 Open Access
Open Access
ARTICLE
Endocrine stimulation in men with non-obstructive azoospermia and low serum testosterone prior to micro-TESE: hormonal response as a predictor of sperm retrieval
1 Obstetrics and Gynecology, Assuta Ashdod University Hospital, Faculty of Health Sciences, Ben-Gurion University of the Negev, Ashdod, 7747629, Israel
2 Obstetrics and Gynecology, Shaare Zedek Medical Center, The Hebrew University Hadassah Medical School, Jerusalem, 9103102, Israel
3 Urology Department, Tel Aviv Sourasky Medical Center, The Gray School of Medical Sciences, Tel Aviv University, Tel Aviv, 6423906, Israel
* Corresponding Author: Snir Dekalo. Email:
Canadian Journal of Urology 2026, 33(1), 135-142. https://doi.org/10.32604/cju.2025.066936
Received 21 April 2025; Accepted 27 October 2025; Issue published 28 February 2026
Abstract
Background: Hormonal treatment and response as a predictor of sperm retrieval prior to microdissection testicular sperm extraction (micro-TESE) are not well established in the current literature. This study aimed to investigate the hormonal response as a predictor of sperm retrieval among men with nonobstructive azoospermia (NOA). Methods: Seventy-seven consecutive patients who had testosterone levels ≤ 14 nmol/L were treated medically with an aromatase inhibitor or recombinant human chorionic gonadotropin (rec-hCG) prior to micro-TESE and were included. Thirty-four (44.2%) had unexplained NOA (UNEX), 25 (32.5%) had Klinefelter syndrome (KS), 8 (10.4%) had a history of cryptorchidism (UDT), 4 (5.2%) had microdeletion of the Azoospermia factor C (AZFc), and 6 (7.8%) were treated previously with chemotherapy. Baseline and post-treatment serum hormonal levels were documented. Pre-op testosterone levels were entered into binary logistic regressions with age, Follicle-stimulating hormone (FSH), and Luteinizing hormone (LH) levels to test for significance with sperm retrieval. We then built logistic regression models to identify predictors of successful surgical sperm retrieval (SSR). Results: Forty-five patients (58%) had successful retrieval. In 32 patients (42%), no sperm was retrieved. Both the mean pre-op testosterone and the mean testosterone change between the two groups were significant (p = 0.02 and p = 0.011, respectively). Receiver operating characteristic (ROC) analysis demonstrated an area under the curve (AUC) of 0.785 (95%CI = 0.685–0.886, p < 0.001). The Youden index coefficient was calculated for KS and UNEX. The cut-off point for KS was established at 0.764 (sensitivity = 0.875, false positive rate [FPR] = 0.111), and 0.215 for UNEX (sensitivity = 0.438, FPR = 0.222). We also observed a correlation between age and SSR (p = 0.05). In KS patients, SSR was determined by pre-op testosterone levels irrespective of age. Conclusion: Pre-operative hormonal response is a predictor for SSR in NOA patients who were treated medically. This data may help during pre-operative counselling.Keywords
Azoospermia is defined as the total absence of spermatozoa in the ejaculate, and it occurs in 10%–15% of men who undergo fertility investigation.1 This needs to be confirmed in repeated tests with vigorous centrifugation of the semen and careful examination of the pellet.2 Non-obstructive azoospermia (NOA), which is associated with spermatogenic failure, is a challenging form of azoospermia that is managed most commonly by surgical sperm retrieval followed by intracytoplasmic sperm injection (ICSI). Many of these patients present with a hormone profile of high Follicle-stimulating hormone (FSH), normal or high Luteinizing hormone (LH) levels, and normal to low testosterone levels.3 Intra-testicular testosterone (ITT) is required for several critical steps associated with spermiogenesis, which include meiosis completion, adhesion of elongated spermatids to Sertoli cells, sperm release, and maintenance of the blood-testis barrier.4,5 Additionally, high ITT is also required for one of the earliest steps of spermatogenesis—the transition from type A to type B spermatogonia.6,7 In view of the pivotal role of testosterone in the regulation of spermatogenesis and spermiogenesis, elevation of ITT may contribute to the optimization of these processes. Hence, NOA patients who present with low circulating testosterone may benefit from hormonal optimization prior to their surgery.8,9
Various sperm retrieval techniques have been used in NOA patients, with significant variability in success rates associated with underlying diagnosis, surgical technique, and several other clinical and laboratory parameters.10–15 The understandable wish of NOA patients who pursue fertility treatment to understand their chances of successful sperm retrieval dictates thorough preoperative counselling. During such counselling sessions, patients should be presented with as much solid evidence to allow them to make an informed decision.
Several clinical variables and biomarkers are predictive tools to estimate the likelihood of sperm retrieval in men with NOA.16–20 In this observational retrospective study, we investigated the surgical outcome in NOA patients who were treated medically prior to their microdissection testicular sperm extraction (micro-TESE), and we assessed the value of their hormonal response as a predictor of SSR.
The records of 117 consecutive NOA patients who underwent micro-TESE by a single surgeon (S.B.) between December 2019 and March 2022 were reviewed retrospectively, and patients who were treated hormonally prior to their operation were identified. Azoospermia was confirmed by two semen analyses according to WHO criteria2 and two extended sperm searches, as described by Ron-El et al.21 Diagnosis of NOA was based on the presence of two or more of the following criteria: histopathology that confirmed no spermatogenesis or maturation arrest, the combination of an elevated FSH level greater than 7.6 IU/L and smaller testicular volumes with a long axis of 4.6 cm or less, absence of sperm on initial sperm search and genetic abnormality or chromosomal abnormality known to be associated with NOA (e.g., Klinefelter syndrome [KS]; Yq microdeletions).22 Patients with proven obstructive azoospermia (i.e., histopathology that confirmed normal spermatogenesis, presence of unilateral or bilateral absence of the vas deferens, presence of an abundant number of sperm on initial sperm search) and men with complete microdeletions on azoospermia factor region (AZF) A or B were excluded from this study.
The study was approved by the local Institutional Review Board of the Assuta Medical Center (No. 0014-22-ASMC). Due to its retrospective nature, informed consent was waived.
Patients’ ages were recorded. Preoperative evaluation included a complete history, physical examination, and routine scrotal ultrasound. Hormonal evaluation included serum testosterone, estradiol, LH, and FSH hormonal analyses.
Blood samples were collected between 7:00 and 10:00 a.m. after 8–10 h of overnight fasting in serum tubes for the estimation of total testosterone, estradiol, FSH, and LH. The same laboratory and kits were used for all samples. Testosterone, estradiol, FSH, and LH levels were analyzed and quantified by a Siemens ADVIA Centaur® XP (Siemens Healthineers, Forchheim, Germany). The laboratory reference ranges were 6.5–24 nmol/L for total testosterone, <223 pmol/L for estradiol, 1–9 IU/L for LH, and 1.4–18.1 IU/L for FSH. All azoospermic men with testosterone levels ≤14 nmol/L23 were included in the study.
Patients with serum testosterone of <14 nmol/L were treated with aromatase inhibitors (1 mg anastrozole daily) for >3 months prior to their surgery. Patients were given oral aromatase inhibitors as first-line treatment regardless of their body mass index (BMI), testosterone/estradiol (T/E) ratio, or estradiol levels. Those who had less than a 15% increase in their testosterone levels after 6 weeks of aromatase inhibitor therapy were converted to recombinant human chorionic gonadotropin (rec-hCG).
Treatment with rec-hCG was initiated at a dose of 2500 IU twice weekly, after 1 month, patients who didn’t respond with elevated testosterone levels to normal limits were given a dose of 5000 IU three times weekly. Hormonal treatment was continued until the time of micro-TESE. Median overall treatment time was 5 months (interquartile range [IQR]: 4–7).
Micro-TESE procedures were all performed by a single surgeon (S.B.) with over 20 years of experience. The procedure was done as described by Schlegel et al.24
Tissue processing and initial and extended sperm search
Seminiferous tubules obtained during micro-TESE were first dissected mechanically under a stereomicroscope with the use of bent 18-gauge needles. The initial search for spermatozoa was performed at 200× magnification with the use of an inverted microscope (Olympus IX81, Olympus Group, Tokyo, Japan).
For an extended sperm search, dissected tubules were dissected further using bent 18-gauge needles for another 20 min and then centrifuged at 1000× g for 15 min. Most of the supernatant was removed, and about 0.5 mL was left above the pellet. The pellet was then resuspended back into the supernatant. Drops of 10–20 μL of the suspension were put in a FluoroDish (FD5040, World Precision Instruments Inc., Sarasota, FL, USA) and covered with light mineral oil (Oil for Embryo Culture; Irvine Scientific, Santa Ana, CA, USA). The extended sperm search was performed with an inverted microscope (Olympus IX81, Olympus Group, Tokyo, Japan) equipped with Nomarski optics, a Uplan Apo X100 or X40 objective lens, and a 0.55 NA condenser lens. Motile or immotile spermatozoa were collected with the use of an injection pipette and Eppendorf TransferMan Nk2 micromanipulator (Eppendorf, Hamburg, Germany). The collected spermatozoa were placed in drops prepared in the same dish made of MHM-C medium (Multipurpose Handling Medium Complete; Irvine Scientific) supplemented with 30% Human Serum Albumin (Irvine Scientific) for further injection into oocytes or cryopreservation.
All analyses were performed with SPSS 29 (IBM Corp., Armonk, NY, USA). Descriptive statistics were derived for all variables; normal distribution was tested, and means and standard deviations (SDs) were calculated for hormones and testosterone measures, and paired t-tests were performed between testosterone measures. Following these tests, the pre-op testosterone measure was entered into binary logistic regressions with age, FSH, and LH hormone levels to test for significance with SSR. The predicted sperm retrieval probability was saved for further analysis and entered into a one-way ANOVA to establish the effect of pre-op testosterone level on sperm retrieval for each diagnosis and pre-op treatment. A Least Significant Difference (LSD) post hoc analysis was performed to highlight differences between the pre- and post-treatment groups. The saved predicted sperm retrieval predictor was further processed in a Receiver Operating Characteristic (ROC) analysis to derive Area Under the Curve (AUC) and Youden Index estimates. Age and diagnosis effects on sperm retrieval rates were then derived by sensitivity/specificity analysis.
Seventy-seven patients with NOA who were medically treated prior to their micro-TESE were included in this study (Table 1). The mean age of subjects was 31.4 (range: 20–47). Thirty-four patients (44.2%) presented with unexplained (idiopathic) NOA (UNEX), 25 (32.5%) had KS, eight (10.4%) had a history of cryptorchidism (UDT), four (5.2%) had microdeletion of the AZFc (Yq del), and six (7.8%) were treated previously with chemotherapy (Post Chemo). Sperm was retrieved successfully in 45 patients (58.4%) (SR+) and failed in 32 patients (42%) (SR−). Successful retrieval varied between 47.1% in UNEX cases to 87.5% in UDT ones. Initial, pre-op, and changes in testosterone were analyzed by sperm retrieval (Table 2).
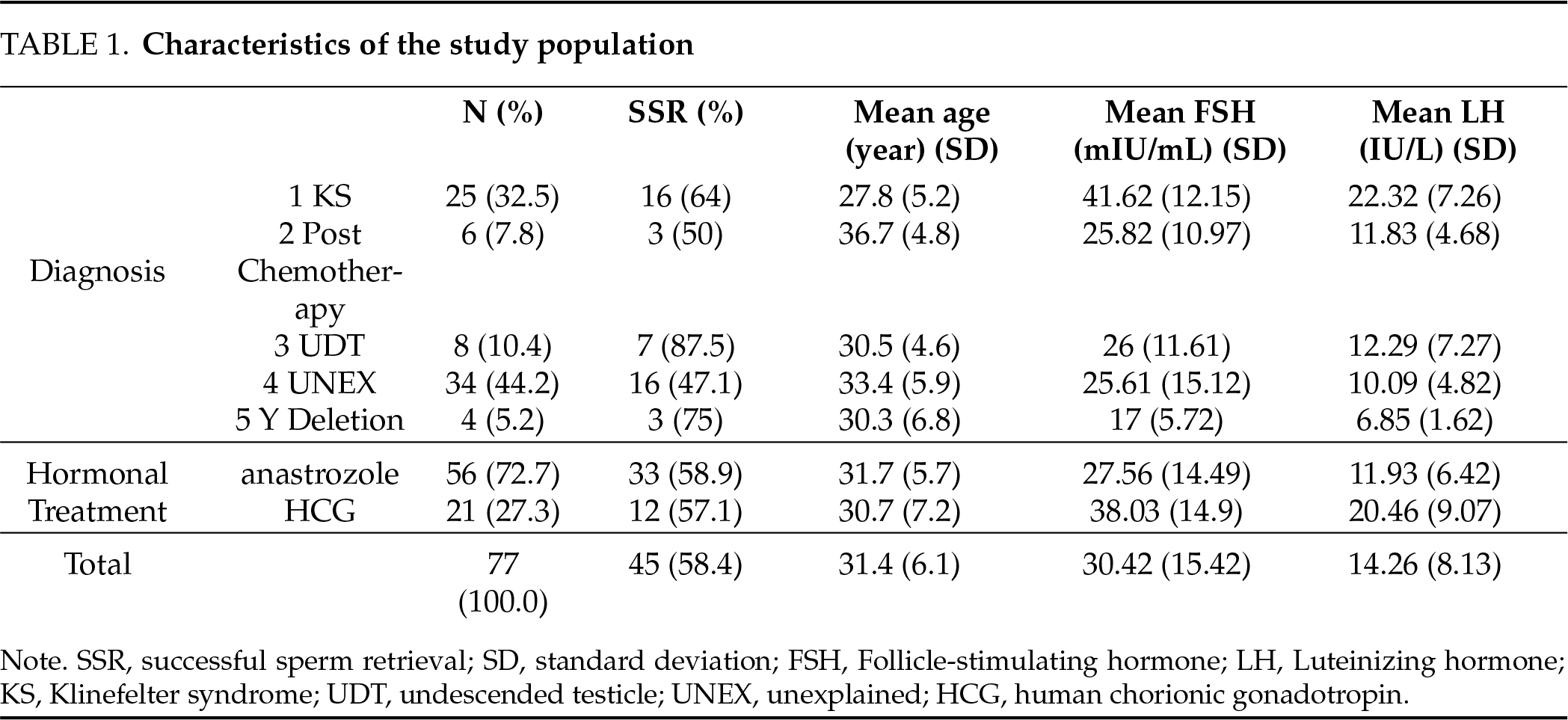
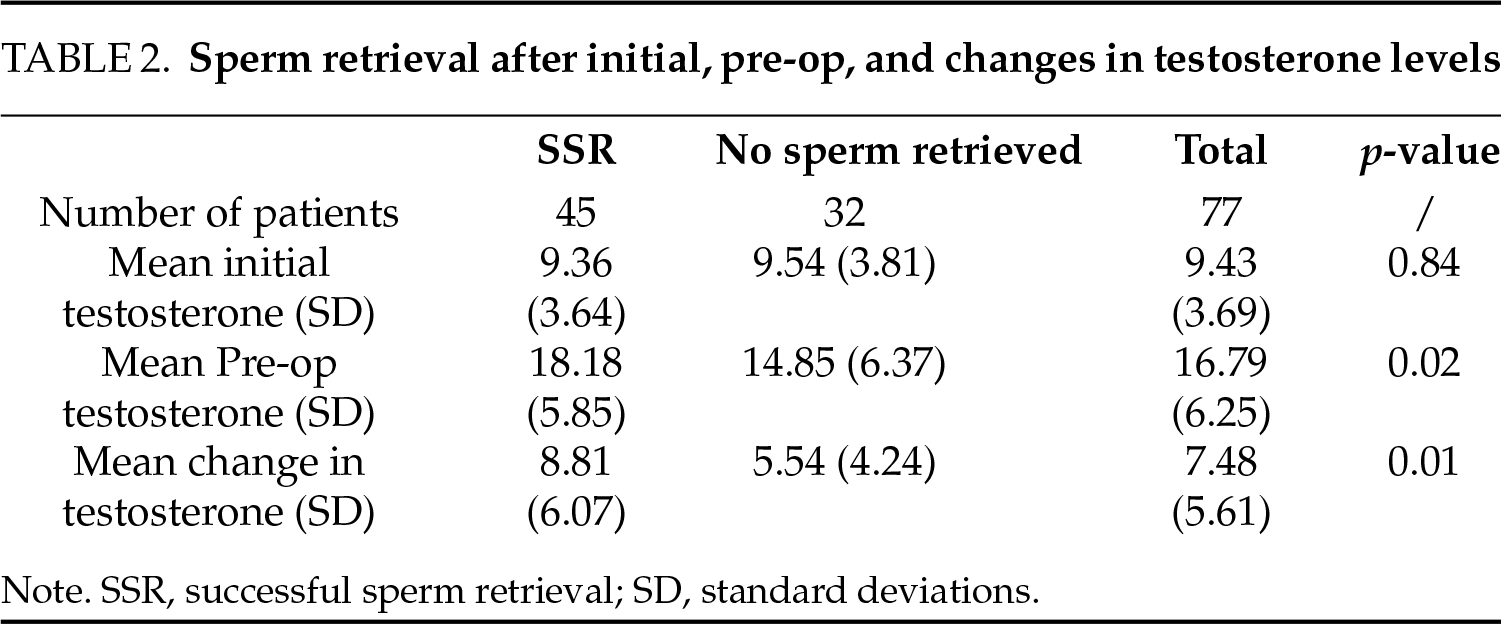
Although initial testosterone differences between the SR+ and SR− groups were small and not significant, both the mean pre-op testosterone and the mean change in testosterone levels between the two groups were significant (p = 0.02 and p = 0.011, respectively). Because pre-op testosterone accounted for most of the change (R2 = 0.656, p < 0.001), it was adopted for further analysis and entered with age, FSH, and LH as independent variables into a binary logistic regression to test sperm retrieval. The saved predicted pre-op sperm retrieval was submitted to a ROC analysis (Figure 1). AUC of 0.785 (95%CI = 0.685–0.886, p < 0.001) was obtained, and a Youden Index coefficient was estimated at a 0.524 cut-off point that conformed to a sensitivity of 0.711 and a false positive rate (FPR) of 0.188.
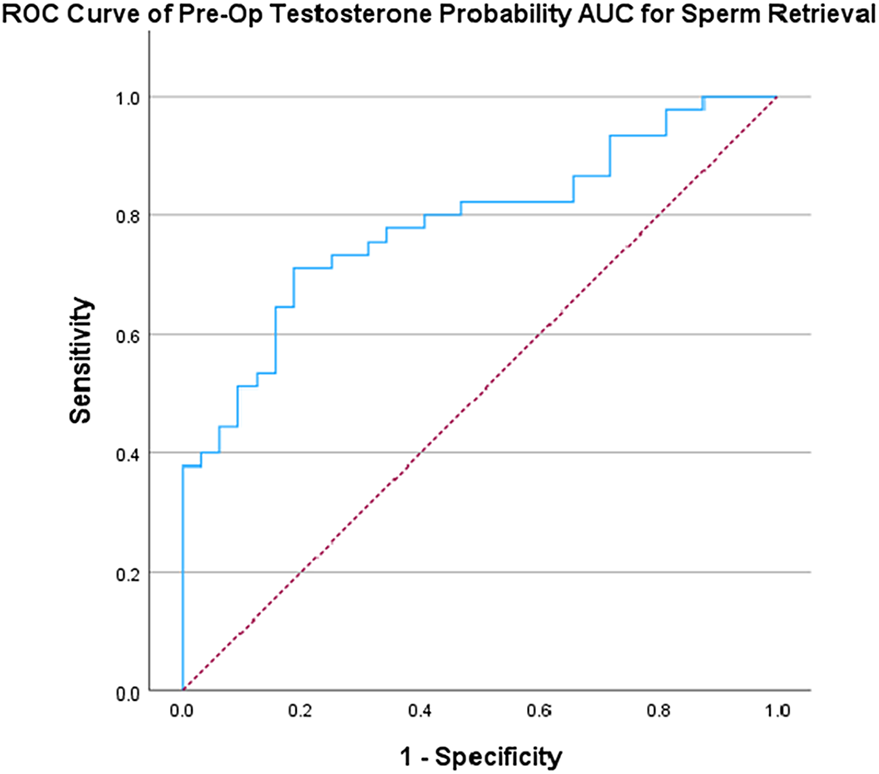
FIGURE 1. Receiver operating characteristic (ROC) analysis of sperm retrieval by pre-op testosterone level
We then calculated the Youden Index Coefficient for the two largest diagnostic categories, KS and UNEX, which together comprised 77% of all cases. The cut-off point for KS was 0.764 (sensitivity = 0.875, FPR = 0.111) and 0.215 for UNEX (sensitivity = 0.438, FPR = 0.222).
To address potential effects of age on sperm retrieval success, the pre-op testosterone predictor was plotted against age and regressed by the observed sperm retrieval (Figure 2). We found a weak, but significant, correlation between age and predicted successful sperm retrieval (R2 = 0.109, p = 0.05).
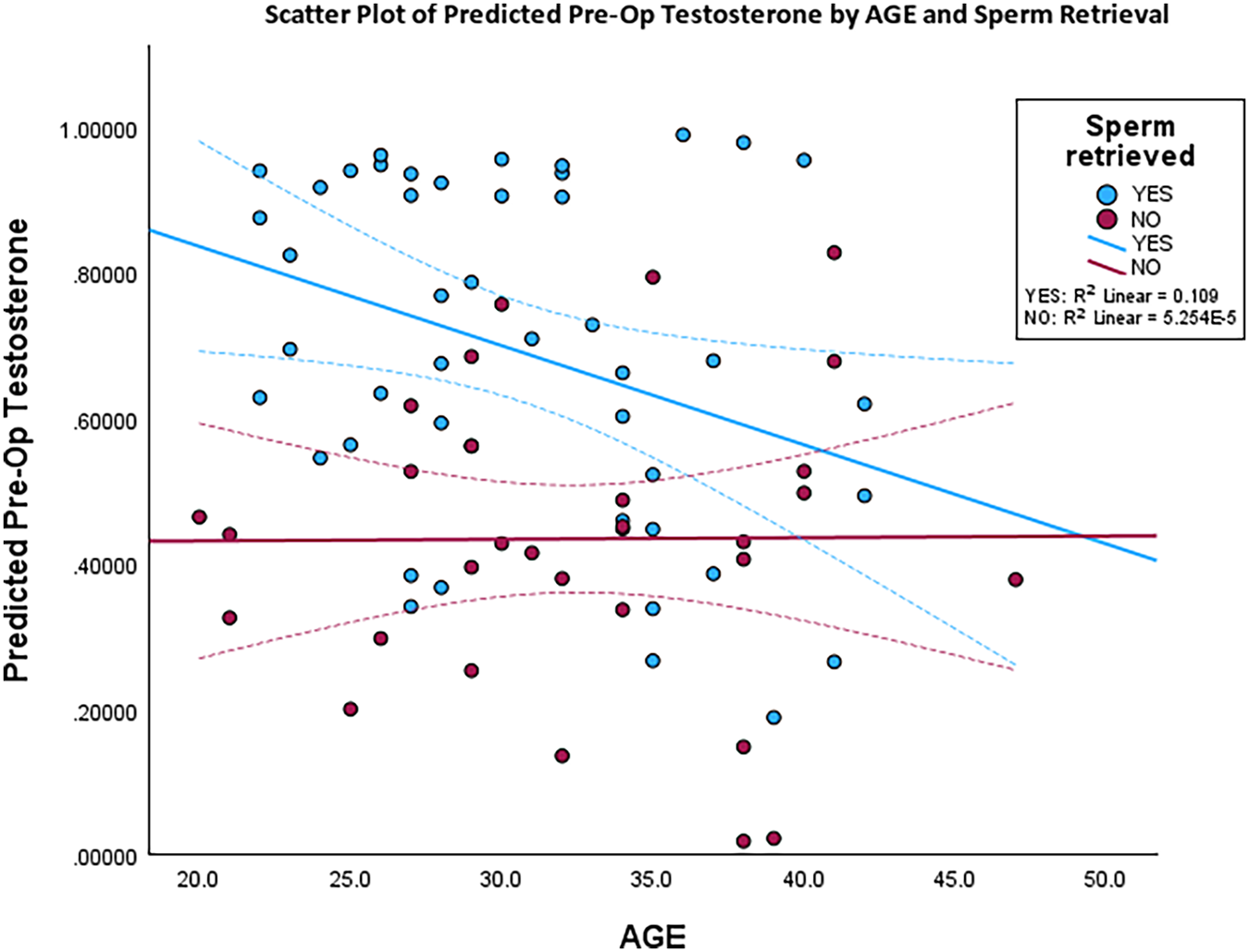
FIGURE 2. Scatter plot of all cases with pre-op testosterone and age, with regression lines and coefficients for sperm retrieval success and failure
To investigate further the interactions among age, testosterone, and sperm retrieval, patients were coded into “young” (20–30) and “old” (30+), and sperm retrieval results for KS and UNEX groups were plotted and compared to reveal different treatment response dynamics (Figure 3). In both groups, younger patients responded better to the testosterone-raising treatment. Among KS patients, sperm retrieval success was determined predominantly by pre-op testosterone levels, irrespective of age, but in UNEX patients, success, in both young and old, was largely independent of the pre-op testosterone level.
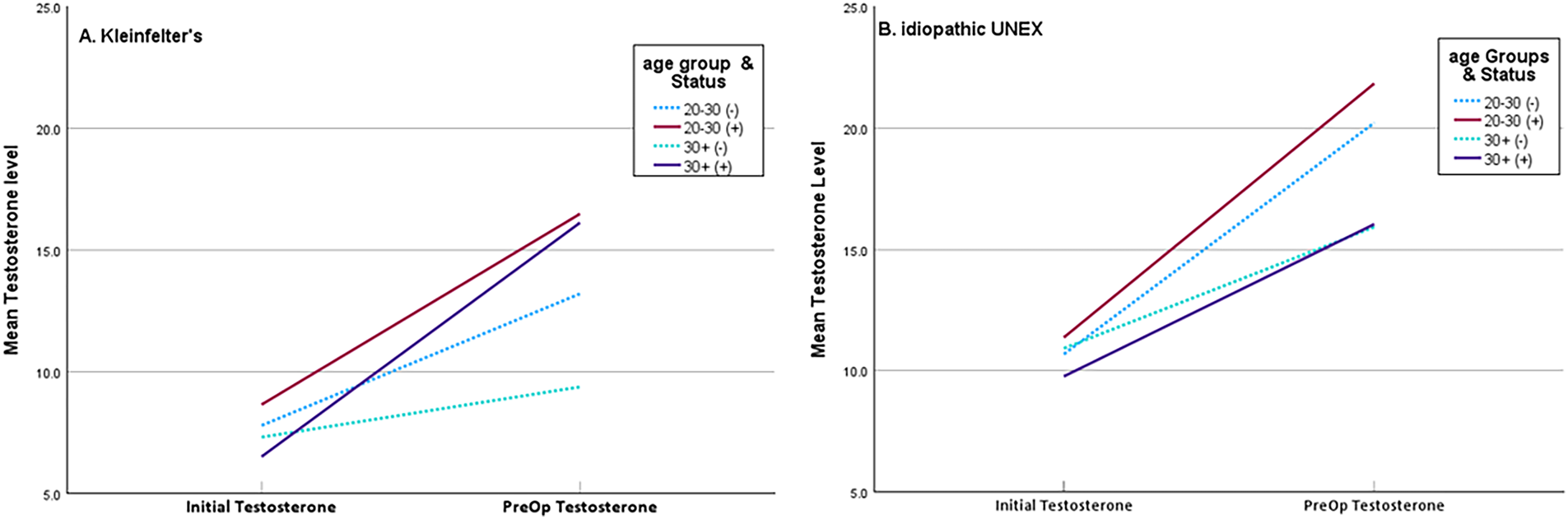
FIGURE 3. Comparison between (A) men with Klinefelter’s and (B) men with unexplained nonobstructive azoospermia (NOA) (idiopathic UNEX) by age, mean initial testosterone, pre-op testosterone, and sperm retrieval. Solid lines denote sperm retrieval success, and dotted lines represent failure
This study investigated the surgical outcome in NOA patients who were treated medically prior to their micro-TESE, and we assessed the value of their hormonal response as a predictor of sperm retrieval. Proper counselling of men undergoing micro-TESE is essential. Patients should be presented with solid evidence, so decisions can be made, and the psychological burden managed. Patients with a poor prognosis might decide to refrain from surgical treatment and consider other options, such as donor sperm or adoption.
Micro-TESE, which is an effective and safe surgical technique, is currently considered a first-line approach for men with NOA undergoing sperm retrieval.25,26 In a meta-analysis comparing sperm retrieval rates from micro-TESE with traditional surgical techniques, micro-TESE was 50% more likely to retrieve sperm successfully than conventional testicular sperm extraction (cTESE).27 Elevated ITT levels are essential for the final stage of spermatogenesis, specifically, for differentiation of spermatids into fully developed spermatozoa. Exogenous testosterone administration in humans demonstrated that decreased ITT levels were correlated with inhibition of spermiogenesis.28,29 NOA patients who presented with hypogonadism may benefit from hormonal stimulation prior to their SSR to maintain sufficient ITT levels, thereby improving the likelihood of effective sperm retrieval.8,30,31
Although there are no meta-analyses or large-scale studies that investigated the use of hCG therapy or aromatase inhibitor therapy in men with hypergonadotropic hypogonadism and NOA, our practice, as reflected in the current report, suggested that such hormonal stimulation therapies prior to micro-TESE may improve sperm retrieval rates.
Our analysis consisted of patients with various NOA causes. The prevalence of the different etiologies can be found in the existing literature.32,33 The current study reports an overall SSR of 58.4%. These findings are consistent with a recent meta-analysis, which recorded an overall SSR success of 47% among NOA subjects.34
In the overall study group, our analysis demonstrated that a higher hormonal response was predictive of successful SR in NOA patients who were treated medically prior to their micro-TESE.35–37 To the best of our knowledge, this is the first report that has investigated the predictive value of post-medical stimulation, pre-operative testosterone levels.
Pre-op testosterone levels had good sensitivity and specificity for predicting sperm retrieval in our medically treated NOA group in total (sensitivity = 71.1%, specificity = 81.2%, and AUC = 0.785). Sensitivity and specificity of pre-op testosterone levels were even better for the KS patient group (sensitivity = 87.5%, specificity = 88.9%).
Although we were unable to fully analyze the underlying determinants of sperm retrieval in the UDT, Yq Deletions, and post-chemo patients due to the limited number of cases, we compared and analyzed the differences between KS and UNEX patients—the two major groups. In both groups, younger age predicted a better response to testosterone-raising treatment. However, in KS patients, sperm retrieval success was determined predominantly by pre-operative testosterone levels, regardless of age. Conversely, in UNEX patients, success was largely independent of pre-operative testosterone levels for both “young” and “old” individuals.
We hypothesize that the higher preoperative testosterone levels among successful KS NOA patients reflected more intact seminiferous tubules, which contained functionally normal Leydig cells with a preserved capacity to secrete testosterone.
The current UNEX data suggest that sperm retrieval success may also be determined by other variables that have not yet been identified. The heterogeneous nature of the idiopathic NOA group makes the identification of clear determinants of sperm retrieval in this group extremely challenging.
We believe that NOA patients may benefit from an individualized pre-op counselling approach. We suggest three supporting pillars for such counselling:
1. The likelihood of a successful SSR based on current relevant literature;
2. The likelihood of a successful SSR based on the specific experience of the clinic/surgeons who are conducting the treatment (local up-to-date database);
3. Individual hormonal response (change in testosterone levels) when pre-op medical treatment is indicated and implemented. This can be used as a stand-alone predictive parameter or as an addition to predictive models developed previously.
The limitations of this study were related primarily to its relatively small sample size. Given its retrospective design, it was not immune to selection bias. Although we didn’t notice a major difference between patients with or without preoperative therapy, scoring methods such as the Johnsen’s score were not available while interpreting the data due to the retrospective nature. Additionally, the patient cohort featured homogeneous ethnicity, which suggested that our findings might not extend to NOA patients across diverse ethnic backgrounds. Consequently, we advocate for further research that involves larger cohorts to ascertain definitively the predictors of successful sperm retrieval in NOA patients. Lastly, we did not measured ITT intraoperatively. Despite these limitations, this study stands as the first report that assesses the predictive significance of post-medical stimulation, pre-operative testosterone levels for successful sperm retrieval, which marks an important contribution to the field.
This study demonstrated that a favorable hormonal response was predictive of successful SR in medically-treated NOA patients, and it distinguished between patients with a good or a poor prognosis. SR success was associated with the change between pre- and post-treatment testosterone levels. We propose using this predictive parameter as part of individualized counselling and to consider its implementation in predictive models that were developed previously.38–40
Acknowledgement
Not applicable.
Funding Statement
Not applicable.
Author Contributions
Shlomi Barak designed the study, conducted the data analysis and drafted and revised the manuscript. Netanel Waldenberg, Aharon Peretz and Reut Bartoov assisted with the data analysis and revised the manuscript. Guy Bar designed the study and revised the manuscript. Snir Dekalo assisted with the data analysis and revised the manuscript. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials
Data will be made available when contacting the corresponding author.
Ethics Approval
The study was approved by the local Institutional Review Board of the Assuta Medical Center (No. 0014-22-ASMC). Due to its retrospective nature, informed consent was waived.
Conflicts of Interest
The authors declare no conflicts of interest to report regarding the present study.
References
1. Hamoda T, Shah R, Mostafa T et al. Global andrology forum (GAF) clinical guidelines on the management of non-obstructive azoospermia: bridging the gap between controversy and consensus. World J Mens Health 2025;43:e26. doi:10.5534/wjmh.250037. [Google Scholar] [PubMed] [CrossRef]
2. Björndahl L, Kirkman Brown J. The sixth edition of the WHO Laboratory Manual for the Examination and Processing of Human Semen: ensuring quality and standardization in basic examination of human ejaculates. Fertil Steril 2022;117(2):246–251. doi:10.1016/j.fertnstert.2021.12.012. [Google Scholar] [PubMed] [CrossRef]
3. Tharakan T, Salonia A, Corona G, Dhillo W, Minhas S, Jayasena C. The role of hormone stimulation in men with nonobstructive azoospermia undergoing surgical sperm retrieval. J Clin Endocrinol Metab 2020;105(12):dgaa556. doi:10.1210/clinem/dgaa556. [Google Scholar] [PubMed] [CrossRef]
4. Esteves SC, Viana MC, Achermann APP, Santi D. Human chorionic gonadotropin-based clinical treatments for infertile men with non-obstructive azoospermia. Andrology 2025:1–35. doi:10.1111/andr.70003. [Google Scholar] [PubMed] [CrossRef]
5. Smith LB, Walker WH. The regulation of spermatogenesis by androgens. Semin Cell Dev Biol 2014;30:2–13. doi:10.1016/j.semcdb.2014.02.012. [Google Scholar] [PubMed] [CrossRef]
6. McLachlan RI. Identification of specific sites of hormonal regulation in spermatogenesis in rats, monkeys, and man. Recent Prog Horm Res 2002;57(1):149–179. doi:10.1210/rp.57.1.149. [Google Scholar] [PubMed] [CrossRef]
7. Shiraishi K, Matsuyama H. Gonadotoropin actions on spermatogenesis and hormonal therapies for spermatogenic disorders. Endocr J 2017;64(2):123–131. doi:10.1507/endocrj.EJ17-0001. [Google Scholar] [PubMed] [CrossRef]
8. Hussein A, Ozgok Y, Ross L, Rao P, Niederberger C. Optimization of spermatogenesis-regulating hormones in patients with non-obstructive azoospermia and its impact on sperm retrieval: a multicentre study. BJU Int 2013;111(3 Pt B):E110–E114. doi:10.1111/j.1464-410X.2012.11485.x. [Google Scholar] [PubMed] [CrossRef]
9. Reifsnyder JE, Ramasamy R, Husseini J, Schlegel PN. Role of optimizing testosterone before microdissection testicular sperm extraction in men with nonobstructive azoospermia. J Urol 2012;188(2):532–536. doi:10.1016/j.juro.2012.04.002. [Google Scholar] [PubMed] [CrossRef]
10. Tsujimura A, Matsumiya K, Miyagawa Y et al. Prediction of successful outcome of microdissection testicular sperm extraction in men with idiopathic nonobstructive azoospermia. J Urol 2004;172(5 Pt 1):1944–1947. doi:10.1097/01.ju.0000142885.20116.60. [Google Scholar] [PubMed] [CrossRef]
11. Kavoussi PK, Atmoko W, Pinggera GM. Technologies to improve sperm retrieval in men undergoing micro-TESE for NOA. Asian J Androl 2025;27(3):375–382. doi:10.4103/aja202450. [Google Scholar] [PubMed] [CrossRef]
12. Sharma A, Minhas S, Dhillo WS, Jayasena CN. Male infertility due to testicular disorders. J Clin Endocrinol Metab 2021;106(2):e442–e459. doi:10.1210/clinem/dgaa781. [Google Scholar] [PubMed] [CrossRef]
13. Deruyver Y, Vanderschueren D, Van der Aa F. Outcome of microdissection TESE compared with conventional TESE in non-obstructive azoospermia: a systematic review. Andrology 2014;2(1):20–24. doi:10.1111/j.2047-2927.2013.00148.x. [Google Scholar] [PubMed] [CrossRef]
14. Kandil H. Integrating artificial intelligence in surgical sperm retrieval techniques: a narrative review. UroPrecision 2025;3(1):17–26. doi:10.1002/uro2.100. [Google Scholar] [CrossRef]
15. Schwarzer JU, Steinfatt H, Schleyer M et al. Microdissection TESE is superior to conventional TESE in patients with nonobstructive azoospermia caused by Y chromosome microdeletions. Andrologia 2016;48(4):402–405. doi:10.1111/and.12460. [Google Scholar] [PubMed] [CrossRef]
16. Zarezadeh R, Fattahi A, Nikanfar S et al. Hormonal markers as noninvasive predictors of sperm retrieval in non-obstructive azoospermia. J Assist Reprod Genet 2021;38(8):2049–5059. doi:10.1007/s10815-021-02176-3. [Google Scholar] [PubMed] [CrossRef]
17. Arshad MA, Majzoub A, Esteves SC. Predictors of surgical sperm retrieval in non-obstructive azoospermia: summary of current literature. Int Urol Nephrol 2020;52(11):2015–2038. doi:10.1007/s11255-020-02529-4. [Google Scholar] [PubMed] [CrossRef]
18. Ishikawa T, Yamaguchi K, Takaya Y, Nishiyama R, Kitaya K, Matsubayashi H. Predictors for sperm retrieval in microdissection sperm extraction for non-obstructive azoospermia. Fertil Steril 2015;104(3):e294. doi:10.1016/j.fertnstert.2015.07.920. [Google Scholar] [CrossRef]
19. Çayan S, Orhan İ, Altay B et al. Fertility outcomes and predictors for successful sperm retrieval and pregnancy in 327 azoospermic men with a history of cryptorchidism who underwent microdissection testicular sperm extraction. Andrology 2021;9(1):253–259. doi:10.1111/andr.12910. [Google Scholar] [PubMed] [CrossRef]
20. Qi L, Liu YP, Zhang NN, Su YC. Predictors of testicular sperm retrieval in patients with non-obstructive azoospermia: a review. J Int Med Res 2021;49(4):03000605211002703. doi:10.1177/03000605211002703. [Google Scholar] [PubMed] [CrossRef]
21. Ron-El R, Strassburger D, Friedler S et al. Extended sperm preparation: an alternative to testicular sperm extraction in non-obstructive azoospermia. Hum Reprod 1997;12(6):1222–1226. doi:10.1093/humrep/12.6.1222. [Google Scholar] [PubMed] [CrossRef]
22. Schoor RA, Elhanbly S, Niederberger CS, Lawrences R. The role of testicular biopsy in the modern management of male infertility. J Urol 2002;167(1):197–200. doi:10.1097/00005392-200201000-00044. [Google Scholar] [CrossRef]
23. Bhasin S, Brito JP, Cunningham GR et al. Testosterone therapy in men with hypogonadism: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 2018;103(5):1715–1744. doi:10.1210/jc.2018-00229. [Google Scholar] [PubMed] [CrossRef]
24. Schlegel P. Microdissection TESE: sperm retrieval in non-obstructive azoopsermia. VIDEO. Hum Reprod Update 1998;4(4):439. doi:10.1093/humupd/4.4.439. [Google Scholar] [PubMed] [CrossRef]
25. Schlegel PN, Sigman M, Collura B et al. Diagnosis and treatment of infertility in men: AUA/ASRM guideline part II. Fertil Steril 2021;115(1):62–69. doi:10.1016/j.fertnstert.2020.11.016. [Google Scholar] [PubMed] [CrossRef]
26. Esteves SC. Microdissection TESE versus conventional TESE for men with nonobstructive azoospermia undergoing sperm retrieval. Int Braz J Urol 2022;48(3):569–578. doi:10.1590/S1677-5538.IBJU.2022.99.14. [Google Scholar] [PubMed] [CrossRef]
27. Bernie AM, Mata DA, Ramasamy R, Schlegel PN. Comparison of microdissection testicular sperm extraction, conventional testicular sperm extraction, and testicular sperm aspiration for nonobstructive azoospermia: a systematic review and meta-analysis. Fertil Steril 2015;104(5):1099–1103.e3. doi:10.1016/j.fertnstert.2015.07.1136. [Google Scholar] [PubMed] [CrossRef]
28. Matthiesson KL, McLachlan RI, O’Donnell L et al. The relative roles of follicle-stimulating hormone and luteinizing hormone in maintaining spermatogonial maturation and spermiation in normal men. J Clin Endocrinol Metab 2006;91(10):3962–3969. doi:10.1210/jc.2006-1145. [Google Scholar] [PubMed] [CrossRef]
29. McLachlan RI, O’Donnell L, Stanton PG et al. Effects of testosterone plus medroxyprogesterone acetate on semen quality, reproductive hormones, and germ cell populations in normal young men. J Clin Endocrinol Metab 2002;87(2):546–556. doi:10.1210/jcem.87.2.8231. [Google Scholar] [PubMed] [CrossRef]
30. Shinjo E, Shiraishi K, Matsuyama H. The effect of human chorionic gonadotropin-based hormonal therapy on intratesticular testosterone levels and spermatogonial DNA synthesis in men with non-obstructive azoospermia. Andrology 2013;1(6):929–935. doi:10.1111/j.2047-2927.2013.00141.x. [Google Scholar] [PubMed] [CrossRef]
31. Shiraishi K, Ohmi C, Shimabukuro T, Matsuyama H. Human chorionic gonadotrophin treatment prior to microdissection testicular sperm extraction in non-obstructive azoospermia. Hum Reprod 2012;27(2):331–339. doi:10.1093/humrep/der404. [Google Scholar] [PubMed] [CrossRef]
32. Zhang HL, Zhao LM, Mao JM et al. Sperm retrieval rates and clinical outcomes for patients with different causes of azoospermia who undergo microdissection testicular sperm extraction-intracytoplasmic sperm injection. Asian J Androl 2021;23(1):59–63. doi:10.4103/aja.aja_12_20. [Google Scholar] [PubMed] [CrossRef]
33. Lantsberg D, Mizrachi Y, Katz DJ. Micro-testicular sperm extraction outcomes for non-obstructive azoospermia in a single large clinic in Victoria. Aust N Z J Obstet Gynaecol 2022;62(2):300–305. doi:10.1111/ajo.13477. [Google Scholar] [PubMed] [CrossRef]
34. Corona G, Minhas S, Giwercman A et al. Sperm recovery and ICSI outcomes in men with non-obstructive azoospermia: a systematic review and meta-analysis. Hum Reprod Update 2019;25(6):733–757. doi:10.1093/humupd/dmz028. [Google Scholar] [PubMed] [CrossRef]
35. Uemura KI, Iwahata T, Ide H et al. Preoperative testosterone and follicle stimulating hormone levels are important predictors for sperm retrieval by microdissection testicular sperm extraction in non-mosaic Klinefelter syndrome. Andrologia 2022;54(11):e14588. doi:10.1111/and.14588. [Google Scholar] [PubMed] [CrossRef]
36. Mehmood S, Aldaweesh S, Junejo NN, Altaweel WM, Kattan SA, Alhathal N. Microdissection testicular sperm extraction: overall results and impact of preoperative testosterone level on sperm retrieval rate in patients with nonobstructive azoospermia. Urol Ann 2019;11(3):287–293. doi:10.4103/UA.UA_36_18. [Google Scholar] [PubMed] [CrossRef]
37. Kim TJ, Koo KC. Testosterone to luteinizing hormone ratio as a potential predictor of sperm retrieval in non-obstructive azoospermia patients. Yonsei Med J 2023;64(7):433–439. doi:10.3349/ymj.2023.0054. [Google Scholar] [PubMed] [CrossRef]
38. Cissen M, Meijerink AM, D’Hauwers KW et al. Prediction model for obtaining spermatozoa with testicular sperm extraction in men with non-obstructive azoospermia. Hum Reprod 2016;31(9):1934–1941. doi:10.1093/humrep/dew147. [Google Scholar] [PubMed] [CrossRef]
39. Bernie AM, Ramasamy R, Schlegel PN. Predictive factors of successful microdissection testicular sperm extraction. Basic Clin Androl 2013;23(1):5. doi:10.1186/2051-4190-23-5. [Google Scholar] [PubMed] [CrossRef]
40. Ramasamy R, Padilla WO, Osterberg EC et al. A comparison of models for predicting sperm retrieval before microdissection testicular sperm extraction in men with nonobstructive azoospermia. J Urol 2013;189(2):638–642. doi:10.1016/j.juro.2012.09.038. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools