 Open Access
Open Access
CASE REPORT
Double blind-ending ureter: diagnostic challenges and robotic-assisted surgical management—case report
1 AOU Pediatric Surgery Department, Meyer Children’s Hospital IRCCS, Florence, 50139, Italy
2 Alma Mater Studiorum, University of Bologna, Bologna, 40126, Italy
3 Pediatric Surgery Department, IRCCS Azienda Ospedaliero, Universitaria Di Bologna, Bologna, 40138, Italy
4 Pediatric and Adult CardioThoracic and Vascular, Oncohematologic and Emergency Radiology, Unit IRCCS Azienda Ospedaliero, Universitaria Di Bologna, Bologna, 40138, Italy
5 Department of NEUROFARBA, University of Florence, Florence, 50134, Italy
6 Department of Urology, Careggi Hospital, Florence, 50134, Italy
* Corresponding Authors: Marco Di Mitri. Email: ,
Canadian Journal of Urology 2026, 33(1), 185-192. https://doi.org/10.32604/cju.2025.067303
Received 29 April 2025; Accepted 18 September 2025; Issue published 28 February 2026
Abstract
Background: Double blind-ending ureter (DBU) is an extremely rare congenital anomaly involving a duplicated ureter with no connection to the renal pelvis or bladder, making diagnosis difficult. Case Description: A 10-year-old girl presented with recurrent abdominal pain and ultrasound evidence of left hydroureteronephrosis. Magnetic resonance imaging (MRI) and three-dimensional (3D) reconstruction revealed a 30 cm blind-ending ureter. Robotic-assisted excision (Da Vinci Xi) was performed safely, preserving adjacent structures. Histology confirmed a nonfunctional ureteral remnant. Conclusions: DBU is a rare duplication variant. Advanced imaging and robotic surgery are essential for accurate diagnosis and effective, minimally invasive treatment.Keywords
Supplementary Material
Supplementary Material FileThe development of the urinary tract, including the kidneys and ureters, begins early in fetal life through a complex process of differentiation and migration. The ureteric bud (UB), an outgrowth from the mesonephric duct, plays a critical role in forming the renal collecting system. Around the fifth week of gestation, the UB interacts with the metanephric mesenchyme, initiating the formation of the renal pelvis, calyces and ureter. In addition to its branching role, the ureteric bud must undergo proper elongation and migration to establish normal anatomical positioning. The interaction between the UB and the cloaca is essential for establishing the ureterovesical junction. Any failure in this process may result in high ureteral insertions, ectopia, or in rare cases, the absence of insertion entirely.1 This critical interaction involves a cascade of molecular signals—most notably the GDNF/RET pathway, as well as BMP4 and WNT11—which guide the branching morphogenesis of the UB and the differentiation of the surrounding mesenchyme into nephrons. Precise regulation of these pathways is essential for the correct anatomical and functional organization of the urinary tract.2 The ureteric bud undergoes a series of branching events to form the renal pelvis, calyces, and collecting ducts. It simultaneously elongates caudally to connect with the urogenital sinus, forming the future bladder insertion. During this process, the ureter becomes invested in the developing bladder wall and eventually forms a muscular orifice through which urine will drain. Any disturbance during this process—whether in the timing of the ureteric bud formation, its branching, or its interaction with the metanephric mesenchyme—can lead to a wide spectrum of anomalies. While ureteral duplication is relatively common, the formation of a blind-ending ureter, or even more rarely, a double blind-ending ureter, represents a significant deviation from typical development. Incomplete apoptosis or failure of canalization may contribute to the persistence of these non-functional structures.3 Any disturbance during this process—whether in the timing of the ureteric bud formation, its branching, or its interaction with the metanephric mesenchyme—can lead to a wide spectrum of anomalies collectively referred to as congenital anomalies of the kidney and urinary tract (CAKUT).1 These anomalies account for a significant proportion of prenatal urinary tract abnormalities and are often diagnosed via antenatal ultrasound. While some defects such as mild hydronephrosis are transient and benign, others—including duplication anomalies or non-functioning ureteral remnants—may lead to clinical symptoms requiring surgical management.4 Among these, ureteral duplication (UD) is relatively common and is observed in approximately 0.8% of the population, with a higher incidence in females. UD typically arises due to an aberrant splitting of the UB before it reaches the metanephric blastema or the formation of two separate ureteric buds. This can lead to complete duplication, in which both ureters enter the bladder separately, or incomplete duplication, where the ureters fuse before insertion into the bladder. Notably, duplicated ureters may be associated with a duplicated renal pelvis or may arise independently from a single renal unit. In some cases, the duplication is limited to the ureter alone, with a solitary renal parenchyma—suggesting a complex embryologic miscommunication between the UB and nephrogenic tissue.5 In extremely rare cases, a duplicated ureter has one branch that ends blindly, not connecting to the renal pelvis or bladder—a condition called blind-ending ureter. This occurs when one limb of the bifurcated UB fails to induce nephron formation or loses connection with the bladder. Incomplete apoptosis or failure of canalization may contribute to the persistence of these non-functional structures. These blind-ending segments can progressively dilate due to accumulation of sterile fluid, resulting in mass effect, discomfort, or diagnostic confusion with other retroperitoneal or pelvic cystic lesions. Even more exceptionally, both ends of the duplicated ureter may terminate blindly. This anomaly, termed a double blind-ending ureter (DBU), is characterized by a tubular remnant lacking anatomical communication with both the renal pelvis and bladder. DBU is an embryological curiosity and remains exceedingly rare in the literature, with only a handful of cases described in both pediatric and adult populations. Its pathogenesis likely involves premature division of the ureteric bud combined with a failure of proper integration into the renal and vesical systems. Because these anomalies may remain asymptomatic or mimic other conditions, they are often discovered incidentally or during imaging for unrelated symptoms.6 Their imaging appearance can mimic that of paraovarian cysts, hydrosalpinx, or lymphatic malformations, especially when they lack peristalsis or urinary tract communication. Advanced imaging techniques such as MRI and 3D reconstruction have become invaluable in differentiating these anomalies preoperatively and guiding surgical planning. The aim of this paper is to describe an extremely rare presentation of a pediatric patient with a DBU. We detail the clinical findings, radiological workup, the role of three-dimensional reconstruction in preoperative planning, and the successful management using a minimally invasive robotic-assisted surgical approach.7,8
UD arises due to an aberrant splitting of the UB before it reaches the metanephric blastema. In extremely rare cases a duplicated ureter has one branch that ends blindly, not connecting to the renal pelvis or bladder, a condition called blind-ending ureter. This condition arises from premature division of the ureteric bud during embryonic development, leading to a bifurcation where one limb is a nonfunctional pouch.3
The term “double blind-ending ureter (DBU) anomaly” refers to an exceptionally rare congenital condition where a duplicated ureter features two blind-ending extremities lacking connections to the renal pelvis and bladder, respectively. Due to the rarity, this anomaly is scarcely documented in medical literature.6,9 The aim of this paper is to describe an extremely rare presentation of a patient with a DBU.
Ethics committee approval was not required for the publication of this case report at our institution. Written informed consent for publication was obtained from the patient and their legal guardians. This report was prepared in accordance with the CARE case report guidelines, and a completed CARE checklist is provided. Please see Supplementary Materials for more details.
A 10-year-old female, with normal growth parameters (body mass index (BMI) 18.1 kg/m²), suffering from recurrent episodes of poorly localized lower abdominal pain over the previous 6 months, with a frequency of 1–2 times per week. Pain episodes were intermittent, not associated with urinary symptoms, and partially relieved by rest or antispasmodics. Her medical history was otherwise unremarkable, with no known urinary tract infections or prior abdominal surgeries.
She came to our attention, at IRCCS Meyer Children’s Hospital, Florence, Italy, with an incidental ultrasound (US) picture of left hydroureteronephrosis. The US showed a renal pelvis (measuring 2 cm) with a tortuous ureter, together with a para-bladder cystic formation of uncertain nature measuring 5–6 cm. Physical examination was not significant. To further evaluate these findings, we performed an abdominal magnetic resonance imaging (MRI), which reported: “A large, fluid-filled tubular structure is present in the left retroperitoneal region, running alongside the left ureter. It extends about 30 cm from the compressed left renal pelvis to the pelvic cavity, following a tortuous path with variable caliber. A saccular dilation (7 × 6 cm) is seen at the lumbosacral transition before it narrows and extends toward the left ovary. There is no clear connection to the renal calyces or bladder, and it remains non-opacified in urographic imaging, indicating it is a non-functioning, dilated ureteral remnant likely from a regressed renal unit.” (Figure 1).
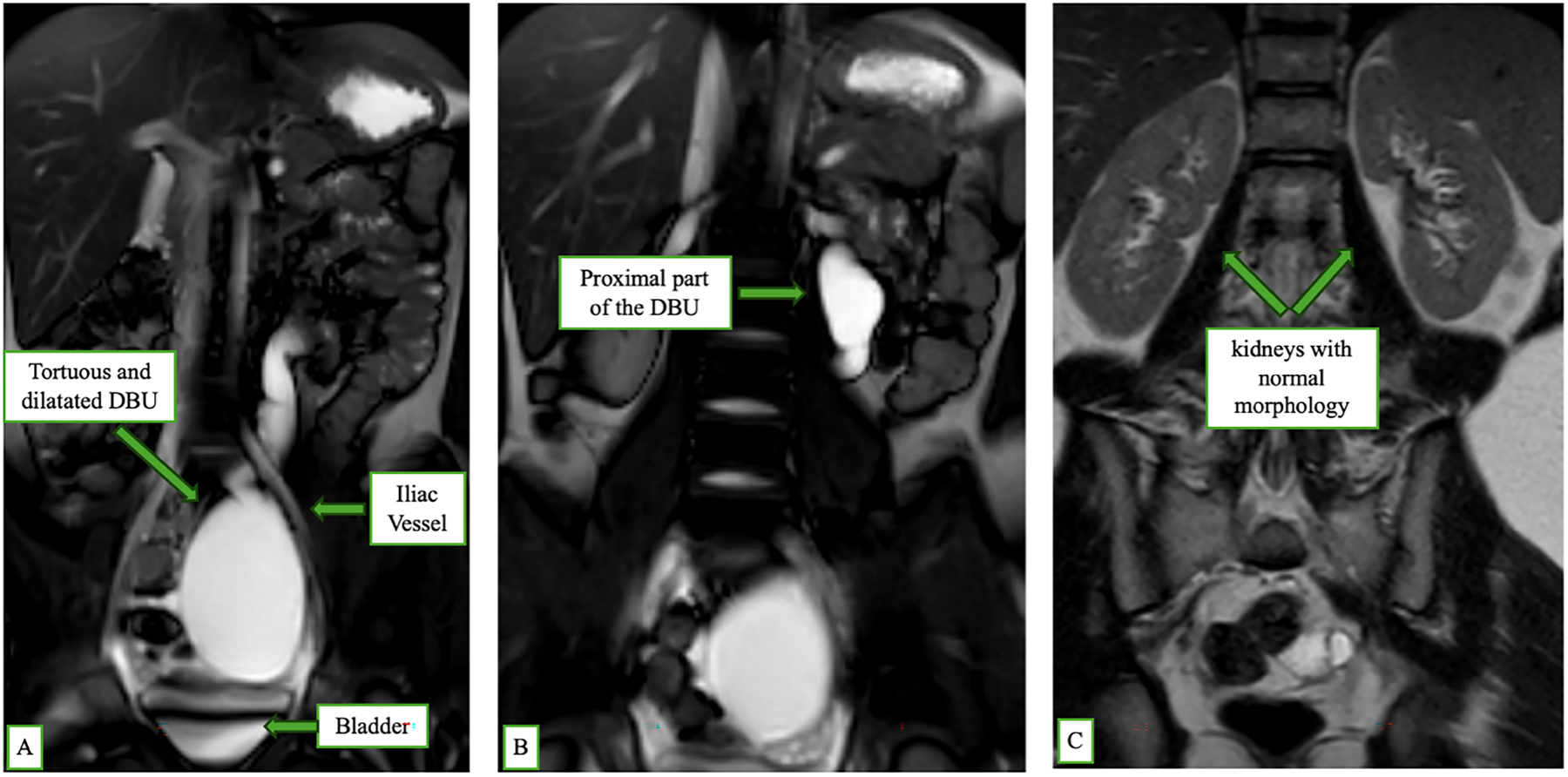
FIGURE 1. Magnetic resonance imaging (MRI) coronal scan showing features of double blind-ending ureter (DBU): (A) A tortuous and dilated DBU running parallel to the normal left ureter, located anterior to the iliac vessel and bladder. (B) Proximal portion of the DBU without connection to the renal pelvis. (C) Bilateral kidneys with normal morphology and no evidence of hydronephrosis
Based on clinical presentation and imaging findings, we suggested surgical removal of the duplicated ureter due to recurring abdominal pain and to avoid the risk of infection or rupture.
Before surgery, we requested Medical-Note Srl to perform a tridimensional (3D) reconstruction of the malformation, to have a clearer view of its anatomy before surgery (Figure 2). The 3D reconstruction highlighted as the dilatated ureter was not connecting to both the renal pelvis and bladder, resulting in a double-blind ending ureter (DBU). Furthermore, by the 3D reconstruction, we had the opportunity to know exactly the relationships between the DBU, the ureter and the vessels.
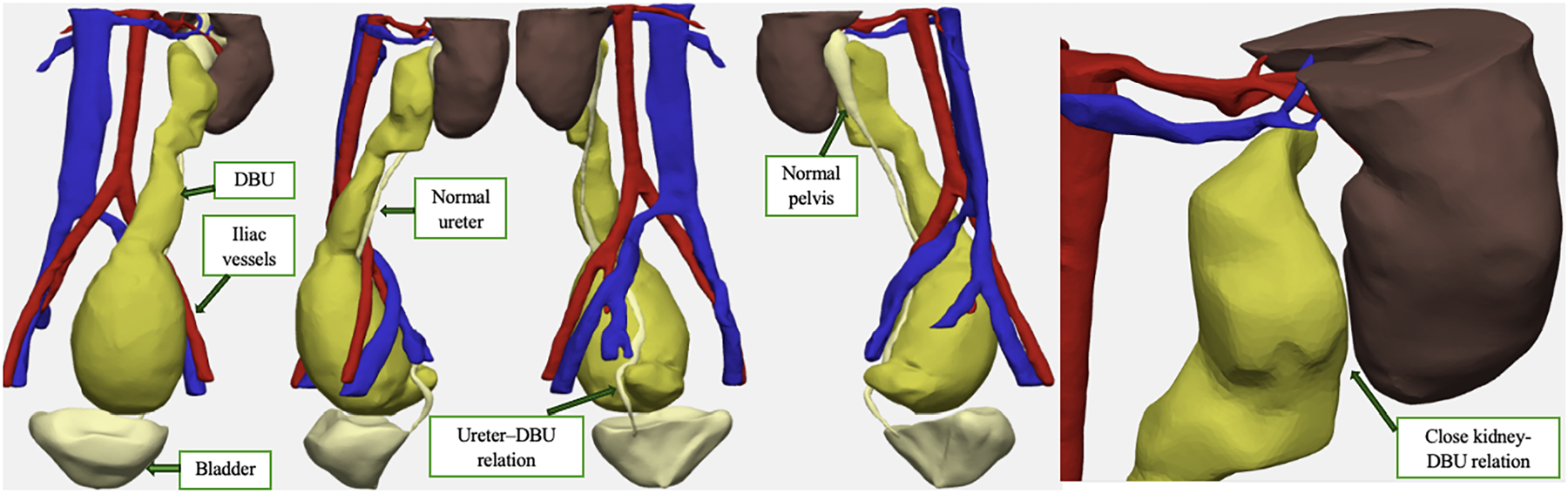
FIGURE 2. 3D MRI reconstruction of the case, with clear and precise visualization of the anatomical relationships among the excluded ureter and the kidney, ureter, bladder and vascular axis
From a diagnostic standpoint, both MRI and 3D reconstruction confirmed the presence of a single, non-bifid renal pelvis on the left side, with no evidence of a duplicated collecting system. The duplicated ureter originated independently, without association with a bifid pelvis, further emphasizing the anomaly of a DBU without upper tract duplication.
The patient underwent a complete excision of the duplex ureter by a robotic-assisted approach (Da Vinci Xi). The patient was placed in a supine position under general anesthesia, with a slight Trendelenburg tilt. Pneumoperitoneum was achieved via an open umbilical access, through which a camera port was inserted. Two additional robotic trocars (8 mm) were placed in the epigastrium and hypogastrium. A 5 mm assistant port was placed in the left iliac fossa. The Da Vinci Xi system was docked on the patient’s left side.
The dilated ureteral remnant was identified in the left retroperitoneum. Dissection began proximally near the renal hilum, where a fibrous attachment to the renal capsule was isolated and divided. The duplicated ureter was meticulously followed distally, with care taken to preserve the native ureter and surrounding vessels. The distal end of the DBU terminated blindly near the bladder but without mucosal communication. The remnant was dissected free along its entire course and extracted through the umbilical port. Total operative time was 60 min, with negligible blood loss. No intraoperative complications occurred (Figure 3). A sample of the fluid contained within the dilated ureter was collected and analyzed, confirming the presence of sterile urine. The post-operative period was uneventful, and the patient was discharged in the second post-operative day. Histological examination of the excised ureter described “a 15 cm long ureteral segment with wall dilation up to 3 cm in diameter. Diagnosis: ureteral wall with normal epithelial lining, subepithelial fibrosis, and muscular layer hypotrophy” (Figure 4). At the three-month follow-up, the patient was asymptomatic. A postoperative abdominal ultrasound confirmed normal kidney and bladder morphology, with no evidence of hydronephrosis or residual ureteral dilation, supporting the success of the surgical excision.
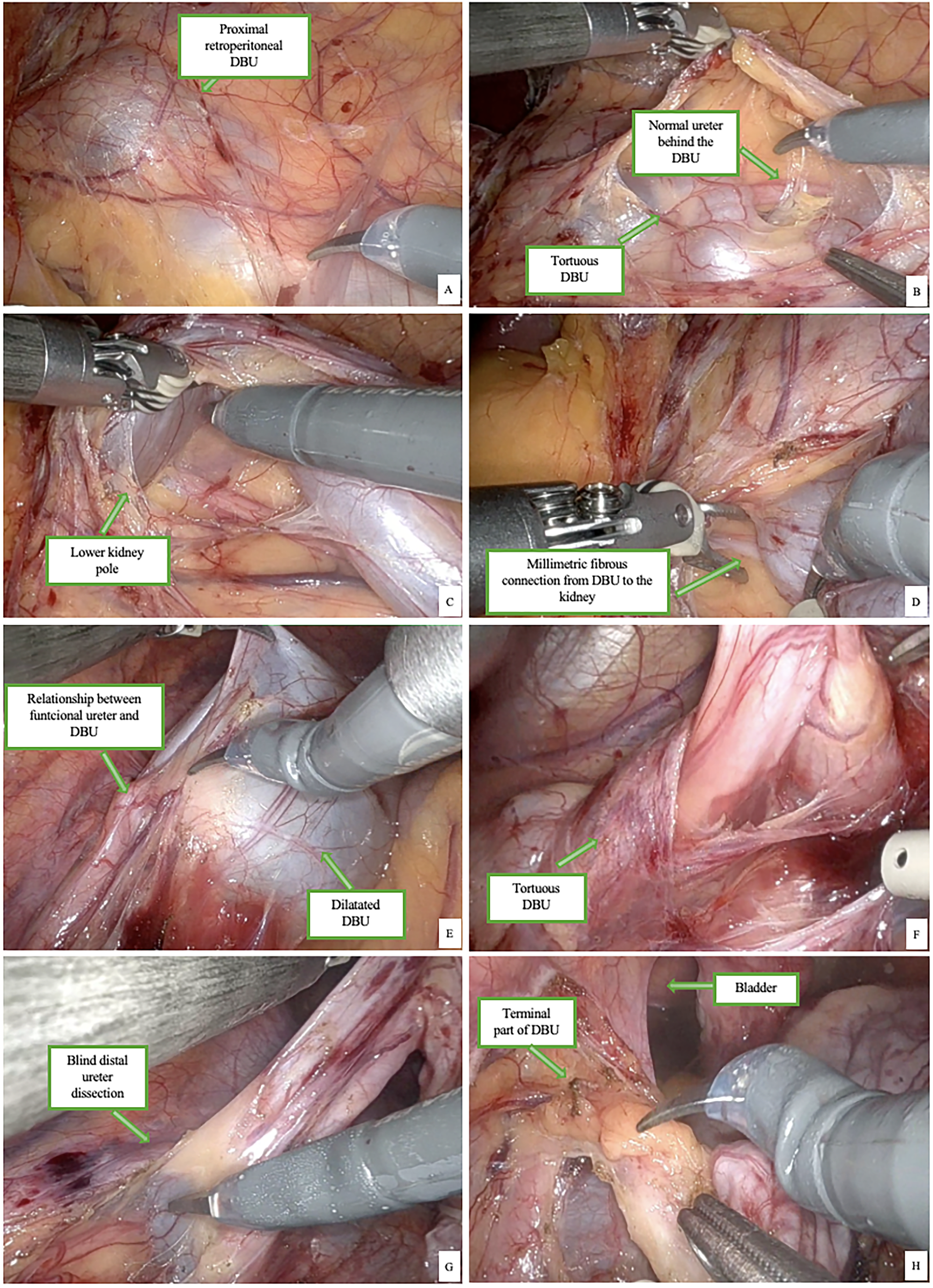
FIGURE 3. Proximal part of the left DBU (A), relationship with normal ureter (B) and kidney (C) and the proximal remaining fibrous tissue (D). The distal part of the dilatated (E) and tortuous DBU and its relationship with the ureter (F). Distal dissection of the DBU (G) and its relationship with the bladder (H)
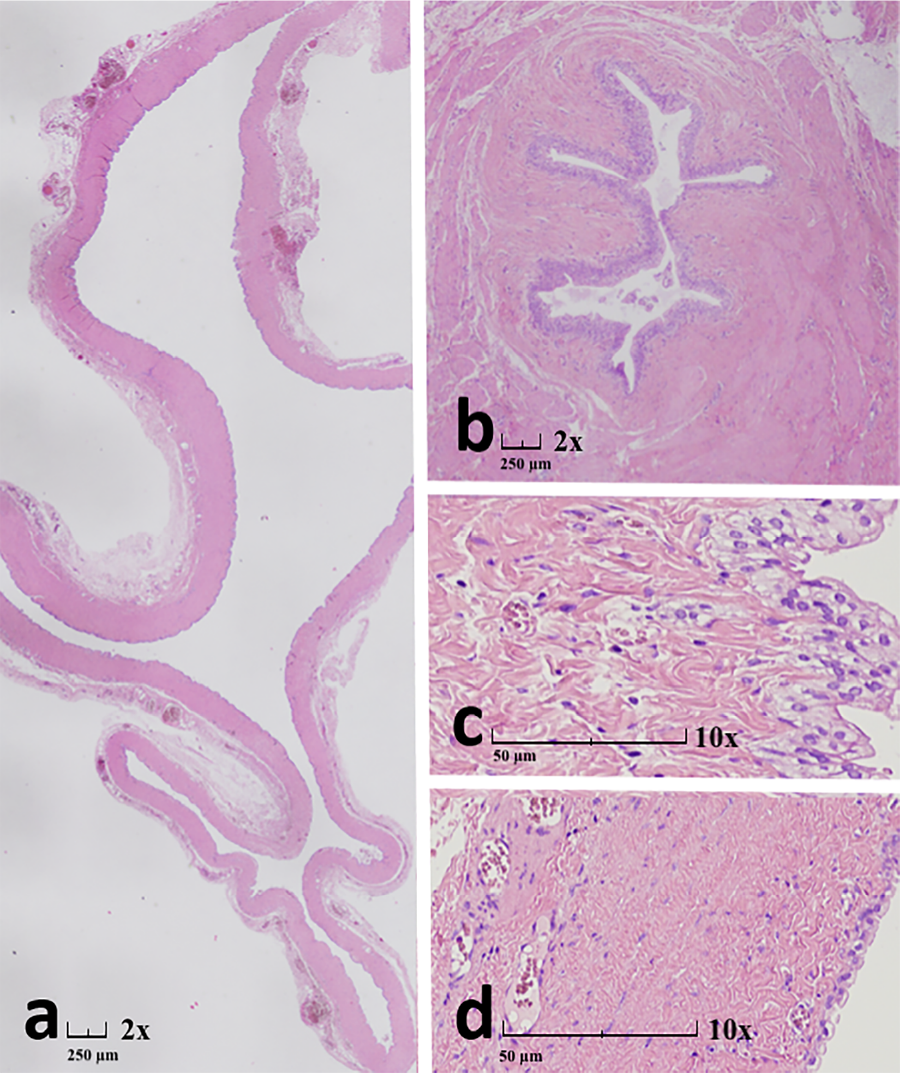
FIGURE 4. The dilated ureter (a) exhibited a flattened urothelium of variable but predominantly marked severity, accompanied by fibrosis of the ureteral wall (c–d). In contrast, the undilated ureter (b) showed preserved urothelial architecture, also with fibrosis of the ureteral wall. Hematoxylin and Eosin staining (a–d); original magnification: a–b 2×, c–d 10×
DBU is an extraordinary, rare anomaly of the urinary system. Ureteral duplication occurs in approximately 0.8% of the population and is more commonly in females.9 It results from an abnormal division of the UB during early embryonic development. Complete UD, in which two distinct ureters drain separately into the bladder, is distinguished from incomplete duplication (bifid ureter), where a single ureteral orifice remains at the bladder level.10 Since in most cases UD is associated with a duplicated renal pelvis, the presentation of a double-blind ending ureter with a single renal pelvis, as seen in our patient, represents an exceptionally rare anomaly. Moreover, the absence of connection with both the kidney and the bladder makes this case highly uncommon. Most reported cases of UD involve a fully functioning duplicated renal unit. When the upper pole continuity is non-functional, it is often associated with congenital conditions such as renal dysplasia, ureterocele or ectopic ureteral insertion. The literature describes various complications linked to UD, including vesicoureteral reflux, ureteropelvic junction obstruction, hydronephrosis and recurrent urinary tract infections (UTIs).11 Our case involves a nonfunctioning, markedly dilated ureter with no evident renal connection, and no communication with the bladder, suggesting a regressed or involuted renal unit.
Similar cases of isolated, non-functional ureteral remnants have been described in the context of ectopic ureters or ureteral dysplasia, where the affected segment fails to contribute to the urinary system. However, these cases typically present with persistent urinary leakage or incontinence due to an ectopic insertion into the vagina, urethra or seminal vesicle in males. Our patient did not exhibit such symptoms, further distinguishing this case from classical ectopic ureter presentations. In the diagnostic workup of a cystic tubular structure adjacent to the bladder or ureter, several differential diagnoses should be considered. These include ureteroceles, paraovarian cysts, lymphangiomas, or mesenteric cysts, which may appear similar on imaging. A dilated blind-ending ureteral remnant can also be mistaken for a hydrosalpinx or Müllerian duct remnant in female patients. Differentiation often relies on detailed imaging, particularly MRI or CT urography, and correlation with anatomical landmarks. The absence of communication with the urinary tract and lack of enhancement or peristalsis are distinguishing features of a non-functioning ureteral segment.
A few case reports have documented non-functioning ureteral remnants, often associated with Meyer-Weigert law, which describes how, in cases of UD, the upper pole continuity drains ectopically while the lower pole continuity maintains a normal insertion.12
Two papers in literature describe similar cases to the one occurred in our unit. Choi et al. reported two cases of DBU in which both ureters terminated blindly without any communication with the urinary system. Their cases involved adult patients and imaging studies revealed cystic, fluid-filled tubular structures extending along the ureteral path. In both instances, surgical excision confirmed the presence of blind-ending ureters with no functional renal contribution or bladder insertion.6 Similarly to these two cases, Hulett and Ozgur, described a pediatric case of complete ureteral duplication with both superior and inferior blind endings. Also, in this case, the patient underwent to a surgical resection confirming the anomaly, with histopathology showing urothelial lining.9
However, it is important to highlight that not all reported cases of DBU required surgical removal; the decision to operate depends on factors such as symptom severity, risk of infection, and ureteral dilation.
Long-term follow-up in such cases is essential to ensure that no secondary complications arise from surgical manipulation or inadvertent injury to the native urinary system. Postoperative ultrasound and renal function monitoring are advised at regular intervals, especially in pediatric patients with long life expectancy. Although recurrence is unlikely following complete excision, residual fibrosis or adhesions may lead to symptoms or obstruction later in life. Moreover, documentation and publication of such rare cases are crucial to raise awareness and expand the surgical and radiological knowledge base for future clinicians encountering similar anomalies.
In our case, the surgical excision was based on significant dilation, risk of infection, and symptom persistence. Furthermore, our case highlights the advantages of moder three-dimensional (3D) imaging reconstruction which provided a highly detailed and spatially accurate visualization of the duplicated ureter allowing a minimally invasive robotic assisted approach. During surgery, two separate targeting steps were performed: the first was the kidney to isolate the proximal ureteral segment and the second was the bladder to dissect the distal portion of the ureter. A meticulous dissection of the ureter was carried out, ensuring that the functional ureter remained unharmed. The detailed anatomical relationships provided by 3D reconstruction were essential in allowing the surgeon to proceed safely, reducing the risk of injury to the functional urinary structures. Our surgery planning highlights the advantages of integrating 3D imaging into the surgical workflow for rare urological anomalies.13 Furthermore, the robotic approach allowed for enhanced dexterity and visualization, especially useful in pediatric patients with complex anatomical variants. The dissection was performed in two phases: the proximal part near the renal hilum and the distal blind end near the bladder dome. Special attention was paid to preserving the native ureter and vascular structures throughout. The use of robotic wristed instruments facilitated delicate maneuvers in a narrow operative field, which would have been more challenging using standard laparoscopy.
The main strengths of this case include the use of advanced three-dimensional imaging for precise preoperative planning, the successful application of a minimally invasive robotic-assisted approach in a pediatric patient, and comprehensive histopathological confirmation. These factors contributed to an accurate diagnosis, safe dissection, and preservation of normal anatomical structures.
This case highlights the importance of considering DBU in the differential diagnosis of cystic pelvic structures and demonstrates how integration of 3D imaging with robotic-assisted surgery can optimize outcomes in rare congenital urological anomalies. Increased awareness and documentation of similar cases may support the development of future diagnostic and surgical guidelines.
This study presents a single pediatric case of double blind-ending ureter (DBU), which inherently limits the generalizability of the findings. Due to the extreme rarity of this anomaly, no standardized diagnostic or management guidelines exist, making it difficult to draw definitive conclusions about the optimal surgical or imaging approach. Although advanced imaging and robotic-assisted surgery were successfully employed in this case, these technologies may not be readily available in all clinical settings, potentially limiting the reproducibility of this approach. Larger case series and multicenter collaborations are needed to better understand the natural history, optimal imaging strategies, and long-term results of surgical intervention in DBU.
This case represents an exceptionally rare form of UD with a single renal pelvis and a nonfunctioning DUB completely disconnected from the urinary tract. The use of 3D imaging was instrumental in accurately diagnosing the anomaly and guiding a safe robotic-assisted surgical approach. The integration of advanced imaging and minimally invasive techniques ensured precise dissection and preservation of functional structures. Further case reports and studies are necessary to enhance the understanding of such rare anomalies and to refine preoperative planning and surgical strategies for optimal patient outcomes.
Acknowledgement
The authors would like to thank the surgical and radiology teams at Meyer Children’s Hospital IRCCS and IRCCS Azienda Ospedaliero-Universitaria Di Bologna for their support in the diagnosis and treatment of this case.
Funding Statement
This research received no external funding.
Author Contributions
The authors confirm contribution to the paper as follows: Study conception and design: Marco Di Mitri, Edoardo Collautti, Lorenzo Masieri, Mario Lima; Data collection: Cristian Bisanti, Annalisa Di Carmine, Andrea Zulli; Analysis and interpretation of results: Riccardo Coletta, Roberto Lo Piccolo, Alberto Mantovani; Draft manuscript preparation: Marco Di Mitri, Edoardo Collautti, Riccardo Coletta, Michelangelo Baldazzi. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials
All relevant data are included in the manuscript. Additional details or supporting materials are available from the corresponding author upon reasonable request.
Ethics Approval
The study is a single retrospective case report. Informed consent was obtained from the patient’s legal guardians for the publication of this case and related images. Ethical approval was not required according to the institutional guidelines for single-case reporting. The CARE Checklist and informed consent forms are available upon request.
Conflicts of Interest
The authors declare no conflicts of interest to report regarding the present study.
Supplementary Materials: The supplementary material is available online at https://www.techscience.com/doi/10.32604/cju.2025.067303/s1.
References
1. Mahmoud AH, Talaat IM, Tlili A, Hamoudi R. Congenital anomalies of the kidney and urinary tract. Front Med 2024;11:1384676. doi:10.3389/fmed.2024.1384676. [Google Scholar] [PubMed] [CrossRef]
2. Majumdar A, Vainio S, Kispert A, McMahon J, McMahon AP. Wnt11 and Ret/Gdnf pathways cooperate in regulating ureteric branching during metanephric kidney development. Development 2003;130(14):3175–3185. doi:10.1242/dev.00520. [Google Scholar] [PubMed] [CrossRef]
3. Moghul S, Liyanage S, Vijayananda S, Tam M, Kapralos I. Bifid ureter with blind-ending branch: a rare anatomic variant detected during antegrade ureteric stent insertion. Radiol Case Rep 2018;13(6):1199–1202. doi:10.1016/j.radcr.2018.07.009. [Google Scholar] [PubMed] [CrossRef]
4. Çaltek HÖ, Çaltek NÇ, Aras D et al. Prenatal diagnosis and postnatal outcomes of congenital kidney and urinary tract anomalies: results from a tertiary center. BMC Pregn Childbir 2025;25(1):598. doi:10.1186/s12884-025-07723-9. [Google Scholar] [PubMed] [CrossRef]
5. Qiao J, Cohen D, Herzlinger D. The metanephric blastema differentiates into collecting system and nephron epithelia in vitro. Development 1995;121(10):3207–3214. doi:10.1242/dev.121.10.3207. [Google Scholar] [PubMed] [CrossRef]
6. Choi JY, Kim S, Kim S. Double-blind ureteral duplication: report of two cases. Eur Radiol 2002;12(3):S136–S139. doi:10.1007/s00330-002-1592-y. [Google Scholar] [PubMed] [CrossRef]
7. Subramaniam R, Springer A. Ureteral duplication and duplex systems. In: Pediatric surgery. Berlin/Heidelberg, Germany: Springer, 2023. p. 327–338. doi:10.1007/978-3-662-43567-0_201. [Google Scholar] [CrossRef]
8. Chang E, Santillan C, O’Boyle MK. Blind-ending branch of a bifid ureter: multidetector CT imaging findings. Br J Radiol 2011;84(998):e38–e40. doi:10.1259/bjr/15001058. [Google Scholar] [PubMed] [CrossRef]
9. Hulett RL, Ozgur HT. Double blind ureteral duplication. Pediatr Radiol 1997;27(6):561–562. doi:10.1007/s002470050183. [Google Scholar] [PubMed] [CrossRef]
10. Society for Maternal-Fetal Medicine (SMFMHopkins LM. Duplicated collecting system. Am J Obstet Gynecol 2021;225(5):B12–B13. doi:10.1016/j.ajog.2021.06.040. [Google Scholar] [PubMed] [CrossRef]
11. Afshar K, Papanikolaou F, Malek R, Bagli D, Pippi-Salle JL, Khoury A. Vesicoureteral reflux and complete ureteral duplication. Conservative or surgical management? J Urol 2005;173(5):1725–1727. doi:10.1097/01.ju.0000154164.99648.ee. [Google Scholar] [PubMed] [CrossRef]
12. Slaughenhoupt BL, Mitcheson HD, Lee DL. Ureteral duplication with lower pole ectopia to the vas: a case report of an exception to the Weigert-Meyer law. Urology 1997;49(2):269–271. doi:10.1016/s0090-4295(96)00431-1. [Google Scholar] [PubMed] [CrossRef]
13. Di Mitri M, Di Carmine A, D’Antonio S et al. Advancing pediatric surgery: the use of HoloLens 2 for 3D anatomical reconstructions in preoperative planning. Children 2025;12(1):32. doi:10.3390/children12010032. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools