 Open Access
Open Access
REVIEW
A comprehensive review about sperm-oocyte interactions and key activation factors of fertilization in mammals
1 Immunopathology-Immunotherapy-Immunomonitoring Laboratory, Faculty of Medicine Mohammed VI University of Sciences and Health, Casablanca, 82403, Morocco
2 In Vitro Fertilization Center IRIFIV, Iris Clinic, Labomac In Vitro Fertilization Center and Clinical Laboratory Medicine, Casablanca, 20100, Morocco
3 Reproductive Medicine and Biology, University Hospital and Faculty of Medicine and PERITOX Laboratory, Picardie Jules Verne University, CURS, Amiens, 80025, France
4 IVF Laboratory, Department of Reproductive Medicine, Mohammed VI International University Hospital, Bouskoura, 27182, Morocco
5 Mohammed VI Center for Research and Innovation (CM6RI), Rabat, 10100, Morocco
* Corresponding Author: Soukaina Azil. Email:
(This article belongs to the Special Issue: Advances and Challenges In Male Infertility: From Molecular Mechanisms to Clinical Applications)
Canadian Journal of Urology 2026, 33(1), 51-62. https://doi.org/10.32604/cju.2025.069420
Received 23 June 2025; Accepted 13 November 2025; Issue published 28 February 2026
Abstract
Mammalian fertilization involves the migration of spermatozoa through the female reproductive system. Early embryonic development is a consequence of several steps and signaling pathways being activated, as well as biochemical and morphological modifications of spermatozoa that enable them to penetrate the membrane of mature oocytes.There are some crucial steps known to clearly explain the process of fertilization, starting with hyperactivation of spermatozoa, mutual recognition, and binding of gametes mediated by receptors located on the surface membranes of both gametes. The final step is followed by oocyte activation, which is primarily triggered via sperm-derived factors, inducing a sharp increase in intracellular calcium levels, eventually leading to polyspermy block.
This review integrates current knowledge of the molecular and physiological events governing fertilization, emphasizing how ion regulation and signaling pathways converge to enable sperm function and oocyte activation. Special attention is given to sperm-derived factors such as phospholipase C zeta (PLCζ) and post-acrosomal sheath WW domain-binding protein (PAWP), which play essential roles in triggering calcium release and supporting early embryonic development.
Keywords
Mammalian reproduction represents a fundamental biological process that ensures the survival, continuity, and evolutionary success of species. It encompasses a complex series of events, beginning with the production of highly specialized gametes (spermatozoa in males and oocytes in females), each of which undergoes precise differentiation and maturation processes to acquire full functional competence. The successful fusion of these gametes is not a random event but the result of finely tuned cellular, molecular, and biochemical mechanisms that coordinate gamete recognition, binding, and membrane fusion, ultimately leading to the formation of a zygote.1
In humans, the functional integrity and interaction of gametes are critical determinants of fertilization efficiency. Human spermatozoa must acquire motility and undergo capacitation, which enables them to respond to oocyte-derived signals and prepare for the acrosomal reaction, while the oocyte provides not only structural and biochemical cues through the zona pellucida but also a regulated cytoplasmic environment that supports sperm entry and activation.2
Firstly, spermatozoa undergo hyperactivation, which is essential to reach a high level of motility and is important for penetrating the zona pellucida during contact with the oocyte. Then, the acrosomal reaction is induced by the sperm acrosome located on the spermatozoon’s head, allowing the sperm to pass through the oocyte’s extracellular matrix (Zona pellucida).3 Gametes can bind easily to other proteins located in each surface membrane of both sperm and the oolemma. At this stage, beyond physical interactions, a series of molecular and ionic signaling events are triggered, involving key factors and ion channels that regulate oocyte activation. Among these, calcium and zinc play central roles in modulating sperm function and initiating intracellular signaling cascades.4
As a consequence of this step, the head of the spermatozoon can get inside the cytoplasm of the oocyte, delivering the genetic material and specific sperm-derived activating factors such as phospholipase C zeta (PLCζ) and post-acrosomal sheath WW domain-binding protein (PAWP). These factors can initiate calcium oscillations via inositol trisphosphate (InsP3) pathways activation,5,6 through InsP3 receptors (InsP3Rs) in the endoplasmic reticulum (ER).7
Finally, oocyte activation leads to the establishment of the polyspermy block, mediated by the secretion of calcium and zinc and by modifications of the oolemma structure, which together prevent the entry of additional spermatozoa.8,9
Ionic, molecular, and hormonal regulation of sperm capacitation and motility
Sperm capacitation is a highly complex and tightly regulated physiological process that enables spermatozoa to acquire the functional competence necessary for successful fertilization. This process involves coordinated ionic exchanges across the sperm plasma membrane, activation of multiple intracellular molecular pathways, and modulation by hormonal signals originating from the female reproductive tract. Among the most critical factors regulating capacitation are the divalent ions calcium (Ca2+) and zinc (Zn2+), which act both as signaling molecules and structural cofactors, influencing enzymatic activity, membrane fluidity, and motility patterns. These ions interact with key sperm-specific ion channels, including the Cation channel of Sperm (CatSper), which mediates calcium influx in response to physiological stimuli; the Voltage-gated proton channel 1 (Hv1), which modulates intracellular pH; the Large-conductance calcium-activated potassium channel 1 (SLO1), and the Sperm-specific potassium channel 3 (SLO3), both of which contribute to hyperpolarization of the sperm membrane. The coordinated activity of these channels regulates intracellular ionic concentrations, membrane potential, and sperm hyperactivation, ultimately ensuring that spermatozoa can efficiently navigate the female reproductive tract and respond to oocyte-derived signals.10
In addition, hormonal factors such as progesterone, secreted by cumulus cells surrounding the oocyte, play a pivotal role in amplifying intracellular signaling cascades. Progesterone binds to sperm receptors and enhances the activity of CatSper channels, triggering localized calcium influx and promoting the acrosomal reaction, a necessary step for penetration of the zona pellucida. The interplay between ionic regulation, channel activity, and hormonal modulation highlights the intricate control mechanisms that govern sperm capacitation. Understanding these processes in detail not only elucidates the fundamental biology of fertilization but also provides critical insights for improving assisted reproductive technologies (ARTs), such as in vitro fertilization (IVF) and intracytoplasmic sperm injection (ICSI), by identifying potential targets for enhancing sperm function and fertilization competence.
Zinc (Zn) is considered an important nutrient for stimulating human spermatogenesis and an indicator of the sperm capacitation state. Several studies have demonstrated a correlation between abnormal semen parameters and altered zinc levels in seminal plasma, with significantly higher concentrations of zinc observed in fertile men compared to infertile men.11,12
The capacitation and hyperactivation of the spermatozoa are important for fertilizing the oocyte, including the increase of sperm membrane fluidity and increasing movements of cholesterol, first one at the tip of the sperm head, the second one within the plasma membrane of the spermatozoa.13 According to some studies, a higher concentration of Zn is expressed in sperm flagella, mitochondria, and spermatids.14 This evidence highlights the importance of zinc levels in spermatogenesis, sperm quality, and the fertilizing ability of spermatozoa. Zinc deficiency contributes to infertility, especially abnormal testicular development, altered spermatogenesis, hormone deficiency, inflammation, secretion of reactive oxygen species, and apoptosis.15 Zinc levels have also been reported to be correlated with hypogonadism, cell damage such as Leydig cells and seminiferous tubules alteration.16,17 Zinc can also play a crucial role in anti-inflammatory function, inducing a significant decrease in Tumor necrosis factor-alpha (TNF-α) expression.16
The elevated Zn2+ concentration negatively regulates the Voltage-gated proton channel 1 (Hv1), responsible for increasing intracellular pH to facilitate the entry of Ca2+ via CatSper. The penetration of calcium ions activates sperm soluble adenylate cyclase (SACY), phosphorylation of protein tyrosine (pY) and protein kinase A (PKA), while also inhibiting the activation of phosphorylating protein tyrosine phosphatase (PTP). As a consequence of this mechanism, the acrosome undergoes remodeling and exocytosis, releasing hydrolytic enzymes that modify the extracellular matrix of the oocyte, particularly the zona pellucida. This modification allows the spermatozoon to penetrate the zona pellucida and establish contact with the oocyte plasma membrane. By orchestrating these ionic and enzymatic changes, zinc contributes to the sequential progression of sperm capacitation, hyperactivation, and the acrosomal reaction, ensuring that only fully competent spermatozoa are capable of successful fertilization18 (Figure 1).
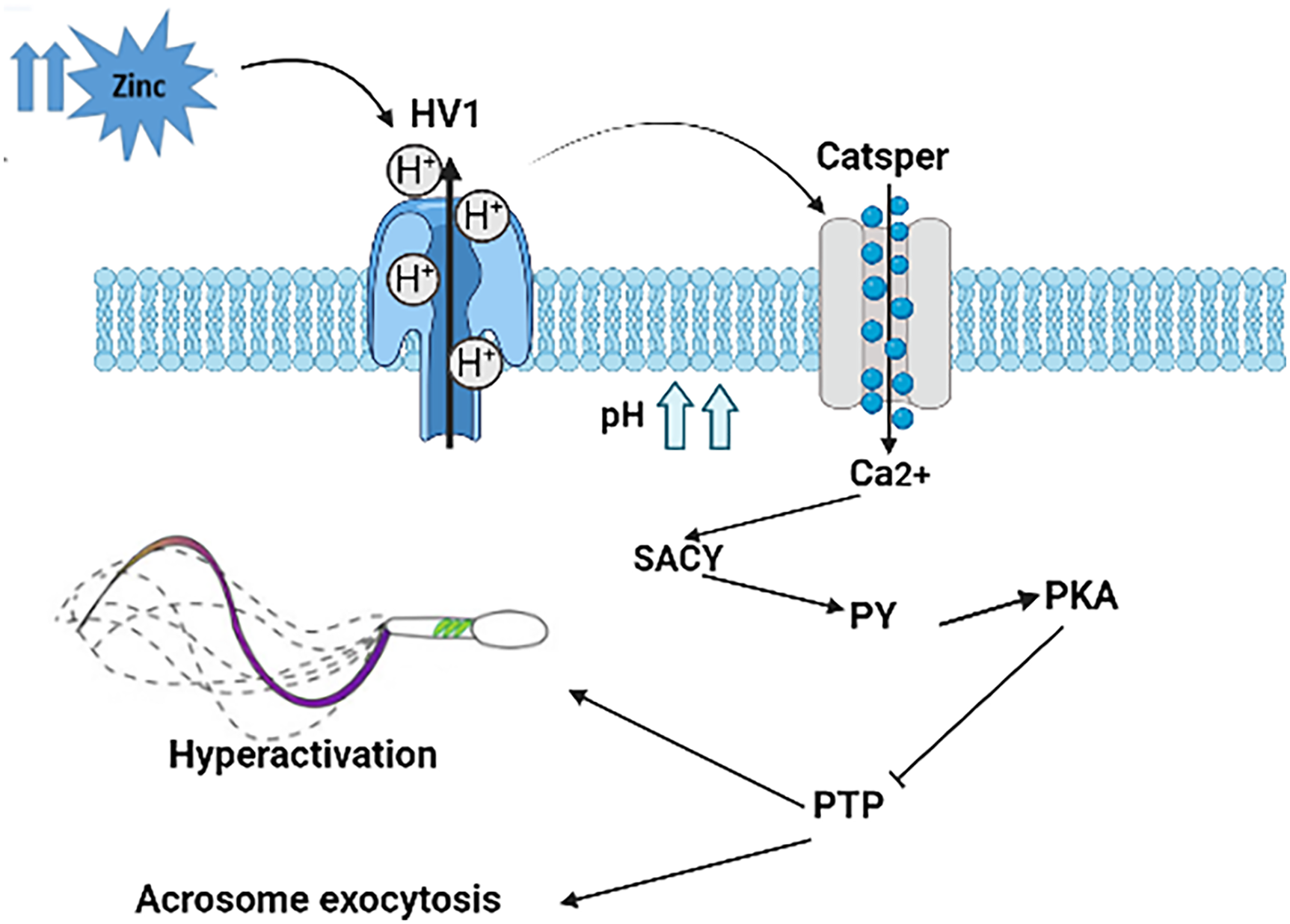
FIGURE 1. Description of mechanisms and molecular pathways involved in acrosomal exocytosis (AE) by Zn2+: The release of Zn2+ activates the secretion of H+, which influences pH; the activation of CatSper increases Ca2+ concentration, activating adenylate cyclase (SACY). The protein kinase A (PKA) is activated, followed by phosphorylation of protein tyrosine (pY), inhibiting the activation of phosphorylating protein tyrosine phosphatase (PTP), and finally, acrosome remodeling and exocytosis occur along with the hyperactivation and motility of the spermatozoa.19 Figure created by the authors using BioRender.com (Version 2024, BioRender, Toronto, ON, Canada)
Calcium ions function as critical intracellular messengers that regulate a wide range of physiological processes in spermatozoa and play a central role in male fertility. Beyond their signaling function, calcium ions are essential for progressive sperm motility, capacitation, and hyperactivation, all of which are necessary for successful navigation through the female reproductive tract and for the fertilization of the oocyte.20 A study reported a strong correlation between altered calcium levels in seminal plasma and impaired male fertility, particularly in conditions such as oligozoospermia and azoospermia, when compared to men with normal sperm parameters.21 During spermatogenesis, calcium ions contribute to the regulation of germ cell proliferation, influencing both spermatogonia and spermatocytes, and thereby supporting the production of functionally competent spermatozoa. After spermatozoa leave the epididymis, calcium continues to regulate motility by modulating flagellar beat patterns and energy metabolism, enabling sperm to respond effectively to environmental cues within the female reproductive tract.22 Experimental evidence has shown that inhibition or dysregulation of calcium signaling pathways can be a major cause of male infertility, as it impairs key events such as motility, capacitation, and hyperactivation.23
Furthermore, calcium signaling is crucial for the fine tuning of sperm hyperactivation, which enhances the mechanical force required for oocyte penetration. Proper calcium dynamics allow sperm to adjust their motility patterns in response to chemotactic signals, the biochemical composition of the reproductive tract, and interactions with the oocyte’s extracellular environment.24 Altogether, these findings highlight the central importance of calcium ions in both the development and functional activation of spermatozoa. Understanding the complex roles of calcium in spermatogenesis, sperm motility, and fertilization not only provides insight into the fundamental biology of reproduction but also offers potential avenues for diagnostic and therapeutic interventions aimed at improving male fertility. In addition, the CatSper channel induces sperm motility by controlling Ca2+ influx25 (Figure 2).
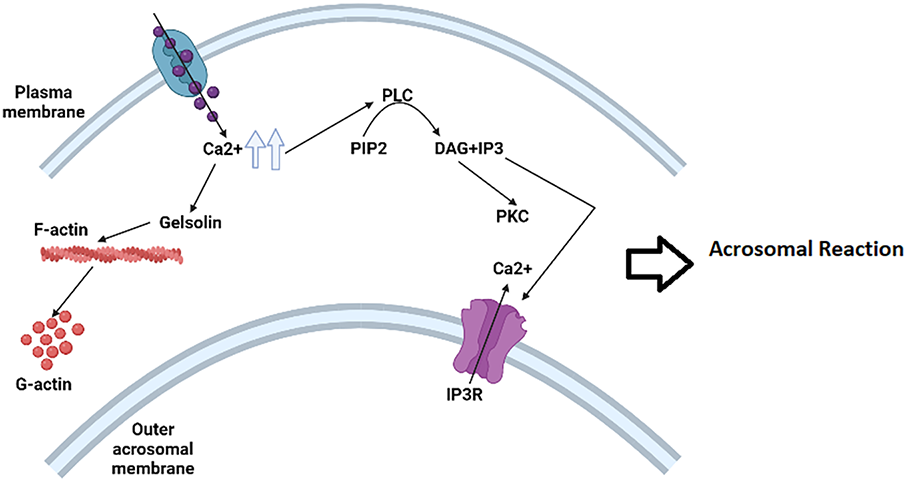
FIGURE 2. Presentation of a biochemical cascade involved in the acrosome reaction (AR); the capacitated spermatozoon binds to the zona pellucida of the oocyte, causing a fast and high increase in intraspermatozoa Ca2+ concentration. As a result, the activation of phospholipase C (PLC) catalyzes the hydrolysis of phosphatidylinositol 4,5-bisphosphate (PIP2) to produce inositol 1,4,5-trisphosphate (IP3) PLC catalyzes and diacylglycerol (DAG); finally, p-gelsolin is activated to convert F-actin to G-actin; meanwhile IP3 activates Ca2+ channel and DAG activates Protein Kinase C (PKC); all these activated molecules can finally mediate the acrosomal reaction.26 Figure created by the authors using BioRender.com (Version 2024, BioRender, Toronto, ON, Canada)
Sperm flagellum presents three functionally different parts: The principal piece, the midpiece, and the end piece. Lots of ion channels are located along the principal piece (Figure 3). The CatSper complex is considered the most essential protein for male fertility. CatSper is also considered voltage-gated, pH-sensitive, and responsive to H+ ions. The protein contains nine units: four alpha pore-forming subunits, CatSper from 1 to 4, and five auxiliary subunits: CatSper ε, β, γ, ζ, and δ. All these proteins are sperm-specific transmembrane proteins located on the surface of the sperm flagellum.26,27 These ion channels do not operate in isolation; progesterone further modulates CatSper activity, amplifying calcium signaling and coordinating with other channels to regulate sperm motility and the acrosome reaction. The involvement of progesterone is important to activate the CatSper channel in human sperm and control the effect of progesterone on hyperactivation.28 For mammalian species, sperm capacitation likely begins inside the uterus, where the concentration of progesterone is higher to facilitate the process.29 It was previously reported that the physiological role of progesterone activates the CatSper channel at 80 nM, stimulating the activation of the porcine spermatozoa incubated with in vitro cultured oviductal cells.29,30 The mutation of the CatSper gene has been clinically linked to male infertility, especially cases presenting astheno-teratozoospermia.31,32
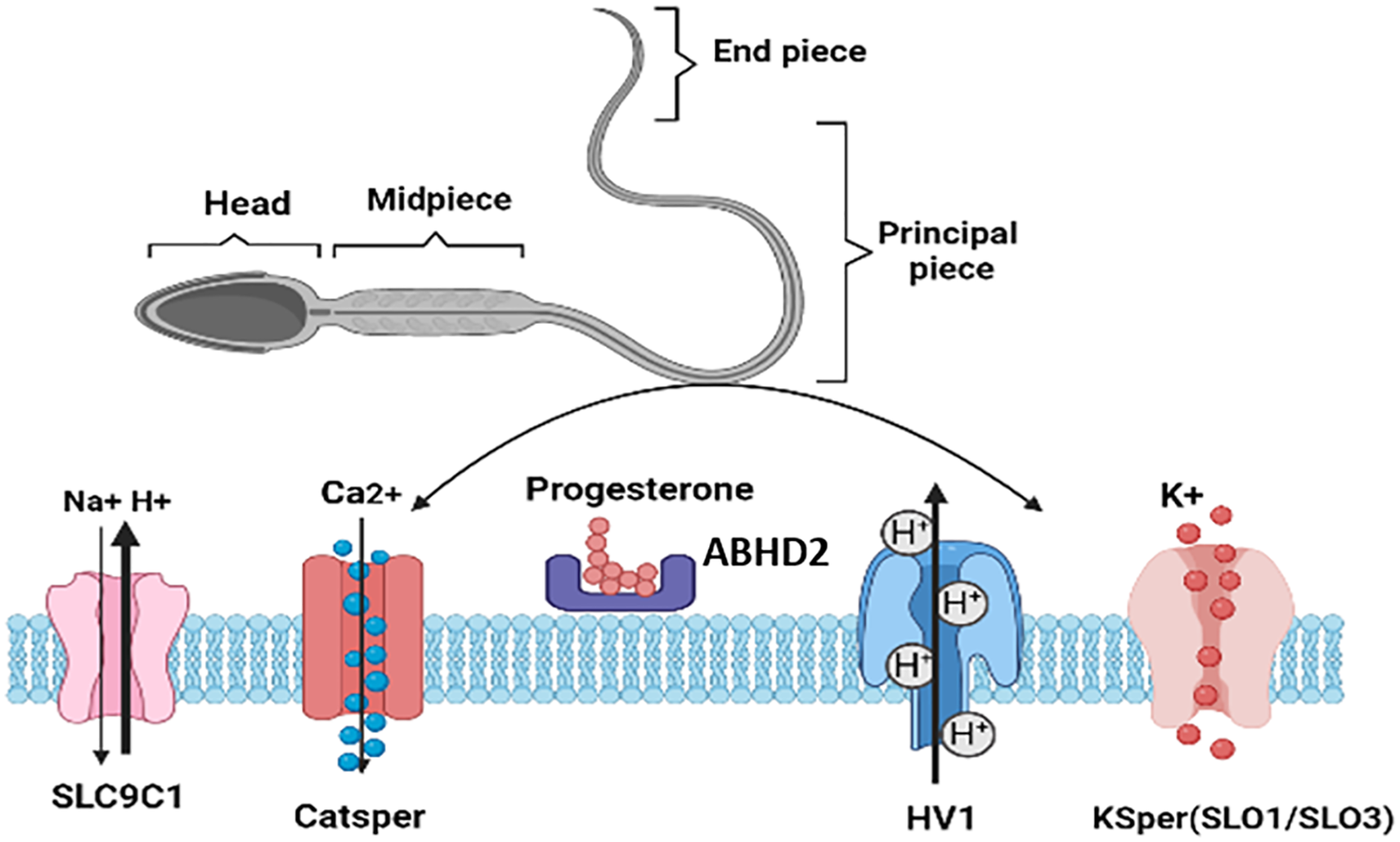
FIGURE 3. Different pathways to induce sperm capacitation and hyperactivation; Sperm Specific Sodium Proton Exchanger (SLC9C1) induces the increase of intracellular Ca2+ in the sperm tail,34 CatSper is activated by intracellular alkalization and progesterone via binding to serine hydrolase, ABHD2; Hv1 is responsible for proton conductance in human sperm and is highly selective for H+ ions, activated during sperm capacitation, membrane depolarization, and alkaline extracellular pH,31 the K+ channel is more sensitive to calcium compared to pH; SLO1 channel and the SLO3 are very sensitive to Ca2+ and weakly dependent on pH; SLO3 regulates the secretion of potassium ions responsible for capacitation and membrane hyperpolarization.42 Figure created by the authors using BioRender.com (Version 2024, BioRender, Toronto, ON, Canada)
Sodium/hydrogen exchangers (Na+/H+ exchangers) play a crucial role in regulating intracellular pH and maintaining ionic balance in spermatozoa. These exchangers are encoded by the NHE gene family, which has been shown to be expressed across multiple species, including rats, mice, and humans. Among these, several isoforms have been identified, such as NHE1, NHE5, and NHE10, each contributing to distinct aspects of cellular homeostasis. Of particular importance in sperm physiology is SLC9C1, a sperm-specific Na+/H+ exchanger that acts as a key gatekeeper in mediating chemotactic responses and guiding sperm toward the oocyte.33
The regulation of calcium concentration within the flagellum represents another critical aspect of sperm activation. Elevated Ca2+ levels can induce conformational changes in inhibitory molecules that normally block specific ion channels. This conformational change causes the dissociation of the blocking molecules, thereby opening the channel and allowing calcium to flow into the sperm. The subsequent calcium influx initiates a cascade of intracellular events that enhance flagellar motility, facilitate hyperactivation, and prime the sperm for processes such as capacitation and acrosomal reaction. Ca2+ concentration in the flagellum can also induce a conformational change in the blocker molecule, leading to the dissociation of the protein that opens the CatSper channel, consequently facilitating Ca2+ release.34
The Hv1 channel is also located on the principal piece of the spermatozoa’s tail, responsible for proton conductance in human sperm and highly selective for H+ ions. The channel is activated during sperm capacitation, membrane depolarization, and alkaline extracellular pH.35,36 A mutation in the Hv1 gene appears not to affect male fertility, as demonstrated by experiments showing that mice lacking the Hv1 channel (Hv1−/−) still produce normal spermatozoa. Although it has been confirmed that Hv1 is responsible for pH control for human spermatozoa, the participation of this channel in the increase of pH during capacitation of spermatozoa has not been discussed yet.36,37
The hyperpolarization is physiologically characterized by an increase in K+ permeability, which also facilitates sperm capacitation.10 The K+ channel is essentially a combination of members of the Slo family: Slo1 is extremely conserved, in contrast to Slo3, which is specifically expressed in mammalian sperm and presents low conservation.38,39 In human sperm, the K+ channel is more sensitive to calcium than to pH and is inhibited by progesterone.40 However, it is indeed clear that the capacitated sperm has a different type of SLO channel, and SLO3 is specifically sensitive to Ca2+.40 To demonstrate the importance of this channel during the capacitation, López-González et al. have found a similarity with the human sperm with inhibitors of both SLO1 and SLO3.41
Calcium channel blockers (CCBs)
Calcium homeostasis and its precise regulation are essential for proper sperm function and, ultimately, for successful fertilization. Calcium ions act as pivotal intracellular messengers, controlling processes such as sperm motility, capacitation, hyperactivation, and acrosomal reaction. Disruption of calcium signaling can therefore profoundly affect male reproductive potential. The use of calcium channel blockers (CCBs) has provided critical insights into these molecular mechanisms by demonstrating how interference with calcium entry or movement within spermatozoa can alter their physiological functions. CCBs are commonly used amlodipine, diltiazem, nicardipine, nifedipine, and verapamil, and they have different effects on cardiac muscle, smooth muscle of blood vessels, and neurons.43 Within sperm cells, these agents block the influx or intracellular movement of free calcium ions, thereby interfering with calcium-dependent signaling pathways that are critical for sperm activation and fertilization.44
Notably, calcium channel blockers (CCBs) have been shown to impair male fertility in several contexts. Their use has been associated with cases of oligozoospermia and azoospermia, as well as a reduced sperm ability to fertilize oocytes, even when semen parameters appear within normal ranges.45 Experimental studies indicate that CCBs, particularly nifedipine, can reduce sperm motility and inhibit the acrosomal reaction, leading to reversible infertility. However, the impact of these drugs on assisted reproductive technologies is less clear, as some investigations have reported no significant reduction in fertilization rates during In Vitro Fertilization (IVF) treatments.46
Several data suggested that Ca2+ plays a prominent role during fertilization in diverse animal species such as mice, rats, pigs, hamsters, and bovines.47,48 Extracellular calcium is particularly essential for the acquisition of progressive sperm motility, which enables spermatozoa to navigate the female reproductive tract and reach the oocyte. The modulation of calcium availability and signaling is therefore a critical determinant of sperm functional competence. In this context, studies using mouse models have demonstrated that amlodipine, a commonly used CCB, can reduce the levels of key reproductive hormones such as testosterone, luteinizing hormone (LH), and follicle-stimulating hormone (FSH), resulting in impaired sperm quality and reduced fertility.49 Beyond hormonal regulation, antihypertensive drugs, including CCBs, may also exert negative effects on erectile function, which has been linked to endothelial dysfunction, further contributing to male reproductive challenges.50
Morakinyo et al. have demonstrated the anti-fertility effects of using different concentrations of nifedipine in male rats. CCBs appear to have a reversible negative effect on male rats. Nevertheless, nifedipine does not inhibit the pituitary-gonadal axis, implying a direct effect on sperm function and testicular physiology.51 Almeida et al. also revealed the effect of calcium antagonist amlodipine decreasing plasma follicle-stimulating hormone, testosterone levels, and a significant reduction in sperm count. The study suggests that administration of amlodipine may impair male reproductive function in rats.52
The union of a mature oocyte and a capacitated spermatozoon represent one of the most critical and tightly regulated events in successful fertilization. This process is not merely a mechanical fusion of gametes but involves a highly coordinated sequence of molecular interactions, structural rearrangements, and biochemical modifications. Initially, the sperm must undergo a series of preparatory events, beginning with capacitation, which confers on the sperm the ability to respond to oocyte-derived signals and to undergo the acrosomal reaction (AR). The AR is an essential and finely regulated physiological process in which the outer acrosomal membrane of the spermatozoon fuses with the plasma membrane, resulting in the release of hydrolytic enzymes. These enzymes, including acrosin and hyaluronidase, play a crucial role in digesting the zona pellucida, the glycoprotein-rich extracellular matrix surrounding the oocyte, thereby facilitating sperm penetration. Following capacitation and AR, the spermatozoon engages in molecular fixation with the oocyte, a process that involves the specific recognition and binding of sperm surface proteins to complementary receptors on the oocyte plasma membrane. This step is accompanied by membrane apposition, lipid reorganization, and localized changes in membrane fluidity, all of which are critical for the subsequent fusion of the sperm and oocyte membranes. The precise modulation of these plasma membrane lipids and associated proteins ensures that only a capacitated and acrosome-reacted sperm can successfully penetrate the oocyte. Ultimately, these complex events culminate in the sperm nucleus entering the oocyte cytoplasm, completing fertilization, and initiating the cascade of molecular and cellular processes required for zygote formation and early embryonic development.53,54
Spermatozoa have the ability to bind specifically to the zona pellucida of an unfertilized oocyte, a process that is essential for the recognition and successful fertilization of the oocyte. The zona pellucida itself is a specialized extracellular matrix that surrounds the oocyte and is primarily composed of four glycoproteins: ZP1, ZP2, ZP3, and ZP4. Each of these glycoproteins has a distinct structural and genetic organization, which contributes to the overall architecture and function of the zona pellucida. Human Zona Pellucida glycoprotein 1 (hZP1) is encoded by 12 exons and consists of approximately 638 amino acids, forming a polypeptide that participates in cross-linking the matrix. ZP2, encoded by 19 exons, presents a longer polypeptide of 745 amino acids, playing a crucial role in maintaining the structural integrity of the zona and in sperm binding. Human ZP3, encoded by 8 exons, consists of 424 amino acids, and is known for its involvement in the initial recognition and binding of sperm. Finally, ZP4, encoded by 13 exons, contains 540 amino acids and contributes to both the mechanical stability and functional properties of the zona pellucida.55–57
ZP2 and ZP3 are expressed in equal amounts within the zona pellucida, while ZP1 and ZP4 are homologs and present the least abundant proteins.57 The hZP1 is composed of a 17% proline (Pro)-rich N-terminal region, while hZP4 presents only 2% of Pro; this Pro-rich region gives flexibility, contributing to a higher resiliency of the ZP before fertilization.58–60 N-terminal proteolytic cleavage of hZP2 compromises the spermatozoa’s binding ability to the oocyte. Several mutations were identified, for hZP1 encoded by nine exons, four exons encoding both hZP2 and hZP3, and two exons encoding hZP4.61–62
The acrosome is an acidic vesicle of lysosomal origin located in the sperm head that accumulates Ca2+ and contains several hydrolytic enzymes released through exocytosis during the acrosomal reaction.63 The interaction between capacitated sperm and zona pellucida induces an increase in Ca2+ concentration, leading to the acrosomal reaction (AR).64,65 The catalyzation of phosphatidylinositol 4,5-bisphosphate (PIP2) produces IP3, diacylglycerol (DAG) and the release of p-gelsolin, which stimulates the conversion of F-actin to G-actin, thus activating the Ca2+ channel in the acrosome membrane to finally mediate the acrosomal reaction. Some studies have confirmed that phospholipase C (PLC) requires 2 μM of Ca2+ for half maximal activity, while 80 μM of Ca2+ is needed for F-actin release from the plasma membrane, essential to achieve AR (Figure 2).66
After reaching the perivitelline space, the spermatozoon binds to the oolemma via receptors uniformly distributed on the surface of a mature oocyte, suggesting that the spermatozoon’s head can bind to any site on the oocyte surface, without forgetting the existence of microdomains facilitating sperm adherence, as fertilization sits “hot spots” on the oocyte membrane. Only three proteins were proven to be crucial for gamete interaction: CD9, IZUMO, and Juno.67
CD9 has a specific role in the fertilization process, located on the oolemma of the mature oocyte and is required for the normal morphology of microvilli.68,69 This protein is involved in gamete fusion. The study of Jégou et al. independently reported the relationship between CD9 and the tight sperm-oocyte fusion.70
IZUMO is an important element of the fertilization expressed on the acrosomal membrane of each spermatozoon. Some studies report that Izumo synthesis takes place after the acrosomal reaction and is secreted from the acrosome, moving to the equatorial segment of the sperm head, where gamete fusion occurs.71 There is some clinical data supporting its implication in idiopathic infertility.72 Mice models presenting IZUMO−/− males, which lack the IZUMO protein essential for sperm-oocyte fusion, are completely sterile despite normal sperm quality. Mutant spermatozoa can get through ZP but fail to fuse with the oocyte.73
Bianchi and colleagues reported the importance of Juno as a principal receptor of IZUMO located on the oolemma.74 This protein is expressed throughout oocyte maturation, precisely in the oolemma and is present in unfertilized mature oocytes.75 In humans, some mutations of this receptor have been related to unexplained fertilization failure.76,77 The binding of IZUMO and Juno is important to facilitate membrane adhesion, and still not sufficient, indicating the requirement for the accumulation of CD9 after the binding of IZUMO and Juno.73
The maturation of the acrosome can be defective in some cases due to mutation of DPY19-like 2 (DPY19L2), a key causative factor associated with human globozoospermia, characterized by the presence of 100% round-headed spermatozoa.78 Spermatozoa obtained from patients with ICSI failure were unable to induce calcium oscillations. Most studies found that several cases could be overcome by intracytoplasmic sperm injection (ICSI) following assisted oocyte activation (AOA).79 The detection of complete or partial deletion of DPY19L2 has a very important implication for diagnosis and treatment for this category of infertile men.80,81
Oocyte activation is an important event in most mammalian species that derives from several events, including cortical granule exocytosis, meiosis resumption, liberation of the second polar body, and the formation of the pronucleus (PN), and finally, the initiation of early embryo development.82,83
This activation is characterized by calcium oscillations due to the secretion of some spermatic factors into the oocyte cytoplasm, especially PLCζ, which was discovered and is expressed on the equatorial and post-acrosomal localizations of the spermatozoa.84,85 The release of sperm-derived phospholipase C zeta (PLCζ) hydrolyses phosphatidyl inositol 4,5-biphosphate (PIP2) to inositol 1,4,5-trisphosphate (IP3).5 These activated pathways lead to calcium oscillations responsible for oocyte activation.86,87
The second spermatic factor recently discovered, the post-acrosomal WW domain binding protein (PAWP), can indirectly increase calcium levels by its interaction with other proteins in the oocyte. PAWP binds to the WW1 domain of the YAP protein to activate the PLC Gamma, which subsequently hydrolyzes PIP2 into a second messenger IP3, and finally, the release of calcium concentration from the endoplasmic reticulum. Subsequently, the oocyte is activated similarly to the effect of PLCζ (Figure 4).88–90
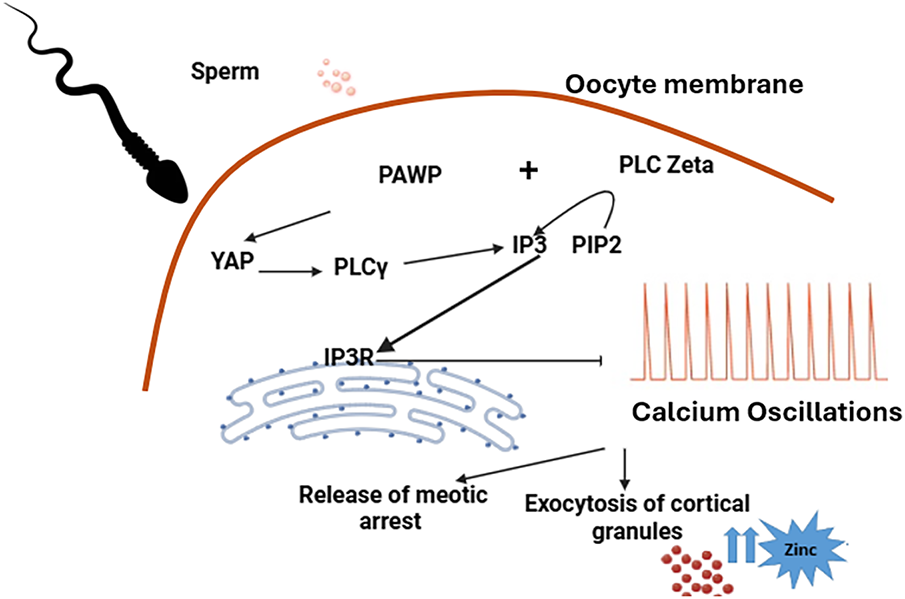
FIGURE 4. Presentation of oocyte activation mechanisms involving the importance of phospholipase C zeta and PAWP; PLCζ induces the hydrolyze of phosphatidyl inositol 4,5-biphosphate (PIP2) to produce inositol 1,4,5-trisphosphate (IP3) and diacylglycerol (DG). IP3 binds to its receptor (IP3R), which is distributed along the endoplasmic reticulum, inducing periodic intracellular Ca2+ increases; PAWP can also increase the calcium levels indirectly by its interaction with other proteins on the oocyte. PAWP binds to the WW1 domain of the YAP protein to activate the PLC Gamma, which subsequently hydrolyzes PIP2 into a second messenger IP3. The secretion of zinc is also observed following oocyte activation. These oscillations stimulate many other events such as exocytosis of cortical granules, release of meiotic arrest, regulation of gene expression, recruitment of maternal mRNA, pronuclear formation, and initiation of embryogenesis.96,97 Figure created by the authors using BioRender.com (Version 2024, BioRender, Toronto, ON, Canada)
Zinc sparks are immediately secreted after the first Ca2+ oscillations observed in human oocytes following fertilization and oocyte activation.91,92 Zinc spark controls the regulation of meiosis from the germinal vesicle (GV) to Metaphase II (MII). Some studies suggest that intracellular Zn2+ could be accumulated in cortical granules, each of which contains ~1 million Zn2+ atoms.93 The release of Zinc during oocyte activation is important to prevent some cases from undergoing ICSI in clinical practice to increase the efficacy of artificial oocyte activation (AOA), especially for calcium ionophore-resistant cases.94,95
Fertilization failure is closely linked to oocyte activation deficiency (OAD), a condition that is most frequently observed in patients diagnosed with either partial or complete globozoospermia. Globozoospermia is a rare sperm morphological disorder characterized by round-headed spermatozoa that lack an acrosome, the specialized organelle essential for normal fertilization. Individuals with this condition typically face severe challenges in achieving natural conception because their spermatozoa are incapable of initiating the cascade of molecular events required for successful oocyte activation. The underlying mechanism is often associated with significantly reduced levels or a complete absence of phospholipase C zeta (PLCζ), a sperm-specific protein crucial for triggering calcium oscillations within the oocyte upon sperm entry. PLCζ functions by hydrolyzing phosphatidylinositol 4,5-bisphosphate (PIP2) to generate inositol 1,4,5-trisphosphate (IP3), which in turn mobilizes calcium from intracellular stores in the oocyte. These calcium oscillations are essential for resuming meiosis, cortical granule exocytosis, and the initiation of zygotic development.90
After the fusion of gametes, the interaction between sperm and oocyte leads to their transformation into a new diploid organism.98 Following fertilization, the oocyte acquires mechanisms to prevent additional spermatozoa from fusing with it, thereby avoiding the generation of polyploid embryos, which would be non-viable. This critical regulation involves modifications to both the zona pellucida (ZP) and the oolemma, rendering them temporarily resistant to further sperm penetration. The block to polyspermy is mediated by two distinct mechanisms. The first, known as the “oocyte membrane block” or “fast block,” occurs within seconds after sperm-oocyte fusion and is primarily characterized by rapid depolarization of the oocyte membrane, triggered by an influx of Na+ ions. This swift alteration in membrane potential effectively prevents additional sperm from fusing with the oocyte during the initial moments of fertilization.99
The second mechanism, referred to as the “slow block,” is activated minutes after the first sperm has entered the oocyte and involves oscillatory increases in intracellular Ca2+ concentration. These calcium oscillations, generated by the activation of phospholipase C zeta (PLCζ) introduced by the sperm, lead to the exocytosis of cortical granules from the oocyte membrane into the perivitelline space. The cortical granules, which are approximately 1 μm in diameter, are rich in hydrolytic enzymes including hydrolases, proteinases, and peroxidases. These enzymes modify the structural components of the human zona pellucida, particularly hZP2 and hZP3, thereby preventing the binding and penetration of additional spermatozoa. Cortical granule exocytosis typically occurs within 5 to 8 min after oocyte activation and represents the primary mechanism of polyspermy block in humans.100
In addition to these well-characterized mechanisms, other molecular factors may contribute to the regulation of polyspermy, including the release of soluble factors from the oocyte, changes in the composition and glycosylation of the zona pellucida, and the temporal modulation of receptor availability on the oolemma. Understanding these intricate events is not only essential for elucidating the fundamental biology of fertilization but also has practical implications for improving Assisted Reproductive Technologies (ARTs). For instance, precise timing of sperm-oocyte interaction in vitro, manipulation of Ca2+ signaling pathways, and modulation of zona pellucida properties can all influence fertilization efficiency and embryo quality. Detailed insights into the fast and slow blocks provide valuable information for clinicians and embryologists seeking to optimize in vitro fertilization (IVF) and intracytoplasmic sperm injection (ICSI) protocols, minimize the risk of polyspermy, and improve the overall success rates of ART procedures.
All these activated pathways and molecular events play critical roles in both the female and male reproductive tracts, contributing to the production of high-quality embryos capable of successful development. Key ions such as calcium and zinc exert pivotal regulatory effects on fertilization by modulating several essential sperm functions, including motility, capacitation, hyperactivation, and acrosomal reaction. Calcium signaling, mediated through channels and intracellular stores, orchestrates precise temporal and spatial events within the sperm, whereas zinc acts both as a structural cofactor and as a signaling molecule influencing enzymatic activity and membrane stabilization. Although the precise interactions between these ions and sperm molecular machinery remain incompletely understood, further studies exploring the dynamic interplay between calcium and zinc concentrations, sperm functional parameters, and the involvement of specific proteins such as PLCζ, PAWP, IZUMO, and CatSper are critical. Such investigations could not only clarify the mechanistic basis of sperm–oocyte recognition and fusion but also identify potential biomarkers for sperm quality and fertilization competence. Enhancing our understanding of these processes can directly impact the optimization of Assisted Reproductive Technologies (ARTs), improving the selection of gametes, the design of culture conditions, and strategies to overcome fertilization failure. Furthermore, insights into ion-mediated regulation of sperm physiology could facilitate the development of novel diagnostic tools for male infertility and lead to personalized interventions aimed at increasing the success rates of in vitro fertilization (IVF) and intracytoplasmic sperm injection (ICSI) procedures. Ultimately, a deeper comprehension of the molecular and ionic mechanisms governing fertilization will contribute to more effective reproductive therapies and a better overall understanding of human fertility.
Acknowledgement
Not applicable.
Funding Statement
The authors received no specific funding for this study.
Author Contributions
The authors confirm contribution to the paper as follows: Conceptualization, Soukaina Azil and Moncef Benkhalifa; methodology, Soukaina Azil; software, Soukaina Azil; validation, Bouchra Ghazi, Soukaina Azil, Yassmine Louanjli, Noureddine Louanjli and Moncef Benkhalifa; formal analysis, Soukaina Azil; investigation, Soukaina Azil; resources, Soukaina Azil; data curation, Soukaina Azil; writing—original draft preparation, Soukaina Azil; writing—review and editing, Bouchra Ghazi; visualization, Soukaina Azil; supervision, Bouchra Ghazi and Moncef Benkhalifa; project administration, Bouchra Ghazi. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials
Not applicable.
Ethics Approval
Not applicable.
Conflicts of Interest
The authors declare no conflicts of interest to report regarding the present study.
References
1. Palumbi SR. Speciation and the evolution of gamete recognition genes: pattern and process. Heredity 2009;102(1):66–76. doi:10.1038/hdy.2008.104. [Google Scholar] [PubMed] [CrossRef]
2. Webb SE, Miller AL. Calcium signalling during embryonic development. Nat Rev Mol Cell Biol 2003;4(7):539–551. doi:10.1038/nrm1149. [Google Scholar] [PubMed] [CrossRef]
3. Hirohashi N, Yanagimachi R. Sperm acrosome reaction: its site and role in fertilization. Biol Reprod 2018;99(1):127–133. doi:10.1093/biolre/ioy045. [Google Scholar] [PubMed] [CrossRef]
4. Sugita H, Takarabe S, Kageyama A, Kawata Y, Ito J. Molecular mechanism of oocyte activation in mammals: past, present, and future directions. Biomolecules 2024;14(3):359. doi:10.3390/biom14030359. [Google Scholar] [PubMed] [CrossRef]
5. Kashir J, Nomikos M, Lai FA, Swann K. Sperm-induced Ca2+ release during egg activation in mammals. Biochem Biophys Res Commun 2014;450(3):1204–1211. doi:10.1016/j.bbrc.2014.04.078. [Google Scholar] [PubMed] [CrossRef]
6. Zafar MI, Lu S, Li H. Sperm-oocyte interplay: an overview of spermatozoon’s role in oocyte activation and current perspectives in diagnosis and fertility treatment. Cell Biosci 2021;11(1):4. doi:10.1186/s13578-020-00520-1. [Google Scholar] [PubMed] [CrossRef]
7. Putney JW, Steinckwich-Besançon N, Numaga-Tomita T et al. The functions of store-operated calcium channels. Biochim Biophys Acta Mol Cell Res 2017;1864(6):900–906. doi:10.1016/j.bbamcr.2016.11.028. [Google Scholar] [PubMed] [CrossRef]
8. Evans JP. Preventing polyspermy in mammalian eggs—contributions of the membrane block and other mechanisms. Mol Reprod Dev 2020;87(3):341–349. doi:10.1002/mrd.23331. [Google Scholar] [PubMed] [CrossRef]
9. Florman HM. 101 on: how we understand polyspermy functions and defenses after E.E. Just’s 1919 study. Mol Reprod Dev 2020;87(3):323–325. doi:10.1002/mrd.23326. [Google Scholar] [PubMed] [CrossRef]
10. Pinto FM, Odriozola A, Candenas L, Subirán N. The role of sperm membrane potential and ion channels in regulating sperm function. Int J Mol Sci 2023;24(8):6995. doi:10.3390/ijms24086995. [Google Scholar] [PubMed] [CrossRef]
11. Kothari RP, Chaudhari AR. Zinc levels in seminal fluid in infertile males and its relation with serum free testosterone. J Clin Diagn Res 2016;10(5):CC05–CC08. doi:10.7860/JCDR/2016/14393.7723. [Google Scholar] [PubMed] [CrossRef]
12. Colagar AH, Marzony ET, Chaichi MJ. Zinc levels in seminal plasma are associated with sperm quality in fertile and infertile men. Nutr Res 2009;29(2):82–88. doi:10.1016/j.nutres.2008.11.007. [Google Scholar] [PubMed] [CrossRef]
13. Aitken RJ, Nixon B. Sperm capacitation: a distant landscape glimpsed but unexplored. Mol Hum Reprod 2013;19(12):785–793. doi:10.1093/molehr/gat067. [Google Scholar] [PubMed] [CrossRef]
14. Yamaguchi S, Miura C, Kikuchi K et al. Zinc is an essential trace element for spermatogenesis. Proc Natl Acad Sci U S A 2009;106(26):10859–10864. doi:10.1073/pnas.0900602106. [Google Scholar] [PubMed] [CrossRef]
15. De Jesús SM, Vigueras-Villaseñor RM, Cortés-Barberena E et al. Zinc and its impact on the function of the testicle and epididymis. Int J Mol Sci 2024;25(16):8991. doi:10.3390/ijms25168991. [Google Scholar] [PubMed] [CrossRef]
16. Beigi Harchegani A, Dahan H, Tahmasbpour E, Kaboutaraki Bakhtiari H, Shahriary A. Effects of zinc deficiency on impaired spermatogenesis and male infertility: the role of oxidative stress, inflammation and apoptosis. Hum Fertil 2020;23(1):5–16. doi:10.1080/14647273.2018.1494390. [Google Scholar] [PubMed] [CrossRef]
17. Fallah A, Mohammad-Hasani A, Colagar AH. Zinc is an essential element for male fertility: a review of Zn roles in men’s health, germination, sperm quality, and fertilization. J Reprod Infertil 2018;19(2):69–81. [Google Scholar] [PubMed]
18. Kerns K, Zigo M, Sutovsky P. Zinc: a necessary ion for mammalian sperm fertilization competency. Int J Mol Sci 2018;19(12):4097. doi:10.3390/ijms19124097. [Google Scholar] [PubMed] [CrossRef]
19. Kerns K, Morales P, Sutovsky P. Regulation of sperm capacitation by the 26S proteasome: an emerging new paradigm in spermatology. Biol Reprod 2016;94(5):117. doi:10.1095/biolreprod.115.136622. [Google Scholar] [PubMed] [CrossRef]
20. Darszon A, Nishigaki T, Wood C, Treviño CL, Felix R, Beltrán C. Calcium channels and Ca2+ fluctuations in sperm physiology. Int Rev Cytol 2005;243:79–172. doi:10.1016/S0074-7696(05)43002-8. [Google Scholar] [PubMed] [CrossRef]
21. Fafula RV, Meskalo ОI, Lychkovskyy EI, Vorobets ZD. ATP-induced changes in [Ca2+]i in spermatozoa of infertile men with oligo- and asthenozoospermia. Biol Stud 2018;12(3–4):47–54. doi:10.30970/sbi.1203.580. [Google Scholar] [CrossRef]
22. Darszon A, Nishigaki T, Beltran C, Treviño CL. Calcium channels in the development, maturation, and function of spermatozoa. Physiol Rev 2011;91(4):1305–1355. doi:10.1152/physrev.00028.2010. [Google Scholar] [PubMed] [CrossRef]
23. Espino J, Mediero M, Lozano GM et al. Reduced levels of intracellular calcium releasing in spermatozoa from asthenozoospermic patients. Reprod Biol Endocrinol 2009;7(1):11. doi:10.1186/1477-7827-7-11. [Google Scholar] [PubMed] [CrossRef]
24. Ho HC, Suarez SS. Hyperactivation of mammalian spermatozoa: function and regulation. Reproduction 2001;122(4):519–526. doi:10.1530/rep.0.1220519. [Google Scholar] [PubMed] [CrossRef]
25. Ren D, Navarro B, Perez G et al. A sperm ion channel required for sperm motility and male fertility. Nature 2001;413(6856):603–609. doi:10.1038/35098027. [Google Scholar] [PubMed] [CrossRef]
26. Fukami K, Yoshida M, Inoue T et al. Phospholipase Cδ4 is required for Ca2+ mobilization essential for acrosome reaction in sperm. J Cell Biol 2003;161(1):79–88. doi:10.1083/jcb.200210057. [Google Scholar] [PubMed] [CrossRef]
27. Chung JJ, Navarro B, Krapivinsky G, Krapivinsky L, Clapham DE. A novel gene required for male fertility and functional CATSPER channel formation in spermatozoa. Nat Commun 2011;2(1):153. doi:10.1038/ncomms1153. [Google Scholar] [PubMed] [CrossRef]
28. Strünker T, Goodwin N, Brenker C et al. The CatSper channel mediates progesterone-induced Ca2+ influx in human sperm. Nature 2011;471(7338):382–386. doi:10.1038/nature09769. [Google Scholar] [PubMed] [CrossRef]
29. De Jonge C. Biological basis for human capacitation-revisited. Hum Reprod Update 2017;23(3):289–299. doi:10.1093/humupd/dmw048. [Google Scholar] [PubMed] [CrossRef]
30. Lamy J, Liere P, Pianos A, Aprahamian F, Mermillod P, Saint-Dizier M. Steroid hormones in bovine oviductal fluid during the estrous cycle. Theriogenology 2016;86(6):1409–1420. doi:10.1016/j.theriogenology.2016.04.086. [Google Scholar] [PubMed] [CrossRef]
31. Williams HL, Mansell S, Alasmari W et al. Specific loss of CatSper function is sufficient to compromise fertilizing capacity of human spermatozoa. Hum Reprod 2015;30(12):2737–2746. doi:10.1093/humrep/dev243. [Google Scholar] [PubMed] [CrossRef]
32. Bhilawadikar R, Zaveri K, Mukadam L et al. Levels of Tektin 2 and CatSper 2 in normozoospermic and oligoasthenozoospermic men and its association with motility, fertilization rate, embryo quality and pregnancy rate. J Assist Reprod Genet 2013;30(4):513–523. doi:10.1007/s10815-013-9972-6. [Google Scholar] [PubMed] [CrossRef]
33. Windler F, Bönigk W, Körschen HG et al. The solute carrier SLC9C1 is a Na+/H+-exchanger gated by an S4-type voltage-sensor and cyclic-nucleotide binding. Nat Commun 2018;9(1):2809. doi:10.1038/s41467-018-05253-x. [Google Scholar] [PubMed] [CrossRef]
34. Hwang JY, Mannowetz N, Zhang Y et al. Dual sensing of physiologic pH and calcium by EFCAB9 regulates sperm motility. Cell 2019;177(6):1480–1494.e19. doi:10.1016/j.cell.2019.03.047. [Google Scholar] [PubMed] [CrossRef]
35. Musset B, Decoursey T. Biophysical properties of the voltage gated proton channel HV1. Wiley Interdiscip Rev Membr Transp Signal 2012;1(5):605–620. doi:10.1002/wmts.55. [Google Scholar] [PubMed] [CrossRef]
36. Dudev T, Musset B, Morgan D et al. Selectivity mechanism of the voltage-gated proton channel, HV1. Sci Rep 2015;5(1):10320. doi:10.1038/srep10320. [Google Scholar] [PubMed] [CrossRef]
37. Ramsey IS, Ruchti E, Kaczmarek JS, Clapham DE. Hv1 proton channels are required for high-level NADPH oxidase-dependent superoxide production during the phagocyte respiratory burst. Proc Natl Acad Sci U S A 2009;106(18):7642–7647. doi:10.1073/pnas.0902761106. [Google Scholar] [PubMed] [CrossRef]
38. Santi CM, Martínez-López P, de la Vega-Beltrán JL et al. The SLO3 sperm-specific potassium channel plays a vital role in male fertility. FEBS Lett 2010;584(5):1041–1046. doi:10.1016/j.febslet.2010.02.005. [Google Scholar] [PubMed] [CrossRef]
39. Miller MR, Mansell SA, Meyers SA, Lishko PV. Flagellar ion channels of sperm: similarities and differences between species. Cell Calcium 2015;58(1):105–113. doi:10.1016/j.ceca.2014.10.009. [Google Scholar] [PubMed] [CrossRef]
40. Brenker C, Zhou Y, Müller A et al. The Ca2+-activated K+ current of human sperm is mediated by Slo3. eLife 2014;3:e01438. doi:10.7554/eLife.01438. [Google Scholar] [PubMed] [CrossRef]
41. López-González I, Torres-Rodríguez P, Sánchez-Carranza O et al. Membrane hyperpolarization during human sperm capacitation. Mol Hum Reprod 2014;20(7):619–629. doi:10.1093/molehr/gau029. [Google Scholar] [PubMed] [CrossRef]
42. Lyon M, Li P, Ferreira JJ et al. A selective inhibitor of the sperm-specific potassium channel SLO3 impairs human sperm function. Proc Natl Acad Sci U S A 2023;120(4):e2212338120. doi:10.1073/pnas.2212338120. [Google Scholar] [PubMed] [CrossRef]
43. Elliott WJ, Ram CVS. Calcium channel blockers: calcium channel blockers. J Clin Hypertens 2011;13(9):687–689. doi:10.1111/j.1751-7176.2011.00513.x. [Google Scholar] [PubMed] [CrossRef]
44. Godfraind T. Discovery and development of calcium channel blockers. Front Pharmacol 2017;8:286. doi:10.3389/fphar.2017.00286. [Google Scholar] [PubMed] [CrossRef]
45. Benoff S, Cooper GW, Hurley I et al. The effect of calcium ion channel blockers on sperm fertilization potential. Fertil Steril 1994;62(3):606–617. doi:10.1016/s0015-0282(16)56953-2. [Google Scholar] [CrossRef]
46. Allard-Phillips E, Kolli S, Rhoton-Vlasak A, Campbell K. Systematic review of the impact of calcium channel blockers on sperm function. Reprod Toxicol 2025;132(4):108841. doi:10.1016/j.reprotox.2025.108841. [Google Scholar] [PubMed] [CrossRef]
47. Kashir J, Deguchi R, Jones C, Coward K, Stricker SA. Comparative biology of sperm factors and fertilization-induced calcium signals across the animal kingdom. Mol Reprod Dev 2013;80(10):787–815. doi:10.1002/mrd.22222. [Google Scholar] [PubMed] [CrossRef]
48. Wakai T, Vanderheyden V, Fissore RA. Ca2+ signaling during mammalian fertilization: requirements, players, and adaptations. Cold Spring Harb Perspect Biol 2011;3(4):a006767. doi:10.1101/cshperspect.a006767. [Google Scholar] [PubMed] [CrossRef]
49. Ghomeshi A, Yang B, Masterson TA. The adverse effects of commonly used medications on male fertility: a comprehensive review. FS Rev 2023;4(3):176–186. doi:10.1016/j.xfnr.2023.08.001. [Google Scholar] [CrossRef]
50. Giuliano FA, Leriche A, Jaudinot EO, de Gendre AS. Prevalence of erectile dysfunction among 7689 patients with diabetes or hypertension, or both. Urology 2004;64(6):1196–1201. doi:10.1016/j.urology.2004.08.059. [Google Scholar] [PubMed] [CrossRef]
51. Morakinyo AO, Iranloye BO, Adegoke OA. Antireproductive effect of calcium channel blockers on male rats. Reprod Med Biol 2009;8(3):97–102. doi:10.1007/s12522-009-0018-9. [Google Scholar] [PubMed] [CrossRef]
52. Lamano-Carvalho TL, Almeida SA, Teófilo JM, Anselmo Franci JA, Brentegani LG. Antireproductive effect of the calcium channel blocker amlodipine in male rats. Exp Toxicol Pathol 2000;52(4):353–356. doi:10.1016/S0940-2993(00)80062-7. [Google Scholar] [PubMed] [CrossRef]
53. Ikawa M, Inoue N, Benham AM, Okabe M. Fertilization: a sperm’s journey to and interaction with the oocyte. J Clin Invest 2010;120(4):984–994. doi:10.1172/JCI41585. [Google Scholar] [PubMed] [CrossRef]
54. Alasmari W, Costello S, Correia J et al. Ca2+ signals generated by CatSper and Ca2+ stores regulate different behaviors in human sperm. J Biol Chem 2013;288(9):6248–6258. doi:10.1074/jbc.m112.439356. [Google Scholar] [PubMed] [CrossRef]
55. Louros NN, Chrysina ED, Baltatzis GE, Patsouris ES, Hamodrakas SJ, Iconomidou VA. A common ‘aggregation-prone’ interface possibly participates in the self-assembly of human zona Pellucida proteins. FEBS Lett 2016;590(5):619–630. doi:10.1002/1873-3468.12099. [Google Scholar] [PubMed] [CrossRef]
56. Törmälä RM, Jääskeläinen M, Lakkakorpi J, Liakka A, Tapanainen JS, Vaskivuo TE. Zona Pellucida components are present in human fetal ovary before follicle formation. Mol Cell Endocrinol 2008;289(1–2):10–15. doi:10.1016/j.mce.2008.01.029. [Google Scholar] [PubMed] [CrossRef]
57. Pökkylä RM, Lakkakorpi JT, Nuojua-Huttunen SH, Tapanainen JS. Sequence variations in human ZP genes as potential modifiers of zona Pellucida architecture. Fertil Steril 2011;95(8):2669–2672. doi:10.1016/j.fertnstert.2011.01.168. [Google Scholar] [PubMed] [CrossRef]
58. Killingbeck EE, Swanson WJ. Egg coat proteins across metazoan evolution. Curr Top Dev Biol 2018;130(1):443–488. doi:10.1016/bs.ctdb.2018.03.005. [Google Scholar] [PubMed] [CrossRef]
59. Goudet G, Mugnier S, Callebaut I, Monget P. Phylogenetic analysis and identification of Pseudogenes reveal a progressive loss of zona Pellucida genes during evolution of vertebrates. Biol Reprod 2008;78(5):796–806. doi:10.1095/biolreprod.107.064568. [Google Scholar] [PubMed] [CrossRef]
60. Litscher ES, Wassarman PM. Zona Pellucida proteins, fibrils, and matrix. Annu Rev Biochem 2020;89(1):695–715. doi:10.1146/annurev-biochem-011520-105310. [Google Scholar] [PubMed] [CrossRef]
61. Rankin TL, O’Brien M, Lee E, Wigglesworth K, Eppig J, Dean J. Defective zonae Pellucidae in Zp2-null mice disrupt folliculogenesis, fertility and development. Development 2001;128(7):1119–1126. doi:10.1242/dev.128.7.1119. [Google Scholar] [PubMed] [CrossRef]
62. Baibakov B, Boggs NA, Yauger B, Baibakov G, Dean J. Human sperm bind to the N-terminal domain of ZP2 in humanized zonae Pellucidae in transgenic mice. J Cell Biol 2012;197(7):897–905. doi:10.1083/jcb.201203062. [Google Scholar] [PubMed] [CrossRef]
63. Beltrán C, Treviño CL, Mata-Martínez E et al. Role of ion channels in the sperm acrosome reaction. Adv Anat Embryol Cell Biol 2016;220(1):35–69. doi:10.1007/978-3-319-30567-7_3. [Google Scholar] [PubMed] [CrossRef]
64. Publicover S, Harper CV, Barratt C. [Ca2+]i signalling in sperm—making the most of what you’ve got. Nat Cell Biol 2007;9(3):235–242. doi:10.1038/ncb0307-235. [Google Scholar] [PubMed] [CrossRef]
65. Stival C, Puga Molina LDC, Paudel B, Buffone MG, Visconti PE, Krapf D. Sperm capacitation and acrosome reaction in mammalian sperm. Adv Anat Embryol Cell Biol 2016;220(Suppl 7):93–106. doi:10.1007/978-3-319-30567-7_5. [Google Scholar] [PubMed] [CrossRef]
66. Spungin B, Breitbart H. Calcium mobilization and influx during sperm exocytosis. J Cell Sci 1996;109(Pt 7):1947–1955. doi:10.1242/jcs.109.7.1947. [Google Scholar] [PubMed] [CrossRef]
67. Van B, Caltrider K. Sperm attachment and penetration competence in the human oocyte: a possible aetiology of fertilization failure involving the organization of oolemmal lipid raft microdomains influenced by the ΔΨm of subplasmalemmal mitochondria. Reprod Biomed Online 2013;27(6):690–701. doi:10.1016/j.rbmo.2013.09.011. [Google Scholar] [PubMed] [CrossRef]
68. Rubinstein E, Ziyyat A, Prenant M et al. Reduced fertility of female mice lacking CD81. Dev Biol 2006;290(2):351–358. doi:10.1016/j.ydbio.2005.11.031. [Google Scholar] [PubMed] [CrossRef]
69. Runge KE, Evans JE, He ZY et al. Oocyte CD9 is enriched on the microvillar membrane and required for normal microvillar shape and distribution. Dev Biol 2007;304(1):317–325. doi:10.1016/j.ydbio.2006.12.041. [Google Scholar] [PubMed] [CrossRef]
70. Jégou A, Ziyyat A, Barraud-Lange V et al. CD9 tetraspanin generates fusion competent sites on the egg membrane for mammalian fertilization. Proc Natl Acad Sci U S A 2011;108(27):10946–10951. doi:10.1073/pnas.1017400108. [Google Scholar] [PubMed] [CrossRef]
71. Satouh Y, Inoue N, Ikawa M, Okabe M. Visualization of the moment of mouse sperm-egg fusion and dynamic localization of IZUMO1. J Cell Sci 2012;125(Pt 21):4985–4990. doi:10.1242/jcs.100867. [Google Scholar] [PubMed] [CrossRef]
72. Granados-Gonzalez V, Aknin-Seifer I, Touraine RL, Chouteau J, Wolf JP, Levy R. Preliminary study on the role of the human IZUMO gene in oocyte-spermatozoa fusion failure. Fertil Steril 2008;90(4):1246–1248. doi:10.1016/j.fertnstert.2007.09.008. [Google Scholar] [PubMed] [CrossRef]
73. Inoue N, Hamada D, Kamikubo H et al. Molecular dissection of IZUMO1, a sperm protein essential for sperm-egg fusion. Development 2013;140(15):3221–3229. doi:10.1242/dev.094854. [Google Scholar] [PubMed] [CrossRef]
74. Bianchi E, Doe B, Goulding D, Wright GJ. Juno is the egg Izumo receptor and is essential for mammalian fertilization. Nature 2014;508(7497):483–487. doi:10.1038/nature13203. [Google Scholar] [PubMed] [CrossRef]
75. Bianchi E, Wright GJ. Sperm meets egg: the genetics of mammalian fertilization. Annu Rev Genet 2016;50(1):93–111. doi:10.1146/annurev-genet-121415-121834. [Google Scholar] [PubMed] [CrossRef]
76. Yu M, Zhao H, Chen T et al. Mutational analysis of IZUMO1R in women with fertilization failure and polyspermy after in vitro fertilization. J Assist Reprod Genet 2018;35(3):539–544. doi:10.1007/s10815-017-1101-5. [Google Scholar] [PubMed] [CrossRef]
77. Chalbi M, Barraud-Lange V, Ravaux B et al. Binding of sperm protein Izumo1 and its egg receptor Juno drives Cd9 accumulation in the intercellular contact area prior to fusion during mammalian fertilization. Development 2014;141(19):3732–3739. doi:10.1242/dev.111534. [Google Scholar] [PubMed] [CrossRef]
78. Abdelhedi F, Chalas C, Petit JM et al. Altered three-dimensional organization of sperm genome in DPY19L2-deficient globozoospermic patients. J Assist Reprod Genet 2019;36(1):69–77. doi:10.1007/s10815-018-1342-y. [Google Scholar] [PubMed] [CrossRef]
79. Nasr-Esfahani MH, Deemeh MR, Tavalaee M. Artificial oocyte activation and intracytoplasmic sperm injection. Fertil Steril 2010;94(2):520–526. doi:10.1016/j.fertnstert.2009.03.061. [Google Scholar] [PubMed] [CrossRef]
80. Ferlin A, Dipresa S, Delbarba A et al. Contemporary genetics-based diagnostics of male infertility. Expert Rev Mol Diagn 2019;19(7):623–633. doi:10.1080/14737159.2019.1633917. [Google Scholar] [PubMed] [CrossRef]
81. Okutman O, Ben Rhouma M, Benkhalifa M, Muller J, Viville S. Genetic evaluation of patients with non-syndromic male infertility. J Assist Reprod Genet 2018;35(11):1939–1951. doi:10.1007/s10815-018-1301-7. [Google Scholar] [PubMed] [CrossRef]
82. Ito J, Parrington J, Fissore RA. PLCζ and its role as a trigger of development in vertebrates. Mol Reprod Dev 2011;78(10–11):846–853. doi:10.1002/mrd.21359. [Google Scholar] [PubMed] [CrossRef]
83. Ducibella T, Fissore R. The roles of Ca2+, downstream protein kinases, and oscillatory signaling in regulating fertilization and the activation of development. Dev Biol 2008;315(2):257–279. doi:10.1016/j.ydbio.2007.12.012. [Google Scholar] [PubMed] [CrossRef]
84. Escoffier J, Lee HC, Yassine S et al. Homozygous mutation of PLCZ1 leads to defective human oocyte activation and infertility that is not rescued by the WW-binding protein PAWP. Hum Mol Genet 2016;25(5):878–891. doi:10.1093/hmg/ddv617. [Google Scholar] [PubMed] [CrossRef]
85. Cox LJ, Larman MG, Saunders CM, Hashimoto K, Swann K, Lai FA. Sperm phospholipase Czeta from humans and cynomolgus monkeys triggers Ca2+ oscillations, activation and development of mouse oocytes. Reproduction 2002;124(5):611–623. doi:10.1530/rep.0.1240611. [Google Scholar] [PubMed] [CrossRef]
86. Sanusi R, Yu Y, Nomikos M, Lai FA, Swann K. Rescue of failed oocyte activation after ICSI in a mouse model of male factor infertility by recombinant phospholipase Cζ. Mol Hum Reprod 2015;21(10):783–791. doi:10.1093/molehr/gav042. [Google Scholar] [PubMed] [CrossRef]
87. Dale B, Wilding M, Coppola G, Tosti E. How do spermatozoa activate oocytes? Reprod Biomed Online 2010;21(1):1–3. doi:10.1016/j.rbmo.2010.02.015. [Google Scholar] [PubMed] [CrossRef]
88. Amdani SN, Yeste M, Jones C, Coward K. Phospholipase C Zeta (PLCζ) and male infertility: clinical update and topical developments. Adv Biol Regul 2016;61:58–67. doi:10.1016/j.jbior.2015.11.009. [Google Scholar] [PubMed] [CrossRef]
89. Kamali-Dolat Abadi M, Tavalaee M, Shahverdi A, Nasr-Esfahani MH. Evaluation of PLCζ and PAWP expression in globozoospermic individuals. Cell J. 2016;18(3):438–445. doi:10.22074/cellj.2016.4572. [Google Scholar] [PubMed] [CrossRef]
90. Azil S, Mbaye MM, Louanjli N, Ghazi B, Benkhalifa M. Phospholipase C Zeta: a hidden face of sperm for oocyte activation and early embryonic development. Obstet Gynecol Sci 2024;67(5):467–480. doi:10.5468/ogs.24019. [Google Scholar] [PubMed] [CrossRef]
91. Que EL, Duncan FE, Lee HC et al. Bovine eggs release zinc in response to parthenogenetic and sperm-induced egg activation. Theriogenology 2019;127(11):41–48. doi:10.1016/j.theriogenology.2018.12.031. [Google Scholar] [PubMed] [CrossRef]
92. Duncan FE, Que EL, Zhang N, Feinberg EC, O’Halloran TV, Woodruff TK. The zinc spark is an inorganic signature of human egg activation. Sci Rep 2016;6(1):24737. doi:10.1038/srep24737. [Google Scholar] [PubMed] [CrossRef]
93. Que EL, Bleher R, Duncan FE et al. Quantitative mapping of zinc fluxes in the mammalian egg reveals the origin of fertilization-induced zinc Sparks. Nat Chem 2015;7(2):130–139. doi:10.1038/nchem.2133. [Google Scholar] [PubMed] [CrossRef]
94. Lee K, Davis A, Zhang L et al. Pig oocyte activation using a Zn2+ chelator. TPEN Theriogenology 2015;84(6):1024–1032. doi:10.1016/j.theriogenology.2015.05.036. [Google Scholar] [PubMed] [CrossRef]
95. De Macedo MP, Glanzner WG, Rissi VB et al. A fast and reliable protocol for activation of porcine oocytes. Theriogenology 2019;123(2):22–29. doi:10.1016/j.theriogenology.2018.09.021. [Google Scholar] [PubMed] [CrossRef]
96. Nomikos M, Sanders JR, Kashir J et al. Functional disparity between human PAWP and PLCζ in the generation of Ca2+ oscillations for oocyte activation. Mol Hum Reprod 2015;21(9):702–710. doi:10.1093/molehr/gav034. [Google Scholar] [PubMed] [CrossRef]
97. Azil S, Kaarouch I, Montjean D et al. Effect of sperm DNA fragmentation and chromatin decondensation on PLCζ efficacy in infertile patients. Curr Issues Mol Biol 2025;47(9):707. doi:10.3390/cimb47090707. [Google Scholar] [PubMed] [CrossRef]
98. Gardner AJ, Evans JP. Mammalian membrane block to polyspermy: new insights into how mammalian eggs prevent fertilisation by multiple sperm. Reprod Fertil Dev 2006;18(1–2):53–61. doi:10.1071/rd05122. [Google Scholar] [PubMed] [CrossRef]
99. Coy P, Avilés M. What controls polyspermy in mammals, the oviduct or the oocyte? Biol Rev Camb Philos Soc 2010;85(3):593–605. doi:10.1111/j.1469-185X.2009.00117.x. [Google Scholar] [PubMed] [CrossRef]
100. Wong JL, Wessel GM. Defending the zygote: search for the ancestral animal block to polyspermy. Curr Top Dev Biol 2006;72(Pt. 1):1–151. doi:10.1016/S0070-2153(05)72001-9. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools