 Open Access
Open Access
ARTICLE
PSMA PET/CT-guided pelvic lymph node dissection in patients with unfavorable intermediate- or high-risk prostate cancer
1 Department of Urology, ERN UROGEN Accredited Center, Ghent University Hospital, Ghent, 9000, Belgium
2 Department of Pathology, Ghent University Hospital, Ghent, 9000, Belgium
3 Department of Radiology, Ghent University Hospital, Ghent, 9000, Belgium
4 Department of Radiation Oncology, Ghent University Hospital, Ghent, 9000, Belgium
5 Department of Medical Imaging (Nuclear Medicine), Ghent University Hospital, Ghent, 9000, Belgium
* Corresponding Author: Eva Donck. Email:
(This article belongs to the Special Issue: Advances in Molecular Imaging and Targeted Therapies for Prostate Cancer)
Canadian Journal of Urology 2026, 33(1), 83-92. https://doi.org/10.32604/cju.2025.068589
Received 01 June 2025; Accepted 27 August 2025; Issue published 28 February 2026
Abstract
Objectives: PSMA PET/CT (Prostate-Specific Membrane Antigen Positron Emission Tomography/Computed Tomography) offers improved accuracy in detecting lymph node invasion (LNI) in prostate cancer (PC) patients, potentially reducing the need for extended pelvic lymph node dissection (ePLND). This study aims to evaluate a patient-tailored care pathway in which ePLND is performed only in patients with unfavorable intermediate- or high-risk PC who are deemed at risk for LNI based on PSMA PET/CT findings. Methods: In this interventional cohort study, 81 patients were managed according to the new care pathway. ePLND was omitted in cases of negative PSMA PET/CT findings (N0M0), while those with positive PSMA PET/CT findings (N1M0) underwent ePLND. A comparator group of 81 patients was selected from a prospectively generated database for comparison. Results: The intervention group experienced a 75% reduction in the number of ePLNDs performed compared to the comparator group (p < 0.001). ePLND-related complications were significantly lower in the intervention group (p = 0.008). No significant difference was observed in 3-year biochemical-recurrence free survival (BRFS) between the two groups (p = 0.958). Conclusion: Omitting ePLND in patients with negative PSMA PET/CT findings (N0M0) leads to a substantial reduction in the number of ePLNDs performed, resulting in a decrease in morbidity, without compromising early oncological outcomes.Keywords
The introduction of PSMA PET/CT (Prostate-Specific Membrane Antigen Positron Emission Tomography/Computed Tomography) has markedly improved the accuracy of lymph node invasion (LNI) detection in prostate cancer (PC) patients compared to conventional imaging modalities such as computed tomography (CT) and magnetic resonance imaging (MRI).1 With a sensitivity of approximately 40–55% and a specificity of around 94–97%, PSMA PET/CT represents a reliable staging tool for assessing LNI.2–4 However, small lymph node metastases may still go undetected. Histopathological examination of lymph nodes obtained through extended pelvic lymph node dissection (ePLND) remains the most reliable method for N-staging. The clinical necessity of staging PC patients for regional LNI has been a subject of ongoing debate. Over the past few years, the European guidelines have adopted a more conservative approach to performing PLND, due to a lack of clear oncological benefit.5–7 As a result, PLND is no longer actively recommended in patients with high-risk and locally advanced PC according to the most recent EAU guidelines. However, if a PLND is to be performed, it is strongly recommended that it be done as an ePLND.7
Several nomograms have been developed to estimate the risk of LNI. Examples include the ROACH formula, various iterations of the Briganti nomogram, and the MSKCC nomogram.8 Using one of these risk calculators, nearly all patients with unfavorable intermediate-risk (Grade group 3) and high-risk PC are considered candidates for ePLND, resulting in a substantial number of potentially unnecessary procedures.8 Notably, these nomograms were developed prior to the widespread adoption of PSMA PET/CT and therefore do not incorporate its findings.8,9 In response to this limitation, newer nomograms are being developed that integrate PSMA PET/CT data to more accurately identify patients who may benefit from PLND.9–11 According to the systematic review by Stabile et al., staging with PSMA PET/CT may help reduce the number of ePLND procedures in men with a lower risk of LNI, due to its high negative predictive value (NPV) in this subgroup.12 However, in high-risk patients, a negative PSMA PET/CT scan does not reliably exclude LNI, as the NPV is significantly lower in this population.12 Further research is needed to evaluate the impact of PSMA PET/CT on the clinical necessity of PLND.
The aim of this study is to evaluate a patient-tailored care pathway for men with unfavorable intermediate- and high-risk PC opting for radical prostatectomy (RP), in which PSMA PET/CT is used as the primary staging modality and PLND is reserved for patients identified as at risk based on PSMA PET/CT findings.
Study design and patient population
This interventional cohort study included patients with unfavorable intermediate- and high-risk PC undergoing RP. Unfavorable intermediate- or high-risk PC was defined by the presence of at least one of the following criteria: ISUP grade group > 3, prostate-specific antigen (PSA) level > 20 ng/dl, clinical T-stage > cT2c as assessed by digital rectal examination (DRE), and/or cN+ on conventional imaging (MRI or CT). The ISUP grade group was determined based on histopathological evaluation of prostate biopsy specimens. All patients underwent pelvic MRI for locoregional staging and biopsy planning. Conventional staging with bone scan and CT was permitted if already performed prior to entry into the care pathway. In cases where conventional imaging had not yet been performed, it was recommended to omit it. Patients were excluded if they were unwilling to provide written informed consent, were unable to comply with the care pathway protocol, had evidence of M1 disease on conventional imaging, had low- or favourable intermediate-risk PC, had a contraindication for PSMA PET/CT, or were unable to undergo pelvic MRI. This study was approved by the Ethics Committee of Ghent University Hospital (reference number BC-10297).
All patients in the intervention cohort underwent either 18F PSMA-11 PET/CT or 68Ga-PSMA-11 PET/CT for N- and M-staging. In cases where both PSMA PET/CT and pelvic MRI showed no evidence of lymph node or distant metastases (N0M0), PLND was omitted during RP. If PSMA PET/CT indicated suspected LNI (N1M0), an ePLND was performed during RP. In cases with negative findings on PSMA PET/CT (N0M0) but suspicious findings on pelvic MRI (N1M0), a selective PLND (removal of the suspicious lymph nodes only) was carried out. The ePLND template included lymph nodes overlying the external iliac vessels, along the internal iliac artery, and within the obturator fossa. Patients were followed at regular intervals: every 3 months during the first year after diagnosis, every 6 months during years 2 to 5, and annually thereafter. Follow-up consisted of history taking, physical examination, and serum PSA measurements. Additional diagnostic tests, including imaging, were performed as deemed necessary by the treating physician. Adjuvant radiotherapy combined with androgen deprivation therapy (ADT) was administered in cases of >3 positive lymph nodes detected during ePLND (whole pelvis radiotherapy), or in cases with 1–2 positive lymph nodes and a pathological stage of ≥pT3b. Early salvage radiotherapy was offered to patients with biochemical recurrence (BCR), defined as PSA > 0.2 ng/mL or three consecutive rises in PSA above the detection limit. Deviations from the protocol were allowed following consultation with the multidisciplinary cancer team.
Since 2015, all patients with PC have been invited to provide informed consent for participation in a prospective registry and follow-up study (EC UZG 2019/1506). Data from this cohort are stored in a pseudonymized database. A total of 81 patients with unfavorable- and high-risk PC were selected from this database to serve as the comparator group. This group was managed according to the pre-existing institutional treatment protocol, which was later also applied to the intervention cohort, ensuring identical radiation and systemic therapy approaches.
The primary outcomes are: (1) reduction in the number of PLNDs performed, and (2) metastasis-free survival (MFS) at 3 years.
Secondary outcomes include: (1) reduction in postoperative complications within 90 days, assessed using the Clavien-Dindo classification system,13 (2) biochemical recurrence-free survival (BRFS) at 3 years, and (3) overall survival (OS) at 3 years.
Statistical analyses were performed using SPSS version 28 and 29 (IBM Corp., Armonk, NY, USA).
A power analysis was conducted prior to the study. It was assumed that 90% of patients in the comparator cohort would undergo ePLND. The new care pathway would be superior if it could achieve a 20% reduction in ePLND without compromising MFS. To achieve a power of 90% with a significance level of 5%, a sample size of 79 patients per group (158 total) was required. To account for potential dropout, 81 patients were enrolled in the intervention cohort.
Patient characteristics were analyzed using descriptive statistics. For continuous variables, normal distribution was assessed using the Shapiro-Wilk test. Normally distributed data were reported as mean with standard deviation; non-normally distributed data as median with interquartile range (IQR). Categorical variables were summarized using frequencies and percentages. Differences between groups were evaluated using the Mann-Whitney U test for continuous variables and the chi-square test for categorical variables. Kaplan-Meier analysis was used to estimate BRFS, MFS, cancer-specific survival (CSS), and OS. Comparisons between groups were made using the Log-Rank test. All analyses were conducted according to the intention-to-treat principle. A p-value < 0.05 was considered statistically significant.
A total of 162 patients were included in the study: 81 in the intervention group and 81 in the comparator group. Preoperative patient characteristics are summarized in Table 1. There were no significant differences between the two groups with regard to age (p = 0.810), preoperative PSA levels (p = 0.296), EAU risk group (p = 0.282), ISUP grade group on prostate biopsy specimen (p = 0.871), clinical T-stage on MRI (p = 0.286), N-stage on conventional imaging (CT or MRI) (p > 0.99), or N-stage on PSMA PET/CT (p = 0.383). In the intervention group, 11 patients were found to have suspected LNI on PSMA PET/CT, of whom 7 did not show LNI on conventional imaging. Notably, no patients had suspected LNI on conventional imaging that was not also detected on PSMA PET/CT; therefore, no selective PLNDs were performed. Postoperative patient characteristics are presented in Table 2. There were no significant differences in pathological tumor stage (p = 0.310), grade group on prostatectomy specimen (p = 0.352), or surgical margin status (p = 0.329) between groups. In the intervention group, LNI was confirmed on final pathology examination in 6 out of 11 (55%) patients who underwent ePLND. In contrast, LNI was detected in 13 out of 72 (18%) patients in the comparator group (p = 0.007). Among the 7 patients with suspected LNI on PSMA PET/CT but not on conventional imaging, 2 had confirmed LNI on final pathology examination. All 4 patients with LNI suspected on both PSMA PET/CT and conventional imaging had pathologically confirmed LNI. In total, 54 patients received adjuvant or salvage radiotherapy: respectively 8 (10%) and 12 (15%) in the intervention group, and 16 (20%) and 18 (22%) in the comparator group (p = 0.058). The median follow-up duration was 35 months (IQR: 26–59), with significantly longer follow-up in the comparator group compared to the intervention group (59 vs. 27 months; p < 0.001).
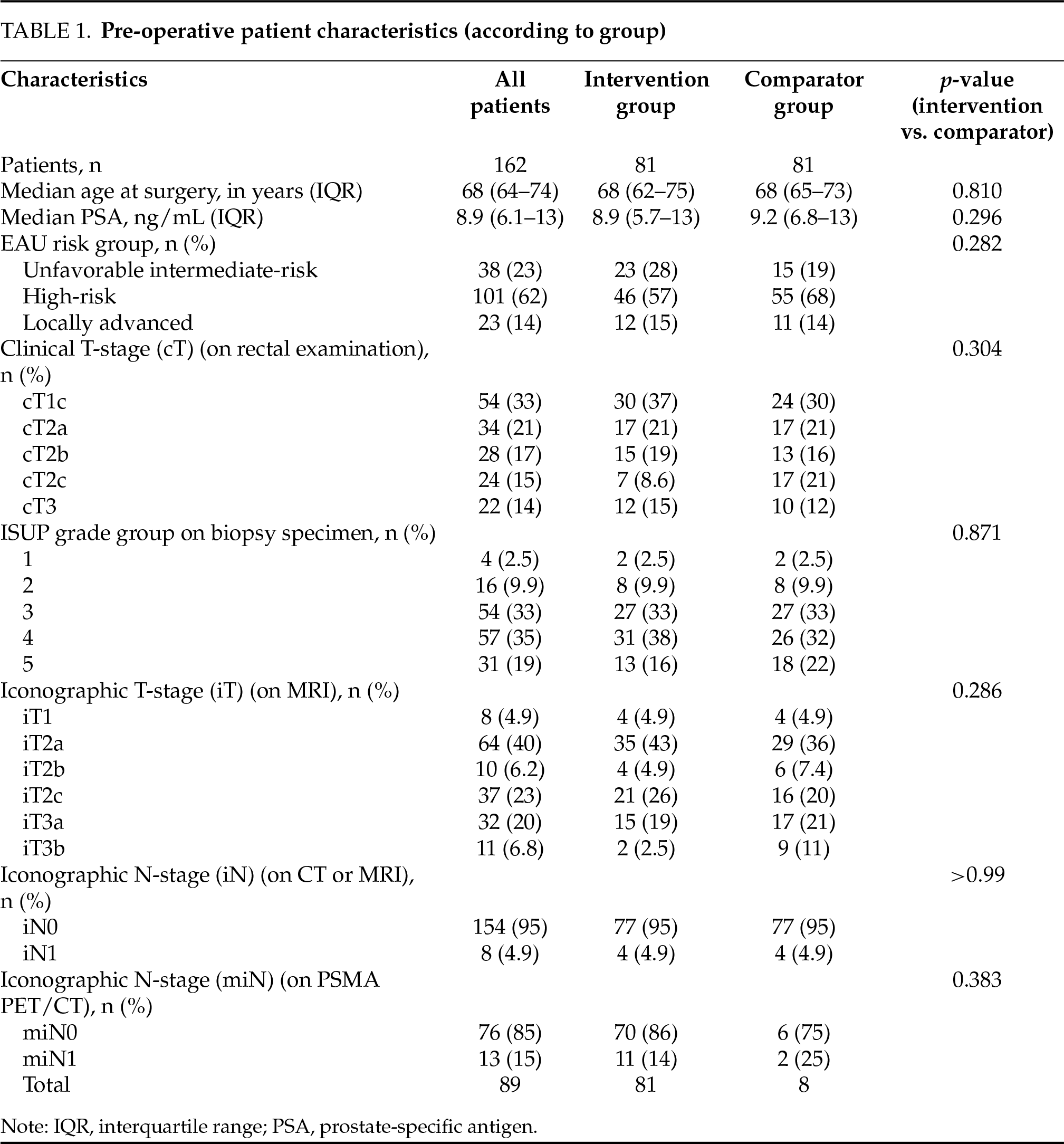
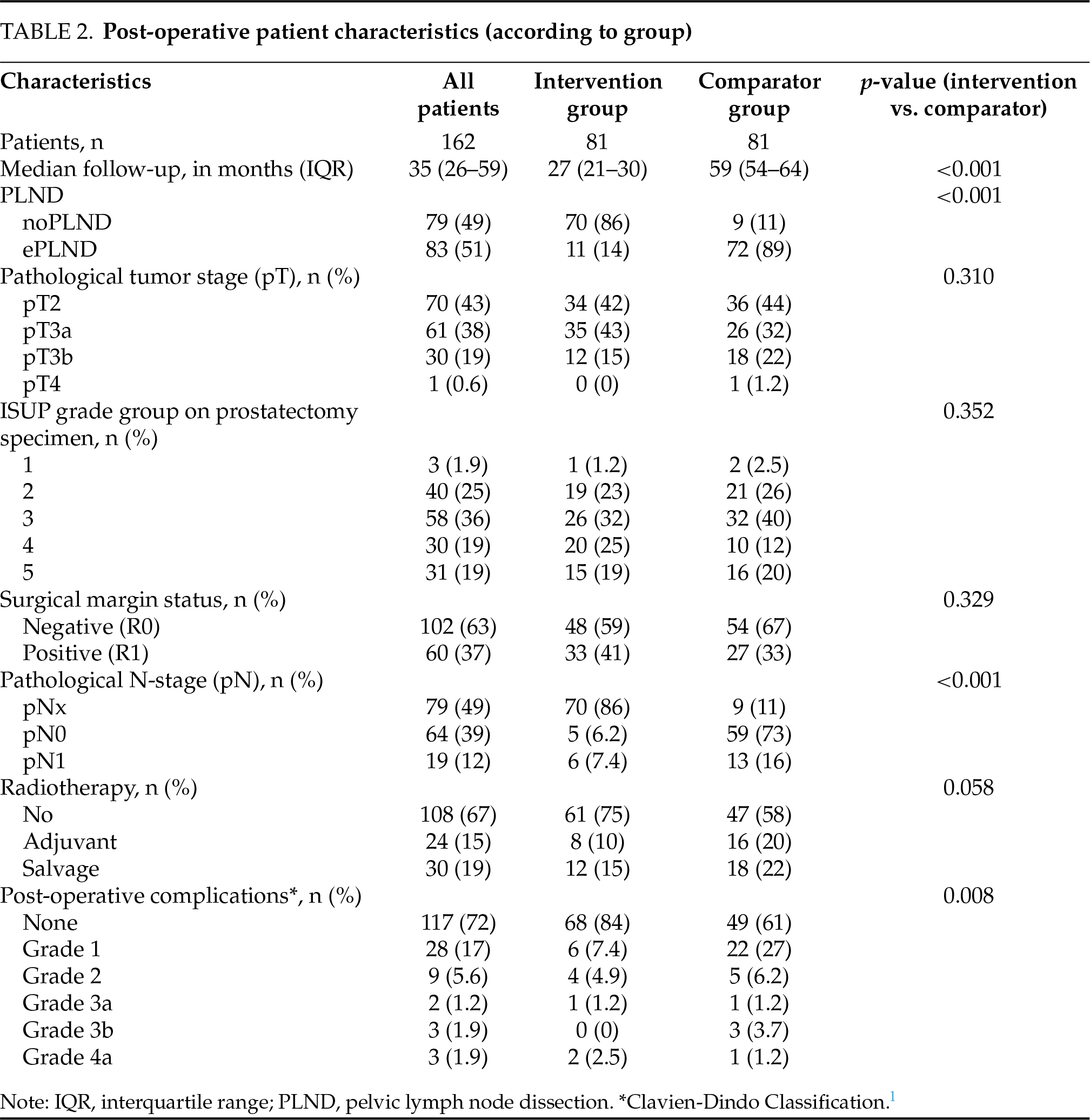
Only 11 (14%) patients in the intervention group underwent ePLND, compared to 72 (89%) in the comparator group (p < 0.001), representing a 75% reduction in the number of ePLNDs performed. During a median follow-up of 27 months, 2 (2.5%) patients in the intervention group developed metastases compared to 8 (9.9%) patients in the comparator group over a median follow-up of 59 months. Estimated 3-year MFS was 93% (±5.8) in the intervention group and 91% (±3.2) in the comparator group (p = 0.324) (Figure 1A). Postoperative complications were significantly fewer in the intervention group (13 events) compared to the comparator group (32 events) (p = 0.008), as shown in Table 2. A detailed list of the post-operative complications, according to the classification by Cacciamani et al., is available in Table 3.14 In the intervention group, 1 (1.2%) patient and 2 (2.5%) patients had a complication that was respectively “strongly related to PLND” and “likely related to PLND”. In comparison, 7 (8.6%) and 9 (11%) patients in the comparator group experienced complications in these respective categories.
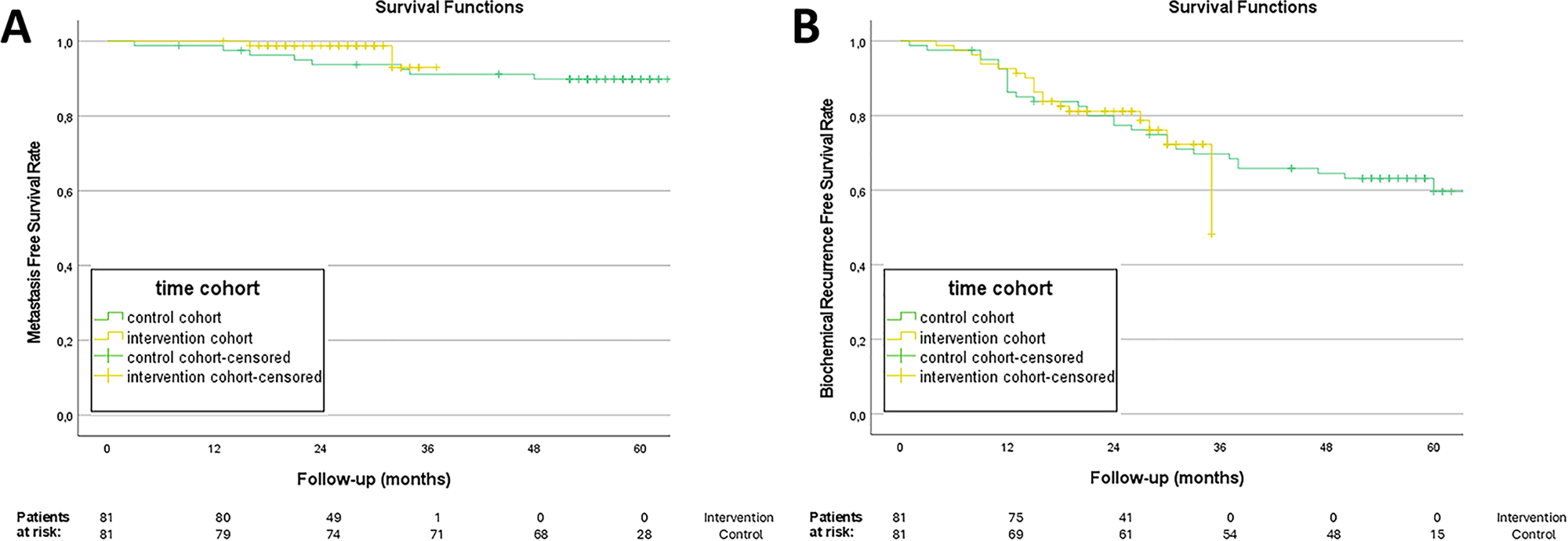
FIGURE 1. Survival functions. (A) Metastasis-free survival (MFS) rate; (B) Biochemical recurrence-free survival (BRFS) rate
BCR occurred in 19 (23%) patients in the intervention group and in 30 (37%) patients in the comparator group. Estimated 3-year BRFS was 48 (±20)% for the intervention group and 70 (±5.2)% for the comparator group (p = 0.958) (Figure 1B). When stratified by EAU risk group, no significant differences in BRFS were observed (p = 0.882). Estimated 3-year OS was 96 (±2.1)% in the comparator group and 100% in the intervention group, with no deaths occurring in the latter (p = 0.118).
The role of ePLND in PC management remains a topic of ongoing debate. To date, no definitive oncological benefit of performing an ePLND has been consistently demonstrated. Two randomized controlled trials (RCTs) have compared extended with limited PLND templates in recent years.15,16 While both studies failed to demonstrate a significant advantage of ePLND in terms of early outcomes, such as BRFS, updated findings from one of these trials now suggest a potential benefit in reducing the risk of developing distant metastases.15–17 However, robust evidence comparing ePLND with no PLND at all is still lacking. The advent of PSMA PET/CT as a novel staging modality—with its superior accuracy compared to conventional imaging—has further fueled the debate around the role of ePLND in primary PC staging.1,9,12 The high NPV of PSMA PET/CT, reaching approximately 95% for the detection of pelvic LNI, may support omitting PLND in patients with negative PSMA PET/CT findings.1 This notion is further supported by a very recent systematic review and meta-analysis, which confirmed that PSMA PET/CT provides clinically relevant information that can influence surgical planning, including the decision whether or not to perform a PLND.4 Nonetheless, patient-specific factors such as PSA level and ISUP grade group remain essential components of the risk assessment, especially given the possibility of microscopic nodal metastases that may be missed on PSMA PET/CT.4
In this study, we evaluate a patient-tailored care pathway in which PLND is performed only in patients with unfavorable intermediate- and high-risk PC deemed at risk for LNI as identified by PSMA PET/CT. As expected, this approach led to a substantial reduction in the number of PLNDs performed, with a 75% decrease compared to the control group. This reduction did not compromise short-term oncological outcomes and was accompanied by a significant drop in PLND-related complications. In the comparator group, a significantly smaller percentage of patients undergoing ePLND had histologically confirmed LNI, further underlining the potential overuse of PLND in the pre-PSMA PET/CT era. That the use of PSMA PET/CT in staging could reduce the number of unnecessary PLND procedures has been brought up by Stabile et al.12 Their systematic review and meta-analysis demonstrated a difference in NPV between risk groups, with a lower NPV in high-risk patients (83%) compared to intermediate-risk patients (96%). This differential diagnostic performance suggests that ePLND may be safely omitted in men with a nomogram-predicted LNI risk below 20% and a negative PSMA PET/CT scan. Conversely, in patients with a higher a priori risk of LNI, forgoing PLND may result in missed nodal disease and is therefore not recommended. These findings are in line with the most recent systematic review and meta-analysis by Mazzone et al., which confirmed the lower NPV of PSMA PET/CT in high-risk patients compared to those with intermediate-risk disease, further reinforcing the need for a risk-adapted approach to interpreting negative PSMA PET/CT findings.4
However, definitive proof of an oncological benefit from performing an ePLND remains lacking and was also not demonstrated in the present study. Early oncologic outcomes, such as improved BRFS, could not be confirmed in the comparator group. Even after stratifying for risk group, no significant differences were observed between the intervention and control arms. It is noteworthy that BRFS appears lower in the intervention group, despite the lack of statistical significance, which may be explained by the smaller sample size and the shorter follow-up period, concentrating events earlier. Only two patients in the intervention group developed metastases, and no deaths occurred in the intervention group during a median follow-up of 27 months. However, this relatively short follow-up—particularly when compared to the 59-month median follow-up in the comparator group—raises the possibility of underestimating late biochemical recurrences. Given the often indolent course of prostate cancer, longer follow-up is essential before drawing firm conclusions regarding long-term oncologic outcomes such as MFS and OS.
We also observed relatively high positive surgical margin (PSM) rates (41% vs. 33%), which are consistent with other studies involving high-risk prostate cancer cohorts.18,19 This likely reflects the unfavorable disease characteristics of our population. Additionally, the routine use of whole-mount histopathological processing in our teaching center may lead to more accurate detection of positive margins. The frequent application of Retzius-sparing robot-assisted laparoscopic radical prostatectomy (RALP), a technique associated with improved functional outcomes but potentially higher apical PSM rates, may also contribute.
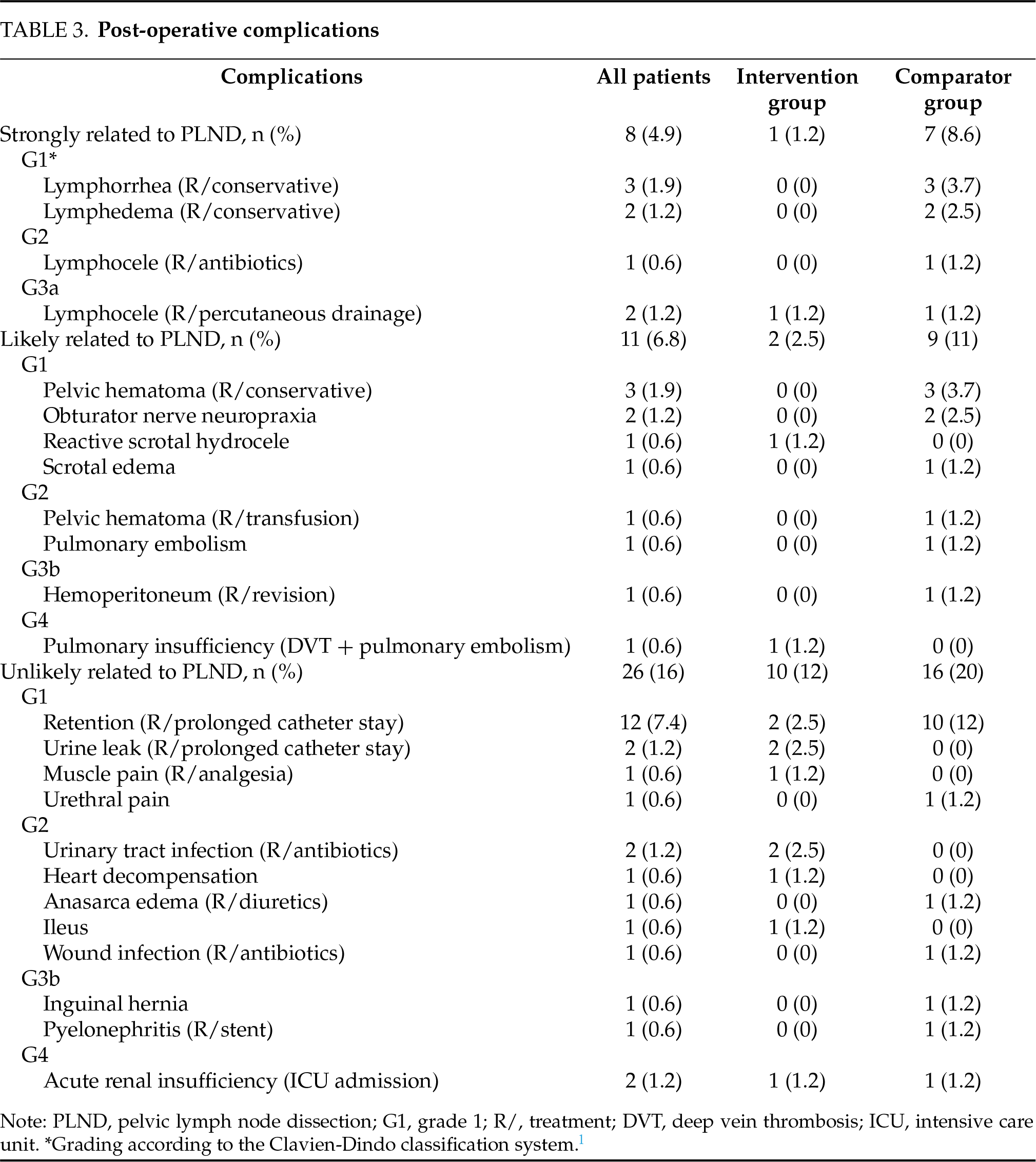
PLND is known to be associated with increased and more severe postoperative complication rates.8,14,20 According to the systematic review by Fossati et al., it is associated with a nearly 20% complication risk, mainly related to significant lymphoceles.7,20 The results from this study are in line with these data: 19 out of 83 (23%) patients who underwent ePLND experienced a complication either strongly or likely related to the procedure according to the classification by Cacciamani et al.14 Most of these were Clavien-Dindo grade 1 or 2 complications (79%). Given that 75% fewer ePLNDs were performed in the intervention group, a significantly lower complication rate was observed in this group compared to the control group.
Despite the increased morbidity and the lack of evidence for an oncological benefit, there is still insufficient proof that PLND provides no oncological advantage. Notably, the prostate-only versus whole-pelvic radiation therapy (POP-RT) trial demonstrated that in high-risk PC patients with negative PSMA PET/CT, whole-pelvic nodal radiotherapy improved BRFS compared to prostate-only radiotherapy.21 This finding suggests that proactive treatment of pelvic nodes may yield oncologic benefits. Furthermore, PLND may still play a role as a staging and stratification tool, identifying patients who could benefit from adjuvant therapies.20 However, according to the systematic review and meta-analysis by Vale et al., there is no clear evidence that adjuvant radiotherapy improves event-free survival compared to early salvage radiotherapy in men with localized or locally advanced PC.22 One limitation of this analysis is the low number of node-positive patients, making it difficult to assess the effect of adjuvant radiotherapy in this subgroup.22 Additional research is therefore warranted to better define the role of adjuvant treatment specifically in the node-positive population. Another important consideration is the diagnostic limitation of PSMA PET/CT. Despite its overall good accuracy, PSMA PET/CT may miss micrometastatic disease due to its relatively low sensitivity (around 55%).4 In PSMA PET/CT-negative patients at high risk for LNI, this may lead to under-staging and potentially under-treatment. However, it remains unclear whether micrometastases missed at PSMA PET/CT have a significant impact on patient prognosis.4 Future clinical pathways should weigh this diagnostic limitation when omitting PLND solely based on negative PSMA findings. Given these complex considerations, the integration of PSMA PET/CT into a tailored surgical approach needs further validation. Ongoing randomized controlled trials such as the PREDICT-study (NCT04269512) and NCT05109910, comparing ePLND with no PLND, are expected to provide more definitive insights into the oncological contribution of PLND.
This study is not free of limitations. A key limitation of this study is its non-randomized design. The intervention group was compared to a historical control cohort derived from a prospective registry, with patients treated approximately two years earlier. Although matching was performed to balance both groups for key baseline characteristics, the use of a historical comparator introduces the potential for temporal bias. Over the intervening period, significant developments in prostate cancer management—such as advancements in surgical techniques, refinement of adjuvant therapies, and broader availability of novel systemic treatments—may have contributed to improved survival outcomes in the intervention group. These improvements in care, which are unrelated to the performance or omission of PLND, may confound the interpretation of oncological benefit associated with the intervention. Furthermore, by including only patients in the intervention group who were staged using PSMA PET/CT, it is possible that individuals with micrometastatic disease located outside the pelvic lymph nodes and detectable only through PSMA PET/CT were excluded, whereas similar patients may have been inadvertently included in the control group due to reliance on conventional imaging. Finally, with a median follow-up of 27 months in the intervention group, our data are currently too immature to evaluate late-oncological outcomes. Extended follow-up will be crucial to validate the long-term safety and efficacy of this proposed care pathway.
Omitting ePLND in patients with unfavorable intermediate- and high-risk PC who have negative PSMA PET/CT findings (N0M0) significantly reduces the number of ePLNDs performed, thereby lowering treatment-related morbidity without compromising early oncological outcomes. However, given the relatively short follow-up duration in this study, the long-term oncological safety of omitting ePLND remains uncertain.
Acknowledgement
No acknowlegdements to declare.
Funding Statement
This research was supported by a grant from Kom op tegen Kanker (Stand Up to Cancer, Belgium).
Author Contributions
The authors confirm contribution to the paper as follows: study conception and design: Nicolaas Lumen, Kathia De Man; data collection: Eva Donck, Nicolaas Lumen, Kathia De Man, Pieter De Visschere, Sofie Verbeke; analysis and interpretation of results: Eva Donck, Nicolaas Lumen; draft manuscript preparation: Eva Donck, Nicolaas Lumen, Valérie Fonteyne, Charles Van Praet. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials
The data generated during the current study are available from the corresponding author on reasonable request.
Ethics Approval
This study was approved by the Ethics Committee of Ghent University Hospital (reference number BC-10297).
Informed Consent
Written informed consent was obtained from all individual participants included in the study.
Conflicts of Interest
The authors declare no conflicts of interest to report regarding the present study.
References
1. Hofman MS, Lawrentschuk N, Francis RJ et al. Prostate-specific membrane antigen PET-CT in patients with high-risk prostate cancer before curative-intent surgery or radiotherapy (proPSMAa prospective, randomised, multicentre study. The Lancet 2020;395(10231):1208–1216. doi:10.1016/S0140-6736(20)30314-7. [Google Scholar] [PubMed] [CrossRef]
2. Hope TA, Eiber M, Armstrong WR et al. Diagnostic accuracy of 68Ga-PSMA-11 PET for pelvic nodal metastasis detection prior to radical prostatectomy and pelvic lymph node dissection: a multicenter prospective phase 3 imaging trial. JAMA Oncol 2021;7(11):1635–1642. doi:10.1001/jamaoncol.2021.3771. [Google Scholar] [PubMed] [CrossRef]
3. Pienta KJ, Gorin MA, Rowe SP et al. A phase 2/3 prospective multicenter study of the diagnostic accuracy of prostate specific membrane antigen PET/CT with 18F-DCFPyL in prostate cancer patients (OSPREY). J Urol 2021;206(1):52–61. doi:10.1097/JU.0000000000001698. [Google Scholar] [PubMed] [CrossRef]
4. Mazzone E, Cannoletta D, Quarta L et al. A comprehensive systematic review and meta-analysis of the role of prostate-specific membrane antigen positron emission tomography for prostate cancer diagnosis and primary staging before definitive treatment. Eur Urol 2025;87(6):654–671. doi:10.1016/j.eururo.2025.03.003. [Google Scholar] [PubMed] [CrossRef]
5. Mottet N, Cornford P, van den Bergh RCN et al. EAU-EANM-ESTRO-ESUR-ISUP-SIOG guidelines on prostate cancer. In: The EAU Annual Congress Milan 2023; 2023; Arnhem, The Netherlands: EAU. p. 67–105. [Google Scholar]
6. Mottet N, van den Bergh RCN, Briers E et al. EAU-EANM-ESTRO-ESUR-SIOG guidelines on prostate cancer—2020 update. Part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol 2021;79(2):243–262. doi:10.1016/j.eururo.2020.09.042. [Google Scholar] [PubMed] [CrossRef]
7. Cornford P, van den Bergh RCN, Briers E et al. EAU-EANM-ESTRO-ESUR-ISUP-SIOG guidelines on prostate cancer—2024 update. Part I: screening, diagnosis, and local treatment with curative intent. Eur Urol 2024;86(2):148–163. doi:10.1016/j.eururo.2024.03.027. [Google Scholar] [PubMed] [CrossRef]
8. Oderda M, Diamand R, Albisinni S et al. Indications for and complications of pelvic lymph node dissection in prostate cancer: accuracy of available nomograms for the prediction of lymph node invasion. BJU Int 2021;127(3):318–325. doi:10.1111/bju.15220. [Google Scholar] [PubMed] [CrossRef]
9. Gandaglia G, Barletta F, Robesti D et al. Identification of the optimal candidates for nodal staging with extended pelvic lymph node dissection among prostate cancer patients who underwent preoperative prostate-specific membrane antigen positron emission tomography. External validation of the memorial sloan kettering cancer center and briganti nomograms and development of a novel tool. Eur Urol Oncol 2023;6(6):543–552. doi:10.1016/j.euo.2023.05.003. [Google Scholar] [PubMed] [CrossRef]
10. Vis AN, Meijer D, Roberts MJ et al. Development and external validation of a novel nomogram to predict the probability of pelvic lymph-node metastases in prostate cancer patients using magnetic resonance imaging and molecular imaging with prostate-specific membrane antigen positron emission. Eur Urol Oncol 2023;6(6):553–563. doi:10.1016/j.euo.2023.03.010. [Google Scholar] [PubMed] [CrossRef]
11. Muehlematter UJ, Schweiger L, Ferraro DA et al. Development and external validation of a multivariable [68Ga]Ga-PSMA-11 PET-based prediction model for lymph node involvement in men with intermediate or high-risk prostate cancer. Eur J Nucl Med Mol Imaging 2023;50(10):3137–3146. doi:10.1007/s00259-023-06278-1. [Google Scholar] [PubMed] [CrossRef]
12. Stabile A, Pellegrino A, Mazzone E et al. Can Negative prostate-specific membrane antigen positron emission tomography/computed tomography avoid the need for pelvic lymph node dissection in newly diagnosed prostate cancer patients? A systematic review and meta-analysis with backup histology as reference standard. Eur Urol Oncol 2022;5(1):1–17. doi:10.1016/j.euo.2021.08.001. [Google Scholar] [PubMed] [CrossRef]
13. Mitropoulos D, Artibani W, Graefen M, Remzi M, Rouprêt M, Truss M. Reporting and grading of complications after urologic surgical procedures: an ad hoc EAU guidelines panel assessment and recommendations. Eur Urol 2012;61(2):341–349. doi:10.1016/j.eururo.2011.10.033. [Google Scholar] [PubMed] [CrossRef]
14. Cacciamani GE, Maas M, Nassiri N et al. Impact of pelvic lymph node dissection and its extent on perioperative morbidity in patients undergoing radical prostatectomy for prostate cancer: a comprehensive systematic review and meta-analysis. Eur Urol Oncol 2021;4(2):134–149. doi:10.1016/j.euo.2021.02.001. [Google Scholar] [PubMed] [CrossRef]
15. Lestingi JFP, Guglielmetti GB, Trinh QD et al. Extended versus limited pelvic lymph node dissection during radical prostatectomy for intermediate- and high-risk prostate cancer: early oncological outcomes from a randomized phase 3 trial. Eur Urol 2021;79(5):595–604. doi:10.1016/j.eururo.2020.11.040. [Google Scholar] [PubMed] [CrossRef]
16. Touijer KA, Sjoberg DD, Benfante N et al. Limited versus extended pelvic lymph node dissection for prostate cancer: a randomized clinical trial. Eur Urol Oncol 2021;4(4):532–539. doi:10.1016/j.euo.2021.03.006. [Google Scholar] [PubMed] [CrossRef]
17. Touijer KA, Vertosick EA, Sjoberg DD et al. Pelvic lymph node dissection in prostate cancer: update from a randomized clinical trial of limited versus extended dissection. Eur Urol 2025;87(2):253–260. doi:10.1016/j.eururo.2024.10.006. [Google Scholar] [PubMed] [CrossRef]
18. Hagman A, Lantz A, Grannas D et al. Positive surgical margin and oncological outcomes after robot-assisted radical prostatectomy in different Cancer of the Prostate Risk Assessment risk groups. BJU Int 2025;136(1):135–142. doi:10.1111/bju.16732. [Google Scholar] [PubMed] [CrossRef]
19. Harty NJ, Kozinn SI, Canes D, Sorcini A, Moinzadeh A. Comparison of positive surgical margin rates in high risk prostate cancer: Open versus minimally invasive radical prostatectomy. Int Braz J Urol 2013;39(5):639–648. doi:10.1590/S1677-5538.IBJU.2013.05.05. [Google Scholar] [PubMed] [CrossRef]
20. Fossati N, Willemse PPM, Van den Broeck T et al. The benefits and harms of different extents of lymph node dissection during radical prostatectomy for prostate cancer: a systematic review. Eur Urol 2017;72(1):84–109. doi:10.1016/j.eururo.2016.12.003. [Google Scholar] [PubMed] [CrossRef]
21. Murthy V, Maitre P, Kannan S et al. Prostate-only versus whole-pelvic radiation therapy in high-risk and very high-risk prostate cancer (POP-RToutcomes from phase III randomized controlled trial. J Clin Oncol 2021;39(11):1234–1242. doi:10.1200/JCO.20.03282. [Google Scholar] [PubMed] [CrossRef]
22. Vale CL, Fisher D, Kneebone A et al. Adjuvant or early salvage radiotherapy for the treatment of localised and locally advanced prostate cancer: a prospectively planned systematic review and meta-analysis of aggregate data. Lancet 2020;396(10260):1422–1431. doi:10.1016/S0140-6736(20)31952-8. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools