 Open Access
Open Access
REVIEW
Association between periodontal disease and prostatic disease: a systematic review and meta-analysis of observational studies
1 Department of Urology, The First Affiliated Hospital of Shihezi University, Shihezi, China
2 Department of Stomatology, Zhongnan Hospital of Wuhan University, Wuhan, China
3 Center for Evidence-Based and Translational Medicine, Zhongnan Hospital of Wuhan University, Wuhan, China
4 Department of Epidemiology and Biostatistics, School of Public Health, Wuhan University, Wuhan, China
5 Department of Urology, Lanzhou University Second Hospital, Lanzhou, China
* Corresponding Authors: Bing-Hui Li. Email: ; Xian-Tao Zeng. Email:
Canadian Journal of Urology 2026, 33(1), 7-20. https://doi.org/10.32604/cju.2026.076741
Received 26 November 2025; Accepted 16 January 2026; Issue published 28 February 2026
Abstract
Objective: Current research highlights periodontal disease as a systemic inflammatory condition that may influence extra-oral diseases such as prostatic diseases, which prompted us to explore the potential association. To evaluate whether periodontal disease is associated with an increased risk of prostatic disease, including prostate cancer, benign prostatic hyperplasia (BPH), and prostatitis. Methods: A systematic search of observational studies concerning the relationship between periodontal disease and prostatic disease was performed in online databases PubMed, Embase, Web of Science, Scopus, CENTRAL, CNKI, and WanFang. Searches were conducted from database inception to 31 July 2025. Pooled hazard ratio (HR) or odds ratio (OR) with 95% confidence intervals (CIs) were synthesized. Subgroup analysis was used to detect the origin of heterogeneity, sensitivity analysis was employed to evaluate the robustness of the results, and publication bias analyses were also performed. R software was used to perform statistical analyses. Results: Sixteen studies that met the preset criteria were included in this study. In the pooled analysis, periodontal disease was associated with increased risk of prostate cancer (HR = 1.23, 95%CI: 1.16–1.29, p < 0.001) or BPH (OR = 1.55, 95%CI: 1.41–1.70, p < 0.001). Sensitivity analysis confirmed the robustness of the results. No obvious publication bias was found in the meta-analysis. Only one cohort study reported that chronic periodontitis increases the risk of prostatitis (HR = 2.521, 95%CI: 1.685–4.005, p < 0.001). The effect of periodontal treatment on prostatic disease is still unclear. Conclusions: The systematic review and meta-analysis identified an observational association between periodontal disease and increased risks of prostate cancer and BPH. Because all included studies were observational, these results indicate association rather than causation, and further prospective and mechanistic studies are required to clarify temporality and causality.Keywords
Supplementary Material
Supplementary Material FileProstatic diseases, including prostate cancer, prostatitis, and benign prostatic hyperplasia (BPH), are common male urological conditions worldwide and impose substantial morbidity. Prostate cancer is the most frequently diagnosed cancer in males, with incidence rates rising annually.1,2 It is estimated that there will be 1.47 million new cases and 0.397 million deaths worldwide in 2022.2 Prostatitis and BPH are inflammatory conditions of the prostate gland that affect almost half of all men in their lifetime.3 The prostate diseases prevalence increases substantially with age, contributing to significant morbidity, reduced quality of life, and increased healthcare utilization.4 Understanding modifiable risk factors for these conditions is therefore of considerable clinical and public health interest.
Periodontal disease is a polymicrobial chronic inflammation, which mainly consists of gingivitis and periodontitis that are associated with the damage of the periodontal ligaments and collapse of the adjacent alveolar bone.5 Recent studies have shed light on the relationship between periodontal disease and systemic diseases, including cardiovascular disease, arterial hypertension, diabetes mellitus, and some extra-oral remote diseases, and have represented a potential risk.5
Increasing mechanistic studies are attempting to explain the biological link between periodontal disease and prostate disease. Periodontitis is a chronic inflammatory condition characterized by the persistent dissemination of periodontal pathogens and their virulence factors, which can induce systemic inflammation.6 Common periodontal pathogens, including Porphyromonas gingivalis (P. gingivalis), have been identified in prostatic fluid and tissue samples from patients with BPH and concomitant periodontitis, suggesting the existence of a potential oral–prostate microbial axis.7,8 An experimental study has demonstrated that P. gingivalis infection or its lipopolysaccharide can activate the interleukin-6/interleukin-6 receptor α (IL-6/IL-6Rα) signaling pathway, thereby amplifying inflammatory responses and disrupting the balance between cellular proliferation and apoptosis. These processes may directly promote prostatic epithelial and stromal hyperplasia, collagen deposition, and tissue fibrosis.8 Alterations in the oral microbiome and its associated metabolic profile, including the upregulation of pro-inflammatory lipid metabolites such as arachidonic acid, have been observed in patients with both BPH and periodontitis. These findings further support the involvement of a microbiome–metabolite–inflammation axis in disease progression.7 Emerging evidence suggests that dysbiosis, particularly involving the oral microbiome, may influence prostate pathology and carcinogenesis by modulating systemic immune responses and inflammatory signaling pathways, thereby contributing to genitourinary diseases.9,10
Several epidemiological studies have explored the potential link between periodontal disease and prostatic conditions. A Taiwan population-based study enrolling 683,854 participants revealed a higher risk of prostate cancer in men with periodontal disease, however, no association was observed in an Atherosclerosis Risk in Communities (ARIC) study conducted in the USA from 1987 to 2012.11,12 Four studies explored the risk of BPH in the community-based population, three studies showed a higher risk in those with periodontal disease,13–15 while the risk of BPH was similar between the two groups in a cohort study.16 In 2021, two meta-analyses provided evidence that periodontal disease is associated with an increased risk of prostate cancer.17,18 A meta-analysis conducted in 2023 showed that periodontal disease could not increase the risk of the incidence of prostate inflammation.19 No meta-analysis on the association between periodontal disease and BPH was found in the databases we searched. With more studies based on large populations on prostate cancer emerging, interest in the role of periodontal disease in prostatic disease has been strengthened. Hence, we conducted a systematic review and meta-analysis to evaluate the association between periodontal disease (exposure) and major prostatic diseases (outcomes). The aim of this meta-analysis is to discuss whether periodontal disease is associated with increased risk of prostate disease.
This systematic review/meta-analysis was conducted following the PRISMA 2020 statement and is registered with PROSPERO (CRD420251245649). A completed PRISMA 2020 checklist is provided in the Supplementary Material.
To be included in the meta-analysis, studied should met the following criteria: (1) studies on adult populations; (2) studies with periodontal disease (periodontitis and gingivitis) or its treatment as exposure, and with prostatic disease risk, including prostate cancer incidence or mortality, prostatitis and BPH incidence as outcome; (3) observational studies (cohort study, case-control study and cross-sectional study), with prospective or retrospective design. Studies were excluded for the following reasons: (1) duplicated data; (2) data could not be obtained even by contacting the original author. For studies on the same outcome involving subjects from the same population or community, only the most comprehensive one was included.
Observational studies concerning the relationship between periodontal disease and prostatic disease were identified through searching PubMed, Embase, CENTRAL, CNKI, and WanFang databases. Electronic searches were conducted from database inception to inception to 31 July 2025. The reference lists of included studies, relevant published original articles, and meta-analyses were also screened to find any other qualified studies. The search terms were structured as follows: (“periodontal disease” OR periodontitis OR gingivitis OR periodont*) AND (“prostatic disease” OR prostate cancer OR prostatitis OR “benign prostatic hyperplasia”).
Study selection and data extraction
Titles and abstracts were screened by two authors (QL and LW) according to eligibility criteria. After removing duplicated and irrelevant studies, the full texts of the potentially eligible studies were then examined. Disagreements were resolved through discussion with the third author (DH).
Data was independently extracted by two authors (YZ and SOY) using pre-designed Excel spreadsheets. The information and data extracted from the included studies were as follows: name of first author, year of publication, country or location of study, study period, study design, study population, sample size, mean age of subjects, follow-up duration, ascertainment of periodontal disease and prostatic disease, and relevant data on outcomes.
For periodontal disease, we extracted the case definitions used by each study, e.g., the Centers for Disease Control and Prevention-American Academy of Periodontology (CDC-AAP) clinical criteria, dentist diagnosis recorded by the International Classification of Diseases (ICD) codes, self-report questionnaires. For prostatitis/prostatic outcomes, we similarly extracted ascertainment (ICD codes, urologist diagnosis, pathology reports, B-ultrasonography or symptom questionnaires).
To detect overlapping study populations, we compared data sources, study periods, and population descriptions. When two studies used the same underlying database and reported overlapping populations/outcomes, we included the most comprehensive study and excluded duplicates. If disagreements occurred, a final consensus was finally achieved through discussion. For each study, we extracted effect estimates from the most fully adjusted multivariable model. When multiple models were reported, the model adjusting for the largest set of clinically relevant confounders was selected.
The quality of included cohort and case-control studies was assessed using the Newcastle–Ottawa (NOS) quality assessment scale for cohort studies and case-control studies,20 respectively. The NOS scale consists of three aspects that evaluate “selection”, “comparability”, and “exposure”. For each item within the “selection” and “exposure” categories, a study can be awarded a maximum of one star, and a maximum of two stars can be given for “Comparability”. Studies with 0–3 stars are considered low quality, while studies with 4–6 stars and 7–9 stars are considered moderate- and high-quality. The quality of cross-sectional studies was evaluated using the Joanna Briggs Institute (JBI) critical appraisal checklists21 for analytical cross-sectional studies. The JBI checklist includes eight questions, and each question can be answered with “yes”, “no”, “unclear”, or “Not applicable”. “Yes” corresponds to one point, and total scores are calculated for each study. We considered studies with 0–3 scores as low risk, and those with 4–6 scores and 7–8 scores as moderate- and high-quality. These two tools are still widely used in epidemiological systematic reviews, are easy to operate, and are comparable to previous similar reviews in this field. Two authors (QL and JYY) independently assessed the quality of all included studies. Disagreements in ratings were resolved by discussion.
Effect estimates reported in the original studies (hazard ratios [HRs], odds ratios [ORs], mortality rate ratios [MRRs], etc.) were extracted with their 95% confidence intervals (CI) and pooled separately without conversion between effect types. We utilized a random-effects model for meta-analyses. To maintain methodological precision, HRs, ORs, and MRRs and 95% CI were pooled separately due to variations in outcome definitions and measurement contexts. The forest plot was generated using the ‘meta’ package in R version 4.4.2. Heterogeneity was evaluated using the I² statistic (with I² > 50% indicating substantial heterogeneity) and the Q-test p-value (p < 0.1 indicating significant heterogeneity). Measures that could not be harmonized (e.g., single-study MRRs with unique definitions) were described qualitatively rather than pooled. We performed separate analyses for prostate cancer, BPH, and prostatitis. Subgroup analyses stratified by study design, mean age of subjects, and quality assessment were conducted to detect the origin of heterogeneity. Sensitivity analysis was performed by omitting one study at a time to evaluate the reliability of the results. Funnel plots were constructed and visually assessed for the possibility of publication biases. Egger’s and Begg’s tests were also used to explore potential publication bias, and p < 0.10 was considered suggestive of potential small-study effects or publication bias. All statistical analyses were performed using R 4.4.2 with the ‘meta’ package (version 8.0-1).
A total of 1428 studies were identified from the online databases and other sources. 453 duplicated studies were first removed, then 865 studies were excluded after reviewing titles and abstracts. 110 potentially relevant studies with their full-text were retrieved and carefully read for eligibility examination, in which 94 studies were eliminated for ineligible publication type, duplicated data, insufficient or inappropriate data, and irrelevance. 16 studies were finally included in the systematic review. The process of study selection is shown in Figure 1.
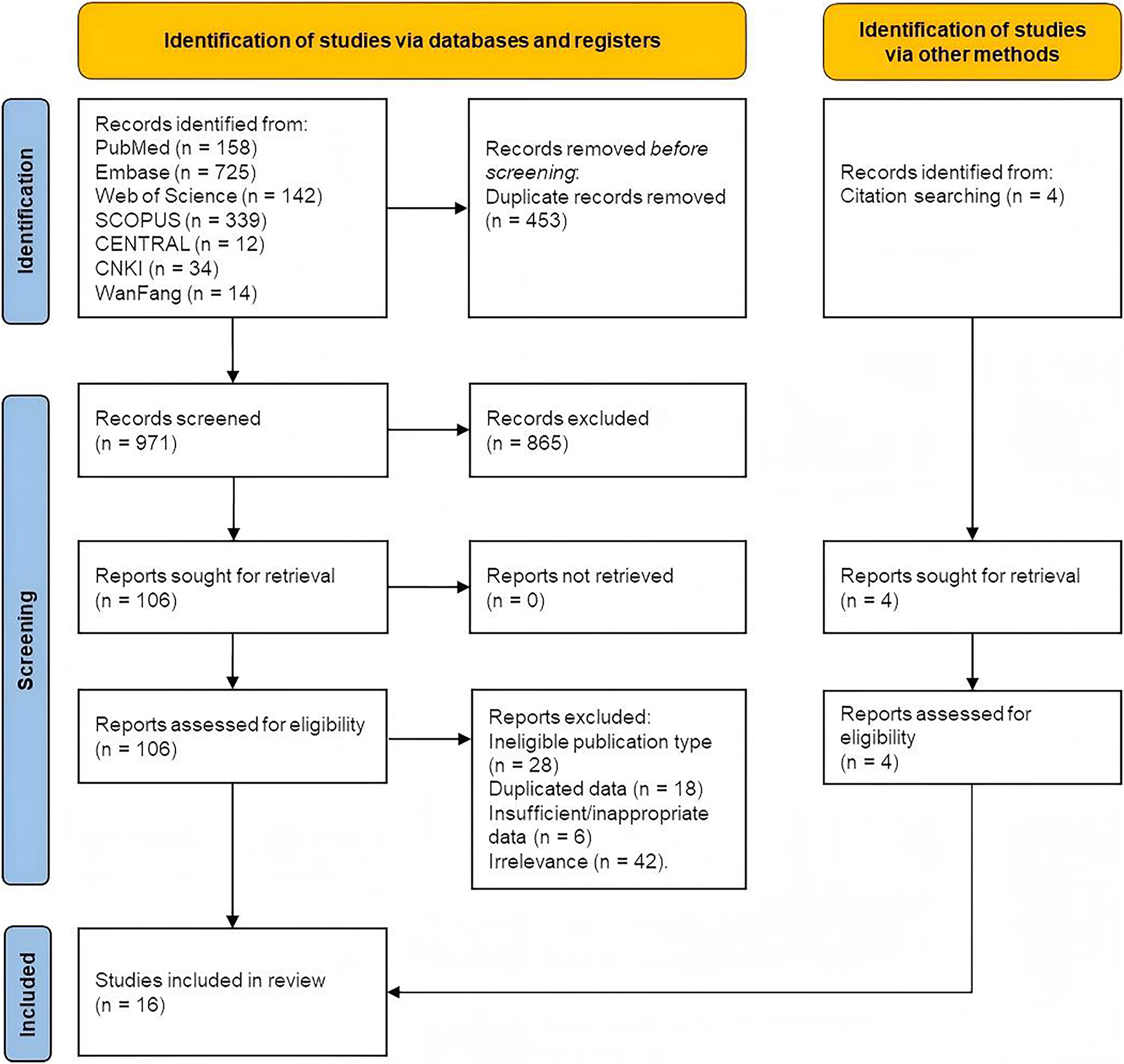
FIGURE 1. Flow diagram of study selection in the meta-analysis
Characteristics of the studies included in the systematic review and meta-analysis are summarized in Table 1. Among 16 included studies, all explored the association between periodontal disease and prostatic disease, and 12 for prostate cancer, 4 for BPH, and 1 for prostatitis. These studies consist of 13 cohort studies, 2 cross-sectional studies, and 1 case-control study, which were published between 2013 and 2023 with a prospective or retrospective design. The mean age of 60 years and above of the subjects was found in 5 studies, and less than 60 years in 9 studies. The follow-up period of 10 years or more was reported in 11 studies and less than 10 years in 2 studies. All the studies reported the ascertainment of periodontal and prostatic disease.
The NOS scale was employed to assess the quality of cohort (Table S1) and case-control studies (Table S2). Among 13 cohort studies, 11 studies, for 5 with 9 stars, 2 with 8 stars and 4 with 7 stars, were considered high quality, and 2 studies with 6 stars were rated as of moderate quality. The one case-control study with 6 stars was of moderate quality. The JBI checklist was applied to assess the quality of 2 cross-sectional studies. The quality of one study with 7 scores was considered high and the other with 6 scores was moderate (Table S3). In general, the results of quality assessment indicated that all studies were of moderate to high methodological quality and low risk of bias.
Periodontal disease and prostate cancer
Eight cohort studies reported the risk of prostate cancer development. Of these, one study involving 1032 subjects reported that periodontitis did not increase the risk of prostate cancer (OR = 1.734, 95%CI: 0.932–3.227, p = 0.082).23 Another study from Turkey reported that periodontal disease increased the risk of prostate cancer, with age- and sex-standardized incidence rates (SIRs) of 1.84 (95%CI: 1.34–2.49, p < 0.001).26 The remaining six cohort studies reported HRs. One of these studies reported the prostate cancer risk for mild, moderate, and severe periodontitis separately.12 Therefore, all three results were included in our meta-analysis. The results showed that periodontal disease increased the risk of prostate cancer compared with individuals without periodontal disease (HR = 1.23, 95%CI: 1.16–1.29, p < 0.001), with moderate heterogeneity (I2 = 48.3%, p = 0.06) (Figure 2).
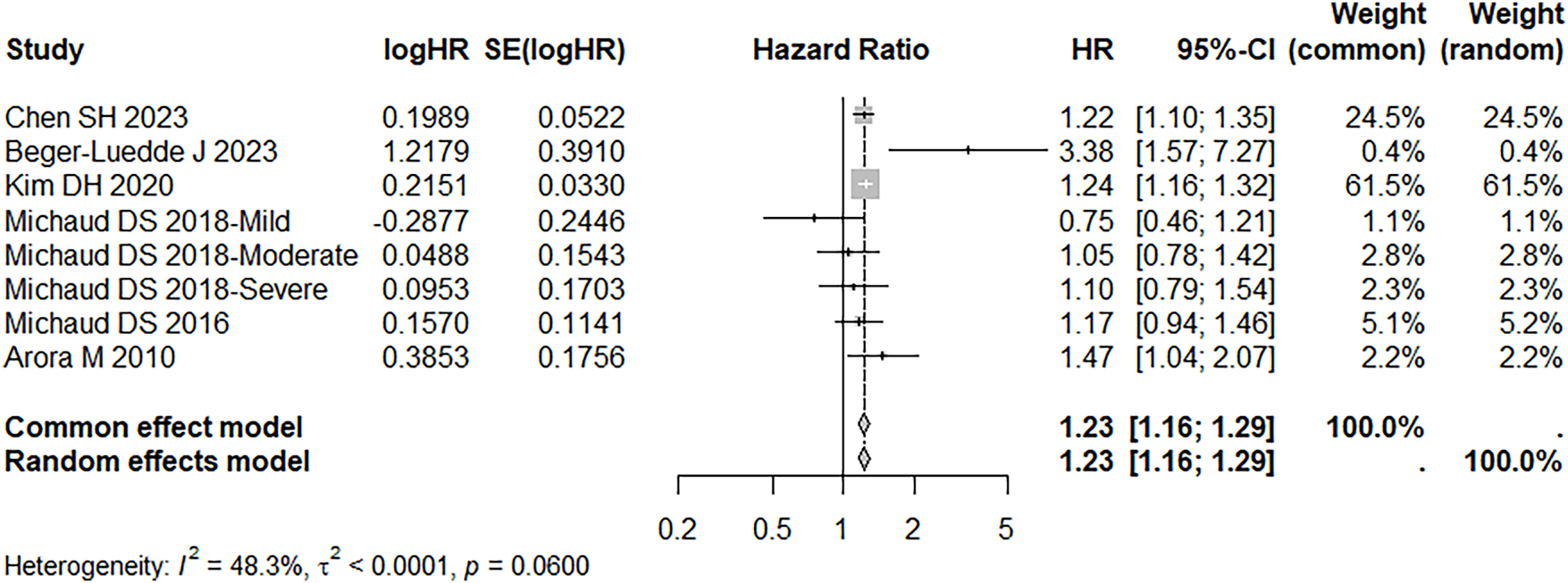
FIGURE 2. Forest plot of the association between periodontal disease and prostate cancer. Data source references.11,12,22,25,28,29
Given potential heterogeneity, subgroup analyses were conducted with stratification by study design, mean age of the included populations, and methodological quality. In prospective cohort studies, heterogeneity was markedly reduced (I2 = 25.9%), and the results indicate that periodontitis is not associated with an increased risk of prostate cancer incidence (HR = 1.13, 95% CI 0.99–1.30). When stratified by mean age (<60 vs. ≥60 years) and by methodological quality, the direction of the association remained positive across strata: HRs were 1.24 (95% CI 1.14–1.36) for populations under 60 years and 1.12 (95% CI 0.94–1.32) for those 60 years or older; and HRs were 1.22 (95% CI 1.16–1.28) for high-quality studies and 1.47 (95% CI 1.04–2.07) for moderate-quality studies.
Sensitivity analysis supported the robustness of the association between periodontal disease and prostate cancer (Fig. S1). Visual inspection of the funnel plot revealed modest asymmetry (Fig. S2). Egger’s test (p = 0.96) and Begg’s test (p = 0.80) did not provide statistically significant evidence of publication bias.
Three studies reported prostate cancer-specific mortality risk.24,27,30 Because these studies used different effect measures—HR, MRR, and OR—a meta-analysis was not feasible. There were notable differences in confounding control across studies: Chung 202024 adjusted for age and sex only; Heikkilä 201827 adjusted more broadly (including age, sex, socioeconomic status, number of teeth, dental treatments, oral health indices, periodontal treatment, diabetes), and Hujoel 200330 adjusted mainly for age and gender, with exposure definitions spanning both periodontitis and gingivitis and reporting different effect measures. Chung 202024 reported adjusted HR 1.34 (95% CI: 1.02–1.76), indicating a statistically significant increased risk. In contrast, Heikkilä 201827 reported adjusted MRR 1.75 (95%CI: 0.63–4.87), and Hujoel 200330 reported periodontitis OR 1.81 (95%CI: 0.76–4.34) and gingivitis OR 1.48 (95%CI: 0.56–3.94); none reached statistical significance, reflecting substantial imprecision.
Four studies evaluated the risk of BPH in individuals with periodontal disease compared with those without. Meta-analysis showed that periodontal disease increased the risk of BPH (OR = 1.55, 95%CI: 1.41–1.70), with no significant heterogeneity (I2 = 0.00%, p = 0.679) (Figure 3). Sensitivity analyses supported the meta-analytic result (Fig. S3). Because the number of studies was small, a funnel plot could not be constructed. Egger’s test (p = 0.629) and Begg’s test (p = 0.654) did not provide statistically significant evidence of publication bias.
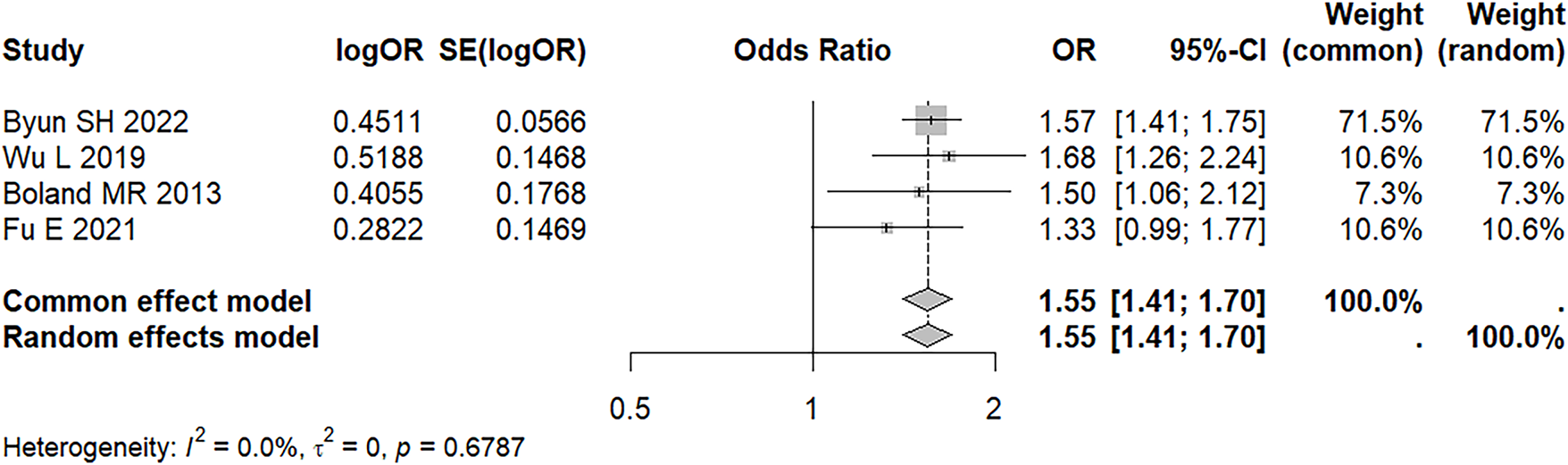
FIGURE 3. Forest plot of the association between periodontal disease and BPH. Data source reference13–16
Periodontal disease and prostatitis
Only one retrospective cohort examined prostatitis as an outcome.16 This cohort included 16,530 participants, with a mean age of approximately 64 years and about 15 years of follow-up. In that study, the exposure was physician-diagnosed chronic periodontitis (identified by ICD codes). For prostatitis as a standalone outcome, the adjusted HR was 2.521 (95% CI 1.685–4.005, p < 0.001). The multivariable model adjusted for age, marital status, education, urbanization level, level of medical care, and several comorbidities (hypertension, hyperlipidemia, depression, diabetes mellitus, coronary heart disease, stroke, obesity, etc.). This study, therefore, suggests a potential association between chronic periodontitis and an increased risk of prostatitis, with a stronger association for prostatitis; however, for the composite outcome, significance was limited, and the evidence comes from a single study, potentially affected by observational bias, information bias, and residual confounding.
Periodontal treatment and prostate disease
Three studies examined the impact of periodontal therapy on prostate disease. A retrospective cohort study by Hwang et al., using a health-insurance database, included 116,706 patients with periodontal disease.31 Compared with untreated individuals, those who received conventional periodontal treatment showed a significantly reduced overall cancer risk. However, the risk of prostate cancer was higher in the treatment group (HR 2.11, 95% CI 1.63–2.73, p < 0.01), potentially reflecting more frequent PSA screening among treated individuals.
In contrast, Chen SH (2023)11 included a cohort diagnosed with periodontal disease from 2000 to 2017 (N = 683,854; mean age 43 years; follow-up 12 years). For incident prostate cancer, the adjusted HR in the periodontal-treatment group was 0.60 (95% CI 0.45–0.80), indicating a significantly reduced risk among patients who received periodontal treatment.
A retrospective matched cohort using Taiwan’s National Health Insurance Research Database analyzed patients newly diagnosed with chronic periodontitis between 2000 and 2015.16 Participants were divided into a periodontal treatment group (n = 5510) and an untreated group (n = 5510). Follow-up through 2015 showed that after adjustment for age, marital status, education, urbanization, level of medical care, and comorbidities, the incidence of BPH did not differ significantly between periodontal treatment and non-treatment groups (adjusted HR 0.998, 95% CI 0.888–1.257). Similarly, the risk of prostatitis remained unchanged (adjusted HR 0.967, 95% CI 0.690–1.362). Therefore, periodontal treatment did not appear to reduce the risk of either BPH or prostatitis.
Recently, a variety of studies have proposed periodontal disease as a risk indicator for prostate disease. We once performed reviews to explore the association between periodontal disease and prostatic disease, as well as potential molecular and cellular mechanisms of oral microbiota in prostatic disease. Although the role of oral microbiota in various diseases has been reported in both experimental animals and humans, indicating that oral microbiota may regulate the progression of prostatic disease through direct (distant dissemination of oral pathobionts) or indirect pathways (inflammation and immune dysregulation), the underlying mechanisms remain elucidated.7,32,33
Several biological mechanisms have been proposed: (1) pathogens or their components may disseminate to the prostate via the bloodstream or lymphatic system, thereby directly inducing local inflammation;34 (2) chronic periodontal inflammation may trigger a systemic inflammatory response that alters the prostatic microenvironment;8 and (3) periodontal disease may influence the urogenital system through alterations in oral microbiota composition and related metabolites.7 Furthermore, aging may represent a key factor linking periodontal disease and prostate disease. Both conditions are highly prevalent among older men and are closely associated with immunosenescence and chronic low-grade systemic inflammation.35–37 Periodontal disease may exacerbate the age-related inflammatory burden by persistently releasing pro-inflammatory cytokines and microbial products, thereby amplifying inflammatory signaling within prostatic tissue.38,39
In addition to inflammation, emerging evidence suggests that microbiota-metabolite-host interactions play a role in regulating the oral-prostate axis.40–42 Periodontal dysbiosis can influence distant organs not only through direct microbial dissemination but also via systemic metabolic alterations.43 Changes in lipid mediators, short-chain fatty acids, and oxidative stress-related metabolites associated with periodontal disease may modulate androgen signaling, stromal-epithelial interactions, and fibrotic processes within the prostate.8,42–46 These mechanisms are particularly relevant to BPH, which is increasingly recognized as a fibroinflammatory condition rather than a purely hormone-driven disorder. These mechanisms are supported in part by animal and histological studies; however, further validation in human populations is required.
The epidemiological relationship between periodontal and prostatic disease also attracted increasing attention, however, conflicting results were shared in several studies.13,14,19 By analyzing the data from the Taiwan National Health Insurance Database, Fu E reported that the patients with periodontitis had a higher risk of prostate disorder in 2021,16 and Chen SH revealed a higher risk of prostate cancer in men with periodontitis in 2023.11 We conducted a cross-sectional study in 2019 and found the relationship between periodontal disease and BPH.14 In addition, a retrospective cohort study published in 2023 involving 19,782 outpatients from the UK demonstrated a significant association between chronic gingivitis and prostate cancer.22 On the contrary, no associations were observed between periodontitis and prostate cancer in a cohort study followed up for more than 30 years in Sweden.23 And a prospective cohort study of 19,933 subjects conducted in the USA also showed that periodontitis was not associated with prostate cancer.28
The newly published large population-based studies with previous studies on the association between periodontal disease and prostatic disease were included in the meta-analysis with the aim of examining if periodontal disease is related to increased risk of prostatic disease. One studies47 exploring the risk of prostate cancer was excluded because of the same data sources and outcome measures as Chen SH’s study in 2023.11 The data sources of the two studies26,48 were consistent and partially duplicated, the more comprehensive one26 was included. The study of Lee JH49 was excluded for reporting the same outcome using the data from the National Health Insurance Service–Elderly Cohort (NHIS-EC) database, as Kim DH’s study.25 Three studies performed by Michaud DS were retrieved,12,28,50 two studies were included according to the inclusion criteria.12,28 Finally, a total of 16 observational studies were included in this systematic review and found that periodontal disease could increase the risk of prostatic disease, either for prostate cancer or BPH. Subgroup analyses stratified by study design, mean age of subjects, follow-up period and methodological quality also achieved similar results, whereas no significant difference was observed in subgroups with a prospective design and with subjects aged 60 years or older. Which is worthy of our attention, the conflicting effects in different subgroups did not necessarily mean that there is no relationship between periodontal disease and prostatic disease in certain subgroups, which may be explained by limited studies and rare outcome events. Sensitivity analyses further confirmed the robustness of our findings. However, the source of heterogeneity across the studies could not be clearly illustrated through subgroup and sensitivity analyses. And the analysis of the association between periodontal disease and prostatitis could not be carried out due to the limited data reported in the included studies.
Previous meta-analyses on this topic were also reviewed. 4 cohort studies and 3 case-control studies were combined to analyze the association between periodontal disease and prostate inflammation in the meta-analysis by Ortíz de Urbina Comerón et al.19 The pooled risk estimate of 4 cohort studies indicated the absence of a significant association, whereas 3 case-control studies exhibited a statistically significant association. Due to a small number of articles included, the evidence is not enough to define the effect of periodontal disease on prostatic disease in this meta-analysis, indicating that periodontal disease could not increase the risk of prostate inflammation. 2 meta-analyses in 2021 investigated the association between periodontal disease and prostate cancer, with 9 cohort studies and 7 cohort or case-control studies included, respectively.17,18 And they both demonstrated that periodontal disease is associated with an increased risk of prostate cancer. In our study, we included more recently published large-scale studies, including 2 cross-sectional studies, and removed those studies with duplicated data compared to recent research. Considering the new findings of the role of periodontal disease in prostatic disease, and what’s more, we separately analyze the association between periodontal disease and prostate cancer, as well as BPH, and found that periodontal disease could increase the risk, either prostate cancer or BPH. And we also attempted to search for sources of between-study heterogeneity through subgroup and sensitivity analyses. Although the stratification according to study design, mean age of subjects, follow-up period, and methodological quality could not be used to explain the source of heterogeneity, the reliability of the findings was confirmed by sensitivity analysis. In terms of the high heterogeneity among the studies, it may be explained by the differences in the ascertainment of periodontal and prostatic disease, severity of periodontal disease, or adjusted variables included in the analytical model between studies, which needs to be further tested as more information is refined.
From an epidemiological perspective, it is important to consider the challenges of causal inference when interpreting the observed association between periodontal disease and prostatic diseases. Most available evidence, including the studies synthesized in the present meta-analysis, is derived from observational designs, which are inherently susceptible to confounding and reverse causality. For example, men with prostatic diseases may have reduced health-seeking behaviors, impaired self-care capacity, or shared lifestyle risk factors such as smoking, obesity, and metabolic diseases, all of which may contribute to poorer periodontal health. Although most included studies adjusted for major confounders, residual confounding cannot be completely excluded. Therefore, the observed association should be interpreted as an indicator of increased risk rather than direct causation.
The association between periodontal therapy and prostate disease remains inconclusive. Only a single large-scale cohort study reported an increased risk of prostate cancer among individuals undergoing regular periodontal treatment; however, this finding may have been confounded by more frequent PSA testing in the treated group.31 A prospective study from Germany enrolled 47 patients with asymptomatic chronic periodontitis and assessed PSA levels before and after periodontal therapy.51 The results indicated that periodontal treatment had no significant effect on total PSA, free PSA, or the fPSA/tPSA ratio. Interestingly, PSA concentrations were lower in smokers than in non-smokers, suggesting that smoking may act as a confounding factor in PSA assessment. These findings do not support a direct effect of periodontal therapy on PSA levels.51 In an American study, 27 men with chronic periodontitis received non-surgical periodontal treatment.52 Following therapy, participants exhibited significant improvements in clinical periodontal parameters and a marked reduction in International Prostate Symptom Score (IPSS). Notably, among patients with elevated baseline PSA levels (>4 ng/mL), average serum PSA levels decreased significantly compared to those with lower PSA levels (≤4 ng/mL). These findings suggest that periodontal therapy may improve urinary symptoms and have a beneficial effect on prostate health, particularly in individuals with chronic prostatitis.52
The strengths of this study include: (1) the inclusion of recently developed, large-scale population-based cohort and registry studies, thereby enhancing the volume and generalizability of the evidence; (2) the conduct of separate meta-analyses for prostate cancer and BPH, which facilitates clearer differentiation between disease-specific outcomes; and (3) the application of prespecified sensitivity and subgroup analyses (e.g., study design, age stratification, and methodological quality) to explore potential sources of heterogeneity. From a clinical perspective, a reproducible association between periodontal disease and prostate disease would suggest that oral health management may represent a component of comprehensive male health strategies. However, caution is warranted, as the current evidence is derived primarily from observational studies and does not establish causality.
Future research should aim to address several key gaps. First, prospective cohort studies with repeated assessments of periodontal status and prostate diseases would improve the level of evidence. Second, standardized and validated diagnostic criteria for both periodontal and prostate diseases are essential to enhance comparability across studies. Third, integrative studies combining epidemiological data with microbiome profiling, metabolomics, and immune phenotyping may help clarify the mechanistic pathways linking oral and prostatic health. Finally, stratified analyses by age, metabolic status, and inflammatory biomarkers may identify susceptible subpopulations who could benefit most from targeted preventive strategies.
Although more recent studies with moderate- to high-quality evidence were included, which may make this meta-analysis the most comprehensive one concerning the association between periodontal disease and prostatic disease, there were still several limitations. First, all included studies were observational in design and therefore cannot establish causal relationships. Second, the diagnostic criteria for periodontal and prostate diseases varied substantially across studies, which may have led to non-differential misclassification of exposures or outcomes. Third, the sets of confounding variables adjusted for differed among studies, and residual confounding may have influenced the effect estimates. Finally, the evidence for certain outcomes, such as prostatitis, was limited and based on a single study.
This systematic review and meta-analysis demonstrate that periodontal disease is associated with an increased risk of major prostatic diseases, particularly prostate cancer and BPH. These findings highlight a potential link between oral health and male urological conditions and underscore the importance of considering periodontal status in the broader assessment of men’s health. Further high-quality prospective studies and mechanistic investigations are needed to delineate the pathways underlying this association and to clarify its clinical implications.
Acknowledgement
None.
Funding Statement
The authors received no specific funding.
Author Contributions
Conceptualization, Bing-Hui Li and Xian-Tao Zeng; resources, Qiang Li and Lan Wu; data curation, Yi Zhang, Song Ou-Yang and Di Huang; writing—original draft preparation, Qiang Li; writing—review and editing, Bing-Hui Li and Xian-Tao Zeng; visualization, Bing-Hui Li; supervision, Xian-Tao Zeng; project administration, Jia-Yu Yang. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials
All data supporting the results of this study are included within the article and its supplementary materials.
Ethics Approval
Not applicable.
Informed Consent
Not applicable.
Conflicts of Interest
The authors declare no conflicts of interest.
Supplementary Materials: The supplementary material is available online at https://www.techscience.com/doi/10.32604/cju.2026.076741/s1.
References
1. Culp MB, Soerjomataram I, Efstathiou JA, Bray F, Jemal A. Recent global patterns in prostate cancer incidence and mortality rates. Eur Urol 2020;77(1):38–52. [Google Scholar] [PubMed]
2. Schafer EJ, Laversanne M, Sung H et al. Recent patterns and trends in global prostate cancer incidence and mortality: an update. Eur Urol 2025;87(3):302–313. [Google Scholar] [PubMed]
3. Guan X, Lao Y, Wang J et al. The methodological quality assessment of systematic reviews/meta-analyses of chronic prostatitis/chronic pelvic pain syndrome using AMSTAR2. BMC Med Res Methodol 2023;23(1):281. [Google Scholar] [PubMed]
4. Zi H, Liu MY, Luo LS et al. Global burden of benign prostatic hyperplasia, urinary tract infections, urolithiasis, bladder cancer, kidney cancer, and prostate cancer from 1990 to 2021. Mil Med Res 2024;11(1):64. doi:10.1186/s40779-024-00569-w. [Google Scholar] [PubMed] [CrossRef]
5. Huang D, Wang YY, Li BH et al. Association between periodontal disease and systemic diseases: a cross-sectional analysis of current evidence. Mil Med Res 2024;11(1):74. [Google Scholar] [PubMed]
6. Martinez-Garcia M, Hernandez-Lemus E. Periodontal inflammation and systemic diseases: an overview. Front Physiol 2021;12:709438. [Google Scholar] [PubMed]
7. Zhu C, Li LY, Li C et al. Profiles of oral microbiota and metabolites in periodontitis and benign prostatic hyperplasia patients: a pilot study. Microbiol Spectr 2025;13(10):e0337624. [Google Scholar] [PubMed]
8. Wang SY, Cai Y, Hu X et al. P. gingivalis in oral-prostate axis exacerbates benign prostatic hyperplasia via IL-6/IL-6R pathway. Mil Med Res 2024;11(1):30. [Google Scholar] [PubMed]
9. Xu XF, Cui JL, Li WH et al. Host-microbiota interactions in genitourinary cancer immunotherapy. Semin Cancer Biol 2025;115:1–15. doi:10.1016/j.semcancer.2025.07.005. [Google Scholar] [PubMed] [CrossRef]
10. Yuan S, Fang C, Leng WD et al. Oral microbiota in the oral-genitourinary axis: identifying periodontitis as a potential risk of genitourinary cancers. Mil Med Res 2021;8(1):54. [Google Scholar] [PubMed]
11. Chen SH, Chen JF, Hung YT et al. Exploring the relationship between periodontitis, anti-periodontitis therapy, and extra-oral cancer risk: findings from a nationwide population-based study. Biomedicines 2023;11(7):1949. [Google Scholar] [PubMed]
12. Michaud DS, Lu J, Peacock-Villada AY et al. Periodontal disease assessed using clinical dental measurements and cancer risk in the ARIC study. J Natl Cancer Inst 2018;110(8):843–854. [Google Scholar] [PubMed]
13. Byun S-H, Min C, Bang W et al. A cross-sectional study for association between periodontitis and benign prostatic hyperplasia using the Korean genome and epidemiology study data. Coatings 2022;12(2):265. [Google Scholar]
14. Wu L, Li BH, Wang YY et al. Periodontal disease and risk of benign prostate hyperplasia: a cross-sectional study. Mil Med Res 2019;6(1):34. [Google Scholar] [PubMed]
15. Boland MR, Hripcsak G, Albers DJ et al. Discovering medical conditions associated with periodontitis using linked electronic health records. J Clin Periodontol 2013;40(5):474–482. [Google Scholar] [PubMed]
16. Fu E, Cheng CM, Chung CH et al. Association of chronic periodontitis with prostatic hyperplasia and prostatitis: a population-based cohort study in Taiwan. J Periodontol 2021;92(1):72–86. [Google Scholar] [PubMed]
17. Guo Z, Gu C, Li S et al. Periodontal disease and the risk of prostate cancer: a meta-analysis of cohort studies. Int Braz J Urol 2021;47(6):1120–1130. [Google Scholar] [PubMed]
18. Wei Y, Zhong Y, Wang Y, Huang R. Association between periodontal disease and prostate cancer: a systematic review and meta-analysis. Med Oral Patol Oral Cir Bucal 2021;26(4):e459–e465. [Google Scholar] [PubMed]
19. Ortíz de Urbina Comerón P, Zubizarreta-Macho A, Lobo Galindo AB et al. Relationship between prostate inflammation and periodontal disease-a systematic review and meta-analysis. J Clin Med 2023;12(18):6070. [Google Scholar]
20. Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 2010;25(9):603–605. [Google Scholar] [PubMed]
21. Ma LL, Wang YY, Yang ZH et al. Methodological quality (risk of bias) assessment tools for primary and secondary medical studies: what are they and which is better? Mil Med Res 2020;7(1):7. [Google Scholar] [PubMed]
22. Beger-Luedde J, Loosen SH, Luedde T, Roderburg C, Kostev K. Association between chronic gingivitis and cancer: a retrospective cohort study of 19,782 outpatients from the United Kingdom. Cancers 2023;15(7):2007. [Google Scholar] [PubMed]
23. Meurman JH, Kallmen H, Andersson LC, Yucel-Lindberg T, Soder B. Prevalence of cancer in relation to signs of periodontal inflammation. PLoS One 2022;17(10):e0276375. [Google Scholar] [PubMed]
24. Chung PC, Chan TC. Association between periodontitis and all-cause and cancer mortality: retrospective elderly community cohort study. BMC Oral Health 2020;20(1):168. [Google Scholar] [PubMed]
25. Kim DH, Jeong SN, Lee JH. Chronic periodontal disease increases risk for prostate cancer in elderly individuals in South Korea: a retrospective nationwide population-based cohort study. J Cancer 2020;11(16):4716–4723. [Google Scholar] [PubMed]
26. Güven DC, Dizdar Ö, Akman AC et al. Evaluation of cancer risk in patients with periodontal diseases. Turk J Med Sci 2019;49(3):826–831. [Google Scholar]
27. Heikkilä P, But A, Sorsa T, Haukka J. Periodontitis and cancer mortality: register-based cohort study of 68,273 adults in 10-year follow-up. Int J Cancer 2018;142(11):2244–2253. [Google Scholar] [PubMed]
28. Michaud DS, Kelsey KT, Papathanasiou E, Genco CA, Giovannucci E. Periodontal disease and risk of all cancers among male never smokers: an updated analysis of the health professionals follow-up study. Ann Oncol 2016;27(5):941–947. [Google Scholar] [PubMed]
29. Arora M, Weuve J, Fall K, Pedersen NL, Mucci LA. An exploration of shared genetic risk factors between periodontal disease and cancers: a prospective co-twin study. Am J Epidemiol 2010;171(2):253–259. [Google Scholar]
30. Hujoel PP, Drangsholt M, Spiekerman C, Weiss NS. An exploration of the periodontitis-cancer association. Ann Epidemiol 2003;13(5):312–316. [Google Scholar] [PubMed]
31. Hwang IM, Sun LM, Lin CL, Lee CF, Kao CH. Periodontal disease with treatment reduces subsequent cancer risks. QJM 2014;107(10):805–812. [Google Scholar] [PubMed]
32. Guo XP, Yang J, Wu L et al. Periodontitis relates to benign prostatic hyperplasia via the gut microbiota and fecal metabolome. Front Microbiol 2023;14:1280628. [Google Scholar] [PubMed]
33. Alluri LSC, Paes Batista da Silva A, Verma S et al. Presence of specific periodontal pathogens in prostate gland diagnosed with chronic inflammation and adenocarcinoma. Cureus 2021;13(9):e17742. [Google Scholar] [PubMed]
34. Zhu C, Li LY, Shi MH et al. Salmonella enterica mediated epigenetic promotion of fibrosis is a novel factor in benign prostatic hyperplasia. Mil Med Res 2025;12(1):24. [Google Scholar] [PubMed]
35. Baima G, Minoli M, Michaud DS et al. Periodontitis and risk of cancer: Mechanistic evidence. Periodontol 2000 2024;96(1):83–94. [Google Scholar] [PubMed]
36. Liu S, Liu F, Zhang B et al. CD4(+) T helper 17 cell response of aged mice promotes prostate cancer cell migration and invasion. Prostate 2020;80(10):764–776. [Google Scholar] [PubMed]
37. Liu Z, Liang Q, Ren Y et al. Immunosenescence: molecular mechanisms and diseases. Signal Transduct Target Ther 2023;8(1):200. [Google Scholar] [PubMed]
38. Bertolini M, Clark D. Periodontal disease as a model to study chronic inflammation in aging. Geroscience 2024;46(4):3695–3709. [Google Scholar] [PubMed]
39. Cao D, Sun R, Peng L et al. Immune cell proinflammatory microenvironment and androgen-related metabolic regulation during benign prostatic hyperplasia in aging. Front Immunol 2022;13:842008. [Google Scholar] [PubMed]
40. Du X, Wu J, Xia S, Zheng H, Zhang B. Unraveling the microbial landscape in prostate cancer: pathogenesis to therapy. NPJ Biofilms Microbiomes; 2026. [Google Scholar]
41. Groeger S, Wu F, Wagenlehner F et al. PD-L1 up-regulation in prostate cancer cells by porphyromonas gingivalis. Front Cell Infect Microbiol 2022;12:935806. [Google Scholar] [PubMed]
42. Li S, Cao H, Zhang Y et al. Association between periodontal disease and prostate disease: a mini review. Front Cell Infect Microbiol 2025;15:1669490. [Google Scholar] [PubMed]
43. Hajishengallis G. Periodontitis: from microbial immune subversion to systemic inflammation. Nat Rev Immunol 2015;15(1):30–44. [Google Scholar] [PubMed]
44. Yang HJ, Kim JH. Role of microbiome and its metabolite, short chain fatty acid in prostate cancer. Investig Clin Urol 2023;64(1):3–12. [Google Scholar] [PubMed]
45. Kumar J, Teoh SL, Das S, Mahakknaukrauh P. Oxidative stress in oral diseases: understanding its relation with other systemic diseases. Front Physiol 2017;8:693. [Google Scholar] [PubMed]
46. Oh B, Figtree G, Costa D et al. Oxidative stress in prostate cancer patients: a systematic review of case control studies. Prostate Int 2016;4(3):71–87. [Google Scholar] [PubMed]
47. Wen BW, Tsai CS, Lin CL et al. Cancer risk among gingivitis and periodontitis patients: a nationwide cohort study. QJM 2014;107(4):283–290. [Google Scholar] [PubMed]
48. Dizdar O, Hayran M, Guven DC et al. Increased cancer risk in patients with periodontitis. Curr Med Res Opin 2017;33(12):2195–2200. [Google Scholar] [PubMed]
49. Lee JH, Kweon HH, Choi JK, Kim YT, Choi SH. Association between periodontal disease and prostate cancer: results of a 12-year longitudinal cohort study in South Korea. J Cancer 2017;8(15):2959–2965. [Google Scholar] [PubMed]
50. Michaud DS, Liu Y, Meyer M, Giovannucci E, Joshipura K. Periodontal disease, tooth loss, and cancer risk in male health professionals: a prospective cohort study. Lancet Oncol 2008;9(6):550–558. [Google Scholar] [PubMed]
51. Kruck S, Hennenlotter J, Amend B et al. Chronic periodontitis does not impact serum levels of prostate-specific antigen. Anticancer Res 2017;37(6):3163–3167. [Google Scholar] [PubMed]
52. Alwithanani N, Bissada NF, Joshi N et al. Periodontal treatment improves prostate symptoms and lowers serum PSA in men with high PSA and chronic periodontitis. Dentistry 2015;5(3):1000284. [Google Scholar]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF
 Downloads
Downloads
 Citation Tools
Citation Tools