 Open Access
Open Access
REVIEW
Progress of the pathogenesis in varicocele: a narrative review
1 School of Medical and Life Sciences, Chengdu University of Traditional Chinese Medicine, Chengdu, 611137, China
2 TCM Regulating Metabolic Diseases Key Laboratory of Sichuan Province, Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu, 610075, China
3 Assisted Reproduction Medicine Center, Chengdu Fifth People’s Hospital/The Fifth People’s Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu, 611130, China
* Corresponding Authors: Junjun Li. Email: ; Xujun Yu. Email:
# These authors contributed equally to this work
Canadian Journal of Urology 2026, 33(1), 63-74. https://doi.org/10.32604/cju.2025.068263
Received 24 May 2025; Accepted 27 October 2025; Issue published 28 February 2026
Abstract
Varicocele (VC) is widely recognized as a prevalent and clinically significant cause of male infertility. However, the comprehensive pathogenic mechanisms underlying VC development and progression remain incompletely understood, creating an important knowledge gap in the field of andrology. This review establishes that VC pathogenesis centers on abnormal vascular remodeling and integrates multiple contributing elements, including anatomical abnormalities, biochemical disturbances, genetic factors, low body mass index (BMI), age, and specific sports habits, while secondary varicoceles are primarily induced by compressive pathologies. Through a systematic synthesis of current evidence and recent advances, this review aims to elucidate the complex pathogenic network of VC and provide valuable insights to guide future research directions and inform the development of targeted clinical applications.Keywords
In the spermatic cord, VC is an aberrant dilatation, elongation, and tortuosity of the venous plexus, which represents the most common correctable cause of male infertility. It can also lead to pain and discomfort in the ipsilateral groin, scrotum, and testicular contents.1 The pathogenesis of VC-induced male infertility is primarily attributed to oxidative stress-mediated sperm damage. Additional contributing mechanisms include impaired venous drainage, which creates a hostile microenvironment characterized by local hyperthermia, hypoxia, and the reflux and accumulation of toxic metabolites.1 VC is prevalent among young adults and is found with a significantly higher frequency in infertile men. The estimated incidence of VC is approximately 15% in the general adult male population,1 compared to 45% in men with primary infertility and up to 80% in those with secondary infertility.2
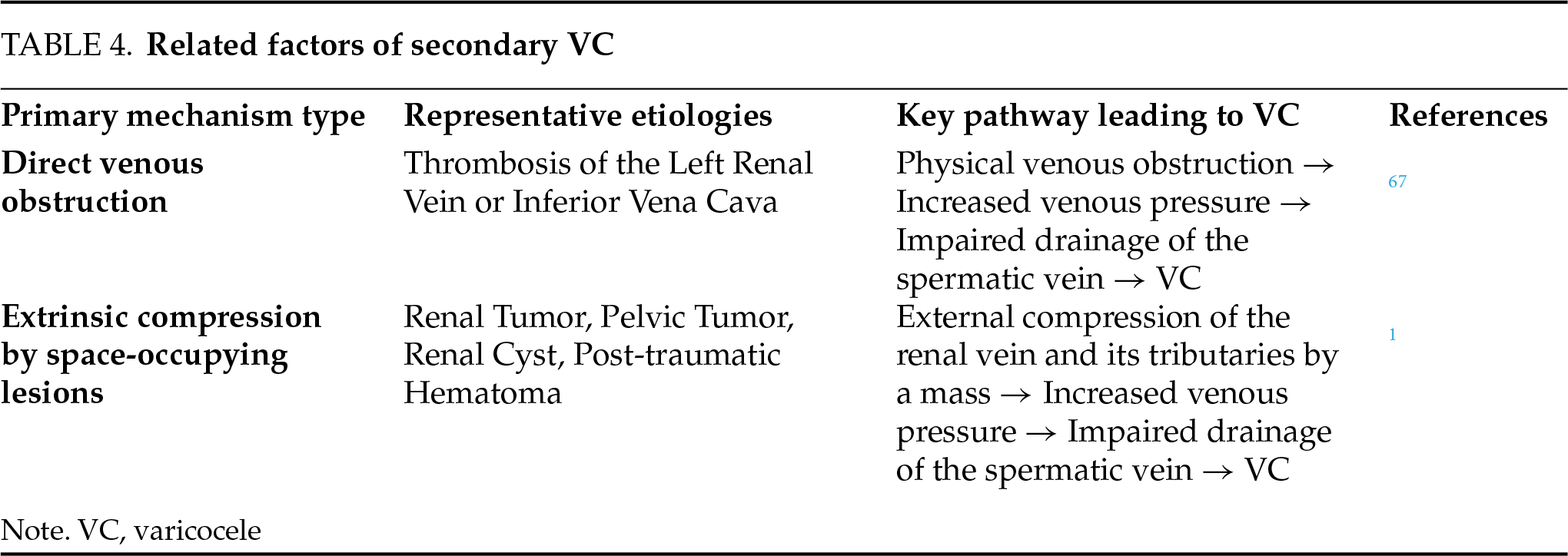
The precise pathophysiology of VC remains incompletely understood; however, it is clinically categorized into primary and secondary types based on etiology. Primary VC is characterized by symptoms that become apparent upon standing or walking and typically subside with rest. The etiology of primary VC is multifactorial, potentially involving congenital anomalies such as incompetent or absent valves within the internal spermatic vein (ISV), inherent weakness of the venous wall and its supporting tissues, a deficiency of smooth muscle or elastic fibers within the venous wall, or underdevelopment of the cremasteric muscle. Secondary VC, which is less common than the primary form, predominantly results from pathologies that impede venous return within the spermatic cord. The main causes include pathological obstruction of the left renal vein or inferior vena cava, and extrinsic compression due to space-occupying lesions such as renal, retroperitoneal, or pelvic tumors.1 Additional compressive etiologies encompass severe hydronephrosis, renal cysts, and retroperitoneal ectopic blood vessels. These conditions lead to mechanical blockage of the venous drainage pathway, ultimately resulting in VC formation.3 The current mainstream management strategy for VC involves conservative observation for asymptomatic patients. For those with palpable VC and significant symptoms, surgical intervention is recommended. The preferred surgical options include microsurgical varicocelectomy, which is generally considered the gold standard, or laparoscopic varicocelectomy, depending on the specific clinical scenario.4 This review aims to synthesize recent advances in our understanding of the pathophysiology of VC, thereby providing a novel framework to inform future strategies for its prevention, diagnosis, and clinical management.
Primary VC exhibits a clear predilection for the left side, occurs bilaterally with lower frequency, and is found in isolation on the right side only rarely.5 Guan et al.6 reported a significant reduction in testicular consistency on the ipsilateral side in infertile patients with left-sided VC. Their study further identified an inverse correlation between the degree of testicular tissue softening and the clinical grade of the VC. Notably, as the severity of the condition progressed, the contralateral testicular tissue also demonstrated compromised consistency and texture. This bilateral deterioration in testicular integrity is associated with impaired spermatogenesis, ultimately contributing to the pathogenesis of male infertility.
Mechanisms of vascular remodeling
Vascular remodeling refers to the process of structural and functional alterations in the blood vessel wall. This process is driven by a complex interplay of inflammatory responses, smooth muscle cell proliferation, apoptosis, migration, and the dynamic turnover of the extracellular matrix. These cellular and molecular events can result in either adaptive or maladaptive outcomes. Adaptive alterations are typically initiated in response to stimuli such as growth factors, vasoactive substances, or hemodynamic changes.7 However, dysregulation of the venous wall’s compensatory mechanisms to such imbalances can lead to maladaptive remodeling, which manifests clinically as varicose veins.8
The pathophysiology of varicose veins is characterized by significant abnormalities in both the extracellular matrix and smooth muscle cells, which collectively lead to vascular dysfunction. The extracellular matrix, primarily composed of collagen types I and III, confers tensile strength and structural support to blood vessels. Consequently, aberrations in extracellular matrix composition directly compromise vascular integrity and morphology. Morphological and biochemical studies indicate that the dissociation and degradation of smooth muscle cells within the venous wall represent key initiating events in varicose vein pathogenesis. Histologically, smooth muscle cells in varicose veins exhibit distinct features, including enlarged nuclei and cytoplasm, increased organelles, irregular shape, loss of myofilaments, and a synthetic phenotype. Additionally, substantial deposits of collagen fibers, predominantly type III, are observed in the intima, while collagen aggregates in the medial and adventitial layers display increased abundance and disorganization.7,9 The irregular thickness of collagen fibers between smooth muscle cells results in widened intercellular spacing, collectively impairing the mechanical properties of the venous wall.9
Liu10 observed a significant reduction in nerve fiber density within the peripheral and tubular walls of varicose ISVs compared to normal veins, a finding that was positively correlated with the degree of varicosity. This suggests that aberrant development of the vascular-associated nerves may constitute an important etiological factor in VC. In addition to this, Tilki reported a significant reduction in nerve fiber density within the outer smooth muscle layer and tubular walls of varicose spermatic veins compared to normal controls. They attributed this reduction to impaired blood supply to the vessel wall. Furthermore, they proposed that the degeneration of the vessel wall and a diminished neurotrophic effect, both resulting from the compromised vasculature, disrupt the contractile function of the spermatic vein’s smooth muscle tissue. This mechanism may represent a key pathogenetic pathway in VC development.11
The unique upright posture of humans is a significant predisposing factor for the development of VC. In humans, the ISV assumes a vertical orientation when standing. This anatomical arrangement results in substantially higher hydrostatic pressure within the spermatic vein lumen compared to the venous systems of quadrupeds.12 According to Xu et al.,13 the standing position increases lower abdominal pressure, thereby exacerbating both the likelihood and severity of VC development and enhancing their visibility on angiographic imaging. This finding is further supported by a study by Li et al.,14 which demonstrated that individuals with VC tend to be taller than those without the condition.
Discrepancy between Right and Left
Significant anatomical asymmetries exist between the left and right spermatic veins. The left ISV is approximately 8–10 cm longer than the right and typically joins the left renal vein at an approximately right angle. In contrast, the right spermatic vein drains obliquely into the inferior vena cava. From a fluid dynamics perspective, the increased length and near-right-angled entry on the left side result in a longer flow path, effectively increasing the vessel’s functional length as described by Poiseuille’s law. This anatomical configuration consequently leads to greater hemodynamic resistance to blood flow on the left side.6 Consequently, primary VCs are considerably more prevalent on the left side, while isolated right-sided cases are uncommon. This clinical pattern is further supported by surgical outcomes. In a study15 of 50 boys (aged 6–14 years) undergoing left-sided VC repair, preoperative pressure measurement in 34 patients combined with intraoperative venography revealed that 38 patients had impaired renal venous drainage and elevated left renal vein pressure. During a follow-up of up to five years, all five patients who experienced recurrence belonged to this group with preoperative elevated pressure. These findings reinforce the strong correlation between the anatomical disparities of the spermatic veins and both the initial development and recurrence of left-sided VC.15
Furthermore, an embryological hypothesis has been proposed suggesting that the left spermatic vein may originate from remnants of the regressed left posterior cardinal vein during embryonic development. The alterations in the thickness and structural strength of the inferior wall of the left spermatic vein resulting from this developmental process are considered a potential contributing factor to the high incidence of VC on the left side.16
Venous Valve Defects or Insufficiency
Under physiological conditions, competent valves prevent venous reflux. Their absence or insufficiency leads to pathological blood reflux and stasis. Although venous valves were historically thought to be absent in intra-abdominal veins, recent evidence indicates that functional valves can be present in the renal veins and inferior vena cava. Consequently, the absence or dysfunction of valves at critical junctions—specifically within the left renal vein and the segment of the ISV near its confluence—constitutes a significant risk factor for VC. This is corroborated by retrograde venography studies, which demonstrate that approximately 75% of left-sided VC are associated with venous valve insufficiency leading to reflux. The present status of research on pertinent venous vascular valves and their connection to VC will be discussed separately in the sections that follow.17
It is generally accepted that competent renal vein valves demonstrate a distinct left-sided predominance.18 This may be related to the fact that the left renal vein needs to receive additional blood from the left ISV for confluence. Accordingly, as the internal pressure in the renal vein is greater than the internal pressure in the ISV in the physiological state, if the renal vein valves become deficient or dysfunctional, blood will continue to reflux along the left renal vein and pool in the left ISV, thereby increasing the risk of VC. Some studies19 have shown that some patients with VC do not have valves in their renal vein anatomy. We believe that the absence or dysfunction of renal vein valves may be a risk factor for the development of VC, which will increase the risk of renal vein blood reflux and pooling in the ISV, but the related hemodynamic changes and the corresponding morphological changes of the spermatic vein need to be further investigated.
The valves in the spermatic veins are mostly located in the scrotal and inguinal regions. The overall number of valves was greater on the left than on the right. At the same time, the frequency of valve loss was twice as high on the left side as on the right side.20 The traditional pathogenic view attributed a significant role to the absence of valves in the spermatic veins as a key factor, or even a primary risk factor, for VC development. However, this perspective has been challenged by recent clinical observations indicating that the presence or absence of valves does not strictly correlate with clinical status. Specifically, patients with symptomatic VC may possess intact valves, whereas individuals without VC can exhibit valve agenesis or dysfunction. Consequently, isolated valvular absence or hypoplasia is now considered an uncommon and insufficient sole cause of VC, underscoring the multifactorial nature of its pathogenesis.21 The striking disparity in VC prevalence between the left and right sides, coupled with the established hemodynamic influence of venous valves,1,4 highlights a critical area for further research. However, the specific pathophysiological mechanisms responsible for the asymmetrical distribution of valves and the differing rates of valvular deficiency between the sides remain poorly understood. Therefore, a systematic investigation into the precise role of spermatic vein valve morphology and function in the pathogenesis of VC is strongly warranted.
Renal Venous Hypertension
Renal venous hypertension is a key mechanistic driver in VC pathogenesis. This is directly substantiated by the findings of Yasumoto,22 who measured significantly elevated pressure in the renal veins of patients with stenotic varicose veins and abundant collateral circulation. Such excessive pressure exerts a direct mechanical effect: beyond a critical threshold, it forces the venous wall to bend and elongate, resulting in VC.23
Deficiencies of the Spermatic Cord Coverings
The spermatic cord coverings, which encase the ISVs and the vas deferens, contain a muscular component composed primarily of the cremaster muscle and the internal and external spermatic fasciae. This sheath functions as a pumping mechanism that facilitates venous return and restricts excessive venous dilation.24 Atrophy or relaxation of this muscular tissue compromises the structural integrity of the venous wall, promoting the formation of varicose veins. Experimental evidence supporting this concept comes from a study in which unilateral excision of the spermatic fascial sheaths in dogs resulted in varicose changes on the operated side, underscoring the critical role of these structures in maintaining normal venous hemodynamics.25
A meta-analysis by Liu et al.26 indicated a lower recurrence rate following endovascular treatment for VC compared to surgical intervention. It has been postulated that this difference may be attributable to the surgical disruption of the spermatic cord coverings, which normally provide a pumping action that aids venous return and restricts dilatation. However, this hypothesis is challenged by a contrasting study of 40 patients who underwent inguinal hernia repair with removal of the spermatic sheath, none of whom developed a VC within 6–18 months postoperatively.27 Given the conflicting evidence and the limited number of studies specifically examining the consequences of sheath excision, it can be inferred that while impairment of the spermatic cord envelope may increase susceptibility to VC, it is unlikely to be the primary causative factor.
Vascular Compression Phenomena
Nutcracker syndrome, also referred to as left renal vein compression syndrome, encompasses the clinical manifestations resulting from chronic compression of the left renal vein. Anatomically, the left renal vein courses through the angle between the superior mesenteric artery and the abdominal aorta, which normally ranges from 19.4° to 70.7°.28 A reduction of this angle due to physiological or pathological conditions can lead to the anterior nutcracker phenomenon.29 Conversely, alterations in the anatomical position of the left renal vein may subject it to compression between the abdominal aorta and the spinal column, resulting in the posterior nutcracker phenomenon.30 The nutcracker phenomenon can lead to compression and elevated internal pressure within the left renal vein. This increased pressure may impair drainage of the left spermatic vein, potentially resulting in VC formation. Epidemiological studies indicate that about half of all patients with a left-sided VC are found to have the compression syndrome.31 This condition is particularly prevalent among adolescents, likely due to rapid growth phases that involve excessive height gain, vertebral hyperextension, and significant changes in body habitus, which can reduce the aortomesenteric angle.31 Additionally, the relative paucity of protective adipose and connective tissue around the superior mesenteric artery origin in adolescents may further predispose them to left renal vein compression compared to adult males.32 Other analogous vascular compression etiologies are summarized in Table 1.
The epidemiologic data indicate that first-degree relatives of individuals with VC exhibit a significantly higher prevalence of the condition compared to the first-degree relatives of unaffected individuals.36 Evidence from genetic association studies implicates specific polymorphic loci in VC susceptibility. These include the GSTM1 locus within the glutathione transferase family (GSTF) and the A1298C locus of the methylenetetrahydrofolate reductase (MTHFR) gene.37 Notably, the MTHFR 1298AA genotype is associated with a 2.3-fold increased risk of VC compared to other genotypes.36 The endothelial nitric oxide synthase gene (NOS3) encodes endothelial nitric oxide synthase protein (eNOS), a key enzyme that regulates plasma nitric oxide (NO) levels. Specific polymorphisms in this gene, such as G894T and 4b/a, have been studied. These variations have shown a significant link to the development of VC.38 Additionally, research by Heidari et al.39 indicates that specific CAG repeat sequence genotypes (e.g., 10/10 and 10/#10) in the polymerase gamma (POLG) gene are significantly negatively correlated with VC severity, with distinct genotypic frequencies observed across different severity classes.
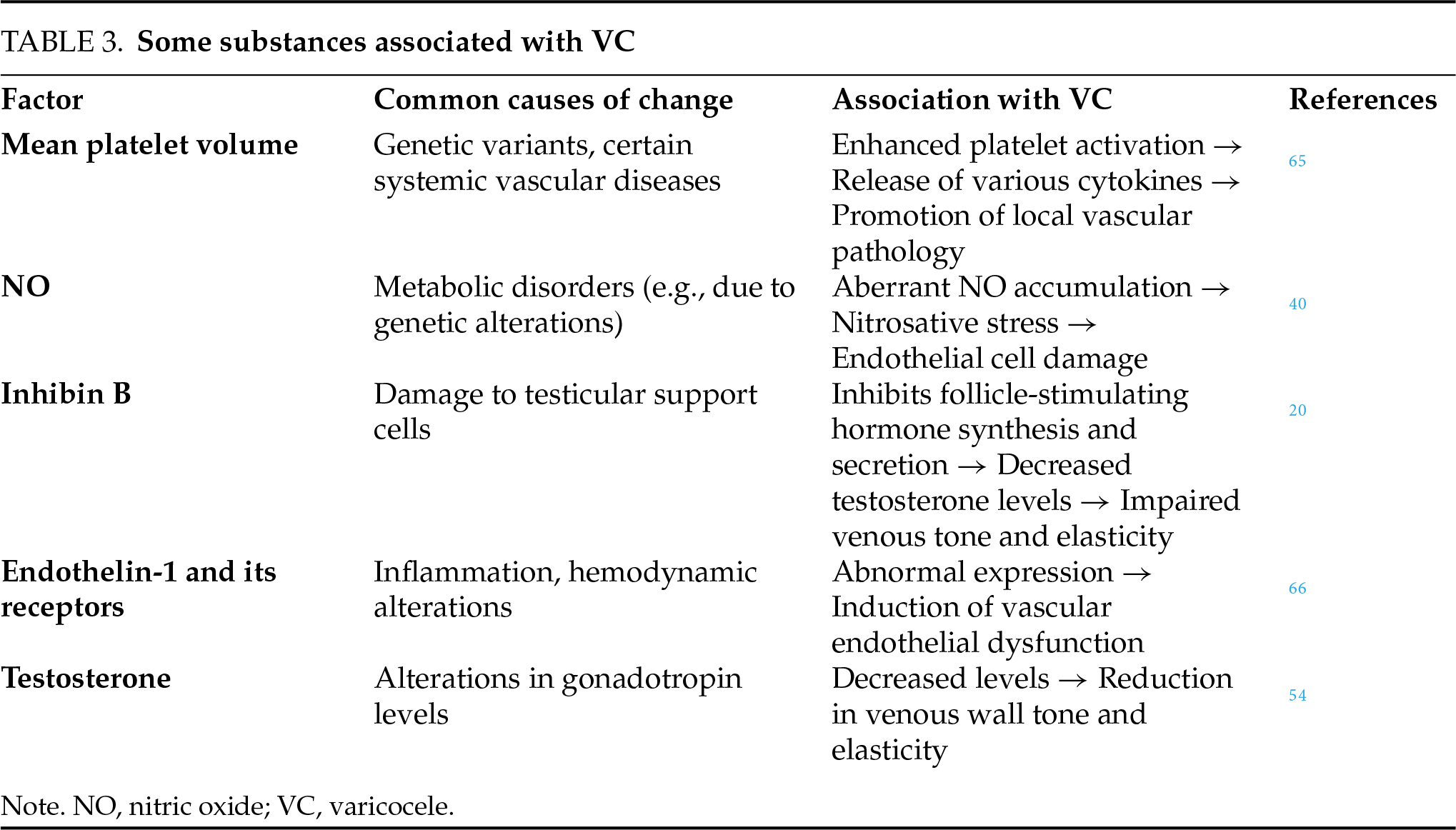
Through data mining and analysis of deleterious nonsynonymous single-nucleotide polymorphisms in the neuronal nitric oxide synthase gene (NOS1) and arginase 2 (ARG2) genes, Karthikeyan et al.40 concluded that the A52P mutation in ARG2 can severely compromise protein stability, leading to loss of function and consequent accumulation of NO. Furthermore, they suggested that the SNP rs200091597 (T718M) in the NOS1 gene could enhance the stability and thus the activity of the NOS1 enzyme. This increased activity would promote the utilization of L-arginine and overproduction of NO, potentially resulting in nitrosative stress—a proposed mechanism contributing to VC pathogenesis.41 However, these findings are derived from computational analyses and require validation through subsequent basic science experiments and clinical studies.
Yang et al.36 conducted the first comprehensive molecular characterization of VC using an integrated whole-exome and transcriptome sequencing approach. Their analysis identified three candidate genes—serine protease inhibitor Kunitz type 1 (SPINT1) gene, Angio-associated, migratory cell protein (AAMP) gene and proliferation marker Ki-67 (MKI67) gene and four key biological pathways—mitogen-activated protein kinase (MAPK) signaling, focal adhesion pathway, extracellular matrix (ECM)-receptor interaction, and hematopoietic cell regulatory signaling—as potentially critical to VC pathogenesis. This foundational work provides important insights into the molecular mechanisms underlying VC development and offers a valuable resource for future research.
The Forkhead box protein C2 (FOXC2) gene, a critical regulator of vascular and lymphatic development, has been implicated in venous valve insufficiency.42,43 Evidence suggests its involvement extends to pathological venous remodeling. For instance, Surendran et al.44 linked the upregulation of the FoxC2-Dll4 pathway to arterialization and abnormal wall remodeling (characterized by smooth muscle cell hypertrophy and hyperplasia) in varicose saphenous veins. Complementing this, Zhang et al.45 demonstrated through in vitro analysis that overexpression of FOXC2-AS1 promotes the phenotypic transformation, proliferation, and migration of saphenous vein smooth muscle cells—effects reversible upon FOXC2 silencing or Notch pathway inhibition. Separately, the vascular endothelial growth factor A (VEGFA) gene and its receptor VEGFR2, which share functional parallels with NOS3 in vascular regulation, are also significantly upregulated in varicose vein walls, a phenomenon potentially mediated by nitric oxide synthase (NOS) activity.46 Based on these findings in other venous pathologies, it is hypothesized that the FOXC2 and VEGFA/VEGFR2 pathways may contribute to VC pathogenesis, although direct evidence specific to VC remains to be established.
Rao et al.47 reported a higher incidence of genetic abnormalities in infertile men with VC compared to those with idiopathic infertility. In their cohort of 251 patients, chromosomal defects were found in 19.3% of VC patients versus 8.76% in the idiopathic group, while Y chromosome microdeletion rates were 5.26% and 3.60%, respectively. Overall, chromosomal abnormalities and Y microdeletions were present in 11.5% and 3.98% of the total infertile cohort, reinforcing an association between VC and these genetic anomalies. Supporting this, Song et al.48 identified microdeletions in AZF regions (AZFa, b, c, or b + c) in approximately 19% (12/62) of idiopathic infertility patients. Notably, a high frequency of microdeletions (11.1%, 9/81) was also detected in a subgroup of non-idiopathic infertility patients who had VC and cryptorchidism. These collective findings suggest a potential link between VC, infertility, and underlying chromosomal abnormalities.
An epidemiological study by Kumanov et al.49 reported two notable associations. First, a higher incidence of VC was observed in boys with dark brown eyes, suggesting a potential link to genetic factors influencing scleral pigmentation; however, this preliminary finding requires validation in larger, specifically designed studies due to limited sample size and a lack of similar research. Second, significant regional disparities were noted, with a higher prevalence among children in large cities compared to those in ordinary towns, possibly attributable to environmental or nutritional factors. A substantial difference in prevalence between Greek and Turkish boys was also recorded, pointing to potential ethnogenetic variations—a hypothesis that likewise warrants confirmation through larger population-based cohort studies.
In summary, the pathogenesis of VC involves a complex interplay of genetic factors, including chromosomal abnormalities, diverse gene poly morphisms, dysregulated biological pathways, and potential ethnogenetic backgrounds. These elements collectively promote VC development by disrupting vascular function, amplifying oxidative stress, and altering ECM homeostasis. The specific roles, biological processes, and pathogenic links of these genetic components are systematically detailed in Table 2, Genetic Factors. These insights hold considerable promise for advancing VC management—enabling genetic screening for high-risk individuals, guiding genotype-specific treatment strategies, and facilitating targeted drug development. Nevertheless, further validation through mechanistic studies and large-scale population cohorts remains essential to firmly establish causal relationships and translational applicability.

The association between physical exercise and the risk of developing VC remains contentious, with conflicting evidence reported in the literature. Di Luigi et al.50 reported a notably high prevalence of VC (29%) among male athletes, a rate substantially exceeding that observed in the general male population. A subsequent study by Hariri et al.51 found a significant positive correlation between VC prevalence and the frequency of weekly exercise sessions, with individuals exercising more than three times per week demonstrating a higher risk. However, no significant association was observed with the type of exercise (e.g., strength training versus aerobics) or the duration of individual sessions. Conversely, Rigano et al.52 reported no significant difference in VC incidence between a cohort of 150 adolescent athletes (aged 10–16 years) and a matched group of 150 non-athletes, leading them to propose that physical activity may not initiate VC but rather exacerbate pre-existing conditions. These discrepant findings may be attributable to key methodological variations across studies, particularly differences in the age cohorts examined (adults versus adolescents), which likely reflect divergent physiological susceptibilities, as well as potential disparities in exercise intensity thresholds. Di Luigi et al.53 further postulated that exercise-induced systemic and local factors—such as suppressed gonadotropin levels, elevated anti-gonadal hormones (which directly reduce testosterone synthesis, thereby impairing venous wall integrity; see Table 3 for detailed mechanisms),54 and transient increases in intra-abdominal pressure may collectively contribute to the development of VC in athletes.
In a key study, Atar and colleagues demonstrated that acute exercise leads to a significant dilation of the spermatic veins and a prolonged duration of venous reflux in individuals with VC.55 While the precise pathophysiological mechanisms underlying this association require further elucidation, the cumulative evidence points to a potential exacerbating role of physical activity in VC. Critical modulatory factors are thought to encompass the frequency, specific type, intensity, and duration of exercise, in addition to underlying individual susceptibility. Hence, future investigations employing rigorous, prospective cohort designs are essential to definitively establish the causal relationship between exercise parameters and VC onset and progression.
BMI is a widely adopted anthropometric measure for estimating adiposity. The meta-analyses has established an inverse correlation between elevated BMI and the risk of developing VC, indicating that men with lower body weight and BMI demonstrate a higher prevalence of VC.14 There is one hypothesis posits that increased perivascular and retroperitoneal adipose tissue in individuals with higher BMI may cushion the left renal vein, thereby mitigating the “nutcracker phenomenon”.56 An alternative, though less supported, explanation suggested that the apparent lower incidence of VC in obese individuals could be attributable to the masking effect of substantial adipose tissue during physical examination.56 However, this diagnostic bias was effectively refuted by Walters57 through the utilization of ultrasound imaging, which provides an objective assessment unaffected by body habitus.
Unusually, a survey of 211,989 men aged 18–65 years in China showed that the incidence of VC on the right side of the spermatic vein was increased by 63.3 per cent in obese men, and the researchers proposed that the possible reason for the discrepancy was that higher BMI tends to imply a higher intra-abdominal pressure and this causes high pressure in the inferior vena cava.58 The right spermatic vein generally drains directly into the inferior vena cava, and higher inferior vena cava pressure can impede venous blood return and venous valve abnormalities, ultimately leading to VC.2 A probable cause for the increased frequency of VC on the right side is that fat can diminish the Nutcracker effect on the left side, which is stronger than the effect on the left side brought about by elevated inferior vena cava pressure. In fact, there is a lack of research on the overall effect of BMI on VC, and there are many close and complex relationships between adipose and vascular tissues. Furthermore, obesity has been traditionally thought to have many other adverse effects on the vasculature.59 Therefore, the proposed mechanisms should be considered preliminary and partial explanations derived from specific observational findings, rather than conclusions drawn from comprehensive mechanistic studies. Consequently, there is a clear necessity for more specialized cohort research to definitively elucidate the relationship between BMI and VC. Nonetheless, it is imperative to reiterate that an elevated BMI is generally associated with a decreased risk of developing VC. It is also critically important to emphasize that although obesity may correlate with a lower incidence of VC, it should under no circumstances be misconstrued as a scientifically valid preventive measure. This is due to the well-documented detrimental effects of obesity on systemic vascular health, which far outweigh any potential local protective association.
Akbay et al.60 conducted a cross-sectional investigation of VC prevalence among 4052 boys aged 2–19 years, categorized into two groups: 2–10 years (n = 1521 children) and 11–19 years (n = 2531 adolescents). The reported prevalence was markedly different between cohorts, at 0.92% in the children group compared to 11.02% in the adolescent group. This striking disparity is likely attributable to the rapid testicular growth and consequent abrupt increase in testicular blood flow during puberty, which may exceed the drainage capacity of the spermatic venous system, promoting VC development.61 Additionally, the anatomical changes associated with puberty are hypothesized to exacerbate the nutcracker phenomenon, further compromising venous return.62 In contrast to the developmental focus of Akbay et al.,60 there is a study1 reported the age-related progression of VC. The data revealed that VC prevalence increases by approximately 10% per decade in this older demographic. Collectively, these findings underscore the chronic and progressive nature of VC. The gradual rise in prevalence with advancing age may involve age-related degenerative changes, such as venous valve incompetence and loss of venous wall elasticity, which occur over extended periods during development and aging.
Other possible contributing factors
Clinical evidence indicates a strong association between smoking and a greater than twofold increase in the incidence of VC. This observation is supported by animal studies identifying gonadal toxins as potential pathogenic cofactors, which are also implicated in the development of concomitant conditions such as oligospermia and scleropathy of the spermatic cord.63 The underlying mechanism likely involves direct gonadotoxic effects and oxidative stress damage induced by nicotine and other harmful compounds. In the context of VC-related oxidative stress, several molecular factors with antioxidant and anti-inflammatory functions have been studied. For example, the transcription factor nuclear factor erythroid 2-related factor 2 (Nrf2) is a key regulator of antioxidant responses. Separately, the enzyme heme oxygenase-1 (HO-1) also exerts potent cytoprotective effects. Xu64 reported that the expression of Nrf2 and HO-1 was significantly higher in VC compared to normal controls. The upregulation of these factors is considered part of the cellular defense mechanism against oxidative stress. Given the potential roles of Nrf2, HO-1, and other molecules, Table 3 summarizes key factors associated with VC, detailing their functions and proposed links to pathogenesis.
Secondary VC is brought on by pathological or local compression of the left renal vein and its refluxing veins, which blocks venous return in the spermatic cord. It is observed in conditions that impact the return of blood, such as thromboembolism-induced blockage of the left renal vein or vena cava, renal tumors, retroperitoneal or pelvic tumors, and so on.1
Furthermore, a significantly higher prevalence of VC has been reported in patients with coronary artery ectasia, suggesting a potential association between VC and other systemic vascular abnormalities.35 Supporting this notion, this study35 also reported a statistically significant correlation between VC and sapheno-femoral junction insufficiency. Collectively, these findings indicate that VC may be part of a broader systemic vascular disorder. However, larger-scale studies are necessary to definitively establish this relationship. The common etiological factors associated with secondary VC are summarized in Table 4.
This review consolidates current knowledge on VC pathogenesis, framing vascular remodeling as a central mechanism. Despite evidence for the involvement of multiple pathways, significant deficiencies persist in our understanding of the genetic determinants and precise molecular triggers of VC. The disease’s complex etiology and variable clinical presentation underpin the considerable limitations of current diagnostic and therapeutic strategies. It is therefore imperative to translate emerging mechanistic insights into clinical applications that address these shortcomings. Future research should prioritize the use of high-throughput sequencing to identify key genes and signaling pathways, thereby providing a foundation for developing targeted and gene-based therapies. Furthermore, proteomic and metabolomic studies on large patient cohorts are warranted to identify robust diagnostic biomarkers. The subsequent application of molecular profiling could potentially supersede conventional diagnostic methods like ultrasound or palpation, enabling precise subtyping and personalized treatment.68 The development of more accurate animal and in vitro models that faithfully recapitulate human VC is also crucial for rigorously evaluating the long-term efficacy of minimally invasive surgeries and pharmacological interventions. In conclusion, large-scale, multi-dimensional research initiatives are essential to drive a comprehensive innovation in the diagnosis and management of VC.
Acknowledgement
Not applicable.
Funding Statement
This research was funded by China Postdoctoral Science Foundation (Grant Number: 2025M773939), National Natural Science Foundation of China (Grant Number: 82205131), and Sichuan Science and Technology Program (Grant Number: 2025ZNSFSC1798).
Author Contributions
The authors confirm contribution to the paper as follows: Conceptualization, Liang Dong and Jialu Xin; methodology, Jialu Xin; validation, Liang Dong, Jingyi Zhang and Zheng Liu; formal analysis, Jialu Xin; investigation, Xujun Yu; resources, Junjun Li; data curation, Jialu Xin; writing—original draft preparation, Liang Dong and Jialu Xin; writing—review and editing, Xujun Yu and Junjun Li; visualization, Junjun Li; supervision, Xujun Yu; project administration, Liang Dong; funding acquisition, Liang Dong. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials
Not applicable.
Ethics Approval
Not applicable.
Conflicts of Interest
The authors declare no conflicts of interest to report regarding the present study.
Abbreviations
| AAMP | Angio-associated, migratory cell protein |
| ARG2 | Arginase 2 |
| BMI | Body mass index |
| eNOS | Endothelial nitric oxide synthase protein |
| ECM | Extracellular matrix |
| FOXC2 | Forkhead box protein C2 |
| GSTF | Glutathione transferase family |
| HO-1 | Heme oxygenase-1 |
| ISV | Internal spermatic vein |
| MAPK | Mitogen-activated protein kinase |
| MTHFR | Methylenetetrahydrofolate reductase |
| MKI67 | Proliferation marker Ki-67 |
| NO | Nitric oxide |
| NOS | Nitric oxide synthase |
| NOS1 | Neuronal nitric oxide synthase gene |
| NOS3 | Endothelial nitric oxide synthase gene |
| Nrf2 | Nuclear factor erythroid 2-related factor |
| POLG | Polymerase gamma |
| SPINT1 | Serine protease inhibitor Kunitz type 1 |
| VC | Varicocele |
| VEGFA | Vascular endothelial growth factor A |
References
1. Chinese Medical Association Andrology Branch. Guidelines for diagnosis and treatment of varicocele. Zhonghua Nan Ke Xue Za Zhi 2022;28(8):756–767. (In Chinese). doi:10.13263/j.cnki.nja.2022.08.010. [Google Scholar] [CrossRef]
2. Hu X, Yang X, Zhao J et al. Association between body mass index and varicocele among 211,989 Chinese reproductive-age males. Int J Urol 2022;29(8):853–859. doi:10.1111/iju.14915. [Google Scholar] [PubMed] [CrossRef]
3. Sun SZ, Yu L, Wang HQ et al. Analysis of the cause of varicocele recurrence and the application of sub-inguinal microsurgical varicocelectomy in recurrent varicocele. Chin J Urol 2021;42(3):208–213. (In Chinese). doi:10.3760/cma.j.cn112330-20210115-00022. [Google Scholar] [CrossRef]
4. Chiba K, Fujisawa M. Clinical outcomes of varicocele repair in infertile men: a review. World J Mens Health 2016;34(2):101–109. doi:10.5534/wjmh.2016.34.2.101. [Google Scholar] [PubMed] [CrossRef]
5. Clavijo RI, Carrasquillo R, Ramasamy R. Varicoceles: prevalence and pathogenesis in adult men. Fertil Steril 2017;108(3):364–369. doi:10.1016/j.fertnstert.2017.06.036. [Google Scholar] [PubMed] [CrossRef]
6. Guan X, Cao W, Lei HE et al. Application value of elastic ultrasound and contrast-enhanced ultrasonography in evaluating testicular blood supply after blockage of the vas deferens artery. Natl J Androl 2019;25(11):990–995. (In Chinese). doi:10.13263/j.cnki.nja.2019.11.005. [Google Scholar] [CrossRef]
7. Sun SZ. Study on the pathological mechanism of varicocele vessel remodeling [dissertation]. Qingdao, China: Qingdao University; 2021. (In Chinese). doi:10.27262/d.cnki.gqdau.2021.001186. [Google Scholar] [CrossRef]
8. Méndez-Barbero N, Gutiérrez-Muñoz C, Blanco-Colio LM. Cellular crosstalk between endothelial and smooth muscle cells in vascular wall remodeling. Int J Mol Sci 2021;22(14):7284. doi:10.3390/ijms22147284. [Google Scholar] [PubMed] [CrossRef]
9. Liu XY. Experimental study on vascular remodeling of varicocele [dissertation]. Chongqing, China: Chongqing Medical University; 2008. (In Chinese). [Google Scholar]
10. Liu YQ. Research on pathology of internal spermatic vein of varicocele and approach to it’s relative pathogenesis [dissertation]. Hefei, China: Anhui Medical University; 2009. (In Chinese). [Google Scholar]
11. Tilki D, Kilic E, Tauber R et al. The complex structure of the smooth muscle layer of spermatic veins and its potential role in the development of varicocele testis. Eur Urol 2007;51(5):1402–1410. doi:10.1016/j.eururo.2006.11.010. [Google Scholar] [PubMed] [CrossRef]
12. Gat Y, Joshua S, Vuk‐Pavlović S, Goren M. Paying the price for standing tall: fluid mechanics of prostate pathology. Prostate 2020;80(15):1297–1303. doi:10.1002/pros.24051. [Google Scholar] [PubMed] [CrossRef]
13. Xu Z, Li J, Li S, Huang H. Combined supine and standing imaging for varicocele: an improved diagnostic approach. J Vis Exp 2024;(213):10.3791/66370. doi:10.3791/66370. [Google Scholar] [PubMed] [CrossRef]
14. Li R, Liu J, Li Y, Wang Q. Effect of somatometric parameters on the prevalence and severity of varicocele: a systematic review and meta-analysis. Reprod Biol Endocrinol 2021;19(1):11. doi:10.1186/s12958-021-00695-3. [Google Scholar] [PubMed] [CrossRef]
15. Gorenstein A, Katz S, Schiller M. Surgical treatment of varicocele in children. Isr J Med Sci 1988;24(3):172–174. [Google Scholar] [PubMed]
16. Lund L, Ernst E, Sørensen HT, Oxlund H. Biomechanical properties of normal and varicose internal spermatic veins. Scand J Urol Nephrol 1998;32(1):47–50. doi:10.1080/003655998750014684. [Google Scholar] [PubMed] [CrossRef]
17. Zavattaro M, Ceruti C, Motta G et al. Treating varicocele in 2018: current knowledge and treatment options. J Endocrinol Invest 2018;41(12):1365–1375. doi:10.1007/s40618-018-0952-7. [Google Scholar] [PubMed] [CrossRef]
18. Shekhawat D, Chaiyamoon A, Cardona JJ et al. Renal vein valves: a prevalence, microanatomical and histological study. Surg Radiol Anat 2024;46(4):535–541. doi:10.1007/s00276-024-03330-z. [Google Scholar] [PubMed] [CrossRef]
19. Braedel HU, Steffens J, Ziegler M, Polsky MS, Platt ML. A possible ontogenic etiology for idiopathic left varicocele. J Urol 1994;151(1):62–66. doi:10.1016/s0022-5347(17)34872-3. [Google Scholar] [PubMed] [CrossRef]
20. Liang S. Clinical research on the diagnosis and management of different types of primary varicocele [dissertation]. Guangzhou, China: Southern Medical University; 2018. (In Chinese). [Google Scholar]
21. Cao ZZ. Study on the relationship between varicocele and nutcracker phenomenon [dissertation]. Changsha, China: Central South University; 2013. (In Chinese). [Google Scholar]
22. Yasumoto R, Asakawa M, Kakinoki K, et al. Clinical studies of varicocele. 2: radiographic examination and measurement of spermatic vein pressure in varicocele patients. Hinyokika Kiyo 1988;34(2):312–315. (In Japanese). [Google Scholar] [PubMed]
23. Fan CJ, Zhang CD, Ye FT. Analysis of lower limb varicose veins from the perspective of hemodynamics. J Vasc Endovasc Surg 2023;9(7):890–896. (In Chinese). doi:10.19418/j.cnki.issn2096-0646.2023.07.26. [Google Scholar] [CrossRef]
24. Wu WC, Cao J. Research progress on etiology of varicocele. Guowai Yixue Miniao Xitong Fence 1987;6:1–2. (In Chinese). [Google Scholar]
25. Shafik A, Khalil AM, Saleh M. The fasciomuscular tube of the spermatic cord: a study of its surgical anatomy and relation to varicocele: a new concept for the pathogenesis of varicocele. Br J Urol 1972;44(2):147–151. doi:10.1111/j.1464-410x.1972.tb10057.x. [Google Scholar] [PubMed] [CrossRef]
26. Liu Q, Zhang X, Zhou F, Xi X, Lian S, Lian Q. Comparing endovascular and surgical treatments for varicocele: a systematic review and meta-analysis. J Vasc Interv Radiol 2022;33(7):834–840.e2. doi:10.1016/j.jvir.2022.03.013. [Google Scholar] [PubMed] [CrossRef]
27. Sayfan J, Halevy A, Shperber Y, Oland J. The role of the spermatic cord layers in the development of varicoceles. J Urol 1985;133(2):223–224. doi:10.1016/s0022-5347(17)48890-2. [Google Scholar] [PubMed] [CrossRef]
28. Ozkurt H, Cenker MM, Bas N, Erturk SM, Basak M. Measurement of the distance and angle between the aorta and superior mesenteric artery: normal values in different BMI categories. Surg Radiol Anat 2007;29(7):595–599. doi:10.1007/s00276-007-0238-9. [Google Scholar] [PubMed] [CrossRef]
29. Wei YX. The relationship between hemodynamic changes of left renal and varicocele [dissertation]. Nanning, China: Guangxi Medical University; 2008. (In Chinese). [Google Scholar]
30. Yang Y-R, Ling W-W, Shi S-H, Li Y-Z, Zhou J-J. Ultrasound features of posterior nutcracker syndrome: case series and literature analysis. Quant Imaging Med Surg 2022;12(10):4984–4989. doi:10.21037/qims-22-166. [Google Scholar] [PubMed] [CrossRef]
31. Liu WY, Jiang M, Wang RT et al. Correlation between left varicocele and nutcracker syndrome. Jujie Shoushuxue Zazhi 2021;30(11):980–982. (In Chinese). doi:10.11659/jjssx.07e021012. [Google Scholar] [CrossRef]
32. Qin SY, Hu HF, Zhang HC et al. Progress in the treatment of nutcracker syndrome. Sichuan Med J 2023;44(10):1104–1107. (In Chinese). doi:10.16252/j.cnki.issn1004-0501-2023.10.020. [Google Scholar] [CrossRef]
33. Arslan H, Etlik Ö, Ceylan K, Temizoz O, Harman M, Kavan M. Incidence of retro-aortic left renal vein and its relationship with varicocele. Eur Radiol 2005;15(8):1717–1720. doi:10.1007/s00330-004-2563-2. [Google Scholar] [PubMed] [CrossRef]
34. Wang FG. The anatomic study of the left spermatic vein on 40 cases of adult males [dissertation]. Changchun, China: Jilin University; 2014. (In Chinese). [Google Scholar]
35. Cai Y. Study on the relationship between varicocele and the prostatic venous plexus by color doppler flow imaging [dissertation]. Nanning, China: Guangxi Medical University; 2012. (In Chinese). [Google Scholar]
36. Yang B, Yang Y, Liu Y et al. Molecular characteristics of varicocele: integration of whole-exome and transcriptome sequencing. Fertil Steril 2021;115(2):363–372. doi:10.1016/j.fertnstert.2020.08.004. [Google Scholar] [PubMed] [CrossRef]
37. Zhu B, Yin L, Zhang JY. Glutathione S-transferase polymorphisms in varicocele patients: a meta-analysis. Genet Mol Res 2015;14(4):18851–18858. doi:10.4238/2015.december.28.34. [Google Scholar] [PubMed] [CrossRef]
38. Kahraman CY, Tasdemir S, Sahin I et al. The relationship between endothelial nitric oxide synthase gene (NOS3) polymorphisms, NOS3 expression, and varicocele. Genet Test Mol Biomark 2016;20(4):191–196. doi:10.1089/gtmb.2015.0294. [Google Scholar] [PubMed] [CrossRef]
39. Heidari MM, Khatami M, Talebi AR. The POLG gene polymorphism in Iranian varicocele-associated infertility patients. Iran J Basic Med Sci 2012;15(2):739–744. [Google Scholar] [PubMed]
40. Karthikeyan V, Vickram AS, Manian R. Estimation of varicocele associated human ARG2 and NOS1 proteins and computational analysis on the effect of its nsSNPs. Int J Biol Macromol 2020;164:735–747. doi:10.1016/j.ijbiomac.2020.07.159. [Google Scholar] [PubMed] [CrossRef]
41. Bromberg Y, Rost B. Correlating protein function and stability through the analysis of single amino acid substitutions. BMC Bioinform 2009;10(Suppl 8):S8. doi:10.1186/1471-2105-10-s8-s8. [Google Scholar] [PubMed] [CrossRef]
42. Kume T. Foxc2 transcription factor: a newly described regulator of angiogenesis. Trends Cardiovasc Med 2008;18(6):224–228. doi:10.1016/j.tcm.2008.11.003. [Google Scholar] [PubMed] [CrossRef]
43. Lyons O, Saha P, Seet C et al. Human venous valve disease caused by mutations in FOXC2 and GJC2. J Exp Med 2017;214(8):2437–2452. doi:10.1084/jem.20160875. [Google Scholar] [PubMed] [CrossRef]
44. Surendran S, Ramegowda KS, Suresh A et al. Arterialization and anomalous vein wall remodeling in varicose veins is associated with upregulated FoxC2-Dll4 pathway. Lab Invest 2016;96(4):399–408. doi:10.1038/labinvest.2015.167. [Google Scholar] [PubMed] [CrossRef]
45. Zhang C, Li H, Guo X. FOXC2-AS1 regulates phenotypic transition, proliferation and migration of human great saphenous vein smooth muscle cells. Biol Res 2019;52(1):59. doi:10.1186/s40659-019-0266-z. [Google Scholar] [CrossRef]
46. Hollingsworth SJ, Powell GL, Barker SGE, Cooper DG. Primary varicose veins: altered transcription of VEGF and its receptors (KDR, flt-1, soluble flt-1) with sapheno-femoral junction incompetence. Eur J Vasc Endovasc Surg 2004;27(3):259–268. doi:10.1016/j.ejvs.2003.12.015. [Google Scholar] [PubMed] [CrossRef]
47. Rao L, Babu A, Kanakavalli M et al. Chromosomal abnormalities and Y chromosome microdeletions in infertile men with varicocele and idiopathic infertility of South Indian origin. J Androl 2004;25(1):147–153. doi:10.1002/j.1939-4640.2004.tb02770.x. [Google Scholar] [PubMed] [CrossRef]
48. Song NH, Wu HF, Zhang W et al. Screening for Y chromosome microdeletions in idiopathic and nonidiopathic infertile men with varicocele and cryptorchidism. Chin Med J Engl 2005;118(17):1462–1467. [Google Scholar] [PubMed]
49. Kumanov P, Robeva RN, Tomova A. Adolescent varicocele: who is at risk? Pediatrics 2008;121(1):e53–e57. doi:10.1542/peds.2007-0340. [Google Scholar] [PubMed] [CrossRef]
50. Di Luigi L, Gentile V, Pigozzi F et al. Physical activity as a possible aggravating factor for athletes with varicocele: impact on the semen profile. Hum Reprod 2001;16(6):1180–1184. doi:10.1093/humrep/16.6.1180. [Google Scholar] [PubMed] [CrossRef]
51. Hariri A, Abbarh S, Ameen A et al. Prevalence and awareness of varicocele among athletes in Riyadh, Saudi Arabia: impact on the semen profile. Res Rep Urol 2019;11:21–27. doi:10.2147/rru.s195582. [Google Scholar] [PubMed] [CrossRef]
52. Rigano E, Santoro G, Impellizzeri P et al. Varicocele and sport in the adolescent age: preliminary report on the effects of physical training. J Endocrinol Invest 2004;27(2):130–132. doi:10.1007/bf03346257. [Google Scholar] [PubMed] [CrossRef]
53. Di Luigi L, Romanelli F, Sgrò P, Lenzi A. Andrological aspects of physical exercise and sport medicine. Endocrine 2012;42(2):278–284. doi:10.1007/s12020-012-9655-6. [Google Scholar] [PubMed] [CrossRef]
54. Hayden RP, Tanrikut C. Testosterone and varicocele. Urol Clin North Am 2016;43(2):223–232. doi:10.1016/j.ucl.2016.01.009. [Google Scholar] [PubMed] [CrossRef]
55. Atar M, Söylemez H, Oguz F et al. Effects of acute exercise on the diameter of the spermatic vein, and duration of reflux in patients with varicocele. Scand J Urol 2013;47(3):206–210. doi:10.3109/00365599.2012.727467. [Google Scholar] [PubMed] [CrossRef]
56. Esteves SC, Cocuzza MS. The obesity paradox in varicocele—is the protective effect real? Int Braz J Urol 2020;47(1):20–22. doi:10.1590/s1677-5538.ibju.2019.0210.1. [Google Scholar] [PubMed] [CrossRef]
57. Chanc Walters R, Marguet CG, Crain DS. Lower prevalence of varicoceles in obese patients found on routine scrotal ultrasound. J Urol 2012;187(2):599–601. doi:10.1016/j.juro.2011.10.039. [Google Scholar] [PubMed] [CrossRef]
58. Cobb WS, Burns JM, Kercher KW et al. Normal intraabdominal pressure in healthy adults. J Surg Res 2005;129(2):231–235. doi:10.1016/j.jss.2005.06.015. [Google Scholar] [PubMed] [CrossRef]
59. Gu P, Xu A. Interplay between adipose tissue and blood vessels in obesity and vascular dysfunction. Rev Endocr Metab Disord 2013;14(1):49–58. doi:10.1007/s11154-012-9230-8. [Google Scholar] [PubMed] [CrossRef]
60. Akbay E, Çayan S, Doruk E, Duce MN, Bozlu M. The prevalence of varicocele and varicocele-related testicular atrophy in Turkish children and adolescents. BJU Int 2000;86(4):490–493. doi:10.1046/j.1464-410x.2000.00735.x. [Google Scholar] [PubMed] [CrossRef]
61. Sandlow J. Pathogenesis and treatment of varicoceles. BMJ 2004;328(7446):967–968. doi:10.1136/bmj.328.7446.967. [Google Scholar] [PubMed] [CrossRef]
62. Sasson DC, Kashanian JA. Varicoceles. JAMA 2020;323(21):2210. doi:10.1001/jama.2020.0397. [Google Scholar] [PubMed] [CrossRef]
63. Fretz PC, Sandlow JI. Varicocele: current concepts in pathophysiology, diagnosis, and treatment. Urol Clin North Am 2002;29(4):921–937. doi:10.1016/s0094-0143(02)00075-7. [Google Scholar] [PubMed] [CrossRef]
64. Xu S. The expression and clinical significance of Nrf2 and HO-1 in varicocele [dissertation]. Shijiazhuang, China: Hebei Medical University; 2023. (In Chinese). 10.27111/d.cnki.ghyku.2023.000659. [Google Scholar] [CrossRef]
65. Erdogan O, Ok F, Carkci S. What is the role of pre-operative blood parameters in forecasting varicocelectomy success? Andrology 2021;9(3):916–921. doi:10.1111/andr.12976. [Google Scholar] [PubMed] [CrossRef]
66. Gyftopoulos K, Chondrogianni C, Papadaki H. Increased expression of endothelin-1 and its receptors in varicocele: an immunohistochemical study. Fertil Steril 2011;95(8):2554–2556.e62. doi:10.1016/j.fertnstert.2011.04.062. [Google Scholar] [PubMed] [CrossRef]
67. Mohammed A, Chinegwundoh F. Testicular varicocele: an overview. Urol Int 2009;82(4):373–379. doi:10.1159/000218523. [Google Scholar] [PubMed] [CrossRef]
68. Kaltsas A, Zikopoulos A, Markou E et al. Proteomics and metabolomics in varicocele-associated male infertility: advancing precision diagnostics and therapy. J Clin Med 2024;13(23):7390. doi:10.3390/jcm13237390. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF
 Downloads
Downloads
 Citation Tools
Citation Tools