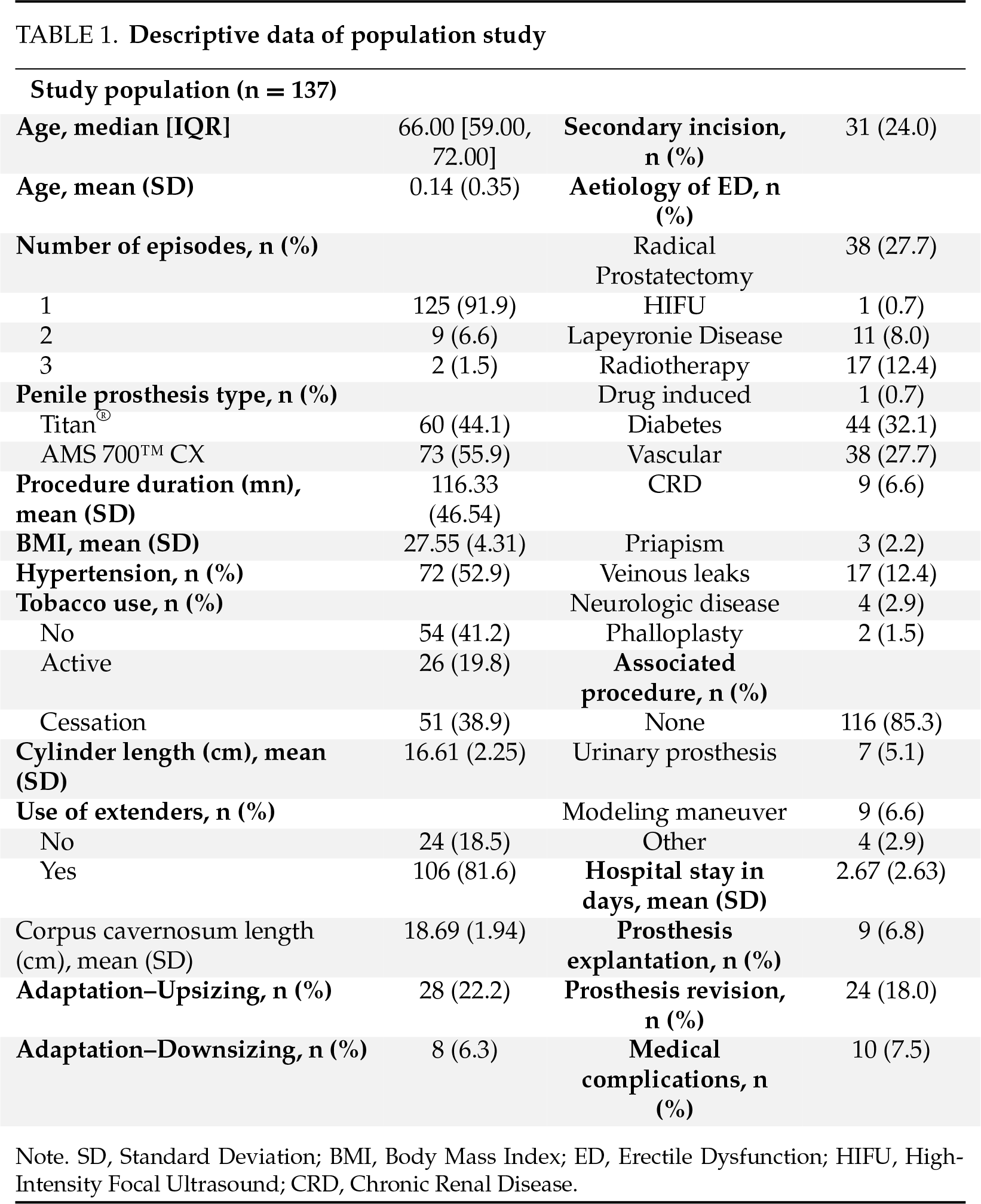
137 patients were included in the study. The median age was 66 years, IQR [59.00, 72.00]. This was a primary IPP implantation in 125 patients (91.9%). Active smoking was found in 26 patients (19.8%), and 32.1% of the patients had diabetes (n = 44). A history of radical prostatectomy was found in 38 patients (27.7%). The average operative time was 116 min. Intraoperatively, the mean corpus cavernosum length was 18.69 cm. Mean cylinder length was 16.61 cm. Rear-tip extenders (RTE) were used in 106 patients (81.6%). Upsizing or downsizing adjustment was used in 36 patients (28.5%). An associated procedure was performed in 20 patients (14.6%); the manual modeling maneuver was the most frequent in nine patients (6.6%). The mean length of hospital stay was 2.67 days. Descriptive characteristics of the population are summarized in Table 1.
The median follow-up was 39 months, IQR [9.00, 62.00]. Out of 137 patients, 33 (24.1%) experienced a complication requiring reintervention. Most were due to mechanical failure (24 patients, 17.5%), while 9 patients (6.6%) required explantation due to infection. The median survival without reintervention was 7 years (Figure 1). The overall rate of reoperation within 4 months following IPP implantation was found to be 7.4%. In univariate analysis, only downsizing (p = 0.046) was associated with the occurrence of complications as defined previously (Table 2). As shown in Figure 2, patients who underwent downsizing had a significantly shorter median time to complication (42 vs. 84 months, p = 0.028). Smoking (p = 0.003) and age (median age: 60 years, IQR, [55, 61] for those with IPP infection, versus 67 years, IQR [59.75, 72.00] for those without infection, p = 0.034) were the two factors significantly associated with explantation for prosthesis infection (Table 3). The number of implantation episodes (p = 0.009) was the only factor significantly associated with prosthetic revision for confirmed mechanical dysfunction during surgical exploration (Table 4). The multivariate analysis by the Cox model did not identify any independent predictive factors for reintervention (Figure 3).
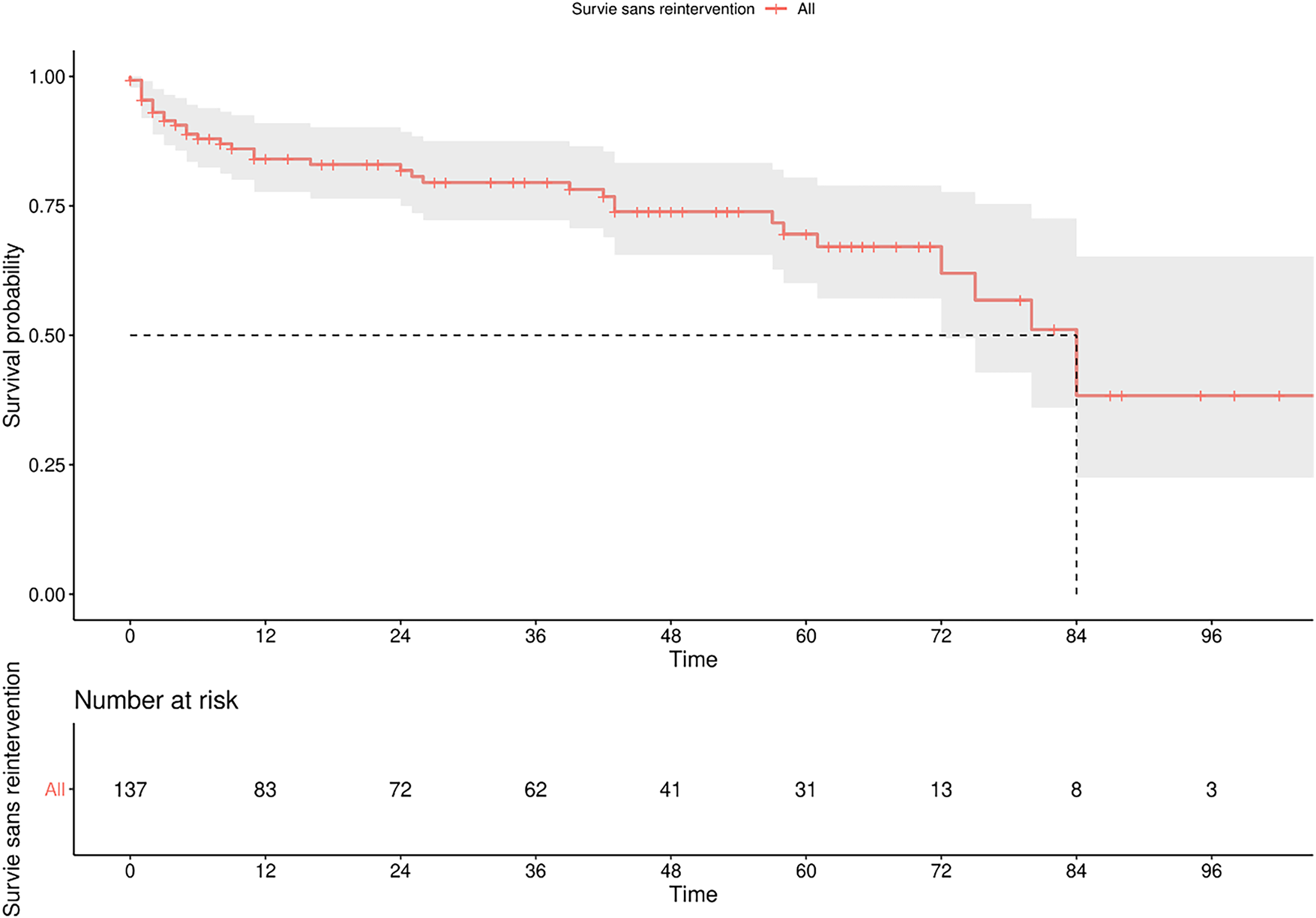
FIGURE 1. Penile prosthesis survival using Kaplan-Meier curve
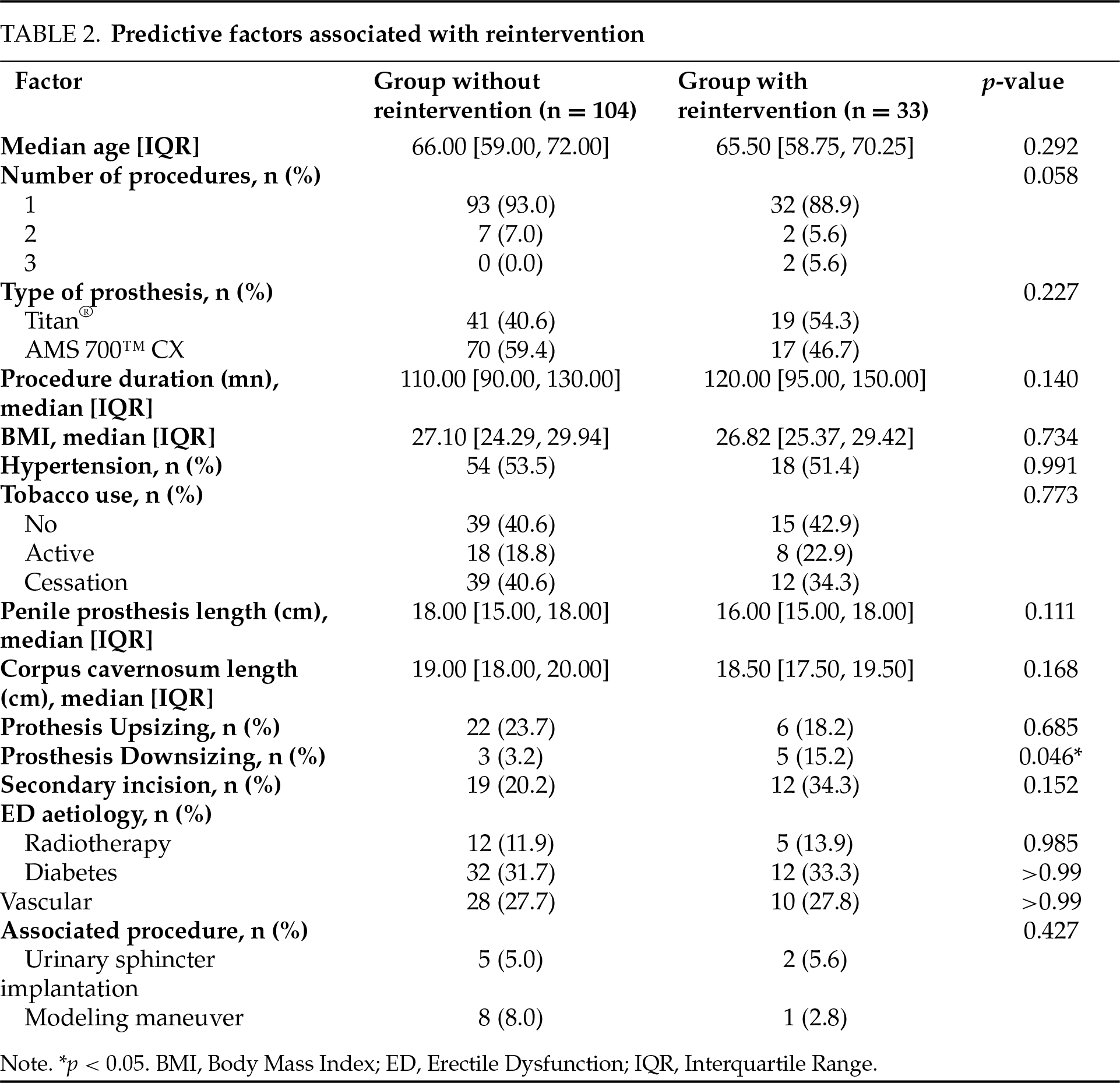
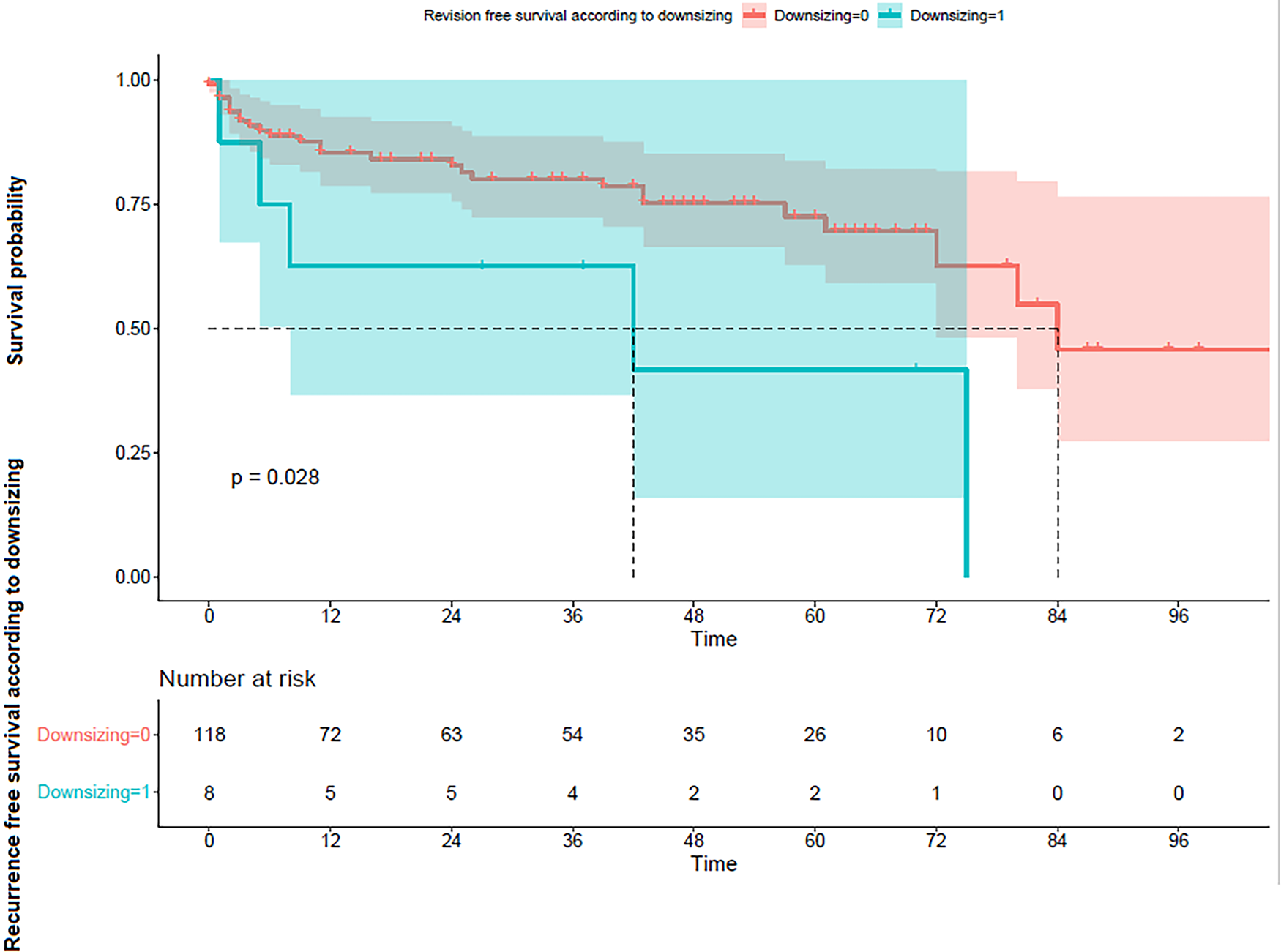
FIGURE 2. Survival according to subgroups analysis with (1) and without (0) downsizing using the Kaplan-Meier curve
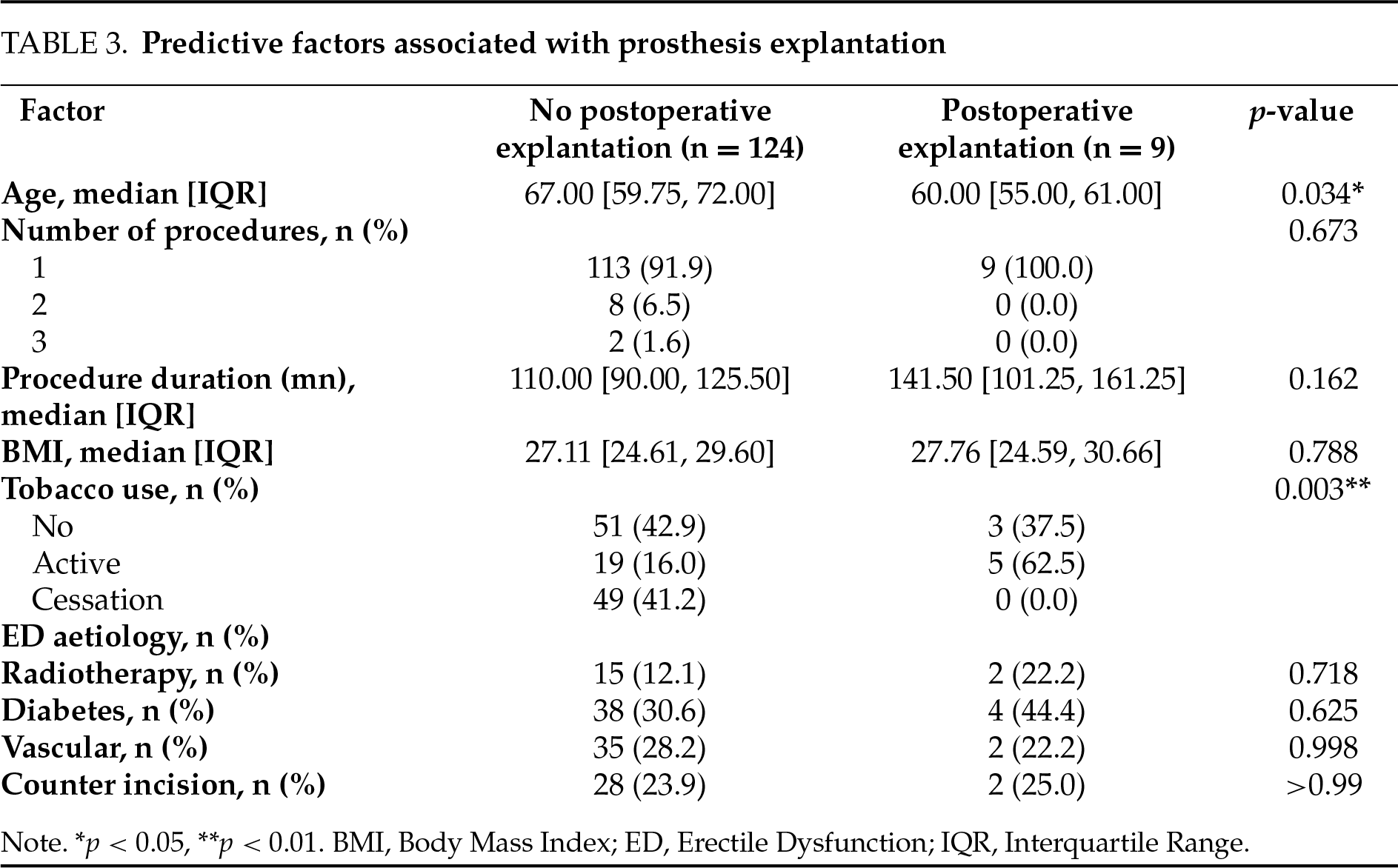
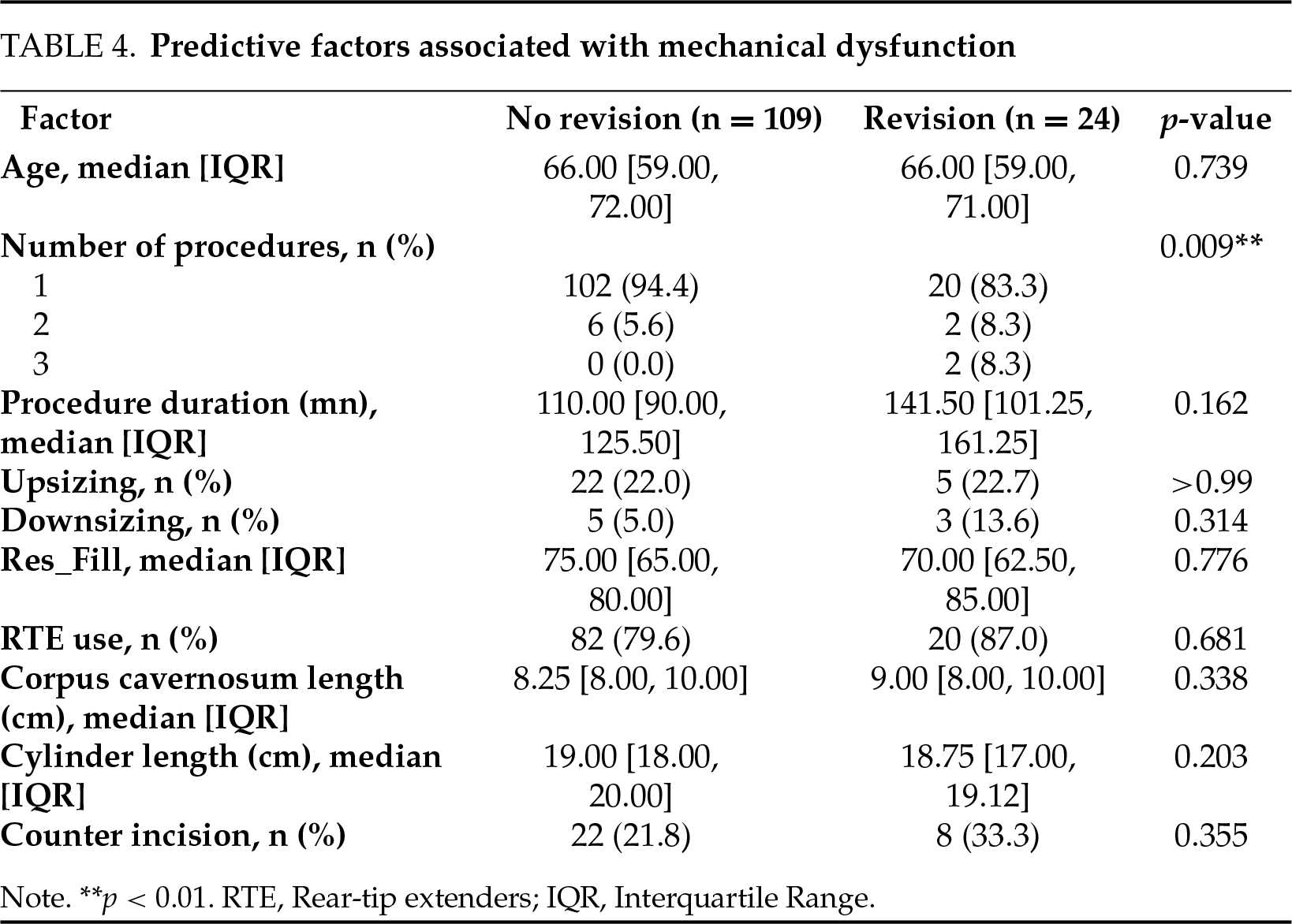
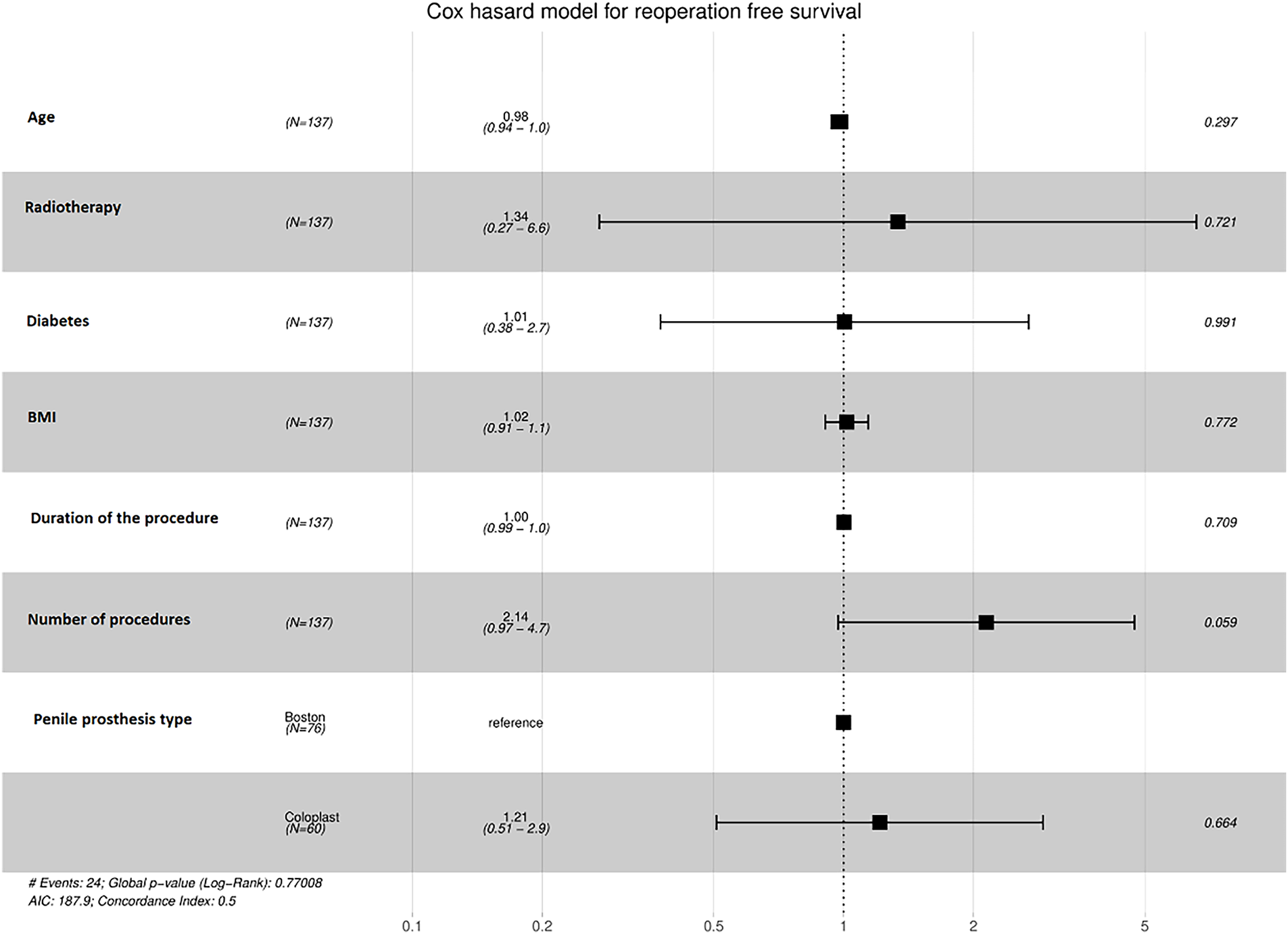
FIGURE 3. Cox hazard model for reintervention-free survival. Note: BMI, body mass index
There remains controversy over the long-term survival outcomes of IPP implantation and the factors associated to its failure. Two major complications can affect IPP survival: prosthesis infection leading to explantation and mechanical dysfunction requiring revision.
One of the largest retrospective series published to date, based on a Californian database including 7666 patients, found a reoperation rate of 11.2% at 5 years, lower than the rate observed in our series.11 These results may be explained by the monocentric nature of our series and the fact that the number of cases operated on in France is lower than that practiced in the United States. However, they are closer to those of Miller and Bennett,10 who found a revision rate of 20.7%. These results remain inconsistent in the literature, as some authors report an overall surgical revision rate exceeding 40%.12,13
One of the most serious complications is IPP infection. The average occurrence of IPP infection exhibited a decreasing trend over time, from an incidence ranging from 8% to 11% in the 1980s and 1990s, to 3 to 5% in the early 2000s. It is estimated to the current range of approximately 0.3% to 2.7% according to a recent meta-analysis.14 In a recent narrative review, infection rates were estimated to be as high as 1–3% in virgin cases and 7–18% in revision cases.15 However, many studies have excluded high-risk patients, such as those with a history of prior IPP implantation or infection.4,7,16 After adjusting for the inclusion of these at-risk characteristics, our study found the postoperative infection rate to be 6.8%, which is higher than the rates reported in recent studies and close to the rate of 8.2% found by Miller et al. in their study.10
The other major complication that may affect IPP survival is device malfunction. In the latest series to date about mechanical indications for IPP revisions, Smelser found a rate of 56.6% of IPP revisions due to mechanical failure,17 which was close to the previous rates reported before.18,19 In all these studies, tubing fracture was the most found, followed by reservoir leaks and pump malfunction. In our study, we did not delve into the specific causes of mechanical dysfunction, as the cause was not always clearly specified in the surgery reports.
Digging into the possible reasons for IPP reoperation after infection or mechanical dysfunction, implicated risk factors can be classified into patient-related and procedure-related factors.
Smoking has been explored in the context of IPP surgeries. It has been identified as a risk factor for glans necrosis, therefore infection following implantation, likely due to poor blood circulation.20 A comprehensive meta-analysis demonstrated a surgical site infection odds ratio of 1.79 among smokers compared to non-smokers. Additionally, it revealed a decreased infection risk (odds ratio 0.43) in patients who ceased smoking at least four weeks prior to their operation.21 Although there is a general lack of specific data regarding smoking and its influence on infection in IPP surgery, one retrospective review observed no significant rise in infection rates among patients who smoked,22 whereas some authors reported a significant relation between smoking and perioperative complications after IPP implantation.23,24 In our study, we found that active smoking was significantly correlated with IPP explantation (p = 0.003).
Regarding the role of diabetes, previous studies presented conflicting evidence for its implication as a predictive factor for postoperative infection. A study by Menshchikov found that diabetes, particularly when indicated by elevated HbA1c levels, is associated with an increased risk of penile prosthesis infection.24 However, more recent studies have demonstrated that patients with diabetes have a higher risk of IPP explantation and fungal infections,25,26 as well as a higher risk of developing fungal infections.27 Our findings didn’t establish a correlation between DM and groups of patients with surviving IPPs and those who developed an infection following revision surgery (44% and 30.6%, respectively; Fisher’s exact test p = 0.625), which was the same conclusion as in Henry et al. results (23% and 25%, respectively; p = 0.5077).28
Etiology of ED may play a role in the occurrence of IPP complications. Spinal cord injury, nerve damage, and neurogenic bladder have been linked to an increased risk of infection and overall IPP complication rate.22,29 The only study about the potential role of phalloplasty involved 247 patients undergoing prosthesis insertion, demonstrating an infection rate of 8.5% with a lack of significant predictive value for infection risk. Multiple studies indicated a minimal risk of infection in patients undergoing IPP post-prostatectomy, as the infection rate did not exceed 2.6% and no significant difference in infection rates between post-RP and vasculogenic erectile dysfunction patients has been found.30,31 In our study, no specific etiology appeared to be linked to the reoperation rate after IPP insertion.
Patients with a prior IPP may present a higher risk of IPP complications, as it was demonstrated in some previous studies.32–34 Regardless of their infection status, prior insertion of an IPP increased the likelihood of requiring explantation compared to IPP-naïve patients. Furthermore, while initial IPP typically carries a 1–3% infection rate, the risk of reinfection in salvage cases has been reported to escalate to as high as 10%.25 In our study, we found a correlation between prior IPP insertions and the reoperation rate due to mechanical dysfunction, but not overall complications or IPP-linked infection.
Many surgical related characteristics such as intraoperative findings (size of the corpora cavernosa or the presence of fibrosis); technical procedure (adaptation in upsizing or downsizing as an adjustment in the cylinder longer or shorter than corpus cavernosum length, final prosthesis length or the choice of making an inguinal counter incision or not to place the reservoir, concomitant procedure) or related to the IPP (prosthesis manufacturer, cylinder size, use and length of RTE and reservoir volume) have been scarcely or not studied at all in the literature. Bellaiche et al., in their study, found that IPP manufacturer and final size of the prosthesis were predictive factors influencing IPP survival (1.89, 95% CI [1.03–3.45]) and (0.27, 95% CI [0.09–0.77]), respectively.12 A retrospective review conducted by Henry et al. in 2009 revealed notably shorter cylinder lengths in prostheses placed, elevated complication rates, and reduced revision-free survival associated with IPP implantation when performed by low-volume surgeons.35 Vanderstappen et al. demonstrated that the more distal the corporotomy, the higher the risk of cylinder dislocation among a series of 15 patients who had a rerouting surgery after IPP insertion.36 In our study, cylinder downsizing was significantly associated with earlier reintervention. Downsizing typically occurs when intraoperative findings—such as corporal fibrosis, anatomical mismatch, or difficulty achieving optimal cylinder fit—necessitate the use of a shorter implant than initially anticipated. As such, downsizing may serve as a surrogate marker for greater surgical complexity. These complex cases may involve altered corporal compliance, prior scarring, or technical limitations that predispose to device instability or malfunction. It was correlated in our study with the occurrence of complications, whether due to infection or mechanical dysfunction. This suggests that technical or anatomical challenges contributing to downsizing may ultimately compromise prosthesis durability. To our knowledge, this is the first study to report a correlation between downsizing and IPP survival.
Several other factors have been assessed in the literature in their role with infection following IPP implantation, such as immunosuppression, prior use of radiation, antibiotic-coated IPP use, preoperative and perioperative use of antibiotics, and surgeon experience;10,12,15,37 but have not been explored in our study due to the lack of data.
However, we found that the postoperative IPP infection rate was correlated with age. Age as a patient’s related characteristic has not been reported as a factor for IPP infection in the literature.38 The discrepancy between the previously cited data and prior study findings may stem from potential non-linear associations, alongside confounding factors like higher rates of smoking, diabetes mellitus, and predominant neurogenic etiology among young patients, ultimately resulting in the insignificance of the association between past IPP procedures and complications when controlling for other variables.
Our study has several limitations: Firstly, its retrospective nature, relatively limited sample size, and heterogeneous patient follow-up. While the median follow-up period is 39 months, some patients had short follow-ups due to data collection cessation, while others were followed for over 10 years, leading to unequal follow-up bias. Four different surgeons, with varying levels of experience, some of whom operated independently while others supervised junior surgeons, performed the surgeries in our study. This may have influenced the complication rates.
Secondly, we defined the time of participation from device implantation to the first removal or surgical revision event, which may not necessarily correlate with the onset of implant dysfunction.
Furthermore, in our study, we defined complications as either revising IPP for mechanical dysfunction or explantation due to infection. However, there are other non-mechanical dysfunctions besides infection that may require reintervention and are considered complications that have not been included in our sample study.17
Another limitation is the lack of detailed data on the specific causes of mechanical dysfunction, as these were not consistently reported in the surgical reports for each revision. This absence of data limits the ability to fully assess the impact of mechanical failure on the overall outcomes of IPP implantation. Preoperative urine culture and antibiotic sensitivity data were also not consistently available for all patients, limiting our ability to include this variable in our statistical analysis.
Lastly, this study’s inability to identify strong independent predictors for reintervention in the multivariate analysis may be attributed to the limited sample size and variable selection. Alternatively, it could reflect a genuine lack of dominant risk factors, highlighting the multifactorial nature of IPP complications.
Strengths of our study included the ability to evaluate cohort outcomes with a substantial follow-up period and the opportunity to study and highlight other prognostic factors for IPP survival that have not been previously described in the literature, particularly regarding surgical procedural characteristics. We have looked specifically at risk factors for IPP outcomes, including patients who have been excluded from many studies in the past.
Our study also found that smoking may be a significant risk factor for infection following IPP implantation. This result, although relevant in our cohort, is not widely supported by existing literature. While several studies have explored risk factors for post-operative infections in this context, very few have specifically identified smoking as a major risk factor for infection after penile prosthesis surgery. This finding underscores the need for further investigation in order to confirm these results.
Postoperative complication rates after IPP implantation may be underestimated in the literature, particularly when revision cases are excluded. Our study provides a broader perspective by including both primary and revision procedures and examining real-life surgical variability.
We found an overall complication rate of 24.1%. Smoking and younger age were significantly associated with infection, while prior IPP surgeries were linked to mechanical malfunction. We also observed that cylinder downsizing was associated with earlier reintervention, possibly reflecting greater surgical complexity.
Although our study is retrospective and limited by its sample size, it offers insight into procedural and patient-related factors that are often underreported. The fact that surgeries were performed by multiple surgeons using different devices, whilst a limitation, also mirrors real-world diversity and practice.
Our findings highlight the need for further studies, ideally prospective and multicentric, to better define the key predictors of IPP outcomes. Including patients and surgical scenarios often excluded in other series, it will contribute meaningfully to the evolving understanding of prosthesis survival.
Acknowledgement
Not applicable.
Funding Statement: The authors received no specific funding for this study.
Author Contributions
Skander Zouari: Conceptualization, methodology, data collection, manuscript drafting. Flora Barthe: Data collection, literature review, manuscript drafting, and revision. Arnoult Morrone: Data collection, statistical analysis, and manuscript editing. Lionel Mendel: Data collection, patient follow-up, and manuscript review. Imad Bentellis: Statistical analysis, data interpretation, and manuscript revision. Louis Vignot: Data collection, literature review, and manuscript editing. Romain Haider: Patient follow-up, data interpretation, and manuscript revision.
Younes Ahallal: Supervision, conceptualization, and critical revision of the manuscript. Daniel Chevallier: Supervision, study design, manuscript revision, and approval of the final version. Matthieu Durand: Supervision, conceptualization, methodology, manuscript revision, and final approval. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials
The data that support the findings of this study are available from the Corresponding Author, Skander Zouari, upon reasonable request.
Ethics Approval
Not applicable.
Conflicts of Interest
The authors declare no conflicts of interest to report regarding the present study.
References
1. Levine LA, Becher EF, Bella AJ et al. Penile prosthesis surgery: current recommendations from the international consultation on sexual medicine. J Sex Med 2016 Apr;13(4):489–518. [Google Scholar]
2. Rajpurkar A, Dhabuwala CB. Comparison of satisfaction rates and erectile function in patients treated with sildenafil, intracavernous prostaglandin E1 and penile implant surgery for erectile dysfunction in urology practice. J Urol 2003 Jul;170(1):159–163. [Google Scholar]
3. Govier FE, Gibbons RP, Correa RJ, Pritchett TR, Kramer-Levien D. Mechanical reliability, surgical complications, and patient and partner satisfaction of the modern three-piece inflatable penile prosthesis. Urology 1998 Aug;52(2):282–286. doi:10.1016/s0090-4295(98)00177-0. [Google Scholar] [PubMed] [CrossRef]
4. Carson CC 3rd. Efficacy of antibiotic impregnation of inflatable penile prostheses in decreasing infection in original implants. J Urol 2004 Apr;171(4):1611–1614. doi:10.1097/01.ju.0000118245.66976.e1. Erratum in: J Urol. 2004 Aug;172(2):781. [Google Scholar] [PubMed] [CrossRef]
5. Carvajal A, Benavides J, García-Perdomo HA, Henry GD. Risk factors associated with penile prosthesis infection: systematic review and meta-analysis. Int J Impot Res 2020;32(6):587–597. [Google Scholar] [PubMed]
6. Miller LE, Khera M, Bhattacharyya S, Patel M, Nitschelm K, Burnett AL. Long-term survival rates of inflatable penile prostheses: systematic review and meta-analysis. Urology 2022;166:6–10. [Google Scholar] [PubMed]
7. Carson CC, Mulcahy JJ, Harsch MR. Long-term infection outcomes after original antibiotic impregnated inflatable penile prosthesis implants: up to 7.7 years of followup. J Urol 2011 Feb;185(2):614–618. doi:10.1016/j.juro.2010.09.094. [Google Scholar] [PubMed] [CrossRef]
8. Henry GD, Wilson SK, Delk JR et al. Revision washout decreases penile prosthesis infection in revision surgery: a multicenter study. J Urol 2005 Jan;173(1):89–92. doi:10.1097/01.ju.0000146717.62215.6f. [Google Scholar] [PubMed] [CrossRef]
9. Montague DK. Does antiseptic washout reduce penile prosthesis infection following revision surgery? Nat Clin Pract Urol 2025;2(5):222–223. doi:10.1038/ncpuro0180. [Google Scholar] [CrossRef]
10. Miller JA, Bennett NE. Comparing risk factors for adverse outcomes in virgin inflatable penile prosthesis implantations and revisions: a retrospective cohort study. Sex Med 2020 Sep;8(3):388–395. [Google Scholar]
11. Mirheydar H, Zhou T, Chang DC, Hsieh TC. Reoperation rates for penile prosthetic surgery. J Sex Med 2016 Jan 1;13(1):129–133. [Google Scholar]
12. Bellaiche J, Gonzalez Serrano A, Larre S. Survie et facteurs associes à la durée de vie des prothèses péniennes a long terme. Prog En Urol 2021 May 1;31:374–380 (In French). doi:10.1016/j.purol.2020.09.018. [Google Scholar] [PubMed] [CrossRef]
13. Chierigo F, Capogrosso P, Dehò F et al. Long-term follow-up after penile prosthesis implantation-survival and quality of life outcomes. J Sex Med 2019 Nov;16(11):1827–1833. [Google Scholar]
14. Gon LM, De Campos CCC, Voris BRI, Passeri LA, Fregonesi A, Riccetto CLZ. A systematic review of penile prosthesis infection and meta-analysis of diabetes mellitus role. BMC Urol 2021 Mar 10;21(1):35. [Google Scholar]
15. Matthew-Onabanjo AN, Matthew AN, Famati E, Nguyen V, Rogers MJ. Perioperative infection prevention during inflatable penile prosthesis surgery: a narrative review. Transl Androl Urol 2024 Aug 31;13(8):1628–1640. [Google Scholar]
16. Mandava SH, Serefoglu EC, Freier MT, Wilson SK, Hellstrom WJG. Infection retardant coated inflatable penile prostheses decrease the incidence of infection: a systematic review and meta-analysis. J Urol 2012 Nov 1;188(5):1855–1860. [Google Scholar]
17. Smelser AM, VanDyke ME, Nealon SW et al. Mechanical indications for inflatable penile prosthesis revision: analysis and implications for revision surgery. J Sex Med 2023 Jun 28;20(7):1044–1051. [Google Scholar]
18. Parikh KA, Pathak RA, Wilson RRA, Patel RJ, Broderick GA. Revision surgery for inflatable penile prosthesis (IPPa single-center experience and pictorial representation. Urology 2021 Jun;152(Suppl 5):42–51. doi:10.1016/j.urology.2020.11.072. [Google Scholar] [PubMed] [CrossRef]
19. Chan EP, Punjani N, Campbell JD, Abed H, Brock G. Indications for penile prosthesis revision: lessons learned to limit technical causes of reoperation. J Sex Med 2019 Sep;16(9):1444–1450. [Google Scholar]
20. Wilson SK, Mora-Estaves C, Egydio P et al. Glans necrosis following penile prosthesis implantation: prevention and treatment suggestions. Urology 2017 Sep;107:144–148. [Google Scholar]
21. Sørensen LT. Wound healing and infection in surgery. The clinical impact of smoking and smoking cessation: a systematic review and meta-analysis. Arch Surg 2012 Apr;147(4):373–383. [Google Scholar]
22. Cakan M, Demirel F, Karabacak O, Yalçinkaya F, Altuğ U. Risk factors for penile prosthetic infection. Int Urol Nephrol 2003;35(2):209–213. [Google Scholar] [PubMed]
23. Lacy JM, Walker J, Gupta S, Davenport DL, Preston DM. Risk factors for removal or revision of penile prostheses in the veteran population. Urology 2016 Dec;98:189–194. doi:10.1016/j.urology.2016.06.039. [Google Scholar] [PubMed] [CrossRef]
24. Menshchikov K, Menshchikov M, Yurasov D, Artamonov A. Risk factors for penile prosthesis infection: an umbrella review and meta-analysis. Arab J Urol 2023;22(2):96–101. [Google Scholar] [PubMed]
25. Adamsky MA, Boysen WR, Cohen AJ et al. Evaluating the role of postoperative oral antibiotic administration in artificial urinary sphincter and inflatable penile prosthesis explantation: a nationwide analysis. Urology 2018 Jan;111:92–98. [Google Scholar]
26. Lipsky MJ, Onyeji I, Golan R et al. Diabetes is a risk factor for inflatable penile prosthesis infection: analysis of a large statewide database. Sex Med 2019 Mar;7(1):35–40. [Google Scholar]
27. Gross MS, Reinstatler L, Henry GD et al. Multicenter investigation of fungal infections of inflatable penile prostheses. J Sex Med 2019 Jul;16(7):1100–1105. [Google Scholar]
28. Henry GD, Donatucci CF, Conners W et al. An outcomes analysis of over 200 revision surgeries for penile prosthesis implantation: a multicenter study. J Sex Med 2012 Jan;9(1):309–315. [Google Scholar]
29. Dave CN, Khalaf A, Patel HD, Kohn TP, Burnett AL. Neurogenic bladder is an independent risk factor for complications associated with inflatable penile prosthesis implantation. Int J Impot Res 2020 Sep;32(5):520–524. [Google Scholar]
30. Yiou R, Binhas M. Combined implantation of a penile prosthesis and adjustable continence therapy ProACT in patients with erectile dysfunction and urinary incontinence after radical prostatectomy: results of a prospective pilot study. J Sex Med 2015 Dec;12(12):2481–2484. [Google Scholar]
31. Lane BR, Abouassaly R, Angermeier KW, Montague DK. Three-piece inflatable penile prostheses can be safely implanted after radical prostatectomy through a transverse scrotal incision. Urology 2007 Sep;70(3):539–542. [Google Scholar]
32. Lotan Y, Roehrborn CG, McConnell JD, Hendin BN. Factors influencing the outcomes of penile prosthesis surgery at a teaching institution. Urology 2003 Nov 1;62(5):918–921. [Google Scholar]
33. Wilson SK, Zumbe J, Henry GD, Salem EA, Delk JR, Cleves MA. Infection reduction using antibiotic-coated inflatable penile prosthesis. Urology 2007 Aug 1;70(2):337–340. [Google Scholar]
34. Elmussareh M, Goddard JC, Summerton DJ, Terry TR. Minimising the risk of device infection in penile prosthetic surgery: a UK perspective. J Clin Urol 2013;6(5):280–288. doi:10.1177/2051415813488367. [Google Scholar] [CrossRef]
35. Henry GD, Kansal NS, Callaway M et al. Centers of excellence concept and penile prostheses: an outcome analysis. J Urol 2009 Mar;181(3):1264–1268. [Google Scholar]
36. Vanderstappen CL, Van Renterghem K. Distal corporotomy localisation is a risk factor for distal cylinder extrusion and rerouting surgery in penile prosthesis implantation. J Sex Med 2022 Nov 1;19(11 Supplement 4):S115–S116. [Google Scholar]
37. Baird BA, Parikh K, Broderick G. Penile implant infection factors: a contemporary narrative review of literature. Transl Androl Urol 2021 Oct;10(10):3873884. [Google Scholar]
38. Madbouly K, AlHajeri D, Habous M, Binsaleh S. Association of the modified frailty index with adverse outcomes after penile prosthesis implantation. Aging Male 2017 Jun;20(2):119–124. [Google Scholar]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools