 Open Access
Open Access
ARTICLE
Association of tertiary lymphoid structures and benign lymphoepithelial lesions in NIH-category IV prostatitis: pathophysiological correlations
1 Department of Pathology, Jossigny Hospital, Jossigny, 75000, France
2 Department of General and Clinical Pathology, Faculty of Medicine, Medical University of Plovdiv, Plovdiv, 4000, Bulgaria
3 Department of Analytical Chemistry and Computer Chemistry, University of Plovdiv “Paisii Hilendarski”, Plovdiv, 4000, Bulgaria
4 Department of Medical Biochemistry, Faculty of Pharmacy, Medical University of Plovdiv, 15A Vasil Aprilov Blvd., Plovdiv, 4002, Bulgaria
5 Department of Pathology, Military Medical Academy, Sofia, 1606, Bulgaria
6 Department of Medical Biology, Medical University of Plovdiv, Plovdiv, 4000, Bulgaria
7 Technological Center for Emergency Medicine, Plovdiv, 4000, Bulgaria
* Corresponding Author: Maria Koleva. Email:
(This article belongs to the Special Issue: Prostate Cancer: Biomarkers, Diagnosis and Treatment)
Canadian Journal of Urology 2026, 33(1), 125-134. https://doi.org/10.32604/cju.2025.068575
Received 01 June 2025; Accepted 12 November 2025; Issue published 28 February 2026
Abstract
Background: Chronic inflammation is closely associated with the most common and socially significant prostate conditions, including benign prostatic hyperplasia (BPH), prostate cancer (PCa), and prostatitis syndromes. NIH-category IV prostatitis (histologic prostatitis, HP) is defined as asymptomatic chronic inflammation of the prostate. The presence of lymphoid follicles, referred to as tertiary lymphoid structures (TLSs), along with benign lympho-epithelial lesions (BLELs), is among the key histological indicators of immune inflammation and can be assessed relatively easily. This study aimed to quantitatively assess TLSs and BLELs, as well as their relationship with the severity of HP. Methods: We investigated TLSs and BLELs in 110 prostatic specimens, including inflammatory and normal tissues, within the context of common prostate pathologies such as BPH and PCa. HP was graded as low-grade (LG) or high-grade (HG) based on the severity of inflammation. Results: TLSs were observed in 51 out of 110 cases (46.4%), while BLELs were identified in 78 cases (70.44%). Both TLSs and BLELs co-occurred in 45 cases (40.9%). Statistical analysis revealed a significant correlation between the presence of TLSs, BLELs (individually or combined), and HG-HP (p < 0.001). Conclusions: This study is the first to quantitatively evaluate the immunopathologic patterns in the inflamed human prostate by analyzing the presence and co-occurrence of TLSs and BLELs. Their formation, likely triggered by antigenic stimuli and external factors, indicates a chronic inflammatory microenvironment. The strong association between TLSs, BLELs, and HG-HP underscores their potential role in HP aggressiveness. These findings suggest that TLSs and BLELs may be crucial contributors to the pathophysiology and morphogenesis of NIH-category IV prostatitis. Furthermore, TLS/BLEL formation may represent a hallmark of tissue autoimmunity, reflecting the immune or autoimmune phase of this prostatitis subtype.Keywords
Chronic inflammation (CI) is a well-established feature of several common prostate pathologies, including benign prostatic hyperplasia (BPH), prostate cancer (PCa), and prostatitis syndromes.1–3 Prostatitis of NIH-category IV, or histologic prostatitis (HP, or lymphocytic prostatitis, or asymptomatic chronic nonspecific prostatitis) is an incidental pathological finding, which appears to be the most common type of prostatic CI in men without clinical symptoms related to urinary tract infection or pelvic pain.4,5 Although CI has been associated with all major socially significant prostatic diseases, the etiology, pathogenesis, and clinical significance of HP remain poorly understood to this day. One increasingly recognized hypothesis in recent years proposes that, in its advanced stage, HP evolves through a mechanism resembling an immune or autoimmune-mediated tissue response.3,6
Tertiary lymphoid structures (TLSs) and benign lymphoepithelial lesions (BLELs) represent key histological components of immune-mediated inflammation and are relatively accessible for both quantitative and qualitative morphological assessment. TLSs are lymphoid aggregates that form in non-lymphoid tissues in the context of CI, autoimmunity, or cancer, and they share characteristics typically confined to secondary lymphoid organs.7–10 Their prognostic value remains under investigation. TLSs in the human prostate have been known for a long time, described in the form of lymph follicles in the context of HP.5,11 In the only study so far examining prostatic TLSs, they have been investigated mainly qualitatively in patients with spontaneous prostate cancer regression.12 To our knowledge, the presence and role of TLSs in inflammatory prostate has not been explored in detail.
The human prostate is endowed with a lympho-epithelial component, which is responsible for the genitourinary mucosal immunity, and so the concept of the prostate-associated lymphoid tissue (PALT) was created.11 Its immunological and microarchitectural features are comparable with those of the other secondary lymphoid organs (like tonsils), especially regarding the lymphoid component, while prostatic epithelial changes are poorly explored.11–13 Our previous research describes BLELs within the double-layered ductal epithelium of the prostate and shows strong membranous PD-L1 expression in cases of high-grade HP.7
To date, no studies have investigated the co-occurrence of TLSs and BLELs in the context of both normal and chronically inflamed prostate tissue. We evaluated quantitatively the presence and the association of TLSs and BLELs immediately adjacent to HP in a large cohort of patients with different types of surgical and autopsy-derived prostate specimens. They were studied in the context of patients’ age, intensity of HP, and immune status (both normal and compromised). Our aim was to clarify the occurrence and pathological role of TLSs and BLELs in HP and to explore their potential clinical relevance.
A retrospective record review was performed on a large volume of prostatic specimens of all types: radical prostatectomies (RP), transurethral resection of the prostate (TURP), adenomectomies (AD), and autopsies, at the Departments of General and Clinical Pathology and Forensic Medicine of St. George University Hospital of Plovdiv; the General Hospital of Lovetch, Bulgaria; Rothschild Hospital, Paris; and the Grand Hôpital de l’Est Francilien, Jossigny, France. A total of 110 prostatic specimens were examined in the basic group of patients: 30 RP, 37 TURP/AD, and 43 autopsies. Prostatic needle biopsies were not examined because of the small amount of prostatic tissue available for histological analysis.
The study was approved by the local Ethics Committees of the hospitals. Informed consent was obtained from all patients and the study was approved by the Ethics Committees at the St. George University Hospital of Plovdiv, Bulgaria (Protocol No 6/08/06/2023), the General Hospital of Lovetch, Bulgaria (Protocol No 1/03/04/2008), Rothschild Hospital, Paris, France (Protocol No 1/16/06/1994), and the Grand Hôpital de l’Est Francilien, Jossigny, France (Protocol No 4/30/05/2008).
Inclusion and exclusion criteria
During the primary review of tissue samples and clinical records of the basic group of patients, the following selection criteria were set: optimal tissue preservation with few or no autolysis according to light microscopic evaluation; histologically present tissue inflammatory infiltrate; no clinical and histological data for systemic granulomatous disease or granulomatous prostatitis; no clinical record of urological disease before death (autopsy patients); only cases without any signs of prostatitis or urinary symptoms (autopsy patients); neither radiation treatment of the genital area nor chemotherapy within the last months before surgery or death.
Selection of the control group
The usage of control material while examining the inflammatory pathology of the prostate gland is a difficult and challenging task due to the frequent presence of CI and the strict clinical indications of prostate sampling.6 For this reason, the selected autopsy prostates served as appropriate control samples for the present immunomorphological assessment of TLSs and BLELs. The control group comprised histologically negative cases for HP, BPH, prostatic intraepithelial neoplasia (PIN), and PCa. These included both medico-legal autopsy specimens and prostates obtained from relatively young individuals. In the current investigation, we examine 3 control groups of autopsy prostates: (1) 6 prostates of newborns and children in prepuberty with age from 1 day to 13 years (mean 2.79 years or 33.5 months); (2) 19 cases of adult prostates, in which the prostate tissue demonstrated normal histological structure without significant inflammatory changes, BPH, PIN, and PCa, with age ranging from 23 to 81 years (mean 44.89 years). The autopsy prostates studied included 16 medico-legal cases of young men who died in traffic accidents with no documented illness; (3) 17 autopsy prostates of men who died from acquired immunodeficiency syndrome (AIDS) in the period before the introduction of highly active antiretroviral treatments (1992–1995), whose ages ranged from 29 to 49 years (mean 38.88 years). The cause of death in the control groups of the autopsy patients was not related to the genitourinary system.
No morphological evidence of prostatic lymphoma was found in any of the examined cases, including those from both the study and control groups.
All specimens were routinely fixed in 10% buffered formalin and embedded in paraffin for histological evaluation. Standard 4-μm-thick sections were cut from paraffin blocks. Sections were stained with haematoxylin-eosin-safran (HES).
Whole-mounted organ sections of RP and between 3 to 15 blocks of TURP/PP specimens were studied. One to three whole-organ sections from each autopsy prostate were examined to view the three anatomic parts of the gland.14 Retrospectively, tissue sections from each case were independently observed by two pathologists (DD and MK). The quantitative morphologic and statistical analysis of HP was performed by the use of a simplified grading system, introduced by Delongchamps et al.,2 based on the classical HP-score by Nickel et al.:5 HP of low grade (LG-HP) and HP of high grade (HG-HP). Briefly, LG-HP is characterized by focal presence of small clusters of inflammatory mononuclear cells, predominantly located within the stroma, separated by extensive areas of non-inflamed tissue, or fewer than 100 inflammatory cells/mm2 (Figure 1A) and HG-HP begins in the moment, when multifocal and diffuse, both stromal and peri-glandular confluent sheets of inflammatory cells (>100 cells/mm2) (Figure 1B), are histologically observed in prostatic parenchyma.2,5 For cases with varying intensity of the inflammatory infiltrate, the highest grade was recorded. Because of a non-significant statistical association with either BPH or PCa, the areas of acute inflammation (the so-called acute prostatitis), were not investigated in this study.2
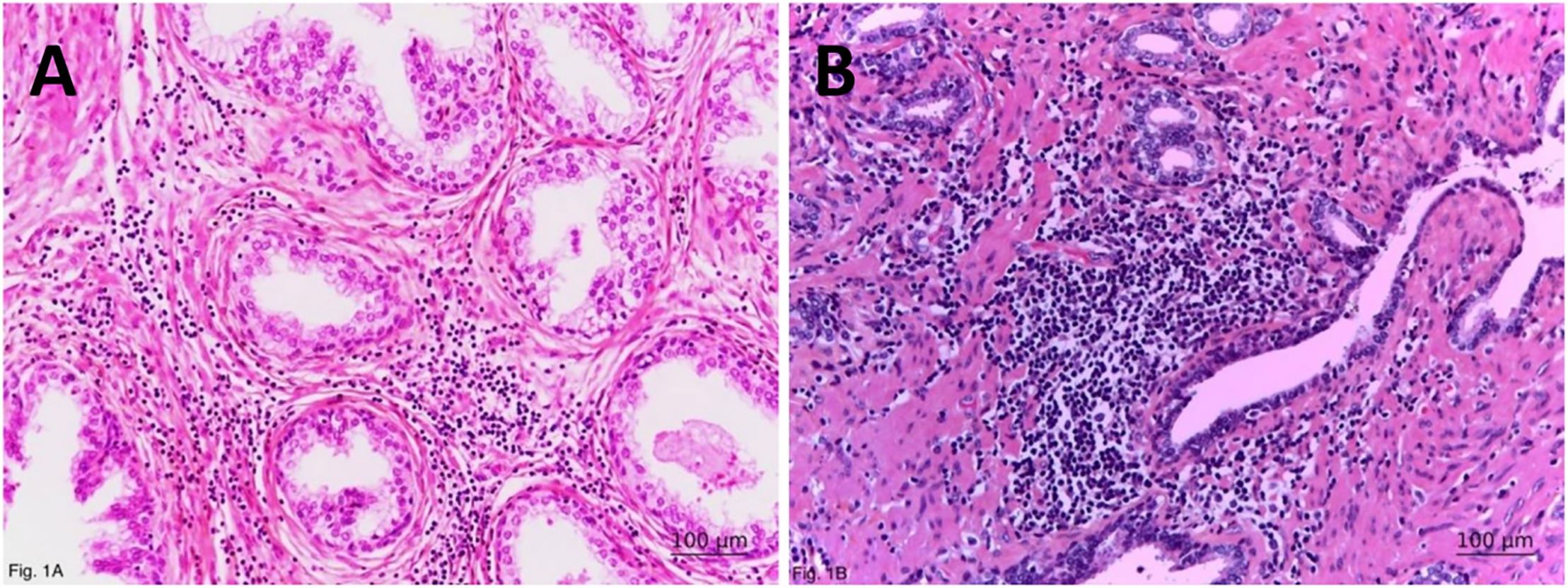
FIGURE 1. Morphological characteristics of histologic prostatitis. General view of (A) low- and (B) high-grade histologic prostatitis with prominent periductal inflammatory infiltrates, duct dilatation, and acinar atrophy (in the upper part of the picture). Haematoxylin-eosin-safran; ×200
Like secondary lymphoid organs, the prostatic TLSs are characterized by T- and B-cell zones, mature dendritic cells within the T-cell zone, and specialized blood vessels with features of high endothelial venules.10 Similarly to other locations,8,9 structures possessing all these three features have been termed as “classical” or “mature” TLSs,10,11 in contrast to “non-classical” TLSs—i.e., lymphoid aggregates that share some but not all of the three primary defining characteristics. In the present study, we quantitatively analyze only the “classical/mature” TLSs, representing lymphoid tissue aggregates with a germinal center, arranged around a central blood vessel lined with high endothelium (Figure 2A).
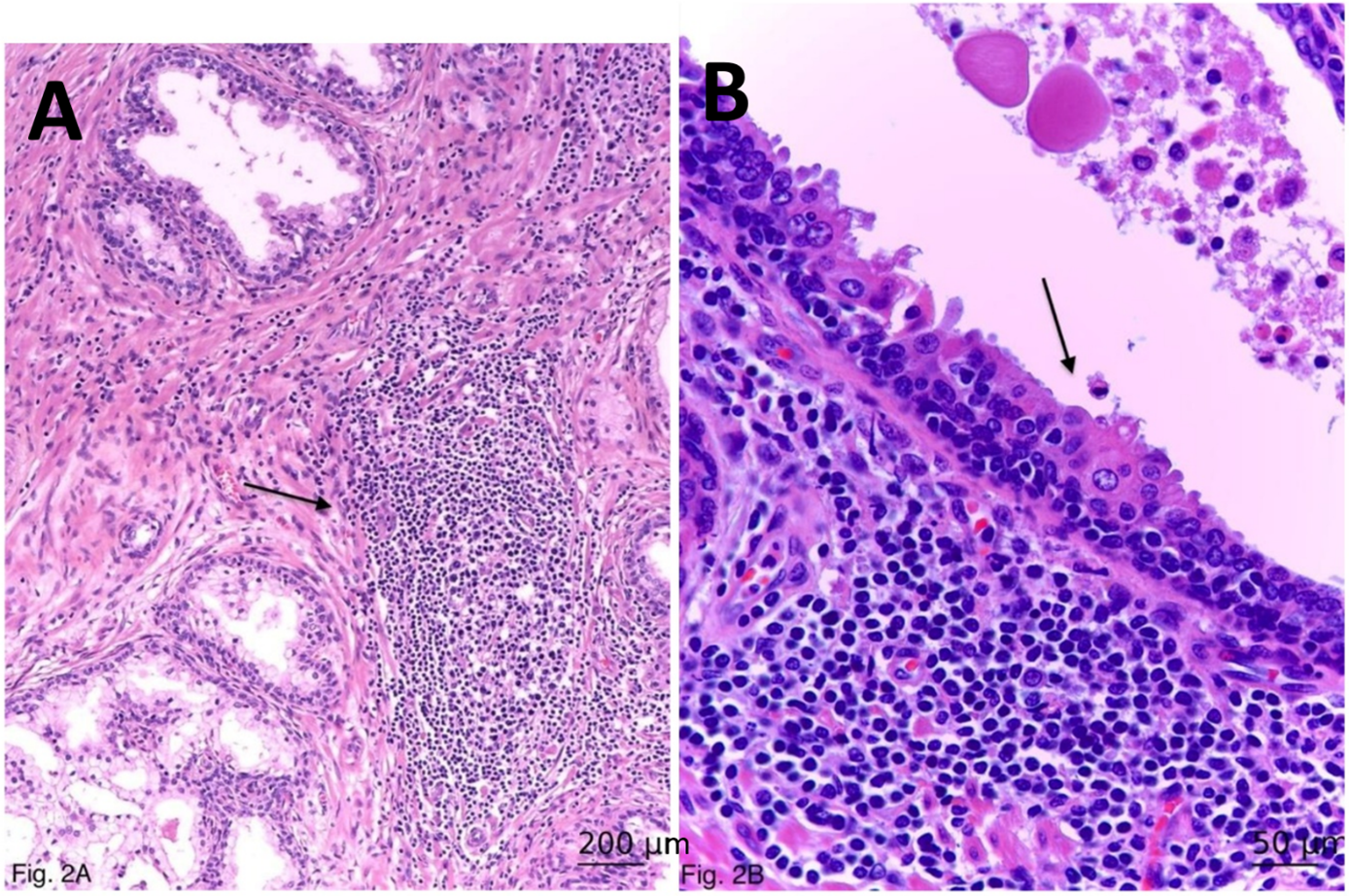
FIGURE 2. Immunomorphological characteristics of histologic prostatitis. (A) Periductal and interstitial tertiary lymphoid structure (lymph follicle; arrow), ×100. (B) A benign lymphoepithelial lesion (arrow), whose ductal epithelium is higher and eosinophilic with irregular and short apical blebs; presence of intra-epithelial lymphocytes. Haematoxylin-eosin-safran, ×400
Although the presence of lymph follicles (TLSs) is part of HG-HP,5 we examined TLSs separately due to the fact that they were not always observed in HG-HP. By definition, BLELs are aggregates of clusters of three or more lymphoid cells with distortion or destruction of the epithelium, and morphological changes within epithelial cells, including distinct eosinophilia.15 We assessed quantitatively the presence of BLELs in the prostate in so-called stage 0 BLELs, characterised by ductal epithelial eosinophilia and the presence of intraepithelial lymphocytes without ductal hyperplasia (Figure 2B).16
Patient age, as well as the presence of TLSs and BLELs in prostate specimens within each group, were evaluated using mean values and ranges. Fisher’s exact test was applied to distinguish associations among the categorical variables. A p-value < 0.05 was considered statistically significant. The statisticals softwares packagse and versions that are used are MS Excel 2016 and SPSS for Windows, v.23.
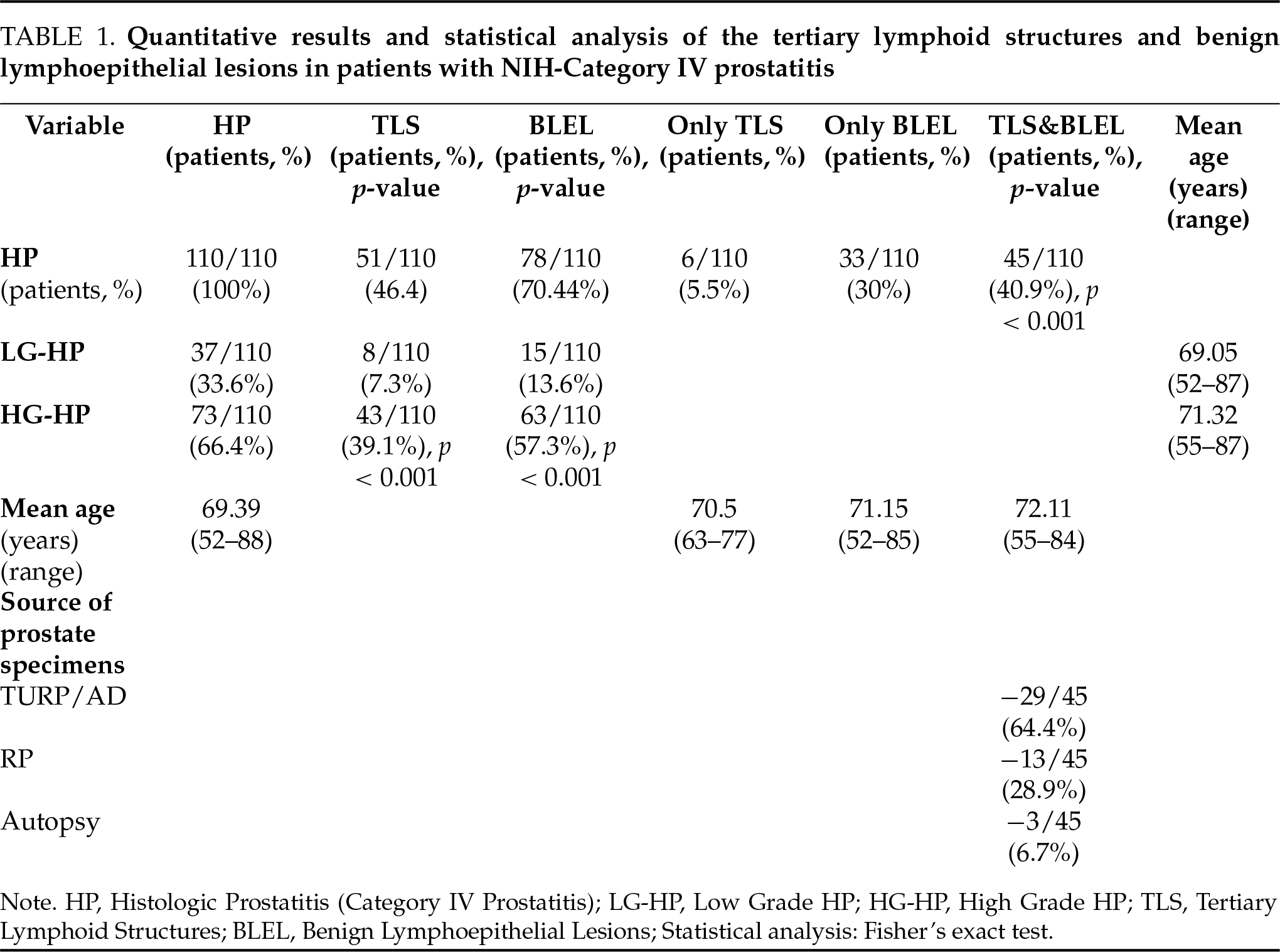
All examined prostates from the basic group showed more or less pronounced HP (Figure 1) in the peripheral (RP, biopsies, and autopsy) and transition (TURP/AD) zones of the prostate, as the mean age of the patients was 69.39 years (range, 52–88). LG-HP was identified in 37/110 cases (33.6%), and the mean age of the patients was 69.05 years (range, 52–88 years). HG-HP was detected in 73/110 cases (66.4%) with a mean age of the patients 71.32 years (range, 55–87 years).
In the control groups (1) and (3), HG-HP was not detected; only LG-HP was found in, respectively, 2/6 cases (33.33%) and 3/17 cases (17.64%). In 3 cases of the control group (3), prostate involvement was observed in the context of generalized opportunistic infections developed in the terminal stage of AIDS (Cytomegalovirus, Adenovirus, and Cryptococcus).
Histologically, the inflammatory infiltrate is equivalent to that in LG-HP. TLSs of “classical” type were detected in the investigated prostates with HP. They exhibited similarities in the structural organization with both peri-ductal and stromal localization (Figure 2A). In all cases where at least one “classical” TLS was present, multiple TLSs were also identified. TLSs were shown in 51/110 cases (46.4%) from the basic groups of patients. In none of the 3 control groups of patients, TLSs were detected. TLSs were combined in 8/110 cases with LG-HP (7.3%) and in 43/110 cases (39.1%) with HG-HP. The statistical analysis revealed a significant association of the presence of TLSs and HG-HP (p < 0.001, Fisher’s exact test).
BLELs were identified in 78/110 cases (70.44%) from the basic group of patients (Figure 2B). BLELs were combined in 15/110 cases with LG-HP (13.6%) and in 63/110 cases (57.3%) with HG-HP. The statistical analysis showed a significant association of the presence of BLEL and HG-HP (p < 0.001; Fisher’s exact test). BLELs were found in none of the 3 control patient groups, including the cases with opportunistic prostatitis in AIDS patients.
The majority of the TLSs surrounding prostatic ducts showed evidence of injury to the ductal epithelial cells and formation of BLEL (Figure 3). In the investigated 110 patients from the basic group with HP, a combination of both TLSs and BLELs was found in 45/110 cases (40.9%). In 6/110 patients (5.5%), TLSs were an independent finding, and in 33/110 prostates, only BLELs were noticed. In 26/110 of the patients with HP was not detected any of the lesions. The statistical analysis showed a significant association of the simultaneous presence of both TLSs and BLELs in HG inflammatory prostate (p < 0.001; Fisher’s exact test). The mean age of the patients with TLSs only was 70.5 years (ranging from 63 to 77 years). The mean age of the patients with BLELs only was 71.15 years (ranging from 52 to 85 years). The mean age of the patients with both TLSs and BLELs was 72.11 years (ranging from 55 to 84 years).
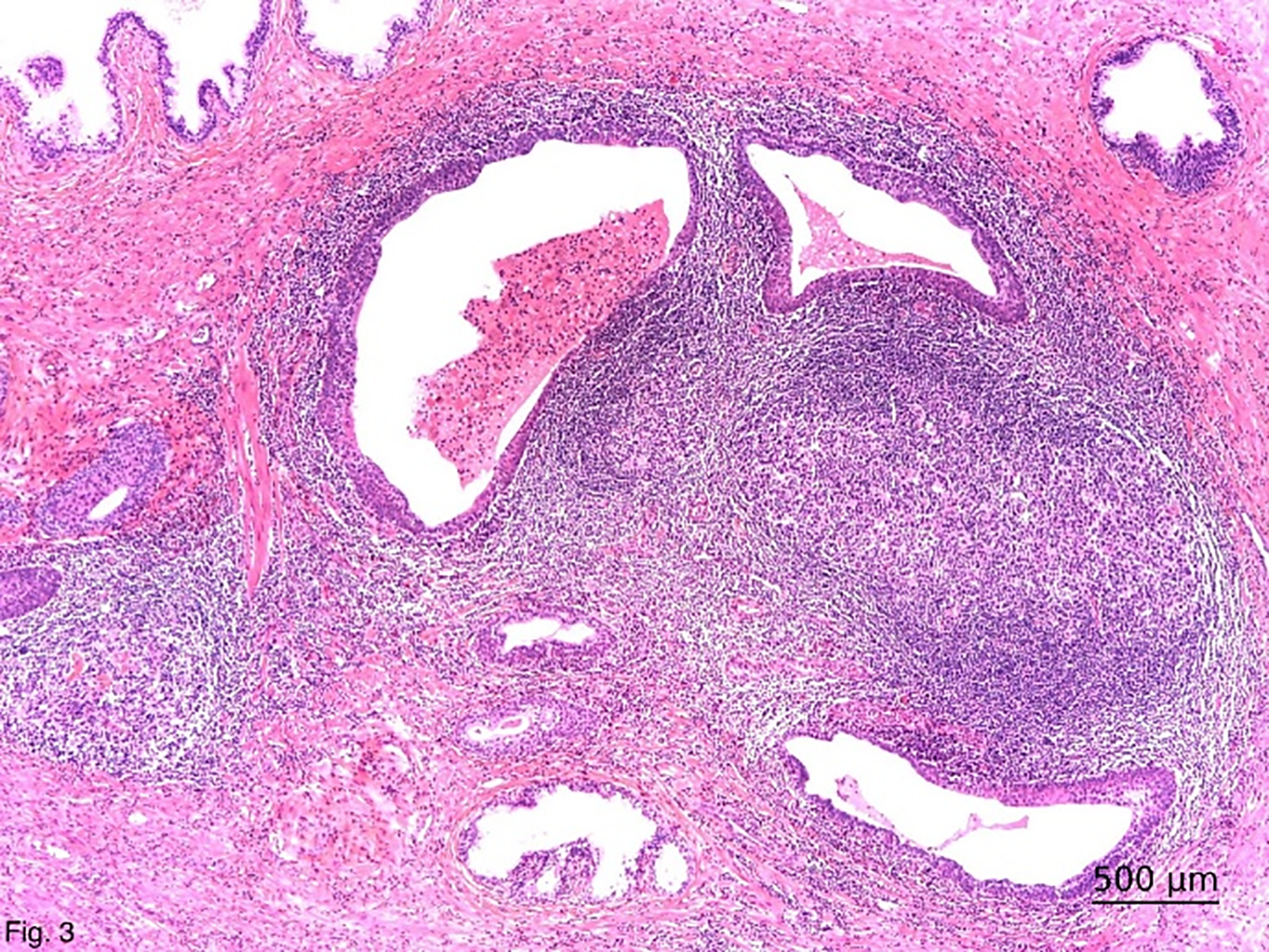
FIGURE 3. Close histologic association of periductal tertiary lymphoid structure and ductal benign lymphoepithelial lesions. Haematoxylin-eosin-safran; ×50
In regard to the source of inflammatory prostate specimens, the simultaneous combination of both TLSs and BLELs were observed most often in TURP/AD (29/45 cases or 64.4%), followed by RP (13/45 cases or 28.9%), and autopsies (3/45 cases or 6.7%).
The quantitative results and the statistical analysis are summarized in Table 1.
In contrast to the urologist, the pathologist definеs prostatitis as the presence of “more” inflammatory cells within the prostatic parenchyma.5 Although CI is a well-established feature of several common prostate pathologies, including prostatitis syndromes, BPH, and PCa,1–4 TLSs and BLELs—key histological features of immune-mediated inflammation, have remained underexplored in the human prostate.
Since the prostate is an organ with constant exposure to pathogens, an early response to such stimuli is essential for survival. However, the characteristics of the antigens that promote HP and TLSs, and BLELs formation are poorly understood so far.13 In the present study, we believe we are taking a step forward in clarifying the pathogenesis of HP. We show that the presence of prostatic TLS increases with the severity of CI: from low levels in LG-HP to significantly higher levels in HG-HP (p < 0.001), suggesting a link between TLS formation and HP intensity. TLSs form postnatally in non-lymphoid tissues in response to chronic antigenic stimulation.9,17 Unlike primary and secondary lymphoid organs, TLSs develop due to excess antigen-self in autoimmune diseases, altered-self in cancer, or foreign during infection and transplant rejection.17 Their primary functions include germinal center formation and the production of antibodies or autoantibodies,17 and they are frequently found in the mucosal tissues. Probably, through their ectopic germinal centres, TLSs can also support the production of autoreactive antibodies by plasma cells in the local sites of CI.8,17,18 In autoimmune diseases, TLSs mostly develop in the context of CI, contribute to the maintenance of the disease process, and are often associated with a more severe disease course.8,19,20 In the prostate, TLSs (lymph follicles) appear during severe CI and are familiar to pathologists.5,21 While previous studies have focused on TLSs in PALT and cancer regression,22 only one quantitative study shows their presence in 10% of young men with BPH.12
We quantitatively demonstrate that TLSs can undergo structural adaptation to support local antigen presentation in HG-HP, thereby enhancing CI, and contributing to disease progression. The quantitative findings of our current study show an evident connection between the incidence of BLELs in the prostate and the degree of antigenic stimulation. Proof of this is the presence of a small amount of BLELs in the group of patients with LG-HP and their significant increase in patients with HG-HP (p < 0.001). Obviously, similarly to TLSs, the morphogenesis of the prostatic BLELs is in direct association with the intensity of HP.
Our previous study describes that BLELs in the prostate are characterized by ductal epithelial distortion with intraepithelial lymphocytosis.6,7 Our observations demonstrate that, similar to glandular epithelium in other organs such as the pancreas, salivary glands, thyroid, and breast,23–26 the prostatic ductal epithelium can undergo transformation into specialized lymphoepithelium. The epithelium, overlying the ductal-associated lymphoid tissue in BLELs, changes its functional morphology and immunohistochemical profile in order to become specialized for the transport of antigens directly from the ductal lumen. Furthermore, we observed a strong membranous PD-L1 expression in these BLELs in cases of HG-HP. PD-L1 expression was found exclusively in inflamed prostatic tissue, particularly within the eosinophilic epithelial cells of BLELs, and was absent in normal or non-inflamed prostates.7 Its expression correlated with CI severity and mirrors immune checkpoint activity seen in autoimmune diseases like Sjögren’s syndrome and inflammatory bowel disease.22,25,26 We assume that the specialized PD-L1 positive BLEL plays a critical role as a gateway to the prostatic parenchyma. It has been shown to serve as an entry portal for antigens, like small intestinal and nasopharyngeal lymphoid tissues.27–29 It is obvious that BLEL are basic patho- and morphogenetic elements of HG-HP, reflecting morphologically the moment of epithelial barrier disruption (Figure 4). Obviously, this is followed by an influx of luminal antigenic substances that interact with the immune periductal cells (autoimmunisation), leading to dysbiosis of the prostate microbiome and an increased susceptibility to infection.3 The latter, once started, could establish a feed-forward mechanism resulting in a chronic, persistent HG inflammatory state (HG-HP).3
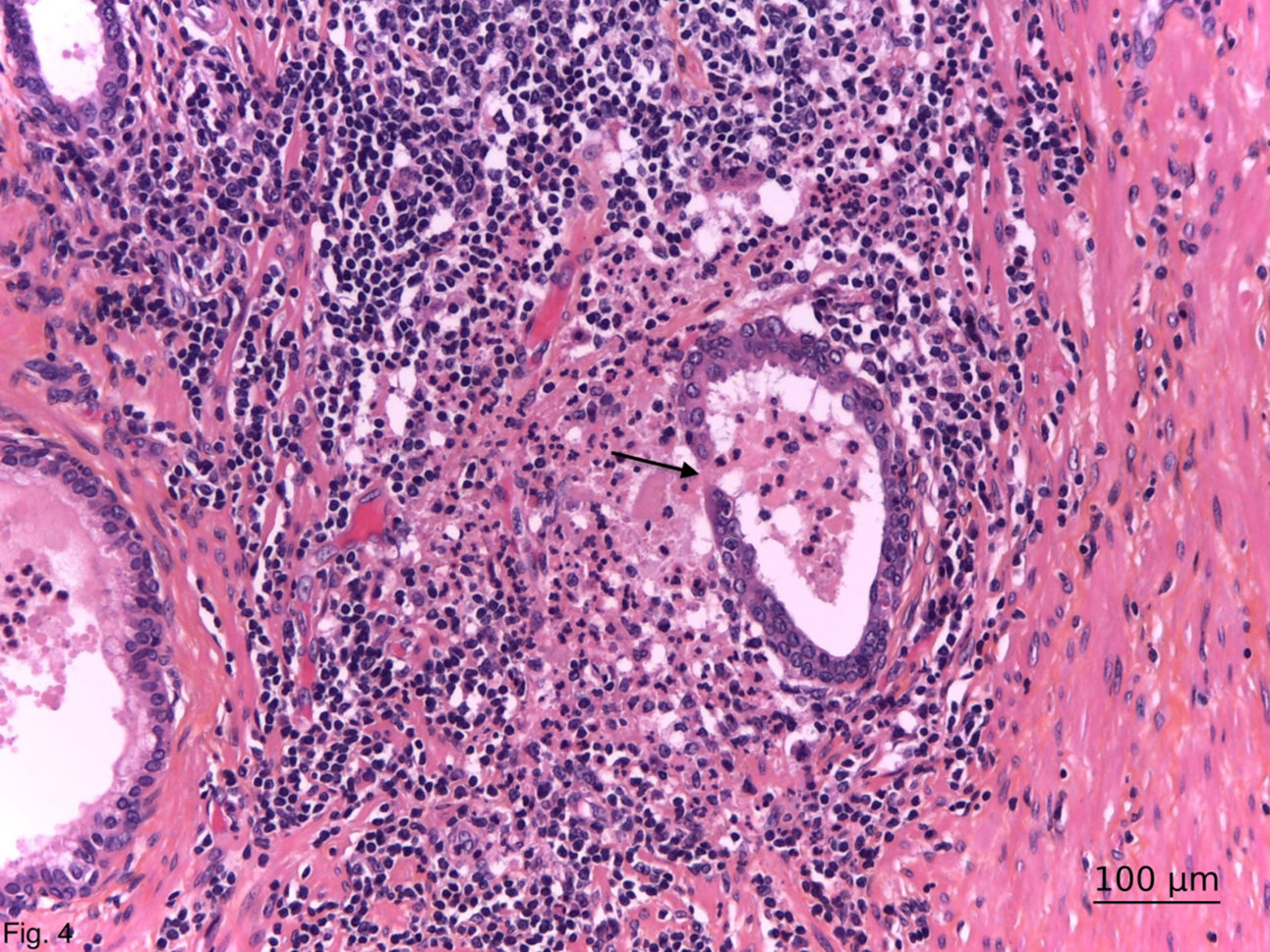
FIGURE 4. General view of high-grade histologic prostatitis with rupture of a ductal benign lymphoepithelial lesion (arrow) and diffusion of the intralumenal substances among immune periductal cells. Haematoxylin-eosin-safran; ×200
Our statistical analysis shows a significant association of the simultaneous presence of both TLSs and BLELs in the context of HG-HP (p < 0.001). Prostatic epithelium can produce various cytokines due to the activation of macrophages residing in the periductal areas and formation of TLSs.20,22 The ductal origin of prostatic BLELs could explain the preferential periductal location of prostatic TLSs. Apart from the necessary proximity to BLELs, this localization is also explained by the need of specialized for lymphoid tissue podoplanine/gp38+ fibroblast-like cells (pericytes, smooth muscle cells and myofibroblasts), which delineate the ductal basal membrane.18 The presence of TLSs, mostly close to the BLELs, is suggestive of their role as the first line of immune defence in the ductal area, which is the entry point of agents/pathogens. Simultaneously or sequentially, all these events might be the key driver of an inflammatory microenvironment and HP progression.3
A similar combination between TLSs and BLELs, called follicle-associated epithelial morphologic changes, is well known from the typical examples of mucosa-associated lymphoid tissue (MALT). By definition, MALT consists of multiple or solitary lymphoid follicles covered by the dome-like shaped follicle-associated epithelium.29–31 These well-organized lymphoid structures are constitutively found in the intestinal Peyer’s patches in the ileum (M-cells) and the nasopharyngeal tract and are inducible in the bronchial tissue.29,30 Our results show that PALT behaves like bronchus-associated lymphoid tissue (BALT),32 which is non-existent in the neonatal period and in childhood but is inducible by external factors. Its development correlates with the degree of HP. Prostatic TLS, BLEL, and PALT are induced and progress at a certain stage of the ontogenetic development of the prostate in the condition of HG-HP. It is obvious that TLSs and BLELs present an integral element of PALT, which is a part of the normal genito-urinary tract mucosal immune system6 and of the inflammatory prostatic microenvironment.3
The co-existence of TLSs and BLELs often develops at sites of inflammation in target tissues in typical autoimmune diseases:8,9 rheumatoid arthritis, Sjogren’s syndrome, multiple sclerosis, Hashimoto thyroiditis, myasthenia gravis, systemic lupus erythematosus.16,22,26,27 It is hypothesized that their presence and persistence contribute to disease progression.25,27 We believe that TLSs and BLELs represent a serious morphological argument supporting the immune/autoimmune pathogenesis of HP.
None of the patients in the three control groups of autopsy prostates had TLSs and BLELs. To our knowledge, any quantitative pathological study of prostatic TLSs and BLELs using control material. Autopsied prostates gave us the opportunity to evaluate the prevalence of TLSs and BLELs in HG-HP, their sporadic presence in LG-HP, and their complete lack in the three control groups of patients. Prostatic TLSs and BLELs, like other organs9, are not present in embryonic life, in newborns, children, and in normal non-inflammatory adult human prostate (control groups (1) and (2)). As our results show, TLSs and BLELs—both individually and in combination—are components of PALT. Their absence in end-stage AIDS cases (control group (3)) indicates a dependеnce on immune activity.
A chronological analysis of the mean age of the patients included in our study suggests a likely pathophysiological and morphogenetic sequence of events. Initially, HP may begin as LG CI, with the mean age of patients with LG-HP being 69.05 years. Over time, and under the influence of a persistent damaging factor, this CI progresses to a HG form, with a mean age of 71.32 years for patients with HG-HP. The appearance of histological features of immune-mediated prostatic inflammation is also observed within this age range—70.5 years for TLSs and 71.15 years for BLELs. The peak occurrence of the combined presence of both immune structures is noted approximately one year later, at a mean age of 72.11 years.
The localization of TLSs and BLELs most commonly overlaps with the typical localization of HP: in the transition zone in patients with BPH (TURP/AD) and in the peripheral zone in patients with PCa (RP). Therefore, we frequently observed the combined presence of these two immune structures in specimens from patients with pronounced clinical symptoms that required surgical intervention (TURP/AD and RP). In contrast, this combination was only sporadically identified in autopsy-derived prostate samples from patients without urological symptoms.
As prostatic TLSs and BLELs have no diagnostic organ specificity, although they are a constant finding in HG-HP, the morphological definition of the former may need to be expanded.5 HG-HP is characterized by four histological signs: lymphocytic inflammatory infiltration, acinar atrophy, alteration of prostatic ducts with TLS and BLEL formation.
However, this study has several limitations. First, we did not quantify TLSs and BLELs (on mm2 or grams of prostatic tissue). Second, we did not assess the presence of other prostatic lesions, presumably precancerous and post-inflammatory, such as high-grade PIN, post-atrophic hyperplasia, and proliferative inflammatory atrophy. Finally, the lack of clinical urological data, including serum level of PSA in autopsy patients, slightly limits, although insignificantly, the complexity of the clinicopathological associations of the presented records. Further investigations of a large series of patients with statistical data processing are necessary in order to gain a more detailed insight into the close relationships of TLSs and BLELs, and TLSs with PSA and precancerous and PIN prostatic lesions.
In conclusion, our results provide evidence that: a) TLSs and BLELs are not observed in the normal human prostate; b) PALT, like BALT, which is non-existent after birth and in childhood, is inducible by external factors and its development is in association with the degree of the NIH group IV prostatitis; c) TLSs and BLELs present an integral part of HG-HP; d) NIH category IV prostatitis occurs in a number of histologic patterns. We describe a new pattern in prostatic inflammation, characterized by the presence of TLSs in close association with BLEL in HG-HP; e) Like other organs, TLSs and BLELs formation in human prostate can be considered as a hallmark of tissue autoimmunity and probably reflects the immune/autoimmune phase of NIH category IV prostatitis; f) The association between TLSs and BLELs and HG-HP reveals the aggressiveness grade of the subclinical NIH-category IV prostatitis. Our results could serve as an accurate guide for the clinician. We believe that except for the aggressiveness grades of the inflammation, the presence of TLSs and BLELs should be noted in the pathologist’s report, especially in initial prostate biopsies, in order to help the timing of the further biopsy; g) The actual NIH consensus grading classification system based on morphological criteria for the chronic prostatic inflammation, confirms that the higher level of TLSs depends significantly on the grade of prostatic tissue inflammation.5 We suggest that the presence of BLELs must be added to it.
The novel data from this study suggest, for the first time, that TLSs and BLELs may be key players in the pathophysiology and morphogenesis of NIH-category IV prostatitis by mediating the self-sustained inflammatory immune/autoimmune processes in this chronic subclinical disease.
Acknowledgement
Not applicable.
Funding Statement
The research received no external funding.
Author Contributions
Dorian Dikov: conceptualization, methodology, writing—review and editing; Maria Koleva: software, writing—original draft preparation; Kiril Simitchiev, Anelia Bivolarska: data duration; Albena Fakirova, Victoria Srafian: supervision, formal analysis. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials
The datasets generated and analyzed during the current study are fully anonymized and contain no personally identifiable information. These datasets are available from the corresponding author upon reasonable request.
Ethics Approval
The study was approved by the Ethics Committees at the St. George University Hospital of Plovdiv, Bulgaria (Protocol No 6/08/06/2023), the General Hospital of Lovetch, Bulgaria (Protocol No 1/03/04/2008), Rothschild Hospital, Paris, France (Protocol No 1/16/06/1994), and the Grand Hôpital de l’Est Francilien, Jossigny, France (Protocol No 4/30/05/2008). No personal or identifiable patient data were collected or disclosed; all analyzed data were fully anonymized in compliance with data protection regulations.
Informed Consent
Informed consent was obtained from all patients.
Conflicts of Interest
Some of the results have been presented at three European congresses (Dublin, Ireland, 2023; Florence, Italy, 2024; Vienna, Austria, 2025).
References
1. De Marzo AM, Platz EA, Sutcliffe S et al. Inflammation in prostate carcinogenesis. Nat Rev Cancer 2007;7(4):256–269. doi:10.1038/nrc2090. [Google Scholar] [PubMed] [CrossRef]
2. Delongchamps NB, de la Roza G, Chandan V et al. Evaluation of prostatitis in autopsied prostates: is chronic inflammation more associated with BPH or cancer? J Urol 2008;179(5):1736–1740. doi:10.1016/j.juro.2008.01.034. [Google Scholar] [PubMed] [CrossRef]
3. Sfanos KS, Yegnasubramanian S, Nelson WG et al. The inflammatory microenvironment and microbiome in prostate cancer development. Nat Rev Urol 2018;15(1):11–24. doi:10.1038/nrurol.2017.167. [Google Scholar] [PubMed] [CrossRef]
4. Krieger JN, Nyberg L, Nickel JC. NIH consensus definition and classification of prostatitis. JAMA 1999;282:236–237. [Google Scholar] [PubMed]
5. Nickel JC, True LD, Krieger JN et al. Consensus development of a histopathological classification system for chronic prostatic inflammation. BJU Int 2001;87(9):797–805. doi:10.1046/j.1464-410x.2001.02193.x. [Google Scholar] [PubMed] [CrossRef]
6. Dikov D, Bachurska S, Staikov D et al. Intraepithelial lymphocytes in relation to NIH category IV prostatitis in autopsy prostate. Prostate 2015;75(10):1074–1084. doi:10.1002/pros.22991. [Google Scholar] [PubMed] [CrossRef]
7. Dikov D, Koleva M, Simitchiev K et al. PD-L1 positive lympho-epithelial lesions in inflammatory prostate. Histol Histopathol 2022;37(8):749–755. [Google Scholar] [PubMed]
8. Corsiero E, Nerviani A, Bombardieri M, Pitzalis C. Ectopic lymphoid structures: powerhouse of autoimmunity. Front Immunol 2016;7(10):430. doi:10.3389/fimmu.2016.00430. [Google Scholar] [PubMed] [CrossRef]
9. Pipi E, Nayar S, Gardner DH et al. Tertiary lymphoid structures: autoimmunity goes local. Front Immunol 2018 Sep 12;9:1952. doi:10.3389/fimmu.2018.01952. [Google Scholar] [PubMed] [CrossRef]
10. Zhu J. Research review of the mechanism and clinical application prospects of tertiary lymphoid structures in the immune micro-environment of gastrointestinal tumors. Oncol Res 2025 Jun 26;33(7):1571–1580. doi:10.32604/or.2025.058957. [Google Scholar] [PubMed] [CrossRef]
11. Carlo ED, Magnasco S, D’Antuono T, Tenaglia R, Sorrentino C. The prostate-associated lyphoide tissue (PATL) is linked to the expression of homing chemokines CXCLI3 and CCL2I. Prostate 2007;67(10):1070–1080. doi:10.1002/pros.20604. [Google Scholar] [PubMed] [CrossRef]
12. García-Hernández ML, Uribe-Uribe NO, Espinosa-González R, Kast WM, Khader SA, Rangel-Moreno J. A Unique cellular and molecular microenvironment is present in tertiary lymphoid organs of patients with spontaneous prostate cancer regression. Front Immunol 2017;8:563. doi:10.3389/fimmu.2017.00563. [Google Scholar] [PubMed] [CrossRef]
13. Blumenfeld W, Tucci S, Narayan P. Incidental lymphocytic prostatitic: selective involvement with nonmalignant glands. Am J Surg Pathol 1992;16(10):975–981. doi:10.1097/00000478-199210000-00008. [Google Scholar] [CrossRef]
14. McNeal JE. Regional morphology and pathology of the prostate. Am J Clin Pathol 1968;49(3):347–357. doi:10.1093/ajcp/49.3.347. [Google Scholar] [PubMed] [CrossRef]
15. Isaacson PG, Chott A, Nakamura S et al. Extranodal marginal zone lymphoma of mucosa-associatedlymphoid tissue (MALT lymphoma). In: Swerdlow SH et al., editors. WHO classification of tumours of hematopoietic and lymphoid tissues. 4th ed. Lyon, France: WHO Classification of Tumours; 2008. doi:10.1016/b978-0-323-47779-6.50067-3. [Google Scholar] [CrossRef]
16. Nakshbandi U, van Ginkel MS, Verstappen GMPJ et al. Histopathological comparison of Sjögren-related features between paired labial and parotid salivary gland biopsies of sicca patients. Rheumatology 2024;63(10):2670–2677. doi:10.1093/rheumatology/keae154. [Google Scholar] [PubMed] [CrossRef]
17. Barone F, Gardner DH, Nayar S, Steinthal N, Buckley CD, Luther SA. Stromal fibroblasts in tertiary lymphoid structures: a novel target in chronic inflammation. Front Immunol 2016;7(4):477. doi:10.3389/fimmu.2016.00477. [Google Scholar] [PubMed] [CrossRef]
18. Zhao R, Gao D. Innate immunity and tertiary lymphoid structures. Immunol Rev 2025;332(1):e70052. doi:10.1111/imr.70052. [Google Scholar] [PubMed] [CrossRef]
19. Alexander RB, Brady F, Ponniah S. Autoimmune prostatitis: evidence of T cell reactivity with normal prostatic proteins. Urology 1997;50(6):893–899. doi:10.1016/s0090-4295(97)00456-1. [Google Scholar] [CrossRef]
20. Goronzy JJ, Weyand CM. Perivascular tertiary lymphoid structures in autoimmune disease. Immunol Rev 2025 Jul;332(1):1543322. doi:10.1111/imr.70047. [Google Scholar] [PubMed] [CrossRef]
21. Bennett B, Culberson DE, Petty CS et al. Histopathology of prostatitis. J Urol 1990;143:265A. [Google Scholar]
22. Pitzalis C, Jones GW, Bombardieri M, Jones SA. Ectopic lymphoid-like structures in infection, cancer and autoimmunity. Nat Rev Immunol 2014;14(7):447–462. doi:10.1038/nri3700. [Google Scholar] [PubMed] [CrossRef]
23. Jing F, Choi EY. Potential of cells and cytokines/chemokines to regulate tertiary lymphoid structures in human diseases. Immune Netw 2016;16:271–280. doi:10.4110/in.2016.16.5.271. [Google Scholar] [PubMed] [CrossRef]
24. Giday SA, Khashab MA, Buscaglia JM et al. Autoimmune pancreatitis: current diagnostic criteria are suboptimal. J Gastroenterol Hepatol 2011;26(6):970–973. doi:10.1111/j.1440-1746.2011.06683.x. [Google Scholar] [PubMed] [CrossRef]
25. Daniels TE. Benign lymphoepithelial lesion and Sjogren syndrome. In: Ellis GL, Auclair PL, Gnepp DR, editors. Surgical pathology of the salivary glands. Philadelphia, PA, USA: Saunders; 1992. p. 83–106. [Google Scholar]
26. Pringle S, Verstappen GM, van Ginkel MS et al. Lymphoepithelial lesions in the salivary glands of primary Sjögren’s syndrome patients: the perfect storm? Clin Exp Rheumatol 2022 Dec;40(12):2434–2442. doi:10.55563/clinexprheumatol/06an99. [Google Scholar] [PubMed] [CrossRef]
27. Matias-Guiu X, Esquius J. Lymphoepithelial lesion in the thyroid. A non-specific histological finding. Pathol Res Pract 1991;187:296–300. doi:10.1016/s0344-0338(11)80787-9. [Google Scholar] [PubMed] [CrossRef]
28. Yamamoto M, Bharti A, Li Y, Kufe D. Interaction of the DF3/MUC1 breast carcinoma-associated antigen and beta-catenin in cell adhesion. J Biol Chem 1997;272:12492–12494. doi:10.1074/jbc.272.19.12492. [Google Scholar] [PubMed] [CrossRef]
29. Tamborrino T, Bonente D, Regoli M et al. Morphological characterization of intrafollicular epithelial bodies (IFEBs) in rabbit peyer’s patches. Int J Mol Sci 2025 Mar 30;26(7):3207. doi:10.3390/ijms26073207. [Google Scholar] [PubMed] [CrossRef]
30. Oya Y, Kimura S, Uemura M, Fujimura Y, Hase K. Tear duct M cells exacerbate allergic conjunctivitis by facilitating germinal-center reactions. Mucosal Immunol 2025 Jun;18(3):549–561. doi:10.1016/j.mucimm.2025.01.009. [Google Scholar] [PubMed] [CrossRef]
31. Kim N, Ju IG, Jeon SH et al. Inhibition of microfold cells ameliorates early pathological phenotypes by modulating microglial functions in Alzheimer’s disease mouse model. J Neuroinflammation 2023 Nov 27;20(1):282. doi:10.1186/s12974-023-02966-9. [Google Scholar] [PubMed] [CrossRef]
32. Moyron-Quiroz JE, Rangel-Moreno J, Kusser K et al. Role of inducible bronchus associated lymphoid tissue (iBALT) in respiratory immunity. Nat Med 2004;10(9):927–934. doi:10.1038/nm1091. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools