 Open Access
Open Access
CASE REPORT
Clear cell sarcoma of the kidney with inferior vena cava tumor thrombus in a pediatric patient: a case report
1 Department of Urology, Hunan Children’s Hospital, Changsha, 410007, China
2 School of Pediatrics, University of South China (Hunan Children’s Hospital), Changsha, 410007, China
* Corresponding Author: Jun He. Email:
# The author contributed to the work equally and should be regarded as co-first authors
Canadian Journal of Urology 2026, 33(1), 201-210. https://doi.org/10.32604/cju.2025.067189
Received 26 April 2025; Accepted 04 September 2025; Issue published 28 February 2026
Abstract
Background: Clear cell sarcoma of the kidney (CCSK) is a rare and highly aggressive pediatric renal malignancy with a marked propensity for metastatic spread. Cases of CCSK associated with inferior vena cava (IVC) tumor thrombus (IVCTT) are exceptionally uncommon in the literature. We report a case of CCSK with IVCTT in a 15-month-old male infant. Case Description: We reported a case admitted in May 2020 for a 3-day history of fever and hematuria. Abdominal CT revealed an unevenly enhanced mass and low-density shadows within the IVC. The diagnosis of CCSK was confirmed via needle biopsy. The patient received 4 cycles of adjuvant chemotherapy. The initial surgery lasted 10 h with 600 mL blood loss, and primary closure was achieved. Postoperative management included 6 cycles of radiotherapy and 5 cycles of chemotherapy. In March 2022, CT detected IVCTT recurrence, requiring surgical intervention involving thrombus removal and partial IVC resection. This procedure lasted 8 h with 300 mL blood loss, followed by 5 additional chemotherapy cycles. The patient showed no sign of IVC obstruction, including varicose veins or lower limb edema, and maintained renal function throughout follow-up. However, Intracranial metastases were detected 15 months postoperatively. After the family opted against additional treatment, the patient succumbed to the disease. Conclusions: The management of CCSK associated with IVCTT should include consideration of IVC thrombectomy. In case of recurrent IVCTT with preserved collateral circulation, combined thrombus excision and partial IVC resection may be warranted.Keywords
Supplementary Material
Supplementary Material FileClear cell sarcoma of the kidney (CCSK) is the second most common pediatric renal malignancy after Wilms Tumor (WT), comprising 3% to 5% of malignant kidney tumors in children.1 CCSK shares many clinical and radiological features with WT and was historically considered a high-risk variant of WT until 1978, when Beckwith first classified it as a distinct entity, “clear cell sarcoma,” based on its unique pathological characteristics.2 In recent years, multimodal therapy combining chemotherapy, surgery, and radiotherapy has significantly improved CCSK outcomes.3 The co-occurrence of CCSK and inferior vena cava tumor thrombus (IVCTT) is exceptionally rare.4–6 A review of seven published studies identified only eight pediatric cases of CCSK with IVCTT. Unlike WT, CCSK has a higher propensity for metastasis, particularly to the bones, a pattern historically termed the “pediatric renal tumor with bone metastases”.7 Recently, brain metastases have emerged as the primary metastatic site, likely due to intensified early-stage treatment.8
Notably, we encountered a case of a child with CCSK and IVCTT at Hunan Children’s Hospital. This study was approved by the Clinical Research Ethics Committee of Hunan Children’s Hospital (No. HCHLL-2024-30). Informed consent was obtained from the patient’s father. After biopsy confirmation, the child received five cycles of chemotherapy followed by CCSK resection and IVCTT removal. Subsequent IVCTT recurrence required partial thrombectomy and with inferior vena cava (IVC) intervention. Postoperative recovery was favorable in the short term; however, the child later developed intracranial metastases. Following the family’s decision to discontinue treatment, the child passed away. This article summarizes the clinical features of this case and reviews relevant literature to analyze surgical indications and assess the evaluate treatment efficacy in pediatric CCSK with IVCTT and thrombus recurrence.
Clinical findings at initial diagnosis
A 15-month-old male infant presented to Hunan Children’s Hospital on 19 May 2020, with a three-day history of fever and hematuria. The initial evaluation raised suspicion of a right renal tumor. The patient had no significant personal or family history of tumors, and his developmental history was unremarkable. Physical examination revealed a blood pressure of 142/93 mmHg. Abdominal examination demonstrated a soft, non-tender abdomen without rebound tenderness. A palpable mass measuring approximately 10 cm in diameter was measuring in the right upper quadrant, characterized by ill-defined borders and moderate mobility. The timeline of the diagnosis and treatment process was shown in Figure 1.
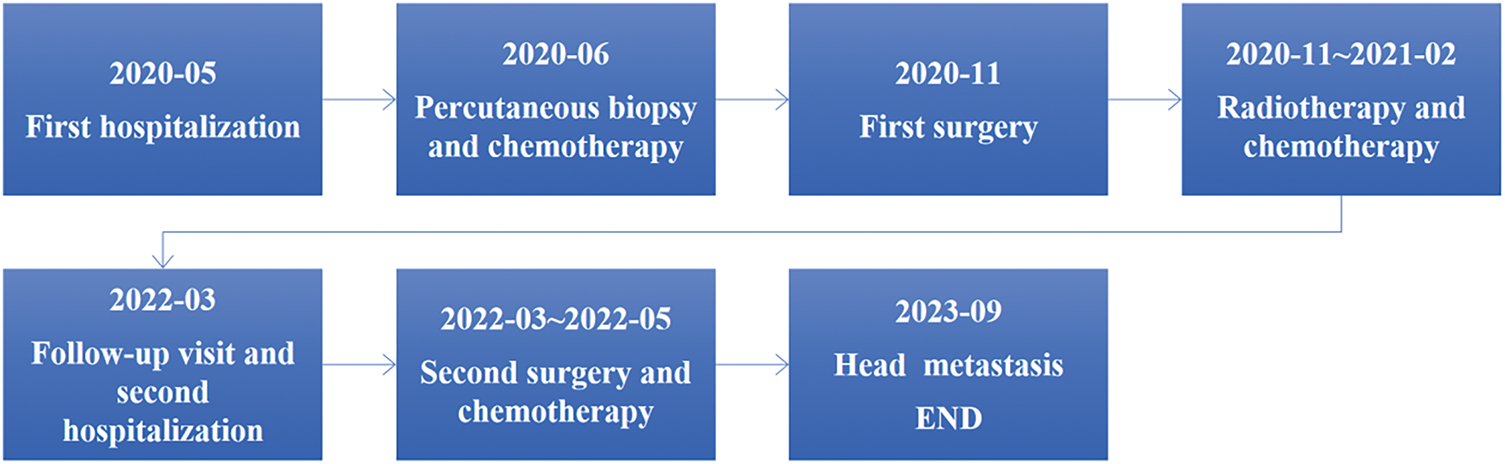
FIGURE 1. The timeline of the diagnosis and treatment process
Cross-sectional imaging demonstrated a large right renal mass measuring 12.2 × 9.9 × 8.9 cm with heterogeneous density (8–53 Hounsfield units) and irregular contrast enhancement, causing significant compression of adjacent structures. The IVC below the hepatic segment showed hypodense lesions with heterogeneous enhancement, alongside multiple enlarged retroperitoneal lymph nodes (Figure 2A). IVC ultrasonography revealed a 53 × 16 mm hypoechoic mass extending from the right renal vein-IVC junction to the retrohepatic IVC segment (Figure 2B). Plain chest radiography and contrast-enhanced cranial CT demonstrated interstitial inflammatory changes. Renal function was normal.
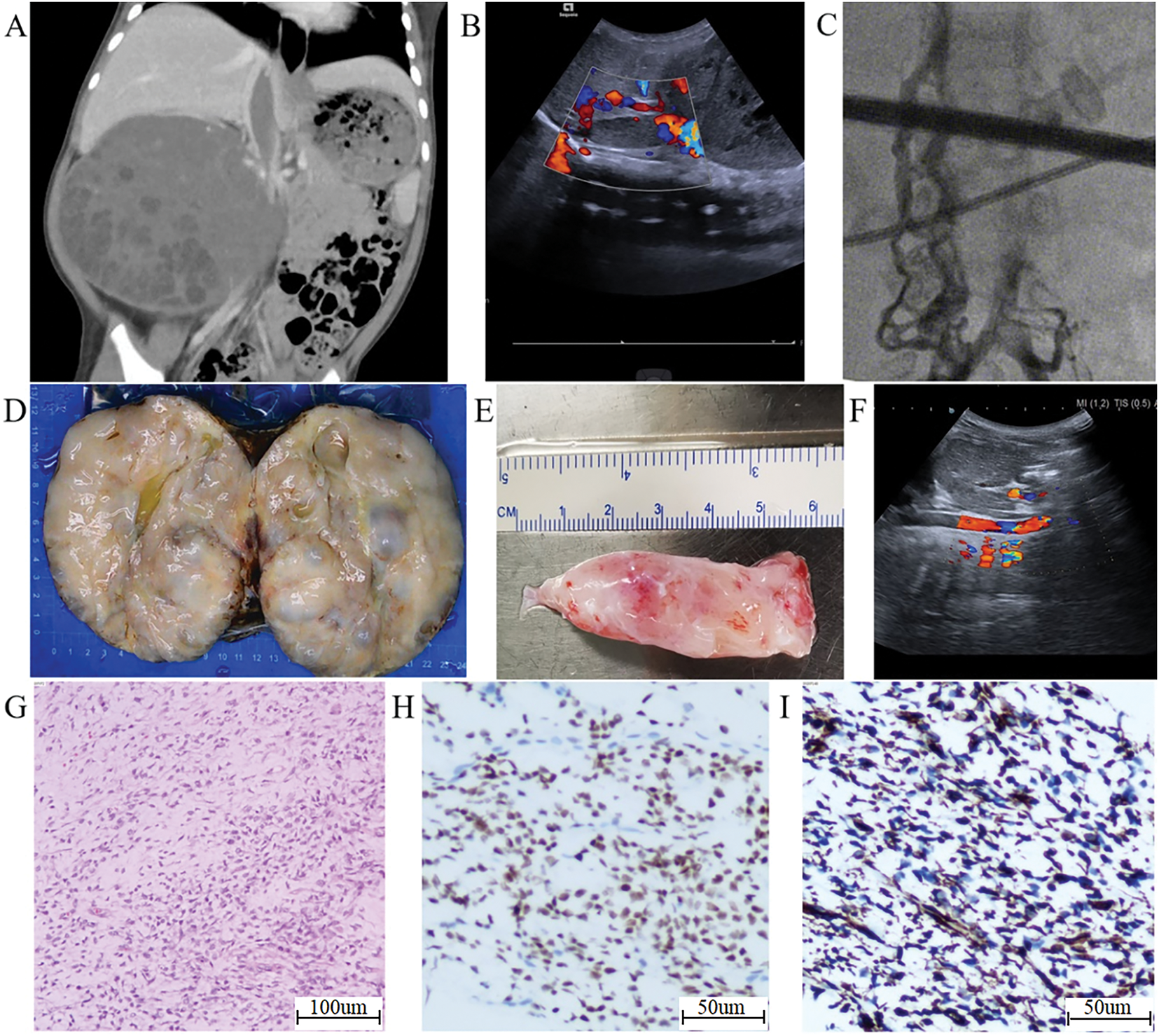
FIGURE 2. Radiological and pathological findings in a clear cell sarcoma of the kidney (CCSK) patient treated with right radical nephrectomy and inferior vena cava (IVC) thrombectomy. (A) Abdominal computed tomography (CT) shows a large mass in the right kidney area, with low-density filling in the IVC, compressing surrounding organs. (B) Color Doppler ultrasound shows a hypoechoic mass filling the IVC. (C) Digital subtraction angiography (DSA) shows interrupted contrast flow in the IVC with reflux from the right lumbar vein. (D) Macroscopic specimen of the resected tumor. (E) The thrombus extracted from the opened IVC. (F) Esophageal Doppler ultrasound confirms unobstructed IVC blood flow after thrombus extraction. (G) Microscopic image of tumor cells (H&E staining ×100). (H) BCOR staining of the nuclei of tumor cells (2+). (I) Vimentin staining of the cytoplasm of tumor cells (3+)
In June 2020, an ultrasound-guided percutaneous biopsy of the right renal tumor was performed. Pathology confirmed CCSK with BCOR gene abnormality. Staging revealed Stage III CCSK with Level III IVCTT.9 After diagnosis, the patient received four cycles of chemotherapy (cyclophosphamide, doxorubicin, vincristine, etoposide, and ifosfamide) combined with oral captopril for hypertension management. Post-chemotherapy CT demonstrated extensive tumor necrosis without a significant reduction in tumor or thrombus size.
In November 2020, preoperative digital subtraction angiography (DSA) demonstrated non-visualization of the right iliac vein distal to its junction with the IVC, with extensive collateral circulation bilaterally. The collateral flow coursed through the paravertebral veins to the azygos vein and into the right atrium. A filling defect was observed at the IVC-hepatic vein confluence (Figure 2C).
During surgery, a reverse T incision was made. The procedure involved exploration around the kidney followed by radical resection of the right renal tumor, including right ureterectomy, retroperitoneal lymph node dissection, and IVC thrombectomy. The resected tumor measured approximately 13 × 12.5 × 10 cm, with an intact capsule, dark purple-gray coloration, and soft consistency. An enlarged lymph node (~1 cm) was identified near the right renal hilum and IVC. After mobilizing the liver, the IVC and its major branches were exposed and occluded. The surgical procedure involved incising the IVC, removing the thrombus, repairing the vessel with sutures, releasing vascular clamps, completing perirenal lymph node clearance, and finally closing the abdominal incision (Figure 2D–F). At the junction of the inferior vena cava and right renal vein, the tumor thrombus was found to be tightly adhered to the vessel wall. To avoid damaging the IVC, this portion was carefully excised using sharp dissection.
Histopathological examination with hematoxylin and eosin (H&E) staining demonstrated features consistent with CCSK, including focal cystic degeneration. The tumor infiltrated the renal parenchyma without capsule penetration but showed involvement of the renal pelvis wall. No tumor cells were identified in the renal sinus or hilar vessels. The IVCTT consisted of tumor cells. No tumor cells were identified at the ureter resection margin or in the lymph nodes adjacent to the right renal vein (0/1). Immunohistochemistry results were as follows: Ki-67 (20%), Vim (3+), CyclinD1 (2+), BCOR (2+), CD56 (+), INI-1 (2+) (Figure 2G–I).
After surgery, the patient received six radiotherapy courses at Xinhua Hospital in Shanghai, with 10.8 Gy/6Fx delivered to the tumor bed (including the IVC). Four chemotherapy courses were subsequently administered at our hospital using cyclophosphamide, adriamycin, vincristine, etoposide, and doxorubicin. Treatment concluded in February 2021. A May 2021 positron emission tomography/computed tomography (PET/CT) scan showed post-right nephrectomy changes without detectable soft tissue masses or localized radioactive uptake. Renal function remained normal, and no other abnormalities were observed, indicating complete remission.
Clinical findings following IVCTT recurrence
During a follow-up visit in March 2022, an abdominal CT scan revealed narrowing in narrowing in the hepatic segment of the IVC, along with partial contrast filling defects in the lower and infra-hepatic IVC segments. Additionally, numerous vascular shadows were observed distally (Figure 3A), consistent with IVCTT recurrence. Subsequent DSA findings confirmed IVC obstruction at the L1/2 level, with contrast medium draining into the bilateral vertebral venous plexus and ultimately into the superior vena cava. Notably, the distal left renal vein did not empty into the IVC but instead communicated with the hemiazygos vein via the perirenal venous plexus (Figure 3B,C).
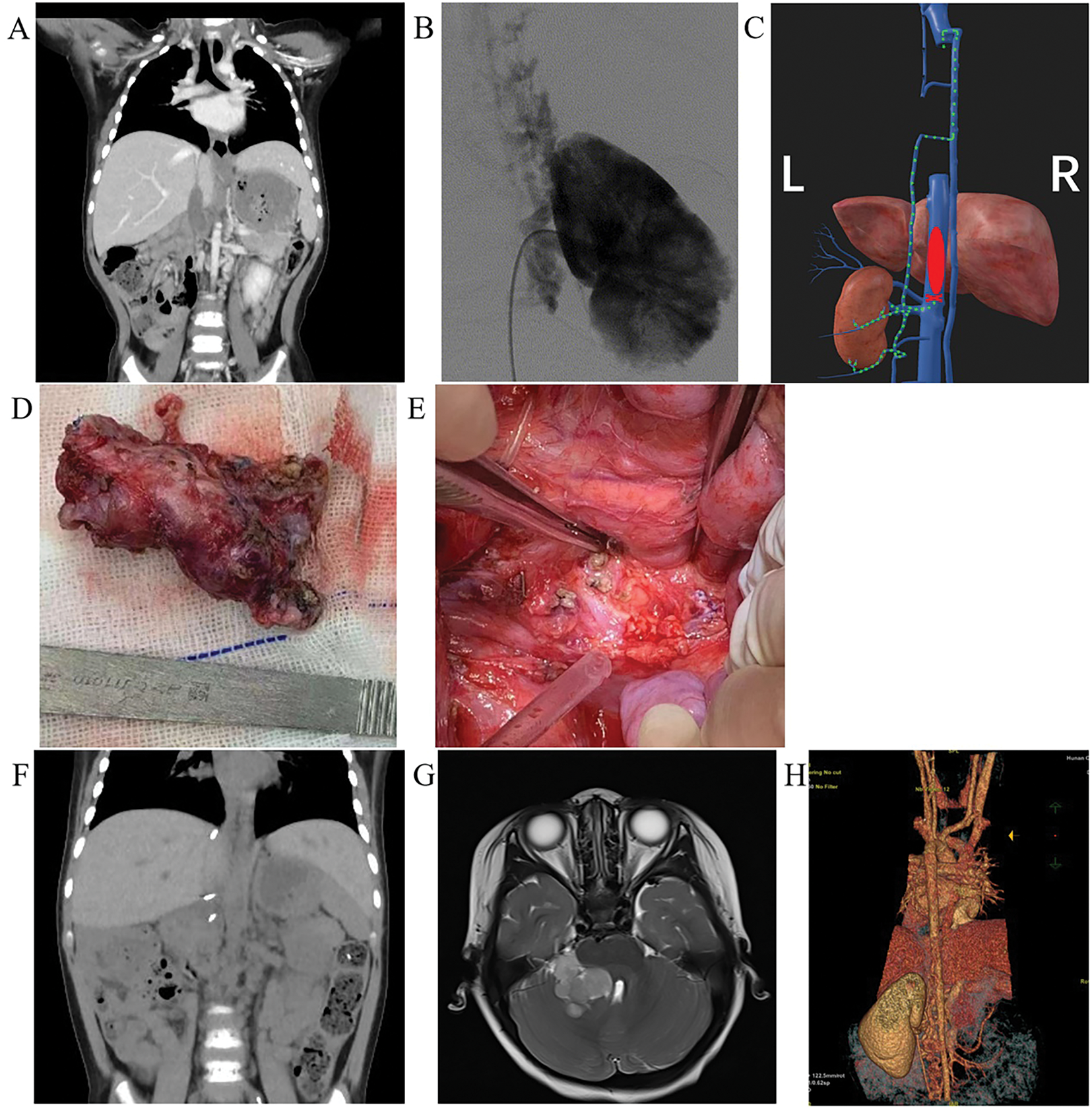
FIGURE 3. Imaging and intraoperative findings in a patient undergoing partial inferior vena cava (IVC) resection and thrombectomy. (A) Computed tomography (CT) imaging in March 2022 shows inferior vena cava tumor thrombus (IVCTT) recurrence at the hepatic segment and the infrahepatic segment of the IVC. (B) Intraoperative digital subtraction angiography (DSA) reveals contrast agent draining from the left renal vein into the hemiazygos vein. (C) Schematic diagram shows retrograde left renal blood flow (green dashed arrow) secondary to IVC occlusion by the thrombus (red ellipse). (D) The intraoperative findings show the resected thrombus and partial IVC. (E) The exposed part of the retroperitoneum after the resection of the thrombus and partial IVC. (F) Postoperative reassessment at three months based on CT shows complete resection of the thrombus with no recurrence. (G) Postoperative cranial magnetic resonance imaging (MRI) at 15 months shows a mass in the right cerebellopontine angle area, measuring approximately 3.4 × 3.2 × 3.7 cm, with slightly prolonged T1 and T2 signals, and significant enhancement observed after contrast. (H) Postoperative single-energy vascular imaging (posterior view) at 15 months shows occlusion of the left renal vein and tortuous dilatation of the hemiazygos vein and left spermatic vein
The surgical procedure involved partial resection of the inferior vena cava, tumor thrombectomy of the inferior vena cava, and partial resection of the left renal vein through the same incision site used in previous operations. The left renal vein was dissected and suspended. Working from inferior to superior, the inferior vena cava was dissected and encircled with a horizontal sling placed 2 cm below the left renal vein. The scar tissue surrounding the liver was released, and the second hepatic portal was mobilized. The anterior, left, right and posterior walls of the IVC were fully dissociated, and a blocking band was placed around the IVC below the diaphragm. Intraoperative ultrasound confirmed the thrombus was contained within the occluded segments of each vessel. Sequential vascular occlusion was performed in the following order: the subphrenic IVC, followed by a segment 2 cm below the left renal vein, and then 1.5 cm distal to the left renal vein. After occluding the thrombus-containing IVC segment proximally and distally, there was no evidence of abdominal organ congestion, with maintained cardiac output, normal urine production, and absence of lower limb edema, indicating adequate IVC collateral circulation. The inferior vena cava and its branches were transected between the second hepatic portal to the left renal vein. A 3 cm segment of inferior vena cava containing the tumor thrombus were resected (Figure 3D,E), with the proximal stump closed using 5-0 PROLENE sutures. The subdiaphragmatic IVC occlusion was then released after 10 min of hepatic vascular isolation. After all the blood vessels were sutured, the remaining vascular flows were blocked for approximately 30 min. The abdomen was then closed, completing the surgical procedure. Pathological examination demonstrated tumor cells within the IVC lumen and adjacent vascular wall, without extension into surrounding tissues or metastasis involvement of regional lymph nodes.
Following the surgical procedure, the patient received five cycles of chemotherapy (ifosfamide, etoposide, vincristine, and irinotecan) and underwent regular follow-up at Hunan Children’s Hospital. A postoperative abdominal CT scan performed three months after surgery showed no tumor recurrence or residual thrombus (Figure 3F). During a subsequent outpatient review in June, the patient was in good general condition, with no evidence of superficial lymph node enlargement, abdominal pain, lower limb edema, varicose veins, or other symptoms. Renal function tests were normal. Unfortunately, the patient returned to our hospital 15 months after surgery due to persistent head tilt and along with right eye deviation lasting for more than 10 days. While an abdominal CT scan showed no recurrent lesions, a head magnetic resonance imaging (MRI) revealed a space-occupying lesion in the right cerebellopontine angle, suggesting a malignant brain tumor (Figure 3G). Despite recommendations for further treatment, the patient’s parents declined. The patient subsequently died.
CCSK primarily occurs in children aged 2 to 4 years, with a male-to-female predominance of approximately 2:1.3 The standard treatment consists of radical nephrectomy combined with chemotherapy. In cases where primary tumor resection is difficult, neoadjuvant chemotherapy is frequently administered following a definitive biopsy diagnosis to reduce tumor volume and simplify surgical intervention. This case was classified as stage III CCSK based on the National Wilms’ Tumor Study Group (NWTSG) protocol10 and treated using the CCCG-WT-2009(3) regimen. A literature review identified eight pediatric patients diagnosed with CCSK and IVCTT across seven articles (Table 1), with a median follow-up duration of 16 months (range: 9 to 23 months). Among these cases, five achieved complete remission during follow-up, while three died following bone metastasis. Five patients received neoadjuvant chemotherapy after biopsy. In Patient 1, tumor volume decreased by 82% after 12 weeks of neoadjuvant chemotherapy. However, patients 3, 5, and 7 exhibited no reduction in tumor or thrombus size, indicating chemotherapy insensitivity, a finding consistent with our case.
A small number of CCSK cases may present with IVCTT.5 In 2016, Zamperlini-Netto et al.6 reviewed clinical data from 12 children with CCSK and found that 8.3% (1/12) exhibited IVCTT. IVC DSA can accurately simulate blood flow through the IVC using contrast agents, helping assess the degree of obstruction and guide surgical planning by evaluating collateral circulation. The presence of IVCTT significantly increases surgical procedural complexity and places substantial demands on the operating surgeon. Currently, the established surgical approach for renal tumors with IVCTT involves radical nephrectomy combined with IVC incision for thrombus extraction.11
The surgical approach, complexity, blood loss, and postoperative complications vary according to thrombus grade. In this case, the tightly adherent thrombus required excision to ensure complete removal while preserving the vena cava. This technique, however, made negative margin confirmation challenging, underscoring the necessity for adjuvant radiation and chemotherapy to prevent tumor recurrence. Based on current findings, the surgical strategy for managing adherent thrombus during the initial procedure may contribute to the recurrence in this case. A similar situation was observed in Patient 3 (Table 1), who had a tightly adherent intracardiac thrombus. Due to the limited number of specific cases, current research is insufficient regarding the correlation between the thrombus adhesion extent and tumor recurrence, and established surgical protocols are still lacking, requiring further investigation.
Thrombus recurrence with scar adhesions may enhance its attachment to the vessel wall, resulting in longer surgical time for venous incision during thrombus removal and making complete resection more difficult due to increased adhesion. For radical tumor resection, removal of adherent vascular segments is recommended. Indications for IVC resection include complete IVC occlusion by the thrombus, severe adhesion to vascular endothelium, venous wall invasion, and tight tumor encasement of the IVC.12 Intraoperative transesophageal ultrasound combined with preoperative IVC DSA helps accurately determine the extent of IVC resection while protecting the collateral circulation trunk. In this case, preoperative IVC DSA showed that specific IVC segments were not visualized, with patent collateral circulation. Notably, in complete occlusion cases, compensatory collateral circulation from the vertebral venous plexus and the azygos vein provided nearly complete compensation. Clinical evidence has confirmed that partial IVC resection without reconstruction is safe and feasible in IVCTT surgery, as collateral circulation can prevent serious hemodynamic complications in patients.13,14
Currently, the necessity of reconstruction following IVC resection remains controversial. In a single-center study, Ye et al.15 retrospectively analyzed 17 patients undergoing segmental IVC resection for renal tumor thrombus, among whom 6 developed postoperative complications related to resection. Thrombosis and elevated creatinine levels resolved within approximately 2 weeks through symptomatic management, including anticoagulation and hemodialysis, with two patients demonstrating lower limb edema during short-term follow-up. Lifante et al.16 performed long-term follow-up (up to 15 years) on patients who underwent partial IVC resection, showing no recurrence or sequelae with continuous anticoagulant therapy. The possible cause of acute renal failure in elderly patients after partial IVC resection was identified as pre-existing renal function decline.17 In children, renal function reaches adult levels at approximately 1.5 years of age. The function of a solitary kidney, as assessed by total glomerular filtration rate and 24-h urine protein, can remain stable long-term without showing deterioration.18 In this case, the child did not undergo IVC reconstruction after resection. While IVC reconstruction more closely matches physiological anatomy, it carries multiple complications and risks, especially in pediatric patients. These include increased surgical costs, longer operation times, possible artificial vascular thrombosis, and rejection risks. Therefore, we do not recommend it as the preferred option.
The recurrence rate of CCSK is relatively high, ranging from 15% to 39%,3 with approximately 30% of patients experiencing recurrence two or more years after initial diagnosis.19 In the present case, the first recurrence, involving IVCTT, occurred 20 months after the initial surgery, while the second recurrence, which presented as brain metastasis, was detected 15 months following the second surgery. These findings align with existing literature (Table 1). Given this pattern, regular and systematic follow-up examinations are critical for early detection of disease recurrence. Notably, the pediatric patient in this case exhibited no obvious clinical symptoms during the first recurrence. The tumor demonstrated slow progression within the IVC, allowing for collateral circulation compensation. This physiological adaptation also facilitated partial IVC resection during the surgical intervention. Published reports describe up to four CCSK recurrences in some patients, all confined to the abdominal region. In one documented case, despite three surgical resections and 11 chemotherapy cycles, the patient succumbed to the disease 2.8 years after recurrence. The largest cohort study on recurrent CCSK, involving 37 patients, reported a 5-year event-free survival (EFS) rate of 18% and a 5-year overall survival (OS) rate of 26%. The study concluded that metastasectomy, combined with localized radiotherapy and intensified chemotherapy, could induce a second complete remission and improve survival outcomes.3 Isolated case reports suggest potential benefits of autologous stem cell rescue (ASCR) and targeted therapies such as anlotinib20 in treating recurrent CCSK, however, the efficacy and long-term prognosis of these interventions require further investigation. Due to the rarity of recurrent CCSK cases, no standardized optimal treatment protocol currently exists.
Literature review from the follow-up period of 9 to 23 months revealed bone metastasis occurring in 12.5% (1/8) of the pediatric cases, with all affected patients ultimately succumbing to the disease. CCSK exhibits marked invasive potential and a pronounced tendency for distant metastasis, with preferential dissemination to the brain, followed by osseous structures.2 In the present case, brain metastasis developed during the second recurrence, with strabismus as the presenting clinical manifestation. The blood-brain barrier functions as a protective sanctuary for tumor cells, shielding microscopic metastatic deposits from the effects of radiotherapy and chemotherapy.21 Published studies document complete remission in 12 CCSK patients with central nervous system recurrence. These include 6 cases reported by Radulescu et al. (median follow-up 2.5 years),22 2 cases by Goosken et al.,23 and 4 cases by Friesenbichler et al.24 (median follow-up 9.3 years). All patients received multimodal therapy combining metastasectomy, radiotherapy, chemotherapy, and autologous stem cell transplantation. In contrast to these reported outcomes, the parents of the patient in our case declined further therapeutic interventions, the patient ultimately died.
Tumor metastasis mechanisms are highly complex. Tumor cells exhibit characteristics of seeking advantage and avoiding harm, hypothesized to result from adverse conditions in the original tumor environment, and promote tumor cells migration.25 Additionally, the existence of tumor stem cells and the support from the tumor microenvironment greatly facilitate tumor metastasis. Although the blood-brain barrier normally prevents tumor cells from entering brain tissue, several factors can increase blood-brain barrier permeability. These include the opening of calcium-activated potassium channels,26 substance P stimulation of brain microvascular endothelial cells to produce TNF-α and angiogenic factors,27 and tissue proteinase S cleavage of junctional adhesion molecule JAM-B.28 These mechanisms may allow tumor cells to enter the brain tissue. The growth of metastatic tumors in the brain depends on complex and specific tumor factors such as cyclooxygenase COX2 and the epidermal growth factor receptor (EGFR) ligand HBEGF.29 Current treatments for brain metastases include neurosurgical resection, chemotherapy, brain radiotherapy, and biological agents. Chemotherapy often uses drugs like temozolomide that can penetrate the blood-brain barrier, while new radiotherapy techniques help eradicate lesions while minimizing damage to normal tissues. Some researchers suggest that prophylactic brain radiotherapy combined with VEGF-A inhibitors may be promising for treating tumors prone to brain metastasis.30
In summary, this pediatric case demonstrates CCSK complicated by IVCTT, leading to recurrence that required a second surgery. For CCSK patients with IVCTT who meet the criteria for IVC resection, simultaneous removal is recommended following the “no residual tumor” surgical principle to achieve cure. The mechanisms of CCSK metastasis and recurrence are not fully understood. Treatments mainly involve lesion resection and increased therapeutic doses. Future multicenter clinical studies with large sample sizes and long-term follow-up are needed to guide treatment strategies and predict prognosis in clinical practice.
Acknowledgement
We gratefully acknowledge the medical officer and physicians in the Departments of Pediatrics and Pediatric Surgery for their clinical support.
Funding Statement
Research Project of Hunan Health Commission in 2023 (D202304058618); Innovation Project of Hunan Science and Technology Department (2018SK504); National Nature Science Foundation of China (No. 82203414).
Author Contributions
Feng Ning drafted the article, obtained research funds; Xingyu Long collected and analyzed data, repaired and wrote articles; Zhi Wang, Bo Yin, and Kan Wang guided and supported contributions; He Jun reviewed and guided the knowledge of the article content. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials
Not applicable.
Ethics Approval
Publication of this case report was approved by the Clinical Research Ethics Committee of Hunan Children’s Hospital, China (No. HCHLL-2024-30).
Informed Consent
Got the informed consent from the patient’s guardian. Written informed consent was obtained from the patient’s legal guardian for publication of this case report and accompanying images. The consent documentation is available for review by the Editor-in-Chief of this journal upon request.
Conflicts of Interest
The authors declare no conflicts of interest to report regarding the present study.
Supplementary Materials
The supplementary material is available online at https://www.techscience.com/doi/10.32604/cju.2025.067189/s1.
References
1. El Kababri M, Khattab M, El Khorassani M et al. Clear cell sarcoma of the kidney. A study of 13 cases. Arch Pediatr 2004;11(7):794–799. doi:10.1016/j.arcped.2004.02.023. [Google Scholar] [PubMed] [CrossRef]
2. Aldera AP, Pillay K. Clear cell sarcoma of the kidney. Arch Pathol Lab Med 2020;144(1):119–123. doi:10.5858/arpa.2018-0353-RS. [Google Scholar] [PubMed] [CrossRef]
3. Gooskens SL, Graf N, Furtwangler R et al. Position paper: rationale for the treatment of children with CCSK in the UMBRELLA SIOP-RTSG 2016 protocol. Nat Rev Urol 2018;15(5):309–319. doi:10.1038/nrurol.2018.14. [Google Scholar] [PubMed] [CrossRef]
4. Hiradfar M, Zabolinejad N, Shojaeian R et al. Pediatric clear cell sarcoma of the kidney with atriocaval thrombus. Pediatr Surg Int 2012;28(11):1141–1145. doi:10.1007/s00383-012-3160-0. [Google Scholar] [PubMed] [CrossRef]
5. Smets AM, de Kraker J. Malignant tumours of the kidney: imaging strategy. Pediatr Radiol 2010;40(6):1010–1018. doi:10.1007/s00247-010-1584-z. [Google Scholar] [PubMed] [CrossRef]
6. Zamperlini-Netto G, Zanette A, Wehbi E et al. PO-60-Renal tumors with extensive vascular disease: management challenges in a pediatric series from the Hospital for Sick Children. Thromb Res 2016;140(Suppl 1):S198–S199. doi:10.1016/S0049-3848(16)30193-1. [Google Scholar] [PubMed] [CrossRef]
7. Zhang A, Yuan X, Jiang S et al. Outcomes of children with clear cell sarcoma of kidney following NWTS strategies in Shanghai China (2003–2021). PLoS One 2024;19(7):e0306863. doi:10.1371/journal.pone.0306863. [Google Scholar] [PubMed] [CrossRef]
8. Benedetti DJ, Renfro LA, Tfirn I et al. Treatment and outcomes of clear cell sarcoma of the kidney: a report from the Children’s Oncology Group studies AREN0321 and AREN03B2. Cancer 2024;130(13):2361–2371. doi:10.1002/cncr.35266. [Google Scholar] [PubMed] [CrossRef]
9. Onal B, Simsekoglu MF, Gultekin MH, Demirdag C, Citgez S, Erozenci A. Clinical outcomes of radical surgery in patients with renal carcinoma and associated venous thrombosis: single-centre experience in a tertiary care institution. Int J Clin Pract 2021;75(4):e13811. doi:10.1111/ijcp.13811. [Google Scholar] [PubMed] [CrossRef]
10. Geller JI, Renfro LA, Grundy PE et al. Rhabdoid tumor of the kidney and soft tissues: results from national wilms tumor study-5 and children’s oncology group study AREN0321. Pediatr Blood Cancer 2025;72(3):e31490. doi:10.1002/pbc.31490. [Google Scholar] [PubMed] [CrossRef]
11. Ciancio G, Gonzalez J, Shirodkar SP, Angulo JC, Soloway MS. Liver transplantation techniques for the surgical management of renal cell carcinoma with tumor thrombus in the inferior vena cava: step-by-step description. Eur Urol 2011;59(3):401–406. doi:10.1016/j.eururo.2010.07.028. [Google Scholar] [PubMed] [CrossRef]
12. Liu Z, Zhang Q, Zhao X et al. Inferior vena cava interruption in renal cell carcinoma with tumor thrombus: surgical strategy and perioperative results. BMC Surg 2021;21(1):402. doi:10.1186/s12893-021-01400-2. [Google Scholar] [PubMed] [CrossRef]
13. Liu C, Zheng Y, Yang X, Shao J, Song X, Liu B. Robot-assisted laparoscopic inferior vena cava segmental resection for renal tumor with tumor thrombus invading the vascular wall. Beijing Da Xue Xue Bao Yi Xue Ban 2025;57(4):796–802. doi:10.1016/j.avsg.2009.12.015. [Google Scholar] [PubMed] [CrossRef]
14. Wang S, Gaurab P, Cui J, Yu S, Fan Y, Zhu Z. Impact of left renal vein ligation on renal function following en bloc resection of segmental inferior vena cava and right kidney. Ann Surg Oncol 2024;31(7):4787–4794. doi:10.1245/s10434-024-15324-7. [Google Scholar] [PubMed] [CrossRef]
15. Ye JF, Ma LL, Zhao L, Wang GL. Segmental vena cava resection for the treatment of renal tumor with invading tumor thrombus. Beijing Da Xue Xue Bao Yi Xue Ban 2018;50(1):183–187. [Google Scholar] [PubMed]
16. Lifante JC, Glehen O, Peyrat P et al. Segmental resection of tumoral invasion of the inferior vena cava without reconstruction. Ann Chir 2001;126(1):46–50. doi:10.1016/s0003-3944(00)00457-0. [Google Scholar] [PubMed] [CrossRef]
17. Daylami R, Amiri A, Goldsmith B, Troppmann C, Schneider PD, Khatri VP. Inferior vena cava leiomyosarcoma: is reconstruction necessary after resection? J Am Coll Surg 2010;210(2):185–190. doi:10.1016/j.jamcollsurg.2009.10.010. [Google Scholar] [PubMed] [CrossRef]
18. Önal HG, Semet Y. Clinical outcomes and risk factors in pediatric patients with solitary functioning kidney: a comparative analysis of congenital and acquired etiologies. Front Pediatr 2025;13:1574000. doi:10.3389/fped.2025.1574000. [Google Scholar] [PubMed] [CrossRef]
19. Furtwängler R, Gooskens SL, van Tinteren H et al. Clear cell sarcomas of the kidney registered on International Society of Pediatric Oncology (SIOP) 93-01 and SIOP, 2001 protocols: a report of the SIOP Renal Tumour Study Group. Eur J Cancer 2013;49(16):3497–3506. doi:10.1016/j.ejca.2013.06.036. [Google Scholar] [PubMed] [CrossRef]
20. Tao J, Yang H, Hao Z et al. Positive response of a recurrent clear cell sarcoma to anlotinib combined with chemotherapy: a case report. Medicine 2022;101(48):e32109. doi:10.1097/MD.0000000000032109. [Google Scholar] [PubMed] [CrossRef]
21. Roubil JG, Harris TJ. Radiation therapy for cerebral metastases: non-stereotactic treatment options. Adv Cancer Res 2025;165:231–254. doi:10.1016/bs.acr.2025.04.008. [Google Scholar] [PubMed] [CrossRef]
22. Radulescu VC, Gerrard M, Moertel C et al. Treatment of recurrent clear cell sarcoma of the kidney with brain metastasis. Pediatr Blood Cancer 2008;50(2):246–249. doi:10.1002/pbc.21131. [Google Scholar] [PubMed] [CrossRef]
23. Gooskens SL, Furtwängler R, Spreafico F et al. Treatment and outcome of patients with relapsed clear cell sarcoma of the kidney: a combined SIOP and AIEOP study. Br J Cancer 2014;111(2):227–233. doi:10.1038/bjc.2014.291. [Google Scholar] [PubMed] [CrossRef]
24. Friesenbichler W, Lüftinger R, Kropshofer G et al. Clear cell sarcoma of the kidney in Austrian children: long-term survival after relapse. Pediat Blood Canc 2021;68(5):e28860. doi:10.1002/pbc.28860. [Google Scholar] [PubMed] [CrossRef]
25. Wang RA, Lu YY, Fan DM. Reasons for cancer metastasis: a holistic perspective. Mol Clin Oncol 2015;3(6):1199–1202. doi:10.3892/mco.2015.623. [Google Scholar] [PubMed] [CrossRef]
26. Ningaraj NS, Rao M, Hashizume K, Asotra K, Black KL. Regulation of blood-brain tumor barrier permeability by calcium-activated potassium channels. J Pharmacol Exp Ther 2002;301(3):838–851. doi:10.1124/jpet.301.3.838. [Google Scholar] [PubMed] [CrossRef]
27. Rodriguez PL, Jiang S, Fu Y, Avraham S, Avraham HK. The proinflammatory peptide substance P promotes blood-brain barrier breaching by breast cancer cells through changes in microvascular endothelial cell tight junctions. Int J Cancer 2014;134(5):1034–1044. doi:10.1002/ijc.28433. [Google Scholar] [PubMed] [CrossRef]
28. Sevenich L, Bowman RL, Mason SD et al. Analysis of tumour- and stroma-supplied proteolytic networks reveals a brain-metastasis-promoting role for cathepsin S. Nat Cell Biol 2014;16(9):876–888. doi:10.1038/ncb3011. [Google Scholar] [PubMed] [CrossRef]
29. Bos PD, Zhang XH-F, Nadal C et al. Genes that mediate breast cancer metastasis to the brain. Nature 2009;459(7249):1005–1009. doi:10.1038/nature08021. [Google Scholar] [PubMed] [CrossRef]
30. Kienast Y, Winkler F. Therapy and prophylaxis of brain metastases. Expert Rev Anticancer Ther 2014;10(11):1763–1777. doi:10.1586/era.10.165. [Google Scholar] [PubMed] [CrossRef]
31. Han H, Bertrand KC, Patel KR et al. BCOR-CCNB3 fusion-positive clear cell sarcoma of the kidney. Pedia Blood Canc 2020;67(4):e28151. doi:10.1002/pbc.28151. [Google Scholar] [PubMed] [CrossRef]
32. Zigman A, Shen I. Clear cell sarcoma of the kidney with cavo-atrial tumor thrombus: complete resection in a child. J Pediatr Surg 2006;41(8):1464–1466. doi:10.1016/j.jpedsurg.2006.04.027. [Google Scholar] [PubMed] [CrossRef]
33. Sugandhi N, Munghate G, Malankar DP et al. Pediatric clear cell sarcoma of the kidney with cavoatrial thrombus. J Pediatr Surg 2011;46(12):2387–2390. doi:10.1016/j.jpedsurg.2011.09.050. [Google Scholar] [PubMed] [CrossRef]
34. Doya LJ, Alyousef K, Oukan M et al. Clear cell sarcoma of the kidney with inferior vena cava thrombus: a case report. J Med Case Rep 2022;16(1):295. doi:10.1186/s13256-022-03489-2. [Google Scholar] [PubMed] [CrossRef]
35. Khozeimeh N, Sinha P, Dome JS, Guzzetta PCJr. Strategy for management of retroperitoneal tumors with caval tumor thrombus. J Pediatr Surg 2011;46(11):2065–2070. doi:10.1016/j.jpedsurg.2011.06.041. [Google Scholar] [PubMed] [CrossRef]
36. Ohtake N, Shiono A, Okabe K et al. Clear cell sarcoma extending into the inferior vena cava. Nihon Hinyokika Gakkai Zasshi 1995;86(7):1298–1301. doi:10.5980/jpnjurol1989.86.1298. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF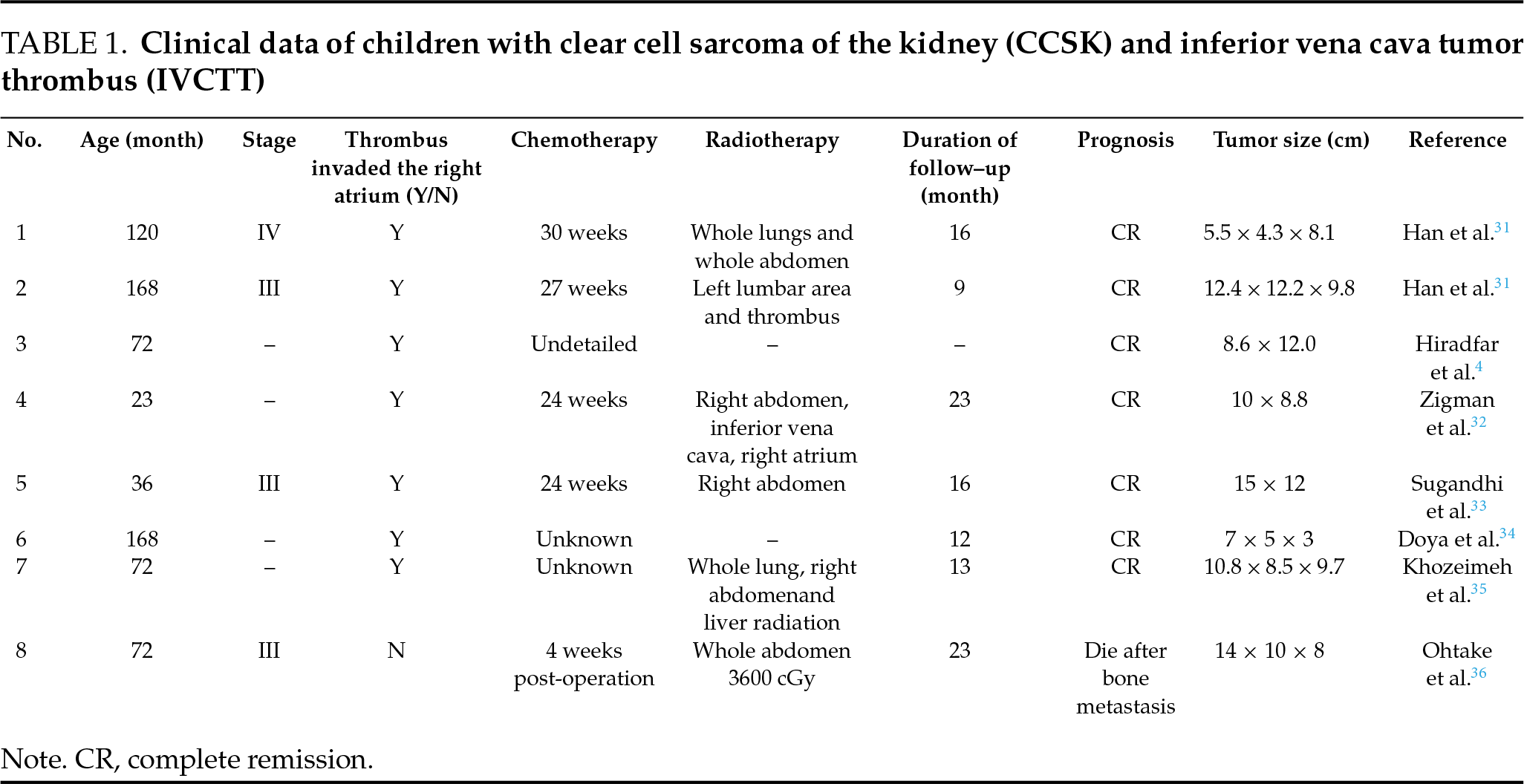
 Downloads
Downloads
 Citation Tools
Citation Tools