 Open Access
Open Access
REVIEW
Prostate specific membrane antigen (PSMA) as a biomarker in early and localized advanced prostate cancer: a narrative review
1 Department of Surgery, University of Melbourne, Melbourne, VIC 3052, Australia
2 Department of Urology, The Royal Melbourne Hospital, Melbourne, VIC 3052, Australia
3 EJ Whitten Prostate Cancer Research Centre, Epworth Healthcare, Melbourne, VIC 3005, Australia
4 Department of Surgery, Austin Healthcare, Heidelberg, VIC 3084, Australia
5 Epworth Healthcare, Melbourne, VIC 3005, Australia
6 Department of Surgery, Peter MacCallum Cancer Centre, Melbourne, VIC 3052, Australia
* Corresponding Author: Jonathon Carll. Email:
Canadian Journal of Urology 2026, 33(1), 21-34. https://doi.org/10.32604/cju.2025.068908
Received 10 June 2025; Accepted 11 October 2025; Issue published 28 February 2026
Abstract
Prostate-specific membrane antigen (PSMA) is a surface membrane antigen that is highly overexpressed in prostate cancer, with heterogenous expression throughout the natural history of the disease. This has generated significant interest as a potential biomarker for use in early diagnosis and treatment of prostate cancer. We reviewed the literature surrounding PSMA and its current clinical applications in diagnosing and managing early prostate cancer that is confined to the prostate and local lymph nodes. A search on PubMed, Medline, and Web of Science was performed using the following keywords: “PSMA”, “Prostate Specific Membrane Antigen”, “Prostate cancer”, “Biomarker”, “Diagnosis”. We considered all available articles relevant to the topic of PSMA as a biomarker in early prostate cancer when developing this narrative review. Key articles assessing the biology of PSMA, as well as its use as a potential diagnostic and therapeutic target in early prostate cancer, were assessed. The role of PSMA PET as a potential diagnostic and risk stratification tool was assessed. The current use of antibody-drug conjugates and radioligand therapy targeting PSMA was assessed, along with any current evidence to support their use in early prostate cancer. PSMA is heavily expressed throughout the early stages of prostate cancer, and this has significant therapeutic implications. There is a growing body of evidence that shows PSMA PET can play a role in the diagnosis, risk stratification, and prognostication of localised prostate cancer. PSMA-targeted therapies such as Lu-177 currently do not have any proven benefit in treating early prostate cancer; however, this remains an area of ongoing research.Keywords
Prostate cancer is one of the most common cancers worldwide, with an increasing incidence with age amongst men.1 Given the large disease burden worldwide, there has been significant interest in identifying early biomarkers that can allow for screening and early diagnosis of prostate cancer. This led to the development of the serum prostate specific antigen (PSA) test, which has proven useful in decreasing the number of men diagnosed with metastatic and advanced prostate cancer.2 However, population-wide PSA screening for prostate cancer remains a controversial concept,3,4 due to the poor specificity of the test, and uncertain impact on overall mortality.5 Given the imperfection of PSA as a standalone marker of early prostate cancer, there has been significant interest in the development of additional biomarkers and imaging modalities to aid in the detection and diagnosis of early prostate cancer.
One of the more useful markers identified for prostate cancer is prostate specific membrane antigen (PSMA)—a transmembrane protein that is highly expressed in normal prostate epithelium, as well as proximal renal tubules, and to a lesser extent in the small bowel, salivary glands, and brain.6 More importantly, PSMA is highly over-expressed in most prostate cancers—it was first discovered by assessing monoclonal antibodies developed by inoculating mice with a human prostate cancer cell line, LNCaP.7 From when it was first described, this has attracted significant interest for its potential use as a target for immune-based therapies, and molecular imaging of prostate cancer.8,9 However further research has demonstrated that PSMA can also be over-expressed in a wide variety of solid-organ tumours, including renal cell carcinoma, breast, bladder, and colorectal cancer.10 This is largely related to its role in mediating angiogenesis and neovascularization within the tumour.11 Whilst the role PSMA plays in angiogenesis is not fully understood,11 mouse models have suggested that it achieves this by modulating integrin signal transduction, and PSMA inhibition suppresses angiogenesis.12 As such, increases in PSMA expression may transform vascular endothelium into an angiogenic state, providing a survival advantage to solid organ tumours.13
PSMA is a type II transmembrane glycoprotein encoded by the FOLH1 gene. It is also known as glutamate carboxypeptidase II (GCPII) or N-acetyl-l-aspartyl-l-glutamate peptidase I (NAALADase),14 which refers to its biological functions. Outside the prostate, PSMA cleaves terminal glutamate from the neurodipeptide, N-acetyl-aspartyl-glutamate (NAAG) in neuronal synapses. It also catalyzes the cleavage of terminal glutamates from poly- and gamma-glutamate folates present within the small intestine, allowing for uptake of folates via other carriers. Within the prostate, PSMA is most heavily expressed within the secretory cells of the luminal epithelium, particularly in cells that are androgen receptor positive.14 Although its physiological role within the prostate is not well understood, there is a clear association with prostate cancer. This is likely due to its effect on folate metabolism, which could confer a survival benefit in folate-depleted conditions.15 Given the necessity of folate in a variety of cellular processes essential for rapid cell division, this likely applies selection pressure leading to over-expression within cancerous cells.
However, the expression of PSMA throughout the natural history of prostate cancer can be heterogenous and can change with treatment such as androgen deprivation.14 Whilst in the earlier stages of cancer it is usually highly expressed, as the cancer advances, it can lose PSMA expression.14 This is particularly noted in castrate-resistant disease, as well as prostate cancers showing neuroendocrine differentiation.16 Lab-based and pre-clinical studies have demonstrated increased PSMA expression after the treatment with androgen receptor (AR) inhibitors,17,18 suggesting that AR signalling may play a role in suppressing PSMA expression in advanced prostate cancer. However, the role of AR signalling and PSMA expression is a nuanced one and not fully understood.19 Prostate cancers can demonstrate heterogenous expression at similar stages of disease, suggesting factors such as tumour micro-environment play an important role in regulating PSMA expression.20 Ultimately, the regulation of PSMA expression throughout the natural history of prostate cancer is an area of ongoing research, and has clinical implications given the role PSMA has as a target for diagnostic imaging and clinical therapy.
The characterisation and study of PSMA has led to many advances in diagnostics and therapeutics of prostate cancer. Although these advances have been most significant in advanced prostate cancer, there is a growing body of evidence to support their use in characterising and diagnosing early prostate cancers. At the forefront of the clinical utility of PSMA is its use in staging advanced prostate cancers to identify metastatic disease.
Radioligands that bind to PSMA have been used to develop PSMA positron emission tomography (PET) scans, which far exceed conventional imaging in specificity and sensitivity of identifying metastatic disease.21 However, further evidence has started to demonstrate that PSMA-targeted imaging may be useful in earlier prostate cancer. As the use of PSMA PET scans has moved into routine clinical practice, there has been growing interest in assessing their role in the diagnosis and detection of early prostate cancer. There has also been a growing body of evidence to demonstrate that PSMA PET can provide valuable information in risk-stratifying patients with earlier prostate cancer. A five-point PRIMARY score can be applied to intraprostatic lesions that are avid on PSMA PET, which is predictive of the likelihood of clinically significant prostate cancer being present.22 PSMA is also actively under investigation as a potential molecular target to deliver drugs and therapeutics for men with early prostate cancer. This approach has been promising in men with advanced disease, and therapeutics such as radioligand therapy are currently under investigation for use in earlier stages of prostate cancer. Overall, PSMA is an important molecular target that is under investigation for a variety of diagnostic, prognostic, and therapeutic uses in early prostate cancer.
Given the growing body of literature demonstrating the utility of PSMA as a biomarker in prostate cancer, it is important to explore and assess how this may impact clinical practice. The goal of this review is to assess, analyse and critically appraise the evidence regarding the use of PSMA as a molecular target for diagnosis, prognosis and treatment of prostate cancer, with a focus on early and localized advanced prostate cancer.
PSMA in Diagnosing Early Prostate Cancer
Regardless of the complex regulation expression of PSMA in advanced prostate cancer, it is most often highly expressed in early stages of the disease. This has led to the development of the PSMA PET scan, which uses a radiolabelled tracer that binds to the PSMA on the surface of cells, allowing for a PSMA-specific PET scan. This has revolutionised imaging within prostate cancer, as it is far more sensitive and specific for detecting metastatic disease than conventional computed tomography (CT) and bone scan’s.23,24 Recently, emerging evidence has also shown that PSMA PET can have a role in the diagnosis and risk stratification of prostate cancer. The maximum standardised uptake value (SUVmax) of an intraprostatic lesion on PSMA PET is closely correlated with both the presence of prostate cancer and its grade group.25 The PRIMARY trial developed a 5-point scoring system for intra-prostatic lesions that are avid on PSMA PET.22 The PRIMARY scoring system assigns a score from 1–5 for the primary intraprostatic lesion, based on the pattern of uptake, its SUVmax (Table 1). This score correlates strongly with the likelihood of clinically significant prostate cancer being present.22,26,27 The PRIMARY trial demonstrated that combining a PSMA PET with a multiparametric magnetic resonance imaging (mpMRI) prior to prostate biopsy can increase sensitivity and negative predictive value in diagnosing clinically significant prostate cancer.26 The PRIMARY score itself has also proven to be a good tool at detecting clinically significant prostate cancer, with a PSMA PET scan showing equivalent diagnostic accuracy at detecting clinically significant prostate cancer when compared to mpMRI.22 This demonstrates that the utility of PSMA as a target for molecular-based imaging is relevant not only in metastatic disease but also earlier in the disease course in detecting early prostate cancer.
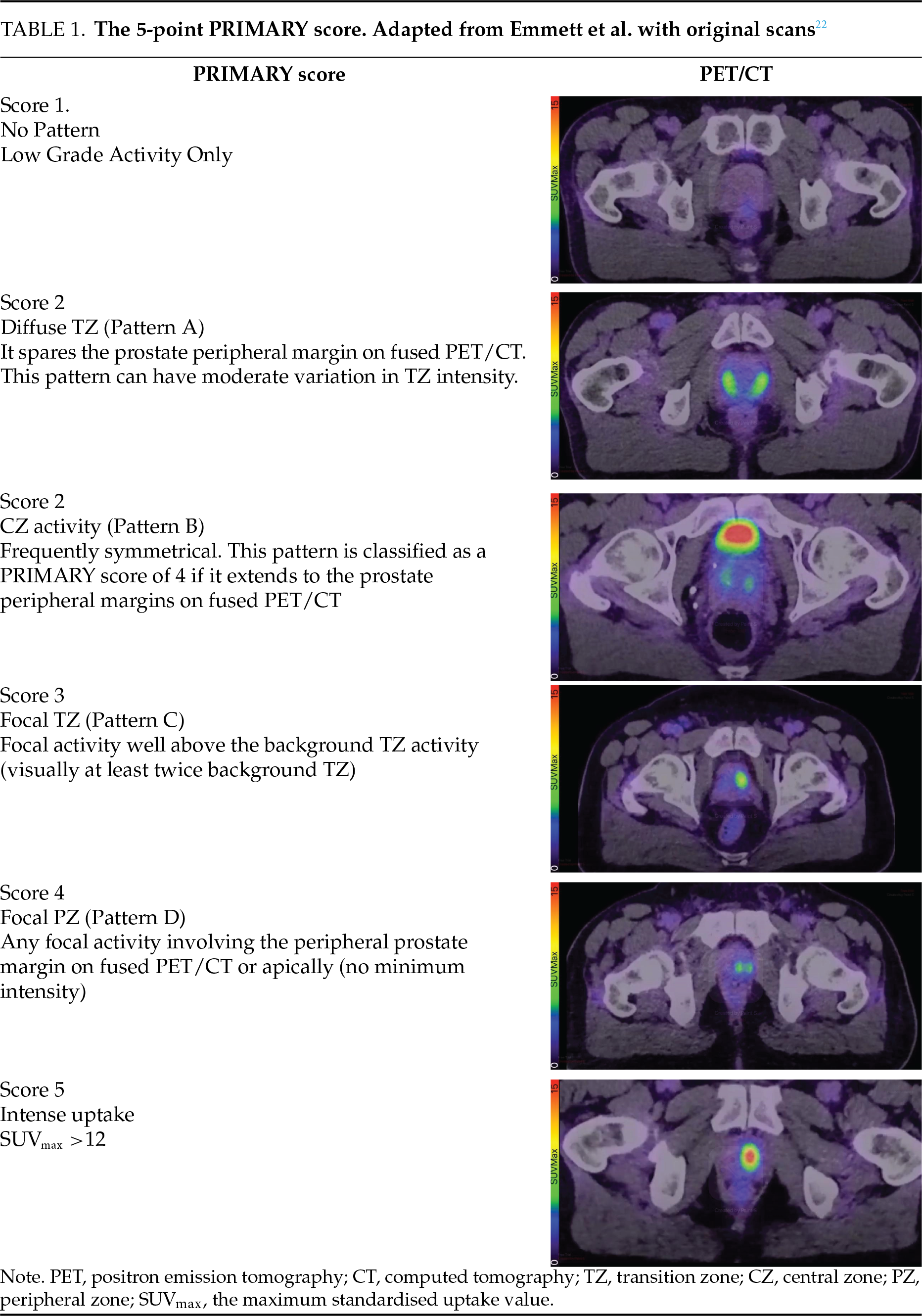
The diagnostic utility of PSMA PET in detecting early prostate cancer is also confirmed in studies assessing the utility of PSMA PET to guide transperineal prostate biopsies. A systematic review assessed 10 different studies, and in a meta-analysis of 6 studies, found that for clinically statistically significant prostate cancer, a PSMA PET-guided prostate biopsy had a sensitivity of 0.89 and a specificity of 0.65.28 This high level of diagnostic accuracy demonstrates the strong correlation between molecular imaging and pathological diagnosis of prostate cancer. Although the authors were unable to identify any studies directly comparing MRI-guided to PSMA-guided prostate biopsies, this is a promising approach. With the high accuracy of the PRIMARY score,22 and promising results of PSMA PET guided biopsies, PSMA PET may become a viable alternative in first-line imaging for men with suspected prostate cancer, or be incorporated in pre-biopsy diagnostic algorithms. Although the evidence base remains immature, this demonstrates the promising role PSMA PET has as a tool to guide prostate biopsies and accurately detect clinically significant prostate cancer.
Another recent comprehensive systematic review and meta-analysis was able to identify 12 studies involving 1533 patients that assessed the utility of PSMA PET in detecting prostate cancer23 The review that PSMA PET had a sensitivity of 82%, specificity of 67%, positive predictive value (PPV) of 77% and negative predictive value (NPV) of 73% for detecting clinically significant (International Society of Urological Pathology [ISUP] 2 or above) prostate cancer. They also found that when combined with mpMRI, the sensitivity increased to 91%, with a comparable specificity of 68%. This reflects the utility that PSMA has as a biomarker for early prostate cancer and suggests that it may have a complementary effect in identifying primary prostate cancers when combined with mpMRI. This suggests that there may be a role in using PSMA to help select patients for prostate biopsy, allowing for the reduction of the number of biopsies and reducing the number of insignificant prostate cancers detected at biopsy.
These results are highly promising and suggest PSMA PET has the potential to become a useful adjunct, or even an alternative to mpMRI in selecting patients to undergo transperineal prostate biopsy. MRI is a time-consuming scan that has a number of contraindications and safety concerns. Gadolinium-based MRI contrast agents are not suitable for men who have significantly impaired renal function29 whilst a hepatically excreted PSMA PET tracer 18F PSMA-1007 is available,30 and can provide similar diagnostic performance in both local lesions and distant disease—although it has higher rates of benign bone uptakes.31 Patients with implantable devices such as cardiac pacemakers or cochlear implants require additional screening and safety assessment of the device prior to undergoing MRI, and some older devices may be MRI-incompatible.29 Image acquisition may be impaired by prosthetic hip joints, leading to decreased cancer detection rate.32 Many men find MRI scanners to be claustrophobic, making them intolerable to a certain percentage of the population. The emergence of PSMA PET may provide an alternative for selecting men for prostate biopsy in these groups of men who would otherwise face significant barriers to or be unable to undergo an mpMRI for the detection of prostate cancer, although further evidence is required to establish it in this role.
Given that many men may have contraindications to mpMRI, PSMA PET may also have a role in the management of low-risk prostate cancer for men placed on active surveillance. There is a growing use of interval mpMRI throughout the course of active surveillance,33 meaning that men may potentially be subjected to multiple scans throughout the course of their lifetime. PSMA PET may therefore be of even more importance in this cohort of men, however, a recent review found that data to support its routine use in active surveillance is lacking.34 There are currently two prospective trials currently enrolling patients that will provide further data regarding the potential use of PSMA PET in active surveillance,35–37 and early studies have suggested that PSMA PET may be useful in active surveillance by detecting occult clinically significant prostate cancer,38 and as a tool to decrease the need for repeat confirmatory biopsy.39
PSMA as a Prognostic Indicator in Prostate Cancer
Lymph node involvement is perhaps one of the most important prognostic factors in prostate cancer, as it demonstrates advanced disease. It is associated with worse rates of biochemical recurrence-free survival and overall survival.40 Traditionally, detection of lymph nodes was done via extended pelvic lymph node dissection (ePLND), as conventional imaging modalities such as CT have a low sensitivity for the detection of lymph node metastasis. However, ePLND is a comorbid procedure and is associated with high rates of lymphocele, infection, and venous thromboembolism.41 Given that systematic reviews have failed to demonstrate an oncological benefit from ePLND, it remains a highly morbid way of providing prognostic information for men with prostate cancer.42 To select patients for lymph node dissection, clinicians have traditionally relied on various nomograms that risk-stratify patients based on clinicopathological features.43 This allows for the selection of only high-risk patients for ePLND, reducing its use in men with low risk of lymph node metastasis, where it is likely to be of little diagnostic or prognostic value. However, in the era of highly specific PSMA-PET scans, there has been a significant debate regarding the ongoing utility of ePLND and whether it can be supplanted by medical imaging with or without the use of a nomogram.
PSMA-PET has shown significant potential in improving these nomograms. Traditional nomograms such as Briganti 201944 and Memorial Sloan Kettering Cancer Center (MSKCC)45 combine various clinico-pathological features to predict the rate of lymph node involvement. These features include age, PSA, type of biopsy (MRI-Targeted vs. systemic), Gleason score, and clinical T stage.44,45 However, given the high specificity for detecting lymph node invasion, there has been considerable research into potentially incorporating PSMA-PET findings into pre-operative nomograms. Meijer et al.46 first reported that incorporation of PSMA-PET findings into a nomogram significantly improved the predictive ability of the MSKCC, Briganti 2017 and Briganti 2019 nomograms. Muehlematter et al.47 subsequently developed their own nomogram, which incorporated volumetric factors of PSMA-avid lesions and the presence of miN1 disease into their model.47 Their PSMA-based model showed similar calibration, with a superior AUC, suggesting that with a risk cut-off of 17%, 50% of lymph node dissections could be spared, whilst only missing 4.8% of patients with lymph node invasion.47 Both of these models were recently externally validated and compared to the MSKCC and Briganti 2012 models by van Bergen et al.48 This study confirmed the finding that the models developed using PSMA-PET parameters outperformed traditional nomograms based on clinicopathological features alone.48 Although a key shortcoming of these models is that they systematically underperform in assessing patients without evidence of lymph node invasion on PSMA PET (miN0 disease). Subsequently, the Briganti 2023 model has been developed,48 and externally validated in this patient group without using parameters from PSMA PET. This demonstrates the importance of patient selection when using nomograms in risk assessment, as they will only be applicable to similar populations in which they were developed. These nomograms, developed by Meijer et al.46 and Muehlematter et al., likely overestimated the prevalence of positive lymph nodes, as they included miN1 (patients with detectable nodes on PSMA-PET) in the population they developed their nomogram. Multiple attempts at external validation of shown that this leads to a decreased accuracy and reduced AUC when examining populations that are miN0.48,49 This limits the applicability of these models in this sub-group of patients, who are the most important to risk-stratify, as PSMA-PET’s high specificity all but confirms the presence of nodal disease in miN1 patients. Ultimately, whether PSMA-PET is a useful parameter to be incorporated into pre-operative nomograms to help identify patients for ePLND is an area of ongoing research, but remains uncertain given recent results suggesting the Briganti 2023 nomogram outperforms nomograms incorporating PSMA parameters when assessing miN0 patients.49
PSMA PET has ushered in a new era of staging lymph nodes for prostate cancer. A meta-analysis pooled specificity for detecting lymph node invasion of 95%, with a sensitivity of 54%.50 This allows for accurate, non-invasive assessment of lymph nodes prior to definitive treatment, giving the clinician and patient valuable prognostic information which can be used to guide treatment choices. However, the relatively low sensitivity of PSMA PET at detecting lymph nodes limits its ability to entirely replace traditional methods of staging lymph nodes, as it will miss a significant proportion of patients with positive lymph nodes. PSMA PET may also fail to involve lymph nodes in ductal prostate cancers, which can express variable PSMA uptake.51,52 Although data suggest that a high percentage of men can safely be excluded from ePLND based on PSMA PET findings,53 whether it is possible to fully omit patients from lymph node dissection based on PET alone remains a controversial topic. A systematic review and meta-analysis of the data suggested that high-risk patients could not be safely excluded from ePLND due to the lower negative predictive value within this cohort of 83%.50 This suggests that within high-risk cohorts, 17% of men with lymph node metastasis may be missed based on PSMA PET alone, although there may be some value within the intermediate-risk group, which had a higher negative predictive value.50 Whether it is safe to exclude patients from ePLND based on PSMA-PET alone remains an ongoing area of research, with an ongoing randomised control trial in the Netherlands investigating this topic. The PSMA-SELECT trial will randomise patients to either receive routine ePLND or ePLND guided by PSMA findings, with a primary endpoint of biochemical recurrence at 2 years. The results of this trial are eagerly awaited and should help clarify the oncological safety of omitting nodal dissection based on PSMA findings. Ultimately, PSMA-PET represents a fundamentally new way to stage for lymph-node invasion. Given the significant morbidity of staging via ePLND, and that a systematic review failed to demonstrate a strong oncological benefit from the nodal dissection,42 PSMA PET represents a new paradigm in lymph node assessment due to its non-invasive nature.
The prognostic value of PSMA PET is not limited to its assessment of lymph node invasion. In early prostate cancer, PSMA PET is also proving to be useful in risk-stratifying men with newly diagnosed prostate cancer based on radiomic features of the primary tumour.54 This is particularly evident when examining the relationship of PSMA PET SUVmax with the Gleason score of the cancer. Studies have consistently demonstrated that a higher SUVmax is correlated with a higher percentage of pattern 4 disease at the time of final prostatectomy.55,56 This pattern was found across multiple studies, in a systematic review of 14 studies involving 1975 patients, which demonstrated that the pooled mean SUVmax increased with ISUP grade group.25 A higher SUVmax was also associated with a higher pathological T stage group, and may be associated increased risk of biochemical recurrence post-surgery. This is an unsurprising finding, as it is well-established that patients with a higher percentage of pattern 4 and increasing grade group demonstrate an increased risk of biochemical recurrence post-surgery.57–59 This demonstrates that PSMA PET may be able to play an important role in risk-stratification and prognostication prior to definitive management, as it can non-invasively provide characterise the risk of the primary cancer.
More interestingly, patients with T3/T4 disease were found to have a higher SUVmax than those with organ-confined disease, although this finding was limited by heterogeneity between studies and evidence of small study effects. This finding is in keeping with a different review by Mazzone et al., which found a pooled sensitivity 71% and specificity of 84% for extracapsular detection (pT3a disease), and a pooled sensitivity 71% and specificity of 93% for detecting seminal vesicle invasion (T3b disease).23 Chen et al. also found that a higher SUVmax was predictive of a shorter time to biochemical recurrence across all cohorts.25 This provides compelling evidence that PSMA can be used as a marker of worse grades and advanced stage of localised disease. Given that higher grade groups, extra-prostatic disease, and short time to biochemical recurrence are all predictors of worse outcomes,60 this implies that PSMA PET assessment of the primary tumour can provide some prognostic information regarding the risk of the underlying disease. Although the data is promising, it remains to be seen whether PSMA PET will become routinely incorporated into risk assessment and prognostication of localised prostate cancers.
PSMA is also a potentially useful tool for risk-stratifying prostate cancer on immunohistochemistry. Multiple studies have demonstrated that PSMA is a useful stain in detecting and diagnosing prostate cancer on both biopsy and radical prostatectomy specimens. PSMA expression is higher in cancerous glands than in benign glands, and increases with cancer’s Gleason score, and is correlated with a higher PSA.61 This suggests that PSMA immunostaining may have potential utility in providing prognostic information regarding cancer aggressiveness. This is also implied by numerous studies, which have demonstrated a significant correlation between PSMA immunostaining and SUVmax on PSMA PET.62–65 This finding has also been confirmed in a recent prospective study, which identified that PSMA staining as measured by the immunoreactive score of both prostate biopsies and radical prostatectomy specimens was predictive of SUVmax on a linear regression model.63 Whilst logically unsurprising, research has demonstrated that low levels of PSMA staining on the primary tumour at the time of radical prostatectomy are a significant predictor of having a negative PSMA scan—when assessing for biochemical recurrent disease.64 Given the association between SUVmax and PSMA immunostaining, biopsy results may be useful in selecting patients for which patients may have positive findings on PSMA-PET.66 Droghetti et al. found that not only is the amount of PSMA expression correlated with SUVmax and PSMA-PET findings, but that the pattern of PSMA expression is relevant as well.66 They found that a membranous pattern of PSMA expression was associated with a higher SUVmax and total lesion activity than a cytosolic expression pattern. This is likely due to membranous PSMA being more available to bind to the radiotracer.
It is a logical progression to assume that if PSMA immunostaining is associated with SUVmax, and SUVmax is associated with aggressiveness of a tumour, and markers for adverse pathological outcomes, then PSMA staining may directly correlate with disease aggressiveness. This was actually the finding of one study by Hupe et al.,67 which examined the use of PSMA staining in risk-stratification. The authors found that PSMA expression on biopsy was correlated with established prognostic biomarkers, including lymph node metastasis, locally recurrent tumours, and distant disease.67 Most importantly, they found that higher levels of PSMA expression on both prostate biopsy and final radical prostatectomy specimen predicted biochemical recurrence post-surgery. This is an important finding, which needs to be investigated further, as it suggests that immunohistostaining performed at the time of initial biopsy can provide important prognostic information regarding the tumour. If confirmed in more studies, then PSMA staining could potentially become a routine part of tumour risk assessment, and could be incorporated into clinical decision-making tools, such as pre-operative nomograms to help select patients for ePLND. However, further evidence is needed prior to PSMA IHC becoming routinely implemented into clinical practice.
Overall, PSMA is a highly promising biomarker for risk-stratifying prostate cancer. PSMA staining may be associated with higher-risk disease, and is associated with SUVmax on PSMA PET scan. SUVmax is an important radiomic marker on PSMA PET, which has been associated with worsened disease grade group. Whether measurements from a PSMA PET, such as the SUVmax, can be incorporated into pre-operative nomograms to improve performance is an ongoing area of research, as it may help select patients for ePLND.
PSMA as a Therapeutic Target for Early Prostate Cancer
PSMA has also garnered significant interest as a potential molecular target for drugs to treat prostate cancer. There are two main approaches to PSMA-targeted drug therapy been developed: Antibody-drug conjugates (ADCs), and radioligand therapy. Antibody drug conjugates bind a cytotoxic agent to a monoclonal antibody specific for a cellular antibody, such as PSMA. The cytotoxic agent is then internalised by endocytosis at the target cell, allowing for targeted delivery of the cytotoxic agent to specific cells expressing that receptor.68 This approach has proven successful in other cancers, with multiple FDA-approved Antibody-drug conjugates for leukaemia, lymphoma, breast, cervical, and urothelial cancer.69 To date, clinical trials of antibody-drug conjugates targeting PSMA in prostate cancer have undergone early clinical trials.68 however, these have been limited to phase I/II trials in advanced metastatic prostate cancer. Results from these trials have also been far from promising and have been limited by significant treatment-related side effects. We were able to identify three separate ADCs that have been trialled in humans. Monomethylauristatin E (MMAE) has undergone both phase I and II trials with results published.70,71 In the phase II trial, 119 patients with metastatic castrate resistant prostate cancer (mCRPC) were recruited, with a plan to complete 8 cycles of treatment. Unfortunately, 62% withdrew before the 5th cycle due to adverse effects, with 51% suffering severe adverse events. There was a rather limited response as well, with a >50% decline in PSA only observed in 14% of patients, which is a relatively modest response. Similarly, the ADC MLN2704 was highly limited by its treatment-related toxicity in a phase I/II trial for men with mCRPC.72 71% of patients experienced peripheral neuropathy whilst on treatment, and only a limited effect was identified, with only 8% of patients having a PSA decrease greater than 50%.72 One other ADC has been studied in a phase I trial for men with mCRPC. MEDI 3726 was studied in 33 men with mCRPC, however, the trial was limited by poor tolerability.73 90% of the patients had treatment-related adverse events, with 45% of these being grade 3 or 4. As such, no maximum tolerated dose (MTD) was able to be identified.73 Overall, so far, human trials involving ADCs have been limited to men with mCRPC and have all demonstrated significant toxicities, with very limited response demonstrated. No phase III trial has been published. This suggests that ADCs may have a limited effect in prostate cancer and significant side effects. Whilst it is certainly possible for safer and more efficacious ADCs to be developed for prostate cancer, more research is needed. All studies so far have been conducted in men with mCRPC, so any potential use for ADCs treating early prostate cancer is purely speculative.
In contrast, the clinical utility of radioligand therapy for prostate cancer has progressed beyond the clinical trial stage and is now approved for select men with metastatic prostate cancer. Radioligand therapy conjugates a radioactive isotope to a monoclonal antibody for a certain antigen. This then delivers the isotope to the targeted cells, where it undergoes decay and delivers a dose of radiation to the cells and the surrounding microenvironment, inducing a cytotoxic effect.74 The most promising agent is Luteitum-177-PSMA-617 (Lu-177), which demonstrated a survival benefit in metastatic castrate-resistant prostate cancer in patients previously treated with androgen deprivation, ARPI, and chemotherapy.75 As a result of the ground-breaking vision trial, Lu-177 got approval for use in humans and has increasingly been used and investigated in earlier stages of the disease process in treatment algorithms. A phase II trial suggested that Lu-177 was superior to the second-line chemotherapy agent Cabazitaxel.76 More recently, trials have assessed Lu-177 for efficacy at earlier stages of the disease process and have shown improved progression-free survival in patients naïve to taxane-based chemotherapy.77,78 A phase II trial also demonstrated a benefit when commenced at the same time as ARPI therapy.79 All these trials contribute to a growing body of evidence that Lu-177 is effective in earlier stages of mCRPC, and that earlier initiation of Lu-177 treatment may offer some survival benefit over the standard-of-care approach to introduction of ARPI, then taxane-based chemotherapy.
Alternative conjugates for delivering Lu-177 for radioligand therapy have also been investigated. Lu-PNT2002, also known as Lu-PSMA-I&T, is the second most widely used lutetium-based radiopharmaceutical in prostate cancer.80 It is currently being evaluated in the setting of castrate resistant prostate cancer that has progressed after a single ARPi.81 The phase I results demonstrated a favourable dosimetry and safety profile, with early promising results, although the final outcomes of the trial are still pending.81 Other Lutetium-based radiopharmaceuticals which have been investigated for use in prostate cancer include [177Lu]-LNC1011,82 177Lu-rhPSMA-10.1,83 177Lu-Ludotadipep,84 177Lu-labeled J591 monoclonal antibodies,85 and PSMA-Targetted CT140386 (Table 2). However, so far, all of these have only been studied in the pre-clinical, or stage I/II setting, and are not yet approved for human use. All these therapies were investigated for metastatic disease and are not applicable to early prostate cancer.
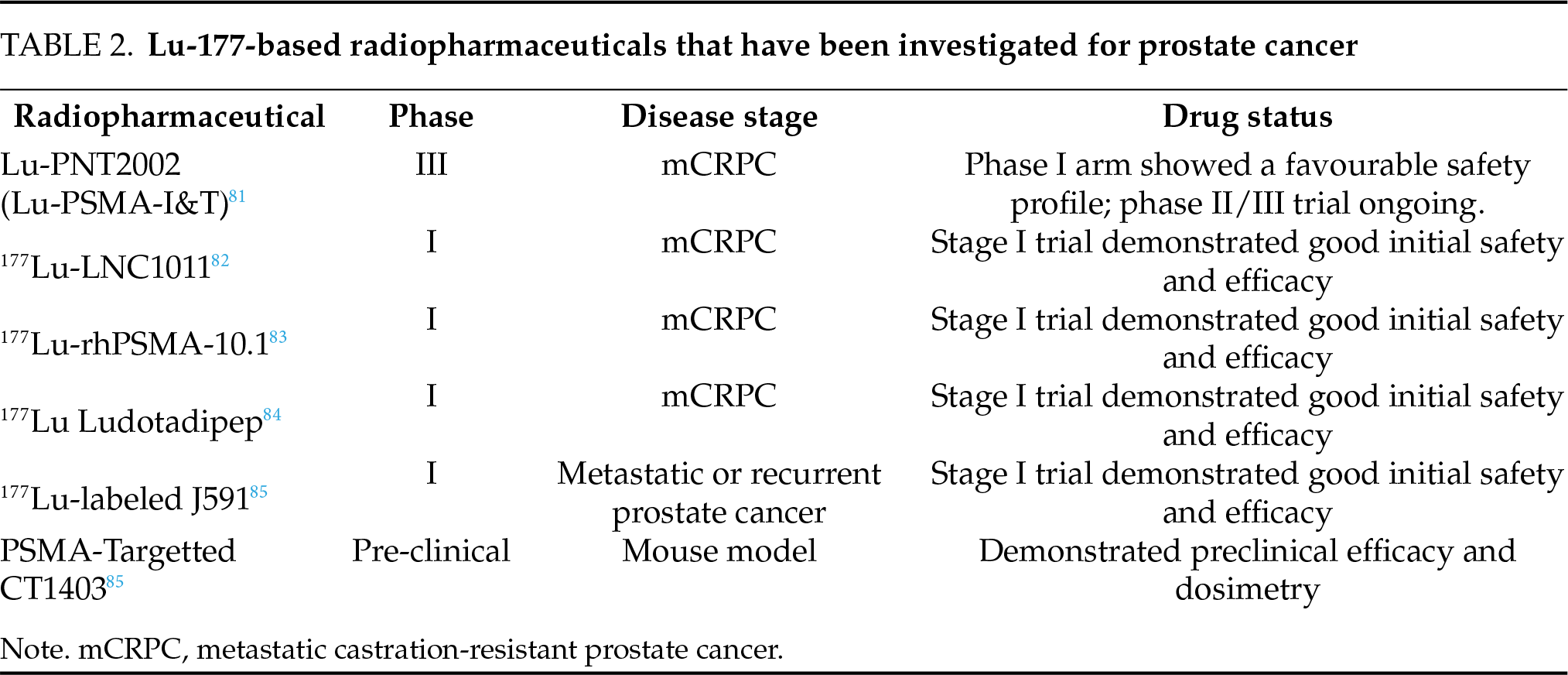
Yet at this stage, radioligand therapy for prostate cancer remains limited to patients with advanced disease. PSMA is heavily expressed in earlier stages of prostate cancer, as such, there is significant interest in its potential clinical utility earlier in the disease course. There is currently an ongoing phase III clinical trial trying to assess for a benefit in the metastatic hormone-sensitive setting.87 If successful, this would represent a major breakthrough as the use of Lu-177 would be pushed back prior to the development of castrate-resistant disease. However, the use of Lu-177 has also been investigated even earlier in the disease process. Recently, the LuTect omy trial demonstrated the safety and feasibility of using Lu-177 as neoadjuvant therapy prior to radical prostatectomy in men with high-risk prostate cancer.88 Although the phase I/II trial was not designed or powered to detect any benefit to survival or recurrence rate, it has proven the feasibility of radioligand therapy in earlier disease stages, prior to radical surgery. Whether Lu-177 may have a role in the treatment of early prostate cancer is a promising topic for further research, however, more evidence is needed before it can be considered to have a role in the treatment of localised prostate cancer. Key clinical trials are progressively investigating the use of Lu-177 at the earlier stages of prostate cancer treatment, demonstrating that there is intense research interest in this topic (Table 3).

In summary, our review highlights the role PSMA plays as a biomarker in the diagnosis, risk-stratification, and prognostication of early and localized advanced prostate cancer. PSMA PET scans allow for far more accurate staging of high-risk disease, and can aid in selecting patients for lymph node dissection. PSMA PET also offers comparable accuracy to mpMRI in detecting clinically significant prostate cancer and targeting transperineal biopsies. PSMA is also an important stain for pathologists and may offer insights into underlying disease aggressiveness. PSMA also represents an emerging therapeutic target, with PSMA-targeted radioligand therapy demonstrating benefit in metastatic disease. Subsequently, clinical trials have progressively assessed the use of Lu-177 in earlier disease stages, including in the adjuvant setting. Overall, PSMA is a useful biomarker in early and localized advanced prostate cancer with a role in disease detection and risk stratification, and potentially as a target for therapeutics.
Acknowledgement
The authors have no acknowledgements to declare.
Funding Statement
The authors received no specific funding.
Author Contributions
Conception and design: Marlon Perera and Jonathon Carll. Data collection: Jonathon Carll and Jacinta Bonaddio. Analysis and interpretation of results: Jonathon Carll, Nathan Lawrentschuk, Marlon Perera. Draft manuscript preparation: Jonathon Carll, Thilakavathi Chengodu, Dixon Woon. Final manuscript review and revision: Jonathon Carll, Jacinta Bonaddio, Marlon Perera, Nathan Lawrentschuk, Thilakavathi Chengodu, Dixon Woon. All authors reviewed the results and approved the final version of this manuscript.
Availability of Data and Materials
This is a review article with no original data collected or stored. All data analyzed in this review article are derived from publicly available sources, primarily peer-reviewed journal articles. All sources cited in this review are accessible through standard academic databases and repositories.
Ethics Approval
Not applicable.
Conflicts of Interest
Jonathon Carll—No conflicts exist. Jacinta Bonaddio—No conflicts exist. Marlon Perera—No conflicts exist. Nathan Lawrentschuk declares the following conflicts: Proctor for both Robotic Surgery (Device Technologies Australia) and Focal Therapy with Nanoknife (Getz Healthcare Australia). Thilakavathi Chengodu—No conflicts exist. Dixon Woon—No conflicts exist.
References
1. Bergengren O, Pekala KR, Matsoukas K et al. Update on prostate cancer epidemiology and risk factors-a systematic review. Eur Urol 2023 Aug;84(2):191–206. doi:10.1016/j.eururo.2023.04.021. [Google Scholar] [PubMed] [CrossRef]
2. Schröder FH, Hugosson J, Carlsson S et al. Screening for prostate cancer decreases the risk of developing metastatic disease: findings from the European Randomized Study of Screening for Prostate Cancer (ERSPC). Eur Urol 2012 Nov;62(5):745–752. doi:10.1016/j.eururo.2012.05.068. [Google Scholar] [PubMed] [CrossRef]
3. Force UPST. Screening for prostate cancer: US preventive services task force recommendation statement. JAMA 2018;319(18):1901–1913. doi:10.1001/jama.2018.3710. [Google Scholar] [PubMed] [CrossRef]
4. Cornford P, Tilki D, van den Bergh RCN et al. EAU-EANM-ESTRO-ESUR-ISUP-SIOG Guidelines on Prostate Cancer. EAU Guidelines Office; 2024. [cited 2025 Mar 19]. https://d56bochluxqnz.cloudfront.net/documents/full-guideline/EAU-EANM-ESTRO-ESUR-ISUP-SIOG-Guidelines-on-Prostate-Cancer-2024_2024-04-09-132035_ypmy_2024-04-16-122605_lqpk.pdf. [Google Scholar]
5. Andriole GL, Crawford ED, Grubb RL3rd et al. Prostate cancer screening in the randomized prostate, lung, colorectal, and ovarian cancer screening trial: mortality results after 13 years of follow-up. J Natl Cancer Inst 2012 Jan 18;104(2):125–132. doi:10.1093/jnci/djr500. [Google Scholar] [PubMed] [CrossRef]
6. Silver DA, Pellicer I, Fair WR, Heston WD, Cordon-Cardo C. Prostate-specific membrane antigen expression in normal and malignant human tissues. Clin Cancer Res 1997;3(1):81–85. [Google Scholar] [PubMed]
7. Horoszewicz JS, Kawinski E, Murphy GP. Monoclonal antibodies to a new antigenic marker in epithelial prostatic cells and serum of prostatic cancer patients. Anticancer Res 1987;7(5B):927–935. doi:10.1016/0734-9750(93)90273-p. [Google Scholar] [CrossRef]
8. Israeli RS, Powell CT, Fair WR, Heston WDW. Molecular cloning of a complementary DNA encoding a prostate-specific membrane antigen. Cancer Res 1993;53(2):227–230. [Google Scholar] [PubMed]
9. O’Keefe DS, Su SL, Bacich DJ et al. Mapping, genomic organization and promoter analysis of the human prostate-specific membrane antigen gene. Biochim Biophys Acta (BBA)-Gene Struct Express 1998;1443(1):113–127. doi:10.1016/S0167-4781(98)00200-0. [Google Scholar] [PubMed] [CrossRef]
10. Salas Fragomeni RA, Amir T, Sheikhbahaei S et al. Imaging of nonprostate cancers using PSMA-targeted radiotracers: rationale, current state of the field, and a call to arms. J Nucl Med 2018;59(6):871–877. doi:10.2967/jnumed.117.203570. [Google Scholar] [PubMed] [CrossRef]
11. Puik JR, Le C, Kazemier G et al. Prostate-specific membrane antigen as target for vasculature-directed therapeutic strategies in solid tumors. Crit Rev Oncol Hematol 2025;205(10):104556. doi:10.1016/j.critrevonc.2024.104556. [Google Scholar] [PubMed] [CrossRef]
12. Conway RE, Petrovic N, Li Z, Heston W, Wu D, Shapiro LH. Prostate-specific membrane antigen regulates angiogenesis by modulating integrin signal transduction. Mol Cell Biol 2006;26(14):5310–5324. doi:10.1128/MCB.00084-06. [Google Scholar] [PubMed] [CrossRef]
13. Watanabe R, Maekawa M, Kiyoi T et al. PSMA-positive membranes secreted from prostate cancer cells have potency to transform vascular endothelial cells into an angiogenic state. Prostate 2021;81(16):1390–1401. doi:10.1002/pros.24237. [Google Scholar] [PubMed] [CrossRef]
14. Bakht MK, Beltran H. Biological determinants of PSMA expression, regulation and heterogeneity in prostate cancer. Nature Rev Urol 2025;22(1):26–45. doi:10.1038/s41585-024-00900-z. [Google Scholar] [PubMed] [CrossRef]
15. Yao V, Bacich DJ. Prostate specific membrane antigen (PSMA) expression gives prostate cancer cells a growth advantage in a physiologically relevant folate environment in vitro. The Prostate 2006;66(8):867–875. doi:10.1002/pros.20361. [Google Scholar] [PubMed] [CrossRef]
16. Beltran H, Prandi D, Mosquera JM et al. Divergent clonal evolution of castration-resistant neuroendocrine prostate cancer. Nat Med 2016 Mar;22(3):298–305. doi:10.1038/nm.4045. [Google Scholar] [PubMed] [CrossRef]
17. Meller B, Bremmer F, Sahlmann CO et al. Alterations in androgen deprivation enhanced prostate-specific membrane antigen (PSMA) expression in prostate cancer cells as a target for diagnostics and therapy. EJNMMI Res 2015;5(1):66. doi:10.1186/s13550-015-0145-8. [Google Scholar] [PubMed] [CrossRef]
18. Evans MJ, Smith-Jones PM, Wongvipat J et al. Noninvasive measurement of androgen receptor signaling with a positron-emitting radiopharmaceutical that targets prostate-specific membrane antigen. Proc Natl Acad Sci U S A 2011;108(23):9578–9582. doi:10.1073/pnas.1106383108. [Google Scholar] [PubMed] [CrossRef]
19. Sommer U, Siciliano T, Ebersbach C et al. Impact of androgen receptor activity on prostate-specific membrane antigen expression in prostate cancer cells. Int J Mol Sci 2022;23(3):1046. doi:10.3390/ijms23031046. [Google Scholar] [PubMed] [CrossRef]
20. Bakht MK, Yamada Y, Ku S-Y et al. Landscape of prostate-specific membrane antigen heterogeneity and regulation in AR-positive and AR-negative metastatic prostate cancer. Nat Cancer 2023;4(5):699–715. doi:10.1038/s43018-023-00539-6. [Google Scholar] [PubMed] [CrossRef]
21. Hofman MS, Lawrentschuk N, Francis RJ et al. Prostate-specific membrane antigen PET-CT in patients with high-risk prostate cancer before curative-intent surgery or radiotherapy (proPSMAa prospective, randomised, multicentre study. Lancet 2020;395(10231):1208–1216. doi:10.1016/S0140-6736(20)30314-7. [Google Scholar] [PubMed] [CrossRef]
22. Emmett L, Papa N, Buteau J et al. The PRIMARY score: using intraprostatic 68Ga-PSMA PET/CT patterns to optimize prostate cancer diagnosis. J Nucl Med 2022;63(11):1644–1650. doi:10.2967/jnumed.121.263448. [Google Scholar] [PubMed] [CrossRef]
23. Mazzone E, Cannoletta D, Quarta L et al. A comprehensive systematic review and meta-analysis of the role of prostate-specific membrane antigen positron emission tomography for prostate cancer diagnosis and primary staging before definitive treatment. Eur Urol 2025;87(6):654–671. doi:10.1016/j.eururo.2025.03.003. [Google Scholar] [PubMed] [CrossRef]
24. Mazzone E, Thomson A, Chen DC et al. The role of prostate-specific membrane antigen positron emission tomography for assessment of local recurrence and distant metastases in patients with biochemical recurrence of prostate cancer after definitive treatment: a systematic review and meta-analysis. Eur Urol 2025;88(2):129–141. doi:10.1016/j.eururo.2025.05.006. [Google Scholar] [PubMed] [CrossRef]
25. Chen DC, Huang S, Papa N et al. Impact of intraprostatic PSMA maximum standardised uptake value following prostatectomy: a systematic review and meta-analysis. BJU Int 2025;135(5):720–732. doi:10.1111/bju.16608. [Google Scholar] [PubMed] [CrossRef]
26. Emmett L, Buteau J, Papa N et al. The additive diagnostic value of prostate-specific membrane antigen positron emission tomography computed tomography to multiparametric magnetic resonance imaging triage in the diagnosis of prostate cancer (PRIMARYa prospective multicentre study. Eur Urol 2021;80(6):682–689. doi:10.1016/j.eururo.2021.08.002. [Google Scholar] [PubMed] [CrossRef]
27. Emmett L, Papa N, Counter W et al. Reproducibility and accuracy of the PRIMARY score on PSMA PET and of PI-RADS on multiparametric MRI for prostate cancer diagnosis within a real-world database. J Nucl Med 2024;65(1):94–99. doi:10.2967/jnumed.123.266164. [Google Scholar] [PubMed] [CrossRef]
28. Aggarwal P, Gunasekaran V, Singh H, Kumar R, Satapathy S, Mittal BR. Diagnostic accuracy of PSMA PET-guided prostate biopsy in prostate cancer—a systematic review and meta-analysis. Clin Nucl Med 2025;50(1):e26–e33. doi:10.1097/rlu.0000000000005501. [Google Scholar] [PubMed] [CrossRef]
29. MRI Safety Guidelines, 3.0. The Royal Australian and New Zealand College of Radiologists; 2021. [cited 2025 Jun 19]. Available from: https://www.ranzcr.com/college/document-library/mri-safety-guidelines. [Google Scholar]
30. Giesel FL, Hadaschik B, Cardinale J et al. F-18 labelled PSMA-1007: biodistribution, radiation dosimetry and histopathological validation of tumor lesions in prostate cancer patients. Eur J Nucl Med Mol Imaging 2017;44(4):678–688. doi:10.1007/s00259-016-3573-4. [Google Scholar] [PubMed] [CrossRef]
31. Huang S, Ong S, McKenzie D et al. Comparison of 18F-based PSMA radiotracers with [68Ga]Ga-PSMA-11 in PET/CT imaging of prostate cancer-a systematic review and meta-analysis. Prostate Cancer Prostatic Dis 2024;27(4):654–664. doi:10.1038/s41391-023-00755-2. [Google Scholar] [PubMed] [CrossRef]
32. Nakai H, Takahashi H, Adamo DA et al. Decreased prostate MRI cancer detection rate due to moderate to severe susceptibility artifacts from hip prosthesis. Eur Radiol 2024;34(5):3387–3399. doi:10.1007/s00330-023-10345-4. [Google Scholar] [PubMed] [CrossRef]
33. Ploussard G, Rouvière O, Rouprêt M, van den Bergh R, Renard-Penna R. The current role of MRI for guiding active surveillance in prostate cancer. Nature Rev Urol 2022;19(6):357–365. doi:10.1038/s41585-022-00587-0. [Google Scholar] [PubMed] [CrossRef]
34. Carll J, Bonaddio J, Lawrentschuk N. PSMA PET as a tool for active surveillance of prostate cancer-where are we at? J Clin Med 2025;14(10):3580. doi:10.3390/jcm14103580. [Google Scholar] [PubMed] [CrossRef]
35. Liu J, Harewood L, Bagguley D et al. Early results from the CONFIRM Trial: utility of prostate-specific membrane antigen positron emission tomography/computed tomography in active surveillance for prostate cancer. Eur Urol Onc 2025;8(4):1118–1125. doi:10.1016/j.euo.2025.02.010. [Google Scholar] [PubMed] [CrossRef]
36. Bagguley D, Harewood L, McKenzie D et al. The CONFIRM trial protocol: the utility of prostate-specific membrane antigen positron emission tomography/computed tomography in active surveillance for prostate cancer. BJU Int 2024;133(Suppl 4):27–36. doi:10.1111/bju.16214. [Google Scholar] [PubMed] [CrossRef]
37. Gondoputro W, Doan P, Katelaris A et al. 68Ga-PSMA-PET/CT in addition to mpMRI in men undergoing biopsy during active surveillance for low- to intermediate-risk prostate cancer: study protocol for a prospective cross-sectional study. Transl Androl Urol 2023;12(10):1598–1606. doi:10.21037/tau-22-708. [Google Scholar] [PubMed] [CrossRef]
38. Heetman JG, Lavalaye J, Polm PD et al. Gallium-68 prostate-specific membrane antigen positron emission tomography/computed tomography in active surveillance for prostate cancer trial (PASPoRT). Eur Urol Oncol 2024;7(2):204–210. doi:10.1016/j.euo.2023.05.004. [Google Scholar] [PubMed] [CrossRef]
39. Pepe P, Pepe L, Tamburo M, Marletta G, Pennisi M, Fraggetta F. 68Ga-PSMA PET/CT evaluation in men enrolled in prostate cancer active surveillance. Arch Ital Urol Androl 2023;22 95(2):11322. doi:10.4081/aiua.2023.11322. [Google Scholar] [PubMed] [CrossRef]
40. Marra G, Valerio M, Heidegger I et al. Management of patients with node-positive prostate cancer at radical prostatectomy and pelvic lymph node dissection: a systematic review. Eur Urol Oncol 2020;3(5):565–581. doi:10.1016/j.euo.2020.08.005. [Google Scholar] [PubMed] [CrossRef]
41. Cacciamani GE, Maas M, Nassiri N et al. Impact of pelvic lymph node dissection and its extent on perioperative morbidity in patients undergoing radical prostatectomy for prostate cancer: a comprehensive systematic review and meta-analysis. Eur Urol Oncol 2021;4(2):134–149. doi:10.1016/j.euo.2021.02.001. [Google Scholar] [PubMed] [CrossRef]
42. Fossati N, Willemse PM, Van den Broeck T et al. The benefits and harms of different extents of lymph node dissection during radical prostatectomy for prostate cancer: a systematic review. Eur Urol 2017;72(1):84–109. doi:10.1016/j.eururo.2016.12.003. [Google Scholar] [PubMed] [CrossRef]
43. Cimino S, Reale G, Castelli T et al. Comparison between Briganti, Partin and MSKCC tools in predicting positive lymph nodes in prostate cancer: a systematic review and meta-analysis. Scandinavian J Urol 2017;51(5):345–350. doi:10.1080/21681805.2017.1332680. [Google Scholar] [PubMed] [CrossRef]
44. Gandaglia G, Ploussard G, Valerio M et al. A novel nomogram to identify candidates for extended pelvic lymph node dissection among patients with clinically localized prostate cancer diagnosed with magnetic resonance imaging-targeted and systematic biopsies. Eur Urol 2019;75(3):506–514. doi:10.1016/j.eururo.2018.10.012. [Google Scholar] [PubMed] [CrossRef]
45. Prostate cancer nomograms: pre-radical prostatectomy. Memorial Sloan Kettering Cancer Centre. [cited 2025 Jun 19]. Available from: https://www.mskcc.org/nomograms/prostate/pre_op. [Google Scholar]
46. Meijer D, van Leeuwen PJ, Roberts MJ et al. External validation and addition of prostate-specific membrane antigen positron emission tomography to the most frequently used nomograms for the prediction of pelvic lymph-node metastases: an international multicenter study. Eur Urol 2021;80(2):234–242. doi:10.1016/j.eururo.2021.05.006. [Google Scholar] [PubMed] [CrossRef]
47. Muehlematter UJ, Schweiger L, Ferraro DA et al. Development and external validation of a multivariable [68Ga]Ga-PSMA-11 PET-based prediction model for lymph node involvement in men with intermediate or high-risk prostate cancer. Eur J Nucl Med Mol Imaging 2023;50(10):3137–3146. doi:10.1007/s00259-023-06278-1. [Google Scholar] [PubMed] [CrossRef]
48. Van Bergen TD, Braat AJAT, Hermsen R et al. External validation of nomograms including PSMA PET information for the prediction of lymph node involvement of prostate cancer. Eur J Nucl Med Mol Imaging 2025;52(10):3744–3756. doi:10.1007/s00259-025-07241-y. [Google Scholar] [PubMed] [CrossRef]
49. Gandaglia G, Barletta F, Scuderi S et al. External validation of nomograms for the identification of pelvic nodal dissection candidates among prostate cancer patients with negative preoperative prostate-specific membrane antigen positron emission tomography. Eur Urol Oncol 2025;79(Suppl 2):595. doi:10.1016/j.euo.2025.01.004. [Google Scholar] [PubMed] [CrossRef]
50. Stabile A, Pellegrino A, Mazzone E et al. Can negative prostate-specific membrane antigen positron emission tomography/computed tomography avoid the need for pelvic lymph node dissection in newly diagnosed prostate cancer patients? a systematic review and meta-analysis with backup histology as reference standard. Eur Urol Oncol 2022;5(1):1–17. doi:10.1016/j.euo.2021.08.001. [Google Scholar] [PubMed] [CrossRef]
51. Pepe P, Pepe L, Curduman M, Pennisi M, Fraggetta F. Ductal prostate cancer staging: role of PSMA PET/CT. Arch Ital Urol Androl 2024;96(1):12132. doi:10.4081/aiua.2024.12132. [Google Scholar] [PubMed] [CrossRef]
52. Zhao Q, Dong A, Bai Y, Zuo C. Prostate-specific membrane antigen uptake heterogeneity in mixed ductal-acinar adenocarcinoma of the prostate. Clin Nucl Med 2023;48(8):750–752. doi:10.1097/rlu.0000000000004742. [Google Scholar] [PubMed] [CrossRef]
53. Yaxley JW, Raveenthiran S, Nouhaud FX et al. Outcomes of primary lymph node staging of intermediate and high risk prostate cancer with 68Ga-PSMA positron emission tomography/computerized tomography compared to histological correlation of pelvic lymph node pathology. J Urol 2019 Apr;201(4):815–820. doi:10.1097/ju.0000000000000053. [Google Scholar] [PubMed] [CrossRef]
54. Dondi F, Antonelli A, Suardi N, Treglia G, Bertagna F. The role of PSMA PET imaging in the classification of the risk of prostate cancer patients: a systematic review on the insights to guide an active surveillance approach. Cancers 2024;16(6):1122. doi:10.3390/cancers16061122. [Google Scholar] [PubMed] [CrossRef]
55. Xue AL, Kalapara AA, Ballok ZE et al. 68Ga-prostate-specific membrane antigen positron emission tomography maximum standardized uptake value as a predictor of gleason pattern 4 and pathological upgrading in intermediate-risk prostate cancer. J Urol 2022;207(2):341–349. doi:10.1097/ju.0000000000002254. [Google Scholar] [PubMed] [CrossRef]
56. Raveenthiran S, Yaxley WJ, Franklin T et al. Findings in 1,123 men with preoperative 68Ga-prostate-specific membrane antigen positron emission tomography/computerized tomography and multiparametric magnetic resonance imaging compared to totally embedded radical prostatectomy histopathology: implications for the diagnosis and management of prostate cancer. J Urol 2022 Mar;207(3):573–580. doi:10.1097/ju.0000000000002293. [Google Scholar] [PubMed] [CrossRef]
57. Sharma M, Miyamoto H. Percent Gleason pattern 4 in stratifying the prognosis of patients with intermediate-risk prostate cancer. Transl Androl Urol 2018;7(Suppl 4):S484–s489. doi:10.21037/tau.2018.03.20. [Google Scholar] [PubMed] [CrossRef]
58. Perera M, Assel MJ, Benfante NE et al. Oncologic outcomes of total length gleason pattern 4 on biopsy in men with grade group 2 prostate cancer. J Urol 2022;208(2):309–316. doi:10.1097/ju.0000000000002685. [Google Scholar] [PubMed] [CrossRef]
59. van Leenders G, van der Kwast TH, Grignon DJ et al. The 2019 international society of urological pathology (ISUP) consensus conference on grading of prostatic carcinoma. Am J Surg Pathol 2020;44(8):e87–e99. doi:10.1097/pas.0000000000001497. [Google Scholar] [PubMed] [CrossRef]
60. Van den Broeck T, van den Bergh RCN, Arfi N et al. Prognostic value of biochemical recurrence following treatment with curative intent for prostate cancer: a systematic review. Eur Urol 2019;75(6):967–987. doi:10.1016/j.eururo.2018.10.011. [Google Scholar] [PubMed] [CrossRef]
61. Bravaccini S, Puccetti M, Bocchini M et al. PSMA expression: a potential ally for the pathologist in prostate cancer diagnosis. Sci Rep 2018;8(1):4254. doi:10.1038/s41598-018-22594-1. [Google Scholar] [PubMed] [CrossRef]
62. Rüschoff JH, Ferraro DA, Muehlematter UJ et al. What’s behind 68Ga-PSMA-11 uptake in primary prostate cancer PET? investigation of histopathological parameters and immunohistochemical PSMA expression patterns. Eur J Nucl Med Mol Imaging 2021;48(12):4042–4053. doi:10.1007/s00259-021-05501-1. [Google Scholar] [PubMed] [CrossRef]
63. Ambrosini F, Piol N, Bauckneht M et al. Immunohistochemical prostate-specific membrane antigen (PSMA) expression patterns of primary prostate cancer tissue as a determining factor for prostate cancer staging with PSMA positron emission tomography/computed tomography. Eur Urol Oncol 2025;395(1):1208. doi:10.1016/j.euo.2025.02.012. [Google Scholar] [PubMed] [CrossRef]
64. Ferraro DA, Rüschoff JH, Muehlematter UJ et al. Immunohistochemical PSMA expression patterns of primary prostate cancer tissue are associated with the detection rate of biochemical recurrence with 68Ga-PSMA-11-PET. Theranostics 2020;10(14):6082–6094. doi:10.7150/thno.44584. [Google Scholar] [PubMed] [CrossRef]
65. Woythal N, Arsenic R, Kempkensteffen C et al. Immunohistochemical validation of PSMA expression measured by 68Ga-PSMA PET/CT in primary prostate cancer. J Nucl Med 2018;59(2):238. doi:10.2967/jnumed.117.195172. [Google Scholar] [PubMed] [CrossRef]
66. Droghetti M, Bianchi L, Presutti M et al. Immunohistochemistry analysis of PSMA expression at prostatic biopsy in high-risk prostate cancer: potential implications for PSMA-PET patient selection. Front Oncol 2024;14:1324631. doi:10.3389/fonc.2024.1324631. [Google Scholar] [PubMed] [CrossRef]
67. Hupe MC, Philippi C, Roth D et al. Expression of prostate-specific membrane antigen (PSMA) on biopsies is an independent risk stratifier of prostate cancer patients at time of initial diagnosis. Front Oncol 2018;8:623. doi:10.3389/fonc.2018.00623. [Google Scholar] [PubMed] [CrossRef]
68. Shih C-H, Hsieh T-Y, Sung W-W. Prostate-specific membrane antigen-targeted antibody-drug conjugates: a promising approach for metastatic castration-resistant prostate cancer. Cells 2025;14(7):513. doi:10.3390/cells14070513. [Google Scholar] [PubMed] [CrossRef]
69. Gogia P, Ashraf H, Bhasin S, Xu Y. Antibody-drug conjugates: a review of approved drugs and their clinical level of evidence. Cancers 2023;15(15):3886. doi:10.3390/cancers15153886. [Google Scholar] [PubMed] [CrossRef]
70. Petrylak DP, Kantoff P, Vogelzang NJ et al. Phase 1 study of PSMA ADC, an antibody-drug conjugate targeting prostate-specific membrane antigen, in chemotherapy-refractory prostate cancer. The Prostate 2019;79(6):604–613. doi:10.1002/pros.23765. [Google Scholar] [PubMed] [CrossRef]
71. Petrylak DP, Vogelzang NJ, Chatta K et al. PSMA ADC monotherapy in patients with progressive metastatic castration-resistant prostate cancer following abiraterone and/or enzalutamide: Efficacy and safety in open-label single-arm phase 2 study. The Prostate 2020;80(1):99–108. doi:10.1002/pros.23922. [Google Scholar] [PubMed] [CrossRef]
72. Milowsky MI, Galsky MD, Morris MJ et al. Phase 1/2 multiple ascending dose trial of the prostate-specific membrane antigen-targeted antibody drug conjugate MLN2704 in metastatic castration-resistant prostate cancer. Urol Oncol: Sem Orig Investigat 2016;34(12):530.e15–530.e21. doi:10.1016/j.urolonc.2016.07.005. [Google Scholar] [PubMed] [CrossRef]
73. de Bono JS, Fleming MT, Wang JS et al. Phase I study of MEDI3726: a prostate-specific membrane antigen-targeted antibody-drug conjugate, in patients with mCRPC after failure of abiraterone or enzalutamide. Clin Cancer Res 2021;27(13):3602–3609. doi:10.1158/1078-0432.CCR-20-4528. [Google Scholar] [PubMed] [CrossRef]
74. Banerjee S, Pillai MRA, Knapp FF. Lutetium-177 therapeutic radiopharmaceuticals: linking chemistry, radiochemistry, and practical applications. Chem Rev 2015;115(8):2934–2974. doi:10.1021/cr500171e. [Google Scholar] [PubMed] [CrossRef]
75. Sartor O, Bono JD, Chi KN et al. Lutetium-177-PSMA-617 for metastatic castration-resistant prostate cancer. N Engl J Med 2021;385(12):1091–1103. doi:10.1056/NEJMoa2107322. [Google Scholar] [PubMed] [CrossRef]
76. Hofman MS, Emmett L, Sandhu S et al. [177Lu]Lu-PSMA-617 versus cabazitaxel in patients with metastatic castration-resistant prostate cancer (TheraPa randomised, open-label, phase 2 trial. The Lancet 2021 Feb 27;397(10276):797–804. doi:10.1016/S0140-6736(21)00237-3. [Google Scholar] [PubMed] [CrossRef]
77. Azad AA, Bressel M, Tan H et al. Sequential [177Lu]Lu-PSMA-617 and docetaxel versus docetaxel in patients with metastatic hormone-sensitive prostate cancer (UpFrontPSMAa multicentre, open-label, randomised, phase 2 study. Lancet Oncol 2024;25(10):1267–1276. doi:10.1016/S1470-2045(24)00440-6. [Google Scholar] [PubMed] [CrossRef]
78. Morris MJ, Castellano D, Herrmann K et al. 177Lu-PSMA-617 versus a change of androgen receptor pathway inhibitor therapy for taxane-naive patients with progressive metastatic castration-resistant prostate cancer (PSMAforea phase 3, randomised, controlled trial. Lancet 2024;404(10459):1227–1239. doi:10.1016/S0140-6736(24)01653-2. [Google Scholar] [PubMed] [CrossRef]
79. Emmett L, Subramaniam S, Crumbaker M et al. [177Lu]Lu-PSMA-617 plus enzalutamide in patients with metastatic castration-resistant prostate cancer (ENZA-pan open-label, multicentre, randomised, phase 2 trial. Lancet Oncol 2024;25(5):563–571. doi:10.1016/S1470-2045(24)00135-9. [Google Scholar] [PubMed] [CrossRef]
80. Karimzadeh A, Heck M, Tauber R et al. 177Lu-PSMA-I&T for treatment of metastatic castration-resistant prostate cancer: prognostic value of scintigraphic and clinical biomarkers. J Nucl Med 2023 Mar;64(3):402–409. doi:10.2967/jnumed.122.264402. [Google Scholar] [PubMed] [CrossRef]
81. Hansen AR, Probst S, Beauregard JM et al. Initial clinical experience with [177Lu]Lu-PNT2002 radioligand therapy in metastatic castration-resistant prostate cancer: dosimetry, safety, and efficacy from the lead-in cohort of the SPLASH trial. Front Oncol 2024;14:1483953. doi:10.3389/fonc.2024.1483953. [Google Scholar] [PubMed] [CrossRef]
82. Hou L, Wang Y, Fu H et al. PSMA-targeted radioligand therapy with [177Lu]Lu-LNC1011 for metastatic castration-resistant prostate cancer: a pilot study. Eur J Nucl Med Mol Imaging 2025;52(11):4033–4043. doi:10.1007/s00259-025-07245-8. [Google Scholar] [PubMed] [CrossRef]
83. Dierks A, Gäble A, Rinscheid A et al. First safety and efficacy data with the radiohybrid 177Lu-rhPSMA-10.1 for the treatment of metastatic prostate cancer. J Nucl Med 2024 Mar;1(3):432–437. doi:10.2967/jnumed.123.266741. [Google Scholar] [PubMed] [CrossRef]
84. Shin D, Ha S, O JH et al. A single dose of novel PSMA-targeting radiopharmaceutical agent [177Lu]ludotadipep for patients with metastatic castration-resistant prostate cancer: phase I clinical trial. Cancers 2022;14(24):6225. doi:10.3390/cancers14246225. [Google Scholar] [PubMed] [CrossRef]
85. Vallabhajosula S, Goldsmith SJ, Kostakoglu L, Milowsky MI, Nanus DM, Bander NH. Radioimmunotherapy of prostate cancer using 90Y- and 177Lu-Labeled J591 monoclonal antibodies: effect of multiple treatments on myelotoxicity. Clin Cancer Res 2005;11(19):7195s–7200s. doi:10.1158/1078-0432.Ccr-1004-0023. [Google Scholar] [PubMed] [CrossRef]
86. Ling X, Latoche JD, Choy CJ et al. Preclinical dosimetry, imaging, and targeted radionuclide therapy studies of Lu-177-labeled albumin-binding, PSMA-targeted CTT1403. Mol Imaging Biol 2020;22(2):274–284. doi:10.1007/s11307-019-01404-8. [Google Scholar] [PubMed] [CrossRef]
87. Kim H, Tagawa ST, Sartor AO et al. PSMAddition: phase 3 trial of Lu-PSMA-617 plus standard of care (SoC) vs. soc alone in patients with metastatic hormone-sensitive prostate cancer. Int J Radiat Oncol Biol Phys 2024;120(2):e546. doi:10.1016/j.ijrobp.2024.07.1209. [Google Scholar] [CrossRef]
88. Eapen RS, Buteau JP, Jackson P et al. Administering [177Lu]Lu-PSMA-617 prior to radical prostatectomy in men with high-risk localised prostate cancer (LuTectomya single-centre, single-arm, phase 1/2 study. Eur Urol 2024;85(3):217–226. doi:10.1016/j.eururo.2023.08.026. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools