 Open Access
Open Access
ARTICLE
SLC16A1 Activates the STAT3/SLC7A11 Pathway to Mediate Ferroptosis Resistance and Tumor Progression in Head and Neck Squamous Cell Carcinoma
1 Department of Otorhinolaryngology Head and Neck Surgery, The First Affiliated Hospital of Anhui Medical University, Hefei, China
2 Department of Otorhinolaryngology Head and Neck Surgery, Suzhou Hospital Affiliated of Anhui Medical University, Suzhou, China
3 Department of Biochemistry & Molecular Biology, School of Basic Medicine, Anhui Medical University, Hefei, China
* Corresponding Authors: Busheng Tong. Email: ; Yehai Liu. Email:
; Huaiyuan Zong. Email:
(This article belongs to the Special Issue: Next-Generation Oncology: Unearthing and Validating Novel Therapeutic Targets)
Oncology Research 2026, 34(5), 34 https://doi.org/10.32604/or.2026.077171
Received 03 December 2025; Accepted 28 February 2026; Issue published 22 April 2026
Abstract
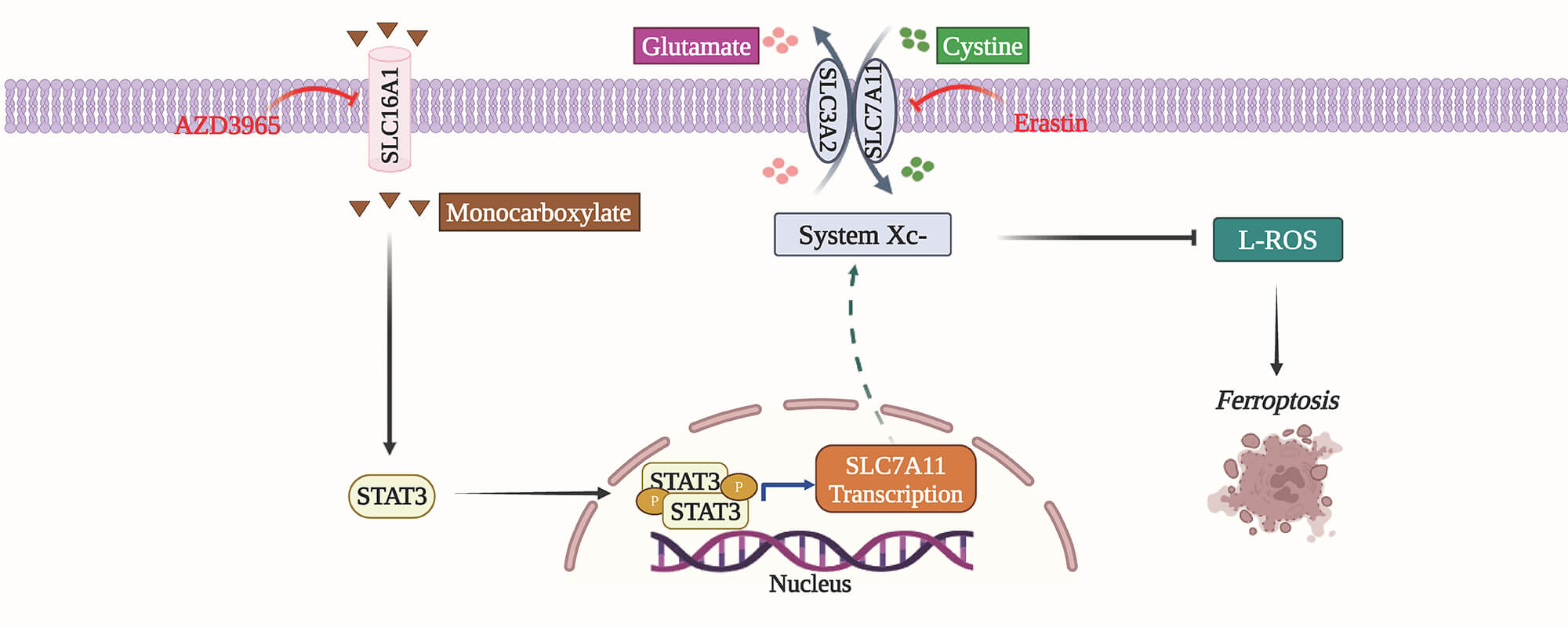
Background: In head and neck squamous cell carcinoma (HNSCC), solute carrier family 16 member 1 (SLC16A1) is associated with tumor advancement and reduced sensitivity to ferroptosis, yet the molecular basis of these effects remains unclear. This study seeks to uncover how SLC16A1 contributes to HNSCC tumorigenesis. Methods: To elucidate how SLC16A1 drives HNSCC progression via ferroptosis resistance, we performed RNA sequencing on SLC16A1-knockdown HNSCC cells and controls, followed by functional validation. We next systematically assessed the role of the candidate molecule solute carrier family 7 member 11 (SLC7A11) in HNSCC progression and resistance to ferroptosis using loss- and gain-of-function experiments in vitro and xenograft-based assays in vivo. Finally, we applied RNA interference and validated expression changes by quantitative real-time polymerase chain reaction and immunoblotting to map the signaling pathway by which SLC16A1 controls SLC7A11 expression. Results: Integrated RNA sequencing and functional assays identified SLC7A11 as a key downstream effector of SLC16A1. SLC7A11 mediates SLC16A1-driven tumor cell proliferation, ferroptosis resistance, and tumorigenesis. Mechanistically, SLC16A1 activates signal transducer and activator of transcription 3 (STAT3) to transcriptionally upregulate SLC7A11 expression. Conclusion: Our study defines a novel SLC16A1–STAT3–SLC7A11 signaling axis that promotes HNSCC progression by conferring robust resistance to ferroptosis. This axis may be leveraged as a therapeutic target to mitigate treatment resistance.Graphic Abstract
Keywords
Supplementary Material
Supplementary Material FileCite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools