 Open Access
Open Access
REVIEW
A Comprehensive Review and Algorithmic Analysis of Histogram-Based Contrast Enhancement Techniques for Medical Imaging
1 School of Electrical Engineering and Computer Science, National University of Sciences and Technology, Islamabad, Pakistan
2 College of Engineering and Computing, American University of Bahrain, Riffa, Bahrain
3 Faculty of Computing and IT, Sohar University, Sohar, Sultanate of Oman
4 IRC for Finance and Digital Economy, King Fahd University of Petroleum and Minerals, Dhahran, Saudi Arabia
5 Department of Information Systems, College of Computer and Information Sciences, Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia
6 Department of Computer Science, College of Computer Science and Information Systems, Najran University, Najran, Saudi Arabia
7 Immersive Virtual Reality Research Group, Department of Computer Science, King Abdulaziz University, Jeddah, Saudi Arabia
* Corresponding Authors: Maqbool Khan. Email: ; Wadee Alhalabi. Email:
Computer Modeling in Engineering & Sciences 2026, 146(3), 4 https://doi.org/10.32604/cmes.2026.074688
Received 16 October 2025; Accepted 09 February 2026; Issue published 30 March 2026
Abstract
Medical imaging is essential in modern health care, allowing accurate diagnosis and effective treatment planning. These images, however, often demonstrate low contrast, noise, and brightness distortion that reduce their diagnostic reliability. This review presents a structured and comprehensive analysis of advanced histogram equalization (HE)-based techniques for medical image enhancement. Our review methodology encompasses: (1) classical HE approaches and related limitations in medical domains; (2) adaptive schemes like Adaptive Histogram Equalization (AHE) and Contrast Limited Adaptive Histogrma Equalization (CLAHE) and their advance variants; (3) brightness-preserving schemes like BBHE and MMBEBHE and related algorithms; (4) dynamic and recursive histogram equalization methods incorporating DHE and RMSHE; (5) fuzzy logic-based enhancement methodologies addressing uncertainty and noise in medical images; and (6) hybrid optimization methodologies through the application of metaheuristic algorithms (World Cup Optimization, Particle Swarm Optimization, Genetic Algorithms, along with histogram-based methodologies.) There is also a comparative discussion given based on contrast improvement, image brightness preservation, noise management, and computational efficiency. Such advancements have better capabilities of improving image quality, which is more important for improved diagnosis and image analysis.Graphic Abstract
Keywords
Medical imaging, which visualizes the internal structure of the body, plays a crucial role in the diagnosis and treatment of many diseases. It includes X-ray, computed tomography (CT), positron emission tomography (PET), magnetic resonance imaging (MRI), sonography, and more. However, accurate diagnosis depends on the quality of the image. Specific issues such as low contrast, noise, blur anatomical borders, and obscure minute yet critical diagnostic details, such as early-stage tumors [1]. These quality drawbacks can result in “clinical misinterpretation” and a significant decline in the reliability of image-based decision-making. This can hinder the analysis process. These may drastically lower diagnosis accuracy, resulting in inaccurate interpretations and a decline in clinical confidence in image-based decision-making.
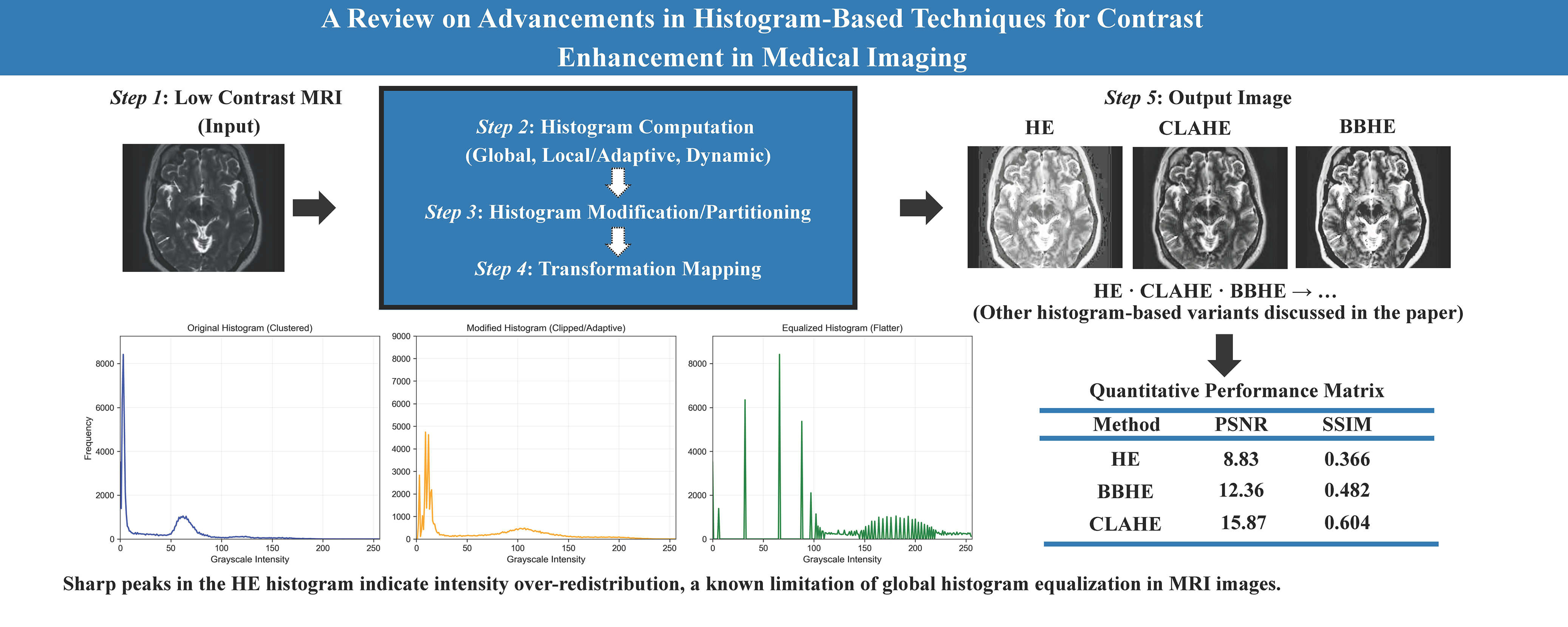
To overcome these problems, image enhancement techniques were developed, such as base–detail layer decomposition combined with adaptive gamma correction, which has been shown to improve CT image visibility without introducing noise or halo artifacts [2]. Image enhancement is one of the primary operations that improves the clarity and visibility of unclear or less visible parts of an image, as well as enhances low contrast. Its main goal is to improve the quality, clarity and to enhance interpretability of images [3]. Contrast enhancement is a crucial part of image improvement. The techniques have changed over time. Among the many methods created over the past few decades, histogram-based techniques stand out due to their computational efficiency and adaptability to different imaging modalities. Fig. 1 shows the MRI images of the brain. We used this dataset to further do analysis (Table 1).
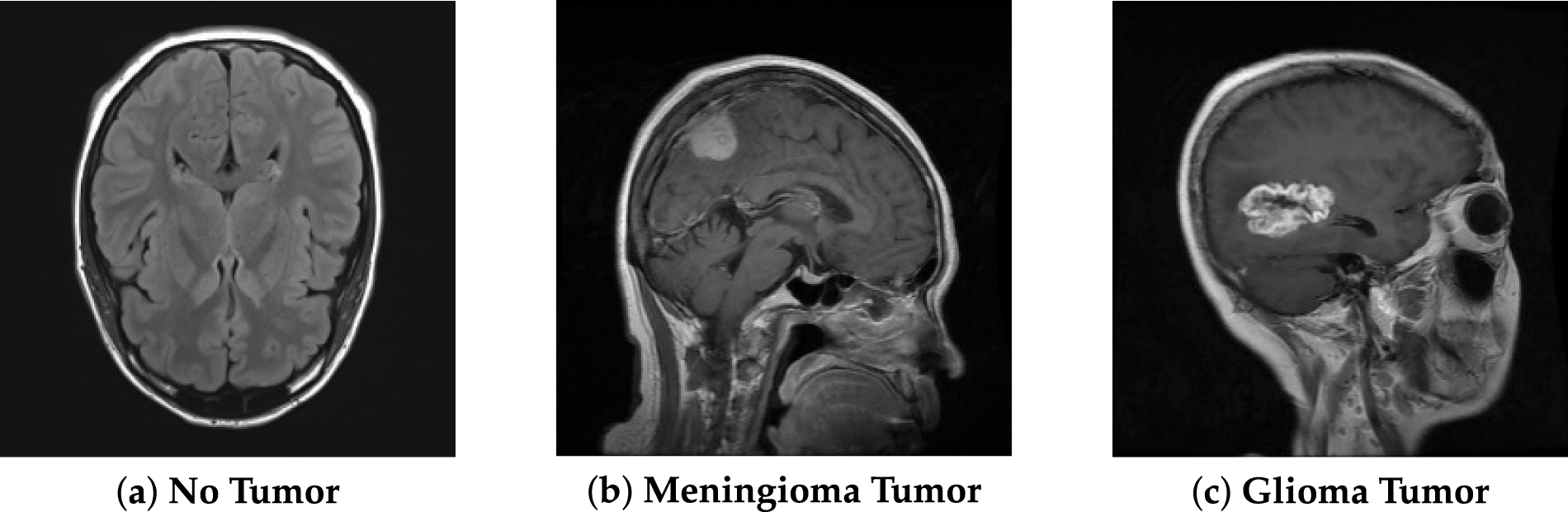
Figure 1: Brain MRI, data from Kaggle [4].
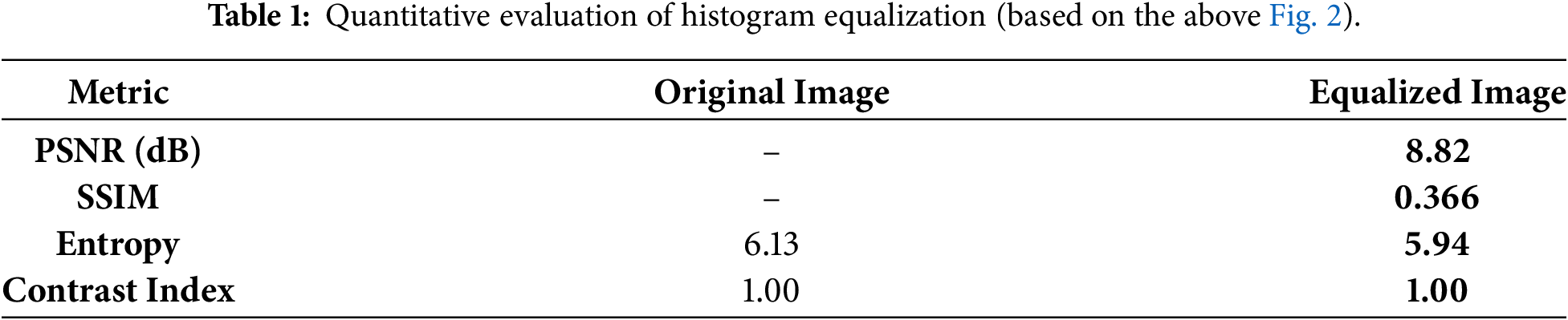
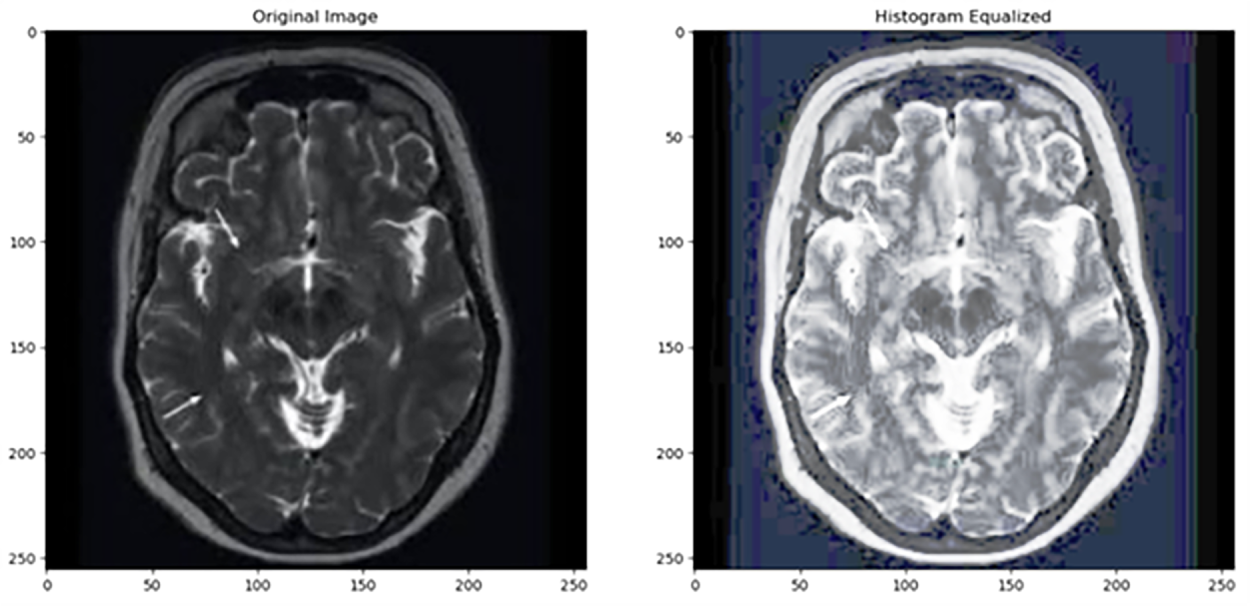
Figure 2: As shown in this figure, histogram equalization introduces excessive contrast in homogeneous regions. Source: Kaggle dataset [4].
A crucial step in the analysis and diagnosis of medical images is the enhancement of contrast. To enhance the appearance, contrast enhancement can be used to darken the dark areas and brighten the light areas. Image Enhancement methods fall into two categories: the Frequency domain and the Spatial domain [5].
Spatial Domain: In spatial domain, in order to enhance the image, pixel values are changed directly [6]. Three major categories can be used to group these techniques:
(a) A global approach, in which the entire image is used, to apply a single transformation algorithm.
(b) A local process, in which every pixel is transformed by taking neighboring pixels into account.
(c) A hybrid strategy that blends local and global methods [7].
The main benefit of spatial-based domain techniques is their simplicity and ease of use [8].
Frequency Domain: As authors discussed in [9], this technique uses Fourier transform to work on images in the spatial domain to refine them and modifies pixel clusters directly. It encompasses Fourier transform, DWT (Discrete Wavelet Transform), and DCT (Discrete Cosine Transform).
Histogram Equalization (HE) is a spatial domain technique. It increases contrast, improves image quality, and enhances the visibility of features in MRI brain images [3]. It is widely used and is one of the simplest methods for contrast enhancement. For instance, the authors in [10] compared histogram equalization and filtering techniques for medical thermal imaging in breast cancer detection. They found that while histogram equalization enhances contrast, it skews the temperature profile, that is a fundamental aspect for diagnosis, and potentially resulting in misinterpretation. Since filtering methods such as Wiener filtering preserve the original temperature information, they are more appropriate for accurate clinical analysis. Fig. 2 shows that histogram equalization (HE) can lead to over-enhancement, resulting in exaggerated contrast in certain regions of the image. Recently in 2022, Histogram Equalization with Minimum Cross-Entropy-Otsu [11] was proposed; it enhances medical image contrast while preserving brightness, improving diagnostic quality with low computational complexity, but it may not handle very noisy images.
Table 1 provides a quantitative comparison between the original and histogram-equalized image from Fig. 2. The equalized image shows a very low PSNR (8.82 dB) and SSIM (0.366), indicating substantial distortion and low similarity to the original. Although both images have the same contrast index (1.00), entropy slightly decreases after equalization (from 6.13 to 5.94), suggesting a small loss in information content. Overall, these metrics confirm that histogram equalization, while enhancing contrast, significantly degrades image quality and alters its statistical properties.
Later, a contrast enhancement framework that combines Reformed Histogram Equalization (RHE), Krill Herd Optimization (KH), and Gray Level Co-Occurrence Matrix (GLCM) was introduced [12]. RHE is a more sophisticated version of conventional histogram equalization, especially designed for improving medical images. This method reduces over-enhancement while maintaining the image’s naturalness by methodically moving residue pixels to empty histogram bins and using a dynamic plateau limit to clip histogram peaks. In order to improve the strategy, we employ a bio-inspired metaheuristic algorithm named KH that automatically learns the optimal plateau limit parameter without any human intervention. For texture feature preservation during the enhancement process, the technique uses a novel fitness function that combines two objective functions: the first takes edge information and entropy measures into account, while the second assesses GLCM contrast and energy parameters.
Fig. 3 shows the output for both the original and the histogram-equalized image. The original image has low contrast, most bars are bunched on the left. While the histogram-equalized image improved the contrast but over-enhanced it. Medical images, particularly skeletal and lung images, were used in a method that combines histogram equalization, the Laplace transform, and the Sobel operator [13]. To sharpen the image and bring out details, the Laplace transform, also known as the Laplacian operator, is used to identify edges and highlight regions where the brightness of the image changes rapidly. The Sobel operator is an edge detection algorithm based on the first-order differential of an image’s intensity function [14]. Then, HE is used to improve overall contrast and brightness, which facilitates the separation of delicate tissues and structures. Recent research work introduced Range Limited Weighted Histogram Equalization (RLWHE) [15], which was particularly developed to enhance the quality of low-contrast medical images. This technique combines weighted histogram equalization (It cleverly extends only selected levels of brightness to enhance contrast without losing details), adaptive gamma correction (It automatically adjusts the brightness transfer function to brighten up dark regions and bring out concealed details), and homomorphic filtering (It is a kind of photo filter that selectively brightens up dark regions and reduces harsh highlights to correct lighting irregularities) to enhance contrast, preserve brightness, and details. This method is computationally intensive and has been validated only on grayscale images.
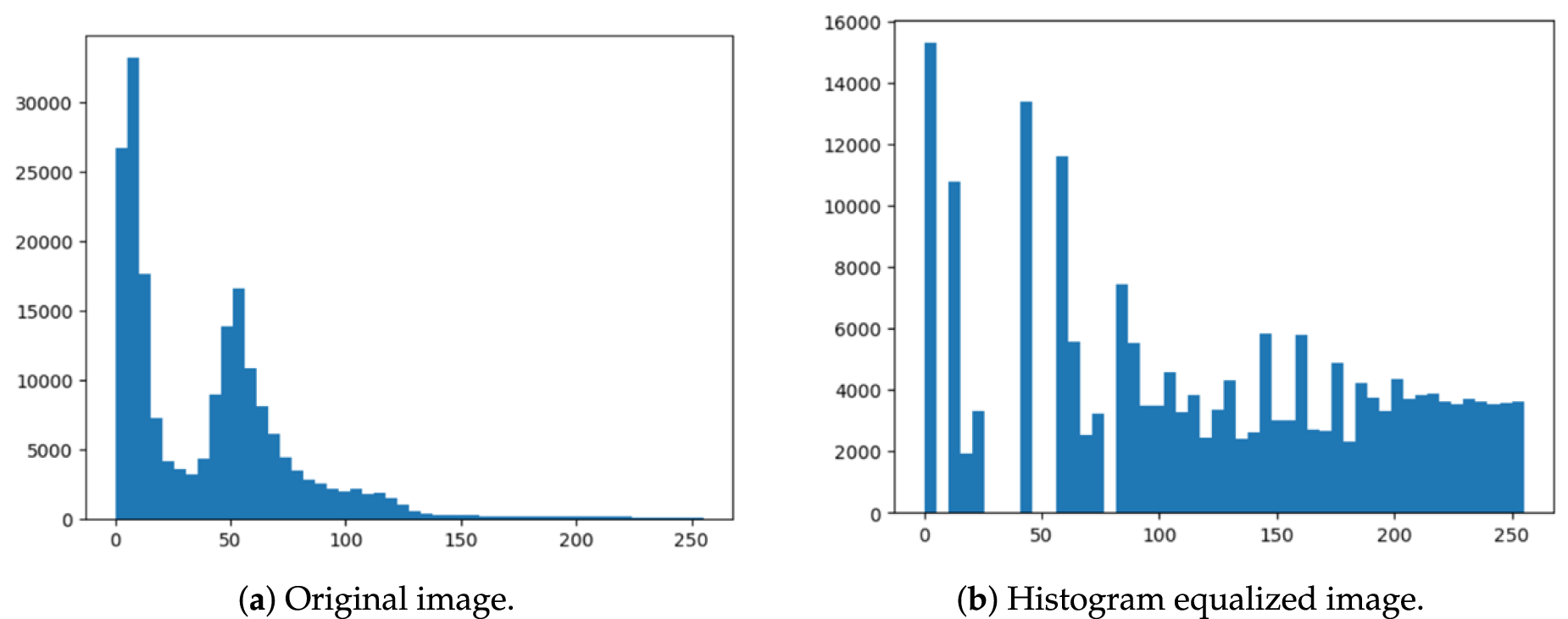
Figure 3: Referring to the images in Fig. 2, here is the comparison of the original and histogram equalized images.
Fig. 4 presents a comprehensive overview that we discussed in this review paper. A list of abbreviations used in the manuscript is provided at the end of the document.
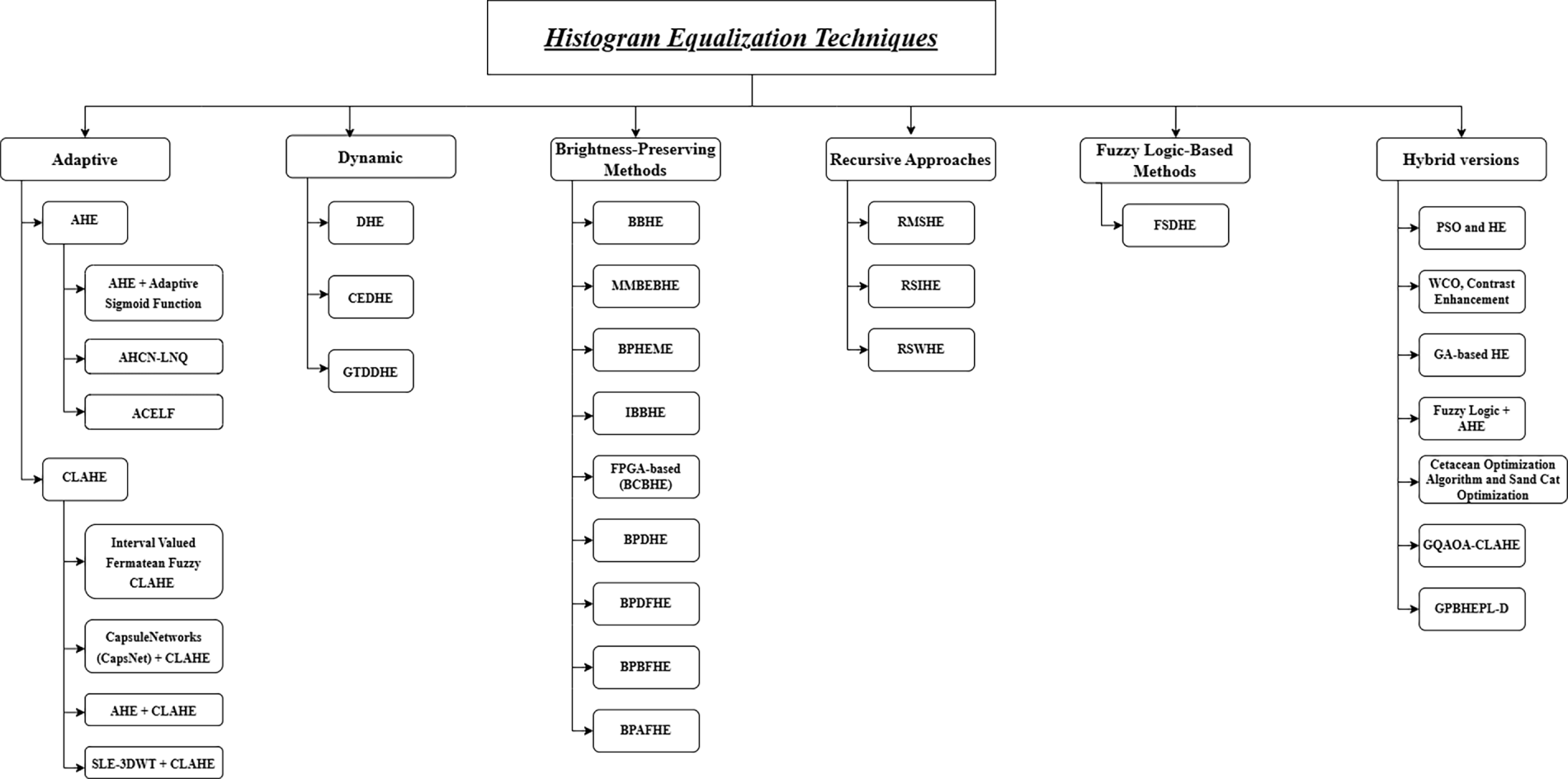
Figure 4: Overall framework and classification of histogram equalization techniques for medical image contrast enhancement.
2 Advanced Histogram-Based Contrast Enhancement Techniques
Adaptive Histogram Equalization (AHE) [16] was first introduced as a solution to the side effects of conventional histogram equalization (HE). Unlike global HE, AHE computes histograms for local neighborhood regions, thereby generating region-specific transformations that better preserve local contrast. In radiology, where precise image interpretation is essential for diagnosis and treatment, AHE is helpful in a variety of medical procedures. Its ability to automate contrast enhancement offers consistent results across large image datasets, which is highly beneficial in clinical environments that require batch processing and reliable visualization for diagnostic purposes [17]. Regionally adaptive histogram equalization [18] is used to increase the contrast of chest X-rays by selectively improving the contrast of low-contrast areas like the mediastinum while maintaining the appearance of the lungs and reducing noise. Although it improves the visibility of structures of interest, it depends on predefined anatomy and is computationally more complex.
Recently, Adaptive Histogram Equalization based on an Adaptive Sigmoid Function (AHEASF) was proposed [19]. The suggested approach aims to enhance the visual quality of medical images and adapt them to specific diagnostic tasks better than the originals. The input image is separated into numerous smaller sub-images using a “split-and-conquer” approach. For each sub-image, an adaptive sigmoid function is employed to generate a smooth enhancement curve that is applied independently, thereby overcoming some limitations of conventional HE through adaptive contrast control and generalization.
Another study introduced an enhanced image pre-processing technique called Adaptive Histogram Contrast Normalization with Learning-based Neural Quantization (AHCN-LNQ) [20]. The method increases the specificity, accuracy, and precision of brain tumor detection by combining intelligent neural network-based classification with contrast enhancement. To improve the visual quality of MRI images for brain tumor detection, this study employs AHCN as a preprocessing step. By redistributing pixel intensity values according to their histogram, the procedure improves the contrast of the input images. The adaptive approach separates the image into tiles and applies local histogram equalization to each tile, in contrast to global histogram equalization, which applies the same transformation to the entire image.
Adaptive Contrast Enhancement With Lesion Focusing (ACELF) [21] is an advanced contrast-enhancement scheme tailored for brain lesion detection in MRI and CT scans, where low contrast and poor detail resolution often hinder diagnosis. The method begins by segmenting brain tissue using Otsu’s thresholding [22], which selects a threshold that minimizes the overlap between foreground (lesion-like) and background (normal tissue) pixel intensities. By comparing local intensities with expected normal-tissue values, ACELF identifies candidate lesion regions. Next, ACELF applies adaptive contrast enhancement via custom histogram matching and gamma correction to globally improve contrast while preserving fine anatomical details. The algorithm implements entropy scaling to control enhancement strength because it needs to protect noisy or complex areas from becoming over-enhanced while increasing visibility in simpler regions where lesions might be hidden and maintaining detailed elements of the image. Finally, lesion-specific enhancement is performed by adjusting intensity values and applying localized histogram equalization, either AHE or CLAHE, to sharpen lesion boundaries and improve diagnostic clarity.
Huang et al. [23] proposed Contrast Limited Dynamic Quadri-Histogram Equalization (CLDQHE), a histogram equalization (HE)-based method designed to enhance image contrast while effectively preserving brightness and structural details. To address limitations in traditional HE approaches—such as over-enhancement and over-smoothing—the method divides the original image histogram into four sub-histograms using a novel threshold scheme. It then applies an adaptive histogram clipping technique with a dynamically adjusted clipping threshold to regulate the enhancement rate and mitigate excessive contrast amplification. Finally, each sub-histogram is independently mapped to a new dynamic range and equalized. Another research proposed the Region-based modified histogram equalization enhances non-uniformly illuminated chest X-rays by independently processing underexposed, well-exposed, and overexposed regions, improving contrast and preserving naturalness [24]. For clinical applications, the choice of enhancement method should be driven by the specific imaging modality and diagnostic goal. Recent studies have leveraged pre-processing like CLAHE with CNNs for improved TB detection [25]. We will look into CLAHE in the next section. Below, Table 2 provides a guide by mapping each technique’s profile to typical applications.
2.1 Contrast Limited Adaptive Histogram Equalization
Contrast-limited adaptive histogram equalization (CLAHE) is helpful and greatly effective for low-contrast images, it restrains contrast enhancements in homogeneous areas to avoid noise amplification [26]. Compared to conventional histogram equalization, CLAHE is applied to small regions of the image (tiles). It focuses only on local contrast and may ignore the overall (global) brightness/contrast structure. An application of this algorithm to a medical image is shown in Fig. 5.
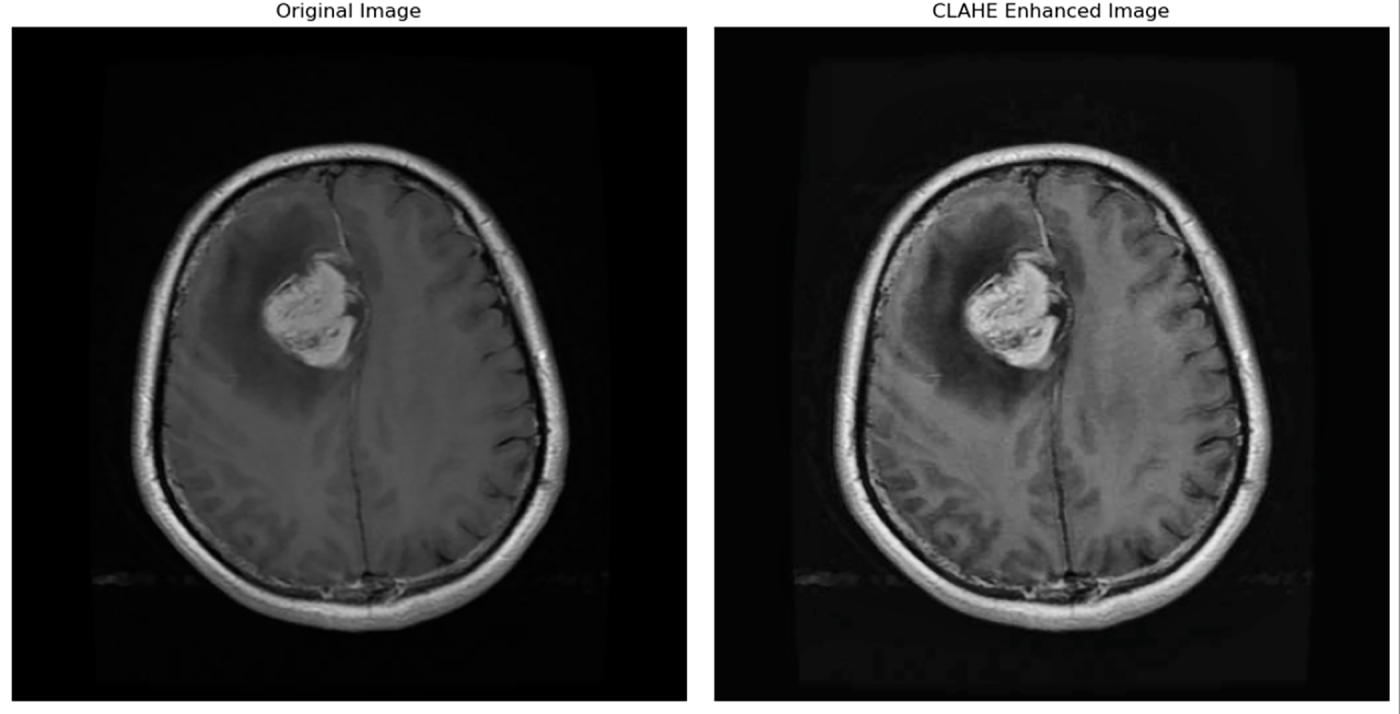
Figure 5: Image before and after applying CLAHE [4].
In 1998, Pisano et al. [27] highlighted CLAHE, stating that it constrains noise amplification by clipping the histogram at a specified limit. In medical imaging, where too much noise can mask clinically significant details, this kind of clipping is especially crucial. To maintain significant details in medical imaging, it is essential. Without increasing noise, CLAHE improves visibility by locally enhancing contrast and adjusting to both high-contrast and low-contrast areas.
GHE (Global Histogram Equalization) preserves global characteristics but loses local details [28]. To simultaneously preserve local details and the global brightness distribution in medical X-rays, Nia and Shih [29] propose Global CLAHE, which combines the advantages of GHE and CLAHE. First, GHE is applied to the input image. The clipping factor is then adjusted each time CLAHE is applied iteratively. After each iteration, the result is compared to the global reference using a similarity metric (like SSIM). Until similarity stops improving, the process is repeated to produce an image with better local details while preserving global brightness and contrast.
Research indicates that the use of CLAHE greatly improves the visibility of microcalcifications, which are tiny white dots mainly made of calcium, in mammograms, enabling radiologists to spot possible breast cancer symptoms much earlier [30]. In addition to revealing significant features, CLAHE’s capability enhances overall diagnostic confidence by reducing false negatives in areas that are typically problematic for conventional contrast enhancement techniques [31]. In a related study, used CLAHE on low-contrast CT scan images in particular. Using it to enhance image contrast without amplifying noise excessively locally [32].
The N-CLAHE algorithm, which combines log normalization with CLAHE, has been proven to improve digital chest X-ray images by increasing local contrast and maintaining anatomical structures. The algorithm performs better than traditional image enhancement algorithms, including histogram equalization, unsharp masking, and CLAHE, in terms of image quality [33]. In a related study, the 3D CLAHE method [34] enhances MRI volumes by improving local contrast and reducing artifacts, aiding diagnosis and treatment; it is effective but may increase noise with larger block sizes. Another paper introduced a method to decompose an input medical image into three separate components: structure (
Opoku et al. [38] combined Capsule Networks (CapsNet) with CLAHE. CapsNet is a type of neural network that serves as an alternative to traditional Convolutional Neural Networks (CNNs). Prior to feature extraction, CLAHE improves contrast and lowers noise in OCT images. This technique enables ophthalmologists to diagnose macular diseases more quickly and accurately. A group of eye conditions known as macular diseases affects the macula, which facilitates the transmission of images from the eye’s optic nerve to the brain.
Härtinger and Steger [39] suggested a novel algorithm that blends AHE and CLAHE. When calculating the histogram, this has a constant-time complexity. Regardless of the size of the filter mask used, this innovation enables efficient image processing with significantly faster execution times without compromising the high quality of the images.
Several advancements have been made to overcome the limitations of classical CLAHE. A recent method, Contrast Limited Adaptive Local Histogram Equalization (CLALHE) [40], enhances image contrast locally and adaptively without requiring manual parameter selection. The approach applies multiple enhancement iterations to identify optimal parameters, then divides the image into subimages and applies these parameters independently to emphasize local features. Evaluated on DIARETDB1, Pasadena-Houses 2000, and Faces 1999 datasets, CLALHE outperformed conventional methods in terms of PSNR, entropy, absolute mean brightness error, SSIM, contrast improvement index, and RMSE, producing well-contrasted images with better-defined details and reduced artifacts.
A 3D U-Net model for hippocampus segmentation in MRI scans was developed [41] to aid in the diagnosis and tracking of Alzheimer’s disease and other neurodegenerative diseases. They used SCE-3DWT (Selective Coefficient-Enhanced 3D Wavelet Transform) and 3D CLAHE as their two primary pre-processing methods. Hippocampus segmentation tasks can benefit from the 3D CLAHE, which is an extension of the classic CLAHE algorithm modified for 3D data.
Using advanced Artificial Intelligence (AI) techniques, specifically by combining image fusion and deep learning methods, another paper [42] proposed a method that increases the accuracy of brain tumor classification in MRI images. By enhancing contrast (CLAHE) and capturing spatial/frequency details (DWT), image fusion (CLAHE + DWT) improves the quality of MRIs. Additionally, it extracts a variety of hierarchical features by combining DenseNet121 and DenseNet169. Through the attention mechanism, the model is able to concentrate on the main components of the image. Finally, the Chirplet Transform, an advanced signal processing technique, is used to detect subtle tumor features.
Contrast Limited Adaptive Histogram Equalization (CLAHE) is widely used for local contrast enhancement in medical imaging. Recent advancements have focused on improving its adaptability and effectiveness. A notable example is the Neighbourhood-based Improved CLAHE (NICLAHE) method [43], which dynamically adjusts the clip limit and tile size relative to local pixel values, instead of using fixed values as in classical CLAHE. NICLAHE significantly enhances retinal image contrast and the visibility of fine structures, improving segmentation performance in automated diagnosis using deep learning frameworks such as ResNet. Evaluations on the DRIVE and HRF datasets demonstrated superior PSNR, RMSE, and overall contrast compared to traditional CLAHE, highlighting its effectiveness in retinal image preprocessing. There’s an advanced medical image enhancement method that optimizes CLAHE parameters (Clip Limit and Region Size) using an improved cuckoo search algorithm with dynamic parameters and hybrid distribution to improve contrast in MRI images [44]. It gives highest PSNR, and lowest distortion. It maintains edges and textures. One of the disadvantages is that it is slow (25 min). Its not for real time use. Another methods, comprising of two-step that uses PSO to optimize CLAHE parameters for image enhancement, then applies Active Contour Model to segment retinal blood vessels for diagnosing eye diseases [45]. It can work best on any dataset. It can automatically select parameters (no manual tuning). It’s best for diabetic retinopathy and vascular disease screening but struggles with pathological regions and lacks real-time diagnosis. In a different application domain, another automated machine learning framework integrating CLAHE image enhancement and ANN classification was developed for quick and accurate burn severity assessment, particularly valuable in emergency and resource-limited settings [46]. Another machine learning-enhanced image enhancement technique that automatically optimizes CLAHE parameters, Dynamic Region-Based CLAHE (DR-CLAHE) [47] employs XGBoost and Random Forest models to automatically determine optimal CLAHE parameters from image statistical and structural features. FLTSCLAHE [48], a fuzzy logic-based technique combining two-stage CLAHE and Laplacian filtering, has demonstrated superior performance in MRI brain image enhancement. A diabetic retinopathy detection system [49] utilizing CLAHE for preprocessing, combined with Median filtering and Geometric Active Contour for optic disc segmentation, achieved 99.006% accuracy using Bag of Visual Words (BOW) based SVM classification on 130 color fundus images.
In recent research [50], a hybrid machine learning pipeline for early skin cancer detection was proposed. The system combines multiple techniques:
• CLAHE (Contrast Limited Adaptive Histogram Equalization) for image enhancement
• FCM (Fuzzy C-Means) for lesion segmentation
• CNN (Convolutional Neural Network) for deep feature extraction
• SVM (Support Vector Machine) for final classification
Another method fuses multi-modal medical images (CT and MRI) using CLAHE for contrast enhancement [51], Non-Subsampled Contourlet Transform (NSCT) for multi-scale decomposition, and guided filtering for edge-preserving detail fusion. Studies on breast cancer [52] and liver tumor [53] datasets show that CLAHE boosts classification accuracy and efficiency, especially when combined with data augmentation techniques. Table 3 summarizes core strengths, practical limitations, and specific applications in medical imaging, highlighting the trade-offs between enhancement quality, computational complexity, and clinical suitability.
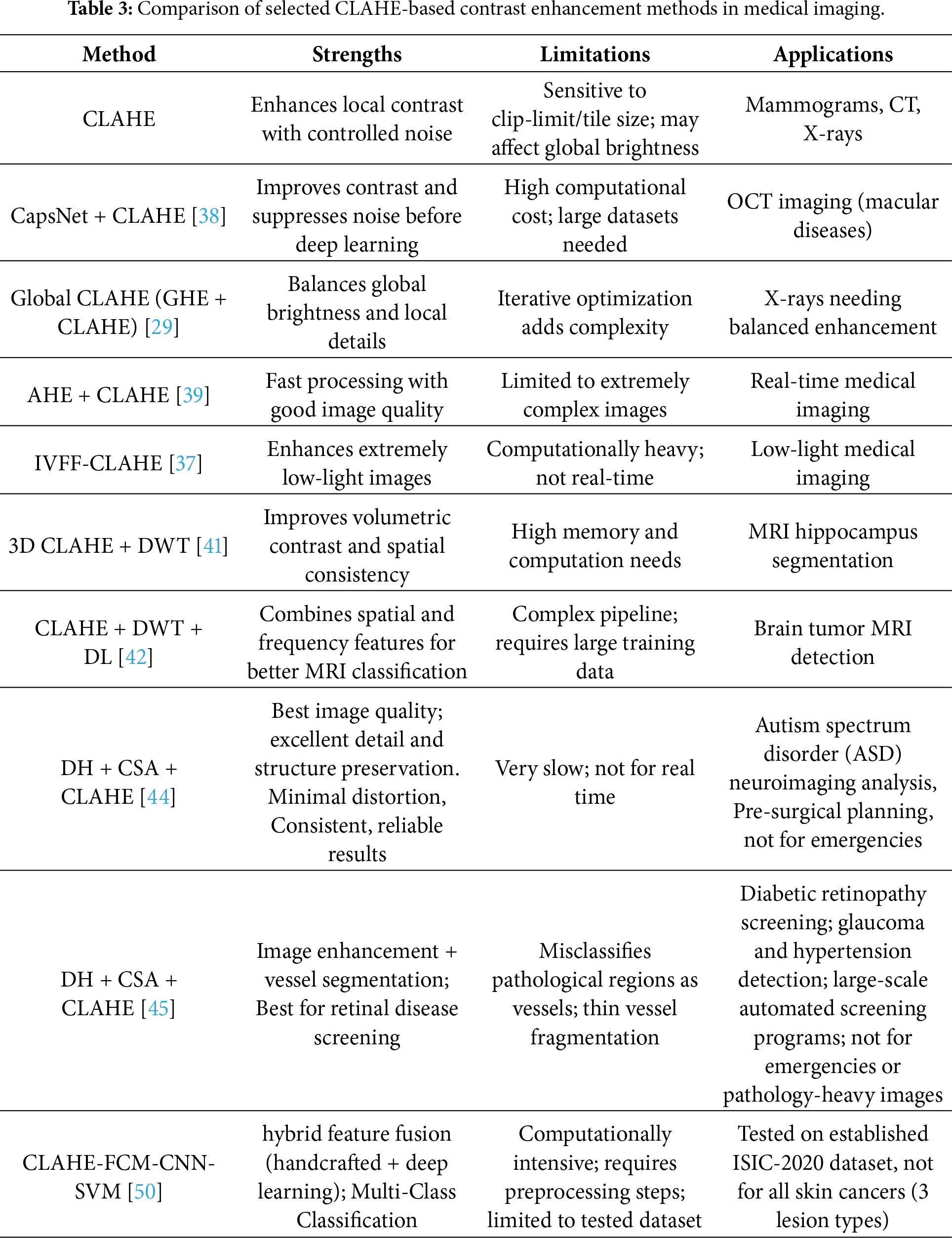
2.2 Brightness Preserving Bi Histogram Equalization
Brightness Preserving Bi-Histogram Equalization (BBHE) was developed for contrast enhancement in order to improve the quality of diagnostic images, including MRIs, liver cancer scans, and X-rays [54]. This method avoids one of the main drawbacks of conventional histogram equalization, which is that it can change the brightness of images and mask crucial diagnostic data. Based on the original image’s mean intensity, BBHE splits the input image into two sub-images. In one sub-image, the pixel values are higher than the mean, whereas in the other, they are either equal to or lower than the mean. Each sub-image is then histogram equalized separately to improve contrast while maintaining the overall brightness of the original image. The paper shows equations like:
Iterative Brightness Bi-Histogram Equalization (IBBHE), as proposed by [55], divides an image into two parts by iteratively adjusting a threshold, improving contrast while maintaining the image’s mean brightness. To ensure better contrast enhancement and brightness preservation, particularly useful in medical imaging. Each component is histogram equalized separately, and the threshold is adjusted through several iterations until it is optimal. Moniruzzaman et al. [56] proposed an enhanced histogram equalization (HE) method to improve Brightness-Biased HE (BBHE). Their approach first detects edges using the Canny method, then determines the most frequent gray level (M) among edge pixels. The image is split into two sub-images at M-darker (
An image contrast enhancement technique called FPGA-based Brightness Configurable Bi-Histogram Equalization (BCBHE) system [57] is based on BBHE. However, it is implemented in hardware using an FPGA (Field Programmable Gate Array) for real-time processing. A programmable logic unit is added, allowing users to adjust the brightness for optimal visual quality. In another paper, developed an automated system for detecting pneumonia using chest X-ray images [58]. To enhance image quality and draw attention to pertinent features, they employed image enhancement techniques, particularly CLAHE and BBHE. To extract features and categorize images into normal and pneumonia groups, they used a custom deep convolutional neural network (CNN) in conjunction with transfer learning, model VGG19.
BBHE and the Pelican Optimization Algorithm (POA) were combined in a recent study [59]. It performs better than conventional HE variants thanks to its optimization-driven methodology. It is applicable to medical imaging. By preserving brightness and boosting contrast, BBHE enhances medical imaging. Although its ICF (Image Contrast Function) values are higher than those of some other algorithms, ERBMAHE (Exposure Region Based Multi-Algorithm Histogram Equalization) [60] method improves local contrast more successfully. However, the method requires higher computational resources due to its multi-stage processing. Despite this, ERBMAHE consistently outperforms several state-of-the-art methods in both qualitative and quantitative evaluations, making it a valuable tool for improving diagnostic accuracy in medical imaging. Another research work [61] presented an exposure-based modified histogram equalization approach for improving non-uniformly exposed chest X-ray images. In contrast to conventional global histogram equalization methods, their approach segments CXR images into under-exposed (UE) and over-exposed (OE) parts based on an exposure value. Non-linear histogram equalization is applied to each part independently, with intensity transformation depending on the exposure value. The processed parts are then combined without over-enhancing. The results, obtained on the COVID-19 Radiography Database, showed improved entropy, PSNR, MSE and AMBE. The method is fully automatic and has potential applications in medical CAD (Computer-Aided Design) systems.
2.3 Dualistic Sub-Image Histogram Equalization
Dualistic Sub-Image Histogram Equalization (DSIHE) was later proposed [62]. It claims that when the median of the input image brightness is used as the histogram separation level, completing two independent histogram equalizations results in maximum entropy. By splitting the histogram into two segments and applying equalization to each separately, DSIHE improves medical imaging. This method helps improve lung CT scans and aid in the early detection of lung cancer by boosting contrast. Furthermore, DSIHE addresses the common issues with conventional histogram equalization, such as noise amplification, under-enhancement, and over-enhancement [63].
Based on the median gray level, input image is divided into two sub-images, a method similar to other approaches. Sub-image will include pixels with gray levels greater than and pixels with gray levels less than or equal to. After that, sub-images are extracted. Determine their PDFs and CDFs. Each sub-image is equalized independently. The enhanced output image is created by combining these equalized sub-images, which guarantees balanced contrast enhancement and better image quality while maintaining brightness. On CT scans, it has shown remarkable success in improving the visibility of tiny nodules and other early indicators of lung cancer. Ullah et al. [64], conducted a study on a deep learning model called DeepEBTDNet, which is intended to identify brain tumors from MRI pictures. The authors used the DSIHE technique. Before being fed into the DeepEBTDNet model, DSIHE was used to improve the quality of MRI images.
Mohan and Thirugnanam [65] demonstrated that by recursively partitioning histograms to preserve brightness better and enhance contrast, Recursive Mean-Separate Histogram Equalization (RMSHE) performs noticeably better than BBHE and DSIHE. In the sections that follow, we will provide an in-depth look at RMSHE and its benefits.
2.4 Dynamic Histogram Equalization
Dynamic Histogram Equalization (DHE) [66] can enhance image contrast but maintain more detail than conventional HE. It is less likely to produce unnatural-looking artifacts within the image. By dividing the histogram into sub-histograms at local minima and further dividing them if one sub-histogram predominates, DHE improves image contrast. To prevent artifacts, gray-level ranges are assigned to sub-histograms according to span and frequency (managed by factor x), and HE is applied separately to each sub-histogram within its allotted range. For further details regarding DHE workflow, look into Fig. 6. The figure depicts its core procedure.
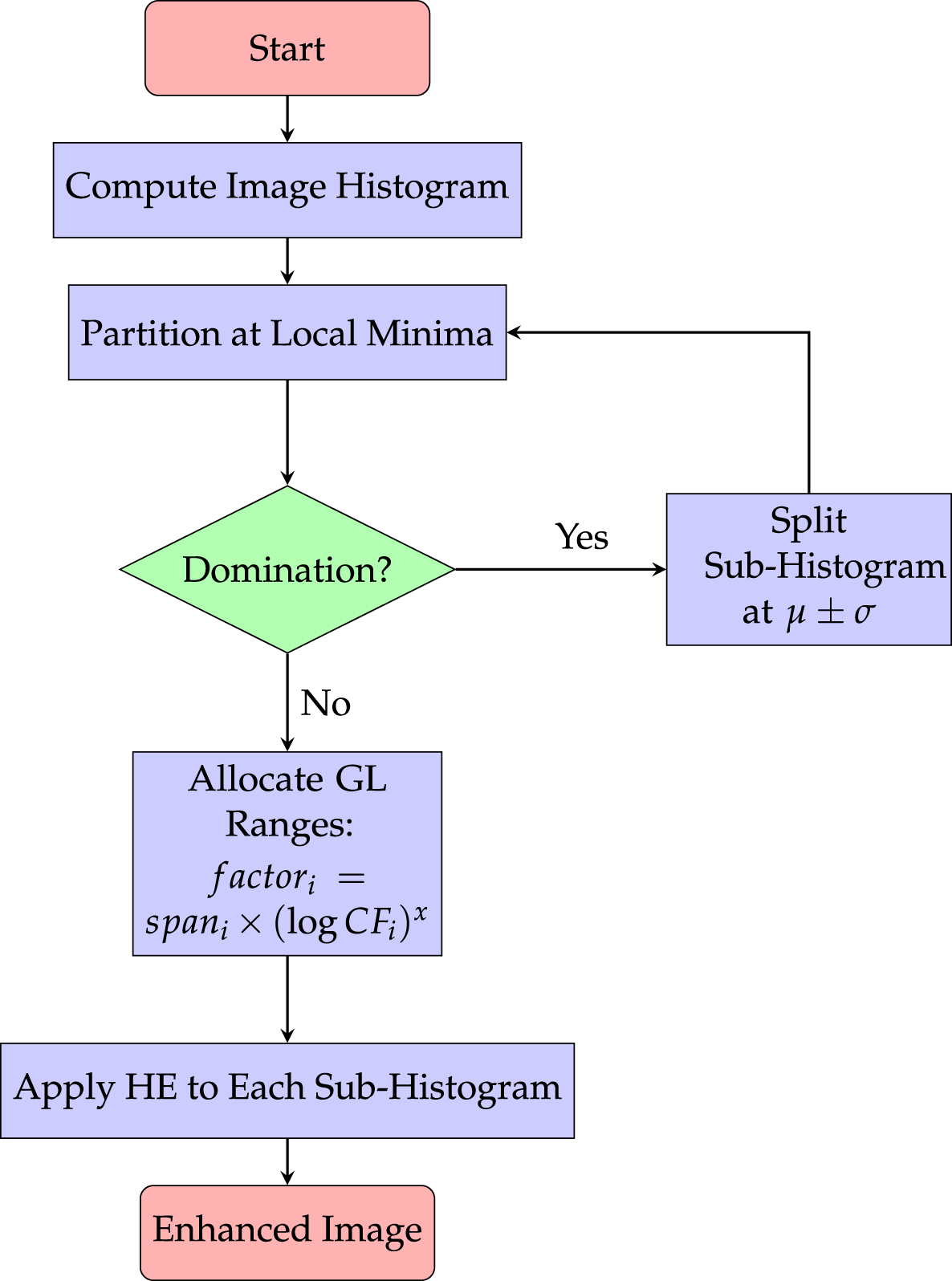
Figure 6: Workflow of Dynamic Histogram Equalization (DHE), illustrating adaptive contrast enhancement that preserves local brightness while improving global image details [66].
Abbasi et al. [67] claimed that DHE preserves the image’s brightness and original appearance while enabling reversible data hiding. This facilitates secure transmission and effective cancer prediction through appropriate image analysis. It does not, however, completely preserve the initial brightness. This restriction lessens its applicability in fields where constant brightness is essential, such as medical imaging.
Contrast enhancement dynamic histogram equalization (CEDHE) [68] is a technique for medical images that enhances visibility and makes fine details in brain images more visible without over-processing. This is addressed by later variations such as BPDHE, which use variance-based segmentation and brightness normalization [69].
Another proposed method, TCDHE-DWT-SVD [70] enhances low-contrast CT and X-ray images by improving brightness, contrast, and structural details using histogram clipping, wavelet decomposition, and spatial frequency-based fusion. It preserves edges and features while reducing errors, though it is computationally more complex and may occasionally underperform on some images.
Vidyasaraswathi and Hanumantharaju [71] proposed a novel method for enhancing medical images, Gradient Texture-Driven Dynamic Histogram Equalization (GTDDHE). It helps physicians make more accurate diagnoses by enhancing the brightness and clarity of images while preserving crucial details like textures and boundaries. The technique improves quality without sacrificing important patterns by using information about the gradients and textures of the image. Another research proposed RDH-based Dynamic Weighted Histogram Equalization [72], it has the ability to improve contrast in medical images while maintaining brightness and originality, provides secure data transmission, and facilitates the classification of brain tumors. The advantages of the proposed method include reversible data hiding, ROI-based enhancement, and the preservation of diagnostic data, while the disadvantages include the increased complexity of computation and the need for accurate ROI segmentation.
Chen et al. [73] presented a technique for enhancing brightness in CMY (cyan, magenta, yellow) electrowetting displays by combining DHE with multi-scale gamma correction. Its adaptive histogram weighting and scale-dependent gamma principles apply to color medical images that need to preserve detail across brightness ranges, even though they were created for reflective displays.
Despite its effectiveness in contrast enhancement, DHE does not strictly preserve image brightness and may amplify noise, limiting its reliability for quantitative clinical analysis [66,67,69]. Among recent advancements, dynamic and dual-domain histogram equalization techniques have gained attention for their ability to regulate enhancement strength adaptively [74]. By combining adaptive histogram segmentation and contrast-limited clipping, the method achieved improved structural preservation and visual quality across multiple application domains.
Another research proposed the technique that combines histogram equalization with the multi-level dyadic wavelet transform [75]. A dyadic wavelet is a wavelet used in signal/image processing that originates from the discretization of the scale of a continuous wavelet by powers of two. The low-frequency coefficients are enhanced using a single-threshold function, and the high-frequency coefficients are adaptively enhanced using double-threshold and directional functions. This technique is capable of improving the contrast of images, reducing noise, and offering better visual clarity for medical diagnosis than existing techniques.
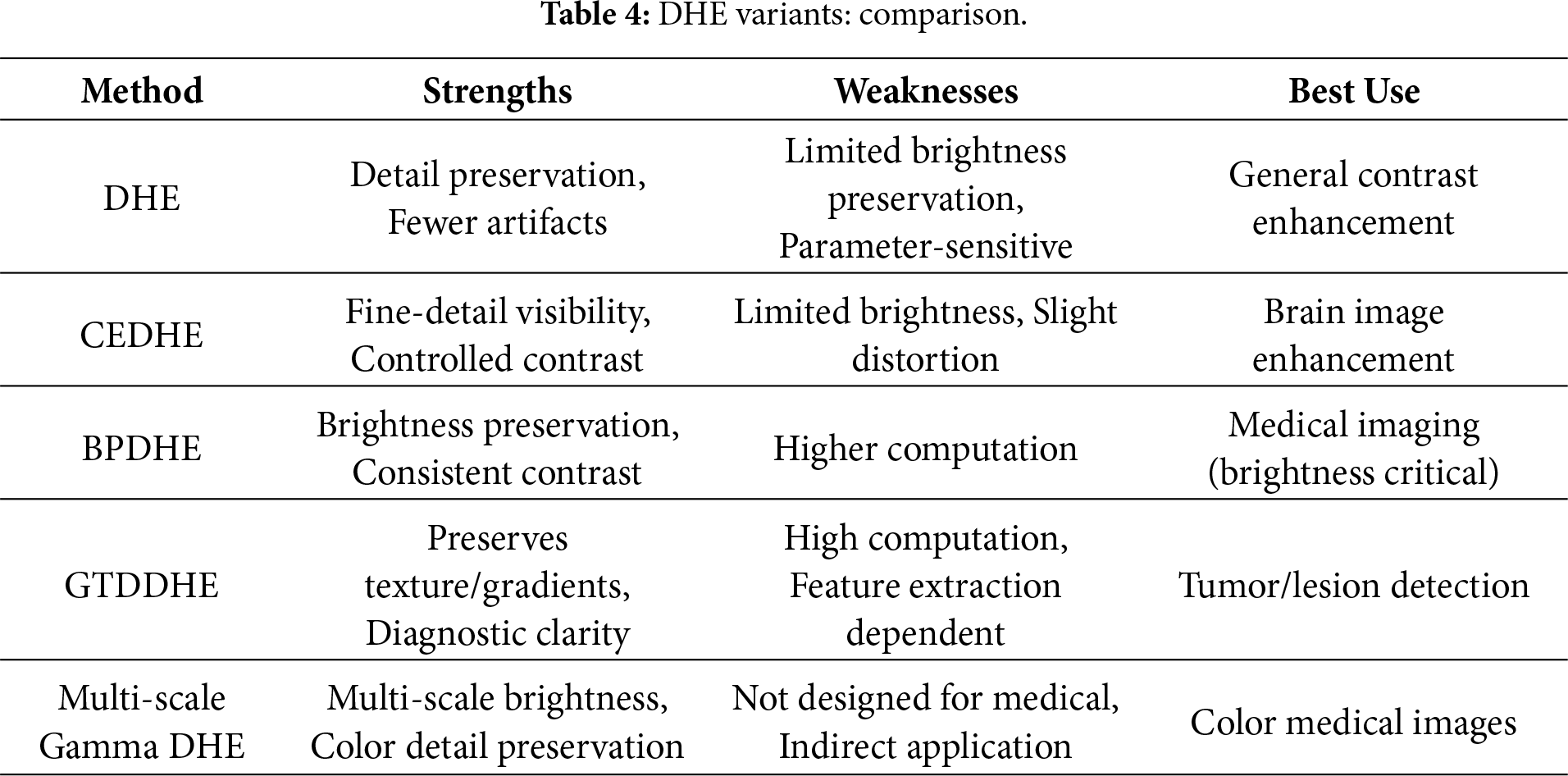
To provide a clearer comparison of Dynamic Histogram Equalization (DHE) and its major variants, Table 4 presents their respective strengths, weaknesses, and suitable use cases, including multi-scale gamma DHE, which extends DHE concepts to color images.
2.5 Adaptive Contrast Enhancement with Lesion Focusing
Adaptive Contrast Enhancement With Lesion Focusing (ACELF) [21] addresses challenges in detecting brain lesions (e.g., hemorrhagic and ischemic) due to limitations in contrast and detail resolution in MRI/CT scans. In order to improve the visibility of brain lesions in medical images, they suggested a sophisticated image processing algorithm. By first segmenting brain tissue using Otsu’s thresholding [22] and comparing pixel intensities with normal tissue values, they were able to identify lesions. By examining the image’s histogram, Otsu’s thresholding determines the threshold that reduces the overlap between the foreground (bright) and background (dark) pixels. Next, they applied adaptive contrast enhancement through custom histogram matching and gamma correction to optimize overall image contrast while preserving details. The algorithm then uses entropy scaling to adjust enhancement levels based on local complexity intelligently, preserving complex structures while boosting simpler regions where lesions may be obscured. Finally, they performed lesion-specific enhancement by modulating intensity values and applying localized HE (AHE or CLAHE) to sharpen lesion boundaries and improve diagnostic clarity.
2.6 Brightness-Preserving Histogram Equalization Variants: A Comparative Perspective
Minimum Mean Brightness Error Bi-Histogram Equalization (MMBEBHE) [76] finds the optimal threshold to split and equalize an image’s histogram, minimizing the absolute mean brightness error. This preserves the perceived brightness while enhancing contrast, which is valuable in medical imaging. Showkat and Sodhi [77] integrated MMBEBHE with a Non-Local Mean filter and BPDFHE within a framework for denoising and enhanced contrast preservation. Brightness Preserving Histogram Equalization with Maximum Entropy (BPHEME) [78] maximizes histogram entropy for contrast enhancement while strictly preserving the original mean brightness. Its global optimization can suppress fine local details and is computationally intensive. The Maximum Entropy Distribution-based Histogram Specification (MEDHS) method [79] refines this using a Gaussian-based entropy model to better preserve structural information. Brightness Preserving Dynamic Histogram Equalization (BPDHE) [80] enhances local contrast while preserving mean brightness. It reduces noise amplification and is particularly effective for subtle tissue contrasts in MRI and X-ray imaging. Brightness Preserving Dynamic Histogram Equalization (BPDHE) [81] enhances local contrast while preserving mean brightness. It reduces noise amplification and is particularly effective for subtle tissue contrasts in MRI and X-ray imaging. BPDFHE [82] utilizes fuzzy statistics to represent uncertainty in gray levels, thus providing a strong enhancement of a low contrast image, such as a medical MR image, while maintaining brightness and dynamic range. The brightening preserving bi-level fuzzy histogram equalization (BPBFHE) [83] and brightening preserving adaptive fuzzy histogram equalization (BPAFHE) [84], which emphasize the reduction of artifacts and the adaptation of fuzzy logic for improved diagnostic reliability, are the extensions. It improves image quality in medical images but can still generate minor artifacts. Building upon these fuzzy-based enhancements, the recently proposed Min–Max Peak Fuzzy Histogram Equalization (MMPFHE) [85], enhances low-contrast images while maintaining their original brightness. The method partitions the histogram using fuzzy logic, identifies maximum and minimum peaks, and applies an entropy-controlled factor to redistribute pixel intensities. By preserving key information in both bright and dark regions, MMPFHE prevents over-amplification of specific intensity ranges and improves overall image quality compared to traditional BBHE methods.
Extending brightness-preserving histogram equalization toward spatial–frequency awareness, a stationary wavelet transform based brightness preserving joint histogram equalization (SWT-BPJHE) technique [86] was proposed for contrast enhancement of brain MR images. The method enhances low-frequency sub-band coefficients using joint histogram equalization while isolating high-frequency components to preserve structural details. By explicitly enforcing brightness preservation, SWT-BPJHE avoids intensity distortion commonly observed in global HE methods. Experimental validation on BrainWeb and Howard Whole Atlas datasets demonstrated superior performance across multiple quantitative indices compared to state-of-the-art approaches. However, the reliance on wavelet decomposition introduces additional computational complexity and may limit real-time applicability. Another paper, purposed a method brightness-preserving optimized weighted bi-histogram equalization that enhances MR brain image contrast while preserving brightness to support accurate and reliable brain tissue segmentation [87]. Its main clinical applications include improved diagnosis and analysis of neurodegenerative diseases such as Alzheimer’s disease, multiple sclerosis, and brain tumors, as well as support for surgical planning and quantitative brain studies. By reducing the effects of noise and intensity inhomogeneity, the method enables better automated segmentation of white matter, gray matter, and CSF. However, a key limitation is its computational complexity due to PSO optimization and fuzzy level set segmentation. Additionally, validation is limited to the BRATS 2015 dataset, which may restrict generalizability to other MRI modalities or datasets.
Comparative Perspective: Fig. 7 shows how brightness-preserving HE methods have changed over time. Early methods, like BBHE worked on making the whole picture look better with brightness. Then BPHEME came along. Added a way to make the picture look more detailed by using something called entropy optimization. After that BPDHE was introduced, which made the picture look even better by changing the way the colors are shown. Some other methods, like BPDFHE, BPBFHE and BPAFHE used logic to make the gray areas of the picture look more natural and to reduce unwanted artifacts in the picture. Now we have methods that combine old ideas, like BBHE with new ways of making the picture look better such as metaheuristic optimization. These new methods try to balance making the picture look good keeping the brightness right and not using much computer power. Brightness-preserving HE methods are still getting better. Overall, the progression shows a shift from simple global enhancement to adaptive, fuzzy, and hybrid techniques for robust, diagnostically reliable medical image enhancement.
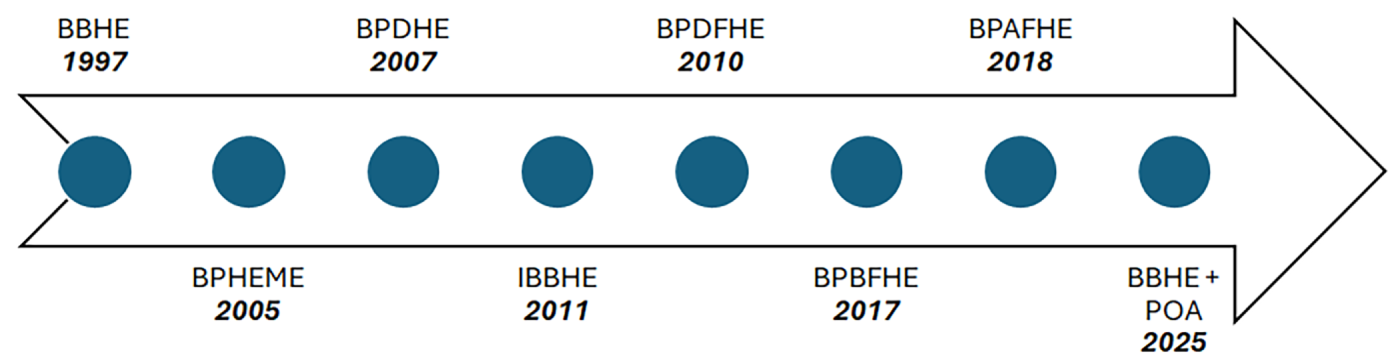
Figure 7: Evolution of brightness-preserving histogram equalization (BBHE) methods in medical imaging.
To highlight the trade-offs among brightness-preserving histogram equalization methods, Table 5 shows a comparison in terms of contrast enhancement capability, limitations, and clinical applicability.
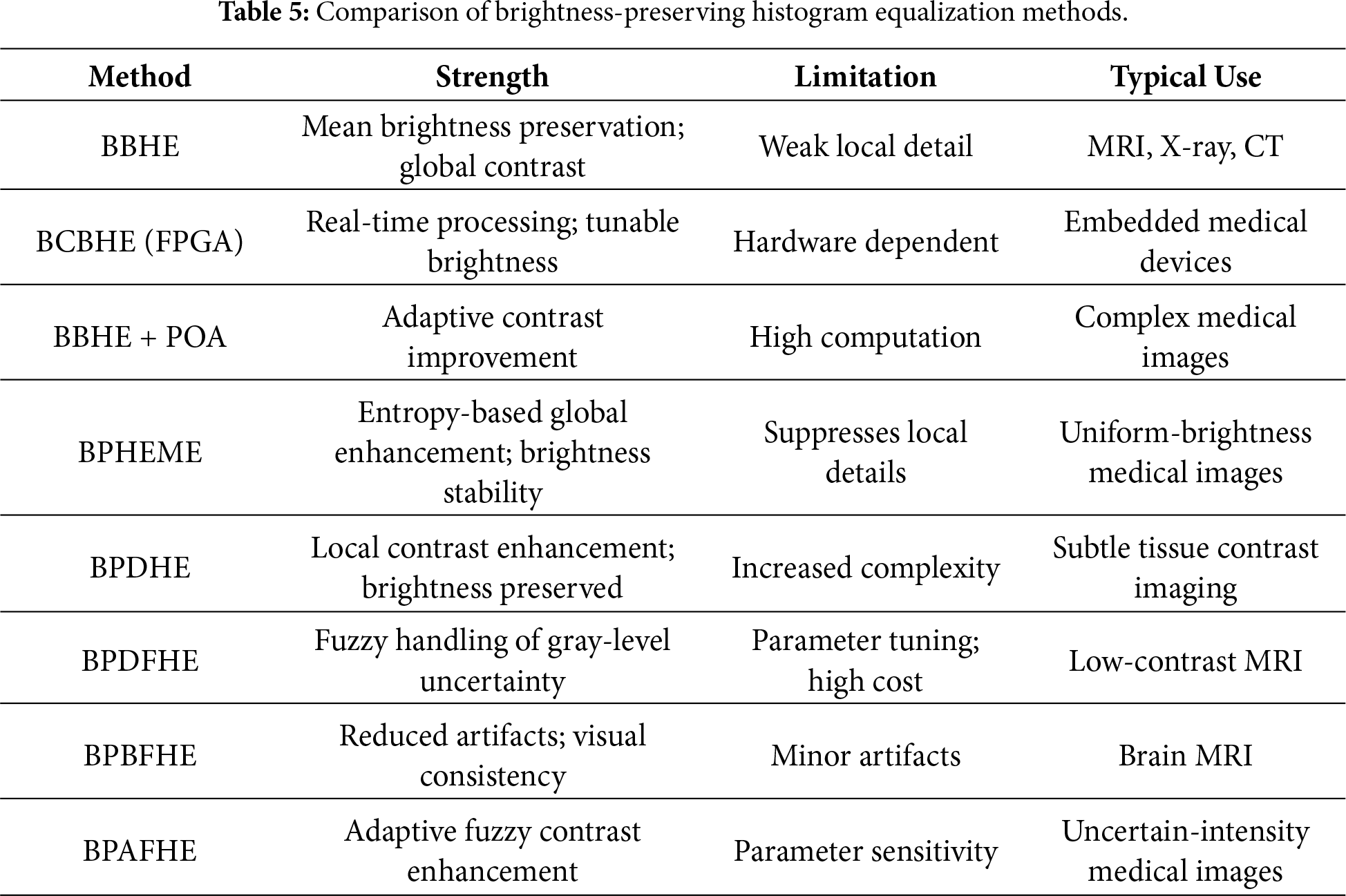
The historical progression of brightness-preserving histogram equalization methods and their adoption in medical imaging is illustrated in Fig. 7.
2.7 Recursive Approaches in Histogram Equalization
A further development of BBHE is Recursive Mean-Separate Histogram Equalization (RMSHE) [88]. RMSHE maintains brightness while increasing contrast by recursively splitting an image’s histogram into sub-histograms based on their respective means. As the recursion level “
However, selecting an appropriate recursion depth
Then Recursively Separated and Weighted Histogram Equalization (RSWHE) technique was proposed [90], it uses a weighting procedure with a normalized power law function, recursively splits the input histogram into sub-histograms, and independently equalizes the sub-histograms to improve image contrast and preserve brightness. In brief, it is an advanced algorithm for image enhancement that combines weighted adjustments and recursive histogram division to enhance contrast while preserving brightness. The more significant regions receive more enhancement thanks to the weighting in RSWHE, which raises the overall quality of the image.
RSWHE effectively maintains the original brightness of images, which is crucial for applications like aerial photography and medical imaging [91]. The results of RSWHE are found to outperform other competing techniques in terms of maintaining contrast enhancement and brightness preservation, in contrast to other comparative methods like RSIHE and RMSHE.
2.8 Fuzzy and Spline Based Dynamic Histogram Equalization
Fuzzy and spline-based dynamic histogram equalization (FSDHE) is a novel technique that enhances image contrast, particularly in the field of medical imaging. The technique combines fuzzy logic with spline interpolation to produce higher-quality images with poor contrast. The procedural steps of the FSDHE algorithm are illustrated in Fig. 8.
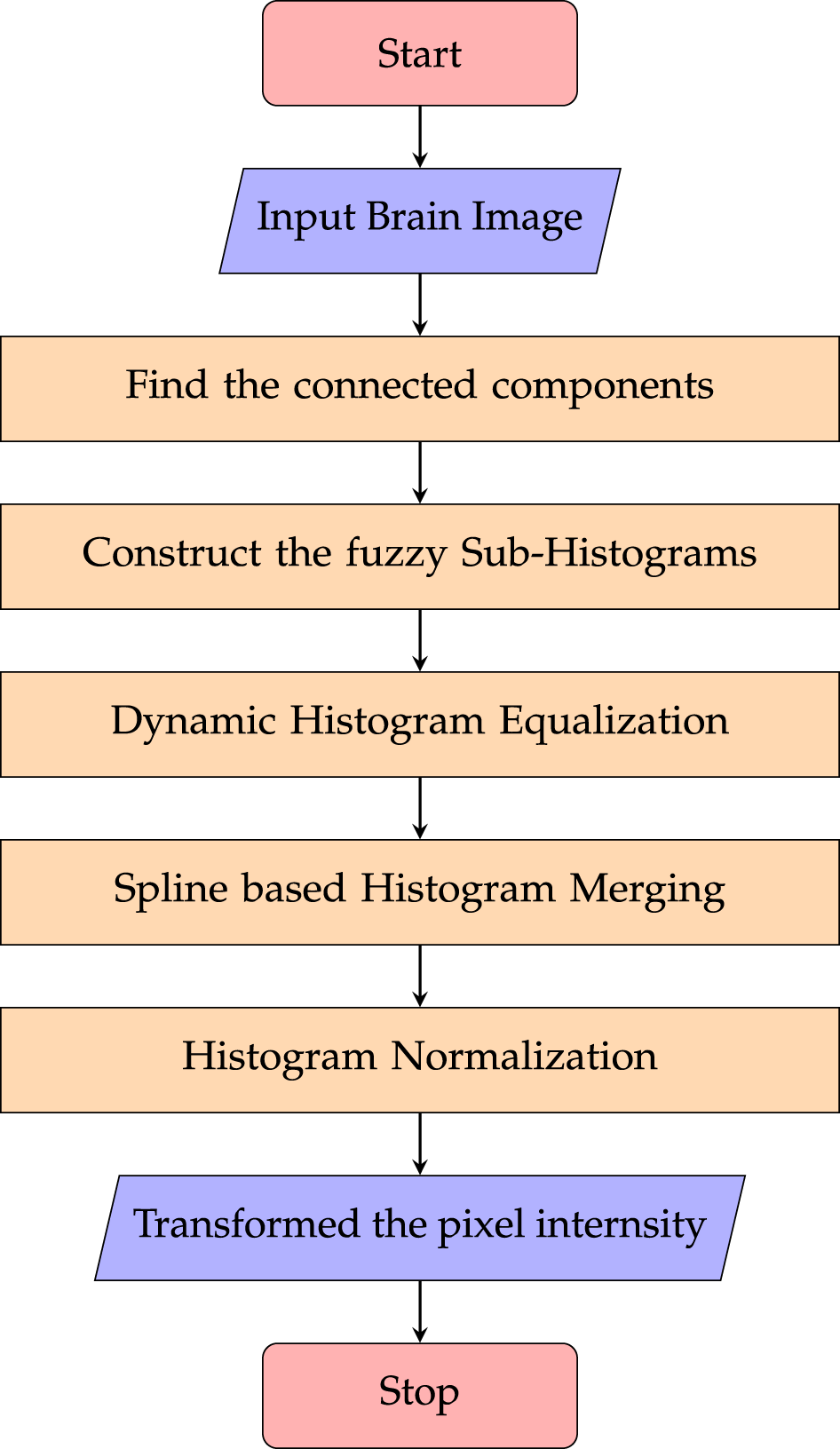
Figure 8: Flowchart of the Fuzzy Sub-Histogram Equalization process, demonstrating enhanced contrast in regions with low intensity variation while maintaining overall image brightness.
As shown in Fig. 8, the process begins with a medical image (for example, a brain scan) as input. The image is then intelligently segmented into regions by identifying connected components using fuzzy logic. After that, fuzzy sub-histograms are produced for every component that has been identified. Every sub-histogram is subjected to AHE separately. The improved sub-histograms are then seamlessly combined using spline interpolation. The final output is then created by normalizing and transforming pixel intensities. An intuitionistic fuzzy logic-based image fusion method [85] has been proposed to enhance medical image fusion. It effectively improves contrast, suppresses noise, and preserves color and details, yielding clearer fused images. Quantitative evaluations show superior edge preservation, detail retention, and overall similarity compared to previous techniques. To implement specific enhancements based on the characteristics of each segment, fuzzy logic is used to extract connected components of an image [92]. When compared to conventional methods, the FSDHE approach has demonstrated superior performance in several parameters, including Absolute Mean Brightness Error (AMBE) and Peak Signal-to-Noise Ratio (PSNR) [93]. The smooth intensity level transitions provided by spline interpolation in FSDHE reduce artifacts and improve the overall visual quality of the enhanced images. In order to improve contrast in brain images, FSDHE separates medical images into linked components, performs dynamic histogram equalization on each component independently, and then uses spline interpolation to smooth the composite histogram. Fuzzy Contextual Dissimilarity Adaptive Histogram Equalization (FCDAHE) [94] enhances medical images by using a fuzzy inference system to compute pixel-wise dissimilarity and clip limits, followed by Contextual Intensity Transformation. Tested on MR images, it improves contrast, preserves fine details, and outperforms traditional methods in PSNR, entropy, contrast ratio, and EME, making it useful for disease diagnosis.
A bi-histogram equalization technique using fuzzy logic was developed to overcome the drawbacks of traditional histogram partitioning methods and mean/median-based histogram partitioning techniques [95]. This technique uses asymmetrical triangular fuzzy membership functions, which are defined based on the mean and standard deviation of the image, to find an optimal threshold value using a fuzzy level-snip technique. The technique has the ability to adaptively partition the histogram and separately equalize the sub-histograms. Another paper proposed Fuzzy Gray Level Difference Histogram Equalization (FGLDHE) [96], an adaptive contrast enhancement method that uses fuzzy logic to deal with uncertainties and noise in medical images. FGLDHE uses the concept of fuzzy gray level difference and a fuzzy clip limit before histogram equalization to enhance fine details in tissues without over-enhancement. FGLDHE provides better visual clarity and noise reduction in MR images than histogram-based methods. Another paper proposed FICDHE, a fuzzy logic-based method for enhancing medical images by improving contrast and detail without artifacts [97]. It adapts to pixel uncertainty and local context, yielding clearer, more natural images, which is beneficial for medical image clarity and diagnostic accuracy. A limitation of this approach is that it relies on multiple parameters and fuzzy inference processes that may increase computational complexity, potentially affecting real-time application performance.
There are also hybrid variations, for example, a hybrid fusion of HE and CLAHE. This approach aims to leverage the strengths of both methods, enhancing overall image quality while preserving local features crucial for accurate brain tumor segmentation [98].
2.9.1 World Cup Organization (WCO) Algorithms for Contrast Enhancement
Metaheuristic and population-based optimization algorithms have been widely explored for medical image contrast enhancement, owing to their ability to optimize complex and non-linear objective functions. These approaches typically aim to enhance visual quality by maximizing contrast-related criteria such as entropy, edge information, and intensity distribution, while preserving diagnostically relevant structures.
Fig. 9 illustrates a generalized workflow of a World Cup Organization (WCO)-based optimization framework for contrast enhancement. In such approaches, candidate solutions represent enhancement parameters and are iteratively evaluated using a predefined objective function. Competitive mechanisms inspired by tournament structures are employed to rank and refine solutions, allowing the algorithm to converge toward optimal or near-optimal contrast configurations.
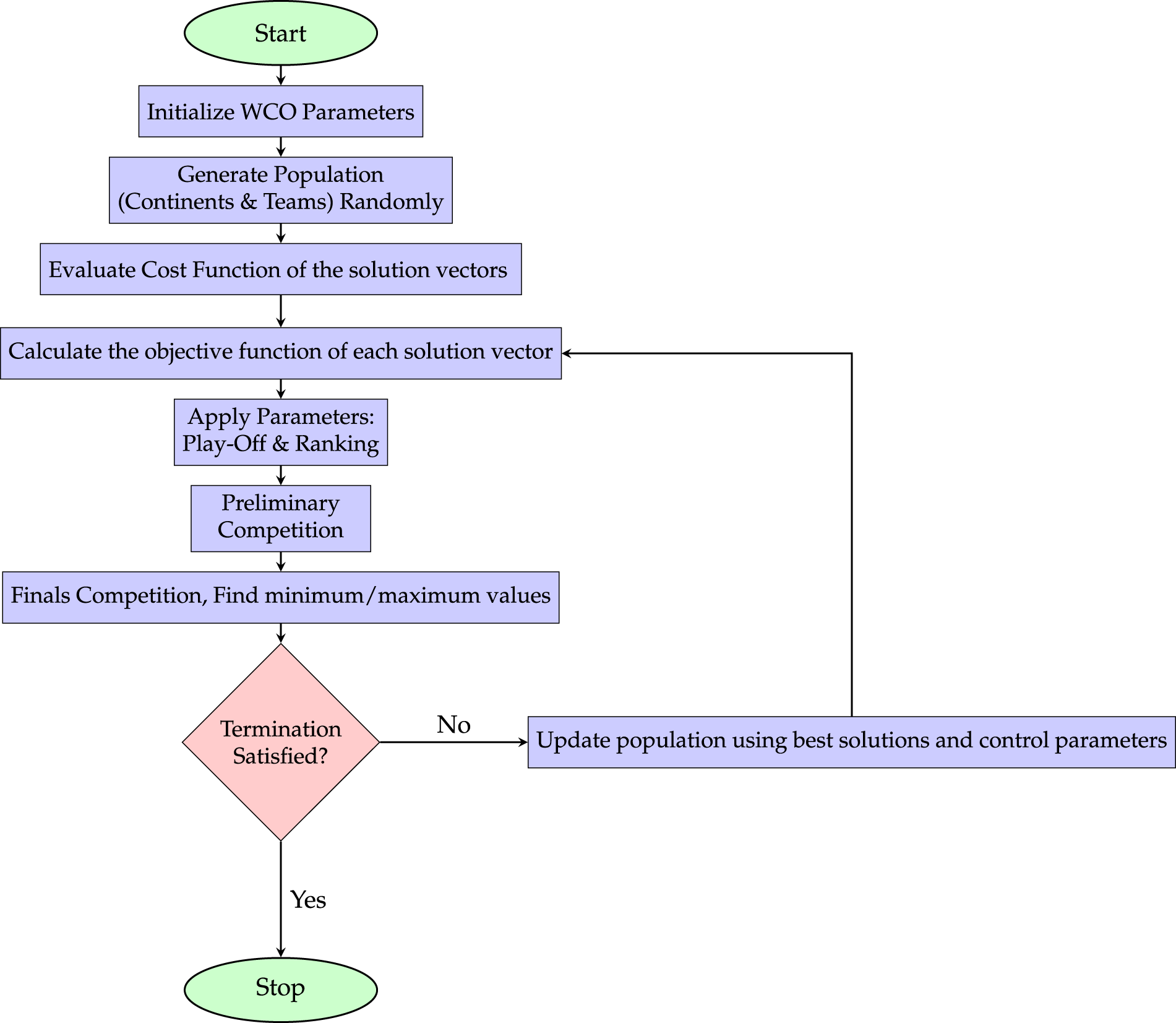
Figure 9: General workflow of a WCO-based optimization framework for medical image contrast enhancement, illustrating the iterative process of population initialization, evaluation, parameter tuning, and competition stages to achieve optimal contrast enhancement solutions.
Wang et al. [99] proposed a chaos-based optimization algorithm for medical image contrast enhancement in order to get around the limitations of conventional methods, like HE and Gamma correction, which frequently lead to artifacts or information loss. Their hybrid approach combined HE, Gamma correction, and edge preservation into a weighted cost function that was optimized using the Chaos World Cup Optimization (CWCO) algorithm. The method demonstrated exceptional performance in metrics such as contrast, entropy, and edge preservation across various medical datasets, outperforming CLAHE, BBHE, and BPDFHE. The algorithm’s versatility and efficacy in enhancing diagnostic clarity in low-contrast medical images were emphasized in the study.
2.9.2 Genetic Algorithm Based Image Enhancement
A genetic algorithm (GA), an evolutionary algorithm that draws inspiration from natural selection and the biological processes of the fittest individual’s reproduction. Currently used in many real-world applications, GA is one of the most widely used optimization algorithms. Image enhancement is one of its many practical uses.
For adaptive image enhancement, Mu et al. [100] suggested a hybrid genetic algorithm (HGA) in conjunction with differential evolution, employing a normalized incomplete Beta function to dynamically modify transformation parameters. Although their approach showed better Contrast Increase Index (CII) and Edge Protection Index (EPI) in general images, its parameter optimization principles may encourage similar modifications in medical imaging, where histogram-based methods are more common. This method demonstrates how evolutionary algorithms can be used in conjunction with conventional contrast enhancement frameworks.
Integrating GA-based optimization, as in [101], with adaptive histogram matching could enhance cross-modal alignment (e.g., positron emission tomography-MRI) by dynamically tuning intensity mappings while preserving diagnostic features. A genetic algorithm-based technique to improve embryonic images in IVF is presented in this paper [102]. It outperforms current methods by optimizing a composite filter from a filter bank to enhance image clarity and detail. The technique is easy to use, efficient, and enhances embryo evaluation; it may be further optimized.
MedGA [103] is a genetic-algorithm-based image enhancement method tailored for medical images with bimodal histograms, improving contrast in key sub-regions while preserving mean brightness. Its strengths include superior signal and perceived image quality compared to conventional methods and suitability for clinical decision support. Limitations include higher computational cost and the need for GPU acceleration for large images.
Acharya and Kumar [104] proposed Genetic Algorithm-based Adaptive Histogram Equalization (GAAHE). It uses an organized workflow to process medical images. After calculating and clipping the input image’s histogram, the method determines an exposure threshold to split the histogram. A genetic algorithm that maximizes a fitness function determines the ideal thresholds for further partitioning each sub-histogram. The cumulative distribution function (CDF) is calculated after the probability density function (PDF) for each partition has been calculated and adjusted to improve contrast. The improved output image is then created by combining the processed sub-histograms after mapping functions have been applied.
A Genetic Algorithm-Based Histogram Equalization (GAHE) method was presented by [105] for the purpose of enhancing and diagnosing breast cancer in mammography images. They used a combination of genetic algorithms and histogram equalization to improve the contrast of low-quality mammograms. In order to provide improved image quality, maintain brightness, and improve feature visibility, the proposed method uses genetic algorithms to solve multiple constraints and improve contrast enhancement. In order to make it easier to detect masses, microcalcifications, and other cancerous areas in low-quality mammograms, GAHE is used to improve contrast and visibility.
2.9.3 Hybrid Histogram Equalization and Fuzzy Logic–Based Image Processing Frameworks
Padmavathy et al. [106] introduced a novel approach to enhancing low-contrast images by combining HE, fuzzy logic, and wavelet transform techniques. Their main objective is to preserve image quality while resolving the issues with conventional histogram equalization techniques, like the creation of artifacts and subpar edge preservation. Their method, which incorporates fuzzy logic, enables more precise contrast adjustment, preserving fine details while enhancing brightness globally.
Decomposing images into spectral components for additional resolution and detail enhancement is also made possible by the inclusion of wavelet transforms. The framework is incredibly user-friendly and adaptable to a wide range of applications, such as remote sensing and medical imaging, thanks to its automatic filtering and denoising techniques. The current study provides a robust solution for long-term image enhancement and outlines future research directions, including examining novel fuzzy membership functions and conducting comparative analyses with alternative methods.
Several hybrid frameworks combine traditional contrast enhancement techniques with intelligent segmentation methods to improve diagnostic accuracy in medical imaging [107]. An automated framework for brain tumor detection integrates histogram equalization for contrast enhancement with morphological preprocessing steps, including bias field correction, anisotropic diffusion filtering, and unsharp masking, to improve MRI image quality. Fuzzy C-Means (FCM) clustering is then employed for tumor segmentation, followed by classification into benign and malignant categories and tumor area estimation. This approach addresses challenges such as noise, low contrast, and manual interpretation errors in MRI scans. However, the reliance on FCM introduces sensitivity to initialization and difficulty in separating regions with similar intensity, and the framework is limited by older datasets and lack of real-time capability.
Building upon fuzzy-based HE methods like BPDFHE and BPBFHE, a recent study proposed a multi-histogram equalization framework using adaptive fuzzy clustering and optimized clipping [108]. The method segments image brightness via fuzzy C-means clustering, applies optimized clipping to suppress over-enhancement, and adjusts the dynamic range of each sub-image prior to independent equalization. Evaluations showed improved contrast, detail preservation, and performance across diverse medical imaging datasets, demonstrating the advantages of combining fuzzy logic with adaptive multi-histogram processing. The Dualistic Fuzzy Histogram Equalization (DFHE) method [109] combines fuzzy image histogram computation with dualistic partitioning to enhance low-contrast images while preserving mean brightness and entropy, achieving superior performance (PSNR: 20.8175, CI: 72.5657) compared to traditional methods. The technique minimizes common artifacts such as over-brightening and darkening effects, making it suitable for medical imaging, satellite imaging, and real-time systems. The fuzzy bi-histogram equalization (FBHE) [110] method enhances low-contrast images through its implementation of a fuzzy image histogram and adaptive histogram partitioning to preserve mean brightness while reducing artifacts. Experimental results on the CSIQ dataset demonstrate that FBHE outperforms conventional and fuzzy contrast enhancement methods in terms of visual quality, PSNR, MSSIM, entropy, and brightness preservation. Recently, hybrid enhancement schemes have been proposed by combining histogram equalization with low-rank and sparse decomposition models [111]. One of the representative schemes combines robust principal component analysis with Log-Schatten norm regularization and adaptive histogram equalization. The Log-Schatten norm improves the penalization of singular values and structure preservation, and adaptive histogram equalization further improves the local contrast. The scheme is formulated as an optimization problem and solved using the ADMM framework, and it shows promising results on retinal fundus and cancer image datasets.
Another study shows that a hybrid image compression scheme for medical images, combining Lempel-Ziv-Welch and clipped histogram equalization for the ROI part, with EZW for the non-ROI part [112], enhances the compression ratio, PSNR, and MSE. The thresholds are optimized by particle swarm and firefly algorithms, resolving the problems of storage and transmission, although the computational complexity is increased due to the hybrid processing.
Another hybrid approach is an adaptive compression algorithm for medical images that combines edge enhancement contrast-limited adaptive histogram equalization (EE-CLAHE) enhancement [113], clustering-based ROI (region of interest) segmentation, and a mix of lossy and lossless techniques, achieving high compression ratios while preserving diagnostic quality in MRI, CT, and X-ray images.
2.9.4 Metaheuristic and Evolutionary Optimization for Contrast Enhancement
Particle swarm optimization (PSO) is used in the Particle Swarm Optimized Texture-Based Histogram Equalization (PSOTHE) method [114] to improve image quality. To eliminate noise and artifacts, PSOTHE focuses on enhancing the texture areas of the image while rejecting non-texture areas. The framework is adaptive and requires less human intervention because the PSO algorithm itself determines the threshold for texture region detection and the parameters for modifying the probability density function (PDF) automatically [114].
PSO and HE were combined in another paper, to improve medical image segmentation [115]. This technique is crucial for improving image quality and precisely identifying regions of interest in medical images, particularly chest X-rays and lung CT scans. Therefore, the goal of the proposed study is to demonstrate empirically the applicability of the combined approach.
Optimized Bi-Histogram Equalization (OBHE) [116], which leverages PSO to preserve brightness fidelity, a key requirement for medical images where intensity values correlate with tissue properties (e.g., T1/T2-weighted MRI). Unlike conventional HE, OBHE avoids over-enhancement and washout effects by:
1. Splitting the histogram at the mean intensity to separately process dark/bright regions (e.g., CSF vs. white matter).
2. PSO-optimized weighting to balance contrast improvement and brightness retention.
As shown in Fig. 10, the PSO-HE segmentation approach first preprocesses the input medical image through grayscale conversion and histogram equalization to enhance contrast. The enhanced image is then segmented using Particle Swarm Optimization, where particle positions are iteratively updated based on predefined cost functions. The dashed box details the key PSO optimization steps.
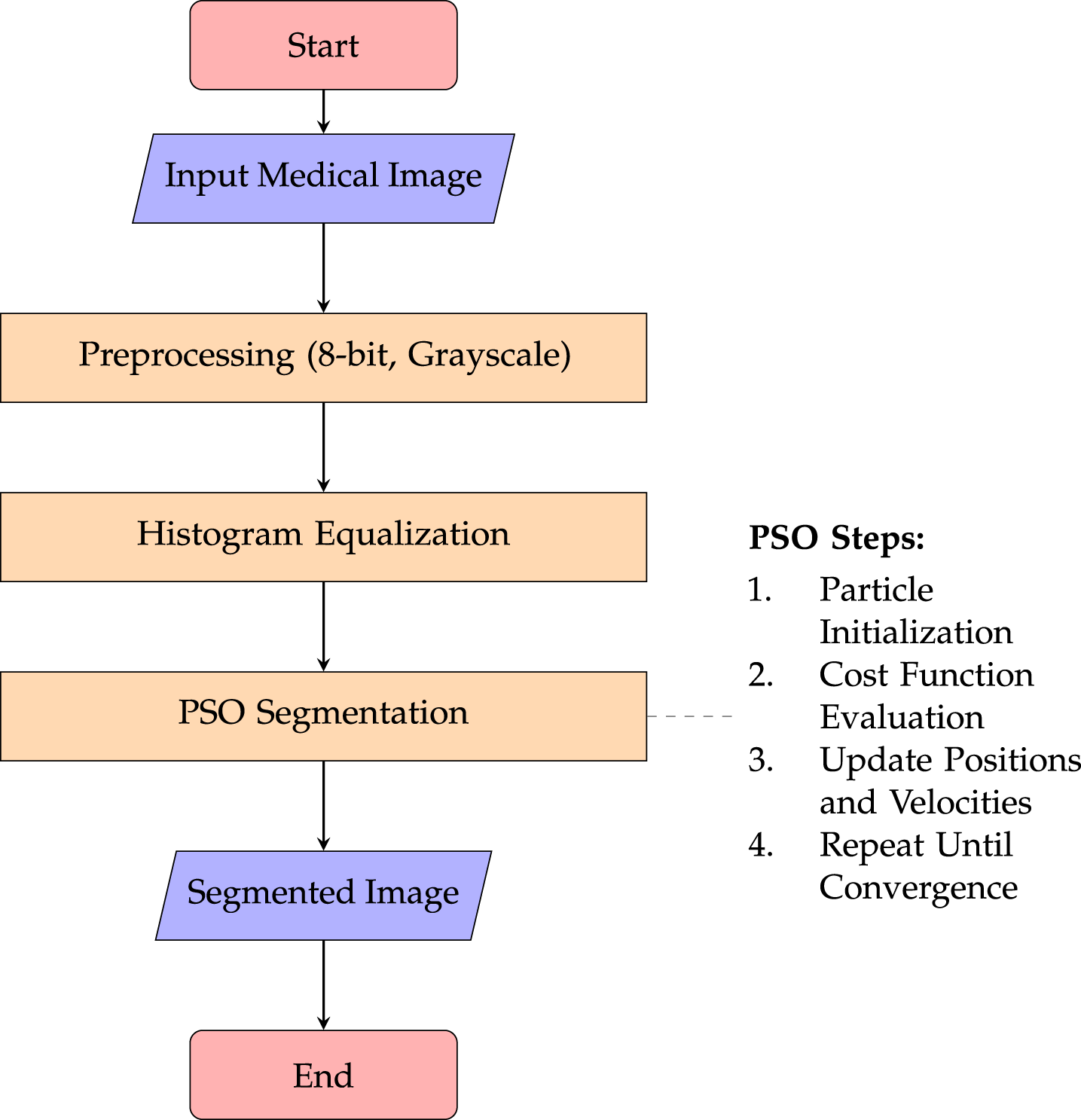
Figure 10: Workflow of the PSO-HE medical image segmentation approach.
To enhance the contrast of cardiac MRI medical images, there’s a novel hybrid optimization technique known as the Cetacean Optimization Algorithm and Sand Cat Swarm Optimization (COA-SCSO) [117]. The technique applies the novel COA-SCSO optimization after processing input MRI images through histogram stretching and fitness function evaluation, as shown in Fig. 11.
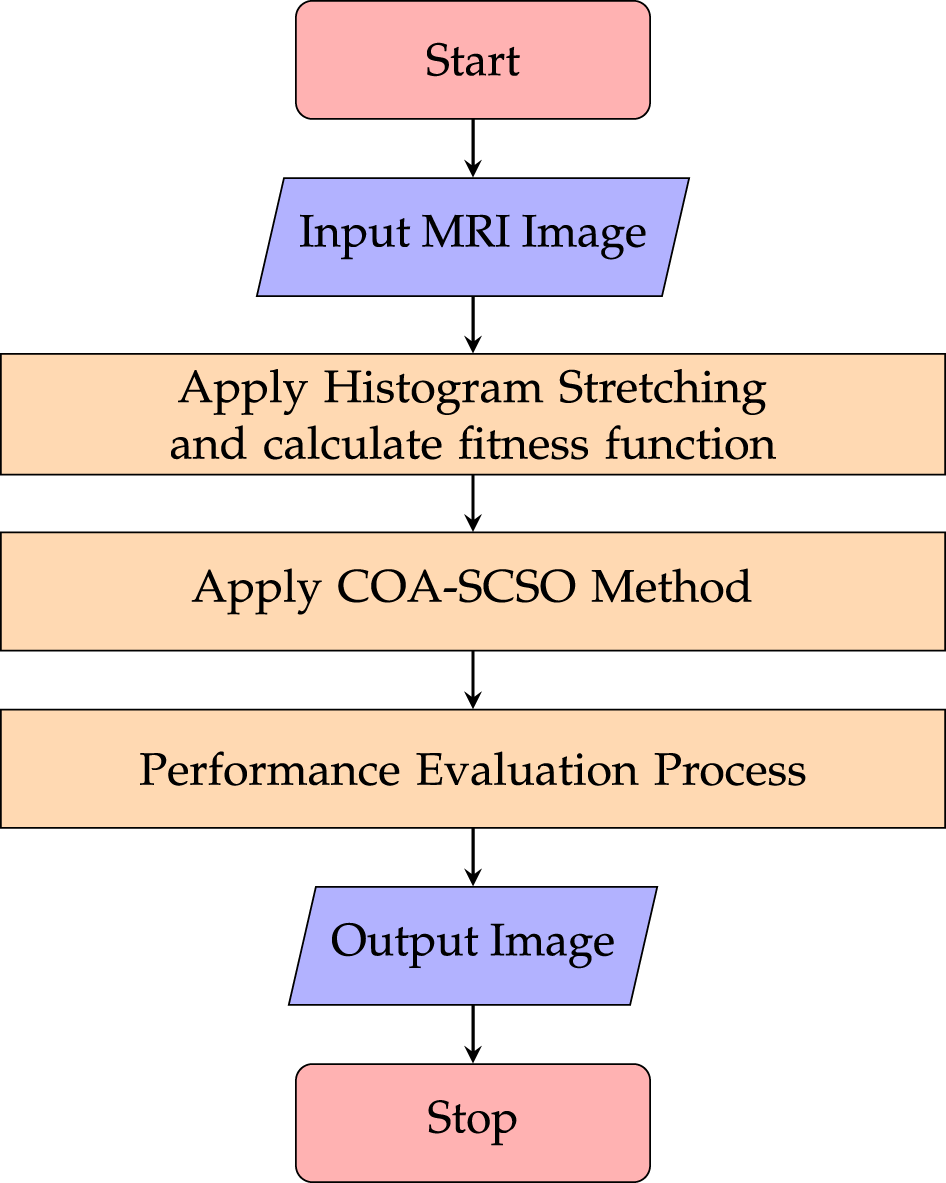
Figure 11: Flowchart of the cardiac MRI enhancement process, illustrating the sequential application of histogram stretching, COA-SCSO optimization, and performance evaluation to produce an enhanced output image with improved contrast and diagnostic quality.
This study successfully addresses the drawbacks of current contrast enhancement techniques by combining two algorithms to reduce noise and improve image quality. The study confirms that a greater range of medical imaging applications could benefit from the application of the COA-SCSO technique. It emphasizes how important it is to enhance image contrast in order to diagnose diseases and develop treatment plans. The Sand Cat Swarm Optimization (SCSO) method presents a robust optimization model that enhances the COA-SCSO approach in generating high-quality images. COA-SCSO is one of the more effective hybrid optimization techniques made possible by advancements in the SCSO algorithm.
Samraj and Karuppusamy [118] proposed a metaheuristic-based histogram equalization framework for mammogram enhancement by integrating the Cuckoo Search Algorithm (CSA) with conventional HE. The method optimizes HE parameters using CSA’s global search capability to enhance contrast while preserving brightness and reducing over-enhancement artifacts. The primary objective is to address low-contrast mammography images that hinder accurate breast cancer detection, with validation conducted on the DDSM dataset. Although the approach demonstrates improved visual quality and quantitative performance, it may incur increased computational cost due to iterative optimization and shows potential sensitivity to CSA parameter tuning and dataset dependency. Clinically, the enhanced mammograms support early breast cancer screening by improving lesion visibility, assisting radiologists in diagnosis and treatment planning. Pashaei and Pashaei [119] introduced a hybrid method for improving medical images called GQAOA-CLAHE, which combines a Gaussian Quantum Arithmetic Optimization Algorithm (GQAOA) with CLAHE. GQAOA to automate the tuning of CLAHE’s parameters and CLAHE for localized contrast enhancement. The suggested GQAOA uses quantum-inspired arithmetic operations enhanced with Gaussian mutation to automatically optimize CLAHE’s critical parameters (clip limit and tile size) in order to avoid suboptimal solutions. Fig. 12 illustrates the overall workflow of the proposed GQAOA-CLAHE framework for medical image enhancement, showing the integration of an optimization algorithm with CLAHE for adaptive contrast improvement. Vijayalakshmi et al. [120] developed a smart image enhancement solution that effectively improves low-contrast X-rays and MRIs. The developed system improves diagnostic visibility through multiple medical imaging tests, which include COVID-19 chest X-rays and MRI scans, but it suffers from three major limitations, including higher computational cost, parameter dependence, and challenges in real-time processing.
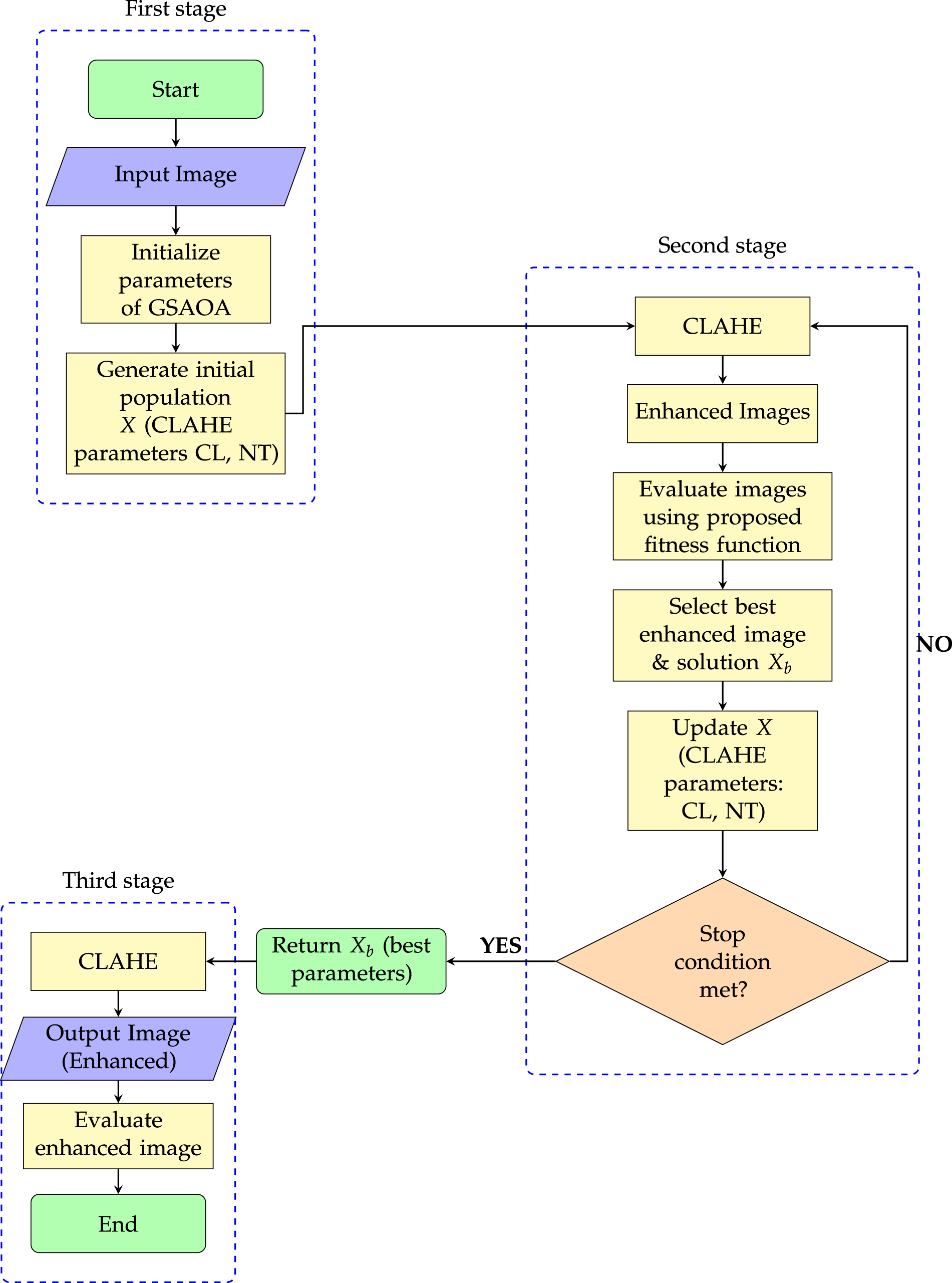
Figure 12: GQAOA-CLAHE schema for medical image enhancement.
A hybrid method for medical image enhancement (mainly X-ray images) by optimizing CLAHE (Contrast Limited Adaptive Histogram Equalization) parameters using a Modified Camel Algorithm (MCA), an improved version of the Camel Optimization Algorithm [121]. It is used for preprocessing low-quality X-ray images to improve contrast for better visualization of bones, lungs, chest, etc, aiding radiologists in detecting abnormalities. The paper compares it with other techniques and shows that MCA performs better for local region enhancement.
An approach to automatically enhance image contrast in X-ray images, including a combination of noise reduction, high-pass filtering, and CLAHE with parameter optimization, was presented [122]. The process was found to be significantly better than CLAHE and window level adjustments in clinical evaluations in enhancing tissue visibility, facilitating proper patient setup in radiation therapy, and so forth.
2.9.5 Gaussian Probability Model + Bi-Histogram Equalization
Gaussian Probability Bi-Histogram Equalization (GPBHEPL-D) [123] analyzed brain MRI, lung CT, and X-ray images. This method was combined with the Gaussian Probability Model, which concentrates on particular pathological areas. Images are split into two segments by Bi-HE for separate enhancement. Plateau limiting prevents over-enhancement and saturation. To achieve the best segmentation, Thresholding Median utilizes median values. Instead of uniformly enhancing entire images, the technique preserves important image details, detects pathological regions of interest (ROI), uses Gaussian probability to suppress background tissue, and precisely enhances contrast in pathological areas.
2.9.6 Deep Learning–Based Hybrid Enhancement
Recent development in image enhancement [124] proposes deep learning algorithms for predicting optimal imaging parameters to enhance the vitreoretinal surface during 3D heads-up vitreoretinal surgery. The hybrid model comprises two steps: a GAN-based architecture, Pix2Pix, for generating visually optimized fake images, followed by a CNN-based architecture-the ResNet-which provides predictions of eight control parameters in the enhancement model. The primary objective is the optimization of images with improved visibility and surgical accuracy, leading to safe and effective surgeries. Limitations include that the algorithm cannot presently be implemented directly in live surgeries due to technical and licensing problems. Presently, this technology works in highly controlled experiments and with high-quality images, but not in seamless, real-time clinical settings. Despite these challenges, this technique holds a promising future for optimizing surgical outcomes and may become a useful tool in future ophthalmic surgery.
ECFusion [125], it is a novel technique aimed at the fusion of multimodal medical images, including computed tomography, magnetic resonance imaging, and single photon emission computed tomography, among others. Its key application is the enhancement of major anatomical contours and functional information. This, in turn, enhances the physician’s view of tissues and their related abnormalities in a better and more detailed manner. However, this technique still has a number of drawbacks, like most image processing techniques. Although the results are promising, there is still a need for further work. For example, testing has been done mostly on selected datasets; therefore, there needs to be stronger validation with regard to diverse clinical scenarios. Also, in its current form, this approach has based its analysis on edge detection through Sobel operators, which can hardly function optimally with noisy or low-contrast images. Further optimization could be necessary for real-time clinical use.
Patient-informed deep learning frameworks have also been applied to optimize image acquisition parameters. For instance, a neural network-based clinical tool was developed to predict optimal contrast injection and scanning parameters for liver CT imaging [126]. By incorporating patient attributes such as height, weight, sex, and age, the model suggested adjustments that improved contrast consistency and reduced underenhanced scans. Although this approach does not directly modify image pixels, it represents a hybrid strategy combining clinical information and deep learning to enhance medical image quality in practice.
Retinexformer [127] is a low-light image enhancement technique based on the Transformer architecture, and it is an extension of the Retinex theory that considers the hidden corruptions in underexposed images. The advantages of this technique include modeling long-range dependencies, removing noise and color distortion, and achieving better performance than state-of-the-art approaches on various benchmarks. The disadvantages include the need for large datasets for training and the possibility of being computationally expensive due to the Transformer structure.
A CNN-based medical image segmentation framework was enhanced using histogram equalization (HE) as a preprocessing step to improve contrast and feature visibility [128]. Evaluations on Lung CT-Scan and Chest X-ray datasets showed that incorporating HE increased CNN segmentation accuracy in both training and validation phases, with improvements in dice similarity coefficient (DSC) and structural similarity index (SSIM) values approaching 0.95. The study demonstrates that simple contrast enhancement via HE can significantly improve deep learning-based segmentation outcomes. Limitations include dependence on dataset characteristics and potential sensitivity to HE parameters, while clinical applications encompass improved automated analysis of thoracic imaging for disease detection.
A recent advancement is UniMIE [129], a training-free diffusion model that carries out unsupervised medical image enhancement across different modalities without fine-tuning. Using the pre-trained ImageNet model, UniMIE improves contrast, brightness, and structural detail in different medical images, including microscopy images, X-rays, brain, and cardiac scans. Performance evaluations demonstrate its superior performance relative to modality-specific deep learning-based approaches. Another paper proposed Hybrid CLAHE + CNN + InceptionV3 [53]; it improves liver tumor detection with high accuracy and enhanced contrast but lacks automatic segmentation and is computationally intensive. Another research work suggests a framework that initially removes speckle noise from ultrasound images using a CNN and then enhances key features using CLAHE and multifractal analysis [130]. This helps in improving contrast, texture, and details, which in turn facilitates accurate medical diagnosis. A recent research work explored the effect of using Contrast Limited Adaptive Histogram Equalization (CLAHE) as a preprocessing technique on mammography images for the detection of breast cancer using the ResNet-18 deep learning model [131]. CLAHE enhanced the accuracy and decreased the computational processing time from 243 to 109 min. The authors of the research work also proposed the potential use of CLAHE and deep learning models for other imaging modalities like MRI, CT, and ultrasound scans. Another study illustrates that CLAHE preprocessing has a substantial impact on medical image classification by improving contrast and preserving edges [132], which enables the VGG-16 model to extract more discriminative features. When combined with data augmentation, it further enhances the accuracy and robustness of brain MRI image classification. Similarly, CLAHE has been applied to post-COVID pneumonia X-ray images to improve local contrast and accelerate deep learning-based detection using VGG16 [133]. Although CLAHE and histogram equalization had minimal effect on accuracy (91–92%), they nearly halved the prediction time, demonstrating their utility in efficient medical image analysis. The CLAHE preprocessing technique has been used for the classification of chest X-ray images intended for tuberculosis detection [134], where the classification model using the DenseNet201 technique demonstrates high accuracy (94.84%) in the classification of medical images, proving that CLAHE preprocessing makes an impact in terms of medical image classification using deep learning techniques.
3 Performance Evaluation of Techniques
Evaluating histogram-based contrast enhancement in medical imaging is challenging due to the wide range of image types, diverse clinical requirements, and the need for accurate diagnosis. The classical HE is low in computational cost but introduces an excessive alteration in brightness along with artifacts. AHE increases the local contrast at the same time, amplifying the noise. Brightness-preserving techniques, such as BBHE, DSIHE, or particularly MMBEBHE, preserve more effectively the luminance, with MMBEBHE displaying the highest fidelity. In summary, among all methods, CLAHE best balances noise reduction with local adaptation and remains the most reliable and clinically widely used method, especially in mammography. CLAHE has been widely used in medical image processing.
Techniques such as DHE and BPDHE have the advantage of maintaining brightness and improving the natural view, which is significant when considering MR and multimodal imaging. Techniques like BPDFHE and FSDHE, which are advanced versions of fuzzy logic, have the advantage of producing smooth and artifact-free images, but are computationally intensive. Techniques like PSO, GA, and the advanced metaheuristics have the advantage of giving the best results, but are computationally intensive, which makes them more suitable for the research environment rather than the routine environment.
PSO-based methods, such as PSOTHE [114] and OBHE [116], enhance medical images by optimizing histogram equalization for texture preservation and brightness fidelity, improving segmentation and contrast in MRI and CT scans. Hybrid optimization techniques, including COA-SCSO [117], CSA-based HE [118], and GQAOA-CLAHE [119], combine metaheuristic algorithms with HE to achieve higher-quality enhancement and automated parameter tuning. While these methods excel in research studies, their high computational cost and iterative processing limit routine clinical adoption.
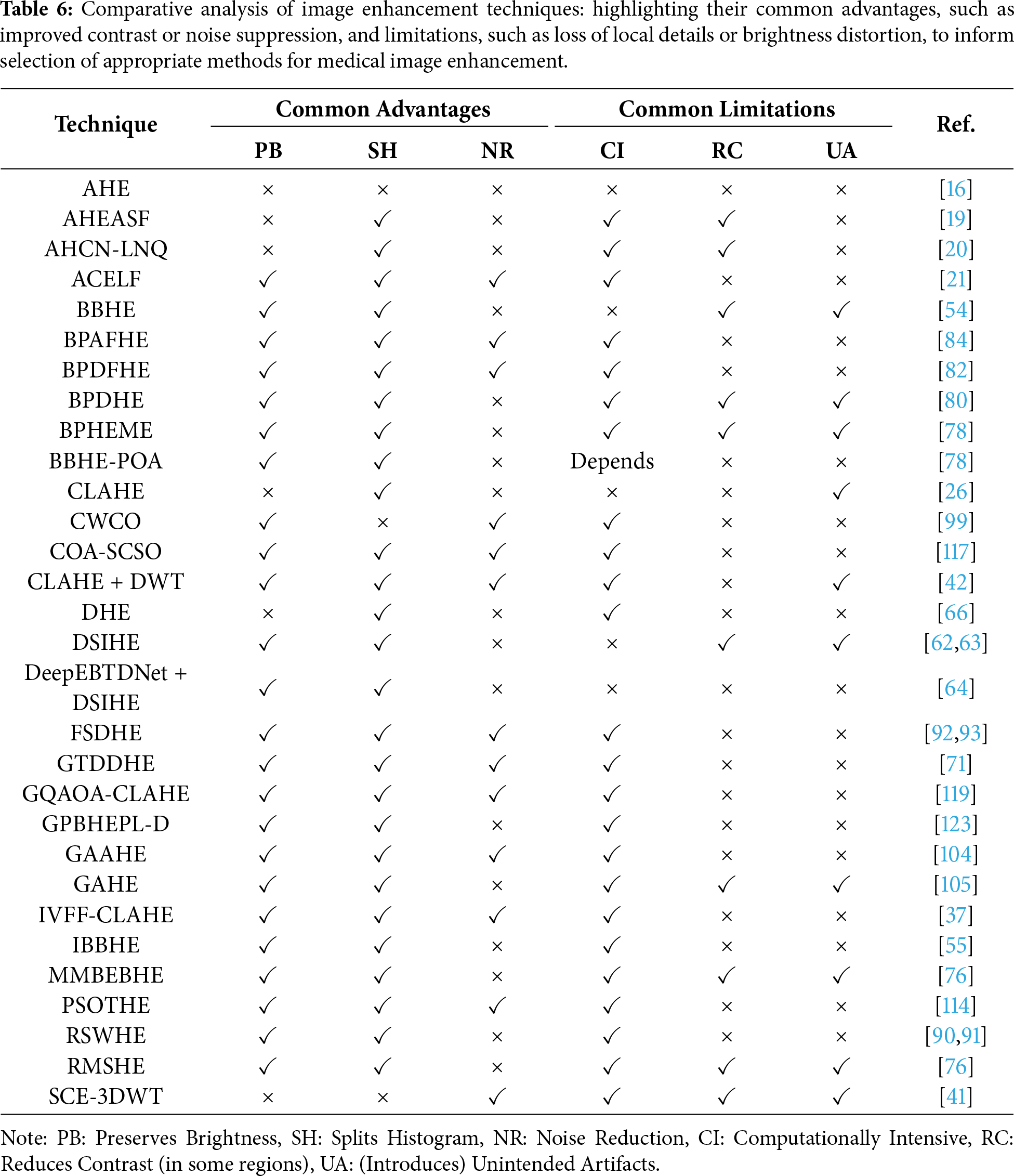
Vijayalakshmi et al. [120] proposed a smart enhancement approach for low-contrast X-rays and MRIs that preserves structural details and improves diagnostic visibility, but its computational demands and parameter sensitivity highlight the trade-off between research-level performance and clinical practicality. This is further supported by application-specific analysis, CLAHE better highlights the lesions in MRI and mammography, whereas tissue contrast is better preserved by BPDHE. The nodules in chest radiography are better enhanced by BBHE and DSIHE. In CT imaging, brightness-preserving algorithms ensure the quantitative correctness of the enhancement. Advanced optimization-based methods have the potential for higher image quality but remain largely theoretical due to the current lack of computational efficiency. From this, one important trade-off can be highlighted: on the one hand, methods proven useful clinically—in practice, CLAHE and MMBEBHE—are preferred, while more advanced optimization methods remain in research. In order to help select the appropriate enhancement technique for given clinical needs, Table 6 compares the reviewed approaches in terms of their general advantages (preserving the image brightness, histogram splitting, or noise reduction) as well as typical shortcomings (computational intensity, regional contrast reduction, introduction of artifacts).
Many deep learning models are either integrated with image enhancement techniques, or there are special deep learning models that improve the image quality. For instance, [135] used three preprocessing techniques, HE, CLAHE, and AD. They saw an effective impact on the performance of deep learning models (U-Net, CNN) in segmenting lumbar spine X-ray images. They concluded that CLAHE and AD can significantly improve the segmentation for models such as Unet. AD helped in noise removal. But the main problem arose that not all deep learning models benefit from the enhancement. HE increased the noise and artifacts, negatively impacting segmentation. Additionally, when performing image preprocessing, it adds additional overhead.
Recently [136], study proposes Residual Refinement Enhancement Network (RRENet), a novel end-to-end deep learning framework, to enhance accelerated Magnetic Resonance Imaging (MRI) quality for use in image-guided radiotherapy (MRigRT). RRENet combines a Generative Adversarial Network (GAN) for initial low-frequency content prediction and a Diffusion Model (DM) with a High-frequency Separation Training Module (HSTM) to refine residual errors and preserve fine details. Deep learning methods, including U-Nets, Convolutional Neural Networks (CNNs), GANs, and DMs, have shown promise in translating low-quality (LQ) images to high-quality (HQ) images, but often fail to adequately recover fine details. To address these limitations, RRENet is proposed as a dual-stage network where a GAN predicts the general structure and a conditional DM with HSTM refines the output by recovering high-frequency details. RRENet successfully generates high-quality MRI images from high-speed scanning sequences, enabling accurate correction of radiotherapy setup errors. With potential future advancements in acceleration and dataset expansion, the proposed method holds promise for implementing adaptive workflows in clinical settings.
Future research should explore the fusion of histogram-based methods with Generative Adversarial Networks (GANs) and conditional GANs—similar to the PixMed-Enhancer approach [137]. To build clinical trust, future contrast enhancement tools should integrate explainable AI (XAI) components, highlighting which regions were enhanced and why, based on histogram distributions and clinical relevance.
Retinex-based framework [138], designed to enhance low-light images in real-time across domains like security surveillance, medical imaging, and autonomous navigation. The method integrates Single-Scale Retinex (SSR) and Multi-Scale Retinex (MSR) to decompose images into illumination and reflectance components, improving brightness, contrast, and detail visibility while reducing noise and preserving edges. This Retinex-based enhancement method demonstrates significant strengths including high accuracy, real-time processing capability, robustness across diverse lighting conditions, and effective noise suppression with detail preservation. However, it faces limitations such as occasional color imbalance in extreme low-light scenarios, dependence on dataset quality, and the need for parameter tuning in new environments. Looking forward, future trends point toward integration with deep learning, edge/IoT deployment, and expansion into video and 3D medical imaging. Its best use cases include real-time surveillance, medical imaging pipelines, and autonomous vehicle perception, where reliable low-light enhancement is critical.
Future research should also focus on enhancing 3D medical image segmentation [139] techniques by integrating advanced models like the Mamba-Enhanced Diffusion Model. This approach, which utilizes a Semantic Hierarchical Embedding mechanism, shows promise in preserving fine-grained features and structural consistency, addressing the limitations of traditional 2D methods. Continued exploration of multi-scale feature extraction and improved conditional information guidance will be crucial for achieving higher accuracy in complex medical scenarios. Recent studies have shown that popular histogram equalization techniques, including Global Histogram Equalization (GHE), CLAHE, Histogram Specification, and BPDHE, can introduce significant data loss (5–40%) in medical images [140]. For example, tests on brain tumor MRI and colorectal cancer datasets demonstrated reduced correlation with original images and a substantial drop in CNN model accuracy, with VGG-16 accuracy decreasing from 82.5% to 51% when trained on CLAHE-processed images. To mitigate such issues, linear contrast enhancement methods, such as the Reinhard technique, are recommended, as they preserve nearly all original information while still improving contrast. This highlights the need for careful application of histogram-based methods, especially in clinical AI workflows where accurate disease detection is critical.
By looking at Table 7, we see that conventional histogram equalization techniques continue to play an important role in medical image enhancement due to their simplicity, interpretability, robustness, and low computational overhead, making them reliable and trustworthy tools in both standalone applications and as complementary preprocessing steps in deep learning pipelines.
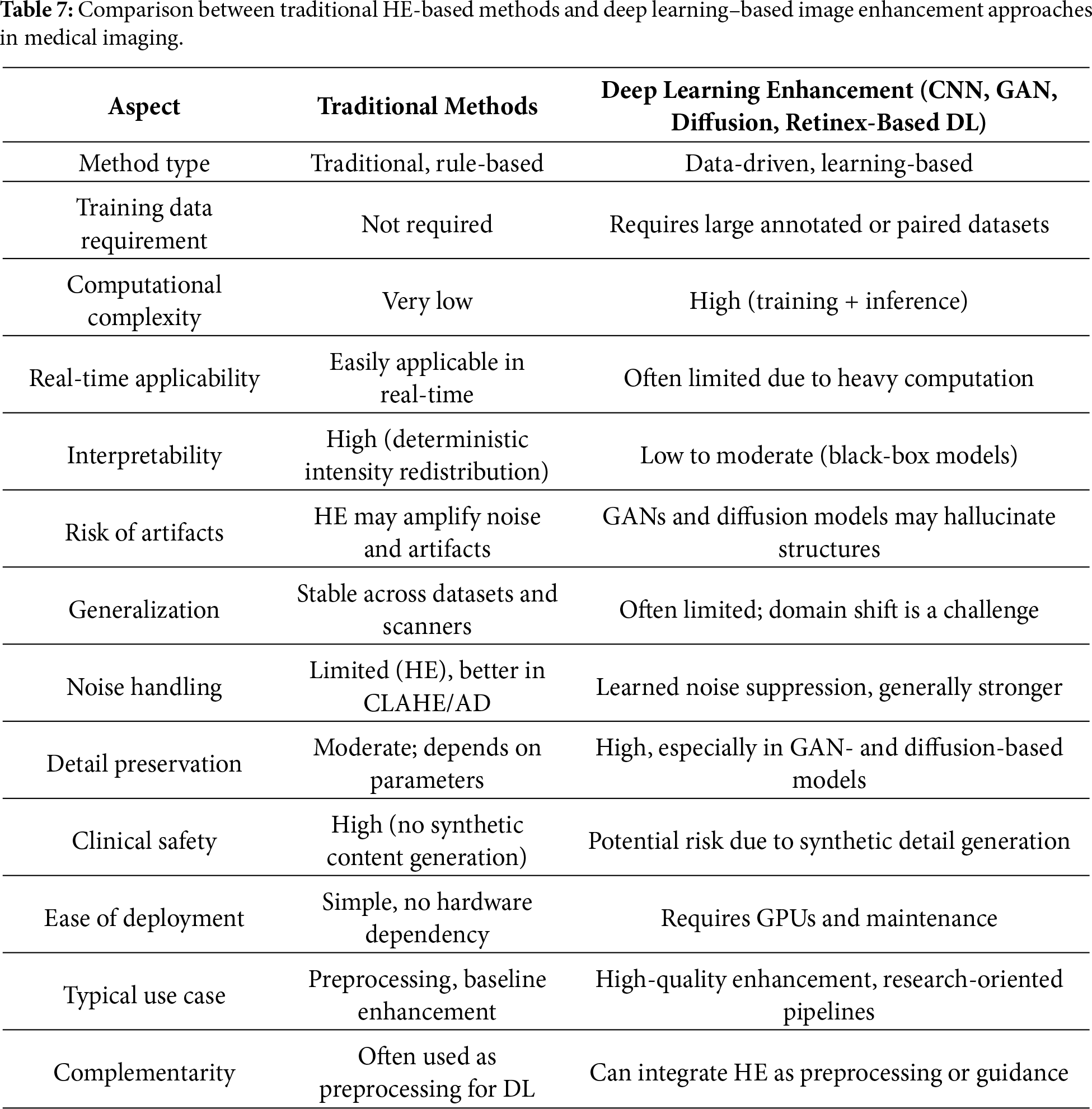
Fig. 13 illustrates the overall evolution of HE-based methodologies and highlights key areas requiring attention. Based on this, the following future challenges have been identified:
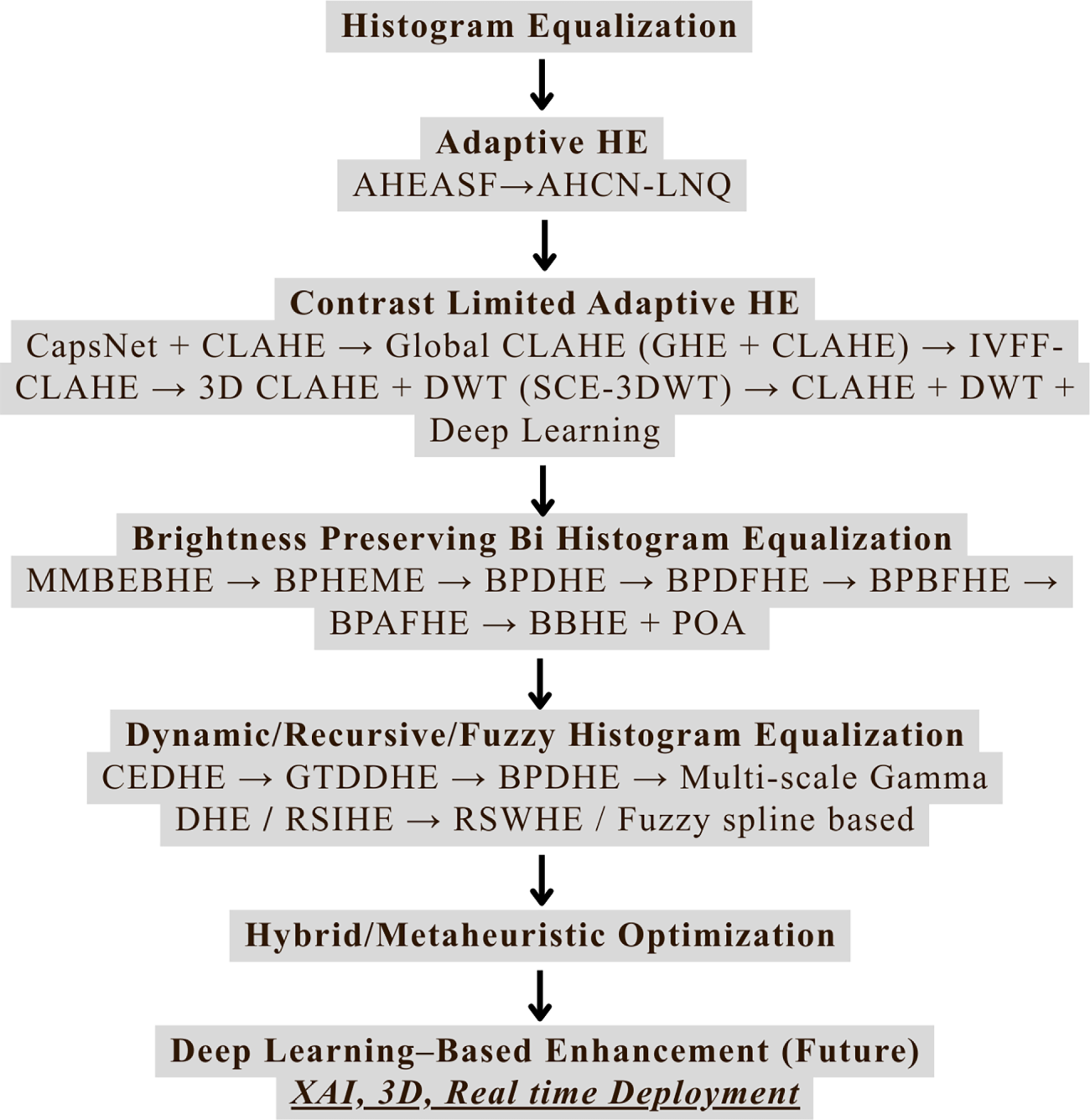
Figure 13: Overall methodological evolution and future challenges.
1. Diagnostic consistency
2. Interpretability
3. Generalization
4. Clinical validation
5. Regulatory compliance
5 Clinical Feasibility, Risks, and Limitations
Histogram equalization (HE) and its modifications, although successful in image contrast enhancement, can pose difficulties when implemented in a clinical environment. Firstly, the ability to disrupt the grayscale consistency of tissues can be misleading and result in the misinterpretation of actual tissue pathologies. This can be particularly problematic when over-enhanced areas, such as tumors, are misrepresented, or when the loss of tissue detail is inadvertently interpreted as a pathological condition. Furthermore, the clinical application of medical imaging is governed by strict regulations and quality control requirements, as set by the FDA and ISO. Any image contrast enhancement algorithm to be used in a clinical setting must therefore adhere to validation and quality control procedures to ensure consistency and accuracy of results. Lastly, the actual implementation of HE-based algorithms can be modality-dependent (CT, MRI, and X-ray), as well as dependent on the quality of the image acquisition process. Other factors include computational complexity, reproducibility of results, and the need for operator training to accurately interpret enhanced images.
We discussed the development of HE-based contrast enhancement methods in medical imaging, which have made significant strides toward addressing the core problems with medical image analysis. The examination of different methods, ranging from traditional HE to complex hybrid optimization techniques, shows how clinical judgment and diagnostic accuracy are always improving. Even though classical HE has good computational performance, it suffers from over-enhancement and brightness distortion errors, which reduces its accuracy. The need for local contrast enhancement gave rise to adaptive methods like AHE and CLAHE. In contrast, brightness-preserving techniques addressed the critical requirement to preserve original intensity relationships necessary for quantitative medical analysis.
Furthermore, through fuzzy logic, metaheuristic optimization algorithms, and other hybrid methods, these approaches have been optimized to balance the competing requirements of contrast improvement, noise reduction, brightness preservation, and low computational cost. Clinically relevant techniques, such as BPDHE and various modifications of CLAHE, as well as more clinically useful optimization-based approaches, are of particular interest because they have been shown to improve performance indicators.
CLAHE is the gold standard for many medical imaging applications because it achieves the perfect balance between usefulness and performance. The ability to identify important characteristics, such as lesions in brain MRIs, microscopic nodules in lung CT scans, and microcalcifications in mammograms, has improved over time. More precise quantitative measurements are made possible by brightness-preserving techniques, which are crucial for monitoring disease progression and developing effective treatment plans. Fuzzy and dynamic techniques are enhanced while retaining a natural look. These methods are helpful for sophisticated medical images that need sensitive refining. The performance potential of hybrid optimization techniques is highest with adaptive parameter optimization; however, a tradeoff between clinical usefulness and computational cost must be exercised. In terms of drawbacks, a large number of advanced optimization techniques are too computationally intensive for clinical use. These methods may not be convenient for everyday clinical practice because they require significant parameter adjustments without automated optimization. In particular, capturing faint images with low-dose imaging becomes especially challenging when balancing noise and contrast. The review paper describes several limitations of histogram-based and deep learning-based contrast enhancement techniques, which show better performance than standard methods because they create visual artifacts, they lose vital information, their effectiveness depends on specific models, and their demands require excessive processing power. The effectiveness of enhancement methods displays variation because different imaging modalities and clinical tasks create distinct requirements, which results in different deep learning performance outcomes. Future research should therefore focus on hybrid enhancement frameworks, information-preserving methods, explainable AI integration, and advanced generative models to ensure robust, clinically reliable image enhancement.
Figs. 14–16 provide a comprehensive overview of the literature selection and analysis process adopted in this review. Fig. 14, presented as a bar graph, depicts the distribution of selected papers across different publication sources. Fig. 15 categorizes the selected literature based on publication type. Fig. 16, presented as a line chart, illustrates the yearly trend of publications considered in this study.
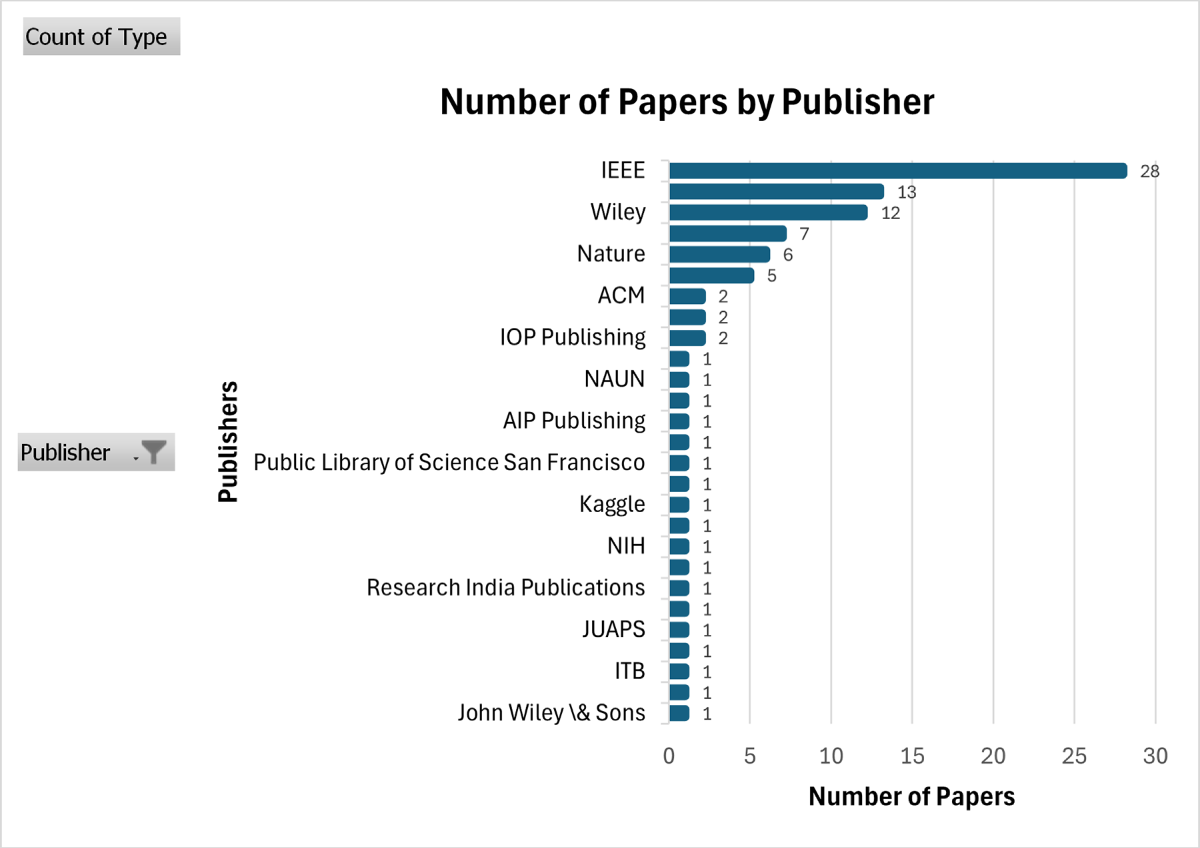
Figure 14: Number of papers.
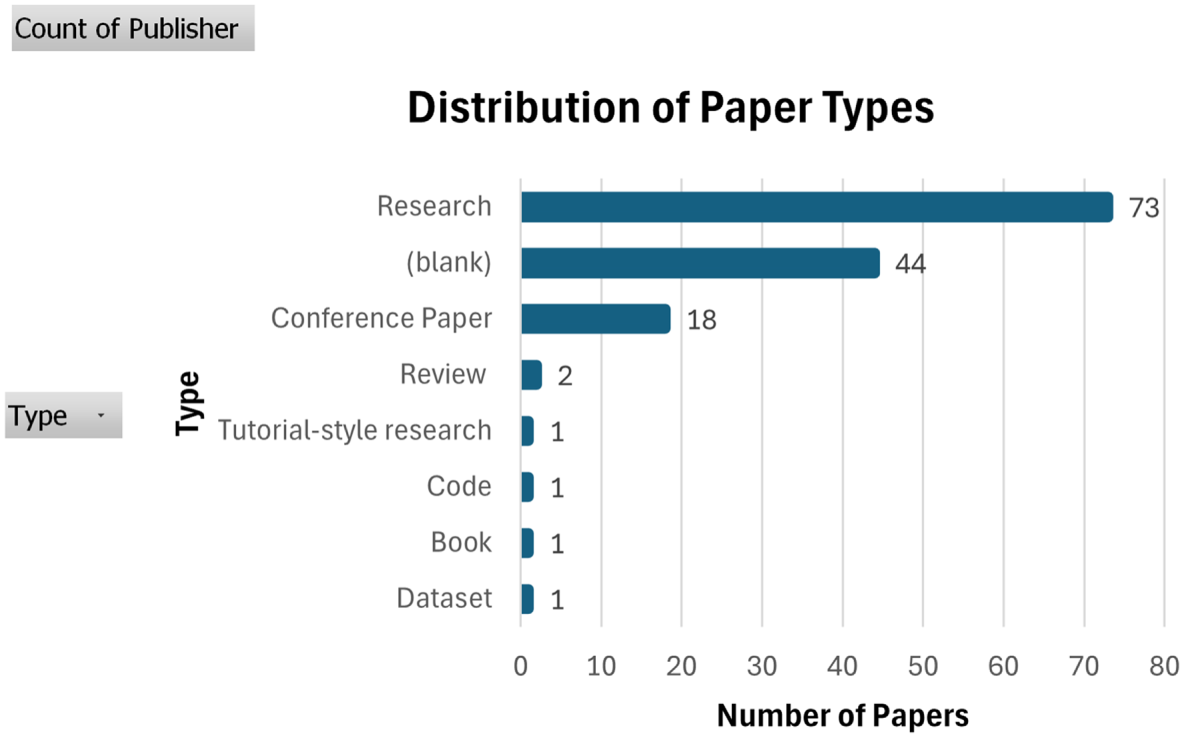
Figure 15: The distribution of papers with publication library. (Data for these figure were collected from the references listed in the study and visualized using Excel).
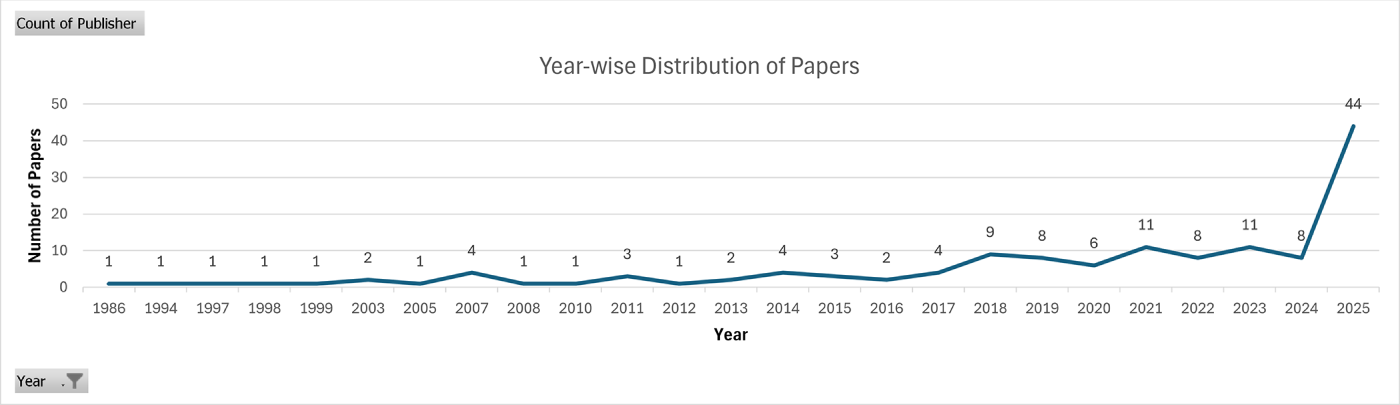
Figure 16: Year-wise distribution of publications on histogram-based contrast enhancement techniques. (Data for this figure were collected from the references listed in the study and visualized using Excel).
Acknowledgement: The authors acknowledge with thanks to the technical and financial support provided by King Abdulaziz University, Jeddah, Saudi Arabia.
Funding Statement: This project was funded by the Deanship of Scientific Research (DSR) at King Abdulaziz University, Jeddah, under grant No. (IFPDP-261-22).
Author Contributions: The authors confirm contribution to the paper as follows: Conceptualization, Saira Ali Bhatti, Maqbool Khan, and Wadee Alhalabi; methodology, Saira Ali Bhatti, Maqbool Khan, and Arshad Ahmad; validation, Muhammad Shahid Anwar, Leila Jamel, and Aisha M. Mashraqi; formal analysis, Saira Ali Bhatti and Arshad Ahmad; investigation, Saira Ali Bhatti, Maqbool Khan, Arshad Ahmad, Muhammad Shahid Anwar, Leila Jamel, Aisha M. Mashraqi, and Wadee Alhalabi; data curation, Saira Ali Bhatti and Maqbool Khan; writing—original draft preparation, Saira Ali Bhatti, Maqbool Khan, and Arshad Ahmad; writing—review and editing, Saira Ali Bhatti, Maqbool Khan, Arshad Ahmad, Muhammad Shahid Anwar, Leila Jamel, Aisha M. Mashraqi, and Wadee Alhalabi; visualization, Leila Jamel; supervision, Maqbool Khan and Wadee Alhalabi; project administration, Maqbool Khan; funding acquisition, Wadee Alhalabi. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: The brain tumor MRI dataset used for illustrative examples in this review is openly available at Kaggle: https://www.kaggle.com/datasets/masoudnickparvar/brain-tumor-mri-dataset.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
List of Abbreviations
| Abbreviation | Full Form |
| AHE | Adaptive Histogram Equalization |
| AMBE | Absolute Mean Brightness Error |
| AHEASF | Adaptive Histogram Equalization based on an Adaptive Sigmoid Function |
| AHCN-LNQ | Adaptive Histogram Contrast Normalization with Learning-based Neural Quantization |
| ACELF | Adaptive Contrast Enhancement With Lesion Focusing |
| BBHE | Brightness Preserving Bi-Histogram Equalization |
| BPBFHE | Brightness Preserving Bi-level Fuzzy Histogram Equalization |
| BPAFHE | Brightness Preserving Adaptive Fuzzy Histogram Equalization |
| BPDHE | Brightness Preserving Dynamic Histogram Equalization |
| BPDFHE | Brightness Preserving Dynamic Fuzzy Histogram Equalization |
| BPHEME | Brightness Preserving Histogram Equalization with Maximum Entropy |
| POA | Pelican Optimization Algorithm |
| CLAHE | Contrast Limited Adaptive Histogram Equalization |
| CNN | Convolutional Neural Network |
| CDF | Cumulative Distribution Function |
| CWCO | Chaos World Cup Optimization |
| ERBMAHE | Exposure Region Based Multi-Algorithm Histogram Equalization |
| CT | Computed Tomography |
| DCT | Discrete Cosine Transform |
| DHE | Dynamic Histogram Equalization |
| DSIHE | Dualistic Sub-Image Histogram Equalization |
| DWT | Discrete Wavelet Transform |
| FPGA | Field-Programmable Gate Array |
| FSDHE | Fuzzy Spline-based Histogram Equalization |
| GAN | Generative Adversarial Network |
| GLCM | Gray Level Co-occurrence Matrix |
| GQAOA | Gaussian Quantum Arithmetic Optimization Algorithm |
| HE | Histogram Equalization |
| IBBHE | Iterative Brightness Bi-Histogram Equalization |
| IVFF-CLAHE | Interval-Valued Fermatean Fuzzy CLAHE |
| KHO | Krill Herd Optimization |
| LMWCO | Logistic Map World Cup Optimization |
| MDSIHE | Modified Dualistic Sub-Image Histogram Equalization |
| MEDHS | Maximum Entropy Distribution-based Histogram Specification |
| MMBEBHE | Minimum Mean Brightness Error Bi-Histogram Equalization |
| MRI | Magnetic Resonance Imaging |
| PSNR | Peak Signal-to-Noise Ratio |
| PSO | Particle Swarm Optimization |
| PSOTHE | Particle Swarm Optimized Texture Based Histogram Equalization |
| Probability Density Function | |
| RHE | Reformed Histogram Equalization |
| RF Radio | Frequency |
| RMSHE | Recursive Mean-Separate Histogram Equalization |
| RSIHE | Recursive Sub-Image Histogram Equalization |
| RSWHE | Recursively Separated and Weighted Histogram Equalization |
| ROI | Region of interest |
| SSIM | Structural Similarity Index |
| SCE-3DWT | Selective Coefficient-Enhanced 3D Wavelet Transform and 3D CLAHE |
| GAAHE | Genetic Algorithm based Adaptive Histogram Equalization |
| GAHE | Genetic Algorithm-Based Histogram Equalization |
| WCO | World Cup Optimization |
References
1. Hussain S, Mubeen I, Ullah N, Shah SSUD, Khan BA, Zahoor M, et al. Modern diagnostic imaging technique applications and risk factors in the medical field: a review. BioMed Res Int. 2022;2022(1):5164970. doi:10.1155/2022/5164970. [Google Scholar] [PubMed] [CrossRef]
2. Khan IR, Mirza W, Siddiq A, Shim SO. Improving visual quality of CT images through enhancement of base and detail layers. J Adv Sci Eng. 2024;27(3):2147–57. [Google Scholar]
3. Kaur H, Rani J. MRI brain image enhancement using Histogram Equalization techniques. In: Proceedings of the 2016 International Conference on Wireless Communications, Signal Processing and Networking (WiSPNET); 2016 Mar 23–25; Chennai, India. p. 770–3. doi:10.1109/wispnet.2016.7566237. [Google Scholar] [CrossRef]
4. Nickparvar M. Brain tumor MRI dataset. Kaggle. 2021 [cited 2026 Jan 1]. Available from: https://www.kaggle.com/dsv/2645886. [Google Scholar]
5. Dawood DFA. The importance of contrast enhancement in medical images analysis and diagnosis. Int J Eng Res Technol. 2018;7(12):21–4. doi:10.17577/ijertv7is120006. [Google Scholar] [CrossRef]
6. Solomon C, Breckon T. Fundamentals of digital image processing: a practical approach with examples in Matlab. Hoboken, NJ, USA: John Wiley & Sons; 2011. doi:10.1002/9780470689776. [Google Scholar] [CrossRef]
7. Rahman S, Rahman MM, Hussain K, Khaled SM, Shoyaib M. Contrast enhancement of medical images using a new version of the World Cup Optimization algorithm. In: Proceedings of the 2014 17th International Conference on Computer and Information Technology (ICCIT); 2014 Dec 22–23; Dhaka, Bangladesh. p. 368–73. [Google Scholar]
8. Singh G, Mittal A. Various image enhancement techniques-a critical review. Int J Innov Sci Res. 2014;10(2):267–74. [Google Scholar]
9. Dynamsoft. Image processing 101—image enhancement. 2023 [cited 2025 Jul 24]. Available from: https://www.dynamsoft.com/blog/insights/image-processing/image-processing-101-image-enhancement/. [Google Scholar]
10. Abdul Wahab A, Mohamad Salim MI, Yunus J, Ramlee MH. Comparative evaluation of medical thermal image enhancement techniques for breast cancer detection. J Eng Technol Sci. 2018;50(1):40–52. doi:10.5614/j.eng.technol.sci.2018.50.1.3. [Google Scholar] [CrossRef]
11. Mousania Y, Karimi S, Farmani A. Optical remote sensing, brightness preserving and contrast enhancement of medical images using histogram equalization with minimum cross-entropy-Otsu algorithm. Opt Quantum Electron. 2022;55(2):105. doi:10.1007/s11082-022-04341-z. [Google Scholar] [CrossRef]
12. Kandhway P, Bhandari AK, Singh A. A novel reformed histogram equalization based medical image contrast enhancement using krill herd optimization. Biomed Signal Process Control. 2020;56(1):101677. doi:10.1016/j.bspc.2019.101677. [Google Scholar] [CrossRef]
13. Ren X, Lai S. Medical image enhancement based on laplace transform, sobel operator and histogram equalization. Acad J Comput Inf Sci. 2022;5(6):48–54. doi:10.25236/ajcis.2022.050608. [Google Scholar] [CrossRef]
14. Deng C, Ma W, Yin Y. An edge detection approach of image fusion based on improved Sobel operator. In: Proceedings of the 2011 4th International Congress on Image and Signal Processing; 2011 Oct 15–17; Shanghai, China. p. 1189–93. doi:10.1109/cisp.2011.6100499. [Google Scholar] [CrossRef]
15. Agarwal M, Mahajan R. Medical image contrast enhancement using range limited weighted histogram equalization. Procedia Comput Sci. 2018;125(1):149–56. doi:10.1016/j.procs.2017.12.021. [Google Scholar] [CrossRef]
16. Pizer SM, Amburn EP, Austin JD, Cromartie R, Geselowitz A, Greer T, et al. Adaptive histogram equalization and its variations. Comput Graph Image Process. 1977;6(3):184–95. doi:10.1016/s0734-189x(87)80186-x. [Google Scholar] [CrossRef]
17. Pizer SM, Austin JD, Perry JR, Safrit HD, Zimmerman JB. Adaptive histogram equalization for automatic contrast enhancement of medical images. In: Proceedings of the Application of Optical Instrumentation in Medicine XIV and Picture Archiving and Communication Systems; 1986 Feb 2–7; Newport Beach, CA, USA. p. 242–50. [Google Scholar]
18. Sherrier RH, Johnson GA. Regionally adaptive histogram equalization of the chest. IEEE Trans Med Imaging. 1987;6(1):1–7. doi:10.1109/tmi.1987.4307791. [Google Scholar] [PubMed] [CrossRef]
19. Vidyasagar KB, Ajay S, Sachin BD, Sharath HS, Indrakumar K. Enhancement of microscopic medical images using adaptive sigmoid function. In: Proceedings of the 2025 3rd International Conference on Data Science and Information System (ICDSIS); 2025 May 16–17; Hassan, India. p. 1–5. doi:10.1109/icdsis65355.2025.11070370. [Google Scholar] [CrossRef]
20. Ramamoorthy M, Qamar S, Manikandan R, Jhanjhi NZ, Masud M, AlZain MA. Earlier detection of brain tumor by pre-processing based on histogram equalization with neural network. Healthcare. 2022;10(7):1218. doi:10.3390/healthcare10071218. [Google Scholar] [PubMed] [CrossRef]
21. Ang J, Sim KS, Tan SC, Lim CP. Adaptive contrast enhancement with lesion focusing (ACELF). IEEE Access. 2025;13(2):41785–96. doi:10.1109/access.2025.3546913. [Google Scholar] [CrossRef]
22. Bangare SL, Dubal A, Bangare PS, Patil ST. Reviewing otsu’s method for image thresholding. Int J Appl Eng Res. 2015;10(9):21777–83. doi:10.37622/ijaer/10.9.2015.21777-21783. [Google Scholar] [CrossRef]
23. Huang Z, Wang Z, Zhang J, Li Q, Shi Y. Image enhancement with the preservation of brightness and structures by employing contrast limited dynamic quadri-histogram equalization. Optik. 2021;226(2):165877. doi:10.1016/j.ijleo.2020.165877. [Google Scholar] [CrossRef]
24. Saini K, Gangwar S, Devi R. A novel region-based modified histogram equalization for enhancing non-uniformly illuminated chest X-ray images. Procedia Comput Sci. 2025;260(2):191–200. doi:10.1016/j.procs.2025.03.193. [Google Scholar] [CrossRef]
25. Shilpa N, Ayeesha Banu W. Tuberculosis detection using deep and hybrid learning techniques using X-ray images. Neural Comput Appl. 2025;37(18):12373–406. doi:10.1007/s00521-025-11173-x. [Google Scholar] [CrossRef]
26. Zuiderveld K. Contrast limited adaptive histogram equalization. Utrecht, The Netherlands: Computer Vision Research Group, Utrecht University; 1994 [cited 2026 Jan 1]. Available from: https://www.cse.unr.edu/bebis/CS474/StudentPaperPresentations/Constrast-Limited. [Google Scholar]
27. Pisano ED, Zong S, Hemminger BM, DeLuca M, Johnston RE, Muller K, et al. Contrast limited adaptive histogram equalization image processing to improve the detection of simulated spiculations in dense mammograms. J Digit Imag. 1998;11(4):193. doi:10.1007/BF03178082. [Google Scholar] [PubMed] [CrossRef]
28. Somal S. Image enhancement using local and global histogram equalization technique and their comparison. In: Proceedings of the First International Conference on Sustainable Technologies for Computational Intelligence; 2019 Mar 29–30; Rajasthan, India. Singapore: Springer; 2019. p. 739–53. doi:10.1007/978-981-15-0029-9_58. [Google Scholar] [CrossRef]
29. Nia SN, Shih FY. Medical X-ray image enhancement using global contrast-limited adaptive histogram equalization. Int J Patt Recogn Artif Intell. 2024;38(12):2457010. doi:10.1142/s0218001424570106. [Google Scholar] [CrossRef]
30. Moradmand H, Setayeshi S, Karimian AR, Sirous M, Akbari ME. Comparing the performance of image enhancement methods to detect microcalcification clusters in digital mammography. Iran J Cancer Prev. 2012;5(2):61. [Google Scholar] [PubMed]
31. Yadav G, Maheshwari S, Agarwal A. Contrast limited adaptive histogram equalization based enhancement for real time video system. In: Proceedings of the 2014 International Conference on Advances in Computing, Communications and Informatics (ICACCI); 2014 Sep 24–27; Delhi, India. p. 2392–7. doi:10.1109/icacci.2014.6968381. [Google Scholar] [CrossRef]
32. Singh P, Mukundan R, De Ryke R. Feature enhancement in medical ultrasound videos using contrast-limited adaptive histogram equalization. J Digit Imag. 2020;33(1):273–85. doi:10.1007/s10278-019-00211-5. [Google Scholar] [PubMed] [CrossRef]
33. Koonsanit K, Thongvigitmanee S, Pongnapang N, Thajchayapong P. Image enhancement on digital X-ray images using N-CLAHE. In: Proceedings of the 2017 10th Biomedical Engineering International Conference (BMEiCON); 2017 Aug 31–Sep 2; Hokkaido, Japan. p. 1–4. doi:10.1109/bmeicon.2017.8229130. [Google Scholar] [CrossRef]
34. Amorim PH, Moraes TF, Silva J, Pedrini H. 3D adaptive histogram equalization method for medical volumes. In: Proceedings of the VISIGRAPP (4: VISAPP); 2018 Jan 27–29; Madeira, Portugal. p. 363–70. [Google Scholar]
35. Dinh PH, Giang NL. A new medical image enhancement algorithm using adaptive parameters. Int J Imaging Syst Technol. 2022;32(6):2198–218. doi:10.1002/ima.22778. [Google Scholar] [CrossRef]
36. Mortatha MB, Thabit SS, Ameer HRA, Nuiaa RR. Dynamic clip limit window size histogram equalization for poor information images. Int J Intell Eng Syst. 2022;15(5):57. doi:10.22266/ijies2022.1031.06. [Google Scholar] [CrossRef]
37. Wang H, Wang D, Wang Z, Li C. Enhancing CLAHE with interval-valued fermatean fuzziness for robust low-light image enhancement. In: Proceedings of the 2024 IEEE 9th International Conference on Data Science in Cyberspace (DSC); 2024 Aug 23–26; Jinan, China. p. 345–53. doi:10.1109/dsc63484.2024.00053. [Google Scholar] [CrossRef]
38. Opoku M, Weyori BA, Adekoya AF, Adu K. CLAHE-CapsNet: efficient retina optical coherence tomography classification using capsule networks with contrast limited adaptive histogram equalization. PLoS One. 2023;18(11):e0288663. doi:10.1371/journal.pone.0288663. [Google Scholar] [PubMed] [CrossRef]
39. Hrtinger P, Steger C. Adaptive histogram equalization in constant time. J Real Time Image Process. 2024;21(3):93. doi:10.1007/s11554-024-01465-1. [Google Scholar] [CrossRef]
40. Majid Mohammed I, Ashidi Mat Isa N. Contrast limited adaptive local histogram equalization method for poor contrast image enhancement. IEEE Access. 2025;13(2):62600–32. doi:10.1109/access.2025.3558506. [Google Scholar] [CrossRef]
41. Khan FF, Kim JH, Park CS, Kim JI, Kwon GR. A resource-efficient 3D U-Net for hippocampus segmentation using CLAHE and SCE-3DWT techniques. IEEE Access. 2025;13(10):99923–38. doi:10.1109/access.2025.3577266. [Google Scholar] [CrossRef]
42. Hekmat A, Zhang Z, Rehman Khan SU, Bilal O. Brain tumor diagnosis redefined: leveraging image fusion for MRI enhancement classification. Biomed Signal Process Control. 2025;109:108040. doi:10.1016/j.bspc.2025.108040. [Google Scholar] [CrossRef]
43. Arulraj A, Ronjalis RR. An improved neighbourhood-based contrast limited adaptive histogram equalization method for contrast enhancement on retinal images. Int J Ophthalmol. 2025;18(12):2225–36. doi:10.18240/ijo.2025.12.02. [Google Scholar] [PubMed] [CrossRef]
44. Anwariningsih SH, Wahyono, Sumiharto R. An improved cuckoo search algorithm with dynamic parameters and hybrid distribution for enhanced CLAHE. Eng Technol Appl Sci Res. 2025;15(4):24148–58. doi:10.48084/etasr.10652. [Google Scholar] [CrossRef]
45. Halder N, Roy D, Sarkar PP, Patra SN, Bandyopadhyay S. PSO-optimized CLAHE for image enhancement and active contour-based segmentation of retinal vessels. Quad J. 2025;13:49–64. [Google Scholar]
46. Rangaiah PKB, Pradeep Kumar BP, Augustine R. Improving burn diagnosis in medical image retrieval from grafting burn samples using B-coefficients and the CLAHE algorithm. Biomed Signal Process Control. 2025;99(3):106814. doi:10.1016/j.bspc.2024.106814. [Google Scholar] [CrossRef]
47. Rifat AH, Rahman MM, Tanzin LA, Ahammed MS, Farid Uddin MS, Rahman MM. An image enhancement technique by optimizing CLAHE parameter through machine learning. In: Proceedings of the 2025 IEEE International Symposium on Consumer Technology (ISCT); 2025 Sep 16–18; Bali, Indonesia. p. 212–7. doi:10.1109/isct66099.2025.11297358. [Google Scholar] [CrossRef]
48. Krayla J, Kumar S, Acharya UK, Bharti M. Fuzzy logic based two stage contrast limited adaptive histogram equalization (FLTSCLAHE) technique for MRI brain image enhancement. Sādhanā. 2025;50(4):307. doi:10.1007/s12046-025-02983-9. [Google Scholar] [CrossRef]
49. Vinuja G, Devi SR, Salma SN, Ahamed AN, Kanmani R. Improved detection of early diabetic retinopathy using contrast limited adaptive histogram equalization and principal component analysis with bag of visual words based SVM. In: Proceedings of the 2025 International Conference on Intelligent Computing and Knowledge Extraction (ICICKE); 2025 Jun 6–7; Bengaluru, India. p. 1–8. doi:10.1109/icicke65317.2025.11136603. [Google Scholar] [CrossRef]
50. Dubey S, Choudhary SK, Dwivedi P, Pandey A, Niranjan DS, Saxena GK. CLAHE-enhanced FCM-CNN-SVM pipeline for early skin cancer detection using ISIC-2020 dataset. In: Proceedings of the 2025 International Conference on Engineering Innovations and Technologies (ICoEIT); 2025 Jul 4–5; Bhopal, India. p. 1341–7. doi:10.1109/icoeit63558.2025.11211759. [Google Scholar] [CrossRef]
51. Nagasirisha B, Reddy GVK, Sugunamma K, Abhishek G, Teja KS. An optimized edge preserving medical image fusion framework using NSCT for improved diagnostic accuracy. GAMANAM Glob Adv Multidiscip Appl Next-Gen Mod Technol. 2025;1(2):113–22. [Google Scholar]
52. Priyah R, Kamalakkannan S. Contrast limited adaptive histogram equalization approach contrast enhancement in liver tumor. In: Proceedings of the 2025 9th International Conference on Inventive Systems and Control (ICISC); 2025 Aug 12–13; Coimbatore, India. p. 1–7. doi:10.1109/icisc65841.2025.11187445. [Google Scholar] [CrossRef]
53. Priyah RR, Kamalakkannan S. Hybrid contrast-limited adaptive histogram equalization and Deep Learning techniques for improving liver tumor detection. Futech. 2025;4(3):67–75. doi:10.55670/fpll.futech.4.3.7. [Google Scholar] [CrossRef]
54. Kim YT. Contrast enhancement using brightness preserving bi-histogram equalization. IEEE Trans Consumer Electron. 1997;43(1):1–8. doi:10.1109/30.580378. [Google Scholar] [CrossRef]
55. Shen H, Sun S, Lei B, Zheng S. An adaptive brightness preserving bi-histogram equalization. MIPPR 2011 Parallel Process Images Optim Med Imag Process. 2011;8005:80050U. doi:10.1117/12.902215. [Google Scholar] [CrossRef]
56. Moniruzzaman M, Shafuzzaman M, Hossain MF. Brightness preserving bi-histogram equalization using edge pixels information. In: Proceedings of the 2013 International Conference on Electrical Information and Communication Technology (EICT); 2014 Feb 13–15; Khulna, Bangladesh. p. 1–5. doi:10.1109/eict.2014.6777872. [Google Scholar] [CrossRef]
57. Wang Z, Yue H, Wang J, Tao H, Wang L. Design and implementation of brightness configurable bi-histogram equalization based on FPGA. IOP Conf Ser Earth Environ Sci. 2021;693:012087. doi:10.1088/1755-1315/693/1/012087. [Google Scholar] [CrossRef]
58. Dahmane O, Khelifi M, Beladgham M, Kadri I. Pneumonia detection based on transfer learning and a combination of VGG19 and a CNN Built from scratch. Indones J Electr Eng Comput Sci. 2021;24(3):1469. doi:10.11591/ijeecs.v24.i3.pp1469-1480. [Google Scholar] [CrossRef]
59. Ismael AN, Khazal IF. Improved brightness-preserving bi-histogram equalization (BBHE) technique based on the pelican optimization algorithm (POA) for image enhancement. J Univ Anbar Pure Sci. 2025;19(1):248–59. [Google Scholar]
60. Gangwar S, Devi R, Mat Isa NA. Optimized exposer region-based modified adaptive histogram equalization method for contrast enhancement in CXR imaging. Sci Rep. 2025;15(1):6693. doi:10.1038/s41598-025-90876-6. [Google Scholar] [PubMed] [CrossRef]
61. Gangwar S, Saini K, Devi R. A novel modified histogram equalization approach for enhancement of non-uniform illumination of Chest X-ray images. Procedia Comput Sci. 2025;258:1736–46. doi:10.1016/j.procs.2025.04.425. [Google Scholar] [CrossRef]
62. Wang Y, Chen Q, Zhang B. Image enhancement based on equal area dualistic sub-image histogram equalization method. IEEE Trans Consumer Electron. 1999;45(1):68–75. doi:10.1109/30.754419. [Google Scholar] [CrossRef]
63. Ezhilraja K, Shanmugavadivu P. Contrast enhancement of lung CT scan images using multi-level modified dualistic sub-image histogram equalization. In: Proceedings of the 2022 International Conference on Automation, Computing and Renewable Systems (ICACRS); 2022 Dec 13–15; Pudukkottai, India. p. 1009–14. doi:10.1109/icacrs55517.2022.10029217. [Google Scholar] [CrossRef]
64. Ullah N, Hassan M, Ali Khan J, Anwar MS, Aurangzeb K. Enhancing explainability in brain tumor detection: a novel DeepEBTDNet model with LIME on MRI images. Int J Imaging Syst Tech. 2024;34(1):e23012. doi:10.1002/ima.23012. [Google Scholar] [CrossRef]
65. Mohan KR, Thirugnanam G. A dualistic sub-image histogram equalization based enhancement and segmentation techniques for medical images. In: Proceedings of the 2013 IEEE Second International Conference on Image Information Processing (ICIIP-2013); 2013 Dec 9–11; Shimla, India. p. 566–9. doi:10.1109/iciip.2013.6707655. [Google Scholar] [CrossRef]
66. Abdullah-Al-Wadud M, Kabir M, Dewan MA, Chae O. A dynamic histogram equalization for image contrast enhancement. IEEE Trans Consumer Electron. 2007;53(2):593–600. doi:10.1109/tce.2007.381734. [Google Scholar] [CrossRef]
67. Abbasi R, Xu L, Wang Z, Chughtai GR, Amin F, Luo B. Dynamic weighted histogram equalization for contrast enhancement using for Cancer Progression Detection in medical imaging. In: Proceedings of the 2018 International Conference on Signal Processing and Machine Learning; 2018 Nov 28–30; Shanghai China. p. 93–8. doi:10.1145/3297067.3297086. [Google Scholar] [CrossRef]
68. Ismail WZW, Sim KS. Contrast enhancement dynamic histogram equalization for medical image processing application. Int J Imaging Syst Tech. 2011;21(3):280–9. doi:10.1002/ima.20295. [Google Scholar] [CrossRef]
69. Dhal KG, Das A, Ghoshal N, Das S. Variance based brightness preserved dynamic histogram equalization for image contrast enhancement. Pattern Recognit Image Anal. 2018;28(4):747–57. doi:10.1134/S1054661818040211. [Google Scholar] [CrossRef]
70. Kumar S, Bhandari AK, Raj A, Swaraj K. Triple clipped histogram-based medical image enhancement using spatial frequency. IEEE Trans Nanobioscience. 2021;20(3):278–86. doi:10.1109/tnb.2021.3064077. [Google Scholar] [PubMed] [CrossRef]
71. Vidyasaraswathi HN, Hanumantharaju MC. Gradient, texture driven based dynamic-histogram equalization for medical image enhancement. Int J Biol Biomed Eng. 2021;15:303–10. doi:10.46300/91011.2021.15.36. [Google Scholar] [CrossRef]
72. Abbasi R, Chen J, Al-Otaibi Y, Rehman A, Abbas A, Cui W. RDH-based dynamic weighted histogram equalization using for secure transmission and cancer prediction. Multimed Syst. 2021;27(2):177–89. doi:10.1007/s00530-020-00718-w. [Google Scholar] [CrossRef]
73. Chen M, Lin Z, Lin S, Lin J, Guo T. Electrowetting display of multiscale Gamma based on dynamic histogram equilibrium. Sci Rep. 2025;15(1):20471. doi:10.1038/s41598-025-04172-4. [Google Scholar] [PubMed] [CrossRef]
74. Ayaz S. Brightness-preserving and structure-enhanced image enhancement using dual-domain histogram equalization. J Innov Comput Emerg Technol. 2025;5(2):1–16. doi:10.56536/jicet.v5i2.227. [Google Scholar] [CrossRef]
75. Aosiman A, Abulijiang A, Abulikemu A, Abdukirim T, Maimaiti M. Medical image enhancement algorithm based on histogram equalization and dyadic wavelet transform. In: Proceedings of the 2020 3rd International Conference on Computer Science and Software Engineering; 2020 May 22–24; Beijing, China. p. 181–5. doi:10.1145/3403746.3403925. [Google Scholar] [CrossRef]
76. Chen SD, Ramli AR. Minimum mean brightness error bi-histogram equalization in contrast enhancement. IEEE Trans Consumer Electron. 2003;49(4):1310–9. doi:10.1109/tce.2003.1261234. [Google Scholar] [CrossRef]
77. Showkat EE, Sodhi DGK. Image enhancement in wavelet domain using histogram equalization and Median filters. Int J Innov Res Comput Sci Technol. 2023;11(1):25–31. doi:10.55524/ijircst.2023.11.1.6. [Google Scholar] [CrossRef]
78. Wang C, Ye Z. Brightness preserving histogram equalization with maximum entropy: a variational perspective. IEEE Trans Consumer Electron. 2005;51(4):1326–34. doi:10.1109/tce.2005.1561863. [Google Scholar] [CrossRef]
79. Yoo JH, Ohm SY, Chung MG. Brightness preservation and image enhancement based on maximum entropy distribution. In: Convergence and hybrid information technology. Berlin, Heidelberg: Springer; 2012. p. 365–72. doi:10.1007/978-3-642-32645-5_46. [Google Scholar] [CrossRef]
80. Ibrahim H, Pik Kong N. Brightness preserving dynamic histogram equalization for image contrast enhancement. IEEE Trans Consumer Electron. 2007;53(4):1752–8. doi:10.1109/tce.2007.4429280. [Google Scholar] [CrossRef]
81. Yang F, Li R. An improved method for brightness preserving dynamic histogram equalization. In: Proceedings of the 2022 International Conference on Artificial Intelligence and Computer Information Technology (AICIT); 2022 Sep 16–18; Yichang, China, p. 1–4. doi:10.1109/aicit55386.2022.9930196. [Google Scholar] [CrossRef]
82. Sheet D, Garud H, Suveer A, Mahadevappa M, Chatterjee J. Brightness preserving dynamic fuzzy histogram equalization. IEEE Trans Consumer Electron. 2010;56(4):2475–80. doi:10.1109/tce.2010.5681130. [Google Scholar] [CrossRef]
83. Magudeeswaran V, Ravichandran CG, Thirumurugan P. Brightness preserving bi-level fuzzy histogram equalization for MRI brain image contrast enhancement. Int J Imaging Syst Tech. 2017;27(2):153–61. doi:10.1002/ima.22219. [Google Scholar] [CrossRef]
84. Subramani B, Veluchamy M. MRI brain image enhancement using brightness preserving adaptive fuzzy histogram equalization. Int J Imaging Syst Tech. 2018;28(3):217–22. doi:10.1002/ima.22272. [Google Scholar] [CrossRef]
85. Kumar A, Kumar S, Kar A. Enhancing image contrast and preserving brightness using Min-max peak fuzzy histogram equalization. Circuits Syst Signal Process. 2025;44(7):4847–89. doi:10.1007/s00034-025-03017-9. [Google Scholar] [CrossRef]
86. Mishro PK, Agrawal S, Panda R, Abraham A. A novel brightness preserving joint histogram equalization technique for contrast enhancement of brain MR images. Biocybern Biomed Eng. 2021;41(2):540–53. doi:10.1016/j.bbe.2021.04.003. [Google Scholar] [CrossRef]
87. Veluchamy M, Mayathevar K, Subramani B. Brightness preserving optimized weighted bi-histogram equalization algorithm and its application to MR brain image segmentation. Int J Imaging Syst Tech. 2019;29(3):339–52. doi:10.1002/ima.22330. [Google Scholar] [CrossRef]
88. Shajy L, Smitha P, Marichami P. Enhancement of sputum cytology images through recursive mean separate histogram equalization and SVM classification. In: Proceedings of the 2014 IEEE International Conference on Computational Intelligence and Computing Research; 2014 Dec 18–20; Coimbatore, India. p. 1–5. doi:10.1109/iccic.2014.7238495. [Google Scholar] [CrossRef]
89. Sim KS, Tso CP, Tan YY. Recursive sub-image histogram equalization applied to gray scale images. Pattern Recognit Lett. 2007;28(10):1209–21. doi:10.1016/j.patrec.2007.02.003. [Google Scholar] [CrossRef]
90. Kim M, Chung M. Recursively separated and weighted histogram equalization for brightness preservation and contrast enhancement. IEEE Trans Consumer Electron. 2008;54(3):1389–97. doi:10.1109/tce.2008.4637632. [Google Scholar] [CrossRef]
91. Ali Qadar M, Yan Z, Rehman A, Alvi MA. Recursive weighted multi-plateau histogram equalization for image enhancement. Optik. 2015;126(24):5890–8. doi:10.1016/j.ijleo.2015.08.278. [Google Scholar] [CrossRef]
92. Saravanan S, Karthigaivel R. A fuzzy and spline based dynamic histogram equalization for contrast enhancement of brain images. Int J Imaging Syst Tech. 2021;31(2):802–27. doi:10.1002/ima.22483. [Google Scholar] [CrossRef]
93. Bhandari AK, Shahnawazuddin S, Meena AK. A novel fuzzy clustering-based histogram model for image contrast enhancement. IEEE Trans Fuzzy Syst. 2020;28(9):2009–21. doi:10.1109/tfuzz.2019.2930028. [Google Scholar] [CrossRef]
94. Subramani B, Veluchamy M. Fuzzy contextual inference system for medical image enhancement. Measurement. 2019;148(1):106967. doi:10.1016/j.measurement.2019.106967. [Google Scholar] [CrossRef]
95. Khan MF, Goyal D, Nofal MM, Khan E, Al-Hmouz R, Herrera-Viedma E. Fuzzy-based histogram partitioning for bi-histogram equalisation of low contrast images. IEEE Access. 2020;8:11595–614. doi:10.1109/access.2020.2965174. [Google Scholar] [CrossRef]
96. Subramani B, Veluchamy M. Fuzzy gray level difference histogram equalization for medical image enhancement. J Med Syst. 2020;44(6):103. doi:10.1007/s10916-020-01568-9. [Google Scholar] [PubMed] [CrossRef]
97. Janabi S, Ali MH. Image improvement via fuzzy inference centered contextual-dissimilarity histogram equalization. Int J Intell Eng Syst. 2022;15(6):101–110. doi:10.22266/ijies2022.1231.11. [Google Scholar] [CrossRef]
98. Saifullah S, Suryotomo AP. Thresholding and hybrid CLAHE-HE for chicken egg embryo segmentation. In: Proceedings of the 2021 International Conference on Communication & Information Technology (ICICT); 2021 Jun 5–6; Basrah, Iraq. p. 268–73. doi:10.1109/icict52195.2021.9568444. [Google Scholar] [CrossRef]
99. Wang C, Liu W, Jimenez G. Using chaos world cup optimization algorithm for medical images contrast enhancement. Concurr Comput Pract Exp. 2020;32(5):e5482. doi:10.1002/cpe.5482. [Google Scholar] [CrossRef]
100. Mu D, Xu C, Ge H. Hybrid genetic algorithm based image enhancement technology. In: Proceedings of the 2011 International Conference on Internet Technology and Applications; 2011 Aug 16–18; Wuhan, China. p. 1–4. doi:10.1109/itap.2011.6006336. [Google Scholar] [CrossRef]
101. Kaur P, Kaur R. A novel approach using image enhancement based on genetic algorithm. In: Proceedings of the International Conference on Advances in Engineering and Technology, ICAET14, IIT Roorkee; 2014 Feb 7–8; Chandigarh, India. p. 584–9. [Google Scholar]
102. Momeni M, Nezhad ZH, Moghaddam ME. Embryonic image enhancement based on genetic algorithm and generic filter. In: Proceedings of the 2017 3rd International Conference on Frontiers of Signal Processing (ICFSP); 2017 Sep 6–8; Paris, France. p. 141–5. doi:10.1109/icfsp.2017.8097158. [Google Scholar] [CrossRef]
103. Rundo L, Tangherloni A, Nobile MS, Militello C, Besozzi D, Mauri G, et al. MedGA: a novel evolutionary method for image enhancement in medical imaging systems. Expert Syst Appl. 2019;119(13):387–99. doi:10.1016/j.eswa.2018.11.013. [Google Scholar] [CrossRef]
104. Acharya UK, Kumar S. Genetic algorithm based adaptive histogram equalization (GAAHE) technique for medical image enhancement. Optik. 2021;230(3):166273. doi:10.1016/j.ijleo.2021.166273. [Google Scholar] [CrossRef]
105. Samraj D, Ramasamy K, Krishnasamy B. Enhancement and diagnosis of breast cancer in mammography images using histogram equalization and genetic algorithm. Multidimens Syst Signal Process. 2023;34(3):681–702. doi:10.1007/s11045-023-00880-0. [Google Scholar] [CrossRef]
106. Padmavathy N, Naidu NS, Harshitha V. A fusion of histogram equalization technique and fuzzy logic for sustained enhancement of images. IOP Conf Ser Earth Environ Sci. 2024;1375:012009. doi:10.1088/1755-1315/1375/1/012009. [Google Scholar] [CrossRef]
107. Sunanda P, Rani KA. Automatic brain tumor segmentation and detection with histogram equalization of morphological image processing. In: International Conference on Advanced Materials, Manufacturing and Sustainable Development (ICAMMSD 2024); 2024 Nov 22–23; Kurnool, India. p. 343–55. [Google Scholar]
108. Li C, Zhang C, Liu Z, Yang X. Multi-histogram equalization for image enhancement using adaptive fuzzy clustering and optimized clipping. Digit Signal Process. 2026;168(9):105466. doi:10.1016/j.dsp.2025.105466. [Google Scholar] [CrossRef]
109. Rahman H, Shimamura T. Dualistic fuzzy histogram equalization for enhancement of low-contrast images. In: Proceedings of the 2025 International Conference on Image and Video Processing (ICIVP); 2025 Apr 23–25; Jeju Island, Republic of Korea. p. 94–8. doi:10.1109/icivp66296.2025.00024. [Google Scholar] [CrossRef]
110. Rahman H, Shimamura T. Fuzzy bi-histogram equalization for enhancement of images. In: Proceedings of the 2025 International Conference on Image and Video Processing (ICIVP); 2025 Apr 23–25; Jeju Island, Republic of Korea. p. 63–8. doi:10.1109/icivp66296.2025.00019. [Google Scholar] [CrossRef]
111. Likassa HT, Chen DG. RPCA with log-schatten norm and adaptive histogram equalization for medical imaging. Int J Stat Med Res. 2025;14:274–88. doi:10.6000/1929-6029.2025.14.27. [Google Scholar] [CrossRef]
112. Manimekalai MAP, Vasanthi NA. Hybrid Lempel–Ziv–Welch and clipped histogram equalization based medical image compression. Clust Comput. 2019;22(5):12805–16. doi:10.1007/s10586-018-1761-7. [Google Scholar] [CrossRef]
113. David PF, Kothandapani SD, Pugalendhi GK. Adaptive compression and reconstruction for multidimensional medical image data: a hybrid algorithm for enhanced image quality. J Imag Inform Med. 2025;38(5):3148–67. doi:10.1007/s10278-024-01353-x. [Google Scholar] [PubMed] [CrossRef]
114. Acharya UK, Kumar S. Particle swarm optimized texture based histogram equalization (PSOTHE) for MRI brain image enhancement. Optik. 2020;224(3):165760. doi:10.1016/j.ijleo.2020.165760. [Google Scholar] [CrossRef]
115. Saifullah S, Dreżewski R. Advanced medical image segmentation enhancement: a particle-swarm-optimization-based histogram equalization approach. Appl Sci. 2024;14(2):923. doi:10.3390/app14020923. [Google Scholar] [CrossRef]
116. Shanmugavadivu P, Balasubramanian K, Muruganandam A. Particle swarm optimized bi-histogram equalization for contrast enhancement and brightness preservation of images. Vis Comput. 2014;30(4):387–99. doi:10.1007/s00371-013-0863-8. [Google Scholar] [CrossRef]
117. Radhika R, Mahajan R. A novel framework of hybrid optimization techniques for contrast-enhancement in cardiac MRI medical images. J Angiother. 2024;8(3):1–11. doi:10.25163/angiotherapy.839504. [Google Scholar] [CrossRef]
118. Samraj D, Karuppusamy M. A metaheuristic-based histogram equalization method for mammogram enhancement using a brightness preserving cuckoo search algorithm. J Opt. 2025;85(1):105749. doi:10.1007/s12596-025-02834-0. [Google Scholar] [CrossRef]
119. Pashaei E, Pashaei E. Gaussian quantum arithmetic optimization-based histogram equalization for medical image enhancement. Multimed Tools Appl. 2023;82(22):34725–48. doi:10.1007/s11042-023-15025-5. [Google Scholar] [CrossRef]
120. Vijayalakshmi D, Elangovan P, Kumari TS, Kumar Nath M. Optimized multi-scale framework for image enhancement using spatial information-based histogram equalization. Imag Sci J. 2025;73(2):176–203. doi:10.1080/13682199.2024.2343979. [Google Scholar] [CrossRef]
121. Omran KM, Ismael AN, Shakir SD. Medical image enhancement applying modified camel algorithm. Int J Comput Digit Syst. 2025;18(1):1–12. doi:10.12785/ijcds/1571111860. [Google Scholar] [CrossRef]
122. Qiu J, Li HH, Zhang T, Ma F, Yang D. Automatic X-ray image contrast enhancement based on parameter auto-optimization. J Appl Clin Med Phys. 2017;18(6):218–23. doi:10.1002/acm2.12172. [Google Scholar] [PubMed] [CrossRef]
123. Kuo CJ, Wu HC. Gaussian probability bi-histogram equalization for enhancement of the pathological features in medical images. Int J Imaging Syst Tech. 2019;29(2):132–45. doi:10.1002/ima.22307. [Google Scholar] [CrossRef]
124. Hwang SH, Kim YJ, Cho JB, Kim KG, Nam DH. Digital image enhancement using deep learning algorithm in 3D heads-up vitreoretinal surgery. Sci Rep. 2025;15(1):18429. doi:10.1038/s41598-025-98801-7. [Google Scholar] [PubMed] [CrossRef]
125. Luo F, Wu D, Pino LR, Ding W. A novel multimodel medical image fusion framework with edge enhancement and cross-scale transformer. Sci Rep. 2025;15(1):11657. doi:10.1038/s41598-025-93616-y. [Google Scholar] [PubMed] [CrossRef]
126. Setiawan H, Ria F, Abadi E, Marin D, Molvin L, Samei E. Development and clinical evaluation of a contrast optimizer for contrast-enhanced CT imaging of the liver. J Comput Assist Tomogr. 2025;49(2):239–46. doi:10.1097/rct.0000000000001677. [Google Scholar] [PubMed] [CrossRef]
127. Cai Y, Bian H, Lin J, Wang H, Timofte R, Zhang Y. Retinexformer: one-stage retinex-based transformer for low-light image enhancement. In: Proceedings of the 2023 IEEE/CVF International Conference on Computer Vision (ICCV); 2023 Oct 1–6; Paris, France. p. 12504–13. doi:10.1109/iccv51070.2023.01149. [Google Scholar] [CrossRef]
128. Saifullah S, Dreżewski R. Enhanced medical image segmentation using CNN based on histogram equalization. In: Proceedings of the 2023 2nd International Conference on Applied Artificial Intelligence and Computing (ICAAIC); 2023 May 4–6; Salem, India. p. 121–6. doi:10.1109/icaaic56838.2023.10141065. [Google Scholar] [CrossRef]
129. Fei B, Li Y, Yang W, Gao H, Xu J, Ma L, et al. A diffusion model for universal medical image enhancement. Commun Med. 2025;5:294. doi:10.1038/s43856-025-00998-1. [Google Scholar] [PubMed] [CrossRef]
130. Singh P, Mukundan R, de Ryke R. Feature enhancement in medical ultrasound videos using multifractal and contrast adaptive histogram equalization techniques. In: Proceedings of the 2019 IEEE Conference on Multimedia Information Processing and Retrieval (MIPR); 2019 Mar 28–30; San Jose, CA, USA. p. 240–5. doi:10.1109/mipr.2019.00050. [Google Scholar] [CrossRef]
131. Alpan K, Arman B, Dimililer K. Effect of contrast limited adaptive histogram equalization (CLAHE) on breast cancer detection using residual network (ResNet). In: Proceedings of the 2025 1st International Conference on Computational Intelligence Approaches and Applications (ICCIAA); 2025 Apr 28–30; Amman, Jordan. p. 1–5. doi:10.1109/icciaa65327.2025.11013776. [Google Scholar] [CrossRef]
132. Halloum K, Ez-Zahraouy H. Enhancing medical image classification through transfer learning and CLAHE optimization. Curr Med Imag Former Curr Med Imag Rev. 2025;21:e15734056342623. doi:10.2174/0115734056342623241119061744. [Google Scholar] [PubMed] [CrossRef]
133. M V, Rajkumar S, Reddy MVK, Janesh V. Detection of post COVID-pneumonia using histogram equalization, CLAHE deep learning techniques. Intel Artif. 2023;26(72):137–45. doi:10.4114/intartif.vol26iss72pp137-145. [Google Scholar] [CrossRef]
134. Risma VM, Utami E. Tuberculosis diagnosis from X-ray images using deep learning and contrast enhancement techniques. J Tek Inform (JUTIF). 2025;6(2):981–94. doi:10.52436/1.jutif.2025.6.2.4315. [Google Scholar] [CrossRef]
135. Ahmad MSZ, Aziz NAA, Lim HS, Ghazali AK, Latiff AA. Impact of image enhancement using contrast-limited adaptive histogram equalization (CLAHEanisotropic diffusion, and histogram equalization on spine X-ray segmentation with U-net, mask R-CNN, and transfer learning. Algorithms. 2025;18(12):796. doi:10.3390/a18120796. [Google Scholar] [CrossRef]
136. Zhong Q, Zhu S, He J, Wang H, Zhong R. Image enhancement for accelerated MRI using a joint GAN and diffusion model framework. Med Phys. 2026;53(1):e70242. doi:10.1002/mp.70242. [Google Scholar] [PubMed] [CrossRef]
137. Aashik Rasool MJ, Abdusalomov A, Kutlimuratov A, Akeel Ahamed MJ, Mirzakhalilov S, Shavkatovich Buriboev A, et al. PixMed-enhancer: an efficient approach for medical image augmentation. Bioengineering. 2025;12(3):235. doi:10.3390/bioengineering12030235. [Google Scholar] [PubMed] [CrossRef]
138. Vimala Ithayan J, Shiney G, Akshitha M, Lavanya K. Enhancing low-light images across divers domain using retinex models for real-time quality enhancement. In: Proceedings of the 2025 9th International Conference on Inventive Systems and Control (ICISC); 2025 Aug 12–13; Coimbatore, India. p. 1–7. doi:10.1109/icisc65841.2025.11188352. [Google Scholar] [CrossRef]
139. Liu Y, Feng Y, Cheng J, Zhan H, MambaDiff Zhu Z. Mamba-enhanced diffusion model for 3D medical image segmentation. IEEE Trans Image Process. 2025;34:5761–75. doi:10.1109/tip.2025.3607615. [Google Scholar] [PubMed] [CrossRef]
140. Roy S, Bhalla K, Patel R. Mathematical analysis of histogram equalization techniques for medical image enhancement: a tutorial from the perspective of data loss. Multimed Tools Appl. 2024;83(5):14363–92. doi:10.1007/s11042-023-15799-8. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools