 Open Access
Open Access
ARTICLE
Low-Temperature Stress Effects on Germination, Antioxidant Enzyme Activity, and Gene Expression in Hybrid and Conventional Rice from Southern China
1 College of Plant Science and Technology, Hunan Biological Electromechanical Vocational Technical College, Changsha, China
2 College of Agriculture and Horticulture, Huaihua Vocational and Technical College, Huaihua, China
3 College of Agronomy, Hunan Agricultural University, Changsha, China
4 Yuelu Mountain Laboratory of Hunan Province, Hunan Agricultural University, Changsha, China
* Corresponding Authors: Cheng Qu. Email: ; Yue Wang. Email:
(This article belongs to the Special Issue: Plant Responses to Abiotic Stress Mechanisms)
Phyton-International Journal of Experimental Botany 2026, 95(3), 10 https://doi.org/10.32604/phyton.2026.077734
Received 16 December 2025; Accepted 05 February 2026; Issue published 31 March 2026
Abstract
To explore the physiological mechanism of rice seeds’ tolerance to cold damage stress, the hybrid rice variety H-You 518 and the conventional rice variety Zhongjiazao 17 were used as experimental materials. A low-temperature cold damage treatment (4°C) and a normal germination treatment (25°C) were used to measure seed vitality, antioxidant enzyme activity, soluble sugar content, soluble protein content, α-amylase activity, endopeptidase, and related gene expression levels. The results showed that low temperature inhibited seed vitality and germination ability, and after restoring normal growth conditions, the activities of POD and CAT, as well as the content of MDA in the seedlings, were significantly increased. Between these two rice varieties, the seed vitality and antioxidant enzyme activity of the Zhongjiazao 17 conventional rice variety were more sensitive to low-temperature stress. During seed germination, both the total soluble sugar and soluble protein levels showed a trend of first decreasing, then increasing, and finally decreasing under low-temperature cold damage. The total soluble sugar content of H-You 518 was higher than that of Zhongjiazao 17. Through correlation analysis, it was found that under low-temperature damage, the activities of α-amylase and endopeptidases were significantly positively correlated with the soluble protein content, with correlation coefficients of 0.55 and 0.61, respectively, indicating that the trends of these changes were basically consistent. During the germination process, the expression levels of the α-AMY and EP genes gradually increased, but low-temperature stress reduced the expression levels of these genes. In summary, low-temperature cold damage conditions reduced the activities of α-amylase and endopeptidases, thereby slowing down the production of soluble sugars and soluble proteins. Moreover, the activities of α-amylase and endopeptidases were regulated by the α-AMY and EP genes. This study provides a reference for the physiological mechanism analysis of rice seed germination in low temperatures and under cold stress.Keywords
China is the world’s largest producer of rice, accounting for approximately 50% of global rice production [1,2]. The southern region of China is located in the tropical and subtropical monsoon climate zone, with abundant light energy and heat, and is the most important double-cropping rice production area in China. In recent years, frequent catastrophic weather events and global warming have caused considerable abiotic stress. Early rice germination in southern China is often affected by low temperatures below 15°C, such as “late spring cold” or “cold dew wind”, resulting in slow or obstructed seed germination, irregular emergence, and serious damage to yield and quality. This has gradually become one of the main reasons for safe rice production in southern rice regions [3,4]. Therefore, enhancing the low-temperature resistance of rice, exploring the defense mechanisms during seed germination, and breeding varieties with strong cold resistance have become popular research topics for many scholars.
Low temperature is one of the main stress factors that plants suffer from during their growth and development, which can easily cause rice seedlings to form dead or stiff seedlings, and even affect the normal growth, development, and yield of rice. Many researchers have conducted in-depth studies on the breeding of cold-tolerant varieties [5,6,7], the effects of low temperature on plant physiology and biochemistry [8,9,10], and hormone regulation of plant response to low temperature [11,12,13]. Studies have shown that seeds become active during the early stages of germination, and the sugars required to maintain their life activities first come from soluble sugars stored in cells to provide energy for embryo growth and various life activities. After seed germination, stored substances such as starch begin to decompose into soluble sugars, which are transported to the embryo for growth and utilization [14]. Alpha-amylase in seeds is the main enzyme that hydrolyzes starch. A low temperature can reduce the activity of a-amylase, resulting in a decrease in soluble sugar content. In the early stages, cells will actively adapt to this change in order to provide energy for the hydrolysis of starch in an energy-saving way [15]. Soluble proteins provide energy and material basis for metabolic activities during seed germination, participate in various biochemical reactions, and promote the formation of embryo morphology. On the one hand, soluble proteins are continuously hydrolyzed and stored by proteases. On the other hand, seed germination is accompanied by a large amount of protein expression, so the content of soluble proteins continues to increase during the seed germination process. A low temperature has an inhibitory effect on protein hydrolysis and expression, thus reducing the synthesis rate of soluble proteins. In addition, studies have shown that low-temperature cold stress can slow down the fluidity of cell membranes and the speed of substances entering and leaving through these membranes, leading to plant cell osmotic stress [16] Although a low temperature can increase the content of some osmoregulatory substances, such as soluble sugars and soluble proteins, it is crucial to increase cell fluid concentration, enhance cell water retention capacity, and prevent tissues from losing too much water and dying. However, the alleviating effect of osmoregulatory substances on water stress in plant tissues under dehydration is limited. Rice needs to absorb and transport water upwards through its roots to maintain the aboveground tissues. The water metabolism balance under low-temperature stress can slow down the growth rate of roots, weaken their ability to absorb water, and cause water metabolism imbalance in rice seedlings [17]. Han et al. [18] found that different rice varieties are affected by their own physiological differences, resulting in different minimum temperatures that seeds can withstand during germination. Generally, low temperatures below 5°C can reduce the germination rate of rice seeds. Duan Licheng et al. [19] suggested that under low-temperature stress, rice is more sensitive to low temperatures when sown using seeds with a germination degree of bud valley than using seeds with broken hulls. Seedlings of an older age are more sensitive to low temperatures than seedlings of a younger age because the content of malondialdehyde increases, while the content of proline gradually decreases, with increasing seedling age. Shinkawa et al. [20] found that exogenous application of ABA can enhance the cold resistance of rice seedlings and suspended cells during low-temperature damage.
Currently, studies on the effects of low temperature on seed germination in maize, soybean, and wheat have been relatively extensive, with a primary focus on seed vigor and physiological-biochemical indicators during the seedling stage. However, there remains a scarcity of reports addressing the changes in amylase activity, protease activity, and associated gene expression during rice seed germination under low-temperature stress [21,22,23]. This study used the hybrid rice variety H-You 518 and the conventional rice variety Zhongjiazao 17 as experimental materials to investigate the effects of low-temperature cold damage on seed germination characteristics, as well as the changes in total soluble sugar content, soluble protein content, α-amylase and endo-peptidase activities, and related gene expression levels during seed germination. The mechanism of rice response to low-temperature cold damage was clarified, providing theoretical support for the application and cultivation of low-temperature-tolerant rice varieties in double-season early rice live broadcasting cultivation.
The two rice varieties used in this study, H-You 518 (a hybrid indica rice) and Zhongjiazao 17 (a conventional indica rice), were selected based on their agronomic significance and representativeness within the target region of Southern China, particularly Hunan Province, a core area of double-cropping rice production. Both varieties were provided by the Rice Science Research Institute of Hunan Agricultural University, ensuring the authenticity and local relevance of the germplasm. The experiment was conducted at the Rice and Rapeseed Disease Resistance Breeding Laboratory of the Life Science Building of Hunan Agricultural University.
Experiment 1: Healthy, plump, uniformly sized, and pest-free seeds were selected and disinfected with a 10% sodium hypochlorite solution. Two treatments were designed for the experiment. (1) Standard germination as the control (CK): after soaking the seeds in distilled water at 37°C for 12 h, they were cultured on germination paper at 25°C. (2) Low-temperature cold damage treatment (CS): after soaking the seeds in distilled water at 4°C for 12 h, they were incubated on germination paper at 4°C for two days before being returned to the standard germination mode. Each treatment was established with four biological replicates, each containing 100 seeds. The seeds were placed in an artificial climate incubator under 12 h of light and 12 h of darkness, with 10,000 LX of light. The seeds were observed and recorded daily. The seeds were placed in the incubator until the seventh day, when they were taken out and frozen in liquid nitrogen; each biological replicate were analyzed in four technical replicates.
Experiment 2: This experiment explored the mechanism of intrinsic material changes during seed swelling process. The seed treatment was the same as in Experiment 1. Each treatment was established with four biological replicates, each containing 100 seeds. The seeds were neatly placed in a germination box covered with germination paper, and treated with distilled water as the swelling solution under dark conditions. The embryos and endosperms were taken at 0, 6, 12, 24, 36, 48, 72, and 96 h of seed swelling and germination, and each biological replicate were analyzed in four technical replicates.
2.3 Measurement Items and Methods
After four days of seed germination, the germination potential was investigated and calculated. On the seventh day, the germination rate was investigated and calculated, and the root length and bud length of 10 plants were measured. Finally, normal seedlings were sterilized at 105°C for 30 min and dried at 80°C until reaching a constant weight, and the dry weight of the seedlings and roots was measured.
Germination potential (%) = number of normal seedlings within four days/number of tested seeds × 100%,
Germination rate (%) = number of normal seedlings within seven days/number of tested seeds × 100%,
Germination index = ∑Gt/Dt (where Gt represents the number of germinated seeds at time t and Dt represents the corresponding number of germinating days),
Vitality index = GI × S (where GI is the germination index and S is the dry weight of individual seedlings).
2.3.2 Determination of Antioxidant Activity and Lipid Peroxidation Content
Each sample was homogenized in a precooled mortar with 4 mL of 0.05 M phosphate buffer (pH 7.8). The resulting slurry was transferred to a 10 mL centrifuge tube, frozen for 20 min, and then centrifuged. The supernatant was collected for subsequent determination of malondialdehyde (MDA) content and enzyme activities.
Peroxidase (POD) activity was measured according to the guaiacol method [24]. An aliquot of the enzyme extract was added to 3 mL of reaction mixture containing 1.4 μL of guaiacol, 0.85 μL of 30% H2O2, and 0.1 mol/L of sodium phosphate buffer (pH 5.5). The increase in absorbance at 470 nm was recorded at 30 s intervals. POD activity was expressed as the change in absorbance per minute, with one unit (U) defined as an increase of 0.01 absorbance units per minute per gram of fresh weight (U·min−1·g−1 FW).
Catalase (CAT) activity was determined via UV spectrophotometry [25]. A 2.5 mL aliquot of reaction solution (prepared by mixing 25 mL of 0.1 mol/L H2O2 with 20 mL of 0.1 mol/L phosphate buffer, pH 7.0) was added to the enzyme extract. The decrease in absorbance at 240 nm was measured every 30 s for a total of five readings. CAT activity was calculated from the linear decrease in absorbance per minute. One unit of activity (U) was defined as a decrease of 0.01 absorbance units per minute per gram of fresh weight (U·min−1·g−1 FW).
The MDA content was quantified using the thiobarbituric acid (TCA-TBA) colorimetric method [26]. An aliquot of the supernatant was mixed with 5 mL of 10% trichloroacetic acid, homogenized, and centrifuged at 4000 rpm for 20 min. Then, 2 mL of the resulting supernatant was reacted with 2 mL of 0.6% TBA in a boiling water bath for 15 min, rapidly cooled, and centrifuged again. Absorbance of the final supernatant was measured at 450, 532, and 600 nm. MDA content was expressed as micromoles per gram of fresh weight (μmol·g−1 FW).
2.3.3 Determination of Soluble Sugar Content and Alpha-Amylase Activity
The total soluble sugar content was analyzed using the anthrone-sulfuric acid assay [27]. Approximately 0.1 g of dried sample was boiled in 7 mL of distilled water in a boiling water bath for 30 min, and the supernatant was collected after centrifugation. The extraction was repeated twice, and the combined supernatants were adjusted to a fixed volume. A 100 μL aliquot of the extract was reacted with 3 mL of freshly prepared 0.15% (w/v) anthrone-sulfuric acid reagent. After heating in a boiling water bath for 10 min and rapid cooling, absorbance was recorded at 620 nm. Ssugar content was e quantified against a glucose standard curve and expressed as glucose equivalents per gram dry weight (mg·g−1 DW).
α-Amylase activity was measured according to the 3,5-dinitrosalicylic acid (DNS) method [28]. Germinated seeds (0.5 g) were homogenized in 4 mL of ice-cold extraction buffer (0.1 M sodium citrate-phosphate buffer, pH 5.6, containing 1 mM CaCl2) and centrifuged at 12,000× g for 20 min at 4°C. The resulting supernatant was used as the crude enzyme extract. For β-amylase inactivation, 1 mL of the extract was heated at 70°C for 15 min and then rapidly cooled. After adding 1 mL of citrate buffer (pH 5.6) and pre-incubating the mixture was pre-incubated at 40°C for 15 min. The reaction was initiated by introducing 2 mL of pre-warmed 1% (w/v) soluble starch solution and allowed to proceed for 5 min at 40°C. The reaction was terminated by adding 4 mL of 0.4 M NaOH. An aliquot of the mixture was combined with DNS reagent, heated in for 5 min in a boiling water bath, cooled, and the absorbance was measured at 520 nm. Activity was calculated using a maltose standard curve and expressed as milligrams of maltose generated per minute per gram of fresh weight (mg·g−1 FW·min−1).
2.3.4 Determination of Endopeptidase Activity and Soluble Protein Content
Endopeptidase activity was assayed following the protocol described by Zhang et al. [29]. Endosperm tissue was homogenized in 4 mL of pre-cooled extraction buffer (50 mM Tris HCl, pH 7.5, containing 1 mM EDTA, 15 mM β-mercaptoethanol, 1% PVP) using a pre-chilled mortar. The homogenate was centrifuged at 15,000× g for 20 min at 4°C, and the supernatant was collected as the crude enzyme extract. The reaction mixture consisted of 0.2 mL of 200 mM acetate buffer (pH 5.2), 0.2 mL of 2% hemoglobin solution, and 0.2 mL of enzyme solution. The mixture was incubated in a constant-temperature water bath at 42°C for 1 h, and then the reaction was terminated by adding 0.8 mL of 7.5% trichloroacetic acid. The mixture was kept at 4°C for 30 min and then centrifuged at 10,000× g for 10 min. The absorbance of the supernatant measured at 278 nm. One unit of enzyme activity (U) was defined as an absorbance change of 0.001 at 278 nm per hour per gram of fresh weight sample (or expressed as ΔA278·g−1 FW·h−1).
Soluble protein concentration was determined using a modified Bradford assay [30]. A 20 μL aliquot of the crude enzyme extract was mixed with 3 mL of Coomassie Brilliant Blue G-250 reaction. A blank was prepared by substituting the enzyme extract with 20 μL of extraction buffer (pH 7.5). After incubation at room temperature for 2 min, absorbance was recorded at 595 nm. Protein concentration was quantified against a bovine serum albumin (BSA) standard curve and expressed as milligrams of protein per gram of fresh weight (mg·g−1 FW).
2.3.5 Analysis of Gene Expression Levels
The expression levels of the α-AMY (α-amylase) and EP (endopeptidase) genes were measured in the embryos, using the UBQ gene as an internal reference gene and Primer Premier 5.0 for primer design (Table 1). RNA was extracted using a kit manufactured by Shanghai Plomega Biological Products Co., Ltd. The quality of RNA was detected via 1% agarose gel electrophoresis, and its concentration and purity were determined using a microplate reader. cDNA synthesis and fluorescence quantitative analysis of the gene expression levels were performed using reagent kits. The qPCR reaction mixture (20 μL) consisted of 10 μL SYBR Green Premix Pro Taq HS, 0.8 μL each of forward and reverse primers, 2 μL cDNA template, and 6.4 μL nuclease-free water. The thermal cycling protocol was as follows: initial denaturation at 95°C for 1 min, followed by 40 cycles of denaturation at 95°C for 10 s and annealing/extension at 60°C for 30 s, ending with a melting curve analysis. Three technical replicates were included for each sample. Cycle threshold (Ct) values were obtained using the 2−ΔΔCt method.
Table 1: Primer sequences applied to qRT-PCR.
| Gene | GenBank Accession No. | Forward Primer Sequence (5′-3′) | Reverse Primer Sequence (5′-3′) | Amplicon |
|---|---|---|---|---|
| UBQ | LOC_Os03g13170 | CGCAAGAAGAAGTGTGGTCA | GGGAGATAACAACGGAAGCA | 102 |
| α-AMY | LOC_Os08g36910 | TCCCTTTCGGTCCTCATCG | CGCGTCCAGATCGTACAGC | 115 |
| EP | LOC_Os05g10640 | TGGTGGCTGCTTTCATGGC | CGCTTGTTGTGCTCGTGGAT | 108 |
The experimental data were statistically analyzed using Excel 2007 software. Variance analysis and correlation analysis (n = 64) were conducted using DPS7.05 for significance testing. Duncan’s new complex difference method was used to analyze the significant differences between treatments (α = 0.05), and GraphPad Prism 8.0 was used for plotting.
3.1 Effects of Low Temperature on Rice Seed Vitality
It was observed that low-temperature cold damage during the germination period has an inhibitory effect on the germination of rice seeds, which is manifested by the inhibition of root and shoot growth, and a decrease in seedling emergence ability (Table 2). Compared with the standard germination conditions, the germination potential, germination rate, germination index, and vitality index of rice seeds significantly decreased after the low-temperature cold damage, decreasing by 19.5%, 17.5%, 22.1%, and 29.3%, respectively, for H-You 518, and by 88.6%, 71.9%, 81.4%, and 96%, respectively, for Zhongjiazao 17. Under the low-temperature cold damage conditions, the germination index of Zhongjiazao 17 decreased more significantly, while H-You 518 was able to maintain the germination ability of the seeds well.
Table 2: Effect of cold damage on seed germination of different rice varieties.
| Variety | Treatment | Root Length/cm | Germinal Length/cm | Germination Energy/% | Germination Rate/% | Germination Index | Vital Index |
|---|---|---|---|---|---|---|---|
| H-You 518 | CK | 7.58 ± 0.21a | 6.82 ± 0.08a | 85.33 ± 1.33a | 89.33 ± 0.88a | 43.53 ± 0.53a | 10.17 ± 0.49a |
| CS | 6.96 ± 0.2a | 6.78 ± 0.16a | 68.67 ± 3.18b | 73.67 ± 2.03b | 35.49 ± 1.29b | 7.19 ± 0.51b | |
| Zhongjiazao 17 | CK | 6.65 ± 0.52a | 5.39 ± 0.06a | 72.01 ± 2.52a | 79.33 ± 1.45a | 36.54 ± 1.68a | 6.23 ± 0.83a |
| CS | 5.93 ± 0.24b | 5.32 ± 0.53a | 7.67 ± 0.67b | 22.33 ± 2.41b | 6.79 ± 1.03b | 0.25 ± 0.11b |
3.2 Effects of Low Temperature on Antioxidant Enzyme Activities and Lipid Peroxidation in Rice Seedlings
There were significant differences in antioxidant enzyme activities and lipid peroxidation in rice seedlings after the low-temperature cold damage treatment. The POD and CAT activities and the MDA content of rice seedlings were measured (Fig. 1). Compared with the standard germination conditions, the POD and CAT activities and the MDA content of the two varieties increased by 6% and 16.9%, 9.9% and 14.1%, and 46.9% and 19.8%, respectively, after the low-temperature cold damage. Under the same conditions, the POD and CAT activities in the rice seedlings of H-You 518 were higher, while the MDA content was slightly lower, than those of Zhongjiazao 17.
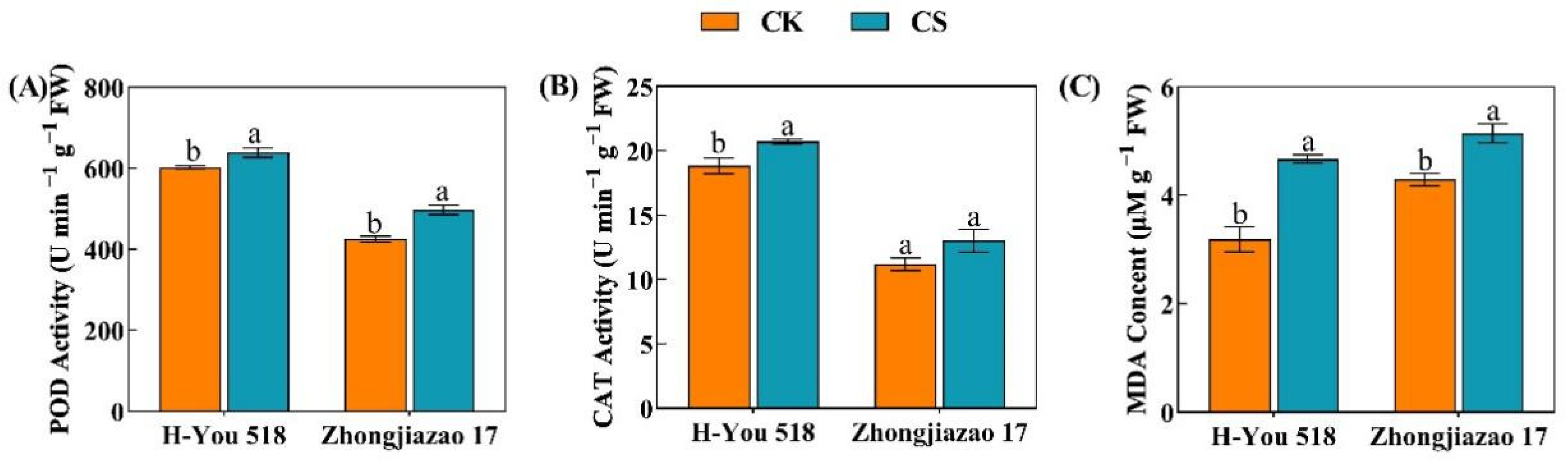
Figure 1: Effect of cold damage on antioxidant enzyme activities and lipid peroxidation in seedlings of different rice cultivars. In the figure, (A) represents peroxidase activity; (B) represents catalase activity; and (C) represents malondialdehyde content. Means ± SEs with different letters denote significant statistical differences (p < 0.05).
3.3 Effects of Low Temperature on Total Soluble Sugar Content during Rice Seed Germination Process
During seed germination, the prestored soluble sugars inside rice seeds are first consumed to provide energy for embryo growth and various life activities. After seed germination, stored substances such as starch begin to decompose into soluble sugars, which are transported to the embryo for growth and utilization. The total soluble sugar content of the two rice varieties showed a trend of first increasing, then decreasing, and finally increasing again under the standard germination and low-temperature cold damage conditions (Fig. 2). In the control group, the soluble sugar content at 6 h, 12 h, and 24 h was 1.85, 1.87, and 1.85 times higher than that at 0 h, respectively. Due to the decrease in starch hydrolysis efficiency caused by the low temperature, the increase in soluble sugar content was affected. The soluble sugar content at 6 h, 12 h, and 24 h after the low-temperature treatment was 1.45, 1.57, and 1.64 times higher than that at 0 h, respectively. However, there were differences in the changes in soluble sugar content between the hybrid and conventional rice varieties over time. During the low-temperature treatment, the total soluble sugar content of H-You 518 rapidly increased and reached its maximum value of 26.1% at 36 h, while that of Zhongjiazao 17 steadily increased, reaching its maximum value of 15.2% after 48 h. It seems that the inhibition of starch hydrolysis in the hybrid rice variety under low-temperature stress is lower than that in the conventional rice variety. After 72 h of low-temperature treatment, the total soluble sugar content of the two rice varieties sharply decreased, but at 96 h, the total soluble sugar content began to increase. After 96 h, the total soluble sugar content of H-You 518 reached the control level, while that of Zhongjiazao 17 only recovered to 30.63% of the control level.
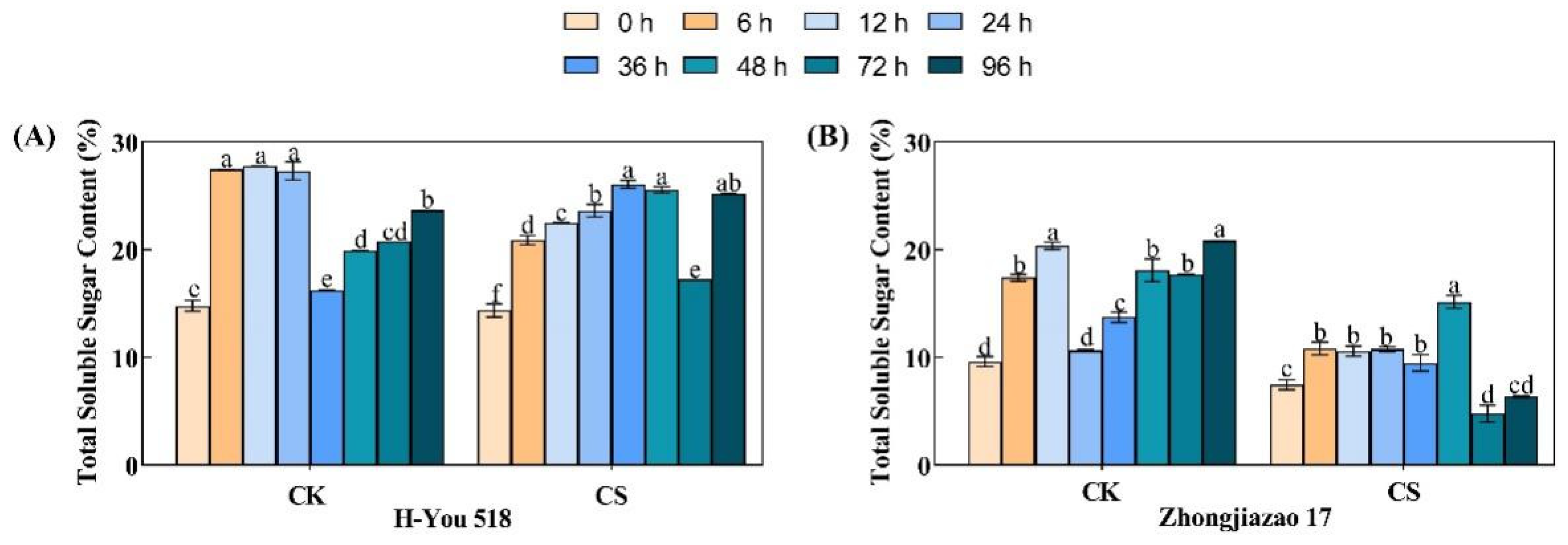
Figure 2: Changes in total soluble sugar content in different rice cultivars during seed germination after low-temperature treatment. In the figure, (A) represents total soluble sugar content of H-You 518; and (B) represents total soluble sugar content of Zhongjiazao 17. Means ± SEs with different letters denote significant statistical differences (p < 0.05).
3.4 Effects of Low Temperature on Soluble Protein Content during Rice Seed Germination Process
During seed germination, the storage proteins in rice seedlings are soluble; that is, they are partially hydrolyzed into water-soluble proteins with smaller molecular weights under the action of protease A. According to Fig. 3, the soluble protein content of the two rice varieties showed a gradually increasing trend during seed germination. The content of soluble proteins in the control group at 12 h, 24 h, and 36 h was 2.29, 2.46, and 2.75 times that at 0 h, respectively. Due to the decrease in protease activity caused by the low-temperature cold damage treatment, the content of soluble proteins was reduced; at 12 h, 24 h, and 36 h, it was 2.08, 2.19, and 2.42 times that at 0 h, respectively. Similarly, there were differences in the changes in soluble protein content over time between the hybrid and conventional rice varieties. Under low-temperature cold damage, the soluble protein content of H-You 518 and Zhongjiazao 17 reached its highest levels of 23.8 mg g−1 Fw and 20.8 mg g−1 Fw at 48 h and 36 h, respectively. This also confirms the inference that the inhibition of protein hydrolysis under low-temperature stress is lower in the hybrid rice variety than in the conventional rice variety. After 72 h of low-temperature treatment, the soluble protein content of the two rice varieties significantly decreased, but after 96 h, the soluble protein content began to increase. At 96 h, the soluble protein content of H-You 518 was 88.79%, while that of Zhongjiazao 17 was 65.80%, compared with the content at 48 h.
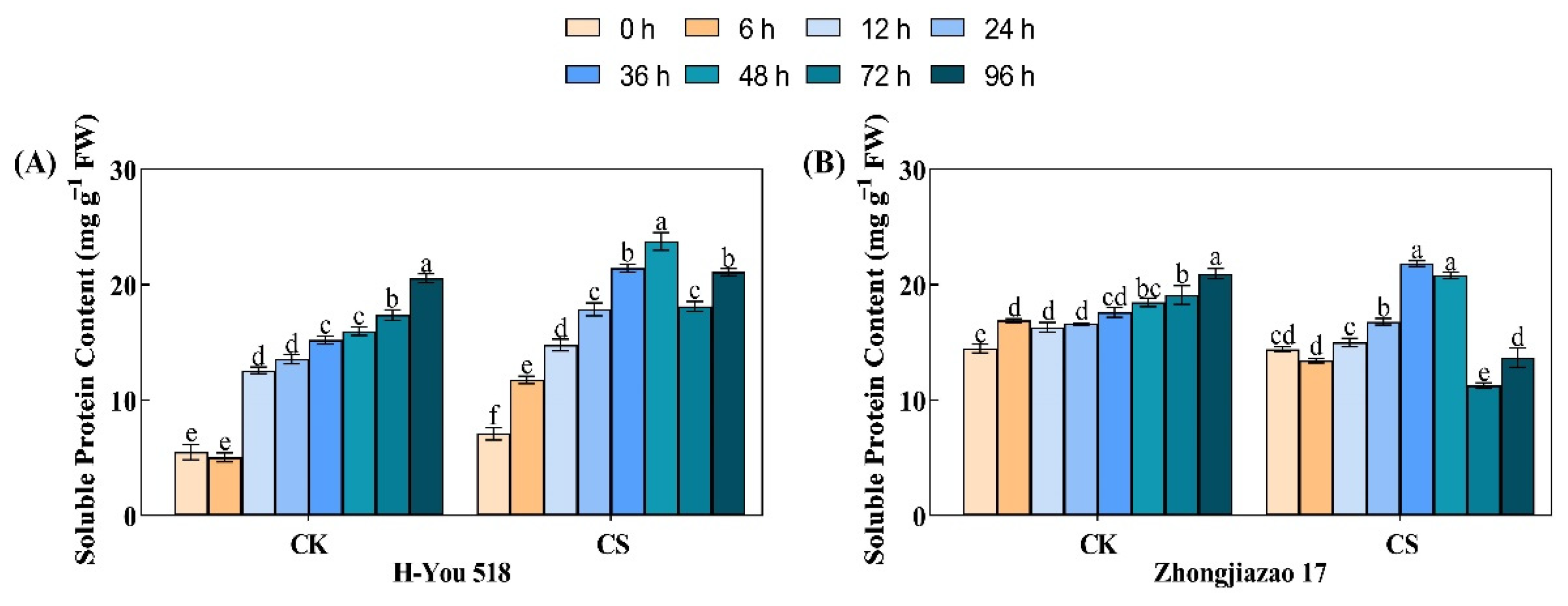
Figure 3: Changes in soluble protein content in different rice varieties during seed germination after low-temperature treatment. In the figure, (A) represents soluble protein content of H-You 518; and (B) represents soluble protein content of Zhongjiazao 17. Means ± SEs with different letters denote significant statistical differences (p < 0.05).
3.5 Effects of Low Temperature on Alpha-Amylase Activity during Rice Seed Germination Process
During the germination stage of rice seeds, the hydrolysis of starch is mainly carried out by alpha-amylase. According to Fig. 4, the α-amylase activity in the control group continued to increase during the period of 0–96 h. The low-temperature cold damage treatment also had a significant inhibitory effect on α-amylase activity, but there were significant differences between the hybrid and conventional rice varieties. The α-amylase activity of H-You 518 treated with low temperature at 12, 24, and 36 h was 1.42, 1.73, and 2.42 times that at 0 h, respectively. The α-amylase activity of Zhongjiazao 17 treated with low temperature at 12, 24, and 36 h was 1.32, 1.65, and 2.19 times that at 0 h, respectively. The inhibition degree of α-amylase activity in the hybrid rice variety under low temperature and cold stress was lower than that in the conventional rice variety. The two rice varieties showed a significant decrease in α-amylase activity at 72 h, and a significant increase in α-amylase activity at 96 h, reaching its maximum value. Under the low-temperature damage treatment, the α-amylase activity of H-You 518 at 96 h was 6.51 times that at 0 h, while that of Zhongjiazao 17 at 96 h was 3.84 times that at 0 h.
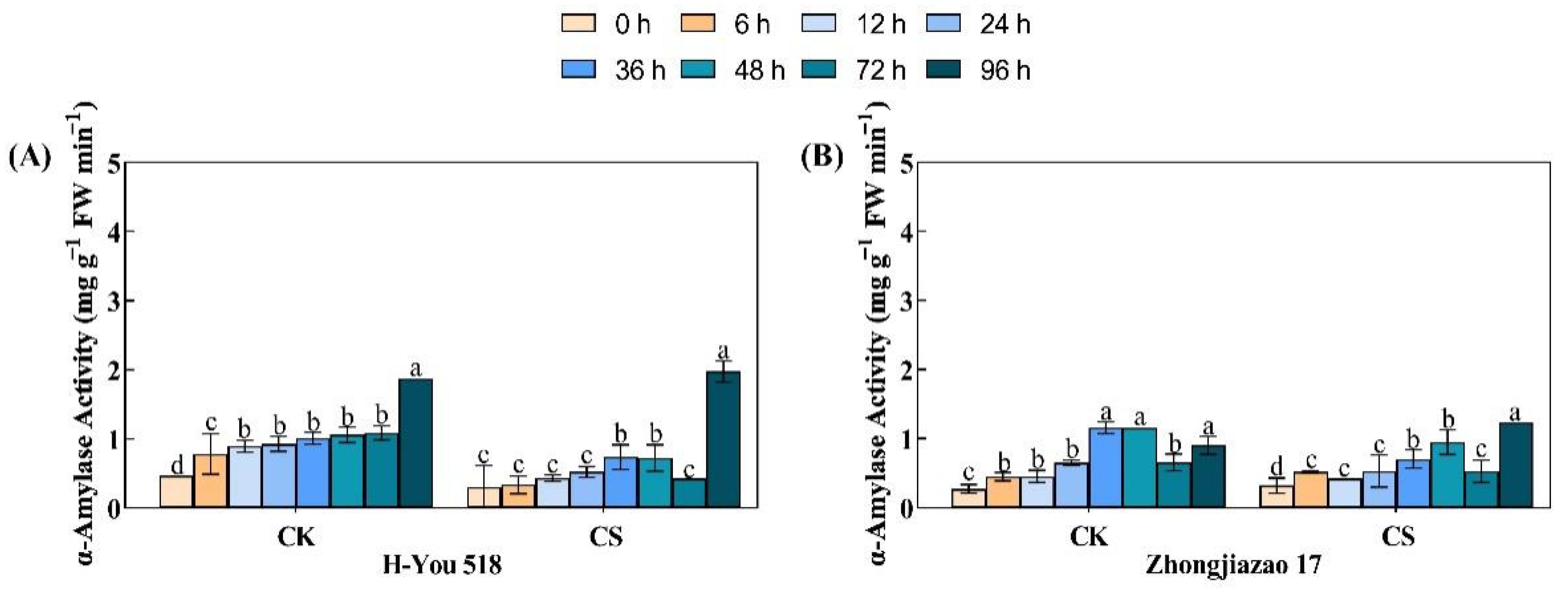
Figure 4: Changes in α-amylase activity in different rice cultivars during seed germination after low-temperature treatment. In the figure, (A) represents alpha-amylase activity of H-You 518; and (B) represents alpha-amylase activity of Zhongjiazao 17. Means ± SEs with different letters denote significant statistical differences (p < 0.05).
3.6 Effects of Low Temperature on Endopeptidase Activity during Rice Seed Germination
Peptidases are enzymes that hydrolyze peptide chains. Endopeptidases and exopeptidases are two common enzymes involved in protein degradation. During the germination and emergence stages of rice seeds, protein degradation is completed by endopeptidases. According to Fig. 5, the activity of endopeptidases increased continuously from 0 to 96 h during seed germination and emergence, while low-temperature cold stress had a strong inhibitory effect on the activity of endopeptidases. Under the control conditions, the endopeptidase activity of H-You 518 at 6, 12, 24, 36, and 48 h was 1.52, 1.81, 2.28, 2.17, and 2.79 times that at 0 h, respectively, while under the low-temperature conditions, it was 1.37, 1.45, 1.80, 1.73, and 1.89 times that at 0 h, respectively. The endopeptidase activity of Zhongjiazao 17 under the low-temperature conditions at 6, 12, 24, 36, and 48 h was 1.09, 1.11, 1.17, 1.15, and 1.31 times that at 0 h, respectively, indicating that the changes in endopeptidase activity were slower in the conventional rice variety. The activity of endopeptidases increased rapidly after 96 h of control treatment, with the activity being 3.62 and 1.84 times higher than that at 0 h for both H-You 518 and Zhongjiazao 17, while the endopeptidase activity of the two rice varieties was only 2.48 and 1.79 times higher than that at 0 h after 96 h of low-temperature cold damage treatment.
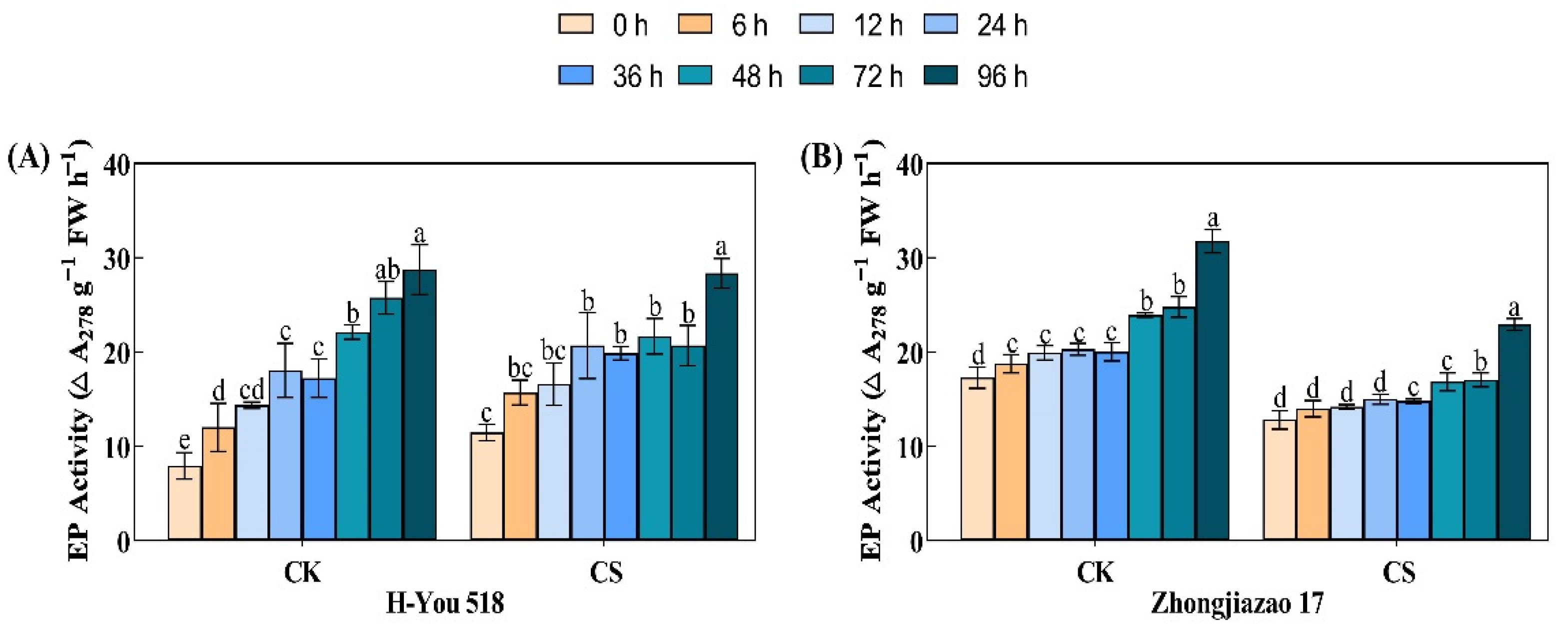
Figure 5: Changes in endopeptidase activity in different rice cultivars during seed germination after low-temperature treatment. In the figure, (A) represents endopeptidase activity of H-You 518; and (B) represents endopeptidase activity of Zhongjiazao 17. Means ± SEs with different letters denote significant statistical differences (p < 0.05).
3.7 Effects of Low Temperature on the Expression of Related Genes during Rice Seed Germination
According to Fig. 6A,B, the expression of the α-AMY gene showed an upward trend from 0 to 96 h of seed germination. The expression of the α-AMY gene reached its maximum value at 96 h, with the expression increasing the fastest in the H-You 518 variety. The expression of the α-AMY gene at 96 h was 10.65 times that at 0 h. Under the control conditions, the expression level of the α-AMY gene in H-You 518 at 6 h, 12 h, 24 h, 36 h, 48 h, 72 h, and 96 h was 6.04, 8.20, 1.96, 5.7, 6.53, 6.97, and 10.65 times higher than that at 0 h, respectively. Under the low-temperature conditions, the expression level of the α-AMY gene in H-You 518 at 6 h, 12 h, 24 h, 36 h, 48 h, 72 h, and 96 h was 0.30, 0.40, 1.39, 0.75, 1.24, 2.49, and 5.17 times higher than that at 0 h, respectively, indicating that the low-temperature treatment significantly reduced the expression level of the α-AMY gene. Under the low-temperature conditions, the expression level of the α-AMY gene first increased steadily and then rapidly.
According to Fig. 6C,D, the expression level of the EP gene showed an upward trend from 0 to 96 h of germination, reaching its maximum value at 96 h. Under the control conditions, the EP gene expression level in H-You 518 at 6 h, 12 h, 24 h, 36 h, 48 h, 72 h, and 96 h was 1.24, 1.35, 1.67, 2.00, 3.62, 4.81, and 6.48 times higher than that at 0 h, respectively. Zhongjiazao 17 showed a similar pattern, indicating that the EP gene expression level slowed down in the early stages of germination and showed a more significant upward trend after 36 h. After 12 h of low-temperature cold damage treatment, the expression level of the EP gene began to be lower than that of the control treatment, and as the low-temperature cold damage time prolonged, the difference in the expression level of the EP gene between the two treatments became greater.
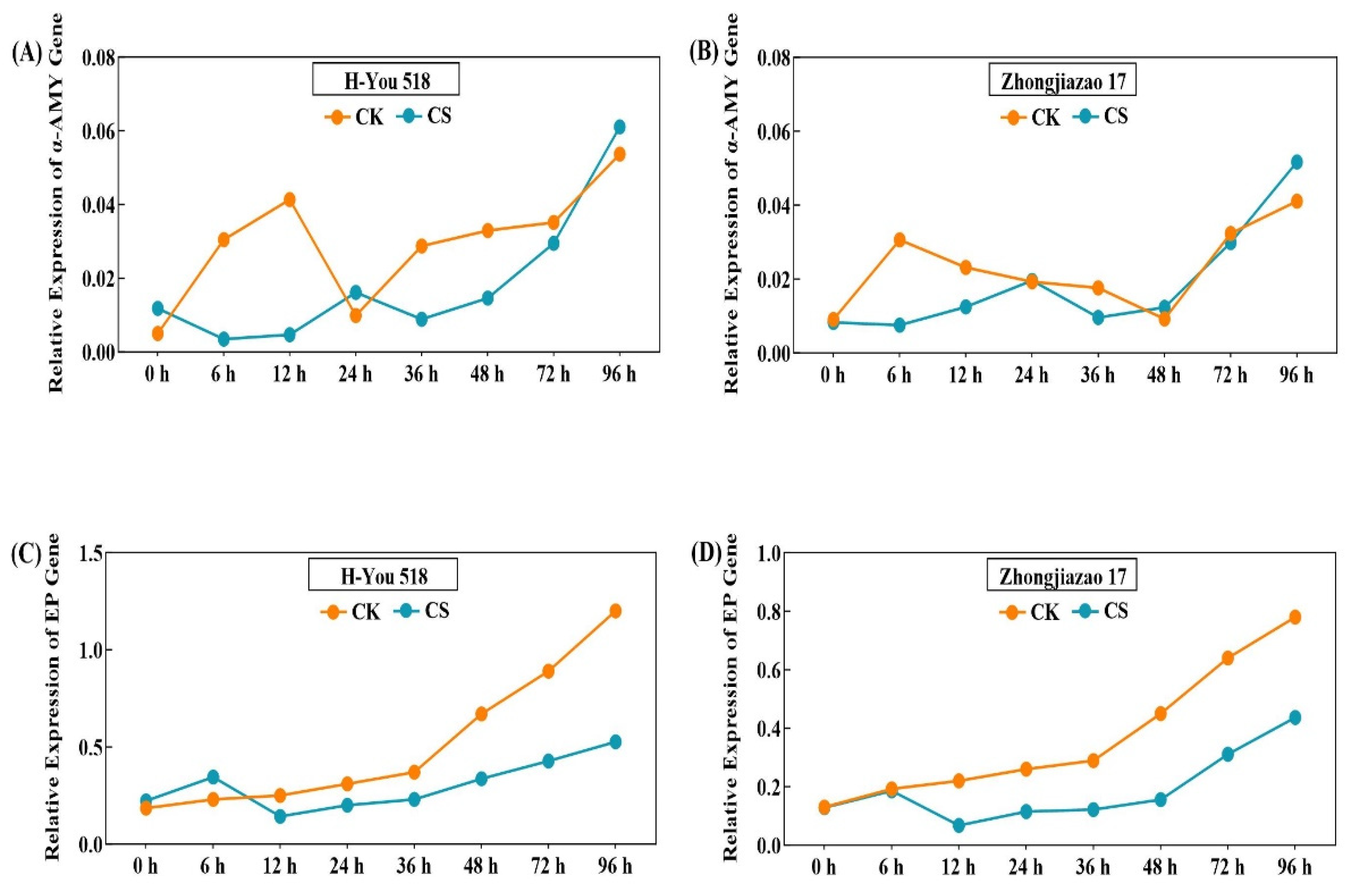
Figure 6: Effects of low temperature on the expression of the α-AMY and EP genes during rice seed germination. (A,C) in the figure represent the variety H-You 518, while (B,D) represent the variety Zhongjiazao 17; (A,B) present the expression levels of the α-amylase gene, while (C,D) present the expression levels of the endopeptidase gene.
3.8 Correlation between Enzyme Activity and Gene Expression Levels, As Well As Storage Substance Content, during Seed Germination
According to Table 3, the correlation between alpha-amylase activity and total soluble sugar content during seed germination was not significant under different germination conditions. After low-temperature damage, the correlation coefficient between α-amylase activity and soluble protein content was 0.55, showing a significant positive correlation, and the trends of these two parameters were consistent. The activity of α-amylase was negatively correlated with the expression level of α-AMY. The correlation between endopeptidase activity and total soluble sugar content was not significant under different germination conditions. The trend of changes in the contents of endopeptidases and soluble proteins was consistent, and these two parameters were significantly positively correlated, with correlation coefficients of 0.83 and 0.61 under the standard and low-temperature conditions, respectively. Under the standard germination conditions, the correlation coefficient between endopeptidase activity and EP expression level was −0.73, showing a significant negative correlation.
Table 3: Correlation coefficients between enzyme activity, gene expression, and storage protein content during seed germination.
| Relativity Analysis | CK | CS |
|---|---|---|
| α-amylase activity and total soluble sugar content | 0.37 | 0.14 |
| α-amylase activity and soluble protein content | 0.07 | 0.55* |
| α-amylase activity and expression level | 0.29 | −0.02 |
| EP activity and total soluble sugar content | −0.21 | 0.26 |
| EP activity and soluble protein content | 0.83** | 0.61** |
| EP activity and expression level | −0.73** | 0.07 |
Rice is a temperature-sensitive and temperature-loving crop. During seed germination, the optimal temperature is generally controlled between 25°C and 28°C. A low temperature has a significant inhibitory effect on seed germination rate, germination vigor, germination index, and vitality index. In this experiment, after three days of low-temperature treatment at 4°C, the germination potential, germination rate, germination index, and vigor index of rice seeds were significantly reduced. At the same time, the seed germination vigor of the convention rice variety Zhongjiazao 17 was more sensitive to low temperatures than that of the hybrid rice variety H-You 518. Liu Yuchen et al. [31] found that the germination ability under low-temperature conditions varies among different varieties, and genotype may determine the cold tolerance of seeds during germination.
In normal environments, the production of reactive oxygen species (ROS) in plants is generally in equilibrium with clearance. However, under adverse conditions, this equilibrium is disrupted, triggering the formation of a large amount of ROS and causing the cell membrane to undergo lipid peroxidation, resulting in damage to the cell membrane structure [32]. To resist the damage caused by reactive oxygen species, plants activate the antioxidant protection system to maintain a dynamic balance of reactive oxygen species in order to tolerate, slow down, or resist stress damage [33]. Low temperatures can induce an increase in antioxidant enzyme activities of rice plants, enhance their antioxidant defense system, help stabilize the cell membrane, delay leaf aging, and improve cold resistance [34,35].
Studies have shown that the short-term growth of plants under low-temperature conditions can increase the activities of enzymes such as SOD and POD, which is beneficial for plants to maintain high cold resistance when subjected to longer periods of low-temperature stress [36]. This experiment showed that after low-temperature stress during the germination stage, the activities of POD and CAT and the content of MDA in the rice seeds significantly increased. Under low-temperature stress, an increase in antioxidant enzyme activities is induced to resist the damage caused by reactive oxygen species to the cell membrane, while the increase in MDA content reflects the level of lipid peroxidation and the degree of damage to the cell membrane structure. This study shows that after the low-temperature cold damage treatment, the antioxidant enzyme activities of the hybrid rice variety H-You 518 are higher, but the MDA content is lower, than those of the conventional rice variety Zhongjiazao 17, indicating that the hybrid rice variety H-You 518 presents stronger cold resistance.
During seed germination, life activities are enhanced and a large amount of carbon source is required. Starch is continuously catalyzed and broken down by amylase and hydrolase enzymes, producing a large amount of carbohydrates to meet the needs of seedling growth [37]. Sucrose is the main soluble sugar in carbohydrate substances. Stachyose plays a key role in the seed germination and dehydration tolerance of some grasses, such as rice seeds [38]. The results of this study indicate that the soluble sugar content during seed germination of the hybrid rice variety H-You 518 and conventional rice variety Zhongjiazao 17 first increased, then decreased, and finally increased again. The synthesis rate of soluble sugars after the low-temperature cold damage treatment was slower in the conventional rice variety compared to the hybrid rice variety. After continuous low-temperature treatment for 72 h, the soluble sugar content decreased, while rice seeds subjected to the control treatment showed a decrease in soluble sugar content after 36 h of germination. This may be because the soluble sugars required for seed germination initially come from small-molecule sugars in the embryo. As the embryo and embryonic root grow, more soluble sugars need to be decomposed into storage substances, synthesized, and converted into sugars. When the synthesis rate is greater than the decomposition rate, the content of soluble sugars can be accumulated.
Soluble proteins provide energy and material basis for metabolic activities during seed germination, participate in various biochemical reactions, and promote the formation of embryo morphology. Changes in the content of soluble proteins also reflect the internal metabolic activities of seeds [39]. During the seed germination process, soluble proteins show a regular and gradual increase, but it is evident that low-temperatures reduce the magnitude of their increase, with the increase in the conventional rice variety being lower than that in the hybrid rice variety. The low-temperature treatment lasted for 72 h, and the soluble protein content showed a decreasing trend. After returning to the normal temperature, the soluble protein content slowly increased again. After 24 h of recovery, the soluble protein content of the hybrid rice variety recovered faster.
Alpha-amylase is the most important hydrolytic enzyme during the germination of rice seeds. When the soluble sugar level drops to a certain extent, alpha-amylase is activated and catalyzes the hydrolysis of starch, which is hydrolyzed into soluble sugars such as glucose under the action of this enzyme [40] Under normal circumstances, the activity of alpha-amylase shows a slow and stable upward trend in the early stage, and rapidly increases at 96 h. However, the low-temperature treatment had a significant inhibitory effect on alpha-amylase activity, and the degree of inhibition was even stronger in the conventional rice variety. In addition, after 72 h of low-temperature treatment, the activity of α-amylase decreased, but after returning to the normal temperature, α-amylase was activated again.
Endopeptidases are a type of enzymes that hydrolyze peptide bonds within peptides or proteins to produce amino acids [41]. During seed germination, endopeptidases break down peptide substances to produce amino acids. In this experiment, endopeptidases showed a gradually increasing trend, and the activity of endopeptidases reached the maximum value at 96 h of seed germination. Under low-temperature damage, the activity of endopeptidases decreased and the synthesis rate slowed down. The activity of endopeptidases in the conventional rice variety was slower than that in the hybrid rice variety.
The alpha-amylase gene is a gene encoding alpha-amylase. During seed germination, the expression of this gene increases, leading to an increase in alpha-amylase content and accelerating the degradation of starch stored in the seeds, thus providing the necessary energy and nutrients for seed germination [42,43]. In this experiment, the expression level of the α-Amy gene showed a continuous upward trend, while low temperature reduced its expression level, showing a gradual increase followed by a rapid increase. The expression level of the α-Amy gene in the conventional rice variety was lower than that in the hybrid rice variety, indicating that the expression of the α-Amy gene can respond quickly to cold stress.
The EP gene encodes enzymes that hydrolyze peptide bonds inside proteins, degrading them into smaller peptides or amino acids [44]. In this experiment, the expression level of the EP gene first slowly and then rapidly increased, indicating that the expression of the EP gene is regulated by low-temperature cold damage. Under the cold damage treatment, although the expression level of the EP gene slowly increased, it remained lower than that in the normal environment. After seed germination for 36 h, the expression level of the EP gene showed a greater difference compared to the control. This study suggests that low-temperature cold damage conditions can reduce the activity of α-amylase and endopeptidases, thereby slowing down the production of soluble sugars and soluble proteins. Moreover, the activities of α-amylase and endo-peptidases are regulated by the α-AMY and EP genes.
It should be noted that the low-temperature treatments applied in this study were selected to elicit strong physiological responses under controlled conditions. While this approach effectively revealed key mechanisms, it did not include an intermediate temperature range (e.g., 10–15°C) that more accurately represents mild cold stress often encountered in field environments. Future studies incorporating such temperature gradients would be valuable to better understand the gradual acclimation responses and to refine threshold temperatures for cold tolerance in practical agricultural settings.
In summary, this study suggests that during seed germination, stored substances inside of rice seeds, such as starch and protein, gradually decompose into soluble sugars and soluble proteins, which are then transported to the embryo to provide nutrient support for its growth and development. However, under low-temperature cold damage conditions, the activities of α-amylase and endopeptidases are inhibited and reduced, directly leading to a slowdown in the synthesis rate of soluble sugars and soluble proteins. The supply of soluble sugars cannot meet the demand, which leads to an imbalance in the physiological functions of the antioxidant system, ultimately resulting in a decrease in the vitality and germination rate of rice seeds.
Acknowledgement:
Funding Statement: This work was funded by the National Key R&D Program of China (2023YFD2301404); Hunan Provincial Natural Science Foundation Project (2024JJ8071; 2023JJ50463); Hunan Province Graduate Research Innovation Project (QL20230173; CX20240636).
Author Contributions: Cheng Qu and Yue Wang conceived and supervised the work; Fen Liu and Hongliang Yuan conducted the experiments, analyzed the data and prepared the figures; Xinpeng Xiang, Yingbo Chen and Yuxuan Cai assisted in the data analysis; Cheng Qu and Yue Wang drafted the manuscript, together with Fen Liu, Hongliang Yuan, Xinpeng Xiang, Yingbo Chen and Yuxuan Cai. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: Not applicable.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Wang Y , Xue Y , Li J . Towards molecular breeding and improvement of rice in China. Trends Plant Sci. 2005; 10( 12): 610– 4. doi:10.1016/j.tplants.2005.10.008. [Google Scholar] [CrossRef]
2. Zhang Q . Strategies for developing green super rice. Proc Natl Acad Sci U S A. 2007; 104( 42): 16402– 9. doi:10.1073/pnas.0708013104. [Google Scholar] [CrossRef]
3. Wu L , Huo ZG , Yang JY , Xiao JJ , Zhang L , Yu CX , et al. Prediction of levels of low temperature disaster to double cropping rice in Southern China. Chin J Ecol. 2016; 35: 925– 33. [Google Scholar]
4. Chen BX , Zhang Q , Dai ZY , Zhou X , Liu J . Physiological and molecular effects of salicylic acid on rice seed germination at low temperature. Sci Agric Sin. 2024; 57( 7): 1220– 36. (In Chinese). [Google Scholar]
5. Guo H , Chen C , Zhang XL , Qin XY , Feng R . Study on cold tolerance identification and genetic homozygosity of wild rice in Guangxi. Southwest China J Agric Sci. 2017; 30: 1245– 50. [Google Scholar]
6. Zhang WY , Lei YB , Zhang YJ , Guo ZH , Liu NS , Ma WD , et al. Research status on cold tolerance in rice and biotechnological breeding strategies for cold-tolerant early geng/japonica in Heilongjiang Province. J Plant Genet Resour. 2024; 25: 1428– 40. (In Chinese). [Google Scholar]
7. Cao W , Wang Y , Tan B , Liu W , Chu L , Pan Z , et al. Evaluation of seed germination vigor of rice cultivars under low temperature or hypoxic condition related with direct seeding rice. Mol Plant Breed. 2018; 16: 3259– 68. (In Chinese). [Google Scholar]
8. Zheng GJ , Ye C , Xu CM , Chen S , Chu G , Chen LP , et al. Effect of low-temperature stress on glucose and water status in rice germ and its relationship with seedling emergence. Acta Agronom Sin. 2024; 50( 12): 3055– 68. doi:10.3724/sp.j.1006.2024.42014. [Google Scholar] [CrossRef]
9. Talukder S , Mamun MAA , Hossain MS , Khan MAR , Rahman MM , Talukder MR , et al. Duration of low temperature changes physiological and biochemical attributes of rice seedling. Agron Res. 2022; 20: 1163. doi:10.15159/AR.22.051. [Google Scholar] [CrossRef]
10. Zeng ZC , Zhang SC , Shi XC , Chen BS , Li WL . Physiological and biochemical indexes of response to low temperature stress in near isogenic lines of wild rice. Guihaia. 2021; 41( 05): 813– 22. [Google Scholar]
11. Soliman MH , Alayafi AAM , El Kelish AA , Abu-Elsaoud AM . Acetylsalicylic acid enhance tolerance of Phaseolus vulgaris L. to chilling stress, improving photosynthesis, antioxidants and expression of cold stress responsive genes. Bot Stud. 2018; 59( 1): 6. doi:10.1186/s40529-018-0222-1. [Google Scholar] [CrossRef]
12. Hu Z , Fan J , Chen K , Amombo E , Chen L , Fu J . Effects of ethylene on photosystem II and antioxidant enzyme activity in Bermuda grass under low temperature. Photosynth Res. 2016; 128( 1): 59– 72. doi:10.1007/s11120-015-0199-5. [Google Scholar] [CrossRef]
13. Yuan J , Meng J , Liang X , Yang E , Yang X , Chen WF . Biochar’s leacheates affect the abscisic acid pathway in rice seedlings under low temperature. Front Plant Sci. 2021; 12: 646910. doi:10.3389/fpls.2021.646910. [Google Scholar] [CrossRef]
14. Farooq MA , Ma W , Shen S , Gu A . Underlying biochemical and molecular mechanisms for seed germination. Int J Mol Sci. 2022; 23( 15): 8502. doi:10.3390/ijms23158502. [Google Scholar] [CrossRef]
15. Waghorn JJ , del Pozo T , Acevedo EA , Cardemil LA . The role of two isoenzymes of α-amylase of Araucaria araucana (Araucariaceae) on the digestion of starch granules during germination. J Exp Bot. 2003; 54( 384): 901– 11. doi:10.1093/jxb/erg093. [Google Scholar] [CrossRef]
16. Guy C . Cold acclimation and freezing stress tolerance: Role of protein metabolism. Annu Rev Plant Physiol Plant Mol Biol. 1990; 41: 187– 223. doi:10.1146/annurev.pp.41.060190.001155. [Google Scholar] [CrossRef]
17. Wang CP , Lei KP , Li ZG , Lin Q , Wu H . Effects of chilling stress on chlorophyll fluorescence characteristics of seedling leaves with different leaf ages of Oryza sativa. J Plant Resour Environ. 2012; 21: 38– 43. [Google Scholar]
18. Han LZ , Zhang YY , Qiao YL , Cao GL , Zhang SY , Kim JH , et al. Genetic and QTL analysis for low-temperature vigor of germination in rice. Acta Genet Sin. 2006; 33( 11): 998– 1006. doi:10.1016/S0379-4172(06)60135-2. [Google Scholar] [CrossRef]
19. Duan LC , Zhang K , Zhang QM , Liu D , Gui BY , Cai Z , et al. Effects of low temperature stress on seedling emergence and seedling growth of direct-seeding early rice. Guangdong Agric Sci. 2018; 45: 1– 6. [Google Scholar]
20. Shinkawa R , Morishita A , Amikura K , Machida R , Murakawa H , Kuchitsu K , et al. Abscisic acid induced freezing tolerance in chilling-sensitive suspension cultures and seedlings of rice. BMC Res Notes. 2013; 6: 351. doi:10.1186/1756-0500-6-351. [Google Scholar] [CrossRef]
21. Han Q , Ren Z , Zhu Q , Zhou Y , Zhu M , He J , et al. Maize SERRATE 1B positively regulates seed germinability under low-temperature. Plant Sci. 2025; 355: 112458. doi:10.1016/j.plantsci.2025.112458. [Google Scholar] [CrossRef]
22. Suo R , Kulbir S , You F , Conner R , Cober E , Wang M , et al. Low temperature and excess moisture affect seed germination of soybean (Glycine max L.) under controlled environments. Can J Plant Sci. 2024; 104( 4): 375– 87. doi:10.1139/cjps-2023-0162. [Google Scholar] [CrossRef]
23. Li J , Li Z , Tang Y , Xiao J , Nangia V , Liu Y . Arginine promotes seed energy metabolism, increasing wheat seed germination at low temperature. Crop J. 2024; 12( 4): 1185– 95. doi:10.1016/j.cj.2024.05.018. [Google Scholar] [CrossRef]
24. Gong H , Zhu X , Chen K , Wang S , Zhang C . Silicon alleviates oxidative damage of wheat plants in pots under drought. Plant Sci. 2005; 169( 2): 313– 21. doi:10.1016/j.plantsci.2005.02.023. [Google Scholar] [CrossRef]
25. Albuquerque RV , Malcher NS , Amado LL , Coleman MD , Dos Santos DC , Borges RS , et al. In vitro protective effect and antioxidant mechanism of resveratrol induced by dapsone hydroxylamine in human cells. PLoS One. 2015; 10( 8): e0134768. doi:10.1371/journal.pone.0134768. [Google Scholar] [CrossRef]
26. Hodges DM , De Long JM , Forney CF , Prange RK . Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta. 1999; 207( 4): 604– 11. doi:10.1007/s004250050524. [Google Scholar] [CrossRef]
27. Huan DU , Tongtong MA , Guo S , Zhang Y , Bai ZY , Li CD . Response of root morphology and leaf osmoregulation substances of seedling in barley genotypes with different heights to PEG stress. Sci Agric Sin. 2017; 50( 13): 2423– 32. [Google Scholar]
28. Liu L , Xia W , Li H , Zeng H , Wei B , Han S , et al. Salinity inhibits rice seed germination by reducing α-amylase activity via decreased bioactive gibberellin content. Front Plant Sci. 2018; 9: 275. doi:10.3389/fpls.2018.00275. [Google Scholar] [CrossRef]
29. Zhang ZG , Rui Q , Xu LL . Relationship between endopeptidase and H2O2 during the aging of wheat leaf. J Integr Plant Biol. 2001; 43: 127– 31. [Google Scholar]
30. Bradford MM . A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976; 72: 248– 54. doi:10.1016/0003-2697(76)90527-3. [Google Scholar] [CrossRef]
31. Liu YC , Yang DG , Li L , Chai MZ , Ju R , Ban B , et al. Effect of low temperature stress on maize seed germination and starch decomposition enzyme activity. J Maize Sci. 2018; 26: 64– 8. doi:10.13597/j.cnki.maize.science.20180110. [Google Scholar] [CrossRef]
32. Desikan R , A-H-Mackerness S , Hancock JT , Neill SJ . Regulation of the Arabidopsis transcriptome by oxidative stress. Plant Physiol. 2001; 127( 1): 159– 72. doi:10.1104/pp.127.1.159. [Google Scholar] [CrossRef]
33. Polle A . Dissecting the superoxide dismutase-ascorbate-glutathione-pathway in chloroplasts by metabolic modeling. Computer simulations as a step towards flux analysis. Plant Physiol. 2001; 126( 1): 445– 62. doi:10.1104/pp.126.1.445. [Google Scholar] [CrossRef]
34. Mittler R , Vanderauwera S , Gollery M , Van Breusegem F . Reactive oxygen gene network of plants. Trends Plant Sci. 2004; 9( 10): 490– 8. doi:10.1016/j.tplants.2004.08.009. [Google Scholar] [CrossRef]
35. Park S , Choi BY , Seol YB , Kim J . Low-temperature plasma modulates seed germination through reactive oxygen species in dose-dependent manner. Free Radic Res. 2025; 59( 6–7): 545– 56. doi:10.1080/10715762.2025.2544800. [Google Scholar] [CrossRef]
36. Zhou Y . Effects of chilling and low light on cucumber seedlings growth and their antioxidative enzyme activities. Chin J Appl Ecol. 2003; 14: 921– 4. [Google Scholar]
37. Chen H , Shi J , Tao L , Han X , Lin G , Cheng X . Exogenous spermidine priming mitigates the osmotic damage in germinating seeds of Leymus chinensis under salt-alkali stress. Front Plant Sci. 2021; 12: 701538. doi:10.3389/fpls.2021.701538. [Google Scholar] [CrossRef]
38. MacLeod AM , McCorquodale H . Water-soluble carbohydrates of seeds of the Gramineae. New Phytol. 1958; 57( 2): 168– 82. doi:10.1111/j.1469-8137.1958.tb05303.x. [Google Scholar] [CrossRef]
39. Begum N , Hasanuzzaman M , Li Y , Akhtar K , Zhang C , Zhao T . Seed germination behavior, growth, physiology and antioxidant metabolism of four contrasting cultivars under combined drought and salinity in soybean. Antioxidants. 2022; 11( 3): 498. doi:10.3390/antiox11030498. [Google Scholar] [CrossRef]
40. Damaris RN , Lin Z , Yang P , He D . The rice alpha-amylase, conserved regulator of seed maturation and germination. Int J Mol Sci. 2019; 20( 2): 450. doi:10.3390/ijms20020450. [Google Scholar] [CrossRef]
41. Mitsuhashi W , Oaks A . Development of endopeptidase activities in maize (Zea mays L.) endosperms. Plant Physiol. 1994; 104( 2): 401– 7. doi:10.1104/pp.104.2.401. [Google Scholar] [CrossRef]
42. Yu H , Teng Z , Liu B , Lv J , Chen Y , Qin Z , et al. Transcription factor OsMYB30 increases trehalose content to inhibit α-amylase and seed germination at low temperature. Plant Physiol. 2024; 194( 3): 1815– 33. doi:10.1093/plphys/kiad650. [Google Scholar] [CrossRef]
43. Xu C , Yang F , Tang X , Lu B , Li Z , Liu Z , et al. Super rice with high sink activities has superior adaptability to low filling stage temperature. Front Plant Sci. 2021; 12: 729021. doi:10.3389/fpls.2021.729021. [Google Scholar] [CrossRef]
44. Zhang N , Jones BL . Purification and partial characterization of a 31-kDa cysteine endopeptidase from germinated barley. Planta. 1996; 199( 4): 565– 72. doi:10.1007/BF00195188. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools