 Open Access
Open Access
ARTICLE
Anti-Inflammatory Effects of Veratramine against Lipopolysaccharide-Induced Inflammation
College of Pharmacy, CMRI Research Institute of Pharmaceutical Sciences, Kyungpook National University, Daegu, Republic of Korea
* Corresponding Author: Jong-Sup Bae. Email:
# These authors contributed equally to this work as the first author
(This article belongs to the Special Issue: Bioactive Natural Components as Regulators of Cellular Pathways and Disease Progression)
BIOCELL 2026, 50(5), 6 https://doi.org/10.32604/biocell.2026.075139
Received 25 October 2025; Accepted 28 January 2026; Issue published 13 May 2026
Abstract
Objectives: Plant-derived bioactive molecules are increasingly recognized as valuable therapeutic resources for managing diverse pathological conditions, particularly those involving vascular inflammation. This study aimed to determine whether veratramine (VRT), a naturally occurring steroidal alkaloid found in Veratrum species of the Liliaceae family, attenuates LPS-induced vascular and pulmonary inflammation by upregulating heme oxygenase-1 (HO-1) and modulating the Nrf2, nuclear factor (NF)-κB, and signal transducer and activator of transcription (STAT1) signaling pathways. Methods: The study assessed the modulatory effects of VRT on HO-1, cyclooxygenase-2 (COX-2), and inducible nitric oxide synthase (iNOS) in LPS-activated human umbilical vein endothelial cells (HUVECs), as well as on iNOS, tumor necrosis factor-α (TNF-α), and interleukin-1β (IL-1β) expression in LPS-treated mice. The VRT doses (0.06–0.6 mg/kg) in mice were chosen to approximate peripheral concentrations of 2–20 μM, corresponding to the non-cytotoxic, anti-inflammatory range in HUVECs, based on calculations using mouse blood volume and body weight, and no overt acute toxicity was observed at these doses in this model. Results: Treatment with VRT enhanced HO-1 induction, inhibited NF-κB activation, diminished COX-2/PGE2 and iNOS/NO production, and attenuated STAT1 phosphorylation. In addition, VRT promoted nuclear translocation of Nrf2 and its binding to antioxidant response elements (AREs), thereby suppressing IL-1β expression in HUVECs. The VRT-mediated inhibition of iNOS/NO was abolished by HO-1 silencing through RNA interference. In vivo, VRT markedly reduced iNOS expression in lung tissue and lowered TNF-α levels in bronchoalveolar lavage fluid. Conclusion: Altogether, these findings demonstrate that VRT exhibits substantial anti-inflammatory potential and may serve as a promising candidate for managing inflammation-related vascular disorders.Graphic Abstract
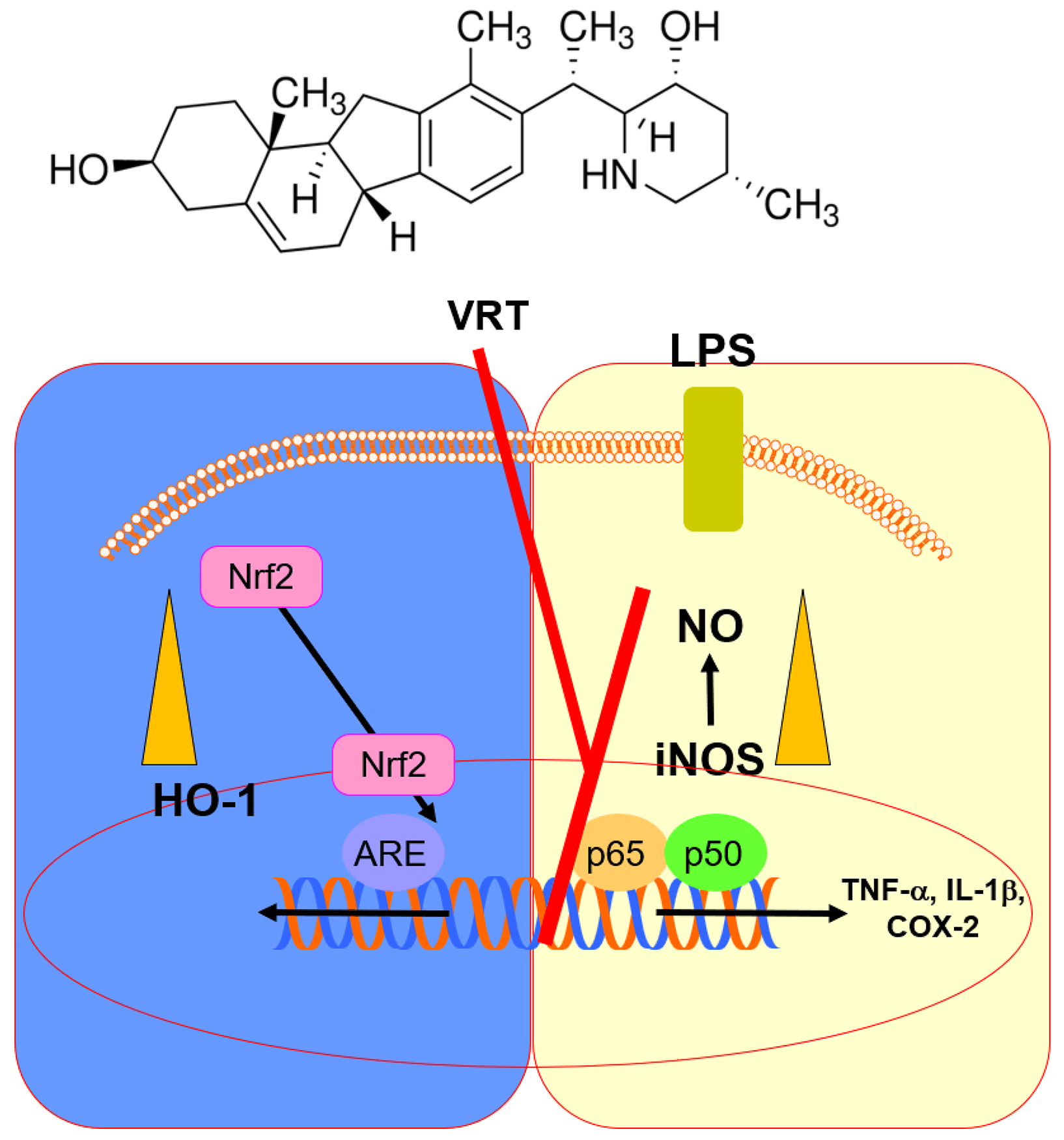
Keywords
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools