 Open Access
Open Access
ARTICLE
Photocatalytic Performance of Kaolin/Cu2CdSnS4 Composite Synthesized via Solvothermal Route
School of Materials Science and Engineering, Yancheng Institute of Technology, Yancheng, China
* Corresponding Author: Hao Guan. Email:
Chalcogenide Letters 2026, 23(2), 7 https://doi.org/10.32604/cl.2026.075941
Received 11 November 2025; Accepted 27 January 2026; Issue published 28 February 2026
Abstract
Cu2CdSnS4 (CCTS) has positioned itself as an efficient option used as a photocatalyst, while kaolin is an ideal catalyst carrier material. In the study, kaolin/Cu2CdSnS4 composite catalyst is effectively synthesized by a one-step solvothermal route and applied to remove methylene blue (MB), a typical pollutant in the textile industry. The structural, morphological, and photocatalytic properties of the composite catalyst are characterized by XRD, SEM, FTIR, PL, etc. The results show that a pure kaolin/Cu2CdSnS4 composite is obtained. The flower-like structure of Cu2CdSnS4 exhibits higher photocatalytic activity due to its relatively large specific surface area, while kaolin creates an ideal carrier. The band gap of the as-obtained kaolin/Cu2CdSnS4 composite is estimated to be 1.69 eV. MB’s degradation efficiency under simulated solar light irradiation is around 94% within 100 min, with a high rate constant of 0.03 min−1. The recyclability and stability of the composite are investigated. Eventually, the mechanism of photocatalysis was explained. This study depicts that kaolin/Cu2CdSnS4 composite is a potential material for effective solar light photocatalytic application.Keywords
Industrial water pollution containing dyes without proper pretreatment has become an ever-increasing challenge, therefore, many new environmentally-friendly technologies in wastewater treatment employing physical, chemical, and biological methods to remove organic contaminants have attracted the interest of many researchers. Among the successful methods, photocatalysis has drawn great attention due to its efficiency, simplicity, and applicability, which can degrade organic contaminants using light energy in the presence of a catalyst. However, the need for UV light for photocatalyst activation limits its practical application due to its low content in solar light; thus, the absorption for improving visible light is enormously important. Many semiconductor materials have been studied as catalysts due to their large absorption range in the visible region. However, traditional photocatalytic materials are hard to simultaneously meet the requirements of broad-spectrum response, high efficiency, durability, and cost-effectiveness for sunlight. Metal sulfides such as In2S3, CdS, and PbS have been widely studied as visible-light-driven photocatalysts because of their narrow band gap. As a novel quaternary material, Cu2ZnSnS4 (CZTS) has earned special interest among researchers for photocatalysis applications owing to its suitable band gap of around 1.5 eV as well as a large absorption coefficient (>104 cm−1) in the visible light region, well carrier transport properties, and adjustable bandgap, etc. [1]. Cu2CdSnS4 with a bandgap of 1.4 eV, as well as a large absorption coefficient of 105 cm−1, has gotten attention as a proper semiconductor photocatalyst because of its similar structure to CZTS [2]. To date, Cu2CdSnS4 compounds have been prepared by various methods, including solvothermal synthesis, microwave-assisted route, wet chemical technique, successive ionic layer adsorption and reaction technique, thermal decomposition method, and direct liquid coating technology, etc. [3,4,5,6,7]. Meanwhile, Cu2CdSnS4 nanoparticles are also researched in the field of photocatalysis and thin film solar cells. However, the aggregation tendency of Cu2CdSnS4 nanoparticles may reduce specific surface area and limit adsorption capacity, and conventional synthesis methods are often energy-intensive and environmentally hazardous [8]. These drawbacks have driven recent intensive research into seeking a simple and effective preparation method and powder dispersion process. In order to achieve better dispersion, Kaolin (Al2Si2O5(OH)4) is introduced during the solvothermal synthesis. Kaolin is a common mineral in nature. The unique two-dimensional layer structure makes it have a high surface area and good adsorption capacity, indicating an ideal catalyst carrier material [9,10,11]. In the last two decades, kaolin has been considered as an ideal carrier for constructing composite catalysts to degrade pollutants under UV/visible light irradiation [12,13,14,15]. As far as we know, the study of Cu2CdSnS4 loading onto kaolin has not been reported until recently.
In this work, a solvothermal method is adopted to prepare kaolin/Cu2CdSnS4 composite. Then, the photocatalytic performance of the obtained kaolin/Cu2CdSnS4 composite is investigated by degrading MB as a pollutant model under visible light.
Kaolin (Al2Si2O5(OH)4), Cu(NO3)2·3H2O, CdCl2·2.5H2O, SnCl2·2H2O and H2NCSNH2 are obtained from Sinopharm Chemical Reagent Co. and are of analytical grade. Based on the previous work, Al2Si2O5(OH)4 (0.1g), Cu(NO3)2·3H2O (0.002mol), CdCl2·2.5H2O (0.001 mol), SnCl2·2H2O (0.001 mol) and H2NCSNH2 (0.006 mol) are dispersed in 80 mL ethylene glycol (EG) under the intensive stirring at room temperature for 2 h, then the solution is transferred into autoclaves and maintained at 200°C for 12 h. The autoclaves are naturally cooled down to room temperature. Finally, the produced samples are collected, washed with deionized water, and dried in vacuum at 80°C for 3 h to obtain the kaolin/Cu2CdSnS4 composite.
The phase compositions are measured using X-ray diffraction (X-Pert3 Powder). The surface morphology is observed using scanning electron microscope (Nova Nano SEM 450). The Fourier transform-infrared (FT-IR) spectra are tested by infrared spectrometer (JASCO 3600). The photoluminescence (PL) spectra are analyzed using spectrofluorophotometer (RF-5301 PC). The optical and photodegradation performances are obtained using UV-VIS-NIR spectrometer (UV2450).
The phase compositions of kaolin, Cu2CdSnS4 and kaolin/Cu2CdSnS4 samples are acquired using XRD analysis, as shown in Fig. 1. It is noticed that the main reflection peaks at angles of 2θ = 28.1°, 46.9° and 55.8° can be indexed to (112), (220) and (312) crystal planes of Cu2CdSnS4 structure, in agreement well with JCPDS 29-0537 card [16]. The average crystallite size estimated from the FWHM of the 28.1° peak using the Debye-Scherrer equation is around 45 nm. Meanwhile, the other diffraction peaks appeared at (2θ) values of 16.4°, 26.2°, 31.2°, 33.2°, 35.4°, 39.3°, 40.8°, 42.6°, 53.6°, 57.5° and 60.8° are well-matched with kaolin structure. All of the diffraction peaks for kaolin and Cu2CdSnS4 are present in the XRD pattern of the composite, with no detectable impurities, indicating the successful formation of kaolin/Cu2CdSnS4 composite, and the kaolin structure does not change in the reaction process. Simultaneously, we can observe that the diffraction peaks of kaolin/Cu2CdSnS4 compared to their pure substances are lower, it is indicated that the crystallinity of kaolin/Cu2CdSnS4 composite decreases obviously, revealing a small particle size.
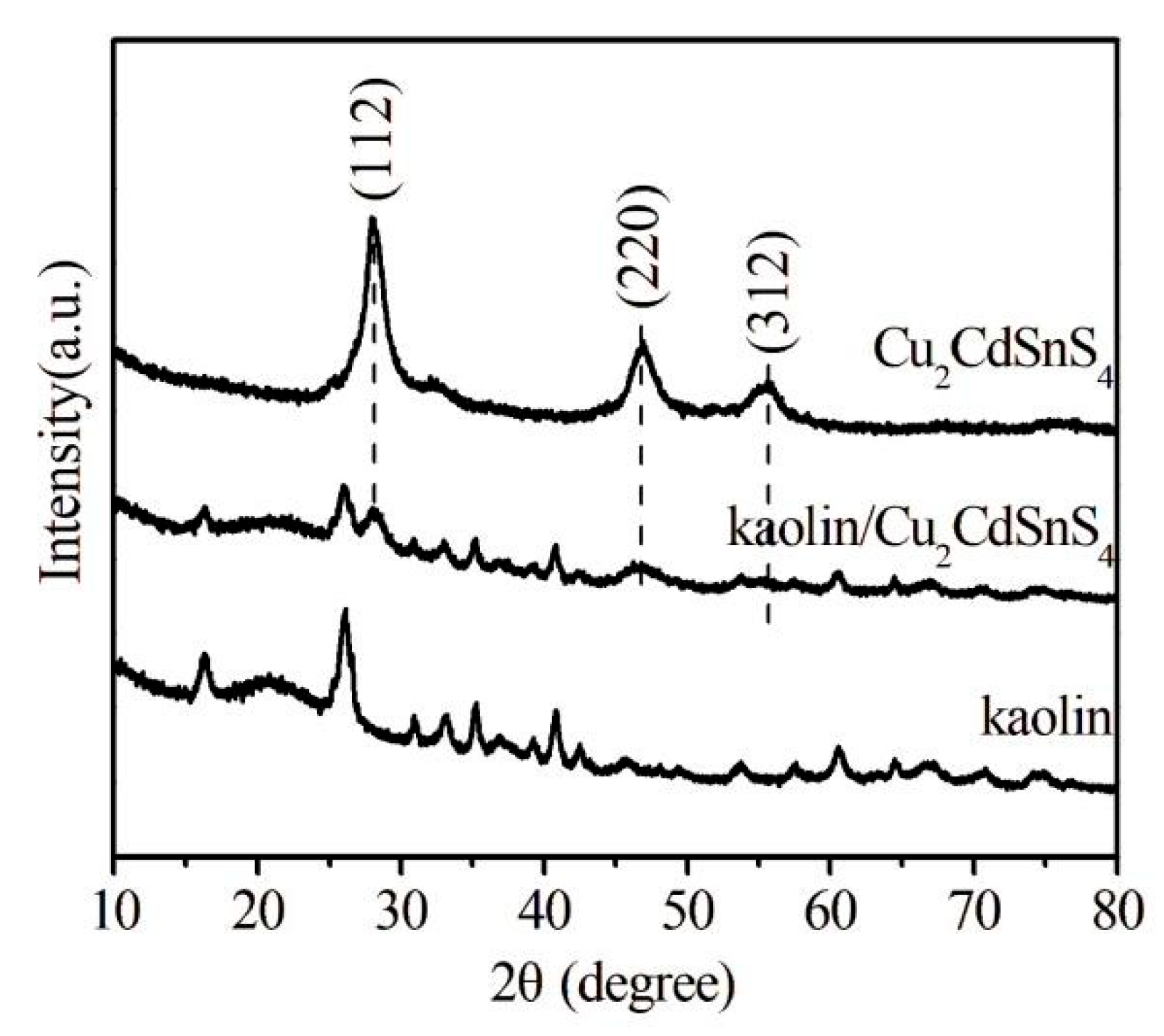
Figure 1: XRD patterns of Cu2CdSnS4, kaolin, and kaolin/Cu2CdSnS4 samples.
Fig. 2 shows the SEM images of all synthesized samples. Pristine Cu2CdSnS4 particles display flower-like hierarchical structures with the peak thickness of about 40–50 nm, in agreement with the result in literature [6]. The kaolin appears as agglomerates of stacked platelets with a thickness of approximately 0.2 μm. Each of these platelets is considered a disposition of several layers, which can supply more adhesion sites for Cu2CdSnS4. For kaolin/Cu2CdSnS4 composite, after introducing kaolin, the particles of Cu2CdSnS4 seem to be distributed on the surface of kaolin. It is indicated that kaolin, as a carrier, effectively inhibits the formation of large-scale aggregates, which is beneficial to photocatalysis.

Figure 2: SEM images of (a) Cu2CdSnS4, (b) kaolin, and (c) kaolin/Cu2CdSnS4 samples.
The FTIR spectra of Cu2CdSnS4 and kaolin/Cu2CdSnS4 composite is displayed in Fig. 3. The absorption bands occur at 3414 cm−1 (characteristic of thiourea or water), 2917 cm−1 (S-H thiol functional group or -C=CH- stretching mode), 1632 cm−1 (OH bending), 1092 cm−1 (metal-thiourea complex), 816 cm−1 (C-S stretching or Al-O-Si bond) and 555 cm−1 (Al-O-Si bond) [17,18,19]. Compared to pure Cu2CdSnS4, the peak intensity of kaolin/Cu2CdSnS4 composite is more apparent, indicating that the prepared composite samples require higher infrared vibration energy, and the internal chemical bonds of Cu2CdSnS4 become more stable after dispersion on the surface of kaolin.
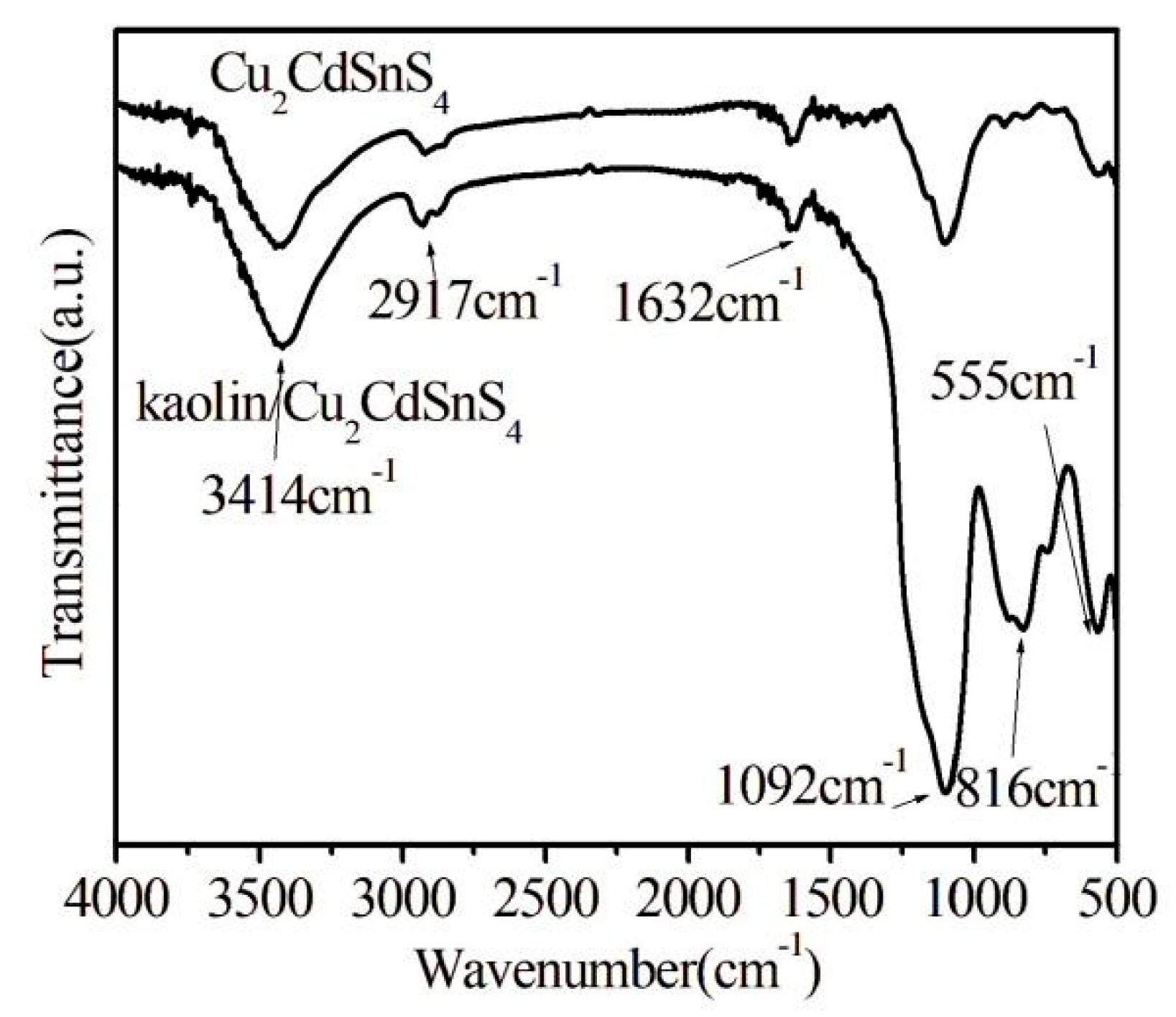
Figure 3: FTIR spectra of Cu2CdSnS4 and kaolin/Cu2CdSnS4 samples.
It is well known that the photocatalytic properties of many photocatalysts mainly depend on their optical characteristics. The band gap is one of the optical characteristics that can be detected based on Kubelka-Munk theory. The calculated values of all prepared products using Tauc formula by plotting the linear region of (αhν)2 vs. hν are exhibited in Fig. 4. It can be observed that the band gap of Cu2CdSnS4 is estimated at 1.65 eV while the one of the Kaolin is around 3.22 eV, in agreement with those reported in literature [20,21]. For the kaolin/Cu2CdSnS4 composite, the band gap is around 1.69 eV, slightly more significant than pure Cu2CdSnS4, implying the usage of full spectrum of visible light for good photocatalytic activity.
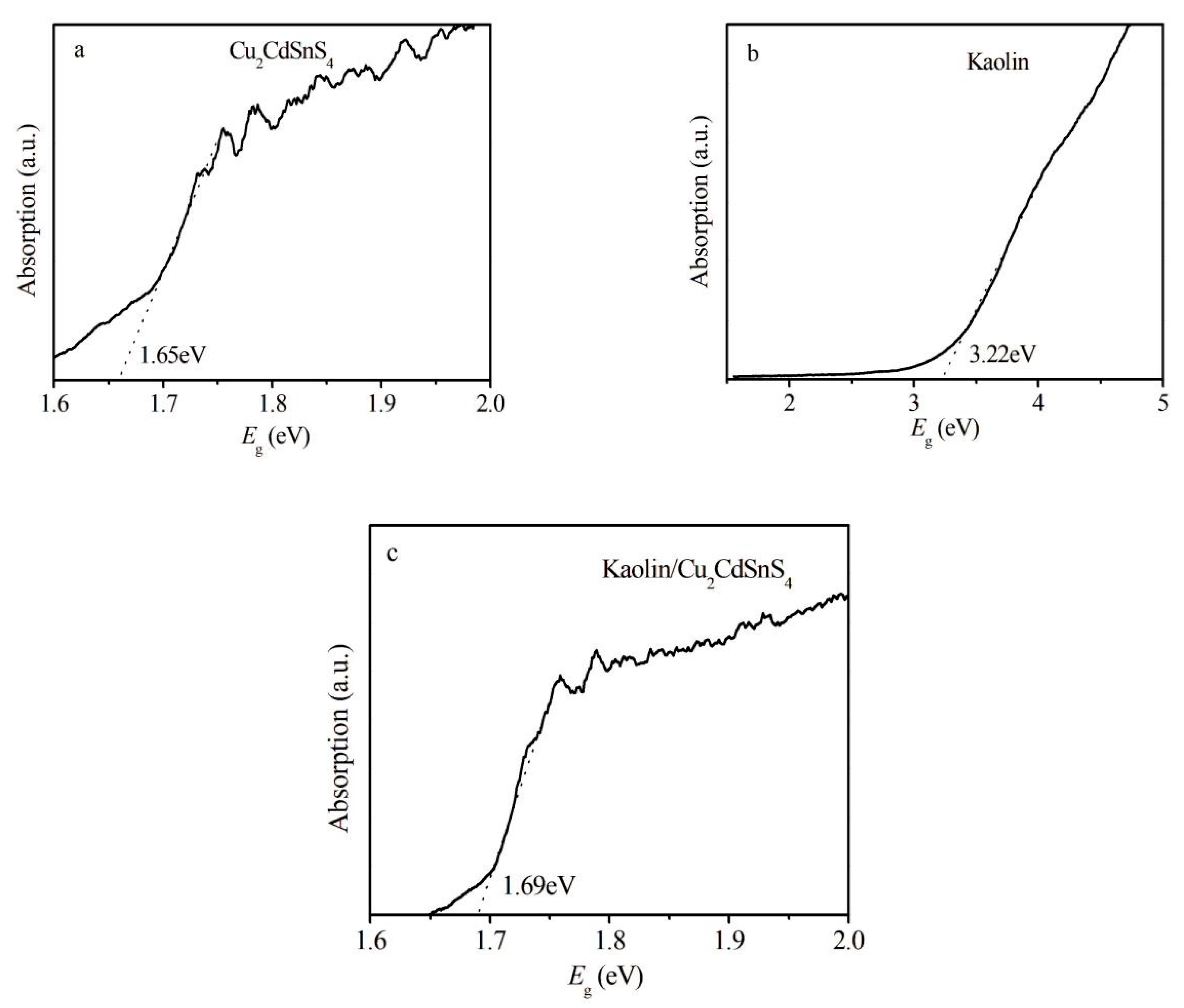
Figure 4: Optical bandgap estimations of (a) Cu2CdSnS4, (b) kaolin and (c) kaolin/Cu2CdSnS4 samples.
For catalysts, the spatial separation and recombination behavior of photo-induced carriers plays a key role in the photodegradation process. The fluorescence intensity can reflect the probability of recombining photo-induced electrons and holes. Photoluminescence (PL) spectroscopy can be utilized to scrutinize the recombination ability of photogenerated charges. The PL spectra of Cu2CdSnS4 and kaolin/Cu2CdSnS4 are shown in Fig. 5. The results show that the intensity of PL emission peak of kaolin/Cu2CdSnS4 composite is lower reduced compared to pure Cu2CdSnS4, indicating that the combination of kaolin/Cu2CdSnS4 can effectively inhibit the recombination of electrons and holes. Therefore, Kaolin/Cu2CdSnS4 composite is conducive to the transportation and separation of photogenerated electrons-holes, and is expected to exhibit high photocatalytic activity in the visible light.
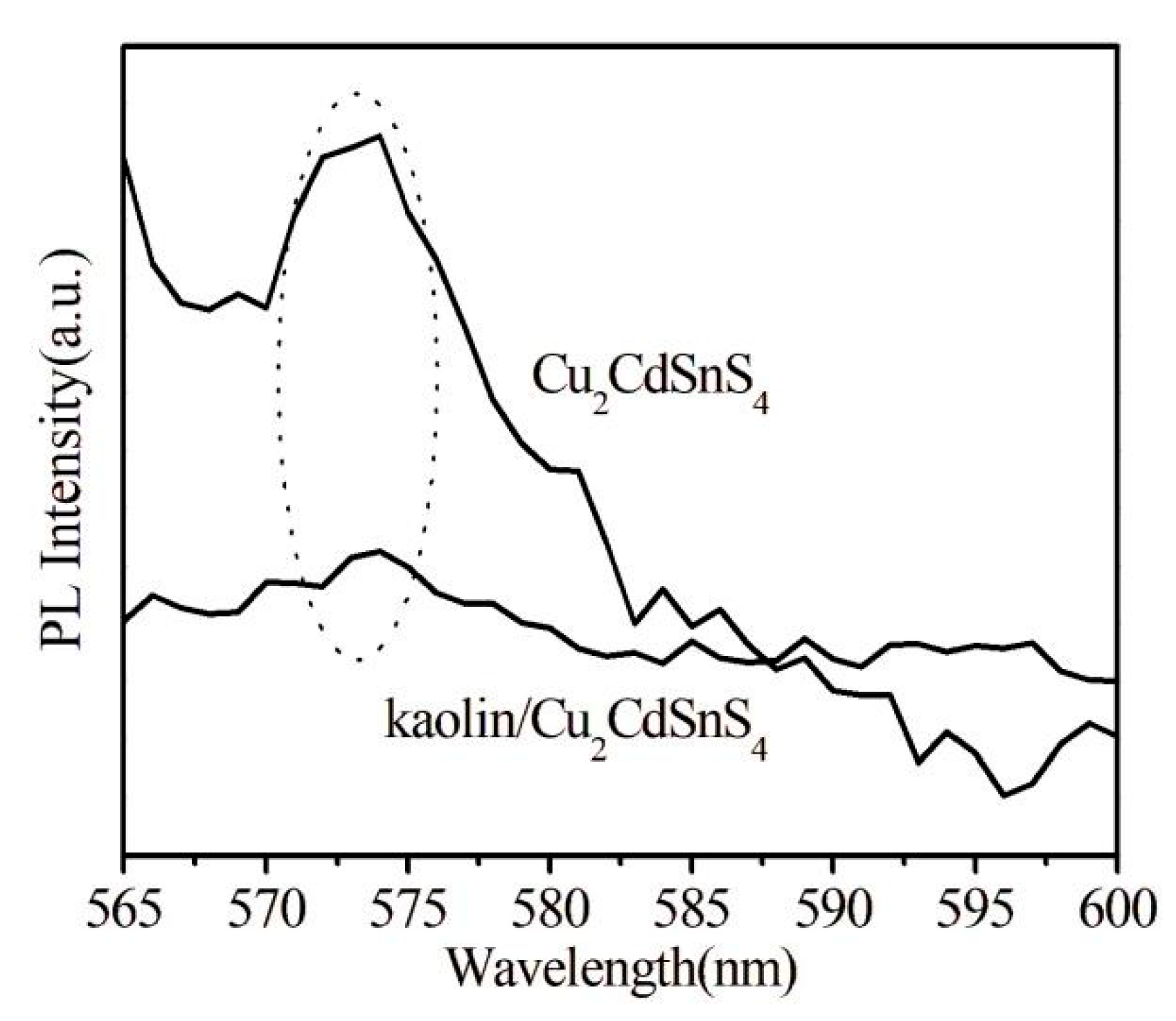
Figure 5: Photoluminescence spectra of Cu2CdSnS4 and kaolin/Cu2CdSnS4 samples.
The photocatalysis performances of the synthesized products are characterized by degrading methylene blue (MB) as a model for industrial wastewater under the irradiation of visible light for different times. The efficiencies of degradation against the times of light exposure are presented in Fig. 6a. The prepared samples show no significant adsorption for MB in a dark environment for 30 min, implying that MB is a non-biodegradable dye. After 100 min illumination, MB is stable without a photocatalyst and decomposes by only about 2%. When Cu2CdSnS4 is added as a catalyst, the MB degradation can increase to 79%. Furthermore, the addition of kaolin/Cu2CdSnS4 composite can decompose about 94%, which may be caused by the large surface area as well as the proper band gap value. Compared to other photocatalysts reported in the literature, kaolin/Cu2CdSnS4 exhibits higher photocatalytic performance than in previous literature, as shown in Table 1. The result proves that Cu2CdSnS4 material opens up a door to visible-light photocatalytic application, and kaolin plays an important role in providing active sites for catalytic Cu2CdSnS4. The reaction data are fitted by the pseudo-first-order reaction kinetics model: ln(C0/C) = kt + b, where k, C0, and C are the apparent reaction rate constant, MB concentration after adsorption equilibrium and at illumination time t, respectively. As shown in Fig. 6b, the rate constant k is estimated to be 0.01 min−1 for Cu2CdSnS4 while that of kaolin/Cu2CdSnS4 increase to 0.03 min−1. It is clear that kaolin/Cu2CdSnS4 composite exhibits the higher reaction rate, inducing the good photocatalytic performance. The cycling performance of kaolin/Cu2CdSnS4 composite for the degradation of MB is further examined. It is exhibited in Fig. 6c that the degradation efficiency decreases from 94% to 88% under the identical environments after reuse three times, which may be caused by the reduction of active sites. However, the degradation efficiency is still excellent, indicating reuse stability of kaolin/Cu2CdSnS4 photocatalyst. Furthermore, Fig. 6d shows that the XRD patterns of kaolin/Cu2CdSnS4 composite after use has no obviously change compared to that before use, confirming the structural stability, which is in agreement with SEM image of kaolin/Cu2CdSnS4 composite after use (Fig. 6e).
Table 1: The similarity of the photocatalytic activity in MB photodegradation from kaolin/Cu2CdSnS4 photocatalyst.
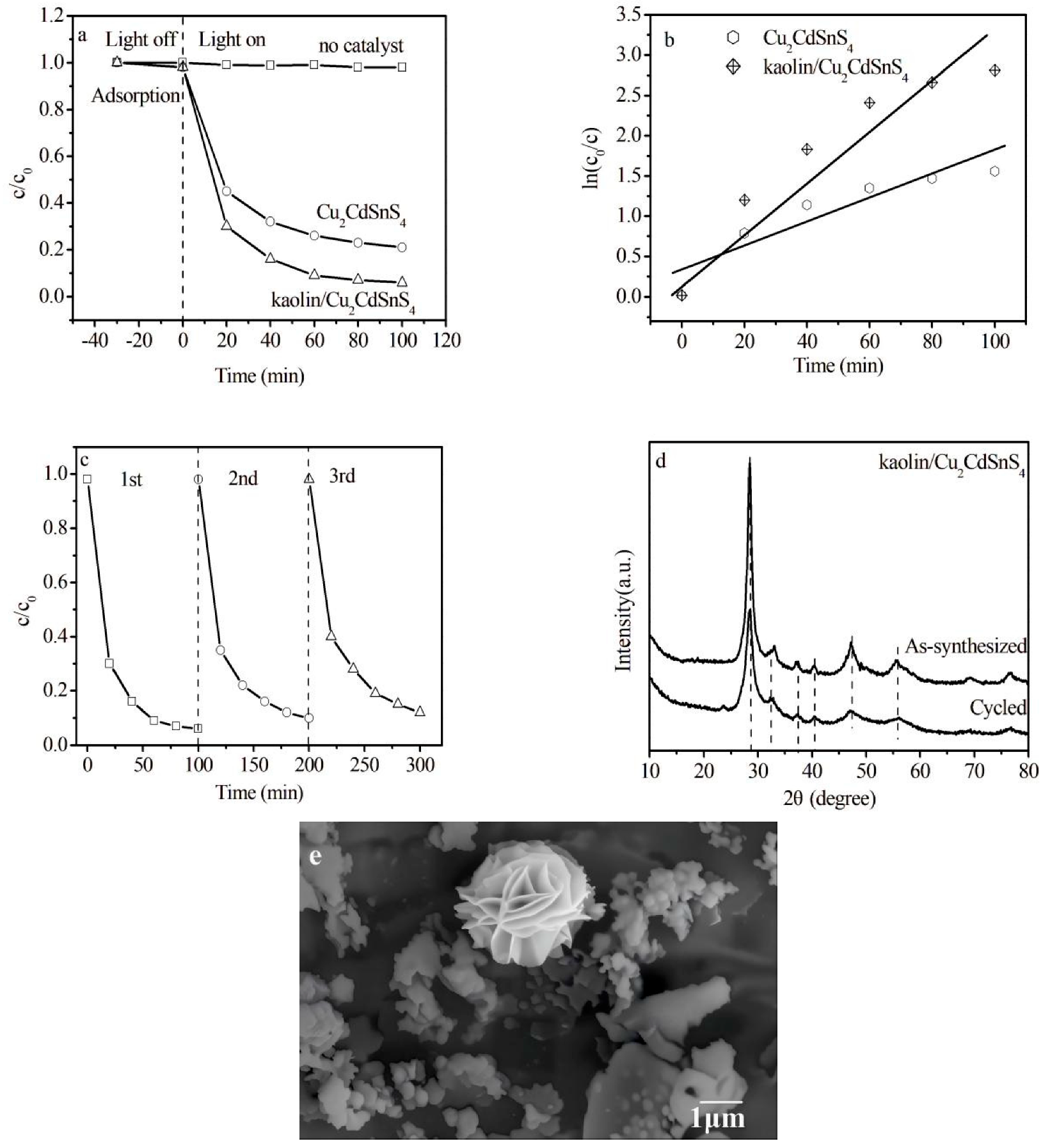
Figure 6: (a) the efficiencies of degradation of MB as a function of different time (b) ln(c0/c) as a function of different time of Cu2CdSnS4 and kaolin/Cu2CdSnS4 composite (c) the photo stability of kaolin/Cu2CdSnS4 composite (d) the structural stability of kaolin/Cu2CdSnS4 composite (e) SEM image of kaolin/Cu2CdSnS4 composite after cycling.
A possible schematic mechanism is shown in Fig. 7. When Uv-Vis irradiation is introduced over CCTS nanoparticles, generation of h+-e− takes place. Then these charge carriers migrate on the surface of CCTS nanoparticles, where a redox reaction takes place with other species which are present on the CCTS surface. h+ reacts with H2O or OH easily to produce •OH radical. In addition, e− reacts with O2 to produce •O2− radical, and •OH can be obtained further. The produced •OH can oxidize degradation the contaminant due to its high oxidizing potential.
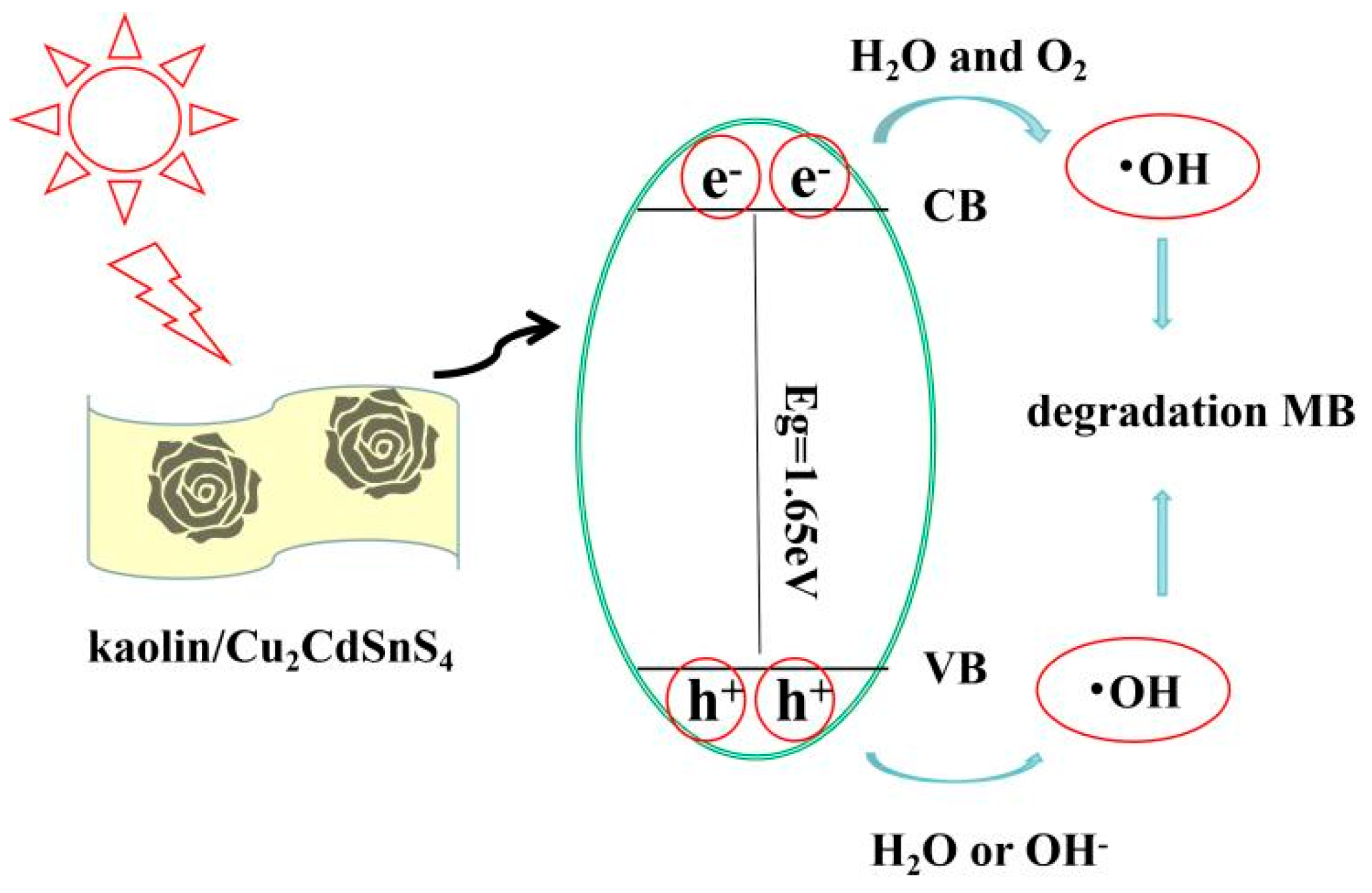
Figure 7: Illustration of photocatalytic redox reaction occurring via photocatalysis to produce •OH to react with MB dye.
In this experiment, kaolin/Cu2CdSnS4 composite is effectively synthesized using a one-step solvothermal route. It can be seen that Cu2CdSnS4 particles exhibit a flower-like hierarchical structure, and the peak thickness is about 40–50 nm. The optical bandgap of the kaolin/Cu2CdSnS4 composite is around 1.69 eV, providing a large absorption range in the visible region. Compared to pure Cu2CdSnS4, kaolin/Cu2CdSnS4 composite exhibits amazing photocatalytic activities for photodegradation of MB of 94% within 100 min irradiation. After repeated use for three cycles, the photodegradation efficiency only decreases to 88%, proving good stability. The work provides a simple and effective method to prepare kaolin/Cu2CdSnS4 composite towards photocatalytic applications.
Acknowledgement:
Funding Statement: This work was supported by ongoing funding of Yancheng Institute of Technology. No additional grants to carry out or direct this particular research were obtained.
Author Contributions: Study conception and design: Hao Guan; data collection: Zicheng Wang and Ruiyang Dai; analysis and interpretation of results: Hao Guan and Tao Dai; draft manuscript preparation: Hao Guan and Zicheng Wang. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: Data sharing is not applicable to this article as no datasets were created or analyzed during the current study.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Shahzad W , Hassan R , Badawi AK , Ismail B . Novel insights from XRD and photophysical properties of Cu2ZnSnS4: Na thin films deposited by aqueous chemical route. J Phys Chem Solids. 2026; 208: 113116. doi:10.1016/j.jpcs.2025.113116. [Google Scholar] [CrossRef]
2. Ashebir GY , Dong C . A Cd-diffusion strategy to prepare Cu2CdSnS4 thin films for solar cells. Sol Energy Mater Sol Cells. 2026; 296: 114026. doi:10.1016/j.solmat.2025.114026. [Google Scholar] [CrossRef]
3. Ţălu Ş . Morphological study of Cu2CdSnS4 nanostructures synthesized via spin coating method at different temperatures. Mater Today Commun. 2025; 48: 113696. doi:10.1016/j.mtcomm.2025.113696. [Google Scholar] [CrossRef]
4. Kamalakannan S , Balasubramaniyan N , Neppolian B . Z-scheme based fabrication of Cu2CdSnS4/Au/g-C3N4 ternary heterojunction with enhanced photocatalytic hydrogen production. Opt Mater. 2025; 164: 117051. doi:10.1016/j.optmat.2025.117051. [Google Scholar] [CrossRef]
5. Vishnupriya S , Choudhari NJ , Kavya DM , Srujana BS , Raviprakash Y . Effect of stacking order on the formation of Cu2CdSnS4 thin films synthesized using thermal evaporation technique. J Mater Eng Perform. 2025; 34( 12): 11119– 27. doi:10.1007/s11665-025-10993-1. [Google Scholar] [CrossRef]
6. Xu Q , Wang Z , Yang H , Xiang Y , Nie G , Yue W . Synthesis of hierarchical Cu2CdSnS4 by microwave-assisted transformation from precursor for photodegradation to malachite green. J Alloys Compd. 2022; 904: 163966. doi:10.1016/j.jallcom.2022.163966. [Google Scholar] [CrossRef]
7. Henry J , Prema P , Padiyan DP , Mohanraj K , Sivakumar G . Shape-dependent optoelectrical investigation of Cu2+xCd1−xSnS4 thin films for solar cell applications. New J Chem. 2016; 40( 3): 2609– 18. doi:10.1039/C5NJ03154K. [Google Scholar] [CrossRef]
8. Yang Y , Waterhouse GIN , Chen Y , Sun-Waterhouse D , Li D . Microbial-enabled green biosynthesis of nanomaterials: current status and future prospects. Biotechnol Adv. 2022; 55: 107914. doi:10.1016/j.biotechadv.2022.107914. [Google Scholar] [CrossRef]
9. Boonphan S , Prachakiew S , Klinbumrung K , Thongrote C , Klinbumrung A . Enhancing photocatalytic performance of Kaolin clay: an overview of treatment strategies and applications. Arch Environ Prot. 2024; 50: 54– 64. doi:10.24425/aep.2024.151686. [Google Scholar] [CrossRef]
10. Liu Z , Zhang Y , Tai Y , Gao R , Liu X , Niu B , et al. Synthesis of CoFe2O4/natural Kaolin activated peroxymonosulfate for visible light degradation of organic contaminants. Opt Mater. 2022; 134: 113088. doi:10.1016/j.optmat.2022.113088. [Google Scholar] [CrossRef]
11. Awugichew DT , Yu G , Noman M , Jiang R , Yun K , Chen Y . Synthesis and application of Kaolin-based catalyst for the degradation of ciprofloxacin in catalytic ozonation. J Water Process Eng. 2025; 70: 107070. doi:10.1016/j.jwpe.2025.107070. [Google Scholar] [CrossRef]
12. Mirbagheri NS , Sabbaghi S . A natural Kaolin/γ-Fe2O3 composite as an efficient nano-adsorbent for removal of phenol from aqueous solutions. Microporous Mesoporous Mater. 2018; 259: 134– 41. doi:10.1016/j.micromeso.2017.10.007. [Google Scholar] [CrossRef]
13. Ma W , Hu J , Yoza BA , Wang Q , Zhang X , Li QX , et al. Kaolinite based catalysts for efficient ozonation of recalcitrant organic chemicals in water. Appl Clay Sci. 2019; 175: 159– 68. doi:10.1016/j.clay.2019.04.011. [Google Scholar] [CrossRef]
14. Gao L , Zhai Y , Ma H , Wang B . Degradation of cationic dye methylene blue by ozonation assisted with Kaolin. Appl Clay Sci. 2009; 46( 2): 226– 9. doi:10.1016/j.clay.2009.08.030. [Google Scholar] [CrossRef]
15. Gong Y , Gai L , Tang J , Fu J , Wang Q , Zeng EY . Reduction of Cr(VI) in simulated groundwater by FeS-coated iron magnetic nanoparticles. Sci Total Environ. 2017; 595: 743– 51. doi:10.1016/j.scitotenv.2017.03.282. [Google Scholar] [CrossRef]
16. Hao G , Xuye S , Tao G , Lin C . Photocatalytic properties of Cu2CdSnS4-CdS nanocomposites synthesized via solvothermal method. Chalcogenide Lett. 2024; 21( 6): 475– 82. doi:10.15251/cl.2024.216.475. [Google Scholar] [CrossRef]
17. Patel M , Mukhopadhyay I , Ray A . Structural, optical and electrical properties of spray-deposited CZTS thin films under a non-equilibrium growth condition. J Phys D Appl Phys. 2012; 45( 44): 445103. doi:10.1088/0022-3727/45/44/445103. [Google Scholar] [CrossRef]
18. Das K , Panda SK , Gorai S , Mishra P , Chaudhuri S . Effect of Cu/In molar ratio on the microstructural and optical properties of microcrystalline CuInS2 prepared by solvothermal route. Mater Res Bull. 2008; 43( 10): 2742– 50. doi:10.1016/j.materresbull.2007.10.013. [Google Scholar] [CrossRef]
19. Gadallah AS , Salim MA , Atwee T , Ghander AM . Effect of Al doping on structural, morphological, optical, and electrical properties of Cu2ZnSnS4 thin films prepared by sol-gel spin coating. Optik. 2018; 159: 275– 82. doi:10.1016/j.ijleo.2018.01.086. [Google Scholar] [CrossRef]
20. Li C , Cao M , Huang J , Wang LJ , Shen Y . Facile synthesis of ultra-long Cu2CdSnS4 nanowires with wurtzite-derived structure. Mater Lett. 2015; 140: 170– 3. doi:10.1016/j.matlet.2014.10.165. [Google Scholar] [CrossRef]
21. de Freitas VAA , de Souza Pinheiro D , Peixoto CF , de Oliveira LCA , da Costa Couceiro PR . Synthesis of Fe-zeolites from Amazonian kaolinite for methylene blue removal: adsorption and photocatalytic activity. Dalton Trans. 2025; 54( 20): 8287– 97. doi:10.1039/d5dt00439j. [Google Scholar] [CrossRef]
22. Abbas A , Li K , Guo X , Wu A , Ali F , Attique S , et al. Solvothermal synthesis of 3D hierarchical Cu2FeSnS4 microspheres for photocatalytic degradation of organic pollutants. Environ Res. 2022; 205: 112539. doi:10.1016/j.envres.2021.112539. [Google Scholar] [CrossRef]
23. Gao Y , Long F , Wang J , Zhang J , Mo S , Zou Z . Understanding the growth mechanism of wurtzite Cu2ZnSnS4 nanocrystals and the photodegradation properties. Mater Des. 2017; 123: 24– 31. doi:10.1016/j.matdes.2017.03.012. [Google Scholar] [CrossRef]
24. Shi K , Wang Z , Li X , Xiao Q , Ji W , Zhang J , et al. A dual-functional adsorption-photocatalysis system driven by interfacial charge dynamics in a type-II 3D/2D CdIn2S4/nickel metal–organic layer heterojunction for environmental purification and water splitting. J Colloid Interface Sci. 2026; 703: 139230. doi:10.1016/j.jcis.2025.139230. [Google Scholar] [CrossRef]
25. Li J , Weng Q , Huang J , Zou Y . Fabrication of 3D hierarchical CdS@CuS hollow heterostructure nanocages for enhanced visible-light-driven dye degradation. Mater Sci Semicond Process. 2026; 202: 110178. doi:10.1016/j.mssp.2025.110178. [Google Scholar] [CrossRef]
26. Lü Y , Qiao QA , Wang R , Dong Y , Cai H , Gao H . Facile construction of Zn0.6Cd0.4S/Bi3O4Br S-scheme heterojunction with enhanced performance under visible light: an experimental and theoretical study. J Environ Chem Eng. 2026; 14( 1): 120748. doi:10.1016/j.jece.2025.120748. [Google Scholar] [CrossRef]
27. Wu Y , Zhang C , Pan Q , Su X , Yi G , Oderinde O . Excellent performance of AgBiS2/UiO-66 Z-scheme heterojunction for amplified CO2 reduction enhancement and pollutants photodegradation: interface effect and DFT studies. J Photochem Photobiol A Chem. 2026; 473: 116915. doi:10.1016/j.jphotochem.2025.116915. [Google Scholar] [CrossRef]
28. Maji SK . Single-source derived ZnS–AgInS2 nanocrystals as tuneable dual-function nanoprobes for photocatalytic and bioimaging applications. Inorg Chem Commun. 2026; 183: 115862. doi:10.1016/j.inoche.2025.115862. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools