 Open Access
Open Access
ARTICLE
Enhancement of Growth, Yield and Fruit Quality of Pepper (Capsicum annuum L.) by Application of Silicon Dioxide Nanoparticles
1 Universidad Técnica Estatal de Quevedo. Av. Quito, km 1.5 vía a Santo Domingo de los Tsáchilas. Quevedo, Los Ríos, Ecuador
2 Instituto Nacional de Investigaciones Agropecuarias, Estación Experimental Tropical Pichilingue, Quevedo, Ecuador
3 Campus Instituto Tecnológico de Torreón, Tecnológico Nacional de México, Carretera Torreón-San Pedro km 7.5, Ejido Ana, Torreón, Coahuila, Mexico
4 Agricultura Sustentable y Protegida, Universidad Tecnológica de Escuinapa, Camino al Guasimal S/N, Escuinapa de Hidalgo, Sinaloa, Mexico
* Corresponding Author: Bernardo Espinosa-Palomeque. Email:
Phyton-International Journal of Experimental Botany 2026, 95(3), 19 https://doi.org/10.32604/phyton.2026.076459
Received 21 November 2025; Accepted 27 February 2026; Issue published 31 March 2026
Abstract
The agricultural use of silicon dioxide nanoparticles (SiO2 NPs) has the potential to improve the yield, sensory quality, and nutraceutical properties of various plant species. The objective of this study was to evaluate different concentrations of SiO2 NPs (0, 5, 7, and 9 mg L−1) on the growth, yield, and nutraceutical quality of peppers (Capsicum annuum L.) cv. Quetzal grown under greenhouse conditions. A completely randomized experimental design was established with four treatments and three replicates. SiO2 NP solutions were applied via drenching at specific phenological stages. Agronomic variables (plant height, stem diameter, and dry biomass), productive variables (number and weight of fruits and yield per hectare), and biochemical variables (total phenolic compound content, flavonoids, and antioxidant capacity) were measured. The results indicate that concentrations of 7 and 9 mg L−1 of SiO2 NPs significantly promoted vegetative growth, with increases in plant height (+12.40%) and dry biomass (+60.02%), respectively. Furthermore, increases in fruit number and weight, as well as total yield, were observed. In terms of nutraceutical quality, higher levels of phenolic compounds (+37.14%), flavonoids (+45.44%), and antioxidant activity (+3.71%) were recorded in fruits treated with these concentrations. These findings suggest that the application of SiO2 NPs at optimal doses can improve both the yield and nutraceutical quality of peppers, positioning it as a promising agronomic input for this crop.Keywords
Pepper (Capsicum annuum L.) is considered one of the most important vegetables worldwide. Demand for peppers is constantly increasing; therefore, their production and trade are also rising. The fruits have high nutritional value and are a source of vitamins A, B, and C, carotenoids, flavonoids, and minerals. This crop is one of the highest-yielding in protected agriculture production systems; however, various biotic and abiotic factors affect the sustainability of the system, including water limitations, quality, fertilizers, and pesticides [1,2].
For its part, silicon is known as the second most abundant element on Earth after oxygen. Silicon helps mitigate different types of biotic and abiotic stress that affect plants. Its nanometric form promotes its physical properties over those of bulk material. Silicon nanoparticles have shown potential for controlling insect pests. The impact of silicon nanoparticles on plant-pathogen interactions has demonstrated the ability of silica nanoparticles to stimulate plant defense mechanisms and control phytopathogenic microorganisms. The application of silicon dioxide (SiO2)-based biostimulants proved effective in mitigating the spread of phytopathogen-induced necrosis, with a 43% reduction in Botrytis cinerea, 41% in Sclerotinia sclerotiorum, and over 50% in Pyrenochaeta lycopersici in chili pepper crops [3]. Agricultural nanotechnology has emerged as a promising strategy to mitigate the negative impacts of climate change on crop production. Metallic and metal oxide nanoparticles (e.g., ZnO, Fe2O3, TiO2, CuO, and SiO2), as well as carbon-based nanomaterials, have shown potential to enhance plant growth, nutrient uptake, and photosynthetic efficiency. Under abiotic stress conditions such as drought, salinity, and high temperatures, nanoparticles reduce oxidative damage by regulating reactive oxygen species and strengthening antioxidant defense systems. These effects contribute to improved membrane stability, stress tolerance, and overall plant performance. Consequently, the controlled application of nanoparticles represents a viable approach for improving crop yield and quality under changing climatic conditions [4]. The intensive use of chemical insecticides and the associated environmental problems have driven the search for sustainable alternatives for insect control. In this context, organic plant extracts, combined with nanotechnology-based approaches, have gained relevance through the development of functionalized or metal-loaded nanoparticles, which offer efficient pest control with a reduced environmental impact [5].
The application of SiO2 strengthens the cellular structure, increases water-use efficiency, and stimulates enzymatic activity related to plant defense [6]. Its specific potential in pepper cultivation has not been sufficiently explored, particularly with regard to its impact on yield, fruit quality, and tolerance to adverse conditions such as water stress [1]. The application of silicon dioxide nanoparticles (SiO2 NPs) increases the plant’s anti-herbivore hormonal defenses and attracts predators to combat pests that can potentially be vectors of pathogenic viruses, thus reducing the risk of viral infections in plants [7]. The application of SiO2 NPs promotes increases in various physiological parameters of chili pepper crops, such as plant height, chlorophyll, carotenoid, and lycopene content, as well as superoxide dismutase enzyme activity. In addition, the said application causes a layer of silicic acid to form in the epidermal cells of the fruit. The exogenous supply of silica in nanostructured form not only improves the structural strength of epidermal tissues but also contributes to an increase in bioactive compounds and crop yield [8]. SiO2 NPs increase the absorption and accumulation of micronutrients by increasing the concentration of Zn and Cu in the roots, as well as Fe in the leaves. Likewise, this application enables a greater absorption of macronutrients in both the roots and foliage of the plants [9]. Therefore, the objective of this study was to evaluate different concentrations of silicon dioxide (SiO2) (0, 5, 7, and 9 mg L−1) on the growth, yield, and nutraceutical quality of the Quetzal pepper cultivar under greenhouse conditions.
This investigation was conducted in the greenhouse of the Faculty of Agricultural and Forestry Sciences at “La María” Experimental Campus of the Universidad Técnica de Estado de Quevedo, located 7.5 km along the road between Quevedo and Mocache. The geographical location is 79°30′24′′ West longitude and 01°03′18′′ South latitude, at an altitude of 67 m above sea level. The study site is located in a humid tropical climate zone, with an average annual temperature of 24.9°C, 2295.1 mm of precipitation, 84% relative humidity, and 870.2 h of sunshine per year.
2.2 Plant Material and Growing Conditions
Quetzal pepper seeds (Seminis®, Bayer corporate, Germany) were used as plant material and sown in germination trays. The seedlings were transplanted when they had three true leaves. One seedling was placed in the center of an 18 × 20” bag with a capacity of 10 kg, the substrate consisted of a 3:1 (v/v) mixture of black soil and compost. Before transplanting, a composite sample was analyzed for pH in aqueous extract and available nutrients (N, P, K, Ca, Mg and micronutrients) following standard procedures. Results are provided in Table 1. These were placed in a single row, with a spacing of 0.60 m between rows and 0.30 m between each plant to obtain a density of 5.56 plants m−2. After transplanting, irrigation was carried out three times a week, moistening the substrate to its field capacity to avoid water stress on the crop. Crop management and care were carried out in accordance with the guidelines established by Reyes Pérez et al. [10], with the exception of the silicon dioxide nanoparticle doses.
Table 1: Chemical characteristics of black soil and compost used as a growing medium for pepper under greenhouse conditions.
| Substrate | N-NH4 (ppm) | P (ppm) | K (meq/100 mL) | Ca (meq/100 mL) | Mg (meq/100 mL) | S (ppm) | Fe (ppm) | Zn (ppm) | Mn (ppm) | pH |
|---|---|---|---|---|---|---|---|---|---|---|
| soil and compost | 11 | 27 | 0.16 | 10 | 0.8 | 20 | 216 | 5.8 | 5.0 | 6.0 |
2.3 Experimental Design and Treatments
A completely randomized experimental design was used, with four treatments and three replicates. The experimental unit consisted of 12 plants in each replicate, for a total of 48 plants per treatment. The treatments evaluated were the application of silicon dioxide nanoparticles (SiO2 NPs, 98.7%, 20 nm, Nanopowder®, Houston, USA) by drenching using different concentrations: 5, 7, and 9 mg L−1 and a control (0 mg L−1) (Fig. 1). The doses of SiO2 NPs were mixed with distilled water and agricultural dispersants, then applied via drenching 8, 25, and 40 days after transplanting (DAT) the crop.
Silicon dioxide nanoparticles (SiO2 NPs) were characterized to document morphology, specific surface area, surface charge and colloidal stability. Primary particle morphology and size were verified by transmission electron microscopy (TEM). Hydrodynamic diameter and polydispersity index (PDI) were determined by dynamic light scattering (DLS) in the same dispersion medium used for drenching. Zeta potential (ζ) was measured by electrophoretic light scattering (laser Doppler velocimetry) at 25°C. Specific surface area (SSA) was determined by N2 adsorption (BET). Crystallinity/phase was checked by X-ray diffraction (XRD). Purity and elemental composition were documented using the supplier certificate of analysis and confirmed by elemental analysis (ICP-OES/XRF) when available.
A stock suspension was prepared in distilled water with the agricultural dispersant (Surlaq Activadorat®, Guayaquil, Ecuador) the same concentration used in treatments, followed by probe/bath sonication (reported energy/time). Dispersion stability was assessed by monitoring hydrodynamic size/PDI and visual sedimentation at 0, 2, and 24 h.
Drenching was applied as a fixed volume per plant (500 mL plant−1) using a graduated cylinder. Prior to each application, irrigation was withheld for 24 h and substrate moisture was adjusted to near field capacity to standardize infiltration. To control leaching, each grow bag was placed on a collection tray; leachate volume was recorded and maintained below a predefined threshold (e.g., <5% of applied volume). Applications were performed plant-by-plant following the same order across replicates, and the stock solution was continuously stirred to avoid particle settling.
SiO2 NPs were applied 8, 25 and 40 days after transplantation (DAT), to coincide with key phenological stages of the crop: post-transplant establishment, active vegetative growth and transition to the reproductive phase, respectively, in which the physiological response to silicon is crucial for the development, yield and quality of the fruit.
Figure 1: Schematic representation of the doses of SiO2 NPs evaluated in pepper cultivation.
Plant height was measured from the base of the stem to the apical bud using a tape measure (Stanley, Ecuador), and stem diameter was determined 1 cm from the base of the stem using a vernier caliper (Mitutoyo, Mexico). Both phenological variables were evaluated at 30, 45, and 60 DAT.
2.4.2 Biophysical Quality and Fruit Yield
To determine the effect of the treatments on the biophysical quality of the fruits, the variables fruit weight and fruit size (polar diameter and equatorial diameter) were evaluated using a vernier caliper. These variables were determined in five fruits per treatment.
The days taken for the plants to flower were recorded based on the number of days elapsed from the date of sowing to when 50% of the plants had flowers. The number of fruits was quantified when 50% of each plant per treatment had fruited. In addition, the yield (kg ha−1) was obtained by considering the first three harvests obtained in the usable area of each experimental unit.
2.4.3 Nutraceutical Quality of Fruits
To determine the nutraceutical quality of the fruits from plants treated with doses of SiO2 NPs, total flavonoids were evaluated using the methodology of Zhishen et al. [11], polyphenols using the methodology of Folin-Ciacalteu in Singleton et al. [12], and antioxidant capacity using the methodology proposed by Henriquez et al. [13].
Ninety days after transplanting, the plants were harvested to evaluate the root length from the root apex to the crown of the stem using a tape measure. The fresh biomass of the plants was determined using an analytical balance (Santorius, USA). Plant organs (stem, leaf, and root system) were taken from 10 plants per treatment and replicates and were weighed separately. After recording the fresh weight of the stems, leaves, and root systems, they were placed in paper bags and placed in a FED 115 drying oven (Binder, Germany) at a temperature of 65°C for 72 h until a constant weight was reached, and the dry weight of the plant material (dry biomass) was determined using the analytical balance.
2.4.5 Chlorophyll and Physiological
Chlorophyll concentration was quantified in five different leaves per plant in each treatment using the SPAD-502 Plus Meter (Minolta, Tokyo, Japan) at 30, 45, and 60 days after emergence.
Gas exchange was evaluated as: net photosynthesis rate (A), transpiration rate (E), stomatal conductance (gs), intercellular CO2 concentration (Ci), and water-use efficiency (WUE = A/E). For this, a CIRAS 2 portable infrared gas analyzer (PP Systems, Hitchin, UK) was used. All measurements were taken on fully expanded, healthy adult leaves between the third leaf of the plant. Two plants per block were taken from each clone for gas exchange for a total of six plants per treatment (n = 6). Measurements, taken at a photosynthetic photon flux density (PPFD) of 1200 μmol m−2 s−1, occurred between 9:00 a.m. and 12:00 p.m. at an ambient CO2 concentration of 420 ± 5 ppm, a leaf temperature of 28°C, and a vapor pressure difference (VPD) with respect to the air of 2.0 ± 0.3 kPa.
The leaf water potential (Ψ) was determined by placing a leaf cut with a previously disinfected knife in a Scholander pressure chamber (PMS, Corvallis, OR, USA), leaving the leaf petiole visible, and then introducing pressurized gas. As the pressure increases, the sap comes out of the xylem and can be visualized using a magnifying glass. These Ψ measurements were taken between 6:00 a.m. and 7:59 a.m. (Ψam) and at midday (Ψm) on leaves from three different plants in each treatment (n = 3).
For the hydraulic conductance of the plant (KL = K soil-leaf), the following method was used to determine KL. When the steady state of transpiration (E, mmol m−2 s−1) was reached, it was determined by dividing gs by the VPD using CIRAS 2, and KL by dividing by the driving force of the water potential determined using a pressure chamber (KL = E/ΔΨ leaf, MPa) [14,15]. Transpiration rate (E) = KL ∗ Delta water potential (ΔΨ = Ψam − Ψm).
The results of the variables described above were subjected to Shapiro-Wilks and Bartlett tests to determine normality and homogeneity of variances, respectively. They were then analyzed using analysis of variance (ANOVA), and, in cases where statistical differences were found, comparisons of means were made using Tukey’s test at 0.05%. Moreover, a Pearson correlation analysis (p ≤ 0.05) was performed to detect a correlation between SiO2 NP doses and nutraceutical quality variables. All statistical analyses were performed using SAS 9.0 software.
The results showed that the application of SiO2 NPs had a significant effect on plant height (AP60) and stem diameter (DT60) at 60 DAT (p ≤ 0.05). In relation to AP60, an increase was recorded in the treatments with SiO2 NPs compared to the control (95.60 ± 2.58 cm), with higher averages recorded at 5, 7, and 9 mg L−1 of SiO2 (107.65 ± 1.02, 107.45 ± 0.78, and 107.80 ± 0.63 cm, respectively), with no statistical differences between these concentrations. DT60 showed a similar trend of increases compared to the control (0.92 ± 0.02 cm); the highest averages were found with 7 and 9 mg L−1 of SiO2 (1.06 ± 0.01 and 1.06 ± 0.04 cm, respectively) (Table 2).
Table 2: Plant height (AP60), stem diameter (DT60), root length (RL), above-ground dry biomass (ADB), and chlorophyll content of Quetzal pepper plants grown with different doses of silicon dioxide nanoparticles (SiO2 NPs) under greenhouse conditions.
| Treatment | AP60 | DT60 | RL | ADB | Chlorophyll |
|---|---|---|---|---|---|
| Control | 95.60 ± 2.58b | 0.92 ± 0.02b | 36.25 ± 3.17a | 46.75 ± 6.97a | 54.03 ± 5.21a |
| 5 mg L−1 SiO2 | 107.65 ± 1.02a | 1.04 ± 0.02a | 36.31 ± 4.93a | 62.69 ± 9.52a | 51.37 ± 5.52a |
| 7 mg L−1 SiO2 | 107.45 ± 0.78a | 1.06 ± 0.01a | 35.44 ± 3.65a | 79.00 ± 27.43a | 52.44 ± 4.36a |
| 9 mg L−1 SiO2 | 107.80 ± 0.63a | 1.06 ± 0.04a | 34.63 ± 3.92a | 74.81 ± 15.20a | 52.44 ± 3.97a |
| CV (%) | 1.41 | 2.37 | 11.13 | 25.46 | 9.07 |
| Significance | <0.0001** | <0.0001** | 0.9209 NS | 0.0738NS | 0.4281NS |
| HSD | 3.1031 | 0.0507 | 8.3319 | 35.175 | 5.2429 |
In contrast, root length (RL) did not show significant differences between treatments (p = 0.9209), with values ranging from 34.63 ± 3.92 to 36.31 ± 4.93 cm. This suggests that SiO2 NPs did not have a direct impact on root development under the experiment’s conditions, although they may have exerted indirect effects on root physiological efficiency that are not detectable by morphometric parameters. Above-ground dry biomass (ADB) exhibited a tendency to increase with higher doses of SiO2 nanoparticles, reaching the highest mean value at 7 mg L−1 (79.00 ± 27.43 g), followed by 9 mg L−1 (74.81 ± 15.20 g). However, despite this apparent increasing trend, the differences among treatments were not statistically significant (p = 0.0738) (Table 2). Despite this, the observed trend suggests a possible cumulative effect of silicon on dry matter production, probably associated with improvements in photosynthesis or the foliage’s greater structural resistance. Overall, these results confirm that the application of SiO2 NPs at concentrations between 5 and 9 mg L−1 promotes significant improvements in above-ground growth variables of the Quetzal pepper cultivar without compromising root morphology. These findings support the potential use of silicon as a plant biostimulant in protected production systems, contributing to the structural development of the crop without apparent adverse effects.
Regarding chlorophyll content, no statistically significant differences were observed among the evaluated treatments. Although the control treatment showed the highest mean chlorophyll value (54.03 ± 5.21 SPAD), this value did not differ significantly from those of the other treatments (Table 2).
Several studies have demonstrated the positive effects of silicon on plant growth. The application of SiO2 NPs significantly increased the height and stem diameter of pepper plants, for example with applications of 750 mg L−1 of SiO2 NPs compared to control treatments [8]. The incorporation of silicon in nutrient solutions applied to the root system at concentrations of 360 mg L−1 promoted stem diameter and plant height [16]. The presence of silicon aids in the structure of collenchyma tissue, which translates into more robust stems, a desirable characteristic in intensive agricultural production systems. This effect may be related to improved efficiency in the absorption and utilization of water and nutrients, especially potassium, as a result of the physiological action of silicon [6].
SiO2 NP applications have had positive and significant effects on plant growth, which is consistent with observations made in other horticultural crops. According to Cázarez-Flores et al. [17], in cucumber plants, an increase in silicon concentration leads to progressive stem elongation. Similarly, Villalón Mendoza et al. [18] found an increase in the height of pequin pepper plants after the application of silicon dioxide, suggesting that supplementation with this element provokes a positive growth response. These morphological effects coincide with those reported by de Sousa et al. [19], who attribute the changes in plant architecture to the regulatory role of silicon on hormonal activity. In particular, it has been noted that silicon modulates the biosynthesis and sensitivity of phytohormones such as abscisic acid (ABA), auxins, cytokinins, and gibberellins, which directly impacts the growth of meristematic tissues and parenchyma, thus favoring the structural development of the plant.
The results obtained showed a significant effect between the different doses of SiO2 NPs on the days taken to flower, number of fruits, fruit weight, and yield in pepper cultivation (p ≤ 0.05). A reduction in days taken to flower was observed as the concentration of SiO2 NPs increased. The shortest times were recorded at doses of 7 mg L−1 (38.27 ± 0.50 days) and 9 mg L−1 (38.50 ± 0.57 days), which were not statistically different from each other but were statistically different from the control treatment (44.50 ± 0.58 days), suggesting a biostimulant effect of silicon on floral induction (Table 3).
Table 3: Days taken to flower, number of fruits per plant, fruit weight, and yield of the Quetzal pepper crop grown with different doses of silicon dioxide nanoparticles (SiO2 NPs) under greenhouse conditions.
| Treatment | Days Taken to Flower | Number of Fruits Per Plant | Fruit Weight (g) | Yield (t ha−1) |
|---|---|---|---|---|
| Control | 44.50 ± 0.58a | 2.05 ± 0.15c | 52.16 ± 2.69c | 18.81 ± 2.76c |
| 5 mg L−1 SiO2 | 41.23 ± 0.50b | 3.00 ± 0.23b | 65.10 ± 0.73b | 34.10 ± 3.10b |
| 7 mg L−1 SiO2 | 38.27 ± 0.50c | 3.20 ± 0.22b | 66.78 ± 0.46b | 36.71 ± 2.64b |
| 9 mg L−1 SiO2 | 38.50 ± 0.57c | 3.71 ± 0.22a | 70.91 ± 1.10a | 44.28 ± 3.24a |
| CV (%) | 8.80 | 6.98 | 2.38 | 1.33 |
| Significance | <0.0001** | <0.0001** | <0.0001** | <0.0001** |
| HSD | 6.1841 | 0.4381 | 3.1855 | 1.1337 |
In terms of the number of fruits per plant, the treatment with 9 mg L−1 of SiO2 NPs had the highest average (3.71 ± 0.22), significantly exceeding the rest of the treatments, including the control (2.05 ± 0.15) and the intermediate doses (5 and 7 mg L−1), the latter showing increases that were statistically lower than the 9 mg L−1 average. Regarding the average fruit weight, there was a significant increase with the increase in the SiO2 NP dose, reaching the highest average with 9 mg L−1 (70.91 ± 1.10 g) in contrast to the lowest average presented in the control (52.16 ± 2.69 g). This trend suggests that silicon has a positive effect on biomass accumulation in reproductive organs (Table 3). Equally, yield per hectare showed an upward trend, reaching the highest average with 9 mg L−1 of SiO2 NPs (44.28 ± 3.24 t ha−1), which was statistically higher than the rest of the treatments. This result represents an increase of 135.40% compared to the control treatment (18.81 ± 2.76 t ha−1), confirming the potential of silicon as a yield-promoting element under controlled conditions. These results support the hypothesis that the application of SiO2 NPs in adequate doses can optimize the phenological development and productive parameters of pepper cultivation under greenhouse conditions. The dose of 9 mg L−1 showed the highest efficiency within the evaluated range, standing out for its significant effect on the analyzed variables (Table 3).
In rose crops, the application of silicon reduced the number of days it took for the flower to open, suggesting the potential for earlier flowering under certain conditions [20]. The role of silicon in improving plant growth and physiological attributes, such as photosynthetic efficiency, could contribute to changes in flowering time, as has been observed in studies with other plant species [21]. Silicon’s ability to improve essential oil production and the modification of oil composition in plants (such as Damask roses) under stress conditions indicate its broad impact on plant metabolic processes, which could influence flowering dynamics [22].
These findings are consistent with several previous studies that indicate the positive effects of silicon on the productivity of different crop species. In this regard, increased yields have been reported in crops such as Saccharum officinarum [23], Helianthus annuus [24], Triticum aestivum [25], and Oryza sativa [26]. This demonstrates the consistency of the beneficial effect of silicon in different agricultural systems. The positive impact of silicon on yield has been largely associated with its influence on photosynthetic processes, particularly in the functional improvement of photosystems I and II (PSI and PSII), which leads to increased chlorophyll synthesis [27]. In addition, silicon optimizes gas exchange through stomatal regulation, promoting carbon fixation and the generation of photoassimilates. This translates into an increase in biomass accumulation and, consequently, a substantial improvement in plant productive performance [28]. Silicon application can lead to increases in photosynthesis rates, longer leaves, improved fruit thickness, and a reduction in non-marketable yields in crops such as chili peppers [16].
Silicon applications promote a linear increase in the number of fruits per plant. This behavior suggests an improvement in the reproductive performance of the crop, attributed to the influence of silicon on key physiological processes such as flowering, fruit set, and initial fruit development [29]. It has been found that under saline stress conditions, the foliar application of silicon is effective in mitigating the effects of the growth environment. This allows productivity to be maintained or even increased compared to untreated plants [30] and reinforces its value as a tool for stabilizing yield under adverse conditions. In addition to the number of fruits, a significant increase in their individual weight has also been observed, an effect that can be explained by greater efficiency in the translocation of assimilates, improved nutrient absorption, and increased photosynthetic activity induced by silicon. The positive correlation between the application of this element and the fruit weight supports the hypothesis that silicon promotes more complete fruit development, aiding fruit filling and commercial quality [29].
These improvements in yield components contribute directly to a significant increase in productivity per hectare. The foliar application of silicon increases commercial crop yields due to the combination of a greater number of fruits and higher average fruit weight [29,30]. Likewise, it has been proven that the combination of silicon with other biostimulants, such as humic acid, enhances the beneficial effects, evidencing a possible synergy that maximizes agronomic benefits [31]. Despite the promising results, it is important to note that silicon’s effectiveness can vary depending on soil and climate conditions and the genetic characteristics of the cultivar, which justifies the need for differentiated management strategies. Viewed together, these findings confirm silicon’s role as a functional element in plant physiology, optimizing nutrient absorption efficiency, and promoting more vigorous and productive plant development. The results shown in Table 4 indicate that the application of SiO2 NPs is statistically different in the morphometric and biochemical parameters compared to the control in Quetzal peppers. Regarding the morphometric variables, fruit diameter increased as the concentration of SiO2 NPs increased, reaching the highest average of 4.88 ± 0.11 cm with the dose of 9 mg L−1, which represented an increase of 26.10% compared to the control (3.87 ± 0.16 cm). Similarly, fruit length showed no statistical differences between the 7 and 9 mg L−1 treatments, but they were significantly higher than the control, with increases ranging from 15.2 to 21.3%.
In terms of bioactive compounds, the application of 7 mg L−1 of SiO2 NPs showed the highest accumulation of total phenolic compounds at 331.67 ± 5.77, exceeding the control by 77.7%. This same treatment significantly enhanced the flavonoid content, registering an average of 237.67 ± 2.52, an increase of 109.1% in comparison to the control. The total antioxidant capacity was 136.00 ± 1.00 with the application of 7 mg L−1 of SiO2 NPs; this was a more modest increase (16.6%) compared to the control (Table 4).
Table 4: Fruit diameter (FD), fruit length (FL), total phenolic compounds (TPC), flavonoids (FLAV), and antioxidant capacity (AC) of the Quetzal pepper crop developed with different doses of silicon dioxide nanoparticles (SiO2 NPs) under greenhouse conditions.
| Treatment | FD | FL | TPC | FLAV | AC |
|---|---|---|---|---|---|
| Control | 3.87 ± 0.16c | 9.76b | 186.67 ± 1.53d | 113.67 ± 1.53d | 116.67 ± 1.53c |
| 5 mg L−1 SiO2 | 4.37 ± 0.07b | 11.24a | 223.33 ± 12.58c | 146.00 ± 1.73c | 116.00 ± 2.00c |
| 7 mg L−1 SiO2 | 4.68 ± 0.13a | 11.52a | 331.67 ± 5.77a | 237.67 ± 2.52a | 136.00 ± 1.00a |
| 9 mg L−1 SiO2 | 4.88 ± 0.11a | 11.84a | 256.00 ± 1.73b | 165.33 ± 1.53b | 121.00 ± 1.00b |
| CV (%) | 2.69 | 2.59 | 2.81 | 1.13 | 1.18 |
| Significance | <0.0001** | <0.0001** | <0.0001** | <0.0001** | <0.0001** |
| HSD | 0.2514 | 0.6034 | 18.35 | 4.89 | 3.77 |
In addition to the above, a positive and highly significant correlation was observed between the dose of SiO2 NPs and total phenolic compounds (r = 0.973, p < 0.001), flavonoids (r = 0.961, p < 0.001), and antioxidant capacity (r = 0.863, p < 0.001). These results indicate that SiO2 NP doses promote the accumulation of secondary metabolites and improve the nutraceutical profile of pepper fruits (Table 5). It is noteworthy that the 9 mg L−1 dose favored morphometric parameters, registering a decrease in the content of bioactive compounds compared to the 7 mg L−1 dose. This implies that silicon may have a hermetic effect on the crop’s secondary metabolism. The coefficients of variation obtained (1.13% to 2.81%) indicate high precision and reliability in the experimental data obtained (Table 4).
Table 5: Pearson’s correlation coefficients between the dose of silicon dioxide nanoparticles (SiO2 NPs), total phenolic compounds (TPC), flavonoids (FLAV), and antioxidant capacity (AC) in Quetzal pepper fruits.
| TPC | FLAV | AC | Dose of SiO2 | |
|---|---|---|---|---|
| TPC | 1 | |||
| FLAV | 0.992** | 1 | ||
| <0.0001 | ||||
| AC | 0.945** | 0.948** | 1 | |
| <0.0001 | <0.0001 | |||
| Dose of SiO2 | 0.973** | 0.961** | 0.863** | 1 |
| <0.0001 | <0.0001 | <0.0001 |
These results suggest that supplementation with 7 mg L−1 of SiO2 NPs is an efficient agronomic strategy to simultaneously enhance the fruit’s morphometric development and its nutraceutical quality, particularly in terms of phenolic compounds and antioxidant capacity. This coincides with reports by various authors on the beneficial effect of silicon in inducing adaptive responses in horticultural crops subjected to intensive production conditions.
Several studies have shown that the application of SiO2 NPs can significantly increase the size and weight of pepper fruits. In particular, combinations of potassium silicate with organic by-products such as vinasse have been shown to be effective in improving these physical attributes of the fruit [32]. An increase in the accumulation of phenolic compounds and flavonoids has been reported in fruits treated with SiO2 NPs, especially when used in the form of nanoparticles, which translates into greater antioxidant capacity. This coincides with the present study, where a positive and significant correlation was found between antioxidant capacity and total phenolic content (r = 0.945, p < 0.001), as well as antioxidant capacity and flavonoids (r = 0.948, p < 0.001). This is of utmost importance from a nutritional and nutraceutical point of view [33]. By strengthening plants’ physiological mechanisms, silicon dioxide not only contributes to maintaining the crops’ nutritional profile but also increases the concentration of beneficial compounds in harvested products, thus improving their overall nutraceutical quality. In chili crops, the foliar application of silicon mitigated adverse effects, such as salinity, by improving the water status, photosynthetic efficiency, and mineral nutrient content. Furthermore, it reduced oxidative damage and stabilized cell membranes. These findings support the potential of silicon as an effective strategy for improving salt stress tolerance in horticultural production systems [30].
The role of silicon goes beyond promoting growth; it is essential for improving plant tolerance to abiotic stresses such as high temperature, drought, and salinity. Research has shown that silicon can mitigate the negative effects of heat stress on plants by inducing the production of heat shock proteins and improving the antioxidant defense system. What is more, silicon application can improve water absorption through aquaporins, resulting in better hydration under stress conditions. This is particularly important when facing abiotic stress factors that alter the water and nutritional balance of plants [34].
The net photosynthetic rate, transpiration, midday water potential, and delta water potential were statistically different in the treatments with SiO2 NP doses (p ≤ 0.05). However, for stomatal conductance, intercellular CO2 concentration, water use efficiency, and leaf-to-air water vapor pressure deficit, water potential at dawn, and hydraulic conductivity, there were no significant differences between the treatments evaluated in the cultivation of Quetzal peppers (p > 0.05) (Fig. 2 and Fig. 3). The treatment with the highest net photosynthetic rate was the 9 mg L−1 SiO2 dose with a value of 17.54 μmol m−2 s−1, which was 26.28% higher than the control treatment. The 5 mg L−1 SiO2 dose presented the lowest values in the net photosynthetic rate, which were on average 13.68% lower than the control treatment. Similarly, the 9 mg L−1 SiO2 treatment had the highest averages in the transpiration variable, with a value of 4.14 mmol H2O m−2 s−1, exceeding the control treatment by 21.41% (Fig. 2).
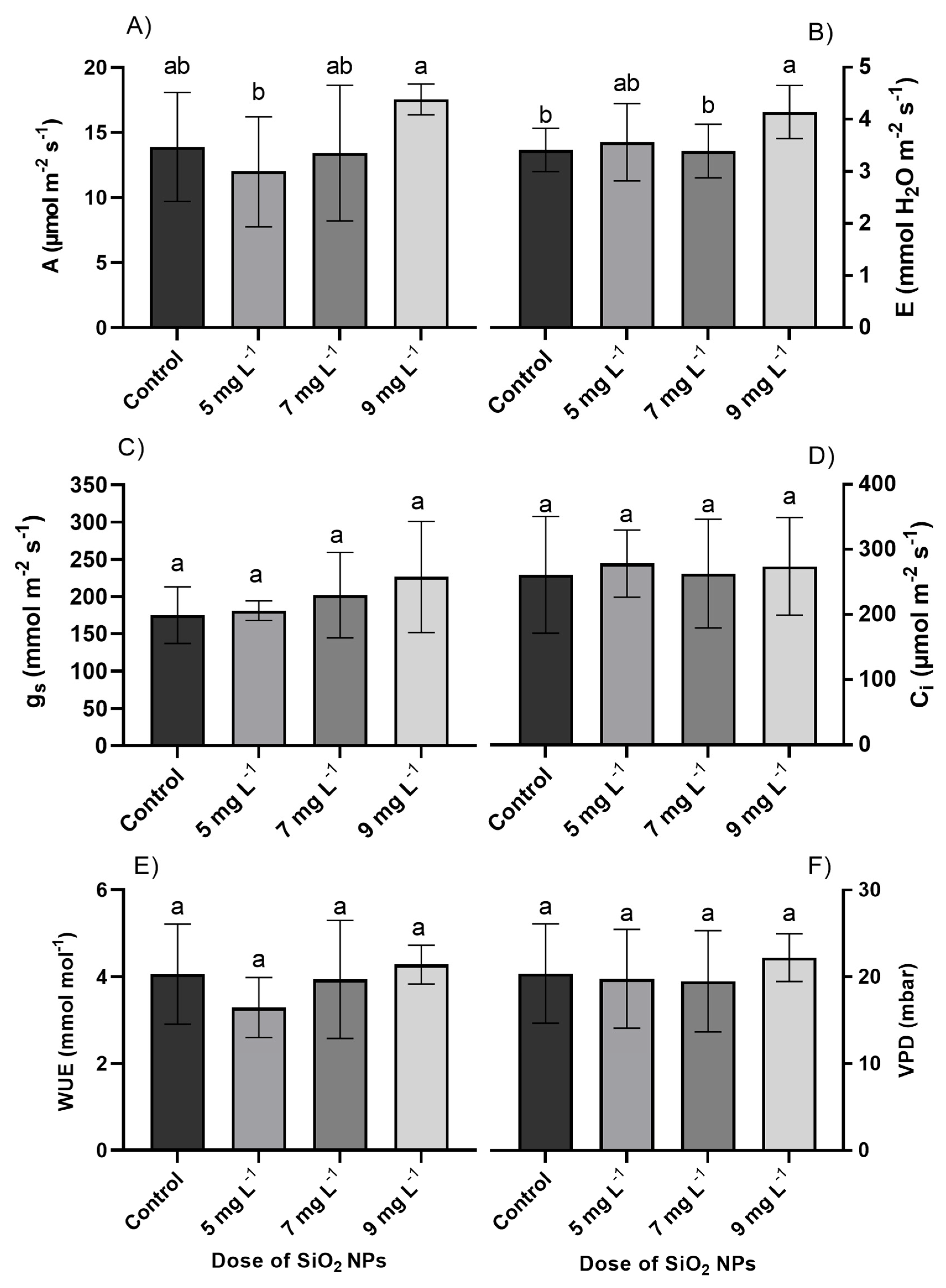
Figure 2: (A) Net photosynthetic rate, (B) transpiration, (C) stomatal conductance, (D) intercellular CO2 concentration, (E) water-use efficiency, and (F) leaf-to-air water vapor pressure deficit in the cultivation of Quetzal peppers with different doses of silicon dioxide nanoparticles (SiO2 NPs). Bars are means ± standard deviation; different letters indicate significant differences according to Tukey’s test (p ≤ 0.05).
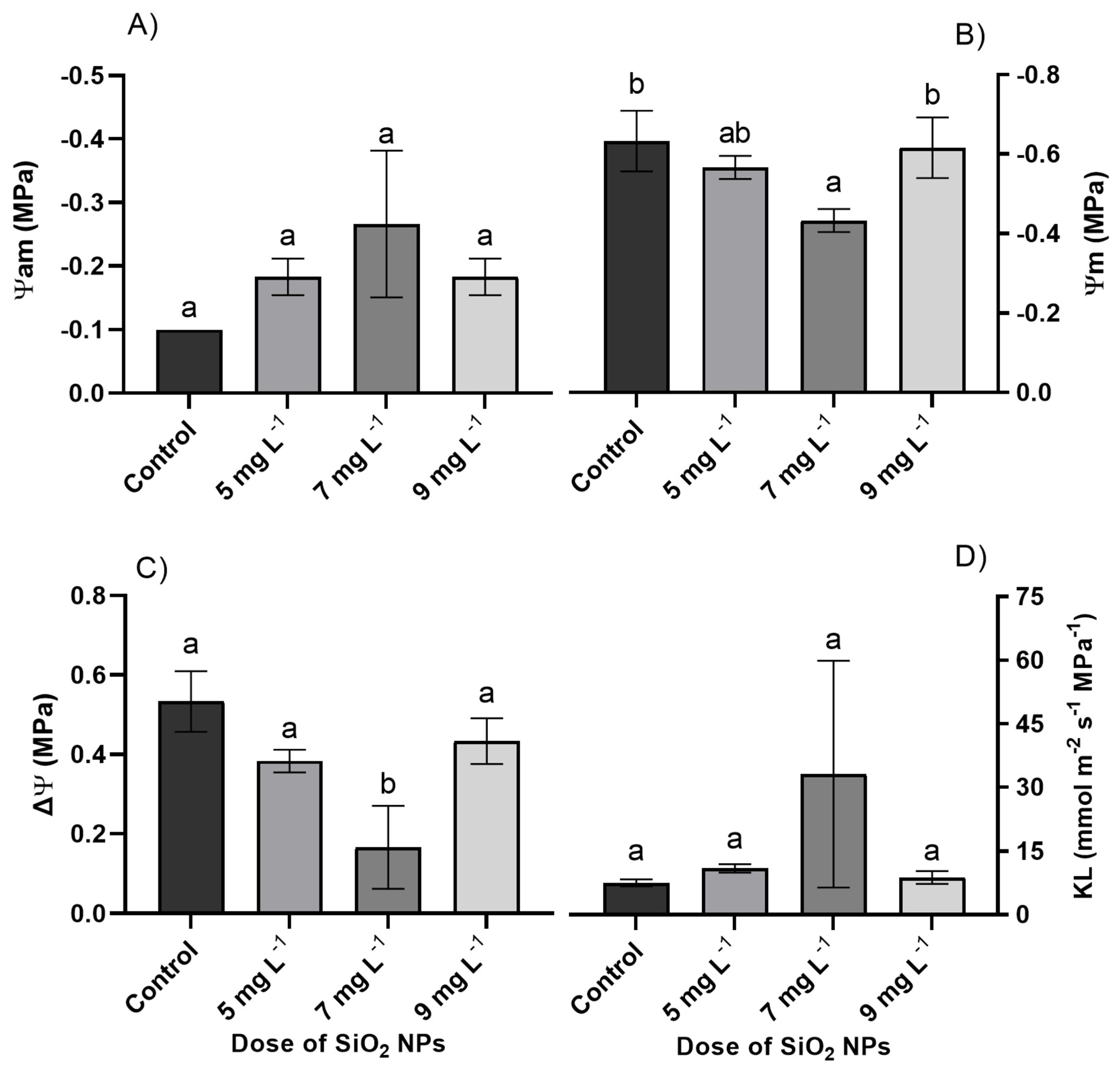
Figure 3: (A) Water potential at dawn (Ψam), (B) water potential at midday (Ψm), (C) delta water potential (ΔΨ), and (D) plant hydraulic conductivity in Quetzal pepper cultivation with different doses of silicon dioxide nanoparticles (SiO2 NPs). The bars represent means ± standard deviation; different letters indicate significant differences according to Tukey’s test (p ≤ 0.05).
Silicon is the second most abundant element in soil and has numerous beneficial effects on plants, especially in mitigating damage caused by stress. The incorporation of silicon into an environment of growth has shown positive effects on plant physiology and yield, reflected in increased photosynthesis, stomatal conductance, leaf water content, and cell membrane stability. These functional changes have resulted in greater biomass accumulation in pepper plants under saline stress, with more notable responses in sensitive genotypes. From an agronomic perspective, this effect is relevant when considering that the application of silicon can be an effective strategy to increase productivity in sensitive genotypes grown under moderate salinity, as well as in tolerant genotypes subjected to high levels of salinity, which demonstrates its potential as a management tool in saline environments [35].
Various studies have shown that the application of SiO2 nanoparticles plays a key role in improving the physiological and biochemical performance of plants. In fava bean (Vicia faba) cultivation, exposure to 1.5 mM SiO2 NPs significantly increased the photosynthetic rate, relative water content, and nutrient absorption. It also reduced the levels of reactive oxygen species such as hydrogen peroxide (H2O2) and molecular oxygen (O2), reflecting a protective effect associated with the regulation of redox homeostasis [36]. According to Ahmed et al. [37], in rapeseed (Brassica napus L.) cultivation, the application of SiO2 NPs mitigated cadmium stress by increasing chlorophyll content and net photosynthesis. This effect is linked to the activation of antioxidant enzymes (SOD, POD, CAT, and APX) and the restriction of heavy metal accumulation and translocation in tissues. These findings reinforce the hypothesis that SiO2 NPs act as modulators of plant physiology by optimizing photosynthetic efficiency, maintaining redox balance, and improving tolerance to stress conditions. Kumari et al. [38] reported that both nSiO2 and nTiO2, as well as their nanocomposites, promote the growth of corn plants (Zea mays), noting that the greatest stimulation occurred with 200 ppm of NC SiO2/TiO2. This positive effect was related to an increase in chlorophyll content, photosynthetic activity, transpiration, and stomatal conductance, in addition to promoting beneficial rhizosphere interactions through an increase in nitrogen-fixing and phosphate-solubilizing microorganisms. This resulted in more efficient nutrient absorption. Overall, scientific evidence confirms that metal oxide nanoparticles and their nanocomposites have high potential as biotechnological tools for improving the physiological performance and adaptive capacity of plants under both optimal and adverse conditions.
The results obtained in this study show that the application of silicon dioxide nanoparticles (SiO2 NPs) at concentrations of 7 and 9 mg L−1 had a significant positive effect on agronomic, physiological, and biochemical variables in the cultivation of Quetzal peppers grown in greenhouses. These doses promoted plant growth, plant height, stem diameter, and above-ground dry biomass, as well as increases in fruit production and quality, reflected in a greater number of fruits, fruit weight, and yield. Likewise, an improvement was observed in the accumulation of total phenolic compounds, flavonoids, and antioxidant capacity, suggesting the potential of SiO2 NPs in the nutraceutical quality of the fruit. Taken together, these findings indicate that silicon dioxide is a useful agronomic input for optimizing pepper productivity and quality. They also reinforce the need to continue evaluating SiO2 NP implementation in different agroecological conditions and cultivars.
Acknowledgement:
Funding Statement: The authors received no specific funding for this study.
Author Contributions: Conceptualization, Juan Jose Reyes-Pérez and Roger Alexander Pincay-Ganchozo; methodology, Juan Jose Reyes-Pérez; software, Bernardo Espinosa-Palomeque; validation, Luis Tarquino Llerena-Ramos; formal analysis, Juan Jose Reyes-Pérez and Bernardo Espinosa-Palomeque; investigation, Juan Antonio Torres-Rodríguez; resources, Juan José Reyes-Pérez; data curation, Bernardo Espinosa-Palomeque; writing—original draft preparation, Bernardo Espinosa-Palomeque; writing—review and editing, Juan Jose Reyes-Pérez and Bernardo Espinosa-Palomeque; visualization, Juan Antonio Torres-Rodríguez; supervision, Pablo Preciado-Rangel; project administration, Mercedes Susana Carranza-Patiño All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: The data that support the findings of this study are available from the corresponding author, upon reasonable request.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Rohitha K , Beena R , Jayalekshmy VG , Nivedhitha MS , Vijayakumar A , Gopinath PP . Changes in water stress indicators and antioxidant systems in chilli by chemical seed priming under water stress condition. Vegetos. 2023; 37( 4): 1489– 502. doi:10.1007/s42535-023-00695-1. [Google Scholar] [CrossRef]
2. Terry AE , Ruiz PJ , Rivera ER , Falcón RA , Carrillo SY . Bioproductos como sustitutos parciales de la nutrición mineral del cultivo de pimiento (Capsicum annuum L.). Acta Agronómica. 2022; 70( 3): 266– 73. doi:10.15446/acag.v70n3.86626. [Google Scholar] [CrossRef]
3. Belhedi M , Sghaier-Hammami B , Masiello M , Nafati H , Somma S , Gambacorta L , et al. Silicon dioxide (SiO2) nanoparticles affect the morphology, sporulation, mycotoxin production, and pathogenicity of Fusarium brachygibbosum infecting olive trees. Front Nanotechnol. 2025; 7: 1– 18. doi:10.3389/fnano.2025.1569453. [Google Scholar] [CrossRef]
4. Muhammad HMD , Naz S , Bibi Z , Ahmed MAA , Stanciu A-S , Ahmad R . Nanoparticles and phytohormonal synergy in plants: Sustainable agriculture approach. Phyton. 2025; 94( 9): 2631– 48. doi:10.32604/phyton.2025.069474. [Google Scholar] [CrossRef]
5. Gorge WA and Abood MF . Magnesium oxide nanoparticles synthesized from Solanum elaeagnifolium and their effect against Mealybugs Phenacoccus solenopsis Tinsley. J Glob Innov Agric Sci. 2025: 391– 5. doi:10.22194/jgias/25.1306. [Google Scholar] [CrossRef]
6. Martos-García I , Fernández-Escobar R , Benlloch-González M . Silicon is a non-essential element but promotes growth in olive plants. Sci Hortic. 2024; 323: 112541. doi:10.1016/j.scienta.2023.112541. [Google Scholar] [CrossRef]
7. Thabet AF , Boraei HA , Galal OA , El-Samahy MFM , Mousa KM , Zhang YZ , et al. Silica nanoparticles as pesticide against insects of different feeding types and their non-target attraction of predators. Sci Rep. 2021; 11( 1): 14484. doi:10.1038/s41598-021-93518-9. [Google Scholar] [CrossRef]
8. Prasad TNVKV , Satisha GC , Kumar ARN , Swethasree M , Girish BP , Sudhakar P , et al. Particulate nanoscale silica induced novel morphological and biochemical stimulus effects in Chilli (Capsicum annuum L.). ACS Agric Sci Technol. 2022; 2( 3): 555– 63. doi:10.1021/acsagscitech.2c00008. [Google Scholar] [CrossRef]
9. González-Moscoso M , Martínez-Villegas NV , Meza-Figueroa D , Rivera-Cruz MC , Cadenas-Pliego G , Juárez-Maldonado A . SiO2 Nanoparticles Improve Nutrient Uptake in Tomato Plants Developed in the Presence of Arsenic. Rev Bio Cienc. 2021; 8: 1– 25. (In Spanish). doi:10.15741/revbio.08.e1084. [Google Scholar] [CrossRef]
10. Reyes Pérez JJ , Llerena Ramos LT , Menace Almea MA , Castro Klinger EV , Aragón Sánchez E , de la Peña RdL , et al. Productividad y calidad del pimiento (Capsicum annumm L.) en respuesta a concentraciones de silicio. Chil J Agric Anim Sci. 2025; 41( 2): 279– 89. doi:10.29393/chjaas41-25pujj80025. [Google Scholar] [CrossRef]
11. Zhishen J , Mengcheng T , Jianming W . The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999; 64( 1): 555– 9. doi:10.1016/S0308-8146(98)00102-2. [Google Scholar] [CrossRef]
12. Singleton V , Orthofer R , Lamuela-Raventós RM . Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciacalteu reagent. Am J Enol Vitic. 1974; 25( 1): 152– 3. [Google Scholar]
13. Henriquez C , Aliaga C , Lissi E . Formation and decay of the ABTS derived radical cation: A comparison of different preparation procedures. Int J Chem Kinet. 2002; 34( 12): 659– 65. doi:10.1002/kin.10094. [Google Scholar] [CrossRef]
14. Sack L , Melcher PJ , Zwieniecki MA , Holbrook NM . The hydraulic conductance of the angiosperm leaf lamina: A comparison of three measurement methods. J Exp Bot. 2002; 53( 378): 2177– 84. doi:10.1093/jxb/erf069. [Google Scholar] [CrossRef]
15. Sack L and Scoffoni C . Measurement of leaf hydraulic conductance and stomatal conductance and their responses to irradiance and dehydration using the Evaporative Flux Method (EFM). Jove-J Vis Exp. 2012; 70( 70): 1– 7. doi:10.3791/4179. [Google Scholar] [CrossRef]
16. Lob S , Sa’ad NS , Ibrahim NF , Che Soh N , Mohd Shah R , Zaudin MSH . Enhanced growth of chili (Capsicum annuum L.) by silicon nutrient application in fertigation system. Malays Appl Biol. 2023; 52( 2): 13– 9. doi:10.55230/mabjournal.v52i2.2648. [Google Scholar] [CrossRef]
17. Cázarez-Flores LL , Partida-Ruvalcaba L , Velázquez-Alcaraz TdJ , Ayala-Tafoya F , Díaz-Valdés T , Yáñez-Juárez MG , et al. Silicio y cloro en el crecimiento, rendimiento y calidad postcosecha de pepino y tomate. Rev Terra Latinoam. 2022; 40: 1– 11. doi:10.28940/terra.v40i0.994. [Google Scholar] [CrossRef]
18. Villalón Mendoza H , Castillo-Villarreal MA , Garza-Ocañas F , Guevara-González JA , Sánchez-Castillo L . Dióxido de silicio como estimulante del índice de calidad de plantas de chile piquín (Capsicum annuum L. var. glabriusculum) producidas en vivero. Rev Mex De Cienc For. 2018; 9( 50): 294– 303. doi:10.29298/rmcf.v9i50.247. [Google Scholar] [CrossRef]
19. de Sousa A , Saleh AM , Habeeb TH , Hassan YM , Zrieq R , Wadaan MAM , et al. Silicon dioxide nanoparticles ameliorate the phytotoxic hazards of aluminum in maize grown on acidic soil. Sci Total Environ. 2019; 693: 133636. doi:10.1016/j.scitotenv.2019.133636. [Google Scholar] [CrossRef]
20. Jalilzadeh Khoie E , Jabbarzadeh Z , Norouzi P , Barin M , Razavi M . Silicon spray affect floricultural traits and leaf elemental nutrient concentrations of Rose ‘Beverly Watson’. J Plant Nutr. 2023; 47( 1): 145– 56. doi:10.1080/01904167.2023.2262513. [Google Scholar] [CrossRef]
21. Uddin M , Bhat UH , Singh S , Singh S , Chishti AS , Khan MMA . Combined application of SiO2 and TiO2 nanoparticles enhances growth characters, physiological attributes and essential oil production of Coleus aromatics Benth. Heliyon. 2023; 9( 11): e21646. doi:10.1016/j.heliyon.2023.e21646. [Google Scholar] [CrossRef]
22. Farahani H , Sajedi NA , Madani H , Changizi M , Naeini MR . Effect of foliar-applied silicon on flower yield and essential oil composition of damask rose (Rosa damascena Miller) under water deficit stress. Silicon. 2020; 13( 12): 4463– 72. doi:10.1007/s12633-020-00762-1. [Google Scholar] [CrossRef]
23. Jain R , Singh A , Jain N , Tripathi P , Chandra A , Shukla SK , et al. Response of silixol sugarcane to growth and physio-biochemical characteristics of sugarcane. Sugar Tech. 2017; 20( 4): 439– 44. doi:10.1007/s12355-017-0557-z. [Google Scholar] [CrossRef]
24. Flores RA , Arruda EM , Souza Junior JPd , de Mello Prado R , Santos ACAd , Aragão AS , et al. Nutrition and production of Helianthus annuus in a function of application of leaf silicon. J Plant Nutr. 2018; 42( 2): 137– 44. doi:10.1080/01904167.2018.1549678. [Google Scholar] [CrossRef]
25. Helal NM , Khattab HI , Emam MM , Niedbala G , Wojciechowski T , Hammami I , et al. Improving yield components and desirable eating quality of two wheat genotypes using Si and nanosi particles under heat stress. Plants. 2022; 11( 14): 1819. doi:10.3390/plants11141819. [Google Scholar] [CrossRef]
26. Jeer M , Suman K , Maheswari TU , Voleti SR , Padmakumari AP . Rice husk ash and imidazole application enhances silicon availability to rice plants and reduces yellow stem borer damage. Field Crops Res. 2018; 224: 60– 6. doi:10.1016/j.fcr.2018.05.002. [Google Scholar] [CrossRef]
27. Helaly MN , El-Hoseiny H , El-Sheery NI , Rastogi A , Kalaji HM . Regulation and physiological role of silicon in alleviating drought stress of mango. Plant Physiol Biochem. 2017; 118: 31– 44. doi:10.1016/j.plaphy.2017.05.021. [Google Scholar] [CrossRef]
28. Wang Y , Zhang B , Jiang D , Chen G . Silicon improves photosynthetic performance by optimizing thylakoid membrane protein components in rice under drought stress. Environ Exp Bot. 2019; 158: 117– 24. doi:10.1016/j.envexpbot.2018.11.022. [Google Scholar] [CrossRef]
29. Reyes-Pérez JJ , Rivas-García T , Rodríguez-Rodríguez S , Torres-Rodríguez JA , Llerena-Ramos LT , Preciado-Rangel P , et al. Relations between the mass of the fruits, the number of fruits per plant and the agricultural yield in pepper, with increasing silicon doses. Trop Subtrop Agroecosystems. 2024; 27( 2): 5190. doi:10.56369/tsaes.5190. [Google Scholar] [CrossRef]
30. Abdelaal KAA , Mazrou YSA , Hafez YM . Silicon foliar application mitigates salt stress in sweet pepper plants by enhancing water status, photosynthesis, antioxidant enzyme activity and fruit yield. Plants. 2020; 9( 6): 733. doi:10.3390/plants9060733. [Google Scholar] [CrossRef]
31. El Sayed HA , Farid SM , El-Zehery R . The effect of some soil treatment and foliar applications on the productivity and quality of pepper crop. J Plant Prod. 2015; 6( 4): 623– 43. doi:10.21608/jpp.2015.49587. [Google Scholar] [CrossRef]
32. Rady MS , Ghoneim IM , Feleafel MN , Hassan SM . Enhancing biometric characters of perennial sweet pepper (Capsicum annuum L.) treated by potassium silicate and vinasse grown under greenhouse conditions. Preprints. 2023. doi:10.21203/rs.3.rs-3312756/v1. [Google Scholar] [CrossRef]
33. González-García Y , Flores-Robles V , Cadenas-Pliego G , Benavides-Mendoza A , Cabrera De La Fuente M , Sandoval-Rangel A , et al. Application of two forms of silicon and their impact on the postharvest and the content of bioactive compounds in cucumber (Cucumis sativus L.) fruits. Biocell. 2022; 46( 11): 2497– 506. doi:10.32604/biocell.2022.021861. [Google Scholar] [CrossRef]
34. Mir RA , Bhat BA , Yousuf H , Islam ST , Raza A , Rizvi MA , et al. Multidimensional role of silicon to activate resilient plant growth and to mitigate abiotic stress. Front Plant Sci. 2022; 13: 819658. doi:10.3389/fpls.2022.819658. [Google Scholar] [CrossRef]
35. Altuntas O , Dasgan HY , Akhoundnejad Y . Silicon-induced salinity tolerance improves photosynthesis, leaf water status, membrane stability, and growth in pepper (Capsicum annuum L.). HortScience. 2018; 53( 12): 1820– 6. doi:10.21273/hortsci13411-18. [Google Scholar] [CrossRef]
36. Desoky EM , Mansour E , El-Sobky EEA , Abdul-Hamid MI , Taha TF , Elakkad HA , et al. Physio-biochemical and agronomic responses of faba beans to exogenously applied nano-silicon under drought stress conditions. Front Plant Sci. 2021; 12: 637783. doi:10.3389/fpls.2021.637783. [Google Scholar] [CrossRef]
37. Ahmed T , Masood HA , Noman M , AL-Huqail AA , Alghanem SM , Khan MM , et al. Biogenic silicon nanoparticles mitigate cadmium (Cd) toxicity in rapeseed (Brassica napus L.) by modulating the cellular oxidative stress metabolism and reducing Cd translocation. J Hazard Mater. 2023; 459: 132070. doi:10.1016/j.jhazmat.2023.132070. [Google Scholar] [CrossRef]
38. Kumari K , Rani N , Hooda V . Unravelling the effects of nano SiO2, nano TiO2 and their nanocomposites on Zea mays L. growth and soil health. Sci Rep. 2024; 14( 1): 13996. doi:10.1038/s41598-024-61456-x. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools