 Open Access
Open Access
ARTICLE
Oncological outcomes of I125 low dose brachytherapy in localized prostate cancer
1 Instituto Médico Tecnológico and Prostate Institute, Barcelona, 08028, Spain
2 Statistics Department, Universidad Nacional de Córdoba, Córdoba, 5000, Argentina
* Corresponding Author: Nuria Jornet. Email:
Canadian Journal of Urology 2026, 33(1), 93-103. https://doi.org/10.32604/cju.2026.069182
Received 17 June 2025; Accepted 22 December 2025; Issue published 28 February 2026
Abstract
Background: Low-dose rate (LDR) prostate brachytherapy is a recommended treatment of localized prostate cancer in current guidelines. The study aimed to determine biochemical relapse-free survival (BRFS) in patients treated with dynamic real-time low-dose rate (LDR) brachytherapy using Iodine 125 (I125). Methods: We retrospectively reviewed 499 patients with localized prostate cancer treated with I125 LDR real-time brachytherapy between 2003 and 2021. The mean patient age was 65 years (range: 45–84 years). Based on the National Comprehensive Cancer Network (NCCN) risk classification, 230 patients (46.1%) were categorized as low risk, 235 (47.1%) as intermediate risk, and 34 (6.8%) as high risk. Gleason scores were distributed as follows: 3+3 in 283 cases (56.7%), 3+4 in 157 cases (31.5%), 4+3 in 46 cases (9.2%), and 4+4 in 13 cases (2.6%). The mean follow-up was 70.5 months. Results: Tumor relapse was observed in 47 patients (9.4%) over a mean follow-up period of 6.26 years (SD 4.16). Local recurrence within the prostate occurred in 20 cases (4%). Patients with nadir PSA <0.2 ng/mL at 5 years of follow-up had a significantly lower incidence of tumor recurrence (3%) compared to those with a nadir PSA >0.2 ng/mL (21.9%) (p = 0.0001). Biochemical relapse-free (BRFS) rates at 5, 10 and 15 years were 96%, 91.5% and 88.9%, respectively. When stratified by NCCCN risk groups, 5-year BRFS was 96% in low risk, 98% in intermediate risk and 85% in high risk patients (p = 0.003). In multivariate analysis, only age at the time of brachytherapy (p = 0.009), initial PSA (p = 0.007) and Gleason grade (p = 0.007) were significantly associated with tumor recurrence. Cancer-specific survival and overall survival were 99.8% and 98.0%, respectively Conclusions: LDR with I125 has excellent long-term oncological outcomes for patients with low and intermediate-risk prostate cancer, in particular, patients achieving a nadir PSA <0.2 ng/mL at 5 years post-treatment.Keywords
Prostate cancer is one of the most frequently diagnosed malignancies in men and represents a major global health concern. For patients with localized disease, several curative treatment options exist, including surgery, external beam radiotherapy, and brachytherapy.1 Low-dose-rate (LDR) brachytherapy with permanent iodine-125 (I125) seed implantation has gained widespread acceptance as a highly effective, minimally invasive treatment modality, particularly for patients with low- and intermediate-risk tumors. LDR brachytherapy with I125 is one of the treatments recommended by the National Comprehensive Cancer Network (NCCN) for the treatment of localized prostate cancer.1
A significant proportion of patients opt for LDR brachytherapy due to its favorable side effect profile—specifically, the lower risk of erectile dysfunction and urinary incontinence—along with shorter recovery times and oncological outcomes that are comparable to, or even better than, those of radical prostatectomy or external beam radiation therapy.2
Although robotic-assisted surgery has become increasingly popular in recent years, it has yet not demonstrate superior oncological efficacy or functional outcomes compared to existing treatment modalities.3 A pivotal development in the field occurred in 1995 when Stone and Stock introduced the technique of dynamic real-time brachytherapy. This approach significantly improved the precision of seed placement, enhancing both oncological control and functional preservation.4
The primary objective of this study was to determine biochemical relapse-free survival (BRFS) in patients treated with dynamic real-time low-dose rate (LDR) brachytherapy using I125.
Secondary objectives included assessing overall survival (OS), prostate cancer-specific survival (PCSS), and identifying independent predictors of BRFS. Variables analyzed included NCCN risk group, Gleason score, clinical stage, age, pretreatment prostate-specific antigen (PSA), prior hormonal therapy, prostate volume, and radiation dose delivered to 90% of the prostate (D90).
Patient population and initial staging
Between 2003 and 2021, a total of 499 patients with localized prostate cancer were treated with real-time dynamic LDR brachytherapy using I125 loose seeds at Instituto Médico Tecnológico and Prostate Institute Barcelona. The study was approved by the Clinical Research Ethics Committee at the Hospital Universitari Vall d’ Hebron, Barcelona (session Number 701).
Clinical staging was performed with an abdominal computerized axial tomography (CAT) scan, bone scan and magnetic resonance imaging (MRI). For patients with a PSA greater than 20 ng/mL, a choline or prostate-specific membrane antigen positron emission tomography (PSMA PET) scan was performed to rule out metastasis.
Risk stratification and treatment protocol
Patients were stratified into risk groups based on NCCN guidelines.1 The low-risk group included patients with clinical stage T1c–T2a, PSA ≤10 ng/mL, and Gleason score 6. The intermediate-risk group comprised patients with clinical stage T2b, or PSA between 10 and 20 ng/mL, or Gleason score 7. The high-risk group included patients with clinical stage T2c or higher, PSA >20 ng/mL, or Gleason score ≥8. In cases where the T2 substage was not specified, patients with PSA <10 ng/mL and Gleason 6 were conservatively assigned to the low-risk category to avoid overestimation of risk.
External beam radiotherapy and hormonal treatment were offered to patients with high-risk tumors. Only patients who agreed to be treated with brachytherapy alone were included in this study. The majority of patients with high-risk tumors were older than 75 years.
An overview of patient demographics, clinical features and risk group distribution is presented in Table 1.
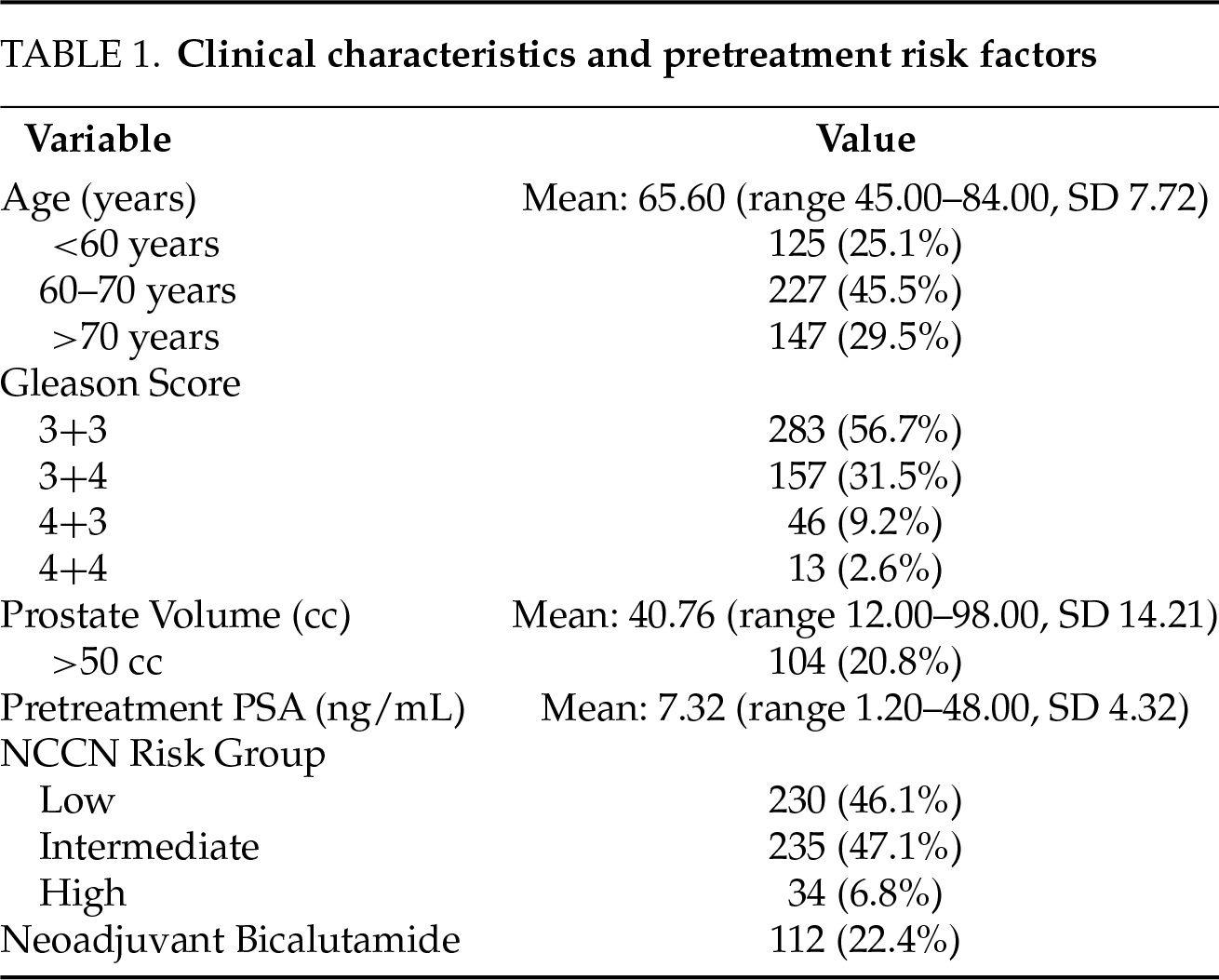
Neoadjuvant treatment with bicalutamide (50 mg/day) was administered to 112 patients (22.4%) prior to LDR brachytherapy.
Bicalutamide was used in patients with severe infravesical urinary obstruction (flow rate less than 10 mL/sec). These patients underwent prostate transurethral resection 2 months before brachytherapy to reduce obstruction. It was also used in patients with prostates larger than 70 mL to avoid interference from the pubic arch when inserting needles laterally into the prostate.
Treatment duration with bicalutamide was less than 3 months in 92.8% of patients.
Prior to the implant day, prostate volume was assessed using transrectal ultrasound or magnetic resonance imaging. The number and activity of I125 seeds were determined using a nomogram based on prostate volume.
Patients received a cleansing laxative solution the evening before and the morning of the procedure to prepare the rectum. On the day of implantation, spinal anesthesia combined with intravenous sedation was administered. The rectum was irrigated with saline solution to remove any residual fecal matter. A 14F Foley catheter was inserted to aid in the visualization of the urethra during treatment planning.
All implants were performed using a dynamic real-time technique with loose I125 seeds (Bard™, now part of BD, Athens, TX, USA) delivered via a Mick™ applicator (Mick Radio-Nuclear Instruments Inc., Bronx, NY, USA). The minimum prescribed peripheral dose was 160 Gy. All procedures were carried out by a consistent multidisciplinary team comprising a medical physicist, a radiation oncologist, and a urologist.
Patients were placed in the lithotomy position. Transverse ultrasound images were obtained at 5-mm intervals from the base to the apex of the prostate using a biplanar probe (BK™ Medical Aps, Herlev, Denmark). These images were imported into the VariSeed™ treatment planning system (Varian Medical Systems, Palo Alto, CA, USA), version 7.0 (before 2008) or version 8.0 (after 2008). The prostate, urethra, and rectum were contoured on the ultrasound images. Using the real-time module in the Variseed™, an initial plan for needle placement and seed distribution was obtained and optimized to meet the dosimetry goals: a minimum prostate dose (D100) of 160 Gy, maximum rectal dose of 90 Gy, and maximum urethral dose below 220 Gy. The volume of the prostate receiving more than 150% of the prescribed dose was monitored.
After placement of peripheral needles, new ultrasound images were acquired. Prostate, rectum, and urethra contours were updated to account for intraoperative changes such as prostate swelling, hematomas, or shifts in position due to needle insertion.
Each source was implanted under direct continuous ultrasound guidance, and dose calculations were updated in real-time. The actual position of each radioactive source was recorded in the treatment planning system.
All procedures were performed in the afternoon. Patients remained under observation overnight and were discharged the following morning after removal of the urinary catheter.
One month after implantation, a computed tomography (CT) scan was performed, and the images were imported into the VariSeed™ treatment planning system software for post-plan dosimetry analysis. The prostate, urethra, bladder, rectum, and seminal vesicles were contoured. The radioactive sources were identified, and the final dose distribution was calculated.
The following dose volume histogram (DVH) parameters were recorded: D90 (minimum dose received by 90% of the prostate), V100 (volume receiving 100% of the prescribed dose), and V150 (volume receiving 150% of the prescribed dose). For the organs at risk we recorded the maximum dose received to 0.1cc of the urethra, D0.1cc, the length of the urethra receiving more than 220 Gy, and the maximum dose to the rectum (D0.1cc) and the rectal volume receiving more than 90 Gy.
Clinical follow-up was conducted at baseline (pre-implantation) and at 3, 6, 12, 24, 48, and 60 months, post-treatment. After 5 years, patients were controlled yearly. PSA determination was performed at each visit.
No patients received adjuvant hormonal therapy or external beam radiation therapy following brachytherapy. Tamsulosin was routinely prescribed postoperatively for a minimum of three months following implantation to relieve lower urinary tract symptoms.
Biochemical recurrence was defined according to the Phoenix definition of nadir +2 ng/mL, with PSA bounces excluded from recurrence classification.5 In cases of biochemical recurrence, defined according to the Phoenix criteria, functional imaging with choline or PSMA PET/CT was performed to localize the site of relapse. If intra-prostatic recurrence was suspected, a confirmatory prostate biopsy was performed.
At the time of last clinical follow-up, patients were categorized into 6 outcome groups: 1—alive without recurrence; 2—alive with recurrence; 3—dead without cancer; 4—dead by prostate cancer; 5—alive with another cancer; 6—dead from another cancer.
All clinical data were entered into a Microsoft Excel database and analyzed using STATA™ (StataCorp LLC, College Station, TX, USA) version 18. For continuous variables, measures of center and dispersion (mean, median, and standard deviation) were calculated. The Shapiro-Willk test was then applied to assess normality.
For categorical variables, absolute and percentage distributions were calculated. Continuous variables were compared using the Student t test or Mann-Whitney test (depending on whether normality was met), and associations between categorical variables were determined using the Fisher exact test or chi-square test.
BRFS, CSS, and OS were calculated using the Kaplan-Meier method. Differences between Kaplan-Meier curves and comparisons between groups were examined using the log-rank test. Overall survival was measured from the date of implantation to the date of death from any cause.
Univariate and multivariate logistic regression analyses were performed to identify independent predictors of biochemical recurrence. Variables included in the models were: age, pre-treatment PSA, prostate volume, Gleason score, NCCN risk group, neoadjuvant hormonal therapy, prior prostate surgery, and D90.
In addition, a multivariate Cox proportional hazard regression analysis was conducted to evaluate factors associated with BRFS. Variables included were: clinical stage, Gleason score, age, pre-treatment PSA, D90, prostate volume, and the use of neoadjuvant hormonal therapy. All tests were two-sided, and a p < 0.05 was considered statistically significant.
At four weeks, post-implantation, post-plan dosimetry showed D90 ≤ 140 Gy in 58 patients (11.9%), 141–160 Gy in 173 patients (35.4%), and >160 Gy in 258 patients (52.7%). Post-plan CT-based dosimetry was not performed in 9 patients (1.8%).
The mean follow-up duration was 70.47 months (range: 1–240 months SD 48.88). A follow-up period of at least 60, 90, and 120 months was available in 259 (52%), 135 (27%), and 88 (18%) patients, respectively.
Tumor recurrence was observed in 47 patients (9.4%) at a mean time to recurrence of 6.26 years (SD: 4.16). The most common sites of recurrence were the prostate, bones, and iliac lymph nodes, as shown in Figure 1. Prostate recurrence was observed in 20 cases (4%), which is considered a true local recurrence post-brachytherapy.
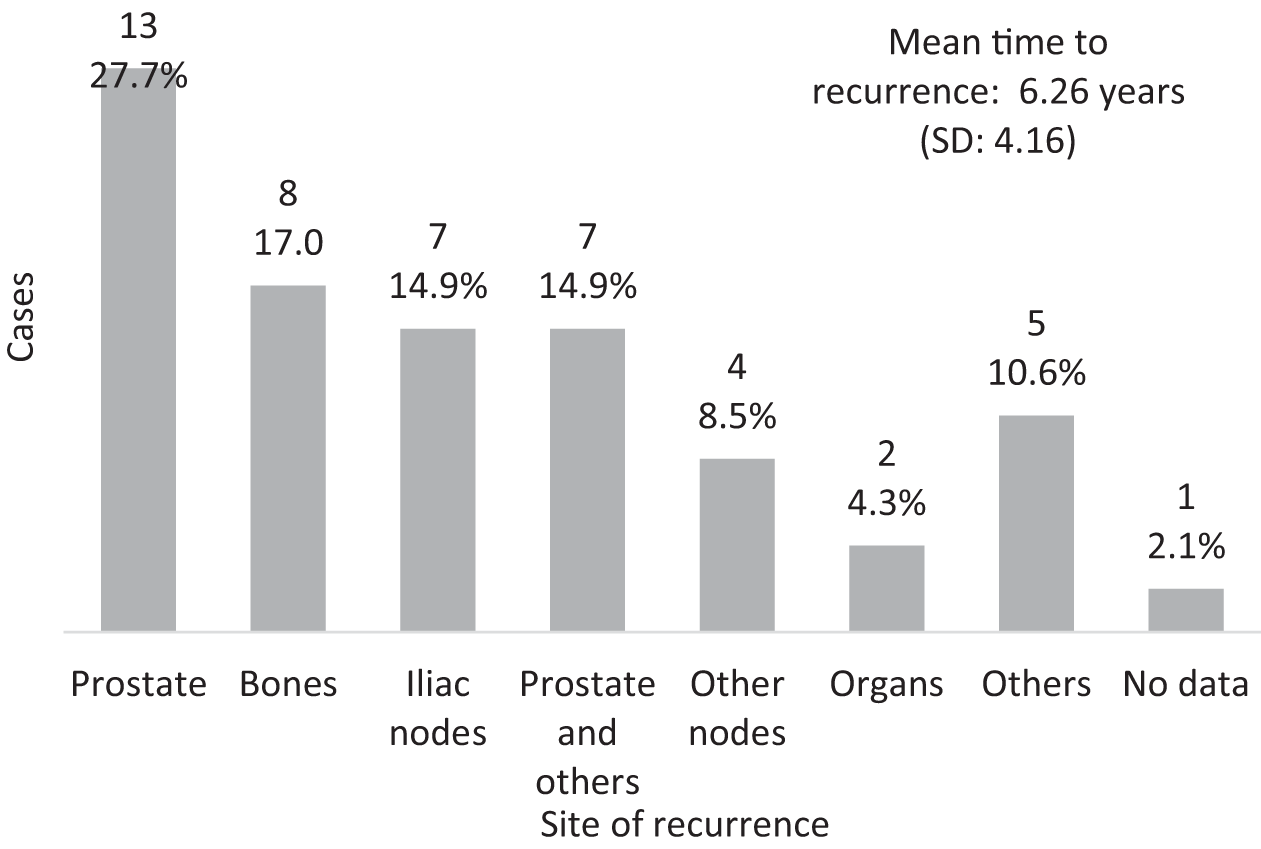
FIGURE 1. Sites of tumor recurrence
By the 4th and 5th year of follow-up, 71.7 and 86.8% of cases respectively had reached a PSA nadir <0.2 ng/mL. Recurrence occurred only in 3.0% of cases with a PSA nadir of <0.2 ng/mL, compared to 21.9% of cases with a PSA nadir >0.2 ng/mL. The difference was statistically significant (p = 0.0001) (Figure 2).
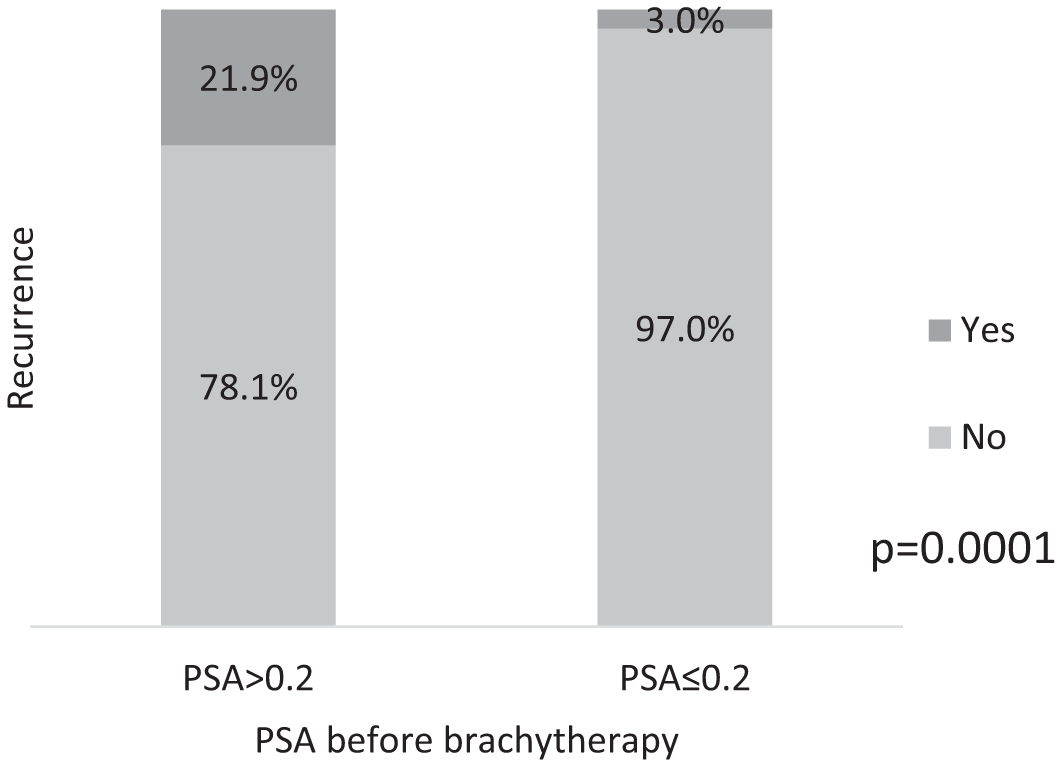
FIGURE 2. Incidence of tumor recurrence in patients with PSA nadir above or below 0.2 ng/mL (p = 0.0001)
The overall BRFS at 5, 10, and 15 years were 96%, 92%, and 91%, respectively (Figure 3a). When stratified by Gleason score (Figure 3b), patients with Gleason score 3+3 had BRFS rates of 96% and 95% at 5 and 10 years, respectively. In cases with a Gleason score 3+4, BRFS rates were 98% and 94% at 5 and 10 years, respectively. For Gleason score 4+3, BRFS rates of 93% and 87% at 5 and 10 years, while for Gleason score 4+4, BRFS rates at 5 and 10 years were 69% and 62%, respectively. Differences were statistically significant (p = 0.0001).
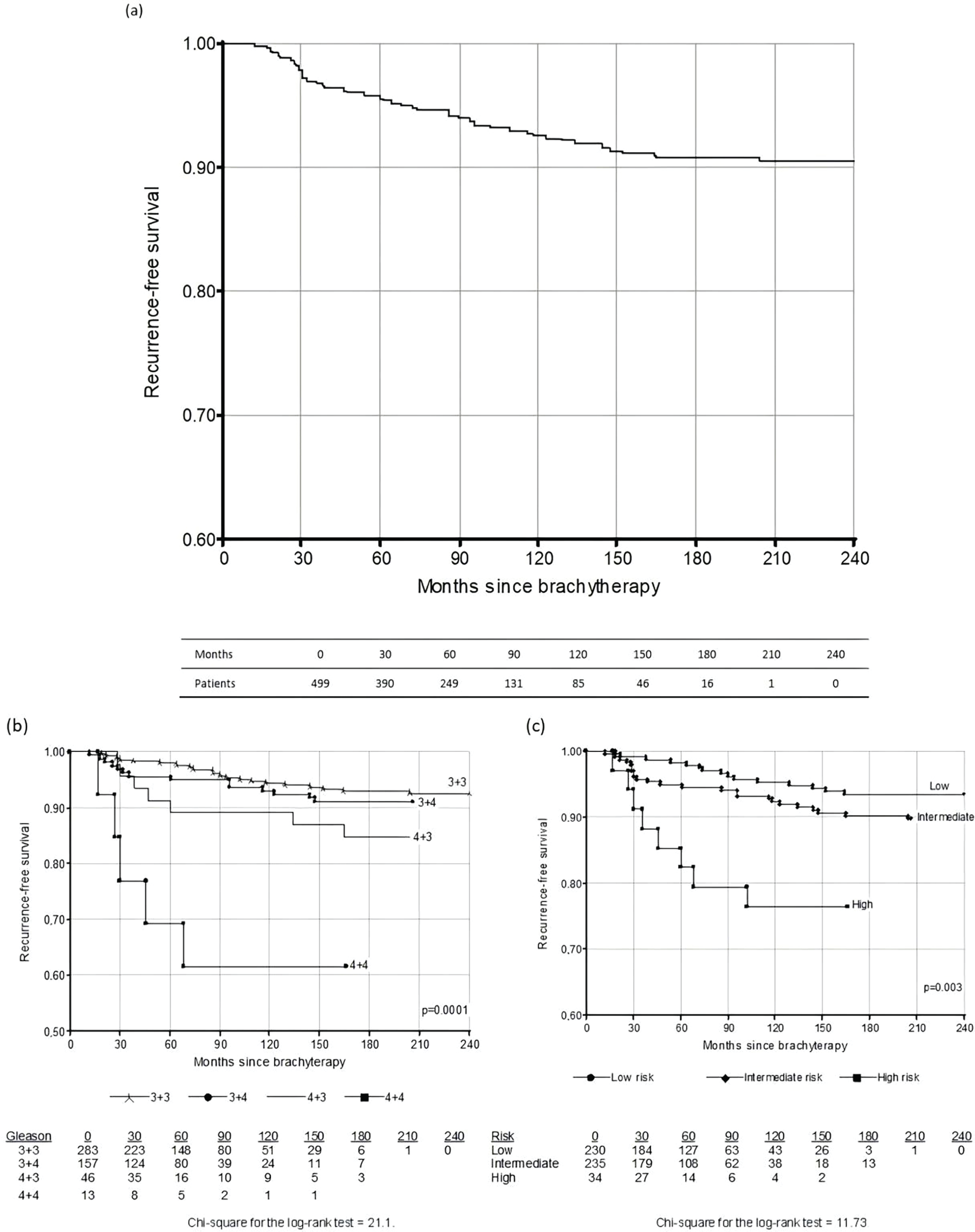
FIGURE 3. Kaplan-Meier recurrence-free survival curves. (a) global; (b) recurrence-free survival according to Gleason score (p = 0.0001); (c) recurrence-free survival according to risk group (p = 0.003)
Stratification by NCCN risk group showed that the 5-year BRFS was 96% in the low-risk group, 98% in the intermediate-risk group, and 85% in the high-risk group. At 10 years, BRFS was 95% for the low-risk group and 87% for the intermediate risk group (Figure 3c). The differences were statistically significant (p = 0.003).
An analysis of BRFS according to post-implant D90 showed recurrence in 17.2% of cases with D90 < 140 Gy, 9.2% in those with D90 between 141 Gy and 160 Gy and 7.4% in those with D90 > 160 Gy (Figure 4). Although this trend suggests improved outcomes with higher D90, the difference did not reach statistical significance (p = 0.063).
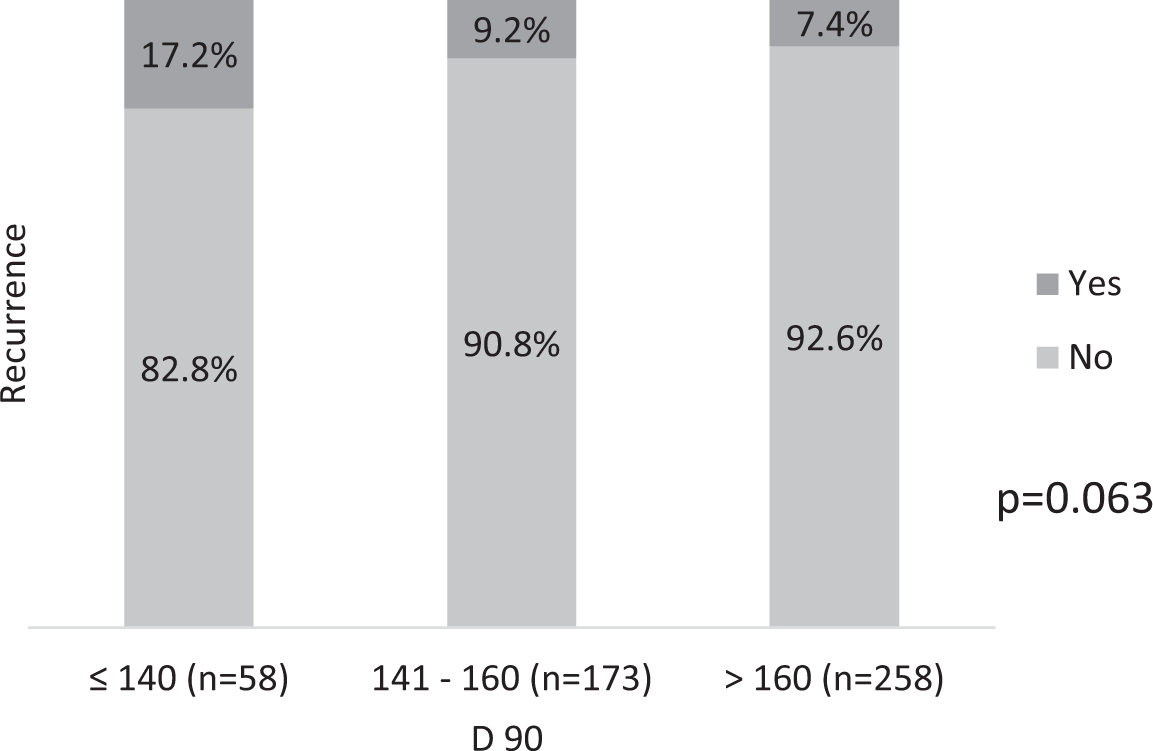
FIGURE 4. Tumor recurrence according to D90 (p = 0.063)
Regarding pretreatment PSA (Figure 5), recurrence was observed in 8% of patients with PSA 4–10 ng/mL, 19% of those with PSA between 10.1–20 ng/mL, and 20.0% in those with PSA >20 ng/mL. This difference was statistically significant (p = 0.020).
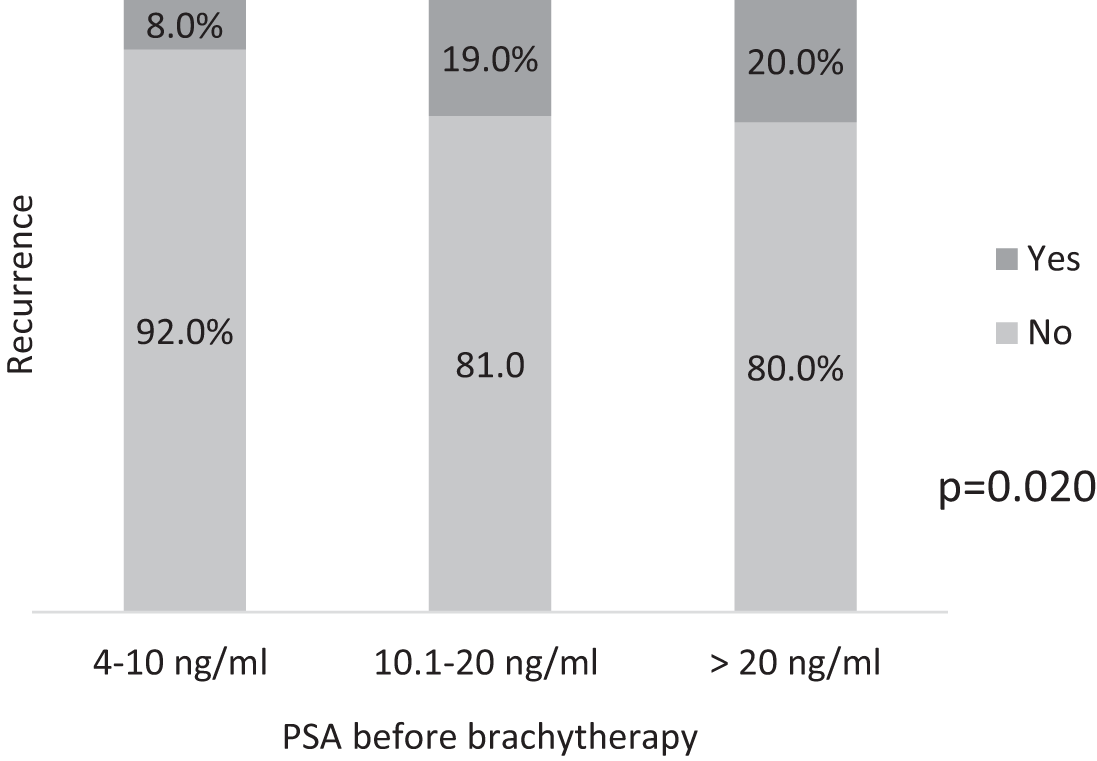
FIGURE 5. Recurrence according to PSA before treatment (p = 0.020)
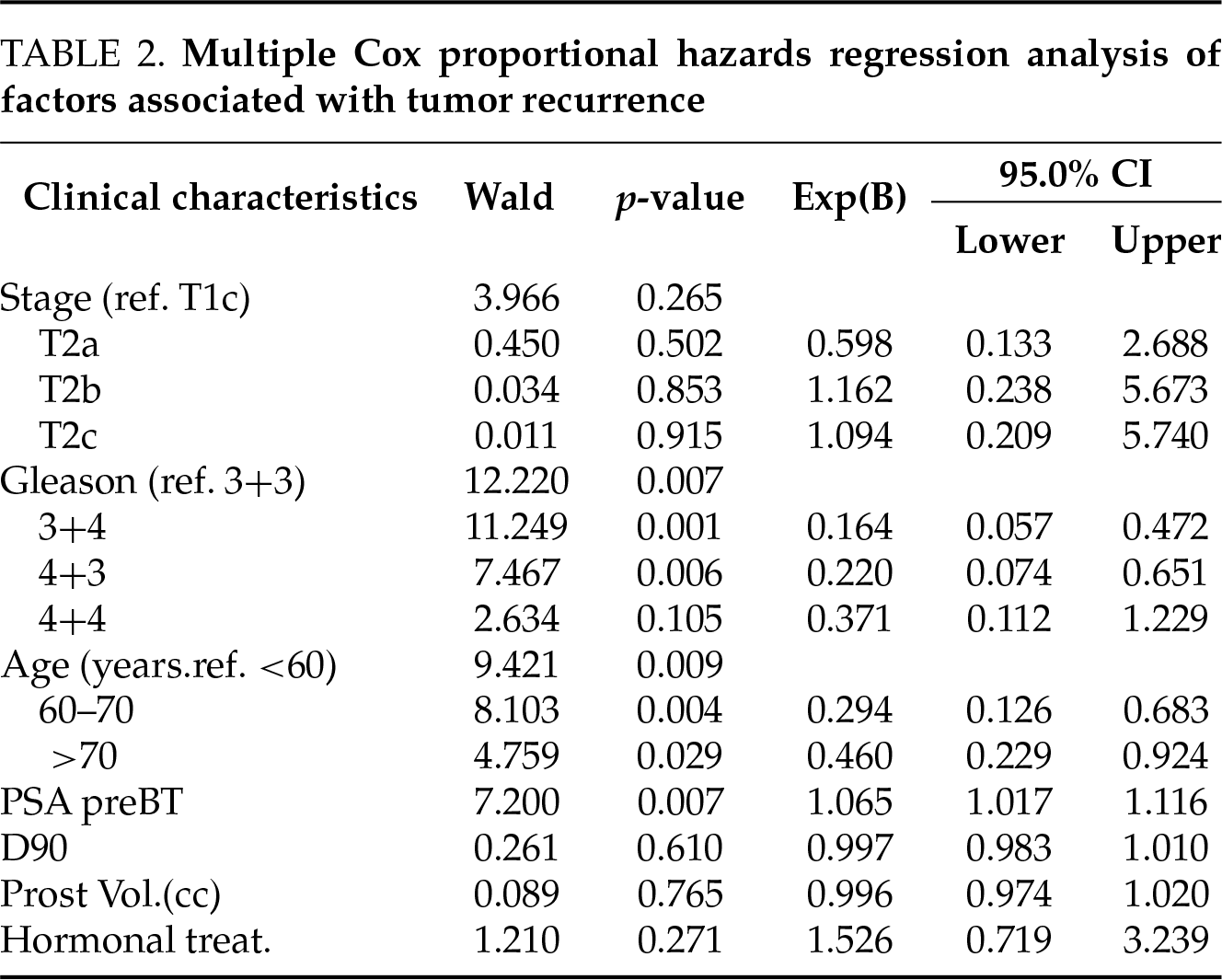
Multivariate analysis using Cox regression identified Gleason score, age, and pre-treatment PSA as independent prognostic factors for tumor recurrence (Table 2). The D90 was not found to be a significant predictor of tumor recurrence either when analyzed as a dichotomous variable (<180 vs. ≥180 Gy) or categorized into three levels (<140 Gy, 141–160 Gy, and >160 Gy).
At the time of final follow-up, two patients had been lost to follow-up. Among the remaining 497 patients followed up, five had died from non-cancerous causes, one had died from prostate cancer, and four had died from other cancers. The OS in the series was 98.0%.
A total of 453 patients (91.0%) were free of prostate cancer or had died from other causes. Only one patient died as a direct result of prostate cancer, resulting in a PCSS rate of 99.8%.
Over the years, seed implantation techniques have evolved significantly. Early procedures relied on a pre-planning technique, where ultrasound images were acquired several days before implantation. This approach made it difficult to replicate prostate positioning accurately on the treatment day, leading to suboptimal seed placement.6,7
In contrast, the dynamic real-time technique, introduced in the mid-to-late 1990s, integrates planning and implantation during the same surgical procedure, so the changes produced in the prostate by the placement of peripheral needles can be adjusted immediately before implanting the sources.8 This ensures more accurate seed localization under real-time ultrasound guidance. The result is improved dosimetry accuracy, with a more consistent delivery of the prescribed dose and notably higher BRFS at 5–8 years of follow-up (95%–97%) compared to the pre-planning approach (70%–91%).9–11 Moreover, the dynamic real-time technique enables a higher prescribed dose (160 Gy) without increasing urethral or rectal toxicity, whereas pre-planning protocols were limited to approximately 145 Gy.
Many earlier studies were heterogeneous, mixing different techniques (pre-planning and real-time) or seed types (stranded and loose),11,12 which complicated outcome comparison.
The distinguishing feature of our series is its methodological consistency: all 499 patients received dynamic real-time implantation with loose 125I seeds at 160 Gy, in a standardized prospective protocol.
Several studies have shown that the real-time implantation technique offers higher BRFS rates at 5 years after treatment than the p re-planning technique (70%–91% vs. 95%–97%), although the differences were not statistically significant.10,13,14
Interestingly, in our cohort, the intermediate-risk NCCN group showed better BRFS outcomes than the low-risk group, a finding that may be influenced by patient selection, sample size, or other unaccounted clinical variables, and which should therefore be interpreted with caution.
In order to compare the results of prostate cancer treatment with radical surgery vs. brachytherapy, Taussky et al. and Soyano et al. have shown that PSA level <0.2 ng/mL should be considered as an indicator of cure after brachytherapy instead of using the Phoenix definition.15,16
In our series, 71.7% and 86.8% of cases had a nadir PSA <0.2 ng/mL after 4 and 5 years of follow-up, respectively. The incidence of tumor recurrence was significantly lower in cases with a nadir PSA <0.2 ng/mL (3.0%) compared to cases with a nadir PSA >0.2 ng/mL (21.9%). In our study, the BRFS rate in patients with a nadir PSA <0.2 ng/mL was 97%. These data are consistent with other series showing that 10-year BRFS ranges from 96.9% to 98.7% in cases with PSA nadir <0.2 ng/mL.17–20 A study of 14,220 patients with localized prostate cancer treated with low-dose brachytherapy showed that 98.7% of cases with nadir PSA ≤0.2 ng/mL were free from recurrence after 10 years of follow-up.21
In our study, tumor recurrence was observed in 47 patients (9.4%), a figure similar to other recent studies, such as Pons et al.11 in Spain with 10.2%, Langley et al.22 in the United Kingdom, and Schubert et al.23 in Germany with tumor recurrence in 7.4% and 6.7%, respectively. Prostate tumor recurrence, which constitutes a local recurrence of treatment, was observed in 4% of patients in our series, a percentage similar to other recently published series with local recurrence in 3.3%–4.8% of cases.11,23
Early work by Stock and Stone in 2002 highlighted the importance of post-implant CAT scan dosimetry, showing that D90 > 140 Gy correlated with superior disease control.24 Our data showed a non-significant trend toward lower recurrence rates with increasing D90, but statistical significance was not reached, echoing mixed findings from Morris et al., who also did not find a correlation between the D90 value and the presence of recurrence and Tanaka et al., who observed that a D90 > 180 Gy was associated with a lower incidence of biochemical recurrence.12,25 The incidence of tumor recurrence was higher in cases with pre-treatment PSA level > 10 ng/mL, and the differences were statistically significant (p = 0.020).
In our series, the 5-year BRFS rate was 96%, which is comparable to that reported in earlier studies, such as Zelefsky et al.26 in 2007 (98%) and Morris et al.12 in 2013 (96.7% at 5 years and 94.1% at 10 years). Similar outcomes have also been observed in more recent series, including Peacock et al.27 in 2017 (95%), Chao et al.28 in 2018 (95%), Pons et al.14 in 2018 (92.7% at 5 years), and Amstrong et al.29 in 2024 (94% at 8 years). Stratified by risk group, we observed 5-year BRFS rates of 96%, 98%, and 85% in low, intermediate, and high risk groups, respectively (p = 0.003). These results are comparable to those obtained by other series.22,30,31
Multivariate Cox regression analyses identified age, Gleason score, and baseline PSA as independent prognostic factors for tumor recurrence, consistent with Prada et al.’s results.32
PCSS reached 99.8%, similar to that reported by Amstrong et al.29 (99.4%), Langley et al.22 (99%), Yaxley et al.17 (98%), and Fellin et al.33 (98%).
Our findings confirm that real-time dynamic LDR brachytherapy using loose I125 seeds offers excellent long-term oncological outcomes in patients with localized prostate cancer, particularly those with low and intermediate-risk disease. The BRFS rates observed at 5,10 and 15 years, combined with a PCSS of 99.8%, support its role as an effective and minimally invasive alternative to radical prostatectomy.
A notable strength of our study is the uniformity of treatment technique across all patients, with standardized implantation and dosimetry protocols. The majority of patients (86.8%) achieved a nadir PSA <0.2 ng/mL within five years, and this subgroup showed significantly lower recurrence rates (3%) compared to those with higher PSA nadir values (21.9%). These results align with emerging evidence suggesting that a nadir PSA <0.2 ng/mL may be a more accurate surrogate for long-term cure of patients treated with LDR brachytherapy than the Phoenix definition.
Although tumor recurrence was rare overall, younger patients (<60 years old) demonstrated better disease control, suggesting that age may be an independent prognostic factor. While dosimetry thresholds such as D90 > 160 Gy showed a trend toward improved outcomes, this did not reach significance in our cohort.
Real-time dynamic LDR brachytherapy is a highly effective and safe modality for the treatment of localized prostate cancer, achieving durable tumor control with minimal morbidity, and should be considered a first-line option in appropriately selected patients.
Acknowledgement
None.
Funding Statement
This research did not receive external funding.
Author Contributions
Gilberto Chéchile Toniolo: Conceptualization of the study and preparation of the initial manuscript draft; Núria Jornet: Critical review of the manuscript and authorship of the technique description from a medical physics perspective; Jady Rojas: Data collection, experimental implementation, and final approval of the submitted version; Natalia Tejedor: Data collection, experimental implementation, and final approval of the submitted version; Santiaga Carrara: Data collection and data analysis; Alicia Macagno: Statistical analysis and preparation of figures and tables; Teresa Brufau: Data collection, experimental implementation, and final approval of the submitted version. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials
All the data generated or analyzed during this study are included in this article.
Ethics Approval
All patients had provided consent for the use of their clinical information for scientific and research purposes at the time of data collection. This study is a retrospective observational analysis based exclusively on previously collected clinical data. No intervention was performed and no aspect of patient management or treatment was altered for the purposes of this research. In this project, all data were obtained from routine clinical practice and were fully anonymized prior to analysis, ensuring that no identifiable personal information was accessible to the researchers. The study was approved by the Clinical Research Ethics Committee at the Hospital Universitari Vall d’Hebron, Barcelona (session Number 701).
Conflicts of Interest
The authors declare no conflicts of interest to report regarding the present study.
References
1. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) Prostate Cancer Version 1.2025; 2024 Dec 4 [cited 2025 Sep 20]. Available from: https://www.scribd.com/document/752145559/NCCN-Guidelines-Prostate-Cancer-Version-1-2024. [Google Scholar]
2. Tsumura H, Tanaka N, Oguchi T et al. Direct comparison of low dose rate brachytherapy versus radical prostatectomy using the surgical definition of biochemical recurrence for patients with intermediate risk prostate cancer. Radiat Oncol 2022;17(1):71. doi:10.1186/s13014-022-02046-x. [Google Scholar] [PubMed] [CrossRef]
3. Giberti C, Gallo F, Schenone M, Cortese P, Gastaldi E, Becco D. Robotic prostatectomy versus brachytherapy for the treatment of low risk prostate cancer. Can J Urol 2017;24:8728–8733. doi:10.1016/s1569-9056(16)60685-1. [Google Scholar] [CrossRef]
4. Stone NN, Stock RG. Brachytherapy for prostate cancer: real-time three-dimensional interactive seed implantation. Tech Urol 1995;1(2):72–80. [Google Scholar] [PubMed]
5. Roach MT, Hanks G, Thames HJ, et al. Defining biochemical failure following radiotherapy with or without hormonal therapy in men with clinically localized prostate cancer: Recommendations of the RTOG ASTRO Phoenix consensus conference. Int J Radiat Oncol Biol. Phys. 2006;65(4):965–974. doi:10.1016/j.ijrobp.2006.07.010. [Google Scholar] [PubMed] [CrossRef]
6. Cox JD, Stetz J, Pajak TF. Toxicity criteria of the Radiation Therapy Oncology Group (RTOG) and the European organization for research and treatment of cancer (EORTC). Int J Radiat Oncol Biol Phys 1995;31(5):1341–1346. [Google Scholar] [PubMed]
7. Sylvester J, Blasko JC, Grimm P, Ragde H. Interstitial implantation techniques in prostate cancer. J Surg Oncol 1997;66(1):65–75. doi:10.1002/(sici)1096-9098(199709)66:1<65::aid-jso13>3.0.co;2-n. [Google Scholar] [CrossRef]
8. Stish BJ, Davis BJ, Mynderse LA, McLaren RH, Deufel CL, Choo R. Low dose rate brachytherapy. Transl Androl Urol 2018;7(3):341–356 doi: 10.21037/tau.2017.12.15. [Google Scholar] [PubMed] [CrossRef]
9. Zelefsky MJ, Yamada Y, Marion C, et al. Improved conformality and decerased toxicity with intraoperative computer-optimized transperineal ultrasound-guided prostate brachytherapy. Int J Radiat Oncol Biol Phys 2003;55(4):956–963 doi: 10.1016/s0360-3016(02)04142-1. [Google Scholar] [PubMed] [CrossRef]
10. Matzkin H, Chen J, German L, Mabjeesh N. Comparison between preoperative and real-time intraoperative planning 125I permanent prostate brachytherapy: long term clinical biochemical outcome. Radiot Oncol 2013;8:288 doi: 10.1186/1748-717x-8-288. [Google Scholar] [PubMed] [CrossRef]
11. Pons-Llanas O, Collado-Ballesteros E, Roldan-Ortega S, et al. Impact of real-time, dose-escaleted permanente seed implant brachytherapy in intermediate-risk prostate cancer. Rep Pract Oncol Radiother 2020;25(4):463–469. doi:10.1016/j.rpor.2020.04.014. [Google Scholar] [PubMed] [CrossRef]
12. Morris WJ, Keyes M, Spadinger I, et al. Population-based 10-year oncologic outcome after low dose-rate brachytherapy for low risk and intermediate-risk prostate cancer. Cancer 2013;119(8):1537–1546. doi:10.1002/cncr.27911. [Google Scholar] [PubMed] [CrossRef]
13. Guinot JL, Ricos JV, Tortajada MI, et al. Comparison of permanent 125I seeds implants with two different techniques in 500 cases of prostate cancer. J Contemp Brachyther 2015;7(4):258–264. doi:10.5114/jcb.2015.53525. [Google Scholar] [PubMed] [CrossRef]
14. Pons-Llanas O, Roldan-Ortega S, Celada-Alvarez F, et al. Permanent seed implant brachytherapy in low risk Prostate cancer: preoperative planning with 145 Gy versus real-time intraoperative planning with 160 Gy. Rep Practl Oncol Radiother 2018;23(4):290–297. doi:10.1016/j.rpor.2018.06.009. [Google Scholar] [PubMed] [CrossRef]
15. Taussky D, Lambert C, Meissner N, Bahary JP, Delouya G. Risk factors for biochemical recurrence after a tissue-ablative prostate-specific antigen <0,2 ng/mL. Brachytherapy 2018;17(5):794–798. doi:10.1016/j.brachy.2018.05.011. [Google Scholar] [PubMed] [CrossRef]
16. Soyano T, Yorozu A, Natsume N, et al. Time to achieve a prostate-specific antigen nadir of <0,2 ng/mL and related factors after permanent prostate brachytherapy. Brachytherapy 2021;20(1):29–37. doi:10.1016/j.brachy.2020.07.006. [Google Scholar] [PubMed] [CrossRef]
17. Yaxley WJ, Mackean J, Desai DJ, et al. Oncological and urinary outcome following low-dose-rate brachytherapy with a median follow up of 11.8 years. BJU Int 2022;130(Suppl 3):40–45. doi:10.1111/bju.15845. [Google Scholar] [PubMed] [CrossRef]
18. Takeuchi S, Iinuma K, Nakane K, et al. Direct comparison of two different definitios with biochemical recurrence after low-dose-rate brachytherapy for prostate cancer. Curr Oncol 2023;30(3):2792–2800. doi:10.3390/curroncol30030212. [Google Scholar] [PubMed] [CrossRef]
19. Ueno Y, Fukumori T, Kusuhara Y, et al. Prostate-specific antigen levels following brachytherapy impact late biochemical recurrence in Japanese patients with localized prostate cancer. Vivo 2023;37(2):738–746. doi:10.21873/invivo.13136. [Google Scholar] [PubMed] [CrossRef]
20. Boehle A, Zywietz D, Robrahn-Nitschke I, Koenig IR, Lusch A. Definition of cure after low-dose-rate brachytherapy in low-and intermediate-risk prostate cancer: Phoenix or surgical? Adv Radiat Oncol 2022;8(2):101112. doi:10.1016/j.adro.2022.101112. [Google Scholar] [PubMed] [CrossRef]
21. Crook JM, Tang C, Thames H, et al. A biochemical definition of cure after brachytherapy for prostate cancer. Radiother Oncol 2020;149:64–69. doi:10.1016/j.radonc.2020.04.038. [Google Scholar] [PubMed] [CrossRef]
22. Langley SEM, Soares R, Uribe J, et al. Long term oncological outcome and toxicity in 597 men aged ≤60 years at time of low-dose-rate brachytherapy for localized prostate cancer. BJU Int 2018;121(1):38–45. doi:10.1111/bju.13946. [Google Scholar] [PubMed] [CrossRef]
23. Schubert P, Strnad V, Höfler D, et al. Outcome of I-125 low-dose-rate brachytherapy in patients with localized prostate cancer: a comprehensive analysis from a specialized tertiary referral center. J Pers Med 2024;14(8):882. doi:10.3390/jpm14080882. [Google Scholar] [PubMed] [CrossRef]
24. Stock RG, Stone NN. Importance of post-implant dosimetry in permanent prostate brachytherapy. Eur Urol 2002;41(4):434–439. doi:10.1016/s0302-2838(02)00018-0. [Google Scholar] [PubMed] [CrossRef]
25. Tanaka N, Nakai Y, Asakawa I, et al. Local dose (biologically effective dose ≥180 Gy2) is an important predictor of biochemical recurrence in patients undergoing low-dos-rate brachytherapy. Int J Urol 2022;29(12):1560–1568. doi:10.1111/iju.15051. [Google Scholar] [PubMed] [CrossRef]
26. Zelefsky MJ, Yamada Y, Cohen GN, et al. Five-year outcome of intraoperative conformal permanent I-125 interstitial implantation for patients with clinically localized prostate cancer. Int J Radiat Oncol Biol Phys 2007;67(1):65–70. doi:10.1016/j.ijrobp.2006.08.030. [Google Scholar] [PubMed] [CrossRef]
27. Peacock M, Martell K, Taggar A, et al. Institutional long-term outcomes at the first Canadian center performing intraoperatively planned low-dose-rate brachytherapy alone in low- and intermediate-risk prostate cancer. Brachytherapy 2017;16(4):822–830. doi:10.1016/j.brachy.2017.03.011. [Google Scholar] [PubMed] [CrossRef]
28. Chao M, Spencer S, Guerrieri M, et al. A single institution analysis of low-dose-rate brachytherapy: 5-year reported survival and late toxicity outcomes. J Contemp Brachytherapy 2018;10(2):155–161. doi:10.5114/jcb.2018.75600. [Google Scholar] [PubMed] [CrossRef]
29. Amstrong A, Huong H, Tacey M, et al. Low-dose-rate brachytherapy and long-term treatment outcomes in patients younger than 60 years of age. J Contemp Brachytherapy 2024;16(1):6–11. doi:10.5114/jcb.2024.135630. [Google Scholar] [PubMed] [CrossRef]
30. Lazarev S, Thompson MR, Stone NS, Stock RG. Low-dose-rate brachytherapy for prostate cancer: outcomes at >10 years of follow-up. BJU Int 2018;121(5):781–790. doi:10.1111/bju.14122. [Google Scholar] [CrossRef]
31. Nakiri M, Ogasawara N, Kurose H, et al. Clinical outcomes of iodine-125 low-dose-rate brachytherapy for localized prostate cancer: a single-institution review in Japan. J Contemp Brachytherapy 2022;14(2):157–168. doi:10.5114/jcb.2022.115380. [Google Scholar] [CrossRef]
32. Prada PJ, Juan G, González-Suárez H et al. Prostate-specific antigen relapse-free survival and side-effects in 734 patients with up to 10 years of follow-up with localized prostate cancer treated by permanent iodine implants. BJU Int 2010;106(1):32–36. doi:10.1111/j.1464-410x.2009.09096.x. [Google Scholar] [PubMed] [CrossRef]
33. Fellin G, Mirri MA, Santoro L, et al. Low dose rate brachytherapy (LDR-BT) as monotherapy for early stage prostate cancer in Italy: practice and outcome analysis in a series of 2237 patients from 11 institutions. Br J Radiol 2016;89(1065):20150981. doi:10.1259/bjr.20150981. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools