 Open Access
Open Access
CASE REPORT
Case report: minimally invasive management of two major complications of colonic perforation and pseudoaneurysm formation following nephrostomy tube placement
1 Urology Service, CHU Tours, 2 boulevard Tonnellé, Tours, 37000, France
2 GRC n°20, Groupe de Recherche Clinique sur la Lithiase Urinaire, Hôpital Tenon, Sorbonne Université, Paris, 75020, France
3 Urology Service, AP-HP, Hopital Tenon, Assistance-Publique Hopitaux de Paris, Sorbonne Universite, 4 Rue de la Chine, Paris, 75020, France
* Corresponding Author: Marie-Lou Letouche. Email:
Canadian Journal of Urology 2026, 33(1), 227-232. https://doi.org/10.32604/cju.2025.067253
Received 28 April 2025; Accepted 01 September 2025; Issue published 28 February 2026
Abstract
Background: We present a case of two major complications following insertion of a nephrostomy managed in a minimally invasive way. Our case is the first in the literature to describe this minimally invasive treatment technique for colon perforation in a completely asymptomatic patient. Case Description: A 75-year-old female patient with a history of bilateral obstructive uropathy secondary to bilateral radiation-induced ureteric strictures attended for bilateral nephrostomy placement. The patient had a history of endometrial cancer, treated previously by total hysterectomy and bilateral salpingo-oophorectomy with adjuvant chemo-radiotherapy and brachytherapy. Her recovery had been further complicated by the development of radiation cystitis, small bowel resection, and short gut syndrome. The strictures had previously been managed with bilateral double J stents, which have now failed. A left-sided nephrostomy insertion was performed. A CT-scan prompted by a repeat deterioration in renal function revealed a right-sided hydronephrosis and suggested transcolic passage of the previously placed left-sided nephrostomy. She had remained clinically well and apyretic. A right-sided nephrostomy was inserted, and the left re-sited following a colonoscopy-guided nephrostomy removal and clip occlusion of the nephrostomy tract. The patient then developed a pseudoaneurysm, which was managed with embolization. The patient was able to return home with corrected kidney function. The three-monthly checks for changes in nephrostomy catheters did not reveal any complications. Conclusions: The patient therefore presented with two major complications of nephrostomy placement: colonic perforation and pseudoaneurysm, classified as Clavien IIIb and IIIa, respectively. Multidisciplinary management, including urologists, gastroenterologists, general surgeons, and radiologists, enabled a minimally invasive management. Minimally invasive management with endoscopic clip placement appears to be a safe alternative to directed fistula of colonic perforation.Keywords
Supplementary Material
Supplementary Material FileNephrostomy placement allows percutaneous drainage and/or decompression of the renal cavities when retrograde access via the natural orifice may not be favoured or has failed. Nephrostomy placement is a common image-guided procedure performed under the remit of urologists and/or interventional radiologists worldwide, depending on local service expertise and practice arrangements.1 This procedure may be performed electively or emergently depending on the indication. Access to the lumbar fossa may be achieved in either a prone or supine position and may be guided by either ultrasound, fluoroscopy, or computed tomography (CT)-based imaging.2 A needle trocar is placed within the pelvicalyceal system and then exchanged for a guidewire, over which a final nephrostomy catheter can be placed, with or without serial fascial dilatation.
Procedure-specific complications include iatrogenic placement-related injury to surrounding structures, including liver, spleen, digestive tract, diaphragm, etc., as well as bleeding complications, including major vessel injury and pseudoaneurysm.3 The reported incidence of colonic perforation during nephrostomy placement is 0.2%–0.5%.4 Hemorrhagic complications account for 2.3% of complications.5 We describe in this case the endoscopic and conservative management of two simultaneous complications due to nephrostomy placement.
A 75-year-old female patient with a history of bilateral obstructive uropathy secondary to bilateral radiation-induced ureteric strictures attended for an elective left-sided nephrostomy placement. The patient’s history is notable for a history of endometrial cancer, currently in remission, and treated previously by total hysterectomy and bilateral salpingo-oophorectomy with adjuvant chemo-radiotherapy and brachytherapy. Her recovery had been further complicated by the development of radiation cystitis and small bowel obstruction, treated with small bowel resection, with subsequent development of short gut syndrome. The strictures had previously been managed with bilateral internalized ureteric stents. However, secondary to extrinsic compression from a fibrotic and fixed retroperitoneum, these had failed with the onset of left-sided ipsilateral hydronephrosis and accompanying renal impairment (please see Figure 1A). There was no evidence of disease progression on her most recent surveillance imaging.
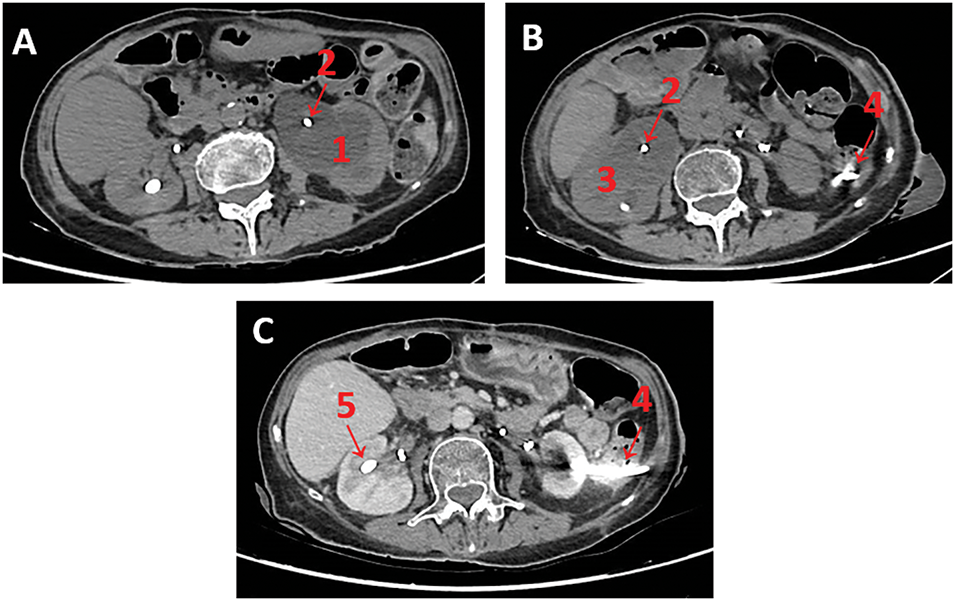
FIGURE 1. Sequential abdominopelvic computed tomography (CT) images. (A) Non-contrast scan showing significant (1) left hydronephrosis and (2) occluded left double J stent; (B) Non-contrast scan raising suspicion for transcolic passage of the (4) left nephrostomy tube; (C) Contrast-enhanced still showing suspicion for transcolic passage of the (4) left nephrostomy tube. Note: (1) left hydronephrosis, (2) occluded double J stent, (3) right hydronephrosis, (4) suspected intracolic trajectory of left nephrostomy, (5) right nephrostomy tube.
On admission, the patient presents as cachectic, with a body mass index (BMI) of only 16 kg/m2. She was not receiving any anticoagulant medication. She required daily total parenteral nutrition to meet nutritional requirements due to his short bowel syndrome.
On routine blood test monitoring with her oncology service recent blood tests had demonstrated the following: a white blood count of 8.02 G/L, a haemaglobin of 9.6 g/dL, a platelet counts of 373 G/L, an elevated serum creatinine of 168 µmol/L, a urea of 26.3 mmol/L, mild elevated potassium at 5.6 mmol/L and an estimated Glomerular Filtration Rate (eGFR) of 25.3 mL/min. Her baseline creatinine and eGFR are 131 µmol/L and 30.3 mL/min, respectively. This acute kidney injury prompted a CT scan that led to her diagnosis with new left-sided hydronephrosis.
In the context of an acute left-sided hydronephrosis and accompanying acute kidney injury, despite appropriate positioning of a left-sided ureteric stent, a left nephrostomy insertion was recommended in order to preserve renal function and reduce the likelihood of obstructive urosepsis and symptomatic hydronephrosis. The hypothesis of an obstructive cause was initially retained due to the improvement in renal function after each change of double J stents and the high probability of retroperitoneal fibrosis induced by the patient’s history of radiotherapy. The procedure was carried out without initial complications and performed under full general anaesthesia with prophylactic intravenous antibiotics on induction, in a modified supine position under ultrasound and fluoroscopic guidance, using a single puncture targeting the posterior calyx. A Kolibri kit from Coloplast (Porges, France) was used. An 18 Fr Chiba needle was placed, allowing easy puncture of a dilated posterior calyx. This immediately drained clear urine, and a guidewire (straight tip, Terumo) was placed. A skin incision was extended, and fascial dilators were used to allow serial dilation prior to introduction of a 12 Fr nephrostomy tube over the guidewire. The correct intrarenal position was confirmed via a confirmatory nephrogram. The nephrostomy was secured using 2 points of Mersuture 1, and the patient was observed for a routine 4–6 h post-procedure. Her post-procedure recovery was uncomplicated, and she remained afebrile. Day one post-procedure left-sided urine output was clear (1200 mL), serum creatinine had improved to 147 µmol/L, and 3 days after, serum creatinine was 131 µmol/L. The patient was discharged home once recovery in renal function had recovered to baseline.
Three weeks later, her renal function again started to deteriorate despite an adequately draining nephrostomy with simultaneous oliguria via the bladder. This prompted a non-contrast abdomino-pelvic CT-scan, which revealed interim development of contralateral right-sided hydronephrosis despite appropriate ureteric stent placement. Also reported was an unexpected finding of a possible trans-colic passage of the previously placed left nephrostomy (please see Figure 1B).
The patient had remained otherwise well and asymptomatic, denying any abdominal discomfort or bowel habit altered from baseline (gastrointestinal symptoms at baseline included diarrhoea, requiring transit slowing agents, see usual treatments). She was apyretic, normotensive, and had a resting pulse rate of 82 beats per minute. No abdominal tenderness or signs of peritonism were elicited on abdominal examination. A right nephrostomy was inserted in the first instance without any immediate complications. Management of a possible left-sided colonic perforation was deferred to allow recovery of renal function, given the patient’s clinical stability.
An abdominopelvic CT scan was repeated with intravenous contrast. The diagnosis is not straightforward due to artefacts caused by the nephrostomy tube. In fact, our radiologists are unable to give a definitive diagnosis of whether a nephrostomy tube is trans-colic, sub-serosal, or in the pericolic fat (please see Figure 1C).
Despite multidisciplinary input from the general surgery team, gastroenterology and radiology colleagues, there remained ambiguity in terms of a definite diagnosis of iatrogenic colonic perforation based on imaging and clinical picture alone. As a primary diagnostic intervention, a colonoscopy was arranged, with a view to a possible therapeutic intervention if confirmed. The patient was counselled regarding the possibility of uncontained colonic perforation and potential need to convert to a laparoscopic +/– open bowel repair, as well as the potential need to create a controlled fistula, either as an intended management strategy or a persistent complication. The risk of potentially exacerbating a colonic perforation with air insufflation during colonoscopy was explained to the patient, as well as the potential need for conversion to a laparoscopic and/or open procedure.
The patient received 2 L of polyethyleneglycol as bowel preparation. The procedure was conducted by a gastroenterologist under general anesthetic to allow for conversion to a laparoscopic or open procedure if required, with prophylactic intravenous antibiotics on induction. The 12 Fr nephrostomy tube was clearly visualized transversing the descending colon as illustrated. Two endoscopic clips (Resolution ClipTM, Boston Scientific, Boston, MA, USA) were applied to both the entry point and exit point, and the nephrostomy was completely retracted. Full-thickness closure was achieved, and non-prophylactic clips were placed (please see Figure 2).
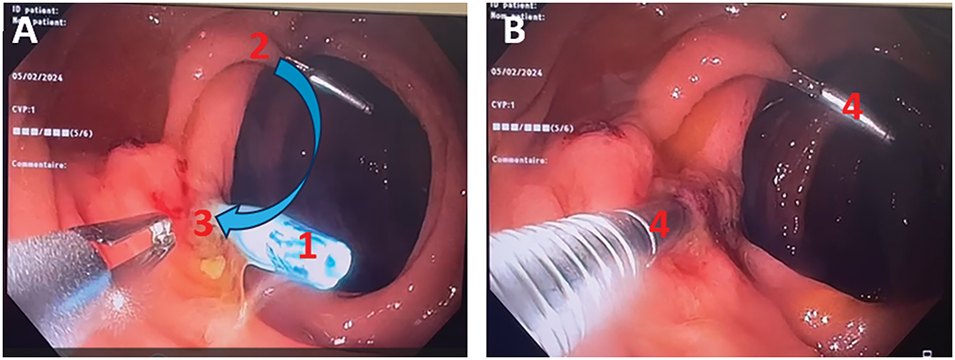
FIGURE 2. Endoscopic view of clip placement on the entry and exit points of the nephrostomy tube, which passes intracolonically, with adequate bowel preparation. (A) Endoscopic view of nephrostomy tube removal; (B) Endoscopic view of endoscopic clip allowing closure of the colonic fistula. Note: (1) nephrostomy tube, (2) entry point, (3) exit point, (4) metallic clip, blue arrow: nephrostomy catheter trajectory.
The patient’s postoperative recovery was uncomplicated: she had no fever, no pain, and no gastrointestinal symptoms (bar baseline symptoms relating to her small bowel syndrome). A left nephrostomy was reinserted after a few days, once dilation of the left cavities could be confirmed. This puncture was more challenging and required 2 attempts to the lower pole calyx.
On day 1 of insertion of the new left nephrostomy tube, the patient developed visible haematuria. The patient was haemodynamically stable, and no blood loss was found. An arterial phase contrast-enhanced abdominopelvic CT-scan revealed a left inferior polar false aneurysm associated with extensive clotting of the cavities, likely related to the first renal puncture-dilatation (please see Figure 3A,B).
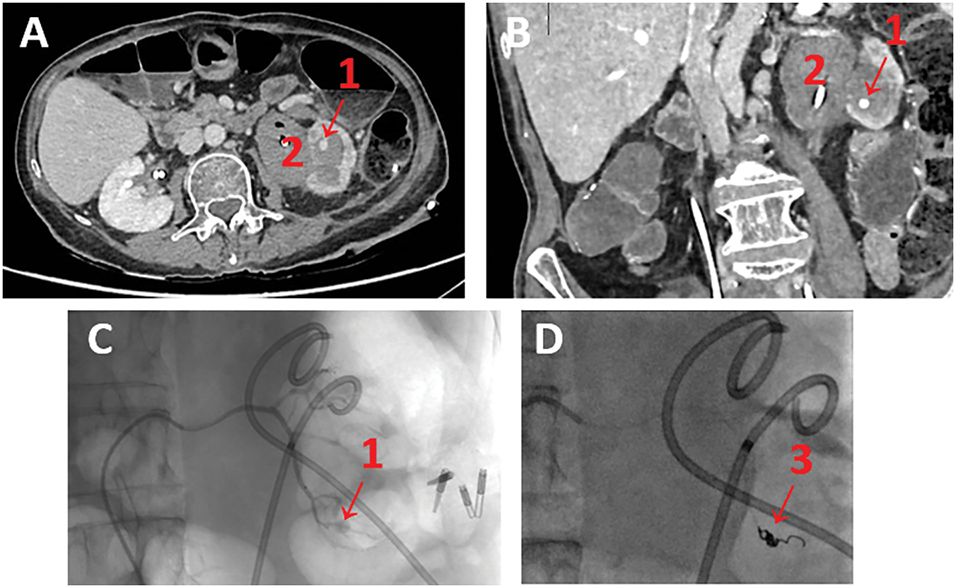
FIGURE 3. Visualisation of pseudoaneurysm. (A) Injected abdominopelvic CT-scan showing left inferior polar arterial pseudoaneurysm—axial view; (B) Injected abdominopelvic CT-scan showing left inferior polar arterial pseudoaneurysm—coronal view; (C,D) Intraoperative arteriography of left renal false aneurysm embolization. Note: (1) arterial pseudoaneurysm, (2) extensive clotting of the cavities, (3) Coil.
Super-elective radioembolization was performed by the interventional radiologists on the same day without any immediate complications (please see Figure 3C and D).
The patient was finally discharged with clear urine and good renal function on day 5. Renal function was satisfactory at 3 months, with creatinine at 129 µmol/L.
It should be noted that the double J tubes were removed during the consultation when the nephrostomy tubes were changed for the first time.
Our patient developed two major complications based on Clavien-Dindo classification following nephrostomy placement: colonic perforation and renal pseudoaneurysm (Clavien IIIb and IIIa, respectively). It should be noted that a colonic perforation following a nephrostomy may sometimes be associated with a Clavien IV complication. Both were managed in a minimally invasive fashion.
The rate of colonic perforation during percutaneous nephrolithotomy is between 0.2 and 0.8%.4 Within the literature, risk factors for colonic perforation during nephrostomy include anatomical variations (such as a retro-renal colon or horseshoe-shaped kidney), modified supine position, reduced retroperitoneal fat, left side, kyphoscoliosis, and previous colonic surgery.6 Our patient had several pre-procedure risk factors for colonic perforation, including previous laparotomy, low BMI, and left modified supine position. The abdominopelvic CT-scan performed prior to the operation is particularly important in guiding the surgical procedure.7
A 2020 meta-analysis found no difference in the choice of puncture technique (ultrasound and/or fluoroscopy). Instead, the choice should depend on the experience of the surgeon and the patient.2 In our case, the surgeon performed an ultrasound-guided puncture with fluoroscopic control.
Traditional teaching in relation to colonic perforation detected following nephrostomy insertion is to favour staged withdrawal of the nephrostomy tube in order to create a controlled colonic fistula, with deferred removal of the nephrostomy tube after a few days. This should be combined with a urethral catheter, a residue-free diet, and broad-spectrum antibiotics.8
In our case, our patient was diagnosed with a colonic perforation incidentally on an abdominopelvic CT scan. In our patient’s case, we decided not to start antibiotic therapy due to the absence of any symptoms, a negative pre-procedure urine culture, and normal inflammatory markers. This decision was the subject of multidisciplinary consultation. This excludes antibiotic prophylaxis recommended prior to various surgical procedures. Given the patient’s pre-existing abdominal pathology, there was no need to tailor the diet as she was already adhering to a low-residue diet.
The multidisciplinary management allowed complete conservative management, without leaving any drainage after the procedure (no retroperitoneal drain to control the digestive fistula) because the fistula could be closed straight away by endoscopy. The closure of iatrogenic colonic perforation is clearly described in the literature, allowing surgery to be avoided as a first-line treatment.9
There is one other reported case within the literature of primary fistula closure without using a retroperitoneal drain.10 Contrary to our records, the patient was not stable at the time of the colonoscopy, which also revealed active bleeding at the site of the colonic perforation. The technique of clipping the perforation has not been developed. It should be noted that the patient had a history of colon resection for locally recurrent colon cancer, particularly at the colonic anastomosis, which is why the colonoscopy had been initially scheduled. Our case is the first in a totally asymptomatic patient.
Bleeding complications during nephrostomy tube insertion are common (26%),5 but can range from benign to potentially serious arterial bleeding. Arterial bleeds require additional management by embolization or surgery. Risk factors described for bleeding during nephrostomy tube insertion include the use of anticoagulants or antiplatelet agents, multiple renal punctures, the size of the nephrostomy, renal parenchymal atrophy, and diabetes. Our patient presented a risk factor: multiple renal punctures. Most of these complications can be controlled by embolization,5 but a nephrectomy may still be required, particularly in a centre without an interventional radiology service.
In our case, multidisciplinary management of the patient (urologists, radiologists, gastroenterologists, general surgeons, and interventional radiology) enabled a minimally invasive management of these 2 major complications (Clavien III) following nephrostomy tube placement.
The insertion of a nephrostomy tube is not a trivial procedure, and it is essential that both urologists, interventional radiologists, and a wider multidisciplinary team are aware of these complications and the potential management strategies in each case. The management of these complications must be multidisciplinary in order to optimise the care of our patients. A minimally invasive approach may be feasible and should be considered.
Acknowledgement
We would like to express our sincere gratitude to our patient for agreeing to the publication of this unique case. We would also like to thank those involved in the patient’s care who are not mentioned in the article.
Funding Statement
The authors received no specific funding for this study.
Author Contributions
All authors contributed to this case study. Conceptualization: Marie-Lou Letouche, Steeve Doizi and Olivier Traxer; Writing—original draft preparation: Marie-Lou Letouche; Writing—review and editing: Marie-Lou Letouche, Aideen Madden and Mariela Corrales; Funding acquisition: None; Supervision: Olivier Traxer. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials
Our patient’s medical data is collected in the patient file available at our hospital.
Ethics Approval
We did not consult an ethics committee as the care provided to the patient was part of standard care.
Informed Consent
The patient gave its written informed consent for publication of this case report and accompanying images.
Conflicts of Interest
Olivier Traxer has declared as a consultant for Karl Storz, Coloplast, IPG photonics, Ambu, Quanta System and Rocamed. Aideen Madden is a consultant for Coloplast. All other authors declare no conflicts of interest to report regarding the present study.
Supplementary Materials: The supplementary material is available online at https://www.techscience.com/doi/10.32604/cju.2025.067253/s1.
References
1. Ghoulian J, Nourian A, Dalimov Z, Ghiraldi EM, Friedlander JI. Percutaneous nephrolithotomy access: a meta-analysis comparing access by urologist vs radiologist. J Endourol 2023;37(1):8–14. [Google Scholar] [PubMed]
2. Corrales M, Doizi S, Barghouthy Y, Kamkoum H, Somani B, Traxer O. Ultrasound or fluoroscopy for percutaneous nephrolithotomy access, is there really a difference? A review of literature. J Endourol 2021;35(3):241–248. [Google Scholar] [PubMed]
3. Turo R, Horsu S, Broome J et al. Complications of percutaneous nephrostomy in a district general hospital. Turk J Urol 2018;44(6):478–483. [Google Scholar] [PubMed]
4. Öztürk H. Treatment of colonic injury during percutaneous nephrolithotomy. Rev Urol 2015;17(3):194–201. [Google Scholar]
5. Ramchandani P, Cardella JF, Grassi CJ et al. Quality improvement guidelines for percutaneous nephrostomy. J Vasc Interv Radiol 2003;14(9 Pt 2):S277–S281. [Google Scholar] [PubMed]
6. Hur KJ, Moon HW, Kang SM, Kim KS, Choi YS, Cho H. Incidence of posterolateral and retrorenal colon in supine and prone position in percutaneous nephrolithotomy. Urolithiasis 2021;49(6):585–590. [Google Scholar] [PubMed]
7. Savin Z, Nevo A, Sofer M. Bowel perforation during percutaneous nephrolithotomy: diagnosis, management, and follow-up. Eur Urol Open Sci 2024;66:1–4. [Google Scholar] [PubMed]
8. Öztürk H. Gastrointestinal system complications in percutaneous nephrolithotomy: a systematic review. J Endourol 2014;28(11):1256–1267. [Google Scholar]
9. Cho SB, Lee WS, Joo YE et al. Therapeutic options for iatrogenic colon perforation: feasibility of endoscopic clip closure and predictors of the need for early surgery. Surg Endosc 2012;26(2):473–479. [Google Scholar] [PubMed]
10. Barros PL, Oliveira B, Pereira R, Dores M, Coutinho A, Peyroteo I. A possible novel approach to small iatrogenic colonic perforation during nephrostomy placement. Urol Case Rep 2023;50(4):102495. [Google Scholar] [PubMed]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools