 Open Access
Open Access
ARTICLE
Proximal/Distal ureteral diameter ratio as a predictor of spontaneous passage in <10 mm ureteral stones
1 Department of Urology, School of Medicine, Ahi Evran University, Kirsehir, Turkiye
2 Department of Urology, Medipol University, Esenler Hospital, Istanbul, Turkiye
* Corresponding Author: İbrahim Üntan. Email:
(This article belongs to the Special Issue: Advances in Endoscopic Management of Urolithiasis)
Canadian Journal of Urology 2026, 33(2), 239-248. https://doi.org/10.32604/cju.2026.076805
Received 27 November 2025; Accepted 29 January 2026; Issue published 20 April 2026
Abstract
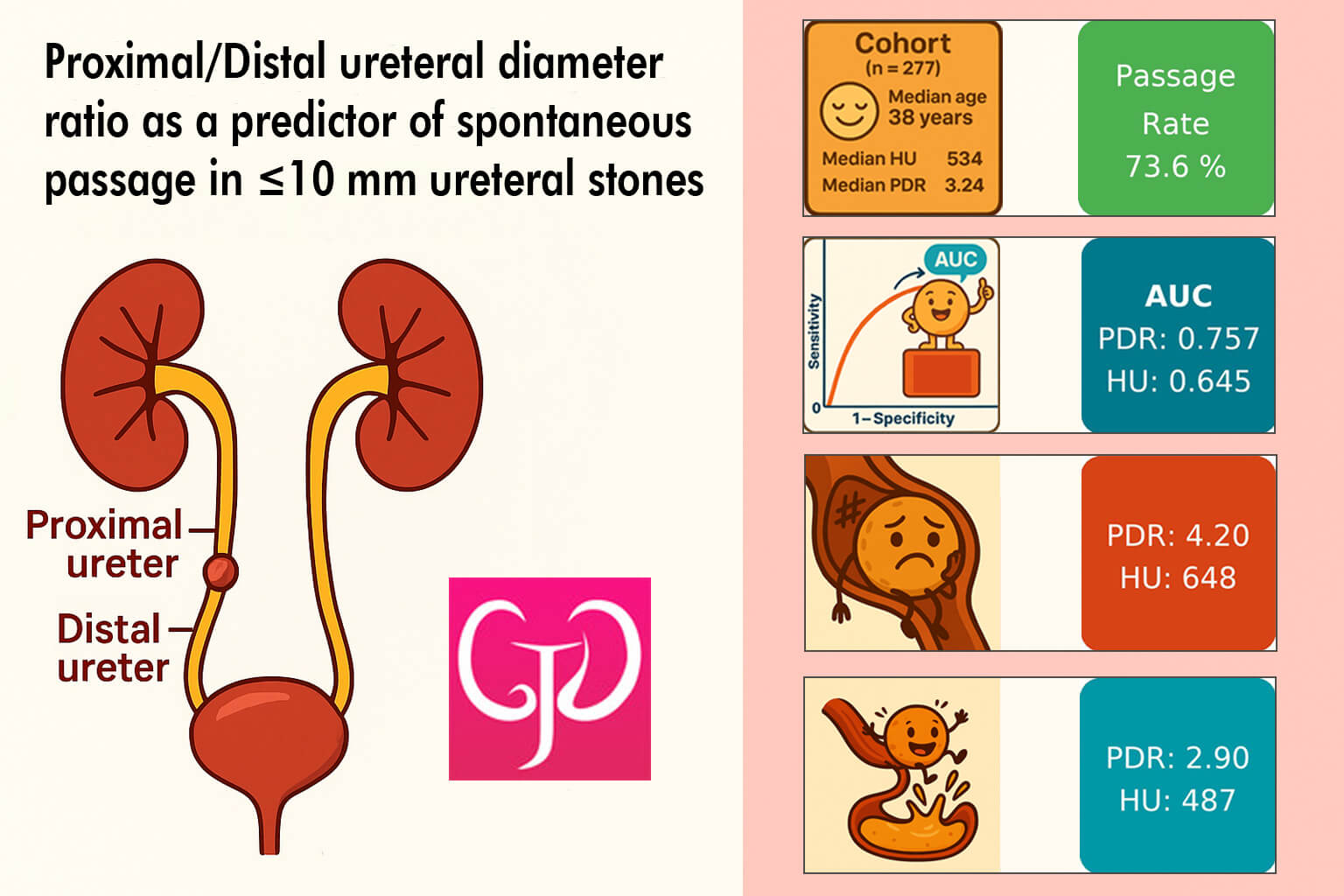
Background: Ureteral stones are a common cause of acute renal colic, and while most small stones pass spontaneously, reliable predictors of passage are needed to optimize patient selection for conservative management. This study aims to evaluate the role of radiologic, clinical and laboratory parameters—particularly the proximal/distal (P/D) ureteral diameter ratio—in predicting spontaneous passage of <10 mm ureteral stones. Materials and Methods: This retrospective cohort study included 277 patients diagnosed with ureteral calculi <10 mm on non-contrast computed tomography between September 2022 and September 2025. Pregnant patients, solitary kidney cases, congenital anomalies, active urinary infection, and incomplete computed tomography (CT) data were excluded. Proximal and distal ureteral diameters were measured 1 cm above and below the stone on axial CT images, and their ratio (P/D ratio) was calculated. Stones located at the ureterovesical or ureteropelvic junction were excluded from ratio analysis due to anatomic constraints. Spontaneous passage within 4 weeks was confirmed by follow-up ultrasonography in all patients. Patients were grouped into passage (n = 204) and no-passage (n = 73). Mann–Whitney U and chi-square tests were used for group comparisons. Predictive performance was assessed by receiver operating characteristic (ROC) analysis. Results: The spontaneous passage rate was 73.6%. Age, sex, and body mass index did not differ significantly between groups (p ≥ 0.05). Stone size (p < 0.001), location (p = 0.003), CT Hounsfield Unit (HU) (p < 0.001), and P/D ureteral diameter ratio (p < 0.001) were significantly associated with passage. Distal and ≤5 mm stones demonstrated markedly higher spontaneous passage rates. Median HU was lower in the passage group (487.00 [348.75–707.00]) than in the no-passage group (648.00 [467.00–846.00]). The P/D ratio was significantly lower in the passage group (2.90 [2.21–3.50]) compared with the no-passage group (4.20 [3.05–5.33]). Alpha-blocker use was not associated with passage (p = 0.232). ROC analysis demonstrated that stone size (area under the curve [AUC] 0.773) and P/D ratio (AUC 0.757) were the strongest predictors, outperforming HU (AUC 0.645) and location (AUC 0.617). Conclusions: Stone size, location, HU value, and the proximal/distal ureteral diameter ratio are significant markers associated with spontaneous passage of <10 mm ureteral stones. Lower HU values, distal location, smaller stone size, and a reduced P/D ratio favor spontaneous passage. The P/D ratio, a simple and practical CT-derived measurement, showed superior discriminative ability compared with HU and may assist clinicians in selecting appropriate candidates for conservative management.Graphic Abstract
Keywords
Ureteral stones are a common cause of acute renal colic and represent a significant proportion of urological emergency visits worldwide.1–3 The lifetime prevalence of urolithiasis ranges from 5% to 15%, and the incidence continues to rise globally.4,5 Spontaneous stone passage depends on multiple factors, including stone size, location, degree of ureteral obstruction, and local anatomic dynamics.6,7
Non-contrast computed tomography (CT) has become the preferred imaging modality for evaluating suspected ureteral calculi due to its high sensitivity and specificity.8,9 In addition to confirming stone presence, CT provides detailed morphologic and structural information that may help predict the likelihood of spontaneous passage.10 Prior studies have explored several CT-derived parameters—such as ureteral wall thickness, periureteral fat stranding, and hydronephrosis severity—as potential indicators of stone impaction and passage probability.11 However, the clinical utility and reproducibility of these parameters remain variable.12
Measurement of proximal and distal ureteral diameters relative to the stone has recently emerged as a promising imaging feature.13 Differences in luminal diameter above and below the stone reflect intraluminal pressure changes and the degree of functional obstruction, potentially offering insight into stone dynamics.14 Although the relationship between ureteral dilatation and obstruction has been described, the predictive value of the proximal/distal (P/D) ureteral diameter ratio for spontaneous passage of small ureteral stones (<10 mm) has not been adequately investigated.15,16
Given the clinical importance of distinguishing patients who will pass their stones spontaneously from those who may require intervention, practical and easily measurable CT parameters are needed. This study aims to evaluate the association between radiologic, clinical, and laboratory characteristics—including the P/D ureteral diameter ratio—and spontaneous passage of <10 mm ureteral stones within a 4-week follow-up period. By examining the discriminative performance of these parameters, we seek to determine their potential role in guiding conservative versus interventional management strategies.
Study design and patient selection
This retrospective cohort study was conducted in the Department of Urology at Medipol University Esenler Hospital after obtaining approval from the Istanbul Medipol University Non-Interventional Clinical Research Ethics Committee (Decision No.: 1282; Date: 30 October 2025). The requirement for written informed consent was waived due to the retrospective nature of the study. Medical records and imaging data of patients who presented with ureteral colic and were diagnosed with ureteral calculi on non-contrast CT between September 2022 and September 2025 were reviewed.
The inclusion criteria were as follows:
1. Age ≥18 years
2. Single ureteral stone measuring <10 mm in maximum transverse diameter on non-contrast CT
3. Stone located in the proximal, mid, or distal ureter
4. Clinical follow-up for spontaneous stone passage with conservative management
5. No intervening urological procedure during the initial 4-week observation period
6. Completion of follow-up ultrasonography at 4 weeks
The exclusion criteria were as follows:
1. Pregnancy
2. Solitary kidney
3. Congenital urinary tract anomalies (e.g., duplicated collecting system, horseshoe kidney, ectopic ureter)
4. Active urinary tract infection at presentation
5. Prior ureteral stenting or ureteral surgery
6. Multiple ureteral stones
7. Bilateral ureteral stones
8. Incomplete or inadequate CT data for measurement
9. UPJ stones precluding reliable proximal diameter measurement
10. UVJ stones precluding reliable distal diameter measurement
11. Loss to follow-up or incomplete follow-up data
Demographic characteristics (age, sex, body mass index), stone side, size, and location (proximal, mid-ureter, distal) were recorded. Alpha-blocker use (yes/no), serum creatinine level, and urinary red blood cell (RBC) categories (0–3) were also collected.
CT acquisition and measurement protocol
All CT scans were non-contrast studies performed on a 64-slice multidetector CT scanner (Siemens Somatom Definition, Siemens Healthcare, Erlangen, Germany) using a standardized institutional protocol. Acquisition parameters included 3-mm slice thickness, 120 kVp, and automatic tube current modulation. All measurements were performed on axial images with consistent window settings. Stone size was measured on axial images using the maximum transverse diameter.
To evaluate ureteral morphology, ureteral diameters were measured 1 cm proximal (above) and 1 cm distal (below) to the stone on axial CT images. Measurements were obtained by placing electronic calipers from outer wall to outer wall at the widest external diameter on the slice where the ureter was best visualized (Figure 1). The proximal/distal ureteral diameter ratio (P/D ratio) was calculated by dividing the proximal diameter by the distal diameter. All measurements were performed by a single experienced urologist blinded to clinical outcomes using a standardized measurement protocol to ensure consistency.

FIGURE 1. Coronal reformatted non-contrast computed tomography image illustrating the measurement protocol for the proximal/distal (P/D) ureteral diameter ratio. Although all measurements were performed on axial images, a coronal reconstruction is presented here to better demonstrate the spatial relationship between the measurement sites and the stone. The proximal ureteral diameter (0.92 cm) is measured 1.00 cm cranial to the stone, and the distal ureteral diameter (0.59 cm) is measured 1.00 cm caudal to the stone. Electronic calipers are placed from outer wall to outer wall at the widest external diameter of each segment. The ureteral calculus appears as a bright hyperattenuating focus (center of image). The P/D ratio in this representative case is 1.56 (0.92 ÷ 0.59), indicating moderate proximal ureteral dilatation relative to the distal segment
The Hounsfield Unit (HU) value of each stone was obtained using a region-of-interest (ROI) placed over the densest portion of the calculus.
Definition of spontaneous stone passage
Spontaneous stone passage was confirmed by follow-up imaging performed at approximately 4 weeks (range: 26–30 days). All patients underwent confirmatory ultrasonography performed by an experienced radiologist, including those reporting symptomatic improvement or witnessed stone expulsion. Stone passage was defined by absence of acoustic shadowing on ultrasonographic examination with resolution of hydronephrosis when present. Patients with persistent stone visualization on imaging were assigned to the no-passage group. Imaging findings were concordant with clinical assessment: all patients who reported witnessed stone expulsion demonstrated absence of stone on imaging, and symptom resolution was consistently associated with imaging confirmation of stone passage.
All analyses were performed using IBM SPSS Statistics 22.0 (IBM Corp., Armonk, NY, USA). Distribution of continuous variables was assessed using the Shapiro–Wilk test. Non-normally distributed variables were expressed as median [interquartile range (IQR)] and compared using the Mann–Whitney U test. Categorical variables were expressed as number (percentage) and compared using the Pearson chi-square test.
The discriminatory performance of predictors for spontaneous passage was evaluated using receiver operating characteristic (ROC) curve analysis, and the area under the curve (AUC) was calculated. Multivariable logistic regression was performed to assess the independent predictive value of the P/D ratio after adjusting for stone size and location. A p-value <0.05 was considered statistically significant.
A total of 277 patients with <10 mm ureteral stones were included in the analysis. All stones in the final cohort were located in the proximal ureter, mid-ureter, or distal ureter where reliable P/D ratio measurement is feasible; patients with stones at the UPJ or UVJ had been excluded during patient selection. Of these 277 patients, 204 (73.6%) experienced spontaneous stone passage within 4 weeks, while 73 (26.4%) did not.
Patient demographics (Table 1) were comparable between the passage and no-passage groups. Age (p = 0.624), sex distribution (p = 0.372), and body mass index (BMI) (p = 0.050) did not differ significantly between groups. Median age was 37.5 years [32.0–46.0] in the passage group and 38.0 years [33.0–46.0] in the no-passage group. The proportion of female patients was similar between groups (26.5% vs. 32.9%).

Stone characteristics (Table 2) showed significant associations with passage. Stone side did not differ between groups (p = 0.99). Stone size was strongly associated with spontaneous passage (p < 0.001). Compared with ≤5 mm stones (reference), stones measuring 5–7 mm had 3.6-fold higher odds of non-passage (OR 3.60; 95% CI 1.68–7.73), while 7–9 mm stones had 14.9-fold higher odds of non-passage (OR 14.86; 95% CI 7.06–31.27). Distal ureteral stones had the highest passage rate, followed by mid-ureteral and proximal stones (p = 0.003). Compared with distal location (reference), proximal stones had 3.0-fold higher odds of non-passage (OR 3.04; 95% CI 1.59–5.81).

CT findings (Table 3) demonstrated significant differences between groups. Median Hounsfield Unit (HU) values were significantly lower in the passage group compared with the no-passage group (487.00 [348.75–707.00] vs. 648.00 [467.00–846.00], p < 0.001). Stones with HU ≥500 had 2.3-fold higher odds of non-passage compared with HU <500 (OR 2.30; 95% CI 1.31–4.02, p = 0.005).

Proximal ureteral diameter was significantly greater among patients without passage (8.9 mm [6.60–10.30]) than those with passage (6.1 mm [4.67–7.00], p < 0.001), whereas distal diameter did not differ significantly (p = 0.502).
The proximal/distal ureteral diameter ratio (P/D ratio) was markedly higher in the no-passage group (4.20 [3.05–5.33] vs. 2.90 [2.21–3.50], p < 0.001). A P/D ratio ≥4.0 was associated with 8.2-fold higher odds of non-passage compared with P/D ratio <4.0 (OR 8.18; 95% CI 4.45–15.02), while P/D ratio ≥3.0 demonstrated 4.4-fold higher odds of non-passage (OR 4.43; 95% CI 2.41–8.15).
Laboratory findings (Table 3) showed that urinary red blood cell (RBC) category differed significantly between groups (p = 0.036); negative hematuria was more common in the passage group (51.5% vs. 32.9%). Serum creatinine levels were similar between groups (p = 0.257).
ROC analysis (Table 4, Figure 2) demonstrated that stone size had the highest discriminative performance (AUC 0.773; 95% CI 0.710–0.832), followed closely by the P/D ureteral diameter ratio (AUC 0.757; 95% CI 0.682–0.829). Both HU value (AUC 0.645; 95% CI 0.574–0.718) and stone location (AUC 0.617; 95% CI 0.546–0.689) showed moderate discriminative ability. The P/D ratio demonstrated superior predictive performance compared with both established CT parameters (HU and location).


FIGURE 2. Receiver operating characteristic curves for prediction of spontaneous passage in <10 mm ureteral stones. AUC: Area under the curve; CI: Confidence interval; P/D: Proximal/distal; HU: Hounsfield unit
In multivariable logistic regression adjusting for stone size and location (Table 5), stone size remained the strongest independent predictor (OR 3.72, 95% CI 2.46–5.64, p < 0.001). The P/D ratio showed a trend toward significance (OR 1.05 per unit, 95% CI 0.98–1.11, p = 0.148), while stone location was not significant (p = 0.992). The addition of P/D ratio to a model containing size and location improved discriminative ability (AUC: 0.775 vs. 0.819, ΔAUC + 0.044).

In this retrospective study, we evaluated the role of clinical, laboratory, and imaging parameters in predicting spontaneous passage of <10 mm ureteral stones. Our findings demonstrate that stone size, stone location, CT Hounsfield Unit (HU) value, and the proximal/distal ureteral diameter ratio are significant determinants of spontaneous passage. In contrast, patient-related factors such as sex, body mass index, serum creatinine level, and use of alpha-blocker therapy showed no meaningful association with passage outcomes.
The overall spontaneous passage rate of 73.6% in our cohort aligns well with previously reported natural-history data within 4–6-week follow-up windows. Existing natural-history evidence suggests that the majority of <10 mm ureteral stones pass spontaneously within 4–6 weeks, with reported 4-week rates of around 50%–75% and substantially higher cumulative rates by 6 weeks.17,18 Evidence from randomized controlled studies indicates that spontaneous passage at 4 weeks typically falls between 50% and 70% among patients managed conservatively.19,20 Together, these data support the appropriateness of the 4-week conservative follow-up period used in our study.
Stone size was the strongest predictor of spontaneous passage, demonstrating both the highest odds ratio (OR 14.86; 95% CI 7.06–31.27 for 7–9 mm vs. ≤5 mm stones) and discriminative performance (AUC 0.773; 95% CI 0.710–0.832). Our findings—with passage rates of 90.3% for ≤5 mm stones compared with 38.5% for 7–9 mm stones—mirror multiple prior analyses showing diminishing passage rates with increasing size. Published data consistently show that ≤5 mm ureteral stones have substantially higher spontaneous passage rates, whereas stones measuring 5–10 mm exhibit markedly reduced likelihood of passage within the early follow-up period.21–23 These convergent findings reinforce stone size as a principal determinant of conservative management success.
Stone location was another major determinant of passage probability. Existing evidence consistently shows that distal ureteral stones have a substantially higher likelihood of spontaneous passage compared with stones located in the mid or proximal ureter.24–26 Anatomical factors, ureteral peristalsis, and lower functional resistance in the distal ureter may explain these differences and support the long-established principle that distal stones are more amenable to expectant management.23,27
Lower stone density on CT is generally linked to higher rates of spontaneous passage, indicating that less dense calculi are more likely to migrate distally without intervention.28,29 Higher HU values generally reflect greater mineral content and stone hardness, which may resist spontaneous dislodgement.30 Our finding that the non-passage group exhibited significantly higher HU values (648 vs. 487 HU, p < 0.001) corroborates this interpretation.
The proximal/distal ureteral diameter ratio demonstrated strong predictive performance (AUC 0.757; 95% CI 0.682–0.829), approaching that of stone size (AUC 0.773; 95% CI 0.710–0.832) and outperforming stone location (AUC 0.617; 95% CI 0.546–0.689), both of which are routinely used in clinical decision-making. While the improvement over HU (ΔAUC = 0.112) is moderate, the clinically relevant comparison is that the P/D ratio approaches the discriminative ability of stone size (ΔAUC = 0.016), the strongest predictor, while substantially outperforming stone location (ΔAUC = 0.140), both of which are routinely used in clinical practice. The P/D ratio’s value lies not in replacing stone size assessment, but in providing complementary information about the functional degree of obstruction when clinical decision-making is uncertain.
In multivariable analysis adjusting for stone size and location, the P/D ratio showed a trend toward independent predictive value (p = 0.148) and improved model discrimination (ΔAUC + 0.044). While not achieving statistical independence, this likely reflects shared pathophysiology between stone size and ureteral obstruction dynamics—larger stones create greater proximal dilatation. The P/D ratio may offer particular value as a confirmatory measurement in borderline or equivocal cases where stone size alone does not clearly indicate optimal management strategy, or when image quality limits precise size assessment. As a simple, reproducible CT measurement, it provides insight into the functional degree of obstruction that complements anatomic stone characteristics.
Recent evidence highlights the predictive value of morphometric CT parameters, particularly those reflecting ureteral wall changes and impaction, suggesting that local inflammatory and obstructive dynamics may improve clinical risk stratification.14,29 Abat et al. previously demonstrated that an elevated upper-to-lower ureteral diameter ratio was associated with stone impaction.13 Our findings extend this work by demonstrating that the P/D ratio also predicts spontaneous passage outcomes. An increased P/D ratio in our study was indicative of elevated proximal pressure and potential impaction, explaining its association with reduced passage rates. The concordance between our findings and those of Abat et al. supports the concept that ureteral diameter asymmetry reflects clinically meaningful obstruction dynamics.
Alpha-blocker therapy did not significantly influence passage outcomes in our cohort (p = 0.232). These results are in keeping with recent large randomized studies showing that tamsulosin does not significantly improve spontaneous passage in small distal stones.20,31–33 While some meta-analyses suggest potential benefit for stones >5 mm, smaller stones typically exhibit high baseline passage rates, limiting the additive effect of medical expulsive therapy.34
Higher urine red blood cell levels in the non-passage group are consistent with evidence linking microscopic hematuria to ureteral impaction.35 Increased mucosal irritation and mechanical embedding of the stone may account for this relationship, suggesting that microscopic hematuria may serve as a subtle indicator of impaction during follow-up.
Overall, our results support the clinical relevance of stone size, stone location, HU value, and especially the proximal/distal ureteral diameter ratio in predicting spontaneous passage. These parameters reflect both the physical characteristics of the stone and the functional dynamics of ureteral obstruction, offering a more nuanced assessment than size alone. Future prospective, multicenter studies with larger cohorts are warranted to validate the predictive performance of this ratio and to explore its incorporation into individualized management algorithms.
The P/D ratio may be influenced by hydration status, baseline ureteral anatomy, and acute ureteral spasm. However, measurements during acute presentation when obstructive hemodynamics are maximal showed consistent associations with outcomes. The retrospective, single-center design may limit generalizability. Alpha-blocker use was not randomized; treated patients had smaller stones (62.3% vs. 25.6% ≤5 mm, p < 0.001) and more distal location (74.4% vs. 14.1%, p < 0.001). This selection bias toward better-prognosis stones would favor finding a treatment effect; the absence of benefit (p = 0.232) aligns with recent randomized trials.
This study demonstrates that stone size, stone location, CT HU value, and the proximal/distal ureteral diameter ratio are important predictors of spontaneous passage in <10 mm ureteral stones. Passage likelihood decreases with increasing stone size and more proximal locations, while higher HU values and elevated proximal/distal diameter ratios are associated with reduced passage rates. In contrast, alpha-blocker therapy, sex, body mass index, and serum creatinine level do not significantly influence spontaneous passage outcomes.
The superior discriminative ability of the proximal/distal ureteral diameter ratio compared with HU value highlights its potential utility as a simple, reproducible imaging metric that reflects underlying obstruction dynamics. Our findings suggest that a 4-week conservative management period is both safe and effective for appropriately selected patients, particularly those with distal stones and lower HU values.
Incorporating morphometric parameters such as the proximal/distal diameter ratio into clinical assessment may contribute to more individualized and accurate decision-making in the management of small ureteral stones. Further large-scale prospective studies are needed to validate these results and support integration into predictive modeling frameworks.
Acknowledgement
Not applicable.
Funding Statement
This research received no external funding.
Author Contributions
Nuh Aldemir conceived and designed the study, collected data, and wrote the draft. İbrahim Üntan reviewed the literature, performed the analyses, and prepared the manuscript. All authors provided critical feedback to each other and contributed to the final manuscript after discussing the results and commenting on the manuscript. The manuscript has been read and approved by all authors. There are no other persons who met the authorship criteria but are not listed. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials
The datasets supporting the findings of the current study are openly available at 10.5281/zenodo.17683073.
Ethics Approval
This study was approved by the Istanbul Medipol University Non-Interventional Clinical Research Ethics Committee (Decision No.: 1282; Date: 30 October 2025). The requirement for written informed consent was waived due to the retrospective nature of the study. Informed consent was waived by the ethics committee due to the retrospective nature of this study.
Conflicts of Interest
The authors declare no conflicts of interest.
References
1. Patti L, Leslie SW. Acute renal colic. In: StatPearls. Updated 2024 Dec 23 ed. Treasure Island, FL, USA: StatPearls Publishing; 2025. doi:10.1080/15424065.2024.2389325. [Google Scholar] [CrossRef]
2. Stamatelou K, Goldfarb DS. Epidemiology of kidney stones. Healthcare 2023;11(3):424. doi:10.3390/healthcare11030424. [Google Scholar] [PubMed] [CrossRef]
3. Shilling M, Faber LS, Doan H et al. Emergency room management of urolithiasis: a sex-based comparison. Can J Urol 2025;32(2):81–88. doi:10.32604/cju.2025.063415. [Google Scholar] [PubMed] [CrossRef]
4. Romero V, Akpinar H, Assimos DG. Kidney stones: a global picture of prevalence, incidence, and associated risk factors. Rev Urol 2010;12(2–3):e86–96. [Google Scholar] [PubMed]
5. Tae BS, Balpukov U, Cho SY, Jeong CW. Eleven-year cumulative incidence and estimated lifetime prevalence of urolithiasis in Korea: a National Health Insurance Service-National Sample Cohort based study. J Korean Med Sci 2018;33(2):e13. doi:10.3346/jkms.2018.33.e13. [Google Scholar] [PubMed] [CrossRef]
6. Coll DM, Varanelli MJ, Smith RC. Relationship of spontaneous passage of ureteral calculi to stone size and location as revealed by unenhanced helical CT. Am J Roentgenol 2002;178(1):101–103. doi:10.2214/ajr.178.1.1780101. [Google Scholar] [PubMed] [CrossRef]
7. Wang C, Di M, Qin J, Wang F, He T, Zhang R. Applying urinary ultrasound to predict the risk of spontaneous ureteral stone passage: a retrospective cohort study. BMC Urol 2024;24(1):171. doi:10.1186/s12894-024-01558-w. [Google Scholar] [PubMed] [CrossRef]
8. Fulgham PF, Assimos DG, Pearle MS, Preminger GM. Clinical effectiveness protocols for imaging in the management of ureteral calculous disease: AUA technology assessment. J Urol 2013;189(4):1203–1213. doi:10.1016/j.juro.2012.10.031. [Google Scholar] [PubMed] [CrossRef]
9. Rapp DE, Wood NL, Bassignani M, Gergoudis L, Caulkins S, Kramolowsky EV. Clinical variables and stone detection in patients with flank pain. Can J Urol 2016;23(5):8441–8445. [Google Scholar] [PubMed]
10. Selvi I, Baydilli N, Tokmak TT, Akinsal EC, Basar H. CT-related parameters and Framingham score as predictors of spontaneous passage of ureteral stones <10 mm: results from a prospective, observational, multicenter study. Urolithiasis 2021;49(3):227–237. doi:10.1007/s00240-020-01214-6. [Google Scholar] [PubMed] [CrossRef]
11. Guzel R, Yildirim S, Temizkan HT, Asik A, Yasar H, Sarica K. Prediction of the impaction of proximal ureteral stones: a critical evaluation of the patient- and stone-related factors affecting the ureteral wall thickness. J Clin Med 2025;14(17):6081. doi:10.3390/jcm14176081. [Google Scholar] [PubMed] [CrossRef]
12. Popiolek M, Lidén M, Georgouleas P, Sahlén K, Sundqvist P, Jendeberg J. Radiological signs of stone impaction add no value in predicting spontaneous stone passage. Urolithiasis 2024;52(1):114. doi:10.21203/rs.3.rs-4376792/v1. [Google Scholar] [CrossRef]
13. Abat D, Börekoğlu A, Altunkol A, Köse I, Boğa MS. Is there any predictive value of the ratio of the upper to the lower diameter of the ureter for ureteral stone impaction? Curr Urol 2021;15(3):161–166. doi:10.1097/cu9.0000000000000019. [Google Scholar] [PubMed] [CrossRef]
14. Dean NS, Millan B, Uy M et al. Ureteral wall thickness is an effective predictor of ureteral stone impaction and management outcomes: a systematic review and meta-analysis. J Urol 2023;210(3):430–437. doi:10.1097/ju.0000000000003561. [Google Scholar] [PubMed] [CrossRef]
15. Heiniger Y, Foerster B, Bodmer NS et al. Prediction for spontaneous passage of ureteral stones with indwelling ureteral stent: PASS score. World J Urol 2025;43(1):259. doi:10.1007/s00345-025-05616-2. [Google Scholar] [PubMed] [CrossRef]
16. Kadirhan O, Aydin S, Keskin E, Kantarci M. Role of ureteral wall thickness and computed tomography imaging in predicting spontaneous passage of ureteral stones. Diagn Interv Radiol 2025. doi:10.4274/dir.2025.253304. [Google Scholar] [PubMed] [CrossRef]
17. Ahmed AF, Gabr AH, Emara AA, Ali M, Abdel-Aziz AS, Alshahrani S. Factors predicting the spontaneous passage of a ureteric calculus of ⩽10 mm. Arab J Urol 2015;13(2):84–90. doi:10.1016/j.aju.2014.11.004. [Google Scholar] [PubMed] [CrossRef]
18. Yallappa S, Amer T, Jones P et al. Natural history of conservatively managed ureteral stones: analysis of 6600 patients. J Endourol 2018;32(5):371–379. doi:10.1089/end.2017.0848. [Google Scholar] [PubMed] [CrossRef]
19. Pickard R, Starr K, MacLennan G et al. Medical expulsive therapy in adults with ureteric colic: a multicentre, randomised, placebo-controlled trial. Lancet 2015;386(9991):341–349. doi:10.1016/s0140-6736(15)60933-3. [Google Scholar] [PubMed] [CrossRef]
20. Meltzer AC, Burrows PK, Wolfson AB et al. Effect of tamsulosin on passage of symptomatic ureteral stones: a randomized clinical trial. JAMA Intern Med 2018;178(8):1051–1057. doi:10.1001/jamainternmed.2018.2259. [Google Scholar] [PubMed] [CrossRef]
21. Ongün Ş., Dursun M. The effect of distal ureteral stone size measurements on spontaneous passage. J Urol Surg 2019;6:139–143. doi:10.4274/jus.galenos.2018.2335. [Google Scholar] [CrossRef]
22. Park JS, Kim DW, Lee D et al. Development of prediction models of spontaneous ureteral stone passage through machine learning: comparison with conventional statistical analysis. PLoS One 2021;16(12):e0260517. doi:10.1371/journal.pone.0260517. [Google Scholar] [PubMed] [CrossRef]
23. Baumgarten L, Desai A, Shipman S et al. Spontaneous passage of ureteral stones in patients with indwelling ureteral stents. Can J Urol 2017;24(5):9024–9029. doi:10.1016/j.juro.2016.02.695. [Google Scholar] [CrossRef]
24. Jendeberg J, Geijer H, Alshamari M, Cierzniak B, Lidén M. Size matters: the width and location of a ureteral stone accurately predict the chance of spontaneous passage. Eur Radiol 2017;27(11):4775–4785. doi:10.1007/s00330-017-4852-6. [Google Scholar] [PubMed] [CrossRef]
25. Tchey DU, Ha YS, Kim WT, Yun SJ, Lee SC, Kim WJ. Expectant management of ureter stones: outcome and clinical factors of spontaneous passage in a single institution’s experience. Korean J Urol 2011;52(12):847–851. doi:10.4111/kju.2011.52.12.847. [Google Scholar] [PubMed] [CrossRef]
26. Ordon M, Andonian S, Blew B, Schuler T, Chew B, Pace KT. CUA guideline: management of ureteral calculi. Can Urol Assoc J 2015;9(11–12):E837–E851. doi:10.5489/cuaj.3483. [Google Scholar] [PubMed] [CrossRef]
27. Modai J, Barda N, Avda Y, Shpunt I, Leibovici D, Shilo Y. Do patients with ureterolithiasis treated conservatively return to follow up? Can J Urol 2021;28(1):10536–10541. [Google Scholar] [PubMed]
28. Erturhan S, Bayrak O, Mete A, Seckiner I, Urgun G, Sarica K. Can the Hounsfield unit predict the success of medically expulsive therapy? Can Urol Assoc J 2013;7(11–12):E677–E680. doi:10.5489/cuaj.352. [Google Scholar] [PubMed] [CrossRef]
29. Khan RU, Nazim SM, Anwar S. CT-based predictors of spontaneous ureteral stone passage. J Coll Physicians Surg Pak 2024;34(8):879–884. doi:10.29271/jcpsp.2024.08.879. [Google Scholar] [PubMed] [CrossRef]
30. Cohen A, Anderson B, Gerber G. Hounsfield Units for nephrolithiasis: predictive power for the clinical urologist. Can J Urol 2017;24(3):8832–8837. [Google Scholar] [PubMed]
31. Al-Ansari A, Al-Naimi A, Alobaidy A, Assadiq K, Azmi MD, Shokeir AA. Efficacy of tamsulosin in the management of lower ureteral stones: a randomized double-blind placebo-controlled study of 100 patients. Urology 2010;75(1):4–7. doi:10.1016/j.urology.2009.09.073. [Google Scholar] [PubMed] [CrossRef]
32. Furyk JS, Chu K, Banks C et al. Distal ureteric stones and tamsulosin: a double-blind, placebo-controlled, randomized, multicenter trial. Ann Emerg Med 2016;67(1):86–95. doi:10.1016/j.annemergmed.2015.06.001. [Google Scholar] [PubMed] [CrossRef]
33. Abdel-Meguid TA, Tayib A, Al-Sayyad A. Tamsulosin to treat uncomplicated distal ureteral calculi: a double blind randomized placebo-controlled trial. Can J Urol 2010;17(3):5178–5183. [Google Scholar] [PubMed]
34. Wang RC, Smith-Bindman R, Whitaker E et al. Effect of tamsulosin on stone passage for ureteral stones: a systematic review and meta-analysis. Ann Emerg Med 2017;69(3):353–61. doi:10.1016/j.annemergmed.2016.06.044. [Google Scholar] [PubMed] [CrossRef]
35. Katayama S, Yoshioka T, Sako T et al. Association of severe microscopic hematuria with successful conservative treatment of single uncomplicated ureteral calculus: a multicenter cohort study. Eur Urol Focus 2021;7(4):812–817. doi:10.1016/j.euf.2020.04.004. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools