 Open Access
Open Access
ARTICLE
Evaluation of the Antifungal Activity of Aqueous Extracts of Corrigiola telephiifolia and Marrubium vulgare against Major Post-Harvest Citrus Diseases
1 Phytopathology Unit, Department of Plant Protection, Ecole Nationale d’Agriculture de Meknès, Km10, Rte Haj Kaddour, Meknes, Morocco
2 Laboratory of Functional Ecology and Environmental Engineering, Sidi Mohamed Ben Abdellah University, Fez, Morocco
3 Laboratory of Biotechnology, Conservation and Valorisation of Bioresources (LBCVBR), Faculty of Sciences, Dhar El Mehraz, Sidi Mohamed Ben Abdallah University, Fez, Morocco
4 Department of Plant and Environment Protection, National School of Agriculture, Ecole Nationale d’Agriculture de Meknes, Route Haj Kaddour, Meknes, Morocco
* Corresponding Author: Rachid Lahlali. Email:
(This article belongs to the Special Issue: Innovations in Post-Harvest Disease Control and Quality Preservation of Horticultural Crops)
Phyton-International Journal of Experimental Botany 2026, 95(4), 7 https://doi.org/10.32604/phyton.2026.078088
Received 23 December 2025; Accepted 09 March 2026; Issue published 28 April 2026
Abstract
Citrus fruits are highly susceptible to post-harvest diseases such as green rot (Penicillium digitatum), blue rot (P. italicum), and sour rot (Geotrichum citri-aurantii), causing significant economic losses. Due to the risks associated with synthetic fungicides and the emergence of resistant strains, natural alternatives are needed. This study evaluated the antifungal activity of aqueous extracts of Corrigiola telephiifolia and Marrubium vulgare. In vitro tests were conducted using PDA medium with extract concentrations of 12.5–100 mg/mL and in vivo assays were performed on artificially wounded oranges with the same extract concentrations. In vitro tests showed strong inhibition of mycelial growth and spore germination, with C. telephiifolia reducing pathogen growth by over 70–89%. In vivo assays confirmed its effectiveness, completely preventing green rot at 12.5 mg/mL and significantly reducing sour rot. Phytochemical analysis revealed high levels of polyphenols and flavonoids in C. telephiifolia. These findings suggest that aqueous extracts of C. telephiifolia are a safe and sustainable alternative to synthetic fungicides for managing post-harvest citrus diseases.Graphic Abstract
Keywords
Citrus fruits, belonging to the Rutaceae family, are among the world’s leading fruit crops, with global production reaching 169.39 million tons in 2023 covering 10.2 million hectares, thus confirming their position among the world’s major horticultural products. They are grown in more than 140 countries [1,2]. Citrus fruits, characterized by numerous varieties, are available on the global market. The orange Citrus sinensis (L.) Osbeck is the most produced variety with 89.8 million tons in 2023, followed by Citrus reticulata Blanco with 38.6 million tons [2]. Citrus fruits are among the most widely consumed fruits in the world. They are valued for their distinctive flavor, nutritional value, and health benefits, including the prevention of diseases such as cardiovascular disease, cancer, diabetes, and other chronic diseases.
After harvesting, citrus fruits undergo various stages (transport, packaging, storage) that cause mechanical damage to the fruit, making it more susceptible to fungal infections, which penetrate the fruit through these wounds and cause it to rot [3]. Conditions favorable to their development lead to deterioration in fruit quality, significantly reducing their shelf life and causing significant economic losses [4]. After harvesting, citrus fruits are exposed to three pathogenic fungi responsible for the main diseases affecting this crop: green rot caused by Penicillium digitatum, blue rot caused by P. italicum, and sour rot caused by Geotrichum citri-aurantii. These diseases are a crucial post-harvest health problem, causing 35% of total losses [5]. Green rot is the most serious disease, causing 90% of recorded post-harvest losses [6]. The application of chemical fungicides such as benomyl and imazalil is the traditional method of controlling the development of these pathogens on citrus fruits [7]. However, its effectiveness is decreasing more and more due to the emergence of resistant strains of the fungus P. digitatum and P. italicum, and its use has been associated with significant adverse effects on human health and the environment, including the presence of toxic residues in fruit and soil contamination [8]. In this context, research focuses on developing alternatives to chemical control methods to ensure the quality and safety of citrus fruits, taking into account growing concerns related to food safety, sustainability, fungal resistance to fungicides, and human health [9]. It is recommended to use non-chemical alternatives sensitive to biological methods, including the use of biological agents, bacteria, yeasts and other control microorganisms, for our health and out of respect for the environment [10,11,12]. Another control method involves the use of medicinal and aromatic plants, which are attracting growing interest due to their powerful natural antifungal properties, biodegradability, and lack of toxicity. They contain several bioactive compounds, such as phenols and flavonoids. Another method of control involves using medicinal and aromatic plants, which are attracting increasing interest due to their potent natural antifungal properties, biodegradability, and safety. These properties are due to bioactive compounds such as phenols and flavonoids, demonstrating the potential of their essential oils to combat the pathogens responsible for post-harvest diseases [13]. Numerous studies focus on plant extracts and their antifungal activity, thanks to their richness in biologically active compounds such as terpenes, flavonoids and phenols, their effectiveness against phytopathogenic fungi and their potential as sustainable alternatives to synthetic fungicides in post-harvest disease management [14]. Recently, combining plant extracts with nanomaterials has emerged as an innovative approach to enhancing antifungal efficacy. For example, zinc-based nanoparticles, synthesized from plant extracts such as Trispermum ammi, have demonstrated significant antifungal activity against post-harvest fruit pathogens, while also providing improved stability and reducing environmental impact [15]. Aqueous extracts are among the most effective extracts for post-harvest protection, particularly against citrus diseases, as confirmed by numerous studies [16].
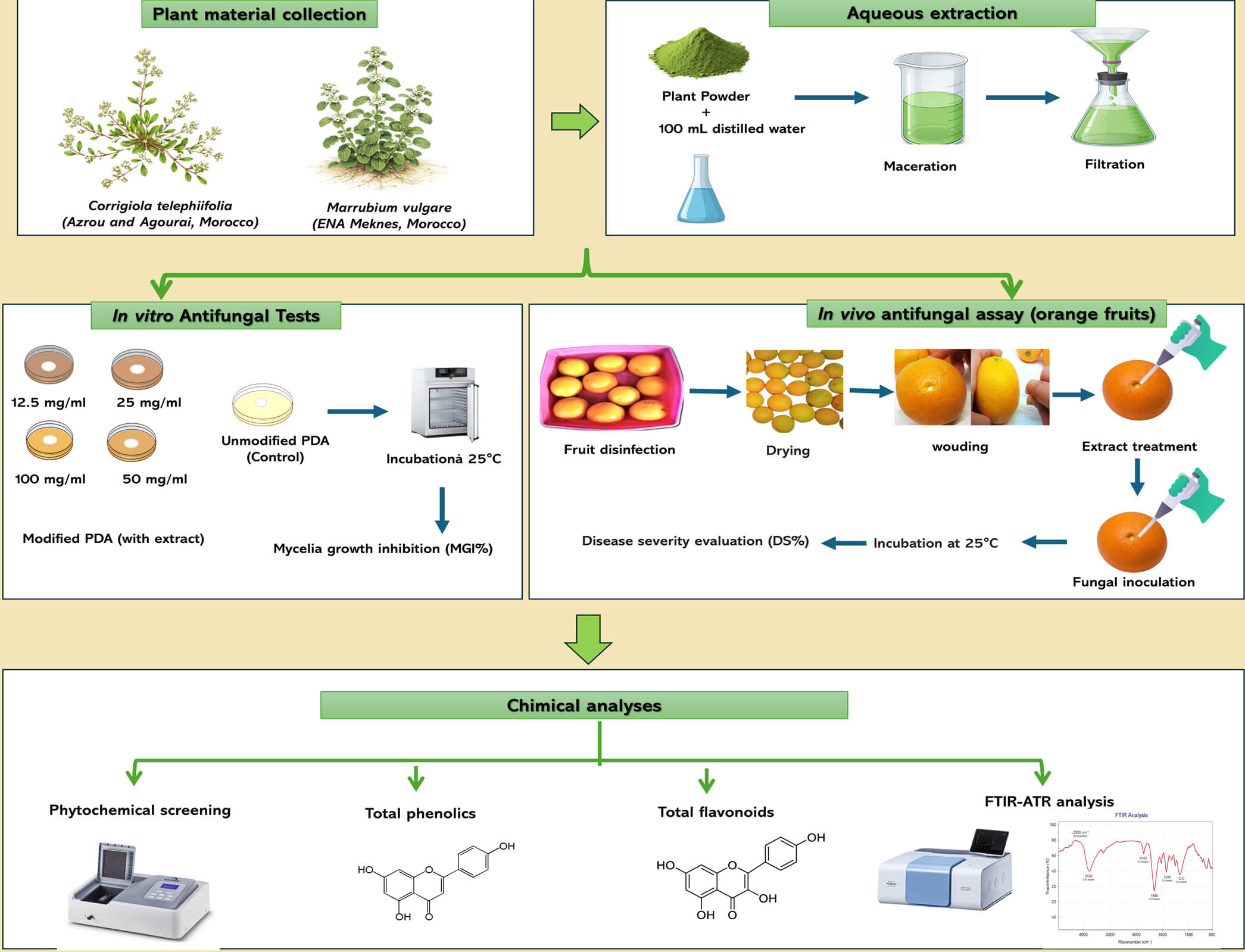
Corrigiola telephiifolia (Polygonaceae) is a halophytic plant native to arid regions. Thanks to its secondary metabolites rich in polyphenols, it is used in traditional medicine to treat microbial infections [17]. Marobium vulgaris (Lamiaceae) is an aromatic plant valued for its antifungal properties against pathogenic fungi, which are attributed to diterpenes and flavonoids [18]. In this context, our study aims to evaluate the antifungal activity of two plant species, The effect of aqueous extracts of Corrigiola telephiifolia harvested from two different regions of Agourai and Azrou, and of Marrubium vulgare, was studied against the main pathogens responsible for post-harvest citrus diseases, namely P. italicum, P. digitatum and G. citri-aurantii. Therefore, the specific objective of this work was to evaluate the inhibitory effect of aqueous extracts of these plants on mycelial growth in vitro, evaluate their inhibitory effect on fungal spore germination; examine their effectiveness in reducing fruit damage in vivo, and finally, perform a phytochemical characterization of the aqueous extracts.
This study focused on two plant species Corrigiola telephiifolia was collected from two separate sites (Agourai and Azrou) in the Fes-Meknes region of Morocco, while Marrubium vulgare was collected near the teaching farm of the National School of Agriculture in Meknes. Both species were harvested in March, at the pre-flowering stage, before the flowers appeared. The samples were dried in an oven at 40°C and then mechanically ground. The resulting powder was stored in the dark pending analysis.
The maceration method in water was used to obtain the aqueous extract [19]: 10 g of plant powder was mixed with 100 mL of distilled water (10% w/v) under continuous stirring at room temperature for 12 h. The extract was filtered and centrifuged (5000 rpm, 10 min). The solvent was removed by lyophilization to avoid thermal degradation and residual water. The aqueous extracts were freshly prepared prior to each experiment and stored at 4°C for no longer than 72 h before use. This short-term storage ensured the maintenance of their antifungal activity. Long-term stability and shelf-life assessments were not performed in the present study and are recommended for future research to determine the practical feasibility of these extracts for post-harvest applications. The extraction yield was calculated using the following equation:
All extract concentrations were expressed consistently as % (w/v). The pH of the extracts was not measured and is acknowledged as a limitation. Fresh extracts were stored at 4°C for no longer than 72 h prior to use.
2.3 Preparation of Fungal Pathogens
The antifungal activity of these plant extracts was tested against three pathogenic fungi from the collection of the phytopathology laboratory at the National School of Agriculture in Meknes, Morocco, which were already isolated from rotten oranges exhibiting the symptoms of the 3 pathogens These fungi were P. italicum (PX640585), P. digitatum (OR616652), and G. citri-aurantii (PX640586). The fungi were grown on potato dextrose agar medium (15 g/L agar, 20 g/L glucose, pH 5.6) and incubated at 25°C for 5 days. The cultures were stored at 4°C until use. A spore suspension was prepared from each 5-day culture by scraping the surface of the culture with a sterile scalpel and suspending the spores in sterile distilled water containing 0.05% Tween 20. To recover only the spores and remove debris from the medium and fungal hyphae, the suspension was then filtered through Watman filter paper. The final concentrations of the spore suspensions were adjusted using a hemocytometer to 1 × 104 spores·mL−1 for P. italicum and P. digitatum, and to 1 × 107 spores mL−1 for G. citri-aurantii [16,20].
2.4 In-Vitro Antifungal Activity of Aqueous Extracts of C. telephiifolia and Marrubium vulgare
2.4.1 Effect on Mycelial Growth
To quantify the inhibition of mycelial growth, the aqueous extracts were mixed with PDA medium using the dilution method to achieve final concentrations of 12.5, 25, 50, and 100 mg·mL−1. Each Petri dish was then inoculated in the center with a 5 mm mycelial disc from a 5-day culture. Control dishes containing only PDA were prepared in parallel. The dishes were then placed at 25°C for 7 days, and the entire treatment was repeated four times. Antifungal activity was expressed as the percentage of mycelial growth inhibition (MGI, %), calculated after seven days of incubation using the following formula [21]:
2.4.2 Effect on Spore Germination
Evaluation of the antifungal activity of aqueous plant extracts on the germination of P. italicum, P. digitatum, and G. citri-aurantii spores, the spore suspension prepared in PDB broth (50 μL) was mixed with extracts at different concentrations (12.5, 25, 50, and 100 mg/mL) in a 1:1 ratio. A control consisting of the spore suspension with distilled water (50 μL) added instead of the extract was included. After incubation for 24 h at 25°C [22]. Germination was studied by optical microscopy (400×). For each repetition, observations were made on 100 spores: the spore was considered germinated if the length of its germination tube exceeded its own diameter. The inhibition rate (GI, %) was calculated using the following formula [23]:
2.5 In-Vivo Evaluation of the Antifungal Activity of Aqueous Extracts of C. telephiifolia and M. vulgare
Citrus fruits (Citrus sinensis var. “Maroc-Late”) were collected from fields in the Moroccan province of El Gharb. No synthetic pesticides were applied preharvest. Only fruits uniform in size, shape, and ripeness, and free of rot or deterioration, were selected. Fruits were either used immediately or stored at 4°C for up to seven days prior to experiments. To ensure surface sterility, fruits were submerged in 2% sodium hypochlorite solution for 2 min, rinsed twice with distilled water, and dried under a laminar flow hood. Each fruit was wounded twice at the equatorial zone using a sterilized stainless steel rod (5 mm in diameter, 4 mm deep), each lesion being treated with 30 μL of plant extract at concentrations of 12.5, 25, 50, and 100 mg/mL. After 2 h of incubation at room temperature under a laminar flow hood, each wound was inoculated with 20 μL of a fungal spore suspension at a concentration of 1 × 104 spores mL−1 for P. italicum and P. digitatum, and 1 × 107 spores mL−1 for G. citri-aurantii [24]. Fruits treated with the fungicide imazalil (1 ppm) and those treated with sterile distilled water served as positive and negative controls, respectively. The treated oranges were incubated in plastic boxes at a temperature of 24°C, with relative humidity controlled at 70%. Five fruits were used per treatment, with three independent replicates. Lesion measurements were performed consistently, and all experiments were conducted independently. Data from replicates were pooled for statistical analysis. Disease severity (DS, %) is determined for each treatment after 7 days of incubation using the following formula [25]:
2.6 Determination of Total Phenolic and Flavonoid Contents in C. telephiifolia and M. vulgare Extracts
The total amount of phenolic compounds was measured using the Folin-Denis reagent [26]. Two ml of Folin-Denis reagent (previously diluted 10 times in distilled water) were added to 400 μL of 1 mg/mL extract. After 4 min, 1600 μL of sodium carbonate solution (75 mg/mL) were added to the solution. After 2 h of incubation in the dark at room temperature, the absorbance was measured at 765 nm using a UV-visible spectrophotometer. Calibration was performed with gallic acid (0–300 μg/mL in methanol). Reagent preparation: 75 mL of distilled water, 10 g of sodium tungstate, 2 g of phosphomolybdic acid, and 15 mL of concentrated phosphoric acid were added to a volumetric flask, and the mixture was boiled for 2 h. After cooling, it was transferred to a 100 mL volumetric flask and diluted to 100 mL with distilled water.
The total flavonoid content was measured using the colorimetric method described by Hssaini et al. [27]. Thus, to 2 mL of the extract previously diluted to 1 mg/mL, an equal amount of AlCl3 solution (2% in methanol) is added and incubated for 10 min in the dark at room temperature before measuring the absorbance at 430 nm by UV-visible spectrophotometry with a blank. For calibration, quercetin was used at a calibrated concentration (0 to 40 μg/mL in the same solvent).
2.7 Phytochemical Screening of C. telephiifolia and M. vulgare Aqueous Extracts
The main objective of phytochemical screening is to identify the main classes of secondary metabolites such as terpenoids, alkaloids, flavonoids, glycosides, tannins, coumarins, saponins, and steroids present in plant extract of C. telephiifolia and M. vulgare. This screening was performed using infused aqueous extracts prepared according to the method described by [27]. The decoction was made using 2 g of treated plant powder and 80 mL of boiling distilled water, which was left to infuse for 24 h. After cooling, the extract was filtered through a Whatman filter.
A Salkowski test was performed by adding 2 mL of chloroform and 3 mL of sulfuric acid to 2 mL of the infused solution. The appearance of a reddish-brown color indicates the presence of terpenoids [28].
The presence of alkaloids was demonstrated by the formation of an orange or red precipitate during alkaloid testing, which consisted of mixing 5 mL of the infused solution with 2 mL of hydrochloric acid and 1 mL of Dragendorff’s reagent (iodobismuthate reagent) [29].
To detect flavonoids, an alkaline test was performed by adding 2 mL of NaOH (2%) to 2 mL of the infused solution. The appearance of a reddish-brown color indicates the presence of terpenoids [30].
The presence of glycosides was demonstrated by the Keller-Kiliani test, consisting of adding 2 mL of acetic acid and 2 mL of sulfuric acid to 2 mL of infused solution. The presence of glycosides was confirmed by the appearance of a red color [28].
One ml of 1% FeCl3 was mixed with 2 mL of infusion. Two cases can be observed: the appearance of a greenish color reveals the presence of catechins. Conversely, a blue-black color indicates the presence of gallic tannins [31].
In a boiling water bath, a mixture made from 2 mL of an infuscate, and 2 mL of hydrochloric acid was infused for 5 min. The appearance of a persistent red color indicates the presence of condensed tannins [32].
In a beaker covered with filter paper soaked in NaOH (10%), 2 mL of infused solution was poured. The mixture was brought to a boil until the paper dried. The presence of coumarins was revealed by the appearance of yellow fluorescence on the paper, observed under UV light at 365 nm [33].
A foam test was performed by vigorously shaking 2 mL of infusion for 30 s. After resting for 15 to 20 min, the appearance of persistent foam ≥1 cm in height indicates the presence of saponins [34].
Steroids were detected using the Liberman-Burchard test with a few drops of acetic anhydride, 2 mL of chloroform, 2 mL of sulfuric acid, and 2 mL of the solution. The presence of steroids was confirmed by the appearance of a red color. The presence of glycosides was determined using the Keller-Kiliani test, which involves adding 2 mL of acetic acid and 2 mL of sulfuric acid to 2 mL of infused solution [28].
2.8 Measurement by Fourier Transform Infrared Spectroscopy
Fourier transform infrared (FTIR) spectroscopy measurements were performed on the studied species using a Bruker Vertex 70 FTIR spectrometer equipped with an Attenuated Total Reflectance (ATR) accessory (Bruker Optics Inc., Ettlingen, Germany). Powder samples from the three plants were deposited onto a 176 μm crystal plate and exposed to an infrared beam with a wavelength between 2.5 and 25 μm. The samples were analyzed in the infrared region between 450 and 4000 cm−1 with a resolution of 4 cm−1 and a peak-to-peak signal-to-noise ratio of 9300:1 for 5 s. The results were then visualized and interpreted using Origin Lab software. The values of the characteristic FTIR peaks were measured, and their functional groups were determined [35]. The integration of areas corresponding to specific spectral bands was performed in order to identify constituents such as proteins, lignin, cellulose, hemicellulose, and pectin [36].
The results obtained from in vitro and in vivo tests, as well as phytochemical analyses, were subjected to one-way ANOVA followed by Tukey’s HSD post-hoc test at a significance level of p < 0.05, using SPSS version 25 software. Each experiment was conducted with five replicates (n = 5), and all tables report mean ± SD values.
The aqueous extract of C. telephiifolia yielded 17.43% for samples taken in Agourai and 18.13% for those taken in Azrou, while the aqueous extract of M. vulgare yielded 12.56%.
3.2 In Vitro Effect of Aqueous Extracts of C. telephiifolia and M. vulgare
3.2.1 Effect on Mycelial Growth
Table 1 shows the inhibitory effect of aqueous extracts of C. telephiifolia and M. vulgare on the mycelial growth of P. italicum, P. digitatum, and G. citri-aurantii in vitro. Both extracts significantly reduced fungal growth compared to the control, and this inhibitory effect was observed at all concentrations and was statistically very significant (p < 0.05).
In particular, aqueous extracts of C. telephiifolia (Agourai and Azrou) proved to be significantly more effective than those of M. vulgare. Extracts of C. telephiifolia from Agourai showed strong inhibitory activity against the three fungi, with inhibition rates exceeding 62% even at the lowest concentration tested (12.5 mg/mL), reaching 76.46% for P. italicum, 89.09% for P. digitatum, and 90.94% for G. citri-aurantii at a concentration of 100 mg/mL. The other aqueous extract from the same species, collected in Azrou, also demonstrated very significant antifungal activity, with inhibition rates of 68.18%, 87.92%, and 86.30% for P. italicum, P. digitatum, and G. candidum, respectively. In contrast, the aqueous extract of M. vulgare showed weaker antifungal activity, with maximum inhibition observed only against P. digitatum (71.85% at 100 mg/mL), while its effect against P. italicum and G. citri-aurantii remained limited (≤15.3%) at all concentrations tested. Overall, these results demonstrate that aqueous extracts of C. telephiifolia, particularly those from the Agourai region, have significant antifungal activity against the three fungal species tested, and that this activity increases proportionally with the concentration of the extract.
Table 1: In vitro effect of concentrations of aqueous extracts of C. telephiifolia and M. vulgare on the mycelial growth of P. italicum, P. digitatum, and G. citri-aurantii after 10 days of incubation at 25°C.
| Inhibition Rate of Mycelial Growth (%) | ||||
|---|---|---|---|---|
| Plant | Concentration (mg/mL) | P. italicum | P. digitatum | G. citri-aurantii |
| C. telephiifolia Agourai | 100 | 76.46 ± 1.86d | 89.09 ± 0.29e | 90.94 ± 0.42h |
| 50 | 70.15 ± 1.02d | 88.74 ± 0.32e | 90.69 ± 0.12h | |
| 25 | 67.20 ± 3.4d | 88.01 ± 0.63e | 90.67 ± 0.24h | |
| 12.5 | 62.78 ± 3.69cd | 88.65 ± 0.84e | 90.38 ± 0.18h | |
| C. telephiifolia Azrou | 100 | 68.18 ± 2.64d | 87.92 ± 0.82de | 86.30 ± 0.34h |
| 50 | 63.76 ± 0.45cd | 87.74 ± 0.53de | 69.46 ± 2.79g | |
| 25 | 61.49 ± 0.97cd | 87.57 ± 0.69de | 57.15 ± 1.54f | |
| 12.5 | 54.42 ± 1.46c | 85.33 ± 0.57d | 52.57 ± 0.78f | |
| M.vulgare | 100 | 5.92 ± 2.99a | 71.85 ± 2.16d | 11.94 ± 0.53e |
| 50 | 11.10 ± 2.8ab | 66.29 ± 2.85d | 7.16 ± 1.43bcd | |
| 25 | 13.3 ± 2.12b | 45.25 ± 2.93c | 6.32 ± 2.28bc | |
| 12.5 | 15.29 ± 2.42b | 24.12 ± 2.14b | 5.12 ± 2.17b | |
| Control (Without extract) | 0 | 0.00 ± 0.00a | 0.00 ± 0.00a | 0.00 ± 0.00a |
3.2.2 Effect on Spore Germination
The results presented in Table 2 highlight a notable antifungal effect of aqueous extracts of C. telephiifolia (Agourai and Azrou) and M. vulgare on the germination of spores from the three pathogenic fungi studied. In general, these aqueous extracts inhibit or reduce spore germination, and the percentage of germination inhibition increases proportionally to the concentration applied, indicating a dose-response relationship. The aqueous extract of C. telephiifolia (Agourai) was the most active, with a minimum concentration of 12.5 mg/mL, inhibition rates above 87% for P. digitatum and G. candidum, and even above 90% at a concentration of 100 mg/mL. In contrast, for P. italicum, the effect remained moderate but showed a dose-dependent trend, with germination inhibition increasing from 3.0% at 12.5 mg/mL to 71.1% at 100 mg/mL. For the extract of C. telephiifolia (Azrou), no effect was observed against P. digitatum (inhibition of 87.4% at 12.5 mg/mL and 92.3% at 100 mg/mL). This extract also reduced the germination rate of G. citri-aurantii spores, with inhibition of 79.17% at 50 mg/mL and 90.28% at 100 mg/mL. However, on P. italicum, the effect remained limited, with rates below 31% even at the maximum concentration. In comparison, M. vulgare extract effectively inhibited the germination of G. citri-aurantii spores, with an inhibition percentage exceeding 93% at 100 mg/mL. However, this same extract was significantly less effective against P. italicum and P. digitatum, where inhibition percentages remained low to moderate.
Table 2: Effect of concentrations of aqueous extracts of C. telephiifolia and M. vulgare on the on-spore germination of P. italicum, P. digitatum, and G. citri-aurantii after 24 h of incubation at 25°C.
| Spore Germination Inhibition Rate (%) | ||||
|---|---|---|---|---|
| Plant | Concentration (mg/mL) | P. italicum | P. digitatum | G. citri-aurantii |
| C. telephiifolia Agourai | 100 | 71.12 ± 3.37j | 92.31 ± 3.43e | 96.53 ± 1.21d |
| 50 | 46.74 ± 1.69ij | 90.11 ± 2.85e | 91.67 ± 2.09d | |
| 25 | 10.46 ± 1.52cde | 87.92 ± 3.43e | 84.03 ± 6.02d | |
| 12.5 | 3.03 ± 3.17ab | 87.37 ± 2.52e | 70.83 ± 12.67d | |
| C. telephiifolia Azrou | 100 | 42.64 ± 3.23hi | 92.32 ± 2.52e | 90.28 ± 1.21d |
| 50 | 27.02 ± 2.5fgh | 92.31 ± 4.15e | 79.17 ± 2.09d | |
| 25 | 15.04 ± 3.37def | 89.57 ± 3.43e | 61.80 ± 3.18c | |
| 12.5 | 6.80 ± 0.48abc | 87.37 ± 2.52e | 42.36 ± 3.89b | |
| M.vulgare | 100 | 30.96 ± 2.86gh | 60.47 ± 5.94d | 93.06 ± 1.20d |
| 50 | 28.21 ± 1.21fgh | 48.37 ± 3.30c | 88.89 ± 1.20d | |
| 25 | 21.49 ± 3.99efg | 47.29 ± 2.24c | 86.81 ± 2.40d | |
| 12.5 | 7.98 ± 2.85bcd | 34.66 ± 3.03b | 80.56 ± 2.40d | |
| Control (Without extract) | 0 | 0.00 ± 0.00a | 0.00 ± 0.00a | 0.00 ± 0.00a |
3.3 Effect of Aqueous Extracts of C. telephiifolia and M. vulgare on the Severity of Orange Rot
In vivo tests showed that aqueous extracts from the three plants studied exhibited varying degrees of activity in reducing the severity of three pathogens (Table 3, Fig. 1).
The aqueous extract of C. telephiifolia from Azrou proved to be the most effective in completely reducing the severity of the disease (0%) caused by P. digitatum green rot at all concentrations tested (25, 50, and 100 mg/mL). A similar effect was observed with the extract of the same species from Agourai, but only at the highest concentration (100 mg/mL), confirming the strong antifungal potential of C. telephiifolia against P. digitatum. Conversely, the filtrate of M. vulgare showed no significant activity against this phytopathogen, with the severity of green rot remaining at over 70%, comparable to the untreated control. With regard to P. italicum, which causes blue mold, the three extracts tested showed no significant effect, with disease levels ranging from 73.13% to 97.01% depending on the concentration, indicating that the extracts tested had little effect against this fungus. Compared to the control, only C. telephiifolia from Azrou reduced the severity of acid rot, by 19.32% at 100 mg/mL and 37.73% at 50 mg/mL. In contrast, the filtrates of Agourai and M. vulgare proved to be ineffective, with the severity of the disease remaining high (>70%) for all concentrations tested (25, 50, and 100 mg/mL). The synthetic fungicide (Imazalil), used as a positive control, showed a disease severity of 81.37 ± 2.05% for P. italicum, and a total reduction for P. digitatum and G. citri-aurantii.
Table 3: Effect of aqueous extracts of C. telephiifolia and M. vulgare on the severity (%) of green, blue, and sour rot of orange after 7 days of incubation at 25°C.
| Disease Severity (DS, %) | ||||
|---|---|---|---|---|
| Plant | Concentration | P. italicum | P. digitatum | G. citri-aurantii |
| C. telephiifolia (Agourai) | 100 mg/mL | 81.46 ± 2.28c | 0.00 ± 0.00a | 71.67 ± 0.49d |
| 50 mg/mL | 88.09 ± 1.54b | 41.30 ± 1.02b | 75.27 ± 0.69c | |
| 25 mg/mL | 93.38 ± 3.34ab | 45.22 ± 0.44b | 82.66 ± 3.96b | |
| 12.5 mg/mL | 96.56 ± 2.45a | 46.78 ± 1.25b | 84.98 ± 2.90b | |
| C. telephiifolia (Azrou) | 100 mg/mL | 85.43 ± 1.78bc | 0.00 ± 0.00a | 19.32 ± 0.63g |
| 50 mg/mL | 94.95 ± 1.50ab | 0.00 ± 0.00a | 37.73 ± 5.61f | |
| 25 mg/mL | 96.73 ± 1.92a | 0.00 ± 0.00a | 66.82 ± 1.28e | |
| 12.5 mg/mL | 98.56 ± 2.78a | 0.00 ± 0.00a | 68.56 ± 1.56e | |
| M.vulgare | 100 mg/mL | 73.13 ± 2.78d | 71.57 ± 1.53c | 59.84 ± 1.03ef |
| 50 mg/mL | 87.81 ± 1.64bc | 89.41 ± 1.81d | 74.03 ± 2.13cd | |
| 25 mg/mL | 97.01 ± 2.80a | 92.61 ± 0.87d | 77.12 ± 1.27c | |
| 12.5 mg/mL | 99.45 ± 1.67a | 96.56 ± 0.12a | 80.45 ± 2.12b | |
| Imazalil fungicide | 1ppm | 81.37 ± 2.05c | 0.00 ± 0.00a | 0.00 ± 0.00a |
| Untreated control | 0 | 100.00 ± 0.00a | 100.00 ± 0.00a | 100.00 ± 0.00a |
| Negative control | 0 | 0.00 ± 0.00a | 0.00 ± 0.00a | 0.00 ± 0.00a |

Figure 1: Effect of aqueous extracts on the development of green, blue, and sour mold lesions on oranges after 10 days of incubation at 25°C. (A) untreated control inoculated with P. digitatum, (B) treated with M. vulgare against P. digitatum, (C) treatment with C. telephiifolia (Agourai) against P. digitatum, (D) treatment with C. telephiifolia (Azrou) against P. digitatum, (E) positive control with the fungicide Imazalil against P. digitatum, (F) control inoculated with G. citri-aurantii, (G) treatment with M. vulgare against G. citri-aurantii, (H) treatment with C. telephiifolia (Agourai) against G. citri-aurantii, (I) treatment with C. telephiifolia (Azrou) against G. citri-aurantii, (J) control treated with Imazalil against G. citri-aurantii, (K) control inoculated with P. italicum, (L) treatment with M. vulgare against P. italicum, (M) treatment with C. telephiifolia (Agourai) against P. italicum, (N) treatment with C. telephiifolia (Azrou) against P. italicum, and (O) control treated with Imazalil against P. italicum.
3.4 Total Polyphenol and Flavonoid Content
The results presented in Table 4 revealed that the aqueous extract of C. telephiifolia from Agourai was the richest in total polyphenols (13.61 μg GAE/mg) and had a relatively high flavonoid content (4.96 μg QE/mg), followed by the aqueous extract of C. telephiifolia from Azrou with (12.91 μg GAE/mg and 3.70 μg QE/mg), while M. vulgare had the lowest concentration of polyphenols (9.93 μg GAE/mg) but the highest flavonoid content compared to the other aqueous extracts (5.28 μg QE/mg).
Table 4: Total polyphenols and flavonoids contents, values with different letters are significantly different, C. telephiifolia Azrou, C. telephiifolia Agourai, M. vulgare.
| Total Polyphenols Content (μg EAG/mg of Extrait) | Total Flavonoids Content (μg EQ/mg of Extrait) | |
|---|---|---|
| C. telephiifolia Azrou | 12.91a | 3.70d |
| C. telephiifolia Agourai | 13.61a | 4.96c |
| M.vulgare | 9.93b | 5.28c |
Qualitative phytochemical analysis (Table 5) revealed marked variations in the secondary metabolic profiles of the three plants studied.
Aqueous extracts of C. telephiofolia from both locations are characterized by a high presence of terpenes, saponins, and unsaturated sterols (+++). However, the Agourai sample has a notable abundance of glycosides (+++), whereas they are completely absent from the Azrou sample. Traces of condensed tannins are observed in the Azrou sample but not detected in the Agourai sample. Saponins, unsaturated sterols, and coumarins are present in both samples, although in greater quantities in the Azrou sample. On the other hand, neither extract contains hydrolysable tannins or steroids. The aqueous extract of M. vulgare is rich in flavonoids (+++) and unsaturated sterols (+++) and has a moderate concentration of glycosides, condensed tannins, terpenes, alkaloids, and coumarins (++). Traces of steroids have been detected, while hydrolysable tannins and saponins are absent.
Table 5: Phytochemical screening results, (+++) = very rich, (++) = moderately rich, (+) = traces, (-) = absent.
| Tests | C. telephiifolia (Azrou) | C. telephiifolia (Agourai) | M. vulgare |
|---|---|---|---|
| Flavonoids | ++ | ++ | +++ |
| Glycosides | - | +++ | ++ |
| Steroids | - | - | + |
| Hydrolyzable tannins | - | - | - |
| Condensed tannins | + | - | ++ |
| Terpenoids | +++ | +++ | ++ |
| Insaturated sterols | +++ | ++ | +++ |
| Saponins | +++ | ++ | - |
| Alkaloids | + | + | ++ |
| Coumarins | ++ | + | ++ |
3.6 Fourier Transform Infrared Spectroscopy Analysis
The peaks of the FT-IR analysis of C. telephiofolia (Azrou and Agourai) and M. vulgare powders are shown in Fig. 2. These spectra allow the determination of the main functional groups of the chemical compounds in the samples. Spectral analysis of the plants studied revealed a broad absorption band around 3313 cm−1, attributed to the stretching vibrations of alcohols, phenols, and proteins (O–H and N–H). The peaks between 2919 cm−1 and 2851 cm−1 correspond to the vibrations of the C–H bonds of lipids and polysaccharides. A well-defined absorption band at 1627 cm−1, characteristic of the amide I region of proteins and pectin’s, corresponds to C=O bonds, while the signal at 1409 cm−1 is associated with O–H and C–N groups in lignin’s and hemicelluloses. In addition, an intense peak, attributed to the C–O vibrations of glycosidic polysaccharides, particularly cellulose and hemicelluloses, is observed at 1027 cm−1. Overall, the three plants have the same functional group, composed of phenolic, polysaccharide, and protein compounds C. telephiofolia (Azrou), with the most intense peaks at 3313, 1627, and 1027 cm−1, provides a differentiation in the relative proportions of these biomolecules among the species studied.

Figure 2: Representation of absorbance bands recorded in the 4000–500 cm−1 region of C. telephiifolia Agourai (C.T.Ag), C. telephiifolia Azrou (C.T.Az), and M. vulgare (M.V).
The absorbance values corresponding to the main functional groups identified by FTIR spectroscopy are presented in Table 6. Quantitative analysis of the FTIR bands reveals clear differences among the three studied plants. The absorbance values are highest for C. telephiofolia (Azrou) for most characteristic bands, particularly those of pectin’s (1760–1720 cm−1), proteins (1710–1620 cm−1), lignin (1615–1590 cm−1), and cellulose (1090–1022 cm−1), with significant differences (p < 0.05) compared to other species. Conversely, C. telephiofolia (Agourai) shows the lowest values for most bands, reflecting a lower content of structural macromolecules. M. vulgare occupies an intermediate position, displaying moderate values for most peaks. These results indicate that the chemical composition of the three plants differs considerably according to species and geographical origin.
Table 6: Comparative analysis of the integrated spectral bands of the FTIR spectra of: C. telephifolia (from Azrou and Agourai) and M. vulgare.
| C. telephiifolia Azrou | C. telephiifolia Agourai | M. vulgare | |
|---|---|---|---|
| Pectin 1760–1720 | 0.177 ± 0.041a | 0.097 ± 0.006b | 0.140 ± 0.012ab |
| Proteins (Amide I) 1710–1620 | 0.671 ± 0.153a | 0.35 ± 0.02b | 0.370 ± 0.030b |
| Lignin (aromatic) 1615–1590 | 0.227 ± 0.052a | 0.12 ± 0.01b | 0.147 ± 0.013b |
| Lignin (aliphatic) 1480–1455 | 0.13 ± 0.03a | 0.070 ± 0.004b | 0.090 ± 0.008b |
| Hemicellulose (aliphatic) 1445–1410 | 0.23 ± 0.06a | 0.120 ± 0.008b | 0.149 ± 0.013b |
| Hemicellulose (glycosidic) 1261–1200 | 1.30 ± 0.31a | 0.156 ± 0.010b | 0.237 ± 0.019b |
| Cellulose 1090–1022 | 0.95 ± 0.23a | 0.499 ± 0.033b | 0.486 ± 0.043b |
Fungicide application is the primary methods for controlling fruit pathogens post-harvest. However, he persistence of residues of these chemicals on the surface of the fruit poses a risk to human health and has a negative impact on the environment. In addition, the continued application of chemical fungicides leads to the emergence of pathogen strains that are resistant to these products [37]. Several studies demonstrate the potential of plant extracts in the formulation of plant-based biological fungicides to inhibit post-harvest infections [21,38]. Plant extracts have gained increasing importance due to their antifungal activity, research has highlighted the abundance of bioactive compounds with antifungal potential and identified these molecules as effective strategies [39]. Natural compounds capable of inhibiting the development and spread of pathogens through direct or indirect mechanisms promote the emergence of resistance and the suppression of fruit diseases [40]. Plant extracts are of interest to researchers studying their antimicrobial and antifungal effects due to the presence of various phenolic compounds. These compounds play an important role, as they allow plant extracts to be used as biocides and preservatives to control fruit diseases post-harvest [41]. Aqueous extracts are generally considered safer than other extracts, particularly ethanol extracts, because their solvent is water, which limits adverse effects and reduces environmental impact. The extraction method is very simple, energy efficient, and uses a renewable solvent. In addition, these extracts are suitable for food applications because they do not alter the quality or taste of the products [42]. To our knowledge, no study has yet examined the efficacy of aqueous extracts of M. vulgare and C. telephiifolia against post-harvest fruit diseases. Our work therefore enabled us to evaluate the antifungal activity of these extracts against P. digitatum, P. italicum, and G. citri-aurantii, which are responsible for green, blue, and sour rot in citrus fruits, respectively. In vitro and in vivo tests were carried out at four concentrations (12.5, 25, 50, and 100 mg/mL), and the results reveal the potential of these extracts for the prevention and control of fruit diseases.
The antifungal activity of aqueous extracts of C. telephiifolia (Agourai and Azrou) and M. vulgare was studied in vitro against P. digitatum, P. italicum, and G. citri-aurantii. The results of this study showed that aqueous extracts of C. telephiifolia had significant antifungal activity, with the Agourai plant inhibiting more than 89% of P. digitatum and G. citri-aurantii, and more than 76% of P. italicum at 100 mg/mL, with the Azrou extract also showing significant antifungal efficacy. These results suggest that C. telephiifolia contains water-soluble compounds whose metabolites may be responsible for the plant’s antifungal effects. Studies have also demonstrated this activity in plants rich in flavonoids, phenolic acids, tannins, saponins, etc. These compounds, produced during growth, can alter the cell wall of certain pathogens, leading to a change in membrane permeability and inhibiting metabolic enzymes [43]. The difference in activity between extracts from Agourai and Azrou, which come from the same plant, could be explained by the influence of soil and climate conditions on the biosynthesis of secondary metabolites [44]. The differences observed between the extracts from Agourai and Azrou could be explained by variations in phytochemical composition linked to geographical origin. Indeed, El Yaagoubi et al. [45] studied C. telephiifolia plants harvested in several regions of Morocco (Agourai, Sefrou, Azrou) and showed that the content of total phenolic compounds and total flavonoids varied considerably depending on the site of origin and the extraction solvent, with maximum values recorded in the raw extract from Agourai (44.76 ± 0.05 mg EAG/g DM and 20.80 ± 0.01 mg QAE/g DM). The same study also demonstrated that Agourai extracts had the highest antioxidant and antimicrobial activity, particularly against Escherichia coli and Pseudomonas aeruginosa. These results confirm that the high phenolic and flavonoid content in the Agourai samples could explain their superior antifungal activity observed in our study. However, FT-IR remains a global characterization technique and does not allow for the precise identification of active compounds or the evaluation of their individual contribution to antifungal activity. Additional analyzes, such as HPLC or LC-MS, combined with fractionation guided by biological activity, are generally required to identify and characterize the molecules responsible for the observed effect [46].
However, M. vulgare extract is only effective against P. digitatum (71.8% at 100 mg/mL), with little effect against P. italicum and G. citri-aurantii. This plant is very rich in antifungal compounds, particularly diterpenes and phenolic compounds [47]. In addition, recent studies have shown that methanolic and acetone extracts of M. vulgare exhibit antifungal activity against several fungi, including Botrytis cinerea and Pythium ultimum, revealing limited inhibition, with marrubiin exerting only a moderate effect (32.4% against B. cinerea) and even stimulating the growth of P. italicum at high concentrations. These observations confirm that the polar components of M. vulgare are weakly active against phytopathogens, which explains the low activity recorded in our study with the aqueous extract [18].
Spore germination tests reveal that C. telephiifolia extract inhibits the germination of P. digitatum and G. citri-aurantii by more than 70% at 12.5 mg/mL. The aqueous extract of M. vulgare inhibits the germination of G. citri-aurantii by more than 93% at 100 mg/mL. In addition, the Agourai extract of C. telephiifolia is the only one to inhibit the germination of P. italicum by 70% at 100 mg/mL. Furthermore, macerated extraction of M. vulgare has recently been shown to have strong antifungal activity against B. cinerea, the agent responsible for gray mold in tomatoes, completely inhibiting spore germination, while the extraction method has a significant impact on biological efficacy. The authors noted that this activity is related to the high quantity of terpenoids and phenolic compounds known for their antifungal activity. This suggests that the low efficacy observed in our research could be due to the aqueous nature of the extract, which has a low extraction capacity for these bioactive compounds [48].
In vivo tests were conducted to evaluate the effectiveness of aqueous plant extracts in mitigating the severity of diseases caused by P. digitatum, P. italicum, and G. citri-aurantii. The aqueous extract of C. telephiifolia from Azrou proved to be the most effective: it completely inhibited green rot at 25 mg/mL, showing its strong antifungal properties in inhibiting the development of P. digitatum on fruit. The same result was observed for the extract of the same species from Agourai, but only at the highest concentration (100 mg/mL). Not all of the extracts tested prevented the development of blue mold caused by P. italicum, as this pathogen quickly colonizes fruit tissue and resists the action of the water-soluble bioactive compounds they contain. Studies confirm that a low dose of imazalil (1 ppm) is insufficient to control Penicillium italiensis, with concentrations between 5 and 10 ppm generally required for effective inhibition, depending on factors such as strain resistance and inoculum quantity. Furthermore, imazalil requires concentrations ≥ 5 ppm for optimal control of Penicillium italiensis in post-harvest citrus, highlighting the variability related to fruit ripening and experimental conditions [49,50]. The C. telephiifolia extract from Azrou reduced the severity of sour rot by 19.32% at 100 mg/mL, while the aqueous extract of M. vulgare proved ineffective. The reduced effectiveness of aqueous antifungal extracts against sour rot caused by the fungus G. citri-aurantii can be explained by several factors. The cell wall of this fungus is rich in complex sugars, chitin, and beta-glucans, which limit the penetration of water-soluble bioactive compounds. Microscopic analysis shows that antifungals inhibit the synthesis of chitin (CHS2 and chsA genes) and glucans (Glu1 and Glu2), thereby increasing the permeability of the cell wall without inducing initial changes in the plasma membrane [51]. In addition, the antifungal compounds present in this fungus may be lipophilic, such as terpenes and nonpolar phenolic compounds. These compounds are difficult to extract with water but are easier to extract with other organic solvents like acetone or methanol. Studies confirm the superior efficacy of alcoholic extracts and essential oils rich in lipophilic terpenes such as citral and trans-2-hexenal, which disrupt the integrity of the fungal membrane by altering β-glucan polysaccharides and ergosterol inhibits the development of G. citri-aurantii [52]. Similarly, methanolic and acetone extracts of ginger spices revealed that fractions of these extracts inhibit the fungal growth of G. citri-aurantii with inhibition diameters of 20–25 mm at 25 mg/mL, compared to only 8–12 mm for aqueous extracts, thanks to better extraction of lipophilic terpene [53]. Thus, the results of this study highlight the limitations of aqueous extracts in controlling G. citri-aurantii, while confirming their targeted efficacy against post-harvest pathogens in citrus fruits. Future prospects include exploring other extraction methods, such as alcoholic extraction or the use of essential oils, as well as combined applications involving plant extracts, antagonistic yeasts, or GRAS salts, in order to improve efficacy against acid rot as part of an integrated and sustainable approach.
The absence of a significant preventive effect of the aqueous plant extracts against sour rot caused by G. citri-aurantii can be attributed to a combination of biological and chemical factors. This pathogen is characterized by a complex cell wall architecture predominantly composed of chitin and β-glucans, which may limit the diffusion and bioavailability of hydrophilic antifungal compounds, thereby reducing their effectiveness [54]. In parallel, several studies have demonstrated that the compounds most active against G. citri-aurantii, including terpenoids and non-polar phenolic molecules, are mainly lipophilic in nature and are therefore poorly extracted using water as a solvent. By contrast, alcoholic extracts and essential oils, which are richer in these lipophilic constituents, generally exhibit higher antifungal activity against sour rot pathogens [55]. For instance, methanolic and acetone extracts, as well as essential oils enriched in monoterpenes and aldehydes such as citral and trans-2-hexenal, have been reported to significantly inhibit the growth of G. citri-aurantii, notably through alterations of cell wall integrity and interference with membrane-associated sterol metabolism [52]. Taken together, these observations highlight the intrinsic limitations of aqueous plant extracts for the control of sour rot, while confirming their selective efficacy against other postharvest citrus pathogens. From a practical perspective, future investigations should therefore explore alternative extraction procedures (e.g., alcoholic extraction or essential oils) or combined application strategies involving plant extracts, antagonistic microorganisms, or GRAS salts, as part of an integrated and sustainable postharvest disease management approach.
With regard to the in vivo development of the three pathogens, significant differences were observed in the phytochemical composition of the extracts of C. telephiifolia and M. vulgare. These differences lie mainly in the presence of saponins in the extracts of C. telephiifolia, which are completely absent from the extracts of M. vulgare. Saponins are compounds characterized by their ability to bind to sterols, proteins, and phospholipids in the fungal cell membrane, causing a loss of structural integrity in this membrane and, simultaneously, an increase in its permeability [56].
Phytochemical examination revealed that the aqueous extract of M. vulgare was very rich in flavonoids and unsaturated sterols, as well as glycosides, condensed tannins, terpenes, alkaloids, and coumarins. No hydrolyzable tannins or saponins were detected. Traces of steroids were found. This observation is consistent with those of Hayat et al. [57], concerning the presence of flavonoids and terpenes, but differs from those of the same author with regard to the presence of coumarins and alkaloids. Similarly, Bouterfas et al. [58] observed the presence of condensed tannins in crude extracts of M. vulgare, but did not detect coumarins. These two authors also note the significant differences in chemical composition, which depend on the growing conditions to which the plants were subjected locally. Phytochemical analysis of aqueous extracts of C. telephiifolia from both sites (Agourai and Azrou) showed the presence of flavonoids in relatively modest quantities, high-density terpenoid compositions, and traces of alkaloids in all samples. Only the Agourai sample contained high levels of glycosides, which were absent from the Azrou sample. Traces of condensed tannins were observed in the Azrou sample, but were absent from the Agourai sample. In this extract, saponins, unsaturated sterols, and coumarins were observed in considerable quantities, but they were more abundant in the Azrou sample. Neither sample contained hydrolyzable tannins or steroids. These observations are consistent with those of [17,59] which mention that the nature and concentration of these compounds depend on the degree of maturity of the plant and the environmental conditions in which it grew. This could explain the differences observed between the two samples of C. telephiifolia.
This study demonstrates that aqueous extracts of C. telephiifolia possess selective antifungal activity against major post-harvest citrus pathogens. Strong inhibition was observed against G. citri-aurantii, moderate activity against P. digitatum, while P. italicum showed limited and inconsistent responses, reflecting its resistance to water-soluble bioactive compounds. Extracts from Agourai displayed higher efficacy than those from Azrou, likely due to higher concentrations of saponins, polyphenols, flavonoids, and terpenoids influenced by geographic origin and environmental conditions. In contrast, aqueous extracts of M. vulgare, which lack saponins and contain mainly flavonoids and sterols, exhibited weak or negligible activity. These findings highlight both the potential and the limitations of aqueous plant extracts for post-harvest disease management. Future research should focus on identifying the specific active compounds, optimizing extraction methods (e.g., alcoholic solvents or essential oils), and evaluating integrated strategies combining plant extracts with other biological or GRAS treatments to improve efficacy against resistant pathogens.
Acknowledgement:
Funding Statement: The authors received no specific funding for this study.
Author Contributions: The authors confirm their contribution to the article as follows: conceptualization, Hajar Zennouhi, Rachid Lahlali; methodology and data collection, Hajar Zennouhi, Mamadou Traoré; analysis and interpretation of results, Hajar Zennouhi, Rachid Ez-zouggari, Mamadou Traoré, Saadia Belmalha, Abderrahim Lazraq; data management, Hajar Zennouhi, Mamadou Traoré; validation, Rachid Lahlali, Abderrahim Lazraq, Saadia Belmalha; first draft, Hajar Zennouhi, Rachid Ez-zouggari, Mamadou Traoré; manuscript writing, Hajar Zennouhi, Rachid Ez-zouggari, Mamadou Traoré, Rachid Lahlali; proofreading and corrections, Abderrahim Lazraq, Rachid Lahlali; project administration, Rachid Lahlali. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: The data are contained within the manuscript.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Richa R , Kohli D , Vishwakarma D , Mishra A , Kabdal B , Kothakota A , et al. Citrus fruit: classification, value addition, nutritional and medicinal values, and relation with pandemic and hidden hunger. J Agric Food Res. 2023; 14: 100718. doi:10.1016/j.jafr.2023.100718. [Google Scholar] [CrossRef]
2. FAOSTAT. [cited 2025 Jan 1]. Available from: https://www.fao.org/faostat/fr/#data/QCL/visualize. [Google Scholar]
3. Bhatta UK . Alternative management approaches of citrus diseases caused by Penicillium digitatum (green mold) and Penicillium italicum (blue mold). Front Plant Sci. 2022; 12: 833328. doi:10.3389/fpls.2021.833328. [Google Scholar] [CrossRef]
4. Yao Y , Li Y , Zhao L , Li S , Zhou Z . Citrus lemon (Citrus limon (L.) Burm. f. cv. Eureka) essential oil controls blue mold in citrus by damaging the cell membrane of Penicillium italicum. LWT. 2023; 188: 115456. doi:10.1016/j.lwt.2023.115456. [Google Scholar] [CrossRef]
5. Khamsaw P , Sangta J , Chaiwan P , Rachtanapun P , Sirilun S , Sringarm K , et al. Bio-circular perspective of citrus fruit loss caused by pathogens: occurrences, active ingredient recovery and applications. Horticulturae. 2022; 8( 8): 748. doi:10.3390/horticulturae8080748. [Google Scholar] [CrossRef]
6. Silva E , Kanashiro AM , Maciel JRF , Dantas Lima Junior R , Botelho MAF , Pereira AK , et al. Decoding the Penicillium italicum-Citrus interaction: untargeted metabolomics sheds light on a neglected postharvest pathogen. J Agric Food Chem. 2025; 73( 43): 27806– 19. doi:10.1021/acs.jafc.5c07618. [Google Scholar] [CrossRef]
7. Elash W , Aborehab M , El-Sehrawy O , Shahin S . Control of green and blue molds of citrus fruits using some biocontrol agents under Egyptian conditions. Egypt J Phytopathol. 2023; 51( 1): 93– 102. doi:10.21608/ejp.2023.199708.1089. [Google Scholar] [CrossRef]
8. Citores L , Valletta M , Singh VP , Pedone PV , Iglesias R , Ferreras JM , et al. Deciphering molecular determinants underlying Penicillium digitatum’s response to biological and chemical antifungal agents by tandem mass tag (TMT)-based high-resolution LC-MS/MS. Int J Mol Sci. 2022; 23( 2): 680. doi:10.3390/ijms23020680. [Google Scholar] [CrossRef]
9. Raynaldo FA , Xu Y , Yolandani , Wang Q , Wu B , Li D . Biological control and other alternatives to chemical fungicides in controlling postharvest disease of fruits caused by Alternaria alternata and Botrytis cinerea. Food Innov Adv. 2024; 3( 2): 135– 43. doi:10.48130/fia-0024-0014. [Google Scholar] [CrossRef]
10. Millan AF , Gamir J , Farran I , Larraya L , Veramendi J . Identification of new antifungal metabolites produced by the yeast Metschnikowia pulcherrima involved in the biocontrol of postharvest plant pathogenic fungi. Postharvest Biol Technol. 2022; 192: 111995. doi:10.1016/j.postharvbio.2022.111995. [Google Scholar] [CrossRef]
11. Castelo Branco Melo NF , de MendonçaSoares BL , Marques Diniz K , Ferreira Leal C , Canto D , Flores MAP , et al. Effects of fungal chitosan nanoparticles as eco-friendly edible coatings on the quality of postharvest table grapes. Postharvest Biol Technol. 2018; 139: 56– 66. doi:10.1016/j.postharvbio.2018.01.014. [Google Scholar] [CrossRef]
12. Zhang X , Yue Q , Xin Y , Ngea GLN , Dhanasekaran S , Luo R , et al. The biocontrol potentiality of Bacillus amyloliquefaciens against postharvest soft rot of tomatoes and insights into the underlying mechanisms. Postharvest Biol Technol. 2024; 214: 112983. doi:10.1016/j.postharvbio.2024.112983. [Google Scholar] [CrossRef]
13. Van Wyk BE . A family-level floristic inventory and analysis of medicinal plants used in Traditional African Medicine. J Ethnopharmacol. 2020; 249: 112351. doi:10.1016/j.jep.2019.112351. [Google Scholar] [CrossRef]
14. Rivas MÁ , Casquete R , Gonçalves Dos Santos MTP , Benito MJ . An overview of the antifungal potential for aromatic plant extracts in agriculture and the food industry: a comprehensive analysis focusing on the Rubus, Cistus and Quercus genera against fungal infections of crops and food. Int J Food Microbiol. 2025; 436: 111209. doi:10.1016/j.ijfoodmicro.2025.111209. [Google Scholar] [CrossRef]
15. Ali M , Wang X , Haroon U , Chaudhary HJ , Kamal A , Ali Q , et al. Antifungal activity of Zinc nitrate derived nano Zno fungicide synthesized from Trachyspermum ammi to control fruit rot disease of grapefruit. Ecotoxicol Environ Saf. 2022; 233: 113311. doi:10.1016/j.ecoenv.2022.113311. [Google Scholar] [CrossRef]
16. Khadiri M , Boubaker H , Askarne L , Farhaoui A , Taoussi M , Haddou LA , et al. Exploration of the antifungal potential of aqueous extracts from two agave species against major postharvest diseases in apples. Postharvest Biol Technol. 2024; 214: 112992. doi:10.1016/j.postharvbio.2024.112992. [Google Scholar] [CrossRef]
17. Amine D , Mohamed B , Jamal I , Laila N . Antibacterial activity of aqueous extracts of Anacyclus pyrethrum (L) link and Corrigiola Telephiifolia pourr. from the middle atlas region-Morocco. Eur Sci J. 2017; 13( 33): 116. doi:10.19044/esj.2017.v13n33p116. [Google Scholar] [CrossRef]
18. Rezgui M , Majdoub N , Mabrouk B , Baldisserotto A , Bino A , Ben Kaab LB , et al. Antioxidant and antifungal activities of marrubiin, extracts and essential oil from Marrubium vulgare L. against pathogenic dermatophyte strains. J Mycol Med. 2020; 30( 1): 100927. doi:10.1016/j.mycmed.2020.100927. [Google Scholar] [CrossRef]
19. El Moussaoui A , Jawhari FZ , Almehdi AM , Elmsellem H , Fikri Benbrahim K , Bousta D , et al. Antibacterial, antifungal and antioxidant activity of total polyphenols of Withania frutescens.L. Bioorg Chem. 2019; 93: 103337. doi:10.1016/j.bioorg.2019.103337. [Google Scholar] [CrossRef]
20. Soto-Muñoz L , Taberner V , de la Fuente B , Jerbi N , Palou L . Curative activity of postharvest GRAS salt treatments to control citrus sour rot caused by Geotrichum citri-aurantii. Int J Food Microbiol. 2020; 335: 108860. doi:10.1016/j.ijfoodmicro.2020.108860. [Google Scholar] [CrossRef]
21. Cherrate M , Echchgadda G , Amiri S , Ezrari S , Radouane N , Oulad El Majdoub Y , et al. Biological control of major postharvest fungal diseases of apple using two Lamiaceae extracts. Arch Phytopathol Plant Prot. 2022; 55( 20): 2356– 81. doi:10.1080/03235408.2023.2166379. [Google Scholar] [CrossRef]
22. Naim N , Fauconnier ML , Ennahli N , Tahiri A , Baala M , Madani I , et al. Chemical composition profiling and antifungal activity of saffron petal extract. Molecules. 2022; 27( 24): 8742. doi:10.3390/molecules27248742. [Google Scholar] [CrossRef]
23. Karim H , Boubaker H , Askarne L , Cherifi K , Lakhtar H , Msanda F , et al. Use of Cistus aqueous extracts as botanical fungicides in the control of citrus sour rot. Microb Pathog. 2017; 104: 263– 7. doi:10.1016/j.micpath.2017.01.041. [Google Scholar] [CrossRef]
24. Santos Moura V , Olandin LD , Mariano BS , Rodrigues J , Devite FT , Arantes ACC , et al. Antifungal activity of citrus essential oil in controlling sour rot in Tahiti acid lime fruits. Plants. 2024; 13( 21): 3075. doi:10.3390/plants13213075. [Google Scholar] [CrossRef]
25. Taha NA , Elsharkawy MM , Shoughy AA , El-Kazzaz MK , Khedr AA . Biological control of postharvest tomato fruit rots using Bacillus spp. and Pseudomonas spp. Egypt J Biol Pest Control. 2023; 33( 1): 106. doi:10.1186/s41938-023-00752-6. [Google Scholar] [CrossRef]
26. Nikolaeva TN , Lapshin PV , Zagoskina NV . Method for determining the total content of phenolic compounds in plant extracts with folin–denis reagent and folin–ciocalteu reagent: modification and comparison. Russ J Bioorg Chem. 2022; 48( 7): 1519– 25. doi:10.1134/S1068162022070214. [Google Scholar] [CrossRef]
27. Hssaini L , Charafi J , Hanine H , Ennahli S , Mekaoui A , Mamouni A , et al. Comparative analysis and physio-biochemical screening of an ex-situ fig (Ficus carica L.) collection. Hortic Environ Biotechnol. 2019; 60( 5): 671– 83. doi:10.1007/s13580-019-00170-4. [Google Scholar] [CrossRef]
28. Iqbal S , Arifeen S , Akbar A , Zahoor S , Maher S , Khan N , et al. Phytochemical screening and antibacterial assay of the crude extract and fractions of Ferula oopoda. Pure Appl Biol. 2019; 8( 1): 742– 9. doi:10.19045/bspab.2019.80016. [Google Scholar] [CrossRef]
29. Mandal SC , Mandal V , Das AK . Qualitative phytochemical screening. In: Essentials of botanical extraction. Amsterdam, The Netherlands: Elsevier; 2015. p. 173– 85. doi:10.1016/B978-0-12-802325-9.00009-4. [Google Scholar] [CrossRef]
30. Solanki SL , Modi CM , Patel HB , Patel UD , Bhadarka DH . Phytochemical screening and thin-layer chromatography of six medicinal plants from the surroundings of Junagadh, Gujarat, India. J Pharmacogn Phytochem. 2019; 8( 4): 3122– 6. [Google Scholar]
31. Boubekeur S , Messaoudi M , Awuchi CG , Otekunrin OA , Sawicka B , Idjeri-Mecherara S , et al. Biological properties and polyphenols content of Algerian Cistus salviifolius L. aerial parts. Eur J Biol Res. 2022; 12( 2): 163– 80. [Google Scholar]
32. Benzeggouta N . Evaluation of the biological effects of aqueous extracts of medicinal plants alone and in combination [ dissertation]. Constantine, Algeria: Université des Frères Mentouri-Constantine 1; 2015. (In French). [cited 2026 Jan 1]. Available from: https://theses-algerie.com/3276558083009490/. [Google Scholar]
33. Alilou H , Bencharki B , Hassani LI , Barka N . Screening phytochimique et identification spectroscopique des flavonoïdes d’Asteriscusgraveolenssubsp. odorus. Afr Sci. 2014; 10( 3): 316– 28. [Google Scholar]
34. Murtiningsih TM , Supriningrum R , Nurhasnawati H . Identification and determination of saponin content from extract of Embelia borneensis bark. Al Ulum J Sains Dan Teknologi. 2023; 9( 3): 117. doi:10.31602/jst.v9i3.12499. [Google Scholar] [CrossRef]
35. El Khetabi A , El Ghadraoui L , Ouaabou R , Ennahli S , Barka EA , Lahlali R . Antifungal activities of aqueous extracts of Moroccan medicinal plants against Monilinia spp. agent of brown rot disease. J Nat Pestic Res. 2023; 5: 100038. doi:10.1016/j.napere.2023.100038. [Google Scholar] [CrossRef]
36. Lahlali R , Kumar S , Wang L , Forseille L , Sylvain N , Korbas M , et al. Cell wall biomolecular composition plays a potential role in the host type II resistance to fusarium head blight in wheat. Front Microbiol. 2016; 7: 910. doi:10.3389/fmicb.2016.00910. [Google Scholar] [CrossRef]
37. Islam T , Danishuddin , Tamanna NT , Matin MN , Barai HR , Haque MA . Resistance mechanisms of plant pathogenic fungi to fungicide, environmental impacts of fungicides, and sustainable solutions. Plants. 2024; 13( 19): 2737. doi:10.3390/plants13192737. [Google Scholar] [CrossRef]
38. Gholamnezhad J . Effect of plant extracts on activity of some defense enzymes of apple fruit in interaction with Botrytis cinerea. J Integr Agric. 2019; 18( 1): 115– 23. doi:10.1016/S2095-3119(18)62104-5. [Google Scholar] [CrossRef]
39. Matrose NA , Obikeze K , Belay ZA , Caleb OJ . Plant extracts and other natural compounds as alternatives for post-harvest management of fruit fungal pathogens: a review. Food Biosci. 2021; 41: 100840. doi:10.1016/j.fbio.2020.100840. [Google Scholar] [CrossRef]
40. Behiry SI , Al-Askar AA , Soliman SA , Alotibi FO , Basile A , Abdelkhalek A , et al. Plantago lagopus extract as a green fungicide induces systemic resistance against Rhizoctonia root rot disease in tomato plants. Front Plant Sci. 2022; 13: 966929. doi:10.3389/fpls.2022.966929. [Google Scholar] [CrossRef]
41. Mastino PM , Marchetti M , Costa J , Juliano C , Usai M . Analytical profiling of phenolic compounds in extracts of three Cistus species from sardinia and their potential antimicrobial and antioxidant activity. Chem Biodivers. 2021; 18( 6): e2100053. doi:10.1002/cbdv.202100053. [Google Scholar] [CrossRef]
42. Mohd Israfi NA , Mohd Ali MIA , Manickam S , Sun X , Goh BH , Tang SY , et al. Essential oils and plant extracts for tropical fruits protection: from farm to table. Front Plant Sci. 2022; 13: 999270. doi:10.3389/fpls.2022.999270. [Google Scholar] [CrossRef]
43. Moges GW , Manahelohe GM , Assege MA , Tasew BS , Molla DK , Belew AA . Phytochemical profiles and biological activity of Myrsine africana L.: a comprehensive review. Front Pharmacol. 2025; 16: 1565656. doi:10.3389/fphar.2025.1565656. [Google Scholar] [CrossRef]
44. Pant P , Pandey S , Dall’Acqua S . The influence of environmental conditions on secondary metabolites in medicinal plants: a literature review. Chem Biodivers. 2021; 18( 11): e2100345. doi:10.1002/cbdv.202100345. [Google Scholar] [CrossRef]
45. El Yaagoubi W , Soussi M , Aazza S , Saadia B , El Ghadraoui L . Phytochemicals, antioxidant activity, and antimicrobial effects of the fractions of Corrigiola telephiifolia pourr. Ecol Eng Environ Technol. 2024; 25( 10): 60– 74. doi:10.12912/27197050/191210. [Google Scholar] [CrossRef]
46. Gashaw AD , Desta MA , Yaya EE . A comprehensive review-current development in spectroscopic and chromatographic techniques for natural product analysis. Results Chem. 2025; 16: 102341. doi:10.1016/j.rechem.2025.102341. [Google Scholar] [CrossRef]
47. Al-Snafi AE , Ali Al-Saedy H , Ali Talab T , Majid WJ , Batiha GE , Abolfazl JS . The bioactive ingredients and therapeutic effects of Marrubium vulgare—a review. Int J Biol Pharm Sci Arch. 2021; 1( 2): 9– 21. doi:10.30574/ijbpsa.2021.1.2.0301. [Google Scholar] [CrossRef]
48. Qessaoui R , Ennasser S , Chafiki S , Ajerrar A , Alouani M , Chabbi N , et al. Antifungal activity of four plant extracts against Botrytis cinerea. Afr Mediterr Agric J Al Awamia. 2023; 140: 19– 36. [Google Scholar]
49. Piancatelli S , Sbodio AO , Michelon TB , Tsen M , Carranza-Rodriguez MC , Gutierrez-Baeza E , et al. Chitosan inhibits the growth of Penicillium digitatum and Penicillium italicum and protects oranges from green and blue mold during postharvest. Postharvest Biol Technol. 2026; 232: 113955. doi:10.1016/j.postharvbio.2025.113955. [Google Scholar] [CrossRef]
50. Thaeder C , Stanek J , Couvreur J , Borrego C , Brunissen F , Allais F , et al. Chemo-enzymatic synthesis and biological assessment of p-coumarate fatty esters: new antifungal agents for potential plant protection. Molecules. 2023; 28( 15): 5803. doi:10.3390/molecules28155803. [Google Scholar] [CrossRef]
51. OuYang Q , Duan X , Li L , Tao N . Cinnamaldehyde exerts its antifungal activity by disrupting the cell wall integrity of Geotrichum citri-aurantii. Front Microbiol. 2019; 10: 55. doi:10.3389/fmicb.2019.00055. [Google Scholar] [CrossRef]
52. Ouyang Q , Shi S , Liu Y , Yang Y , Zhang Y , Yuan X , et al. Inhibitory mechanisms of trans-2-hexenal on the growth of Geotrichum citri-aurantii. J Fungi. 2023; 9( 9): 930. doi:10.3390/jof9090930. [Google Scholar] [CrossRef]
53. Akullo JO , Kiage B , Nakimbugwe D , Kinyuru J . Effect of aqueous and organic solvent extraction on in-vitro antimicrobial activity of two varieties of fresh ginger (Zingiber officinale) and garlic (Allium sativum). Heliyon. 2022; 8( 9): e10457. doi:10.1016/j.heliyon.2022.e10457. [Google Scholar] [CrossRef]
54. Gow NAR , Lenardon MD . Architecture of the dynamic fungal cell wall. Nat Rev Microbiol. 2023; 21( 4): 248– 59. doi:10.1038/s41579-022-00796-9. [Google Scholar] [CrossRef]
55. Pavoni L , Perinelli DR , Bonacucina G , Cespi M , Palmieri GF . An overview of micro- and nanoemulsions as vehicles for essential oils: formulation, preparation and stability. Nanomaterials. 2020; 10( 1): 135. doi:10.3390/nano10010135. [Google Scholar] [CrossRef]
56. Kasmi M , Aourach M , El Boukari M , Barrijal S , Essalmani H . Efficacité des extraits aqueux des plantes aromatiques et médicinales contre la pourriture grise de la tomate au Maroc. Comptes Rendus Biol. 2017; 340( 8): 386– 93. doi:10.1016/j.crvi.2017.07.010. [Google Scholar] [CrossRef]
57. Hayat J , Akodad M , Moumen A , Baghour M , Skalli A , Ezrari S , et al. Phytochemical screening, polyphenols, flavonoids and tannin content, antioxidant activities and FTIR characterization of Marrubium vulgare L. from 2 different localities of Northeast of Morocco. Heliyon. 2020; 6( 11): e05609. doi:10.1016/j.heliyon.2020.e05609. [Google Scholar] [CrossRef]
58. Bouterfas K , Mehdadi Z , Elaoufi MM , Latreche A , Benchiha W . Antioxidant activity and total phenolic and flavonoids content variations of leaves extracts of white Horehound (Marrubium vulgare Linné) from three geographical origins. Ann Pharm Fr. 2016; 74( 6): 453– 62. doi:10.1016/j.pharma.2016.07.002. [Google Scholar] [CrossRef]
59. Doudach L , Meddah B , Faouzi M , Khatib AM , Lalou C , Hammani K , et al. Cytotoxic and antioxidant activity of various extracts of Corrigiola telephiifolia Pourr. Int J Pharm Pharm Sci. 2013; 5( 3): 154– 8. [Google Scholar]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools