 Open Access
Open Access
ARTICLE
Repurposing Artesunate to Combat Progression and Metastasis via Targeting Circulating Tumor Cells
1 Laboratory of Biochemistry/Metastatic Signaling, Section of Genetics, Cell Biology and Development, Department of Biology, University of Patras, University Campus, Patras, Greece
2 Synthetic Organic Chemistry Laboratory, Department of Chemistry, University of Patras, University Campus, Patras, Greece
3 Medical Science Liaison MSL Oncology & Rare Diseases, Medical Department, Ipsen Greece & Cyprus, IPSEN SP LLC, 63 Agiou Dimitriou Str., Alimos–Athens, Greece
4 Department of Pharmacology and Physiology, University of Maryland School of Medicine, Baltimore, MD, USA
5 Department of Medical Oncology, General University Hospital of Larissa, Larissa, Greece
6 Laboratory of Rare Human Circulating Cells and Liquid Biopsy (LCCRH), University Medical Centre of Montpellier, Montpellier, France
7 CREEC, MIVEGEC, University of Montpellier, CNRS, IRD, Montpellier, France
8 European Liquid Biopsy Society (ELBS), Hamburg, Germany
* Corresponding Author: Galatea Kallergi. Email:
# These authors contributed equally to this work
Oncology Research 2026, 34(6), 13 https://doi.org/10.32604/or.2026.075600
Received 04 November 2025; Accepted 18 March 2026; Issue published 21 May 2026
Abstract
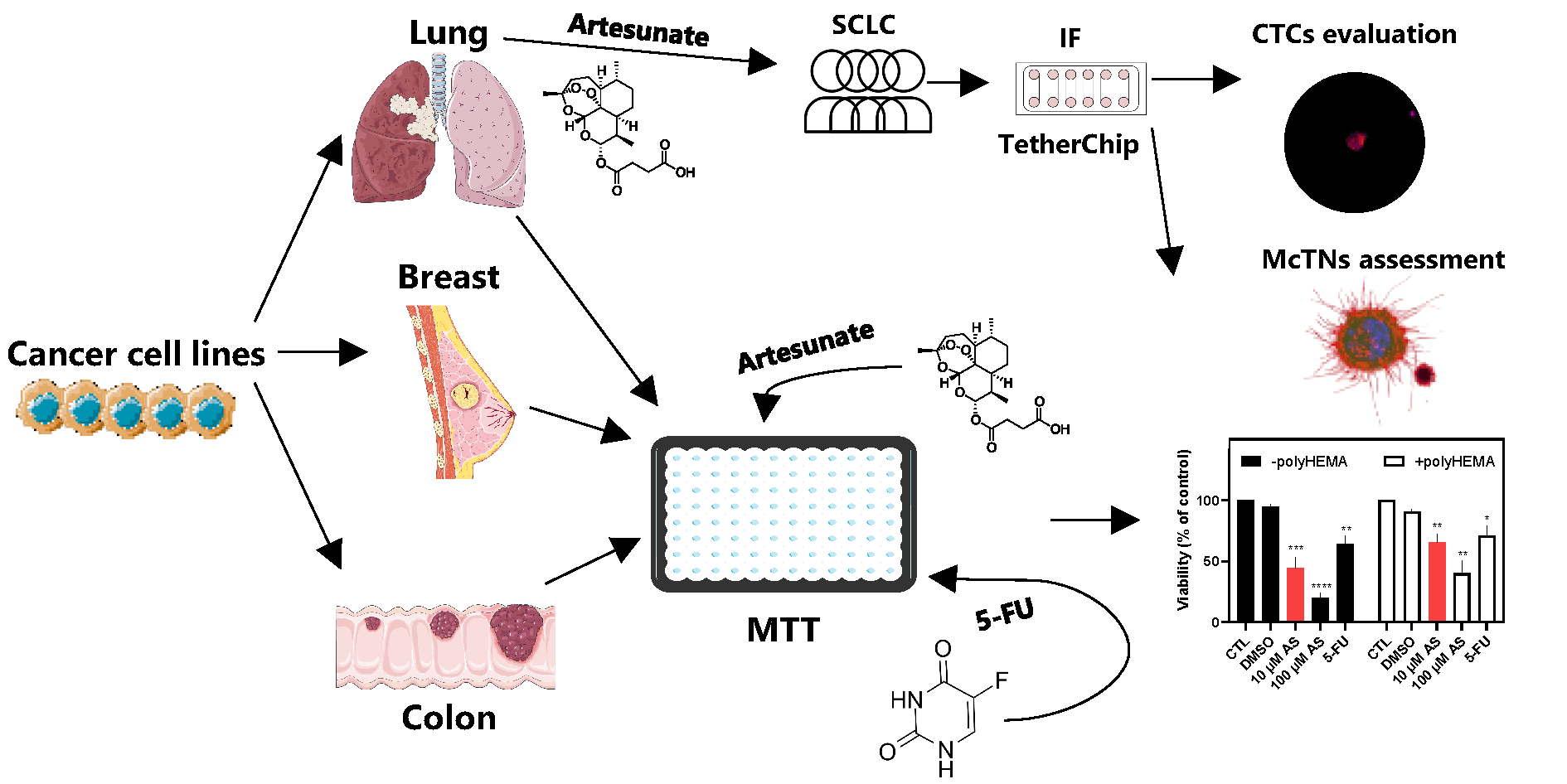
Objectives: Circulating tumor cells (CTCs) drive metastasis and exhibit resistance to conventional therapies, making them crucial therapeutic targets. Artesunate (AS), a derivative of artemisinin, displays anticancer activity, including inhibition of JunB proto-oncogene (JUNB) and programmed death ligand-1 (PD-L1) and upregulation of Vimentin (VIM), markers related to poor prognosis in CTCs. This study aimed to evaluate the effects of AS on adherent and non-adherent cancer cell lines (breast, lung, colon), the patient-derived colon cancer CTC-MCC-41 line, and CTCs from small-cell lung cancer (SCLC) patients. Methods: AS’s effect was evaluated using TetherChip technology. Cell viability was measured using MTT assay, while immunofluorescence staining and the VyCAP platform were applied to characterize and quantify CTCs. Results: AS significantly reduces viability in all tested cell lines in a time- and concentration-dependent manner, with non-adherent cells showing higher resistance. Notably, CTC-MCC-41 cells are the most sensitive to AS treatment. AS demonstrates stronger cytotoxicity than 5-fluorouracil (5-FU) in most cancer models. In SCLC patient samples, AS reduces total CTC counts (p < 0.001), eliminates aggressive phenotypes such as (CK+/CXCR4+/JUNB–) and (CK+/VIM+/GLU+), and increases apoptotic (M30+) CTCs (p = 0.021). AS additionally impairs structural features like microtentacles, which facilitate CTC reattachment. Conclusions: These findings underscore AS’s ability to target metastasis-competent and anoikis-resistant tumor cells, reducing their viability, invasiveness, and survival mechanisms. AS emerges as a promising candidate for anti-metastatic therapy and warrants further investigation in precision oncology.Graphic Abstract
Keywords
Supplementary Material
Supplementary Material FileCite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools