 Open Access
Open Access
HOW I DO IT
Suprapubic transvesical adenoma resection of the prostate (STAR-P): step-by-step technique
1 City Clinic, Urology, Bozen, 39100, Italy
2 Urology Department, Università La Sapienza, Rome, 00185, Italy
3 Urology Department, Klinikum Klagenfurt, 9020, Austria
4 Urology Department, IRCCS “Regina Elena” National Cancer Institute, Rome, 00128, Italy
5 Belgrade Center for Genitourinary Reconstructive Surgery, Serbia, 11000, Belgrade
6 School of Medicine, University of Belgrade, Belgrade, 11000, Serbia
* Corresponding Author: Bruno Bucca. Email:
Canadian Journal of Urology 2026, 33(1), 75-82. https://doi.org/10.32604/cju.2025.068035
Received 20 May 2025; Accepted 29 October 2025; Issue published 28 February 2026
Abstract
Overview: Surgical management of benign prostatic hyperplasia (BPH) has evolved significantly, incorporating various minimally invasive procedures aimed at reducing morbidity and optimizing patient outcomes. Despite advancements, transurethral approaches continue to pose risks such as urethral strictures and urinary incontinence due to mechanical and thermal stress. To address these limitations, the Suprapubic Transvesical Adenoma Resection of the Prostate (STAR-P) was developed, offering a direct suprapubic route that bypasses the urethra entirely. Recent studies have validated STAR-P as both feasible and safe, emphasizing advantages such as enhanced visualization of the urinary sphincter, minimized urethral trauma, effective hemostasis, and reduced operative stress. The procedure utilizes specially designed instrumentation, including a large-caliber bipolar resectoscope (42 Fr), allowing the efficient removal of substantial adenoma tissue in fewer resection passes compared to traditional methods. Objectives: This article provides a comprehensive, step-by-step description of the STAR-P technique. The primary objective is to detail patient selection criteria, preoperative assessments, procedural steps including mini-open suprapubic access, specialized instrumentation usage, resection techniques, and postoperative management protocols. Highlighting technical considerations and procedural innovations aims to inform urologists about the potential benefits of STAR-P, particularly in patients at higher risk for urethral complications or those with large prostate volumes. By documenting the procedural intricacies and outcomes clearly and thoroughly, we seek to encourage informed adoption of STAR-P as an alternative, effective surgical approach for managing benign prostatic hyperplasia, thus contributing to the evolving landscape of minimally invasive urological surgery.Keywords
Supplementary Material
Supplementary Material FileSurgery for benign prostatic hyperplasia (BPH) is widely performed and includes a range of techniques adapted to the severity of symptoms, prostate size, and individual patient characteristics. Advancements in surgical technology have led to the widespread adoption of minimally invasive procedures. In particular, laser enucleation of the prostate (e.g., holmium laser enucleation of the prostate [HoLEP] and thulium laser enucleation of the prostate [ThuLEP]) and robotic-assisted simple prostatectomy (RASP) have gained prominence due to their enhanced safety profiles, reduced blood loss, shorter hospital stays, and overall lower invasiveness. These modern techniques provide effective symptom relief while minimizing perioperative complications, making them increasingly preferred options for both patients and surgeons.1 A recent study investigated the interhospital variability in the surgical treatment of patients with benign prostatic hyperplasia and confirmed an increased use of invasive techniques.2,3 Despite significant improvements in technique, the risk of postoperative complications, like incontinence and urethral stricture, has not decreased.4 The termical and mechanical stress exerted on the urethra and external urinary sphincter during transurethral procedures can result in transient or persistent urethral trauma and urinary incontinence.
In literature, postoperative stricture rates vary widely, ranging from 1% to 12%.4 In 2025, a nomogram was published to predict the risk of post-TURP urethral stricture. This tool allows for the early identification of high-risk patients, enabling targeted preventive interventions. Multivariate analyses identified age, prostate size, preoperative indwelling catheter use, preoperative urethral dilation, and postoperative indwelling catheter duration as independent risk factors for urethral stricture.5
To address this issue, the Suprapubic Transvesical Adenoma Resection of the Prostate (STAR-P) has been ideated to bypass the urethral access. The first STAR-P attempts were described in 2004 in a few patients with BPH and conditions that contraindicated a transurethral approach.6 A recent retrospective analysis of 44 consecutive patients who underwent STAR-P has been published, confirming the surgery as feasible and safe.7 The suprapubic access offers several significant advantages such as the avoidance of the mechanical stress to the whole urethra, the constant visualization of the external urinary sphincter during resection, the freedom of movement and, not the least, the top-down view that allows accurate hemostasis. Moreover, the transurethral access poses a dimensional limit of urethral lumen and instrumentation. Recently, we manufactured a dedicated equipment with a 42 Fr resectoscope and a set of custom-designed loops of various sizes, with the largest capable of resecting up to 7 g of adenoma in a single step.8
In this paper, we present an exhaustive step-by-step description of the STAR-P procedure.
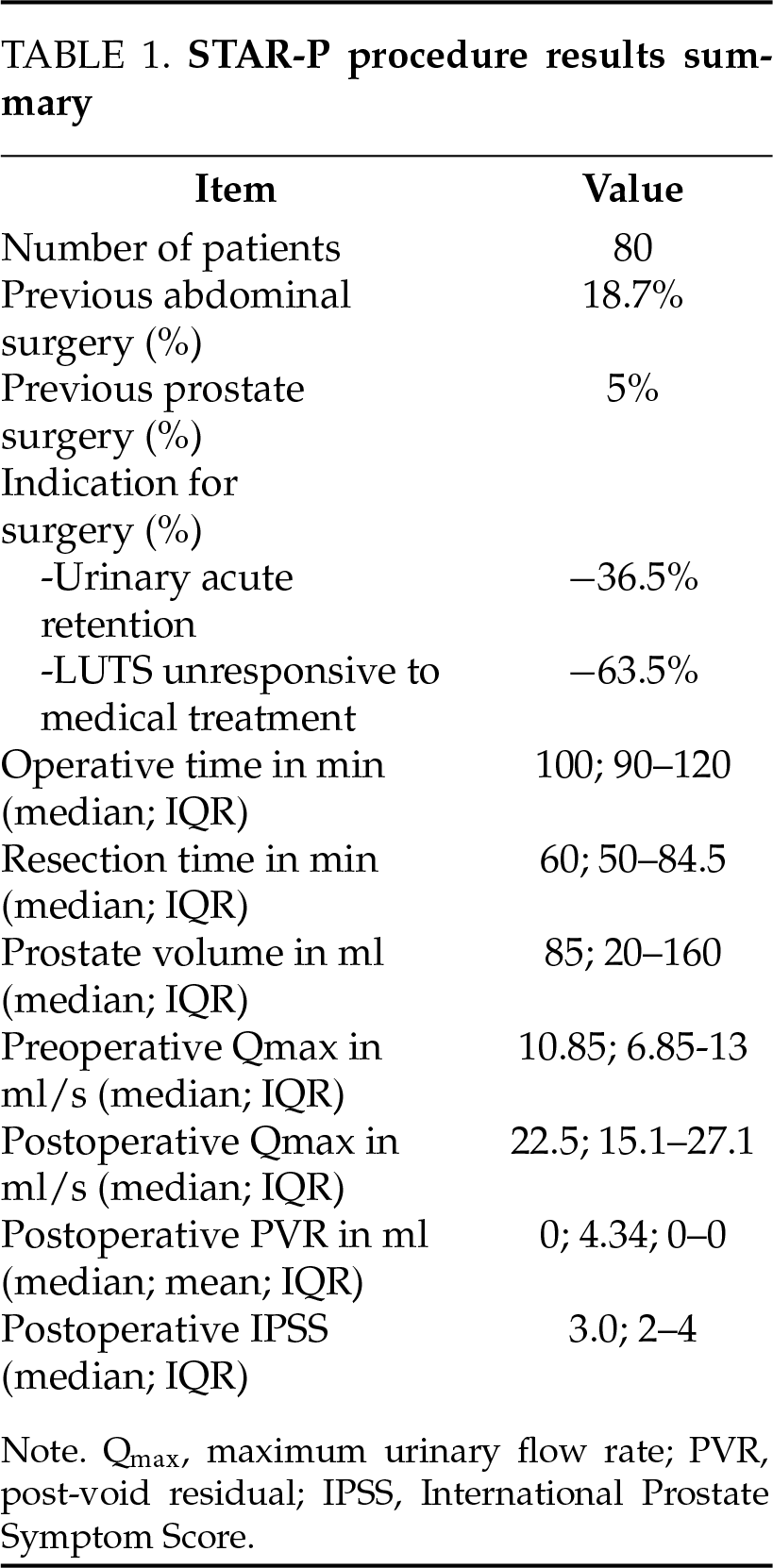
Between 2020 and 2024, in City Clinic Medical Center, a single surgeon consecutively performed the STAR-P procedure on 80 patients diagnosed with benign prostatic obstruction (BPO). Surgical indications included moderate-to-severe lower urinary tract symptoms (LUTS), refractory to medical management, assessed by the International Prostate Symptom Score (IPSS) questionnaire. Patient evaluation involved a thorough medical history and physical examination, preoperative PSA measurement, ultrasound imaging, uroflowmetry, and assessment of post-void residual (PVR) volume. prostate specific antigen (PSA) measurement served primarily to estimate the risk of incidental prostate cancer and secondarily as a postoperative marker of successful adenoma resection. Suprapubic ultrasound evaluated prostate volume, morphology, presence of a median lobe, bladder volume prior to voiding, and identified intestinal loops potentially interposed in the retropubic region, relevant for surgical planning. All patients also underwent urinalysis and urine culture; in cases of bacterial growth, targeted antibiotic therapy was administered until a sterile culture was documented. A flexible urethrocystoscopy was recommended preoperatively to assess urethral lumen integrity, identify significant or subclinical strictures, visualize prostatic urethra anatomy, external urinary sphincter status, localize ureteral orifices, exclude bladder tumors, and confirm bladder capacity. Exclusion criteria included previous pelvic surgery, urothelial cancer history, prior kidney transplantation, inability to temporarily suspend anticoagulant medications, American Society of Anesthesiologists (ASA) score above 3, and significant central adiposity posing risks during transvesical access. All study patients had a body mass index below 27 kg/m². Neither prostate size nor the presence of an indwelling catheter represented exclusion criteria. Patients provided written informed consent after receiving comprehensive information regarding the innovative nature of STAR-P as an alternative approach to conventional bipolar transurethral resection (B-TURP). Ethical approval was obtained from the CityClinic Bolzano ethical board (sign date: 01 September 2020), in accordance with the Declaration of Helsinki. Follow-up at 3, 6 and 12 months included repeated uroflowmetry, IPSS questionnaire administration, and PVR measurement.
Either general or spinal subarachnoid anesthesia can be administered, depending on the patient’s medical history or preference. A single-shot antibiotic is administered. The patient is positioned supine with the feet kept parallel. In obese patients, a slight Trendelenburg position (maximum 20 degrees) is beneficial. A dedicated drape, such as a cesarean section drape with lateral pockets for collecting fluids, is applied. The penis is included in the surgical field to facilitate catheter manipulation. A 16 Fr three-way Foley catheter is gently placed, and the bladder is filled retrogradely. The surgeon stands to the left of the patient if right-handed.
A dedicated equipment was designed by Prof. Christian Gozzi and manufactured by Tontarra Medizintechnik GmbH (Wurmlingen, Germany). The resectoscope has an outer sheath diameter of 42.06 Fr and an operative length of 177 mm, while the inner sheath has an operative length of 193 mm. It features a high-definition optical system with a maximum diameter of 4 mm, a length of 300 mm, and a 30° oblique viewing angle. Loops are available in various calibers, including 24 Fr, 27 Fr, and 30 Fr. An equatorial loop has been developed for smoothing. For the removal of the chips, we use an Ellik evacuator (KARL STORZ Endoskope, Tuttlingen, Germany) or a Toomey syringe (Allwin Medical, Anaheim, CA, USA).
A 2–4 cm midline transverse incision is made approximately 3 cm above the pubic symphysis. If the access site is uncertain, the suprapubic region can be punctured with a needle to locate the bladder and estimate the skin-to-bladder distance. The incision is deepened layer by layer through Camper’s and Scarpa’s fascia until the rectus abdominis fascia is reached. At this point, a longitudinal incision is made along the linea alba using a cold blade, followed by blunt dissection to access the anterior bladder wall. It is advisable to push the peritoneal fold cranially with a small swab while strictly remaining extraperitoneal. The bladder wall is identified and held with two Vicryl 2/0 sutures (Ethicon, Somerville, NJ, USA). A small longitudinal incision is made between them using electrocautery and a 15 mm laparoscopic trocar is inserted.
Resectoscope insertion and cystoscopy
The 42 Fr Gozzi resectoscope is introduced under direct vision through the trocar. A cystoscopy is performed to identify ureteral orifices, bladder neck, and prostate, ensuring proper orientation before resection. The catheter is deflated and retracted up to the veru-montanum. The tip of the catheter is positioned at the seminal colliculus, serving as both a landmark and an outlet pathway. The ureteral orifices are always marked with pinpoint coagulation 5–8 mm away toward the bladder neck. This ensures that their position remains identifiable throughout the procedure, especially when a large prostate displaces the ureteral orifices from their usual location. Similarly, marking with coagulation the circumference of the bladder neck helps to maintain visibility of the peripheral resection limits. For a particularly tight bladder neck, it is advisable to make an incision at the bladder neck before advancing the instrument through the prostatic urethra to prevent traumatic tears.
For bilobed adenomas or small median lobe involvement, resection can begin at the 11–1 o’clock position, initially using a medium loop (42 Fr). If a large median lobe is present, it is recommended to start its resection at the 5–7 o’clock position and complete the median lobe resection to create a good outflow channel. The lateral lobes can be resected using the largest loop available. The advantage is a full-length resection from the verumontanum to the bladder neck, resulting in very large glandular chips (up to 5 g) and allowing the procedure to be completed with fewer passes compared to traditional transurethral resection. After resecting the lateral lobes, a small or medium loop (42 Fr) can be used to refine the apex. Apical refinement is optimal in this procedure, as the external urinary sphincter remains visible at all times, preventing iatrogenic damage. If ejaculatory preservation is intended, the tissue surrounding the verumontanum is preserved. For finer adjustments, consider using an equatorial loop. Anatomically, the translational motion during resection from the verumontanum to the bladder neck naturally follows the contour of the prostate. During STARP-P, coagulation is performed after each bipolar loop pass. The limited number of passes ensures optimal visibility and improves time efficiency. At the end of the resection, there is a clear panoramic view of the prostatic lodge, allowing for meticulous and precise hemostasis.
Chips removal and catheter placement
At the end of the resection, the bladder catheter is temporarily cuffed under direct visualization at the level of the prostatic fossa to prevent chips from becoming lodged in this area. Prostatic chips after STAR-P are few in number and large in size and can be evacuated with an Ellik evacuator or a Toomey syringe. The last pieces may also be easily removed individually by hooking them with the resectoscope. A 16 Fr silicon catheter is placed into the bladder lumen under direct visualization. The trocar is now removed under direct visualization.
Closure of the surgical defect
The bladder wall is closed in two layers with Vicryl 2/0. At the end of the suturing, a leak test is always recommended by filling the bladder lumen with saline solution. The layers above the bladder are closed with sutures in accordance with anatomical planes, and the small skin incision is sealed using 4/0 Monocryl intradermic stitches (Ethicon, Somerville, NJ, USA), ensuring an optimal aesthetic result. In our series, no complications related to the access site were observed, including urinary leakage or fluid extravasation into the surrounding tissues. No suprapubic catheter was ever placed in our series of cases.
Bladder irrigation is maintained on the day of surgery or for 24 h (or as needed). The catheter is removed on the first postoperative day. Discharge occurs on the same day or the day after. Patients are advised to maintain adequate hydration and urinate frequently, avoiding bladder overdistension. A cystogram is not routinely performed, as no complications related to the transvesical access have ever been reported. Regular follow-up is conducted at 4 weeks and then at 3, 6, and 12 months, including uroflowmetry with post-void residual assessment and IPSS questionnaires.
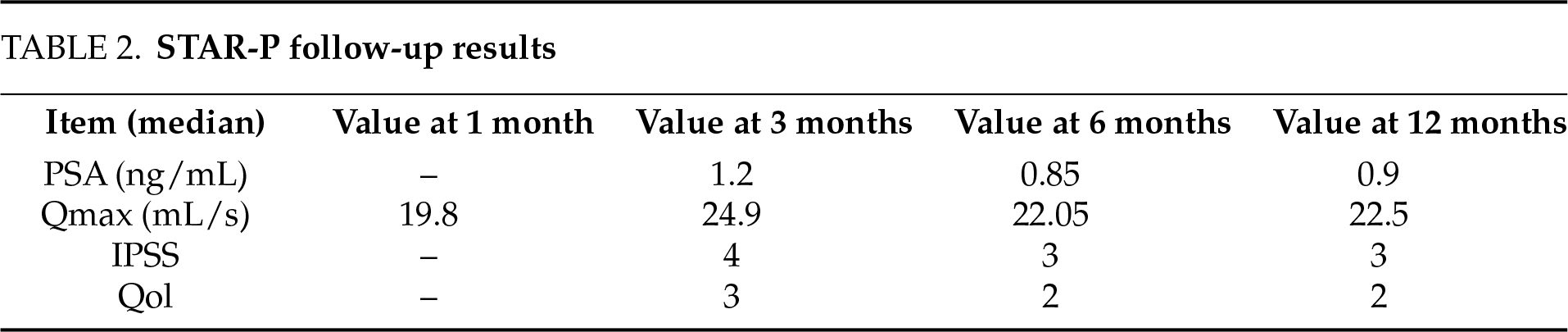
This single-center, single-operator study involved a retrospective analysis of prospectively collected data from 80 consecutive patients who underwent the STAR-P procedure (Table 1). Patient demographics and clinical data were meticulously gathered in a dedicated database and subsequently analyzed using descriptive statistical methods to ensure accuracy and clarity in outcome reporting. The procedure demonstrated a highly favorable safety profile, with no intraoperative complications recorded. Notably, no complications related specifically to the suprapubic access occurred. Importantly, by avoiding transurethral instrumentation, the procedure successfully circumvented all potential complications typically associated with transurethral prostate resections, including urethral strictures, sphincter injury, and related urinary dysfunction. The median overall operative time for the STAR-P procedure was 100 min, including preparation, resection, and closure phases, while the median duration of active adenoma resection was notably shorter at 60 min. The median length of hospitalization, based on all STAR-P procedures performed, is 2 nights, range (1–3) and the mean and median postoperative catheterization time was 2 days (range 1–4), considering that the initial data collection took place during the early phase of technique standardization. All patients included in the analysis exhibited a significant improvement in uroflowmetry indices postoperatively, along with high levels of patient satisfaction as reported in follow-up assessments. Specifically, the median preoperative maximum urinary flow rate (Qmax) was 10.85 mL/s, markedly increasing to a median postoperative Qmax of 22.5 mL/s, underscoring a robust functional improvement. The postoperative evaluation revealed a median post-void residual volume of 0 mL, with a mean residual of 4.34 mL, highlighting an effective bladder emptying capacity post-surgery. At follow-up, median PSA levels decreased from 1.2 ng/mL at 3 months to 0.85 ng/mL at 6 months and remained stable at 0.9 ng/mL after 12 months. Median Qmax improved from 19.8 mL/s at 1 month to 24.9 mL/s at 3 months, maintaining satisfactory flow rates at 6 months (22.05 mL/s) and 12 months (22.5 mL/s). Symptom scores showed progressive improvement, with median IPSS decreasing from 4 at 3 months to 3 at both 6 and 12 months, while quality of life (QoL) scores improved from 3 at 3 months to 2 at 6 and 12 months (Table 2). Patients enrolled in the study exhibited a median prostate volume of 85 mL, with prostate sizes ranging widely from 20 mL to 160 mL, demonstrating the versatility and applicability of the technique across varying prostate sizes. A small minority, specifically three patients out of the 80 evaluated (3.75%), reported transient symptoms of urinary urgency postoperatively. These symptoms resolved spontaneously and completely. No cases of stress urinary incontinence were reported. The total postoperative complication rate was 7.50% with only one complication requiring surgical reintervention. Analyzing the complications observed (considering that this series also includes patients operated on during the standardization phase of the technique), the following events were recorded: four cases of bladder neck contracture; one case of suprapubic access haematoms in a patient who subsequently required a hemostatic TUR on the first postoperative day (The patient did not correctly follow the instructions regarding the discontinuation of the anticoagulant tablet); one patient developed epididymitis within 30 days, which was treated with intramuscular ceftriaxone and oral azithromycin.
In recent years, the evolution of surgical techniques and technological innovation in the management of (BPO) has led to the emergence of a wide array of novel devices and approaches. These advancements have been progressively acknowledged and incorporated into both American and European clinical guidelines, establishing themselves as viable minimally invasive alternatives for selected patients.9,10 Nonetheless, with the notable exception of robot-assisted transvesical adenomectomy, the transurethral route remains the mainstay of surgical management in routine urological practice. Complications associated with transurethral access are well documented, yet they remain frequently underestimated by endoscopic urologists. This is largely due to the fact that the sequelae, such as urethral strictures or sphincteric injury, often fall under the domain of genitourinary reconstructive specialists, thus distancing the procedural act from its potential consequences.11 The array of surgical techniques currently available ranges from traditional monopolar or bipolar TURP to laser-based enucleation methods (HoLEP, ThuLEP), vaporization (PVP), and newer approaches such as water vapor thermal therapy (Rezūm™), prostatic urethral lift (PUL), or prostate artery embolization (PAE). While these techniques differ substantially in terms of invasiveness, learning curve, and functional outcomes, they also vary widely in guideline endorsement and cost effectiveness, highlighting significant discrepancies in international recommendations for emerging therapies such as iTIND™, RASP, and PAE, underscoring a lack of consensus due to insufficient long-term data and heterogeneous evidence grading systems.12 From an economic perspective, as reported by Vuichoud and Loughlin,13 the financial impact of BPO is considerable, with an estimated $4 billion spent annually in the U.S. alone. Direct costs (medications, procedures, follow-up visits), indirect costs (lost productivity), and intangible costs (quality of life impact) all contribute to the burden. Although only a small proportion of patients currently undergo surgery, demographic projections suggest a steep rise in demand for surgical treatment in the coming decades, emphasizing the need for cost-efficient, scalable solutions.
Among the various surgical options currently available for the treatment of large prostatic adenomas, RASP has emerged as a technically valid and functionally effective alternative. The European Association of Urology (EAU) guidelines acknowledge RASP as a suitable approach for patients with BPH and prostate volumes exceeding 80 mL, particularly when performed in high-volume centers with appropriate robotic expertise.14 Both multiport (MP) and single-port (SP) configurations have demonstrated encouraging perioperative outcomes, including reduced bleeding, shorter hospitalization, and lower analgesic requirements compared to open surgery.15,16 However, the economic dimension of robotic surgery must be carefully considered.6 Using data from the National Inpatient Sample in the United States reported that RASP was associated with significantly higher direct hospital costs compared to open simple prostatectomy, with an average cost differential of over $6000 per case. Although reduced transfusion rates, shorter lengths of stay, and fewer complications may help mitigate these costs in well-resourced settings, overall cost-efficiency remains highly dependent on institutional case volume and surgical expertise. The recent development and FDA approval of single-port robotic systems, such as the da Vinci SP, have allowed for refined minimally invasive transvesical approaches combining adenoma enucleation with 360° reconstructive mucosal flaps. Ramos et al.15 demonstrated the feasibility of SP-RASP even in glands exceeding 300 mL, with median blood loss of 100 mL, minimal pain, and over 95% same-day discharge.
However, these results were obtained in an elite academic setting with high robotic proficiency and access to cutting-edge platforms. It is important to emphasize that the infrastructure required for robotic procedures, including acquisition costs, maintenance, and surgeon training; limits their accessibility to a minority of healthcare systems worldwide. As noted by Vince et al.14 the application of such high-cost technology to a benign condition like BPH may be sustainable only in rich countries, where reimbursement models and institutional resources can absorb the expense. For most centers globally, including nations in Europe, Latin America, Asia, and Africa, robotic-assisted simple prostatectomy remains cost-prohibitive compared to other well-established, safe, and more cost-effective techniques, highlighting the need for effective and accessible alternatives.
Situated between the well-recognized risks associated with transurethral techniques and the economic inaccessibility of robotic surgery, the STAR-P procedure presents itself as a rational and potentially scalable alternative. By employing bipolar energy, alongside a newly designed yet cost-conscious instrumentation system, STAR-P avoids the need for capital-intensive infrastructure. Its safety profile, reproducibility, and learning curve appear favorable, especially in the hands of surgeons experienced in both endoscopic and open urological techniques.
While we acknowledge that broader validation through prospective multicenter studies and randomized controlled trials is essential, the results obtained thus far allow us to cautiously affirm the potential role of STAR-P within the modern surgical landscape of benign prostatic obstruction. In our view, its ability to balance clinical efficacy, technical accessibility, and economic sustainability makes it a candidate worthy of continued investigation and thoughtful integration into contemporary urological practice.
In summary, STAR-P represents a compelling addition to the surgical toolbox for BPH [supplementary material], combining the principles of endoscopic control with the accessibility of a mini-open suprapubic approach. Its reproducibility, safety, and economic sustainability make it particularly well suited not only for expert hands but also as a valuable training platform for the next generation of urologists. By bridging classic and contemporary techniques, STAR-P offers both a rational alternative to transurethral surgery and a pragmatic response to the financial and structural limitations of robotic systems.
Acknowledgement
The authors acknowledge Professor Christian Gozzi, who conceived the study and served as its Principal Investigator. Special thanks are extended to Professor Miroslav Djordjevic for his insightful guidance and for enriching the scientific discussion with his profound expertise in reconstructive urology. The support of Tontarra Medizintechnik, which developed and provided the custom equipment essential for this research, is gratefully acknowledged. Furthermore, the authors wish to express their deepest appreciation to the patients and their families for their crucial contribution and steadfast adherence to the study’s protocol.
Funding Statement
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. The necessary equipment for the study was developed and provided as an in-kind contribution by Tontarra Medizintechnik.
Author Contributions
Christian Gozzi: conceived the idea, ideated the research study and supervised manuscript transcription; Bruno Bucca: conducected experimental investigation, managed and analyzed data and wrote the initial draft of the manuscript; Orietta Dalpiaz: provided necessary resources, oversaw the project’s clinical aspects and provided critical revision to the manuscript for publication; Aldo Brassetti: provided scientific critical discussion, interpretation of data and revision of the manuscript; Luca Matteo Gobbi: contibuted in acquisition of data, data analysis and writing of the manuscript; Greta Basile: contibuted in acquisition of data, data analysis and writing of the manuscript; Miroslav Djordjevic: provided necessary resources, oversaw the project’s clinical aspects and provided critical revision to the manuscript for publication. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials
The raw data supporting this study are available from the corresponding author upon reasonable request, in order to ensure patient’s privacy.
Ethics Approval
All patients signed an informed consent. Our study was approved by the CityClinic Bolzano Ethical Board (sign date: 01 September 2020) and was conducted in accordance with the Declaration of Helsinki.
Conflicts of Interest
The authors declare that the equipment was manufactured and furnished by Tontarra Medizintechnik GmbH. However, The Tontarra Medizintechnik GmbH had no role in the study’s design, data, analysis or manuscript preaparation.
Supplementary Materials: The supplementary material is available online at https://www.techscience.com/doi/10.32604/cju.2025.068035/s1.
Abbreviation
| STAR-P | Suprapubic Transvesical Adenoma Resection of the Prostate |
| BPH | Benign Prostatic Hyperplasia |
| TURP | Transurethral Resection of the Prostate |
| HoLEP | Holmium Laser Enucleation of the Prostate |
| ThuLEP | Thullium Laser Enucleation of the Prostate |
| RASP | Robotic-Assisted Simple Prostatectomy |
| PSA | Prostate Specific Antigen |
| LUTS | Lower Urinary Tract Symptoms |
| Qmax | Maximum flow rate |
| BPO | Benign Prostatic Obstruction |
References
1. Madersbacher S, Roehrborn CG, Oelke M. The role of novel minimally invasive treatments for lower urinary tract symptoms associated with benign prostatic hyperplasia. BJU Int 2020;126(3):317–326. doi:10.1111/bju.15154. [Google Scholar] [PubMed] [CrossRef]
2. Iván PM, Alberto BA, Lluís PC et al. A real-world evidence study of interhospital variability in the surgical treatment of patients with benign prostatic hyperplasia: the REVALURO study. Int Urol Nephrol 2025;57(3):775–784. doi:10.1007/s11255-024-04239-7. [Google Scholar] [PubMed] [CrossRef]
3. Marcon J, Keller P, Pyrgidis N et al. Trends and perioperative outcomes of surgical treatments for benign prostatic hyperplasia in Germany: results from the GRAND study. Eur Urol Focus 2025;3:S2405–S4569. doi:10.1016/j.euf.2025.01.002. [Google Scholar] [PubMed] [CrossRef]
4. Bucca B, Gozzi C, Gobbi LM et al. The dark side of transurethral access for LUTS/BPH surgery: a narrative review. Asian J Androl 2025;39:2255. doi:10.4103/aja202523. [Google Scholar] [PubMed] [CrossRef]
5. Elsaqa M, Serag M, Leelani N et al. The incidence of urethral stricture and bladder neck contracture with transurethral resection vs. holmium laser enucleation of prostate: a matched, dual-center study. Can Urol Assoc 2022;17(1). doi:10.5489/cuaj.7967. [Google Scholar] [PubMed] [CrossRef]
6. Wan Mokhter WM, Duan X, Yang J, Mohamed Daud MA. Construction of a nomogram to predict urethral stricture after transurethral resection of the prostate: a retrospective cohort study. PLoS One 2025;20(2):e0313557. doi:10.1371/journal.pone.0313557. [Google Scholar] [PubMed] [CrossRef]
7. Berger AP, Horninger W, Rehder P et al. Suprapubic electroresection of prostate in three patients with benign prostatic hyperplasia and previous surgery for long urethral stricture. Urology 2005;65(4):798. doi:10.1016/j.urology.2004.10.052. [Google Scholar] [PubMed] [CrossRef]
8. Bucca B, Gobbi LM, Dalpiaz O et al. Suprapubic transvesical adenoma resection of the prostate (STAR-Pa novel technique for surgical treatment of benign prostatic hyperplasia. Euro Urol Focus 2024;10(6):991–998. doi:10.1016/j.euf.2024.05.009. [Google Scholar] [PubMed] [CrossRef]
9. Cornu JN, Gacci M, Hashim H et al. Non-neurogenic male lower urinary tract symptoms (LUTS) EAU guidelines on; 2025. [Google Scholar]
10. Sandhu JS, Goueli R, Kirkby E, Wilt TJ. Management of lower urinary tract symptoms attributed to benign prostatic hyperplasia (BPHAUA guideline amendment 2023. J Urol 2024;211(1):11–19. doi:10.1097/ju.0000000000003698. [Google Scholar] [PubMed] [CrossRef]
11. Cornu JN, Ahyai S, Bachmann A et al. A systematic review and meta-analysis of functional outcomes and complications following transurethral procedures for lower urinary tract symptoms resulting from benign prostatic obstruction: an update. Euro Urol 2015;67(6):1066–1096. doi:10.1016/j.eururo.2014.06.017. [Google Scholar] [PubMed] [CrossRef]
12. Matter LS, Burkhardt O, Lyatoshinsky P, Blarer J, Seiler R, Abt D. Surgical treatment of lower urinary tract symptoms secondary to benign prostatic obstruction: an analysis and meta-synthesis of available guidelines. BMC Urol 2025;25(1):99. doi:10.1186/s12894-025-01788-6. [Google Scholar] [PubMed] [CrossRef]
13. Vuichoud C, Loughlin KR. Benign prostatic hyperplasia: epidemiology, economics and evaluation. Can J Urol 2015;22 Suppl 1:1–6. [Google Scholar] [PubMed]
14. Vince R, Hampton LJ, Vartolomei MD, Shariat SF, Porpiglia F, Autorino R. Robotic assisted simple prostatectomy: recent advances. Curr Opin Urol 2018;28(3):309–314. doi:10.1097/MOU.0000000000000499. [Google Scholar] [PubMed] [CrossRef]
15. Ramos R, Ferguson E, Abou Zeinab M et al. Single-port transvesical robot-assisted simple prostatectomy: surgical technique and clinical outcomes. Euro Urol 2024;85(5):445–456. doi:10.1016/j.eururo.2023.11.012. [Google Scholar] [PubMed] [CrossRef]
16. Bhanvadia R, Ashbrook C, Gahan J et al. Perioperative outcomes and cost of robotic vs open simple prostatectomy in the modern robotic era: results from the National Inpatient Sample. BJU Int 2021;128(2):168–177. doi:10.1111/bju.15258. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools