 Open Access
Open Access
REVIEW
From Flavedo to Flavors and Fragrance: Botanical Foundations of Citrus Peel Essential Oils in Bio-Based Value Chains
Department of Plant Cultivation—Botany, Faculty of Agriculture, University of Agricultural Sciences and Veterinary Medicine from Cluj-Napoca, Cluj-Napoca, Romania
* Corresponding Author: Ioana Crișan. Email:
(This article belongs to the Special Issue: The Biological Activity of Essential Oils, Volume II)
Phyton-International Journal of Experimental Botany 2026, 95(3), 5 https://doi.org/10.32604/phyton.2026.078943
Received 11 January 2026; Accepted 26 February 2026; Issue published 31 March 2026
Abstract
Citrus is a genus of great economic importance and a complex history of hybridization. The aim of this work is to present the trajectory of essential oil (EO) from the citrus fruit peel to their main uses. The flavedo is a morpho-functional region of the citrus fruit peel where EO-producing cavity glands are located. The EO produced is the source of the characteristic citrus aroma relevant for flavor (food and beverage industry) and fragrance applications (perfumery). The citrus EO is dominated by monoterpene hydrocarbons. Out of these, D-limonene is the major compound in the peel EO of commercial citrus types, accounting for 30% and over 90% of its composition. Minor oxygenated components are important contributors to the distinct sensory characteristics. Selected studies on various citrus types indicate that EOs exhibit a strain-dependent antimicrobial activity as well as certain antioxidant activity relevant in particular for food preservation and active packaging. The most dynamic area of research is the optimization of delivery systems that seek to improve the stability and functionality across applications. The citrus value chain is integrated vertically, generating value for the customer and is linked horizontally to other value chains. Its main vulnerability resides in upstream production constraints from the agronomic segment that can potentially impact sustainability and supply. For the next decade, growth is projected for citrus flavor and fragrance markets.Keywords
Citrus fruits are cultivated in over 140 countries around the world in the so-called “citrus belt” located mainly in tropical, subtropical and Mediterranean climate between 40° N and 40° S latitude. The place of origin and domestication in Asia has been a smaller area compared to their wide distribution today [1]. The characteristic citrus aroma is the foundation of the global citrus flavor and fragrance market. According to a recent market analysis, the global citrus flavors market is expected to increase between 2026 and 2036, from USD 13.4 billion to USD 21.8 billion, with a compound annual growth rate of over 4% [2]. At the same time, between 2025 and 2033, the global citrus perfume market size was projected to grow from USD 10.6 billion in global market revenue to USD 16.7 billion, at a compound annual growth rate of over 5% [3].
EOs are considered “low volume-high value” commodities due to their market prices [4]. Natural aromas gain more traction than synthetic ones, and this supports the citrus industry growth [2]. Citrus EOs are formed and stored in oil glands at different depths in the fruit peel, mainly the flavedo region. Citrus EO contains between 85 and 99% volatile components and between 1 and 15% non-volatile compounds [5]. Orange, lemon and lime have been listed at the top of the most widely produced and traded EOs [4]. The soft drinks industry is one of the largest destinations of citrus EOs [6,7,8], followed by the candy industry. In the food industry, both flavor and functional properties of citrus EO are important. Citrus flavoring is used across a wide range of food items, from ice cream, desserts, cookies and bakery [6]. Bioactivities are screened particularly for applications related to the food industry [9].
The high economic relevance of citrus EO and increasing market trends bring into focus the citrus plants as sources of economically valuable natural compounds. Despite their extensive use, the literature is often fragmented on citrus EO, focusing on botanical, chemical and applied aspects. The aim of this work is to provide a contextualization of Citrus EO by connecting botany to its uses. In this regard, this work invites the reader to an exploration of the trajectory of EO from the flavedo to their final uses that contribute to human well-being.
The origin of citrus plants is rather interesting, with hypotheses that evolved throughout time. While all scientific efforts of elucidating various hypotheses remain captivating explorations of the subject, the generally accepted idea today is that citrus originated in Southeast Asia [10,11]. Studies on collections of citrus species, hybrids and admixed varieties suggest that genus Citrus is a monophyletic group that diversified during late Miocene throughout Southeast Asia, followed by another radiation during the early Pliocene in Oceania [11,12]. Citrus was domesticated in Southeast Asia many thousands of years ago [13].
Citrus has always been of immense economic value [14,15]. As early as 2000 BC in the Xia Dynasty in China, citrus was listed among the main tributes and taxes in important Chinese citrus-producing regions. In Ancient China, citrus was seen as a symbol of moral integrity and distinguished spirit and had recognized medicinal value [12]. The citrus is supposed to have been first introduced in the Mediterranean region in ancient times. In Europe, the first written mention of the fruit comes from the Greek writings of Theophrastus in the 4th century BC [16]. But archaeological research suggests its presence possibly as far back as the 6th century BC in Sardinia and the Campanian coast. Archaic citrus forms were present in Italy at least by the 1st century BC, according to archaeobotanical evidence [17]. In the New World, the citrus was introduced in the 16th century AD. The etymology of the name derives from the Greek “Kedros” in reference to trees such as cedars, pines or cypresses with their refreshing scent. Important early work in citrus cultivation, “Hesperides sive de malorum aureorum cultura et usu” from the 17th century AD, hinted to an association of citrus fruits and golden apples from the garden of Hesperides, an ancient Greek legend that predates citrus introduction in Europe [12]. Nonetheless, botanically the fruit of this genus is called hesperidium [18].
The majority of hybrids and admixtures of the current commercial importance or the cultivated citrus express phenotypes that are maintained by continuous clonal propagation by grafting or apomixis via nucellar polyembryony [12].
Genus Citrus L. belongs to the family Rutaceae, subfamily Aurantioideae, tribe Citreae, subtribe Citrinae [19,20]. The genus Citrus is one of the genera belonging to the “true citrus” group with almost all of the commercially cultivated citrus situated in this genus [19,20,21,22]. At the time of writing this work, checklist of genus Citrus lists 33 accepted species and hybrids [23], as presented in Table A1. Classification of this genus remains challenging, mainly due to a lack of consensus in what differences may justify species status. Most of the modern cultivars have interspecific origin because citrus species are sexually compatible able to produce fertile hybrids [12].
The members of the genus Citrus are shrubs or small trees, evergreen, rarely deciduous. The young branches are often flat and angled, usually presenting spines at axils. Leaves are most often 1-foliolate, with the petiole usually conspicuously winged and articulated with the base of the leaf blade. The leaf blade is more or less leathery, presenting dense pellucid fragrant oil spots; the leaf margin is crenulate to entire. Flowers are fragrant and hermaphrodite or male, either solitary or arranged in small axillary inflorescence. The calyx is shaped as a cup and is subglabrous. Petals vary in number, are imbricate and thick, and are white in color or pinkish-red outside. Stamens are usually either free or basally fused. The flower has a nectary disk. The ovary has several locules, each with two or more ovules. The characteristic fruit is classed as a hesperidium with adaxially attached seeds. The seed germination is hypogeous [24].
Hesperidium is characterized by a leathery epicarp (syn. exocarp) that presents the color and secretory structures of EO, a soft spongy mesocarp usually white in color and a thin endocarp filled with many succulent trichomes called juice sacs that make the pulp of the fruit [18,25,26]. Citrus fruits have been considered non-climacteric. Experimental evidence suggests that only young citrus fruitlets exhibit a climacteric mechanism that subsequently gets lost during growth and maturation [27]. Hence, citrus fruits ripen before harvest, and this is generally indicated by the rind changing in color with the exception of some orange varieties that change color before ripeness [14]. In the Northern Hemisphere the fruit harvesting starts in October-November and may continue until late spring and early summer of the following year. Crops from regions closer to the equator may flower 2–3 times a year [25].
Early reference taxonomic systems of classification for the genus Citrus such as those proposed by T. Tanaka or W.T. Swingle and P.C. Reece, dealt with morphology, biogeography and sexual compatibility [12,20,28]. To these followed findings that rely on molecular tools, which increase the precision of the insight into this genus [29,30]. Metabolomic approaches are versatile for taxonomic insights, especially applicable for Citrus genotypes characterization and germplasm evaluations [31]. Genomic and phylogenetic research revealed that several industrial citrus types occurred from repeated hybridization among a small ancestral core, primary comprised by: citron (C. medica), mandarin (C. reticulata) and pomelo (C. maxima) [11,30]. This core represents the origin of cultivated Citrus group. Subsequent recombination among these basic taxa gave rise to other economically important citruses such as sweet and bitter/sour orange, grapefruit, lemon and lime [32]. This explains why some commercial citruses genetically are situated within the same group (genetic affinity), but display distinct phenotype which is relevant for industrial purposes and applications.
Based on Table 1, can be observed that POWO (Plants of The World Online) nomenclature reflects genetic ancestry and phylogenetic affinity of interest for botanists, whereas RHS (Royal Horticultural Society) grouping is oriented toward cultivation practice. Industry names have been well-established for decades across literature and emphasize the distinct chemical, sensory, and functional identities of commercially important citrus types. Regarding this last instance, T. Tanaka system of classification [33] had great influence by imbedding in use some of the names for citrus types in circulation in agriculture, trade, EO literature, standards, pharmacopoeias and industry.
Table 1: Correspondence between some commercial Citrus names and nomenclature used in current sources.
| Common Name | Plants of the World Online (POWO)1 | Royal Horticultural Society (RHS)2 | Frequently Used in Industry (ISO)3 | European Pharmacopoeia (EPh)4 |
|---|---|---|---|---|
| bergamot | Citrus × limon (L.) Osbeck | Citrus × limon Bergamot Group | Citrus bergamia Risso & Poit | - |
| bitter orange | Citrus × aurantium L. | Citrus × aurantium Sour Orange Group | Citrus aurantium L. | Citrus aurantium L. ssp. aurantium (C. aurantium L. ssp. amara Engl.) |
| grapefruit | Citrus × aurantium f. aurantium | Citrus × paradisi | Citrus × paradisi Macfad. | - |
| lemon | Citrus × limon (L.) Osbeck | Citrus × limon | Citrus limon (L.) Burman | Citrus limon (L.) Burman |
| key lime | Citrus × aurantiifolia (Christm.) Swingle | Citrus × aurantiifolia | Citrus aurantifolia (Christm.) Swingle | - |
| mandarin | Citrus reticulata Blanco | Citrus reticulata | Citrus reticulata Blanco | Citrus reticulata Blanco |
| sweet orange | Citrus × aurantium f. aurantium | Citrus × aurantium Sweet Orange Group | Citrus sinensis (L.) Osbeck | Citrus × sinensis (L.) Osbeck. |
Citrus has a basic chromosome number of x = 9 [37]. Genetic transformation allows the introduction of traits without altering the elite genetic background. This is important for citrus improvement of commercial species such as sweet orange, lemon and grapefruit where due to existing heterozygous and complex genetic structure, transformation might be one of the best tools [13]. Citrus phylogeny studies remain challenging, particularly due to the difficulty of identifying pure wild progenitor species in the context of consistent interspecific hybridization in this genus [12].
2.3 The Fruit Peel and Secretory Structures
The flavedo is comprised of epicarp, hypodermis, outer mesocarp and oil glands. The epicarp is covered towards the exterior with a cuticle that is complex in its origin, development and structure. The epicuticular waxes are synthesized by epidermal cells and continue to deposit as the fruit grows, hardens and later develops cracks naturally. Studies in some varieties of orange, tangerine and lemon revealed that initially these wax layers are amorphous and later, as surfaces crack, they get lifted to form large and irregular flat plates. Their main function is to prevent water loss through the peel. Stomata are spread over the surface of the fruit between the oil glands, and found in higher numbers in the style region and decreasing towards the stem and calyx region. The stomata get plugged by waxes as the fruit grows and matures [25].
The epicarp presents plastids: chloroplasts that turn into globular chromoplasts (with carotenoids) as the fruit changes color from green to yellow or orange when ripe [38]. Based on the observation of a healthy lemon fruit peel section, the epidermis is made of polygonal isodiametric cells with no intercellular spaces, and the epidermis has a uniform thickness. The cells immediately beneath the epidermis make up the hypoderm and are small thick-walled parenchymatous cells with no intercellular spaces surrounding the oil glands [39].
The secretory glands of the Citrus peel are named rind oil glands [40], or secretory cavities [41]. More precisely, the colorless hypodermis and outer mesocarp located immediately beneath epicarp contain the oil glands [25]. These glands are the main sites of synthesis, secretion and accumulation of EO and develop progressively with the fruit growth. Although the mode of secretory cavity formation in Citrus has been debated, with interpretations ranging from schizogenous to lysigenous or the overlapping of both as model of development, the more recent evidence is supporting the first. Study on the ontogeny of these secretory structures identified four stages. The first is starting in the ovary of the flower (from an epidermal cell and a subepidermal cell) and is leading to the formation of an initial cell group found in young fruits. In the second stage this initial group develops into a glandular structure with a lumen. The third stage corresponds to the lumen-expanding stage. In fourth and final stage of maturity, the lumen reaches its final size. In the mature glands, the secretory cavity consists of a conical cap and a globular gland. The lumen is lined by cell layers comprised of tangentially elongated cells with dense cytoplasm. These are surrounded by a sheath of several layers of elongated cells with higher vacuolization [42]. The characteristics of the rind oil glands can be used for storage time prediction as demonstrated for oranges [40]. The EO glands are ranging in sizes between 10–100 μm [25] or even larger, between 200–400 μm [42]. A study comparing differences between the peel of Pompia (an Italian lemon variety) and pomelo has put in evidence notable differences in the localization variability and size of these EO secretory structures. Firstly, a greater variation in the distance from epidermis of oil secretory cavities was observed in the lemon variety (at depths of 16 μm to 2100 μm) compared to pomelo in which secretory cavities were positioned more superficially under the epidermis (at depths of 9 μm to 216 μm) and were larger than in the lemon variety studied. In addition, in the epidermis of Pompia, having characteristic wrinkled surface, histologic analysis indicated both the presence of stomata but also discontinuities reminiscent of lenticels [41]. The EOs from these secretory glands give the specific citrus aroma [25]. If the glands rupture by impact, fruits develop oleocellosis—a specific lesion of the rind [43].
The albedo is the inner mesocarp and is made of parenchymatous cells with air spaces in between, acting as shock absorbent against impacts. It has varying thickness from thin as in limes (1–2 mm), to medium in oranges (2–5 mm) and thick in pomelos (up to 20 mm). Main vascular bundle strands make up a network along the fruit axis [25], and extend radially from the inner mesocarp towards the epidermis, branching with increased frequency as they approach the epidermis [44]. The flavedo and albedo are blended together, but cell division in albedo tissue ceases after 8–9 weeks and then grows by expansion, while flavedo cell division continues until the fruit maturity [25]. A study on peel of lemon and pomelo showed a change in the arrangement of cells between endocarp and pericarp with an increase in the number of cells per millimeter towards the epidermis [45]. The surface of the flavedo is slick [39].
In Fig. 1, the aspect of the fruit peel sections of some commercial citrus types can be observed. It can be noticed that globular secretory glands with the lumen filled with EO are imbedded at different depths beneath the epidermis, more or less reaching into the white spongy mesocarp, indicating they are not confined to the epicarp sensu stricto.
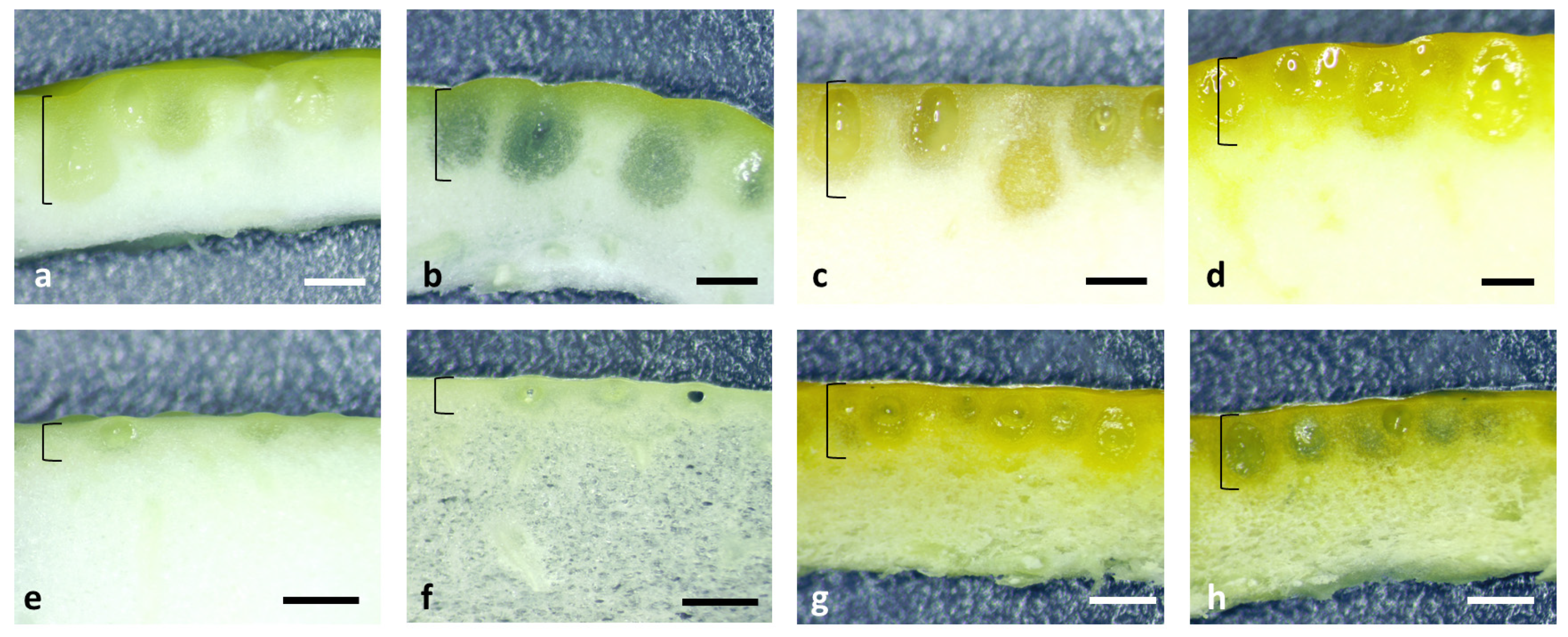
Figure 1: Fruit peel section: lemon (a,b), grapefruit (c), sweet orange (d), pomelo (e,f), clementine (g,h) (scale bar = 1 mm); brackets indicate the flavedo region.
Microscopic examination of the peel section shows that the epicarp surface of the lemon has small depressions at the place where secretory cavities are located (Fig. 1a,b), the grapefruit epicarp surface is smooth over the secretory cavities (Fig. 1c), while in the pomelo the secretory cavities are superficially located and have small convex protrusions towards the exterior (Fig. 1e,f). The flavedo of sweet orange (Fig. 1d) and clementine (Fig. 1g,h) has many secretory cavities one next to another, while in pomelo these are more sparsely arranged with distance between them (Fig. 1e,f).
3.1 Composition and Bioactivity
Citrus EOs are comprised mostly of terpenes (with monoterpenes as dominant) as well as some oxygen-containing compounds. The volatile components represent around 90% of the EO, while non-volatile components are being found in very small amounts. It was determined that terpene hydrocarbons (mainly monoterpenes) while abundant in peel EO, have a lower contribution to specific aroma on their own or may act as flavor carriers and, furthermore, can be removed to increase the intensity of the aroma. Instead, oxygenated compounds, while present in lower amounts in EO were found to be responsible for the characteristic aroma of each citrus type [4]. The citrus EOs are soluble in natural oils, alcohol and ether, but insoluble in water [5]. Based on the EO composition from the seven most important commercial citrus types (Table 2), there are reported components belonging to six main classes of compounds, yet, their presence varies (Table 2). The distinctive aroma of various citrus types is due to a particular profile and represents criteria of selection for destinations of use [46]. Metabolomic profiling based on citrus peel EOs is supporting chemotaxonomic classification and can be a versatile method for germplasm evaluation. Particularly, D-limonene, α-pinene, sabinene and terpinene isomers were shown to be key metabolites relevant for distinction and classification of Citrus genotypes [31].
Table 2: EO peel composition of some commercial Citrus types.
| EO Components | Composition (%) Mininum − Maximum Values | |||||||
|---|---|---|---|---|---|---|---|---|
| Class | Compound* | Bergamot | Bitter Orange | Grapefruit | Lemon | Lime | Mandarin | Sweet Orange |
| Monoterpene hydrocarbons | limonene | 32–47 | 93–95 | 92–96 | 56–78 | 36–46 | 65–75 | 92–97 |
| β-pinene | 4–8.5 | 0.2–1.2 | 0.05–0.2 | 7–17 | 1–3 | 1.2–2.0 | 0.02–0.3 | |
| γ-terpinene | 6–10 | - | - | 6–12 | 10–13 | 16–22 | ||
| sabinene | - | - | 0.1–0.6 | 1–3 | 0.1–0.3 | <0.3 | 0.2–1.1 | |
| α-pinene | - | 0.2–0.7 | 0.2–0.6 | - | 0.8–1.3 | 1.6–3 | 0.4–0.6 | |
| myrcene | - | 1.5–3 | 1.5–2.5 | 1.1–1.5 | 1.5–2.0 | 1.5–2.5 | ||
| p-cymene | - | - | 1.5–2.8 | <1.0 | ||||
| Oxygenated monoterpenes (monotepene alcohols) | γ-terpineol | - | - | - | - | 0.7–1.4 | - | - |
| α-terpineol | - | - | - | <0.6 | 6–8 | - | - | |
| linalool | 3–15 | 0.1–0.4 | - | - | - | - | 0.2–0.7 | |
| fenchol | - | - | - | - | 0.4–0.8 | - | - | |
| borneol | - | - | - | - | 0.5–0.8 | - | - | |
| Oxygenated monoterpenes (monoterpene aldehydes) | geranial | 0.25–0.5 | - | - | 0.5–2.3 | - | - | 0.03–0.2 |
| neral | - | - | 0.02–0.04 | 0.3–1.5 | - | - | 0.02–0.1 | |
| Oxygenated monoterpenes (monoterpene esters) | neryl acetate | - | - | - | 0.2–0.9 | - | - | - |
| linalyl acetate | 22–36 | 0.5–1 | - | - | - | - | ||
| geranyl acetate | - | 0.1–0.3 | - | 0.1–0.8 | - | - | - | |
| Sesquiterpene hydrocarbons | β-caryophyllene | - | t, 0.2 | 0.2–0.5 | <0.5 | 0.4–0.8 | - | - |
| germacrene D | - | t, 0.2 | - | - | - | - | - | |
| valencene | - | - | - | - | - | - | 0.02–0.5 | |
| β-bisabolene | 0.3–0.7 | - | - | - | 1–1.5 | - | - | |
| α-bergamotene | - | - | - | - | 0.5–0.9 | - | - | |
| α-farnesene | - | - | - | - | 0.6–0.9 | - | - | |
| Oxygenated sesquiterpenes | nootkatone | - | - | 0.01–0.8 | - | - | - | - |
| trans-β-nerolidol | - | t, 0.2 | - | - | - | - | - | |
| Aliphatic aldehydes (non-terpenoid) | nonanal | - | - | 0.04–0.1 | - | - | - | - |
| octanal | - | t, 0.1 | 0.2–0.8 | - | - | - | 0.1–0.4 | |
| decanal | - | t, 0.3 | 0.1–0.6 | - | - | - | 0.1–0.4 | |
| Nitrogen-containing aromatic compounds | methyl N-methylanthranilate | - | - | - | - | - | 0.3–0.6 | - |
| Reference source1 | [47] | [48] | [49] | [36] | [50] | [36] | [36] | |
D-limonene is the major component of EOs from the most important commercial citrus types (Table 2), and is responsible for the pleasant lemon-like aroma [51]. This compound has broad ecological role in plants, such as chemical defense of tissue and antifungal protection to name some of the most notable [52]. The body of research in the last 30 years on D-limonene (the main monoterpene in citrus EOs) is particularly rich, and research topics are widely concerned with its biologic activities (Table 3). The structure of this molecule, (R)-4-isopropenyl-1-methylcyclohexene, is characterized by a six-carbon ring with a carbon-carbon double bond and an isopropenyl side chain [51], conferring its lipophilic property, which is important for its biologic activity, especially in the interaction with microbial cell membranes [53]. This compound has a low toxicity [54] and was studied on model animals to prospect the potential development of therapeutic uses [55]. People can be exposed either intentionally or incidentally to D-limonene from cosmetics, additives, supplements, pesticides or flavored foodstuffs. This substance has a short half-life in humans ranging between 12–24 h and is excreted mainly through urine [54].
Table 3: Main biologic activities of D-limonene studied over last 30 years [53].
| Activity | Summarized Mode of Action | Potential Relevance |
|---|---|---|
| Antimicrobial | board-spectrum antibacterial activity; better inhibitory effect against Gram-positive than Gram-negative pathogens | adjuvant in overcoming or treating infections of antibiotic-resistant strains |
| Antioxidant | can enhance the activity of some key antioxidant enzymes in human body (superoxide dismutase, heme oxygenase-1); inhibits lipid peroxidation | protective effect for cells to oxidative damage |
| Anti-cancer | inducing apoptosis, inhibiting cell proliferation and angiogenesis; regulating multiple signaling pathway and autophagy-related pathways | chemoprevention, reducing cancer cell proliferation |
| Anti-inflammatory | inhibition of pro-inflammatory cytokines; reducing the levels of some facors and enzymes involved in inflmamtory response; inhibiting specific signal transduction pathways (of pro-inflamatory genes); | alleviating inflamation |
| Neuroprotective | inhibition of neuroinflamatory markers; regulation of neurotransmitter activity | brain cell protective effect; improvement in congnitive function |
Bergamot EO has a green to greenish-yellow color [4] and is distinguished by its elevated content of monoterpene esters compared to the other citruses (Table 2), with linalyl acetate conferring a floral-fruity scent note [56], in addition to linalool (Table 2) that contributes to the characteristic bergamot scent [57]. A study screened the antibacterial properties of bergamot EO against eight Listeria monocytogenes strains isolated from seafood, poultry and humans. There was observed a variable effectiveness of EO depending on the strain. The minimum inhibitory concentration ranged between 5 μL/mL to 0.625 μL/mL [58]. Distilled extract obtained from bergamot EO, was studied for the kinetics of antimicrobial activity against clinical isolates using a time-kill assay. Results demonstrated significant antimicrobial activity. The microscopic examination revealed compromised microbial cell integrity, including membrane damage and structural disorganization that caused bacterial and fungal cell death [59].
Bitter orange EO has a pale yellow or yellowish-brown color [4], and the volatile profile is strongly dominated by limonene with only a minor fraction of other compounds (Table 2). Bitter orange EO was tested against an eclectic selection of microorganisms, including foodborne, plant-pathogenic and environmental bacteria, highlighting the baseline antibacterial activity across different groups. The results showed that the best activity was against Pectobacterium carotovorum. Furthermore, vapor-phase EO effectively inhibited the spoilage bacteria in a food model (carrot) in a concentration-dependent manner. Overall the study indicates strain-dependent activity, but also a several-times-smaller effect compared to reference antibiotics [60], suggesting its potential rather as supporting or adjuvant antimicrobial role.
Grapefruit EO has a greenish-yellow color [4], and besides being limonene-rich, it is characterized by the presence of the oxygenated sesquiterpene nootkatone, which is responsible for the characteristic grapefruit scent [61], together with minor aliphatic aldehydes that further shape the scent profile, including octanal, described as having a fruity scent [62] and nonanal, associated with rose-orange notes [63]. A study has put in evidence a minimum inhibitory concentration of 2.5 mg/mL of the grapefruit EO against Escherichia coli, with a 2–8 times better effect compared to other citrus EOs tested [64].
Lemon EO has a yellow or pale greenish-yellow color [4], and in addition to limonene as a major component, it also presents higher levels of β-pinene (Table 2), a compound described as having a turpentine-like scent with dry, woody or resinous nuances [65]. A study on Eureka lemon indicated that peel EO obtained by cold-pressing shows higher antioxidant activity than the one obtained by hydro-distillation [66]. A comparative study has put in evidence that lemon EO displayed stronger bactericidal potential against Escherichia coli than other citrus types. The minimum bactericidal concentration against Escherichia coli was 10 mg/mL, lower than that of D-limonene or other citrus EOs tested [64].
Lime EO is colorless or pale yellow [4], displaying a more complex monoterpene profile with a lower proportion of limonene but a higher contribution of α-terpineol (oxygenated monoterpene) (Table 2), described as having a floral (lilac-like) scent [67]. A comparative study revealed that while key lime EO nano-emulsion demonstrated lower antibacterial activity than kaffir lime and particularly calamansi lime across the tested bacteria strains, the key lime EO nano-emulsion proved more stable over storage, showing the smallest reduction in inhibition zone after 1 month. This is relevant for incorporation in beverages or water-based food products as antimicrobial agents and for flavoring [68].
Mandarin EO has a greenish-yellow or reddish-yellow color [4], and besides high limonene content has notably higher levels of γ-terpinene, and the presence of the nitrogen-containing aromatic compound methyl N-methylanthranilate (Table 2); γ-terpinene is responsible for the lemon-like (citrusy) aroma [69]. The antimicrobial potential of EO from Nanfeng mandarins was assessed using the filter paper diffusion method against the most common food-borne pathogens. Results showed the inhibition zone diameter of EO against Pseudomonas aeruginosa was 7.93 mm and against Escherichia coli was 9.48 mm, exceeding ampicillin. Furthermore, the minimum inhibitory concentration against Bacillus subtilis was 6.25 μL/mL of EO, lower than ampicillin of 25 μL/mL, indicating a four times higher potency to inhibit this bacterium than the antibiotic. Regarding the antioxidant activity of mandarin EO, compared to the reference antioxidant butylated hydroxytoluene (BHT) it was lower; however, there were noted some interesting cultivar-dependent differences [70].
Sweet orange EO has a yellow or reddish-yellow color [4], and its limonene-rich profile has only a limited contribution of other compound classes (Table 2). The antimicrobial potential of EO from Gannan Newhall navel oranges was assessed using the filter paper diffusion method against five common food-borne pathogens. Results indicated an inhibition zone of 10.77 mm against Escherichia coli, exceeding ampicillin. The minimum inhibitory concentration of orange EO was 6.25 and 100 μg/mL against Bacillus subtilis and Pseudomonas aeruginosa respectively, both surpassing the effect of ampicillin [70].
The citrus peel EO is obtained by mechanical extraction (cold-pressing of peel) or distillation, the first one resulting in a higher quality and market price compared to the second one [4]. According to the European Pharmacopoeia, cold-pressing involves the expression of the EO from the pericarp (without heat) followed by the subsequent separation by physical methods. The steam distillation by comparison obtains the EO by the passage of water steam through the plant material, which condensates and is collected [36]. On a small-scale, EO can be obtained directly from the peel by hand-pressing, collecting in brine solution on ice followed by centrifugation at 4°C, then by dehydration of the supernatant with anhydrous sodium sulfate and finally by filtration [4]. Modern approaches use ultrasound-assisted extraction, microwave-assisted extraction and supercritical fluid extraction for increasing the efficiency of EO extraction from citrus pericarp [71].
For commercial purposes, there are three industrial approaches for EO extraction from Citrus peel:
- -recovery of EO from fruit waste after extraction of juice;
- -recovery of EO from flavedo after removal from fruit by abrasion or shaving;
- -simultaneous extraction of juice and EO emulsion from fruit [4].
Depending on the equipment used for the extraction, the general principle is that secretory glands from the Citrus pericarp are ruptured. This can happen before juice is extracted or during the extraction of juice from the fruit followed by spraying of water. Then, the water emulsion undergoes centrifugation. In the first stage, the product stream goes into the centrifuge, having an EO content of 0.5 to 2% and exits having 70–90% EO content. In the second stage of centrifugation this is further concentrated to 99%. However, the resulting EO retains dissolved wax residues that remain completely soluble in the EO at temperatures over 15–20°C. Through “winterization” the EO is held at temperatures of 1°C or lower for a varying duration up to 30 days, resulting in the precipitation and settling of the wax component that can be removed [72]. EO is to be recovered immediately due to changes in composition caused by activity of microorganisms [5].
After extraction, the citrus EOs are not always used directly in their native form. Considerable research attention is given to the development of modulating treatment of EO or delivery systems that are meant to improve specific properties, stability, handling and functional performance. Cold plasma treatment of citrus EO was shown to modulate and enhance secondary aroma notes that can further expand the flavor and fragrance applications [73]. Also, deterpenation techniques of citrus EOs enhance the oxygenated fractions, improving characteristics for food applications [74].
Citrus EOs are classified as GRAS (generally recognized as safe) and therefore used for flavoring. Furthermore, these are non-toxic, non-mutagenic and non-carcinogenic [75]. D-limonene, the main component of citrus EO, is safe for all animal species when used as a flavoring agent, except for male rats, at a maximum proposed dose level of 25 mg/kg feed [76].
While the use of citrus EO experienced an increase in cosmetics, there must be considered the challenges related to potential health-damaging effects of EO or its contamination. For decades, bergamot EO has been widely used in cosmetics due to its distinctive refreshing scent, but became restricted in most countries in later years due to phototoxicity and berloque dermatitis risk. In aromatherapy caution shall be considered for psoralen-containing citrus EOs [4]. However, aside from the moderate phototoxicity risk of bergamot and lime EOs, most commercial citrus types EOs have been assessed as not phototoxic or of low risk [75].
The contamination of citrus EO along the production and storage chain is an important safety concern. Firstly, some pesticide residues can persist on the fruit peel due to treatments applied to the crop. Another risk is related to the major plastic contaminants, such as phthalate esters, chloroparaffins and phosphorylated plasticizers during processing or storage. Other contaminants such as hypochlorous acid (HOCl) can have as source either the chlorine-treated water used in EO recovery process or various sanitizers used in the postharvest chain—this compound can further react with certain terpenes resulting in terpene chlorohydrin [4]. A study identified three terpene chlorohydrins in cold-pressed orange EO that occurred by reaction of hypochlorous acid with D-limonene [77].
The ISO 210:2023 provides the guidelines related to the packaging, conditioning and storage of EOs. These consider the potential reactivity of EO components with storage containers [78]. Citrus EO has a typical shelf life of 1–2 years. During storage, oxidation products increase in concentration. Quality depreciation of citrus peel EO may occur during storage due to evaporation, oxidation, polymerization, rearrangement and cyclization of some labile constituents in the presence of ambient factors (e.g., heat, light, oxygen, moisture). Following such processes, the occurrence of off-notes (alcohols, carbonyl compounds, esters, epoxide and hydrocarbons) alters the aroma characteristics. The main drivers of oxidation are autoxidation, photo-oxidation, lipoxygenase-assisted oxidation or thermal oxidation [4]. Particularly, the predominant compound of Citrus EOs, limonene, can generate some allergenic oxidation products during handling and storage such as R-(–)-carvone and R-(+)-limonene 2-hydroperoxide [54].
Cold-pressed citrus EO has been suggested as more stable than distilled EO due to the presence of some non-volatile components (e.g., polymethoxylated flavones and coumarins) that have antioxidative properties. Encapsulation represents an effective approach to protect EO from environmental factors and minimize loss of volatile flavor compounds. As for technique, citrus peel oil can be spray-dried and encapsulated in a double emulsion for cosmetic applications [4]. Optimized delivery systems become indispensable to meet the requirements of modern applications. An experimental study microencapsulated bitter orange EO in maltodextrin and whey protein concentrate, using the spout fluidized bed drying technique to obtain a dry powder comprised of microencapsulated particles that are suitable for handling, storage and controlled release [79]. A recent study successfully used electrospinning to incorporate orange EO into an antimicrobial starch-based fibrous delivery system suitable for packaging [80]. Research demonstrated that bergamot EO can be encapsulated in protein-polysaccharide nanocomplexes that result in enhanced physical stability and bioactivity that emphasizes their potential use as nanocarriers in pharmaceutical and food applications [81]. A study explored the conversion of bergamot EO into solid dosage forms optimized for pharmaceutical and nutraceutical applications [82]. Efficient delivery systems are becoming essential in translating the bioactivity of citrus EOs into more stable and safer applications in food, cosmetics and pharmaceutics.
4 Destinations of Use and Bio-Based Value Chain
4.1 Flavors and Other Food-Related Uses
Citrus peel EO as well as extracted EO finds wide application in food industry. The citrus peel can be used as such, being rich in EO and highly aromatic. The peel of lemon has about 2.5% EO [83]. The level of citrus flavor use is variable, ranging from 200 ppm in drinks to more than ten times more in chewing gum [6]. The grated lemon zest is used in cooking and for lemonades [84]. Cold-pressed citrus EOs are used in soft-drinks, sherbet, confectionary and bakery [4]. It is estimated that about 20% of confectionary (hard candy and boiled sweet) is citrus flavored [6]. Earl Gray tea is flavored with bergamot EO [85]. Carbonated soft drinks, commonly called “sodas” or “fizzy drinks”, can be flavored with citrus EOs. The lemon-lime and orange soft-drinks concentrates are the most commonly used. Citrus EOs are incorporated into proprietary flavor systems which are fractions often referred to as “parts” depending on brand and formulation, a common example being Cola Parts I, II, III or more [86]. Citrus flavoring contributes to a high percentage of all soft drinks produced in the world. The concentrated and terpeneless EOs are used both for their flavor effects and stability in the production of clear beverages [6]. Citrus peel EO is also used in pharmaceutical products to improve the taste of drugs [4].
Besides being a flavor enhancer, citrus EO has potential as a preservative agent of food products [5]. A study demonstrated the effectiveness of guar gum-chitosan edible films enriched with orange EO nano-emulsion 3:1, in the preservation of Inner Mongolian cheese. The results indicated the effectiveness of the packaging material to extend the shelf life and safety of cheese by improving physicochemical stability, inhibiting microbial growth and delaying lipid oxidation [87]. A study created biodegradable films with modified carioca bean starch and 10% orange peel EO, having antifungal properties for cake preservation. During nine days of storage, the cake covered in a package with EO exhibited higher fungal growth control compared to the film with no EO [88]. A study assessed the potential application of xanthan gum and pectin film enriched with orange peel EO in the preservation of meat. Results indicated an EO dose-dependent improvement in the mechanical strength and antioxidant activity, accompanied by enhanced radical scavenging capacity of the coating. After ten days of storage, the chicken meat covered with the bioactive film with 1.5% EO displayed a lower rate of pH increase, indicative of delayed spoilage [89]. Nano-fiber obtained from a blend of polymers (Eudragit and collagen) and functionalized with bitter orange EO, demonstrated parameters suitable for wide food packaging applications [90]. EO can be used as a reducing agent of peroxidase activity in stored leafy vegetables or as an antioxidant in edible oil. Furthermore, citrus EO can potentially inhibit the formation of N-nitrosodimethylamine (NDMA), a carcinogen that may occur in the processing or storage of food products [4].
The preservation of foodstuffs is becoming highly researched due to novel possibilities of application methods such as thin edible films and packaging, microencapsulation and nano-emulsion. However, some practical challenges remain and these are mainly related to the interaction between food products (e.g., meat proteins) and EO components that might result in the destabilization of volatile components, leading to the generation of undesired compounds or reduced effectiveness. Another concern is related to unpleasant organoleptic changes EO might bring to food stuffs [5]. The citrus EO applications for beverages bring different challenges not encountered in other food applications, due to low levels of EO used in water at low pH that may lead to reactions that impact flavor quality [6]. These challenges represent viable future research topics into optimizing citrus EO use for food and beverages.
4.2 Fragrance and Other Non-Food Uses
The fragrance of citrus peel EO finds a diverse non-food destination of use. In the fragrance industry, citrus is an important “top note”. The citrus note has low sillage and is created by a blend of EOs from citruses. It is suitable for either floral, woody, aldehyde or oriental bouquet [4] as presented in Table 4.
Table 4: Citrus EO destination based on fragrance [4].
| Citrus | Characteristics | Recommended Destination |
|---|---|---|
| bergamot | refreshing | perfumes including chypre and fougère |
| grapefruit | well in combination with spicy aromas | perfumes |
| lemon | most widely used | colognes |
| lime | well paired with other citrus or spices | masculine fragrances |
| orange | sweetest out of citruses | cosmetics and soaps |
Top luxury fragrance companies include citrus-scented products in their portfolios. One of the widely used websites for perfume information lists 955 fragrances in the “citrus” group across multiple brands and classified as either unisex or for men or women [91]. A recent market report further indicates that by application, the men segment is projected to maintain a larger share of the citrus scent perfume market, throughout the forecast interval of 2025–2033 [3].
Aromatherapy based on citrus EOs is used for relieving symptoms and increasing well-being, particularly associated with modulation of mood and stress, besides some physiological functions. The most common mediated way of aromatherapy is through inhalation and topic absorption through the skin. When inhaled, the volatiles are delivered to the central nervous system. The fragrant molecules activate the olfactory sensory neurons inside the nasal cavity and send signals to the regions in the brain involved in automatic regulation, emotion and cognition. At the level of respiratory epithelium, EO molecules can be absorbed into the systemic circulation [92]. Retailers recommend bergamot EO for massage therapy, orange and grapefruit for uplifting mood while lemon and lime aroma for stimulant effect over the digestive system [93]. Some citrus EO compounds were associated with different beneficial therapeutic effects relevant for aromatherapy, such as limonene, 1,8 cineole, α- and β-pinene, γ-terpinene, myrcene, p-cymene, linalool and camphene [92].
4.3 Citrus EO Value-Chain Timeline
Citrus is among the most important fruit trees in the world, with two clearly differentiated markets: fresh fruit and processed juice, with orange as predominant in both, followed by mandarins, lemons and limes and finally grapefruit [13]. The citrus value chain is linking vertically various actors from farmer to consumer and is linked horizontally with other value chains [94]. The Citrus EO bio-based value chain starts at the biologic level, where the genotype interacts with the environmental conditions and cultivation technology to determine fruit yield with its quality and peel characteristics (Fig. 2).
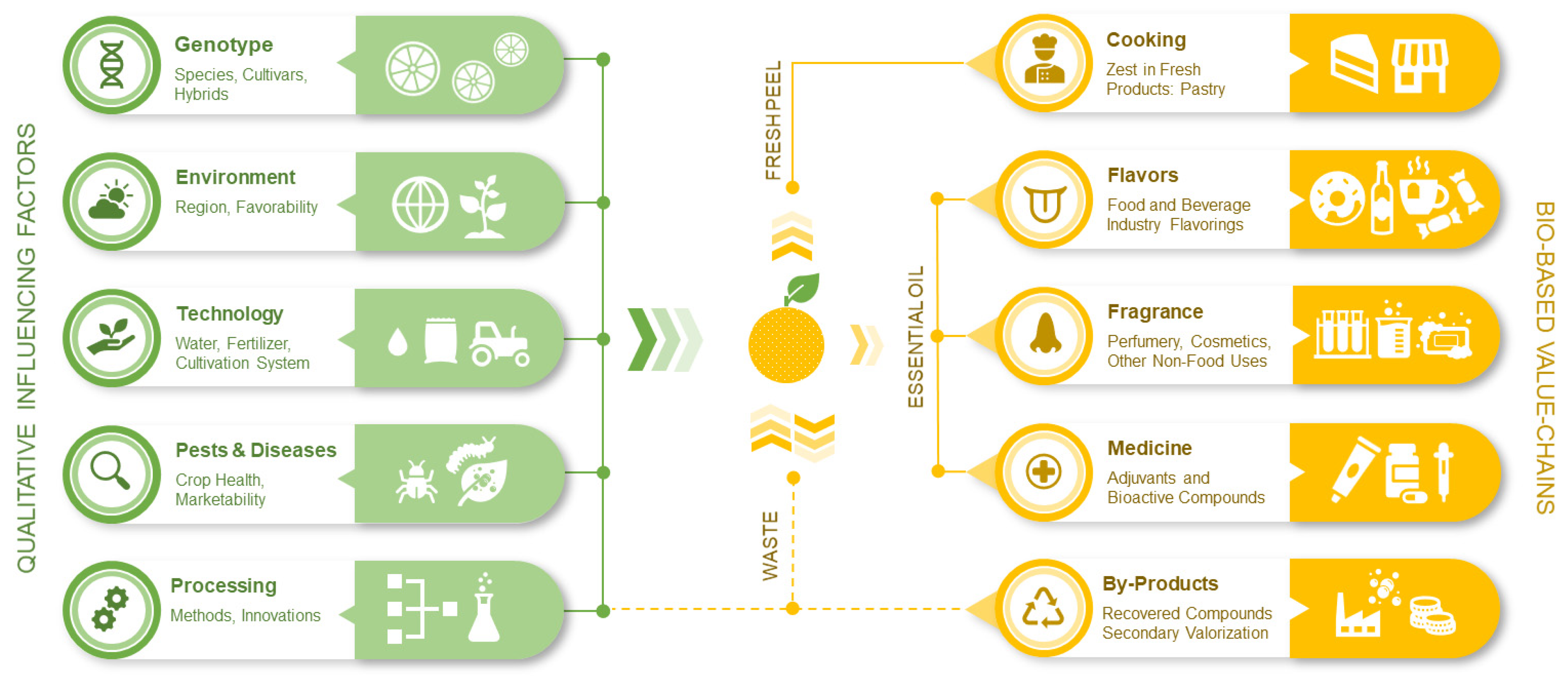
Figure 2: Summary of bio-based value chain of Citrus EO from agriculture to flavor and fragrance markets (original infographic by I.C.).
The composition of the citrus EOs is variable, under the influence of factors such as genotype (species, variety, cultivar), cultivation practices, extraction and separation methods [5]. A large part of raw material for EO extraction is generated by the citrus juice industry [95,96]. The citrus juice industry produces massive waste, accounting for 40–50% of the total fruit weight (comprised of peel and bagasse), and the extracted EO from citrus peel represents one the most important by-product of this Citrus processing industry [66].
The zest of the fresh citrus fruits contains EO that is responsible for the taste, explaining the wide use in bakery and lemonades [84]. Industrial flavor and fragrance applications are relying on EO. The first largest destination is soft drinks, followed secondly by confectionery [6]. In the beverage industry, citrus EOs are typically supplied by large soft-drink licensing companies as concentrate formulations to smaller licensed bottling companies [86]. The residual processing streams present increasing opportunities for the valorization of recovered compounds that anchors this value chain into the bioeconomic framework. The various fractions of peel waste and residue are rich in compounds that can be recovered and used across various industries [95,97,98,99,100].
While consumer trends are in favor of an increased market of citrus flavors and fragrances, the main risks and threats are related to the unavailability or quality of raw materials. These stem from agronomy factors, such as unpredictable weather conditions or fruit diseases, but also from non-compliance with standards and regulations that can lead to recalls and industry restrictions, or thirdly could be impacted by trade politics [2]. Certification schemes remain powerful approaches that can play a role in transnational regulation of the global citrus supply chain. These overcome the limited power of national governments in enforcing conditions of production abroad and therefore remain important leverage points in ensuring quality [101]. As citrus EOs are experiencing increased use as flavoring agents and as their consumption rises, it is expected that requirements for purity and stability will become more strict [6]. Novel directions are shaped by digital shopping, interactive experiences and traceability [2].
Despite increasing demand, with both flavor [2] and fragrance [3] markets projected to increase, a recent report showed that for 2024/2025, production declined for major citrus types across important exporting countries, mainly due to unfavorable weather events that impacted the crops [102]. Furthermore, it has been outlined that a smarter and efficient planning of the production and distribution systems in the citrus supply chain is needed [94]. In this regard, increasing demand might raise prices if the production declines in the following years. For this reason, agronomic aspects must be examined and addressed to ensure a steady and reliable supply of citrus raw materials into the EO value chain.
Flavedo is a region of the citrus fruit peel that presents the secretory structures responsible for EO biosynthesis and accumulation. The flavedo constitutes the critical anatomical interface that connects botany to industrial valorization of citrus EO. The aroma of EO is the basis of fragrance and flavor use.
Citrus EO composition is dominated by D-limonene, a monoterpene hydrocarbon having a generic citrusy aroma. However, its presence may vary widely from less than 50% from EO composition (bergamot, lime) to over 90% (orange and grapefruit). This major compound is complemented by other constituents that confer the specific aroma identity to varying citrus types (e.g., nootkatone, linalool).
Bioactivity of EOs complements their primary flavor and fragrance functions. The first major destination of use for the EO is flavoring of soft drinks (lime, lemon, orange) followed by confectionary. The food industry is sourcing the bioactivity and functional properties for both preservation and health. The fragrance industry is an important destination for EOs, with a projected market increase over the next decade.
The value chain is enjoying the opportunity of abundant raw material (fruit peels) from the juice industry, with citrus EO representing the most valuable by-product of this industry. Evidence on the botany, phytochemistry and applications highlights citrus EO as a valuable plant-derived resource that sustains food, fragrance and other bio-based related value chains. Recent challenges are related to some environmental and agronomic factors that could impact sustainability.
Acknowledgement:
Funding Statement: The author received no specific funding for this study.
Availability of Data and Materials: Not applicable.
Ethics Approval: Not applicable.
Conflicts of Interest: The author declares no conflicts of interest.
Table A1: Citrus genus species and hybrids sensu POWO (alphabetic) [23].
| Citrus | Native Range1 | Notes |
|---|---|---|
| Citrus assamensis R.M.Dutta & Bhattacharya | Assam to Bangladesh | |
| Citrus × aurantiifolia (Christm.) Swingle | E. Himalaya | cultigen |
| Citrus × aurantium L. | S. China | cultigen |
| Citrus australasica F.Muell. | Australia (SE. Queensland to NE. New South Wales) | |
| Citrus australis (Mudie) Planch. | Australia (SE. Queensland) | |
| Citrus cavaleriei H.Lév. ex Cavalerie | Central China (to NW. and W. Hunan) | |
| Citrus × depressa Hayata* | East Asia (intoduced) | natural hybrid |
| Citrus garrawayae F.M.Bailey | New Guinea (Goodenough Island), Australia (Queensland) | |
| Citrus glauca (Lindl.) Burkill | Australia (Queensland to New South Wales) | |
| Citrus gracilis Mabb. | Northern Territory | |
| Citrus halimii B.C.Stone | S. Thailand to Peninsula Malaysia, N. Borneo. | |
| Citrus hystrix DC. | E. Himalaya to China (Yunnan, N. Guangxi) and Wallis Island | |
| Citrus indica Tanaka | Assam to Bangladesh | |
| Citrus inodora F.M.Bailey | Australia (Queensland) | |
| Citrus japonica Thunb. | SE. China to Hainan | |
| Citrus khasya Markovitch | Sikkim to Bangladesh | |
| Citrus latipes (Swingle) Tanaka | Assam to N. Myanmar | |
| Citrus × limon (L.) Osbeck | - | cultigen |
| Citrus mangshanensis S.W.He & G.F.Liu | China (Hunan: S. Nanling Mountains) | |
| Citrus maxima (Burm.) Merr. | Assam to Indo-China | |
| Citrus medica L. | W. Central Himalaya to Myanmar | |
| Citrus neocaledonica Guillaumin | New Caledonia | |
| Citrus oxanthera Beauvis. | New Caledonia | |
| Citrus polyandra Tanaka | NE. New Guinea to Bismarck Archipelago | |
| Citrus polytrifolia Govaerts | China (SE. Yunnan) | |
| Citrus pubinervia D.G.Zhang, Z.H.Xiang & Y.Wu | China (SW. Hubei, NW. Hunan) | |
| Citrus reticulata Blanco | China (Guangxi to Hunan and Jiangxi) | |
| Citrus swinglei Burkill ex Harms | Hainan Malaysia | |
| Citrus trifoliata L. | Central and S. China | |
| Citrus undulata Guillaumin | New Caledonia | |
| Citrus wakonai P.I.Forst. & M.W.Sm. | Papua New Guinea | |
| Citrus warburgiana F.M.Bailey | Papua New Guinea | |
| Citrus wintersii Mabb. | Papua New Guinea |
References
1. Zhong G , Nicolosi E . Citrus origin, diffusion, and economic importance. In: Gentile A , La Malfa S , Deng Z , editors. The Citrus genome. Cham, Switzerland: Springer International Publishing; 2020. p. 5– 21. doi:10.1007/978-3-030-15308-3_2. [Google Scholar] [CrossRef]
2. Future Market Insights . Citrus flavors market forecast and outlook 2026 to 2036. Newark, DE, USA: Future Market Insights, Inc.; 2026. [cited 2026 Feb 28]. Available from: https://www.futuremarketinsights.com/reports/citrus-flavours-market [Google Scholar]
3. Gore A . Citrus scent perfume market analysis 2025. Pune, India: Cognitive Market Research; 2025. [cited 2025 Dec 15]. Available from: https://www.cognitivemarketresearch.com/citrus-scent-perfume-market-report [Google Scholar]
4. Shahidi F , Zhong Y . Citrus oils and essences. kirk-othmer encyclopedia of chemical technology. Hoboken, NJ, USA: John Wiley & Sons, Ltd.; 2012, p. 1– 17. doi:10.1002/0471238961.citrshah.a01. [Google Scholar] [CrossRef]
5. Mahato N , Sharma K , Koteswararao R , Sinha M , Baral E , Cho MH . Citrus essential oils: Extraction, authentication and application in food preservation. Crit Rev Food Sci Nutr. 2019; 59( 4): 611– 25. doi:10.1080/10408398.2017.1384716. [Google Scholar] [CrossRef]
6. Colombo E , Ghizzoni C , Cagni D . Citrus oils in food and beverages: Uses and analyses. In: Citrus. Boca Raton, FL, USA: CRC Press; 2002. [Google Scholar]
7. El-Shemy HA . Potential of essential oils. London, UK: InTech; 2018. doi:10.5772/intechopen.69939. [Google Scholar] [CrossRef]
8. Hausch BJ , Lorjaroenphon Y , Cadwallader KR . Flavor chemistry of lemon-lime carbonated beverages. J Agric Food Chem. 2015; 63( 1): 112– 9. doi:10.1021/jf504852z. [Google Scholar] [CrossRef]
9. Haokip SW , Sheikh KA , Das S , Devi OB , Singh YD , Wangchu L , et al. Unraveling physicochemical profiles and bioactivities of Citrus peel essential oils: A comprehensive review. Eur Food Res Technol. 2023; 249( 11): 2821– 34. doi:10.1007/s00217-023-04330-w. [Google Scholar] [CrossRef]
10. Curk F , Luro F , Hussain S , Ollitrault P . Citrus origins. In: Citrus production. Boca Raton, FL, USA: CRC Press; 2022. doi:10.1201/9781003119852-1. [Google Scholar] [CrossRef]
11. Wu GA , Terol J , Ibanez V , López-García A , Pérez-Román E , Borredá C , et al. Genomics of the origin and evolution of Citrus. Nature. 2018; 554( 7692): 311– 6. doi:10.1038/nature25447. [Google Scholar] [CrossRef]
12. Talon M , Caruso M , Gmitter FG Jr . The genus Citrus. Duxford, UK: Woodhead Publishing; 2020. [Google Scholar]
13. Ollitrault P , Navarro L . Citrus. In: Badenes ML , Byrne DH , editors. Fruit breeding. Boston, MA, USA: Springer US; 2011. p. 623– 62. doi:10.1007/978-1-4419-0763-9_16. [Google Scholar] [CrossRef]
14. Hussain SZ , Naseer B , Qadri T , Fatima T , Ahmad Bhat T . Citrus Fruits—Morphology, taxonomy, composition and health benefits. In: Hussain SZ , Naseer B , Qadri T , Fatima T , Bhat TA , editors. Fruits grown in highland regions of the himalayas: Nutritional and health benefits. Cham, Switzerland: Springer International Publishing; 2021. p. 229– 44. doi:10.1007/978-3-030-75502-7_18. [Google Scholar] [CrossRef]
15. Mabberley DJ . Citrus: A world history. London, UK: Thames & Hudson; 2024. [Google Scholar]
16. Ramon-Laca L . The introduction of cultivated Citrus to Europe via Northern Africa and the Iberian Peninsula. Econ Bot. 2003; 57( 4): 502– 14. doi:10.1663/0013-0001(2003)057[0502:TIOCCT]2.0.CO;2. [Google Scholar] [CrossRef]
17. Pagnoux C , Celant A , Coubray S , Fiorentino G , Zech-Matterne V . The introduction of Citrus to Italy, with reference to the identification problems of seed remains. Veg Hist Archaeobot. 2013; 22( 5): 421– 38. doi:10.1007/s00334-012-0389-4. [Google Scholar] [CrossRef]
18. Simpson MG . Plant systematics. Burlington, MA, USA: Academic Press; 2010. doi:10.1016/B978-0-12-374380-0.50001-4. [Google Scholar] [CrossRef]
19. Ortiz JM . Botany: Taxonomy, morphology and physiology of fruits, leaves and flowers. In: Citrus. Boca Raton, FL, USA: CRC Press; 2002. [Google Scholar]
20. Luro F , Curk F , Froelicher Y , Ollitrault P . Recent insights on Citrus diversity and phylogeny. In: Agrumed: Archaeology and history of citrus fruit in the Mediterranean: Acclimatization, diversifications, uses. Naples, Italy: Publications du Centre Jean Bérard; 2017. doi:10.4000/books.pcjb.2169. [Google Scholar] [CrossRef]
21. Wang X , Xu Y , Zhang S , Cao L , Huang Y , Cheng J , et al. Genomic analyses of primitive, wild and cultivated Citrus provide insights into asexual reproduction. Nat Genet. 2017; 49( 5): 765– 72. doi:10.1038/ng.3839. [Google Scholar] [CrossRef]
22. Ollitrault P , Curk F , Krueger R . Citrus taxonomy. In: The genus Citrus. Amsterdam, The Netherlands: Elsevier; 2020; p. 57– 81. doi:10.1016/b978-0-12-812163-4.00004-8. [Google Scholar] [CrossRef]
23. Citrus L. Plants of the World Online | Kew Science. Plants of the World Online; n.d. [cited 2025 Dec 10]. Available from: http://powo.science.kew.org/taxon/urn:lsid:ipni.org:names:30022289-2. [Google Scholar]
24. Zhang D , Mabberley DJ . eFloras. Flora China. 2008; 11: 52– 94. [cited 2025 Dec 15]. Available from: http://www.efloras.org/florataxon.aspx?flora_id=2&taxon_id=107164 [Google Scholar]
25. Ladaniya M . Citrus fruit: Biology, technology and evaluation. London, UK: Academic Press; 2008. [Google Scholar]
26. Stoie A , Vârban R . Botany: Plant morphology and anatomy. Cluj-Napoca, Romania: Academic Press; 2019. [Google Scholar]
27. Katz E , Lagunes PM , Riov J , Weiss D , Goldschmidt EE . Molecular and physiological evidence suggests the existence of a system II-like pathway of ethylene production in non-climacteric Citrus fruit. Planta. 2004; 219( 2): 243– 52. doi:10.1007/s00425-004-1228-3. [Google Scholar] [CrossRef]
28. Barrett HC , Rhodes AM . A numerical taxonomic study of affinity relationships in cultivated Citrus and its close relatives. Syst Bot. 1976; 1( 2): 105. doi:10.2307/2418763. [Google Scholar] [CrossRef]
29. Nicolosi E , Deng ZN , Gentile A , La Malfa S , Continella G , Tribulato E . Citrus phylogeny and genetic origin of important species as investigated by molecular markers. Theor Appl Genet. 2000; 100( 8): 1155– 66. doi:10.1007/s001220051419. [Google Scholar] [CrossRef]
30. Wu GA , Prochnik S , Jenkins J , Salse J , Hellsten U , Murat F , et al. Sequencing of diverse mandarin, pummelo and orange genomes reveals complex history of admixture during Citrus domestication. Nat Biotechnol. 2014; 32( 7): 656– 62. doi:10.1038/nbt.2906. [Google Scholar] [CrossRef]
31. Jing L , Lei Z , Zhang G , Pilon AC , Huhman DV , Xie R , et al. Metabolite profiles of essential oils in Citrus peels and their taxonomic implications. Metabolomics. 2015; 11( 4): 952– 63. doi:10.1007/s11306-014-0751-x. [Google Scholar] [CrossRef]
32. Ollitrault P , Jaquemond C , Dubois C , Luro F . Citrus. In: Genetic diversity of cultivated tropical plants. Paris, France: Science Publishers Inc.; 2003. p. 193– 217. [Google Scholar]
33. Tanaka T . Misunderstanding with regards citrus classification and nomeclature. Bull Univ Osaka Pref Ser B. 1969; 1: 139– 45. doi:10.24729/00009479. [Google Scholar] [CrossRef]
34. Help, Advice & Tips from the RHS on All Kinds of Plants/RHS n.d. [cited 2025 Dec 22]. Available from: http://www.rhs.org.uk/plants/. [Google Scholar]
35. ISO 4720;2018(en):Essential Oils—Nomenclature n.d. [cited 2025 Dec 22]. Available from: https://www.iso.org/obp/ui/. [Google Scholar]
36. EDQM . European Pharmacopoeia. Vol. 1. 11th ed. Strasbourg, France: European Directorate for the Quality of Medicines & HealthCare of the Council of Europe; 2023. [Google Scholar]
37. Satyawada RR , Hynniewta M , Malik SK . Karyological studies in ten species of Citrus (Linnaeus, 1753) (Rutaceae) of north-east India. Comp Cytogenet. 2011; 5( 4): 277– 87. doi:10.3897/compcytogen.v5i4.1796. [Google Scholar] [CrossRef]
38. Rodrigo MJ , Alquézar B , Alós E , Lado J , Zacarías L . Biochemical bases and molecular regulation of pigmentation in the peel of Citrus fruit. Sci Hortic. 2013; 163: 46– 62. doi:10.1016/j.scienta.2013.08.014. [Google Scholar] [CrossRef]
39. Kaur R , Kaur N , Singh H . Pericarp and pedicel anatomy in relation to fruit cracking in lemon (Citrus limon L Burm.). Sci Hortic. 2019; 246: 462– 8. doi:10.1016/j.scienta.2018.11.040. [Google Scholar] [CrossRef]
40. Gao S , Kang H , An X , Cheng Y , Chen H , Chen Y , et al. Non-destructive storage time prediction of newhall navel oranges based on the characteristics of rind oil glands. Front Plant Sci. 2022; 13: 811630. doi:10.3389/fpls.2022.811630. [Google Scholar] [CrossRef]
41. Cocco E , Giorgi G , Marsigliesi V , Mura F , Alves-Silva JM , Zuzarte M , et al. Histology of Pompia peel and bioactivity of its essential oil: A new Citrus-based approach to skin regeneration. Pharmaceuticals. 2025; 18( 9): 1256. doi:10.3390/ph18091256. [Google Scholar] [CrossRef]
42. Liang SJ , Wu H , Lun X , Lu DW . Secretory cavity development and its relationship with the accumulation of essential oil in fruits of Citrus medica L. var. sarcodactylis (Noot.) Swingle. J Integr Plant Biol. 2006; 48( 5): 573– 83. doi:10.1111/j.1744-7909.2006.00230.x. [Google Scholar] [CrossRef]
43. Knight TG . Structural basis of the rind disorder oleocellosis in Washington navel orange (Citrus sinensis L. Osbeck). Ann Bot. 2002; 90( 6): 765– 73. doi:10.1093/aob/mcf258. [Google Scholar] [CrossRef]
44. Jentzsch M , Albiez V , Kardamakis TC , Speck T . Analysis of the peel structure of different Citrus spp. via light microscopy, SEM and μCT with manual and automatic segmentation. Soft Matter. 2024; 20( 12): 2804– 11. doi:10.1039/d3sm01511d. [Google Scholar] [CrossRef]
45. Jentzsch M , Becker S , Thielen M , Speck T . Functional anatomy, impact behavior and energy dissipation of the peel of Citrus × limon: A comparison of Citrus × limon and Citrus maxima. Plants. 2022; 11( 7): 991. doi:10.3390/plants11070991. [Google Scholar] [CrossRef]
46. González-Mas MC , Rambla JL , López-Gresa MP , Blázquez MA , Granell A . Volatile compounds in Citrus Essential oils: A comprehensive review. Front Plant Sci. 2019; 10: 12. doi:10.3389/fpls.2019.00012. [Google Scholar] [CrossRef]
47. ISO 3520:2022. ISO n.d. [cited 2025 Dec 23]. Available from: https://www.iso.org/standard/81602.html. [Google Scholar]
48. ISO 9844:2006. ISO n.d. [cited 2025 Dec 23]. Available from: https://www.iso.org/standard/37053.html. [Google Scholar]
49. ISO 3053:2004. ISO n.d. [cited 2025 Dec 23]. Available from: https://www.iso.org/standard/32040.html. [Google Scholar]
50. ISO 3519:2005. ISO n.d. [cited 2025 Dec 23]. Available from: https://www.iso.org/standard/37623.html. [Google Scholar]
51. PubChem . (+)-Limonene n.d. [cited 2025 Dec 24]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/440917. [Google Scholar]
52. Erasto P , Viljoen AM . Limonene—A review: Biosynthetic, ecological and pharmacological relevance. Nat Prod Commun. 2008; 3( 7): 1934578X0800300728. doi:10.1177/1934578x0800300728. [Google Scholar] [CrossRef]
53. Wang H , Lu Q , Chen X , Qian Y , Qian B , Tan H . Global trends and biological activity hotspots of d-limonene in essential oils: A 30-year bibliometric study. Naunyn Schmiedebergs Arch Pharmacol. 2025; 398( 5): 5491– 507. doi:10.1007/s00210-024-03607-5. [Google Scholar] [CrossRef]
54. Kim YW , Kim MJ , Chung BY , Bang DY , Lim SK , Choi SM , et al. Safety evaluation and risk assessment of d-limonene. J Toxicol Environ Health Part B. 2013; 16( 1): 17– 38. doi:10.1080/10937404.2013.769418. [Google Scholar] [CrossRef]
55. Anandakumar P , Kamaraj S , Vanitha MK . D-limonene: A multifunctional compound with potent therapeutic effects. J Food Biochem. 2021; 45: e13566. doi:10.1111/jfbc.13566. [Google Scholar] [CrossRef]
56. PubChem . Linalyl Acetate n.d. [cited 2025 Dec 25]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/8294. [Google Scholar]
57. PubChem . Linalool n.d. [cited 2025 Dec 24]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/6549. [Google Scholar]
58. Marotta SM , Giarratana F , Parco A , Neri D , Ziino G , Giuffrida A , et al. Evaluation of the antibacterial activity of bergamot essential oils on different Listeria monocytogenes strains. Ital J Food Saf. 2016; 5: 6176. doi:10.4081/ijfs.2016.6176. [Google Scholar] [CrossRef]
59. Quirino A , Giorgi V , Palma E , Marascio N , Morelli P , Maletta A , et al. Citrus bergamia: Kinetics of antimicrobial activity on clinical isolates. Antibiotics. 2022; 11( 3): 361. doi:10.3390/antibiotics11030361. [Google Scholar] [CrossRef]
60. Kačániová M , Verešová A , Čmiková N . The antimicrobial activity of Citrus aurantium amara. Sci Pap Anim Sci Biotechnol. 2024; 57: 32– 7. [Google Scholar]
61. PubChem . (+)-Nootkatone n.d. [cited 2025 Dec 24]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/1268142. [Google Scholar]
62. PubChem . Octanal n.d. [cited 2025 Dec 24]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/454. [Google Scholar]
63. PubChem . Nonanal n.d. [cited 2025 Dec 24]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/31289. [Google Scholar]
64. Li Y , Liu S , Zhao C , Zhang Z , Nie D , Tang W , et al. The chemical composition and antibacterial and antioxidant activities of five Citrus essential oils. Molecules. 2022; 27( 20): 7044. doi:10.3390/molecules27207044. [Google Scholar] [CrossRef]
65. PubChem . Beta-Pinene n.d. [cited 2025 Dec 24]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/14896. [Google Scholar]
66. Lu Q , Huang N , Peng Y , Zhu C , Pan S . Peel oils from three Citrus species: Volatile constituents, antioxidant activities and related contributions of individual components. J Food Sci Technol. 2019; 56( 10): 4492– 502. doi:10.1007/s13197-019-03937-w. [Google Scholar] [CrossRef]
67. PubChem . Alpha-Terpineol n.d. [cited 2025 Dec 24]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/17100. [Google Scholar]
68. Liew SN , Utra U , Alias AK , Tan TB , Tan CP , Yussof NS . Physical, morphological and antibacterial properties of lime essential oil nanoemulsions prepared via spontaneous emulsification method. LWT. 2020; 128: 109388. doi:10.1016/j.lwt.2020.109388. [Google Scholar] [CrossRef]
69. PubChem . Gamma-Terpinene n.d. [cited 2025 Dec 24]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/7461. [Google Scholar]
70. Lin X , Cao S , Sun J , Lu D , Zhong B , Chun J . The chemical compositions, and antibacterial and antioxidant activities of four types of Citrus essential oils. Molecules. 2021; 26( 11): 3412. doi:10.3390/molecules26113412. [Google Scholar] [CrossRef]
71. Sangeeta A , Gopalakrishnan K , Mishra P . New food product development from Citrus fruits. In: Gupta AK , Kour J , Mishra P , editors. Citrus fruits and juice: Processing and quality profiling. Singapore: Springer Nature; 2024. p. 223– 58. doi:10.1007/978-981-99-8699-6_10. [Google Scholar] [CrossRef]
72. Sharma R , Nanda R , Bhagat N . Citrus flavours. In: Gupta AK , Kour J , Mishra P , editors. Citrus fruits and juice: Processing and quality profiling. Singapore: Springer Nature; 2024. p. 275– 99. doi:10.1007/978-981-99-8699-6_12. [Google Scholar] [CrossRef]
73. Fernandes FAN , Maia DLH , Canuto KM , de Brito ES . Aroma modulation of limonene-rich essential oil using cold plasma technology. Plasma Chem Plasma Process. 2025; 45( 6): 1925– 44. doi:10.1007/s11090-025-10585-w. [Google Scholar] [CrossRef]
74. Türkmenoğlu A , Özmen D . Deterpenation techniques of Citrus essential oils and the role of oxyterpenes in food industry. Flavour Fragr J. 2023; 38( 6): 407– 15. doi:10.1002/ffj.3758. [Google Scholar] [CrossRef]
75. Dosoky NS , Setzer WN . Biological activities and safety of Citrus spp. essential oils. Int J Mol Sci. 2018; 19( 7): 1966. doi:10.3390/ijms19071966. [Google Scholar] [CrossRef]
76. EFSA Panel on Additives and Products or Substances used in Animal Feed (FEEDAP). Scientific opinion on the safety and efficacy of aliphatic and aromatic hydrocarbons (chemical group 31) when used as flavourings for all animal species. EFSA J. 2015; 13: 4053. doi:10.2903/j.efsa.2015.4053. [Google Scholar] [CrossRef]
77. Weiss ER , Pika J , Braddock RJ . Isolation and identification of terpene chlorohydrins found in cold-pressed orange oil. J Agric Food Chem. 2003; 51( 8): 2277– 82. doi:10.1021/jf026122n. [Google Scholar] [CrossRef]
78. ISO 210:2023. ISO n.d. [cited 2025 Dec 9]. Available from: https://www.iso.org/standard/83777.html. [Google Scholar]
79. Sharifpour A , Dakheli MJ , Rahimi S , Bassiri A . Optimization of bitter orange (Citrus aurantium L.) essential oil microencapsulation through spout fluidized bed drying. J Food Meas Charact. 2025; 19( 1): 89– 107. doi:10.1007/s11694-024-02948-2. [Google Scholar] [CrossRef]
80. dos Santos FN , de Souza EJD , Pires JB , Crizel RL , da Cruz EP , Kroning IS , et al. Orange peel essential oil in rice starch encapsulating material for antimicrobial application against Escherichia coli. Int J Biol Macromol. 2025; 289: 138955. doi:10.1016/j.ijbiomac.2024.138955. [Google Scholar] [CrossRef]
81. Rasheed HA , Rehman A , Li C , Bai M , Karim A , Dai J , et al. Fabrication of Citrus bergamia essential oil-loaded sodium caseinate/peach gum nano complexes: Physicochemical, spectral, and structural characterization. Int J Biol Macromol. 2024; 260: 129475. doi:10.1016/j.ijbiomac.2024.129475. [Google Scholar] [CrossRef]
82. Zambito Y , Piras AM , Fabiano A . Bergamot essential oil: A method for introducing it in solid dosage forms. Foods. 2022; 11( 23): 3860. doi:10.3390/foods11233860. [Google Scholar] [CrossRef]
83. Chevallier A . Encyclopedia of herbal medicine. 4th ed. Munich, Germany: Dorling Kinderslay; 2023. [Google Scholar]
84. Sonneman T . Lemon: A global history. London, UK: Reaktion Books; 2013. [Google Scholar]
85. Navarra M , Mannucci C , Delbò M , Calapai G . Citrus bergamia essential oil: From basic research to clinical application. Front Pharmacol. 2015; 6: 36. doi:10.3389/fphar.2015.00036. [Google Scholar] [CrossRef]
86. Ameh SJ , Obodozie-Ofoegbu O . Essential oils as flavors in carbonated cola and citrus soft drinks. In: Essential oils in food preservation, flavor and safety. Amsterdam, The Netherlands: Academic Press; 2016, p. 111– 21. doi:10.1016/B978-0-12-416641-7.00011-0. [Google Scholar] [CrossRef]
87. Cai R , Jia L , Yang R , Tao H , Cui H , Lin L , et al. Fabrication of guar gum/chitosan edible films reinforced with orange essential oil nanoemulsion for cheese preservation. Int J Biol Macromol. 2025; 285: 138285. doi:10.1016/j.ijbiomac.2024.138285. [Google Scholar] [CrossRef]
88. Lemos GS , Vitoria JS , Fonseca LM , Pires JB , da Silva FT , Siebeneichler TJ , et al. Active food packages for cake conservation: Antifungal potential of bean starch biodegradable films with orange peel essential oil. Int J Biol Macromol. 2025; 310: 143441. doi:10.1016/j.ijbiomac.2025.143441. [Google Scholar] [CrossRef]
89. Dalei G , Das S , Mohanty D , Biswal S , Jena D , Dehury P , et al. Xanthan gum-pectin edible coating enriched with sweet orange (Citrus sinensis L.) peel essential oil for chicken meat preservation. Food Biophys. 2025; 20( 1): 52. doi:10.1007/s11483-025-09949-8. [Google Scholar] [CrossRef]
90. Talib Al-Sudani B , Khoshkalampour A , Kamil MM , Al-Musawi MH , Mohammadzadeh V , Ahmadi S , et al. A novel antioxidant and antimicrobial food packaging based on Eudragit ®/collagen electrospun nanofiber incorporated with bitter orange peel essential oil. LWT. 2024; 193: 115730. doi:10.1016/j.lwt.2024.115730. [Google Scholar] [CrossRef]
91. Fragrantica . Citrus—Perfume Group 2025. [cited 2025 Dec 24]. Available from: https://www.fragrantica.com/groups/citrus.html. [Google Scholar]
92. Agarwal P , Sebghatollahi Z , Kamal M , Dhyani A , Shrivastava A , Singh KK , et al. Citrus Essential oils in aromatherapy: Therapeutic effects and mechanisms. Antioxidants. 2022; 11( 12): 2374. doi:10.3390/antiox11122374. [Google Scholar] [CrossRef]
93. Hill DK . The dōTERRA® essential oil chemistry handbook. 3rd ed. Pleasant Grove, UT, USA: dōTERRA International, LLC; 2024. [Google Scholar]
94. Haque EU , Hayat A , Asim M , Afzaal S , Hanif MS , Murtaza MA , et al. The supply and value chain of citrus fruit: Producer to consumer. In: Citrus production. 1st ed. Boca Raton, FL, USA: CRC Press; 2022. doi:10.1201/9781003119852-26. [Google Scholar] [CrossRef]
95. Ahmed A , Imran A , Afzaal M , Rasheed M , Chauhan A , Islam F . Introduction of Citrus waste utilization. In: Chauhan A , Islam F , Imran A , Singh Aswal J , editors. Valorization of citrus food waste. Cham, Switzerland: Springer Nature; 2025. p. 1– 9. doi:10.1007/978-3-031-77999-2_1. [Google Scholar] [CrossRef]
96. Ben Hsouna A , Sadaka C , Generalić Mekinić I , Garzoli S , Švarc-Gajić J , Rodrigues F , et al. The chemical variability, nutraceutical value, and food-industry and cosmetic applications of Citrus Plants: A critical review. Antioxidants. 2023; 12( 2): 481. doi:10.3390/antiox12020481. [Google Scholar] [CrossRef]
97. Gomez-Urios C , Siroli L , Grassi S , Patrignani F , Blesa J , Lanciotti R , et al. Sustainable valorization of Citrus by-products: Natural deep eutectic solvents for bioactive extraction and biological applications of Citrus sinensis peel. Eur Food Res Technol. 2025; 251( 7): 1965– 80. doi:10.1007/s00217-025-04757-3. [Google Scholar] [CrossRef]
98. Negrea M , Cocan I , Jianu C , Alexa E , Berbecea A , Poiana MA , et al. Valorization of Citrus Peel byproducts: A sustainable approach to nutrient-rich jam production. Foods. 2025; 14( 8): 1339. doi:10.3390/foods14081339. [Google Scholar] [CrossRef]
99. Ram B , Jadhav HB , Shewale SR , Annapure US . Demonstrating the sustainable approach in valorizing the Citrus limetta peel. Food Chem Adv. 2025; 9: 101171. doi:10.1016/j.focha.2025.101171. [Google Scholar] [CrossRef]
100. Stavrakakis I , Melidis P , Kavroulakis N , Goliomytis M , Simitzis P , Ntougias S . Bioeconomy-based approaches for the microbial valorization of Citrus Processing waste. Microorganisms. 2025; 13( 8): 1891. doi:10.3390/microorganisms13081891. [Google Scholar] [CrossRef]
101. Mook A , Overdevest C . What drives market construction for fair trade, organic, and GlobalGAP certification in the global Citrus value chain? Evidence at the importer level in the Netherlands and the United States. Bus Strat Environ. 2021; 30( 7): 2996– 3008. doi:10.1002/bse.2784. [Google Scholar] [CrossRef]
102. USDA. Citrus: World markets and trade. Washington, DC, USA: United States Department of Agriculture—Foreign Agricultural Service; 2025. [cited 2025 Dec 15]. Available from: https://www.fas.usda.gov/data/citrus-world-markets-and-trade-01302025 [Google Scholar]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools