 Open Access
Open Access
ARTICLE
Phytochemical Profile, Fatty Acid Composition, Mineral Content, and α-Amylase/α-Glucosidase Inhibitory Activities of Date Seed Extracts from Four Moroccan Varieties Grown in the Figuig Oasis
1 Electrochemistry Team’s Research, Applied Chemistry and Environment Laboratory (LCAE), Faculty of Sciences, University Mohammed First (UMP), Oujda, Morocco
2 Laboratory of Bioresources, Biotechnology, Ethnopharmacology, and Health, Faculty of Sciences, University Mohammed First (UMP), BP 717, Oujda, Morocco
3 Laboratoire d’Amélioration des Productions Agricoles, Biotechnologie et Environnement (LAPABE), Faculté des Sciences, University Mohammed First (UMP), Oujda, Morocco
* Corresponding Author: Amine Elbouzidi. Email:
(This article belongs to the Special Issue: Medicinal Plants and Natural Bioactives: From Pharmacology to Cosmeceutical Innovation)
Phyton-International Journal of Experimental Botany 2026, 95(2), 12 https://doi.org/10.32604/phyton.2026.074293
Received 08 October 2025; Accepted 19 January 2026; Issue published 28 February 2026
Abstract
Date palm (Phoenix dactylifera L.) seeds, often discarded as by-products during date fruit processing, represent approximately 6–20% of the fruit’s mass depending on cultivar and maturity stage. Although traditionally used for producing activated charcoal and in environmental remediation, their pharmacological potential remains underexplored. This study aimed to evaluate the chemical composition, biological activity, and potential commercial value of seeds from four widely consumed Saharan cultivars (Assiane, Boufegouss, Aziza, and Majhoul) collected in the Figuig region. Soxhlet extraction was employed to obtain seed extracts, which were subsequently characterized by qualitative and quantitative phytochemical screening. The analyses revealed the presence of polyphenols, flavonoids, terpenoids, steroids, alkaloids, and other secondary metabolites. The Aziza and Boufegouss varieties were particularly rich in flavonoids (82.09–139.61 mg quercetin equivalents/100 g dry matter) and total polyphenols (69.81–1183.96 mg gallic acid equivalents/100 g dry matter). Mineral profiling confirmed the occurrence of essential trace elements, including Zn, Cu, Fe, and Co. To investigate their antidiabetic potential, in vitro assays of α-glucosidase and α-amylase inhibition were performed. Acetone extracts demonstrated significant inhibitory activity against both enzymes, suggesting a promising role of P. dactylifera seed extracts as natural agents for the management of type 2 diabetes mellitus.Keywords
In Morocco, the date palm (Phoenix dactylifera L.) is far more than a simple agricultural commodity; it is a cornerstone of cultural heritage, traditional knowledge, and economic resilience. Cultivated for centuries in the arid and semi-arid regions of the country, particularly in southern oases, the date palm has shaped both the landscape and the livelihoods of local populations. Its adaptation to harsh environmental conditions, such as extreme heat and low rainfall, underscores its ecological importance as a keystone species for desert agriculture. The palm’s ability to thrive in such climates has long ensured a reliable source of sustenance, fuel, and raw material, embedding it deeply within Morocco’s socio-economic fabric [1].
Among Morocco’s oases, the town of Figuig stands out as a historical center of date palm cultivation. This region is particularly renowned for producing high-quality fruits distinguished by their sweetness, texture, and unique organoleptic properties. Beyond their gastronomic value, date fruits also hold nutritional significance, being rich in natural sugars, dietary fiber, vitamins, and minerals. Morphologically, the fruit is classified as a berry, with variable shapes—oblong, oval, or spherical. It is composed of three main layers: the pericarp (outer skin), mesocarp (fleshy pulp), and endocarp (hard shell), which encloses the seed.
Date seeds, sometimes referred to as date stones or pits, represent a lignocellulosic by-product that constitutes approximately 6–14% of the total fruit weight [2]. With Morocco ranking among the top global producers, seed generation occurs on a massive scale. According to FAOSTAT (2024), an estimated 103,000 tonnes of dates were harvested in the country, corresponding to approximately 6164–14,660 tonnes of seeds arising from both direct consumption and industrial processing. In many cases, these by-products are discarded or used in low-value applications such as animal feed, thereby contributing to waste management challenges. Their underutilization not only raises ecological concerns but also represents a missed opportunity for bioeconomic valorization.
Recent scientific investigations have shed light on the remarkable chemical composition of date seeds, positioning them as a promising raw material for value-added applications. Seeds are reported to contain significant quantities of phenolic compounds and flavonoids, conferring potent antioxidant properties, alongside dietary fiber, proteins [3], carbohydrates, and essential minerals including potassium, calcium, phosphorus, sodium, and iron [4]. This diverse phytochemical and nutritional profiles have sparked interest in both food science and pharmacology. For instance, date seed powder has been incorporated into bakery products to enhance their nutritional quality, while oil extracted from seeds has been evaluated for its fatty acid composition and antioxidant activity.
Beyond food applications, date seeds have also found uses in industrial and pharmaceutical sectors. Activated carbon derived from seed biomass has been applied in water purification and gas adsorption processes [5]. Additionally, roasted date seed powder has entered the market as a caffeine-free natural coffee substitute, offering potential for functional beverage innovation [6]. From an ethnopharmacological perspective, seed extracts have demonstrated bioactivities such as anti-inflammatory, hepatoprotective, and hypoglycemic effects, reinforcing their potential as an untapped source of nutraceuticals.
In light of these findings, the exploration of Moroccan date seeds, particularly those from the Saharan region of Figuig, is of considerable scientific and economic interest. Characterizing their phytochemical content, mineral composition, and bioactive potential not only supports sustainable waste valorization but also contributes to the development of novel applications in food, pharmaceutical, and environmental industries. This study therefore aims to bridge the gap between traditional uses and modern scientific evidence by systematically evaluating the chemical and biological properties of seeds from four locally significant cultivars: Assiane, Boufegouss, Aziza, and Majhoul.
Hexane, chloroform, acetone, ethanol, and methanol of analytical grade (≥99.5%) were procured from Sigma-Aldrich (St Louis, MO, USA). Reagents including DPPH radical were used in their highly pure form.
2.2 Collection of Dates/for Seeds
A total of four date fruit samples were obtained from several oases throughout Morocco during the 2021 harvest season. These cultivars, originating from the Figuig region, comprised Aziza Bouzid, Bouffegous, Assiane, and El Mejhol. The varieties that mature later in the season were collected in October and November. The date seeds were separated from the fruit and transported to the laboratory of the Department of Chemistry at Mohammed First University in Oujda. Subsequently, the seeds were washed with distilled water and air-dried at ambient temperature (25 ± 2°C) and 45–50% relative humidity for 7 days until constant weight was reached, ensuring uniform drying across all samples. The final moisture content was verified by repeated weighing. The dried seeds were then finely ground into powder using an electronic shredder. Soxhlet extraction was performed for each solvent, and the extraction yields were calculated. Residual solvents were checked using gas chromatography (GC) to confirm complete removal. Additionally, selected fractions were analyzed by HPLC to validate the presence of key phytochemicals and ensure the quality and reproducibility of the extracts.
The weight of ten fruits from each cultivar was measured using an electronic analytical balance with an accuracy of ±0.001 g. The mean values were calculated to obtain representative weight data for each variety.
The preparation of crude organic extracts was carried out by successive exhaustion of the powder in a Soxhlet apparatus using four organic solvents of increasing polarity: n-hexane, chloroform, acetone, and finally distilled water. Each solvent extraction was performed for 8 h. The extracts were then concentrated to dryness under reduced pressure at 40°C using a rotary evaporator, and the resulting residues were stored at 4°C.
2.5 Phytochemical Investigation of Date Seeds
The phytochemical screening was based on characteristic colorimetric or precipitation reactions induced by specific reagents.
- Steroids and Terpenoids (Liebermann–Burchard Test): Steroidal and terpenoid constituents were detected by dissolving the dried extract in chloroform, followed by the addition of acetic anhydride. Subsequent introduction of 2–3 drops of concentrated sulfuric acid produced a color shift from red to blue and finally to dark green, confirming their presence [7].
- Alkaloids (Dragendorff’s Test): Alkaloids were identified by mixing 2 mL of the extract with 1 mL of Dragendorff’s reagent. The formation of an orange to reddish precipitate indicated a positive result [8].
- Flavonoids: To test for flavonoids, 0.5 g of magnesium was treated with a few drops of concentrated hydrochloric acid and then added to 0.5 mL of the extract. The appearance of a pinkish-red coloration confirmed their presence [8].
- Phenolics and Tannins: For phenolic compounds and tannins, 2 mL of the extract were treated with a few drops of 1% alcoholic ferric chloride solution. The emergence of a dark blue or bluish-green coloration was taken as evidence of these compounds [9].
- Coumarins: The presence of coumarins was established by mixing 2 mL of the extract with 3 mL of 10% sodium hydroxide solution. A yellow coloration indicated a positive reaction [9].
- Saponins: Saponins were detected by vigorously mixing 2 mL of the extract with distilled water. The persistence of foam after 15 s confirmed their presence [9].
- i.Total phenolic content
The quantification of total phenolic content in the extracts was performed according to the Folin–Ciocalteu colorimetric assay described by Singleton et al. (1999), with minor modifications. Briefly, 50 μL of each crude extract were combined with 250 μL of Folin–Ciocalteu reagent (10%) and left to react in the dark for one hour. Absorbance values were then recorded at 760 nm using a UV–Vis spectrophotometer (Jenway 6300, Staffordshire, UK). Phenolic concentrations were calculated and expressed as milligrams of gallic acid equivalents (GAE) per 100 g of dry matter. Results are presented as the mean ± standard deviation of three independent replicates [10].
- ii.The total flavonoid
The quantification of flavonoids in the extracts was performed using a colorimetric assay adapted from Quettier-Dellen et al. (2000). In practice, 1 mL of the extract was mixed with an equal volume of aluminum chloride hexahydrate (AlCl3·6H2O) solution. After standing for 15 min at ambient temperature, the absorbance of the resulting complex was read at 430 nm. The flavonoid content was expressed as milligrams of quercetin equivalents (QE) per gram of dry material, with values presented as mean ± standard deviation from three independent measurements [11].
2.5.3 Gas Chromatography-Mass Spectroscopy (GC-MS) Analysis
The fatty acid profile of date seed oil was investigated through gas chromatography coupled with mass spectrometry (GC–MS) on a Shimadzu QP2010 system (Shimadzu, Tokyo, Japan). Prior to analysis, the oils were transesterified to their corresponding fatty acid methyl esters using sodium methanolate, following the (ISO 21:1978) protocol [12]. Separation was achieved on a DB-5 capillary column (30 m × 0.25 mm i.d., 0.25 μm film thickness) under helium as the carrier gas at a constant flow of 1 mL/min. The injector was operated in splitless mode, with 1 μL of hexane-diluted sample (1:100, v/v) introduced, while the ion source was maintained at 210°C. The resulting chromatograms allowed for qualitative and quantitative characterization of the fatty acids present in the samples [13].
Dietary minerals are indispensable micronutrients that play critical roles in human health, contributing to skeletal development, cellular homeostasis, enzymatic activity, and numerous biochemical pathways. Seeds of Phoenix dactylifera L. from the Oriental region of Figuig represent a valuable reservoir of these elements, highlighting their potential nutritional and pharmacological relevance.
In this study, the concentrations of essential and trace metallic elements in date seed powders were determined following a modified AOAC (1970) protocol [14]. Briefly, 0.5 g of each powdered seed sample was introduced into a digestion flask, to which 20 mL of concentrated nitric acid (HNO3, 69.5%) and 100 mL of double-distilled water were added. The mixture was subjected to wet digestion at 100°C for three hours to achieve complete mineralization of the organic matrix. After cooling, the digests were filtered, and the filtrates were diluted with double-distilled water to a final volume of 250 mL.
The quantification of trace and essential metals—including zinc (Zn), copper (Cu), iron (Fe), cobalt (Co), nickel (Ni), chromium (Cr), and cadmium (Cd)—was carried out using atomic absorption spectrophotometry (AAS). To ensure reliability and reproducibility, all analyses were performed in triplicate, and results were expressed as mean values ± standard deviation.
2.7 DPPH Radical Scavenging Assay
The antioxidant capacity of the extracts was assessed using the DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging assay, adapted from the procedure originally described by Brand-Williams et al., with minor modifications [15]. A stock solution of DPPH (0.1 mM) was prepared in methanol immediately before use to ensure stability.
For each test, 1 mL of extract at different concentrations (0.062–1 mg/mL) was added to 2 mL of the DPPH solution. The mixtures were gently vortexed and left to stand for 30 min in the dark at room temperature to allow the reaction to occur. The decrease in absorbance was then recorded at 517 nm using methanol as a blank.
The percentage of radical-scavenging activity was calculated using the following equation:
Acontrol is the absorbance of the DPPH solution without extract
Asample is the absorbance after incubation with the extract.
2.8 Investigation of In Vitro Inhibitory Effects on Pancreatic α-Amylase and Intestinal α-Glucosidase
2.8.1 In Vitro Assessment of the Inhibitory Effect on Pancreatic α-Amylase Activity
The inhibitory effect of α-amylase was evaluated following the procedure described by Thalapaneni et al. (2008), with slight modifications [16]. Briefly, 200 µL of phosphate buffer (0.2 M, pH 6.9) were mixed with 200 µL of α-amylase solution (13 IU) and 100 µL of the four extracts or acarbose at different concentrations (0.062, 0.125, 0.25, 0.5, and 1 mg/mL). The reaction mixture was preincubated at 37°C for 10 min, after which 200 µL of a 1% starch solution prepared in phosphate buffer was added as substrate. The mixture was then incubated for 20 min at 37°C, and the reaction was terminated by the addition of 600 µL of 3,5-dinitrosalicylic acid (DNSA) reagent. The samples were subsequently heated in boiling water for 8 min, rapidly cooled in an ice bath, and diluted with 1 mL of distilled water. The absorbance was measured at 540 nm.
The α-amylase inhibitory activity was calculated using the equation below:
2.8.2 In Vitro Assessment of the Inhibitory Effect on Intestinal α-Glucosidase Activity
The inhibitory activity of intestinal α-glucosidase was determined according to the procedure of Elrherabi et al., with slight modifications [17]. The assay was based on quantifying D-glucose released from sucrose hydrolysis. Twenty microliters of acarbose (0.062, 0.125, 0.25, 0.5, and 1 mg/mL), as well as Ethanolic Extract (EET) and Acetonic Extract (EAC) from each variety at equivalent concentrations, were added to the reaction mixture, which consisted of 100 µL sucrose solution (50 mM), 100 µL α-glucosidase (10 IU), and 1000 µL phosphate buffer (50 mM, pH 7.5). A control containing 10 µL phosphate buffer was also prepared. After incubation at 37°C for 25 min, the enzymatic reaction was terminated by heating at 100°C for 5 min. The released D-glucose was quantified using the glucose oxidase–peroxidase GOD/POD kit, and absorbance was measured at 500 nm [18]. The inhibition percentage was then calculated using the standard equation as in 2.8.1.
All results are expressed as mean ± SD (n = 3). Normality of data was assessed using the Shapiro–Wilk test, and homogeneity of variances using Levene’s test. When assumptions were satisfied (p > 0.05), differences among cultivars were evaluated using one-way ANOVA followed by Tukey’s HSD post-hoc test. When normality or homoscedasticity was not met, a Kruskal–Wallis test followed by Dunn’s test (Bonferroni correction) was used. Lowercase letters indicate significant differences between cultivars (p < 0.05), and uppercase letters indicate differences between solvents within each cultivar
The morphometric evaluation of date seeds from the four studied cultivars is presented in Fig. 1. The results indicate that the mean seed weight exceeds 1 g in the El Mejhol and Boufegous varieties, whereas the Aziza and Assiane seeds exhibit lower weights, generally below 1 g.
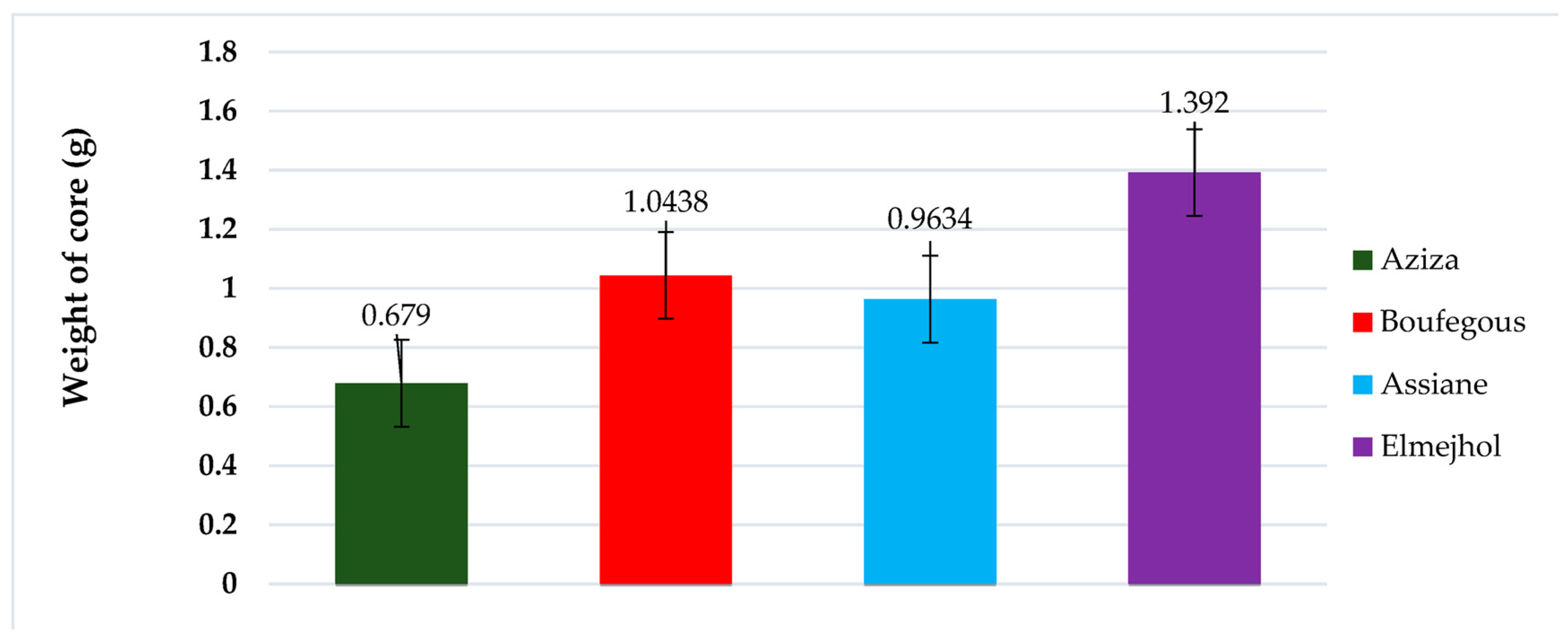
Figure 1: Morphological parameters of date seeds from four cultivars cultivated in the Figuig Oasis. Data represent the mean ± standard deviation (SD) of measurements obtained from 10 individual seeds (n = 10). Error bars indicate variability among samples.
These findings highlight noticeable variability in seed size and weight among the cultivars, reflecting the inherent morphological diversity of P. dactylifera. Such differences may be influenced not only by genetic factors but also by local environmental conditions, including soil composition, irrigation practices, and climatic variability. Previous studies have similarly reported significant inter- and intra-cultivar variation in the morphometric traits of date fruits and seeds, attributing this to both hereditary factors and agro-ecological conditions [19]. Larger seed dimensions, as observed in El Mejhol and Boufegous, are often associated with later-maturing cultivars, which typically develop under extended periods of high temperature and lower humidity, conditions that favor greater biomass accumulation [20].
Morphometric variability is not only of taxonomic or agronomic interest but also has practical implications. Seed size can influence the yield of bioactive compounds and the efficiency of industrial processing, such as extraction for nutraceutical or functional food applications. The observed differences therefore underscore the importance of cultivar selection and local growing conditions when considering the valorization of date seeds as a raw material for food and pharmaceutical industries.
First and foremost, the extraction process for date stone powder employs solvents with increasing polarity, each variety undergoes this process to achieve optimal extraction with the respective solvent used the yield percentages of the various extracts obtained from the powder of each variety (Fig. 2), utilizing the soxhlet apparatus, have been arranged in the following ascending order as: Hexane (HEE) < Chloroform (CFE) < Acetone (ACE) < Ethanol (ETE) < Water (EE).
The Aziza variety exhibits a high total recovery rate (51.30%), with fibre and residues accounting for most of the residual fraction, whereas the Mejhoul variety shows a lower total yield (32.15%).
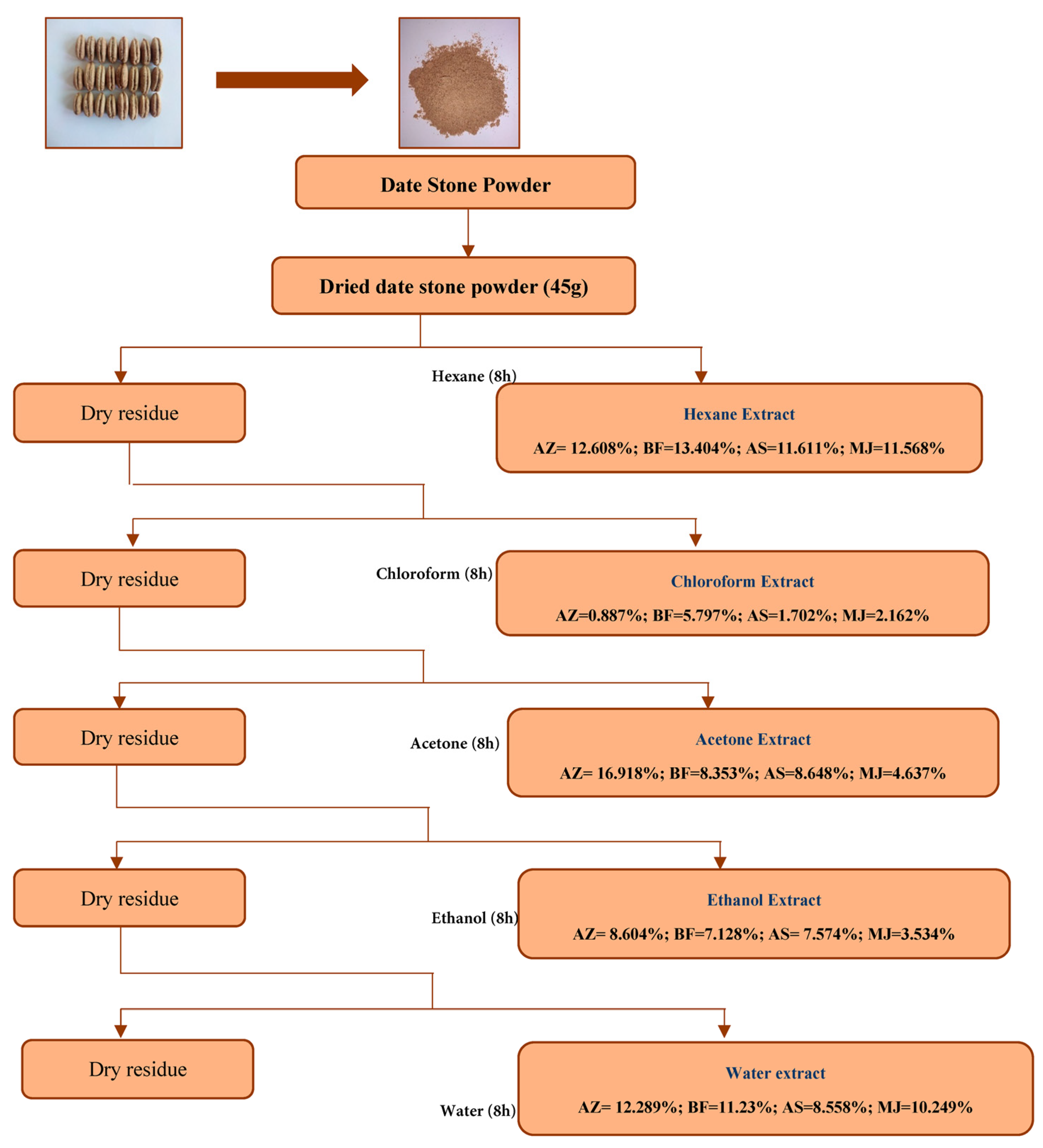
Figure 2: Extraction yields of different solvents (hexane, chloroform, acetone, ethanol, and water) from date stone varieties (AZ: Aziza; BF: Boufegous; AS: Assiane; MJ: Mejhoul).
In this section, we sought to emphasize the qualitative presence of secondary metabolites in the different date seed varieties under investigation, including terpenoids, steroids, alkaloids, flavonoids, and related classes of compounds. The results of the phytochemical screening are summarized in the following Table 1. for each cultivar.
Table 1: Qualitative assessment of phytochemical constituents in extracts from date pits of four varieties. “+”: Present; “−”: Absent.
| Extracts | Varieties | Terpenoids | Steroids | Alkaloids | Flavonoids | Phenolic and Tannins | Coumarins | Saponins |
|---|---|---|---|---|---|---|---|---|
| Chloroform (CHE) | Aziza | + | + | − | − | − | + | − |
| Bouffegous | + | − | − | − | − | + | − | |
| Assiane | + | + | − | − | − | + | − | |
| Mejhoul | + | + | − | − | − | + | − | |
| Acetone (ACE) | Aziza | + | + | + | + | + | + | − |
| Bouffegous | + | + | + | + | + | + | − | |
| Assiane | + | + | − | + | + | + | − | |
| Mejhoul | + | + | − | + | + | + | − | |
| Ethanol (ETE) | Aziza | + | + | + | + | + | + | − |
| Bouffegous | + | + | + | + | + | + | − | |
| Assiane | + | + | + | + | + | + | − | |
| Mejhoul | + | + | + | + | + | + | − | |
| Water (WAE) | Aziza | + | + | + | + | + | − | − |
| Bouffegous | + | + | + | + | + | − | + | |
| Assiane | + | + | + | + | + | − | − | |
| Mejhoul | + | + | + | + | + | − | − |
Overall, our analyses revealed a wide diversity of phytochemicals in the kernels of the four studied varieties. Flavonoids, phenolic compounds, alkaloids, terpenoids, and steroids were consistently detected in acetone, ethanol, and aqueous extracts across all varieties. The only notable exception was the absence of alkaloids in the acetone extract of the Assiane cultivar. By contrast, chloroform extracts generally yielded negative results for alkaloids, flavonoids, phenols, and tannins, indicating the limited solubility of these polar compounds in non-polar solvents. With respect to saponins, none were detected in any extract of the El Mejhol, Assiane, or Aziza cultivars. Interestingly, a positive reaction for saponins was observed exclusively in the aqueous extract of the Boufegous variety, suggesting cultivar-dependent variability in secondary metabolite distribution. Additionally, coumarins were consistently detected in the chloroform, acetone, and ethanol extracts of all four varieties, underlining their widespread occurrence.
These findings corroborate earlier studies. Rav, (2017) reported similar results in their phytochemical assessment of date pits from India, demonstrating the abundant presence of alkaloids, tannins, phenols, sterols, terpenes, as well as flavonoids and saponins, particularly in aqueous extracts [21]. Likewise, our results are in agreement with those obtained for the Saudi Arabian cultivar “Ajwa.” According to Zainab (2024), this variety displayed comparable profiles in terms of polyphenols, flavonoids, and alkaloids, thereby confirming the global consistency of phytochemical signatures among date seed varieties from different regions [22].
Taken together, the phytochemical screening performed here highlights the chemical richness of Moroccan date seeds, while also underlining some inter-varietal differences. Such variability is likely linked to both genetic factors and environmental influences (e.g., soil, climate, cultivation practices). Given the biological relevance of these classes of compounds—particularly flavonoids and phenolics, which are associated with antioxidant, anti-inflammatory, and antidiabetic properties—these results further justify the growing interest in date seeds as a sustainable source of bioactive molecules for nutraceutical and pharmaceutical applications.
Table 2 summarizes the total phenolic contents of date seed extracts from four Moroccan cultivars, expressed as milligrams of gallic acid equivalents (mg GAE) per 100 g of dry matter. As shown, phenolic distribution varied markedly according to both the cultivar and the extraction solvent. For total phenolics (Table 2), ANOVA revealed highly significant cultivar effects for acetone (F(3,8) = 248.6, p < 0.0001), ethanol (F(3,8) = 575.3, p < 0.0001) and water extracts (F(3,8) = 1642.4, p < 0.0001).
The aqueous extract of the Assiane variety exhibited the highest total phenolic content (452.21 ± 0.043 mg GAE/100 g), surpassing all other extracts across the four cultivars. The aqueous extract of Aziza (183.96 ± 0.008 mg GAE/100 g) was lower but still significant. The aqueous extracts of Boufegous (248.15 ± 0.013 mg GAE/100 g) and Mejhoul (253.20 ± 0.074 mg GAE/100 g) showed moderate values. The ethanol extract of Aziza also showed a notably high phenolic level (268.70 ± 0.003 mg GAE/100 g), reinforcing the conclusion that Aziza seeds are particularly rich in polyphenols. Among the acetone extracts, the Assiane variety presented the highest concentration (254.78 ± 0.006 mg GAE/100 g), while Aziza (138.34 ± 0.012 mg GAE/100 g), Mejhoul (83.70 ± 0.056 mg GAE/100 g), and Boufegous (82.61 ± 0.001 mg GAE/100 g) displayed comparatively lower values. Ethanol extracts also showed moderate levels, with Assiane (128.42 ± 0.051 mg GAE/100 g) and Mejhoul (134.05 ± 0.005 mg GAE/100 g) yielding similar contents, slightly higher than Boufegous (69.81 ± 0.009 mg GAE/100 g). In contrast, none of the cultivars showed detectable phenolic compounds in hexane extracts, which is consistent with the polarity of phenolic compounds that makes them insoluble in non-polar solvents.
Table 2: Quantification of total phenolics (expressed as mg gallic acid equivalents per 100 g of DW) in extracts of different date seeds.
| Hexane (HE) | Acetone (ACE) | Ethanol (ETE) | Water (WAE) | |
|---|---|---|---|---|
| Aziza | ND | 138.34 ± 0.012bA | 268.70 ± 0.003cB | 183.96 ± 0.008cC |
| Boufegous | ND | 82.60 ± 0.001aA | 69.81 ± 0.009aA | 248.15 ± 0.013aB |
| Assiane | ND | 254.78 ± 0.006cB | 128.41 ± 0.051bA | 452.21 ± 0.043bC |
| Mejhoul | ND | 83.70 ± 0.056aA | 134.05 ± 0.005bB | 253.20 ± 0.074aC |
These results confirm that the extraction solvent plays a decisive role in the recovery of phenolics, with polar solvents such as water and ethanol yielding higher concentrations than less polar solvents like acetone or non-polar solvents such as hexane. Similar findings have been reported by Al-Farsi and Lee (2008), who demonstrated that aqueous and hydroalcoholic extractions are more efficient in recovering hydrophilic phenolic constituents from date seeds [23]. Moreover, Habib and Ibrahim (2009) highlighted that phenolic yields are highly cultivar-dependent, which aligns with the present observation of particularly high levels in Aziza seeds compared to other Moroccan varieties [24].
The very high phenolic content of Aziza aqueous extract suggests its potential as a rich source of antioxidant compounds. Polyphenols are known to scavenge free radicals, reduce oxidative stress, and provide protective effects against chronic diseases such as diabetes, cardiovascular disorders, and cancer [25]. In addition, the moderate-to-high levels observed in Assiane extracts may also be of pharmacological interest, particularly considering the diversity of phenolic subclasses (flavonoids, tannins, phenolic acids) reported in date seeds [26].
3.4.2 Total Flavonoids Content
Table 3 presents the total flavonoid contents of the four studied date seed varieties, expressed as milligrams of quercetin equivalents (mg EQ) per 100 g of dry matter. The results demonstrate clear inter-varietal and solvent-dependent differences in flavonoid recovery. For total flavonoids (Table 3), significant cultivar differences were also observed for acetone (F(3,8) = 984.7, p < 0.0001) and water extracts (F(3,8) = 721.4, p < 0.0001). For the ethanol extract, normality was not satisfied (Shapiro–Wilk p = 0.031), therefore a non-parametric Kruskal–Wallis test was performed (H = 10.88, p = 0.012), followed by Dunn’s test with Bonferroni adjustment.
The Aziza variety consistently exhibited the highest flavonoid levels, with the aqueous extract yielding the maximum value (138.61 ± 0.15 mg EQ/100 g), followed by the acetone extract (118.92 ± 0.37 mg EQ/100 g). These results are in close agreement with those reported by Halabi et al. (2024), who studied date seeds from the Figuig region and confirmed a flavonoid content of 116.52 mg EQ/100 g for the Aziza cultivar [27]. This consistency strengthens the reliability of Aziza seeds as a particularly rich source of flavonoids.
Among the other varieties, the aqueous extract of Boufegous (82.09 ± 0.20 mg EQ/100 g) ranked second, while Assiane (50.28 ± 0.21 mg EQ/100 g) and Mejhoul (40.74 ± 0.32 mg EQ/100 g) showed lower values. In contrast, ethanol extracts generally exhibited minimal flavonoid recovery, with Assiane showing the lowest concentration (4.72 ± 0.81 mg EQ/100 g). This result corroborates the findings of Djaoudene et al. (2019), who reported a comparable value of 6.52 mg EQ/100 g in date seeds [28]. ANOVA revealed highly significant differences among cultivars for total phenolics and flavonoids (Table 2 and Table 3), with F-values ranging from 248.6 to 1642.4 (p < 0.0001). When assumptions of normality were not fulfilled (ethanol–flavonoid dataset), a Kruskal–Wallis test was used (H = 10.88, p = 0.012). Post-hoc comparisons (Tukey/Dunn) confirmed the cultivar-dependent distribution of phenolics and flavonoids, as indicated by the different superscript letters
The overall range of flavonoid contents observed in this study is consistent with earlier findings by Al-Farsi and Lee (2008), who noted that while relatively few studies have quantified flavonoids in date seeds, reported values range from 63.90 to 153.3 mg/100 g dry matter [6]. Our present results thus align well within this spectrum, particularly for Aziza, further confirming the role of solvent polarity in modulating flavonoid recovery.
Table 3: Flavonoid content (expressed as mg of quercetin equivalents per 100 g of DW) of different variety of date seeds extracts.
| Hexane (HE) | Acetone (ACE) | Ethanol (ETE) | Water (WAE) | |
|---|---|---|---|---|
| Aziza | ND | 118.92 ± 0.379bB | 16.05 ± 0.022cA | 138.61 ± 0.151cB |
| Boufegous | ND | 23.13 ± 0.308aB | 12.94 ± 0.099bcA | 82.08 ± 0.205bC |
| Assiane | ND | 23.07 ± 0.440aB | 4.72 ± 0.805aA | 50.28 ± 0.209aC |
| Mejhoul | ND | 21.09 ± 0.288aB | 10.08 ± 0.015bA | 40.74 ± 0.325aC |
The predominance of flavonoids in aqueous and acetone extracts is expected, as these solvents are more efficient in solubilizing polar polyphenolic compounds compared to ethanol or non-polar solvents such as hexane. The absence of detectable flavonoids in hexane extracts across all varieties further confirms this polarity-driven extraction pattern.
From a functional perspective, flavonoids are bioactive compounds with well-documented antioxidant, anti-inflammatory, and antidiabetic properties. Their high abundance in Aziza and Boufegous varieties suggests strong nutraceutical potential. Previous studies have associated flavonoid-rich date seed extracts with inhibition of lipid peroxidation, scavenging of reactive oxygen species, and modulation of carbohydrate metabolism, making them particularly relevant in the prevention of metabolic disorders such as diabetes and cardiovascular disease [25].
3.5 Gas Chromatography–Mass Spectrometry (GC-MS) Analysis
The fatty acid analysis revealed clear quantitative differences among the four date seed oils. Carvacrol was the major component in the Aziza variety (37.95%), while thymol acetate (12.93–19.87%) and thymol (2.15–10.01%) were present in variable amounts across the samples. Lauric acid ranged from 3.57% (EHE1) to 25.38% (EHE2), whereas palmitic acid varied between 3.32% and 10.41%. Oleic acid showed a wide distribution, from 5.72% to 31.83%, with the highest value observed in the Mejhoul extract (EHE4). Vaccenic acid was detected exclusively in the Assiane variety (36.25%). Several minor fatty acids, including caprylic, myristic, and pentadecanoic acids, were also identified in low concentrations. Overall, both major and minor fatty acids varied considerably between cultivars. The results are presented in Fig. 3, while the identified compounds and their relative percentages are summarized in Table 4.
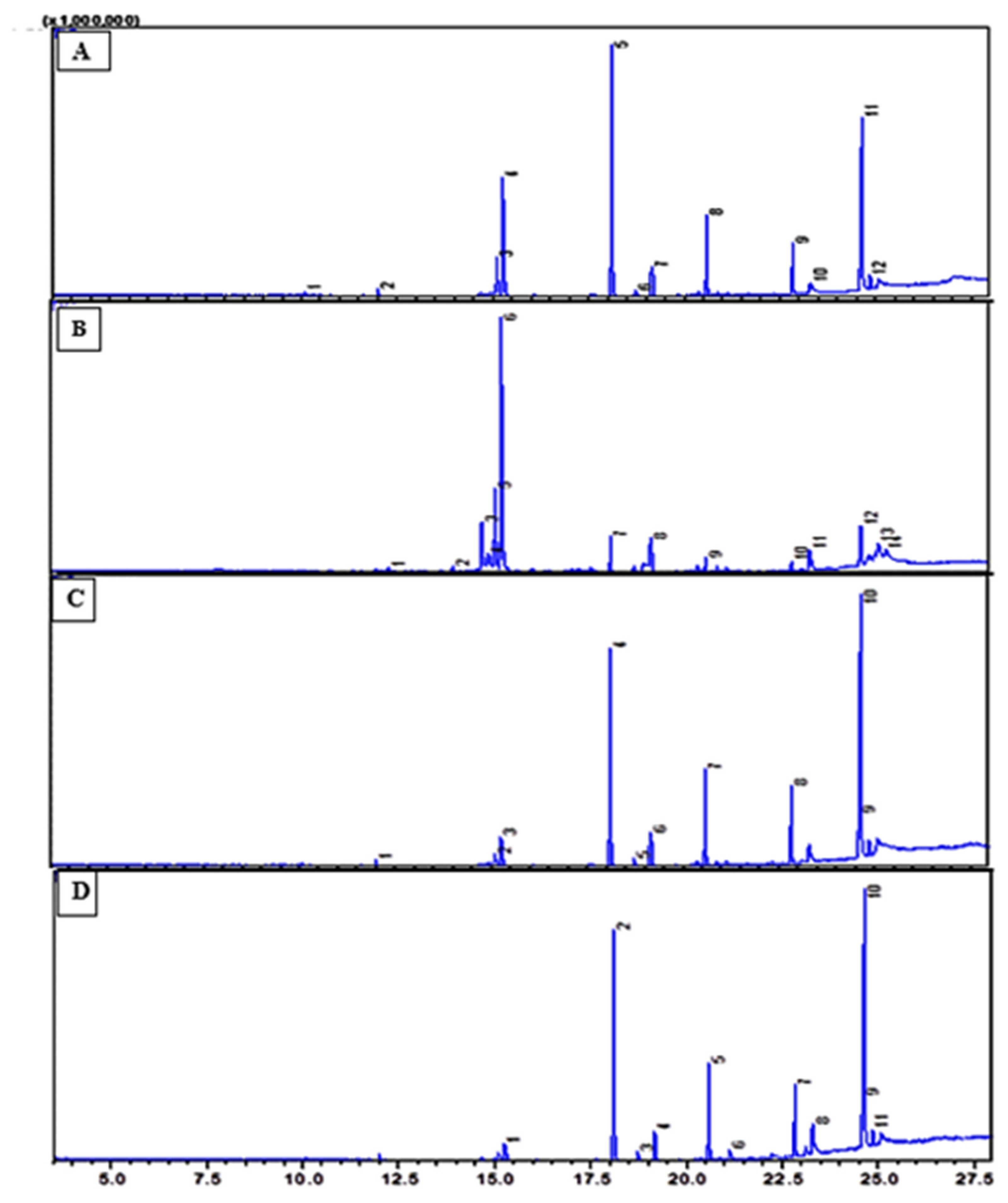
Figure 3: Comparative GC–MS chromatograms of hexane extracts obtained from the seeds of four date varieties: (A) Aziza, (B) Bouffegous, (C) Assiane, and (D) Mejhoul. Retention time: min.
In the four studied date seed oils, the fatty acid profile is mainly defined by the occurrence of four dominant fatty acids—oleic (C18:1), lauric (C12:0), and palmitic (C16:0)—present in different proportions across varieties, and phthalic acid, allyl ethyl ester. Together, these acids comprise over 75% of the total fatty acid content. Notably, certain fatty acids were found in significant quantities, particularly in specific varieties, with vaccenic acid reaching an impressive 36.25% in the Assiane cultivar, carvacrol at 37.95% in the Aziza Bouzid cultivar, and thymol acetate at 19.87% in the Boufegous cultivar. Additionally, Minor fatty acids were also identified, such as caprylic acid (C8:0), myristic acid (C14:0), and pentadecanoic acid (C15:0). Overall, the fatty acid composition of date seed oil is influenced by several factors, including the cultivar, the degree of ripeness, and the extraction technique employed [29]. Date seed oil contains a variety of fatty acids, including saturated (lauric, myristic, caprylic, pentadecanoic, lignoceric and palmitic acids), monounsaturated (oleic, vaccenic and petroselinic acids) fatty acids, comprising approximately 44% and 78% of the total content, respectively. It has been observed that Iranian date varieties (Sayer, Khenizi, Majul, Shekar, Zahedi, Goftar, and Khasuee) as well as Tunisian varieties (Deglet Nour and Allig) had higher levels of unsaturated fatty acids compared to saturated ones, similar to our case [1]. Oleic acid was the predominant unsaturated fatty acid (31%), followed by lauric acid (24.2%) in the Mejhoul variety, confirming the findings of Raza et al. [30]. In the Assiane variety, vaccenic acid was the dominant fatty acid (36.25%), followed by lauric acid (22.65%). For the Boufegous variety, the main saturated fatty acid was lauric acid (25.38%), followed by oleic acid (24.09%), confirming the finding of Raza et al., and Akbari et al. [30,31]. Additionally, the degree of unsaturation in these date seed oils is lower than that typically found in olive oil; however, they are still considered a valuable source of oleic acid, with levels comparable to those reported in rice bran oil [32].
Table 4: Fatty acid profil of date seed oil expressed as percentage of total faty acid quantified (EH1) Hexane Extract of Aziza variety; (EH2) Hexane Extract of Bouffegous variety; (EH3) Hexane Extract of Assiane variety, and (EH4) Hexane Extract of Mejhoul variety.
| Compound | EHE1 A% | EHE2 A% | EHE3 A% | EHE4 A% | Reference |
|---|---|---|---|---|---|
| Caprylic acid | - | 0.61 | - | - | - |
| Thymol | 10.01 | 6.66 | 5.87 | 2.15 | STD |
| Thymol acetate | 12.93 | 19.87 | 1.18 | - | STD |
| carvacrol | 37.95 | - | - | - | STD |
| Lauric acid | 3.57 | 25.38 | 22.65 | 24.23 | [23] |
| Pentadecanoic acid | - | 0.86 | 0.92 | 1.11 | - |
| Phthalic acid.allyl ethyl ester | 7.52 | 7.77 | 8.10 | 4.37 | [1] |
| Myristic acid | 1.59 | 6.98 | - | 10.09 | [31,32] |
| Palmitic acid | 3.32 | 5.94 | 10.41 | 8.22 | [33,34] |
| Lignoceric acid | - | - | - | 7.28 | - |
| Oleic acid | 5.94 | 24.09 | 5.72 | 31.83 | [23,24] |
| Vaccenic acid | - | - | 36.25 | - | - |
| Petroselinic acid | 10.36 | - | - | - | - |
Oleic acid–rich oils are particularly valued for their high stability and health benefits. As a major unsaturated fatty acid, oleic acid plays a key role in human nutrition, known for its ability to help prevent cardiovascular diseases [29], its low saturation, its capacity to lower blood cholesterol, and its strong resistance to oxidation [33]. It is well established that oils rich in unsaturated fatty acids play a protective role against cardiovascular diseases and inflammatory conditions [34]. Additionally, numerous studies emphasize the benefits of lauric acid, including its role in reducing the risk of prostatic hyperplasia [28], Its health advantages over trans fats [35], together with its antimicrobial activity that suppresses microbial growth and toxin formation, further enhance its value [29,34].
Fig. 4, and Table 5 present the micronutrient profile of the four studied date pit varieties, focusing on essential trace elements such as iron (Fe), zinc (Zn), copper (Cu), and cobalt (Co). Among these, Zinc (Zn) was the most abundant element, with values ranging from 0.166 to 0.209 mg/100 g, where Boufegous exhibited the maximum content and Assiane the minimum. Copper (Cu) concentrations extended from 0.067 to 0.109 mg/100 g, reaching their peak in El Mejhol and their lowest in Aziza. Iron (Fe) showed a broader variation, between 0.009 and 0.173 mg/100 g, with Assiane presenting the highest amount and Aziza the lowest. Nickel (Ni) levels were relatively stable, fluctuating between 0.005 and 0.007 mg/100 g; Boufegous and El Mejhol recorded the upper values, whereas Aziza showed the lowest. Lead (Pb) contents spanned from 0.021 to 0.041 mg/100 g, with Boufegous and El Mejhol again at the higher end. Cadmium (Cd) concentrations ranged between 0.009 and 0.015 mg/100 g, being highest in Boufegous and lowest in Aziza. Lastly, cobalt (Co) varied from 0.025 to 0.030 mg/100 g, with Boufegous and Assiane presenting the greatest levels, while El Mejhol registered the smallest concentration.
Figure 4: Development of calibration curves for the spectrophotometric determination of selected heavy metals (Pb, Cu, Zn, Ni, Cd, Fe, and Co).
Table 5: Mineral contents (mg/100 g dry weight) of date stone.
| Minerals | Aziza (mg/100 g) | Bouffegous (mg/100 g) | Assiane (mg/100 g) | Mejhoul (mg/100 g) |
|---|---|---|---|---|
| Copper (Cu) | 0.067 ± 0.001a | - | - | 0.109 ± 0.067b |
| Zinc (Zn) | 0.168 ± 0.004a | 0.209 ± 0.002b | 0.166 ± 0.006a | 0.206 ± 0.017b |
| Iron (Fe) | 0.009 ± 0.000a | 0.043 ± 0.007b | 0.173 ± 0.05c | 0.147 ± 0.014c |
| Nikel (Ni) | 0.005 ± 0.000a | 0.007 ± 0.000a | 0.006 ± 0.000a | 0.007 ± 0.000a |
| Lead (Pb) | 0.021 ± 0.000a | 0.029 ± 0.003a | 0.040 ± 0.001b | 0.041 ± 0.001b |
| Cadmium (Cd) | 0.009 ± 0.001a | 0.015 ± 0.007b | 0.010 ± 0.001a | 0.013 ± 0.000a |
| Cobalt (Co) | - | 0.030 ± 0.000a | 0.030 ± 0.000a | 0.025 ± 0.001a |
Similar levels of macroelements and trace elements have been reported in date pits from various cultivars by Al-Hooti et al. (1998) [36], Al-Showiman (1990) [37], Besbes et al. (2004) [38], Devshony et al. (1992) [39], El-Shurafa et al. (1982) [40], Al-Yahya et al. (1986) [41], Rahman et al. (2007) [42], Sawaya et al. (1984) [43], Sumianah et al. (1984) and Habib et al. (2009) [24,44]. Variations in mineral content between date pit varieties may result from genetic diversity, differences in harvest periods, post-harvest processing methods, fertilizer application, irrigation water quality, soil mineral availability, and environmental influences. The outcomes of this study, alongside prior research, highlight the remarkable mineral composition of date pits. Minerals play a vital role in human health, as they are involved in the development of bones, teeth, soft tissues, hemoglobin, muscles, blood, and nerve cells. In addition, they are crucial for maintaining both physical well-being and mental health [45].
The concentrations of heavy metals detected in the date seed samples were low, with Pb ranging from 0.21 to 0.41 mg/kg and Cd from 0.09 to 0.15 mg/kg (dry weight). When compared with international regulatory limits (Commission Regulation (EC), Codex Alimentarius) [46,47], all Cd values and most Pb values fall within the acceptable ranges for plant-derived food ingredients. Although the Pb levels in Assiane and Mejhoul were slightly higher than the strictest limits for foods consumed directly, date seeds are typically used as a minor ingredient (generally <5–10 g/day), resulting in a negligible dietary exposure. Overall, these levels do not pose a safety concern for potential food or nutraceutical applications.
3.7 DPPH Radical Scavenging Assay
The antioxidant activity of the date seed extracts, evaluated using the DPPH radical scavenging assay, showed marked variations depending on both the cultivar and the extraction solvent (Table 6.). Overall, the hydro-alcoholic (EET) and aqueous (WAE) extracts exhibited lower IC50 values than the acetonic extracts (ACE), indicating stronger radical scavenging efficiency. Among the four studied cultivars, Assiane (EET: 0.10 mg/mL) and Boufegous (WAE: 0.14 mg/mL) demonstrated the highest antioxidant potential, with IC50 values comparable to that of the reference antioxidant, ascorbic acid (0.134 mg/mL). In contrast, the ethanolic and aqueous extracts of Aziza showed higher IC50 values, reflecting weaker DPPH scavenging ability.
Table 6: IC50 values (mg/mL) of DPPH radical scavenging activity of date seed extracts.
| Date Varieties | IC50 Inhibition of DPPH (mg/mL) | ||
|---|---|---|---|
| Acetone (ACE) | Ethanol (EET) | Water (WAE) | |
| Aziza | 0.12 ± 0.00 | 0.21 ± 0.03 | 0.17 ± 0.00 |
| Boufegous | 0.12 ± 0.02 | 0.18 ± 0.00 | 0.14 ± 0.00 |
| Assiane | 0.16 ± 0.01 | 0.10 ± 0.00 | 0.16 ± 0.00 |
| Mejhoul | 0.18 ± 0.0 3 | 0.12 ± 0.00 | 0.15 ± 0.01 |
| Ascorbic acid | 0.134 ± 0.00 | ||
These findings appear closely linked to the phenolic composition of the extracts. Numerous studies have reported a strong positive correlation between total polyphenol content in date seeds and their antioxidant capacity measured through DPPH scavenging [48,49,50]. Our results align with this trend: the Assiane and Boufegous cultivars previously identified as having higher levels of phenolics and flavonoids also displayed the strongest DPPH neutralizing ability.
Overall, the antioxidant profiles obtained in this study highlight the promising potential of Moroccan date seeds as natural sources of radical-scavenging molecules. In particular, the Assiane and Boufegous cultivars exhibited notable activity, with their hydro-alcoholic extracts performing comparably to standard antioxidants. These findings reinforce the value of date seeds as underutilized functional ingredients with significant bioactive properties.
3.8 Inhibitory Activities of Date Pits Extracts Against Pancreatic α-Amylase Activity and Intestinal α-Glucosidase (In Vitro)
3.8.1 In Vitro Investigation of α-Amylase Inhibitory Properties
Diabetes is a long-term metabolic disorder marked by sustained elevations in blood glucose levels, a condition known as hyperglycemia. To manage this condition, recent research has focused on identifying new inhibitors of pancreatic alpha-amylase and intestinal alpha-glucosidase, based on natural extracts [51].
The modulation of pancreatic α-amylase activity is an important therapeutic target for controlling postprandial hyperglycemia. In the present study, acetone (EAC) and ethanol (EET) extracts from four date seed varieties were assessed for their in vitro inhibitory effects on α-amylase (Fig. 5). Overall, all extracts displayed a clear dose-dependent response across the tested concentrations (0.062–1 mg/mL), although the magnitude of inhibition varied depending on the extraction solvent and the specific variety.
For the acetone extracts, inhibition increased sharply with concentration for all varieties. The Mejhoul variety exhibited the highest activity, reaching approximately 82% inhibition at 1 mg/mL, outperforming Assiane (~76%), Aziza (~72%), and Boufegous (~62%). This enhanced performance is consistent with the IC50 value obtained for EAC-Mejhoul, which is very close to that of acarbose, indicating that acetone efficiently extracts highly bioactive compounds from this variety. As expected, acarbose, used as a positive control, showed rapid and strong inhibition even at low concentrations. Nevertheless, at the highest concentration, EAC-MJ approached the inhibitory capacity of acarbose, highlighting its notable potential.
In contrast, the inhibition patterns of the ethanol extracts differed markedly. While the EET fractions also showed concentration-dependent inhibition, their maximum inhibitory percentages were generally lower than those of the acetone extracts. Interestingly, the Boufegous variety weak in the acetone fraction displayed the strongest activity among the ethanolic extracts, reaching nearly 74% inhibition at 1 mg/mL, surpassing the ethanolic extracts of Mejhoul, Assiane, and Aziza. This inversion suggests that different classes of bioactive metabolites are preferentially extracted by ethanol in Boufegous, reflecting the influence of solvent polarity on the extractable phytochemical profile.
These findings demonstrate that both the variety and the extraction solvent significantly affect the inhibitory potential of date seed extracts. In most cases, acetone extraction produced stronger α-amylase inhibition, except for Boufegous, which was more responsive to ethanol. Such variability underscores the chemical complexity of date seeds and the possibility that distinct phytochemical families contribute to enzyme inhibition in each variety.
Overall, the results highlight the promising potential of date seeds as natural inhibitors of α-amylase. In particular, the acetone extract of Mejhoul stands out, displaying an inhibitory capacity comparable to that of acarbose at higher concentrations. These observations support the valorization of date seed by-products as bioactive agents with potential relevance in the management of type 2 diabetes.
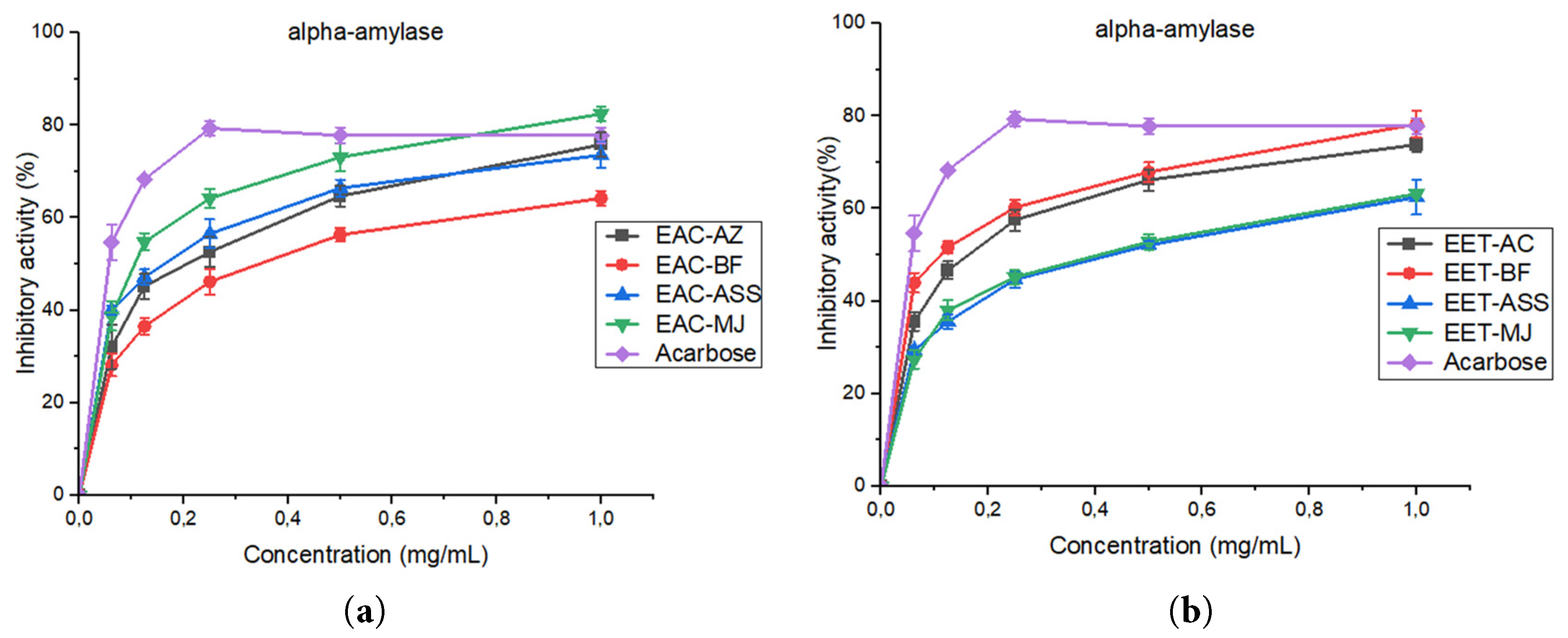
Figure 5: Comparative in vitro inhibition of pancreatic α-amylase by (a) acetone and (b) ethanol extracts of four date seed varieties in comparison with acarbose. Values represent mean ± standard deviation (SD) of three independent measurements (n = 3). Error bars indicate standard deviation.
3.8.2 Assessment of In Vitro α-Glucosidase Inhibitory Activity
In this in vitro study, the inhibitory effects of different concentrations (0.062, 0.125, 0.25, 0.5, and 1 mg/mL) of acetone and ethanol extracts from four date seed varieties were evaluated on intestinal alpha-glucosidase activity. As shown in Fig. 6, the acetone extract of the Boufegous variety exhibited the strongest in vitro inhibition of intestinal α-glucosidase, reaching nearly 80% at 1 mg/mL (IC50 = 0.45 ± 0.0062 mg/mL). This activity is comparable to that of acarbose, which inhibited approximately 95% at the same concentration (IC50 = 0.40 ± 0.0068 mg/mL). Notable inhibitory effects were also observed for Assiane and Mejhoul, while Aziza showed the lowest activity. The inhibitory effects of ethanolic extracts of date pits on intestinal α-glucosidase showed that, among the extracts tested, the ethanolic extract of the Aziza variety (EET-AZ) had the highest inhibitory potential, reaching approximately 75% at 1 mg/mL with an IC50 value of 0.50 ± 0.0081 mg/mL, close to that of acarbose, used as a positive control, which reached approximately 95%. mL with an IC50 value of 0.38 ± 0.0081 mg/mL, close to that of acarbose, used as a positive control, which reached approximately 95% at 1 μg/mL with an IC50 value of IC50 = 0.40 ± 0.0068 mg/mL. In contrast, the ethanolic extract of the Boufegous variety (EET-BF) showed the lowest inhibitory activity.
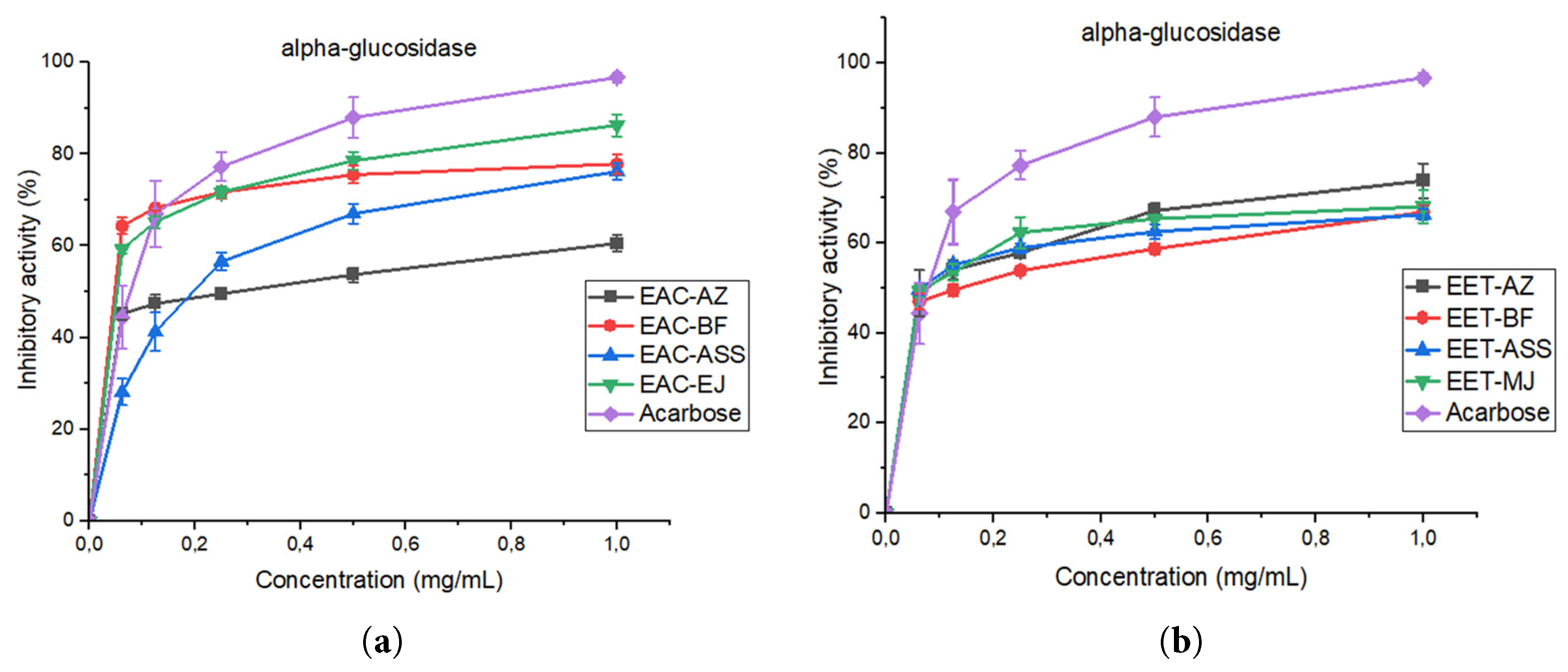
Figure 6: Comparative in vitro inhibition of intestinal α-glucosidase by (a) acetone and (b) ethanol extracts of four date seed varieties versus acarbose. Values represent mean ± standard deviation (SD) of three independent measurements (n = 3). Error bars indicate standard deviation.
This study revealed that date seeds possess a strong capacity to reduce the activity of carbohydrate-digesting enzymes, specifically α-amylase and α-glucosidase. Such enzymatic inhibition suggests that date seeds may serve as a valuable natural source of compounds with antidiabetic potential. The contribution of phenolic acids and flavonoids is particularly important, as these phytochemicals are likely to play a central role in suppressing the function of these enzymes. To confirm these observations, more studies that are detailed are necessary to isolate the responsible molecules and to elucidate their mechanisms of action.
This study demonstrates that Moroccan date seeds are a valuable, yet underexploited, natural resource with significant nutritional, phytochemical, and functional potential. Across the four cultivars examined, date seeds were shown to contain essential fatty acids, minerals, and a rich diversity of bioactive compounds such as phenolics and flavonoids. These components contribute to strong antioxidant and enzyme-inhibitory activities, confirming the functional relevance of date seeds.
Overall, the findings support the use of date seed–derived ingredients in food, nutraceutical, and health-oriented applications. Given their abundance as agro-industrial by-products, date seeds represent a sustainable and cost-effective source of health-promoting compounds. Future studies should focus on identifying the specific active molecules involved, assessing their in vivo bioavailability, and exploring their incorporation into innovative food formulations or therapeutic strategies.
Acknowledgement:
Funding Statement: The authors received no specific funding for this study.
Author Contributions: The authors confirm contribution to the paper as follows: Conceptualization, Manar Bouhchich and Mostafa Mimouni; Formal analysis, Amine Elbouzidi and Mohamed Addi; Investigation, Manar Bouhchich, Abdessadek Rahimi and Rhizlan Abdnim; Methodology, Manar Bouhchich, Abdessadek Rahimi and Rhizlan Abdnim; Project administration, Mostafa Mimouni; Software, Rhizlan Abdnim; Supervision, Mostafa Mimouni; Validation, Amine Elbouzidi and Mohamed Addi; Visualization, Amine Elbouzidi; Writing—original draft, Manar Bouhchich, Abdessadek Rahimi and Rhizlan Abdnim; Writing—review & editing, Amine Elbouzidi, Mohamed Addi and Mostafa Mimouni. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: The data that support the findings of this study are available from the Corresponding Author, M.B., upon reasonable request.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
Abbreviations
| P. dactylifera L. | Phoenix dactylifera Linnaeus |
| HE | Hexane Extract |
| HEE | Hexane Extract (oil, in GC–MS) |
| CHE | Chloroform Extract |
| CFE | Chloroform Extract |
| ACE | Acetone Extract |
| EAC | Acetone Extract (enzyme assays) |
| ETE | Ethanol Extract |
| EET | Ethanol Extract (enzyme assays) |
| WAE | Water Extract |
| EE | Water Extract |
| DNSA | 3,5-Dinitrosalicylic Acid |
| GAE | Gallic Acid Equivalents |
| QE (or EQ) | Quercetin Equivalents |
| IU | International Unit |
| AOAC | Association of Official Analytical Chemists |
| UV–Vis | Ultraviolet–Visible Spectrophotometer |
| GC–MS (or CG–MS) | Gas Chromatography–Mass Spectrometry |
| AAS | Atomic Absorption Spectrophotometry |
| IC50 | Half-Maximal Inhibitory Concentration |
| GOD/POD | Glucose Oxidase/Peroxidase (kit) |
| DPPH | 2,2-Diphenyl-1-picrylhydrazyl |
| AlCl3·6H2O | Aluminum Chloride Hexahydrate |
| HNO3 | Nitric Acid |
References
1. Metoui M , Essid A , Bouzoumita A , Ferchichi A . Chemical composition, antioxidantand antibacterial activity of TunisianDate palm seed. Pol J Environ Stud. 2019; 28( 1): 267– 74. doi:10.15244/pjoes/84918. [Google Scholar] [CrossRef]
2. Adeosun AM , Oni SO , Ighodaro OM , Durosinlorun OH , Oyedele OM . Phytochemical, minerals and free radical scavenging profiles of Phoenix dactilyfera L. seed extract. J Taibah Univ Med Sci. 2016; 11( 1): 1– 6. doi:10.1016/j.jtumed.2015.11.006. [Google Scholar] [CrossRef]
3. Nweze Nwogu C , Nwaiwu U , Uchechukwu Udo V , James Nwosu O , Ezenwa Hart C . Effect of date seed granules on the mechanical properties of Glass fibre reinforced epoxy composite. Clean Mater. 2022; 6: 100160. doi:10.1016/j.clema.2022.100160. [Google Scholar] [CrossRef]
4. Murthy HN , Bapat VA . Bioactive compounds in underutilized fruits and nuts. Cham, Switzerland: Springer International Publishing; 2020. doi:10.1007/978-3-030-06120-3. [Google Scholar] [CrossRef]
5. Alsulaili AD , Refaie AA , Garcia HA . Adsorption capacity of activated carbon derived from date seeds: characterization, optimization, kinetic and equilibrium studies. Chemosphere. 2023; 313: 137554. doi:10.1016/j.chemosphere.2022.137554. [Google Scholar] [CrossRef]
6. Ali Al-Farsi M , Lee CY . Optimization of phenolics and dietary fibre extraction from date seeds. Food Chem. 2008; 108( 3): 977– 85. doi:10.1016/j.foodchem.2007.12.009. [Google Scholar] [CrossRef]
7. Laaraj N , Bouhrim M , Kharchoufa L , Tiji S , Bendaha H , Addi M , et al. Phytochemical analysis, α-glucosidase and α-amylase inhibitory activities and acute toxicity studies of extracts from pomegranate (Punica granatum) bark, a valuable agro-industrial by-product. Foods. 2022; 11( 9): 1353. doi:10.3390/foods11091353. [Google Scholar] [CrossRef]
8. Kalauni SK , Karki J , Sharma M , Khanal LN . Phytochemical screening, evaluation of antioxidant and antidiabetic activities of green tea available in Nepal. Prithvi Acad J. 2024; 7: 9– 19. doi:10.3126/paj.v7i1.65744. [Google Scholar] [CrossRef]
9. Harborne JB . Phytochemical methods: a guide to modern techniques of plant analysis. 2nd ed. Dordrecht, The Netherlands: Springer; 1984. 302 p. [Google Scholar]
10. Elbouzidi A , Taibi M , Baraich A , Haddou M , Loukili EH , Asehraou A , et al. Enhancing secondary metabolite production in Pelargonium graveolens hort. cell cultures: eliciting effects of chitosan and jasmonic acid on bioactive compound production. Horticulturae. 2024; 10( 5): 521. doi:10.3390/horticulturae10050521. [Google Scholar] [CrossRef]
11. Afqir H , Belmalha S , Farihi A , Elbouzidi A , Bouhrim M , Elrherabi A , et al. Comparative analysis of phenolic and flavonoid content, antioxidant, antibacterial activities, and functional groups of chemicals from Hypericum perforatum L., and Papaver rhoeas L. flower extracts. Ecol Eng Environ Technol. 2024; 25( 2): 88– 101. doi:10.12912/27197050/175801. [Google Scholar] [CrossRef]
12. ISO 5509:1978. Oils-preparation of methyl esters of fatty acids. Geneva, Switzerland: International Organization for Standardization; 1978. [Google Scholar]
13. Benayad O , Bouhrim M , Tiji S , Kharchoufa L , Addi M , Drouet S , et al. Phytochemical profile, α-glucosidase, and α-amylase inhibition potential and toxicity evaluation of extracts from Citrus aurantium (L.) peel, a valuable by-product from northeastern Morocco. Biomolecules. 2021; 11( 11): 1555. doi:10.3390/biom11111555. [Google Scholar] [CrossRef]
14. Smith DL , Schrenk WG . Application of atomic absorption spectroscopy to plant analysis. I. comparison of zinc and manganese analysis with official AOAC colorimetric methods. J Assoc Off Anal Chem. 1972; 55( 4): 669– 75. doi:10.1093/jaoac/55.4.669. [Google Scholar] [CrossRef]
15. Brand-Williams W , Cuvelier ME , Berset C . Use of a free radical method to evaluate antioxidant activity. LWT Food Sci Technol. 1995; 28( 1): 25– 30. doi:10.1016/S0023-6438(95)80008-5. [Google Scholar] [CrossRef]
16. Thalapaneni NR , Chidambaram KA , Ellappan T , Sabapathi ML , Mandal SC . Inhibition of carbohydrate digestive enzymes by Talinum portulacifolium (forssk) leaf extract. J Complement Integr Med. 2008; 5( 1): 1120. doi:10.2202/1553-3840.1120. [Google Scholar] [CrossRef]
17. Elrherabi A , Bouhrim M , Abdnim R , Berraaouan A , Ziyyat A , Mekhfi H , et al. Antihyperglycemic potential of the Lavandula stoechas aqueous extract via inhibition of digestive enzymes and reduction of intestinal glucose absorption. J Ayurveda Integr Med. 2023; 14( 5): 100795. doi:10.1016/j.jaim.2023.100795. [Google Scholar] [CrossRef]
18. El Hassania L , Mounime K , Elbouzidi A , Taibi M , Mohamed C , Abdelkhaleq L , et al. Analyzing the bioactive properties and volatile profiles characteristics of Opuntia dillenii (Ker gawl.) haw: exploring its potential for pharmacological applications. Chem Biodivers. 2024; 21( 3): e202301890. doi:10.1002/cbdv.202301890. [Google Scholar] [CrossRef]
19. Bassi D , Cirilli M , Rossini L . Most important fruit crops in Mediterranean Basin (Mb). Milano, Italy: Milano University Press; 2024. 152 p. doi:10.54103/milanoup.155. [Google Scholar] [CrossRef]
20. Noutfia Y , Ropelewska E , Szwejda-Grzybowska J , Mieszczakowska-Frąc M , Siarkowski S , Rutkowski KP , et al. Effects of mild infrared and convective drying on physicochemical properties, polyphenol compounds, and image features of two date palm cultivars: ‘Mejhoul’ and ‘Boufeggous’. LWT. 2025; 218: 117502. doi:10.1016/j.lwt.2025.117502. [Google Scholar] [CrossRef]
21. Ravi L . Bioactivity of Phoenix dactylifera seed and its phytochemical analysis. Int J Green Pharm. 2017; 11( 2): 1– 6. [Google Scholar]
22. Hasan ZYM , Aljeboury GH , Obaid WA , Khalaf RA . Phytochemical and antimicrobial study for Phoenix dactylifera L. (ajwa date) seeds extract: ajwa seeds extract biological activity. J Biotechnol Res Cent. 2024; 18( 2): 119– 33. doi:10.24126/jobrc.2024.18.2.880. [Google Scholar] [CrossRef]
23. Ali Al-Farsi M , Lee CY . Nutritional and functional properties of dates: a review. Crit Rev Food Sci Nutr. 2008; 48( 10): 877– 87. doi:10.1080/10408390701724264. [Google Scholar] [CrossRef]
24. Habib HM , Ibrahim WH . Nutritional quality evaluation of eighteen date pit varieties. Int J Food Sci Nutr. 2009; 60( Suppl 1): 99– 111. doi:10.1080/09637480802314639. [Google Scholar] [CrossRef]
25. Baliga MS , Baliga BRV , Kandathil SM , Bhat HP , Vayalil PK . A review of the chemistry and pharmacology of the date fruits (Phoenix dactylifera L.). Food Res Int. 2011; 44( 7): 1812– 22. doi:10.1016/j.foodres.2010.07.004. [Google Scholar] [CrossRef]
26. Selim S , Abdel-Mawgoud M , Al-Sharary T , Almuhayawi MS , Alruhaili MH , Al Jaouni SK , et al. Pits of date palm: bioactive composition, antibacterial activity and antimutagenicity potentials. Agronomy. 2022; 12( 1): 54. doi:10.3390/agronomy12010054. [Google Scholar] [CrossRef]
27. Halabi Y , Nasri C , El Guezzane C , Harhar H , Chahboun N , Warad I , et al. Qualitative and quantitative analysis of Phoenix dactilifera L. seeds in Morocco with antioxidant activities using chemometrics. Ecol Eng Environ Technol. 2024; 25( 4): 81– 91. doi:10.12912/27197050/183179. [Google Scholar] [CrossRef]
28. Djaoudene O , López V , Cásedas G , Les F , Schisano C , Bachir Bey M , et al. Phoenix dactylifera L. seeds: a by-product as a source of bioactive compounds with antioxidant and enzyme inhibitory properties. Food Funct. 2019; 10( 8): 4953– 65. doi:10.1039/C9FO01125K. [Google Scholar] [CrossRef]
29. Mrabet A , Jiménez-Araujo A , Guillén-Bejarano R , Rodríguez-Arcos R , Sindic M . Date seeds: a promising source of oil with functional properties. Foods. 2020; 9( 6): 787. doi:10.3390/foods9060787. [Google Scholar] [CrossRef]
30. Raza MQ , Arshad MU , Arshad MS , Anjum FM . Characterization of compositional and functional characteristics of date seeds and oil (Phoenix dactylifera L.) from three varieties. Int J Biosci. 2019; 15: 1– 4. doi:10.12692/ijb/15.3.1-14. [Google Scholar] [CrossRef]
31. Akbari M . Oil characteristics and fatty acid profile of seeds from three varieties of date palm (Phoenix dactylifera) cultivars in Bushehr-Iran. Afr J Biotechnol. 2012; 11( 57): 1084. doi:10.5897/AJB12.1084 [Google Scholar] [CrossRef]
32. Afiq MJA , Rahman RA , Man YBC , Al-Kahtani HA , Mansor TST . Date seed and date seed oil. Int Food Res J. 2013; 20: 2035. [Google Scholar]
33. Kirthy Reddy M , Durga Rani H , Naga Deepika C , Samrawat S , Akshara V , Rajesh K . Study on physico-chemical properties of oil and powder of date palm seeds (Phoenix dactylifera). Int J Curr Microbiol App Sci. 2017; 6( 12): 486– 92. doi:10.20546/ijcmas.2017.612.059. [Google Scholar] [CrossRef]
34. Ramadan MF , Sharanabasappa G , Parmjyothi S , Seshagiri M , Moersel JT . Profile and levels of fatty acids and bioactive constituents in Mahua butter from fruit-seeds of buttercup tree [Madhuca longifolia (Koenig). Eur Food Res Technol. 2006; 222( 5): 710– 8. doi:10.1007/s00217-005-0155-2. [Google Scholar] [CrossRef]
35. de Roos N , Schouten E , Katan M . Consumption of a solid fat rich in lauric acid results in a more favorable serum lipid profile in healthy men and women than consumption of a solid fat rich in trans-fatty acids. J Nutr. 2001; 131( 2): 242– 5. doi:10.1093/jn/131.2.242. [Google Scholar] [CrossRef]
36. Al-Hooti S , Sidhu JS , Qabazard H . Chemical composition of seeds of date fruit cultivars of United Arab Emirates. Food Nutr Sci. 1998; 35( 1): 44– 6. [Google Scholar]
37. Fayadh JM , Al-Showiman SS . Chemical composition of date palm (Phoenix dactylifera L.). J Chem Soc Pak. 1990; 12: 84– 103. [Google Scholar]
38. Besbes S , Blecker C , Deroanne C , Drira NE , Attia H . Date seeds: chemical composition and characteristic profiles of the lipid fraction. Food Chem. 2004; 84( 4): 577– 84. doi:10.1016/S0308-8146(03)00281-4. [Google Scholar] [CrossRef]
39. Devshony S , Eteshola E , Shani A . Characteristics and some potential applications of date palm (Phoenix dactylifera L.) seeds and seed oil. J Am Oil Chem Soc. 1992; 69( 6): 595– 7. doi:10.1007/BF02636115. [Google Scholar] [CrossRef]
40. El-Shurafa MY , Ahmed HS , Abou-Naji SE . Organic and inorganic constituents of date palm pit (seed). Date Palm J. 1982; 1( 2): 275– 84. [Google Scholar]
41. Al-Yahya MA . Phytochemical and biological studies on Saudi medicinal plants. Part II. The major component of the spathe oil of Phoenix dactylifera. Fitoterapia. 1986; 57( 4): 284– 7. [Google Scholar]
42. Rahman MS , Kasapis S , Al-Kharusi NSZ , Al-Marhubi IM , Khan AJ . Composition characterisation and thermal transition of date pits powders. J Food Eng. 2007; 80( 1): 1– 10. doi:10.1016/j.jfoodeng.2006.04.030. [Google Scholar] [CrossRef]
43. Sawaya WN , Khalil JK , Safi WJ . Chemical composition and nutritional quality of date seeds. J Food Sci. 1984; 49( 2): 617– 9. doi:10.1111/j.1365-2621.1984.tb12482.x. [Google Scholar] [CrossRef]
44. Sumianah G , Makki YM , Rumney TG . Changes in the chemical composition of three cultivars of date palm seed during germination. Date Palm. 1984; 3( 2): 395– 407. [Google Scholar]
45. O’Dell BL , Sunde RA . Handbook of nutritionally essential mineral elements. New York, NY, USA: Marcel Dekker; 1997. doi:10.1201/9781482273106. [Google Scholar] [CrossRef]
46. Commission of the European Communities. Commission Regulation (EC) No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Brussels, Belgium: Commission of the European Communities; 2006. [Google Scholar]
47. CXS 193-1995. General standard for contaminants and toxins in food and feed. Rome, Italy: Codex Alimentarius Commission; 2016. doi:10.1093/law:epil/9780199231690/e1857. [Google Scholar] [CrossRef]
48. Habib HM , Platat C , Meudec E , Cheynier V , Ibrahim WH . Polyphenolic compounds in date fruit seed (Phoenix dactylifera): characterisation and quantification by using UPLC-DAD-ESI-MS. J Sci Food Agric. 2014; 94( 6): 1084– 9. doi:10.1002/jsfa.6387. [Google Scholar] [CrossRef]
49. Bouhlali EDT , Alem C , Ennassir J , Benlyas M , Mbark AN , Zegzouti YF . Phytochemical compositions and antioxidant capacity of three date (Phoenix dactylifera L.) seeds varieties grown in the South East Morocco. J Saudi Soc Agric Sci. 2017; 16( 4): 350– 7. doi:10.1016/j.jssas.2015.11.002. [Google Scholar] [CrossRef]
50. Haddou S , Elrherabi A , Loukili EH , Abdnim R , Hbika A , Bouhrim M , et al. Chemical analysis of the antihyperglycemic, and pancreatic α-amylase, lipase, and intestinal α-glucosidase inhibitory activities of Cannabis sativa L. seed extracts. Molecules. 2023; 29( 1): 93. doi:10.3390/molecules29010093. [Google Scholar] [CrossRef]
51. Abdalla RSM , Albasheer AA , El-Hussein ARM , Gadkariem EA . Physico-chemical characteristics of date seed oil grown in Sudan. Am J Appl Sci. 2012; 9( 7): 993– 99. doi:10.3844/ajassp.2012.993.999. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools