 Open Access
Open Access
ARTICLE
Experimental Study on the Frosting Characteristics of Corrugated Surfaces under the Influence of Different Surface Properties
1 Production Department, No. 4 Mine, Pingdingshan Tian’an Coal Mining Group Co., Ltd., Pingdingshan, 467000, China
2 School of Civil Engineering, Henan Polytechnic University, Jiaozuo, 454000, China
3 School of Mechanical and Power Engineering, Henan Polytechnic University, Jiaozuo, 454000, China
* Corresponding Author: Haikun Zheng. Email:
(This article belongs to the Special Issue: Advances in Microscale Fluid Flow, Heat Transfer, and Phase Change)
Frontiers in Heat and Mass Transfer 2026, 24(1), 10 https://doi.org/10.32604/fhmt.2025.074404
Received 10 October 2025; Accepted 05 November 2025; Issue published 28 February 2026
Abstract
This study experimentally investigates the influence of surface wettability on the frosting characteristics of three types of corrugated structures (Types A, B, and C) under controlled low-temperature conditions. The experiments were conducted in a constant-temperature bath at a cold surface temperature of –5°C, relative humidity of 90%, and ambient air temperature of 10°C. The results reveal that the variation trends of frost morphology, frost mass, and frost layer thickness are generally consistent across surfaces with different wettability. Among the tested surfaces, frost crystal formation and complete surface coverage occurred latest on the superhydrophobic surface (CA = 153.9–165.8°), next on the bare aluminum surface (75.3–83.2°), and earliest on the hydrophilic surface (5.3–7.5°). At the same frosting duration, the superhydrophobic surface exhibited a sparse and fluffy frost layer, the bare aluminum surface formed a rough and dense frost, while the hydrophilic surface developed a fine and compact frost layer. The amount of frost formation decreased in the order of hydrophilic > bare aluminum > superhydrophobic, indicating that the superhydrophobic surface provides the most significant anti-frosting effect during the initial stages of frost formation. For instance, on the Type A corrugated structure, after 15 min of frosting, the frost mass on the superhydrophobic surface was 38.78% and 68.45% lower than those on the bare aluminum and hydrophilic surfaces, respectively. After 30 min, these differences were 4.99% and 25.26%, respectively. Overall, the superhydrophobic surface exhibited the smallest frost mass and frost layer thickness, demonstrating superior anti-frosting performance compared with the other surface types.Keywords
In engineering applications such as refrigeration, air conditioning, and heat pumps, frosting on heat exchanger surfaces is a common yet highly detrimental phenomenon. Both finned-tube and microchannel heat exchangers, widely used across various systems, are susceptible to this issue [1]. The operation of such low-temperature equipment fundamentally depends on heat exchange with ambient air, and under conditions of low temperature and high humidity, frost formation on the heat transfer surface becomes almost inevitable. The accumulation of frost layers not only increases thermal resistance and reduces heat transfer efficiency, but also results in higher energy consumption and power loss within the system [2,3]. Taking microchannel heat exchangers as an example, the porous frost layer formed during frosting significantly increases heat transfer and flow resistance, alters gas flow distribution and flow characteristics, and consequently leads to performance degradation. In severe cases, it may even cause system failure [4]. Therefore, a thorough understanding of the frosting mechanism on finned surfaces is of great theoretical and practical significance for the performance optimization and energy-efficient design of heat exchangers.
Given the complex structure of finned-tube heat exchangers, directly observing and measuring the frost layer on fin surfaces is challenging. Consequently, most existing studies employ simplified models—using flat plates to represent flat fins and corrugated plates to represent corrugated fins—to analyze frost formation on cold surfaces. Previous research has indicated that the key factors influencing frosting behavior can be categorized into three groups: operating conditions [5,6], surface structure, and surface characteristics [7,8]. Building upon this foundation, the present study focuses on elucidating the influence of surface characteristics on the frosting behavior of corrugated fins with varying geometries. The objective is to provide a theoretical basis and experimental support for the anti-frosting design and performance enhancement of finned-tube heat exchangers.
The corrugated surface possesses distinctive geometric characteristics. When a heat transfer medium flows over such a surface, it readily induces the disruption and instability of the thermal boundary layer, generating local swirling and vortex structures that markedly enhance heat transfer performance [9,10]. Consequently, corrugated surfaces are widely regarded as an effective means of improving heat exchanger performance. Numerous studies have investigated the influence of corrugated structures on heat transfer and frosting characteristics. Sripattanapipat [11] reported that the sharp-edge configuration of corrugated plates significantly enhances local heat transfer, particularly in V-shaped corrugations. Parlak conducted a comparative numerical investigation of V-shaped, sinusoidal, and straight microchannels, revealing that within a Reynolds number range of 100–400, the Nusselt number of the V-shaped corrugated channel was approximately 27.3% higher than that of the straight channel, demonstrating the best overall heat transfer performance [12]. Ma et al. [13] combined numerical simulations with experiments to study the frosting characteristics of corrugated plates and found excellent agreement between simulation and experimental results. Their findings indicated that the frost layer was densest near the leading edge of the corrugated plate, with the frost density at the crest being higher than that at the trough. Furthermore, as the local airflow velocity increased, the frost layer thickness also increased accordingly. It is noteworthy that Wu’s experimental results suggested that under identical frosting conditions, the overall heat transfer performance of a flat fin surface was slightly superior to that of a corrugated one. Nevertheless, the corrugated surface demonstrated a distinct advantage in heat exchanger applications due to its higher local heat transfer coefficient [14]. Therefore, further investigation into the frosting characteristics of corrugated surfaces with different surface wettability is of great importance for optimizing the anti-frosting performance of corrugated fin heat exchangers and improving their overall heat exchange efficiency.
In addition to surface geometry, surface wettability serves as another critical factor influencing frosting behavior. Since the concept of the “lotus leaf effect” was proposed, the potential of superhydrophobic surfaces to inhibit frost formation has attracted considerable research attention. Molecular dynamics simulations indicate that as the surface contact angle increases, both ice nucleation and frost layer growth are significantly delayed, and the self-propelling motion of droplets on hydrophobic surfaces is superior to that on hydrophilic ones [15]. Huang et al. [16] observed microscopically that during the initial stage of frosting, a larger contact angle corresponds to a slower growth rate of ice nuclei. Jiang et al. [17] further reported that at a surface temperature of –10°C, the freezing time on a superhydrophobic surface is approximately twelve times longer than that on a hydrophilic surface, and even at –20°C, it remains about four times longer. Similarly, Mohammad et al. [18] observed comparable frost-suppression behavior on non-metallic hydrophobic graphene surfaces. Visualization experiments conducted by Gao’s team [19] further confirmed that on a superhydrophobic surface with regular micro–nano structures, the freezing time of droplets (10–20 μm in diameter) is about 4.42 times longer than that on a smooth surface lacking such structures. This phenomenon is primarily attributed to the larger contact angle and smaller solid–liquid contact area of superhydrophobic surfaces, which increase the nucleation energy barrier and interfacial thermal resistance, thereby delaying freezing [20,21]. Furthermore, such surfaces demonstrate remarkable integrated performance in heat exchanger applications. Studies [22,23] have shown that under frosting conditions, the heat exchange efficiency of superhydrophobic surfaces significantly surpasses that of conventional ones. Their advantages are mainly reflected in their synergistic response to airflow and droplet gravity, which enables rapid removal of surface moisture and effectively suppresses frost formation, while simultaneously optimizing the fluid flow at the solid–wall interface to reduce flow resistance and enhance heat transfer performance. Numerical simulations by Rahbarshahlan et al. [24] further validated these findings, showing that superhydrophobic surfaces enhanced local heat transfer in microchannel heat exchangers, increasing heat flux by approximately 15%. Similarly, Chen et al. [25] demonstrated that under low heat flux conditions, superhydrophobic surfaces achieved higher overall heat transfer performance. Therefore, superhydrophobic surfaces exhibit dual engineering potential, offering both frost suppression and drag-reduction–driven energy efficiency improvements.
In conclusion, while numerous studies have examined the factors influencing frosting on cold surfaces, research focusing on the frosting characteristics of corrugated surfaces remains relatively limited. The influence of surface properties on frosting behavior, in particular, has not been systematically investigated. This study experimentally investigates the effects of surface characteristics on the frosting morphology, frost accumulation, and frost layer thickness on corrugated surfaces. The findings provide an experimental basis and a valuable reference for further elucidating the frosting mechanism of corrugated surfaces and for optimizing the design of corrugated finned-tube heat exchangers.
2 Experimental System and Working Conditions
2.1 Visual Low-Temperature Frosting Experimental Platform
As illustrated in Fig. 1, the visual cryogenic frosting experimental platform employed in this study primarily comprises three subsystems: a humid air conditioning system, a cryogenic test bench system, and a data and image acquisition system. The humid air conditioning system is designed to precisely regulate the temperature, relative humidity, and flow rate of the humid air in accordance with experimental requirements. The cryogenic test bench system, which includes a low-temperature constant-temperature bath and a liquid-cooled plate, is used to control the surface temperature of the aluminum corrugated samples to achieve different cold surface temperature conditions. The data and image acquisition system consists of a real-time data measurement unit and an image capture unit. This system monitors and records key experimental parameters—including cold surface temperature, humid air temperature, relative humidity, and flow rate—in real time, while simultaneously capturing the growth process and morphological evolution of frost crystals on the cold surface. The specific specifications of the main instruments and measuring equipment used in the experiment are detailed in Table 1.
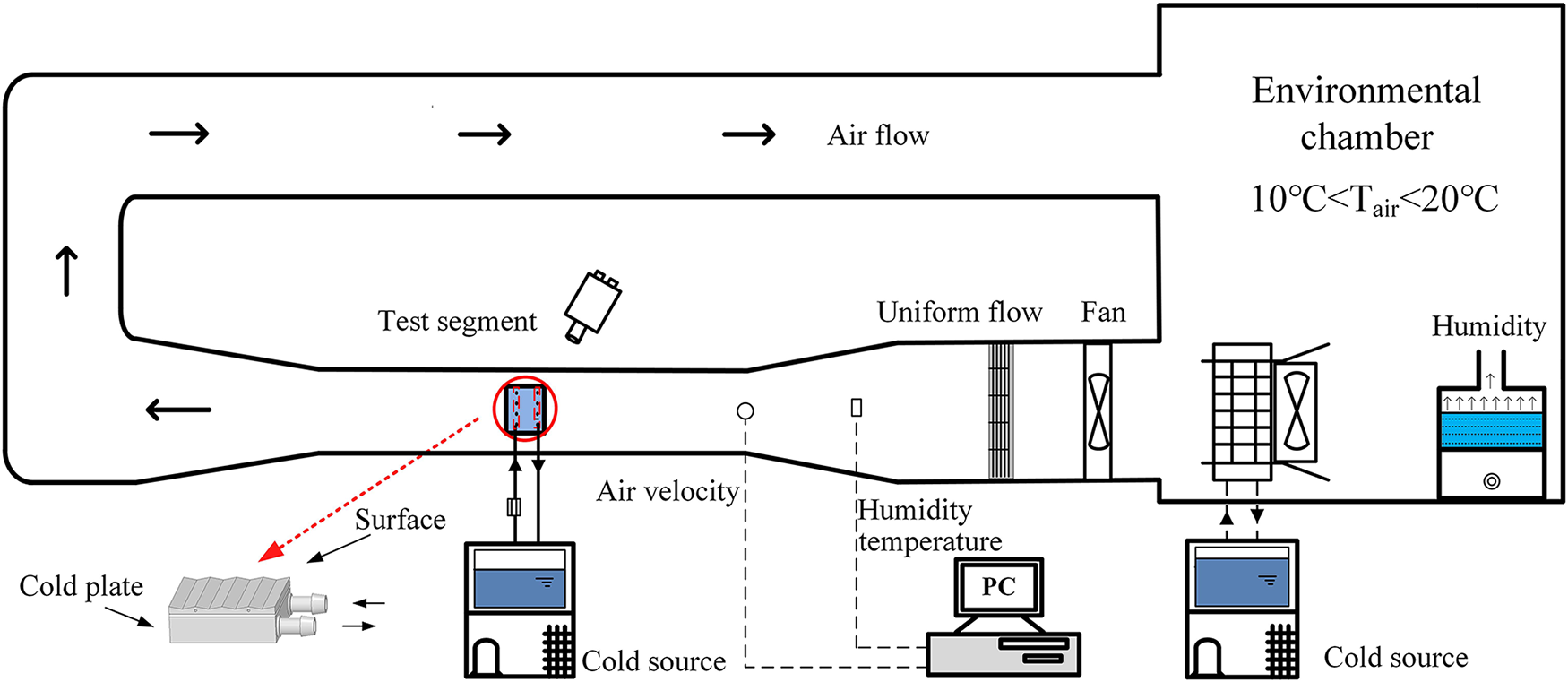
Figure 1: Shows the system diagram of the visual low-temperature frosting experimental platform
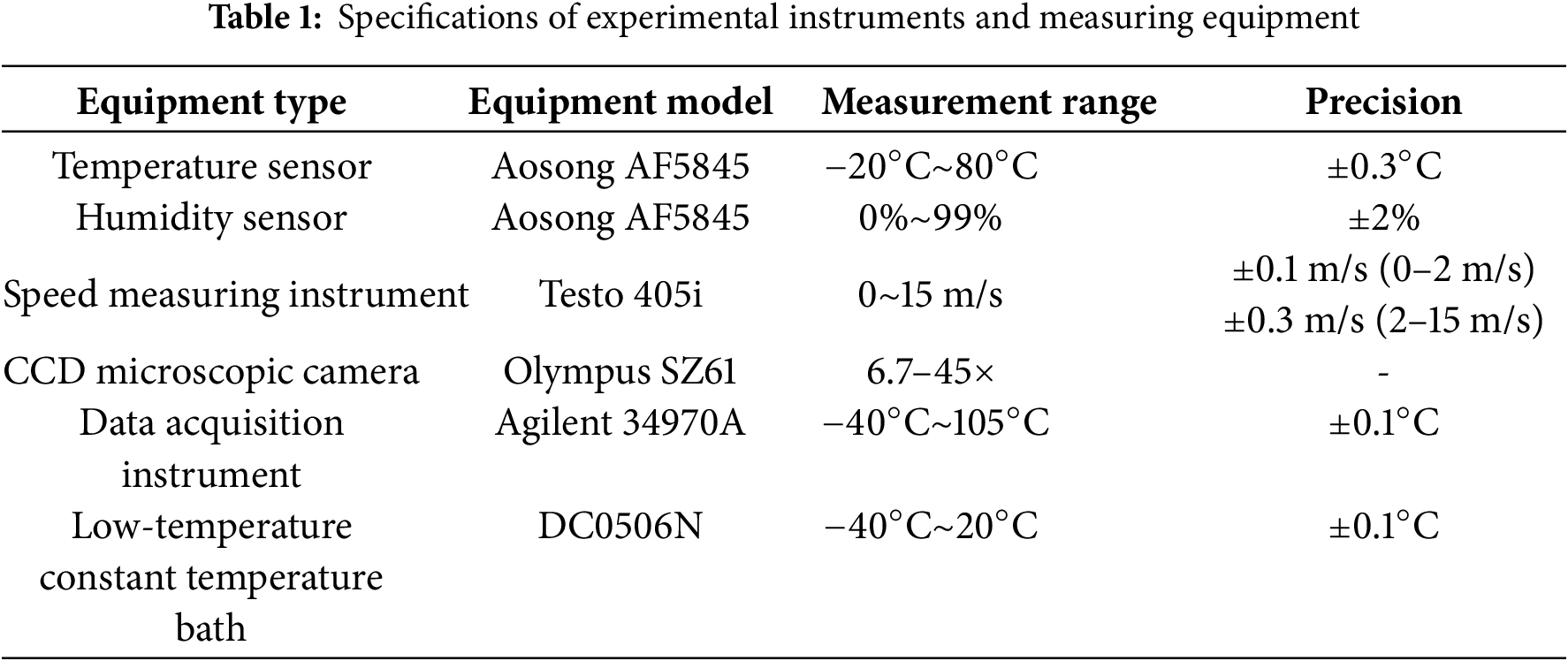
2.2 Experimental Parameters and Working Conditions
The influence of ripple angle and depth on frost formation under different ripple distribution forms has been preliminarily verified in previous experimental studies. Therefore, representative parameters were selected in this work for further analysis. In this study, three types of aluminum corrugated surfaces—Type A, Type B, and Type C—were selected as the base materials. Using chemical etching, the surface of the metal was modified with a micro-nano structure by applying low surface energy substances. This modification reduced the surface energy of the metal, resulting in nine experimental samples with varying wetting characteristics. Table 2 presents the geometric structure parameters, surface roughness data of the three corrugated surface types, and the contact angle measurement results for each sample.
The experimental conditions were as follows: cold surface temperature Tw = −5°C, wet air temperature Tair = 10°C, relative humidity of wet air RH = 90%, and wet air velocity v = 2.5 m/s. During the experiment, the prepared samples with different wetting characteristics were sequentially placed on the liquid cooling table of the low-temperature testing system. The microscopic morphological changes in the frost layer growth were observed in real-time using a CCD microscopic camera.
2.3 Data Processing and Uncertainty Analysis
Frost layer thickness is a key measurement parameter in this study. However, due to the pronounced spatial inhomogeneity of frost growth, accurately determining its thickness presents a significant challenge. To address this issue, the average frost thickness was determined from side-view images by calculating the ratio of the lateral projection area of the frost layer to the length of the corresponding cold surface. This method effectively characterizes the overall growth behavior of the frost layer under morphologically non-uniform conditions, providing a more comprehensive and representative estimation of its thickness.
The image processing procedure was as follows. First, ImageJ was used to adjust the brightness and contrast to highlight the frost layer and the upper surface boundary of the sample. Next, the frost image was binarized through threshold segmentation. The Image Calculator function in ImageJ was then employed to compute the difference between the frost image at time ti and that at the initial time t0, thereby obtaining the binarized image of the frost layer. For each X-coordinate, the corresponding maximum Y-value was extracted, and the resulting points were connected to form the frost surface curve. Based on this curve, the temporal evolution of the frost surface was analyzed. The frost layer thickness was subsequently calculated using Eq. (1), and software such as Origin was used to plot the average thickness and frost surface variation curves.
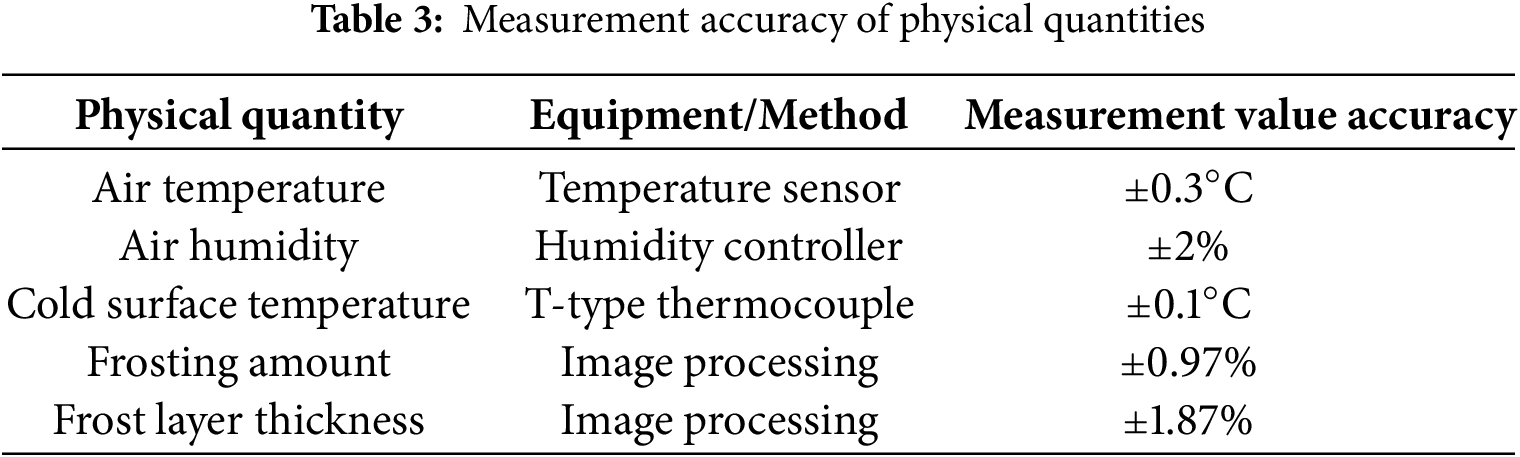
In the uncertainty analysis, experimental errors primarily originated from two sources: measurement instruments and image processing. To evaluate the uncertainty introduced by the image processing procedure, multiple independent analyses were performed on the same set of images, and the relative error of the calculated frost layer thickness was found to be ±1.87%. Meanwhile, the measurement accuracy of all physical quantities was strictly controlled, and the specific accuracies of the instruments used are summarized in Table 3.
3.1 The Influence of Surface Characteristics on the Frosting Characteristics of the Corrugated Surface of Type A Structure
Under identical working conditions, the frosting process on Type A corrugated surfaces exhibits significant variations depending on sample wettability. According to the experimental results presented in Fig. 2, frost crystals initially appeared on the superhydrophobic surface at 191 s, with complete surface coverage occurring at 720 s. In contrast, the bare aluminum surface exhibited frost crystal formation in just 55 s, achieving full coverage within 255 s. The hydrophilic surface demonstrated the most rapid frosting, with initial crystal formation observed at 15 s and complete coverage occurring within 65 s. This temporal progression correlates directly with surface wettability: a larger contact angle corresponds to a higher nucleation energy barrier that water vapor must overcome during phase transition, thereby effectively delaying both the initiation and development of frost crystals.

Figure 2: Frost morphology on the surface of Type A structure with corrugated surface—aerial view
Regarding frost crystal morphology, distinct structural characteristics were observed across different surface types. The superhydrophobic surface developed a fluffy and sparse frost layer structure, while the bare aluminum surface exhibited rough but dense frost features. The hydrophilic surface, conversely, formed a fine and densely packed frost layer. These morphological differences originate from the fundamental regulatory mechanism of surface characteristics on frost crystal growth. Superhydrophobic surfaces, characterized by their low surface energy, substantially increase the nucleation energy barrier for condensate droplets and consequently inhibit frost crystal development. The high surface energy of bare aluminum, combined with its irregular microstructure, promotes accelerated frost crystal growth. The hydrophilic surface, featuring uniform and dense micro-scale groove structures, facilitates the formation of a compact frost layer architecture.
Experimental observations revealed a pronounced spatial nonuniformity in the distribution of frost layers. As shown in the side view of Fig. 3, the geometric configuration of the corrugated structure, together with the airflow direction, exerts a significant influence on the local frost growth characteristics. The crest is directly exposed to the incoming airflow, where the intensified turbulence enhances local convective heat and mass transfer of water vapor, leading to substantial condensation. Consequently, frost initially forms rapidly on the windward side and subsequently spreads outward. In contrast, within the leeward region, the airflow velocity decreases, reducing the local saturation water vapor concentration and constraining the sublimation rate. This results in slower frost growth and the formation of a distinct frost boundary. After 30 min of frosting, the frost thickness varies markedly among different surfaces: it is thinnest on the superhydrophobic surface, followed by the hydrophilic surface, and thickest on bare aluminum. These differences arise from the effects of surface wettability on the formation energy barrier of ice nuclei and on interfacial heat transfer behavior. Superhydrophobic surfaces, characterized by high contact angles and limited solid–liquid contact areas, possess a relatively large energy barrier and higher interfacial thermal resistance during droplet formation or primary crystal nucleation, thereby suppressing the vapor-to-solid phase change rate. Moreover, the weak adhesion between frost crystals and the substrate facilitates detachment under airflow shear, further slowing frost accumulation. Conversely, hydrophilic surfaces promote continuous condensation and strong bonding among frost crystals, producing a denser frost layer that grows more slowly in thickness. Bare aluminum, lacking distinctive wetting properties, allows rapid vapor deposition and substantial frost buildup, resulting in the greatest final thickness. The thickness disparity at the wave crest is particularly pronounced, underscoring that surface geometry and wettability jointly govern local heat and mass transfer, serving as the key physical factors dictating the formation and evolution of frost layers.
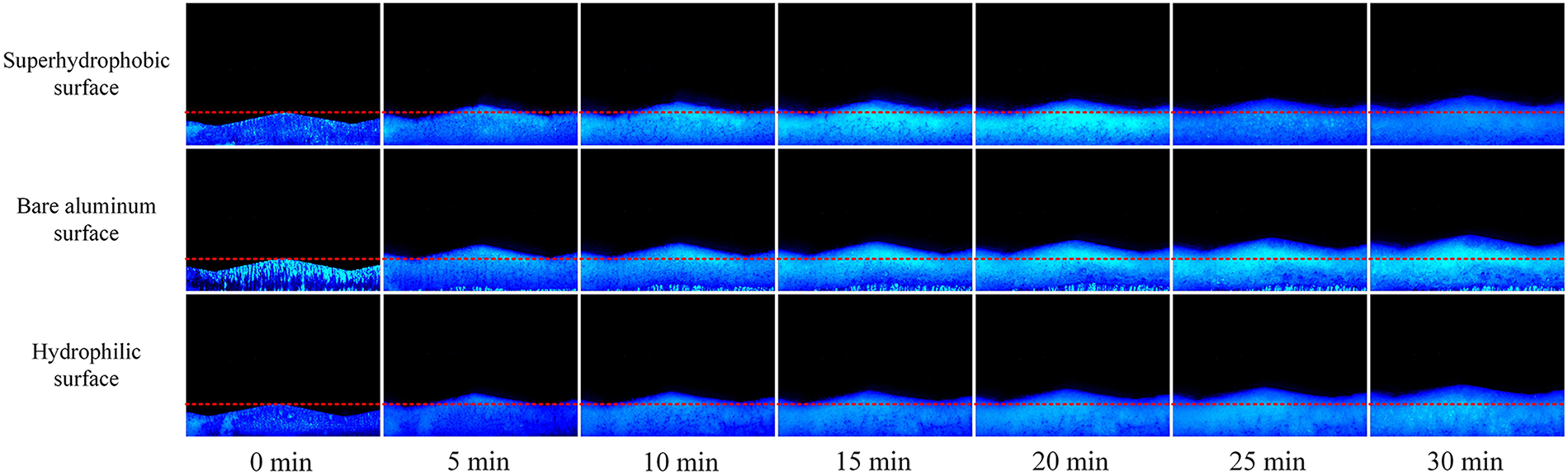
Figure 3: Frost morphology on the surface of corrugated A-type structure—side view
3.1.2 Frost Formation Amount and Frost Layer Thickness
Fig. 4 illustrates the variation patterns of frost accumulation and frost layer thickness on Type A corrugated surfaces with different surface wettability. As shown in Fig. 4a, under identical working conditions, the hydrophilic surface exhibits the highest frost accumulation, followed by the bare aluminum surface, while the superhydrophobic surface demonstrates the least frost accumulation. This phenomenon is primarily attributed to differences in nucleation energy barriers governed by surface wettability: a smaller contact angle corresponds to stronger surface wettability and a lower energy barrier for water vapor phase transition during frosting. Consequently, hydrophilic surfaces are more susceptible to frost formation. The bare aluminum surface, with its relatively higher energy barrier, experiences moderately limited frost accumulation. In contrast, the superhydrophobic surface, characterized by a large contact angle and low surface energy, significantly inhibits frost crystal growth, resulting in minimal frost accumulation.
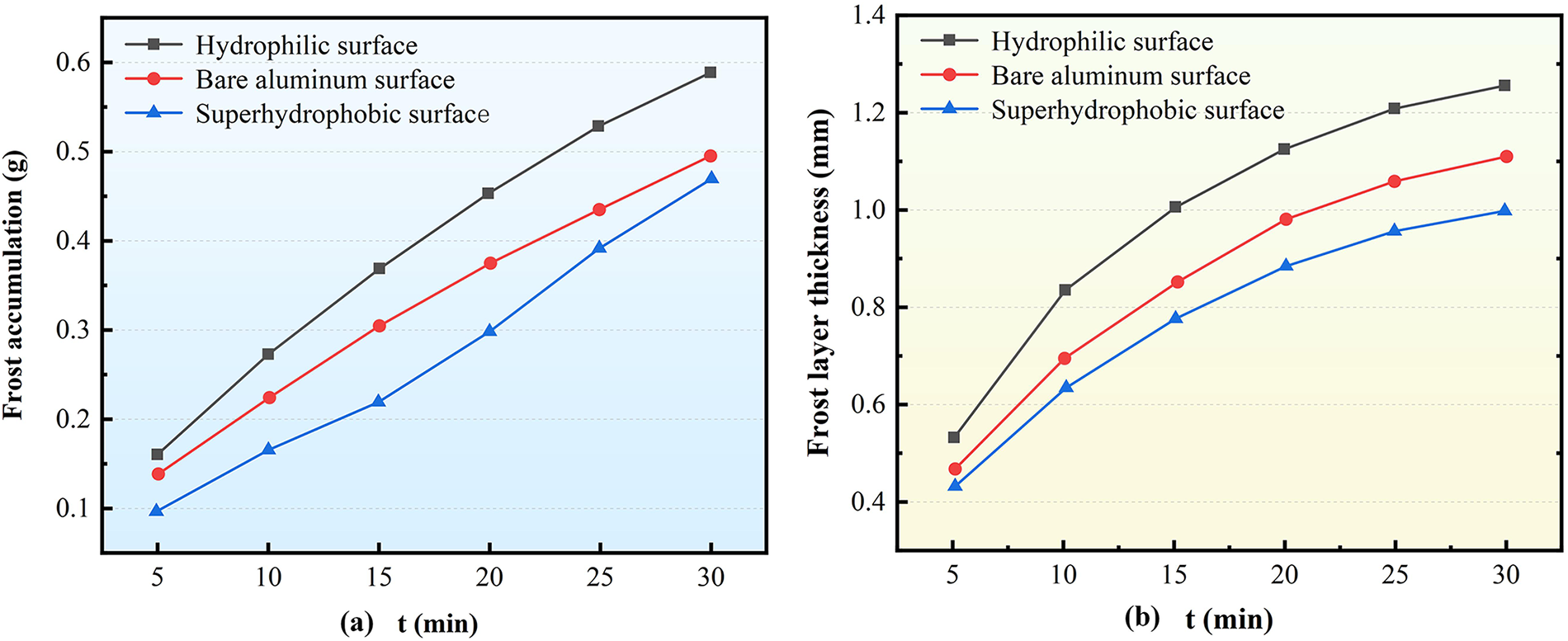
Figure 4: Influence of Surface characteristics on the frosting of the corrugated surface of Type A structure (a) Frosting accumulation; (b) Frost layer thickness
Regarding the temporal evolution of frost accumulation, both the hydrophilic and bare aluminum surfaces exhibit a gradually decelerating growth rate. In comparison, the superhydrophobic surface displays a distinctive fluctuating trend characterized by an initial decrease, followed by an increase, and a subsequent decrease. This behavior can be explained as follows: the growing frost layer increases thermal resistance, reduces the effective temperature difference, and thereby suppresses further frost formation. Initially, the high nucleation energy barrier of the superhydrophobic surface significantly inhibits frost accumulation. As the frost layer progressively covers the hydrophobic areas, this energy barrier diminishes, allowing for a temporary increase in frost accumulation. Eventually, heat transfer limitations again dominate, causing the growth rate to decline. Comparative analysis confirms that the superhydrophobic surface provides the most pronounced anti-frosting effect during the early stages. For instance, at the 15-min mark, the frost accumulation on the superhydrophobic surface was 38.78% and 68.45% lower than that on the bare aluminum and hydrophilic surfaces, respectively. By the 30-min mark, these differences reduced to 4.99% and 25.26%, respectively.
As shown in Fig. 4b, under identical working conditions, the bare aluminum surface exhibits the greatest frost layer thickness, followed by the hydrophilic surface, while the superhydrophobic surface maintains the thinnest frost layer. This variation stems from the combined influence of surface energy characteristics and microstructure on the frost crystal growth process. On bare aluminum surfaces, the relatively high surface energy and distinct microscopic roughness facilitate water vapor condensation and promote frost crystal adhesion and accumulation. As frosting progresses, frost crystals continuously interweave and grow, forming a thick frost layer with a consistently high growth rate. The hydrophilic surface, with its strong wettability and regular, dense micro-scale groove structure, promotes uniform frost distribution and significant densification. Although this results in a large frost mass per unit area, the tight, low-porosity structure restricts overall thickness growth, exhibiting a characteristic “high mass, low thickness” pattern. In contrast, the extremely low surface energy and high nucleation barrier of superhydrophobic surfaces inhibit both water vapor condensation and ice nucleus formation, thereby significantly delaying frost crystal formation and expansion. During the early frosting stage, frost layer growth is nearly stagnant. As the process continues into mid and later stages, the gradual coverage of the surface by frost partially weakens this barrier, allowing limited, slow crystal growth. A comprehensive comparison confirms that after 30 min of frosting, the superhydrophobic surface’s frost layer thickness remains significantly lower than the others—specifically, 25.64% and 10.71% thinner than the bare aluminum and hydrophilic surfaces, respectively. This demonstrates that the superhydrophobic surface effectively suppresses both frost crystal growth and layer accumulation throughout the entire frosting process, exhibiting excellent anti-frosting performance.
As illustrated in Fig. 4, from a macroscopic perspective, the overall accumulation of the frost layer exhibits a two-stage pattern—an initially rapid growth phase followed by a gradual stabilization phase. This behavior primarily arises from the structural evolution of the frost layer and the corresponding variations in its heat and mass transfer characteristics. According to Gibbs free energy theory, the driving force for phase change during frosting is predominantly governed by the absolute humidity of the air and the degree of subcooling of the cold surface. As frosting progresses, the temperature difference between the cold surface (or the frost surface) and the surrounding air gradually decreases. In the initial stage, the cold surface possesses a relatively low temperature and a large exposed area, leading to high rates of water vapor condensation and freezing. Consequently, frost crystals form and accumulate rapidly, resulting in a sharp increase in frost layer thickness. With the continued thickening of the frost layer, its porous microstructure introduces significant thermal and diffusion resistance to heat and mass transfer. The surface temperature of the frost consequently rises, and the rate of vapor phase change declines, thereby inhibiting further crystal growth. As a result, in the later stage of the process, the frost growth rate decreases markedly and eventually approaches a quasi-steady state.
3.2 The Influence of Surface Characteristics on the Frosting Characteristics of the Corrugated Surface of Type B Structure
Fig. 5 reveals significant differences in the frosting behavior of samples with different wettability on Type B corrugated surfaces under experimental conditions. Specifically, frost crystals initially appeared on the superhydrophobic surface at 255 s, with complete surface coverage occurring at 1073 s. In comparison, the bare aluminum surface exhibited initial frosting at 65 s and reached full coverage at 250 s, while the hydrophilic surface demonstrated the most rapid frosting—initial crystals formed at 12 s, with complete coverage achieved in just 60 s. These temporal patterns confirm that surface wettability and the corresponding nucleation barrier substantially influence both the initiation timing and development rate of frost formation. Consistent with the frosting patterns observed on Type A structures, the superhydrophobic surface demonstrated the longest delay in frosting onset, followed by bare aluminum, while the hydrophilic surface exhibited the most rapid frost formation and propagation.
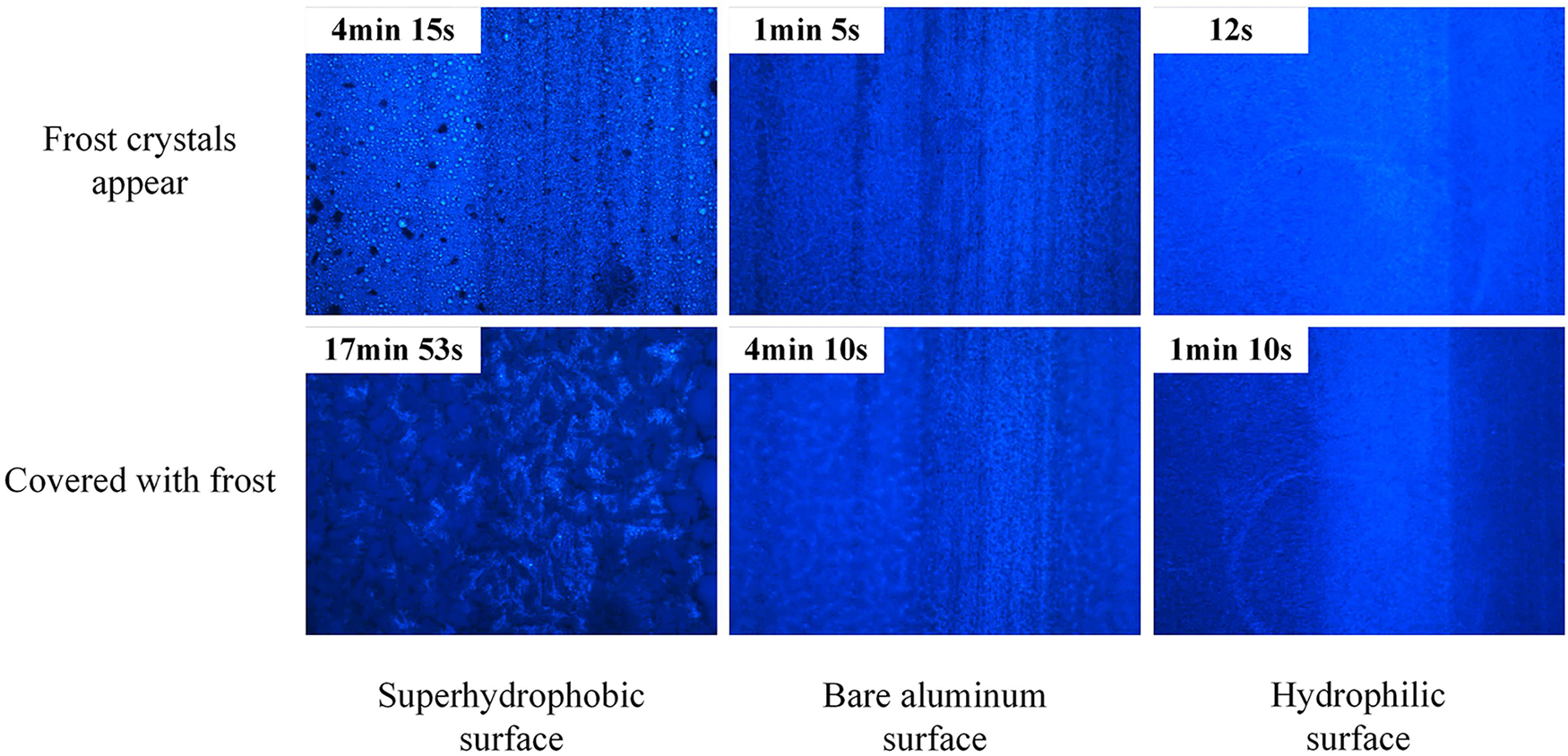
Figure 5: Frost morphology on the surface of Type B structure with corrugated surface—aerial view
Regarding frost crystal morphology, both the top view in Fig. 5 and the side view in Fig. 6 illustrate how surface wettability governs frost layer structure. The superhydrophobic surface developed a fluffy, sparse frost morphology with substantial inter-crystal voids. The bare aluminum surface produced rough, densely packed crystals characteristic of rapid accumulation, whereas the hydrophilic surface formed fine, uniformly distributed frost crystals with a compact structure. Fig. 5 further reveals distinct “boundary lines” in frost distribution across all surfaces, with frost layers transitioning gradually from thick to thin. As illustrated in the side view of Fig. 6, the central region corresponds to the corrugation peak, with humid airflow moving from left to right. The slightly leftward plane represents the windward side, while the slightly rightward trough constitutes the leeward side. Due to direct exposure to incoming airflow and consequently richer water vapor supply, the windward side experiences significantly faster frosting than the leeward side, resulting in clearly demarcated frost boundary regions.
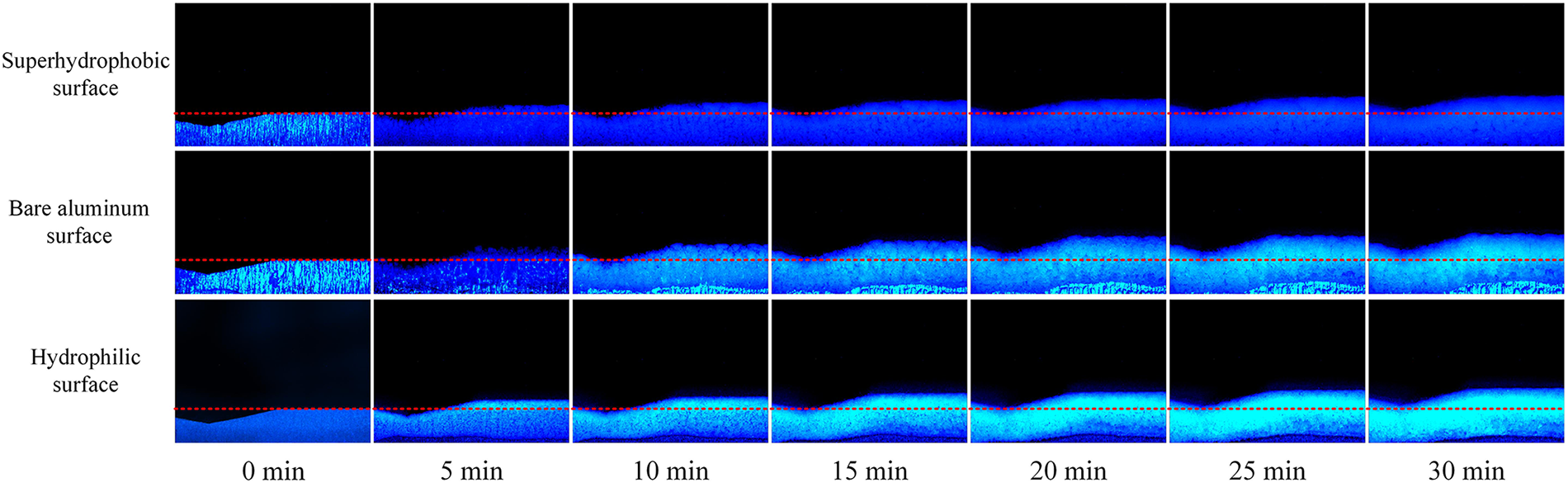
Figure 6: Frost morphology on the surface of corrugated B-type structure—side view
Fig. 6 also demonstrates notable differences in frost layer thickness attributable to surface wettability, particularly at wave peaks. After 30 min of frosting, the superhydrophobic surface maintained the thinnest frost layer, followed by the hydrophilic surface, while the bare aluminum surface accumulated the thickest layer. This hierarchy reflects fundamental inhibition mechanisms: superhydrophobic surfaces effectively suppress frost accumulation through low surface energy and high nucleation barriers; hydrophilic surfaces, despite their dense frost structure, exhibit limited thickness growth; and bare aluminum surfaces, with their high surface energy, actively promote rapid frost crystal development. Overall, while Type B corrugated structures follow the same fundamental frosting patterns as Type A, they exhibit greater susceptibility to flow field effects, resulting in more pronounced spatial heterogeneity in frost layer distribution.
3.2.2 Frost Formation Amount and Frost Layer Thickness
Fig. 7 illustrates the variation patterns of frost accumulation and frost layer thickness on Type B corrugated structures with different surface wettability. Under identical working conditions, the hydrophilic surface exhibits the highest frost accumulation, followed by the bare aluminum surface, while the superhydrophobic surface demonstrates the least frost accumulation. This hierarchy is primarily governed by differences in surface wettability and surface energy. Consistent with the frosting behavior observed on Type A structures, a smaller contact angle corresponds to stronger surface wettability and a lower nucleation energy barrier for water vapor deposition. Consequently, the hydrophilic surface facilitates rapid frost crystal formation and accumulation, resulting in the greatest frost mass. The bare aluminum surface exhibits moderate frost accumulation, whereas the superhydrophobic surface, with its low surface energy and high nucleation barrier, effectively suppresses both frost initiation and growth, leading to minimal frost accumulation.
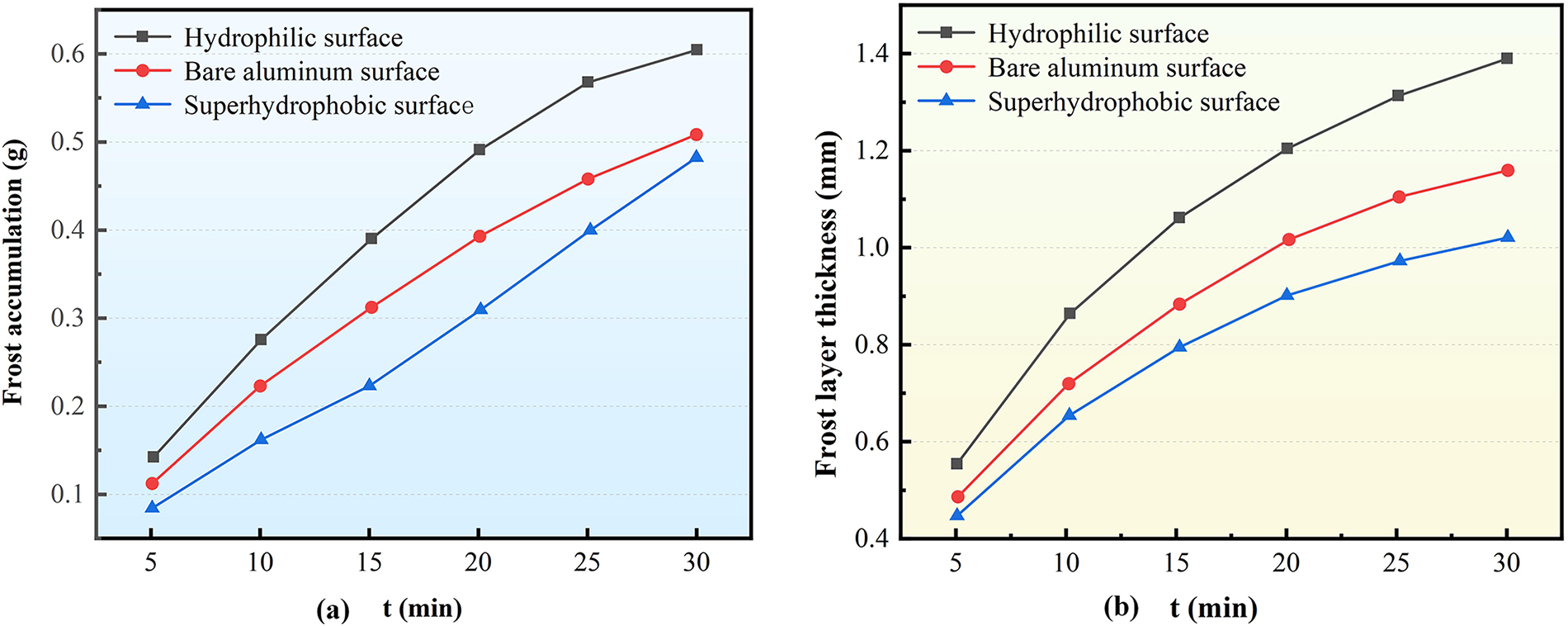
Figure 7: Influence of Surface characteristics on the frosting of the corrugated surface of Type B structure (a) Frosting accumulation; (b) Frost layer thickness
Analysis of the temporal evolution reveals distinct frost accumulation trends across different surfaces. The anti-frosting effect of the superhydrophobic surface is particularly pronounced during the early stages compared to the middle and later phases of frosting. Specifically, at the 15-min mark, the frost accumulation on the superhydrophobic surface was 26.69% and 40.03% lower than that on the bare aluminum and hydrophilic surfaces, respectively. By the 30-min mark, these differences reduced to 4.65% and 23.97%, respectively. This progressive convergence indicates that while the superhydrophobic surface provides strong frost suppression initially, its effectiveness diminishes as the accumulating frost layer gradually compromises the surface’s original hydrophobic properties.
Fig. 7b demonstrates that under identical working conditions, the bare aluminum surface develops the thickest frost layer, followed by the hydrophilic surface, while the superhydrophobic surface maintains the thinnest frost layer. This pattern, consistent with observations on Type A structures, results from the combined effects of surface energy and microstructure. Higher surface energy facilitates water vapor condensation and frost crystal growth, while greater surface roughness promotes crystal adhesion and accumulation. The bare aluminum surface, characterized by high surface energy and distinct microscopic roughness, enables rapid frost crystal adhesion and growth, leading to the fastest thickness increase. The hydrophilic surface, with its uniform and fine groove structure and strong wettability, produces a densely distributed frost layer. Although this results in substantial frost mass accumulation, the high-density, low-porosity structure inherently restricts thickness development. In contrast, superhydrophobic surfaces exhibit the lowest surface energy and poorest wettability, where the elevated nucleation energy barrier effectively suppresses frost crystal growth, resulting in minimal layer thickness.
Experimental measurements after 30 min of frosting show that the superhydrophobic surface reduces frost layer thickness by 35.67% compared to bare aluminum and by 13.30% compared to the hydrophilic surface. These results confirm that superhydrophobic surfaces significantly inhibit frost layer development throughout the frosting process, effectively limiting both frost accumulation and thickness growth. While Type B corrugated surfaces follow the same fundamental frosting mechanisms as Type A structures, differences in peak geometry and resultant flow field distribution lead to more pronounced spatial heterogeneity in frost layer formation. These findings further validate the crucial role of surface wettability and energy characteristics in governing frost crystal growth kinetics and frost layer evolution.
3.3 The Influence of Surface Characteristics on the Frosting Characteristics of the Corrugated Surface of Type C Structure
As shown in Fig. 8, the frosting process on Type C corrugated structures varies significantly across surfaces with different wettability under experimental conditions. Frost crystals initially appeared on the superhydrophobic surface at 210 s, with complete surface coverage occurring at 794 s. In comparison, the bare aluminum surface exhibited initial crystallization at 45 s and reached full coverage within 129 s, while the hydrophilic surface demonstrated the most rapid phase change—nucleation beginning at just 9 s and complete frosting achieved within 50 s. These substantial temporal differences are primarily governed by surface wettability and the magnitude of the corresponding nucleation energy barrier. Consistent with the patterns observed in Types A and B structures, the Type C configuration shows the same hierarchy: the superhydrophobic surface demonstrates the longest delay in both frost initiation and complete coverage, followed by bare aluminum, while the hydrophilic surface exhibits the most rapid frost formation and propagation. This consistent ordering across all three corrugated geometries confirms the dominant influence of surface energy and wetting characteristics on the initial stages of frost formation.

Figure 8: Frost morphology on the surface of Type C structure with corrugated surface—aerial view
Regarding frost crystal morphology, distinct structural characteristics are observed across different surfaces: the superhydrophobic surface exhibits a loose and sparse frost layer, the bare aluminum surface shows a rough and dense structure, and the hydrophilic surface displays a fine and densely packed morphology (see Fig. 8 top view and Fig. 9 side view). Observation further reveals a distinct “boundary” feature in the frost layer distribution. As shown in Fig. 9, the central region corresponds to the peak area of the corrugation, with humid airflow moving from left to right. The left side represents the windward side of the corrugated surface, while the right side constitutes the leeward area. Due to the ample water vapor supply and enhanced local heat transfer on the windward side, frost crystals grow more rapidly in this region, forming clear frost layer boundaries and pronounced thickness gradients. Fig. 9 further demonstrates significant differences in frost crystal growth rates across surfaces with different wettability, leading to notable variations in frost layer thickness under identical frosting durations, particularly at the wave peaks. After 30 min of frosting, the superhydrophobic surface maintains the thinnest frost layer, followed by the hydrophilic surface, while the bare aluminum surface develops the thickest accumulation. This pattern confirms that weaker surface wettability and higher nucleation barriers correspond to stronger suppression of frost crystal growth and slower frost layer thickening. Conversely, high surface energy and strong wettability accelerate both frost nucleation and crystal accumulation, promoting rapid frost layer development.
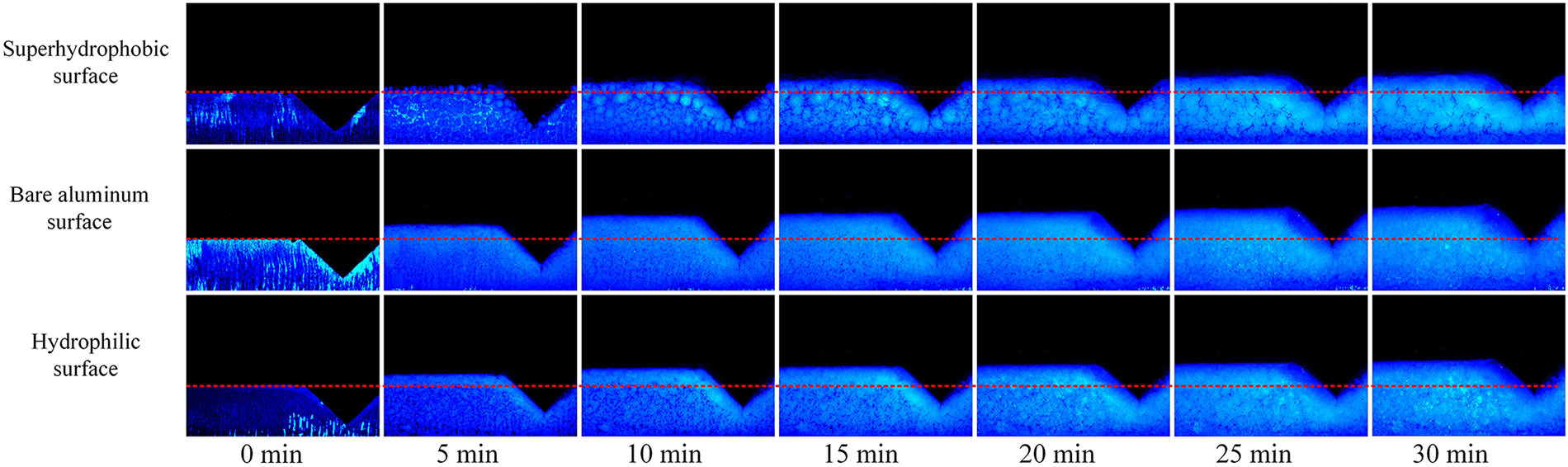
Figure 9: Frost morphology on the surface of corrugated C-type structure—side view
3.3.2 Frost Formation Amount and Frost Layer Thickness
Fig. 10 presents the variation patterns of frost accumulation and frost layer thickness on Type C corrugated surfaces with different wettability under standard experimental conditions (cold surface temperature Tw = −5°C, wet air temperature Tair = 10°C, relative humidity RH = 90%, and air velocity v = 2.5 m/s). As shown in Fig. 10a, under identical conditions, frost accumulation follows a descending order: hydrophilic surface, bare aluminum surface, and superhydrophobic surface. This hierarchy aligns with observations from Types A and B structures, further confirming the dominant role of surface wettability in frosting behavior. Specifically, a smaller contact angle corresponds to a lower phase-change energy barrier, making frost formation more favorable.
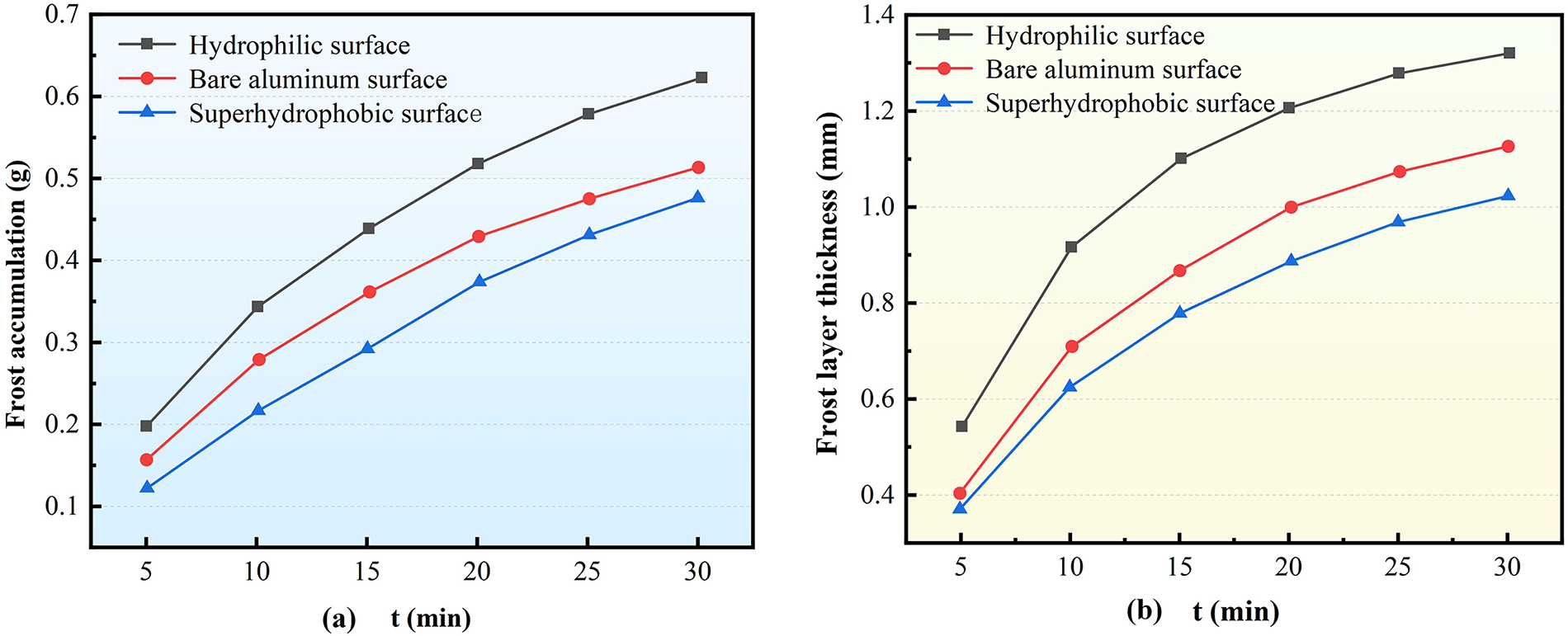
Figure 10: Influence of Surface characteristics on the frosting of the corrugated surface of Type C structure (a) Frosting accumulation; (b) Frost layer thickness
Notably, the superhydrophobic surface exhibits particularly effective frost suppression during the early stages. At the 15-min mark, its frost accumulation was 24.97% and 50.62% lower than that of the bare aluminum and hydrophilic surfaces, respectively. By 30 min, these margins narrowed to 7.50% and 30.91%, indicating a gradual decline in frost suppression efficiency as the process advanced.
Regarding frost layer thickness (Fig. 10b), Type C structures maintain the same trend observed in Types A and B: the bare aluminum surface develops the thickest frost layer, followed by the hydrophilic surface, with the superhydrophobic surface remaining the thinnest. After 30 min of frosting, the superhydrophobic surface reduced frost layer thickness by 29.30% compared to bare aluminum and by 10.42% compared to the hydrophilic surface. These results demonstrate how surface characteristics differentially regulate frost layer growth: superhydrophobic surfaces effectively retard frost crystal development, while the dense structure of frost on hydrophilic surfaces, despite substantial mass accumulation, inherently restricts rapid thickness increase.
From a detailed perspective, the C-type corrugated surface exhibits slightly higher frost accumulation and growth rate compared with the A- and B-type structures. This is primarily attributed to the enhancement of local heat and mass transfer induced by its geometric configuration. The larger corrugation angle intensifies airflow disturbance on the windward side, where turbulence continuously disrupts and renews the temperature and concentration boundary layers. Consequently, both the local convective heat transfer coefficient and the mass transfer rate of water vapor are increased, promoting condensation and freezing. Meanwhile, the corrugation depth of 3 mm not only ensures sufficient air mixing but also generates localized recirculation zones, which facilitate the retention and secondary growth of frost crystals, thereby accelerating frost accumulation. As a combined effect, the C-type corrugated surface demonstrates a slightly greater frost layer thickness and a higher rate of frost growth than the other two configurations.
This study experimentally investigated the influence of surface characteristics on the frosting behavior of three distinct corrugated surface geometries (Types A, B, and C). It systematically documented the microscopic morphology evolution during frosting and quantitatively analyzed the variation patterns of frost accumulation and layer thickness. The principal conclusions are as follows:
(1) Surface characteristics exert a consistent influence on the frosting behavior across all three corrugated surface types. All geometries demonstrate similar trends in frost morphology, accumulation mass, and layer thickness progression. This consistent pattern confirms that the influence of surface wettability on the frosting process is universal and not substantially modulated by variations in corrugated geometric configuration.
(2) Significant temporal differences were observed in the frosting processes across the three surface types. Hydrophilic surfaces exhibited the most rapid frost nucleation, achieving the fastest complete surface coverage. Bare aluminum surfaces demonstrated intermediate frosting speeds, while superhydrophobic surfaces provided the most substantial delay, effectively prolonging both the initial frost crystal formation and the subsequent expansion phases.
(3) The superhydrophobic surface forms a sparse and fluffy frost crystal structure, while the bare aluminum surface exhibits a rough and dense frost layer, and the hydrophilic surface forms a fine and dense frost crystal structure. Due to the aerodynamic characteristics of the corrugated structure, the frost layer growth rate on the windward side is significantly faster than on the leeward side, owing to the higher water vapor flux, resulting in a distinct boundary in the frost layer distribution.
(4) The frost layer thickness measurements show that the frost layer is thickest on the bare aluminum surface, followed by the hydrophilic surface, and thinnest on the superhydrophobic surface.
Acknowledgement: The authors gratefully acknowledge the invaluable assistance and steadfast support provided by their laboratory colleagues.
Funding Statement: This work was financially supported by the Science and Technology Research Project of Henan Province (No. 232102241014), the Key scientific research project of Henan Province Colleges and Universities (No. 22A470002) and Doctoral Fund Project of Henan Polytechnic University (No. B2021-37).
Author Contributions: Kai Song: Writing—original draft, Writing—review & editing, Data curation. Lishan Feng: Methodology, Validation, Formal analysis. Shugang Duan: Project administration, Methodology, Resources. Weilong Zhao: Methodology, Supervision. Haikun Zheng: Project administration, Funding acquisition, Supervision. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials: The authors confirm that the data supporting the findings of this study are available within the article.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest to report regarding the present study.
References
1. Xiong T, Zhao T, Liu G, Zhao L, Jin J, Gao Q, et al. Frosting characteristics of microchannel heat exchangers: parametric studies and correlation development. J Build Eng. 2024;90:109521. doi:10.1016/j.jobe.2024.109521. [Google Scholar] [CrossRef]
2. Ma C, Ge X, Zheng H, Song M, Sheng W, Chen X, et al. From common low temperature to ultra-low temperature: overview of frosting characteristics and defrosting techniques in equipment. Appl Therm Eng. 2025;278:127205. doi:10.1016/j.applthermaleng.2025.127205. [Google Scholar] [CrossRef]
3. Wang F, Liang C, Zhang X. Research of anti-frosting technology in refrigeration and air conditioning fields: a review. Renew Sustain Energy Rev. 2018;81:707–22. doi:10.1016/j.rser.2017.08.046. [Google Scholar] [CrossRef]
4. Han Z, Han B, Wang Z, Liu H. A prediction model for frosting process on inclined cold surface of microchannel heat exchanger. Int J Heat Mass Transf. 2025;253:127584. doi:10.1016/j.ijheatmasstransfer.2025.127584. [Google Scholar] [CrossRef]
5. Ji J, Li P, Pan Y, Zhao J, Sun X. Experimental study on natural convection frosting characteristics of vertical rib plates under different humidity and different materials. Int J Heat Mass Transf. 2025;251:127404. doi:10.1016/j.ijheatmasstransfer.2025.127404. [Google Scholar] [CrossRef]
6. Zhang L, Zhao S, Gu T, Wu L, Zhan B, Xu Z, et al. Frost crystal growth characteristics on surface of frozen water droplets with collapse and reverse melting behaviors considered. Appl Therm Eng. 2025;276:126991. doi:10.1016/j.applthermaleng.2025.126991. [Google Scholar] [CrossRef]
7. Lee H, Yang JB, Kim DR. Anti-frosting characteristics of superhydrophobic-hydrophilic wettability switchable surfaces. Int J Heat Mass Transf. 2024;221:125035. doi:10.1016/j.ijheatmasstransfer.2023.125035. [Google Scholar] [CrossRef]
8. Su W, Ma D, Jin X, Liu Z, Zhao D. Experimental study on frosting and defrosting characteristics for inclined cold plates with surface wettability considered. Appl Therm Eng. 2024;239:122089. doi:10.1016/j.applthermaleng.2023.122089. [Google Scholar] [CrossRef]
9. Fayz-Al-Asad M, Mebarek-Oudina F, Vaidya H, Hasan MS, Sarker MMA, Ismail AI. Finite element analysis for magneto-convection heat transfer performance in vertical wavy surface enclosure: fin size impact. Front Heat Mass Transf. 2024;22(3):817–37. doi:10.32604/fhmt.2024.050814. [Google Scholar] [CrossRef]
10. Saleh ZM, Al-Turaihi RS, Kadhim ZK. Enhancement of thermal performance of counter flow double pipe heat exchanger by inserting wavy-edged tape. Front Heat Mass Transf. 2025;23(2):615–50. doi:10.32604/fhmt.2025.063404. [Google Scholar] [CrossRef]
11. Sripattanapipat S, Jain N, Bhattacharyya S, Chuwattanakul V, Naphon P, Eiamsa-ard S. Flow topology and thermal mechanism in turbulent channel flow with tapered V-shaped baffles. Case Stud Therm Eng. 2025;73(1):106610. doi:10.1016/j.csite.2025.106610. [Google Scholar] [CrossRef]
12. Parlak Z. Optimal design of wavy microchannel and comparison of heat transfer characteristics with zigzag and straight geometries. Heat Mass Transf. 2018;54(11):3317–28. doi:10.1007/s00231-018-2375-6. [Google Scholar] [CrossRef]
13. Ma Q, Wu X, Chu F, Zhu B. Experimental and numerical investigations of frost formation on wavy plates. Appl Therm Eng. 2018;138(1):627–32. doi:10.1016/j.applthermaleng.2018.04.098. [Google Scholar] [CrossRef]
14. Wu W, Luo J, Li D, Feng X, Tang L, Fang Z, et al. Experimental investigation of heat transfer performance of a finned-tube heat exchanger under frosting conditions. Sustain Cities Soc. 2022;80:103752. doi:10.1016/j.scs.2022.103752. [Google Scholar] [CrossRef]
15. Hu H, Chen X, Wang Q, Li Q, Niu D, Du M. Droplet self-driven characteristics on wedge-shaped surface with composite gradients: a molecular dynamics study. Front Heat Mass Transf. 2024;22(4):1071–85. doi:10.32604/fhmt.2024.054218. [Google Scholar] [CrossRef]
16. Huang Q, Gong J, Jin L, Liu G, Wu X, Hou J. A micro-nano composite superhydrophobic surface with good frost suppression performance and durability. J Enh Heat Transf. 2022;29(7):29–49. doi:10.1615/jenhheattransf.2022042325. [Google Scholar] [CrossRef]
17. Jiang J, Lu GY, Tang GH. Inhibition of surface ice nucleation by combination of superhydrophobic coating and alcohol spraying. Int J Heat Mass Transf. 2019;134:628–33. doi:10.1016/j.ijheatmasstransfer.2019.01.080. [Google Scholar] [CrossRef]
18. Haque MR, Das SR, Betz AR. Experimental investigation of condensation and freezing phenomena on hydrophilic and hydrophobic graphene coating. Appl Therm Eng. 2019;160:113987. doi:10.1016/j.applthermaleng.2019.113987. [Google Scholar] [CrossRef]
19. Gao R, Song M, Chao CYH, Lin S, Zhang L, Zhang X. Review on condensation frosting and defrosting experiments for superhydrophobic surfaces. Appl Therm Eng. 2024;236:121691. doi:10.1016/j.applthermaleng.2023.121691. [Google Scholar] [CrossRef]
20. Arianpour F, Farzaneh M, Kulinich SA. Hydrophobic and ice-retarding properties of doped silicone rubber coatings. Appl Surf Sci. 2013;265:546–52. doi:10.1016/j.apsusc.2012.11.042. [Google Scholar] [CrossRef]
21. Alizadeh A, Yamada M, Li R, Shang W, Otta S, Zhong S, et al. Dynamics of ice nucleation on water repellent surfaces. Langmuir. 2012;28(6):3180–6. doi:10.1021/la2045256. [Google Scholar] [PubMed] [CrossRef]
22. Boyina KS, Mahvi AJ, Chavan S, Park D, Kumar K, Lira M, et al. Condensation frosting on meter-scale superhydrophobic and superhydrophilic heat exchangers. Int J Heat Mass Transf. 2019;145:118694. doi:10.1016/j.ijheatmasstransfer.2019.118694. [Google Scholar] [CrossRef]
23. Wang F, Zhou Y, Yang W, Ni M, Zhang X, Liang C. Anti-frosting performance of sprayable superhydrophobic coating suitable for outdoor coil of air source heat pump. Appl Therm Eng. 2020;169:114967. doi:10.1016/j.applthermaleng.2020.114967. [Google Scholar] [CrossRef]
24. Rahbarshahlan S, Esmaeilzadeh E, Rostamzadeh Khosroshahi A, Ghaffarzadeh Bakhshayesh A. Numerical simulation of fluid flow and heat transfer in microchannels with patterns of hydrophobic/hydrophilic walls. Eur Phys J Plus. 2020;135(2):157. doi:10.1140/epjp/s13360-019-00095-y. [Google Scholar] [CrossRef]
25. Chen H, Cheng K, Zhang Y, Zhang W. Effect of surface wettability on the flow and heat transfer performance of pulsating heat pipe. Front Heat Mass Transf. 2025;23(1):361–81. doi:10.32604/fhmt.2025.059837. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools