 Open Access
Open Access
CASE REPORT
Unusual metastatic patterns of urologic malignancies: a case series and literature review
1 Department of Urology, Izmir Katip Celebi University Ataturk Training and Research Hospital, Izmir, 35360, Turkey
2 Department of Urology, Izmir Katip Celebi University, Karabaglar, Izmir, 35620, Turkey
* Corresponding Author: Kursad Donmez. Email:
Canadian Journal of Urology 2026, 33(1), 211-219. https://doi.org/10.32604/cju.2025.068294
Received 25 May 2025; Accepted 22 September 2025; Issue published 28 February 2026
Abstract
Background: Urologic cancers typically metastasize to common organs, while spread to sites such as the heart, hand, cranial bones, penis, or tonsil is exceedingly rare. The clinical relevance and prognostic impact of these unusual metastases remain unclear. This case series highlights six exceptional cases, providing unique insights to support awareness and management. Case Description: We retrospectively analyzed six cases of patients diagnosed with urologic cancers who developed metastases at uncommon sites. Each case was evaluated based on clinical presentation, imaging findings, histopathological confirmation, treatment course, and outcomes. Relevant literature was reviewed to contextualize findings. Among the six cases, metastatic involvement was identified in atypical locations including the right atrium (testicular cancer), hand (bladder cancer), perineum and penis (sarcomatoid bladder cancer), cranial bones (prostate cancer), penis (rectal cancer), and tonsillar region (renal pelvis rhabdomyosarcoma). Timely imaging and histopathological confirmation guided personalized treatment strategies, primarily involving surgical interventions. Despite aggressive treatments, the prognosis remained poor in most cases, reflecting the aggressive nature of these metastases. Conclusion: Rare metastases from urologic malignancies, although uncommon, have significant implications on patient management. Early detection through advanced imaging and aggressive multimodal interventions are pivotal for improving outcomes. Awareness of such atypical metastatic patterns is essential for clinicians managing urologic cancers.Keywords
Supplementary Material
Supplementary Material FileUrologic cancers (prostate, kidney, bladder, testicular, etc.) usually spread to common sites such as regional lymph nodes, lungs, liver, and bones.1 However, metastases to unusual sites do occur, albeit very infrequently. For example, metastases to the hand bones (acrometastases) are exceedingly rare—on the order of only ~0.1% of all skeletal metastases.2 Despite their rarity, such atypical metastases have been reported in patients with advanced genitourinary cancers, often as late manifestations of widespread disease.3 Rare metastatic involvement has been documented in many organ systems. For instance, brain metastases from prostate cancer are extremely uncommon—studies indicate that fewer than 1% of metastatic prostate cancer patients develop brain lesions.4 Similarly, cutaneous metastases from urologic tumors are very rare.5 These examples illustrate that although uncommon, virtually any organ can be a target of metastasis in advanced urologic malignancies.
Given these low incidences, one might question the need for aggressive surveillance imaging. However, improved cancer therapies and prolonged survival mean patients live long enough to develop atypical metastases, and indeed the frequency of such rare metastases is rising with time.6
As patients live longer with advanced urologic malignancies, the likelihood of encountering unusual metastatic presentations grows. Timely recognition of such metastases is essential, as they may significantly alter management strategies and prognostic outlooks. This case series aims to present six distinct cases of urologic cancers with rare metastatic localizations and to review the relevant literature to support early recognition and tailored intervention in such scenarios. This study reported according to the CARE checklist.
Case 1: Testicular cancer with cardiac metastasis
A 19-year-old male presented with a painless right scrotal mass persisting for three weeks. Physical examination revealed a firm, non-tender, 10 cm intratesticular mass. β-Human Chorionic Gonadotropin (β-HCG) was elevated (290 IU/L), while alpha-fetoprotein (AFP) and lactate dehydrogenase (LDH) were within normal ranges. Scrotal Doppler ultrasonography and contrast-enhanced thoracoabdominal CT revealed a 10 cm right testicular mass, an 8 cm para-aortic lymphadenopathy, a 3 cm pulmonary metastasis, and posterior mediastinal lymphadenopathy (Figure 1).
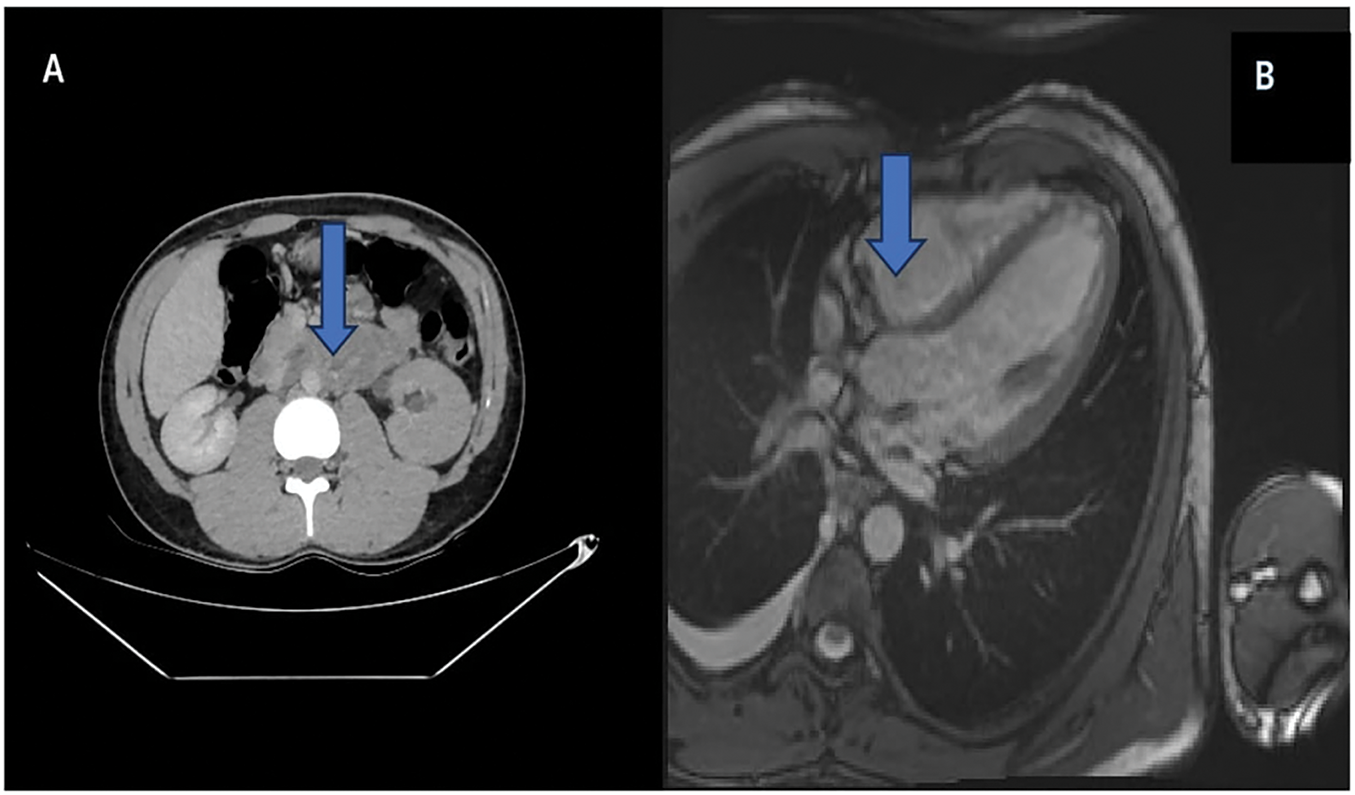
FIGURE 1. Metastasis of testicular tumor. (A) Para-aortic lymph node metastasis of testicular tumor on contrast-enhanced abdominal-pelvic computed tomography (CT). (B) Right atrium posterior metastasis of testicular tumor on contrast-enhanced cardiac magnetic resonance imaging (MRI) (shown with blue arrow, respectively)
The patient underwent right inguinal orchiectomy, and pathology showed 60% teratoma, 30% yolk sac tumor, and 10% embryonal carcinoma. Pre-chemotherapy transthoracic-echo-cardiography revealed a 2 cm hypoechoic mass in the right atrium. Despite three cycles of bleomycin, etoposide, and cisplatin (BEP) chemotherapy normalizing tumor markers, cardiac magnetic resonance imaging (MRI) confirmed a 4 × 3 cm metastatic lesion in the right atrium (Figure 1).
Metastatic spread to the heart is an uncommon occurrence in solid tumors, reported in approximately 1%–1.5% of autopsy series, and is usually asymptomatic. In testicular germ cell tumors, intracardiac metastasis is exceedingly rare, with only isolated cases described in the literature.7 Such occurrences usually indicate disseminated end-stage disease (98.5% of those cases also had metastases elsewhere).8 Renal cell carcinoma can invade the heart via the IVC, but true metastatic implants in the heart muscle or chambers from genitourinary primaries are exceedingly rare (isolated case reports). Intracardiac metastases are found typically in advanced stage cancer patients, often incidentally on imaging or autopsy, since they are frequently asymptomatic.9 Similarly, urothelial carcinoma can very rarely seed the heart. Pouwels et al. reported a case of urothelial cancer with cardiac metastasis found on screening echocardiography.10
We should mention that the right atrium metastasis in a testicular GCT is an extraordinarily unusual event, emphasizing its rarity by noting only isolated cases exist in literature.
These cases illustrate that while routine cardiac screening in all cancer patients is not indicated, early cardiac imaging should be considered in those with suggestive symptoms or extensive disease.10 Echocardiography can detect intracardiac masses or associated findings like unexplained pericardial effusion.11 However, echocardiography may miss small or intracardiac lesions in some cases. We opted for advanced imaging modalities for more definitive assessment. Cardiac MRI provides excellent tissue characterization of cardiac masses and played a significant role in our testicular case (confirming the presence of an RA tumor and its size). Cardiac CT can also delineate masses or tumor thrombi. Additionally, positron emission tomography-computed tomography (PET-CT) can help identify metabolically active cardiac lesions in the context of widespread disease.8 We can cite that echocardiography, while useful as an initial tool, often needs to be followed by MRI or CT for precise localization and characterization of the lesion.
Early detection of an intracardiac metastasis is crucial, as timely intervention (surgical resection or chemotherapy) may prevent life-threatening cardiac complications.7,12 Unfortunately, prognosis remains guarded– most patients with cardiac metastases from solid tumors have advanced disease and poor outcomes.10 In summary, if intracardiac metastasis is clinically suspected (e.g., patient with known cancer develops cardiac symptoms or has extensive metastatic disease), one should perform echocardiography first, and then confirm with cardiac MRI or CT for a comprehensive evaluation.
Case 2: Bladder cancer with metastasis to hand
A 63-year-old male with left flank pain and gross hematuria was diagnosed via CT urography with a left distal ureter and renal pelvis tumor. He underwent radical nephroureterectomy with pathology confirming a high-grade T1 urothelial carcinoma with negative margins.
Following incomplete resection of recurrent bladder tumors, he underwent radical cystoprostatectomy and extended lymphadenectomy. At three-year follow-up, PET-CT revealed a metastatic lesion at the right 2th proximal interphalangeal (PIP) joint. Surgical excision confirmed metastatic urothelial carcinoma (Figure 2).
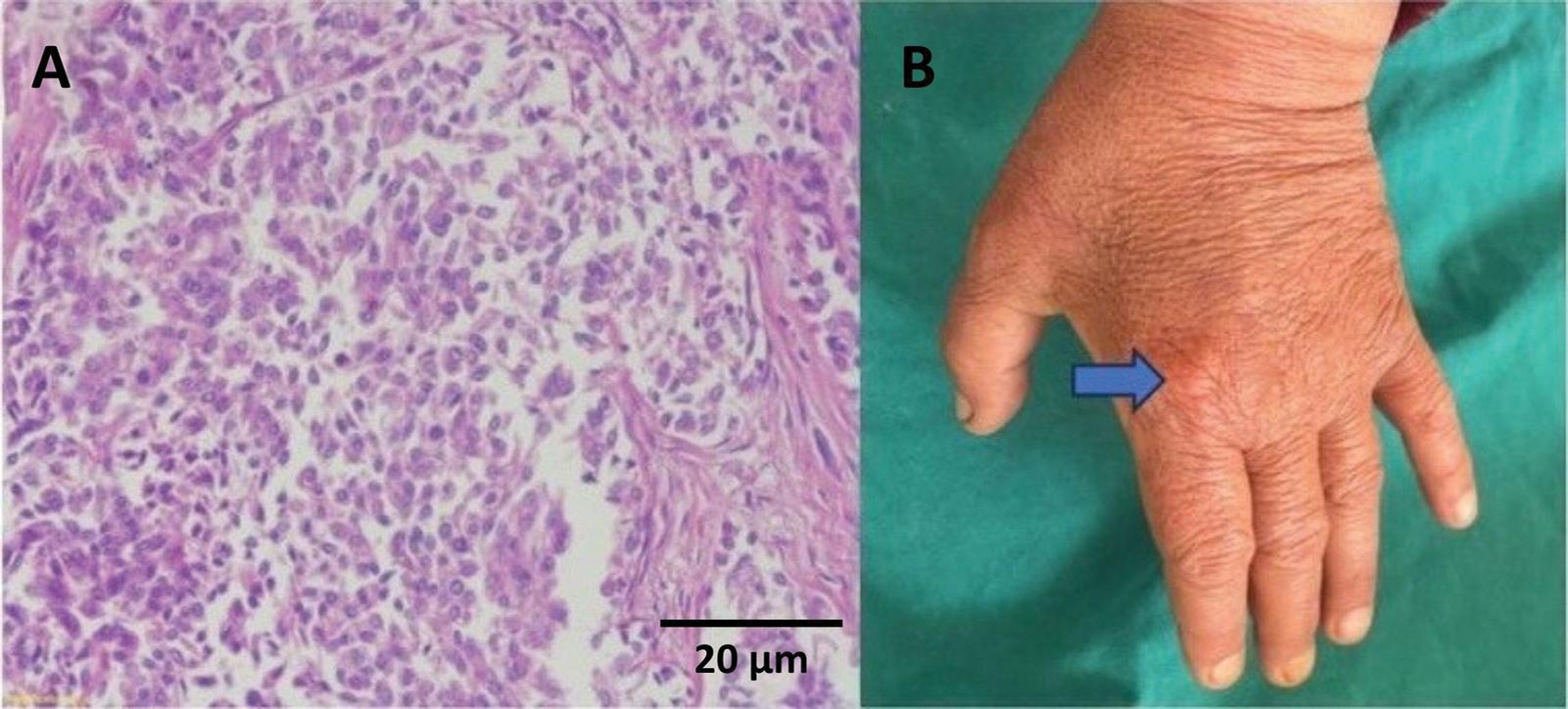
FIGURE 2. Histological and macroscopic images of finger metastasis. (A) Tumor cells with eosinophilic cytoplasm and thin fibrous septa in between, showing a poorly differentiated appearance (hematoxylin-eosin [H&E] staining, 20×) finger metastasis. (B) Macroscopic view of finger metastasis second proximal interphalangeal (PIP) (shown with blue arrow)
A bladder urothelial carcinoma metastasizing to the hand, represents an acrometastasis to the distal extremity. Such occurrences are exceedingly rare, comprising only ~0.1% of all bone metastases.13,14 In a systematic review of hand metastases, the majority originated from lung cancers, with genitourinary tumors accounting for a smaller fraction.6 Acrometastases most often affect the distal phalanges (about 21% of cases), followed by metacarpals (16%) and proximal phalanges (7%).13,14 Metastatic carcinoma to the digits of the hand has been noted only in isolated reports and is often a harbinger of advanced disease.13 Once diagnosed, surgical excision (metastasectomy) can provide symptom relief and local disease control, although the development of an acrometastasis generally signifies aggressive systemic disease.
Case 3: Bladder cancer with perineal and penile metastasis
Two cases in our series involved metastatic tumors to the penis—one from a sarcomatoid variant of bladder carcinoma and another from rectal adenocarcinoma. A 58-year-old male with hematuria was diagnosed with a 5 cm right bladder wall tumor. CT revealed metastatic lymph nodes but no distant metastases. After transurethral resection of bladder tumor (TURBT) and pathology revealing high-grade T2 tumor, the patient underwent radical cystoprostatectomy.
One year postoperatively, a 4 × 5 cm perineal mass infiltrating the penile root developed. Excision and total penectomy pathology revealed sarcomatoid urothelial carcinoma (Figure 3).
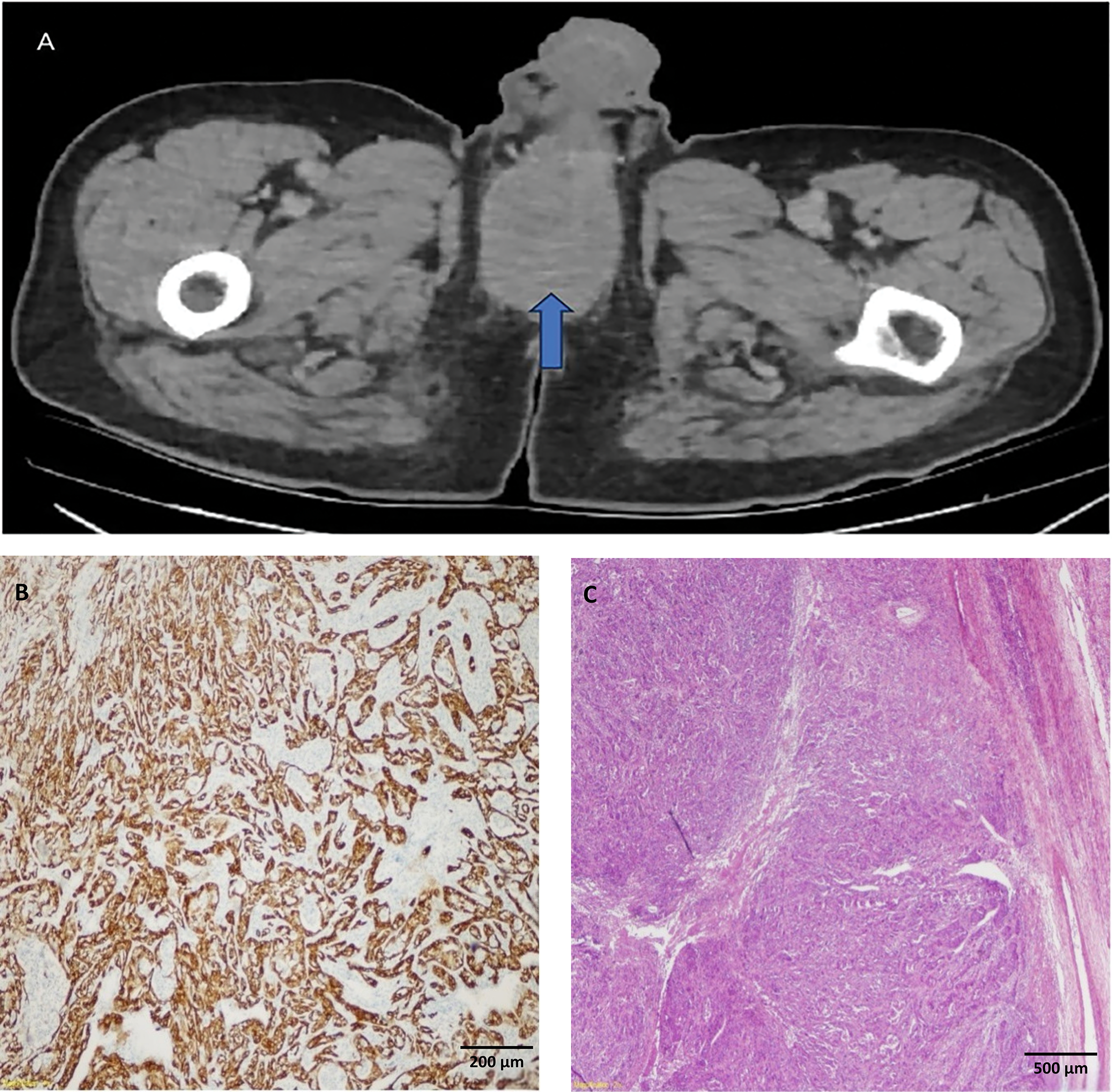
FIGURE 3. Imaginf and histological examination of penile metastasis in a postoperative cystoprostatectomy patient. (A) Computed tomography (CT) showing penile metastasis in the the postoperative first year following radical cystoprostatectomy (indicated by the blue arrow). (B) Cytokeratin 7 immunohistochemical staining of penile infiltration. (C) Hematoxylin-eosin [H&E] staining of penile Infiltration in 2× magnification
Metastases to the penis are extremely infrequent despite the organ’s rich vascularization and interconnected venous plexus. Fewer than 400 cases of penile metastases from all primaries have been reported in the literature and most arise from genitourinary primaries (prostate and bladder being the most common).1
Metastatic spread from bladder cancer to the penis typically occurs via retrograde venous route through the pelvic venous plexus (Santorini’s plexus) or lymphatic channels. When the bladder tumor has sarcomatoid (carcinosarcoma) differentiation, as in our patient, the disease is known to behave very aggressively. Sarcomatoid bladder carcinomas frequently present at advanced stage with a propensity for widespread and unusual metastases. Our case concurs with prior reports of early penile involvement in metastatic bladder cancer, including cases presenting as malignant priapism or penile mass after cystectomy.7
Case 4: Prostate cancer with cranial metastasis
A 72-year-old male under androgen deprivation therapy for high-risk prostate cancer (Gleason 4+5, prostate specific antigen [PSA] 260 ng/mL) presented with body pain and lower urinary symptoms. Despite castration-level testosterone, PSA remained elevated. Prostate spesific membrane antigen (PSMA) revealed multiple skeletal metastases and a suspicious frontal lobe lesion. Cranial MRI confirmed a parieto-occipital bone lesion suggestive of metastasis (Figure 4).
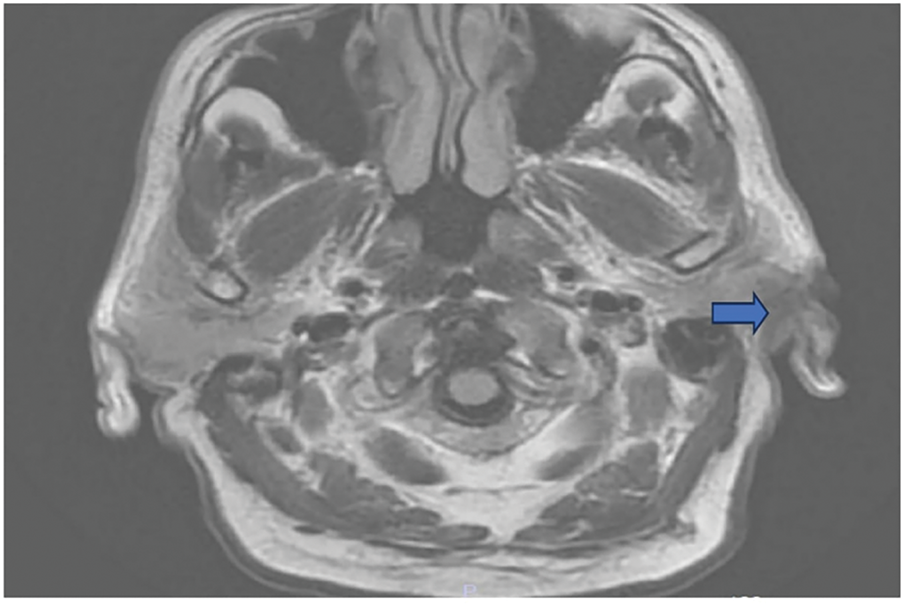
FIGURE 4. Metastatic lesion in the T1 axial bone window reported as compatible with prostate cancer observed in the left parieto-occipital bone (shown with blue arrow)
Prostate cancer commonly metastasizes to bone, but involvement of the skull or cranial bones is uncommon. Brain or skull metastases from prostate cancer, while rare (<2% incidence in clinical series), do occur and have been reported even when serum PSA is not markedly elevated.15 For instance, isolated brain metastases have been documented in prostate cancer patients who had normal PSA levels at presentation.16 In our case, a castration-resistant prostate cancer patient developed a metastasis to the parieto-occipital bone, detected on PSMA and MRI. This scenario, although rare, has been observed in advanced prostate cancer—often manifesting as skull base metastases with cranial nerve palsies or as dural metastases mimicking meningiomas or subdural hematomas.17
Indeed, in many reported cases, a prostate metastasis to the skull was initially mistaken for a benign neurologic lesion on imaging. Multimodal imaging and biopsy are therefore critical to distinguish a metastatic cranial lesion from primary brain tumors or benign conditions.17
The development of brain or skull metastasis in prostate cancer usually signifies widely metastatic disease with a poor prognosis—median survival on the order of months in most series.16 Our case underscores that, although rare, intracranial metastasis should remain in the differential diagnosis for prostate cancer patients with new neurologic symptoms or atypical skull lesions. Awareness of this possibility can lead to prompt neurosurgical consultation and targeted therapy (for example, focal radiation or hormonal therapy adjustments).
Case 5: Rectal cancer metastasis to penis
A 74-year-old male with a history of rectal adenocarcinoma treated with chemoradiotherapy and colostomy presented with scrotal discharge. He developed Fournier gangrene requiring multiple debridements. Due to extensive necrosis, total penectomy was performed.
Histopathology revealed poorly differentiated adenocarcinoma consistent with rectal origin (Figure 5).
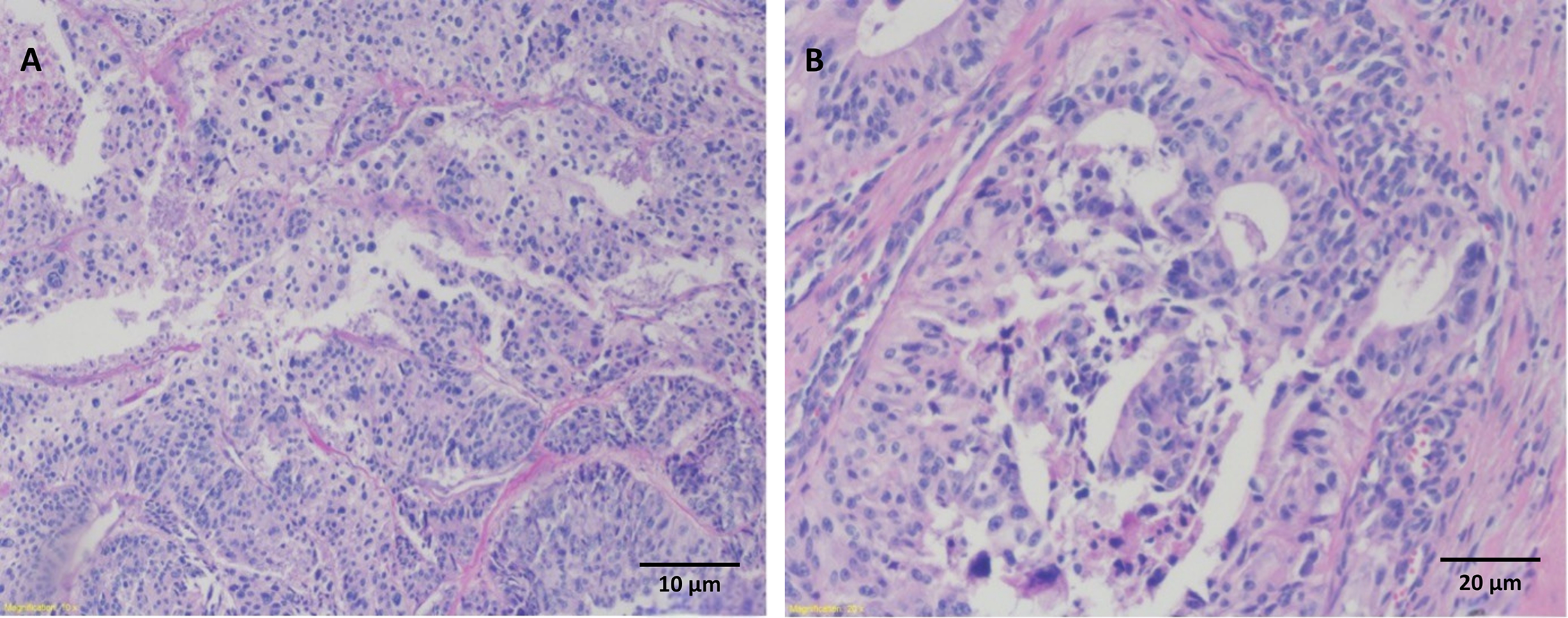
FIGURE 5. Histopathological analysis of colon adenocarcinoma penile metastasis. (A) Same tumor sample, area of poorly differentiated pleomorphic tumor cells, colon adenocarcinoma penile metastasis (Hematoxylin-eosin [H&E] staining, 10×). (B) Poorly differentiated areas as well as selectable gland structures (H&E staining, 20×)
Metastatic spread to the penis from a colorectal primary is even rarer. Fewer than ~80 cases of rectal carcinoma metastasizing to the penis have been documented to date.17 In our case, the rectal cancer patient’s penile metastasis was discovered during surgery for Fournier’s gangrene, illustrating that such metastases can sometimes be obscured by or present as complications like infection.
Literature suggests that penile metastases from rectal adenocarcinoma usually indicate diffuse dissemination and carry a dismal prognosis.18 Regardless of the primary tumor, prognosis of penile metastasis is very poor, with an average survival of only ~3–4 months after diagnosis. Aggressive treatment (combining surgery with systemic therapy) has yielded survival up to 9–12 months in select cases, but cure is exceedingly rare.19 Surgical intervention (such as total or partial penectomy) remains the mainstay for managing penile metastases when limited to the penis, as it can alleviate symptoms and prevent local complications. Our cases reinforce that any unexplained penile lesion in a patient with a pelvic malignancy (or even a history of one) should raise suspicion of metastasis after more common causes are excluded.
Case 6: Renal pelvis embryonal rhabdomyosarcoma with tonsillar metastasis
Perhaps the most extraordinary finding in our series was the metastasis to the palatine tonsil in a patient with an embryonal rhabdomyosarcoma of the renal pelvis. A 62-year-old woman presented with left flank pain and hematuria. Imaging revealed an 11 cm mass arising from the left renal pelvis infiltrating the parenchyma. She underwent radical nephroureterectomy. Histopathology confirmed embryonal rhabdomyosarcoma. One month postoperatively, PET-CT detected metabolic activity in the right tonsillar region. Tru-cut biopsy confirmed metastatic embryonal carcinoma (Figure 6).
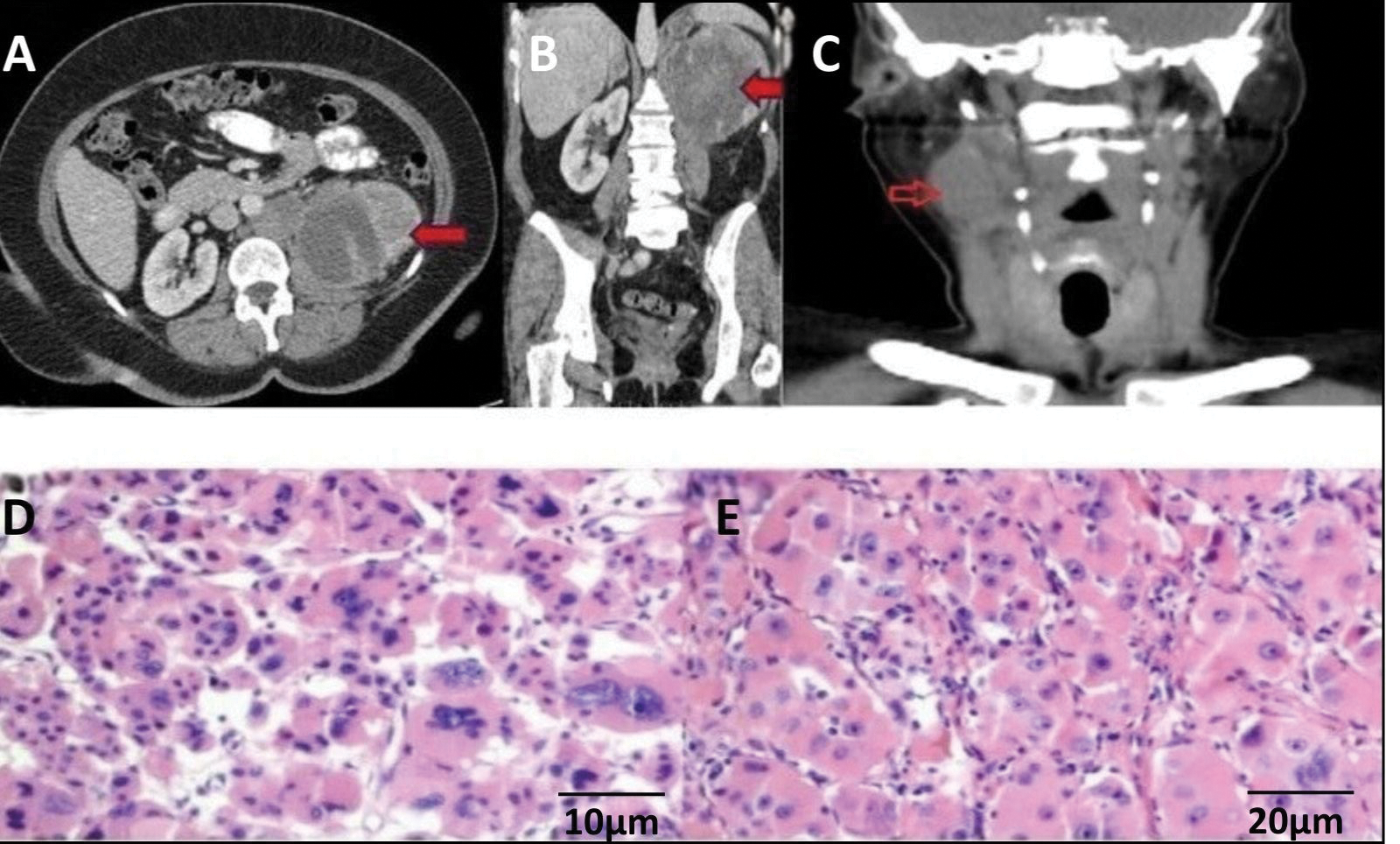
FIGURE 6. Imaging and histopathological features of renal and tonsillar metastasis. (A) Axial CT image of left renal mass (indicated by the red arrow). (B) Coronal CT image of left renal mass (indicated by the red arrow). (C) CT image showing tonsillar metastasis (indicated by the red arrow). (D) Extensive areas of necrosis in pathological examination (Hematoxylin-eosin [H&E] staining, ×10). (E) Areas of spindle cells in fascicular arrangement (H&E staining, ×20)
Metastatic tumors to the palatine tonsil are exceedingly uncommon; even among common cancers, spread to the tonsil is rare (for instance, only about 20 cases from lung cancer and 10 cases from colorectal cancer have ever been reported).20,21 To our knowledge, metastasis of a genitourinary primary to the tonsil has only been described in the most exceptional circumstances, if at all. The mechanism of spread to head–neck lymphoid tissue like the tonsil is not well understood—hematogenous dissemination or retrograde lymphatic flow has been postulated in reported cases.20
In our patient, the tonsillar metastasis was detected merely one month after nephroureterectomy for the primary tumor, highlighting the highly aggressive nature of sarcomatous malignancies. Rhabdomyosarcomas in adults are known to behave aggressively, and the appearance of a metastasis in an unusual site so early in the postoperative period suggests fulminant systemic spread. This finding reinforces the need for thorough evaluation of any new head and neck masses in patients with a history of urologic cancer, even if such metastases are exceedingly rare. Given the rarity, there is no established treatment protocol for a tonsillar metastasis of a urologic tumor; management would be palliative, focusing on local control (e.g., tonsillectomy or radiation) and systemic chemotherapy. Unfortunately, like other sarcomatous metastases, the prognosis is grave in this scenario.
These cases carry important clinical lessons. First, clinicians must maintain a broad differential when evaluating unusual lesions, particularly in patients with a history of urologic malignancy. Atypical presentations—such as a cardiac mass in a young man, a finger lesion in a bladder cancer survivor, or a penile lesion in an elderly patient—should prompt consideration of metastatic disease from a known or even occult primary tumor. Notably, in some patients, such rare metastases may be the initial manifestation of an otherwise asymptomatic malignancy. For example, there are instances where a cardiac tumor was the first indication of testicular cancer or a phalangeal lesion led to the diagnosis of an aggressive bladder carcinoma.22 Obtaining tissue diagnosis in such cases is crucial to avoid misdiagnosis and inappropriate treatment.
Second, early recognition of these rare metastatic sites can directly influence therapeutic decisions. Timely detection enables a tailored, multidisciplinary approach—ranging from surgical resection of isolated metastases (e.g., penectomy or finger amputation) to site-specific radiation or systemic therapy adjustments. In our series, selected patients received metastasis-directed interventions that, while not curative, alleviated symptoms and potentially extended survival. Literature suggests that local treatment of solitary or symptomatic metastases, when feasible, may offer palliation or modest survival benefit in appropriately selected cases.22 However, the presence of these metastases generally indicates widespread disease, and thus necessitates realistic discussions with patients about prognosis and goals of care.
Mechanistically, such atypical metastatic spread can be attributed to several anatomical and biological factors. One critical pathway is the Batson’s vertebral venous plexus—a valveless venous system connecting pelvic organs to the vertebral column and cranial vault.23 This network facilitates retrograde tumor cell migration, especially in prostate cancer, allowing skeletal or cranial metastases without pulmonary involvement.
Additionally, disrupted or aberrant lymphatic drainage—whether due to prior surgery, tumor burden, or congenital variation—can redirect tumor cells to unexpected nodal regions, such as supraclavicular or inguinal lymph nodes in renal or bladder cancers.24 Finally, aggressive histologic subtypes, such as sarcomatoid renal cell carcinoma or neuroendocrine prostate cancer, are prone to early and widespread dissemination, including to rare locations, and may do so with little or no elevation in tumor markers such as PSA.25 These mechanisms collectively explain how urologic malignancies, although typically following predictable routes, may occasionally metastasize to highly unusual sites.
This case series provides valuable insights by documenting a spectrum of exceptionally rare metastatic sites of urologic malignancies, most of which are scarcely reported in the literature. The systematic description of clinical presentation, imaging modalities, histopathological confirmation, and therapeutic strategies enhances its educational value for clinicians who may encounter such atypical manifestations in practice. Furthermore, the inclusion of multiple tumor types and metastatic localizations allows for a broader perspective on the biological heterogeneity and dissemination pathways of urologic cancers.
Nevertheless, several limitations must be acknowledged. The retrospective and single-center design inherently limits the generalizability of the findings. The small sample size, although consistent with the rarity of such cases, restricts the ability to draw definitive conclusions regarding prognostic factors or treatment efficacy. In addition, the lack of long-term follow-up data in some patients prevents a comprehensive assessment of survival outcomes and therapeutic durability. Finally, the descriptive nature of the study precludes statistical analysis, which could have provided stronger evidence for clinical decision-making.
Unusual metastases from urologic malignancies are rare but clinically significant, affecting diagnostic and therapeutic strategies. A high index of suspicion and appropriate imaging are critical for early detection and tailored management.
This vigilant approach can lead to earlier diagnosis, appropriate multidisciplinary treatment, and potentially improved patient outcomes even in the face of these rare and formidable metastatic patterns.
Acknowledgement
The authors declare that there is no need for an acknowledgment section in this manuscript.
Funding Statement
The authors received no specific funding for this study.
Author Contributions
The authors confirm contribution to the paper as follows: study conception and design: Kursad Donmez, Yigit Akin; data collection: Enis Mert Yorulmaz, Enes Candemir; analysis and interpretation of results: Kursad Donmez, Enis Mert Yorulmaz; draft manuscript preparation: Enis Mert Yorulmaz, Enes Candemir; language and editorial revision: Serkan Ozcan; statistical analysis and critical review: Osman Kose; scientific content revision: Sacit Nuri Gorgel; supervision and hypothesis development: Kursad Donmez, Yigit Akin. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request. Due to the retrospective and patient-identifiable nature of the data, public sharing is restricted to protect patient confidentiality.
Ethics Approval
This retrospective case series was conducted in accordance with institutional ethical policies and the principles of the Declaration of Helsinki. Since all data were de-identified and derived from routine clinical care, the study was granted an exemption from formal Institutional Review Board (IRB) approval. The exemption was officially confirmed by the Ethics Committee of İzmir Katip Celebi University Ataturk Training and Research Hospital.
Informed Consent
Informed consent was obtained from all patients or their legal representatives for anonymized publication.
Conflicts of Interest
The authors declare no conflicts of interest to report regarding the present study.
Supplementary Materials: The supplementary material is available online at https://www.techscience.com/doi/10.32604/cju.2025.068294/s1.
References
1. Rajeswaran K, Muzio K, Briones J et al. Prostate cancer brain metastasis: review of a rare complication with limited treatment options and poor prognosis. J Clin Med 2022;11(14):4165. doi:10.3390/jcm11144165. [Google Scholar] [PubMed] [CrossRef]
2. Lander RD, O’Donnell MJ. A case of metastatic prostate cancer to the distal phalanx. Hand 2022;17(3):NP1–NP4. doi:10.1177/15589447211028922. [Google Scholar] [PubMed] [CrossRef]
3. Hayden RJ, Sullivan LG, Jebson PJ. The hand in metastatic disease and acral manifestations of paraneoplastic syndromes. Hand Clin 2004;20(3):335–343. doi:10.1016/j.hcl.2004.03.010. [Google Scholar] [PubMed] [CrossRef]
4. Lungulescu C, Ştefan R, Ştefania D et al. Solitary brain metastasis of prostate adenocarcinoma with normal PSA and PALB2 mutation. J Med Radiat On 2021;1:43–50. doi:10.53011/jmro.2021.01.04. [Google Scholar] [CrossRef]
5. Bazzi N, Jaber S, Saad WA, Al Hassan S, Dbouk S, Alhousseini A. Cutaneous metastasis of bladder cancer: a systematic review. Cureus 2024;16(11):e74439. doi:10.7759/cureus.74439. [Google Scholar] [PubMed] [CrossRef]
6. Umana GE, Scalia G, Palmisciano P et al. Acrometastases to the hand: a systematic review. Medicina 2021;57(9):950. doi:10.3390/medicina57090950. [Google Scholar] [PubMed] [CrossRef]
7. Rojas-Cadena M, Izquierdo-Condoy JS, Arias-Intriago M et al. Unusual presentation of metastatic testicular mixed germ cell tumor with intracardiac extension: a case report. J Clin Med 2025;14(10):3564. doi:10.3390/jcm141023564. [Google Scholar] [CrossRef]
8. Grech N, Camilleri W, Borg A. Multiple cardiac metastases from urothelial carcinoma case report. Egyptian Heart J 2022;74(1):28. doi:10.1186/s43044-022-00264-y. [Google Scholar] [PubMed] [CrossRef]
9. Casey III B, Bahekar A, Patel D, Guddeti R, Sinnapunayagam S. Cardiac metastasis from renal cell carcinoma. Cureus 2023;15:e36439. doi:10.7759/cureus.36439. [Google Scholar] [PubMed] [CrossRef]
10. Pouwels S, Fitski W, Magro M, van der Linden-Foolen Y, Stavast J, Burger DHC. Multiple cardiac complications due to sepsis and cardiac metastasis of a sarcomatoid urothelial carcinoma of the bladder: a case report. Int J Surg Case Rep 2025;133:111514. doi:10.1016/j.ijscr.2025.111514. [Google Scholar] [PubMed] [CrossRef]
11. Reynen K, Köckeritz U, Strasser RH. Metastases to the heart. Anna Oncol 2004;15(3):375–381. doi:10.1093/annonc/mdh086. [Google Scholar] [PubMed] [CrossRef]
12. Shields AM, Pomplun S, Deshpande R, Whitaker DC. Right ventricular metastasis of transitional cell carcinoma of the renal pelvis: successful single stage surgical treatment. Interac Cardiovasc Thorac Surg 2011;12(2):297–300. doi:10.1510/icvts.2010.256230. [Google Scholar] [PubMed] [CrossRef]
13. Assem H, Broeke N, Coleman L, Gotto G, Bismar T. Highly aggressive urothelial carcinoma of the bladder presenting with solitary metastasis to the phalanx. Can Urol Assoc J 2020;14(12):E607–E610. doi:10.5489/cuaj.6263. [Google Scholar] [PubMed] [CrossRef]
14. Dow T, Davis C, ElAbd R, Lalonde D, Williams J. Cancer metastases to the hand: a systematic review and meta-analysis. Hand 2024;19(6):865–874. doi:10.1177/15589447231153175. [Google Scholar] [PubMed] [CrossRef]
15. Fuentes F, Hattab WF, Xiong W et al. Evolution of structural rearrangements in prostate cancer intracranial metastases. npj Precis Oncol 2023;7(1):35. doi:10.1038/s41698-023-00435-3. [Google Scholar] [PubMed] [CrossRef]
16. Nassar DN, Sheikh R. Late metastasis of rectal adenocarcinoma to the penis. BMJ Case Rep 2024;17(1):e262775. doi:10.1136/bcr-2024-262775. [Google Scholar] [PubMed] [CrossRef]
17. Liu KT, Chang YC, Chang JL. Incidental finding of metastatic prostatic adenocarcinoma of frontotemporal bone presenting as subdual hematoma: a case report and review of literature. Ann Med Surg 2012;73:103142. doi:10.1016/j.amsu.2021.103142. [Google Scholar] [PubMed] [CrossRef]
18. Sun JJ, Zhang SY, Tian JJ, Jin BY. Penile metastasis from rectal carcinoma: a case report. World J Clinic Cases 2022;10(19):6609–6616. doi:10.12998/wjcc.v10.i19.6609. [Google Scholar] [PubMed] [CrossRef]
19. Kumar N, Bhattacharyya T, Mandal AK, Gupta N, Rajwanshi A, Kumar R. Penile metastasis secondary to bladder cancer: a report of two cases. Indian J Palliat Care 2014;20(1):57–60. doi:10.4103/0973-1075.125565. [Google Scholar] [PubMed] [CrossRef]
20. Morales Medina G, Vallés Rodríguez C, Rettig Infante I, Bayona Romero M, Oyarzún Bahamonde E, Enterría González Á. Palatine tonsil metastatic colon adenocarcinoma: case report. Braz J Otorhinolaryngol 2022;88(Suppl.4):S212–S214. doi:10.1016/j.bjorl.2021.07.012. [Google Scholar] [PubMed] [CrossRef]
21. Yücel L, Gökmen MF, Büyükatalay Zç, Dizbay Sak S, Beton S. Tonsil metastasis of lung adenocarcinoma: a rare case. Çukurova Med J 2020;45(3):1250–1252. doi:10.17826/cumj.730086. [Google Scholar] [CrossRef]
22. Marghich O, Dkhissi Y, Alila M, El Bouhaddouti H. Penile metastases of rectal adenocarcinoma after abdominoperineal resection: a case report. J Med Case Rep 2019;13(1):233. doi:10.1186/s13256-019-2147-z. [Google Scholar] [PubMed] [CrossRef]
23. Capito PR, Wang H, Brem H, Ahn HS, Bryan RN. Magnetic resonance imaging diagnosis of an intracranial metastasis of adenocarcinoma of the prostate: case report. Md Med J 1991;40(2):113–115. [Google Scholar] [PubMed]
24. Perry L, Schwartz J, Slotman G, Al Ustwani O, Kulkarni N. Occult renal cell carcinoma presenting as a palpable supraclavicular virchow’s node. Am Surg 2019;85:342–344. doi:10.1177/000313481908500711. [Google Scholar] [CrossRef]
25. Dong B, Zhan H, Luan T, Wang J. The role and controversy of pelvic lymph node dissection in prostate cancer treatment: a focused review. World J Surg Oncol 2024;22(1):68. doi:10.1186/s12957-024-03344-2. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools