 Open Access
Open Access
ARTICLE
Integration of Computer Vision and Physicochemical Parameters for Post-Harvest Ripeness Classification of TomEJC Mango
1 Department of Food Science and Technology, Faculty of Agriculture, University of Peradeniya, Peradeniya, Sri Lanka
2 Department of Computer Science, Faculty of Natural Sciences, The Open University of Sri Lanka, Nugegoda, Sri Lanka
3 Department of Computing, Atlantic Technological University, Donegal Campus, Letterkenny, Donegal, Ireland
4 Department of Civil Engineering and Construction, Atlantic Technological University, Sligo, Ireland
* Corresponding Authors: Rasanjali Samarakoon. Email: ; Upaka Rathnayake. Email:
(This article belongs to the Special Issue: Advances in the Physiological, Biochemical and Molecular Mechanisms Regulating Fruit Ripening in Tropical Fruits)
Phyton-International Journal of Experimental Botany 2026, 95(4), 19 https://doi.org/10.32604/phyton.2026.078657
Received 05 January 2026; Accepted 31 March 2026; Issue published 28 April 2026
Abstract
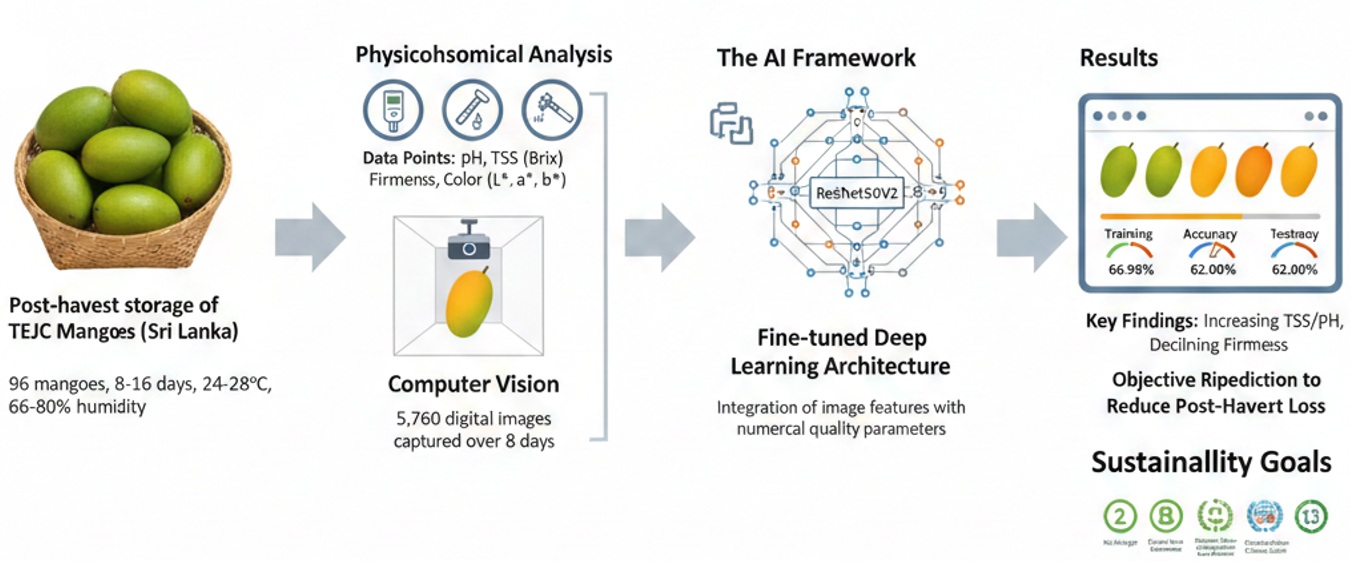
Accurately determining the optimal post-harvest storage period is still a major challenge in mango processing, especially for the Tom EJC (TEJC) variety, due to reliance on subjective visual evaluations, leading to inconsistent product quality and increased post-harvest losses. This study presents an artificial intelligence-based framework combining computer vision and physicochemical analysis to objectively predict the optimal post-harvest storage period of TEJC mango before processing. TEJC mangoes of grade one were stored for eight days at 24–28°C temperature and 66.4–80% relative humidity. Daily measurements of pH, Total Soluble Solids (TSS), firmness, and peel color parameters (L*, a*, b*) were evaluated along with an image dataset of 5760 photos taken under variable lighting. Image data were then combined with numerical quality parameters to train and evaluate a deep learning model based on a fine-tuning architecture of ResNet50V2 for the classification of multi-class ripeness stages. The model achieved 66.96% of training and 62% testing accuracy, demonstrating the feasibility of integrating computer vision and physicochemical parameters for preliminary multi-class ripeness classification under non-uniform real-world conditions. The ripening trends were reflected in increasing TSS and pH values and declining fruit firmness. Among peel colour parameters, a* was strongly associated with ripening advancement. The findings underscore the potential of deep learning tools as non-destructive decision-support systems for post-harvest mango processing. The proposed framework serves as a proof-of-concept demonstrating its potential applicability in real-world scenarios. Nevertheless, the dataset used in this study enabled proof-of-concept evaluation; it represents a potential limitation for deep learning models, which typically benefit from larger and more diverse training sets.Graphic Abstract
Keywords
Mango (Mangifera indica L.) is among the most economically significant tropical fruits globally, valued for its distinctive flavour, nutritional value, and applicability in processed food. Mangoes exhibit noticeable colour changes during ripening, transitioning from green to various shades depending on the cultivar, due to chlorophyll degradation and the emergence of pigments like carotenoids and anthocyanins [1,2]. Key quality parameters of mangoes, including firmness, Total Soluble Solids (TSS), and pH, undergo notable changes after harvesting [3]. Economically, mango cultivation and processing offer a significant source of income, especially in the tropical and subtropical regions, where it is one of the most cultivated fruits in the world [4,5,6]. Economic profitability in mango cultivation is often undermined by excessive post-harvest losses due to improper ripeness assessments and storage practices, leading to inconsistent product quality, more waste, and higher labour costs [2,7,8]. Addressing these economic challenges is critical to guaranteeing profitability and livelihoods, particularly among small-scale farmers.
Harvesting mangoes at the correct time is highly important [9,10,11]. Harvesting either too early or too late disrupts normal ripening. This can adversely affect the flavour development, uneven texture, and reduce the shelf life. To date, many farmers rely on visual appearance and personal experience when determining harvest time, which results in inconsistent end quality [2,12,13,14]. In addition, post-harvesting is always challenging [15,16,17]. Improper temperature and humidity controls are two major factors affecting the mango quality during transportation and storage. The combined effects would enhance the physiological deterioration, thus increasing the fruit damage.
Mangoes are one of the principal fruit crops in Sri Lanka that supply inputs to both local and export markets [18,19]. The post-harvest loss of mangoes in Sri Lanka was put at 17 to 36%, which disturbs the supply of fresh mangoes to the consumer and also the economic sustainability of mango production in the country as a whole [20,21]. When considering the Tom EJC (TEJC) variety, it is one of the many mango cultivars, most renowned for its exceptional flavor, texture, and aroma. However, as with many mango cultivars, it is highly perishable with a comparatively short shelf life owing to its rapid ripening nature and proneness to microbial spoilage [22]. Technological breakthroughs such as image processing and Artificial Intelligence (AI) pose solutions to above stated issues related to mango harvesting and post-harvesting [23,24,25]. As pointed out by Prabhu et al. [23], throughout industrial processing, the visual quality of the fruit is monitored by a combination of human supervision and computer vision technology to ensure optimal levels of production. Computerized systems must accurately grade mangoes according to their stage of ripeness, regardless of orientation. Since mangoes lack uniform coloration, assessing ripeness using overall colour features is challenging. Instead, localized colour features from decomposed pieces are utilized to enhance precision and effectiveness [23].
Over the past decades, it has been significant in agriculture, where it aids in improving productivity by analyzing images in real time. Image processing is about capturing images and processing them to obtain useful information and other significant parameters, making agriculture more efficient and well-informed [26]. Specifically, deep learning platforms such as Convolutional Neural Networks (CNN) significantly contribute to fruit quality assessments, ensuring more objective, faster, and more accurate reviews [27].
Besides, Artificial Intelligence (AI) is increasingly used in agriculture to forecast the shelf life and quality of fruits based on massive data sets to build and develop robust prediction models [28,29]. The mango processing industry, however, usually experiences difficulties related to determining optimal ripeness and storage duration, primarily due to reliance on subjective visual inspections. The resulting consequences of these procedures could be inconsistent product quality, waste accumulation, and high labor costs, as discussed above. The integration of AI technology, particularly image processing and machine learning techniques, in identifying the optimal storage period for TEJC mangoes presents significant opportunities for improving post-harvest management practices. To fully utilize these technologies for improving the quality and shelf life of TEJC mangoes, several knowledge gaps must be filled.
Therefore, the objective of this study was to develop and evaluate a proof-of-concept AI-based framework that integrates image-derived features with physicochemical parameters (pH, TSS, firmness, and peel colour) to classify post-harvest ripening stages of TEJC mangoes under ambient storage conditions. Unlike previous studies that relied solely on visual features or single quality indicators, this work combines non-destructive image analysis with laboratory-validated ripening metrics for a specific cultivar and origin, thereby contributing cultivar-specific insights into AI-assisted post-harvest quality assessment.
Aligning with the United Nations Sustainable Development Goals (SDGs), namely SDG 2 (Zero Hunger), SDG 8 (Decent Work and Economic Growth), SDG 12 (Responsible Consumption and Production), and SDG 13 (Climate Action), the proposed model anticipates drastically reduced food waste, improved utilization of resources, and reduced climate footprint Despite initial high capital costs, the model provides significant long-term economic and environmental savings, promoting sustainable farming practices and resilience.
2.1 Sample Collection and Experimental Design
TEJC mangoes were sourced from CIC Agribusiness (Pvt) Ltd., located at Pelwehera, Dambulla, Sri Lanka (coordinates 7°53′34.05′′ N, 80°40′58.44′′ E). The mango trees from which the fruits were harvested were planted in May 2016 and were located in Orchard/Block No. 1. Harvesting was done after four years of planting. This study focused on the first grade of TEJC mangoes, which typically weigh above 600 g, have smooth skin, and show no dark spots. The study was conducted using two batches of TEJC mangoes.
A day after harvesting, TEJC mangoes were sourced from CIC Agribusiness, Sri Lanka, and transported to the laboratory under ambient conditions. Upon arrival, the mangoes were visually inspected to ensure uniformity in size, shape, and the absence of physical damage. Each batch contained 48 mangoes, making a total of 96 mangoes used for the experiment. It is acknowledged that this sample size, while sufficient for a controlled proof-of-concept study, represents a limitation with respect to the generalizability of deep learning model outputs; future work employing a larger and more diverse mango population would improve model robustness. Six replicates of mangoes were taken per day for image capturing and physicochemical analyses. All the mango fruits were labeled according to their replicates and date for tracking them precisely over the study duration. A preliminary trial was carried out before carrying out the actual experiment to check the ripening pattern of the mangoes and the storage period. The TEJC mangoes were stored in two batches, with each batch stored for 8 days. The storage conditions maintained a temperature range of 24–28°C and an RH range of 66.4–80%. Preliminary comparison of physicochemical trends between the two batches showed similar ripening patterns. Therefore, data from both batches were pooled for analysis.
During storage, mangoes were analyzed regularly to assess changes in weight, pH, total soluble solids (TSS), firmness, and peel color. Additionally, relative humidity (RH) and temperature readings were recorded daily using the psychrometer to monitor environmental conditions. Each mango was individually placed on the weighing balance (Gram EK—Compact Precision Laboratory and Quality Balance), and the weight (in grams) was recorded once the reading stabilized. To ensure accuracy, the balance was positioned on a flat, vibration-free surface to prevent fluctuations. Mangoes were handled carefully to avoid moisture loss or physical damage before weighing. Upon receiving a batch, the initial weight of each mango was recorded. Subsequently, the final weight was measured daily before taking other measurements to monitor weight changes throughout the ripening period. The weight loss percentage was calculated using the following Eq. (1).
The outer peel color of the mango was measured without any modifications to the fruit surface to ensure accurate reading. Before each use, the colorimeter (Colorimeter PCE-CSM 1) was calibrated using a white standard calibration plate. The device was then placed directly on the mango surface, ensuring full contact with the peel for precise measurement. The L* (lightness), a* (red-green), and b* (yellow-blue) values were recorded from the colorimeter. To account for color variations, readings were taken at 30 different points on each mango. Additionally, consistent lighting conditions were maintained throughout the study to minimize discrepancies in color measurement.
Mangoes were placed on a flat, stable surface to ensure accurate firmness measurements. The fruit hardness tester probe [5000 g × 1 g Precision Fruit Sclerometer (Fruit Hardness Tester, Model: FR-5105)] was gently pressed against the mango peel, and a standardized force was applied until the probe penetrated the mango. The firmness value, expressed in Newtons (N), was recorded. To ensure consistency and obtain reliable data, measurements were taken at 30 different points on each mango. The device was reset to zero after each reading to maintain accuracy and prevent measurement errors.
A small portion of mango pulp was extracted using a muslin cloth and placed on a watch glass for pH measurement. From each mango, 15 pieces were cut from each side, homogenized, and pulp was extracted, making a total of 30 pieces per fruit to ensure a representative sample. Before the measurement, the pH meter (Extech PH100 pH meter) was calibrated using standard buffer solutions (pH 4.0, 7.0, and 10.0). The electrode of the pH meter was then immersed in the mango pulp sample, and the pH reading was recorded once the value stabilized. To maintain accuracy, the electrode was cleaned with distilled water before and after each measurement. All measurements were conducted at room temperature to ensure consistency across samples.
A small portion of mango pulp was squeezed to extract the juice. The extracted juice was then filtered using a muslin cloth to remove any solid particles. Before measurement, the pocket refractometer [digital Brix meter—MINT IN BOX—ATAGO Pocket Refractometer, PR-1 (0–32% Brix Range, 9 V)] was calibrated using distilled water. A few drops of the filtered mango juice were carefully placed on the sample plate of the Brix meter. The Brix value (%), which indicates the total soluble solids (TSS) content, was read from the digital display and recorded. To prevent contamination and ensure accuracy, the sample plate was cleaned with distilled water after each measurement. Additionally, all readings were taken at a consistent room temperature to maintain reliability.
2.3 Image Capturing and Dataset Preparation
Mango fruit images were collected across eight ripening stages under controlled lighting conditions to ensure consistency in visual quality. Each fruit sample was photographed using a fixed camera setup to minimize variations caused by distance, illumination, and background noise. Images were stored according to their corresponding ripening stage labels, which ranged from Stage 1 (immature) to Stage 8 (fully ripened).
Each captured image was paired with physicochemical measurements obtained from laboratory testing. The recorded parameters included color measurements (L*, a*, b* values), fruit firmness (Newton), total soluble solids (TSS measured in Brix), and pH level. These values were stored in structured CSV files and mapped to the corresponding image file names to ensure accurate data association.
All images were resized to 224 × 224 pixels and normalized by scaling pixel values between 0 and 1 prior to model training. This preprocessing ensured compatibility with the pre-trained convolutional neural network architecture and improved training stability.
2.4 Dataset Splitting Strategy
The dataset was divided into training and testing subsets using predefined folder structures and corresponding CSV annotation files. The training dataset consisted of images and associated physicochemical parameters used to train the model, while the testing dataset was used exclusively for performance evaluation.
Each dataset contains:
- 1.Image files organized by ripening stage folders.
- 2.Excel file linking image filenames with physicochemical measurements and stage labels.
During training, batches were generated dynamically using a custom data generator to simultaneously feed image data and regression targets into the model. The testing dataset followed the same structure but was used only for validation and performance assessment.
 |
The proposed system uses a transfer learning approach based on the ResNet50V2 deep convolutional neural network pre-trained on the ImageNet dataset. The base convolutional layers were utilized for feature extraction, while the fully connected classification layers were removed.
The implemented model is a multi-output deep learning architecture but operates using a single input modality (image input). The extracted deep image features are used to perform two simultaneous tasks:
- Classification Task: Predicting mango ripening stages using a Softmax output layer.
- Regression Task: Predicting physicochemical properties including L*, a*, b*, firmness, TSS, and pH values using a linear activation layer.
The model structure allows shared feature learning, where visual features extracted from images are used for both classification and regression output simultaneously.
The required resources for the coding process included Google Colab as the coding platform, with Python as the programming language. The libraries utilized were TensorFlow, Keras, NumPy, OpenCV, ImageDataGenerator, and Matplotlib.
In this research study, the technique of rescaling was utilized as a preprocessing step for the model. This involved multiplying the input data by a certain value before any other processing. The original images were in the RGB color space with coefficient values ranging from 0 to 255. However, such values were deemed unsuitable for processing by their models, given a typical learning rate. Thus, the values were scaled between 0 and 1 by multiplying them by a factor of 1/255. This allowed the models to effectively process the images and achieve more accurate results with model convergence and training stability. The images were resized to (224,224,3) and used a CNN model named ResNet50V2, which was a pre-trained model to classify and train the images by removing the top layers of the CNN model (top = false). This classified the trained images into 8 stages and gave the output of classification and numerical values of the 6 data values collected.
Convolutional Neural Networks (CNNs) are a type of neural network designed for image recognition and processing, inspired by the human visual cortex. They use small filters to extract features like edges and textures, which are then processed through multiple layers to identify complex patterns [30]. This structure enables CNNs to perform well in tasks such as object classification and scene recognition [31]. CNNs are a subset of deep learning algorithms that are effective for tasks such as image classification because they can learn to recognize patterns and features in images. It is possible to create a CNN-based image processing model that can successfully detect mango ripening stages by training a convolutional neural network on a TEJC mango image dataset. The CNN-based ripening stages detection of a TEJC mango will aid in determining the optimized storage period and the physicochemical parameters, together with ultimately providing a good quality product for the food processing industry for production. Compared to the traditional method, which is classified manually by human operators, it might lead to personal errors.
The overall accuracy of the algorithm is influenced by the number of layers, wherein an increase in the number of layers results in moderate accuracy. However, an increase in the number of layers also leads to an increase in processing time. These layers execute a mathematical operation that requires the image matrix and filter as input. The flowchart depicting the entire CNN process and the architecture of the CNN algorithm utilized for model creation is shown below in Fig. 1.

Figure 1: Flow chart diagram of model creation.
Following the construction of the Convolutional Neural Network (CNN) architecture, the model was compiled by specifying the optimization algorithm, loss function, and performance metrics for training. The compilation procedure accepts three arguments: Optimizer, Loss function, and Metrics. The model training was performed using the Adam optimization algorithm. The optimizer was configured using default TensorFlow parameters, including an initial learning rate of 0.001. The model was trained using a batch size of 32 to ensure efficient learning and stable gradient updates.
Key training configurations include:
- Optimizer: Adam
- Learning Rate: 0.001
- Batch Size: 32
- Loss Functions:
- Categorical Cross-Entropy for classification
- Mean Squared Error (MSE) for regression
Evaluation Metrics:
- Classification Accuracy for stage prediction
- Mean Squared Error for physicochemical parameter prediction
To leverage transfer learning, the convolutional layers of the ResNet50V2 base model were frozen during training. Only the newly added fully connected output layers were trained, which reduces overfitting and improves generalization when working with limited datasets. No explicit regularization techniques, such as dropout or weight decay, were applied, as the frozen pre-trained layers inherently reduce overfitting risk.
The evaluation metric is to be used for monitoring the performance of the model during training. Metrics were used to evaluate the performance of the model on validation data, and the ‘accuracy’ measure was applied in this classification task research. Moreover, this evaluation metric represents the proportion of correctly classified instances over the total number of instances.
During this process, the CNN model received a large data set consisting of labeled images of TEJC mangoes, categorized as either Day 1, Day 2, Day 3, Day 4, Day 5, Day 6, Day 7, or Day 8, for classification purposes. This constitutes the primary function of the CNN model. Once the training process was completed, the model was used to make predictions, and the resulting probability distribution was analyzed. To train the CNN model, the fit method was used on the training dataset.
Overall, the ripeness prediction in this study was based on the integration of image-derived features with key physicochemical quality parameters known to reflect mango maturation. During training, batches were generated dynamically using a custom data generator to simultaneously feed image data and physicochemical regression targets into the model. It is important to note that the physiochemical parameters, TSS, pH, firmness, L*, a*, and b* serve exclusively as regression targets during model training and are not used as input features. The model receives only image data as input and learns to predict both the ripening stages (classification) and the physiochemical values (regression) from visual features alone. The testing dataset followed the same structure but was used only for validation and performance assessment. Both losses are optimized concurrently during training. Mean Squared Error is reported alongside classification accuracy to evaluate regression performance and ensure accurate estimation of fruit quality attributes.
2.7 Validation Parameters Clarification
Model validation was performed using the testing dataset containing both image data and physicochemical measurements. The evaluation process assessed:
- Classification accuracy based on predicted ripening stages from image inputs.
- Regression accuracy using predicted physicochemical parameters compared with laboratory-measured values.
Although physicochemical parameters were used as regression targets during training, the model predictions relied solely on image inputs. This approach allows the system to estimate fruit quality characteristics directly from visual information without requiring laboratory measurements during deployment.
All statistical analyses were performed using R software (version 4.4.3) and Minitab (version 21.2). Mean values and standard deviations were calculated for all physicochemical parameters. One-way analysis of variance (ANOVA) followed by Tukey’s HSD test was used to evaluate significant differences of physiochemical parameters during the storage period. Pearson correlation analysis was conducted to assess relationships among quality parameters. Statistical significance was determined at α = 0.05.
3.1 Physicochemical Changes in Mangoes during Storage
The pH of TEJC mangoes increased gradually over the storage period, with notable variations depending on the ripening stage. Initially, the pH was lower, indicating higher acidity, but as ripening progressed, acidity decreased due to the degradation of organic acids. During storage, mangoes typically exhibit an increase in pH levels, which correlates with a decrease in titratable acidity as the fruit ripens. Ibarra-Garza et al. [32] highlighted that acidity loss leads to a gradual pH increase in mangoes undergoing postharvest ripening, noting variability in results due to biological differences in mango cultivars. This trend is also supported by Yin et al. [33]. who indicated that continuous ripening and spoilage during prolonged storage decreased acidity levels, thus affecting the pH. Fig. 2a shows the pH of the TEJC mangoes over an 8-day storage period.
The total soluble solids (TSS) content is a critical indicator of the ripeness and palatability of fruits, including mangoes, and significantly influences their sensory attributes. Throughout the storage period of TEJC mangoes, a notable change in TSS levels occurred, which correlates closely with the ripening process. Research indicates that total soluble solids (TSS) in mangoes initially increase during ripening due to the starch converted into sugars, enhancing sweetness, which is a crucial indicator of ripeness [34]. Specifically, Mounika et al. [35] noted that an increase in TSS is associated with the ripening of fresh products, reflecting the metabolic conversions occurring in the fruit. In this study, TEJC mangoes showed a gradual increase in TSS during early storage, consistent with Kumari et al. [36], who observed similar trends in guava and papaya due to polysaccharide conversion. This increase enhances both flavor and market value, as consumers tend to prefer sweeter fruits. Studies, including those by Mounika et al. [35] indicated that prolonged storage activates metabolic pathways that break down sugars, ultimately reducing TSS and compromising fruit quality. The physicochemical trends observed in TEJC mangoes, including an increase in TSS up to ~16° Brix and a marked firmness decline from ~37 N to ~11 N over storage, are consistent with other mango cultivars. In Keitt mangoes, TSS values were reported to increase from ~9 to ~17° Brix while firmness decreased drastically across ripening stages, illustrating a similar pattern of sugar accumulation and softening during ripening [32]. Fig. 2b shows the TSS change of TEJC mangoes over an 8-day storage period.

Figure 2: Changes in the key physicochemical parameters of TEJC mangoes over an 8-day storage period. (a) pH; (b) TSS; (c) Firmness.
The firmness of TEJC mangoes decreased significantly over storage time. This softening is attributed to the enzymatic degradation of cell walls, making mangoes more susceptible to mechanical damage. Firmness is a critical quality parameter that determines mango quality. Studies show that mangoes experience significant texture degradation due to enzymatic activity and water loss. For instance, Sivankalyani et al. [37] indicated that chilling temperatures reduce firmness in mangoes stored below optimal temperatures. Fig. 2c shows the decrease in firmness of TEJC mangoes over the storage time.
The color change of TEJC mangoes during storage is a fundamental indicator of ripening and quality, directly impacting consumer acceptance and marketability. In this research, it was observed that TEJC mangoes transitioned from a light green-yellow color to a rich golden-yellow or orange hue as they ripened. Fig. 3a,b show the changes in color of TEJC mangoes over the storage time. This progression aligns with the well-documented ripening process in mangoes, wherein chlorophyll degradation and the accumulation of carotenoids contribute to the color transformation [38,39]. Multivarietal studies of Indian cultivars further demonstrated distinct ripening profiles in TSS, pH, and color across varieties, indicating genetic influences on ripening dynamics [40]. Comparable increases in sugar and colorimetric changes, with decreases in organic acids and firmness, were also documented in Sri Lankan mango cultivars, reinforcing that these physicochemical transitions are common but cultivar-specific in magnitude [41].


Figure 3: Peel color changes of TEJC mango over an 8-day storage period. (a) L*, a*, b* change; (b) Color change.
Initially, the mangoes exhibited a predominant green peel color, which signifies unripe fruit. As ripening commenced, a notable shift toward yellow hues occurred, indicative of the breakdown of chlorophyll and the synthesis of carotenoids such as beta-carotene [42]. This progression was quantitatively supported by the Li color scale, which rates color change from green (0) to fully yellow (6), demonstrating how, over time, the yellowed area of the fruit skin increased significantly, typically reaching the fully yellow stage within a defined storage period [38]. Notably, this aligns with the findings from Javed et al. [38], who specifically noted that the control of chlorophyll during storage was critical in maintaining quality.
This study aligns with Theanjumpol et al. [43], who found that mangoes transition from green to mature hues during storage, improving sugar content and flavor, key factors for marketability. Ripening enhances overall fruit quality, particularly color, due to biochemical changes. Additionally, environmental conditions play a crucial role; Ngo et al. [44] noted that lower temperatures slow de-greening, delaying ripening.
The assessment of weight loss in TEJC mangoes throughout the storage period reveals crucial insights into the fruit’s ripening dynamics. The mean weight loss percentages over 8 days period of storage of TEJC mangoes are shown in Table 1. As mangoes progress toward ripeness, weight loss becomes increasingly pronounced, primarily due to physiological processes such as transpiration and respiration. The present study observed a notable weight loss in TEJC mangoes, which is consistent with previous findings in various mango cultivars. For instance, Dieye et al. [45] reported that under ambient storage conditions at 25°C, mangoes experienced significant moisture loss, reflecting typical patterns of ripening and subsequent deterioration.
Throughout the storage period, TEJC mangoes exhibited gradual weight loss, particularly after the initial stage of ripening. This pattern has been corroborated by various studies conducted on different varieties of mango. For example, Dieye et al. [45] documented weight losses of approximately 7.1% in ‘Apple’ mangoes and 4.9% in ‘Ngowe’ mangoes after 6 to 7 days of storage at similar temperatures, indicating that the weight loss observed in TEJC mangoes, which is approximately 9.7%, could be comparable. Further emphasizing the climacteric nature of mangoes, respiration rates increase as the fruit ripens, leading to higher weight losses over time [45].
Additionally, other research highlights the role of environmental conditions in weight loss during ripening. Weight loss in climacteric fruits like mangoes often correlates directly with increased metabolic activity. For instance, Gill et al. [46] indicated that physiological weight loss in ripening mangoes is progressive, driven by the fruit’s respiration and transpiration rates, leading to significant reductions in fresh weight over time. This finding suggests that higher respiration rates due to ripening could compound weight loss in TEJC mangoes.
Table 1: Weight loss percentage during 8 days storage period.
| Day | Weight Loss Percentage (%)* |
|---|---|
| 1 | 0.0 ± 0.00e |
| 2 | 1.5 ± 0.20d |
| 3 | 2.34 ± 0.23d |
| 4 | 2.82 ± 0.41d |
| 5 | 4.79 ± 0.54c |
| 6 | 5.08 ± 0.48c |
| 7 | 7.28 ± 1.22b |
| 8 | 9.67 ± 1.61a |
Total soluble solids (TSS) showed a moderate positive correlation with pH (r = 0.65, p < 0.05) and a strong negative correlation with firmness (r = −0.95, p < 0.05), indicating that sugar accumulation during ripening was closely associated with tissue softening in TEJC mangoes. In addition, the colorimetric parameter a* demonstrated a strong positive correlation with TSS (r = 0.95, p < 0.05), suggesting that increased soluble sugar content coincided with enhanced red–yellow pigmentation development.
These interrelationships reflect coordinated biochemical and structural changes during ripening and appear consistent with climacteric fruit physiology. However, given the relatively small sample size (n = 180), these findings should be interpreted as indicative trends within the controlled storage conditions of this study. Further validation using larger datasets and additional storage conditions would strengthen the statistical robustness of these associations.

Figure 4: Correlation Matrix of TEJC mango attributes over 8 days storage period.
3.2 AI-Based Prediction of Mango Ripening and Quality Parameters
The system was successfully compiled, and after completing the training part of the real-time image processing algorithm, which is developed to identify the TEJC mango ripening stage and physicochemical parameters, specific results were obtained, along with an assessment of the overall model performance. A total of 5760 images were used to train the model with 20 epochs to train the overall model for every epoch, which is reasonable considering the size of the dataset and the complexity of the neural network.
Recent advancements in image processing techniques have enabled effective monitoring of mango ripening stages. Utilizing AI models, researchers have developed systems capable of analyzing visual attributes to predict quality parameters. Zhang et al. [47] highlighted the potential of similar technologies in tracking quality changes during storage, framing these insights within broader applications of AI in food science.
Convolutional Neural Networks (CNNs) have shown promise in classifying mango ripeness by processing images. The performance of these AI models has been benchmarked against traditional methods, revealing accuracy in determining ripeness stages along with efficiency advantages in real-time applications. This represents a significant development aligned with emerging trends in real-time quality assurance processes in the food industry [48]. In a study by Nambi et al. [49], the CNN-based RGB image processing method was employed to predict the ripeness levels of “Alphonso” mangoes. While specific accuracy metrics were not explicitly stated, the research emphasizes notable efficiency in recognizing varying ripeness levels non-destructively, supporting the effectiveness of machine vision applications in this context.
Therefore, these results suggest that the CNN-based TEJC mango ripening stage identification algorithm is able to identify the ripening stage along with TSS, pH, firmness, and peel color (L*, a*, b*) of TEJC mango with moderate accuracy. Table 2 indicates the overall CNN model training and testing accuracy of the developed algorithm for identifying the ripening stage of TEJC mangoes.
Table 2: Overall Model Performance Information.
| Training Data | Testing Data | ||
|---|---|---|---|
| Loss | Accuracy | Loss | Accuracy |
| 34% | 67% | 32% | 62% |
The results obtained after completing the model training are shown in Fig. 5a. The graph represents the relationship between accuracy and the number of epochs, for both training accuracy and validation accuracy. The accuracy for each epoch is shown for the training and validation. In addition, Fig. 5b shows both the training and validation loss of the CNN model. The graph depicts the relationship between loss and the number of epochs, showing both the testing and validation loss. This indicates how accurate the model’s predictions were for the given data. The achieved training and validation loss values are relatively low, indicating good performance of the model. Furthermore, Fig. 5c shows the Mean Squared Error (MSE) for training and validation over multiple epochs in the model. Initially, MSE drops steeply, indicating rapid learning, and then gradually stabilizes, suggesting convergence. The training and validation curves follow a similar trend, meaning the model generalizes well without signs of overfitting. If further improvement is needed, consider tuning hyperparameters, increasing epochs, or adjusting the model architecture.

Figure 5: Model accuracy, loss, and regression MSE. (a) For accuracy; (b) For Model loss; (c) For MSE.
The ML algorithm was developed for making a single prediction using a trained CNN model to obtain the prediction result of the 8 ripening stages of the TEJC mango. Therefore, the prediction function of the CNN model is developed to predict the test image. The test image path should feed into the system, and it can predict the ripening stage, the pH, TSS, color, and the firmness values of the TEJC mango. The output of the model is a probability distribution over the possible classes. Finally, based on the class with the highest probability, the algorithm predicts the fruit ripening stage. Fig. 6 shows the output of the model prediction, where the mango is recognized as being in stage 3. The predicted quality parameters include peel color values of L* = 59.48, a* = 0.97, and b* = 35.35, along with a firmness value of 28.04, total soluble solids (TSS) of 7.60, and a pH value of 4.08.

Figure 6: Results for the TEJC mango ripening stage and physiochemical parameters.
This particular CNN-based real-time image processing TEJC mango ripening stage identification algorithm was developed in order to identify the ripening stage. The overall system algorithm is able to obtain overall model performance with 67% of training set accuracy and 62% of testing set accuracy. Therefore, these research findings indicated that the TEJC mango ripening stage identifying image processing algorithm based on CNN can effectively categorize the ripening stages and physicochemical parameters of TEJC mangoes with a notable level of accuracy. Furthermore, the developed CNN model is also able to provide accurate output results for maturity stage prediction, and it ensures the efficiency and effectiveness of the developed algorithm.
AI-driven methods for assessing mango maturity present enhanced efficiency over conventional manual inspections, which are often subjective. While traditional approaches are useful, they lack the precision provided by machine learning algorithms, as noted in recent empirical studies [50]. The objectivity and speed of AI models reflect a transformative shift in quality management for postharvest products.
The model developed with AI demonstrated potential applications in replacing traditional manual sorting and visual inspection with automated analysis of mango ripeness stages. By employing image processing techniques to assess colorimetric values and other physicochemical parameters, processors can ensure that only mangoes of optimal ripeness are utilized for processing. This not only increases product quality but also minimizes the risk of post-harvest losses, as mentioned in research by Meighani et al. [51], on high technologies in post-harvest processing. Therefore, the use of AI-based approaches gives a more standardized and effective system for mango quality management in the food industry.
In addition, countries like Sri Lanka are experiencing skill migration to the developed countries, and therefore, the readily available skillset of the workforce is decaying. On top of that, youth have little or no interest in agriculture-related professions due to inequality with other professions, such as technologically oriented ones. Therefore, it is a challenge to understand the harvest and engage in the related activities solely based on experience, which used to be the case. The proposed AI-based prediction model has a potential to overcome such human factors in decision making. Therefore, the proposed research directly aligns with SDG 8—Decent Work and Economic Growth. In addition, the research presented proposes positive inputs to SDG 2—Zero Hunger, SDG 9—Industry, Innovation and Infrastructure, SDG 12—Responsible Consumption and Production, and SDG 13—Climate Action. It is well known that a significant portion of agricultural production is wasted on harvesting, transportation, and storage. This is a critical issue for today’s world. Therefore, this research helps in seeking solutions for such problems.
Despite the promising results, several limitations in this research are to be acknowledged. This study focused only on first-grade TEJC mangoes, which limits the generalizability of the findings to other cultivars and quality grades. Although the dataset comprised 5760 images linked with physicochemical measurements, these were derived from only 96 mangoes collected from two batches under controlled conditions. While preliminary comparisons indicated similar ripening trends between batches, the batch effect was not statistically analyzed. The relatively small and homogeneous sample size may restrict the model ability to capture the full biological variability associated with mango ripening. Such variability can arise from genetic differences, cultivation practices, and environmental influences. Therefore, it is highly recommended to include more batches in a future study. However, the expenditure has to be taken into account in conducting such experiments.
In addition, the controlled storage and image acquisition conditions, while ensuring consistency, do not fully reflect real-world scenarios. Variations in environmental factors such as temperature, humidity, drought, and excessive rainfall can significantly influence ripening behavior and physicochemical properties. Similarly, the use of standardized lighting and backgrounds during image capture may limit the model robustness when applied under more variable field conditions, including differences in lighting, orientation, and background complexity. Furthermore, the absence of external validation using independent datasets constrains confidence in the model’s predictive performance. Although the AI model demonstrated encouraging results, its reliability could be enhanced through validation with larger and more diverse datasets, as well as under varying imaging conditions. Future studies should therefore incorporate a broader range of mango cultivars, quality grades, environmental conditions, and acquisition settings to improve the robustness, generalizability, and practical applicability of AI-supported mango ripeness assessment.
The study demonstrated clear ripening trends in TEJC mangoes, characterized by increasing pH and TSS, decreasing firmness and L* and b* values, and increasing a* values, confirming progressive maturation. Strong correlations between color parameters and TSS highlighted their effectiveness as ripeness indicators. The CNN-based AI model achieved 67% training accuracy and 62% testing accuracy, showing potential applicability to predict ripening stages and quality attributes under practical conditions. Notably, this study presents the first AI-based ripeness prediction model for TEJC mangoes integrating physicochemical validation with computer vision in a Sri Lankan context. The proposed approach offers a non-destructive, consistent, and labor-efficient tool for postharvest quality management. While the proposed framework demonstrates feasibility, further validation across cultivars, storage conditions, and larger datasets are required before broader application can be considered. Cultivar specificity and environmental variability remain limitations; the findings provide a strong foundation for future improvements through model refinement, inclusion of environmental factors, and validation across cultivars and locations, supporting smarter and more sustainable mango postharvest systems.
Acknowledgement:
Funding Statement: The authors received no specific funding for this study.
Author Contributions: Savindi Thathsarani, Ashan Lakshitha, Pasindu Pramodya, Praveen Perera: writing—original draft, visualization, software, resources, methodology, investigation, formal analysis, data curation. Shagufta Henna: writing—review & editing. Rasanjali Samarakoon, Upaka Rathnayake: supervision, validation, project administration, conceptualization, writing—review & editing. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: Data and Material will be made available on reasonable request.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Shahzaib M , Wang S , Yao R , Ercisli S , Wang R , Fu X , et al. Sandalwood Essential Oil (SEO) readily inhibits Colletotrichum gloeosporioides-mediated anthracnose in post-harvest stored mango (Mangifera indica L. cv. ‘Keitt’). Phyton-Int J Exp Bot. 2025; 94( 7): 2167– 81. doi:10.32604/phyton.2025.065068. [Google Scholar] [CrossRef]
2. Le TD , Viet Nguyen T , Van Muoi N , Toan HT , Lan NM , Pham TN . Supply chain management of mango (Mangifera indica L.) fruit: A review with a focus on product quality during postharvest. Front Sustain Food Syst. 2022; 5: 799431. doi:10.3389/fsufs.2021.799431. [Google Scholar] [CrossRef]
3. Xu D , Wang H , Ji H , Zhang X , Wang Y , Zhang Z , et al. Hyperspectral imaging for evaluating impact damage to mango according to changes in quality attributes. Sensors. 2018; 18( 11): 3920. doi:10.3390/s18113920. [Google Scholar] [CrossRef]
4. Normand F , Lauri PE , Legave JM . Climate change and its probable effects on mango production and cultivation. Acta Hortic. 2015;( 1075): 21– 31. doi:10.17660/actahortic.2015.1075.1. [Google Scholar] [CrossRef]
5. Chay KG . A review on production and marketing of mango fruit. World J Agric Soil Sci. 2019; 2(2). doi:10.33552/wjass.2019.02.000533. [Google Scholar] [CrossRef]
6. Kiloes AM , Azizan FA , Checco J , Joyce D , Abdul Aziz A . What do consumers want in fresh mangoes? A systematic literature review. Int J Food Sci Technol. 2022; 57( 3): 1473– 92. doi:10.1111/ijfs.15503. [Google Scholar] [CrossRef]
7. Tarekegn K , Kelem F . Assessment of mango post-harvest losses along value chain in the gamo zone, southern Ethiopia. Int J Fruit Sci. 2022; 22( 1): 170– 82. doi:10.1080/15538362.2021.2025194. [Google Scholar] [CrossRef]
8. Roy S , Hassan MK , Ahmed QM , Rahman MM , Hasan GN , Sarkar MN . Assessment of postharvest loss and constraints in the supply chain of mango. Int J Bus Manag Soc Res. 2019; 7( 1): 412– 20. doi:10.18801/ijbmsr.070119.43. [Google Scholar] [CrossRef]
9. Kailaku SI , Arkeman Y , Purwanto YA , Udin F . Appropriate harvest age of mango (Mangifera indica cv. Arumanis) for quality assurance in long distance transportation planning in Indonesia. J Agric Food Res. 2023; 14: 100763. doi:10.1016/j.jafr.2023.100763. [Google Scholar] [CrossRef]
10. Gianguzzi G , Farina V , Inglese P , Rodrigo MGL . Effect of harvest date on mango (Mangifera indica L. cultivar osteen) fruit’s qualitative development, shelf life and consumer acceptance. Agronomy. 2021; 11( 4): 811. doi:10.3390/agronomy11040811. [Google Scholar] [CrossRef]
11. Moniruzzaman M , Uddin MS , Akhter MAE , Tripathi A , Rahaman KR . Application of geospatial techniques in evaluating spatial variability of commercially harvested mangoes in Bangladesh. Sustainability. 2022; 14( 20): 13495. doi:10.3390/su142013495. [Google Scholar] [CrossRef]
12. Reddy BR , Vahoniya DR , Bagda B , Damor AB . Post-harvest losses in mango supply chains in India: Causes and preventive measures. J Exp Agric Int. 2025; 47( 10): 293– 302. doi:10.9734/jeai/2025/v47i103813. [Google Scholar] [CrossRef]
13. Wanitchang P , Terdwongworakul A , Wanitchang J , Nakawajana N . Non-destructive maturity classification of mango based on physical, mechanical and optical properties. J Food Eng. 2011; 105( 3): 477– 84. doi:10.1016/j.jfoodeng.2011.03.006. [Google Scholar] [CrossRef]
14. Jha SN , Chopra S , Kingsly ARP . Modeling of color values for nondestructive evaluation of maturity of mango. J Food Eng. 2007; 78( 1): 22– 6. doi:10.1016/j.jfoodeng.2005.08.048. [Google Scholar] [CrossRef]
15. Becerra-Sanchez F , Taylor G . Reducing post-harvest losses and improving quality in sweet corn (Zea mays L.): Challenges and solutions for less food waste and improved food security. Food Energy Secur. 2021; 10( 3): e277. doi:10.1002/fes3.277. [Google Scholar] [CrossRef]
16. Mutungi C , Manda J , Feleke S , Abass A , Bekunda M , Hoschle-Zeledon I , et al. Adoption and impacts of improved post-harvest technologies on food security and welfare of maize-farming households in Tanzania: A comparative assessment. Food Secur. 2023; 15( 4): 1007– 23. doi:10.1007/s12571-023-01365-5. [Google Scholar] [CrossRef]
17. Ali Khan AH , Kiyani A , Santiago-Herrera M , Ibáñez J , Yousaf S , Iqbal M , et al. Sustainability of phytoremediation: Post-harvest stratagems and economic opportunities for the produced metals contaminated biomass. J Environ Manag. 2023; 326: 116700. doi:10.1016/j.jenvman.2022.116700. [Google Scholar] [CrossRef]
18. Wickrama SP , Kandangama NB , Wickramaarachchi T , Weerahewa J . Assessing the impact of non-tariff measures on Sri Lankan mango exports: Insights, challenges, and recommendations. Front Sustain Food Syst. 2024; 8: 1293263. doi:10.3389/fsufs.2024.1293263. [Google Scholar] [CrossRef]
19. Chandrapala AG , Pushpakumara SMV , Kadupitiya HK , Munasinghe MAK . Exploring and mapping of potential areas for fruit crops in Sri Lanka. Trop Agric. 2024; 171( 3): 20– 40. doi:10.4038/ta.v171i3.16. [Google Scholar] [CrossRef]
20. Ekanayake G , Abeywickrama K , Daranagama A , Kannangara S . Morphological characterization and molecular identification of stem-end rot associated fungal species isolated from ‘karutha colomban’ mango fruits in Sri Lanka. J Agric Sciences. 2019; 14( 2): 120– 8. doi:10.4038/jas.v14i2.8514. [Google Scholar] [CrossRef]
21. Rajapaksha L , Gunathilake DC , Pathirana SM , Fernando TN . Reducing post-harvest losses in fruits and vegetables for ensuring food security–Case of Sri Lanka. MOJ Food Process Technol. 2021; 9( 1): 7– 16. doi:10.15406/mojfpt.2021.09.00255. [Google Scholar] [CrossRef]
22. Sivakumar D , Jiang Y , Yahia EM . Maintaining mango (Mangifera indica L.) fruit quality during the export chain. Food Res Int. 2011; 44( 5): 1254– 63. doi:10.1016/j.foodres.2010.11.022. [Google Scholar] [CrossRef]
23. Prabhu A , Rani NS , Nandi CS . Towards importance of comprehensive color features analysis using iterative golden ratio proportions for Alphonso mango ripening stage classification by adapting to natural progressive ripening method. J Food Compos Anal. 2024; 126: 105873. doi:10.1016/j.jfca.2023.105873. [Google Scholar] [CrossRef]
24. Dutta J , Deshpande P , Rai B . AI-based soft-sensor for shelf life prediction of ‘Kesar’ mango. SN Appl Sci. 2021; 3( 6): 657. doi:10.1007/s42452-021-04657-7. [Google Scholar] [CrossRef]
25. Hua X , Lin Q , Gong F , Cai L , Liu J . Mango wine making process optimization based on artificial intelligence deep learning technology. Expert Syst. 2024; 41( 6): e13032. doi:10.1111/exsy.13032. [Google Scholar] [CrossRef]
26. Lakshitha AA , Liyanage MR , Samarakoon ERJ . Maturity detection of scotch bonnet pepper (Capsicum chinense) using image processing. J Res Technol Eng. 2024; 5( 4): 85– 92. [Google Scholar]
27. Rahman MM , Taohidul Islam SM , Biswas MA , Bepery C . Detection of artificially ripen mango using image processing. In: Proceedings of International Conference on Machine Intelligence and Data Science Applications. Singapore: Springer; 2021. p. 759– 69. doi:10.1007/978-981-33-4087-9_61. [Google Scholar] [CrossRef]
28. Sow S , Ranjan S , Seleiman MF , Alkharabsheh HM , Kumar M , Kumar N , et al. Artificial intelligence for maximizing agricultural input use efficiency: Exploring nutrient, water and weed management strategies. Phyton. 2024; 93( 7): 1569– 98. doi:10.32604/phyton.2024.052241. [Google Scholar] [CrossRef]
29. Li D , Bai L , Wang R , Ying S . Research progress of machine learning in extending and regulating the shelf life of fruits and vegetables. Foods. 2024; 13( 19): 3025. doi:10.3390/foods13193025. [Google Scholar] [CrossRef]
30. Li N , Zhao X , Yang Y , Zou X . Objects classification by learning-based visual saliency model and convolutional neural network. Comput Intell Neurosci. 2016; 2016: 7942501. doi:10.1155/2016/7942501. [Google Scholar] [CrossRef]
31. Zoumpourlis G , Doumanoglou A , Vretos N , Daras P . Non-linear convolution filters for CNN-based learning. In: Proceedings of the 2017 IEEE International Conference on Computer Vision (ICCV); 2017 Oct 22–29; Venice, Italy. p. 4771– 9. doi:10.1109/ICCV.2017.510. [Google Scholar] [CrossRef]
32. Ibarra-Garza IP , Ramos-Parra PA , Hernández-Brenes C , Jacobo-Velázquez DA . Effects of postharvest ripening on the nutraceutical and physicochemical properties of mango (Mangifera indica L. cv Keitt). Postharvest Biol Technol. 2015; 103: 45– 54. doi:10.1016/j.postharvbio.2015.02.014. [Google Scholar] [CrossRef]
33. Yin C , Huang C , Wang J , Liu Y , Lu P , Huang L . Effect of chitosan- and alginate-based coatings enriched with cinnamon essential oil microcapsules to improve the postharvest quality of mangoes. Materials. 2019; 12( 13): 2039. doi:10.3390/ma12132039. [Google Scholar] [CrossRef]
34. Wang J , Ren C , Wang J , Fu J , Yin Q , Huang Y , et al. Liquid chromatography quadrupole time-of-flight mass spectrometry-based metabolic characterization of mango ripened by different methods. Foods. 2024; 13( 22): 3548. doi:10.3390/foods13223548. [Google Scholar] [CrossRef]
35. Mounika T , Reddy NN , Lakshmi NJ , Joshi V . Studies on the effect of post harvest treatments on shelf life and quality of mango [Mangifera indica L.] cv. Amrapali. J Appl Nat Sci. 2017; 9( 4): 2055– 61. doi:10.31018/jans.v9i4.1488. [Google Scholar] [CrossRef]
36. Kumari P , Singh R , Kumar A , Sharma MM . Development of RTS from Papaya (Carica papaya L.) and Aloe vera (Aloe barbadensis Miller.) pulp. Int J Chem Stud. 2020; 8( 3): 646– 50. doi:10.22271/chemi.2020.v8.i3h.9279. [Google Scholar] [CrossRef]
37. Sivankalyani V , Maoz I , Feygenberg O , Maurer D , Alkan N . Chilling stress upregulates α-linolenic acid-oxidation pathway and induces volatiles of C(6) and C(9) aldehydes in mango fruit. J Agric Food Chem. 2017; 65( 3): 632– 8. doi:10.1021/acs.jafc.6b04355. [Google Scholar] [CrossRef]
38. Javed S , Fu H , Ali A , Nadeem A , Amin M , Razzaq K , et al. Comparative response of mango fruit towards pre- and post-storage quarantine heat treatments. Agronomy. 2022; 12( 6): 1476. doi:10.3390/agronomy12061476. [Google Scholar] [CrossRef]
39. Zhu X , Wang Q , Cao J , Jiang W . Effects of chitosan coating on postharvest quality of mango (Mangifera indica L. cv. Tainong) fruits. J Food Process Preserv. 2008; 32( 5): 770– 84. doi:10.1111/j.1745-4549.2008.00213.x. [Google Scholar] [CrossRef]
40. Shinde SP , Suraj P , Biswas A , Hon GR , Matche RS , Chaudhari SR . Physiochemical dynamics and biochemical transformations during ripening of Indian Mangifera indica L. varieties: Insights from multi-parameter analysis and NMR spectroscopy. J Food Compos Anal. 2025; 142: 107526. doi:10.1016/j.jfca.2025.107526. [Google Scholar] [CrossRef]
41. Thanaraj T , Terry LA . Temporal change in taste- and health-related compounds during postharvest ripening of Sri Lankan mango fruit (Mangifera indica L.). Acta Hortic. 2010;( 877): 1183– 9. doi:10.17660/actahortic.2010.877.161. [Google Scholar] [CrossRef]
42. Jaya S , Das H . Accelerated storage, shelf life and color of mango powder. J Food Process Preserv. 2005; 29( 1): 45– 62. doi:10.1111/j.1745-4549.2005.00012.x. [Google Scholar] [CrossRef]
43. Theanjumpol P , Self G , Rittiron R , Pankasemsuk T , Sardsud V . Quality control of mango fruit during postharvest by near infrared spectroscopy. Chiang Mai Univ J Nat Sci. 2014; 13( 2). doi:10.12982/cmujns.2014.0028. [Google Scholar] [CrossRef]
44. Ngo TMP , Nguyen TH , Dang TMQ , Van Thanh D , Reungsang A , Chaiwong N , et al. Effect of pectin/nanochitosan-based coatings and storage temperature on shelf-life extension of “elephant” mango (Mangifera indica L.) fruit. Polymers. 2021; 13( 19): 3430. doi:10.3390/polym13193430. [Google Scholar] [CrossRef]
45. Dieye M , Ndiaye ND , Bassama J , Mertz C , Bugaud C , Diatta P , et al. Storage time as an index for varietal prediction of mango ripening: A systemic approach validated on five Senegalese varieties. Foods. 2022; 11( 23): 3759. doi:10.3390/foods11233759. [Google Scholar] [CrossRef]
46. Gill PPS , Jawandha SK , Kaur N , Singh N . Physico-chemical changes during progressive ripening of mango (Mangifera indica L.) cv. Dashehari under different temperature regimes. J Food Sci Technol. 2017; 54( 7): 1964– 70. doi:10.1007/s13197-017-2632-6. [Google Scholar] [CrossRef]
47. Zhang B , Huang C , Zhang L , Wang J , Huang X , Zhao Y , et al. Application of chlorine dioxide microcapsule sustained-release antibacterial films for preservation of mangos. J Food Sci Technol. 2019; 56( 3): 1095– 103. doi:10.1007/s13197-019-03636-6. [Google Scholar] [CrossRef]
48. Xin M , Li C , Khoo HE , Li L , He X , Yi P , et al. Dynamic analyses of transcriptome and metabolic profiling: Revealing molecular insight of aroma synthesis of mango (Mangifera indica L. var. Tainong). Front Plant Sci. 2021; 12: 666805. doi:10.3389/fpls.2021.666805. [Google Scholar] [CrossRef]
49. Eyarkai Nambi V , Thangavel K , Shahir S , Thirupathi V . Comparison of various RGB image features for nondestructive prediction of ripening quality of “alphonso” mangoes for easy adoptability in machine vision applications: A multivariate approach. J Food Qual. 2016; 39( 6): 816– 25. doi:10.1111/jfq.12245. [Google Scholar] [CrossRef]
50. Hailu Z . Effects of controlled atmosphere storage and temperature on quality attributes of mango. J Chem Eng Process Technol. 2016; 7( 5): 1000317. doi:10.4172/2157-7048.1000317. [Google Scholar] [CrossRef]
51. Meighani H , Ghasemnezhad M , Bakhshi D . Effect of different coatings on post-harvest quality and bioactive compounds of pomegranate (Punica granatum L.) fruits. J Food Sci Technol. 2015; 52( 7): 4507– 14. doi:10.1007/s13197-014-1484-6. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools