 Open Access
Open Access
CASE REPORT
Feasibility of water vapor thermal therapy for treating lower urinary tract symptoms in men with localized prostate cancer on active surveillance: a case series
1 Hackensack Meridian Health, Palisades Medical Center, North Bergen, NJ 07047, USA
2 St. George’s University School of Medicine, University Centre, Grenada, P.O. Box 7, West Indies
3 Albert Einstein College of Medicine, Bronx, NY 10461, USA
4 Department of Urology, Montefiore Medical Center, Bronx, NY 10467, USA
5 DSS Urology, Queens Village, NY 11427, USA
* Corresponding Author: Mustufa Babar. Email:
Canadian Journal of Urology 2026, 33(1), 193-199. https://doi.org/10.32604/cju.2025.066654
Received 14 April 2025; Accepted 22 August 2025; Issue published 28 February 2026
Abstract
Background: Prostate cancer is a common malignancy, with many men on active surveillance for localized, low-risk disease also experiencing lower urinary tract symptoms (LUTS) from benign prostatic hyperplasia (BPH). Water Vapor Thermal Therapy (WVTT) is a minimally invasive BPH treatment, but its safety and efficacy in this setting are unclear. Case Description: We report three men with localized PCa on active surveillance who underwent WVTT for LUTS. Conclusions: WVTT appears safe and potentially effective in treating LUTS, especially in those with lower-risk disease and smaller prostate volumes. Further research is needed to confirm safety, efficacy, and optimal patient selection.Keywords
Prostate cancer (PCa) is the second most common cancer and the fifth leading cause of cancer-related death among men worldwide.1 As screening for PCa becomes more widespread and detection methods improve, a considerable number of patients are diagnosed with localized PCa. This trend underscores the increasing importance of active surveillance, particularly for those with low-risk PCa.2,3
Active surveillance aims to maintain the patient’s quality of life (QoL) by deferring or delaying definitive treatment, such as surgery or radiation therapy, until there is evidence of disease progression.2 The American Urological Association and the American Society for Radiation Oncology guidelines outline the principles of active surveillance, which include regular monitoring with prostate-specific antigen (PSA) testing, digital rectal examinations, and periodic prostate biopsies.4 However, patients undergoing active surveillance for PCa may experience moderate and severe lower urinary tract symptoms (LUTS) attributed to an accompanying large benign prostate adenoma affecting the transition zone of the prostate, also known as benign prostatic hyperplasia (BPH).5
Water Vapor Thermal Therapy (WVTT; The Rezum System, Boston Scientific Corporation, Marlborough, MA, USA) is a minimally invasive surgical therapy (MIST) that utilizes convective thermal energy to ablate obstructive prostatic tissue. WVTT has been well studied for the improvement of LUTS in the setting of BPH.6 However, the role of WVTT in the treatment of LUTS from BPH in the setting of PCa has never been investigated. We report outcomes of WVTT for the treatment of LUTS secondary to BPH in a case series of three patients with concurrent localized PCa on active surveillance. Given the small sample size, our aim is to present hypothesis-generating findings that may inform future prospective studies.
This study was approved by the Western Institutional Review Board under protocol #11181739-1. This study was prepared according to the CARE case report guideline, and a CARE checklist was provided.7 Please see Supplementary Material S1 for more details.
Three patients with BPH and localized PCa on active surveillance underwent WVTT at DSS Urology between January and May 2018. Pressure flow urodynamic studies were performed in all patients before WVTT to exclude other causes of LUTS (e.g., detrusor overactivity, neurogenic bladder). Baseline patient characteristics are presented in Table 1, WVTT procedural characteristics are summarized in Table 2, and WVTT outcome measures are summarized in Table 3. Percent change in International Prostate Symptom Score (IPSS) over time is illustrated in Figure 1. Percent changes were calculated using baseline and follow-up values.
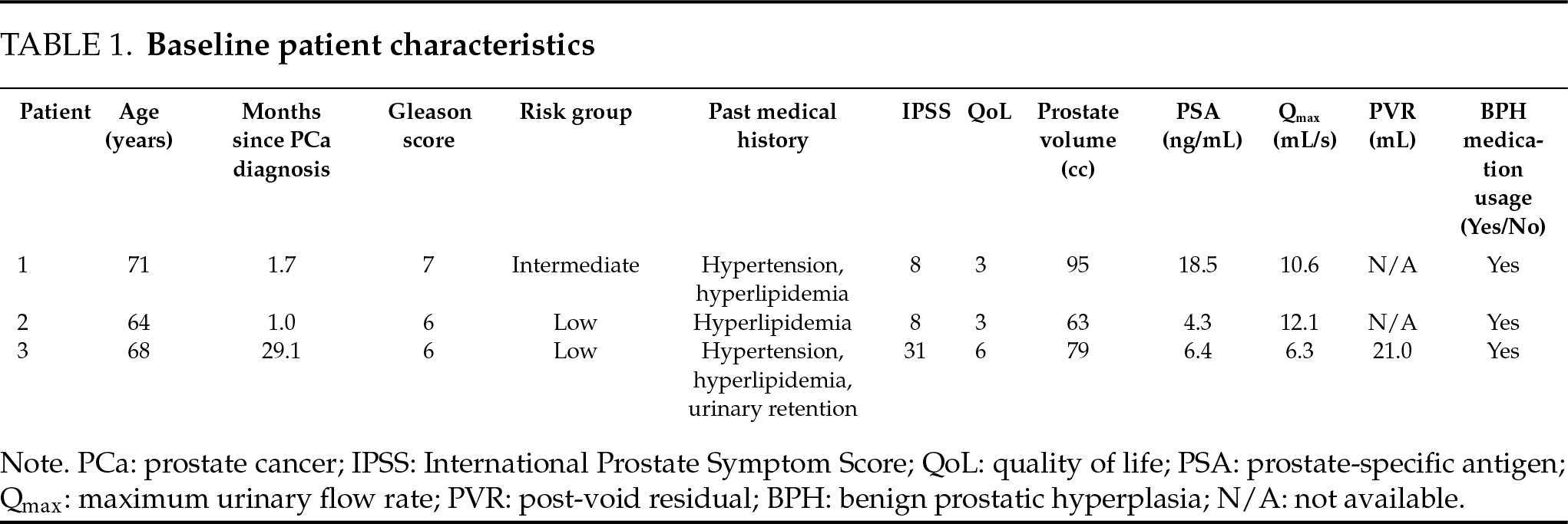

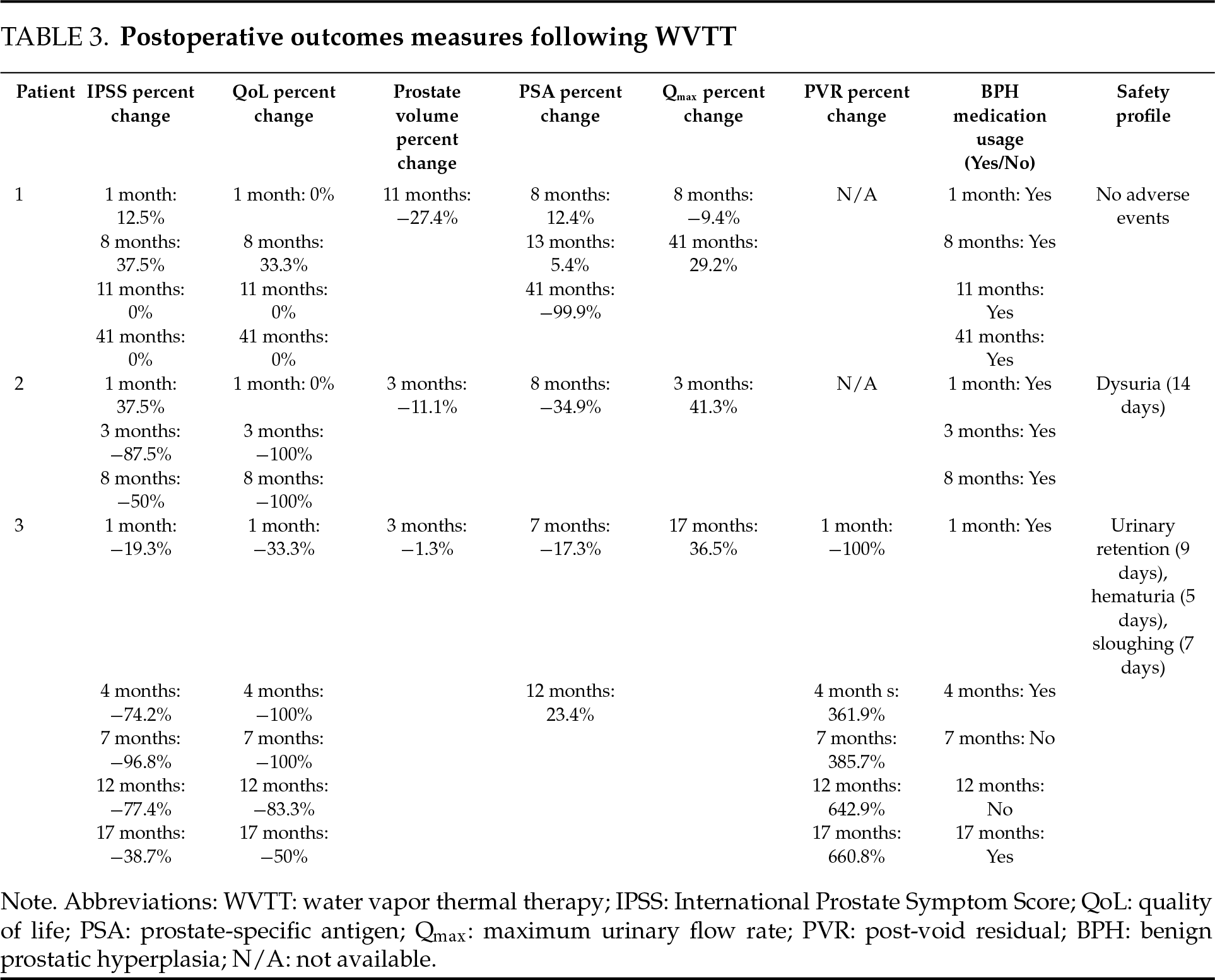
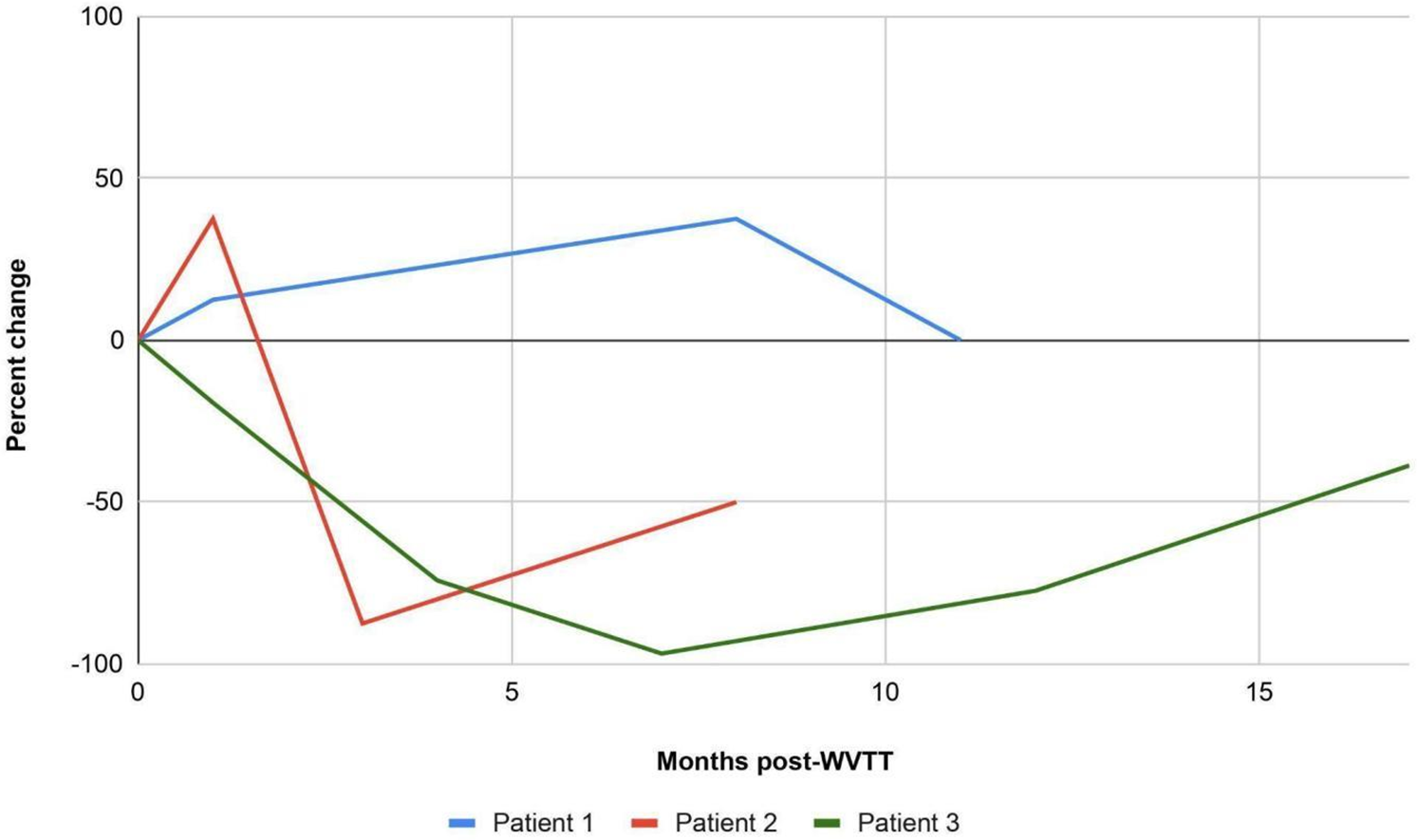
FIGURE 1. Percent change in IPSS over time following WVTT. Abbreviations: WVTT: water vapor thermal therapy
Patient 1 is a 71-year-old male with a past medical history of Gleason 7 PCa (intermediate risk) diagnosed in April 2018 and on active surveillance, BPH, hypertension, and hyperlipidemia who presented to the clinic five weeks after PCa diagnosis with bothersome LUTS that showed minimal improvement with tamsulosin 0.4 mg. His IPSS indicated moderate LUTS (8), with a QoL score of 3. Prostate volume was measured at 95 cc, PSA at 18.5 ng/mL, and maximum urinary flow rate (Qmax) at 10.6 mL/s. UroCuff test indicated borderline obstruction.
The patient underwent WVTT with general anesthesia in May 2018 without any intra- or postoperative complications. Six needles were injected into the lateral prostatic lobes and one into the medial prostatic lobe. A urinary catheter was placed postoperatively and removed after 7 days when the patient passed his trial of void (TOV).
In June 2018 (1 month postoperative), IPSS increased to 9 (point change: 1, percent change: 12.5%) and QoL remained at 3 (point change: 0, percent change: 0%). Computed Tomography (CT) scan of the abdomen and pelvis and whole-body bone scan were negative for metastasis. In January 2019 (8 months postoperative), IPSS increased to 11 (point change: 3, percent change: 37.5%), QoL increased to 4 (point change: 1, percent change: 33.3%), and PSA increased to 20.8 ng/mL (percent change: 12.4%). Qmax remained slow at 9.6 mL/s (percent change: −9.4%) with minimal post-void residual (PVR) on bladder scan. In February 2019 (9 months postoperative), MRI of the prostate revealed a Prostate Imaging-Reporting and Data System (PI-RAD) 5 lesion in the inferior/anterior transition zone, a PI-RAD 4 lesion in the mid gland peripheral zone, and possible seminal vesicle involvement. In April 2019 (11 months postoperative), transrectal ultrasound (TRUS) guided prostate and seminal vesicle biopsy revealed Gleason 7 PCa and benign seminal vesicles, with a reduction in prostate volume to 69 cc (percent change: −27.4%). IPSS remained at 8 (point change: 0, percent change: 0%) and QoL at 3 (point change: 0, percent change: 0%).
In June 2019 (13 months postoperative), PSA remained elevated at 19.5 ng/mL (percent change: 5.4%) and the patient decided to start radiation therapy for two months and hormone therapy with Trelstar 11.25 mg for two years. Over the next two years, PSA steadily decreased from 0.37 ng/mL in September 2019 (16 months postoperative) to <0.01 ng/mL in October 2021 (41 months postoperative), while Qmax remained slow at 13.7 mL/s (percent change: 29.2%) with minimal PVR, and IPSS and QoL remained stable at 8 and 3, respectively (point and percent changes: 0 and 0%, respectively, for both IPSS and QoL). The patient continued tamsulosin 0.4 mg throughout the duration of the follow-ups. In October 2021, the patient moved back to his country and was advised to follow-up with a urologist there.
Patient 2 is a 64-year-old male with a past medical history of Gleason 6 PCa (low risk) diagnosed in January 2018 and on active surveillance, BPH, and hyperlipidemia who presented to the clinic four weeks after PCa diagnosis with bothersome LUTS that showed minimal improvement with tamsulosin 0.4 mg. His IPSS indicated moderate LUTS (8), with a QoL score of 3. Prostate volume was measured at 63 cc, PSA at 4.3 ng/mL, and maximum urinary flow rate (Qmax) at 12.1 mL/s. UroCuff test indicated obstruction.
The patient underwent WVTT with local prostate block anesthesia in February 2018 without any intraoperative complications. Four needles were injected into the lateral prostatic lobes and one into the medial prostatic lobe. A urinary catheter was placed postoperatively and removed after 5 days when the patient passed his trial of void (TOV). The patient experienced dysuria for two weeks following catheter removal.
In March 2018 (1 month postoperative), IPSS increased to 11 (point change: 3, percent change: 37.5%) and QoL remained at 3 (point change: 0, percent change: 0%). In May 2018 (3 months postoperative), IPSS decreased to 1 (point change: −7, percent change: −87.5%), QoL decreased to 0 (point change: −3, percent change: −100%), and Qmax increased to 17.1 mL/s (percent change: 41.3%) with minimal PVR on bladder scan. TRUS of the prostate revealed a 11.1% decrease in prostate volume to 56 cc. In October 2018 (8 months postoperative), IPSS slightly increased to 4 (point change: −4, percent change: −50%) since the last visit but QoL remained at 0 (point change: −3, percent change: −100%). PSA decreased to 2.8 ng/mL (percent change: −34.9%). Patient was scheduled for an interval prostate biopsy but lost to follow-up. The patient continued tamsulosin 0.4 mg throughout the duration of the follow-ups.
Patient 3 is a 68-year-old male with a past medical history of Gleason 6 PCa (low risk) diagnosed in April 2016 and on active surveillance, BPH with a prior transurethral microwave thermotherapy in 2007 and GreenLight laser treatment in 2009, atrial fibrillation, hypertension, urinary retention, and hyperlipidemia who presented to the clinic in May 2018 with bothersome LUTS that showed minimal improvement with tamsulosin 0.4 mg and had been in urinary retention for one month. His IPSS indicated severe LUTS (31), with a QoL score of 6. Prostate volume was measured at 79 cc, PSA at 6.4 ng/mL, and maximum urinary flow rate (Qmax) at 6.3 mL/s. UroCuff test indicated obstruction.
The patient underwent WVTT with general anesthesia in May 2018 without any intraoperative complications. Four needles were injected into the lateral prostatic lobes and one into the medial prostatic lobe. A urinary catheter was placed postoperatively and removed after 7 days when the patient passed his TOV. However, the next day after catheter removal the patient came in with urinary retention. A urinary catheter was placed again which drained 800 cc of clear urine. The catheter was removed 9 days later when the patient passed his TOV again. The patient experienced hematuria for 5 days and sloughing for one week following catheter removal.
In June 2018 (1 month postoperative), IPSS decreased to 25 (point change: −6, percent change: −19.3%), QoL decreased to 4 (point change: −2, percent change: −33.3%), and there was minimal PVR on bladder scan (percent change: −100%). In September 2018 (4 months postoperative), IPSS decreased to 8 (point change: −23, percent change: −74.2%), QoL decreased to 0 (point change: −6, percent change: −100%), and PVR revealed 97 cc on bladder scan (percent change: 361.9%). TRUS of the prostate revealed a negligible 1.3% decrease in prostate volume to 78 cc. In January 2019 (7 months postoperative), IPSS was 1 (point change: −30, percent change: −96.8%), QoL was 0 (point change: −6, percent change: −100%), PVR revealed 102 cc on bladder scan (percent change: 385.7%), PSA was 5.3 ng/mL (percent change: −17.3%), and the patient was able to discontinue tamsulosin. In May 2019 (12 months postoperative), IPSS was 7 (point change: −24, percent change: −77.4%), QoL was 1 (point change: −5, percent change: −83.3%), PVR was 156 cc (percent change: 642.9%), PSA was 7.9 ng/mL (percent change: 23.4%), and the patient remained off BPH medications.
In October 2019 (17 months postoperative), the patient started to experience bothersome LUTS again (IPSS 19 [point change: −12, percent change: −38.7%], QoL 3 [point change: −3, percent change: −50%]) and was restarted on tamsulosin 0.4 mg. Qmax was 8.6 mL/s (percent change: 36.5%) and PVR revealed 159 cc (percent change: 660.8%) on bladder scan.
In December 2019 (19 months postoperative), the patient underwent another WVTT procedure under general anesthesia without any intraoperative complications. Six needles were injected into the lateral prostatic lobes and one into the medial prostatic lobe. The patient moved across the country a few days after the procedure and followed up at a different institution.
We report the first cases of off-label WVTT use to manage LUTS secondary to BPH in men with concurrent localized PCa on active surveillance. In our unique case series of three patients with moderate to severe LUTS, we found that WVTT effectively treated LUTS in two out of the three patients (Patient 2 and Patient 3). Patient 1 experienced clinical worsening of LUTS (≥3 point change in IPSS) up to 8 months before returning to baseline at 11 months prior to undergoing radiation and hormone therapy. Patient 2 experienced clinical worsening of LUTS at 1 month, followed by improvement at 3 months, that remained durable to the last follow-up at 8 months. Finally, Patient 3 experienced gradual improvements in LUTS until 7 months, that remained durable to the last follow-up at 17 months, prior to electing to undergo a repeat WVTT. Additionally, urinary retention in Patient 3 was able to be relieved as a result of the first WVTT. All three patients continued their BPH medications at their last follow-up. Nevertheless, the safety profile in these patients aligned with prior literature,6 with most adverse events related to the endoscopic nature of the procedure.
Patient 1 did not experience improvements in LUTS following WVTT, unlike Patients 2 and 3. This may be attributed to a combination of several factors, including Patient 1 having a higher Gleason score and risk group (Patient 1: Gleason 7, intermediate risk vs. Patient 2: Gleason 6, low risk; Patient 3: Gleason 6, low risk), receiving a greater number of injections (Patient 1: 7 vs. Patient 2: 5, Patient 3: 5), having a higher baseline PSA (Patient 1: 18.5 ng/mL vs. Patient 2: 4.3 ng/mL, Patient 3: 6.4 ng/mL), and having a larger baseline prostate volume (Patient 1: 95 cc vs. Patient 2: 63 cc, Patient 3: 79 cc). While WVTT targets BPH in the transition zone, it is possible that higher-risk or higher-grade cancers may be associated with more periurethral inflammation or tumor burden encroaching on the transition zone, which could affect baseline obstruction or limit the efficacy of tissue ablation with WVTT. Based on these characteristics, it may be plausible that PCa patients with low-risk PCa and prostate volumes under 80 cc may derive the most benefit from WVTT. However, given the limited literature on the use of WVTT to treat LUTS in men with localized PCa on active surveillance, further research is needed to identify the patient cohorts that may benefit the most from this MIST. Additionally, the role of WVTT on oncological outcomes needs to be further investigated.
Besides WVTT, other MISTs, including prostate artery embolization (PAE), holmium laser enucleation of the prostate (HoLEP), and photoselective vaporization of the prostate (GreenLight) have also been investigated in treating LUTS secondary to BPH in men with concurrent localized PCa on active surveillance. In a retrospective study of 21 localized PCa patients on active surveillance, Parikh et al.8 found that PAE was safe and effective for the treatment of men with LUTS, with IPSS significantly improving by a median of 12 and 14 points at 6 and 12 weeks, respectively. Furthermore, in a prospective study of 117 localized PCa patients on active surveillance, Elsaqa et al.9 found HoLEP to not only improve LUTS, with IPSS significantly improving by a median of 19 points by 1 year, but also reduced the contribution of large volume adenoma to PSA level in PCa patients, thereby better reflecting PSA levels and reducing overtreatment in those who pursued treatment after active surveillance. Finally, in a retrospective of 71 localized PCa patients on active surveillance, Jibara et al.10 found GreenLight to provide durable relief in LUTS, with IPSS significantly improving by a mean of 12 points by 1 year, without adverse effects on disease progression rates. Our case series adds to the limited literature of exploring the use of MISTs to manage LUTS in PCa patients on active surveillance. Of note, while definitive oncologic treatments such as surgery or focal therapies may also improve LUTS, they are typically pursued with curative intent rather than as symptom-directed interventions during active surveillance, which is the intended role of MISTs in this context.
This case series is not without limitations. First, our findings are preliminary and intended to be hypothesis-generating. We evaluated WVTT in only three patients who had limited follow-up. Second, the retrospective design poses constraints in data collection and analysis. Lastly, we did not evaluate sexual function since none of the patients in our case series were sexually active at baseline. Despite these limitations, our case series offers insights into exploring the off-label WVTT use to manage LUTS secondary to BPH in men with concurrent localized PCa on active surveillance, and highlights the importance of further research in identifying specific PCa patient cohorts that may benefit from WVTT.
In our case series of three patients, WVTT safely and effectively treated LUTS secondary to BPH in two men with low-risk localized PCa on active surveillance with prostate volumes <80 cc. Notably, WVTT also relieved urinary retention at baseline in one of these patients. WVTT was not effective in managing LUTS in one patient with intermediate-risk localized PCa on active surveillance with a prostate volume >80 cc. Given the novelty of this application, our findings should be interpreted as exploratory and not definitive, and additional studies with larger sample sizes are warranted to explore WVTT outcomes in men with PCa, particularly to clarify whether lower-risk disease and smaller prostate volumes are associated with greater symptomatic benefit.
Acknowledgement
We thank Dr. David S. Schnapp for helping to create the database and the staff at DSS Urology for providing assistance throughout the period of data collection.
Funding Statement
The authors received no specific funding for this study.
Author Contributions
Study conception and design: Mustufa Babar, Farooz Babar, Noah Hawks-Ladds, Justin Loloi, Michael Ciatto; data collection: Mustufa Babar; analysis and interpretation of results: Mustufa Babar, Farooz Babar, Noah Hawks-Ladds; draft manuscript preparation: Mustufa Babar, Farooz Babar, Noah Hawks-Ladds, Justin Loloi, Michael Ciatto. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to their containing information that could compromise the privacy of research participants.
Ethics Approval
Institutional Review Board approval was obtained from the Western Institutional Review Board, under reference number #1-1181739-1. As per the IRB determination, no patient informed consent was required for this case report, as all patient information has been fully de-identified in accordance with applicable ethical and regulatory standards.
Conflicts of Interest
The authors declare no conflicts of interest to report regarding the present study.
References
1. Wang L, Lu B, He M et al. Prostate cancer incidence and mortality: global status and temporal trends in 89 countries from 2000 to 2019. Front Public Health 2022;10:811044. [Google Scholar] [PubMed]
2. Kang SK, Mali RD, Prabhu V, Ferket BS, Loeb S. Active surveillance strategies for low-grade prostate cancer: comparative benefits and cost-effectiveness. Radiology 2021;300(3):594–604. [Google Scholar] [PubMed]
3. Cooperberg MR, Meeks W, Fang R, Gaylis FD, Catalona WJ, Makarov DV. Time trends and variation in the use of active surveillance for management of low-risk prostate cancer in the US. JAMA Netw Open 2023;6(3):e231439. [Google Scholar] [PubMed]
4. Eastham JA, Auffenberg GB, Barocas DA et al. Clinically localized prostate cancer: AUA/ASTRO guideline, part II: principles of active surveillance, principles of surgery, and follow-up. J Urol 2022;208(1):19–25. [Google Scholar] [PubMed]
5. Schenk JM, Kristal AR, Arnold KB et al. Association of symptomatic benign prostatic hyperplasia and prostate cancer: results from the prostate cancer prevention trial. Am J Epidemiol 2011;173(12):1419–1428. [Google Scholar] [PubMed]
6. Babar M, Loloi J, Tang K, Syed U, Ciatto M. Emerging outcomes of water vapor thermal therapy (Rezum) in a broad range of patients with lower urinary tract symptoms secondary to benign prostatic hyperplasia: a systematic review. Low Urin Tract Symptoms 2022;14(3):140–154. [Google Scholar] [PubMed]
7. Riley DS, Barber MS, Kienle GS et al. CARE guidelines for case reports: explanation and elaboration document. J Clin Epi 2017;89:218–235. [Google Scholar]
8. Parikh N, Keshishian E, Manley B et al. Effectiveness and safety of prostatic artery embolization for the treatment of lower urinary tract symptoms from benign prostatic hyperplasia in men with concurrent localized prostate cancer. J Vasc Interv Radiol 2021;32(7):1053–1061. [Google Scholar] [PubMed]
9. Elsaqa M, Slade A, Lingeman J et al. Holmium laser enucleation of prostate in patients with pre-existing localized prostate cancer, dual center study. J Endourol 2023;37(3):330–334. [Google Scholar] [PubMed]
10. Jibara G, Sjoberg DD, Stearns GL et al. Photoselective vaporization of the prostate in the management of lower urinary tract symptoms in prostate cancer patients on active surveillance. Urology 2021;156:225–230. [Google Scholar] [PubMed]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools