 Open Access
Open Access
ARTICLE
The challenge of molecular selection in liver-limited metastatic colorectal cancer for surgical resection: a systematic review and meta-analysis in the context of current and future approaches
1 Experimental and Clinical Pharmacology Unit, Centro di Riferimento Oncologico di Aviano (CRO) IRCCS, Aviano, 33081, Italy
2 Department of Medicine (DMED), University of Udine, Udine, 33100, Italy
3 Unit of Cancer Epidemiology, Centro di Riferimento Oncologico di Aviano (CRO) IRCCS, Aviano, 33081, Italy
4 Department of Oncology and Hemato-Oncology, Università degli Studi di Milano, Milan, 20122, Italy
5 Department of Hematology, Oncology, and Molecular Medicine, Grande Ospedale Metropolitano Niguarda, Milan, 20122, Italy
6 Oncology Pharmacy Unit, IRCCS Istituto Romagnolo per lo Studio dei Tumori Dino Amadori, Meldola, 47014, Italy
7 Pharmacy Unit, Fondazione IRCCS Istituto Tumori di Milano, Milan, 20122, Italy
8 Department of Pharmacy, Ca’ Foncello Treviso Regional Hospital, Piazzale Ospedale 1, Treviso, 31100, Italy
9 Department of Medical Surgical and Health Sciences, University of Trieste, Trieste, 34127, Italy
10 Hospital Pharmacy, Cattinara Hospital, Azienda Sanitaria Universitaria Giuliano Isontina, Trieste, 34148, Italy
11 Department of Oncology and Onco-Hematology, Postgraduate School of Clinical Pharmacology and Toxicology, University of Milan, Milan, 20122, Italy
* Corresponding Authors: FEDERICA TOSI. Email: ; ELENA PERUZZI. Email:
Oncology Research 2024, 32(9), 1407-1422. https://doi.org/10.32604/or.2024.049181
Received 29 December 2023; Accepted 29 February 2024; Issue published 23 August 2024
Abstract
Objectives: Treatment of metastatic colorectal cancer (mCRC) includes resection of liver metastases (LM), however, no validated biomarker identifies patients most likely to benefit from this procedure. This meta-analysis aimed to assess the impact of the most relevant molecular alterations in cancer-related genes of CRC (i.e., RAS, BRAF, SMAD4, PIK3CA) as prognostic markers of survival and disease recurrence in patients with mCRC surgically treated by LM resection. Methods: A systematic literature review was performed to identify studies reporting data regarding survival and/or recurrence in patients that underwent complete liver resection for CRC LM, stratified according to RAS, BRAF, PIK3CA, and SMAD4 mutational status. Hazard ratios (HRs) from multivariate analyses were pooled in the meta-analysis and various adjustment strategies for confounding factors were combined. The search was conducted in numerous databases, including MEDLINE (PubMed), Embase, Cumulative Index to Nursing and Allied Health Literature (CINAHL) (EBSCO host), and WHO Global Index Medicus, through March 18th, 2022. Meta-analyses, editorials, letters to the editor, case reports, studies on other primary cancers, studies with primary metastatic sites other than the liver, studies lacking specific oncological outcome variables or genetic data, non-English language studies, and studies omitting residual disease data from liver metastasectomy were excluded. The remaining 47 studies were summarized in a descriptive table which outlines the key characteristics of each study and final results were graphically presented. Results: RAS mutation status was negatively associated with overall survival (OS) (HR, 1.68; 95% CI, 1.54–1.84) and recurrence free survival (RFS) (HR, 1.46; 95% CI, 1.33–1.61). A negative association was also found for BRAF regarding OS (HR, 2.64; 95% CI, 2.15–3.24) and RFS (HR, 1.89; 95% CI, 1.32–2.73) and SMAD4 regarding OS (HR, 1.93; 95% CI, 1.56–2.38) and RFS (HR, 1.95; 95% CI, 1.31–2.91). For PIK3CA only three studies were eligible and no significant association with either OS or RFS could be highlighted. Conclusion: RAS, BRAF, and SMAD4 are negatively associated with OS and RFS in patients undergoing curative liver metastasectomy from colorectal cancer. No conclusion can be drawn for PIK3CA due to the limited literature availability. These data support the integration of RAS, BRAF, and SMAD4 mutational status in the surgical decision-making for colorectal liver metastasis. Nevertheless, we have to consider several limitations, the major ones being the pooling of results from studies that evaluated patient outcomes as either disease-free survival (DFS) or RFS; the inclusion of patients with minimal residual disease and unconsidered potential confounding factors, such as variability in resectability definitions, chemotherapy use, and a potential interaction between biological markers and pre- and post-resection pharmacological treatments.Graphic Abstract
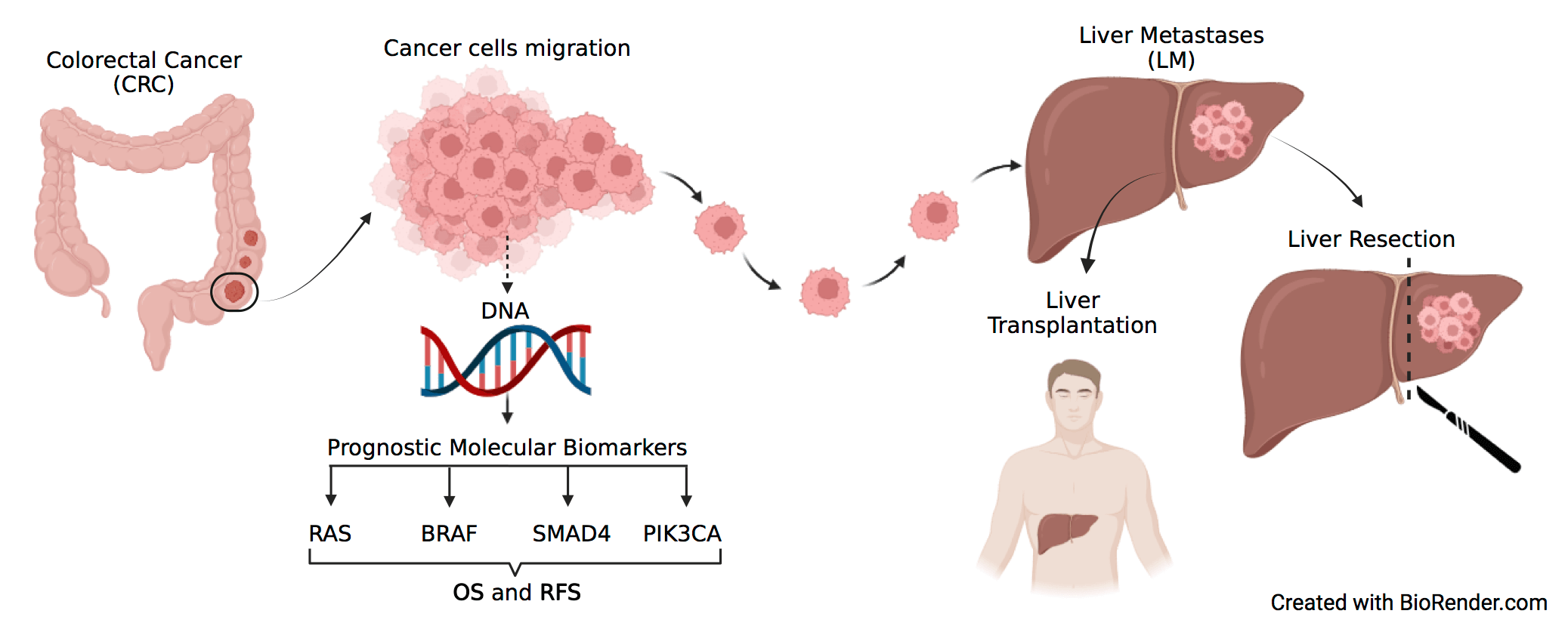
Keywords
Supplementary Material
Supplementary Material FileCite This Article
 Copyright © 2024 The Author(s). Published by Tech Science Press.
Copyright © 2024 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools