 Open Access
Open Access
REVIEW
Global Impact of Potential Use of Phytomelatonin in Crops
1 Phytohormones and Plant Development Laboratory, Department of Plant Biology (Plant Physiology), Faculty of Biology, University of Murcia, Murcia, Spain
2 Life Sciences and Systems Biology Department, Plant Physiology Unit, University of Turin, Turin, Italy
3 Soil-Plant Laboratory, Center of Plant, Soil Interaction and Natural Resources Biotechnology-BIOREN-UFRO, University of La Frontera, Temuco, Chile
* Corresponding Author: Marino B. Arnao. Email:
Phyton-International Journal of Experimental Botany 2026, 95(3), 1 https://doi.org/10.32604/phyton.2026.077309
Received 06 December 2025; Accepted 06 February 2026; Issue published 31 March 2026
Abstract
Phytomelatonin is a versatile compound with important roles in plants, spanning from promoting germination and plant growth to defending against abiotic and biotic stresses. Its natural origin as biostimulator aligns with sustainable agricultural practices that aim to reduce reliance on synthetic agrochemicals while supporting crop productivity. As research progresses and reveals its full potential across various sectors of agriculture and food production systems, phytomelatonin emerges as a promising candidate for integrated crop management, with the goal of achieving more resilient and sustainable food systems worldwide. More agronomic data will be essential to decide whether the use of (phyto)melatonin can be a clear advance in the improvement of crops and their production. This paper presents the possible improvements and challenges to be faced in order to elucidate whether the cost/benefit balance in the use of melatonin in crops will be environmentally and economically sustainable.Keywords
1 Phytomelatonin and Its Conceptual Framework
Phytomelatonin is a naturally occurring compound found in various plant species that shares a similar chemical structure to melatonin, the hormone predominantly known for its role in regulating biological rhythms in animals and humans [1]. The discovery of phytomelatonin in edible plants in 1995 has opened exciting new avenues for its application beyond agriculture into the realm of animal nutrition [2,3,4]. Phytomelatonin is present in all the plants analyzed, both wild and which are consumed by humans and animals, which permit them to explore its potential benefits when incorporated into diets. Because it is derived from natural sources, it aligns well with current trends favoring sustainable agriculture and organic farming practices [5].
Phytomelatonin is synthesized by a wide range of fruits, vegetables, grains, seeds, and other botanical sources. Its presence in such a diverse array of plants suggests that phytomelatonin plays important roles in plant physiology, including plant growth regulation and plant responses to environmental stressors. In a similar way that synthetic melatonin, phytomelatonin has attracted significant scientific interest due to its potential health benefits when included in the human diet. Some authors explored its application as a nutraceutical, a natural supplement that can promote health and prevent disease due to its antioxidant properties and ability to influence biological clock and other processes related to aging and diseases [6,7]. Additionally, melatonin is considered as a phytogenic additive in animal feed, aiming to improve livestock health and productivity through natural means [8]. Melatonin also exhibits potent antioxidant activity by neutralizing harmful free radicals generated during normal metabolic processes or due to environmental stressors such as pollution, heavy metals or UV radiation [9,10,11,12,13,14]. Dietary intake of (phyto)melatonin can influence these biological processes. When consumed through foods rich in this compound or via supplements derived from plant sources, phytomelatonin may act as a natural agent supporting overall health [15,16]. Moreover, its antioxidant capacity helps protect cells from oxidative damage by scavenging reactive oxygen species (ROS), thereby reducing cellular stress and preventing damage to DNA, proteins, and lipids, which has encouraged its consumption as an anti-aging supplement [17,18].
In this work, we present a practical vision to elucidate the concepts surrounding (phyto)melatonin’s mechanisms and possible applications in agriculture from both scientific and legal perspectives within the EU context. It explores the current state of knowledge about (phyto)melatonin’s biological effects on plants, examines regulatory considerations for its approval as an active substance or biostimulant, and discusses future research directions for its integration into sustainable farming practices.
2 Perspectives on Possible Agricultural Applications
In plants, phytomelatonin serves a multitude of vital functions that are essential for growth, development, and resilience. This naturally occurring compound influences key physiological processes such as seed germination, root and shoot development, and photosynthesis efficiency. By promoting germination, rooting, root and shoot elongation and enhancing overall plant vigor, phytomelatonin contributes to healthier and more robust plants. It also plays a significant role in regulating flowering time, fructification (parthenocarpy) and ripening which can impact crop yield and quality [19,20,21,22].
One of the most critical roles of phytomelatonin is its involvement in plant responses to environmental stresses. Plants are constantly exposed to various abiotic stressors such as drought, waterlogging, salinity and alkalinization of soils, extreme temperatures (heat and cold), and oxidative damage (increasing ROS) caused by pollutants such as heavy metals, pesticides, and others. Phytomelatonin helps mitigate these stresses by acting as a potent antioxidant. It scavenges free radicals, unstable molecules that can cause cellular damage, and thereby reduces oxidative stress within plant cells. This protective function enhances plant resilience under adverse conditions, allowing them to maintain plant growth and productivity despite environmental challenges [23,24,25,26,27].
Moreover, phytomelatonin is involved in activating defense mechanisms against pathogen attacks. When plants face threats from bacteria, fungi, or viruses, phytomelatonin levels often increase as part of the plant’s innate immune response. This boost in defense capacity helps limit disease progression and supports overall plant health [10,28,29]. Consequently, the presence of phytomelatonin contributes to increased crop yields by improving plant resistance to both biotic and abiotic stresses.
Beyond its role as an antioxidant, phytomelatonin functions as a regulator of plant hormones which coordinate plant growth, development, and stress responses. It interacts with phytohormone’s pathways such as auxins, cytokinins, gibberellins, abscisic acid, ethylene, jasmonic and salicylic acid to fine-tune physiological processes. This regulatory capacity allows plants to adapt more effectively to changing environmental conditions by modulating plant growth patterns and stress responses [19].
Phytomelatonin is a versatile bioactive compound with significant functions within plants, ranging from promoting vegetative and reproductive development to protecting against environmental stresses, both abiotic and biotic (Fig. 1). According to its actions of agronomic interest, we can classify the advantages of the application of phytomelatonin in crops such as:
- 1.(Phyto)melatonin as a plant biostimulator
A biostimulator is a substance that promotes plant growth or development without being classified as a fertilizer. (Phyto)melatonin’s ability to influence key physiological processes makes it an attractive candidate in:
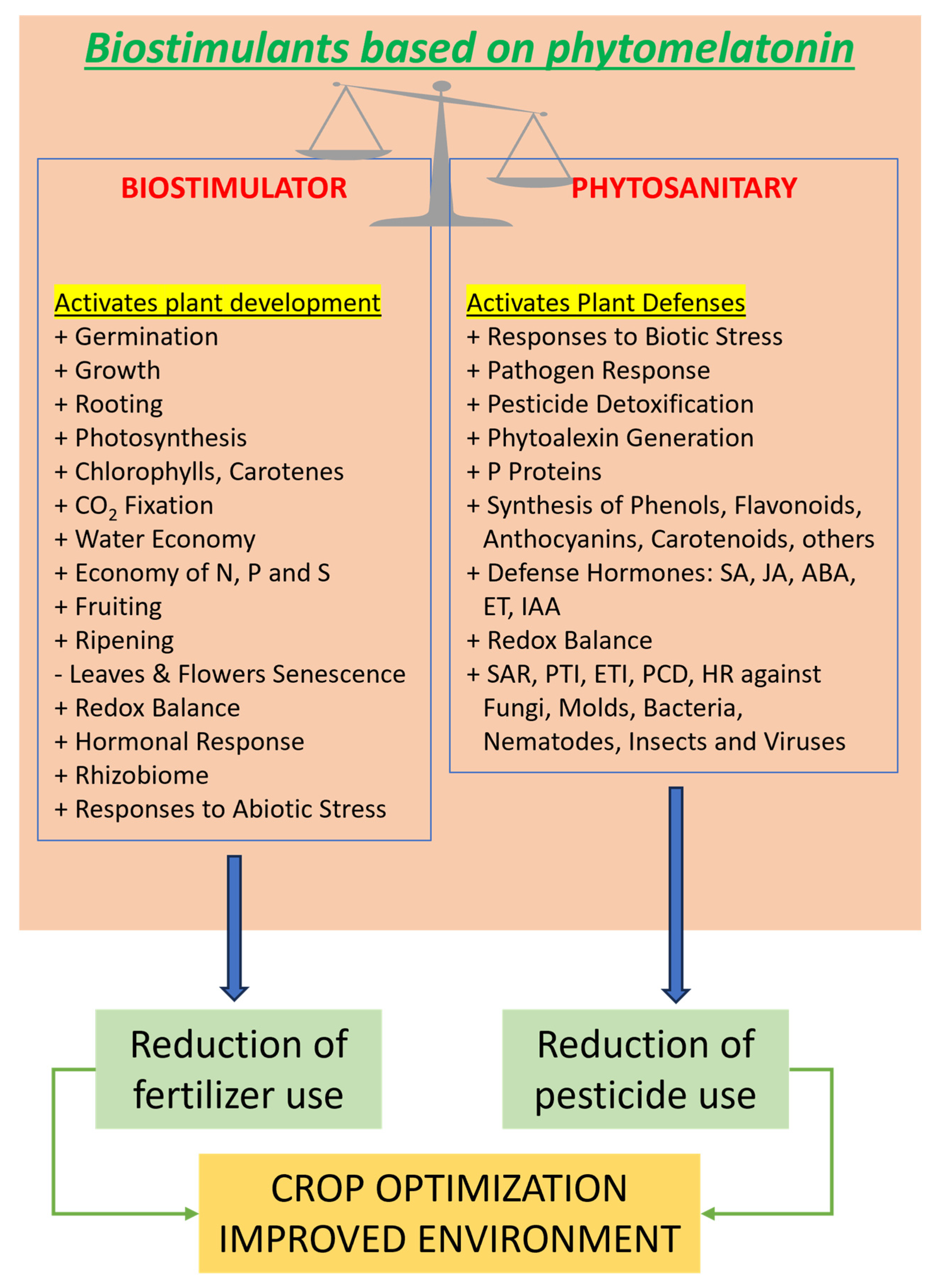
Figure 1: Positive effects of phytomelatonin on several physiological and agronomic aspects.
1a. Vegetative and Reproductive Physiology: Several studies have shown that exogenous application of melatonin can stimulate seed germination, rooting, root elongation, shoot growth, flowering time regulation, and fruit development, for example inducing parthenocarpic responses in some crops. In general, we can act from seedlings, obtaining more vigorous plantlets into the decisive step between greenhouse to open field, to mature plants, where phytomelatonin could reinforce stress responses against abiotic and biotic agents. Also, the induction of endogenous phytomelatonin biosynthesis showed similar physiological responses to those described above [21,23].
1b. Stress Tolerance: Melatonin enhances plants’ resilience against environmental stresses by scavenging ROS, upregulating antioxidant enzymes like superoxide dismutase, catalase and peroxidases, improving ascorbate/glutathione redox forms, and also upregulating the expression of Asada’s cycle enzymes through the regulation of the enzymes implies, increasing GSH/GSSG and ASA/DHA ratios [21]. So, phytomelatonin modulates gene expression related to specific stress factors. Generic abiotic stress transcription factors such as DREB, CBF, MYB and WRKY can be induced by melatonin. For example, in cold stress response, phytomelatonin up-regulates responsive transcription factors such as CAMTA3, ZAT6, ZAT10, and ZAT12, and several cold-responsive genes such as COR, LTI, KIN and RD, all of which improve cold tolerance [26].
Under salinity stress, melatonin treatment counteracts the growth inhibition and reduced photosynthesis caused by salt, thereby enhancing salinity tolerance. Melatonin safeguards the photosynthetic apparatus from biochemical, structural, and physiological damage. It also reduces reactive oxygen species (ROS) levels and membrane injury by activating the antioxidant system and regulating osmoregulation. Regarding transcriptional responses, a group of calcineurin B-like (CBL) proteins, which interact specifically with a family of protein kinases called CBL-interacting protein kinases (CIPKs), play a key role in melatonin-induced salinity response. The physiological functions of CBL and CIPK were initially identified in the salt overly sensitive (SOS) pathway. Arabidopsis mutants sos1, sos2, and sos3 exhibit salt sensitivity under high-salt conditions. After melatonin treatment, SOS1, SOS2, SOS3, along with certain Na+/H+ antiporters (NHXs), are significantly upregulated, leading to improved salt tolerance [30,31].
In soils contaminated with pollutants such as pesticides or heavy metals, plants encounter significant growth challenges not only due to the toxicity of these substances but also because of soil degradation factors like acidification, alkalinization, mineral deficiencies, and the lack of rhizogenic life. Treatment with melatonin helps plants develop greater tolerance to these polluted soils. It reduces oxidative damage and lowers the levels of heavy metals that reach the cells. Melatonin activates the antioxidant system and promotes the production of phytochelatins, specialized proteins that bind and sequester heavy metals into complexes stored in vacuoles and/or cell walls, preventing interference with cellular metabolism. Recent research highlighting melatonin’s role in counteracting heavy metal toxicity and its potential in phytoremediation techniques can be found in studies [32,33,34].
1c. Photosynthesis Efficiency and Yield: Application of melatonin has been linked to improved chlorophyll content and photosynthetic activity through the activation of photosystem elements, especially under stress conditions. Phytomelatonin enhances CO2 uptake and reduces water loss, boosting water-use efficiency (the ratio of moles CO2 fixed to moles H2O used). Consequently, melatonin was shown to elevate photosynthetic rate, PSII efficiency, Rubisco activity, chlorophyll and carotenoid levels, stomatal conductance, and leaf internal CO2. The Rubisco small subunit (RbcS) and many chlorophyll a/b binding protein transcripts were upregulated after melatonin treatment, while the senescence-related transcription factor SAG12 and chlorophyll degradation enzymes such as pheophorbide a oxygenase (PaO) were downregulated [35].
A field of study of great interest is legume crops. Melatonin treatments in grain legumes in different stress situations resulted in an increase in antioxidant enzymes and nitrogen metabolism, along with the induction of antioxidant and osmoprotective metabolites. The result in cultivation was an increase in photosynthetic capacity, growth and yield in several grain legume species. However, further studies in these species would be needed to resolve numerous gaps in their mode of action [36]. Similarly, although with many more studies, tests carried out with melatonin in cereals have clearly shown a benefit in the crop, especially in situations of stress due to drought, salinity and extreme temperatures. Today, multiple field trials are underway in wheat and other cereals to verify their real possibilities of improvement under natural conditions [37,38].
1d. Primary and Secondary Metabolism: Phytomelatonin markedly promotes the overexpression of genes encoding various enzymes involved in carbohydrate, amino acid, and lipid metabolism in plants. This includes enzymes related to sucrose and starch synthesis, glycolysis, fermentation, the Krebs cycle, the pentose phosphate pathway, fatty acid production, and other plant metabolic processes. Also, phytomelatonin regulates N-, P-, and S-metabolism, and plant cell osmoregulation processes. Significant regulated effects on polyphenolic, glucosinolates, terpenoid and alkaloid pathways occur mediated by phytomelatonin [21].
1e. Post-Harvest Preservation: Phytomelatonin can delay senescence and spoilage of fruits and vegetables during storage by reducing oxidative damage. In this case, melatonin application near pre-harvest or already in post-harvest, improves the quality and half-life of products, enlightening their organoleptic qualities and, generally, delaying senescent processes. In short, melatonin-treated fruits and vegetables arrive in better conditions for the consumer, improving transport and extending their shelf life. An extensive list of fruits and vegetables treated with melatonin and post-harvest improved can be consulted [20,39].
To our knowledge, phytomelatonin’s receptor (CAND2/PMTR1)-mediated actions appear to include a dual signal-transduction pathway: one pathway through the activation of successive MAP kinases (MPK3/6 AND SnRK1) and phosphatases (PP2C1); and another through GPA1, the alpha subunit of the heterotrimeric G proteins [20,40]. These actions of phytomelatonin mediated by its receptor would constitute only a part of its action since there are many other direct and indirect responses that we do not know their mechanism or signaling pathway. The multi-response factor of phytomelatonin plus the role as a regulator of the levels and signaling of other phytohormones suggests that its action is better included within the scope of plant master regulator than of just another plant hormone [41].
- 2.(Phyto)melatonin against biotic stressors
Melatonin acts as a versatile protector against biotic stress by activating two levels of plant immunity: PTI and ETI. The recognition of pathogen effectors by NLR receptors triggers a robust defense response characterized by HR, SAR, and PCD, aimed at restricting pathogen spread. Melatonin also modulates the crosstalk between signaling molecules such as NO, JA, SA, and ROS, enhancing the host’s defensive capacity and upregulating protective genes (Fig. 2). In addition to its signaling functions, melatonin contributes to structural defense; studies in Arabidopsis thaliana indicate that it promotes callose deposition and elevates cell wall constituents (cellulose, xylose, and galactose), thereby strengthening the physical barrier against bacterial and fungal invasions.
This action of melatonin seems to be determined by its role in the up-regulation of multiple wall enzymes such as cellulose synthase, callose synthase, pectin esterase, pectin-methyl esterase, and many others. The transcripts of several sucrose-related enzymes such as sucrose synthases, invertases, and sucrose-transporters are up-regulated by melatonin, increasing cellulose, callose, and starch accordingly with reported in melatonin-treated plants [42,43,44,45,46,47].
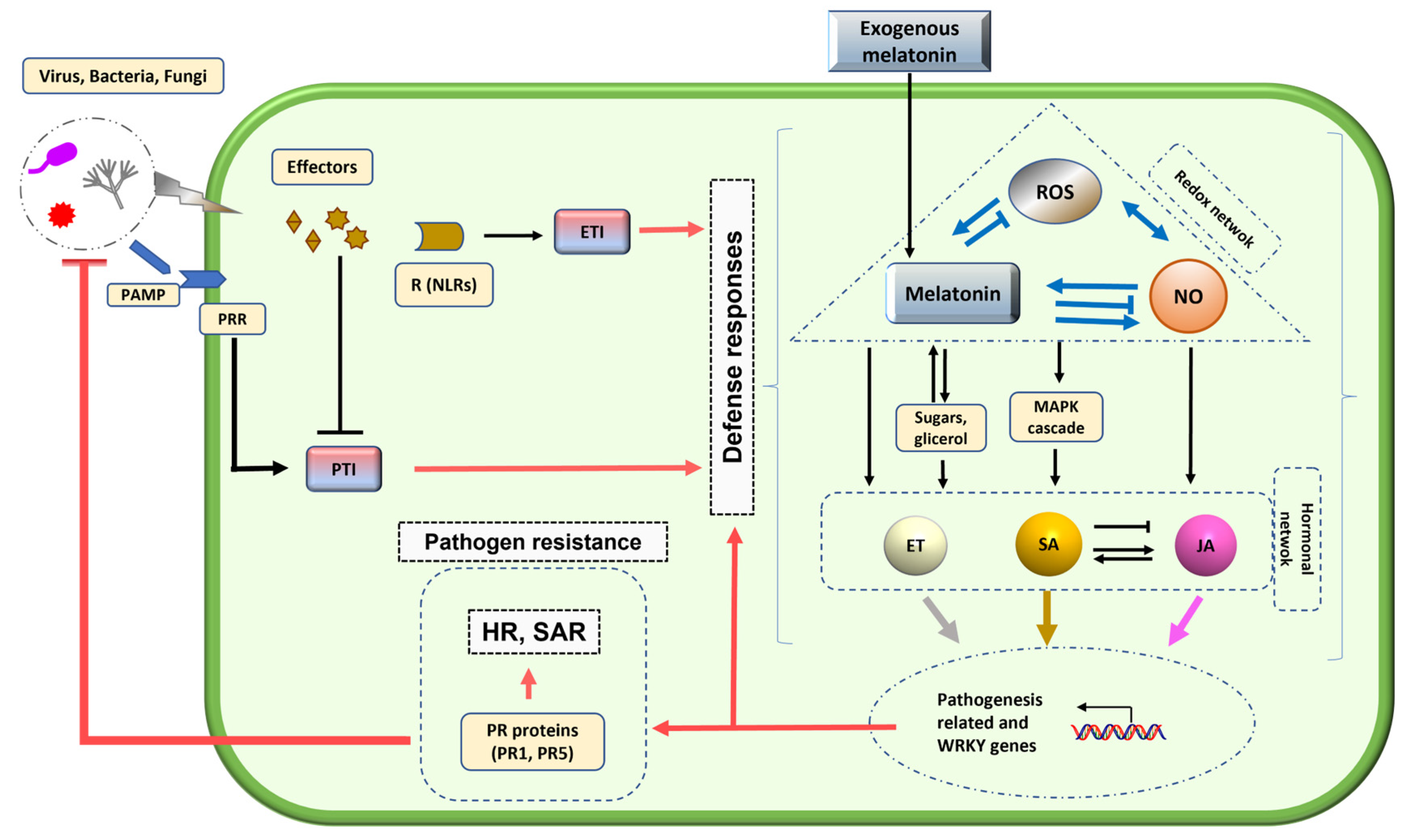
Figure 2: Model proposed for the interaction between plant pathogens and defense responses mediated by melatonin [29].
As consequence, phytomelatonin slows down damage and stimulates systemic acquired resistance, which contributes to increasing both crop health and postharvest health quality, minimizing the spoilage of fruits and vegetables during storage. An interesting aspect of the use of phytomelatonin as plant biotic protector is that it can act synergistically with fungicides in some cases, promoting high plant growth and tolerance to fungicide, with low damage at low fungicide doses applied, all these as an eco-friendly alternative [48]. A similar focus has been proposed by melatonin as a safener versus pesticides such as herbicides, acaricides, etc. [49].
3 Legal Considerations on (Phyto)Melatonin in the European Union
In recent years, the agricultural sector has experienced significant transformations driven by technological advances, environmental concerns, and evolving regulatory frameworks. Among these developments is the exploration of novel substances that can enhance crop productivity, improve plant resilience, and reduce reliance on traditional chemical pesticides and fertilizers. One such promising compound is melatonin. Its multifaceted properties include acting as a plant growth regulator, a biostimulant, and a protector against various abiotic and biotic stresses. These attributes position melatonin as a candidate for sustainable agriculture practices aimed at increasing yields while minimizing environmental impact.
However, integrating melatonin into crop management systems necessitates careful consideration of legal frameworks governing active substances used in plant protection products and fertilizers within the European Union (EU). The EU’s regulatory landscape has undergone numerous reforms to establish clear policies regarding authorization, classification, use, distribution, importation, and management of such substances. This ensures that innovations like melatonin are incorporated responsibly, balancing agricultural benefits with safety for humans, animals, and ecosystems.
The term “biostimulant” was first proposed to denote “materials that, in minute quantities, promote plant growth” by Zhang and Schmidt (1997) [50]. Although extensive revision works on this topic can be consulted [51], under the EC (European Community) regulation (EU 2019/1009) “Plant biostimulants are defined as products stimulating plant’s nutrition processes independently of the products nutrient content with the aim of improving the nutrient use efficiency, tolerance to abiotic stress, crop quality, availability of confined nutrients in the soil and rhizosphere, also through the humification and degradation of organic compounds in the soil”.
The European Biostimulants Industry Council (EBIC) presents a fairly comprehensive definition of biostimulants; refers to them as substances or microorganisms applied to plants or soil that stimulate natural processes by improving nutrient efficiency, tolerance to abiotic stress, and nutrient quality and availability, regardless of their direct nutrient content. This means that biostimulants do not provide nutrients in the traditional sense, as fertilizers tend to do, but rather act by stimulating the physiology and metabolism of the plant, helping it to grow better and cope with difficult conditions. This continuous evolution of biostimulant definition has resulted in a lack of standardized definitions and methods for evaluating the efficacy of these products. It is often unclear which component or combination of components is really responsible for the biostimulator effect, and the composition itself can vary greatly even among seemingly similar commercial products [52].
"Regarding its regulatory status, the European Chemicals Agency (ECHA) classifies melatonin (N-(2-(5-methoxyindol-3-yl)-ethyl)-acetamide; EC No. 200-797-7) as non-hazardous in terms of physical-chemical properties, environmental impact, and most human health endpoints, including mutagenicity and carcinogenicity. However, it is strictly designated as a reproductive toxicant (Hazard Statement H361: 'Suspected of damaging fertility or the unborn child'). This classification reflects its potent biological role as a mammalian hormone; for instance, melatonin is widely used in livestock to synchronize reproductive cycles and enhance conception rates. While environmentally benign, this specific toxicological profile complicates its potential authorization as an active substance under the REACH Regulation (EC) 1907/2006, as reproductive safety remains a critical threshold for chemical registration."
However, if we look at a possible Maximum Residue Limits (MRL) for melatonin, since there have never been cases of toxicity in humans due to melatonin intake and the doses of application are usually very low (<30 ppm), added to the fact that melatonin is rapidly metabolizable and very sensitive to light conditions, there may not be problems of residues in the treatments, as we found in a study on post-harvest conservation of broccoli [53].
4 Future Perspectives on Phytomelatonin Applications in Crops
Lab assays vs. Crop assays
Obviously, the vast majority of the data we have refers to laboratory or small-scale tests. The next step in phytomelatonin research is mandatory medium and large-scale agronomic trials to verify the previous lab assays. In this scope, there are problems to be solved since the dosages and effectiveness of the treatments must be adapted to each type of crop and to the variability of the agricultural environment and the species or varieties studied. Limitations such as the form of dosage, whether it is in greenhouses or in the open field, and whether it is in irrigated or rainfed crops, would be among the first challenges to overcome.
However, there are data from field tests with very positive results about the protective role of phytomelatonin in various situations of stress and multi-stress, especially due to water scarcity and salinity. The field trials of crops such as apple, citrus, cherry, grapes, peach, plums, pistachio and some berries in terms of fruit tree are noteworthy, also in banana. Very attractive data on broccoli, lettuce, watermelon and potato have been obtained; also data on cereals, especially rice, corn and wheat. Some interesting data on legumes such as soybeans and beans. Much more data is available in greenhouse crops, especially in tomato, strawberry, eggplant, cucumber, aromatic, and other plants.
Foliar fertigation seems to be the best technique to apply due to the amphiphilic nature of phytomelatonin that makes it pass through the leaf cuticles without the help of wetting, which is a great saving. However, it has been found that the application of phytomelatonin through droppers together with root fertilization or through the soil, although it involves higher doses due to losses, a great improvement in the rhizobiome has been observed, pointing to a better water and nutrient economy, mainly nitrogen and phosphorus.
As for the valuation of costs and benefits in crops, for now, there are no real agronomically indicative data. Although it should be noted that synthetic melatonin treatments are very economical because the chemical industry has highly optimized the chemical synthesis process, and with very low costs; We could say that the cost would surely be much lower than that of other agrochemicals commonly used in most crops. In addition, the doses to be used, according to the large number of conclusive data, are not too high, generally between 4 and 24 ppm, and between 1–3 treatments, depending on the problem to be improved. In general, we can anticipate that the cost/benefit balance would be clearly inclined towards the benefits, often multiple, both in quality and quantity of crop production.
Synthetic Melatonin vs. Rich-phytomelatonin extracts
Since pure melatonin is restricted for direct agronomic use, phytomelatonin-rich extracts offer a feasible alternative as plant biostimulants. According to EU Regulation 2019/1009, biostimulants are defined by their capacity to improve plant productivity through the emergent properties of their biological constituents. These products are classified into microbial (Subtype A) and non-microbial (Subtype B) categories. The latter includes plant or fungal extracts (Component Material Category 2) processed through physical means such as milling, centrifugation, or supercritical CO2 extraction, excluding chemical synthesis. This classification provides a clear pathway for developing phytomelatonin-based products that enhance crop resilience while meeting modern regulatory standards.
The goal of obtaining phytomelatonin-rich plants as CMC is ambitious, because phytomelatonin levels in plants are typically very low, usually less than 10 ng per gram of plant material. An extensive classification of numerous plants by their phytomelatonin content can be consulted [5,54,55]. There are currently several products on the market from plants that have a high phytomelatonin content. Most of them are marketed as food supplements, generally as products beside jet-lag or other sleep problems. Our recent publication presents the details of these products that could serve as a basis for being used as biostimulants in crops [56]. At the University of Murcia (Spain) has been developed a product focused on its use as a biostimulant by crops. These are aromatic-medicinal plants (AMPs) that after an exclusive elicitation process over-produce a large amount of phytomelatonin. From this raw material, phytomelatonin-rich extracts can be obtained by purification techniques. Our phytomelatonin-rich extracts have been checked in several crops such as tomato, lettuce, cereals, broccoli, and others, with excellent results improving germination, rooting and plant growth against stressors. Also, as postharvest improved, delaying senescence and decreasing the spoilage of fruits and vegetables during storage by reducing oxidative damage and/or pathogen diseases. This product rich in phytomelatonin from cultivated plants does not have the legal restrictions of pure active substances and can be used as applied as a natural biostimulant source in the development of eco-friendly phytochemical broths in accordance with EU 2019/1009 legislation.
Fortunately, the legislation on biostimulants for crops in developed countries has opted for the lower use of active chemical substances, looking more at the use of products obtained from biological material. In the case of phytomelatonin, although we can say that the application in crops of extracts rich in phytomelatonin is in its initiatory stages, it is to be expected that the commitment to plants rich in phytomelatonin will give way to other products rich in melatonin from other biological materials such as fungi, algae or bacteria in culture. The controlled use of these products in crop improvement can provide R+D in the face of the already legible environment changes that can cause several stressful situations in crops in many places around the world.
Acknowledgement:
Funding Statement: The authors received no specific funding for this study.
Author Contributions: Conceptualization, Marino B. Arnao; methodology, Marino B. Arnao, Giuseppe Mannino, Leyla Parra-Almuna; software, Manuela Giraldo-Acosta, Antonio Cano; validation, Soundouss Kaabi, Antonio Cano, Josefa Hernández-Ruiz; investigation, Manuela Giraldo-Acosta, Antonio Cano, Josefa Hernández-Ruiz; writing—original draft preparation, Marino B. Arnao, Antonio Cano, Josefa Hernández-Ruiz; writing—review and editing, Marino B. Arnao, Giuseppe Mannino, Leyla Parra-Almuna; visualization, Marino B. Arnao, Soundouss Kaabi; funding acquisition, Marino B. Arnao. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: Data available on request from the authors.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Lerner AB, Case JD, Takahashi Y, Lee TH, Mori W. Isolation of melatonin, the pineal gland factor that lightens melanocytes. J Am Chem Soc. 1958;80(10):2587. doi:10.1021/ja01543a060. [Google Scholar] [CrossRef]
2. Hattori A, Migitaka H, Iigo M, Itoh M, Yamamoto K, Ohtani-Kaneko R, et al. Identification of melatonin in plants and its effects on plasma melatonin levels and binding to melatonin receptors in vertebrates. Biochem Mol Biol Int. 1995;35(3):627–34. [Google Scholar]
3. Kolar J, Machackova I, Illnerova H, Prinsen E, van Dongen W, van Onckelen H. Melatonin in higher plant determined by radioimmunoassay and liquid chromatography-mass spectrometry. Biol Rhythm Res. 1995;26:406–9. [Google Scholar]
4. Dubbels R, Reiter RJ, Klenke E, Goebel A, Schnakenberg E, Ehlers C, et al. Melatonin in edible plants identified by radioimmunoassay and by high performance liquid chromatography-mass spectrometry. J Pineal Res. 1995;18(1):28–31. doi:10.1111/j.1600-079x.1995.tb00136.x. [Google Scholar] [CrossRef]
5. Arnao MB. Phytomelatonin: discovery, content, and role in plants. Adv Bot. 2014;2014:815769. doi:10.1155/2014/815769. [Google Scholar] [CrossRef]
6. Reiter RJ. The melatonin rhythm: both a clock and a calendar. Experientia. 1993;49(8):654–64. doi:10.1007/BF01923947. [Google Scholar] [CrossRef]
7. Cruz-Sanabria F, Bruno S, Crippa A, Frumento P, Scarselli M, Skene DJ, et al. Optimizing the time and dose of melatonin as a sleep-promoting drug: a systematic review of randomized controlled trials and dose-response meta-analysis. J Pineal Res. 2024;76(5):e12985. doi:10.1111/jpi.12985. [Google Scholar] [CrossRef]
8. Kaabi S, El Bouzdoudi B, El Kbiach ML, Cano A, Hernández-Ruiz J, Arnao MB. Content of phytomelatonin in acorns (Quercus sp.) in its possible use as a phytogenic in animal nutrition. Processes. 2025;13(7):2202. doi:10.3390/pr13072202. [Google Scholar] [CrossRef]
9. Galano A, Tan DX, Reiter RJ. On the free radical scavenging activities of melatonin’s metabolites, AFMK and AMK. J Pineal Res. 2013;54(3):245–57. doi:10.1111/jpi.12010. [Google Scholar] [CrossRef]
10. Ameen M, Zafar A, Mahmood A, Zia MA, Kamran K, Javaid MM, et al. Melatonin as a master regulatory hormone for genetic responses to biotic and abiotic stresses in model plant Arabidopsis thaliana: a comprehensive review. Funct Plant Biol. 2024;51:FP23248. doi:10.1071/FP23248. [Google Scholar] [CrossRef]
11. Ikram M, Mehran M, Rehman HU, Ullah S, Bakhsh MZM, Tahira M, et al. Mechanistic review of melatonin metabolism and signaling pathways in plants: biosynthesis, regulation, and roles under abiotic stress. Plant Stress. 2024;14:100685. doi:10.1016/j.stress.2024.100685. [Google Scholar] [CrossRef]
12. Khan S, Saify S, Sofo A, Khan NA. The mechanisms of melatonin action in shielding photosynthesis during heat stress. CABI Rev. 2024;19(1):1–16. doi:10.1079/cabireviews.2024.0027. [Google Scholar] [CrossRef]
13. Obaid Saeed Alfalahi B, Lamdjad I, Alnujaifi M, Atallah Alheety N, Qayyum A. Melatonin as a protective agent against environmental stresses: a review into its molecular regulation in plants. OBM Genet. 2024;8(2):1–10. doi:10.21926/obm.genet.2402242. [Google Scholar] [CrossRef]
14. Dong W. Melatonin: a key player in alleviating heavy metal stress in plants—current insights and future directions. Hortic Plant J. 2026;12(1):36–48. doi:10.1016/j.hpj.2025.05.005. [Google Scholar] [CrossRef]
15. Dhole AM, Shelat HN. Phytomelatonin: a plant hormone for management of stress. J Anal Pharm Res. 2018;7(2):188–90. doi:10.15406/japlr.2018.07.00224. [Google Scholar] [CrossRef]
16. Mehrzadi S, Sheibani M, Koosha F, Alinaghian N, Pourhanifeh MH, Tabaeian SAP, et al. Protective and therapeutic potential of melatonin against intestinal diseases: updated review of current data based on molecular mechanisms. Expert Rev Gastroenterol Hepatol. 2023;17(10):1011–29. doi:10.1080/17474124.2023.2267439. [Google Scholar] [CrossRef]
17. Galano A, Reiter RJ. Melatonin and its metabolites vs. oxidative stress: from individual actions to collective protection. J Pineal Res. 2018;65(1):e12514. doi:10.1111/jpi.12514. [Google Scholar] [CrossRef]
18. Álvarez-López AI, Cruz-Chamorro I, Lardone PJ, Bejarano I, Aspiazu-Hinostroza K, Ponce-España E, et al. Melatonin, an antitumor necrosis factor therapy. J Pineal Res. 2025;77(1):e70025. doi:10.1111/jpi.70025. [Google Scholar] [CrossRef]
19. Arnao MB, Hernández-Ruiz J. Melatonin against environmental plant stressors: a review. Curr Protein Pept Sci. 2021;22(5):413–29. doi:10.2174/1389203721999210101235422. [Google Scholar] [CrossRef]
20. Aghdam MS, Arnao MB. Phytomelatonin: from intracellular signaling to global horticulture market. J Pineal Res. 2024;76(5):e12990. doi:10.1111/jpi.12990. [Google Scholar] [CrossRef]
21. Arnao MB, Cano A, Hernández-Ruiz J. Phytomelatonin: an unexpected molecule with amazing performances in plants. J Exp Bot. 2022;73(17):5779–800. doi:10.1093/jxb/erac009. [Google Scholar] [CrossRef]
22. Cano A, Hernández-Ruiz J, Arnao MB. Role of exogenous melatonin in plant biotechnology: physiological and applied aspects. Crit Rev Plant Sci. 2024;43(6):395–404. doi:10.1080/07352689.2024.2394002. [Google Scholar] [CrossRef]
23. Wang L, Tanveer M, Wang H, Arnao MB. Melatonin as a key regulator in seed germination under abiotic stress. J Pineal Res. 2024;76(1):e12937. doi:10.1111/jpi.12937. [Google Scholar] [CrossRef]
24. Moustafa-Farag M, Elkelish A, Dafea M, Khan M, Arnao MB, Abdelhamid MT, et al. Role of melatonin in plant tolerance to soil stressors: salinity, pH and heavy metals. Molecules. 2020;25(22):5359. doi:10.3390/molecules25225359. [Google Scholar] [CrossRef]
25. Moustafa-Farag M, Mahmoud A, Arnao MB, Sheteiwy MS, Dafea M, Soltan M, et al. Melatonin-induced water stress tolerance in plants: recent advances. Antioxidants. 2020;9(9):809. doi:10.3390/antiox9090809. [Google Scholar] [CrossRef]
26. Arnao MB, Hernández-Ruiz J. Melatonin as a regulatory hub of plant hormone levels and action in stress situations. Plant Biol. 2021;23(Suppl 1):7–19. doi:10.1111/plb.13202. [Google Scholar] [CrossRef]
27. Ahmad J, Hayat F, Khan U, Ahmed N, Li J, Ercisli S, et al. Melatonin: a promising approach to enhance abiotic stress tolerance in horticultural plants. S Afr J Bot. 2024;164:66–76. doi:10.1016/j.sajb.2023.10.045. [Google Scholar] [CrossRef]
28. Moustafa-Farag M, Almoneafy A, Mahmoud A, Elkelish A, Arnao MB, Li L, et al. Melatonin and its protective role against biotic stress impacts on plants. Biomolecules. 2020;10(1):54. doi:10.3390/biom10010054. [Google Scholar] [CrossRef]
29. Hernández-Ruiz J, Giraldo-Acosta M, El Mihyaoui A, Cano A, Arnao MB. Melatonin as a possible natural anti-viral compound in plant biocontrol. Plants. 2023;12(4):781. doi:10.3390/plants12040781. [Google Scholar] [CrossRef]
30. Mao J, Nuruzzaman Manik SM, Shi S, Chao J, Jin Y, Wang Q, et al. Mechanisms and physiological roles of the CBL-CIPK networking system in Arabidopsis thaliana. Genes. 2016;7(9):62. doi:10.3390/genes7090062. [Google Scholar] [CrossRef]
31. Chen Z, Xie Y, Gu Q, Zhao G, Zhang Y, Cui W, et al. The AtrbohF-dependent regulation of ROS signaling is required for melatonin-induced salinity tolerance in Arabidopsis. Free Radic Biol Med. 2017;108:465–77. doi:10.1016/j.freeradbiomed.2017.04.009. [Google Scholar] [CrossRef]
32. Arnao MB, Hernández-Ruiz J. Role of melatonin to enhance phytoremediation capacity. Appl Sci. 2019;9(24):5293. doi:10.3390/app9245293. [Google Scholar] [CrossRef]
33. Hasan MK, Ahammed GJ, Sun S, Li M, Yin H, Zhou J. Melatonin inhibits cadmium translocation and enhances plant tolerance by regulating sulfur uptake and assimilation in Solanum lycopersicum L. J Agric Food Chem. 2019;67(38):10563–76. doi:10.1021/acs.jafc.9b02404. [Google Scholar] [CrossRef]
34. Ahammed GJ, Li X. Melatonin-induced detoxification of organic pollutants and alleviation of phytotoxicity in selected horticultural crops. Horticulturae. 2022;8(12):1142. doi:10.3390/horticulturae8121142. [Google Scholar] [CrossRef]
35. Ahmad R, Alsahli AA, Alansi S, Altaf MA. Exogenous melatonin confers drought stress by promoting plant growth, photosynthetic efficiency and antioxidant defense system of pea (Pisum sativum L.). Sci Hortic. 2023;322:112431. doi:10.1016/j.scienta.2023.112431. [Google Scholar] [CrossRef]
36. Gajardo HA, González-Villagra J, Arce-Johnson P. Melatonin and grain legume crops: opportunities for abiotic stress tolerance enhancement and food sustainability. Plants. 2025;14(21):3324. doi:10.3390/plants14213324. [Google Scholar] [CrossRef]
37. Yang X, Chen J, Ma Y, Huang M, Qiu T, Bian H, et al. Function, mechanism, and application of plant melatonin: an update with a focus on the cereal crop, barley (Hordeum vulgare L.). Antioxidants. 2022;11(4):634. doi:10.3390/antiox11040634. [Google Scholar] [CrossRef]
38. Muhammad I, Ahmad S, Shen W. Melatonin-mediated molecular responses in plants: enhancing stress tolerance and mitigating environmental challenges in cereal crop production. Int J Mol Sci. 2024;25(8):4551. doi:10.3390/ijms25084551. [Google Scholar] [CrossRef]
39. Hernández-Ruiz J, Ruiz-Cano D, Giraldo-Acosta M, Cano A, Arnao MB. Melatonin in Brassicaceae: role in postharvest and interesting phytochemicals. Molecules. 2022;27(5):1523. doi:10.3390/molecules27051523. [Google Scholar] [CrossRef]
40. Wei J, Li DX, Zhang JR, Shan C, Rengel Z, Song ZB, et al. Phytomelatonin receptor PMTR1-mediated signaling regulates stomatal closure in Arabidopsis thaliana. J Pineal Res. 2018;65(2):e12500. doi:10.1111/jpi.12500. [Google Scholar] [CrossRef]
41. Kouser R, Zaidi S, Arif Y, Hasan SA, Faizan M, Hayat S. Melatonin biosynthesis, signaling regulation, crosstalk with phytohormones and its role in defence mechanism and ROS scavenging. Braz J Bot. 2025;48(1):24. doi:10.1007/s40415-025-01068-5. [Google Scholar] [CrossRef]
42. Qian Y, Tan DX, Reiter RJ, Shi H. Comparative metabolomic analysis highlights the involvement of sugars and glycerol in melatonin-mediated innate immunity against bacterial pathogen in Arabidopsis. Sci Rep. 2015;5:15815. doi:10.1038/srep15815. [Google Scholar] [CrossRef]
43. Zhao H, Xu L, Su T, Jiang Y, Hu L, Ma F. Melatonin regulates carbohydrate metabolism and defenses against Pseudomonas syringae pv. tomato DC3000 infection in Arabidopsis thaliana. J Pineal Res. 2015;59(1):109–19. doi:10.1111/jpi.12245. [Google Scholar] [CrossRef]
44. Zhao L, Chen L, Gu P, Zhan X, Zhang Y, Hou C, et al. Exogenous application of melatonin improves plant resistance to virus infection. Plant Pathol. 2019;68(7):1287–95. doi:10.1111/ppa.13057. [Google Scholar] [CrossRef]
45. Tiwari RK, Lal MK, Kumar R, Mangal V, Altaf MA, Sharma S, et al. Insight into melatonin-mediated response and signaling in the regulation of plant defense under biotic stress. Plant Mol Biol. 2022;109(4–5):385–99. doi:10.1007/s11103-021-01202-3. [Google Scholar] [CrossRef]
46. Zeng W, Mostafa S, Lu Z, Jin B. Melatonin-mediated abiotic stress tolerance in plants. Front Plant Sci. 2022;13:847175. doi:10.3389/fpls.2022.847175. [Google Scholar] [CrossRef]
47. Eke P, Mabou LCN, Tchonang DN, Kouokap LRK, Youmbi DY, Dinango VN, et al. Explicating the role of melatonin in the mitigation of fungal diseases in plants. In: Melatonin in plants: a pleiotropic molecule for abiotic stresses and pathogen infection. Singapore: Springer; 2023. p. 155–76. doi:10.1007/978-981-99-6741-4_9. [Google Scholar] [CrossRef]
48. Zhang S, Liu S, Zhang J, Reiter RJ, Wang Y, Qiu D, et al. Synergistic anti-oomycete effect of melatonin with a biofungicide against oomycetic black shank disease. J Pineal Res. 2018;65(2):e12492. doi:10.1111/jpi.12492. [Google Scholar] [CrossRef]
49. Giraldo-Acosta M, Arnao MB. Melatonin, a promising natural crop safener. Outlooks Pest Manag. 2024;35(3):115–9. doi:10.1564/v35_jun_06. [Google Scholar] [CrossRef]
50. Zhang X, Schmidt R. The impact of growth regulators on the α-tocopherol status in water-stressed Poa pratensis L. Int Turfgrass Soc Res J. 1997;8:1364–73. [Google Scholar]
51. Rouphael Y, Colla G. Toward a sustainable agriculture through plant biostimulants: from experimental data to practical applications. Agronomy. 2020;10(10):1461. doi:10.3390/agronomy10101461. [Google Scholar] [CrossRef]
52. Khoulati A, Ouahhoud S, Taibi M, Ezrari S, Mamri S, Merah O, et al. Harnessing biostimulants for sustainable agriculture: innovations, challenges, and future prospects. Discov Agric. 2025;3(1):56. doi:10.1007/s44279-025-00177-9. [Google Scholar] [CrossRef]
53. Cano A, Giraldo-Acosta M, García-Sánchez S, Hernández-Ruiz J, B Arnao M. Effect of melatonin in broccoli postharvest and possible melatonin ingestion level. Plants. 2022;11(15):2000. doi:10.3390/plants11152000. [Google Scholar] [CrossRef]
54. Arnao MB, Hernández-Ruiz J. The potential of phytomelatonin as a nutraceutical. Molecules. 2018;23(1):238. doi:10.3390/molecules23010238. [Google Scholar] [CrossRef]
55. Cheng G, Ma T, Deng Z, Gutiérrez-Gamboa G, Ge Q, Xu P, et al. Plant-derived melatonin from food: a gift of nature. Food Funct. 2021;12(7):2829–49. doi:10.1039/d0fo03213a. [Google Scholar] [CrossRef]
56. Cano A, Hernández-Ruiz J, Arnao MB. Current state of the natural melatonin: the phytomelatonin market. Melatonin Res. 2024;7(3):242–8. doi:10.32794/mr112500179. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools